Review: Mechanisms of Glyphosate and Glyphosate-Based Herbicides Action in Female and Male Fertility in Humans and Animal Models
Abstract
:1. Introduction
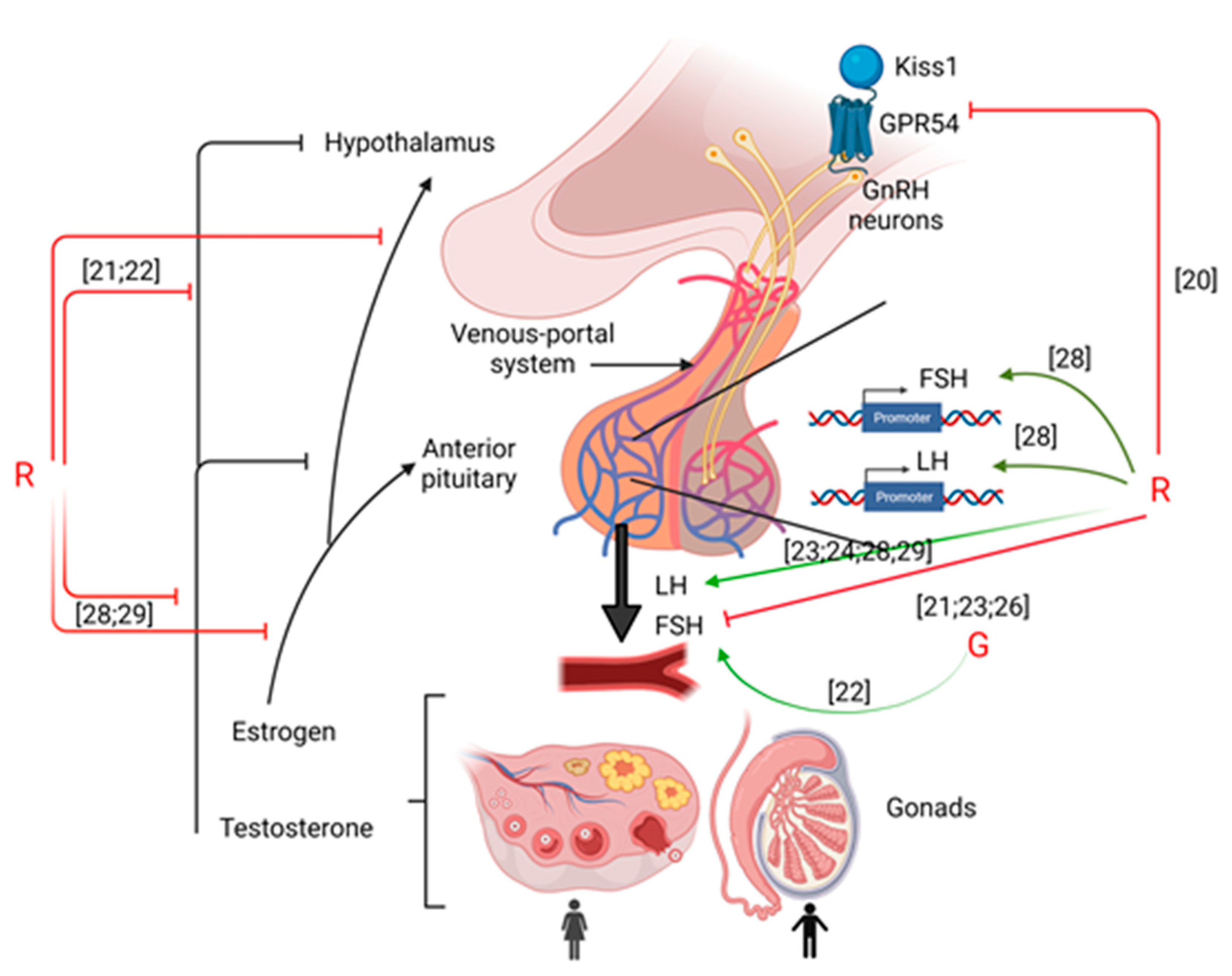
2. Hypothalamus
2.1. Kiss Expression
2.2. GnRH Secretion
3. Pituitary
3.1. Long Term Exposure to GBHs
3.2. Gestational Exposure to GBHs
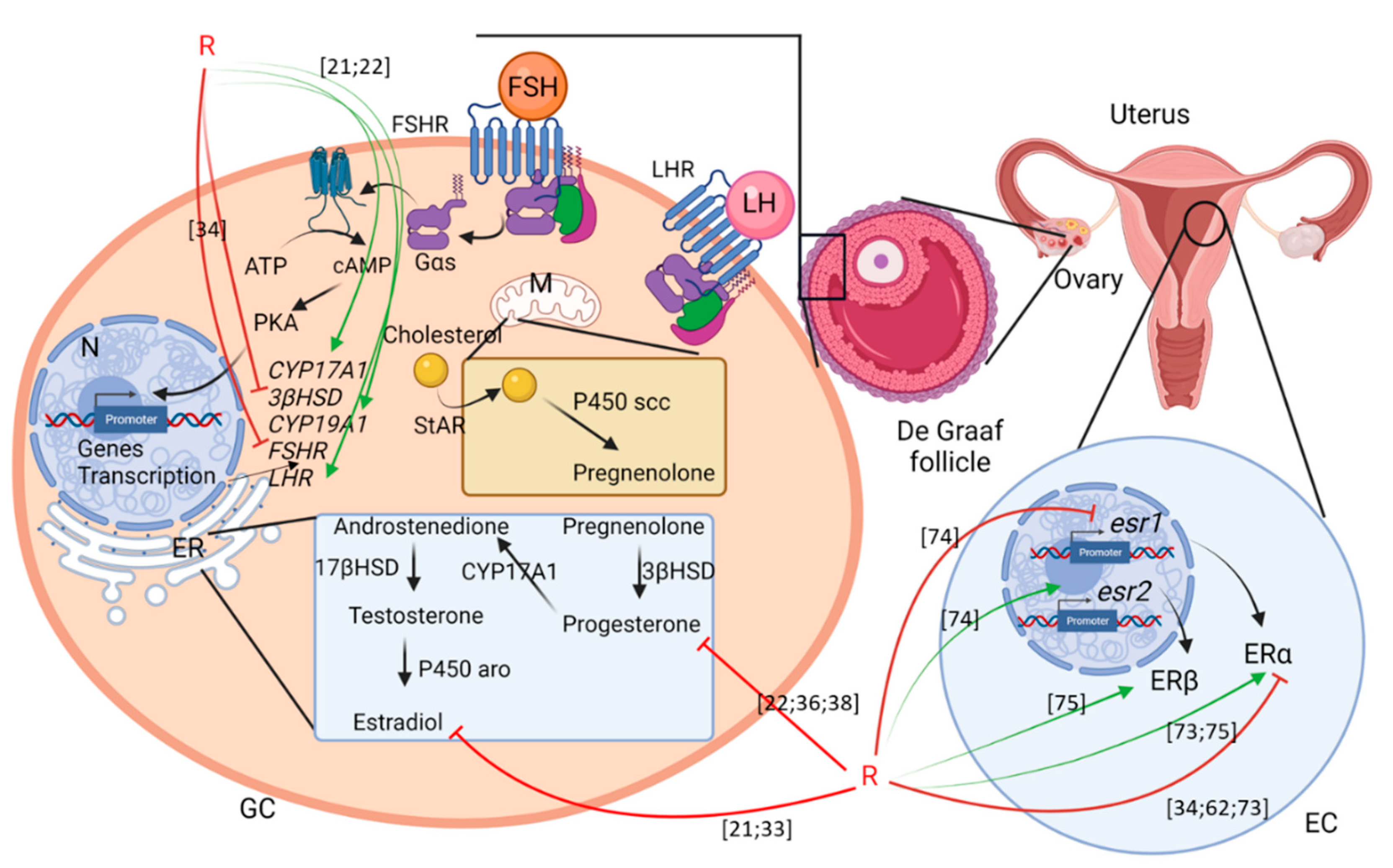
4. Gonads
4.1. Ovary
4.1.1. Steroidogenesis
4.1.2. Ovarian Alterations
In Vivo Studies
In Vitro Studies
4.2. Testis
4.2.1. In Vivo Studies
4.2.2. In Vitro Studies
4.2.3. Sertoli Cells
In Vivo Studies
In Vitro Studies
4.2.4. Germ Cells Alterations
In Vivo Studies
In Vitro Studies
4.2.5. Other Alterations of Male Reproductive System
5. Uterus
5.1. In Utero and Perinatal Development
5.2. Puberty
5.3. Peri-Implantation Period
6. Placenta
7. Embryo Development
7.1. In Vitro Studies
7.2. In Vivo Studies
8. Transgenerational Effects
9. Involvement of Glyphosate or GBH in Reproductive Pathologies
10. Conclusions and Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 3βHSD | 3 β-hydroxysteroid dehydrogenase |
| ADI | Acceptable daily intake |
| AOEL | Acceptable operator exposure level |
| AM | Antimesometrial |
| AMPA | Amino-methyl-phosphonic acid |
| ATP | Adenosine triphosphate |
| BTB | Blood-testis barrier |
| Ca2+ | Calcium |
| cAMP | Cyclic adenosine 3′,5′-monophosphate |
| CAT | Catalase |
| COCs | Cumulus-oocyte complexes |
| CYP11a1 | Cytochrome P450 family 11 subfamily A member 1 |
| CYP17a1 | Cytochrome P450 family 17 subfamily A member 1 |
| CYP19a1 | Cytochrome P450 family 19 subfamily A member 1 |
| Δ4 | Androstenedione |
| DDI | Dietary Daily Intake value |
| DHEA | Dehydroepiandrosterone |
| DHR | Differential histone retention region |
| Dmnt1 | DNA (cytosine-5)-methyltransferase 1 |
| Dmrt1 | Doublesex and mab-3 related transcription factor 1 |
| DMR | DNA methylation region |
| E2 | Estradiol |
| EC | Endometrial cells |
| ECM | extracellular matrix |
| ED | Endocrine disruptors |
| EDC | Endocrine-disrupting chemical |
| EFSA | European Food Safety Authority |
| EPSPS | 5-enolpyruvylshikimate-3-phosphate synthase |
| ER | Estrogen receptor |
| FAO | Food and Agricultural Organization of the United Nation |
| Foxa2 | Forkhead box a2 |
| FSH | Follicle-stimulating hormone |
| G | Glyphosate |
| GBH | Glyphosate-based herbicide |
| GC | Granulosa cells |
| GD | Gestational day |
| GDF9 | Growth differentiation factor 9 |
| GE | Glandular epithelium |
| GnRH | Gonadotropin-releasing hormone |
| HDAC3 | Histone Deacetylase 3 |
| HHR | Hypothalamic-Pituitary-Renal |
| Hox a10 | Homeobox a10 |
| HPG | Hypothalamic-pituitary-gonadal |
| HPT | Hypothalamic-Pituitary-Thyroid |
| ICM | Inner cellular mass |
| IGF1 | Insulin-like growth factor 1 |
| Insl3 | Insuline-like-3 |
| IP3 | Inositol triphosphate |
| KCs | Key characteristics |
| Kiss1 | Kisspeptin protein |
| LA | Alpha-lipoic acid |
| LC | Leydig cells |
| LE | Uterine luminal epithelium |
| LH | Luteinizing hormone |
| M | Mesometrial |
| MDA | Malondialdehyde |
| mg/kg/bw/d | mg/kg bodyweight/day |
| MMP | Mitochondrial membrane potential |
| MRL | Maximum Residues Limits |
| MTOC | MicroTubule-Organizing Center |
| NADPH | Nicotinamide adenine dinucleotide phosphate |
| NO | Nitrogen oxide |
| NOAEL | No observed adverse effect level |
| NR1D1 | Nuclear receptor subfamily 1, group D member 1 |
| PBMC | Peripheral blood mononuclear cell |
| PCOS | Polycystic ovary syndrome |
| P450 scc | Sidechain cytochrome P450 |
| Pg | Progesterone |
| PI3K | Phosphoinositide 3-kinase |
| PKA | Protein Kinase A |
| PKC | Protein Kinase C |
| PM | Pesticide mixture |
| PND | Post-natal day |
| POA | Preoptic area |
| POEA | Polyoxyethylene tallow amine |
| SS | Endometrial stroma |
| R | Roundup |
| ROS | Reactive oxygen species |
| SC | Sertoli cells |
| SOD | Superoxide dismutase |
| ST | Seminiferous tube |
| StAR | Steroidogenic Acute Regulatory protein |
| T | Testosterone |
| TE | Trophectoderm |
| TH | Thyroid hormone |
References
- Muñoz, J.P.; Bleak, T.C.; Calaf, G.M. Glyphosate and the Key Characteristics of an Endocrine Disruptor: A Review. Chemosphere 2021, 270, 128619. [Google Scholar] [CrossRef]
- Defarge, N.; Takács, E.; Lozano, V.; Mesnage, R.; Spiroux de Vendômois, J.; Séralini, G.-E.; Székács, A. Co-Formulants in Glyphosate-Based Herbicides Disrupt Aromatase Activity in Human Cells below Toxic Levels. Int. J. Environ. Res. Public Health 2016, 13, 264. [Google Scholar] [CrossRef] [Green Version]
- Defarge, N.; Spiroux de Vendômois, J.; Séralini, G.E. Toxicity of Formulants and Heavy Metals in Glyphosate-Based Herbicides and Other Pesticides. Toxicol. Rep. 2018, 5, 156–163. [Google Scholar] [CrossRef]
- Agostini, L.P.; Dettogni, R.S.; dos Reis, R.S.; Stur, E.; dos Santos, E.V.W.; Ventorim, D.P.; Garcia, F.M.; Cardoso, R.C.; Graceli, J.B.; Louro, I.D. Effects of Glyphosate Exposure on Human Health: Insights from Epidemiological and in Vitro Studies. Sci. Total Environ. 2020, 705, 135808. [Google Scholar] [CrossRef] [PubMed]
- Mesnage, R.; Defarge, N.; Spiroux de Vendômois, J.; Séralini, G.E. Potential Toxic Effects of Glyphosate and Its Commercial Formulations below Regulatory Limits. Food Chem. Toxicol. 2015, 84, 133–153. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hori, Y.; Fujisawa, M.; Shimada, K.; Hirose, Y. Determination of the Herbicide Glyphosate and its Metabolite in Biological Specimens by Gas chromatography-mass Spectrometry. A Case of Poisoning by Roundup® Herbicide. J. Anal. Toxicol. 2003, 27, 162–166. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- European Food Safety Authority (EFSA). Peer Review of the Pesticide Risk Assessment of the Potential Endocrine Disrupting Properties of Glyphosate. Eur. Food Saf. Auth. J. 2017, 15, e04979. [Google Scholar] [CrossRef]
- Gillezeau, C.; van Gerwen, M.; Shaffer, R.M.; Rana, I.; Zhang, L.; Sheppard, L.; Taioli, E. The Evidence of Human Exposure to Glyphosate: A Review. Environ. Health 2019, 18, 2. [Google Scholar] [CrossRef] [Green Version]
- Taxvig, C.; Hass, U.; Axelstad, M.; Dalgaard, M.; Boberg, J.; Andeasen, H.R.; Vinggaard, A.M. Endocrine-Disrupting Activities In Vivo of the Fungicides Tebuconazole and Epoxiconazole. Toxicol. Sci. 2007, 100, 464–473. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- La Merrill, M.A.; Vandenberg, L.N.; Smith, M.T.; Goodson, W.; Browne, P.; Patisaul, H.B.; Guyton, K.Z.; Kortenkamp, A.; Cogliano, V.J.; Woodruff, T.J.; et al. Consensus on the Key Characteristics of Endocrine-Disrupting Chemicals as a Basis for Hazard Identification. Nat. Rev. Endocrinol. 2020, 16, 45–57. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ingaramo, P.; Alarcón, R.; Muñoz-de-Toro, M.; Luque, E.H. Are Glyphosate and Glyphosate-Based Herbicides Endocrine Disruptors That Alter Female Fertility? Mol. Cell. Endocrinol. 2020, 518, 110934. [Google Scholar] [CrossRef] [PubMed]
- Hao, Y.; Zhang, Y.; Cheng, J.; Xu, W.; Xu, Z.; Gao, J.; Tao, L. Adjuvant Contributes Roundup’s Unexpected Effects on A549 Cells. Environ. Res. 2020, 184, 109306. [Google Scholar] [CrossRef] [PubMed]
- Mesnage, R.; Mazzacuva, F.; Caldwell, A.; Halket, J.; Antoniou, M.N. Urinary Excretion of Herbicide Co-Formulants after Oral Exposure to Roundup MON 52276 in Rats. Environ. Res. 2021, 197, 111103. [Google Scholar] [CrossRef] [PubMed]
- Cao, C.; Ding, Y.; Kong, X.; Feng, G.; Xiang, W.; Chen, L.; Yang, F.; Zhang, K.; Chu, M.; Wang, P.; et al. Reproductive Role of MiRNA in the Hypothalamic-Pituitary Axis. Mol. Cell. Neurosci. 2018, 88, 130–137. [Google Scholar] [CrossRef] [PubMed]
- Uenoyama, Y.; Inoue, N.; Maeda, K.; Tsukamura, H. The Roles of Kisspeptin in the Mechanism Underlying Reproductive Functions in Mammals. J. Reprod. Dev. 2018, 64, 469–476. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cericato, L.; Neto, J.G.M.; Fagundes, M.; Kreutz, L.C.; Quevedo, R.M.; Finco, J.; Centenaro, L.; Pottker, E.; Anziliero, D.; Barcellos, L.J.G. Cortisol Response to Acute Stress in Jundiá Rhamdia Quelen Acutely Exposed to Sub-Lethal Concentrations of Agrichemicals. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2008, 148, 281–286. [Google Scholar] [CrossRef] [PubMed]
- De Souza, J.S.; Kizys, M.M.L.; da Conceição, R.R.; Glebocki, G.; Romano, R.M.; Ortiga-Carvalho, T.M.; Giannocco, G.; da Silva, I.D.C.G.; Dias da Silva, M.R.; Romano, M.A.; et al. Perinatal Exposure to Glyphosate-Based Herbicide Alters the Thyrotrophic Axis and Causes Thyroid Hormone Homeostasis Imbalance in Male Rats. Toxicology 2017, 377, 25–37. [Google Scholar] [CrossRef]
- Martinez, A.; Al-Ahmad, A.J. Effects of Glyphosate and Aminomethylphosphonic Acid on an Isogeneic Model of the Human Blood-Brain Barrier. Toxicol. Lett. 2019, 304, 39–49. [Google Scholar] [CrossRef]
- Smith, C.M.; Vera, M.K.M.; Bhandari, R.K. Developmental and Epigenetic Effects of Roundup and Glyphosate Exposure on Japanese Medaka (Oryzias Latipes). Aquat. Toxicol. 2019, 210, 215–226. [Google Scholar] [CrossRef]
- Fu, H.; Feng, G.; Xiaoxu, W.; Peng, T.; Shengnan, Q.; Baoming, S.; Anshan, S. Effects of Glyphosate-Based Herbicide-Contaminated Diets on Reproductive Organ Toxicity and Hypothalamic-Pituitary-Ovarian Axis Hormones in Weaned Piglets. Environ. Pollut. 2021, 272, 115596. [Google Scholar] [CrossRef] [PubMed]
- Ren, X.; Li, R.; Liu, J.; Huang, K.; Wu, S.; Li, Y.; Li, C. Effects of Glyphosate on the Ovarian Function of Pregnant Mice, the Secretion of Hormones and the Sex Ratio of Their Fetuses. Environ. Pollut. 2018, 243, 833–841. [Google Scholar] [CrossRef]
- Popoola, S.O.; Sakpa, C.L. Hormones of Pituitary-Gonadal Axis and Histology of Pituitary Gland Following Oral Treatment of Male Wistar Rats with Glyphosate. J. Biomed. Res. Clin. Pract. 2018, 1, 164–169. [Google Scholar] [CrossRef]
- Astiz, M.; Hurtado de Catalfo, G.E.; García, M.N.; Galletti, S.M.; Errecalde, A.L.; de Alaniz, M.J.T.; Marra, C.A. Pesticide-Induced Decrease in Rat Testicular Steroidogenesis Is Differentially Prevented by Lipoate and Tocopherol. Ecotoxicol. Environ. Saf. 2013, 91, 129–138. [Google Scholar] [CrossRef]
- Abarikwu, S.O.; Akiri, O.F.; Durojaiye, M.A.; Adenike, A. Combined Effects of Repeated Administration of Bretmont Wipeout (Glyphosate) and Ultrazin (Atrazine) on Testosterone, Oxidative Stress and Sperm Quality of Wistar Rats. Toxicol. Mech. Methods 2015, 25, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Owagboriaye, F.O.; Dedeke, G.A.; Ademolu, K.O.; Olujimi, O.O.; Ashidi, J.S.; Adeyinka, A.A. Reproductive Toxicity of Roundup Herbicide Exposure in Male Albino Rat. Exp. Toxicol. Pathol. 2017, 69, 461–468. [Google Scholar] [CrossRef]
- Séralini, G.-E.; Clair, E.; Mesnage, R.; Gress, S.; Defarge, N.; Malatesta, M.; Hennequin, D.; de Vendômois, J.S. Republished Study: Long-Term Toxicity of a Roundup Herbicide and a Roundup-Tolerantgenetically Modified Maize. Environ. Sci. Eur. 2014, 26, 14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Romano, M.A.; Romano, R.M.; Santos, L.D.; Wisniewski, P.; Campos, D.A.; de Souza, P.B.; Viau, P.; Bernardi, M.M.; Nunes, M.T.; de Oliveira, C.A. Glyphosate Impairs Male Offspring Reproductive Development by Disrupting Gonadotropin Expression. Arch. Toxicol. 2012, 86, 663–673. [Google Scholar] [CrossRef]
- Teleken, J.L.; Gomes, E.C.Z.; Marmentini, C.; Moi, M.B.; Ribeiro, R.A.; Balbo, S.L.; Amorim, E.M.P.; Bonfleur, M.L. Glyphosate-Based Herbicide Exposure during Pregnancy and Lactation Malprograms the Male Reproductive Morphofunction in F1 Offspring. J. Dev. Orig. Health Dis. 2020, 11, 146–153. [Google Scholar] [CrossRef] [PubMed]
- Gigante, P.; Berni, M.; Bussolati, S.; Grasselli, F.; Grolli, S.; Ramoni, R.; Basini, G. Glyphosate Affects Swine Ovarian and Adipose Stromal Cell Functions. Anim. Reprod. Sci. 2018, 195, 185–196. [Google Scholar] [CrossRef] [PubMed]
- Manservisi, F.; Lesseur, C.; Panzacchi, S.; Mandrioli, D.; Falcioni, L.; Bua, L.; Manservigi, M.; Spinaci, M.; Galeati, G.; Mantovani, A.; et al. The Ramazzini Institute 13-Week Pilot Study Glyphosate-Based Herbicides Administered at Human-Equivalent Dose to Sprague Dawley Rats: Effects on Development and Endocrine System. Environ. Health 2019, 18, 15. [Google Scholar] [CrossRef] [Green Version]
- Ganesan, S.; Keating, A.F. Ovarian Mitochondrial and Oxidative Stress Proteins Are Altered by Glyphosate Exposure in Mice. Toxicol. Appl. Pharmacol. 2020, 402, 115116. [Google Scholar] [CrossRef] [PubMed]
- Hamdaoui, L.; Naifar, M.; Rahmouni, F.; Harrabi, B.; Ayadi, F.; Sahnoun, Z.; Rebai, T. Subchronic Exposure to Kalach 360 SL-Induced Endocrine Disruption and Ovary Damage in Female Rats. Arch. Physiol. Biochem. 2018, 124, 27–34. [Google Scholar] [CrossRef]
- Alarcón, R.; Rivera, O.E.; Ingaramo, P.I.; Tschopp, M.V.; Dioguardi, G.H.; Milesi, M.M.; Muñoz-de-Toro, M.; Luque, E.H. Neonatal Exposure to a Glyphosate-Based Herbicide Alters the Uterine Differentiation of Prepubertal Ewe Lambs. Environ. Pollut. 2020, 265, 114874. [Google Scholar] [CrossRef]
- Perego, M.C.; Caloni, F.; Cortinovis, C.; Schutz, L.F.; Albonico, M.; Tsuzukibashi, D.; Spicer, L.J. Influence of a Roundup Formulation on Glyphosate Effects on Steroidogenesis and Proliferation of Bovine Granulosa Cells in Vitro. Chemosphere 2017, 188, 274–279. [Google Scholar] [CrossRef]
- Perego, M.C.; Schutz, L.F.; Caloni, F.; Cortinovis, C.; Albonico, M.; Spicer, L.J. Evidence for Direct Effects of Glyphosate on Ovarian Function: Glyphosate Influences Steroidogenesis and Proliferation of Bovine Granulosa but Not Theca Cells in Vitro: Glyphosate Effects on Bovine Granulosa. J. Appl. Toxicol. 2017, 37, 692–698. [Google Scholar] [CrossRef]
- Wrobel, M.H. Glyphosate Affects the Secretion of Regulators of Uterine Contractions in Cows While It Does Not Directly Impair the Motoric Function of Myometrium in Vitro. Toxicol. Appl. Pharmacol. 2018, 349, 55–61. [Google Scholar] [CrossRef]
- Spinaci, M.; Nerozzi, C.; lo Tamanini, C.; Bucci, D.; Galeati, G. Glyphosate and Its Formulation Roundup Impair Pig Oocyte Maturation. Sci. Rep. 2020, 10, 12007. [Google Scholar] [CrossRef]
- Armiliato, N.; Ammar, D.; Nezzi, L.; Straliotto, M.; Muller, Y.M.R.; Nazari, E.M. Changes in Ultrastructure and Expression of Steroidogenic Factor-1 in Ovaries of Zebrafish Danio Rerio Exposed to Glyphosate. J. Toxicol. Environ. Health A 2014, 77, 405–414. [Google Scholar] [CrossRef] [PubMed]
- Alarcón, R.; Ingaramo, P.I.; Rivera, O.E.; Dioguardi, G.H.; Repetti, M.R.; Demonte, L.D.; Milesi, M.M.; Varayoud, J.; Muñoz-de-Toro, M.; Luque, E.H. Neonatal Exposure to a Glyphosate-Based Herbicide Alters the Histofunctional Differentiation of the Ovaries and Uterus in Lambs. Mol. Cell. Endocrinol. 2019, 482, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Davico, C.E.; Pereira, A.G.; Nezzi, L.; Jaramillo, M.L.; de Melo, M.S.; Müller, Y.M.R.; Nazari, E.M. Reproductive Toxicity of Roundup WG® Herbicide: Impairments in Ovarian Follicles of Model Organism Danio Rerio. Environ. Sci. Pollut. Res. 2021, 28, 15147–15159. [Google Scholar] [CrossRef]
- Bhardwaj, J.K.; Mittal, M.; Saraf, P. Effective Attenuation of Glyphosate-Induced Oxidative Stress and Granulosa Cell Apoptosis by Vitamins C and E in Caprines. Mol. Reprod. Dev. 2019, 86, 42–52. [Google Scholar] [CrossRef] [Green Version]
- Mäkelä, J.-A.; Koskenniemi, J.J.; Virtanen, H.E.; Toppari, J. Testis Development. Endocr. Rev. 2019, 40, 857–905. [Google Scholar] [CrossRef]
- Gorga, A.; Rindone, G.M.; Centola, C.L.; Sobarzo, C.M.; Pellizzari, E.H.; Del Carmen Camberos, M.; Marín-Briggiler, C.I.; Cohen, D.J.; Riera, M.F.; Galardo, M.N.; et al. Low Doses of Glyphosate/Roundup Alter Blood–Testis Barrier Integrity in Juvenile Rats. Front. Endocrinol. 2021, 12, 615678. [Google Scholar] [CrossRef] [PubMed]
- Dallegrave, E.; Mantese, F.D.; Oliveira, R.T.; Andrade, A.J.M.; Dalsenter, P.R.; Langeloh, A. Pre- and Postnatal Toxicity of the Commercial Glyphosate Formulation in Wistar Rats. Arch. Toxicol. 2007, 81, 665–673. [Google Scholar] [CrossRef]
- Romano, R.M.; Romano, M.A.; Bernardi, M.M.; Furtado, P.V.; Oliveira, C.A. Prepubertal Exposure to Commercial Formulation of the Herbicide Glyphosate Alters Testosterone Levels and Testicular Morphology. Arch. Toxicol. 2010, 84, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Nardi, J.; Moras, P.B.; Koeppe, C.; Dallegrave, E.; Leal, M.B.; Rossato-Grando, L.G. Prepubertal Subchronic Exposure to Soy Milk and Glyphosate Leads to Endocrine Disruption. Food Chem. Toxicol. 2017, 100, 247–252. [Google Scholar] [CrossRef]
- Razi, M.; Shahmohamadloo, S. Histological and Histochemical Effects of Gly-Phosate on Testicular Tissue and Function. Iran. J. Reprod. Med. 2012, 10, 181–192. [Google Scholar] [PubMed]
- Cassault-Meyer, E.; Gress, S.; Séralini, G.-É.; Galeraud-Denis, I. An Acute Exposure to Glyphosate-Based Herbicide Alters Aromatase Levels in Testis and Sperm Nuclear Quality. Environ. Toxicol. Pharmacol. 2014, 38, 131–140. [Google Scholar] [CrossRef]
- Dai, P.; Hu, P.; Tang, J.; Li, Y.; Li, C. Effect of Glyphosate on Reproductive Organs in Male Rat. Acta Histochem. 2016, 118, 519–526. [Google Scholar] [CrossRef]
- Johansson, H.K.L.; Schwartz, C.L.; Nielsen, L.N.; Boberg, J.; Vinggaard, A.M.; Bahl, M.I.; Svingen, T. Exposure to a Glyphosate-Based Herbicide Formulation, but Not Glyphosate Alone, Has Only Minor Effects on Adult Rat Testis. Reprod. Toxicol. 2018, 82, 25–31. [Google Scholar] [CrossRef] [Green Version]
- Mutwedu, V.B.; Nyongesa, A.W.; Azine, P.C.; Chiregereza, D.K.; Ngoumtsop, V.H.; Mugumaarhahama, Y.; Ayagirwe, R.B.B. Growth Performance and Reproductive Function Impairment of Glyphosate-based Herbicide in Male Guinea Pig (Cavia Porcellus). Vet. Med. Sci. 2021, 7, 1047–1055. [Google Scholar] [CrossRef]
- Oliveira, A.G.; Telles, L.F.; Hess, R.A.; Mahecha, G.A.B.; Oliveira, C.A. Effects of the Herbicide Roundup on the Epididymal Region of Drakes Anas Platyrhynchos. Reprod. Toxicol. 2007, 23, 182–191. [Google Scholar] [CrossRef]
- Pham, T.H.; Derian, L.; Kervarrec, C.; Kernanec, P.-Y.; Jégou, B.; Smagulova, F.; Gely-Pernot, A. Perinatal Exposure to Glyphosate and a Glyphosate-Based Herbicide Affect Spermatogenesis in Mice. Toxicol. Sci. 2019, 169, 260–271. [Google Scholar] [CrossRef]
- Ruuskanen, S.; Rainio, M.J.; Gómez-Gallego, C.; Selenius, O.; Salminen, S.; Collado, M.C.; Saikkonen, K.; Saloniemi, I.; Helander, M. Glyphosate-Based Herbicides Influence Antioxidants, Reproductive Hormones and Gut Microbiome but Not Reproduction: A Long-Term Experiment in an Avian Model. Environ. Pollut. 2020, 266, 115108. [Google Scholar] [CrossRef] [PubMed]
- Yousef, M.I.; Salem, M.H.; Ibrahim, H.Z.; Helmi, S.; Seehy, M.A.; Bertheussen, K. Toxic Effects of Carbofuran and Glyphosate on Semen Characteristics in Rabbits. J. Environ. Sci. Health Part B 1995, 30, 513–534. [Google Scholar] [CrossRef]
- Zhao, L.; Zhang, J.; Yang, L.; Zhang, H.; Zhang, Y.; Gao, D.; Jiang, H.; Li, Y.; Dong, H.; Ma, T.; et al. Glyphosate Exposure Attenuates Testosterone Synthesis via NR1D1 Inhibition of StAR Expression in Mouse Leydig Cells. Sci. Total Environ. 2021, 785, 147323. [Google Scholar] [CrossRef] [PubMed]
- Clair, É.; Mesnage, R.; Travert, C.; Séralini, G.-É. A Glyphosate-Based Herbicide Induces Necrosis and Apoptosis in Mature Rat Testicular Cells in Vitro, and Testosterone Decrease at Lower Levels. Toxicol. In Vitro 2012, 26, 269–279. [Google Scholar] [CrossRef] [PubMed]
- Vanlaeys, A.; Dubuisson, F.; Seralini, G.-E.; Travert, C. Formulants of Glyphosate-Based Herbicides Have More Deleterious Impact than Glyphosate on TM4 Sertoli Cells. Toxicol. In Vitro 2018, 52, 14–22. [Google Scholar] [CrossRef]
- Walsh, L.P.; McCormick, C.; Martin, C.; Stocco, D.M. Roundup Inhibits Steroidogenesis by Disrupting Steroidogenic Acute Regulatory (StAR) Protein Expression. Environ. Health Perspect. 2000, 108, 769–776. [Google Scholar] [CrossRef]
- Benachour, N.; Sipahutar, H.; Moslemi, S.; Gasnier, C.; Travert, C.; Séralini, G.E. Time- and Dose-Dependent Effects of Roundup on Human Embryonic and Placental Cells. Arch. Environ. Contam. Toxicol. 2007, 53, 126–133. [Google Scholar] [CrossRef]
- Richard, S.; Moslemi, S.; Sipahutar, H.; Benachour, N.; Seralini, G.-E. Differential Effects of Glyphosate and Roundup on Human Placental Cells and Aromatase. Environ. Health Perspect. 2005, 113, 716–720. [Google Scholar] [CrossRef] [Green Version]
- Avdatek, F.; Birdane, Y.O.; Türkmen, R.; Demirel, H.H. Ameliorative Effect of Resveratrol on Testicular Oxidative Stress, Spermatological Parameters and DNA Damage in Glyphosate-Based Herbicide-Exposed Rats. Andrologia 2018, 50, e13036. [Google Scholar] [CrossRef] [PubMed]
- Gress, S.; Laurant, C.; Defarge, N.; Travert, C.; Séralini, G.-É. Dig1 Protects against Locomotor and Biochemical Dysfunctions Provoked by Roundup. BMC Complement. Altern. Med. 2016, 16, 234. [Google Scholar] [CrossRef] [Green Version]
- Xia, Y.; Yang, X.; Lu, J.; Xie, Q.; Ye, A.; Sun, W. The Endoplasmic Reticulum Stress and Related Signal Pathway Mediated the Glyphosate-Induced Testosterone Synthesis Inhibition in TM3 Cells. Environ. Pollut. 2020, 260, 113949. [Google Scholar] [CrossRef]
- De Liz Oliveira Cavalli, V.L.; Cattani, D.; Heinz Rieg, C.E.; Pierozan, P.; Zanatta, L.; Benedetti Parisotto, E.; Wilhelm Filho, D.; Mena Barreto Silva, F.R.; Pessoa-Pureur, R.; Zamoner, A. Roundup Disrupts Male Reproductive Functions by Triggering Calcium-Mediated Cell Death in Rat Testis and Sertoli Cells. Free Radic. Biol. Med. 2013, 65, 335–346. [Google Scholar] [CrossRef] [Green Version]
- Jiang, X.; Zhang, N.; Yin, L.; Zhang, W.; Han, F.; Liu, W.; Chen, H.; Cao, J.; Liu, J. A Commercial Roundup® Formulation Induced Male Germ Cell Apoptosis by Promoting the Expression of XAF1 in Adult Mice. Toxicol. Lett. 2018, 296, 163–172. [Google Scholar] [CrossRef]
- Nerozzi, C.; Recuero, S.; Galeati, G.; Bucci, D.; Spinaci, M.; Yeste, M. Effects of Roundup and Its Main Component, Glyphosate, upon Mammalian Sperm Function and Survival. Sci. Rep. 2020, 10, 11026. [Google Scholar] [CrossRef]
- Anifandis, G.; Amiridis, G.; Dafopoulos, K.; Daponte, A.; Dovolou, E.; Gavriil, E.; Gorgogietas, V.; Kachpani, E.; Mamuris, Z.; Messini, C.; et al. The In Vitro Impact of the Herbicide Roundup on Human Sperm Motility and Sperm Mitochondria. Toxics 2017, 6, 2. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anifandis, G.; Katsanaki, K.; Lagodonti, G.; Messini, C.; Simopoulou, M.; Dafopoulos, K.; Daponte, A. The Effect of Glyphosate on Human Sperm Motility and Sperm DNA Fragmentation. Int. J. Env. Res. Public Health 2018, 15, 1117. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferramosca, A.; Lorenzetti, S.; Di Giacomo, M.; Murrieri, F.; Coppola, L.; Zara, V. Herbicides Glyphosate and Glufosinate Ammonium Negatively Affect Human Sperm Mitochondria Respiration Efficiency. Reprod. Toxicol. 2021, 99, 48–55. [Google Scholar] [CrossRef]
- Ingaramo, P.I.; Varayoud, J.; Milesi, M.M.; Schimpf, M.G.; Muñoz-de-Toro, M.; Luque, E.H. Effects of Neonatal Exposure to a Glyphosate-Based Herbicide on Female Rat Reproduction. Reproduction 2016, 152, 403–415. [Google Scholar] [CrossRef] [Green Version]
- Guerrero Schimpf, M.; Milesi, M.M.; Ingaramo, P.I.; Luque, E.H.; Varayoud, J. Neonatal Exposure to a Glyphosate Based Herbicide Alters the Development of the Rat Uterus. Toxicology 2017, 376, 2–14. [Google Scholar] [CrossRef]
- Guerrero Schimpf, M.; Milesi, M.M.; Luque, E.H.; Varayoud, J. Glyphosate-Based Herbicide Enhances the Uterine Sensitivity to Estradiol in Rats. J. Endocrinol. 2018, 239, 197–213. [Google Scholar] [CrossRef] [Green Version]
- Varayoud, J.; Durando, M.; Ramos, J.G.; Milesi, M.M.; Ingaramo, P.I.; Muñoz-de-Toro, M.; Luque, E.H. Effects of a Glyphosate-Based Herbicide on the Uterus of Adult Ovariectomized Rats: Herbicide’s Effect on Castrated Rat Uterus. Environ. Toxicol. 2017, 32, 1191–1201. [Google Scholar] [CrossRef] [PubMed]
- Lorenz, V.; Pacini, G.; Luque, E.H.; Varayoud, J.; Milesi, M.M. Perinatal Exposure to Glyphosate or a Glyphosate-Based Formulation Disrupts Hormonal and Uterine Milieu during the Receptive State in Rats. Food Chem. Toxicol. 2020, 143, 111560. [Google Scholar] [CrossRef] [PubMed]
- Gasnier, C.; Dumont, C.; Benachour, N.; Clair, E.; Chagnon, M.-C.; Séralini, G.-E. Glyphosate-Based Herbicides Are Toxic and Endocrine Disruptors in Human Cell Lines. Toxicology 2009, 262, 184–191. [Google Scholar] [CrossRef] [PubMed]
- Gastiazoro, M.P.; Durando, M.; Milesi, M.M.; Lorenz, V.; Vollmer, G.; Varayoud, J.; Zierau, O. Glyphosate Induces Epithelial Mesenchymal Transition-Related Changes in Human Endometrial Ishikawa Cells via Estrogen Receptor Pathway. Mol. Cell. Endocrinol. 2020, 510, 110841. [Google Scholar] [CrossRef]
- Benachour, N.; Séralini, G.-E. Glyphosate Formulations Induce Apoptosis and Necrosis in Human Umbilical, Embryonic, and Placental Cells. Chem. Res. Toxicol. 2009, 22, 97–105. [Google Scholar] [CrossRef]
- Kongtip, P.; Nankongnab, N.; Phupancharoensuk, R.; Palarach, C.; Sujirarat, D.; Sangprasert, S.; Sermsuk, M.; Sawattrakool, N.; Woskie, S.R. Glyphosate and Paraquat in Maternal and Fetal Serums in Thai Women. J. Agromedicine 2017, 22, 282–289. [Google Scholar] [CrossRef]
- Mose, T.; Kjaerstad, M.B.; Mathiesen, L.; Nielsen, J.B.; Edelfors, S.; Knudsen, L.E. Placental Passage of Benzoic Acid, Caffeine, and Glyphosate in an Ex Vivo Human Perfusion System. J. Toxicol. Environ. Health A 2008, 71, 984–991. [Google Scholar] [CrossRef]
- Simasotchi, C.; Chissey, A.; Jungers, G.; Fournier, T.; Seralini, G.-E.; Gil, S. A Glyphosate-Based Formulation but Not Glyphosate Alone Alters Human Placental Integrity. Toxics 2021, 9, 220. [Google Scholar] [CrossRef]
- Yahfoufi, Z.A.; Bai, D.; Khan, S.N.; Chatzicharalampous, C.; Kohan-Ghadr, H.-R.; Morris, R.T.; Abu-Soud, H.M. Glyphosate Induces Metaphase II Oocyte Deterioration and Embryo Damage by Zinc Depletion and Overproduction of Reactive Oxygen Species. Toxicology 2020, 439, 152466. [Google Scholar] [CrossRef] [PubMed]
- Cai, W.; Yang, X.; Li, X.; Li, H.; Wang, S.; Wu, Z.; Yu, M.; Ma, S.; Tang, S. Low-Dose Roundup Induces Developmental Toxicity in Bovine Preimplantation Embryos in Vitro. Environ. Sci. Pollut. Res. 2020, 27, 16451–16459. [Google Scholar] [CrossRef] [PubMed]
- Fathi, M.A.; Abdelghani, E.; Shen, D.; Ren, X.; Dai, P.; Li, Z.; Tang, Q.; Li, Y.; Li, C. Effect of in Ovo Glyphosate Injection on Embryonic Development, Serum Biochemistry, Antioxidant Status and Histopathological Changes in Newly Hatched Chicks. J. Anim. Physiol. Anim. Nutr. 2019, 103, 1776–1784. [Google Scholar] [CrossRef]
- Fathi, M.A.; Han, G.; Kang, R.; Shen, D.; Shen, J.; Li, C. Disruption of Cytochrome P450 Enzymes in the Liver and Small Intestine in Chicken Embryos in Ovo Exposed to Glyphosate. Environ. Sci. Pollut. Res. 2020, 27, 16865–16875. [Google Scholar] [CrossRef]
- Szabó, R.; Szemerédy, G.; Kormos, É.; Lehel, J.; Budai, P. Studies on joint toxic effects of a glyphosate herbicide (fozát 480) and a heavy metal (cadmium) on chicken embryos. Agrofor Int. J. 2018, 2, 64–71. [Google Scholar] [CrossRef]
- Uren Webster, T.M.; Laing, L.V.; Florance, H.; Santos, E.M. Effects of Glyphosate and Its Formulation, Roundup, on Reproduction in Zebrafish (Danio Rerio). Environ. Sci. Technol. 2014, 48, 1271–1279. [Google Scholar] [CrossRef]
- Díaz-Martín, R.D.; Valencia-Hernández, J.D.; Betancourt-Lozano, M.; Yáñez-Rivera, B. Changes in Microtubule Stability in Zebrafish (Danio Rerio) Embryos after Glyphosate Exposure. Heliyon 2021, 7, e06027. [Google Scholar] [CrossRef]
- Schweizer, M.; Brilisauer, K.; Triebskorn, R.; Forchhammer, K.; Köhler, H.-R. How Glyphosate and Its Associated Acidity Affect Early Development in Zebrafish (Danio Rerio). PeerJ 2019, 7, e7094. [Google Scholar] [CrossRef] [Green Version]
- Paganelli, A.; Gnazzo, V.; Acosta, H.; López, S.L.; Carrasco, A.E. Glyphosate-Based Herbicides Produce Teratogenic Effects on Vertebrates by Impairing Retinoic Acid Signaling. Chem. Res. Toxicol. 2010, 23, 1586–1595. [Google Scholar] [CrossRef]
- De Almeida, L.L.; Teixeira, Á.A.C.; Soares, A.F.; da Cunha, F.M.; da Silva Junior, V.A.; Vieira Filho, L.D.; Wanderley-Teixeira, V. Effects of Melatonin in Rats in the Initial Third Stage of Pregnancy Exposed to Sub-Lethal Doses of Herbicides. Acta Histochem. 2017, 119, 220–227. [Google Scholar] [CrossRef] [PubMed]
- Daruich, J.; Zirulnik, F.; Sofía Gimenez, M. Effect of the Herbicide Glyphosate on Enzymatic Activity in Pregnant Rats and Their Fetuses. Environ. Res. 2001, 85, 226–231. [Google Scholar] [CrossRef]
- Dallegrave, E.; Mantese, F.D.; Coelho, R.S.; Pereira, J.D.; Dalsenter, P.R.; Langeloh, A. The Teratogenic Potential of the Herbicide Glyphosate-Roundup® in Wistar Rats. Toxicol. Lett. 2003, 142, 45–52. [Google Scholar] [CrossRef]
- De Araujo, J.S.A.; Delgado, I.F.; Paumgartten, F.J.R. Glyphosate and Adverse Pregnancy Outcomes, a Systematic Review of Observational Studies. BMC Public Health 2016, 16, 472. [Google Scholar] [CrossRef] [Green Version]
- Maamar, M.B.; Beck, D.; Nilsson, E.E.; Kubsad, D.; Skinner, M.K. Epigenome-Wide Association Study for Glyphosate Induced Transgenerational Sperm DNA Methylation and Histone Retention Epigenetic Biomarkers for Disease. Epigenetics 2021, 16, 1150–1167. [Google Scholar] [CrossRef]
- Kubsad, D.; Nilsson, E.E.; King, S.E.; Sadler-Riggleman, I.; Beck, D.; Skinner, M.K. Assessment of Glyphosate Induced Epigenetic Transgenerational Inheritance of Pathologies and Sperm Epimutations: Generational Toxicology. Sci. Rep. 2019, 9, 6372. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lorenz, V.; Milesi, M.M.; Schimpf, M.G.; Luque, E.H.; Varayoud, J. Epigenetic Disruption of Estrogen Receptor Alpha Is Induced by a Glyphosate-Based Herbicide in the Preimplantation Uterus of Rats. Mol. Cell. Endocrinol. 2019, 480, 133–141. [Google Scholar] [CrossRef]
- Milesi, M.M.; Lorenz, V.; Pacini, G.; Repetti, M.R.; Demonte, L.D.; Varayoud, J.; Luque, E.H. Perinatal Exposure to a Glyphosate-Based Herbicide Impairs Female Reproductive Outcomes and Induces Second-Generation Adverse Effects in Wistar Rats. Arch. Toxicol. 2018, 92, 2629–2643. [Google Scholar] [CrossRef]
- Gomez, A.L.; Altamirano, G.A.; Leturia, J.; Bosquiazzo, V.L.; Muñoz-de-Toro, M.; Kass, L. Male Mammary Gland Development and Methylation Status of Estrogen Receptor Alpha in Wistar Rats Are Modified by the Developmental Exposure to a Glyphosate-Based Herbicide. Mol. Cell. Endocrinol. 2019, 481, 14–25. [Google Scholar] [CrossRef]
- Brake, D.G.; Evenson, D.P. A Generational Study of Glyphosate-Tolerant Soybeans on Mouse Fetal, Postnatal, Pubertal and Adult Testicular Development. Food Chem. Toxicol. 2004, 42, 29–36. [Google Scholar] [CrossRef]
- Woźniak, E.; Reszka, E.; Jabłońska, E.; Michałowicz, J.; Huras, B.; Bukowska, B. Glyphosate and AMPA Induce Alterations in Expression of Genes Involved in Chromatin Architecture in Human Peripheral Blood Mononuclear Cells (In Vitro). Int. J. Mol. Sci. 2021, 22, 2966. [Google Scholar] [CrossRef] [PubMed]
- Garry, V.F.; Harkins, M.E.; Erickson, L.L.; Long-Simpson, L.K.; Holland, S.E.; Burroughs, B.L. Birth Defects, Season of Conception, and Sex of Children Born to Pesticide Applicators Living in the Red River Valley of Minnesota, USA. Environ. Health Perspect. 2002, 110, 441–449. [Google Scholar] [CrossRef] [Green Version]
- Rappazzo, K.M.; Warren, J.L.; Davalos, A.D.; Meyer, R.E.; Sanders, A.P.; Brownstein, N.C.; Luben, T.J. Maternal Residential Exposure to Specific Agricultural Pesticide Active Ingredients and Birth Defects in a 2003–2005 North Carolina Birth Cohort. Birth Defects Res. 2019, 111, 312–323. [Google Scholar] [CrossRef] [PubMed]
- Savitz, D.A.; Arbuckle, T.; Kaczor, D.; Curtis, K.M. Male Pesticide Exposure and Pregnancy Outcome. Am. J. Epidemiol. 1997, 146, 1025–1036. [Google Scholar] [CrossRef] [Green Version]
- Mink, P.J.; Mandel, J.S.; Lundin, J.I.; Sceurman, B.K. Epidemiologic Studies of Glyphosate and Non-Cancer Health Outcomes: A Review. Regul. Toxicol. Pharmacol. 2011, 61, 172–184. [Google Scholar] [CrossRef] [PubMed]
- Paris, K.; Aris, A. Lien hypothétique entre l’endométriose et l’accumulation de xénobiotiques associés aux aliments génétiquement modifiés. Gynécologie Obs. Fertil. 2010, 38, 747–753. [Google Scholar] [CrossRef] [PubMed]
- Agarwal, A.; Aponte-Mellado, A.; Premkumar, B.J.; Shaman, A.; Gupta, S. The Effects of Oxidative Stress on Female Reproduction: A Review. Reprod. Biol. Endocrinol. 2012, 10, 49. [Google Scholar] [CrossRef] [Green Version]
- Kass, L.; Gomez, A.L.; Altamirano, G.A. Relationship between Agrochemical Compounds and Mammary Gland Development and Breast Cancer. Mol. Cell. Endocrinol. 2020, 508, 110789. [Google Scholar] [CrossRef]
- Pu, Y.; Yang, J.; Chang, L.; Qu, Y.; Wang, S.; Zhang, K.; Xiong, Z.; Zhang, J.; Tan, Y.; Wang, X.; et al. Maternal Glyphosate Exposure Causes Autism-like Behaviors in Offspring through Increased Expression of Soluble Epoxide Hydrolase. Proc. Natl. Acad. Sci. USA 2020, 117, 11753–11759. [Google Scholar] [CrossRef]
- Samsel, A.; Seneff, S. Glyphosate, Pathways to Modern Diseases II: Celiac Sprue and Gluten Intolerance. Interdiscip. Toxicol. 2013, 6, 159–184. [Google Scholar] [CrossRef] [Green Version]
- Zhang, J.; Zhao, C.; Shi, F.; Zhang, S.; Wang, S.; Feng, X. Melatonin Alleviates the Deterioration of Oocytes and Hormonal Disorders from Mice Subjected to Glyphosate. Mol. Cell. Endocrinol. 2021, 520, 111073. [Google Scholar] [CrossRef] [PubMed]
- Gasnier, C.; Benachour, N.; Clair, E.; Travert, C.; Langlois, F.; Laurant, C.; Decroix-Laporte, C.; Séralini, G.-E. Dig1 Protects against Cell Death Provoked by Glyphosate-Based Herbicides in Human Liver Cell Lines. J. Occup. Med. Toxicol. 2010, 5, 29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Çavuşoğlu, K.; Yapar, K.; Oruç, E.; Yalçın, E. Protective Effect of Ginkgo Biloba L. Leaf Extract Against Glyphosate Toxicity in Swiss Albino Mice. J. Med. Food 2011, 14, 1263–1272. [Google Scholar] [CrossRef] [PubMed]


| Hormone | Pesticide | Doses Eq. G | Sex | Species | Stages | Time Exposure | Compartment | Molecules | Effect | References |
|---|---|---|---|---|---|---|---|---|---|---|
| FSH | R | 10–40 mg/kg/bw/d | F | Pig | weaned | 36 days | Serum | Hormone | ↘ | [21] |
| R | 0.5% | F | ICR mice (Pregnant) | 10 weeks | GD1-GD19 | Pituitary | mRNA | ↗ | [22] | |
| R | 50 mg/kg/bw/d | M | Wistar rat | 60 days | GD18-PND5 | Pituitary | mRNA | ↗ | [28] | |
| G | 5 mg/kg * | M | 6 weeks | 52 days | Plasma & Pituitary | Hormone | NS | [25] | ||
| G | 10 mg/kg/bw | M | 2 months (adult) | 3 times a week during 5 weeks | Plasma | Hormone | ↗ | [24] | ||
| R | 400 or 2000 mg/kg/bw | M | Adult | 60 days | Pituitary | Hormone | ↘ | [23] | ||
| R | 1.85 g/L ** | M | C57Bl/6 mice | 50 days | GD4-PND30 | Plasma & Pituitary | Hormone | NS | [29] | |
| R | 3.6–248.4 mg/kg/bw | M | Albino rat | Adult | 12 weeks | Blood | Hormone | ↘ | [26] | |
| LH | R | 0.5% | F | ICR mice (Pregnant) | 10 weeks | GD1-GD19 | Pituitary | mRNA | ↗ | [22] |
| R | 50 mg/kg/bw/d | M | Wistar rat | 60 days | GD18-PND5 | Pituitary | mRNA | ↗ | [28] | |
| Hormone | ↗ | |||||||||
| G | 5 mg/kg | M | 6 weeks | 52 days | Plasma & Pituitary | Hormone | NS | [25] | ||
| G | 10 mg/kg + other pesticides | M | 2 months | 3 times a week during 5 weeks | Plasma | Hormone | ↗ | [24] | ||
| R | 400 or 2000 mg/kg | M | Adult | 60 days | Pituitary | Hormone | ↗ | [23] | ||
| R | 3.6–248.4 mg/kg | M | Albino rat | Adult | 12 weeks | Blood | Hormone | ↘ | [26] |
| Pesticides | Doses Eq. G | Time Exposure | Species | Stages | Compartment | Hormones | References |
|---|---|---|---|---|---|---|---|
| R | 10 mg/kg/bw | 36 days | Pig | weaned | plasma | Estrogen ↘; LHRH ↗; Testosterone ↗; Prolactin ↘ | [21] |
| R | 65 µg/L * | 15 days | Zebrafish | ovary | SF-1 (steroidogenic factor-1) ↗ | [ 39] | |
| R | 2 mg/kg/day | 15 days | Ovine | PND1—PND14 | granulosa | FSHR↘; GDF9↘ | [40] |
| R | 1.75 mg/kg/bw/day | GD6—PND120 | Sprague Dawley rats | pregnancy and weaning | Estrogen NS | [31] | |
| R | 5 g/L ** | GD1—GD19 | ICR mice (Pregnant) | pregnancy | serum | Pg ↘ | [22] |
| ovary | LHR ↘; 3β HSD ↘; Cyp17a1 ↗ | ||||||
| G | 5 g/L ** | ICR mice (Pregnant) | serum | Estrogen ↗; Pg ↘; FSHR ↘; Cyp11a1 ↗; 3β HSD ↘; Cyp19a1 ↗ | |||
| K360 | 126 mg/kg ** | 60 days | Wistar rat | ovary | Estrogen ↘ | [33] | |
| G | 2 mg/kg/bw 5 times a week | 20 weeks | C57BL6 mice | 6 weeks old | Estrogen NS; NS Progesterone | [32] |
| Pesticides | Doses Eq. G | Time Exposure | Species | Stages | Compartment | Hormones | References |
|---|---|---|---|---|---|---|---|
| R + FSH + T + IGF1 | 10 mg/L or 300 mg/L | 2 days | Bovine | granulosa | Estrogen ↘; Pg ↘ | [35] | |
| R + FSH + T+ IGF1 | 10 mg/L | Estrogen ↘; Pg ↘ | |||||
| R + FSH + T + IGF1 | 1 mg/L | Estrogen ↗; Pg ↗ | |||||
| R + FSH + T | 1 mg/L | Estrogen ↘ | |||||
| R | 10 µg/L | 72 h | Bovine | 8th & 12th days of estrus cycle | granulosa | Estrogen NS | [37] |
| G | 10 µg/L | Bovine | Estrogen ↗ | ||||
| G | 200 µg/L | 48 h | Pig | granulosa | Estrogen ↘; Pg ↗ | [30] | |
| G + FSH + IGF1 | 5 mg/L | 2 days | Bovine | granulosa | Estrogen ↘; Pg NS | [36] | |
| 10 mg/L or 300 mg/L | NS | ||||||
| G | 200 mg/L | 44 h | Pig | oocyte | Pg ↗ | [38] | |
| R | 100 mg/L | Pg ↘ |
| Pesticide | Doses Eq. G | Time Exposure | Species | Stages | Compartment | Effect | References |
|---|---|---|---|---|---|---|---|
| R | 50- 450 mg/kg/bw/d | GD21-PND21 | Wistar rat | puberty | ↘ | [45] | |
| R | 50 mg/kg/bw/d | GD18-PND5 | Wistar rat | adult | ↗ | [28] | |
| R | 1.85 g/L * | GD4-PND30 | C57 Bl/6 mice | ↗ | [29] | ||
| R | 5–250 mg/kg/bw/d | 30 days | Wistar rat | prepubertal | ↘ | [46] | |
| serum | [47] | ||||||
| R | 3.6–248.4 mg/kg/bw/d | 12 weeks | Albino rat | adult | blood | ↘ | [26] |
| 15 days | Drakes | adult | serum | [53] | |||
| 6 weeks | Rabbit | adult | [56] | ||||
| G | 5–125 mg/kg/bw/d | 10–20 days | Wistar rat | adult | serum | ↘ | [48] |
| 52 days | Wistar rat | adult | plasma | ↘ | [25] | ||
| 5 weeks | Wistar rat | adult | plasma | ↘ | [24] | ||
| 60 days | Wistar rat | adult | ↘ | [23] | |||
| 4 weeks | BALB/c mice | adult | serum | ↘ | [57] | ||
| G | 0.5–50 mg/kg/bw/d | GD10.5-PN20 | Swiss mice | PND5-8 months | ↘ | [54] |
| Pesticide | Doses Eq. G | Time Exposure | Species | Stages | Compartment | Effect | References |
|---|---|---|---|---|---|---|---|
| R | 360 µg/L | 24 h | Albino Sprague Dawley rats | adult | Rat testis | ↘ | [58] |
| G | 360 µg/L | ↘ | |||||
| 5 mg/L | 1–24 h | Mouse | TM3 mouse Leydig cells | ↘ | [59] | ||
| 16.9 mg/L | 24 h | Mouse | TM3 mouse Leydig cells | ↘ | [57] | ||
| 16.9 mg/L | Mouse | Primary mouse Leydig cells | ↘ |
| Experiment | Pesticide | Doses Eq. G | Time Exposure | Species | Compartment | Hormones | Effect | References |
|---|---|---|---|---|---|---|---|---|
| In vivo | G | 5 g/L * | 4 weeks | BALB/c mice | serum | StAR | ↘ | [57] |
| StAR mRNA | ↘ | |||||||
| G | 2.5 mg/L * | 2 weeks | Sprague Dawley rats | testis | StAR | NS | [51] | |
| StAR mRNA | ↘ | |||||||
| In vitro | G | 5 mg/L | 1–24 h | Mouse | TM3 mouse Leydig cells | StAR | ↘ | [59] |
| G | 16.9 mg/L | 24 h | TM3 mouse Leydig cells | StAR | ↘ | [57] | ||
| StAR mRNA | ↘ | |||||||
| R | 24 mg/L | 4 h | Mouse MA-10 Leydig Tumor cell line | StAR | ↘ | [60] | ||
| StAR mRNA | NS | |||||||
| In vivo | R | 2.25 g/L * | 8 days | Sprague Dawley rats | CYP19A1 | ↗ | [49] | |
| In vitro | G | 360 µg/L | 24 h | Albino Sprague Dawley rats | testis | CYP19A1 | ↘ | [58] |
| R | 360 µg/L | ↘ | ||||||
| R | 2.16 g/L | 18 h | Equine | testis | P450 aromatase | ↘ | [61] | |
| G | 6.48 g/L | ↘ | [62] |
| Experiment | Pesticides | Doses | Protective Molecules (PM) | Dose (PM) | Time Exposure | Sex | Species | Stages | Steroidogenesis | Oxidative Stress | Sperm | Other Effects | References |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| In vivo | Mixture (Zineb+G+Dimethoat) | 10 mg/kg/bw | α-Lipoic acid | 50 mg/kg/bw/d | 5 weeks | M | Wistar rat | 8 weeks | Restored T, LH, FSH, 3 and 17 βHSD level | [24] | |||
| G | 375 mg/kg/bw/d | Resveratrol | 20 mg/kg/bw/d | 8 weeks | M | Wistar Albino rat | 12 weeks | Restored MDA level | ↗ sperm motility ↘ abnormal sperm rate no effect on sperm concentration | ↗ plasma integrity, ↘ DNA damage, no effect on Sertoli cells | [65] | ||
| G | 250–500 mg/kg/bw/d | Melatonin | 15 mg/kg | 7 days | F | Kumming mice | 4 weeks | Normalized Cat, GPx activity, restored mitochondrial ATP, ↘ ROS levels | Enhance meiotic progression, normalize Bax/Bcl2, inhibition of apoptosis and autophagy | [112] | |||
| R | 500 mg/kg/bw | Melatonin | 10 mg/kg | GD1-GD7 | F | Wistar rat | 12 weeks | No effect on the morphology of blastocysts, ↗ weight of ovary and number of corpus lutea, ↗ viability of the embryo implantation | [92] | ||||
| R | 135 mg/kg/bw/d | Dig1 | 1.2 mg/kg/bw/d | 8 days | M | Sprague Dawley rat | adult | Restored plasma estradiol level | [64] | ||||
| R | 50 mg/kg/bw/d | Ginkgo Biloda (leaf extract) | 50 o 150 mg/kg/bw/d | 72 h | / | Swiss Albino mice | 12–14 weeks | Restored ROS level in kidney and liver | ↘ apoptosis | [113] | |||
| In vitro | R | 0.1–4.0 g/L | Vitamin C | 1 mM | 24–72 h | F | Caprine | Antral follicle | Restored MDA and ROS levels, normalized Cat, SOD and GST activity | [42] | |||
| R | 0.72–360 mg/L | Vitamin C + Trolox (Vit E) Nifedipine | 100 μM + 100 μM 10μM | 30 min | M | Wistar rat | Prepubertal testis | ↘ Ca2+ uptake | [66] | ||||
| G | 0.069–169 mg/L | Quercetin | 1 nM | 10 min | M | Human sperm | Normalized mitochondrial respiration | [71] | |||||
| G | 33.8–338.1 μg/L | Fulvestrant (ER antagonist) | 700 nM | 24 h | F | Human endometrial Ishikawa cells | Normalized endometrial cell migration and invasion, normalized level of E-Cadherin | [78] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Serra, L.; Estienne, A.; Vasseur, C.; Froment, P.; Dupont, J. Review: Mechanisms of Glyphosate and Glyphosate-Based Herbicides Action in Female and Male Fertility in Humans and Animal Models. Cells 2021, 10, 3079. https://doi.org/10.3390/cells10113079
Serra L, Estienne A, Vasseur C, Froment P, Dupont J. Review: Mechanisms of Glyphosate and Glyphosate-Based Herbicides Action in Female and Male Fertility in Humans and Animal Models. Cells. 2021; 10(11):3079. https://doi.org/10.3390/cells10113079
Chicago/Turabian StyleSerra, Loïse, Anthony Estienne, Claudine Vasseur, Pascal Froment, and Joëlle Dupont. 2021. "Review: Mechanisms of Glyphosate and Glyphosate-Based Herbicides Action in Female and Male Fertility in Humans and Animal Models" Cells 10, no. 11: 3079. https://doi.org/10.3390/cells10113079
APA StyleSerra, L., Estienne, A., Vasseur, C., Froment, P., & Dupont, J. (2021). Review: Mechanisms of Glyphosate and Glyphosate-Based Herbicides Action in Female and Male Fertility in Humans and Animal Models. Cells, 10(11), 3079. https://doi.org/10.3390/cells10113079







