Steeping of Biofortified Orange Maize Genotypes for Ogi Production Modifies Pasting Properties and Carotenoid Stability
Abstract
Chemical Compounds Studied in This Article:
1. Introduction
2. Materials and Methods
2.1. Chemicals and Standards
2.2. Biofortified Yellow and Orange Endosperm Maize Genotypes
2.3. Processing Method of Maize
2.4. Biofortified Maize Porridge Preparation
2.5. Assessment of Pasting Properties by Rapid Visco Analyser (RVA)
2.6. Carotenoid Analysis
2.7. Statistical Analysis of Data
3. Results
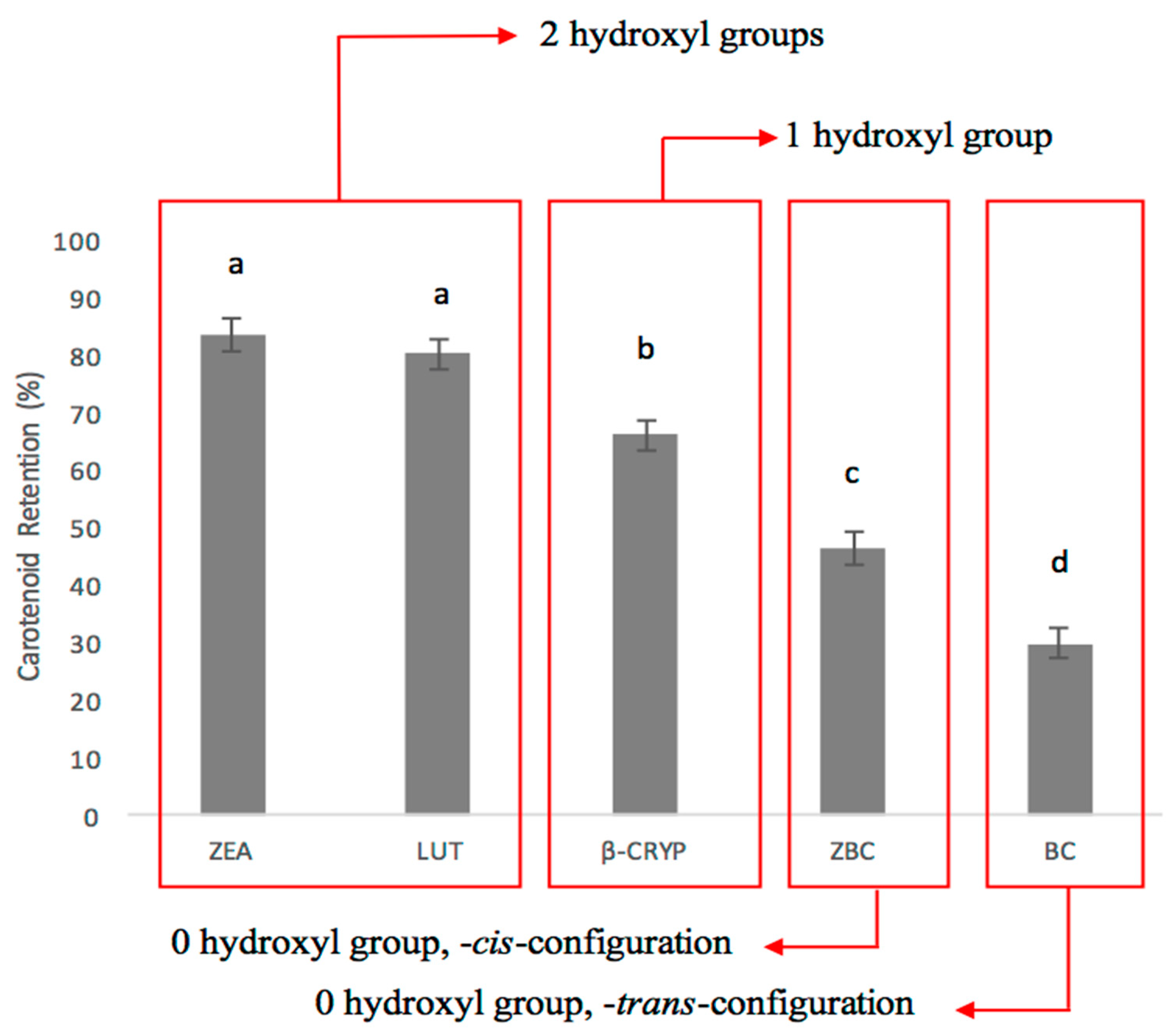
3.1. Carotenoid Profile on Selected Biofortified Maize Genotypes
3.2. Effect of Fermentation on Carotenoid Recovery in Biofortified Maize
3.3. Carotenoids Stability During Porridge Preparation (Wet Cooking)
3.4. Rheological Characterization of Porridges by RVA
4. Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| pVACs | provitamin A carotenoids |
| TCC | total carotenoid content |
| BC | all-trans-β-carotene |
| AC | α-carotene |
| BCRYP | β-cryptoxanthin |
| LUT | all-trans-lutein |
| ZEA | zeaxanthin |
| DW | dry weight basis |
| wb | wet basis |
| RH | relative humidity |
| SEM | standard error of the mean |
| Orange ISO | orange corn in isolated field where it open pollinates |
References
- World Health Organization. Global Prevalence of Vitamin a Deficiency in Populations at Risk 1995–2005: WHO Global Database on Vitamin a Deficiency; World Health Organization: Geneva, Switzerland, 2009. [Google Scholar]
- Pixley, K.; Rojas, N.P.; Babu, R.; Mutale, R.; Surles, R.; Simpungwe, E. Biofortification of maize with provitamin a carotenoids. In Carotenoids and Human Health; Humana Press: Totowa, NJ, USA, 2012; pp. 271–292. [Google Scholar]
- Menkir, A.; Rocheford, T.; Maziya-Dixon, B.; Tanumihardjo, S. Exploiting natural variation in exotic germplasm for increasing provitamin-A carotenoids in tropical maize. Euphytica 2015, 205, 203–217. [Google Scholar] [CrossRef]
- Bouis, H.E.; Saltzman, A. Improving nutrition through biofortification—A review of evidence from HarvestPlus, 2003 through 2016. Glob. Food Secur. 2017, 12, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Menkir, A.; Palacios-Rojas, N.; Alamu, O.; Dias Paes, M.C.; Dhliwayo, T.; Maziya-Dixon, B.; Mengesha, W.; Ndhlela, T.; Oliveira Guimarães, P.E.; Pixley, K.; et al. Vitamin A-Biofortified Maize: Exploiting Native Genetic Variation for Nutrient Enrichment; Science Brief: Biofortification No. 2 (February 2018); CIMMYT, IITA, EMBRAPA, HarvestPlus, and Crop Trust: Bonn, Germany, 2018. [Google Scholar]
- Ortiz, D.; Rocheford, T.; Ferruzzi, M.G. Influence of temperature and humidity on the stability of carotenoids in biofortified maize (Zea mays L.) genotypes during controlled postharvest storage. J. Agric. Food Chem. 2016, 64, 2727–2736. [Google Scholar] [CrossRef] [PubMed]
- Taleon, V.; Mugode, L.; Cabrera-Soto, L.; Palacios-Rojas, N. Carotenoid retention in biofortified maize using different post-harvest storage and packaging methods. Food Chem. 2017, 232, 1–36. [Google Scholar] [CrossRef]
- Palmer, A.C.; Craft, N.E.; Schulze, K.J.; Barffour, M.; Chileshe, J.; Siamusantu, W.; West, K.P. Impact of biofortified maize consumption on serum carotenoid concentrations in Zambian children. Eur. J. Clin. Nutr. 2018, 72, 301–303. [Google Scholar] [CrossRef]
- Palmer, A.C.; Healy, K.; Barffour, M.A.; Siamusantu, W.; Chileshe, J.; Schulze, K.J.; West, K.P., Jr.; Labrique, A.B. Provitamin a carotenoid–biofortified maize consumption increases pupillary responsiveness among zambian children in a randomized controlled trial. J. Nutr. 2016, 146, 2551–2558. [Google Scholar] [CrossRef]
- Palmer, A.C.; Chileshe, J.; Hall, A.G.; Barffour, M.A.; Molobeka, N.; West, K.P., Jr.; Haskell, M.J. Short-term daily consumption of provitamin a carotenoid–biofortified maize has limited impact on breast milk retinol concentrations in zambian women enrolled in a randomized controlled feeding trial. J. Nutr. 2016, 146, 1783–1792. [Google Scholar] [CrossRef][Green Version]
- Gannon, B.; Kaliwile, C.; Arscott, S.A.; Schmaelzle, S.; Chileshe, J.; Kalungwana, N.; Mosonda, M.; Pixley, K.; Masi, C.; Tanumihardjo, S.A. Biofortified orange maize is as efficacious as a vitamin A supplement in Zambian children even in the presence of high liver reserves of vitamin A: A community-based, randomized placebo-controlled trial. Am. J. Clin. Nutr. 2014, 100, 1541–1550. [Google Scholar] [CrossRef]
- Rodriguez-Amaya, D.B. Carotenoids and Food Preparation: The Retention of Provitamin a Carotenoids in Prepared, Processed and Stored Foods; John Snow Incorporated/OMNI Project: Arlington, VA, USA, 1997. [Google Scholar]
- Franz, C.M.A.P.; Huch, M.; Mathara, J.M.; Abriouel, H.; Benomar, N.; Reid, G.; Galvez, A.; Holzapfel, W.H. African fermented foods and probiotics. Int. J. Food Microbiol. 2014, 190, 84–96. [Google Scholar] [CrossRef]
- Gabaza, M.; Muchuweti, M.; Vandamme, P.; Raes, K. Can fermentation be used as a sustainable strategy to reduce iron and zinc binders in traditional African fermented cereal porridges or gruels? Food Rev. Int. 2017, 33, 561–586. [Google Scholar] [CrossRef]
- Svanberg, U.; Lorri, W. Fermentation and nutrient availability. Food Control 1997, 8, 319–327. [Google Scholar] [CrossRef]
- Afify, A.E.-M.M.R.; El-Beltagi, H.S.; Abd El-Salam, S.M.; Omran, A.A. Bioavailability of iron, zinc, phytate and phytase activity during soaking and germination of white sorghum varieties. PLoS ONE 2011, 6, e25512. [Google Scholar] [CrossRef] [PubMed]
- Alka, S.; Neelam, Y.; Shruti, S. Effect of fermentation on physicochemical properties & in vitro starch and protein digestibility of selected cereals. Int. J. Agric. Food Sci. 2012, 2, 66–70. [Google Scholar]
- Cui, L.; Li, D.-J.; Liu, C.-Q. Effect of fermentation on the nutritive value of maize. Int. J. Food Sci. Technol. 2012, 47, 755–760. [Google Scholar] [CrossRef]
- Liu, Y.Q.; Davis, C.R.; Schmaelzle, S.T.; Rocheford, T.; Cook, M.E.; Tanumihardjo, S.A. β-Cryptoxanthin biofortified maize (Zea mays L.) increases β-cryptoxanthin concentration and enhances the color of chicken egg yolk. Poult. Sci. 2012, 91, 432–438. [Google Scholar] [CrossRef]
- Teucher, B.; Olivares, M.; Cori, H. Enhancers of iron absorption: Ascorbic acid and other organic acids. Int. J. Vitam. Nutr. Res. 2004, 74, 403–419. [Google Scholar] [CrossRef]
- Hotz, C.; Gibson, R.S. Traditional food-processing and preparation practices to enhance the bioavailability of micronutrients in plant-based diets. J. Nutr. 2007, 137, 1097–1100. [Google Scholar] [CrossRef]
- Kumar, V.; Sinha, A.K.; Makkar, H.P.S.; Becker, K. Dietary roles of phytate and phytase in human nutrition: A review. Food Chem. 2010, 120, 945–959. [Google Scholar] [CrossRef]
- Thakkar, S.K.; Huo, T.; Maziya-Dixon, B.; Failla, M.L. Impact of style of processing on retention and bioaccessibility of β-carotene in cassava (manihot esculanta, crantz). J. Agric. Food Chem. 2009, 57, 1344–1348. [Google Scholar] [CrossRef]
- Failla, M.L.; Chitchumroonchokchai, C.; Siritunga, D.; de Moura, F.F.; Fregene, M.; Manary, M.J.; Sayre, R.T. Retention during processing and bioaccessibility of β-carotene in high β-carotene transgenic cassava root. J. Agric. Food Chem. 2012, 60, 3861–3866. [Google Scholar] [CrossRef]
- Aragón, I.J.; Ceballos, H.; Dufour, D.; Ferruzzi, M.G. Pro-vitamin a carotenoids stability and bioaccessibility from elite selection of biofortified cassava roots (Manihot esculenta, Crantz) processed to traditional flours and porridges. Food Funct. 2018, 9, 4822–4835. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Tayie, F.A.K.; Young, M.F.; Rocheford, T.; White, W.S. Retention of provitamin a carotenoids in high β-carotene maize (Zea mays L.) during traditional african household processing. J. Agric. Food Chem. 2007, 55, 10744–10750. [Google Scholar] [CrossRef] [PubMed]
- Reboul, E.; Richelle, M.; Perrot, E.; Desmoulins-Malezet, C.; Pirisi, V.; Borel, P. Bioaccessibility of carotenoids and vitamin e from their main dietary sources. J. Agric. Food Chem. 2006, 54, 8749–8755. [Google Scholar] [CrossRef] [PubMed]
- Osungbaro, T.O. Effect of fermentation period on amylose content and textural characteristics of “Ogi” (a fermented maize porridge). J. Ferment. Bioeng. 1990, 70, 22–25. [Google Scholar] [CrossRef]
- Li, M.; Dhital, S.; Wei, Y. Multilevel structure of wheat starch and its relationship to noodle eating qualities. Compr. Rev. Food Sci. Food Saf. 2017, 16, 1042–1055. [Google Scholar] [CrossRef]
- Cozzolino, D. The use of the rapid visco analyser (RVA) in breeding and selection of cereals. J. Cereal Sci. 2016, 70, 282–290. [Google Scholar] [CrossRef]
- Aremu, C. Nutrient composition of corn OGI prepared by a slightly modified traditional technique. Food Chem. 1993, 46, 231–233. [Google Scholar] [CrossRef]
- Lipkie, T.E.; de Moura, F.F.; Zhao, Z.-Y.; Albertsen, M.C.; Che, P.; Glassman, K.; Ferruzzi, M.G. Bioaccessibility of carotenoids from transgenic provitamin a biofortified sorghum. J. Agric. Food Chem. 2013, 61, 5764–5771. [Google Scholar] [CrossRef]
- Shafie, B.; Cheng, S.C.; Lee, H.H.; Yiu, P.H. Characterization and classification of whole-grain rice based on rapid visco analyzer (RVA) pasting profile. Int. Food Res. J. 2016, 23, 2138–2143. [Google Scholar]
- Ortiz, D.; Ferruzzi, M.G. Identification and quantification of carotenoids and tocochromanols in sorghum grain by high-performance liquid chromatography. In Sorghum: Methods and Protocols; Zhao, Z.-Y., Dahlberg, J., Eds.; Methods and Protocols; Springer: New York, NY, USA, 2019; Volume 1931, pp. 141–151. [Google Scholar]
- Carotenoids; Britton, G., Liaaen-Jensen, S., Pfander, H., Eds.; Birkhäuser Basel: Basel, Switzerland, 2004. [Google Scholar]
- Oguntoyinbo, F.A.; Narbad, A. Molecular characterization of lactic acid bacteria and in situ amylase expression during traditional fermentation of cereal foods. Food Microbiol. 2012, 31, 254–262. [Google Scholar] [CrossRef]
- Adeyemi, I.A.; Beckley, O. Effect of period of maize fermentation and souring on chemical properties and amylograph pasting viscosity of ogi. J. Cereal Sci. 1986, 4, 353–360. [Google Scholar] [CrossRef]
- Akinrele, I.A. Fermentation studies on maize during the preparation of a traditional african starch-cake food. J. Sci. Food Agric. 1970, 21, 619–625. [Google Scholar] [CrossRef]
- Teniola, O.D.; Odunfa, S.A. Microbial assessment and quality evaluation of ogi during spoilage. World J. Microbiol. Biotechnol. 2002, 18, 731–737. [Google Scholar] [CrossRef]
- Osungbaro, T.O. Effect of differences in variety and dry milling of maize on textural characteristics of Ogi (fermented maize porridge) and Agidi (fermented maize meal). J. Sci. Food Agric. 1990, 52, 1–11. [Google Scholar] [CrossRef]
- Okoli, E.C.; Adeyemi, I.A. Manufacturing of Ogi from malted (germinated) corn (Zea mays L.): Evaluation of chemical, pasting and sensory properties. J. Food Sci. 1989, 54, 971–973. [Google Scholar] [CrossRef]
- Nago, C.M.; Tétégan, E.; Matencio, F.; Mestres, C. Effects of maize type and fermentation conditions on the quality of beninese traditional Ogi, a fermented maize slurry. J. Cereal Sci. 1998, 28, 215–222. [Google Scholar] [CrossRef]
- Omemu, A.M.; Oyewole, O.B.; Bankole, M.O. Significance of yeasts in the fermentation of maize for ogi production. Food Microbiol. 2007, 24, 571–576. [Google Scholar] [CrossRef]
- Beugre, G.A.M.; Yapo, B.M.; Blei, S.H.; Gnakri, D. Effect of fermentation time on the physico-chemical properties of maize flour. Int. J. Res. Stud. Biosci. 2014, 2, 30–38. [Google Scholar]
- Odunfa, S.A.; Adeyele, S. Microbiological changes during the traditional production of ogi-baba, a west African fermented sorghum gruel. J. Cereal Sci. 1985, 3, 173–180. [Google Scholar] [CrossRef]
- Assohoun-Djeni, N.M.C.; Djeni, N.T.; Messaoudi, S.; Lhomme, E.; Koussemon-Camara, M.; Ouassa, T.; Chobert, J.M.; Onno, B.; Dousset, X. Biodiversity, dynamics and antimicrobial activity of lactic acid bacteria involved in the fermentation of maize flour for doklu production in Côte d’Ivoire. Food Control 2016, 62, 397–404. [Google Scholar] [CrossRef]
- Okeke, C.A.; Ezekiel, C.N.; Nwangburuka, C.C.; Sulyok, M.; Ezeamagu, C.O.; Adeleke, R.A.; Dike, S.K.; Krska, R. Bacterial diversity and mycotoxin reduction during maize fermentation (steeping) for Ogi production. Front. Microbiol. 2015, 6, 1402. [Google Scholar] [CrossRef] [PubMed]
- Boon, C.S.; McClements, D.J.; Weiss, J.; Decker, E.A. Factors influencing the chemical stability of carotenoids in foods. Crit. Rev. Food Sci. Nutr. 2010, 50, 515–532. [Google Scholar] [CrossRef] [PubMed]
- Bechoff, A.; Tomlins, K.I.; Chijioke, U.; Ilona, P.; Westby, A.; Boy, E. Physical losses could partially explain modest carotenoid retention in dried food products from biofortified cassava. PLoS ONE 2018, 13, e0194402. [Google Scholar] [CrossRef] [PubMed]
- Mahajan, S.; Chauhan, B.M. A research note effect of natural fermentation on the extractability of minerals from pearl millet flour. J. Food Sci. 1988, 53, 1576–1577. [Google Scholar] [CrossRef]
- Lopez, H.W.; Duclos, V.; Coudray, C.; Krespine, V.; Feillet-Coudray, C.; Messager, A.; Demigné, C.; Rémésy, C. Making bread with sourdough improves mineral bioavailability from reconstituted whole wheat flour in rats. Nutrition 2003, 19, 524–530. [Google Scholar] [CrossRef]
- Kean, E.G.; Hamaker, B.R.; Ferruzzi, M.G. Carotenoid bioaccessibility from whole grain and degermed maize meal products. J. Agric. Food Chem. 2008, 56, 9918–9926. [Google Scholar] [CrossRef]
- Mellado-Ortega, E.; Hornero-Méndez, D. Lutein esterification in wheat flour increases the carotenoid retention and is induced by storage temperatures. Foods 2017, 6, 111. [Google Scholar] [CrossRef]
- Miller, N.J.; Sampson, J.; Candeias, L.P.; Bramley, P.M.; Rice-Evans, C.A. Antioxidant activities of carotenes and xanthophylls. FEBS Lett. 1996, 384, 240–242. [Google Scholar] [CrossRef]
- Xiao, Y.-D.; Huang, W.-Y.; Li, D.-J.; Song, J.-F.; Liu, C.-Q.; Wei, Q.-Y.; Zhang, M.; Yang, Q.-M. Thermal degradation kinetics of all-trans and cis-carotenoids in a light-induced model system. Food Chem. 2018, 239, 360–368. [Google Scholar] [CrossRef]
- Böhm, V.; Puspitasari-Nienaber, N.L.; Ferruzzi, M.G.; Schwartz, S.J. Trolox equivalent antioxidant capacity of different geometrical isomers of α-carotene, β-carotene, lycopene, and zeaxanthin. J. Agric. Food Chem. 2002, 50, 221–226. [Google Scholar] [CrossRef]
- Liao, L.; Wu, W. Fermentation effect on the properties of sweet potato starch and its noodle’s quality by lactobacillus plantarum. J. Food Process Eng. 2016, 40, e12460. [Google Scholar] [CrossRef]
- Sowa, M.; Yu, J.; Palacios-Rojas, N.; Goltz, S.R.; Howe, J.A.; Davis, C.R.; Rocheford, T.; Tanumihardjo, S.A. Retention of carotenoids in biofortified maize flour and β-cryptoxanthin-enhanced eggs after household cooking. ACS Omega 2017, 2, 7320–7328. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, D.; Ponrajan, A.; Bonnet, J.P.; Rocheford, T.; Ferruzzi, M.G. Carotenoid stability during dry milling, storage, and extrusion processing of biofortified maize genotypes. J. Agric. Food Chem. 2018, 66, 4683–4691. [Google Scholar] [CrossRef] [PubMed]
- Gayral, M.; Bakan, B.; Dalgalarrondo, M.; Elmorjani, K.; Delluc, C.; Brunet, S.; Linossier, L.; Morel, M.-H.; Marion, D. Lipid partitioning in maize (Zea mays L.) endosperm highlights relationships among starch lipids, amylose, and vitreousness. J. Agric. Food Chem. 2015, 63, 3551–3558. [Google Scholar] [CrossRef]
- Gayral, M.; Gaillard, C.; Bakan, B.; Dalgalarrondo, M.; Elmorjani, K.; Delluc, C.; Brunet, S.; Linossier, L.; Morel, M.-H.; Marion, D. Transition from vitreous to floury endosperm in maize (Zea mays L.) kernels is related to protein and starch gradients. J. Cereal Sci. 2016, 68, 148–154. [Google Scholar] [CrossRef]
- Teixeira, C.S.; da Rocha Neves, G.A.; Caliari, M.R.; Júnior, M.S.S. Waxy maize starch modified by sun-drying after spontaneous or backslapping fermentation. Int. J. Biol. Macromol. 2019, 135, 553–559. [Google Scholar] [CrossRef]
- Díaz, A.; Dini, C.; Viña, S.Z.; García, M.A. Technological properties of sour cassava starches—Effect of fermentation and drying processes. LWT Food Sci. Technol. 2018, 93, 116–123. [Google Scholar] [CrossRef]
- Zhao, T.; Li, X.; Zhu, R.; Ma, Z.; Liu, L.; Wang, X.; Hu, X. Effect of natural fermentation on the structure and physicochemical properties of wheat starch. Carbohydr. Polym. 2019, 218, 163–169. [Google Scholar] [CrossRef]
- Karim, A.A.; Norziah, M.H.; Seow, C.C. Methods for the study of starch retrogradation. Food Chem. 2000, 71, 9–36. [Google Scholar] [CrossRef]
- Saulnier, L.; Marot, C.; Chanliaud, E.; Thibault, J.-F. Cell wall polysaccharide interactions in maize bran. Carbohydr. Polym. 1995, 26, 279–287. [Google Scholar] [CrossRef]
- Wolf, M.J.; McMasters, M.M.; Cannon, J.A.; Rosewell, E.C.; Rist, C.E. Preparation and some properties of hemicelluloses from corn hulls. Cereal Chem. 1953, 30, 451–470. [Google Scholar]
- Martínez-Bustos, F.; Martínez-Flores, H.E.; Sanmartín-Martínez, E.; Sánchez-Sinencio, F.; Chang, Y.K.; Barrera-Arellano, D.; Rios, E. Effect of the components of maize on the quality of masa and tortillas during the traditional nixtamalisation process. J. Sci. Food Agric. 2001, 81, 1455–1462. [Google Scholar] [CrossRef]
- Proulx, A.K.; Reddy, M.B. Fermentation and lactic acid addition enhance iron bioavailability of maize. J. Agric. Food Chem. 2007, 55, 2749–2754. [Google Scholar] [CrossRef] [PubMed]
- Simwaka, J.E.; Huiming, Z.; Masamba, K.G. Amino acid profile, mineral, pasting, thermal and protein solubility characteristics of sorghum-finger millet based complementary food as affected by fermentation. J. Acad. Ind. Res. 2015, 3, 504–510. [Google Scholar]
- Fairweather-Tait, S.; Hurrell, R.F. Bioavailability of minerals and trace elements. Nutr. Res. Rev. 1996, 9, 295–324. [Google Scholar] [CrossRef]
| Genotype | Pedigree |
|---|---|
| 1 | C17 × DE3 |
| 2 | Hi27 × CML328 |
| 3 | 2013 Orange ISO |
| 4 | 2015 Orange ISO |
| 5 | [KUI carotenoid syn-FS17-3-1-B-B-B-B-B-B-B-B] × [(MAS[206/312]-23-2-1-1-B-B-B/[BETASYN]BC1-6-5-1xFloridaASYN#-B)-B-1-3-B-B-B] |
| Fermentation (hours) | Genotype 1,2 | ||||
|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | |
| all-trans-lutein | |||||
| 0 | 17.1 ± 0.3a | 8.1 ± 0.2a | 8.9 ± 0.2a | 7.5 ± 0.1a | 7.8 ± 0.5a |
| 24 | 16.3 ± 1.2a | 7.8 ± 0.7a | 8.6 ± 0.3a | 6.4 ± 0.3b | 7.1 ± 0.9a |
| 72 | 17.2 ± 0.8a | 7.1 ± 0.4a | 7.8 ± 0.4b | 5.8 ± 0.4b | 8.9 ± 1.0a |
| 120 | 6.6 ± 0.2b | 2.7 ± 0.2b | 5.2 ± 0.9c | 2.2 ± 0.1c | 3.3 ± 0.3b |
| all-trans-zeaxanthin | |||||
| 0 | 3.7 ± 0.2a | 34 ± 0.6a | 43.3 ± 0.7a | 33.5 ± 0.4a | 29.2 ± 0.9a |
| 24 | 3.7 ± 0.2a | 32.7 ± 3.4b | 45.5 ± 1.6a | 30.0 ± 0.9a | 20.3 ± 3.0b |
| 72 | 3.9 ± 0.2a | 31.3 ± 2.0b | 39.1 ± 2.3b | 26.5 ± 2.6b | 24.8 ± 2.7b |
| 120 | 1.5 ± 0b | 11.5 ± 0.6c | 20.6 ± 5.1c | 9.9 ± 0.1c | 9.0 ± 0.9c |
| β-cryptoxanthin | |||||
| 0 | 0.8 ± 0.1a | 5.3 ± 0.3b | 3.0 ± 0.1a | 2.2 ± 0.2a | 3.6 ± 0.3a |
| 24 | 0.8 ± 1.1a | 7.7 ± 0.5a | 4.0 ± 0.3a | 2.3 ± 0.1a | 3.5 ± 0.5a |
| 72 | 0.8 ± 0.0a | 6.4 ± 0.9b | 3.1 ± 0.2a | 2.1 ± 0.1a | 4.0 ± 0.4a |
| 120 | 0.4 ± 0.1a | 2.6 ± 0.1c | 1.6 ± 0.3b | 0.8 ± 0.0b | 1.5 ± 0.1b |
| all-trans-β-carotene | |||||
| 0 | 6.8 ± 0.3b | 4 ± 0.2a | 2.8 ± 0.1a | 2.1 ± 0.2a | 2.6 ± 0.2a |
| 24 | 7.1 ± 0.5ab | 5.1 ± 0.4a | 2.9 ± 0.3a | 1.9 ± 0.1a | 2.2 ± 0.4a |
| 72 | 8.2 ± 0.3a | 4.8 ± 0.6a | 2.9 ± 0.2a | 1.8 ± 0.2a | 2.7 ± 0.3a |
| 120 | 3.2 ± 0.2c | 1.9 ± 0.1b | 1.5 ± 0.3b | 0.7 ± 0.0b | 1.0 ± 0.1b |
| total carotenoid content | |||||
| 0 | 32.7 ± 0.9a | 55.9 ± 1.5b | 61.0 ± 1.3ab | 47.5 ± 0.9a | 46.1 ± 2.2a |
| 24 | 31.7 ± 2.0a | 58.9 ± 3.3a | 64.1 ± 2.7a | 42.6 ± 1.4b | 35.3 ± 4.8a |
| 72 | 34.4 ± 1.3a | 54.5 ± 3.7b | 55.9 ± 3.1b | 38.2 ± 3.3b | 43.0 ± 4.7a |
| 120 | 13.6 ± 0.4b | 20.6 ± 1c | 30.4 ± 6.9c | 14.5 ± 0.1c | 15.8 ± 1.5b |
| Xanthophylls | |||||
| 0 | 21.6 ± 0.5a | 47.4 ± 1a | 55.2 ± 1.0ab | 43.2 ± 0.6a | 40.6 ± 2.2a |
| 24 | 20.8 ± 1.4a | 48.2 ± 2.5a | 58.1 ± 2.2a | 38.6 ± 1.2b | 30.9 ± 4.9b |
| 72 | 21.9 ± 1.0a | 44.9 ± 3.2b | 50.1 ± 2.9b | 34.4 ± 3.0b | 37.7 ± 4.1b |
| 120 | 8.6 ± 0.2b | 16.8 ± 0.9c | 27.4 ± 6.3c | 13.0 ± 0.1c | 13.7 ± 1.4c |
| provitamin A carotenoids3 | |||||
| 0 | 9.3 ± 0.4a | 8.9 ± 0.5a | 5.9 ± 0.3a | 4.3 ± 0.3a | 5.8 ± 0.6a |
| 24 | 9.4 ± 0.5a | 11.8 ± 0.8a | 6.4 ± 0.5a | 4.0 ± 0.2a | 5.1 ± 0.7a |
| 72 | 10.8 ± 0.3a | 10.4 ± 1.3a | 5.9 ± 0.3a | 3.8 ± 0.3a | 6.0 ± 0.7a |
| 120 | 4.3 ± 0.2b | 4.1 ± 0.1b | 3.0 ± 0.6b | 1.5 ± 0.0b | 2.2 ± 0.2b |
| sum of cis-β-carotene | |||||
| 0 | 4.3 ± 0.2a | 4.5 ± 0.3a | 3.0 ± 0.3a | 2.2 ± 0.1a | 2.9 ± 0.4a |
| 24 | 3.9 ± 0.2a | 5.5 ± 0.4a | 3.0 ± 0.4a | 2.1 ± 0.1a | 2.2 ± 0.3a |
| 72 | 4.3 ± 0.1a | 4.8 ± 0.6a | 2.9 ± 0.1a | 1.9 ± 0.1a | 2.5 ± 0.3a |
| 120 | 1.9 ± 0.1b | 1.9 ± 0.1b | 1.5 ± 0.3b | 0.8 ± 0.0b | 1.1 ± 0.1b |
| Fermentation (hours) | Genotype 1,2 | ||||
|---|---|---|---|---|---|
| 1 | 2 | 3 | 4 | 5 | |
| all-trans-lutein | |||||
| 0 | 10.9 ± 0.4a | 8.0 ± 0.2a | 9.4 ± 0.2a | 6.4 ± 0.1a | 8.2 ± 0.4a |
| 24 | 10.5 ± 1.2a | 6.1 ± 0.5b | 4.8 ± 0.6b | 4.1 ± 0.3b | 6.7 ± 0.8a |
| 72 | 12.2 ± 1.6a | 6.3 ± 0.2b | 5.6 ± 0.1b | 5.1 ± 0.3b | 8.1 ± 0.2a |
| 120 | 10.0 ± 0.3a | 6.6 ± 0.1b | 5.6 ± 0.2b | 4.4 ± 0.2b | 8.0 ± 0.6a |
| all-trans-zeaxanthin | |||||
| 0 | 2.6 ± 0.0a | 29.5 ± 0.9a | 34.5 ± 0.9a | 19.6 ± 0.2ab | 19.7 ± 0.8a |
| 24 | 2.4 ± 0.3a | 29.8 ± 2.4a | 32.6 ± 0.6ab | 25.4 ± 2.4a | 22.4 ± 2.0a |
| 72 | 2.9 ± 0.4a | 27.3 ± 1a | 31.1 ± 1.8ab | 22.5 ± 0.8ab | 22.8 ± 1.0a |
| 120 | 2.3 ± 0.1a | 27.4 ± 0.1a | 28.4 ± 0.5b | 18.6 ± 0.2b | 21.1 ± 1.3a |
| β-cryptoxanthin | |||||
| 0 | 0.9 ± 0.0a | 1.6 ± 0.1a | 2.3 ± 0.1a | 1.4 ± 0.1a | 1.6 ± 0.1a |
| 24 | 0.7 ± 0.1a | 5.1 ± 1.5a | 0.7 ± 0.0b | 0.9 ± 0.3a | 1.5 ± 0.9a |
| 72 | 0.9 ± 0.1a | 5.5 ± 0.2a | 0.9 ± 0.1b | 1.3 ± 0.4a | 2.9 ± 0.7a |
| 120 | 0.7 ± 0.0a | 4.4 ± 1.1a | 0.8 ± 0.0b | 1.1 ± 0.3a | 2.8 ± 0.6a |
| all-trans-β-carotene | |||||
| 0 | 3.5 ± 0.1a | 4.3 ± 0.2a | 2.0 ± 0.1a | 1.2 ± 0.0a | 1.5 ± 0.0a |
| 24 | 4.9 ± 0.5a | 3.0 ± 0.3b | 1.3 ± 0.1b | 1.0 ± 0.0b | 1.6 ± 0.2a |
| 72 | 4.4 ± 0.9a | 2.9 ± 0.1b | 1.7 ± 0.1b | 1.2 ± 0.1a | 1.8 ± 0.0a |
| 120 | 4.5 ± 0.1a | 3.1 ± 0.2b | 1.5 ± 0.1b | 1.0 ± 0.0ab | 1.7 ± 0.0a |
| total carotenoid content | |||||
| 0 | 20.8 ± 0.6a | 47.6 ± 1.4a | 50.8 ± 1.0a | 30.3 ± 0.2ab | 33.2 ± 1.3a |
| 24 | 21.1 ± 2.3a | 46.9 ± 4.7a | 41.5 ± 1.1b | 32.5 ± 2.2a | 33.6 ± 3.8a |
| 72 | 23.6 ± 2.6a | 45.6 ± 1.6a | 41.2 ± 1.9b | 31.6 ± 1.5ab | 37.2 ± 1.6a |
| 120 | 20.4 ± 0.6a | 45.1 ± 1.3a | 38.2 ± 0.7b | 26.4 ± 0.6b | 35.4 ± 2.5a |
| Xanthophylls | |||||
| 0 | 14.4 ± 0.4a | 39.1 ± 1.1a | 46.2 ± 0.9a | 27.4 ± 0.0ab | 29.5 ± 1.2a |
| 24 | 13.6 ± 1.5a | 41.0 ± 4.3a | 38.2 ± 1.0b | 30.4 ± 2.1a | 30.5 ± 3.4a |
| 72 | 16.0 ± 2.1a | 39.1 ± 1.3a | 37.5 ± 1.7b | 29.0 ± 1.4ab | 33.8 ± 1.6a |
| 120 | 13.0 ± 0.5a | 38.3 ± 1.2a | 34.9 ± 0.8b | 24.1 ± 0.6b | 31.8 ± 2.5a |
| provitamin A carotenoids3 | |||||
| 0 | 5.4 ± 0.1a | 7.2 ± 0.3a | 4.4 ± 0.1a | 2.7 ± 0.0a | 3.4 ± 0.1a |
| 24 | 6.6 ± 0.7a | 7.0 ± 1.1a | 2.7 ± 0.3b | 2.0 ± 0.2a | 3.1 ± 0.7a |
| 72 | 6.5 ± 0.9a | 7.5 ± 0.3a | 3.1 ± 0.2b | 2.6 ± 0.3a | 4.1 ± 0.4a |
| 120 | 6.3 ± 0.2a | 7.1 ± 0.6a | 2.8 ± 0.1b | 2.2 ± 0.2a | 4.0 ± 0.3a |
| sum of cis-β-carotene | |||||
| 0 | 2.9 ± 0.1a | 4.1 ± 0.1a | 2.7 ± 0.2a | 1.7 ± 0.2a | 2.3 ± 0.1a |
| 24 | 2.6 ± 0.3a | 3.0 ± 0.3b | 2.1 ± 0.5a | 1.2 ± 0.1a | 1.6 ± 0.3a |
| 72 | 3.2 ± 0.6a | 3.6 ± 0.2a | 2.0 ± 0.1a | 1.5 ± 0.1a | 1.7 ± 0.1a |
| 120 | 2.9 ± 0.1a | 3.6 ± 0.1a | 1.8 ± 0.0a | 1.3 ± 0.0a | 1.9 ± 0.0a |
| Genotype1,2 | Fermentation (hours) | Peak Viscosity (cP) | Pasting Temperature (°C) | Hot Paste Viscosity (cP) | Cool Paste Viscosity (cP) | Breakdown (cP) | Setback (cP) |
|---|---|---|---|---|---|---|---|
| 1 | 0 | 81 ± 3.6c | 74 ± 2.3a | 77 ± 2.9c | 175 ± 5.8c | 4 ± 1.0c | 94 ± 5.3ab |
| 24 | 211 ± 19.1b | 76 ± 0.2a | 187 ± 11.8b | 328 ± 31.1b | 24 ± 7.5c | 117 ± 12.1a | |
| 72 | 412 ± 9.8a | 74 ± 0.5a | 297 ± 9.0a | 545 ± 16.2a | 116 ± 3.2b | 133 ± 7.2a | |
| 120 | 454 ± 14a | 74 ± 0.5a | 284 ± 11.1a | 507 ± 17.7a | 170 ± 8.2a | 53 ± 7.9b | |
| 2 | 0 | 73 ± 2.1b | 80 ± 2.4a | 95 ± 1.1b | 151 ± 1.6b | −23 ± 1.5b | 79 ± 3.5c |
| 24 | 70 ± 3.0b | 76 ± 0.9a | 108 ± 5.9b | 144 ± 8.3b | −38 ± 3.1b | 74 ± 5.6c | |
| 72 | 229 ± 9.1a | 77 ± 0.3a | 265 ± 4.0a | 441 ± 7.9a | −36 ± 9.3b | 212 ± 11.9a | |
| 120 | 282 ± 17.5a | 76 ± 0.7a | 242 ± 21.5a | 426 ± 39.8a | 40 ± 4.5a | 144 ± 22.5b | |
| 3 | 0 | 73 ± 2.0c | 78 ± 0.9a | 89 ± 4.1c | 167 ± 6.0c | −16 ± 2.2c | 94 ± 5.2b |
| 24 | 75 ± 4.0c | 78 ± 2.3a | 88 ± 10.7c | 139 ± 13c | −13 ± 11.7c | 65 ± 13.7b | |
| 72 | 202 ± 15.5b | 75 ± 0.2a | 180 ± 11.6b | 331 ± 22.1b | 22 ± 4.0b | 130 ± 7.3a | |
| 120 | 482 ± 20.5a | 74 ± 1.2a | 301 ± 18.2a | 554 ± 32.9a | 181 ± 8a | 72 ± 18.9b | |
| 4 | 0 | 71 ± 3.3c | 80 ± 1.3ab | 83 ± 4.8b | 165 ± 7.3b | −12 ± 2.6c | 94 ± 4.5bc |
| 24 | 78 ± 7.9c | 80 ± 0.6a | 92 ± 8.2b | 141 ± 15.6b | −14 ± 2.9c | 63 ± 8.9c | |
| 72 | 258 ± 10.3b | 76 ± 0.8ab | 237 ± 7.7a | 467 ± 10.1a | 20 ± 3.7b | 210 ± 3.9a | |
| 120 | 336 ± 28.3a | 74 ± 0.8b | 252 ± 10.3a | 481 ± 17.8a | 85 ± 18a | 145 ± 10.5b | |
| 5 | 0 | 36 ± 1.8c | 75 ± 1.9b | 43 ± 0.8b | 60 ± 1.6b | −7 ± 2.0b | 24 ± 1.7b |
| 24 | 35 ± 10.0c | 82 ± 2.0a | 44 ± 15.0b | 63 ± 26.2b | −9 ± 6.3b | 29 ± 16.8b | |
| 72 | 99 ± 13.6b | 77 ± 0.5b | 130 ± 15.9a | 220 ± 30.8a | −32 ± 3.9b | 121 ± 17.1a | |
| 120 | 223 ± 16.5a | 77 ± 0.4ab | 169 ± 13.0a | 303 ± 25.0a | 54 ± 5.5a | 80 ± 9.5ab |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ortiz, D.; Nkhata, S.G.; Rocheford, T.; Ferruzzi, M.G. Steeping of Biofortified Orange Maize Genotypes for Ogi Production Modifies Pasting Properties and Carotenoid Stability. Agronomy 2019, 9, 771. https://doi.org/10.3390/agronomy9110771
Ortiz D, Nkhata SG, Rocheford T, Ferruzzi MG. Steeping of Biofortified Orange Maize Genotypes for Ogi Production Modifies Pasting Properties and Carotenoid Stability. Agronomy. 2019; 9(11):771. https://doi.org/10.3390/agronomy9110771
Chicago/Turabian StyleOrtiz, Darwin, Smith G. Nkhata, Torbert Rocheford, and Mario G. Ferruzzi. 2019. "Steeping of Biofortified Orange Maize Genotypes for Ogi Production Modifies Pasting Properties and Carotenoid Stability" Agronomy 9, no. 11: 771. https://doi.org/10.3390/agronomy9110771
APA StyleOrtiz, D., Nkhata, S. G., Rocheford, T., & Ferruzzi, M. G. (2019). Steeping of Biofortified Orange Maize Genotypes for Ogi Production Modifies Pasting Properties and Carotenoid Stability. Agronomy, 9(11), 771. https://doi.org/10.3390/agronomy9110771






