Salinity and Low Phosphorus Differentially Affect Shoot and Root Traits in Two Wheat Cultivars with Contrasting Tolerance to Salt
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material and Growth Conditions
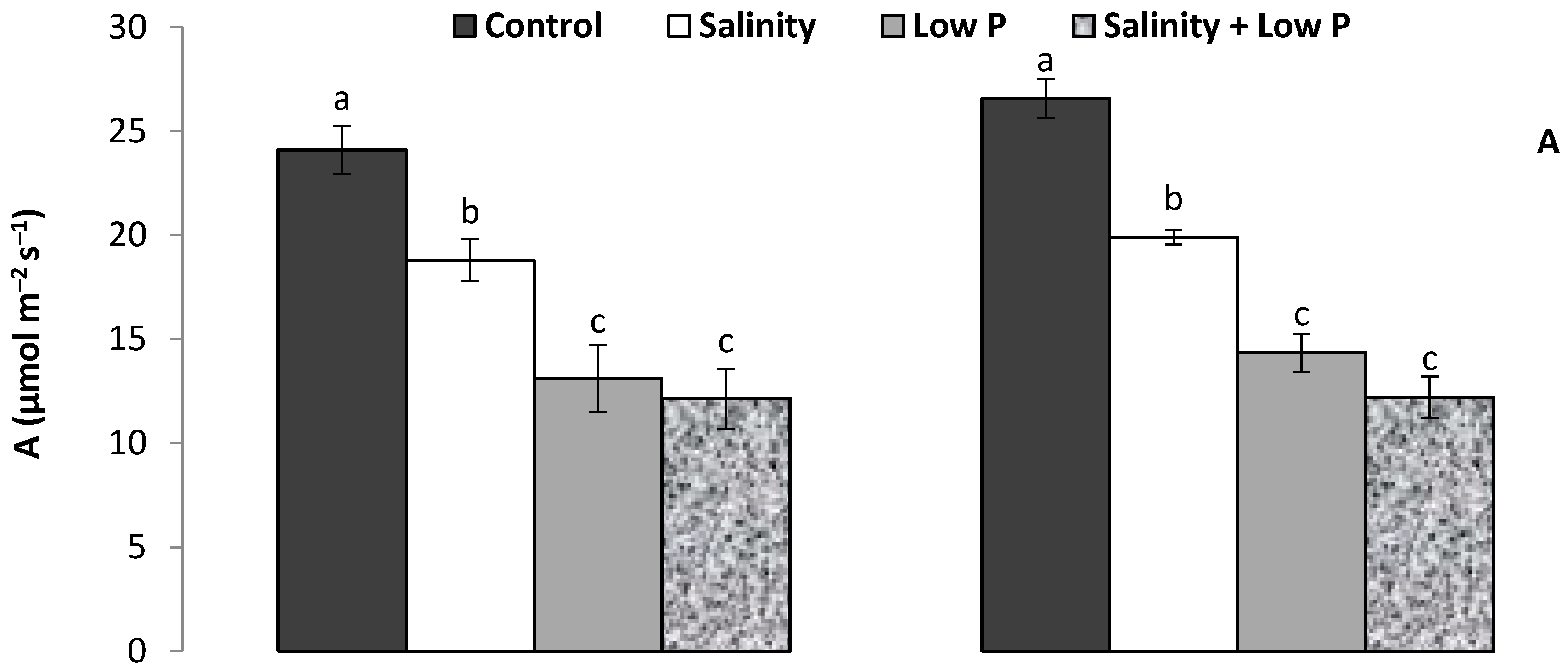
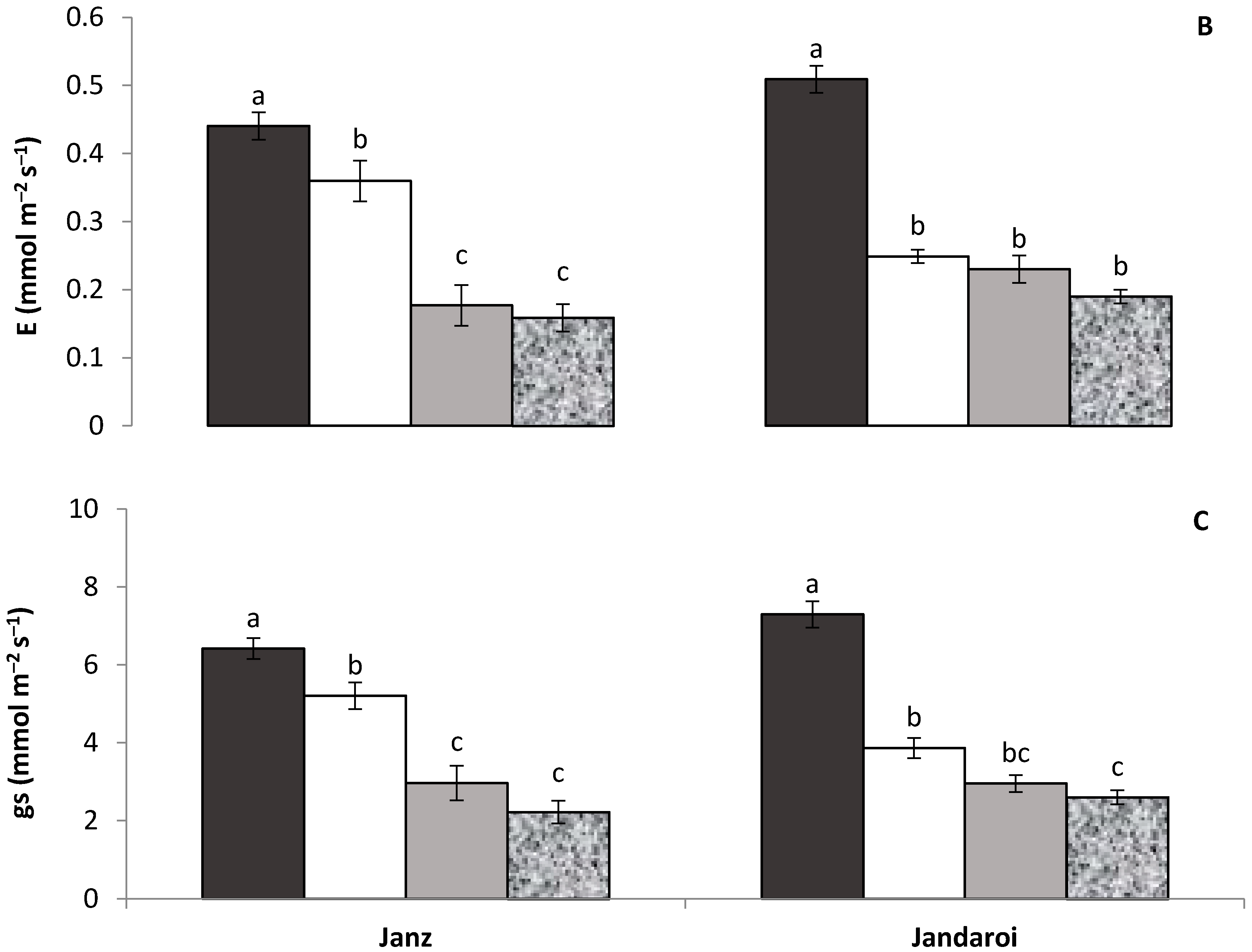
2.2. Gas Exchange and Chlorophyll Content
2.3. Shoot Traits and Nutrients
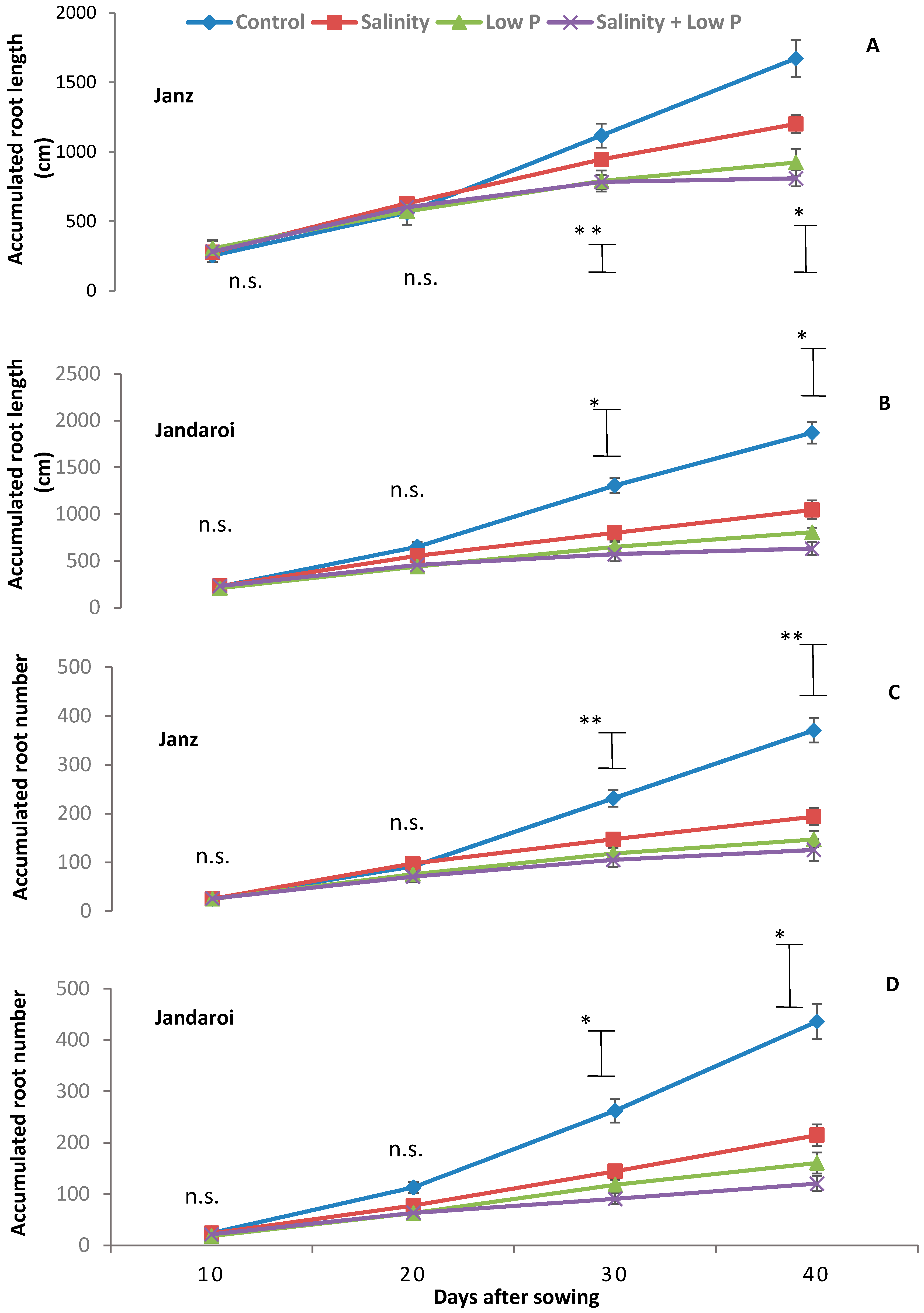
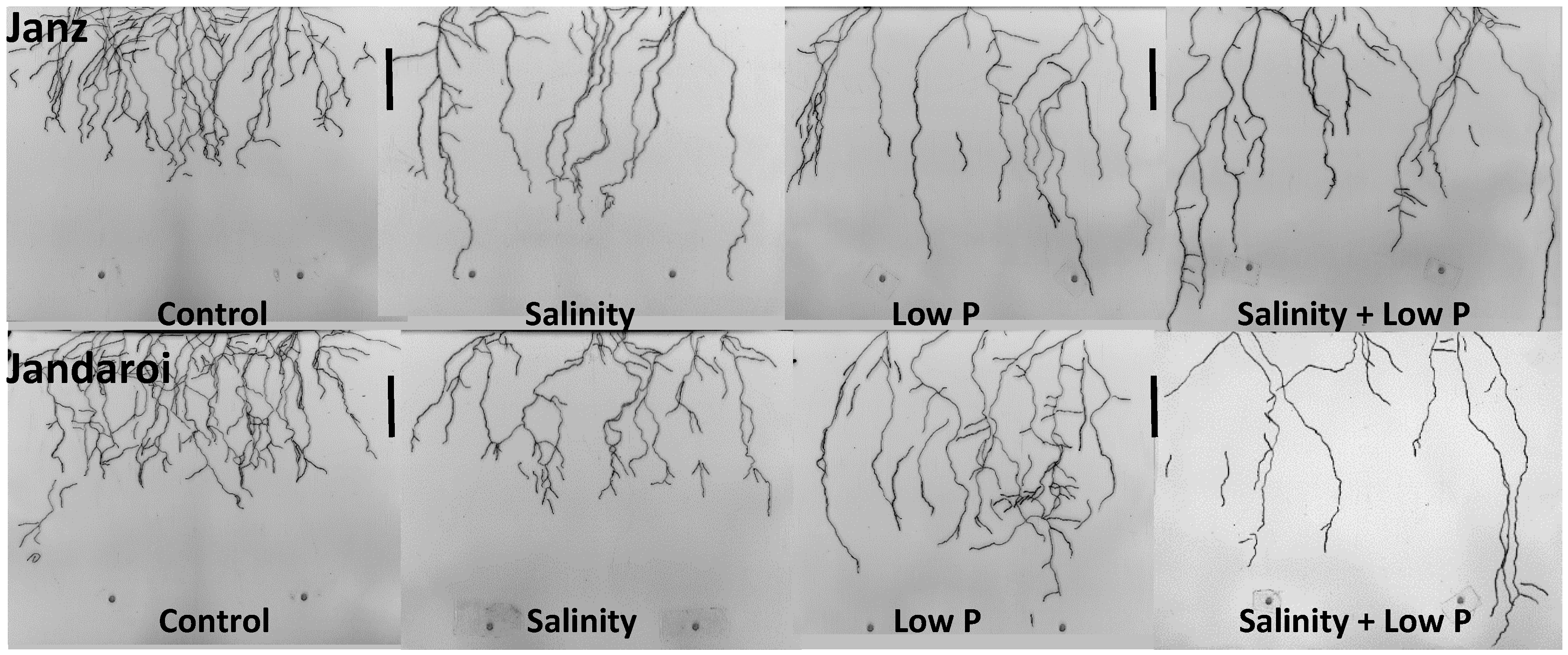
2.4. Root Growth
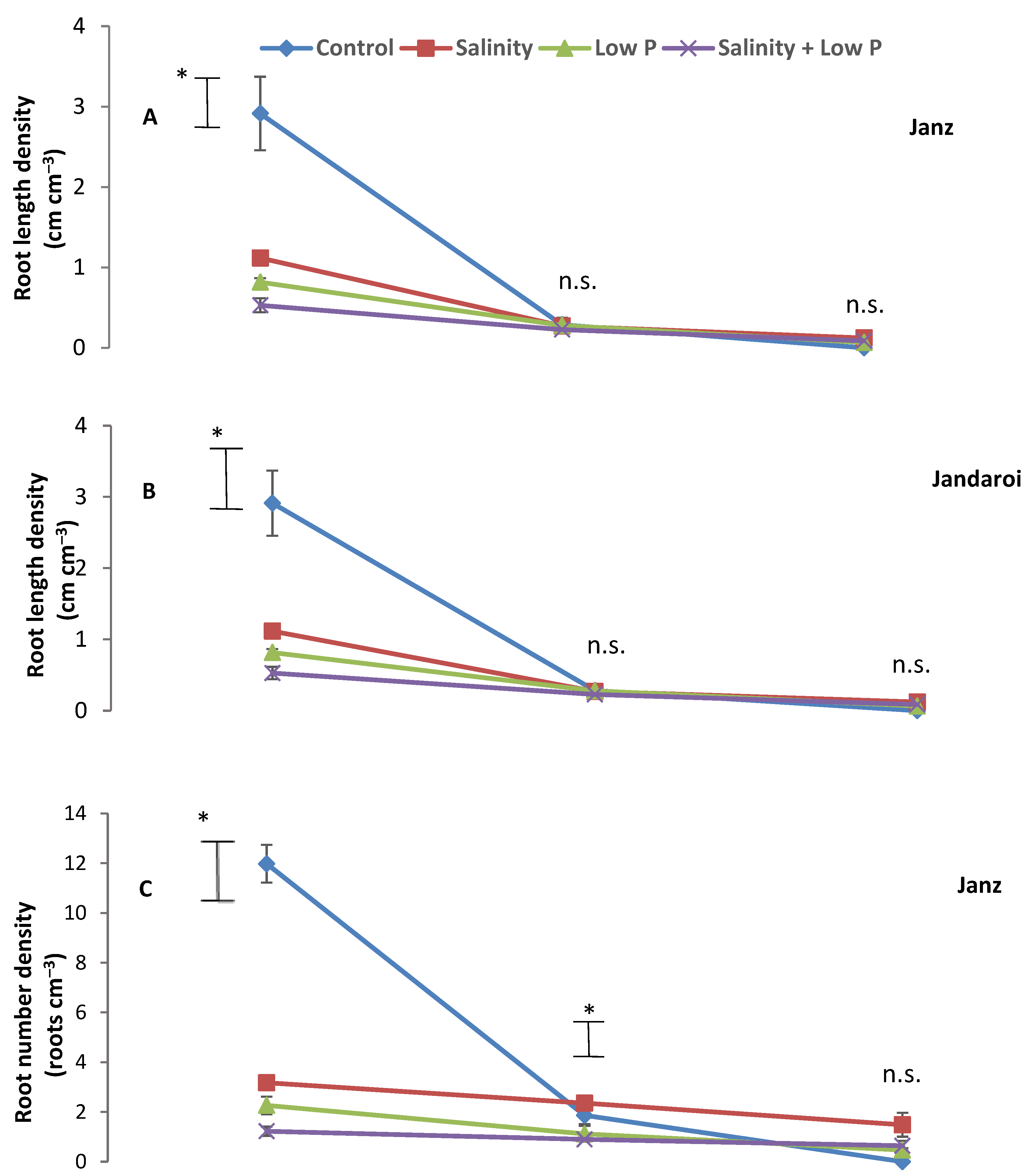
2.5. Root Traits at Harvest
2.6. Statistical Analysis
3. Results
3.1. Plant Growth and Shoot Traits
3.2. Tissue Ionic Contents
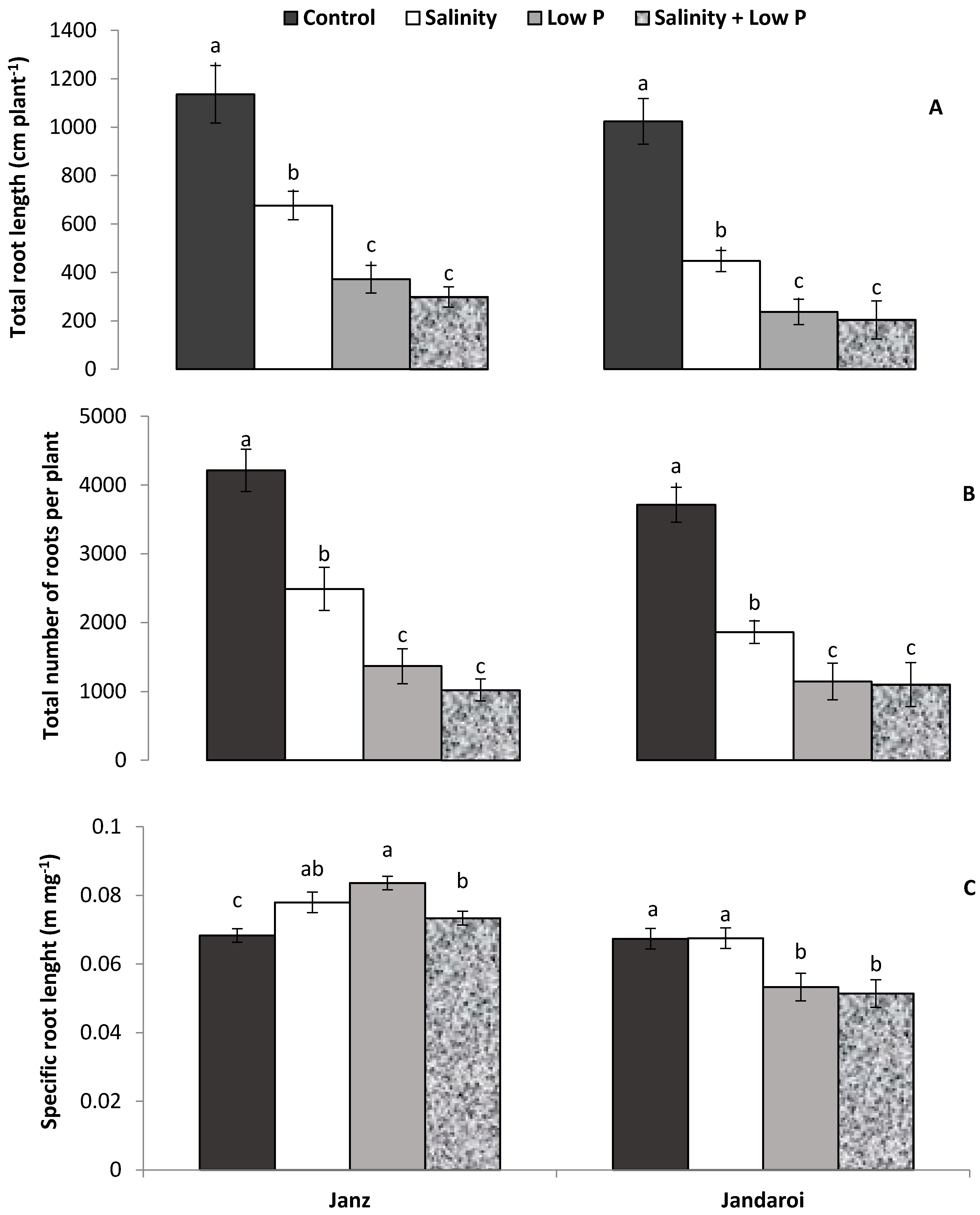
3.3. Root Growth
4. Discussion
4.1. Relative Effect of P and Salt on Growth and Physiology
4.2. Relative Effects of P and Salt on Ionic Balance
4.3. Relative Effects of P and Salt on Root Growth and Morphology
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Munns, R.; Tester, M. Mechanisms of salinity tolerance. Annu. Rev. Plant Biol. 2008, 59, 651–681. [Google Scholar] [CrossRef] [PubMed]
- Qadir, M.; Quillérou, E.; Nangia, V.; Murtaza, G.; Singh, M.; Thomas, R.J.; Drechsel, P.; Noble, A.D. Economics of salt-induced land degradation and restoration. In Natural Resources Forum; Wiley Online Library: Bognor Regis, UK, 2014; pp. 282–295. [Google Scholar]
- Negrão, S.; Schmöckel, S.; Tester, M. Evaluating physiological responses of plants to salinity stress. Ann. Bot. 2017, 119, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Abbas, G.; Saqib, M.; Akhtar, J.; Murtaza, G. Physiological and biochemical characterization of Acacia stenophylla and Acacia albida exposed to salinity under hydroponic conditions. Can. J. For. Res. 2017, 47, 1293–1301. [Google Scholar] [CrossRef]
- Flowers, T.J.; Colmer, T.D. Plant salt tolerance: Adaptations in halophytes. Ann. Bot. 2015, 115, 327–331. [Google Scholar] [CrossRef] [PubMed]
- Saqib, M.; Zörb, C.; Rengel, Z.; Schubert, S. The expression of the endogenous vacuolar Na+/H+ antiporters in roots and shoots correlates positively with the salt resistance of wheat (Triticum aestivum L.). Plant Sci. 2005, 169, 959–965. [Google Scholar] [CrossRef]
- Saqib, M.; Akhtar, J.; Abbas, G.; Nasim, M. Salinity and drought interaction in wheat (Triticum aestivum L.) is affected by the genotype and plant growth stage. Acta Physiol. Plant. 2013, 35, 2761–2768. [Google Scholar] [CrossRef]
- Hu, Y.; Hackl, H.; Schmidhalter, U. Comparative performance of spectral and thermographic properties of plants and physiological traits for phenotyping salinity tolerance of wheat cultivars under simulated field conditions. Funct. Plant Biol. 2017, 44, 134–142. [Google Scholar] [CrossRef]
- Talbi, Z.O.; Abdelly, C.; Debez, A. Interactive effects of salinity and phosphorus availability on growth, water relations, nutritional status and photosynthetic activity of barley (Hordeum vulgare L.). Plant Biol. 2011, 13, 872–880. [Google Scholar] [CrossRef] [PubMed]
- Abbas, G.; Saqib, M.; Akhtar, J. Interactive effects of salinity and iron deficiency on different rice genotypes. J. Plant Nutr. Soil Sci. 2015, 178, 306–311. [Google Scholar] [CrossRef]
- Rahnama, A.; Munns, R.; Poustini, K.; Watt, M. A screening method to identify genetic variation in root growth response to a salinity gradient. J. Exp. Bot. 2011, 62, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Zribi, O.T.; Houmani, H.; Kouas, S.; Slama, I.; Ksouri, R.; Abdelly, C. Comparative study of the interactive effects of salinity and phosphorus availability in wild (Hordeum maritimum) and cultivated barley (H. vulgare). J. Plant Growth Regul. 2014, 33, 860–870. [Google Scholar] [CrossRef]
- Fahad, S.; Hussain, S.; Saud, S.; Hassan, S.; Tanveer, M.; Ihsan, M.Z.; Shah, A.N.; Ullah, A.; Khan, F.; Ullah, S.; et al. A combined application of biochar and phosphorus alleviates heat-induced adversities on physiological, agronomical and quality attributes of rice. Plant Physiol. Biochem. 2016, 103, 191–198. [Google Scholar] [CrossRef] [PubMed]
- Vance, C.P.; Uhde-Stone, C.; Allan, D.L. Phosphorus acquisition and use: Critical adaptations by plants for securing a nonrenewable resource. New Phytol. 2003, 157, 423–447. [Google Scholar] [CrossRef]
- Chen, Y.L.; Dunbabin, V.M.; Diggle, A.J.; Siddique, K.H.; Rengel, Z. Phosphorus starvation boosts carboxylate secretion in P-deficient genotypes of Lupinus angustifolius with contrasting root structure. Crop Pasture Sci. 2013, 64, 588–599. [Google Scholar] [CrossRef]
- Niu, Y.F.; Chai, R.S.; Jin, G.L.; Wang, H.; Tang, C.X.; Zhang, Y.S. Responses of root architecture development to low phosphorus availability: A review. Ann. Bot. 2012, 112, 391–408. [Google Scholar] [CrossRef] [PubMed]
- Holford, I. Soil phosphorus: Its measurement, and its uptake by plants. Soil Res. 1997, 35, 227–240. [Google Scholar] [CrossRef]
- Marschner, P.; Solaiman, Z.; Rengel, Z. Rhizosphere properties of Poaceae genotypes under P-limiting conditions. Plant Soil 2006, 283, 11–24. [Google Scholar] [CrossRef]
- Qadir, M.; Noble, A.D.; Oster, J.; Schubert, S.; Ghafoor, A. Driving forces for sodium removal during phytoremediation of calcareous sodic and saline–sodic soils: A review. Soil Use Manag. 2005, 21, 173–180. [Google Scholar] [CrossRef]
- Martinez, V.; Bernstein, N.; Läuchli, A. Salt-induced inhibition of phosphorus transport in lettuce plants. Physiol. Plant 1996, 97, 118–122. [Google Scholar] [CrossRef]
- Kaya, C.; Higgs, D.; Kirnak, H. The effects of high salinity (NaCl) and supplementary phosphorus and potassium on physiology and nutrition development of spinach. Bulg. J. Plant Physiol. 2001, 27, 47–59. [Google Scholar]
- Zribi, O.T.; Labidi, N.; Slama, I.; Debez, A.; Ksouri, R.; Rabhi, M.; Smaoui, A.; Abdelly, C. Alleviation of phosphorus deficiency stress by moderate salinity in the halophyte Hordeum maritimum L. Plant Growth Regul. 2012, 66, 75–85. [Google Scholar] [CrossRef]
- Phang, T.H.; Shao, G.; Liao, H.; Yan, X.; Lam, H.M. High external phosphate (Pi) increases sodium ion uptake and reduces salt tolerance of ‘Pi-tolerant’soybean. Physiol. Plant. 2009, 135, 412–425. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Schmidhalter, U. Drought and salinity: A comparison of their effects on mineral nutrition of plants. J. Plant Nutr. Soil Sci. 2005, 168, 541–549. [Google Scholar] [CrossRef]
- Maas, E.V.; Hoffman, G.J. Crop salt tolerance–current assessment. J. Irrig. Drain. Div. 1977, 103, 115–134. [Google Scholar]
- Genc, Y.; Mcdonald, G.K.; Tester, M. Reassessment of tissue Na+ concentration as a criterion for salinity tolerance in bread wheat. Plant Cell Environ. 2007, 30, 1486–1498. [Google Scholar] [CrossRef] [PubMed]
- McDonald, G.; Bovill, W.; Taylor, J.; Wheeler, R. Responses to phosphorus among wheat genotypes. Crop Pasture Sci. 2015, 66, 430–444. [Google Scholar] [CrossRef]
- Osborne, L.; Rengel, Z. Genotypic differences in wheat for uptake and utilisation of P from iron phosphate. Crop Pasture Sci. 2002, 53, 837–844. [Google Scholar] [CrossRef]
- Liao, M.; Palta, J.A.; Fillery, I.R. Root characteristics of vigorous wheat improve early nitrogen uptake. Aust. J. Agric. Res. 2006, 57, 1097–1107. [Google Scholar] [CrossRef]
- Zaicou, C.; Curtis, B.; Dhammu, H.; Ellis, S.; Jorgensen, D.; Miyan, S.; Penny, S.; Shackley, B.; Sharma, D. Wheat Variety Guide 2008 Western Australia; Department of Agriculture and Food (Western Australia): Perth, Australia, 2008.
- Schwenke, G.; Simpfendorfer, S.; Collard, B. Confirmation of chloride deficiency as the cause of leaf spotting in durum wheat grown in the Australian northern grains region. Crop Pasture Sci. 2015, 66, 122–134. [Google Scholar] [CrossRef]
- Shamaya, N.J. Genetic Studies of Salinity Tolerance in Wheat. Master’s Thesis, The University of Adelaide, Adelaide, Australia, 2014. [Google Scholar]
- Zhu, M.; Shabala, S.; Shabala, L.; Fan, Y.; Zhou, M. Evaluating predictive values of various physiological indices for salinity stress tolerance in wheat. J. Agron. Crop Sci. 2016, 202, 115–124. [Google Scholar] [CrossRef]
- Aziz, M.M.; Palta, J.A.; Siddique, K.H.; Sadras, V.O. Five decades of selection for yield reduced root length density and increased nitrogen uptake per unit root length in Australian wheat varieties. Plant Soil 2017, 413, 181–192. [Google Scholar] [CrossRef]
- Palta, J.A.; Fillery, I.R.; Rebetzke, G.J. Restricted-tillering wheat does not lead to greater investment in roots and early nitrogen uptake. Field Crop. Res. 2007, 104, 52–59. [Google Scholar] [CrossRef]
- Pfeifer, J. Elucidation of Root-Soil Interactions of Crops in Space and Time by Establishment and Application of Novel Image Based Non-Invasive Root Phenotyping Methods. Doctoral Thesis, ETH-Zürich, Zurich, Switzerland, 2013. [Google Scholar]
- Basett, J.; Denney, R.; Jerrery, G.; Mendham, J. Vogel’s Text Book of Quantitative Inorganic Analysis, 5th ed.; Longman Group: London, UK, 1978. [Google Scholar]
- Chen, Y.L.; Dunbabin, V.M.; Diggle, A.J.; Siddique, K.H.; Rengel, Z. Assessing variability in root traits of wild Lupinus angustifolius germplasm: Basis for modelling root system structure. Plant Soil 2012, 354, 141–155. [Google Scholar] [CrossRef]
- Steel, R.; Torrie, J.; Dickey, D. Principles and Procedures of Statistics: A Biometrical Approach, 3rd ed.; McGraw-Hill Book Co. Inc.: New York, NY, USA, 1997. [Google Scholar]
- Chen, Y.L.; Dunbabin, V.M.; Postma, J.A.; Diggle, A.J.; Siddique, K.H.; Rengel, Z. Modelling root plasticity and response of narrow-leafed lupin to heterogeneous phosphorus supply. Plant Soil 2013, 372, 319–337. [Google Scholar] [CrossRef]
- Wang, J.; Li, B.; Meng, Y.; Ma, X.; Lai, Y.; Si, E.; Yang, K.; Ren, P.; Shang, X.; Wang, H. Transcriptomic profiling of the salt-stress response in the halophyte Halogeton glomeratus. BMC Genom. 2015, 16, 169. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; (The University of Western Australia, Australia). Personal communication, 2017.
- Munns, R.; James, R.A. Screening methods for salinity tolerance: A case study with tetraploid wheat. Plant Soil 2003, 253, 201–218. [Google Scholar] [CrossRef]
- Li, J.; Xie, Y.; Dai, A.; Liu, L.; Li, Z. Root and shoot traits responses to phosphorus deficiency and QTL analysis at seedling stage using introgression lines of rice. J. Genet. Genom. 2009, 36, 173–183. [Google Scholar] [CrossRef]
- Tewari, R.K.; Kumar, P.; Tewari, N.; Srivastava, S.; Sharma, P.N. Macronutrient deficiencies and differential antioxidant responses—Influence on the activity and expression of superoxide dismutase in maize. Plant Sci. 2004, 166, 687–694. [Google Scholar] [CrossRef]
- Sultana, N.; Ikeda, T.; Itoh, R. Effect of NaCl salinity on photosynthesis and dry matter accumulation in developing rice grains. Environ. Exp. Bot. 1999, 42, 211–220. [Google Scholar] [CrossRef]
- He, G.; Zhang, J.; Hu, X.; Wu, J. Effect of aluminum toxicity and phosphorus deficiency on the growth and photosynthesis of oil tea (Camellia oleifera Abel.) seedlings in acidic red soils. Acta Physiol. Plant. 2011, 33, 1285–1292. [Google Scholar] [CrossRef]
- Koyro, H.-W. Effect of salinity on growth, photosynthesis, water relations and solute composition of the potential cash crop halophyte Plantago coronopus (L.). Environ. Exp. Bot. 2006, 56, 136–146. [Google Scholar] [CrossRef]
- Yousfi, S.; Houmani, H.; Zribi, F.; Abdelly, C.; Gharsalli, M. Physiological responses of wild and cultivated barley to the interactive effect of salinity and iron deficiency. ISRN Agron. 2012. [Google Scholar] [CrossRef]
- Gorham, J.; Jones, R.W.; Bristol, A. Partial characterization of the trait for enhanced K+−Na+ discrimination in the D genome of wheat. Planta 1990, 180, 590–597. [Google Scholar] [CrossRef] [PubMed]
- Byrt, C.S.; Xu, B.; Krishnan, M.; Lightfoot, D.J.; Athman, A.; Jacobs, A.K.; Watson-Haigh, N.S.; Plett, D.; Munns, R.; Tester, M.; et al. The Na+ transporter, TaHKT1; 5-D, limits shoot Na+ accumulation in bread wheat. Plant J. 2014, 80, 516–526. [Google Scholar] [CrossRef] [PubMed]
- Grattan, S.; Grieve, C. Salinity–mineral nutrient relations in horticultural crops. Sci. Hortic. 1998, 78, 127–157. [Google Scholar] [CrossRef]
- Adams, E.; Shin, R. Transport, signaling, and homeostasis of potassium and sodium in plants. J. Integr. Plant Biol. 2014, 56, 231–249. [Google Scholar] [CrossRef] [PubMed]
- Marschner, H. Mineral Nutrition of Higher Plants, 3rd ed.; Academic Press: London, UK, 1995. [Google Scholar]
- Fujita, K.; Kai, Y.; Takayanagi, M.; El-Shemy, H.; Adu-Gyamfi, J.J.; Mohapatra, P.K. Genotypic variability of pigeonpea in distribution of photosynthetic carbon at low phosphorus level. Plant Sci. 2004, 166, 641–649. [Google Scholar] [CrossRef]

| Janz | Control | Salinity | Low P | Salinity + Low P |
|---|---|---|---|---|
| SL | 22.2 ± 0.93 a | 19.7 ± 1.0 a | 14.8 ± 1.59 b | 13.7 ± 0.33 b |
| LA | 575.8 ± 19 a | 323.8 ± 18 b | 177.7 ± 28.5 c | 162.2 ± 14.4 c |
| SDW | 0.132 ± 0.01 a | 0.073 ± 0.003 b | 0.029 ± 0.002 c | 0.026 ± 0.006 c |
| RDW | 0.166 ± 0.013 a | 0.087 ± 0.0104 b | 0.044 ± 0.006 c | 0.041 ± 0.003 c |
| RDW: SDW | 1.25 ± 0.06 b | 1.18 ± 0.05 b | 1.52 ± 0.05 a | 1.56 ± 0.04 a |
| CCI | 38 ± 2.88 a | 36.4 ± 2.28 a | 25.6 ± 1.04 b | 24 ± 1.82 c |
| TWC | 8.61 ± 0.17 a | 7.54 ± 0.17 b | 3.6 ± 0.25 c | 3.3 ± 0.3 c |
| Jandaroi | ||||
| SL | 24.2 ± 0.93 a | 17± 0.58 b | 16.5 ± 1.26 b | 14.8 ± 1.74 b |
| LA | 517 ± 24 a | 260 ± 8 b | 180.1 ± 28 c | 165.4 ± 12 c |
| SDW | 0.128 ± 0.007 a | 0.046 ± 0.004 b | 0.032 ± 0.003 c | 0.029 ± 0.005 c |
| RDW | 0.152 ± 0.011 a | 0.066 ± 0.001 b | 0.044 ± 0.002 c | 0.040 ± 0.007 c |
| RDW: SDW | 1.18 ± 0.03 b | 1.42 ± 0.02 a | 1.40 ± 0.02 a | 1.38 ± 0.04 a |
| CCI | 42.37 ± 0.68 a | 28 ± 2.89 b | 27.1 ± 1.91 b | 24 ± 2.95 b |
| TWC | 7.6 ± 0.17 a | 5.4 ± 0.33 b | 4.96 ± 0.3b c | 4 ± 0.13 c |
| Janz | Control | Salinity | Low P | Salinity + Low P |
|---|---|---|---|---|
| Shoot Na | 0.05 ± 0.01 c | 0.65 ± 0.08 a | 0.05 ± 0.02 c | 0.28 ± 0.08 b |
| Shoot K | 0.68 ± 0.06 a | 0.45 ± 0.04 c | 0.57 ± 0.03 b | 0.41 ± 0.04 c |
| Shoot P | 0.08 ± 0.003 a | 0.06 ± 0.004 b | 0.02 ± 0.001 c | 0.01 ± 0.005 c |
| Root Na | 0.04 ± 0.02 c | 0.34 ± 0.04 a | 0.04 ± 0.01 c | 0.20 ± 0.02 b |
| Root K | 0.47 ± 0.03 a | 0.22 ± 0.03 c | 0.31 ± 0.01 b | 0.15 ± 0.04 c |
| Root P | 0.034 ± 0.001 a | 0.026 ± 0.002 b | 0.012 ± 0.000 c | 0.014 ± 0.002 c |
| Shoot K: Na | 13.6 ± 0.60 a | 0.69 ± 0.30 c | 11.4 ± 0.40 b | 1.48 ± 0.60 c |
| Root K: Na | 11.64 ± 0.60 a | 0.63 ± 0.30 c | 8.43 ± 0.80 b | 0.74 ± 0.40 c |
| Jandaroi | ||||
| Shoot Na | 0.08 ± 0.01 c | 0.93 ± 0.09 a | 0.09 ± 0.01 c | 0.66 ± 0.07 b |
| Shoot K | 0.72 ± 0.03 a | 0.38 ± 0.03 c | 0.60 ± 0.04 b | 0.20 ± 0.03 d |
| Shoot P | 0.06 ± 0.002 a | 0.05 ± 0.001 b | 0.02 ± 0.002 c | 0.02 ± 0.004 c |
| Root Na | 0.05 ± 0.02 c | 0.36 ± 0.02 a | 0.05 ± 0.01 c | 0.23 ± 0.03 b |
| Root K | 0.45 ± 0.05 a | 0.16 ± 0.01 c | 0.29 ± 0.02 b | 0.13 ± 0.04 c |
| Root P | 0.029 ± 0.002 a | 0.019 ± 0.001 b | 0.012 ± 0.001 c | 0.013 ± 0.002 c |
| Shoot K: Na | 9.02 ± 0.90 a | 0.41 ± 0.20 c | 6.67 ± 0.80 b | 0.30 ± 0.10 c |
| Root K: Na | 8.99 ± 0.40 a | 0.44 ± 0.20 c | 5.75 ± 0.60 b | 0.57 ± 0.30 c |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Abbas, G.; Chen, Y.; Khan, F.Y.; Feng, Y.; Palta, J.A.; Siddique, K.H.M. Salinity and Low Phosphorus Differentially Affect Shoot and Root Traits in Two Wheat Cultivars with Contrasting Tolerance to Salt. Agronomy 2018, 8, 155. https://doi.org/10.3390/agronomy8080155
Abbas G, Chen Y, Khan FY, Feng Y, Palta JA, Siddique KHM. Salinity and Low Phosphorus Differentially Affect Shoot and Root Traits in Two Wheat Cultivars with Contrasting Tolerance to Salt. Agronomy. 2018; 8(8):155. https://doi.org/10.3390/agronomy8080155
Chicago/Turabian StyleAbbas, Ghulam, Yinglong Chen, Faisal Younus Khan, Yupeng Feng, Jairo A. Palta, and Kadambot H. M. Siddique. 2018. "Salinity and Low Phosphorus Differentially Affect Shoot and Root Traits in Two Wheat Cultivars with Contrasting Tolerance to Salt" Agronomy 8, no. 8: 155. https://doi.org/10.3390/agronomy8080155
APA StyleAbbas, G., Chen, Y., Khan, F. Y., Feng, Y., Palta, J. A., & Siddique, K. H. M. (2018). Salinity and Low Phosphorus Differentially Affect Shoot and Root Traits in Two Wheat Cultivars with Contrasting Tolerance to Salt. Agronomy, 8(8), 155. https://doi.org/10.3390/agronomy8080155






