Arabidopsis thaliana Immunity-Related Compounds Modulate Disease Susceptibility in Barley
Abstract
1. Introduction
2. Materials and Methods
2.1. Plants and Growth Conditions
2.2. Chemicals and Treatments
2.3. Xanthomonas Translucens pv. Cerealis (Xtc) Infection
2.4. Blumeria Graminis f. sp. Hordei (Bgh) Infection
2.5. P. Teres Infection
2.6. Statistics
3. Results
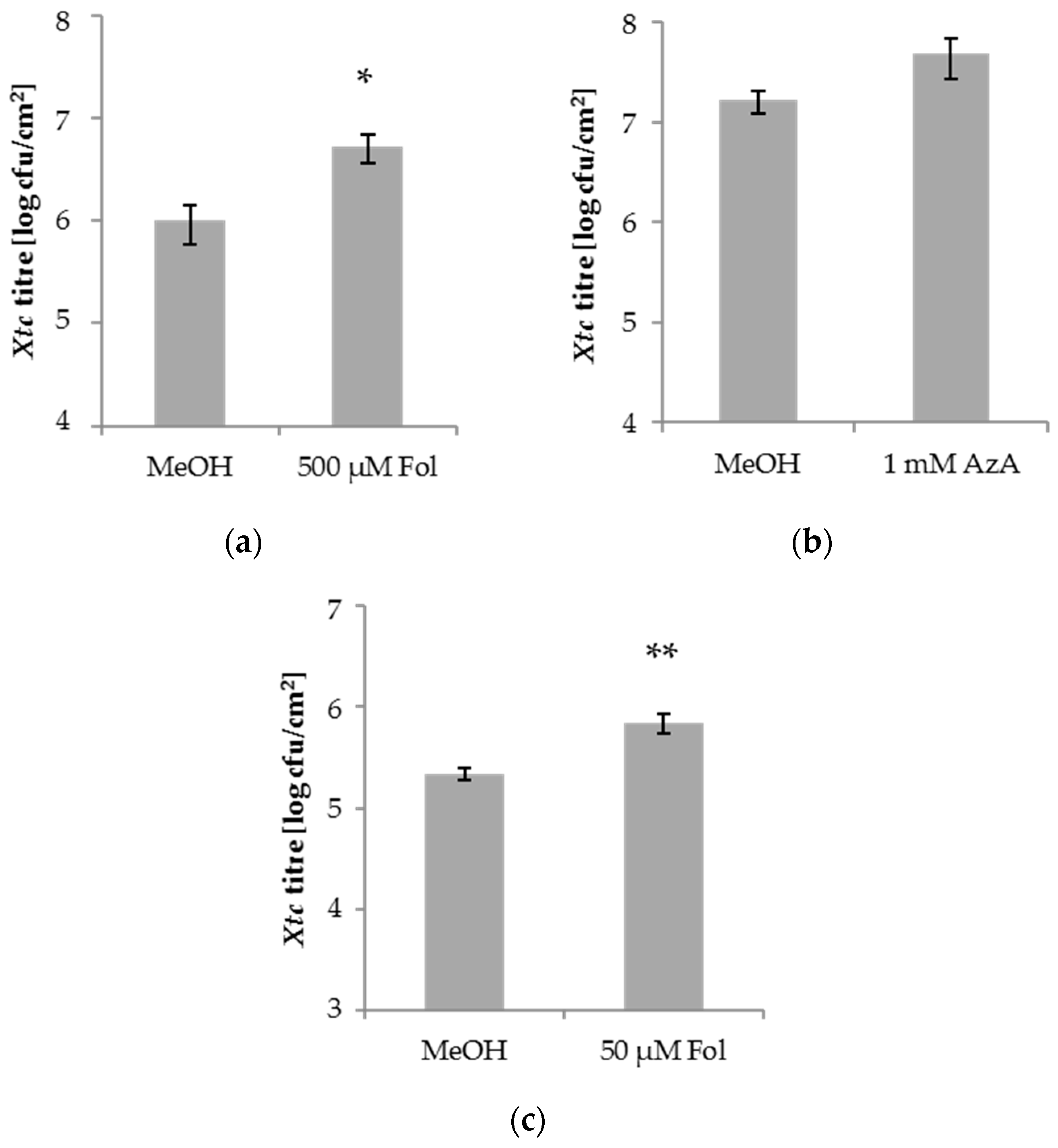
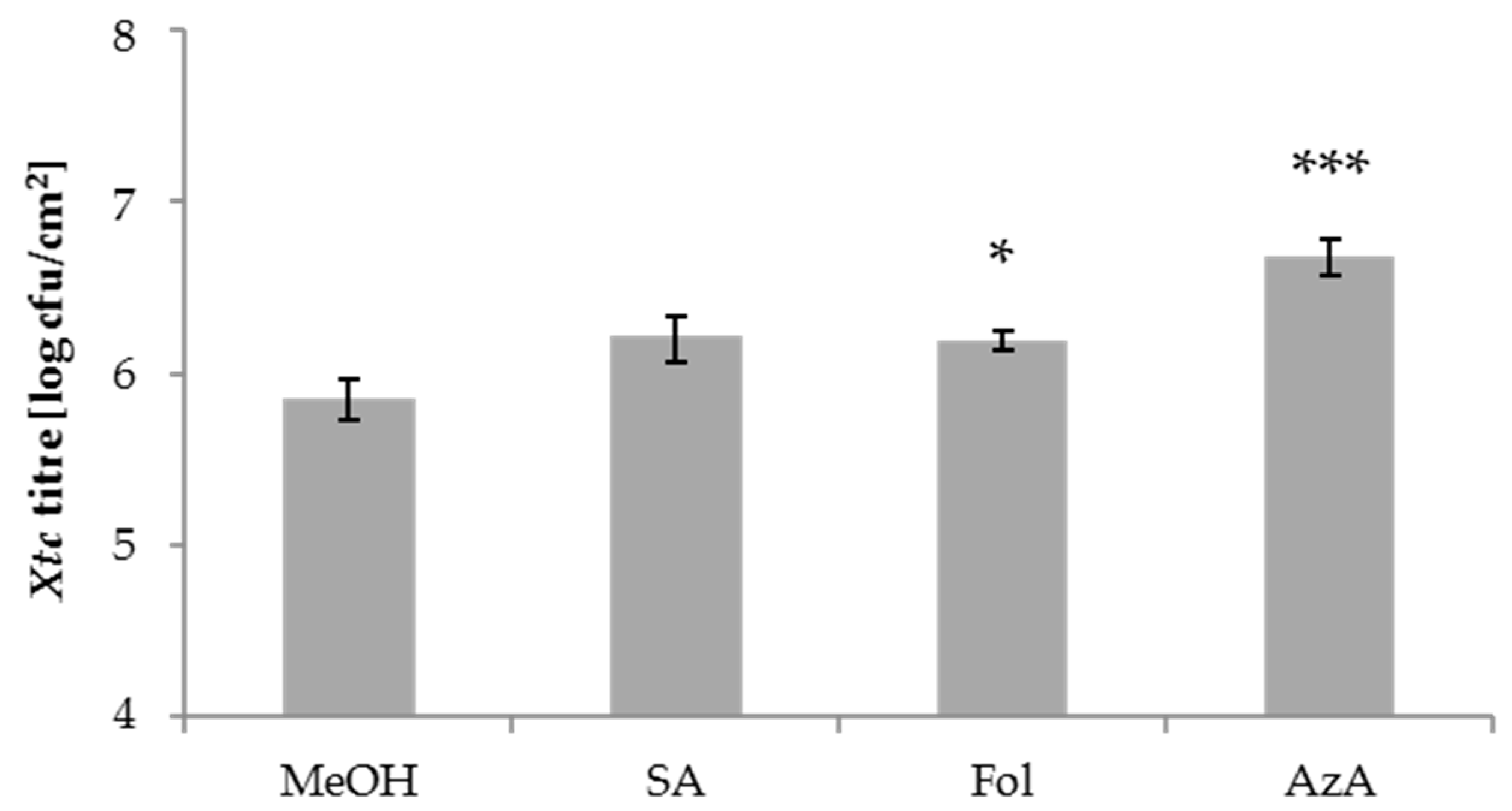
3.1. Folic Acid and Azelaic Acid Enhance the Susceptibility of Barley to Xtc
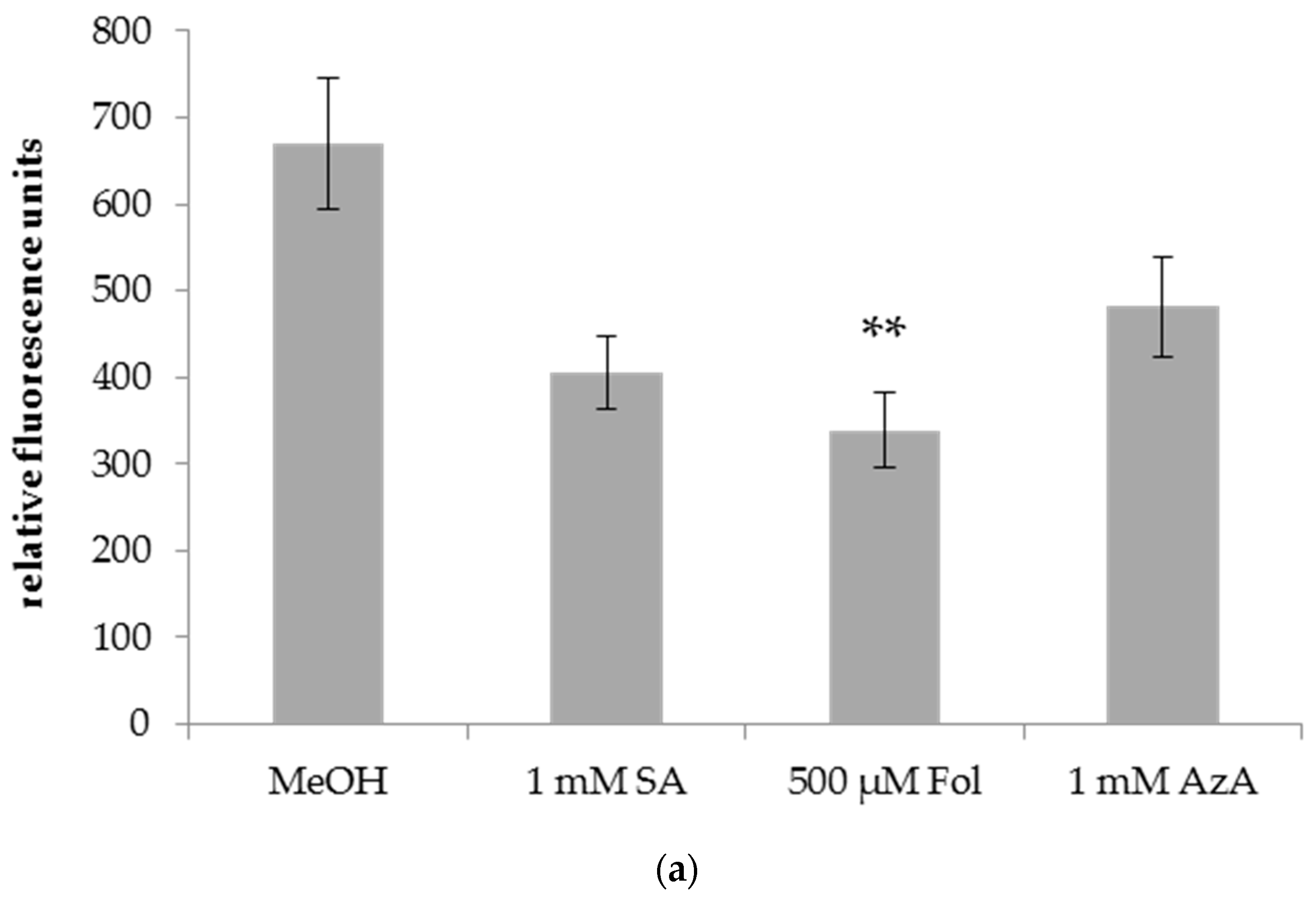
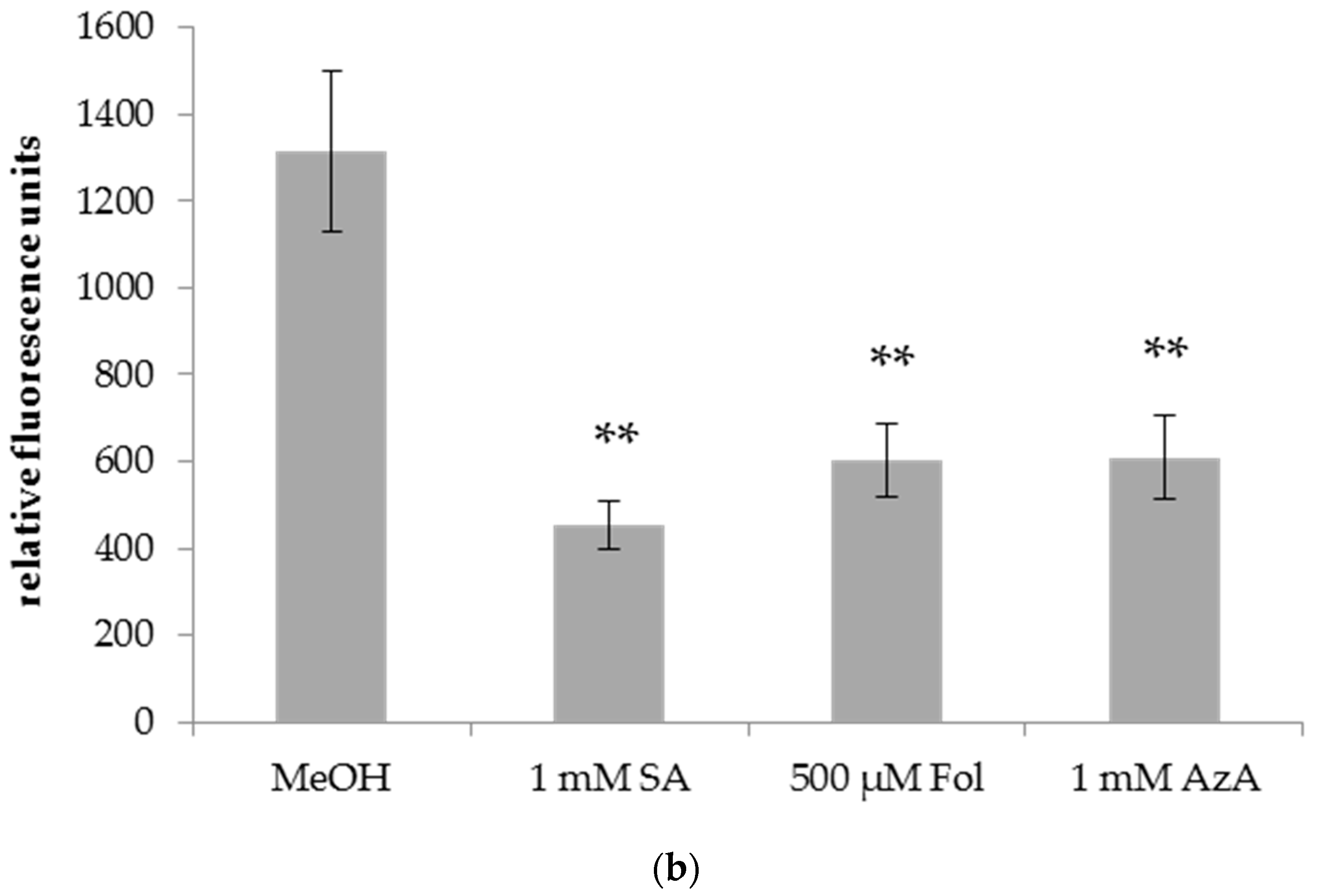
3.2. Folic Acid Enhances the Resistance of Barley to Powdery Mildew (Bgh)
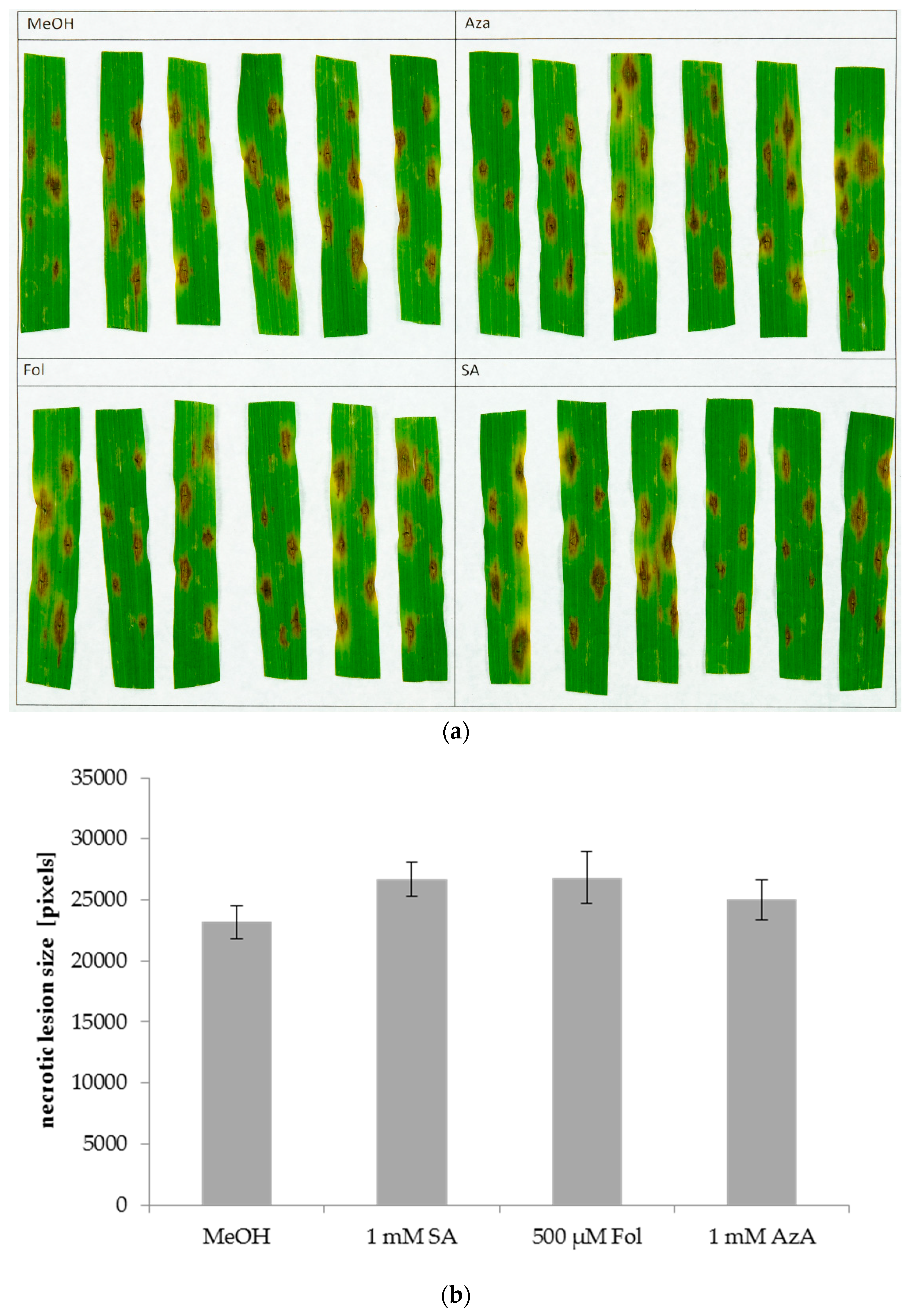
3.3. Arabidopsis Immunity-Related Compounds Do Not Alter Barley Susceptibility to P. Teres
4. Discussion
4.1. Salicylic Acid Has Differential Effects on Pathogens with Different Lifestyles
4.2. Folic Acid Has Differential Effects against Bacteria and Fungi with Similar Lifestyles
4.3. Azelaic Acid Moderately Affects Barley Defence Responses
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Panstruga, R.; Parker, J.E.; Schulze-Lefert, P. SnapShot: Plant Immune Response Pathways. Cell 2009, 136, 978.e1–978.e3. [Google Scholar] [CrossRef] [PubMed]
- Jones, J.D.G.; Dangl, J.L. The plant immune system. Nature 2006, 444, 323–329. [Google Scholar] [CrossRef] [PubMed]
- Glazebrook, J. Contrasting Mechanisms of Defense against Biotrophic and Necrotrophic Pathogens. Annu. Rev. Phytopathol. 2005, 43, 205–227. [Google Scholar] [CrossRef] [PubMed]
- Spoel, S.H.; Dong, X. How do plants achieve immunity? Defence without specialized immune cells. Nat. Rev. Immunol. 2012, 12, 89–100. [Google Scholar] [CrossRef] [PubMed]
- Zipfel, C.; Felix, G. Plants and animals: A different taste for microbes? Curr. Opin. Plant Biol. 2005, 8, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Henry, E.; Yadeta, K.A.; Coaker, G. Recognition of bacterial plant pathogens: Local, systemic and transgenerational immunity. New Phytol. 2013, 199, 908–915. [Google Scholar] [CrossRef] [PubMed]
- Klessig, D.F.; Choi, H.W.; Dempsey, M.A. Systemic Acquired Resistance and Salicylic Acid: Past, Present and Future. Mol. Plant-Microbe Interact. 2018. [Google Scholar] [CrossRef] [PubMed]
- Vlot, A.C.; Dempsey, D.A.; Klessig, D.F. Salicylic Acid, a Multifaceted Hormone to Combat Disease. Annu. Rev. Phytopathol. 2009, 47, 177–206. [Google Scholar] [CrossRef] [PubMed]
- Fu, Z.Q.; Dong, X. Systemic Acquired Resistance: Turning Local Infection into Global Defense. Annu. Rev. Plant Biol. 2013, 64, 839–863. [Google Scholar] [CrossRef] [PubMed]
- Pajerowska-Mukhtar, K.M.; Emerine, D.K.; Mukhtar, M.S. Tell me more: Roles of NPRs in plant immunity. Trends Plant Sci. 2013, 18, 402–411. [Google Scholar] [CrossRef] [PubMed]
- Van Loon, L.C.; Rep, M.; Pieterse, C.M.J. Significance of Inducible Defense-related Proteins in Infected Plants. Annu. Rev. Phytopathol. 2006, 44, 135–162. [Google Scholar] [CrossRef] [PubMed]
- Shah, J.; Chaturvedi, R.; Chowdhury, Z.; Venables, B.; Petros, R.A. Signaling by small metabolites in systemic acquired resistance. Plant J. 2014, 79, 645–658. [Google Scholar] [CrossRef] [PubMed]
- Conrath, U.; Beckers, G.J.M.; Flors, V.; García-Agustín, P.; Jakab, G.; Mauch, F.; Newman, M.-A.; Pieterse, C.M.J.; Poinssot, B.; Pozo, M.J.; et al. Priming: Getting Ready for Battle. Mol. Plant-Microbe Interact. 2006, 19, 1062–1071. [Google Scholar] [CrossRef] [PubMed]
- Gourbal, B.; Pinaud, S.; Beckers, G.J.M.; Van Der Meer, J.W.M.; Conrath, U.; Netea, M.G. Innate immune memory: An evolutionary perspective. Immunol. Rev. 2018, 283, 21–40. [Google Scholar] [CrossRef] [PubMed]
- Riedlmeier, M.; Ghirardo, A.; Wenig, M.; Knappe, C.; Koch, K.; Georgii, E.; Dey, S.; Parker, J.E.; Schnitzler, J.-P.; Vlot, A.C. Monoterpenes Support Systemic Acquired Resistance within and between Plants. Plant Cell 2017, 29, 1440–1459. [Google Scholar] [CrossRef] [PubMed]
- Park, S.W.; Kaimoyo, E.; Kumar, D.; Mosher, S.; Klessig, D.F. Methyl salicylate is a critical mobile signal for plant systemic acquired resistance. Science 2007, 318, 113–116. [Google Scholar] [CrossRef] [PubMed]
- Chanda, B.; Xia, Y.; Mandal, M.K.; Yu, K.; Sekine, K.T.; Gao, Q.M.; Selote, D.; Hu, Y.; Stromberg, A.; Navarre, D.; et al. Glycerol-3-phosphate is a critical mobile inducer of systemic immunity in plants. Nat. Genet. 2011, 43, 421–429. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Liu, R.; Lim, G.-H.; De Lorenzo, L.; Yu, K.; Zhang, K.; Hunt, A.G.; Kachroo, A.; Kachroo, P. Pipecolic acid confers systemic immunity by regulating free radicals. Sci. Adv. 2018, 4, eaar4509. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.W.; Tschaplinski, T.J.; Wang, L.; Glazebrook, J.; Greenberg, J.T. Priming in Systemic Plant Immunity. Science 2009, 324, 89–91. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, R.; Venables, B.; Petros, R.A.; Nalam, V.; Li, M.; Wang, X.; Takemoto, L.J.; Shah, J. An abietane diterpenoid is a potent activator of systemic acquired resistance. Plant J. 2012, 71, 161–172. [Google Scholar] [CrossRef] [PubMed]
- Hartmann, M.; Zeier, T.; Bernsdorff, F.; Reichel-Deland, V.; Kim, D.; Hohmann, M.; Scholten, N.; Schuck, S.; Bräutigam, A.; Hölzel, T.; et al. Flavin Monooxygenase-Generated N-Hydroxypipecolic Acid Is a Critical Element of Plant Systemic Immunity. Cell 2018, 173, 456–469. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.-C.; Holmes, E.C.; Rajniak, J.; Kim, J.-G.; Tang, S.; Fischer, C.R.; Mudgett, M.B.; Sattely, E.S. N-hydroxy-pipecolic acid is a mobile metabolite that induces systemic disease resistance in Arabidopsis. Proc. Natl. Acad. Sci. USA 2018, 115, E4920–E4929. [Google Scholar] [CrossRef] [PubMed]
- Maldonado, A.M.; Doerner, P.; Dixon, R.A.; Lamb, C.J.; Cameron, R.K. A putative lipid transfer protein involved in systemic resistance signalling in Arabidopsis. Nature 2002, 419, 399–403. [Google Scholar] [CrossRef] [PubMed]
- Champigny, M.J.; Isaacs, M.; Carella, P.; Faubert, J.; Fobert, P.R.; Cameron, R.K. Long distance movement of DIR1 and investigation of the role of DIR1-like during systemic acquired resistance in Arabidopsis. Front. Plant Sci. 2013, 4, 230. [Google Scholar] [CrossRef] [PubMed]
- Vlot, A.C.; Pabst, E.; Riedlmeier, M. Systemic Signalling in Plant Defence. eLS 2017, 1–9. [Google Scholar] [CrossRef]
- Gao, Q.-M.; Zhu, S.; Kachroo, P.; Kachroo, A. Signal regulators of systemic acquired resistance. Front. Plant Sci. 2015, 6, 228. [Google Scholar] [CrossRef] [PubMed]
- Métraux, J.P.; Signer, H.; Ryals, J.; Ward, E.; Wyss-Benz, M.; Gaudin, J.; Raschdorf, K.; Schmid, E.; Blum, W.; Inverardi, B. Increase in salicylic acid at the onset of systemic acquired resistance in cucumber. Science 1990, 250, 1004–1006. [Google Scholar] [CrossRef] [PubMed]
- Navarova, H.; Bernsdorff, F.; Doring, A.-C.; Zeier, J. Pipecolic Acid, an Endogenous Mediator of Defense Amplification and Priming, Is a Critical Regulator of Inducible Plant Immunity. Plant Cell 2012, 24, 5123–5141. [Google Scholar] [CrossRef] [PubMed]
- Lim, G.H.; Shine, M.B.; de Lorenzo, L.; Yu, K.; Cui, W.; Navarre, D.; Hunt, A.G.; Lee, J.Y.; Kachroo, A.; Kachroo, P. Plasmodesmata Localizing Proteins Regulate Transport and Signaling during Systemic Acquired Immunity in Plants. Cell Host Microbe 2016, 19, 541–549. [Google Scholar] [CrossRef] [PubMed]
- Yu, K.; Soares, J.; Mandal, M.K.; Wang, C.; Chanda, B.; Gifford, A.N.; Fowler, J.S.; Navarre, D.; Kachroo, A.; Kachroo, P. A feed-back regulatory loop between glycerol-3-phosphate and lipid transfer proteins DIR1 and AZI1 mediates azelaic acid-induced systemic immunity. Cell Rep. 2013, 3, 1266–1278. [Google Scholar] [CrossRef] [PubMed]
- Cecchini, N.M.; Steffes, K.; Schläppi, M.R.; Gifford, A.N.; Greenberg, J.T. Arabidopsis AZI1 family proteins mediate signal mobilization for systemic defence priming. Nat. Commun. 2015, 6, 7658. [Google Scholar] [CrossRef] [PubMed]
- Meuwly, P.; Molders, W.; Buchala, A.; Metraux, J.-P. Local and Systemic Biosynthesis of Salicylic Acid in lnfected Cucumber Plants. Plant Phisiology 1995, 109, 1107–1114. [Google Scholar] [CrossRef]
- Vernooij, B.; Friedrichya, L.; Reist, R.; Kolditzjawhar, R.; Ward, E.; Uknes, S.; Kessmann, H.; Ryals, J. Salicylic Acid Is Not the Translocated Signal Responsible for lnducing Systemic Acquired Resistance but Is Required in Signal Transduction. Plant Cell 1994, 6, 959–965. [Google Scholar] [CrossRef] [PubMed]
- Ding, P.; Rekhter, D.; Ding, Y.; Feussner, K.; Busta, L.; Haroth, S.; Xu, S.; Li, X.; Jetter, R.; Feussner, I.; et al. Characterization of a Pipecolic Acid Biosynthesis Pathway Required for Systemic Acquired Resistance. Plant Cell 2016, 28, 2603–2615. [Google Scholar] [CrossRef] [PubMed]
- Wittek, F.; Kanawati, B.; Wenig, M.; Hoffmann, T.; Franz-Oberdorf, K.; Schwab, W.; Schmitt-Kopplin, P.; Vlot, A.C. Folic acid induces salicylic acid-dependent immunity in Arabidopsis and enhances susceptibility to Alternaria brassicicola. Mol. Plant Pathol. 2015, 16, 616–622. [Google Scholar] [CrossRef] [PubMed]
- Song, G.C.; Choi, H.K.; Ryu, C.M. The folate precursor para-aminobenzoic acid elicits induced resistance against Cucumber mosaic virus and Xanthomonas axonopodis. Ann. Bot. 2013, 111, 925–934. [Google Scholar] [CrossRef] [PubMed]
- Balmer, D.; Planchamp, C.; Mauch-Mani, B. On the move: Induced resistance in monocots. J. Exp. Bot. 2013, 64, 1249–1261. [Google Scholar] [CrossRef] [PubMed]
- Sharma, R.; De Vleesschauwer, D.; Sharma, M.K.; Ronald, P.C. Recent advances in dissecting stress-regulatory crosstalk in rice. Mol. Plant 2013, 6, 250–260. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Yi, G.; Peng, X.; Huang, B.; Liu, E.; Zhang, J. Systemic acquired resistance in Cavendish banana induced by infection with an incompatible strain of Fusarium oxysporum f. sp. cubense. J. Plant Physiol. 2013, 170, 1039–1046. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Zhao, J.; Liu, P.; Xing, H.; Li, C.; Wei, G.; Kang, Z. Glycerol-3-phosphate metabolism in wheat contributes to systemic acquired resistance against Puccinia striiformis f. sp. tritici. PLoS ONE 2013, 8, e81756. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, S.M.; Liu, P.; Xue, Q.; Ji, C.; Qi, T.; Guo, J.; Guo, J.; Kang, Z. TaDIR1-2, a Wheat Ortholog of Lipid Transfer Protein AtDIR1 Contributes to Negative Regulation of Wheat Resistance against Puccinia striiformis f. sp. tritici. Front. Plant Sci. 2017, 8, 521. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, Y.; Liu, P.; Ding, Y.; Mu, X.; Liu, X.; Wang, X.; Zhao, M.; Huai, B.; Huang, L.; et al. TaRar1 Is Involved in Wheat Defense against Stripe Rust Pathogen Mediated by YrSu. Front. Plant Sci. 2017, 8, 156. [Google Scholar] [CrossRef] [PubMed]
- Balmer, D.; De Papajewski, D.V.; Planchamp, C.; Glauser, G.; Mauch-Mani, B. Induced resistance in maize is based on organ-specific defence responses. Plant J. 2013, 74, 213–225. [Google Scholar] [CrossRef] [PubMed]
- Morris, S.W.; Vernooij, B.; Titatarn, S.; Starrett, M.; Thomas, S.; Wiltse, C.C.; Frederiksen, R.A.; Bhandhufalck, A.; Hulbert, S.; Uknes, S. Induced Resistance Responses in Maize. Mol. Plant-Microbe Interact. 1998, 11, 643–658. [Google Scholar] [CrossRef] [PubMed]
- Görlach, J.; Volrath, S.; Knauf-Beiter, G.; Hengy, G.; Beckhove, U.; Kogel, K.; Oostendorp, M.; Staub, T.; Ward, E.; Kessmann, H.; et al. Benzothiadiazole, a Novel Class of Inducers of Systemic Acquired Resistance, Activates Gene Expression and Disease Resistance in Wheat. Plant Cell 1996, 8, 629–643. [Google Scholar] [CrossRef] [PubMed]
- Beßer, K.; Jarosch, B.; Langen, G.; Kogel, K.-H. Expression analysis of genes induced in barley after chemical activation reveals distinct disease resistance pathways. Mol. Plant Pathol. 2000, 1, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Dey, S.; Wenig, M.; Langen, G.; Sharma, S.; Kugler, K.G.; Knappe, C.; Hause, B.; Bichlmeier, M.; Babaeizad, V.; Imani, J.; et al. Bacteria-Triggered Systemic Immunity in Barley Is Associated with WRKY and ETHYLENE RESPONSIVE FACTORs But Not with Salicylic Acid. Plant Physiol. 2014, 166, 2133–2151. [Google Scholar] [CrossRef] [PubMed]
- Jansen, C.; Korell, M.; Eckey, C.; Biedenkopf, D.; Kogel, K.-H. Identification and transcriptional analysis of powdery mildew-induced barley genes. Plant Sci. 2005, 168, 373–380. [Google Scholar] [CrossRef]
- Thordal-Christensen, H.; Gregersen, P.L.; Collinge, D.B. The Barley/Blumeria (Syn. Erysiphe) Graminis Interaction. In Mechanisms of Resistance to Plant Diseases; Springer: Dordrecht, The Netherlands, 2000; pp. 77–100. [Google Scholar]
- Hückelhoven, R.; Panstruga, R. Cell biology of the plant-powdery mildew interaction. Curr. Opin. Plant Biol. 2011, 14, 738–746. [Google Scholar] [CrossRef] [PubMed]
- Dean, R.; Van Kan, J.A.L.; Pretorius, Z.A.; Hammond-Kosack, K.E.; Di Pietro, A.; Spanu, P.D.; Rudd, J.J.; Dickman, M.; Kahmann, R.; Ellis, J.; et al. The Top 10 fungal pathogens in molecular plant pathology. Mol. Plant Pathol. 2012, 13, 414–430. [Google Scholar] [CrossRef] [PubMed]
- Vallelian-Bindschedler, L.; Mösinger, E.; Métraux, J.P.; Schweizer, P. Structure, expression and localization of a germin-like protein in barley (Hordeum vulgare L.) that is insolubilized in stressed leaves. Plant Mol. Biol. 1998, 37, 297–308. [Google Scholar] [CrossRef] [PubMed]
- Hückelhoven, R.; Fodor, J.; Preis, C.; Kogel, K.-H. Hypersensitive Cell Death and Papilla Formation in Barley Attacked by the Powdery Mildew Fungus Are Associated with Hydrogen Peroxide but Not with Salicylic Acid Accumulation1. Plant Physiol. 1999, 119, 1251–1260. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.K.; Langen, G.; Hess, W.; Börner, T.; Hückelhoven, R.; Kogel, K.-H. The white barley mutant albostrians shows enhanced resistance to the biotroph Blumeria graminis f. sp. hordei. Mol. Plant-Microbe Interact. 2004, 17, 374–382. [Google Scholar] [CrossRef] [PubMed]
- Kogel, K.H.; Ortel, B.; Jarosch, B.; Atzorn, R.; Schiffer, R.; Wasternack, C. Resistance in barley against the powdery mildew fungus (Erysiphe graminis f. sp. hordei) is not associated with enhanced levels of endogenous jasmonates. Eur. J. Plant Pathol. 1995, 101, 319–332. [Google Scholar] [CrossRef]
- Cui, J.; Bahrami, A.K.; Pringle, E.G.; Hernandez-Guzman, G.; Bender, C.L.; Pierce, N.E.; Ausubel, F.M. Pseudomonas syringae manipulates systemic plant defenses against pathogens and herbivores. Proc. Natl. Acad. Sci. USA 2005, 102, 1791–1796. [Google Scholar] [CrossRef] [PubMed]
- Yasuda, M.; Ishikawa, A.; Jikumaru, Y.; Seki, M.; Umezawa, T.; Asami, T.; Maruyama-Nakashita, A.; Kudo, T.; Shinozaki, K.; Yoshida, S.; et al. Antagonistic Interaction between Systemic Acquired Resistance and the Abscisic Acid-Mediated Abiotic Stress Response in Arabidopsis. Plant Cell 2008, 20, 1678–1692. [Google Scholar] [CrossRef] [PubMed]
- Shigenaga, A.M.; Argueso, C.T. No hormone to rule them all: Interactions of plant hormones during the responses of plants to pathogens. Semin. Cell Dev. Biol. 2016, 56, 174–189. [Google Scholar] [CrossRef] [PubMed]
- Pieterse, C.M.J.; Van der Does, D.; Zamioudis, C.; Leon-Reyes, A.; Van Wees, S.C.M. Hormonal Modulation of Plant Immunity. Annu. Rev. Cell Dev. Biol. 2012, 28, 489–521. [Google Scholar] [CrossRef] [PubMed]
- Robert-Seilaniantz, A.; Grant, M.; Jones, J.D.G. Hormone Crosstalk in Plant Disease and Defense: More Than Just JASMONATE-SALICYLATE Antagonism. Annu. Rev. Phytopathol. 2011, 49, 317–343. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Z.; An, F.; Feng, Y.; Li, P.; Xue, L.; Mu, A.; Jiang, Z.; Kim, J.-M.; To, T.K.; Li, W.; et al. Derepression of ethylene-stabilized transcription factors (EIN3/EIL1) mediates jasmonate and ethylene signaling synergy in Arabidopsis. Proc. Natl. Acad. Sci. USA 2011, 108, 12539–12544. [Google Scholar] [CrossRef] [PubMed]
- Beckers, G.J.M.; Spoel, S.H. Fine-tuning plant defence signalling: Salicylate versus jasmonate. Plant Biol. 2006, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Spoel, S.H.; Johnson, J.S.; Dong, X. Regulation of tradeoffs between plant defenses against pathogens with different lifestyles. Proc. Natl. Acad. Sci. USA 2007, 104, 18842–18847. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Ellwood, S.R.; Oliver, R.P.; Friesen, T.L. Pyrenophora teres: Profile of an increasingly damaging barley pathogen. Mol. Plant Pathol. 2011, 12, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Vlot, A.C.; Liu, P.P.; Cameron, R.K.; Park, S.W.; Yang, Y.; Kumar, D.; Zhou, F.; Padukkavidana, T.; Gustafsson, C.; Pichersky, E.; et al. Identification of likely orthologs of tobacco salicylic acid-binding protein 2 and their role in systemic acquired resistance in Arabidopsis thaliana. Plant J. 2008, 56, 445–456. [Google Scholar] [CrossRef] [PubMed]
- Delventhal, R.; Rajaraman, J.; Stefanato, F.L.; Rehman, S.; Aghnoum, R.; McGrann, G.R.D.; Bolger, M.; Usadel, B.; Hedley, P.E.; Boyd, L.; et al. A comparative analysis of nonhost resistance across the two Triticeae crop species wheat and barley. BMC Plant Biol. 2017, 17, 232. [Google Scholar] [CrossRef] [PubMed]
- Nair, K.R.S.; Ellingboe, A.H. A method of controlled inoculations with conidiospores of Erysiphe graminis var. tritici. Phytopathology 1962, 52, 714. [Google Scholar]
- Foissner, I.; Wendehenne, D.; Langebartels, C.; Durner, J. In vivo imaging of elicitor-induced nitric oxide burst in tobacco. Plant J. 2000, 23, 817–824. [Google Scholar] [CrossRef] [PubMed]
- Laflamme, B.; Middleton, M.; Lo, T.; Desveaux, D.; Guttman, D.S. Image-Based Quantification of Plant Immunity and Disease. Mol. Plant-Microbe Interact. 2016, 29, MPMI-07-16-0129. [Google Scholar] [CrossRef] [PubMed]
- Kojima, H.; Urano, Y.; Kikuchi, K.; Higuchi, T.; Hirata, Y.; Nagano, T. Fluorescent indicators for imaging nitric oxide production. Angew. Chem. Int. Ed. 1999, 38, 3209–3212. [Google Scholar] [CrossRef]
- Prats, E.; Mur, L.A.J.; Sanderson, R.; Carver, T.L.W. Nitric oxide contributes both to papilla-based resistance and the hypersensitive response in barley attacked by Blumeria graminis f. sp. hordei. Mol. Plant Pathol. 2005, 6, 65–78. [Google Scholar] [CrossRef] [PubMed]
- Prats, E.; Carver, T.L.W.; Mur, L.A.J. Pathogen-derived nitric oxide influences formation of the appressorium infection structure in the phytopathogenic fungus Blumeria graminis. Res. Microbiol. 2008, 159, 476–480. [Google Scholar] [CrossRef] [PubMed]
- Lin, M.-R.; Edwards, H.H. Primary Penetration Process in Powdery Mildewed Barley Related to Host Cell Age, Cell Type, and Occurrence of Basic Staining Material. New Phytol. 1974, 73, 131–137. [Google Scholar] [CrossRef]
- Wendehenne, D.; Gao, Q.; Kachroo, A.; Kachroo, P. Free radical-mediated systemic immunity in plants. Curr. Opin. Plant Biol. 2014, 20, 127–134. [Google Scholar] [CrossRef] [PubMed]

© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lenk, M.; Wenig, M.; Mengel, F.; Häußler, F.; Vlot, A.C. Arabidopsis thaliana Immunity-Related Compounds Modulate Disease Susceptibility in Barley. Agronomy 2018, 8, 142. https://doi.org/10.3390/agronomy8080142
Lenk M, Wenig M, Mengel F, Häußler F, Vlot AC. Arabidopsis thaliana Immunity-Related Compounds Modulate Disease Susceptibility in Barley. Agronomy. 2018; 8(8):142. https://doi.org/10.3390/agronomy8080142
Chicago/Turabian StyleLenk, Miriam, Marion Wenig, Felicitas Mengel, Finni Häußler, and A. Corina Vlot. 2018. "Arabidopsis thaliana Immunity-Related Compounds Modulate Disease Susceptibility in Barley" Agronomy 8, no. 8: 142. https://doi.org/10.3390/agronomy8080142
APA StyleLenk, M., Wenig, M., Mengel, F., Häußler, F., & Vlot, A. C. (2018). Arabidopsis thaliana Immunity-Related Compounds Modulate Disease Susceptibility in Barley. Agronomy, 8(8), 142. https://doi.org/10.3390/agronomy8080142





