Degranulation of Mast Cells as a Target for Drug Development
Abstract
1. Introduction
2. Factors Involved in the Differentiation, Proliferation, and Activation of Mast Cells
2.1. SCF (Stem Cell Factor) and IL-3
2.2. IL-4 and IL-33
2.3. IL-9
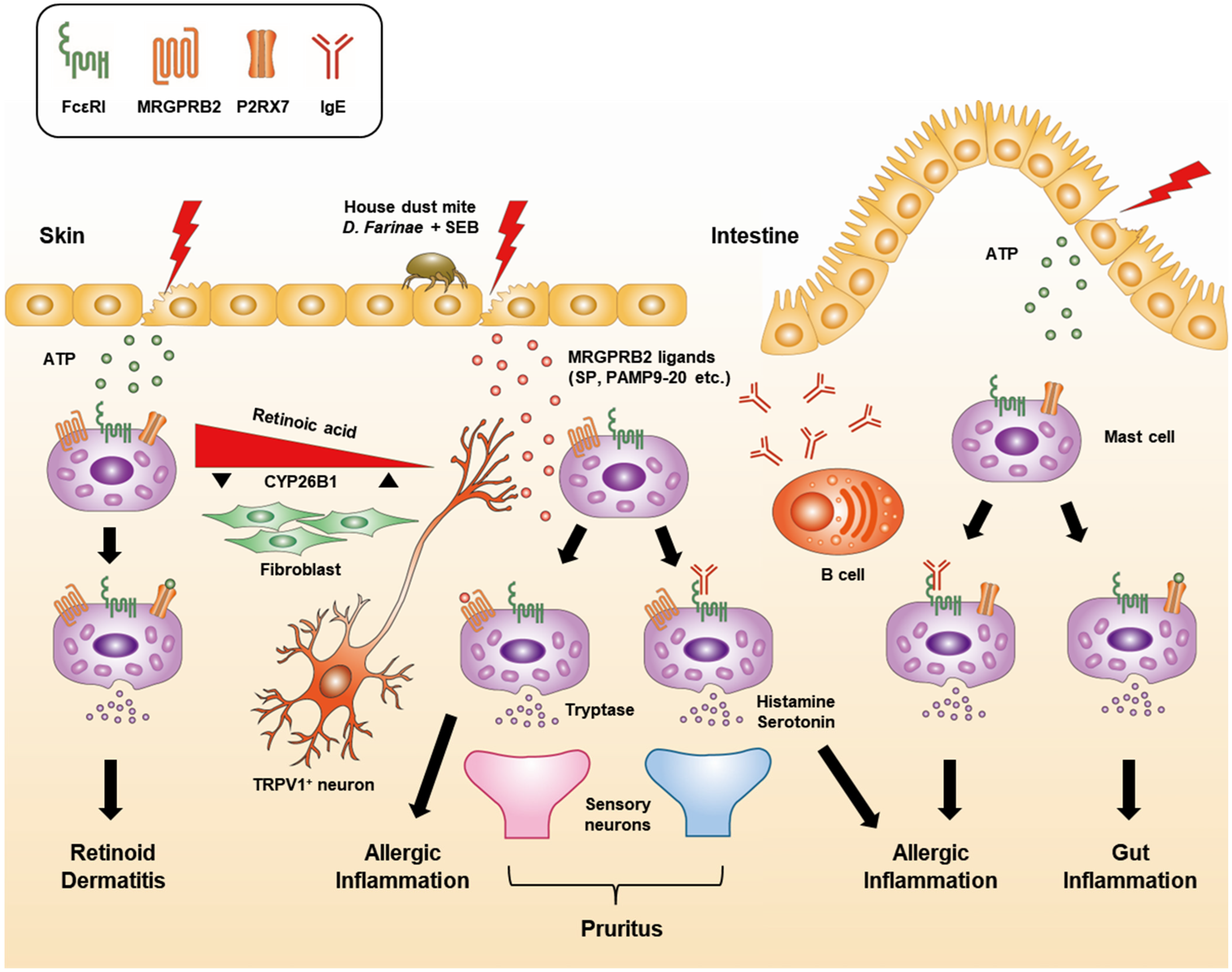
3. Receptors Involved in Mast Cell Activation and Degranulation
3.1. High-Affinity IgE Receptor (FcεRI)
3.2. Low-Affinity IgG Receptors (FcγRs)
3.3. MRGPRX2/B2
3.4. P2X Purinoceptor 7 (P2RX7)
3.5. Adhesion G-Protein-Coupled Receptor E2 (ADGRE2)
4. Development of Drugs Targeting the Inhibition of Mast Cell Degranulation
4.1. Inhibition of IgE-Dependent Degranulation
4.1.1. Omalizumab
4.1.2. Ligelizumab
4.1.3. UB-221
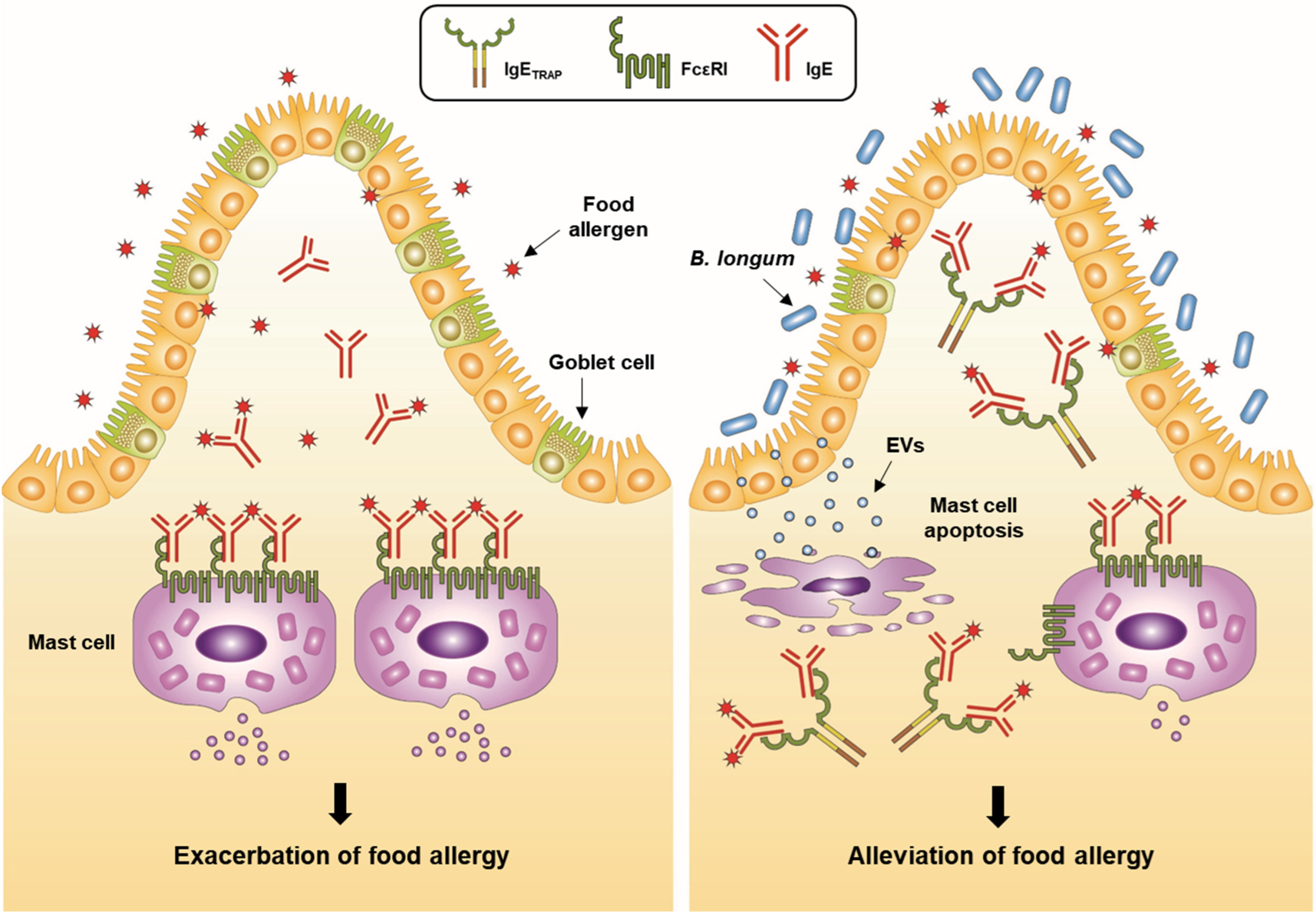
4.1.4. IgETRAP
4.2. Inhibition of IgE-Independent Degranulation
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Gurish, M.F.; Austen, K.F. Developmental Origin and Functional Specialization of Mast Cell Subsets. Immunity 2012, 37, 25–33. [Google Scholar] [CrossRef] [PubMed]
- Galli, S.J.; Borregaard, N.; Wynn, T.A. Phenotypic and functional plasticity of cells of innate immunity: Macrophages, mast cells and neutrophils. Nat. Immunol. 2011, 12, 1035–1044. [Google Scholar] [CrossRef] [PubMed]
- Moon, T.C.; St Laurent, C.D.; Morris, K.E.; Marcet, C.; Yoshimura, T.; Sekar, Y.; Befus, A.D. Advances in mast cell biology: New understanding of heterogeneity and function. Mucosal Immunol. 2010, 3, 111–128. [Google Scholar] [CrossRef]
- Da Silva, E.Z.M.; Jamur, M.C.; Oliver, C. Mast Cell Function: A new vision of an old cell. J. Histochem. Cytochem. 2014, 62, 698–738. [Google Scholar] [CrossRef] [PubMed]
- West, P.W.; Bulfone-Paus, S. Mast cell tissue heterogeneity and specificity of immune cell recruitment. Front. Immunol. 2022, 13, 932090. [Google Scholar] [CrossRef]
- Tsai, M.; Valent, P.; Galli, S.J. KIT as a master regulator of the mast cell lineage. J. Allergy Clin. Immunol. 2022, 149, 1845–1854. [Google Scholar] [CrossRef]
- Lennartsson, J.; Rönnstrand, L. Stem Cell Factor Receptor/c-Kit: From Basic Science to Clinical Implications. Physiol. Rev. 2012, 92, 1619–1649. [Google Scholar] [CrossRef]
- Tajima, Y.; Moore, M.A.S.; Soares, V.; Ono, M.; Kissel, H.; Besmer, P. Consequences of exclusive expression in vivo of kit-ligand lacking the major proteolytic cleavage site. Proc. Natl. Acad. Sci. USA 1998, 95, 11903–11908. [Google Scholar] [CrossRef]
- Kapur, R.; Majumdar, M.; Xiao, X.; McAndrews-Hill, M.; Schindler, K.; Williams, D.A. Signaling through the interaction of membrane-restricted stem cell factor and c-kit receptor tyrosine kinase: Genetic evidence for a differential role in erythropoiesis. Blood 1998, 91, 879–889. [Google Scholar] [CrossRef]
- Takematsu, E.; Massidda, M.; Auster, J.; Chen, P.-C.; Im, B.; Srinath, S.; Canga, S.; Singh, A.; Majid, M.; Sherman, M.; et al. Transmembrane stem cell factor protein therapeutics enhance revascularization in ischemia without mast cell activation. Nat. Commun. 2022, 13, 2497. [Google Scholar] [CrossRef]
- Ho, C.C.M.; Chhabra, A.; Starkl, P.; Schnorr, P.-J.; Wilmes, S.; Moraga, I.; Kwon, H.-S.; Gaudenzio, N.; Sibilano, R.; Wehrman, T.S.; et al. Decoupling the Functional Pleiotropy of Stem Cell Factor by Tuning c-Kit Signaling. Cell 2017, 168, 1041–1052.e18. [Google Scholar] [CrossRef] [PubMed]
- Hundley, T.R.; Gilfillan, A.M.; Tkaczyk, C.; Andrade, M.V.; Metcalfe, D.D.; Beaven, M.A. Kit and FcϵRI mediate unique and convergent signals for release of inflammatory mediators from human mast cells. Blood 2004, 104, 2410–2417. [Google Scholar] [CrossRef]
- Tkaczyk, C.; Horejsi, V.; Iwaki, S.; Draber, P.; Samelson, L.E.; Satterthwaite, A.B.; Nahm, D.H.; Metcalfe, D.D.; Gilfillan, A.M. NTAL phosphorylation is a pivotal link between the signaling cascades leading to human mast cell degranulation following Kit activation and Fc RI aggregation. Blood 2004, 104, 207–214. [Google Scholar] [CrossRef]
- Wang, Z.; Mascarenhas, N.; Eckmann, L.; Miyamoto, Y.; Sun, X.; Kawakami, T.; Di Nardo, A. Skin microbiome promotes mast cell maturation by triggering stem cell factor production in keratinocytes. J. Allergy Clin. Immunol. 2017, 139, 1205–1216.e6. [Google Scholar] [CrossRef] [PubMed]
- Kunii, J.; Takahashi, K.; Kasakura, K.; Tsuda, M.; Nakano, K.; Hosono, A.; Kaminogawa, S. Commensal bacteria promote migration of mast cells into the intestine. Immunobiology 2011, 216, 692–697. [Google Scholar] [CrossRef] [PubMed]
- Schwarzer, M.; Hermanova, P.; Srutkova, D.; Golias, J.; Hudcovic, T.; Zwicker, C.; Sinkora, M.; Akgün, J.; Wiedermann, U.; Tuckova, L.; et al. Germ-Free Mice Exhibit Mast Cells with Impaired Functionality and Gut Homing and Do Not Develop Food Allergy. Front. Immunol. 2019, 10, 205. [Google Scholar] [CrossRef] [PubMed]
- Cahenzli, J.; Köller, Y.; Wyss, M.; Geuking, M.B.; McCoy, K.D. Intestinal Microbial Diversity during Early-Life Colonization Shapes Long-Term IgE Levels. Cell Host Microbe 2013, 14, 559–570. [Google Scholar] [CrossRef]
- Stefka, A.T.; Feehley, T.; Tripathi, P.; Qiu, J.; McCoy, K.; Mazmanian, S.K.; Tjota, M.Y.; Seo, G.-Y.; Cao, S.; Theriault, B.R.; et al. Commensal bacteria protect against food allergen sensitization. Proc. Natl. Acad. Sci. USA 2014, 111, 13145–13150. [Google Scholar] [CrossRef]
- Caslin, H.L.; Kiwanuka, K.N.; Haque, T.T.; Taruselli, M.T.; MacKnight, H.P.; Paranjape, A.; Ryan, J.J. Controlling Mast Cell Activation and Homeostasis: Work Influenced by Bill Paul That Continues Today. Front. Immunol. 2018, 9, 868. [Google Scholar] [CrossRef]
- Lantz, C.S.; Boesiger, J.; Song, C.H.; Mach, N.; Kobayashi, T.; Mulligan, R.C.; Nawa, Y.; Dranoff, G.; Galli, S.J. Role for interleukin-3 in mast-cell and basophil development and in immunity to parasites. Nature 1998, 392, 90–93. [Google Scholar] [CrossRef]
- Brown, M.A.; Pierce, J.H.; Watson, C.J.; Falco, J.; Ihle, J.N.; Paul, W.E. B cell stimulatory factor-1/interleukin-4 mRNA is expressed by normal and transformed mast cells. Cell 1987, 50, 809–818. [Google Scholar] [CrossRef] [PubMed]
- Burton, O.T.; Darling, A.R.; Zhou, J.S.; Noval-Rivas, M.; Jones, T.G.; Gurish, M.F.; Chatila, T.A.; Oettgen, H.C. Direct effects of IL-4 on mast cells drive their intestinal expansion and increase susceptibility to anaphylaxis in a murine model of food allergy. Mucosal Immunol. 2013, 6, 740–750. [Google Scholar] [CrossRef] [PubMed]
- Rivas, M.N.; Burton, O.T.; Oettgen, H.C.; Chatila, T. IL-4 production by group 2 innate lymphoid cells promotes food allergy by blocking regulatory T-cell function. J. Allergy Clin. Immunol. 2016, 138, 801–811.e9. [Google Scholar] [CrossRef] [PubMed]
- McLeod, J.J.; Baker, B.; Ryan, J.J. Mast cell production and response to IL-4 and IL-13. Cytokine 2015, 75, 57–61. [Google Scholar] [CrossRef]
- Dahlin, J.S.; Hallgren, J. Mast cell progenitors: Origin, development and migration to tissues. Mol. Immunol. 2015, 63, 9–17. [Google Scholar] [CrossRef]
- Liew, F.Y.; Girard, J.-P.; Turnquist, H.R. Interleukin-33 in health and disease. Nat. Rev. Immunol. 2016, 16, 676–689. [Google Scholar] [CrossRef]
- Roan, F.; Obata-Ninomiya, K.; Ziegler, S.F. Epithelial cell–derived cytokines: More than just signaling the alarm. J. Clin. Investig. 2019, 129, 1441–1451. [Google Scholar] [CrossRef]
- Wang, J.-X.; Kaieda, S.; Ameri, S.; Fishgal, N.; Dwyer, D.; Dellinger, A.; Kepley, C.L.; Gurish, M.F.; Nigrovic, P.A. IL-33/ST2 axis promotes mast cell survival via BCLXL. Proc. Natl. Acad. Sci. USA 2014, 111, 10281–10286. [Google Scholar] [CrossRef]
- Allakhverdi, Z.; Smith, D.E.; Comeau, M.R.; Delespesse, G. Cutting Edge: The ST2 Ligand IL-33 Potently Activates and Drives Maturation of Human Mast Cells. J. Immunol. 2007, 179, 2051–2054. [Google Scholar] [CrossRef]
- Leyva-Castillo, J.-M.; Galand, C.; Kam, C.; Burton, O.; Gurish, M.; Musser, M.A.; Goldsmith, J.D.; Hait, E.; Nurko, S.; Brombacher, F.; et al. Mechanical Skin Injury Promotes Food Anaphylaxis by Driving Intestinal Mast Cell Expansion. Immunity 2019, 50, 1262–1275.e4. [Google Scholar] [CrossRef]
- Schmitz, J.; Owyang, A.; Oldham, E.; Song, Y.; Murphy, E.; McClanahan, T.K.; Zurawski, G.; Moshrefi, M.; Qin, J.; Li, X.; et al. IL-33, an Interleukin-1-like Cytokine that Signals via the IL-1 Receptor-Related Protein ST2 and Induces T Helper Type 2-Associated Cytokines. Immunity 2005, 23, 479–490. [Google Scholar] [CrossRef] [PubMed]
- Johnston, L.K.; Hsu, C.-L.; Krier-Burris, R.A.; Chhiba, K.D.; Chien, K.B.; McKenzie, A.; Berdnikovs, S.; Bryce, P.J. IL-33 Precedes IL-5 in Regulating Eosinophil Commitment and Is Required for Eosinophil Homeostasis. J. Immunol. 2016, 197, 3445–3453. [Google Scholar] [CrossRef] [PubMed]
- Komai-Koma, M.; Brombacher, F.; Pushparaj, P.N.; Arendse, B.; McSharry, C.; Alexander, J.; Chaudhuri, R.; Thomson, N.; McKenzie, A.N.J.; McInnes, I.; et al. Interleukin-33 amplifies I g E synthesis and triggers mast cell degranulation via interleukin-4 in naïve mice. Allergy 2012, 67, 1118–1126. [Google Scholar] [CrossRef] [PubMed]
- Chu, D.K.; Llop-Guevara, A.; Walker, T.D.; Flader, K.; Goncharova, S.; Boudreau, J.E.; Moore, C.L.; In, T.S.; Waserman, S.; Coyle, A.J.; et al. IL-33, but not thymic stromal lymphopoietin or IL-25, is central to mite and peanut allergic sensitization. J. Allergy Clin. Immunol. 2013, 131, 187–200.e8. [Google Scholar] [CrossRef]
- Bartnikas, L.M.; Gurish, M.F.; Burton, O.T.; Leisten, S.; Janssen, E.; Oettgen, H.C.; Beaupré, J.; Lewis, C.N.; Austen, K.F.; Schulte, S.; et al. Epicutaneous sensitization results in IgE-dependent intestinal mast cell expansion and food-induced anaphylaxis. J. Allergy Clin. Immunol. 2013, 131, 451–460.e6. [Google Scholar] [CrossRef] [PubMed]
- Burton, O.T.; Rivas, M.N.; Zhou, J.S.; Logsdon, S.L.; Darling, A.R.; Koleoglou, K.J.; Roers, A.; Houshyar, H.; Crackower, M.A.; Chatila, T.A.; et al. Immunoglobulin E Signal Inhibition during Allergen Ingestion Leads to Reversal of Established Food Allergy and Induction of Regulatory T Cells. Immunity 2014, 41, 141–151. [Google Scholar] [CrossRef]
- An, S.B.; Yang, B.-G.; Jang, G.; Kim, D.-Y.; Kim, J.; Oh, S.-M.; Oh, N.; Lee, S.; Moon, J.-Y.; Kim, J.-A.; et al. Combined IgE neutralization and Bifidobacterium longum supplementation reduces the allergic response in models of food allergy. Nat. Commun. 2022, 13, 5669. [Google Scholar] [CrossRef]
- Galand, C.; Leyva-Castillo, J.M.; Yoon, J.; Han, A.; Lee, M.S.; McKenzie, A.N.; Stassen, M.; Oyoshi, M.K.; Finkelman, F.D.; Geha, R.S. IL-33 promotes food anaphylaxis in epicutaneously sensitized mice by targeting mast cells. J. Allergy Clin. Immunol. 2016, 138, 1356–1366. [Google Scholar] [CrossRef]
- Hsu, C.-L.; Neilsen, C.V.; Bryce, P.J. IL-33 Is Produced by Mast Cells and Regulates IgE-Dependent Inflammation. PLoS ONE 2010, 5, e11944. [Google Scholar] [CrossRef]
- Shimokawa, C.; Kanaya, T.; Hachisuka, M.; Ishiwata, K.; Hisaeda, H.; Kurashima, Y.; Kiyono, H.; Yoshimoto, T.; Kaisho, T.; Ohno, H. Mast Cells Are Crucial for Induction of Group 2 Innate Lymphoid Cells and Clearance of Helminth Infections. Immunity 2017, 46, 863–874.e4. [Google Scholar] [CrossRef]
- Townsend, M.J.; Fallon, P.G.; Matthews, D.J.; Smith, P.; Jolin, H.E.; McKenzie, A.N. IL-9-Deficient Mice Establish Fundamental Roles for IL-9 in Pulmonary Mastocytosis and Goblet Cell Hyperplasia but Not T Cell Development. Immunity 2000, 13, 573–583. [Google Scholar] [CrossRef] [PubMed]
- Temann, U.-A.; Geba, G.P.; Rankin, J.A.; Flavell, R.A. Expression of Interleukin 9 in the Lungs of Transgenic Mice Causes Airway Inflammation, Mast Cell Hyperplasia, and Bronchial Hyperresponsiveness. J. Exp. Med. 1998, 188, 1307–1320. [Google Scholar] [CrossRef] [PubMed]
- Hültner, L.; Druez, C.; Moeller, J.; Uyttenhove, C.; Schmitt, E.; Rüde, E.; Dörmer, P.; Van Snick, J. Mast cell growth-enhancing activity (MEA) is structurally related and functionally identical to the novel mouse T cell growth factor P40/TCGFIII (interleukin 9). Eur. J. Immunol. 1990, 20, 1413–1416. [Google Scholar] [CrossRef]
- Angkasekwinai, P.; Dong, C. IL-9-producing T cells: Potential players in allergy and cancer. Nat. Rev. Immunol. 2021, 21, 37–48. [Google Scholar] [CrossRef]
- Forbes, E.E.; Groschwitz, K.; Abonia, J.P.; Brandt, E.; Cohen, E.B.; Blanchard, C.; Ahrens, R.; Seidu, L.; McKenzie, A.; Strait, R.; et al. IL-9– and mast cell–mediated intestinal permeability predisposes to oral antigen hypersensitivity. J. Exp. Med. 2008, 205, 897–913. [Google Scholar] [CrossRef] [PubMed]
- Osterfeld, H.; Ahrens, R.; Strait, R.; Finkelman, F.D.; Renauld, J.-C.; Hogan, S.P. Differential roles for the IL-9/IL-9 receptor α-chain pathway in systemic and oral antigen–induced anaphylaxis. J. Allergy Clin. Immunol. 2010, 125, 469–476.e2. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.-Y.; Lee, J.-B.; Liu, B.; Ohta, S.; Wang, P.-Y.; Kartashov, A.V.; Mugge, L.; Abonia, J.P.; Barski, A.; Izuhara, K.; et al. Induction of Interleukin-9-Producing Mucosal Mast Cells Promotes Susceptibility to IgE-Mediated Experimental Food Allergy. Immunity 2015, 43, 788–802. [Google Scholar] [CrossRef]
- Lee, Y.; Kumagai, Y.; Jang, M.S.; Kim, J.-H.; Yang, B.-G.; Lee, E.-J.; Kim, Y.-M.; Akira, S.; Jang, M.H. Intestinal Lin−c-Kit+NKp46−CD4− Population Strongly Produces IL-22 upon IL-1β Stimulation. J. Immunol. 2013, 190, 5296–5305. [Google Scholar] [CrossRef]
- Kraft, S.; Kinet, J.-P.P. New developments in FcεRI regulation, function and inhibition. Nat. Rev. Immunol. 2007, 7, 365–378. [Google Scholar] [CrossRef]
- Kitaura, J.; Song, J.; Tsai, M.; Asai, K.; Maeda-Yamamoto, M.; Mocsai, A.; Kawakami, Y.; Liu, F.-T.; Lowell, C.A.; Barisas, B.G.; et al. Evidence that IgE molecules mediate a spectrum of effects on mast cell survival and activation via aggregation of the FcεRI. Proc. Natl. Acad. Sci. USA 2003, 100, 12911–12916. [Google Scholar] [CrossRef]
- Asai, K.; Kitaura, J.; Kawakami, Y.; Yamagata, N.; Tsai, M.; Carbone, D.P.; Liu, F.-T.; Galli, S.J.; Kawakami, T. Regulation of Mast Cell Survival by IgE. Immunity 2001, 14, 791–800. [Google Scholar] [CrossRef] [PubMed]
- Kalesnikoff, J.; Huber, M.; Lam, V.; Damen, J.E.; Zhang, J.; Siraganian, R.P.; Krystal, G. Monomeric IgE Stimulates Signaling Pathways in Mast Cells that Lead to Cytokine Production and Cell Survival. Immunity 2001, 14, 801–811. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M.; Lantz, C.S.; Oettgen, H.C.; Katona, I.M.; Fleming, T.; Miyajima, I.; Kinet, J.-P.; Galli, S.J. IgE Enhances Mouse Mast Cell FcεRI Expression In Vitro and In Vivo: Evidence for a Novel Amplification Mechanism in IgE-dependent Reactions. J. Exp. Med. 1997, 185, 663–672. [Google Scholar] [CrossRef] [PubMed]
- Rivas, M.N.; Burton, O.T.; Wise, P.; Charbonnier, L.-M.; Georgiev, P.; Oettgen, H.; Rachid, R.; Chatila, T.A. Regulatory T Cell Reprogramming toward a Th2-Cell-like Lineage Impairs Oral Tolerance and Promotes Food Allergy. Immunity 2015, 42, 512–523. [Google Scholar] [CrossRef]
- Tordesillas, L.; Berin, M.C.; Sampson, H.A. Immunology of Food Allergy. Immunity 2017, 47, 32–50. [Google Scholar] [CrossRef]
- PALFORZIA|FDA. Available online: https://www.fda.gov/vaccines-blood-biologics/allergenics/palforzia (accessed on 20 February 2023).
- Schneider, L.C.; Rachid, R.; LeBovidge, J.; Blood, E.; Mittal, M.; Umetsu, D.T. A pilot study of omalizumab to facilitate rapid oral desensitization in high-risk peanut-allergic patients. J. Allergy Clin. Immunol. 2013, 132, 1368–1374. [Google Scholar] [CrossRef]
- Abdel-Gadir, A.; Schneider, L.; Casini, A.; Charbonnier, L.-M.; Little, S.; Harrington, T.; Umetsu, D.T.; Rachid, R.; Chatila, T.A. Oral immunotherapy with omalizumab reverses the Th2 cell-like programme of regulatory T cells and restores their function. Clin. Exp. Allergy 2018, 48, 825–836. [Google Scholar] [CrossRef]
- Reber, L.L.; Hernandez, J.D.; Galli, S.J. The pathophysiology of anaphylaxis. J. Allergy Clin. Immunol. 2017, 140, 335–348. [Google Scholar] [CrossRef]
- Miyajima, I.; Dombrowicz, D.; Martin, T.R.; Ravetch, J.V.; Kinet, J.P.; Galli, S.J. Systemic anaphylaxis in the mouse can be mediated largely through IgG1 and Fc gammaRIII. Assessment of the cardiopulmonary changes, mast cell degranulation, and death associated with active or IgE- or IgG1-dependent passive anaphylaxis. J. Clin. Investig. 1997, 99, 901–914. [Google Scholar] [CrossRef]
- Beutier, H.; Gillis, C.M.; Iannascoli, B.; Godon, O.; England, P.; Sibilano, R.; Reber, L.L.; Galli, S.J.; Cragg, M.S.; Van Rooijen, N.; et al. IgG subclasses determine pathways of anaphylaxis in mice. J. Allergy Clin. Immunol. 2017, 139, 269–280.e7. [Google Scholar] [CrossRef]
- Bruhns, P. Properties of mouse and human IgG receptors and their contribution to disease models. Blood 2012, 119, 5640–5649. [Google Scholar] [CrossRef] [PubMed]
- Dombrowicz, D.; Flamand, V.; Miyajima, I.; Ravetch, J.V.; Galli, S.J.; Kinet, J.P. Absence of Fc epsilonRI alpha chain results in upregulation of Fc gammaRIII-dependent mast cell degranulation and anaphylaxis. Evidence of competition between Fc epsilonRI and Fc gammaRIII for limiting amounts of FcR beta and gamma chains. J. Clin. Investig. 1997, 99, 915–925. [Google Scholar] [CrossRef] [PubMed]
- Gillis, C.M.; Jönsson, F.; Mancardi, D.A.; Tu, N.; Beutier, H.; Van Rooijen, N.; Macdonald, L.E.; Murphy, A.J.; Bruhns, P. Mechanisms of anaphylaxis in human low-affinity IgG receptor locus knock-in mice. J. Allergy Clin. Immunol. 2017, 139, 1253–1265.e14. [Google Scholar] [CrossRef]
- Vadas, P.; Gold, M.; Perelman, B.; Liss, G.M.; Lack, G.; Blyth, T.; Simons, F.E.R.; Simons, K.J.; Cass, D.; Yeung, J. Platelet-Activating Factor, PAF Acetylhydrolase, and Severe Anaphylaxis. N. Engl. J. Med. 2008, 358, 28–35. [Google Scholar] [CrossRef] [PubMed]
- Kajiwara, N.; Sasaki, T.; Bradding, P.; Cruse, G.; Sagara, H.; Ohmori, K.; Saito, H.; Ra, C.; Okayama, Y. Activation of human mast cells through the platelet-activating factor receptor. J. Allergy Clin. Immunol. 2010, 125, 1137–1145.e6. [Google Scholar] [CrossRef]
- Kumar, M.; Duraisamy, K.; Chow, B.-K. Unlocking the Non-IgE-Mediated Pseudo-Allergic Reaction Puzzle with Mas-Related G-Protein Coupled Receptor Member X2 (MRGPRX2). Cells 2021, 10, 1033. [Google Scholar] [CrossRef]
- Roy, S.; Na Ayudhya, C.C.; Thapaliya, M.; Deepak, V.; Ali, H. Multifaceted MRGPRX2: New insight into the role of mast cells in health and disease. J. Allergy Clin. Immunol. 2021, 148, 293–308. [Google Scholar] [CrossRef]
- McNeil, B.D.; Pundir, P.; Meeker, S.; Han, L.; Undem, B.J.; Kulka, M.; Dong, X. Identification of a mast-cell-specific receptor crucial for pseudo-allergic drug reactions. Nature 2014, 519, 237–241. [Google Scholar] [CrossRef]
- Subramanian, H.; Gupta, K.; Ali, H. Roles of Mas-related G protein–coupled receptor X2 on mast cell–mediated host defense, pseudoallergic drug reactions, and chronic inflammatory diseases. J. Allergy Clin. Immunol. 2016, 138, 700–710. [Google Scholar] [CrossRef]
- Pundir, P.; Liu, R.; Vasavda, C.; Serhan, N.; Limjunyawong, N.; Yee, R.; Zhan, Y.; Dong, X.; Wu, X.; Zhang, Y.; et al. A Connective Tissue Mast-Cell-Specific Receptor Detects Bacterial Quorum-Sensing Molecules and Mediates Antibacterial Immunity. Cell Host Microbe 2019, 26, 114–122.e8. [Google Scholar] [CrossRef]
- Serhan, N.; Basso, L.; Sibilano, R.; Petitfils, C.; Meixiong, J.; Bonnart, C.; Reber, L.; Marichal, T.; Starkl, P.; Cenac, N.; et al. House dust mites activate nociceptor–mast cell clusters to drive type 2 skin inflammation. Nat. Immunol. 2019, 20, 1435–1443. [Google Scholar] [CrossRef] [PubMed]
- Järvikallio, A.; Harvima, I.T.; Naukkarinen, A. Mast cells, nerves and neuropeptides in atopic dermatitis and nummular eczema. Arch. Dermatol. Res. 2003, 295, 2–7. [Google Scholar] [CrossRef] [PubMed]
- Sugiura, H.; Maeda, T.; Uehara, M. Mast cell invasion of peripheral nerve in skin lesions of atopic dermatitis. Acta Dermato-Venereologica. Suppl. 1992, 176, 74–76. [Google Scholar]
- Meixiong, J.; Anderson, M.; Limjunyawong, N.; Sabbagh, M.F.; Hu, E.; Mack, M.R.; Oetjen, L.K.; Wang, F.; Kim, B.S.; Dong, X. Activation of Mast-Cell-Expressed Mas-Related G-Protein-Coupled Receptors Drives Non-histaminergic Itch. Immunity 2019, 50, 1163–1171.e5. [Google Scholar] [CrossRef] [PubMed]
- Green, D.P.; Limjunyawong, N.; Gour, N.; Pundir, P.; Dong, X. A Mast-Cell-Specific Receptor Mediates Neurogenic Inflammation and Pain. Neuron 2019, 101, 412–420.e3. [Google Scholar] [CrossRef]
- Kurashima, Y.; Amiya, T.; Fujisawa, K.; Shibata, N.; Suzuki, Y.; Kogure, Y.; Hashimoto, E.; Otsuka, A.; Kabashima, K.; Sato, S.; et al. The Enzyme Cyp26b1 Mediates Inhibition of Mast Cell Activation by Fibroblasts to Maintain Skin-Barrier Homeostasis. Immunity 2014, 40, 530–541. [Google Scholar] [CrossRef]
- Kurashima, Y.; Amiya, T.; Nochi, T.; Fujisawa, K.; Haraguchi, T.; Iba, H.; Tsutsui, H.; Sato, S.; Nakajima, S.; Iijima, H.; et al. Extracellular ATP mediates mast cell-dependent intestinal inflammation through P2X7 purinoceptors. Nat. Commun. 2012, 3, 1034. [Google Scholar] [CrossRef]
- Idzko, M.; Ferrari, D.; Eltzschig, H.K. Nucleotide signalling during inflammation. Nature 2014, 509, 310–317. [Google Scholar] [CrossRef]
- Olivera, A.; Beaven, M.A.; Metcalfe, D.D. Mast cells signal their importance in health and disease. J. Allergy Clin. Immunol. 2018, 142, 381–393. [Google Scholar] [CrossRef]
- Kwakkenbos, M.J.; Chang, G.-W.; Lin, H.-H.; Pouwels, W.; de Jong, E.C.; Lier, R.A.W.V.; Gordon, S.; Hamann, J. The human EGF-TM7 family member EMR2 is a heterodimeric receptor expressed on myeloid cells. J. Leukoc. Biol. 2002, 71, 854–862. [Google Scholar] [CrossRef]
- Florian, S.; Sonneck, K.; Czerny, M.; Hennersdorf, F.; Hauswirth, A.W.; Buhring, H.-J.; Valent, P. Detection of novel leukocyte differentiation antigens on basophils and mast cells by HLDA8 antibodies. Allergy 2006, 61, 1054–1062. [Google Scholar] [CrossRef] [PubMed]
- Boyden, S.E.; Desai, A.; Cruse, G.; Young, M.L.; Bolan, H.C.; Scott, L.M.; Eisch, A.R.; Long, R.D.; Lee, C.-C.R.; Satorius, C.L.; et al. Vibratory Urticaria Associated with a Missense Variant in ADGRE2. N. Engl. J. Med. 2016, 374, 656–663. [Google Scholar] [CrossRef] [PubMed]
- Le, Q.T.; Lyons, J.J.; Naranjo, A.N.; Olivera, A.; Lazarus, R.A.; Metcalfe, D.D.; Milner, J.D.; Schwartz, L.B. Impact of naturally forming human α/β-tryptase heterotetramers in the pathogenesis of hereditary α-tryptasemia. J. Exp. Med. 2019, 216, 2348–2361. [Google Scholar] [CrossRef] [PubMed]
- Acharya, M.; Borland, G.; Edkins, A.L.; MacLellan, L.M.; Matheson, J.; Ozanne, B.W.; Cushley, W. CD23/FcεRII: Molecular multi-tasking. Clin. Exp. Immunol. 2010, 162, 12–23. [Google Scholar] [CrossRef]
- Balbino, B.; Conde, E.; Marichal, T.; Starkl, P.; Reber, L.L. Approaches to target IgE antibodies in allergic diseases. Pharmacol. Ther. 2018, 191, 50–64. [Google Scholar] [CrossRef]
- Guntern, P.; Eggel, A. Past, present, and future of anti-IgE biologics. Allergy 2020, 75, 2491–2502. [Google Scholar] [CrossRef]
- Limb, S.L.; Starke, P.R.; Lee, C.E.; Chowdhury, B.A. Delayed onset and protracted progression of anaphylaxis after omalizumab administration in patients with asthma. J. Allergy Clin. Immunol. 2007, 120, 1378–1381. [Google Scholar] [CrossRef]
- Lieberman, P.L.; Umetsu, D.T.; Carrigan, G.J.; Rahmaoui, A. Anaphylactic reactions associated with omalizumab administration: Analysis of a case-control study. J. Allergy Clin. Immunol. 2016, 138, 913–915.e2. [Google Scholar] [CrossRef]
- Lieberman, P.L.; Jones, I.; Rajwanshi, R.; Rosén, K.; Umetsu, D.T. Anaphylaxis associated with omalizumab administration: Risk factors and patient characteristics. J. Allergy Clin. Immunol. 2017, 140, 1734–1736.e4. [Google Scholar] [CrossRef]
- Balbino, B.; Herviou, P.; Godon, O.; Stackowicz, J.; Goff, O.R.-L.; Iannascoli, B.; Sterlin, D.; Brûlé, S.; Millot, G.A.; Harris, F.M.; et al. The anti-IgE mAb omalizumab induces adverse reactions by engaging Fcγ receptors. J. Clin. Investig. 2020, 130, 1330–1335. [Google Scholar] [CrossRef]
- Xolair Label. Available online: https://www.accessdata.fda.gov/drugsatfda_docs/label/2021/103976s5238lbl.pdf (accessed on 20 February 2023).
- Xolair Breakthrough Therapy Designation. Available online: https://www.gene.com/media/press-releases/14740/2018-08-12/fda-grants-breakthrough-therapy-designat (accessed on 20 February 2023).
- Wang, H.-H.; Li, Y.-C.; Huang, Y.-C. Efficacy of omalizumab in patients with atopic dermatitis: A systematic review and meta-analysis. J. Allergy Clin. Immunol. 2016, 138, 1719–1722.e1. [Google Scholar] [CrossRef] [PubMed]
- Wollenberg, A.; Thomsen, S.F.; Lacour, J.-P.; Jaumont, X.; Lazarewicz, S. Targeting immunoglobulin E in atopic dermatitis: A review of the existing evidence. World Allergy Organ. J. 2021, 14, 100519. [Google Scholar] [CrossRef] [PubMed]
- Hasni, S.; Gupta, S.; Davis, M.; Poncio, E.; Bsn, Y.T.; Joyal, E.; Fike, A.; Manna, Z.; Auh, S.; Shi, Y.; et al. Safety and Tolerability of Omalizumab: A Randomized Clinical Trial of Humanized Anti-IgE Monoclonal Antibody in Systemic Lupus Erythematosus. Arthritis Rheumatol. 2019, 71, 1135–1140. [Google Scholar] [CrossRef]
- Charles, N.; Hardwick, D.; Daugas, E.; Illei, G.G.; Rivera, J. Basophils and the T helper 2 environment can promote the development of lupus nephritis. Nat. Med. 2010, 16, 701–707. [Google Scholar] [CrossRef]
- Dema, B.; Pellefigues, C.; Hasni, S.; Gault, N.; Jiang, C.; Ricks, T.K.; Bonelli, M.M.; Scheffel, J.; Sacré, K.; Jablonski, M.; et al. Autoreactive IgE Is Prevalent in Systemic Lupus Erythematosus and Is Associated with Increased Disease Activity and Nephritis. PLoS ONE 2014, 9, e90424. [Google Scholar] [CrossRef]
- Atta, A.M.; Santiago, M.B.; Guerra, F.G.; Pereira, M.M.; Atta, M.L.B.S. Autoimmune Response of IgE Antibodies to Cellular Self-Antigens in Systemic Lupus Erythematosus. Int. Arch. Allergy Immunol. 2010, 152, 401–406. [Google Scholar] [CrossRef]
- Dema, B.; Charles, N.; Pellefigues, C.; Ricks, T.K.; Suzuki, R.; Jiang, C.; Scheffel, J.; Hasni, S.; Hoffman, V.; Jablonski, M.; et al. Immunoglobulin E plays an immunoregulatory role in lupus. J. Exp. Med. 2014, 211, 2159–2168. [Google Scholar] [CrossRef] [PubMed]
- Chang, T.W.; Chen, C.; Lin, C.-J.; Metz, M.; Church, M.K.; Maurer, M. The potential pharmacologic mechanisms of omalizumab in patients with chronic spontaneous urticaria. J. Allergy Clin. Immunol. 2015, 135, 337–342. [Google Scholar] [CrossRef]
- Gasser, P.; Tarchevskaya, S.S.; Guntern, P.; Brigger, D.; Ruppli, R.; Zbären, N.; Kleinboelting, S.; Heusser, C.; Jardetzky, T.S.; Eggel, A. The mechanistic and functional profile of the therapeutic anti-IgE antibody ligelizumab differs from omalizumab. Nat. Commun. 2020, 11, 165. [Google Scholar] [CrossRef]
- Gauvreau, G.M.; Arm, J.P.; Boulet, L.-P.; Leigh, R.; Cockcroft, D.W.; Davis, B.E.; Mayers, I.; FitzGerald, J.M.; Dahlen, B.; Killian, K.J.; et al. Efficacy and safety of multiple doses of QGE031 (ligelizumab) versus omalizumab and placebo in inhibiting allergen-induced early asthmatic responses. J. Allergy Clin. Immunol. 2016, 138, 1051–1059. [Google Scholar] [CrossRef]
- Trischler, J.; Bottoli, I.; Janocha, R.; Heusser, C.; Jaumont, X.; Lowe, P.; Gautier, A.; Pethe, A.; Woessner, R.; Zerwes, H.; et al. Ligelizumab treatment for severe asthma: Learnings from the clinical development programme. Clin. Transl. Immunol. 2021, 10, e1255. [Google Scholar] [CrossRef] [PubMed]
- Maurer, M.; Giménez-Arnau, A.M.; Sussman, G.; Metz, M.; Baker, D.R.; Bauer, A.; Bernstein, J.A.; Brehler, R.; Chu, C.-Y.; Chung, W.-H.; et al. Ligelizumab for Chronic Spontaneous Urticaria. N. Engl. J. Med. 2019, 381, 1321–1332. [Google Scholar] [CrossRef] [PubMed]
- Ligelizumab Breakthrough Therapy Designation. Available online: https://www.novartis.com/news/media-releases/novartis-ligelizumab-qge031-receives-fda-breakthrough-therapy-designation-patients-chronic-spontaneous-urticaria-csu (accessed on 20 February 2023).
- Ligelizumab CSU Phase III. Available online: https://www.novartis.com/news/media-releases/novartis-provides-update-phase-iii-ligelizumab-qge031-studies-chronic-spontaneous-urticaria-csu (accessed on 20 February 2023).
- Kuo, B.-S.; Li, C.-H.; Chen, J.-B.; Shiung, Y.-Y.; Chu, C.-Y.; Lee, C.-H.; Liu, Y.-J.; Kuo, J.-H.; Hsu, C.; Su, H.-W.; et al. IgE-neutralizing UB-221 mAb, distinct from omalizumab and ligelizumab, exhibits CD23-mediated IgE downregulation and relieves urticaria symptoms. J. Clin. Investig. 2022, 132, e157765. [Google Scholar] [CrossRef] [PubMed]
- Joubert, M.K.; Deshpande, M.; Yang, J.; Reynolds, H.; Bryson, C.; Fogg, M.; Baker, M.P.; Herskovitz, J.; Goletz, T.J.; Zhou, L.; et al. Use of In Vitro Assays to Assess Immunogenicity Risk of Antibody-Based Biotherapeutics. PLoS ONE 2016, 11, e0159328. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-H.; Jeun, E.-J.; Hong, C.-P.; Kim, S.-H.; Jang, M.S.; Lee, E.-J.; Moon, S.J.; Yun, C.H.; Im, S.-H.; Jeong, S.-G.; et al. Extracellular vesicle–derived protein from Bifidobacterium longum alleviates food allergy through mast cell suppression. J. Allergy Clin. Immunol. 2016, 137, 507–516.e8. [Google Scholar] [CrossRef] [PubMed]
- Plum, T.; Wang, X.; Rettel, M.; Krijgsveld, J.; Feyerabend, T.B.; Rodewald, H.-R. Human Mast Cell Proteome Reveals Unique Lineage, Putative Functions, and Structural Basis for Cell Ablation. Immunity 2020, 52, 404–416.e5. [Google Scholar] [CrossRef]
- Dondalska, A.; Rönnberg, E.; Ma, H.; Pålsson, S.A.; Magnusdottir, E.; Gao, T.; Adam, L.; Lerner, E.A.; Nilsson, G.; Lagerström, M.; et al. Amelioration of Compound 48/80-Mediated Itch and LL-37-Induced Inflammation by a Single-Stranded Oligonucleotide. Front. Immunol. 2020, 11, 559589. [Google Scholar] [CrossRef]
- Suzuki, Y.; Liu, S.; Ogasawara, T.; Sawasaki, T.; Takasaki, Y.; Yorozuya, T.; Mogi, M. A novel MRGPRX2-targeting antagonistic DNA aptamer inhibits histamine release and prevents mast cell-mediated anaphylaxis. Eur. J. Pharmacol. 2020, 878, 173104. [Google Scholar] [CrossRef]
- Azimi, E.; Reddy, V.B.; Shade, K.-T.C.; Anthony, R.M.; Talbot, S.; Pereira, P.J.S.; Lerner, E.A. Dual action of neurokinin-1 antagonists on Mas-related GPCRs. J. Clin. Investig. Insight 2016, 1, e89362. [Google Scholar] [CrossRef]
- Ogasawara, H.; Furuno, M.; Edamura, K.; Noguchi, M. Novel MRGPRX2 antagonists inhibit IgE-independent activation of human umbilical cord blood-derived mast cells. J. Leukoc. Biol. 2019, 106, 1069–1077. [Google Scholar] [CrossRef]
- Gulbransen, B.D.; Bashashati, M.; Hirota, S.A.; Gui, X.; Roberts, J.A.; MacDonald, J.; Muruve, D.A.; McKay, D.M.; Beck, P.L.; Mawe, G.M.; et al. Activation of neuronal P2X7 receptor–pannexin-1 mediates death of enteric neurons during colitis. Nat. Med. 2012, 18, 600–604. [Google Scholar] [CrossRef] [PubMed]
- Wan, P.; Liu, X.; Xiong, Y.; Ren, Y.; Chen, J.; Lu, N.; Guo, Y.; Bai, A. Extracellular ATP mediates inflammatory responses in colitis via P2 × 7 receptor signaling. Sci. Rep. 2016, 6, 19108. [Google Scholar] [CrossRef]
- Marques, C.C.; Castelo-Branco, M.T.; Pacheco, R.G.; Buongusto, F.; Rosário, A.D.; Schanaider, A.; Coutinho-Silva, R.; de Souza, H.S. Prophylactic systemic P2X7 receptor blockade prevents experimental colitis. Biochim. Biophys. Acta BBA Mol. Basis Dis. 2014, 1842, 65–78. [Google Scholar] [CrossRef]
- Eser, A.; Colombel, J.-F.; Rutgeerts, P.; Vermeire, S.; Vogelsang, H.; Braddock, M.; Persson, T.; Reinisch, W. Safety and Efficacy of an Oral Inhibitor of the Purinergic Receptor P2X7 in Adult Patients with Moderately to Severely Active Crohn’s Disease: A Randomized Placebo-controlled, Double-blind, Phase IIa Study. Inflamm. Bowel Dis. 2015, 21, 2247–2253. [Google Scholar] [CrossRef] [PubMed]
- Kolkhir, P.; Elieh-Ali-Komi, D.; Metz, M.; Siebenhaar, F.; Maurer, M. Understanding human mast cells: Lesson from therapies for allergic and non-allergic diseases. Nat. Rev. Immunol. 2022, 22, 294–308. [Google Scholar] [CrossRef] [PubMed]
| Omalizumab | Ligelizumab | UB-221 | IgETRAP | |
|---|---|---|---|---|
| Drug form | Anti-IgE IgG1 Ab | Anti-IgE IgG1 Ab | Anti-IgE IgG1 Ab | FcεRIα–IgD/IgG4 Fc fusion protein |
| Binding domain of IgE | Cε3 domain | Cε3 domain | Cε3 domain | Cε3 domain and Cε2–Cε3 linker region |
| Fold increase in IgE affinity compared to omalizumab | 1-fold | 14-fold | 4-fold | 69-fold |
| Potential risk of low affinity FcγRs-associated side effects | + | + | + | − |
| Potential risk of C1q-associated side effects | + | + | + | − |
| Inhibition of CD23–IgE interaction | + | + | − | ? |
| Inhibition of IgE production | − | − | + | ? |
| Reference | [108] | [108] | [108] | [37,86] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yang, B.-G.; Kim, A.-R.; Lee, D.; An, S.B.; Shim, Y.A.; Jang, M.H. Degranulation of Mast Cells as a Target for Drug Development. Cells 2023, 12, 1506. https://doi.org/10.3390/cells12111506
Yang B-G, Kim A-R, Lee D, An SB, Shim YA, Jang MH. Degranulation of Mast Cells as a Target for Drug Development. Cells. 2023; 12(11):1506. https://doi.org/10.3390/cells12111506
Chicago/Turabian StyleYang, Bo-Gie, A-Ram Kim, Dajeong Lee, Seong Beom An, Yaein Amy Shim, and Myoung Ho Jang. 2023. "Degranulation of Mast Cells as a Target for Drug Development" Cells 12, no. 11: 1506. https://doi.org/10.3390/cells12111506
APA StyleYang, B.-G., Kim, A.-R., Lee, D., An, S. B., Shim, Y. A., & Jang, M. H. (2023). Degranulation of Mast Cells as a Target for Drug Development. Cells, 12(11), 1506. https://doi.org/10.3390/cells12111506





