Structure and Development of the Legume-Rhizobial Symbiotic Interface in Infection Threads
Abstract
1. Introduction
2. Molecular Dialogue
3. Attachment
3.1. Attachment of Rhizobia
3.2. Curling of Root Hairs
4. Invasion of Host Cells
4.1. Initiation of the Infection Thread
4.2. The Nodule Primordium and Nodule Meristem
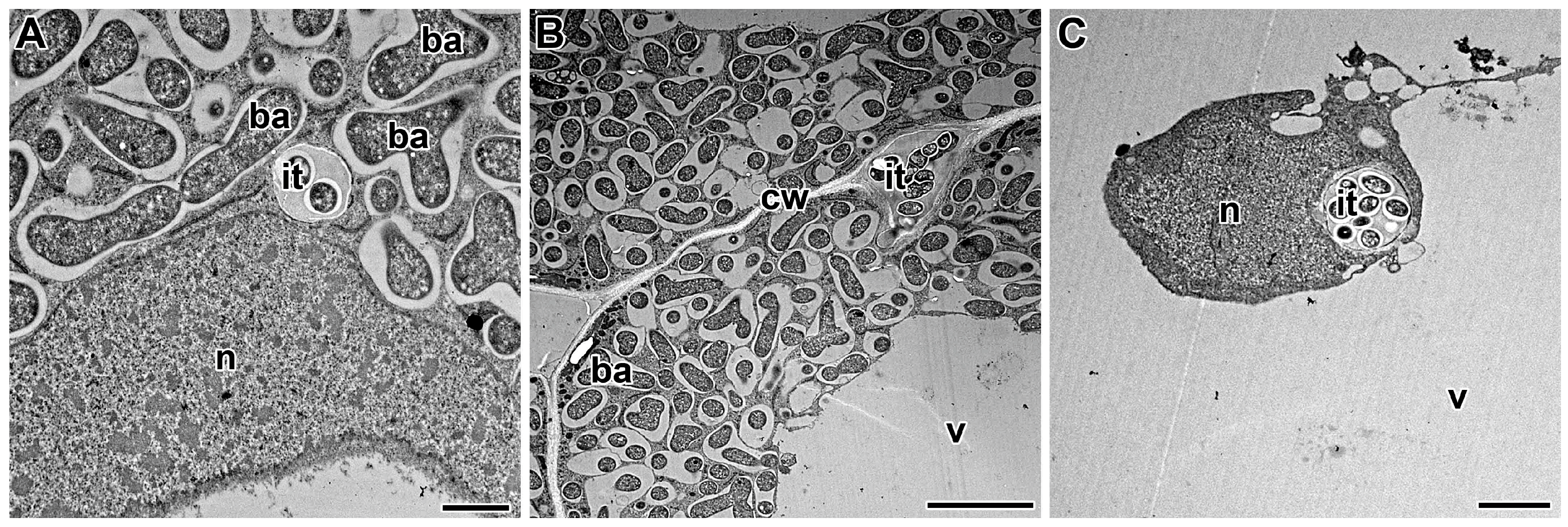
5. Propagation of the Infection Thread
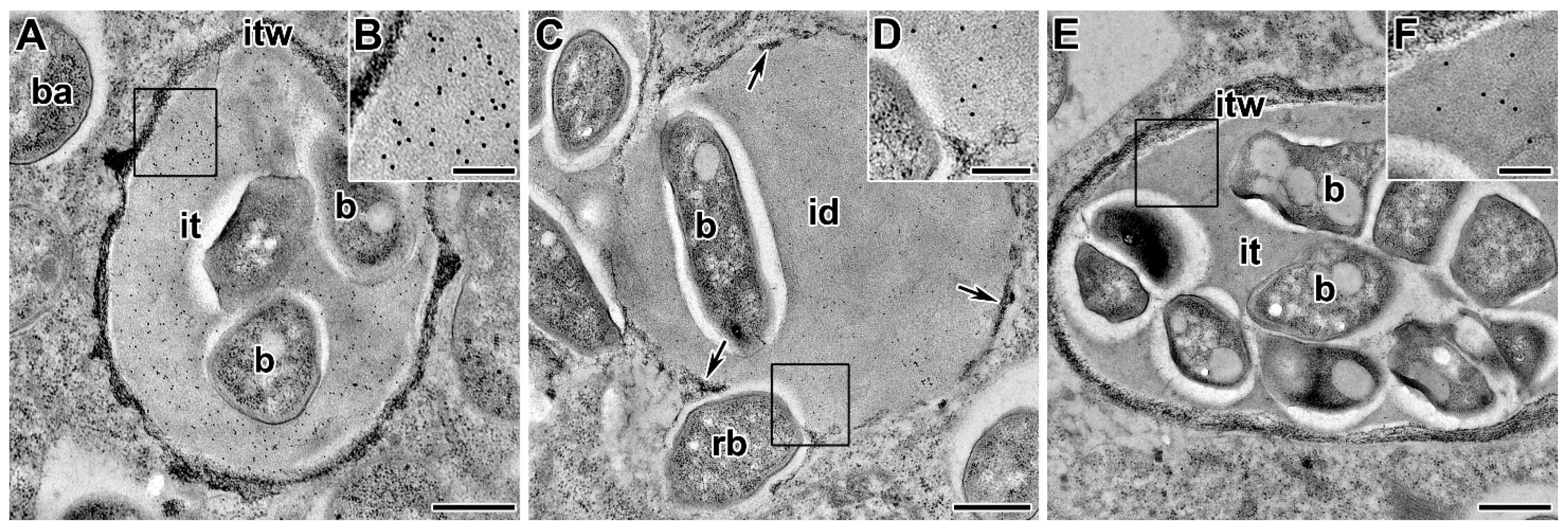
5.1. Infection Thread Wall
5.1.1. Enzymes Involved in the Growth of the Infection Thread
5.1.2. Polysaccharides and Proteins of the Infection Thread Wall
5.2. Infection Thread Matrix
5.3. ROS and NO
5.4. Defense Reactions
6. Release of Bacteria from Infection Threads
7. Nodule Senescence and Release of Bacteria
8. Environmental Influences
9. Conclusions and Perspectives
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Liu, S.; Ratet, P.; Magne, K. Nodule diversity, evolution, organogenesis and identity. In Advances in Botanical Research; Frendo, P., Frugier, F., Masson-Boivin, C., Eds.; Academic Press: London, UK; San Diego, CA, USA; Waltham, MA, USA; Oxford, UK, 2020; Volume 94, pp. 119–148. [Google Scholar]
- Forest, F.; Chase, M.W.; Eurosid, I. The Timetree of Life; Hedges, S.B., Kumar, S., Eds.; Oxford University Press: New York, NY, USA, 2009; pp. 188–196. [Google Scholar]
- Van Rhijn, P.; Vanderleyden, J. The Rhizobium-plant symbiosis. Microbiol. Rev. 1995, 59, 124–142. [Google Scholar] [CrossRef] [PubMed]
- Pawlowski, K.; Sirrenberg, A. Symbiosis between Frankia and actinorhizal plants: Root nodules of non-legumes. Indian J. Exp. Biol. 2003, 41, 1165–1183. [Google Scholar] [PubMed]
- Franche, C.; Lindström, K.; Elmerich, C. Nitrogen-fixing bacteria associated with leguminous and non-leguminous plants. Plant Soil 2009, 321, 35–59. [Google Scholar] [CrossRef]
- Zipfel, C.; Oldroyd, G.E.D. Plant signalling in symbiosis and immunity. Nature 2017, 543, 328–336. [Google Scholar] [CrossRef] [PubMed]
- Ibáñez, F.; Wall, L.; Fabra, A. Starting points in plant-bacteria nitrogen-fixing symbioses: Intercellular invasion of the roots. J. Exp. Bot. 2016, 68, 1905–1918. [Google Scholar] [CrossRef] [PubMed]
- Sprent, J.I. Evolving ideas of legume evolution and diversity: A taxonomic perspective on the occurrence of nodulation. New Phytol. 2007, 174, 11–25. [Google Scholar] [CrossRef]
- Sprent, J.I.; de Faria, S.M. Mechanisms of infection of plants by nitrogen fixing organisms. Plant Soil 1988, 110, 157–165. [Google Scholar] [CrossRef]
- Sprent, J.I.; James, E.K. Legume evolution: Where do nodules and mycorrhizas fit in? Plant Physiol. 2007, 144, 575–581. [Google Scholar] [CrossRef] [PubMed]
- Sharma, V.; Bhattacharyya, S.; Kumar, R.; Kumar, A.; Ibañez, F.; Wang, J.; Guo, B.; Sudini, H.K.; Gopalakrishnan, S.; DasGupta, M.; et al. Molecular basis of root nodule symbiosis between Bradyrhizobium and ‘crack-entry’ legume groundnut (Arachis hypogaea L.). Plants 2020, 9, 276. [Google Scholar] [CrossRef]
- Subba-Rao, N.S.; Mateos, P.F.; Baker, D.; Stuart Pankratz, H.; Palma, J.; Dazzo, F.B.; Sprent, J.I. The unique root-nodule symbiosis between Rhizobium and the aquatic legume, Neptunia natans (L. f.) Druce. Planta 1995, 196, 311–320. [Google Scholar] [CrossRef]
- Sprent, J.I.; Ardley, J.; James, E.K. Biogeography of nodulated legumes and their nitrogen-fixing symbionts. New Phytol. 2017, 215, 40–56. [Google Scholar] [CrossRef] [PubMed]
- Ward, H.M. On the tubercular swellings on the roots of Vicia faba. Proc. R. Soc. Lond. 1887, 42, 539–562. [Google Scholar] [CrossRef]
- McCoy, E. Infection by bact. radicicola in relation to the microchemistry of the host’s cell walls. Proc. R. Soc. Lond. Ser. B Contain. Pap. Biol. Character 1932, 110, 514–533. [Google Scholar] [CrossRef]
- Allen, E.K.; Allen, O.N. Biological aspects of symbiotic nitrogen fixation. In Der Stickstoffumsatz/Nitrogen Metabolism; Allen, E.K., Ed.; Springer: Berlin/Heidelberg, Germany, 1958; Volume 8, pp. 48–118. [Google Scholar]
- Brewin, N.J. Plant cell wall remodelling in the Rhizobium–legume symbiosis. Crit. Rev. Plant Sci. 2004, 23, 293–316. [Google Scholar] [CrossRef]
- Cole, R.A.; Fowler, J.E. Polarized growth: Maintaining focus on the tip. Curr. Opin. Plant Biol. 2006, 9, 579–588. [Google Scholar] [CrossRef] [PubMed]
- Fournier, J.; Timmers, A.C.J.; Sieberer, B.J.; Jauneau, A.; Chabaud, M.; Barker, D.G. Mechanism of infection thread elongation in root hairs of Medicago truncatula and dynamic interplay with associated rhizobial colonization. Plant Physiol. 2008, 148, 1985–1995. [Google Scholar] [CrossRef]
- Fournier, J.; Teillet, A.; Chabaud, M.; Ivanov, S.; Genre, A.; Limpens, E.; de Carvalho-Niebel, F.; Barker, D.G. Remodeling of the infection chamber before infection thread formation reveals a two-step mechanism for rhizobial entry into the host legume root hair. Plant Physiol. 2015, 167, 1233–1242. [Google Scholar] [CrossRef]
- Liu, C.-W.; Breakspear, A.; Stacey, N.; Findlay, K.; Nakashima, J.; Ramakrishnan, K.; Liu, M.; Xie, F.; Endre, G.; de Carvalho-Niebel, F.; et al. A protein complex required for polar growth of rhizobial infection threads. Nat. Commun. 2019, 10, 2848. [Google Scholar] [CrossRef]
- Brewin, N.J. Development of the legume root nodule. Ann. Rev. Cell Biol. 1991, 7, 191–226. [Google Scholar] [CrossRef]
- Perret, X.; Staehelin, C.; Broughton, W.J. Molecular basis of symbiotic promiscuity. Microbiol. Mol. Biol. Rev. 2000, 64, 180–201. [Google Scholar] [CrossRef] [PubMed]
- Spaink, H.P. Root nodulation and infection factors produced by rhizobial bacteria. Ann. Rev. Microbiol. 2000, 54, 257–288. [Google Scholar] [CrossRef] [PubMed]
- Fraysse, N.; Couderc, F.; Poinsot, V. Surface polysaccharide involvement in establishing the rhizobium–legume symbiosis. Eur. J. Biochem. 2003, 270, 1365–1380. [Google Scholar] [CrossRef] [PubMed]
- Denarie, J.; Debelle, F.; Prome, J.-C. Rhizobium lipo-chitooligosaccharide nodulation factors: Signaling molecules mediating recognition and morphogenesis. Ann. Rev. Biochem. 1996, 65, 503–535. [Google Scholar] [CrossRef] [PubMed]
- Buhian, W.P.; Bensmihen, S. Mini-Review: Nod factor regulation of phytohormone signaling and homeostasis during rhizobia-legume symbiosis. Front. Plant Sci. 2018, 9, 1247. [Google Scholar] [CrossRef] [PubMed]
- Mbengue, M.D.; Hervé, C.; Debellé, F. Nod factor signaling in symbiotic nodulation. In Advances in Botanical Research; Frendo, P., Frugier, F., Masson-Boivin, C., Eds.; Academic Press: London, UK; San Diego, CA, USA; Waltham, MA, USA; Oxford, UK, 2020; Volume 94, pp. 1–39. [Google Scholar]
- Tsyganova, A.V.; Tsyganov, V.E. Plant genetic control over infection thread development during legume-Rhizobium symbiosis. In Symbiosis; Rigobelo, E.C., Ed.; IntechOpen: London, UK, 2018; pp. 23–52. [Google Scholar] [CrossRef]
- Roy, S.; Liu, W.; Nandety, R.S.; Crook, A.; Mysore, K.S.; Pislariu, C.I.; Frugoli, J.; Dickstein, R.; Udvardi, M.K. Celebrating 20 years of genetic discoveries in legume nodulation and symbiotic nitrogen fixation. Plant Cell 2020, 32, 15–41. [Google Scholar] [CrossRef] [PubMed]
- Tsyganov, V.E.; Tsyganova, A.V. Symbiotic regulatory genes controlling nodule development in Pisum sativum L. Plants 2020, 9, 1741. [Google Scholar] [CrossRef]
- Oldroyd, G.E. Speak, friend, and enter: Signalling systems that promote beneficial symbiotic associations in plants. Nat. Rev. Microbiol. 2013, 11, 252–263. [Google Scholar] [CrossRef]
- Goedhart, J.; Hink, M.A.; Visser, A.J.W.G.; Bisseling, T.; Gadella, T.W.J., Jr. In Vivo fluorescence correlation microscopy (FCM) reveals accumulation and immobilization of Nod factors in root hair cell walls. Plant J. 2000, 21, 109–119. [Google Scholar] [CrossRef] [PubMed]
- Felle, H.H.; Kondorosi, E.; Kondorosi, A.; Schultze, M. The role of ion fluxes in Nod factor signalling in Medicago sativa. Plant J. 1998, 13, 455–463. [Google Scholar] [CrossRef]
- Damiani, I.; Drain, A.; Guichard, M.; Balzergue, S.; Boscari, A.; Boyer, J.-C.; Brunaud, V.; Cottaz, S.; Rancurel, C.; Da Rocha, M.; et al. Nod factor effects on root hair-specific transcriptome of Medicago truncatula: Focus on plasma membrane transport systems and reactive oxygen species networks. Front. Plant Sci. 2016, 7, 794. [Google Scholar] [CrossRef]
- Ehrhardt, D.W.; Wais, R.; Long, S.R. Calcium spiking in plant root hairs responding to Rhizobium nodulation signals. Cell 1996, 85, 673–681. [Google Scholar] [CrossRef]
- Sieberer, B.J.; Chabaud, M.; Timmers, A.C.; Monin, A.; Fournier, J.; Barker, D.G. A nuclear-targeted cameleon demonstrates intranuclear Ca2+ spiking in Medicago truncatula root hairs in response to rhizobial nodulation factors. Plant Physiol. 2009, 151, 1197–1206. [Google Scholar] [CrossRef] [PubMed]
- Cárdenas, L.; Vidali, L.; Domınguez, J.; Pérez, H.; Sánchez, F.; Hepler, P.K.; Quinto, C. Rearrangement of actin microfilaments in plant root hairs responding to Rhizobium etli nodulation signals. Plant Physiol. 1998, 116, 871–877. [Google Scholar] [CrossRef] [PubMed]
- De Ruijter, N.C.; Bisseling, T.; Emons, A.M.C. Rhizobium Nod factors induce an increase in sub-apical fine bundles of actin filaments in Vicia sativa root hairs within minutes. Mol. Plant Microbe Interact. 1999, 12, 829–832. [Google Scholar] [CrossRef]
- Sieberer, B.; Emons, A.M.C. Cytoarchitecture and pattern of cytoplasmic streaming in root hairs of Medicago truncatula during development and deformation by nodulation factors. Protoplasma 2000, 214, 118–127. [Google Scholar] [CrossRef]
- Sieberer, B.J.; Timmers, A.C.; Emons, A.M.C. Nod factors alter the microtubule cytoskeleton in Medicago truncatula root hairs to allow root hair reorientation. Mol. Plant Microbe Interact. 2005, 18, 1195–1204. [Google Scholar] [CrossRef] [PubMed]
- D’Haeze, W.; Glushka, J.; De Rycke, R.; Holsters, M.; Carlson, R.W. Structural characterization of extracellular polysaccharides of Azorhizobium caulinodans and importance for nodule initiation on Sesbania rostrata. Mol. Microbiol. 2004, 52, 485–500. [Google Scholar] [CrossRef]
- Downie, J.A. The roles of extracellular proteins, polysaccharides and signals in the interactions of rhizobia with legume roots. FEMS Microbiol. Rev. 2010, 34, 150–170. [Google Scholar] [CrossRef]
- Wisniewski-Dyé, F.; Downie, J.A. Quorum-sensing in Rhizobium. Antonie Leeuwenhoek 2002, 81, 397–407. [Google Scholar] [CrossRef]
- Rinaudi, L.V.; Giordano, W. An integrated view of biofilm formation in rhizobia. FEMS Microbiol. Lett. 2010, 304, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Navarro, D.N.; Dardanelli, M.S.; Ruíz-Saínz, J.E. Attachment of bacteria to the roots of higher plants. FEMS Microbiol. Lett. 2007, 272, 127–136. [Google Scholar] [CrossRef]
- Hirsch, A.M. Role of lectins (and rhizobial exopolysaccharides) in legume nodulation. Curr. Opin. Plant Biol. 1999, 2, 320–326. [Google Scholar] [CrossRef]
- Lagarda-Diaz, I.; Guzman-Partida, A.M.; Vazquez-Moreno, L. Legume lectins: Proteins with diverse applications. Int. J. Mol. Sci. 2017, 18, 1242. [Google Scholar] [CrossRef] [PubMed]
- Albareda, M.; Dardanelli, M.S.; Sousa, C.; Megías, M.; Temprano, F.; Rodríguez-Navarro, D.N. Factors affecting the attachment of rhizospheric bacteria to bean and soybean roots. FEMS Microbiol. Lett. 2006, 259, 67–73. [Google Scholar] [CrossRef] [PubMed]
- Laus, M.C.; Logman, T.J.; Lamers, G.E.; Van Brussel, A.A.N.; Carlson, R.W.; Kijne, J.W. A novel polar surface polysaccharide from Rhizobium leguminosarum binds host plant lectin. Mol. Microbiol. 2006, 59, 1704–1713. [Google Scholar] [CrossRef] [PubMed]
- Williams, A.; Wilkinson, A.; Krehenbrink, M.; Russo, D.M.; Zorreguieta, A.; Downie, J.A. Glucomannan-mediated attachment of Rhizobium leguminosarum to pea root hairs is required for competitive nodule infection. J. Bacteriol. 2008, 190, 4706–4715. [Google Scholar] [CrossRef] [PubMed]
- Lodeiro, A.b.R.; Favelukes, G. Early interactions of Bradyrhizobium japonicum and soybean roots: Specificity in the process of adsorption. Soil Biol. Biochem. 1999, 31, 1405–1411. [Google Scholar] [CrossRef]
- Gibson, K.E.; Kobayashi, H.; Walker, G.C. Molecular determinants of a symbiotic chronic infection. Annu. Rev. Genet. 2008, 42, 413–441. [Google Scholar] [CrossRef]
- Robledo, M.; Jiménez-Zurdo, J.I.; Velázquez, E.; Trujillo, M.E.; Zurdo-Piñeiro, J.L.; Ramírez-Bahena, M.H.; Ramos, B.; Díaz-Mínguez, J.M.; Dazzo, F.; Martínez-Molina, E.; et al. Rhizobium cellulase CelC2 is essential for primary symbiotic infection of legume host roots. Proc. Natl. Acad. Sci. USA 2008, 105, 7064–7069. [Google Scholar] [CrossRef]
- Navarro-Gochicoa, M.-T.; Camut, S.; Timmers, A.C.J.; Niebel, A.; Hervé, C.; Boutet, E.; Bono, J.-J.; Imberty, A.; Cullimore, J.V. Characterization of four lectin-like receptor kinases expressed in roots of Medicago truncatula. Structure, location, regulation of expression, and potential role in the symbiosis with Sinorhizobium meliloti. Plant Physiol. 2003, 133, 1893–1910. [Google Scholar] [CrossRef]
- De Hoff, P.L.; Brill, L.M.; Hirsch, A.M. Plant lectins: The ties that bind in root symbiosis and plant defense. Mol. Genet. Genom. 2009, 282, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Swart, S.; Logman, T.J.; Smit, G.; Lugtenberg, B.J.; Kijne, J.W. Purification and partial characterization of a glycoprotein from pea (Pisum sativum) with receptor activity for rhicadhesin, an attachment protein of Rhizobiaceae. Plant Mol. Biol. 1994, 24, 171–183. [Google Scholar] [CrossRef] [PubMed]
- Dardanelli, M.; Angelini, J.; Fabra, A. A calcium-dependent bacterial surface protein is involved in the attachment of rhizobia to peanut roots. Can. J. Microbiol. 2003, 49, 399–405. [Google Scholar] [CrossRef] [PubMed]
- Ausmees, N.; Jacobsson, K.; Lindberg, M. A unipolarly located, cell-surface-associated agglutinin, RapA, belongs to a family of Rhizobium-adhering proteins (Rap) in Rhizobium leguminosarum bv. trifolii. Microbiology 2001, 147, 549–559. [Google Scholar] [CrossRef] [PubMed]
- Mongiardini, E.J.; Ausmees, N.; Pérez-Giménez, J.; Althabegoiti, M.J.; Ignacio Quelas, J.; López-García, S.L.; Lodeiro, A.R. The rhizobial adhesion protein RapA1 is involved in adsorption of rhizobia to plant roots but not in nodulation. FEMS Microbiol. Ecol. 2008, 65, 279–288. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Russo, D.M.; Williams, A.; Edwards, A.; Posadas, D.M.; Finnie, C.; Dankert, M.; Downie, J.A.; Zorreguieta, A. Proteins exported via the PrsD-PrsE type I secretion system and the acidic exopolysaccharide are involved in biofilm formation by Rhizobium leguminosarum. J. Bacteriol. 2006, 188, 4474–4486. [Google Scholar] [CrossRef] [PubMed]
- Krehenbrink, M.; Downie, J.A. Identification of protein secretion systems and novel secreted proteins in Rhizobium leguminosarum bv. viciae. BMC Genom. 2008, 9, 55. [Google Scholar] [CrossRef]
- Talukdar, T.; Gorecka, K.M.; de Carvalho-Niebel, F.; Downie, J.A.; Cullimore, J.; Pikula, S. Annexins - calcium- and membrane-binding proteins in the plant kingdom: Potential role in nodulation and mycorrhization in Medicago truncatula. Acta Biochim. Pol. 2009, 56, 199–210. [Google Scholar] [CrossRef]
- Manthey, K.; Krajinski, F.; Hohnjec, N.; Firnhaber, C.; Pühler, A.; Perlick, A.M.; Küster, H. Transcriptome profiling in root nodules and arbuscular mycorrhiza identifies a collection of novel genes induced during Medicago truncatula root endosymbioses. Mol. Plant Microbe Interact. 2004, 17, 1063–1077. [Google Scholar] [CrossRef]
- Edwards, A.; Heckmann, A.B.; Yousafzai, F.; Duc, G.; Downie, J.A. Structural implications of mutations in the pea SYM8 symbiosis gene, the DMI1 ortholog, encoding a predicted ion channel. Mol. Plant Microbe Interact. 2007, 20, 1183–1191. [Google Scholar] [CrossRef]
- Xie, F.; Williams, A.; Edwards, A.; Downie, J.A. A plant arabinogalactan-like glycoprotein promotes a novel type of polar surface attachment by Rhizobium leguminosarum. Mol. Plant Microbe Interact. 2012, 25, 250–258. [Google Scholar] [CrossRef] [PubMed]
- Esseling, J.J.; Lhuissier, F.G.; Emons, A.M.C. Nod factor-induced root hair curling: Continuous polar growth towards the point of Nod factor application. Plant Physiol. 2003, 132, 1982–1988. [Google Scholar] [CrossRef] [PubMed]
- Dazzo, F.B.; Orgambide, G.G.; Philip-Hollingsworth, S.; Hollingsworth, R.I.; Ninke, K.O.; Salzwedel, J.L. Modulation of development, growth dynamics, wall crystallinity, and infection sites in white clover root hairs by membrane chitolipooligosaccharides from Rhizobium leguminosarum biovar trifolii. J. Bacteriol. 1996, 178, 3621–3627. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gage, D.J.; Margolin, W. Hanging by a thread: Invasion of legume plants by rhizobia. Curr. Opin. Microbiol. 2000, 3, 613–617. [Google Scholar] [CrossRef]
- Carol, R.J.; Dolan, L. Building a hair: Tip growth in Arabidopsis thaliana root hairs. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2002, 357, 815–821. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mollet, J.-C.; Leroux, C.; Dardelle, F.; Lehner, A. Cell wall composition, biosynthesis and remodeling during pollen tube growth. Plants 2013, 2, 107–147. [Google Scholar] [CrossRef]
- Galway, M.E.; Heckman, J.W.; Schiefelbein, J.W. Growth and ultrastructure of Arabidopsis root hairs: The rhd3 mutation alters vacuole enlargement and tip growth. Planta 1997, 201, 209–218. [Google Scholar] [CrossRef]
- Grierson, C.S.; Roberts, K.; Feldmann, K.A.; Dolan, L. The COW1 locus of Arabidopsis acts after RHD2, and in parallel with RHD3 and TIP1, to determine the shape, rate of elongation, and number of root hairs produced from each site of hair formation. Plant Physiol. 1997, 115, 981–990. [Google Scholar] [CrossRef][Green Version]
- Peiter, E.; Sun, J.; Heckmann, A.B.; Venkateshwaran, M.; Riely, B.K.; Otegui, M.S.; Edwards, A.; Freshour, G.; Hahn, M.G.; Cook, D.R.; et al. The Medicago truncatula DMI1 protein modulates cytosolic calcium signaling. Plant Physiol. 2007, 145, 192–203. [Google Scholar] [CrossRef]
- Bibikova, T.N.; Zhigilei, A.; Gilroy, S. Root hair growth in Arabidopsis thaliana is directed by calcium and an endogenous polarity. Planta 1997, 203, 495–505. [Google Scholar] [CrossRef]
- Yokota, K.; Fukai, E.; Madsen, L.H.; Jurkiewicz, A.; Rueda, P.; Radutoiu, S.; Held, M.; Hossain, M.S.; Szczyglowski, K.; Morieri, G. Rearrangement of actin cytoskeleton mediates invasion of Lotus japonicus roots by Mesorhizobium loti. Plant Cell 2009, 21, 267–284. [Google Scholar] [CrossRef] [PubMed]
- Su, C.; Klein, M.-L.; Hernández-Reyes, C.; Batzenschlager, M.; Ditengou, F.A.; Lace, B.; Keller, J.; Delaux, P.-M.; Ott, T. The Medicago truncatula DREPP protein triggers microtubule fragmentation in membrane nanodomains during symbiotic infections. Plant Cell 2020, 32, 1689–1702. [Google Scholar] [CrossRef] [PubMed]
- Murray, J.D. Invasion by invitation: Rhizobial infection in legumes. Mol. Plant Microbe Interact. 2011, 24, 631–639. [Google Scholar] [CrossRef] [PubMed]
- Janczarek, M.; Rachwał, K.; Marzec, A.; Grządziel, J.; Palusińska-Szysz, M. Signal molecules and cell-surface components involved in early stages of the legume–rhizobium interactions. Appl. Soil Ecol. 2015, 85, 94–113. [Google Scholar] [CrossRef]
- Peleg-Grossman, S.; Volpin, H.; Levine, A. Root hair curling and Rhizobium infection in Medicago truncatula are mediated by phosphatidylinositide-regulated endocytosis and reactive oxygen species. J. Exp. Bot. 2007, 58, 1637–1649. [Google Scholar] [CrossRef] [PubMed]
- Lei, M.-J.; Wang, Q.; Li, X.; Chen, A.; Luo, L.; Xie, Y.; Li, G.; Luo, D.; Mysore, K.S.; Wen, J.; et al. The small GTPase ROP10 of Medicago truncatula is required for both tip growth of root hairs and Nod factor-induced root hair deformation. Plant Cell 2015, 27, 806–822. [Google Scholar] [CrossRef] [PubMed]
- Sahlman, K.; Fåhraeus, G. An electron microscope study of root-hair infection by Rhizobium. Microbiology 1963, 33, 425–427. [Google Scholar] [CrossRef] [PubMed]
- Gage, D.J. Infection and invasion of roots by symbiotic, nitrogen-fixing rhizobia during nodulation of temperate legumes. Microbiol. Mol. Biol. Rev. 2004, 68, 280–300. [Google Scholar] [CrossRef] [PubMed]
- Walker, S.A.; Viprey, V.; Downie, J.A. Dissection of nodulation signaling using pea mutants defective for calcium spiking induced by Nod factors and chitin oligomers. Proc. Natl. Acad. Sci. USA 2000, 97, 13413–13418. [Google Scholar] [CrossRef] [PubMed]
- Catoira, R.; Timmers, A.; Maillet, F.; Galera, C.; Penmetsa, R.V.; Cook, D.; Dénarié, J.; Gough, C. The HCL gene of Medicago truncatula controls Rhizobium-induced root hair curling. Development 2001, 128, 1507–1518. [Google Scholar] [CrossRef]
- Sogawa, A.; Yamazaki, A.; Yamasaki, H.; Komi, M.; Manabe, T.; Tajima, S.; Hayashi, M.; Nomura, M. SNARE Proteins LjVAMP72a and LjVAMP72b are required for root symbiosis and root hair formation in Lotus japonicus. Front. Plant Sci. 2019, 9, 1992. [Google Scholar] [CrossRef] [PubMed]
- Yano, K.; Shibata, S.; Chen, W.-L.; Sato, S.; Kaneko, T.; Jurkiewicz, A.; Sandal, N.; Banba, M.; Imaizumi-Anraku, H.; Kojima, T.; et al. CERBERUS, a novel U-box protein containing WD-40 repeats, is required for formation of the infection thread and nodule development in the legume–Rhizobium symbiosis. Plant J. 2009, 60, 168–180. [Google Scholar] [CrossRef] [PubMed]
- Haney, C.H.; Long, S.R. Plant flotillins are required for infection by nitrogen-fixing bacteria. Proc. Natl. Acad. Sci. USA 2010, 107, 478–483. [Google Scholar] [CrossRef] [PubMed]
- Guan, D.; Stacey, N.; Liu, C.; Wen, J.; Mysore, K.S.; Torres-Jerez, I.; Vernié, T.; Tadege, M.; Zhou, C.; Wang, Z.-y.; et al. Rhizobial infection is associated with the development of peripheral vasculature in nodules of Medicago truncatula. Plant Physiol. 2013, 162, 107–115. [Google Scholar] [CrossRef] [PubMed]
- Pii, Y.; Molesini, B.; Pandolfini, T. The involvement of Medicago truncatula non-specific lipid transfer protein N5 in the control of rhizobial infection. Plant Signal. Behav. 2013, 8, e24836. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Suzaki, T.; Takeda, N.; Nishida, H.; Hoshino, M.; Ito, M.; Misawa, F.; Handa, Y.; Miura, K.; Kawaguchi, M. LACK OF SYMBIONT ACCOMMODATION controls intracellular symbiont accommodation in root nodule and arbuscular mycorrhizal symbiosis in Lotus japonicus. PLoS Genet. 2019, 15, e1007865. [Google Scholar] [CrossRef]
- Yano, K.; Yoshida, S.; Müller, J.; Singh, S.; Banba, M.; Vickers, K.; Markmann, K.; White, C.; Schuller, B.; Sato, S.; et al. CYCLOPS, a mediator of symbiotic intracellular accommodation. Proc. Natl. Acad. Sci. USA 2008, 105, 20540–20545. [Google Scholar] [CrossRef]
- Smit, P.; Raedts, J.; Portyanko, V.; Debellé, F.; Gough, C.; Bisseling, T.; Geurts, R. NSP1 of the GRAS protein family is essential for rhizobial Nod factor-induced transcription. Science 2005, 308, 1789–1791. [Google Scholar] [CrossRef]
- Kaló, P.; Gleason, C.; Edwards, A.; Marsh, J.; Mitra, R.M.; Hirsch, S.; Jakab, J.; Sims, S.; Long, S.R.; Rogers, J.; et al. Nodulation signaling in legumes requires NSP2, a member of the GRAS family of transcriptional regulators. Science 2005, 308, 1786–1789. [Google Scholar] [CrossRef]
- Middleton, P.H.; Jakab, J.; Penmetsa, R.V.; Starker, C.G.; Doll, J.; Kaló, P.; Prabhu, R.; Marsh, J.F.; Mitra, R.M.; Kereszt, A.; et al. An ERF transcription factor in Medicago truncatula that is essential for Nod factor signal transduction. Plant Cell 2007, 19, 1221–1234. [Google Scholar] [CrossRef]
- Borisov, A.Y.; Madsen, L.H.; Tsyganov, V.E.; Umehara, Y.; Voroshilova, V.A.; Batagov, A.O.; Sandal, N.; Mortensen, A.; Schauser, L.; Ellis, N.; et al. The Sym35 gene required for root nodule development in pea is an ortholog of Nin from Lotus japonicus. Plant Physiol. 2003, 131, 1009–1017. [Google Scholar] [CrossRef] [PubMed]
- Laloum, T.; Baudin, M.; Frances, L.; Lepage, A.; Billault-Penneteau, B.; Cerri, M.R.; Ariel, F.; Jardinaud, M.-F.; Gamas, P.; de Carvalho-Niebel, F.; et al. Two CCAAT-box-binding transcription factors redundantly regulate early steps of the legume-rhizobia endosymbiosis. Plant J. 2014, 79, 757–768. [Google Scholar] [CrossRef] [PubMed]
- Newman-Griffis, A.H.; del Cerro, P.; Charpentier, M.; Meier, I. Medicago LINC complexes function in nuclear morphology, nuclear movement, and root nodule symbiosis. Plant Physiol. 2019, 179, 491–506. [Google Scholar] [CrossRef] [PubMed]
- Nutman, P.S. The influence of the legume in root-nodule symbiosis. Biol. Rev. 1956, 31, 109–151. [Google Scholar] [CrossRef]
- Ljunggren, H.; Fåhraeus, G. Effect of Rhizobium polysaccharide on the formation of polygalacturonase in lucerne and clover. Nature 1959, 184, 1578–1579. [Google Scholar] [CrossRef]
- Dart, P.J.; Mercer, F.V. The legume rhizosphere. Archiv. Mikrobiol. 1964, 47, 344–378. [Google Scholar] [CrossRef]
- Napoli, C.A.; Hubbell, D.H. Ultrastructure of Rhizobium-induced infection threads in clover root hairs. Appl. Microbiol. 1975, 30, 1003–1009. [Google Scholar] [CrossRef]
- Callaham, D.A.; Torrey, J.G. The structural basis for infection of root hairs of Trifolium repens by Rhizobium. Can. J. Bot. 1981, 59, 1647–1664. [Google Scholar] [CrossRef]
- Turgeon, B.G.; Bauer, W.D. Ultrastructure of infection-thread development during the infection of soybean by Rhizobium japonicum. Planta 1985, 163, 328–349. [Google Scholar] [CrossRef]
- Fåhraeus, G. The infection of clover root hairs by nodule bacteria studied by a simple glass slide technique. J. Gen. Microbiol. 1957, 16, 374–381. [Google Scholar] [CrossRef]
- Fåhraeus, G.; Ljunggren, H. The possible significance of pectic enzymes in root hair infection by nodule bacteria. Physiol. Plant. 1959, 12, 145–154. [Google Scholar] [CrossRef]
- Mateos, P.F.; Jimenez-Zurdo, J.I.; Chen, J.; Squartini, A.S.; Haack, S.K.; Martinez-Molina, E.; Hubbell, D.H.; Dazzo, F.B. Cell-associated pectinolytic and cellulolytic enzymes in Rhizobium leguminosarum biovar trifolii. Appl. Environ. Microbiol. 1992, 58, 1816–1822. [Google Scholar] [CrossRef] [PubMed]
- Roberts, J.A.; Whitelaw, C.A.; Gonzalez-Carranza, Z.H.; McManus, M.T. Cell separation processes in plants—Models, mechanisms and manipulation. Ann. Bot. 2000, 86, 223–235. [Google Scholar] [CrossRef]
- Cosgrove, D.J.; Li, L.C.; Cho, H.-T.; Hoffmann-Benning, S.; Moore, R.C.; Blecker, D. The growing world of expansins. Plant Cell Physiol. 2002, 43, 1436–1444. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, J.A.; Coronado, C.; Pérez-Hormaeche, J.; Kondorosi, A.; Ratet, P.; Palomares, A.J. MsPG3, a Medicago sativa polygalacturonase gene expressed during the alfalfa–Rhizobium meliloti interaction. Proc. Natl. Acad. Sci. USA 1998, 95, 9687–9692. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Llorente, I.D.; Pérez-Hormaeche, J.; Dary, M.; Caviedes, M.A.; Kondorosi, A.; Ratet, P.; Palomares, A.J. Expression of MsPG3-GFP fusions in Medicago truncatula ‘hairy roots’ reveals preferential tip localization of the protein in root hairs. Eur. J. Biochem. 2003, 270, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Xie, F.; Murray, J.D.; Kim, J.; Heckmann, A.B.; Edwards, A.; Oldroyd, G.E.D.; Downie, J.A. Legume pectate lyase required for root infection by rhizobia. Proc. Natl. Acad. Sci. USA 2012, 109, 633–638. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Zhao, J.; Tan, Z.; Zeng, R.; Liao, H. GmEXPB2, a cell wall β-expansin, affects soybean nodulation through modifying root architecture and promoting nodule formation and development. Plant Physiol. 2015, 169, 2640–2653. [Google Scholar] [CrossRef]
- Perrine-Walker, F.M.; Lartaud, M.; Kouchi, H.; Ridge, R.W. Microtubule array formation during root hair infection thread initiation and elongation in the Mesorhizobium-Lotus symbiosis. Protoplasma 2014, 251, 1099–1111. [Google Scholar] [CrossRef]
- Tansengco, M.L.; Hayashi, M.; Kawaguchi, M.; Imaizumi-Anraku, H.; Murooka, Y. crinkle, a novel symbiotic mutant that affects the infection thread growth and alters the root hair, trichome, and seed development in Lotus japonicus. Plant Physiol. 2003, 131, 1054–1063. [Google Scholar] [CrossRef] [PubMed]
- Hossain, M.S.; Liao, J.; James, E.K.; Sato, S.; Tabata, S.; Jurkiewicz, A.; Madsen, L.H.; Stougaard, J.; Ross, L.; Szczyglowski, K. Lotus japonicus ARPC1 is required for rhizobial infection. Plant Physiol. 2012, 160, 917–928. [Google Scholar] [CrossRef] [PubMed]
- Miyahara, A.; Richens, J.; Starker, C.; Morieri, G.; Smith, L.; Long, S.; Downie, J.A.; Oldroyd, G.E.D. Conservation in function of a SCAR/WAVE component during infection thread and root hair growth in Medicago truncatula. Mol. Plant Microbe Interact. 2010, 23, 1553–1562. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Chen, A.-M.; Luo, L.; Sun, J.; Cao, L.-P.; Yu, G.-Q.; Zhu, J.-B.; Wang, Y.-Z. Characterization and expression analysis of Medicago truncatula ROP GTPase family during the early stage of symbiosis. J. Integr. Plant Biol. 2010, 52, 639–652. [Google Scholar] [CrossRef] [PubMed]
- Saarikangas, J.; Zhao, H.; Lappalainen, P. Regulation of the actin cytoskeleton-plasma membrane interplay by phosphoinositides. Physiol. Rev. 2010, 90, 259–289. [Google Scholar] [CrossRef] [PubMed]
- Ortega-Ortega, Y.; Carrasco-Castilla, J.; Juárez-Verdayes, M.A.; Toscano-Morales, R.; Fonseca-García, C.; Nava, N.; Cárdenas, L.; Quinto, C. Actin depolymerizing factor modulates rhizobial infection and nodule organogenesis in common bean. Int. J. Mol. Sci. 2020, 21, 1970. [Google Scholar] [CrossRef]
- Andrio, E.; Marino, D.; Marmeys, A.; de Segonzac, M.D.; Damiani, I.; Genre, A.; Huguet, S.; Frendo, P.; Puppo, A.; Pauly, N. Hydrogen peroxide-regulated genes in the Medicago truncatula–Sinorhizobium meliloti symbiosis. New Phytol. 2013, 198, 179–189. [Google Scholar] [CrossRef] [PubMed]
- Shaw, S.L.; Long, S.R. Nod factor inhibition of reactive oxygen efflux in a host legume. Plant Physiol. 2003, 132, 2196–2204. [Google Scholar] [CrossRef] [PubMed]
- Lohar, D.P.; Haridas, S.; Gantt, J.S.; VandenBosch, K.A. A transient decrease in reactive oxygen species in roots leads to root hair deformation in the legume–rhizobia symbiosis. New Phytol. 2007, 173, 39–49. [Google Scholar] [CrossRef] [PubMed]
- Cárdenas, L.; Quinto, C. Reactive oxygen species (ROS) as early signals in root hair cells responding to rhizobial nodulation factors. Plant Signal. Behav. 2008, 3, 1101–1102. [Google Scholar] [CrossRef]
- Puppo, A.; Pauly, N.; Boscari, A.; Mandon, K.; Brouquisse, R. Hydrogen peroxide and nitric oxide: Key regulators of the legume–Rhizobium and mycorrhizal symbioses. Antioxid. Redox Signal. 2013, 18, 2202–2219. [Google Scholar] [CrossRef] [PubMed]
- Montiel, J.; Arthikala, M.-K.; Cárdenas, L.; Quinto, C. Legume NADPH oxidases have crucial roles at different stages of nodulation. Int. J. Mol. Sci. 2016, 17, 680. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Abarca, F.; Herrera-Cervera, J.A.; Bueno, P.; Sanjuan, J.; Bisseling, T.; Olivares, J. Involvement of salicylic acid in the establishment of the Rhizobium meliloti-alfalfa symbiosis. Mol. Plant Microbe Interact. 1998, 11, 153–155. [Google Scholar] [CrossRef]
- Nagata, M.; Hashimoto, M.; Murakami, E.-i.; Shimoda, Y.; Shimoda-Sasakura, F.; Kucho, K.-i.; Suzuki, A.; Abe, M.; Higashi, S.; Uchiumi, T. A possible role of class 1 plant hemoglobin at the early stage of legume-rhizobium symbiosis. Plant Signal. Behav. 2009, 4, 202–204. [Google Scholar] [CrossRef] [PubMed]
- Del Giudice, J.; Cam, Y.; Damiani, I.; Fung-Chat, F.; Meilhoc, E.; Bruand, C.; Brouquisse, R.; Puppo, A.; Boscari, A. Nitric oxide is required for an optimal establishment of the Medicago truncatula–Sinorhizobium meliloti symbiosis. New Phytol. 2011, 191, 405–417. [Google Scholar] [CrossRef] [PubMed]
- Murakami, E.-i.; Nagata, M.; Shimoda, Y.; Kucho, K.-i.; Higashi, S.; Abe, M.; Hashimoto, M.; Uchiumi, T. Nitric oxide production induced in roots of Lotus japonicus by lipopolysaccharide from Mesorhizobium loti. Plant Cell Physiol. 2011, 52, 610–617. [Google Scholar] [CrossRef] [PubMed]
- Bindschedler, L.V.; Dewdney, J.; Blee, K.A.; Stone, J.M.; Asai, T.; Plotnikov, J.; Denoux, C.; Hayes, T.; Gerrish, C.; Davies, D.R.; et al. Peroxidase-dependent apoplastic oxidative burst in Arabidopsis required for pathogen resistance. Plant J. 2006, 47, 851–863. [Google Scholar] [CrossRef] [PubMed]
- Breakspear, A.; Liu, C.; Roy, S.; Stacey, N.; Rogers, C.; Trick, M.; Morieri, G.; Mysore, K.S.; Wen, J.; Oldroyd, G.E.; et al. The root hair “infectome” of Medicago truncatula uncovers changes in cell cycle genes and reveals a requirement for auxin signaling in rhizobial infection. Plant Cell 2014, 26, 4680–4701. [Google Scholar] [CrossRef] [PubMed]
- van Brussel, A.A.N.; Bakhuizen, R.; van Spronsen, P.C.; Spaink, H.P.; Tak, T.; Lugtenberg, B.J.J.; Kijne, J.W. Induction of pre-infection thread structures in the leguminous host plant by mitogenic lipo-oligosaccharides of Rhizobium. Science 1992, 257, 70–72. [Google Scholar] [CrossRef]
- Timmers, A.C.; Auriac, M.C.; Truchet, G. Refined analysis of early symbiotic steps of the Rhizobium-Medicago interaction in relationship with microtubular cytoskeleton rearrangements. Development 1999, 126, 3617–3628. [Google Scholar] [CrossRef]
- Tsyganov, V.E.; Voroshilova, V.A.; Priefer, U.B.; Borisov, A.Y.; Tikhonovich, I.A. Genetic dissection of the initiation of the infection process and nodule tissue development in the Rhizobium-pea (Pisum sativum L.) symbiosis. Ann. Bot. 2002, 89, 357–366. [Google Scholar] [CrossRef]
- Oldroyd, G.E.; Downie, J.A. Coordinating nodule morphogenesis with rhizobial infection in legumes. Ann. Rev. Plant Biol. 2008, 59, 519–546. [Google Scholar] [CrossRef] [PubMed]
- Madsen, L.H.; Tirichine, L.; Jurkiewicz, A.; Sullivan, J.T.; Heckmann, A.B.; Bek, A.S.; Ronson, C.W.; James, E.K.; Stougaard, J. The molecular network governing nodule organogenesis and infection in the model legume Lotus japonicus. Nat. Commun. 2010, 1, 10. [Google Scholar] [CrossRef] [PubMed]
- Robertson, J.G.; Wells, B.; Brewin, N.J.; Wood, E.; Knight, C.D.; Downie, J.A. The legume-Rhizobium symbiosis: A cell surface interaction. J. Cell Sci. Suppl. 1985, 2, 317–331. [Google Scholar] [CrossRef] [PubMed]
- Gage, D.J. Analysis of infection thread development using Gfp-and DsRed-expressing Sinorhizobium meliloti. J. Bacteriol. 2002, 184, 7042–7046. [Google Scholar] [CrossRef] [PubMed]
- Roth, L.; Stacey, G. Bacterium release into host cells of nitrogen-fixing soybean nodules: The symbiosome membrane comes from three sources. Eur. J. Cell Biol. 1989, 49, 13–23. [Google Scholar] [PubMed]
- Rathbun, E.A.; Naldrett, M.J.; Brewin, N.J. Identification of a family of extensin-like glycoproteins in the lumen of Rhizobium-induced infection threads in pea root nodules. Mol. Plant Microbe Interact. 2002, 15, 350–359. [Google Scholar] [CrossRef]
- Wisniewski, J.-P.; Rathbun, E.A.; Knox, J.P.; Brewin, N.J. Involvement of diamine oxidase and peroxidase in insolubilization of the extracellular matrix: Implications for pea nodule initiation by Rhizobium leguminosarum. Mol. Plant Microbe Interact. 2000, 13, 413–420. [Google Scholar] [CrossRef] [PubMed]
- Hérouart, D.; Baudouin, E.; Frendo, P.; Harrison, J.; Santos, R.; Jamet, A.; Van de Sype, G.; Touati, D.; Puppo, A. Reactive oxygen species, nitric oxide and glutathione: A key role in the establishment of the legume–Rhizobium symbiosis? Plant Physiol. Biochem. 2002, 40, 619–624. [Google Scholar] [CrossRef]
- Brewin, N.; Khodorenko, A.; Tsyganov, V.E.; Borisov, A.Y.; Tikhonovich, I.A.; Rathbun, E. Legume AGP-extensins in Rhizobium infection. In Biological Nitrogen Fixation: Towards Poverty Alleviation through Sustainable Agriculture; Dakora, F.D., Chimphango, S.B.M., Valentine, A.J., Elmerich, C., Newton, W.E., Eds.; Springer: Dordrecht, The Netherlands, 2008; Volume 42, pp. 185–187. [Google Scholar]
- Rathbun, E.A.; Brewin, N.J. Gum arabic glycoprotein and the infection of legumes by Rhizobium: Evidence for tyrosine cross-linking by peroxidase and by inorganic catalysis. Asp. Appl. Biol. 2009, 96, 241–246. [Google Scholar]
- Cheng, H.-P.; Walker, G.C. Succinoglycan is required for initiation and elongation of infection threads during nodulation of alfalfa by Rhizobium meliloti. J. Bacteriol. 1998, 180, 5183–5191. [Google Scholar] [CrossRef] [PubMed]
- Timmers, A.C.; Auriac, M.C.; de Billy, F.; Truchet, G. Nod factor internalization and microtubular cytoskeleton changes occur concomitantly during nodule differentiation in alfalfa. Development 1998, 125, 339–349. [Google Scholar] [CrossRef]
- Tsyganov, V.E.; Voroshilova, V.A.; Herrera-Cervera, J.A.; Sanjuan-Pinilla, J.M.; Borisov, A.Y.; Tikhonovich, I.A.; Priefer, U.B.; Olivares, J.; Sanjuan, J. Developmental downregulation of rhizobial genes as a function of symbiosome differentiation in symbiotic root nodules of Pisum sativum. New Phytol. 2003, 159, 521–530. [Google Scholar] [CrossRef] [PubMed]
- Arrighi, J.-F.; Barre, A.; Amor, B.B.; Bersoult, A.; Soriano, L.C.; Mirabella, R.; de Carvalho-Niebel, F.; Journet, E.-P.; Ghérardi, M.; Huguet, T.; et al. The Medicago truncatula lysine motif-receptor-like kinase gene family includes NFP and new nodule-expressed genes. Plant Physiol. 2006, 142, 265–279. [Google Scholar] [CrossRef]
- Haney, C.H.; Riely, B.K.; Tricoli, D.M.; Cook, D.R.; Ehrhardt, D.W.; Long, S.R. Symbiotic rhizobia bacteria trigger a change in localization and dynamics of the Medicago truncatula receptor kinase LYK3. Plant Cell 2011, 23, 2774–2787. [Google Scholar] [CrossRef] [PubMed]
- Huisman, R.; Geurts, R. A roadmap toward engineered nitrogen-fixing nodule symbiosis. Plant Commun. 2020, 1, 100019. [Google Scholar] [CrossRef] [PubMed]
- Kirienko, A.N.; Porozov, Y.B.; Malkov, N.V.; Akhtemova, G.A.; Le Signor, C.; Thompson, R.; Saffray, C.; Dalmais, M.; Bendahmane, A.; Tikhonovich, I.A.; et al. Role of a receptor-like kinase K1 in pea Rhizobium symbiosis development. Planta 2018, 248, 1101–1120. [Google Scholar] [CrossRef] [PubMed]
- Kawaharada, Y.; Kelly, S.; Nielsen, M.W.; Hjuler, C.T.; Gysel, K.; Muszyński, A.; Carlson, R.W.; Thygesen, M.B.; Sandal, N.; Asmussen, M.H.; et al. Receptor-mediated exopolysaccharide perception controls bacterial infection. Nature 2015, 523, 308–312. [Google Scholar] [CrossRef] [PubMed]
- Kawaharada, Y.; Nielsen, M.W.; Kelly, S.; James, E.K.; Andersen, K.R.; Rasmussen, S.R.; Füchtbauer, W.; Madsen, L.H.; Heckmann, A.B.; Radutoiu, S.; et al. Differential regulation of the Epr3 receptor coordinates membrane-restricted rhizobial colonization of root nodule primordia. Nat. Commun. 2017, 8, 14534. [Google Scholar] [CrossRef] [PubMed]
- Muszyński, A.; Heiss, C.; Hjuler, C.T.; Sullivan, J.T.; Kelly, S.J.; Thygesen, M.B.; Stougaard, J.; Azadi, P.; Carlson, R.W.; Ronson, C.W. Structures of exopolysaccharides involved in receptor-mediated perception of Mesorhizobium loti by Lotus japonicus. J. Biol. Chem. 2016, 291, 20946–20961. [Google Scholar] [CrossRef]
- Maillet, F.; Fournier, J.; Mendis, H.C.; Tadege, M.; Wen, J.; Ratet, P.; Mysore, K.S.; Gough, C.; Jones, K.M. Sinorhizobium meliloti succinylated high-molecular-weight succinoglycan and the Medicago truncatula LysM receptor-like kinase MtLYK10 participate independently in symbiotic infection. Plant J. 2020, 102, 311–326. [Google Scholar] [CrossRef]
- Skorupska, A.; Janczarek, M.; Marczak, M.; Mazur, A.; Król, J. Rhizobial exopolysaccharides: Genetic control and symbiotic functions. Microb. Cell Factories 2006, 5, 7. [Google Scholar] [CrossRef] [PubMed]
- Zgadzaj, R.; James, E.K.; Kelly, S.; Kawaharada, Y.; de Jonge, N.; Jensen, D.B.; Madsen, L.H.; Radutoiu, S. A legume genetic framework controls infection of nodules by symbiotic and endophytic bacteria. PLoS Genet. 2015, 11, e1005280. [Google Scholar] [CrossRef] [PubMed]
- Kiss, E.; Kereszt, A.; Barta, F.; Stephens, S.; Reuhs, B.L.; Kondorosi, Á.; Putnoky, P. The rkp-3 gene region of Sinorhizobium meliloti Rm41 contains strain-specific genes that determine K antigen structure. Mol. Plant Microbe Interact. 2001, 14, 1395–1403. [Google Scholar] [CrossRef] [PubMed]
- Le Quéré, A.J.L.; Deakin, W.J.; Schmeisser, C.; Carlson, R.W.; Streit, W.R.; Broughton, W.J.; Forsberg, L.S. Structural characterization of a K-antigen capsular polysaccharide essential for normal symbiotic infection in Rhizobium sp. NGR234: Deletion of the rkpMNO locus prevents synthesis of 5,7-diacetamido-3,5,7,9-tetradeoxy-non-2-ulosonic acid. J. Biol. Chem. 2006, 281, 28981–28992. [Google Scholar] [CrossRef] [PubMed]
- Pellock, B.J.; Cheng, H.-P.; Walker, G.C. Alfalfa root nodule invasion efficiency is dependent on Sinorhizobium meliloti polysaccharides. J. Bacteriol. 2000, 182, 4310–4318. [Google Scholar] [CrossRef] [PubMed]
- Rachwał, K.; Boguszewska, A.; Kopcińska, J.; Karaś, M.; Tchórzewski, M.; Janczarek, M. The regulatory protein RosR affects Rhizobium leguminosarum bv. trifolii protein profiles, cell surface properties, and symbiosis with clover. Front. Microbiol. 2016, 7, 1302. [Google Scholar] [CrossRef]
- Shimomura, K.; Nomura, M.; Tajima, S.; Kouchi, H. LjnsRING, a novel RING finger protein, is required for symbiotic interactions between Mesorhizobium loti and Lotus japonicus. Plant Cell Physiol. 2006, 47, 1572–1581. [Google Scholar] [CrossRef] [PubMed]
- Den Herder, G.; De Keyser, A.; De Rycke, R.; Rombauts, S.; Van de Velde, W.; Clemente, M.R.; Verplancke, C.; Mergaert, P.; Kondorosi, E.; Holsters, M.; et al. Seven in Absentia proteins affect plant growth and nodulation in Medicago truncatula. Plant Physiol. 2008, 148, 369–382. [Google Scholar] [CrossRef]
- Sinharoy, S.; Liu, C.; Breakspear, A.; Guan, D.; Shailes, S.; Nakashima, J.; Zhang, S.; Wen, J.; Torres-Jerez, I.; Oldroyd, G.; et al. A Medicago truncatula Cystathionine-β-synthase-like domain-containing protein is required for rhizobial infection and symbiotic nitrogen fixation. Plant Physiol. 2016, 170, 2204–2217. [Google Scholar] [CrossRef]
- McAdam, E.L.; Reid, J.B.; Foo, E. Gibberellins promote nodule organogenesis but inhibit the infection stages of nodulation. J. Exp. Bot. 2018, 69, 2117–2130. [Google Scholar] [CrossRef] [PubMed]
- Serova, T.A.; Tsyganova, A.V.; Tikhonovich, I.A.; Tsyganov, V.E. Gibberellins inhibit nodule senescence and stimulate nodule meristem bifurcation in pea (Pisum sativum L.). Front. Plant Sci. 2019, 10, 285. [Google Scholar] [CrossRef] [PubMed]
- Kiss, E.; Oláh, B.; Kaló, P.; Morales, M.; Heckmann, A.B.; Borbola, A.; Lózsa, A.; Kontár, K.; Middleton, P.; Downie, J.A.; et al. LIN, a novel type of U-Box/WD40 protein, controls early infection by rhizobia in legumes. Plant Physiol. 2009, 151, 1239–1249. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Z.; Zogli, P.; Libault, M. Plant hormones differentially control the sub-cellular localization of plasma membrane microdomains during the early stage of soybean nodulation. Genes 2019, 10, 1012. [Google Scholar] [CrossRef]
- Dolgikh, E.A.; Kusakin, P.G.; Kitaeva, A.B.; Tsyganova, A.V.; Kirienko, A.N.; Leppyanen, I.V.; Dolgikh, A.V.; Ilina, E.L.; Demchenko, K.N.; Tikhonovich, I.A.; et al. Mutational analysis indicates that abnormalities in rhizobial infection and subsequent plant cell and bacteroid differentiation in pea (Pisum sativum) nodules coincide with abnormal cytokinin responses and localization. Ann. Bot. 2020, 125, 905–923. [Google Scholar] [CrossRef] [PubMed]
- Serova, T.A.; Tikhonovich, I.A.; Tsyganov, V.E. Analysis of nodule senescence in pea (Pisum sativum L.) using laser microdissection, real-time PCR, and ACC immunolocalization. J. Plant Physiol. 2017, 212, 29–44. [Google Scholar] [CrossRef]
- Rodríguez-López, J.; López, A.H.; Estrada-Navarrete, G.; Sánchez, F.; Díaz-Camino, C. The noncanonical heat shock protein PvNod22 is essential for infection thread progression during rhizobial endosymbiosis in common bean. Mol. Plant Microbe Interact. 2019, 32, 939–948. [Google Scholar] [CrossRef]
- Liang, P.; Stratil, T.F.; Popp, C.; Marín, M.; Folgmann, J.; Mysore, K.S.; Wen, J.; Ott, T. Symbiotic root infections in Medicago truncatula require remorin-mediated receptor stabilization in membrane nanodomains. Proc. Natl. Acad. Sci. USA 2018, 115, 5289–5294. [Google Scholar] [CrossRef]
- Liu, M.; Jia, N.; Li, X.; Liu, R.; Xie, Q.; Murray, J.D.; Downie, J.A.; Xie, F. CERBERUS is critical for stabilization of VAPYRIN during rhizobial infection in Lotus japonicus. New Phytol. 2021, 229, 1684–1700. [Google Scholar] [CrossRef] [PubMed]
- Arrighi, J.-F.; Godfroy, O.; de Billy, F.; Saurat, O.; Jauneau, A.; Gough, C. The RPG gene of Medicago truncatula controls Rhizobium-directed polar growth during infection. Proc. Natl. Acad. Sci. USA 2008, 105, 9817–9822. [Google Scholar] [CrossRef] [PubMed]
- Dalla Via, V.; Traubenik, S.; Rivero, C.; Aguilar, O.M.; Zanetti, M.E.; Blanco, F.A. The monomeric GTPase RabA2 is required for progression and maintenance of membrane integrity of infection threads during root nodule symbiosis. Plant Mol. Biol. 2017, 93, 549–562. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Liu, M.X.; Qiu, L.P.; Xie, F. SPIKE1 activates the GTPase ROP6 to guide the polarized growth of infection threads in Lotus japonicus. Plant Cell 2020, 32, 3774–3791. [Google Scholar] [CrossRef] [PubMed]
- Yano, K.; Aoki, S.; Liu, M.; Umehara, Y.; Suganuma, N.; Iwasaki, W.; Sato, S.; Soyano, T.; Kouchi, H.; Kawaguchi, M. Function and evolution of a Lotus japonicus AP2/ERF family transcription factor that is required for development of infection threads. DNA Res. 2016, 24, 193–203. [Google Scholar] [CrossRef]
- Jaffe, M.J.; Leopold, A.C.; Staples, R.C. Thigmo responses in plants and fungi. Am. J. Bot. 2002, 89, 375–382. [Google Scholar] [CrossRef]
- Zhu, J.-K. Abiotic stress signaling and responses in plants. Cell 2016, 167, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Kitaeva, A.B.; Demchenko, K.N.; Tikhonovich, I.A.; Timmers, A.C.J.; Tsyganov, V.E. Comparative analysis of the tubulin cytoskeleton organization in nodules of Medicago truncatula and Pisum sativum: Bacterial release and bacteroid positioning correlate with characteristic microtubule rearrangements. New Phytol. 2016, 210, 168–183. [Google Scholar] [CrossRef] [PubMed]
- Qiu, L.; Lin, J.-s.; Xu, J.; Sato, S.; Parniske, M.; Wang, T.L.; Downie, J.A.; Xie, F. SCARN a novel class of SCAR protein that is required for root-hair infection during legume nodulation. PLoS Genet. 2015, 11, e1005623. [Google Scholar] [CrossRef]
- Roland, J.-C. The relationship between the plasmalemma and plant cell wall. In International Review of Cytology; Bourne, G.H., Danielli, J.F., Jeon, K.W., Eds.; Academic Press: London, UK; San Diego, CA, USA; Waltham, MA, USA; Oxford, UK, 1973; Volume 36, pp. 45–92. [Google Scholar]
- Hirsch, A.M.; Bang, M.; Ausubel, F.M. Ultrastructural analysis of ineffective alfalfa nodules formed by nif::Tn5 mutants of Rhizobium meliloti. J. Bacteriol. 1983, 155, 367–380. [Google Scholar] [CrossRef]
- Tsyganova, A.V.; Tsyganov, V.E. Organization of the endoplasmic reticulum in cells of effective and ineffective pea nodules (Pisum sativum L.). Ekol. Genet. 2019, 17, 5–14. [Google Scholar] [CrossRef]
- VandenBosch, K.A.; Bradley, D.J.; Knox, J.P.; Perotto, S.; Butcher, G.W.; Brewin, N.J. Common components of the infection thread matrix and the intercellular space identified by immunocytochemical analysis of pea nodules and uninfected roots. EMBO J. 1989, 8, 335–341. [Google Scholar] [CrossRef] [PubMed]
- Dixon, R.O.D. The structure of infection threads, bacteria and bacteroids in pea and clover root nodules. Archiv. Mikrobiol. 1964, 48, 166–178. [Google Scholar] [CrossRef]
- van Spronsen, P.C.; Bakhuizen, R.; van Brussel, A.A.; Kijne, J.W. Cell wall degradation during infection thread formation by the root nodule bacterium Rhizobium leguminosarum is a two-step process. Eur. J. Cell Biol. 1994, 64, 88–94. [Google Scholar] [PubMed]
- Monahan-Giovanelli, H.; Pinedo, C.A.; Gage, D.J. Architecture of infection thread networks in developing root nodules induced by the symbiotic bacterium Sinorhizobium meliloti on Medicago truncatula. Plant Physiol. 2006, 140, 661–670. [Google Scholar] [CrossRef] [PubMed]
- Benaben, V.; Duc, G.; Lefebvre, V.; Huguet, T. TE7, an inefficient symbiotic mutant of Medicago truncatula Gaertn. cv Jemalong. Plant Physiol. 1995, 107, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Veereshlingam, H.; Haynes, J.G.; Penmetsa, R.V.; Cook, D.R.; Sherrier, D.J.; Dickstein, R. nip, a symbiotic Medicago truncatula mutant that forms root nodules with aberrant infection threads and plant defense-like response. Plant Physiol. 2004, 136, 3692–3702. [Google Scholar] [CrossRef] [PubMed]
- Teillet, A.; Garcia, J.; de Billy, F.; Gherardi, M.; Huguet, T.; Barker, D.G.; de Carvalho-Niebel, F.; Journet, E.-P. api, a novel Medicago truncatula symbiotic mutant impaired in nodule primordium invasion. Mol. Plant Microbe Interact. 2008, 21, 535–546. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tsyganov, V.E.; Morzhina, E.V.; Stefanov, S.Y.; Borisov, A.Y.; Lebsky, V.K.; Tikhonovich, I.A. The pea (Pisum sativum L.) genes sym33 and sym40 control infection thread formation and root nodule function. Mol. Gen. Genet. 1998, 259, 491–503. [Google Scholar] [CrossRef] [PubMed]
- Morzhina, E.V.; Tsyganov, V.E.; Borisov, A.Y.; Lebsky, V.K.; Tikhonovich, I.A. Four developmental stages identified by genetic dissection of pea (Pisum sativum L.) root nodule morphogenesis. Plant Sci. 2000, 155, 75–83. [Google Scholar] [CrossRef]
- Voroshilova, V.A.; Boesten, B.; Tsyganov, V.E.; Borisov, A.Y.; Tikhonovich, I.A.; Priefer, U.B. Effect of mutations in Pisum sativum L. genes blocking different stages of nodule development on the expression of late symbiotic genes in Rhizobium leguminosarum bv. viciae. Mol. Plant Microbe Interact. 2001, 14, 471–476. [Google Scholar] [CrossRef] [PubMed]
- Voroshilova, V.A.; Demchenko, K.N.; Brewin, N.J.; Borisov, A.Y.; Tikhonovich, I.A. Initiation of a legume nodule with an indeterminate meristem involves proliferating host cells that harbour infection threads. New Phytol. 2009, 181, 913–923. [Google Scholar] [CrossRef] [PubMed]
- Rae, A.L.; Bonfante-Fasolo, P.; Brewin, N.J. Structure and growth of infection threads in the legume symbiosis with Rhizobium leguminosarum. Plant J. 1992, 2, 385–395. [Google Scholar] [CrossRef]
- Bolaños, L.; El-Hamdaoui, A.; Bonilla, I. Recovery of development and functionality of nodules and plant growth in salt-stressed Pisum sativum-Rhizobium leguminosarum symbiosis by boron and calcium. J. Plant Physiol. 2003, 160, 1493–1497. [Google Scholar] [CrossRef]
- Redondo-Nieto, M.; Wilmot, A.R.; El-Hamdaoui, A.; Bonilla, I.; Bolaños, L. Relationship between boron and calcium in the N2-fixing legume–rhizobia symbiosis. Plant Cell Environ. 2003, 26, 1905–1915. [Google Scholar] [CrossRef]
- Redondo-Nieto, M.; Pulido, L.; Reguera, M.; Bonilla, I.; Bolaños, L. Developmentally regulated membrane glycoproteins sharing antigenicity with rhamnogalacturonan II are not detected in nodulated boron deficient Pisum sativum. Plant Cell Environ. 2007, 30, 1436–1443. [Google Scholar] [CrossRef]
- Ivanova, K.A.; Tsyganova, A.V.; Brewin, N.J.; Tikhonovich, I.A.; Tsyganov, V.E. Induction of host defences by Rhizobium during ineffective nodulation of pea (Pisum sativum L.) carrying symbiotically defective mutations sym40 (PsEFD), sym33 (PsIPD3/PsCYCLOPS) and sym42. Protoplasma 2015, 252, 1505–1517. [Google Scholar] [CrossRef] [PubMed]
- Sujkowska-Rybkowska, M.; Borucki, W. Pectins esterification in the apoplast of aluminum-treated pea root nodules. J. Plant Physiol. 2015, 184, 1–7. [Google Scholar] [CrossRef]
- Tsyganova, A.V.; Seliverstova, E.V.; Brewin, N.J.; Tsyganov, V.E. Comparative analysis of remodelling of the plant–microbe interface in Pisum sativum and Medicago truncatula symbiotic nodules. Protoplasma 2019, 256, 983–996. [Google Scholar] [CrossRef] [PubMed]
- Tsyganova, A.V.; Seliverstova, E.V.; Brewin, N.J.; Tsyganov, V.E. Bacterial release is accompanied by ectopic accumulation of cell wall material around the vacuole in nodules of Pisum sativum sym33-3 allele encoding transcription factor PsCYCLOPS/PsIPD3. Protoplasma 2019, 256, 1449–1453. [Google Scholar] [CrossRef] [PubMed]
- Gavrin, A.; Chiasson, D.; Ovchinnikova, E.; Kaiser, B.N.; Bisseling, T.; Fedorova, E.E. VAMP721a and VAMP721d are important for pectin dynamics and release of bacteria in soybean nodules. New Phytol. 2016, 210, 1011–1021. [Google Scholar] [CrossRef] [PubMed]
- Reguera, M.; Abreu, I.; Brewin, N.J.; Bonilla, I.; Bolaños, L. Borate promotes the formation of a complex between legume AGP-extensin and rhamnogalacturonan II and enhances production of Rhizobium capsular polysaccharide during infection thread development in Pisum sativum symbiotic root nodules. Plant Cell Environ. 2010, 33, 2112–2120. [Google Scholar] [CrossRef] [PubMed]
- Moore, P.J.; Staehelin, L.A. Immunogold localization of the cell-wall-matrix polysaccharides rhamnogalacturonan I and xyloglucan during cell expansion and cytokinesis in Trifolium pratense L.; implication for secretory pathways. Planta 1988, 174, 433–445. [Google Scholar] [CrossRef] [PubMed]
- Rae, A.; Perotto, S.; Knox, J.; Kannenberg, E.; Brewin, N. Expression of extracellular glycoproteins in the uninfected cells of developing pea nodule tissue. Mol. Plant-Microbe Interact. 1991, 4, 563–570. [Google Scholar] [CrossRef]
- Bolanos, L.; Brewin, N.J.; Bonilla, I. Effects of Boron on Rhizobium-legume cell-surface interactions and nodule development. Plant Physiol. 1996, 110, 1249–1256. [Google Scholar] [CrossRef] [PubMed]
- Olsson, P.A.; Kjellbom, P.; Rosendahl, L. Rhizobium colonization induced changes in membrane-bound and soluble hydroxyproline-rich glycoprotein composition in pea. Physiol. Plant. 2002, 114, 652–660. [Google Scholar] [CrossRef] [PubMed]
- Tsyganova, A.V.; Tsyganov, V.E.; Findlay, K.C.; Borisov, A.Y.; Tikhonovich, I.A.; Brewin, N.J. Distribution of legume arabinogalactan protein-extensin (AGPE) glycoproteins in symbiotically defective pea mutants with abnormal infection threads. Cell Tissue Biol. 2009, 3, 93–102. [Google Scholar] [CrossRef]
- Bradley, D.J.; Wood, E.A.; Larkins, A.P.; Galfre, G.; Butcher, G.W.; Brewin, N.J. Isolation of monoclonal antibodies reacting with peribacteriod membranes and other components of pea root nodules containing Rhizobium leguminosarum. Planta 1988, 173, 149–160. [Google Scholar] [CrossRef] [PubMed]
- Tsyganova, A.V.; Brewin, N.; Tsyganov, V.E. Analysis of epitope distribution of arabinogalactan protein-extensins in pea (Pisum sativum) nodules of wild-type and mutants impaired in infection thread growth. Ekol. Genet. 2019, 17, 5–12. [Google Scholar] [CrossRef]
- Benhamou, N.; Lafontaine, P.J.; Mazau, D.; Esquerré-Tugayé, M.-T. Differential accumulation of hydroxyproline-rich glycoproteins in bean root nodule cells infected with a wild-type strain or a C4-dicarboxylic acid mutant of Rhizobium leguminosarum bv. phaseoli. Planta 1991, 184, 457–467. [Google Scholar] [CrossRef] [PubMed]
- Bonilla, I.; Mergold-Villasenor, C.; Campos, M.E.; Sanchez, N.; Perez, H.; Lopez, L.; Castrejon, L.; Sanchez, F.; Cassab, G.I. The aberrant cell walls of boron-deficient bean root nodules have no covalently bound hydroxyproline/proline-rich proteins. Plant Physiol. 1997, 115, 1329–1340. [Google Scholar] [CrossRef]
- Sherrier, D.J.; Taylor, G.S.; Silverstein, K.A.T.; Gonzales, M.B.; VandenBosch, K.A. Accumulation of extracellular proteins bearing unique proline-rich motifs in intercellular spaces of the legume nodule parenchyma. Protoplasma 2005, 225, 43–55. [Google Scholar] [CrossRef]
- Sujkowska, M.; Górska-Czekaj, M.; Bederska, M.; Borucki, W. Vacuolar organization in the nodule parenchyma is important for the functioning of pea root nodules. Symbiosis 2011, 54, 1. [Google Scholar] [CrossRef][Green Version]
- Gardner, C.D.; Sherrier, D.J.; Kardailsky, I.V.; Brewin, N.J. Localization of lipoxygenase proteins and mRNA in pea nodules: Identification of lipoxygenase in the lumen of infection threads. Mol. Plant Microbe Interact. 1996, 9, 282–289. [Google Scholar] [CrossRef]
- Rubio, M.C.; James, E.K.; Clemente, M.R.; Bucciarelli, B.; Fedorova, M.; Vance, C.P.; Becana, M. Localization of superoxide dismutases and hydrogen peroxide in legume root nodules. Mol. Plant Microbe Interact. 2004, 17, 1294–1305. [Google Scholar] [CrossRef] [PubMed]
- Sujkowska-Rybkowska, M.; Borucki, W. Localization of hydrogen peroxide accumulation and diamine oxidase activity in pea root nodules under aluminum stress. Micron 2014, 57, 13–22. [Google Scholar] [CrossRef] [PubMed]
- Santos, R.; Hérouart, D.; Sigaud, S.; Touati, D.; Puppo, A. Oxidative burst in alfalfa-Sinorhizobium meliloti symbiotic interaction. Mol. Plant Microbe Interact. 2001, 14, 86–89. [Google Scholar] [CrossRef] [PubMed]
- Jamet, A.; Mandon, K.; Puppo, A.; Hérouart, D. H2O2 is required for optimal establishment of the Medicago sativa/Sinorhizobium meliloti symbiosis. J. Bacteriol. 2007, 189, 8741–8745. [Google Scholar] [CrossRef] [PubMed]
- Tsyganova, A.V.; Tsyganov, V.; Borisov, A.Y.; Tikhonovich, I.A.; Brewin, N.J. Comparative cytochemical analysis of hydrogen peroxide distribution in pea ineffective mutant SGEFix−-1 (sym40) and initial line SGE. Ekol. Genet. 2009, 7, 3–9. [Google Scholar] [CrossRef]
- Fauvart, M.; Verstraeten, N.; Dombrecht, B.; Venmans, R.; Beullens, S.; Heusdens, C.; Michiels, J. Rhizobium etli HrpW is a pectin-degrading enzyme and differs from phytopathogenic homologues in enzymically crucial tryptophan and glycine residues. Microbiology 2009, 155, 3045–3054. [Google Scholar] [CrossRef]
- Lievens, S.; Goormachtig, S.; Herman, S.; Holsters, M. Patterns of pectin methylesterase transcripts in developing stem nodules of Sesbania rostrata. Mol. Plant Microbe Interact. 2002, 15, 164–168. [Google Scholar] [CrossRef] [PubMed]
- Rodríguez-Llorente, I.D.; Pérez-Hormaeche, J.; Mounadi, K.E.; Dary, M.; Caviedes, M.A.; Cosson, V.; Kondorosi, A.; Ratet, P.; Palomares, A.J. From pollen tubes to infection threads: Recruitment of Medicago floral pectic genes for symbiosis. Plant J. 2004, 39, 587–598. [Google Scholar] [CrossRef]
- Frueauf, J.B.; Dolata, M.; Leykam, J.F.; Lloyd, E.A.; Gonzales, M.; VandenBosch, K.; Kieliszewski, M.J. Peptides isolated from cell walls of Medicago truncatula nodules and uninfected root. Phytochemistry 2000, 55, 429–438. [Google Scholar] [CrossRef]
- Arsenijevic-Maksimovic, I.; Broughton, W.J.; Krause, A. Rhizobia modulate root-hair-specific expression of extensin genes. Mol. Plant Microbe Interact. 1997, 10, 95–101. [Google Scholar] [CrossRef] [PubMed]
- Giordano, W.; Hirsch, A.M. The expression of MaEXP1, a Melilotus alba expansin gene, is upregulated during the sweetclover-Sinorhizobium meliloti interaction. Mol. Plant Microbe Interact. 2004, 17, 613–622. [Google Scholar] [CrossRef] [PubMed]
- Lee, A.; Giordano, W.; Hirsch, A.M. Cytokinin induces expansin gene expression in Melilotus alba Desr. wild-type and the non-nodulating, non-mycorrhizal (Nod-Myc-) mutant Masym3. Plant Signal. Behav. 2008, 3, 218–223. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Greene, E.A.; Erard, M.; Dedieu, A.; Barker, D.G. MtENOD16 and 20 are members of a family of phytocyanin-related early nodulins. Plant Mol. Biol. 1998, 36, 775–783. [Google Scholar] [CrossRef]
- Dolgikh, E.A.; Leppyanen, I.V.; Osipova, M.A.; Savelyeva, N.V.; Borisov, A.Y.; Tsyganov, V.E.; Geurts, R.; Tikhonovich, I.A. Genetic dissection of Rhizobium-induced infection and nodule organogenesis in pea based on ENOD12A and ENOD5 expression analysis. Plant Biol. 2011, 13, 285–296. [Google Scholar] [CrossRef]
- Tsyganova, A.V.; Tsyganov, V.E. Plant cell wall in symbiotic interactions. Pectins. Agric. Biol. 2019, 54, 446–457. [Google Scholar] [CrossRef]
- Ivanov, S.; Fedorova, E.E.; Limpens, E.; De Mita, S.; Genre, A.; Bonfante, P.; Bisseling, T. Rhizobium–legume symbiosis shares an exocytotic pathway required for arbuscule formation. Proc. Natl. Acad. Sci. USA 2012, 109, 8316–8321. [Google Scholar] [CrossRef]
- Atmodjo, M.A.; Hao, Z.; Mohnen, D. Evolving views of pectin biosynthesis. Ann. Rev. Plant Biol. 2013, 64, 747–779. [Google Scholar] [CrossRef]
- Brewin, N.J.; Rae, A.L.; Perotto, S.; Knox, J.P.; Roberts, K.; LeGal, M.F.; Sindhu, S.S.; Wood, E.A.; Kannenberg, E.L. Immunological dissection of the plant-microbe interface in pea nodules. In Nitrogen Fixation; Gresshoff, P.M., Roth, L.E., Stacey, G., Newton, W.E., Eds.; Springer: Boston, MA, USA, 1990; pp. 227–234. [Google Scholar] [CrossRef]
- Cassab, G.I. Arabinogalactan proteins during the development of soybean root nodules. Planta 1986, 168, 441–446. [Google Scholar] [CrossRef]
- Nguema-Ona, E.; Vicré-Gibouin, M.; Cannesan, M.-A.; Driouich, A. Arabinogalactan proteins in root–microbe interactions. Trends Plant Sci. 2013, 18, 440–449. [Google Scholar] [CrossRef]
- Perotto, S.; Vandenbosch, K.A.; Butcher, G.W.; Brewin, N.J. Molecular composition and development of the plant glycocalyx associated with the peribacteroid membrane of pea root-nodules. Development 1991, 112, 763–773. [Google Scholar] [CrossRef]
- Gucciardo, S.; Rathbun, E.A.; Shanks, M.; Jenkyns, S.; Mak, L.; Durrant, M.C.; Brewin, N.J. Epitope tagging of legume root nodule extensin modifies protein structure and crosslinking in cell walls of transformed tobacco leaves. Mol. Plant Microbe Interact. 2005, 18, 24–32. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Legocki, R.P.; Verma, D.P.S. Identification of “nodule-specific” host proteins (nodulins) involved in the development of Rhizobium-legume symbiosis. Cell 1980, 20, 153–163. [Google Scholar] [CrossRef]
- Jeffree, C.E.; Dale, J.E.; Fry, S.C. The genesis of intercellular spaces in developing leaves of Phaseolus vulgaris L. Protoplasma 1986, 132, 90–98. [Google Scholar] [CrossRef]
- Peleg-Grossman, S.; Melamed-Book, N.; Levine, A. ROS production during symbiotic infection suppresses pathogenesis-related gene expression. Plant Signal. Behav. 2012, 7, 409–415. [Google Scholar] [CrossRef] [PubMed]
- Montiel, J.; Nava, N.; Cárdenas, L.; Sánchez-López, R.; Arthikala, M.-K.; Santana, O.; Sánchez, F.; Quinto, C. A Phaseolus vulgaris NADPH oxidase gene is required for root infection by rhizobia. Plant Cell Physiol. 2012, 53, 1751–1767. [Google Scholar] [CrossRef] [PubMed]
- Arthikala, M.-K.; Montiel, J.; Sánchez-López, R.; Nava, N.; Cárdenas, L.; Quinto, C. Respiratory burst oxidase homolog gene A is crucial for Rhizobium infection and nodule maturation and function in common bean. Front. Plant Sci. 2017, 8, 2003. [Google Scholar] [CrossRef]
- Provorov, N.A.; Tsyganova, A.V.; Brewin, N.J.; Tsyganov, V.E.; Vorobyov, N.I. Evolution of symbiotic bacteria within the extra- and intra-cellular plant compartments: Experimental evidence and mathematical simulation (Mini-review). Symbiosis 2012, 58, 39–50. [Google Scholar] [CrossRef]
- Lehman, A.P.; Long, S.R. Exopolysaccharides from Sinorhizobium meliloti can protect against H2O2-dependent damage. J. Bacteriol. 2013, 195, 5362–5369. [Google Scholar] [CrossRef] [PubMed]
- Mithöfer, A. Suppression of plant defence in rhizobia–legume symbiosis. Trends Plant Sci. 2002, 7, 440–444. [Google Scholar] [CrossRef]
- Soto, M.J.; Sanjuán, J.; Olivares, J. Rhizobia and plant-pathogenic bacteria: Common infection weapons. Microbiology 2006, 152, 3167–3174. [Google Scholar] [CrossRef] [PubMed]
- van Workum, W.A.T.; van Slageren, S.; van Brussel, A.A.N.; Kijne, J.W. Role of exopolysaccharides of Rhizobium leguminosarum bv. viciae as host plant-specific molecules required for infection thread formation during nodulation of Vicia sativa. Mol. Plant Microbe Interact. 1998, 11, 1233–1241. [Google Scholar] [CrossRef]
- Niehaus, K.; Kapp, D.; Pühler, A. Plant defence and delayed infection of alfalfa pseudonodules induced by an exopolysaccharide (EPS I)-deficient Rhizobium meliloti mutant. Planta 1993, 190, 415–425. [Google Scholar] [CrossRef]
- Beck, S.; Marlow, V.L.; Woodall, K.; Doerrler, W.T.; James, E.K.; Ferguson, G.P. The Sinorhizobium meliloti MsbA2 protein is essential for the legume symbiosis. Microbiology 2008, 154, 1258–1270. [Google Scholar] [CrossRef] [PubMed]
- Albus, U.; Baier, R.; Holst, O.; Pühler, A.; Niehaus, K. Suppression of an elicitor-induced oxidative burst reaction in Medicago sativa cell cultures by Sinorhizobium meliloti lipopolysaccharides. New Phytol. 2001, 151, 597–606. [Google Scholar] [CrossRef] [PubMed]
- Scheidle, H.; Groß, A.; Niehaus, K. The lipid A substructure of the Sinorhizobium meliloti lipopolysaccharides is sufficient to suppress the oxidative burst in host plants. New Phytol. 2005, 165, 559–566. [Google Scholar] [CrossRef] [PubMed]
- Bright, L.J.; Liang, Y.; Mitchell, D.M.; Harris, J.M. The LATD gene of Medicago truncatula is required for both nodule and root development. Mol. Plant Microbe Interact. 2005, 18, 521–532. [Google Scholar] [CrossRef] [PubMed]
- Yendrek, C.R.; Lee, Y.-C.; Morris, V.; Liang, Y.; Pislariu, C.I.; Burkart, G.; Meckfessel, M.H.; Salehin, M.; Kessler, H.; Wessler, H.; et al. A putative transporter is essential for integrating nutrient and hormone signaling with lateral root growth and nodule development in Medicago truncatula. Plant J. 2010, 62, 100–112. [Google Scholar] [CrossRef] [PubMed]
- Ovchinnikova, E.; Journet, E.-P.; Chabaud, M.; Cosson, V.; Ratet, P.; Duc, G.; Fedorova, E.; Liu, W.; den Camp, R.O.; Zhukov, V.; et al. IPD3 controls the formation of nitrogen-fixing symbiosomes in pea and Medicago Spp. Mol. Plant Microbe Interact. 2011, 24, 1333–1344. [Google Scholar] [CrossRef]
- Tsyganova, A.V.; Kitaeva, A.B.; Tsyganov, V.E. Cell differentiation in nitrogen-fixing nodules hosting symbiosomes. Funct. Plant Biol. 2018, 45, 47–57. [Google Scholar] [CrossRef] [PubMed]
- Bassett, B.; Goodman, R.N.; Novacky, A. Ultrastructure of soybean nodules. I: Release of rhizobia from the infection thread. Can. J. Microbiol. 1977, 23, 573–582. [Google Scholar] [CrossRef] [PubMed]
- Cermola, M.; Fedorova, E.; Taté, R.; Riccio, A.; Favre, R.; Patriarca, E.J. Nodule invasion and symbiosome differentiation during Rhizobium etli–Phaseolus vulgaris symbiosis. Mol. Plant Microbe Interact. 2000, 13, 733–741. [Google Scholar] [CrossRef] [PubMed]
- Robertson, J.G.; Lyttleton, P.; Bullivant, S.; Grayston, G.F. Membranes in lupin root nodules. I. The role of Golgi bodies in the biogenesis of infection threads and peribacteroid membranes. J. Cell Sci. 1978, 30, 129–149. [Google Scholar] [CrossRef] [PubMed]
- Kereszt, A.; Mergaert, P.; Montiel, J.; Endre, G.; Kondorosi, É. Impact of plant peptides on symbiotic nodule development and functioning. Front. Plant Sci. 2018, 9, 1026. [Google Scholar] [CrossRef] [PubMed]
- Ivanov, S.; Fedorova, E.; Bisseling, T. Intracellular plant microbe associations: Secretory pathways and the formation of perimicrobial compartments. Curr. Opin. Plant Biol. 2010, 13, 372–377. [Google Scholar] [CrossRef] [PubMed]
- Bradley, D.J.; Butcher, G.W.; Galfre, G.; Wood, E.A.; Brewin, N.J. Physical association between the peribacteroid membrane and lipopolysaccharide from the bacteroid outer membrane in Rhizobium-infected pea root nodule cells. J. Cell Sci. 1986, 85, 47–61. [Google Scholar] [CrossRef]
- Venado, R.E.; Liang, J.; Marín, M. Rhizobia infection, a journey to the inside of plant cells. In Advances in Botanical Research; Frendo, P., Frugier, F., Masson-Boivin, C., Eds.; Academic Press: London, UK; San Diego, CA, USA; Waltham, MA, USA; Oxford, UK, 2020; Volume 94, pp. 97–118. [Google Scholar]
- Ferguson, G.P.; Roop, R.M.; Walker, G.C. Deficiency of a Sinorhizobium meliloti bacA mutant in alfalfa symbiosis correlates with alteration of the cell envelope. J. Bacteriol. 2002, 184, 5625–5632. [Google Scholar] [CrossRef] [PubMed]
- Kannenberg, E.L.; Perotto, S.; Bianciotto, V.; Rathbun, E.A.; Brewin, N.J. Lipopolysaccharide epitope expression of Rhizobium bacteroids as revealed by in situ immunolabelling of pea root nodule sections. J. Bacteriol. 1994, 176, 2021–2032. [Google Scholar] [CrossRef] [PubMed]
- Bourassa, D.V.; Kannenberg, E.L.; Sherrier, D.J.; Buhr, R.J.; Carlson, R.W. The lipopolysaccharide lipid A long-chain fatty acid is important for Rhizobium leguminosarum growth and stress adaptation in free-living and nodule environments. Mol. Plant Microbe Interact. 2017, 30, 161–175. [Google Scholar] [CrossRef]
- Perotto, S.; Brewin, N.; Kannenberg, E. Cytological evidence for a host defense response that reduces cell and tissue invasion in pea nodules by lipopolysaccharide-defective mutants of Rhizobium leguminosarum strain 3841. Mol. Plant Microbe Interact. 1994, 7, 99–112. [Google Scholar] [CrossRef]
- Serova, T.A.; Tsyganov, V.E. Symbiotic nodule senescence in legumes: Molecular-genetic and cellular aspects (review). Agric. Biol. 2014, 5, 3–15. [Google Scholar] [CrossRef]
- Timmers, A.C.J.; Soupène, E.; Auriac, M.-C.; de Billy, F.; Vasse, J.; Boistard, P.; Truchet, G. Saprophytic intracellular rhizobia in alfalfa nodules. Mol. Plant Microbe Interact. 2000, 13, 1204–1213. [Google Scholar] [CrossRef]
- Skawińska, M.; Sańko-Sawczenko, I.; Dmitruk, D.; Czarnocka, W.; Łotocka, B. Organization and ultrastructure of Medicago truncatula root apical meristem. In The Model Legume Medicago Truncatula; de Bruijn, F., Ed.; John Wiley & Sons: Hoboken, NJ, USA, 2020; p. 709. [Google Scholar] [CrossRef]
- Xi, J.; Chen, Y.; Nakashima, J.; Wang, S.-m.; Chen, R. Medicago truncatula esn1 defines a genetic locus involved in nodule senescence and symbiotic nitrogen fixation. Mol. Plant Microbe Interact. 2013, 26, 893–902. [Google Scholar] [CrossRef] [PubMed]
- Melino, V.J.; Drew, E.A.; Ballard, R.A.; Reeve, W.G.; Thomson, G.; White, R.G.; O’Hara, G.W. Identifying abnormalities in symbiotic development between Trifolium spp. and Rhizobium leguminosarum bv. trifolii leading to sub-optimal and ineffective nodule phenotypes. Ann. Bot. 2012, 110, 1559–1572. [Google Scholar] [CrossRef][Green Version]
- Zahran, H.H. Rhizobium-legume symbiosis and nitrogen fixation under severe conditions and in an arid climate. Microbiol. Mol. Biol. Rev. 1999, 63, 968–989. [Google Scholar] [CrossRef]
- Chaudhary, P.; Dudeja, S.S.; Kapoor, K.K. Effectivity of host-Rhizobium leguminosarum symbiosis in soils receiving sewage water containing heavy metals. Microbiol. Res. 2004, 159, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Gorshkov, A.P.; Tsyganova, A.V.; Vorobiev, M.G.; Tsyganov, V.E. The fungicide tetramethylthiuram disulfide negatively affects plant cell walls, infection thread walls, and symbiosomes in pea (Pisum sativum L.) symbiotic nodules. Plants 2020, 9, 1488. [Google Scholar] [CrossRef]
- Delgadillo, J.; Lafuente, A.; Doukkali, B.; Redondo-Gómez, S.; Mateos-Naranjo, E.; Caviedes, M.A.; Pajuelo, E.; Rodríguez-Llorente, I.D. Improving legume nodulation and Cu rhizostabilization using a genetically modified rhizobia. Environ. Technol. 2015, 36, 1237–1245. [Google Scholar] [CrossRef]
- Fagorzi, C.; Checcucci, A.; DiCenzo, G.C.; Debiec-Andrzejewska, K.; Dziewit, L.; Pini, F.; Mengoni, A. Harnessing rhizobia to improve heavy-metal phytoremediation by legumes. Genes 2018, 9, 542. [Google Scholar] [CrossRef] [PubMed]
- Tsyganov, V.E.; Tsyganova, A.V.; Gorshkov, A.P.; Seliverstova, E.V.; Kim, V.E.; Chizhevskaya, E.P.; Belimov, A.A.; Serova, T.A.; Ivanova, K.A.; Kulaeva, O.A.; et al. Efficacy of a plant-microbe system: Pisum sativum (L.) cadmium-tolerant mutant and Rhizobium leguminosarum strains, expressing pea metallothionein genes PsMT1 and PsMT2, for cadmium phytoremediation. Front. Microbiol. 2020, 11, 15. [Google Scholar] [CrossRef] [PubMed]
- Montiel, J.; Reid, D.; Grønbæk, T.H.; Benfeldt, C.M.; James, E.K.; Ott, T.; Ditengou, F.A.; Nadzieja, M.; Kelly, S.; Stougaard, J. Distinct signaling routes mediate intercellular and intracellular rhizobial infection in Lotus japonicus. Plant Physiol. 2020, 185, 1131–1147. [Google Scholar] [CrossRef] [PubMed]
- Borisov, A.Y.; Danilova, T.N.; Koroleva, T.A.; Kuznetsova, E.V.; Madsen, L.; Mofett, M.; Naumkina, T.S.; Nemankin, T.A.; Ovchinnikova, E.S.; Pavlova, Z.B.; et al. Regulatory genes of garden pea (Pisum sativum L.) controlling the development of nitrogen-fixing nodules and arbuscular mycorrhiza: A review of basic and applied aspects. Appl. Biochem. Microbiol. 2007, 43, 237–243. [Google Scholar] [CrossRef]
- Radhakrishnan, G.V.; Keller, J.; Rich, M.K.; Vernié, T.; Mbadinga Mbadinga, D.L.; Vigneron, N.; Cottret, L.; Clemente, H.S.; Libourel, C.; Cheema, J.; et al. An ancestral signalling pathway is conserved in intracellular symbioses-forming plant lineages. Nat. Plants 2020, 6, 280–289. [Google Scholar] [CrossRef] [PubMed]



 —Ca2+; light blue extracellular matrix is fluid; shaded blue matrix is solid; pink cell wall is newly synthesized; red cell wall is mature or modified. Objects are not scaled.
—Ca2+; light blue extracellular matrix is fluid; shaded blue matrix is solid; pink cell wall is newly synthesized; red cell wall is mature or modified. Objects are not scaled.
 —Ca2+; light blue extracellular matrix is fluid; shaded blue matrix is solid; pink cell wall is newly synthesized; red cell wall is mature or modified. Objects are not scaled.
—Ca2+; light blue extracellular matrix is fluid; shaded blue matrix is solid; pink cell wall is newly synthesized; red cell wall is mature or modified. Objects are not scaled.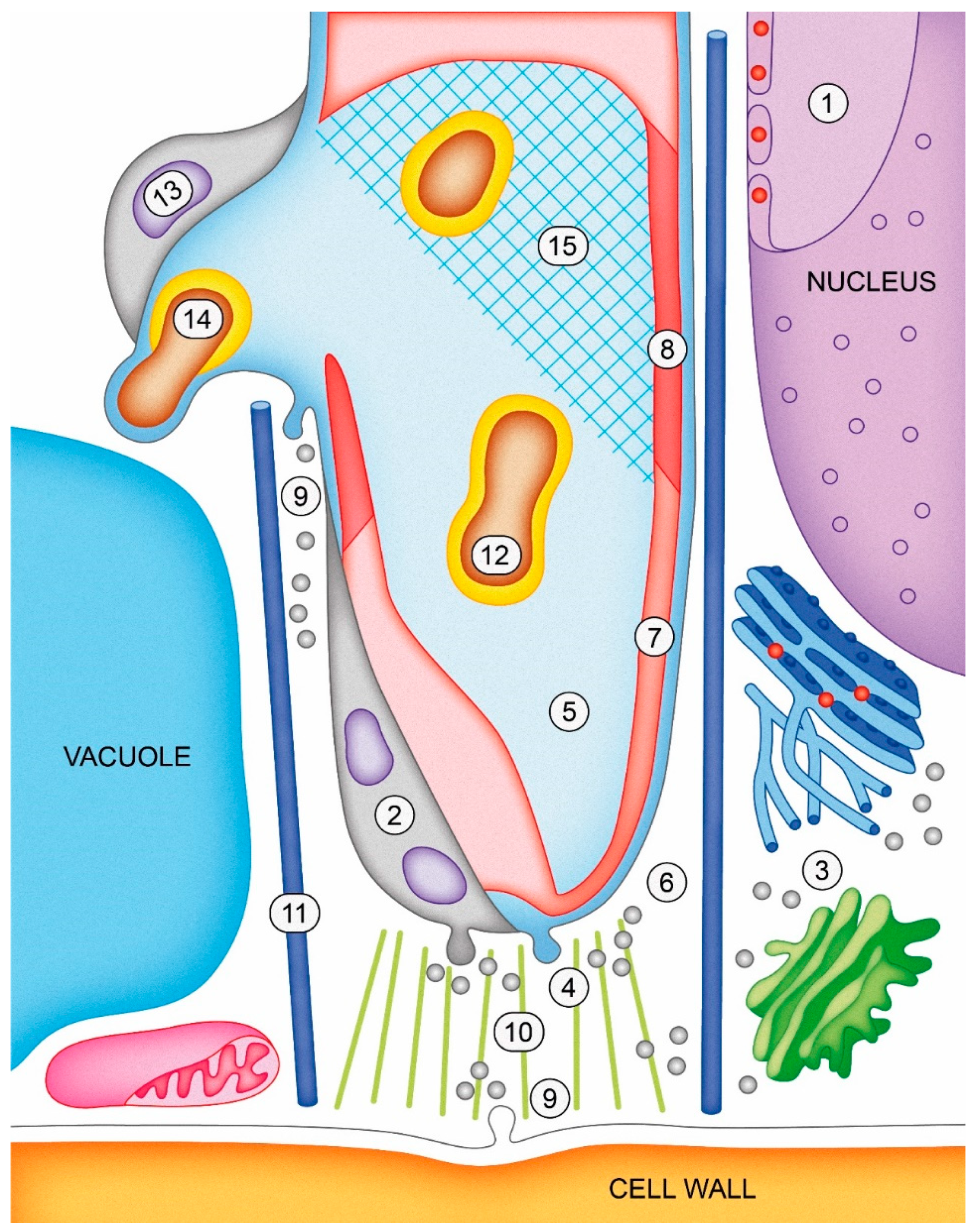
| Probe a | Component b | Epitope Recognized c | References |
|---|---|---|---|
| Antibody | |||
| JIM5 | low methyl-esterified HG | α-MeGalA(2)-(1→4)-α-GalA(3)- (1→4)-α-MeGalA | [186,197,198,199,200,201,202,203,204] |
| JIM7 | high methyl-esterified HG | α-GalA-(1→4)-α- MeGalA(4)-(1→4)-α-GalA | [197,202,203,204] |
| LM19 | low methyl-esterified HG | α-GalA-(1→4)(4) | [205] |
| LM20 | high methyl-esterified HG | α-MeGalA-(1→4)(4) | [205] |
| 2F4 | calcium cross-linked HG | dimer of α-MeGalA-(1→4)(9) and Ca2+(5) | [202] |
| LM5 | (1→4)-β-D-galactan (RG-I) | β-Gal-(1→4)(3) | [203,204] |
| anti-RG- II | monomeric and dimeric RG-II | unknown | [199,200,205,206] |
| anti- XyG | XyG | unknown | [197,207] |
| anti-callose | callose ((1→3)-β-D-glucan) | β-Glc-(1→3)(5) | [205] |
| MAC265 | 95kDa AGPE | unknown | [141,186,197,205,208,209,210,211] |
| MAC204 | 95kDa AGPE | unknown | [186,212,213] |
| MAC236 | 95kDa AGPE | unknown | [186,213] |
| JIM13 | AGP | unknown | [208] |
| anti-HRGP | hydroxyproline-rich glycoproteins (HPGPs) | unknown | [214] |
| anti-ENOD2 | early nodulin2/hydroxyproline-rich glycoproteins (ENOD2/HPGPs) | unknown | [215,216] |
| anti-VAMP721d/VAMP721e | vesicle-associated membrane proteins (VAMPs) | peptide QKLPSTNNKFTYNC | [205] |
| anti- EGL1 | endo-β-1,4-glucanase | peptide CYFPKRIHHRGSSLP | [217] |
| anti-LOX | lipoxygenase (LOX) | unknown | [218] |
| anti-SOD | superoxide dismutase (SOD) | unknown | [219] |
| anti-DAO | diamine oxidase (DAO) | unknown | [220] |
| Cytochemical reagent | |||
| chlor-zinc-iodide | cellulose | na | [15] |
| I2KI (I2, KI, H2SO4) | cellulose | na | [15] |
| cerium chloride (CeCl4) | H2O2 | na | [219,220,221,222,223] |
| ruthenium red | unesterified HG | na | [202,204] |
| aniline blue | callose ((1→3)-β-D-glucan) | na | [201,204] |
| neutral red | suberin | na | [201,204] |
| I2KI (I2, KI, H2SO4) | suberin | na | [204] |
| Protein | |||
| CBH-I | cellulose | na | [197] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsyganova, A.V.; Brewin, N.J.; Tsyganov, V.E. Structure and Development of the Legume-Rhizobial Symbiotic Interface in Infection Threads. Cells 2021, 10, 1050. https://doi.org/10.3390/cells10051050
Tsyganova AV, Brewin NJ, Tsyganov VE. Structure and Development of the Legume-Rhizobial Symbiotic Interface in Infection Threads. Cells. 2021; 10(5):1050. https://doi.org/10.3390/cells10051050
Chicago/Turabian StyleTsyganova, Anna V., Nicholas J. Brewin, and Viktor E. Tsyganov. 2021. "Structure and Development of the Legume-Rhizobial Symbiotic Interface in Infection Threads" Cells 10, no. 5: 1050. https://doi.org/10.3390/cells10051050
APA StyleTsyganova, A. V., Brewin, N. J., & Tsyganov, V. E. (2021). Structure and Development of the Legume-Rhizobial Symbiotic Interface in Infection Threads. Cells, 10(5), 1050. https://doi.org/10.3390/cells10051050








