Mapping Quantitative Trait Loci for Agronomic Traits in Winter Wheat under Different Soil Moisture Levels
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Material
2.2. Field Trials
2.3. Phenotypic Data Analysis
2.4. Molecular Marker Genotyping
2.5. Map Construction and QTL Mapping
3. Results
3.1. Trait Means, Heritability Estimates, and Correlation Analysis
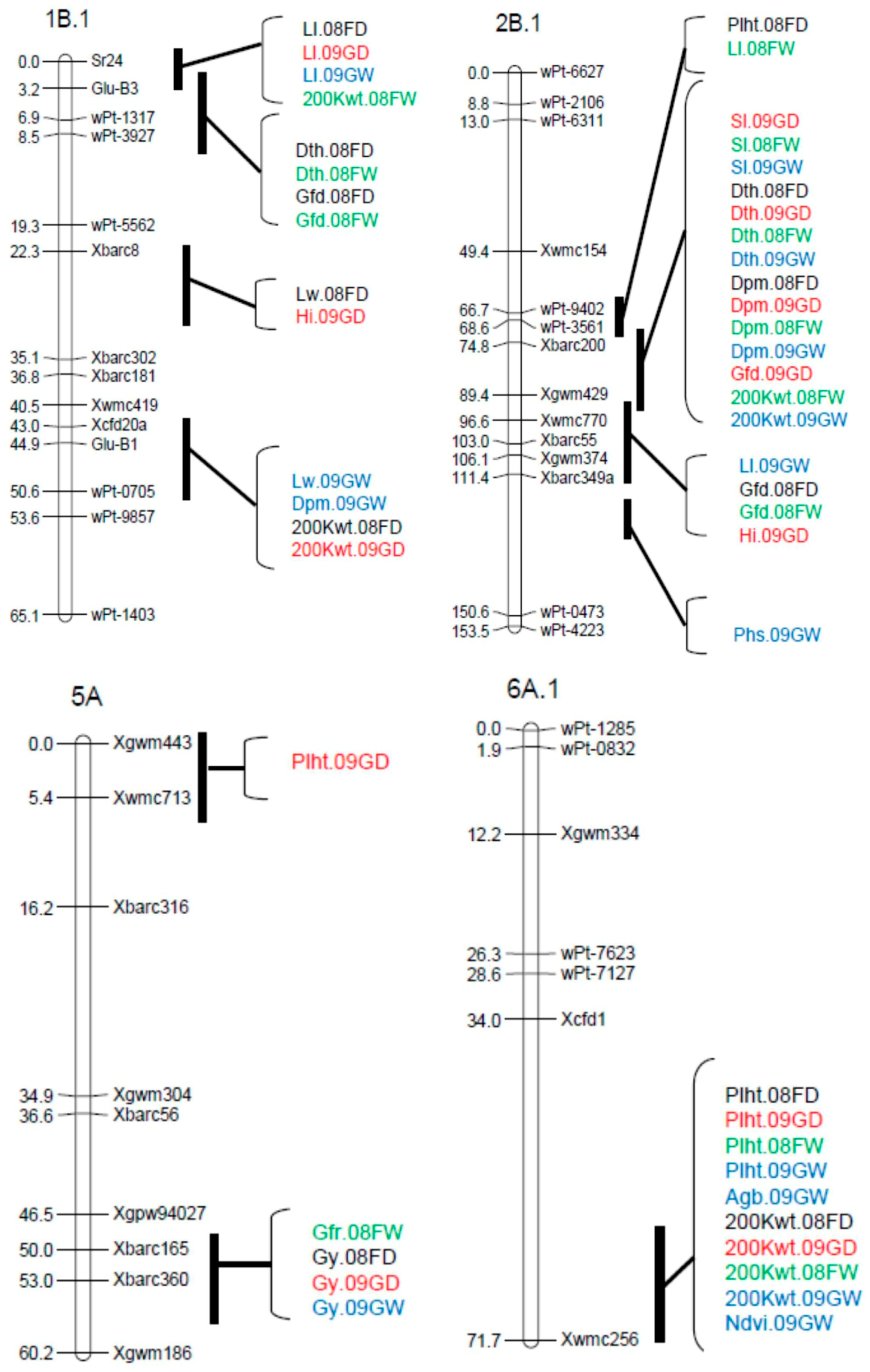
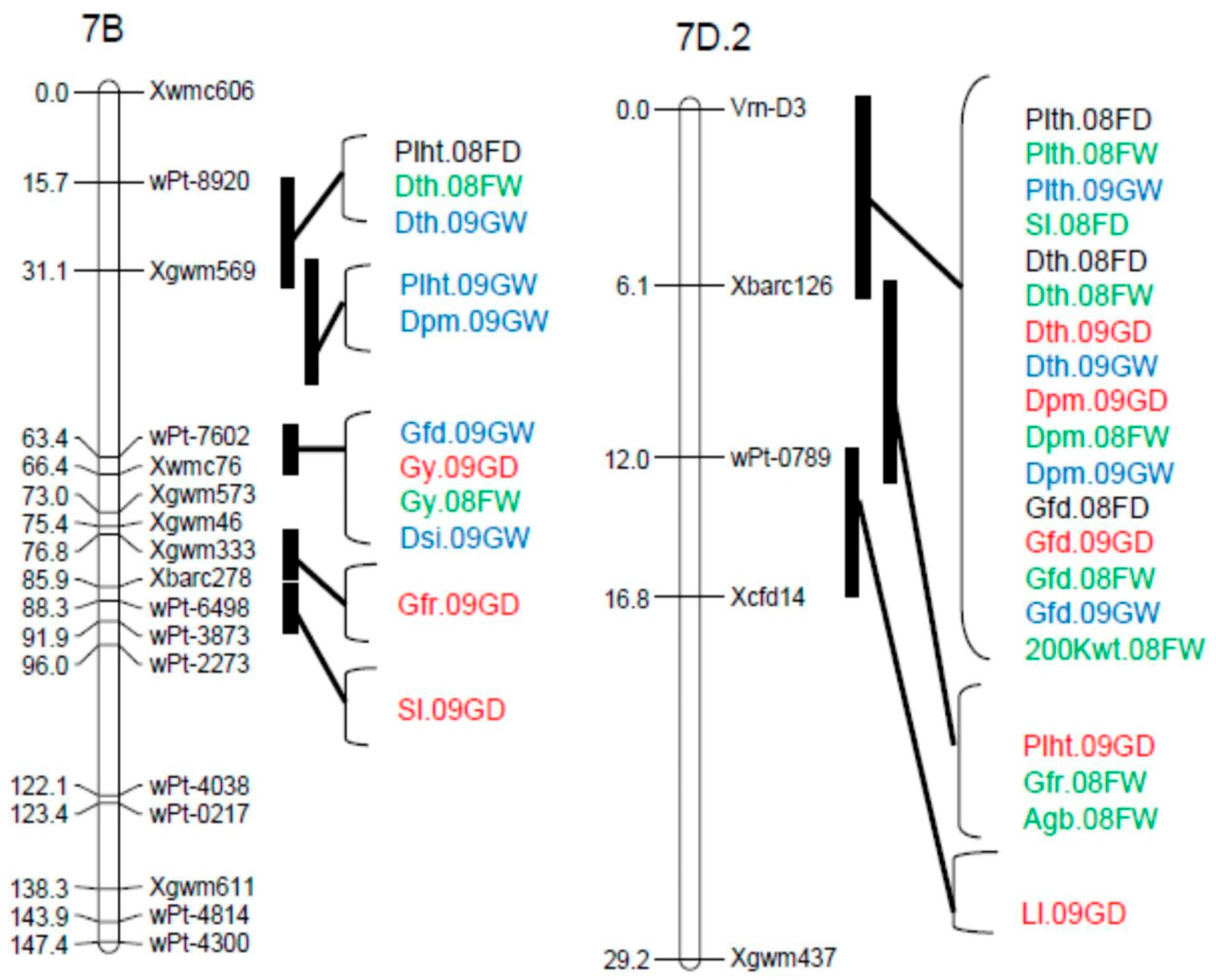
3.2. Marker Analysis and Map Construction
3.3. QTL Analysis
4. Discussion
4.1. Trait Means, Heritability Estimates, and Correlation Analysis
4.2. QTL Mapping
4.2.1. QTL Distribution
4.2.2. Phenological Parameters
4.2.3. Morphological Traits
4.2.4. Yield and Yield Components
4.2.5. Pre-Harvest Sprouting
4.2.6. Normalized Difference Vegetation Index
4.2.7. Drought Susceptibility Index
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Wheat. Wheat: Vital Grain of Civilization and Food Security; 2013 Annual Report; CGIAR Research Program on Wheat: Mexico City, Mexico, 2014. [Google Scholar]
- Weightman, R.M.; Millar, S.; Alava, J.; Foulkes, M.J.; Fish, L.; Snape, J.W. Effects of drought and the presence of the 1BL/1RS translocation on grain vitreosity, hardness and protein content in winter wheat. J. Cereal. Sci. 2008, 47, 457–468. [Google Scholar] [CrossRef]
- Peleg, Z.; Fahima, T.; Krugman, T.; Abbo, S.; Yakir, D.; Korol, A.B.; Saranga, Y. Genomic dissection of drought resistance in durum wheat x wild emmer wheat recombinant inbreed line population. Plant Environ. 2009, 32, 758–779. [Google Scholar] [CrossRef] [PubMed]
- Arumuganathan, K.; Earle, E.D. Nuclear DNA content of some important plant species. Plant Mol. Biol. Rep. 1991, 9, 208–218. [Google Scholar] [CrossRef]
- Doerge, R.W. Mapping and analysis of quantitative trait loci in experimental populations. Nat. Rev. Genet. 2002, 3, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Roder, M.S.; Korzun, V.; Wendehake, K.; Plaschke, J.; Tixier, M.H.; Leroy, P.; Ganal, M.W. A microsatellite map of wheat. Genetics 1998, 149, 2007–2023. [Google Scholar] [PubMed]
- Somers, D.J.; Isaac, P.; Edwards, K. A high-density microsatellite consensus map for bread wheat (Triticum aestivum L.). Theor. Appl. Genet. 2004, 109, 1105–1114. [Google Scholar] [CrossRef] [PubMed]
- McCartney, C.A.; Somers, D.J.; Humphreys, D.G.; Lukow, O.; Ames, N.; Noll, J.; Cloutier, S.; McCallum, B.D. Mapping quantitative trait loci controlling agronomic traits in the spring wheat cross RL4452 x ‘AC Domain’. Genome 2005, 48, 870–883. [Google Scholar] [CrossRef] [PubMed]
- Wenzl, P.; Carling, J.; Kudrna, D.; Jaccoud, D.; Huttner, E.; Kleinhofs, A.; Kilian, A. Diversit Arrays Technology (DArT) for whole-genome profiling of barley. Proc. Natl. Acad. Sci. USA 2004, 101, 9915–9920. [Google Scholar] [CrossRef] [PubMed]
- Crossa, J.; Burgueno, J.; Dreisigacker, S.; Vargas, M.; Herrera-Foessel, S.A.; Lillemo, M.; Singh, R.P.; Trethowan, R.; Warburton, M.; Franco, J.; et al. Association analysis of historical bread wheat germplasm using additive genetic covariance of relatives and population structure. Genetics 2007, 177, 1889–1913. [Google Scholar] [CrossRef] [PubMed]
- Griffiths, S.; Simmonds, J.; Leverington, M.; Wang, Y.; Fish, L.; Sayers, L.; Alibert, L.; Orford, S.; Wingen, L.; Herry, L.; et al. Meta-QTL analysis of the genetic control of ear emergence in elite European winter wheat germplasm. Theor. Appl. Genet. 2009, 119, 383–395. [Google Scholar] [CrossRef] [PubMed]
- Raman, R.; Raman, H.; Martin, P. Functional gene markers for polyphenol oxidase locus in bread wheat (Triticum aestivum L.). Mol. Breed. 2007, 19, 315–328. [Google Scholar] [CrossRef]
- Huang, X.Q.; Cloutier, S.; Lycar, L.; Radovanovic, N.; Humphreys, D.G.; Noll, J.S.; Somers, D.J.; Brown, P.D. Molecular detection of QTLs for agronomic and quality traits in a doubled haploid population derived from two Canadian wheats (Triticum aestivum L.). Theor. Appl. Genet. 2006, 113, 753–766. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.G.; Xu, S.S.; Friesen, T.L.; Faris, J.D. Whole genome mapping in a wheat doubled haploid population using SSRs and TRAPs and the identification of QTL for agronomic traits. Mol. Breed. 2008, 22, 251–266. [Google Scholar] [CrossRef]
- Lin, F.; Xue, S.L.; Tian, D.G.; Li, C.J.; Cao, Y.; Zhang, Z.Z.; Zhang, C.Q.; Ma, Z.Q. Mapping chromosomal regions affecting flowering time in a spring wheat RIL population. Euphytica 2008, 164, 769–777. [Google Scholar] [CrossRef]
- Galiba, G.; Quarrie, S.A.; Sutka, J.; Morgounov, A.; Snape, J.W. RFLP mapping of the vernalization (Vrn1) and frost-resistance (Fr1) genes on chromosome 5A of wheat. Theor. Appl. Genet. 1995, 90, 1174–1179. [Google Scholar] [CrossRef] [PubMed]
- Iwaki, K.; Nishida, J.; Yanagisawa, T.; Yoshida, H.; Kato, K. Genetic analysis of Vrn-B1 for vernalization requirement by using linked dCAPS markers in bread wheat (Triticum aestivum L.). Theor. Appl. Genet. 2002, 104, 571–576. [Google Scholar] [CrossRef] [PubMed]
- Nelson, J.C.; Sorrells, M.E.; VanDeynze, A.E.; Lu, Y.H.; Atkinson, M.; Bernard, M.; Leroy, P.; Faris, J.D.; Anderson, J.A. Molecular mapping of wheat-major genes and rearrangement in homologous group-4, group-5, and group-7. Genetics 1995, 141, 721–731. [Google Scholar] [PubMed]
- Yan, L.; Fu, D.; Li, C.; Blechl, A.; Tranquilli, G.; Bonafede, M.; Sanchez, A.; Valarik, M.; Yasuda, S.; Dubcovsky, J. The wheat and barley vernalization gene VRN3 is an orthologue of FT. Proc. Natl. Acad. Sci. USA 2006, 103, 19581–19586. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.X.; Hai, L.; Zhang, X.Y.; You, G.X.; Yan, C.S.; Xiao, S.H. QTL mapping for grain filling rate and yield-related traits in RILs of the Chinese winter wheat population Heshangmai X Yu8679. Theor. Appl. Genet. 2009, 118, 313–325. [Google Scholar] [CrossRef] [PubMed]
- Scarth, R.; Law, C.N. The location of the photoperiod gene, Ppd2 and an additional genetic-factor for ear-emergence time on chromosome-2B of wheat. Heredity 1983, 51, 607–619. [Google Scholar] [CrossRef]
- Hoogendoorn, J. A reciprocal F1 monosomic analysis of the genetic-control of time of ear emergence, number of leaves and number of spikelets in wheat (Triticum aestivum L.). Euphytica 1985, 34, 545–558. [Google Scholar] [CrossRef]
- Shah, M.M.; Gill, K.S.; Baenziger, P.S.; Yen, Y.; Kaeppler, S.M.; Ariyarathne, H.M. Molecular mapping of loci for agronomic traits on chromosome 3A of bread wheat. Crop Sci. 1999, 39, 1728–1732. [Google Scholar] [CrossRef]
- Araki, E.; Miura, H.; Sawada, S. Identification of genetic loci affecting amylose content and agronomic traits on chromosome 4A of wheat. Theor. Appl. Genet. 1999, 98, 977–984. [Google Scholar] [CrossRef]
- Lopes, M.; Reynolds, P.; McIntyre, L.; Mathews, L.; Jalal Kamali, R.; Mossad, M.; Feltaous, Y.; Tahir, A.; Chatrath, R.; Ogbonnaya, F. QTL for yield and associated traits in the Seri/Babax population grown across several environments in Mexico, in the West Asia, North Africa, and South Asia regions. Theor. Appl. Genet. 2013, 126, 971–984. [Google Scholar] [CrossRef] [PubMed]
- Borner, A.; Schumann, E.; Furste, A.; Coster, H.; Leithold, B.; Roder, M.S.; Weber, W.E. Mapping of quantitative trait loci determining agronomic important characters in hexaploid wheat (Triticum aestivum L.). Theor. Appl. Genet. 2002, 105, 921–936. [Google Scholar] [PubMed]
- Campbell, B.T.; Baenziger, P.S.; Gill, K.S.; Eskridge, K.M.; Budak, H.; Erayman, M.; Dweikat, I.; Yen, Y. Identification of QTLs and environmental interactions associated with agronomic traits on chromosome 3A of wheat. Crop Sci. 2003, 43, 1493–1505. [Google Scholar] [CrossRef]
- Groos, C.; Robert, N.; Bervas, E.; Charme, G. Genetic analysis of grain protein- content, grain yield and thousand-kernel weight in bread wheat. Theor. Appl. Genet. 2003, 106, 1032–1040. [Google Scholar] [CrossRef] [PubMed]
- Quarrie, S.A.; Steed, A.; Calestani, C.; Semikhodskii, A.; Lebreton, C.; Chinoy, C.; Steele, N.; Pljevljakusic, D.; Waterman, E.; Weyen, J.; et al. A high-density genetic map of hexaploid wheat (Triticum aestivum L.) from the cross Chinese Spring X SQ1 and its use to compare QTLs for grain yield across a range of environments. Theor. Appl. Genet. 2005, 110, 865–880. [Google Scholar] [CrossRef] [PubMed]
- Marza, F.; Bai, G.H.; Carver, B.F.; Zhou, W.C. Quantitative trait loci for yield and related traits in the wheat population Ning7840 x Clark. Theor. Appl. Genet. 2006, 112, 688–698. [Google Scholar] [CrossRef] [PubMed]
- Maccaferri, M.; Sanguineti, M.C.; Corneti, S.; Ortega, J.L.A.; Ben Salem, M.; Bort, J.; DeAmbrogio, E.; Del Moral, L.F.G.; Demontis, A.; El-Ahmed, A.; et al. Quantitative trait loci for grain yield and adaptation of durum wheat (Triticumdurum Desf.) across a wide range of water availability. Genetics 2008, 178, 489–511. [Google Scholar] [CrossRef] [PubMed]
- McIntyre, C.L.; Mathews, K.L.; Rattey, A.; Chapman, S.C.; Drenth, J.; Ghaderi, M.; Reynolds, M.; Shorter, R. Molecular detection of genomic regions associated with grain yield and yield-related components in an elite bread wheat cross evaluated under irrigated and rainfed conditions. Theor. Appl. Genet. 2010, 120, 527–541. [Google Scholar] [CrossRef] [PubMed]
- El-Feki, W.M.; Byrne, P.F.; Reid, S.D.; Haley, S.D. Registration of CO940610/‘Platte’ wheat doubled haploid mapping population. J. Plant Reg. 2015, 9, 419–423. [Google Scholar] [CrossRef]
- El-Feki, W.M.; Byrne, P.F.; Reid, S.D.; Lapitan, N.L.V.; Haley, S.D. Quantitative trait locus mapping for end-use quality traits in hard winter wheat under contrasting soil moisture levels. Crop Sci. 2013, 53, 1953–1967. [Google Scholar] [CrossRef]
- Haley, S.D.; Johnson, J.J.; Peairs, F.B.; Quick, J.S.; Stromberger, J.A.; Clayshulte, S.R.; Butler, J.D.; Rudolph, J.B.; Seabourn, B.W.; Bai, G.H.; et al. Registration of ‘Ripper’ Wheat. J. Plant Reg. 2007, 1, 1–6. [Google Scholar] [CrossRef]
- Fischer, R.A.; Maurer, R. Drought resistance in spring wheat cultivars. Part 1, grain yield response. Aust. J. Agric. Res. 1978, 29, 897–912. [Google Scholar] [CrossRef]
- Mares, D.; Mrva, K.; Cheong, J.; Williams, K.; Watson, B.; Storlie, E.; Sutherland, M.; Zou, Y. A QTL located on chromosome 4A associated with dormancy in white and red-grained wheats of diverse origin. Theor. Appl. Genet. 2005, 112, 1357–1364. [Google Scholar] [CrossRef] [PubMed]
- Aparicio, N.; Villegas, D.; Casadesus, J.; Araus, J.L.; Royo, C. Spectral vegetation indices as nondestructive tools for determining durum wheat yield. Agron. J. 2000, 92, 83–91. [Google Scholar] [CrossRef]
- Penuelas, J.; Isla, R.; Filella, I.; Araus, J.L. Visible and near-infrared reflectance assessment of salinity effects on barley. Crop Sci. 1997, 37, 198–202. [Google Scholar] [CrossRef]
- Butler, J.D.; Byrne, P.F.; Mohammadi, V.; Chapman, P.L.; Haley, S.D. Agronomic performance of Rht alleles in a spring wheat population across a range of moisture levels. Crop Sci. 2005, 45, 939–947. [Google Scholar] [CrossRef]
- Hallauer, A.R.; Carena, M.J.; Miranda Filho, J.B. Quantitative Genetics in Maize Breeding; Springer: New York, NY, USA, 2010. [Google Scholar]
- Knapp, S.J.; Stroup, W.W.; Ross, W.M. Exact confidence intervals for heritability on a progeny mean basis. Crop Sci. 1985, 25, 192–194. [Google Scholar] [CrossRef]
- Riede, C.R.; Anderson, J.A. Linkage of RFLP markers to an aluminum tolerance gene in wheat. Crop Sci. 1996, 36, 905–909. [Google Scholar] [CrossRef]
- Sun, D.J.; He, Z.H.; Xia, X.C.; Zhang, L.P.; Morris, C.F.; Appels, R.; Ma, W.J.; Wang, H. A novel STS marker for polyphenol oxidase activity in bread wheat. Mol. Breed. 2005, 16, 209–218. [Google Scholar] [CrossRef]
- Mago, R.; Bariana, H.S.; Dundas, I.S.; Spielmeyer, W.; Lawrence, G.J.; Pryor, A.J.; Ellis, J.G. Development of PCR markers for the selection of wheat stem rust resistance genes Sr24 and Sr26 in diverse wheat germplasm. Theor. Appl. Genet. 2005, 111, 496–504. [Google Scholar] [CrossRef] [PubMed]
- Akbari, M.; Wenzl, P.; Caig, V.; Carling, J.; Xia, L.; Yang, S.Y.; Uszynski, G.; Mohler, V.; Lehmensiek, A.; Kuchel, H.; et al. Diversity arrays technology DArT for high-throughput profiling of the 615 hexaploid wheat genome. Theor. Appl. Genet. 2006, 113, 1409–1420. [Google Scholar] [CrossRef] [PubMed]
- Voorrips, R.E. MapChart, Software for the graphical presentation of linkage maps and QTLs. J. Hered. 2002, 93, 77–78. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Basten, C.; Zeng, Z. Windows QTL Cartographer 2.5; North Carolina State University: Raleigh, NC, USA, 2010; Available online: http://statgen.ncsu.edu/qtlcart/WQTLCart.htm (accessed on 20 November 2017).
- Grogan, S.M.; Brown-Guedira, G.; Haley, S.D.; McMaster, G.S.; Reid, S.D.; Smith, J.; Byrne, P.F. Allelic variation in developmental genes and effects on winter wheat heading date in the U.S. Great Plains. PLoS ONE 2016, 11, E0152852. [Google Scholar] [CrossRef] [PubMed]
- El-Feki, W.M. Mapping Quantitative Trait Loci for Bread Making Quality and Agronomic Traits in Winter Wheat under Different Soil Moisture Levels. Ph.D. Thesis, Colorado State University, Fort Collins, CO, USA, 2010. [Google Scholar]
- Kumar, N.; Kulwal, P.L.; Gaur, A.; Tyagi, A.K.; Khurana, J.P.; Khurana, P.; Balyan, H.S.; Gupta, P.K. QTL analysis for grain weight in common wheat. Euphytica 2006, 151, 135–144. [Google Scholar] [CrossRef]
- Cuthbert, J.L.; Somers, D.J.; Brule-Babel, A.L.; Brown, P.D.; Crow, G.H. Molecular mapping of quantitative trait loci for yield and yield components in spring wheat (Triticum aestivum L.). Theor. Appl. Genet. 2008, 117, 595–608. [Google Scholar] [CrossRef] [PubMed]
- Hai, L.; Guo, H.J.; Wagner, C.; Xiao, S.H.; Friedt, W. Genomic regions for yield and yield parameters in Chinese winter wheat (Triticum aestivum L.) genotypes tested under varying environments correspond to QTL in widely different wheat materials. Plant Sci. 2008, 175, 226–232. [Google Scholar] [CrossRef]
- Quarrie, S.A.; Quarrie, S.P.; Radosevic, R.; Rancic, D.; Kaminska, A.; Barnes, J.D.; Leverington, M.; Ceoloni, C.; Dodig, D. Dissecting a wheat QTL for yield present in a range of environments, from the QTL to candidate genes. J. Exp. Bot. 2006, 57, 2627–2637. [Google Scholar] [CrossRef] [PubMed]
- Wiersma, J.J.; Busch, R.H.; Fulcher, G.G.; Hareland, G.A. Recurrent selection for kernel weight in spring wheat. Crop Sci. 2001, 41, 999–1005. [Google Scholar] [CrossRef]
- Spielmeyer, W.; Hyles, J.; Joaquim, P.; Azanza, F.; Bonnett, D.; Ellis, M.E.; Moore, C.; Richards, R.A. A QTL on chromosome 6A in bread wheat (Triticum aestivum) is associated with longer coleoptiles, greater seedling vigour and final plant height. Theor. Appl. Genet. 2007, 115, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Sourdille, P.; Tixier, M.H.; Charmet, G.; Gay, G.; Cadalen, T.; Bernard, S.; Bernard, M. Location of genes involved in ear compactness in wheat (Triticum aestivum) by means of molecular markers. Mol. Breed. 2000, 6, 247–255. [Google Scholar] [CrossRef]
- Sourdille, P.; Cadalen, T.; Guyomarch, H.; Snape, J.; Perretant, M.R.; Charmet, G.; Boeuf, C.; Bernard, M.; Bernard, M. An update of the Courtot × Chinese Spring intervarietal molecular marker linkage map for the QTL detection of agronomic traits in wheat. Theor. Appl. Genet. 2003, 106, 530–538. [Google Scholar] [CrossRef] [PubMed]
- Hanocq, E.; Niarquin, M.; Heumez, E.; Rousset, M.; Le Gouis, J. Detection and mapping of QTL for earliness components in a bread wheat recombinant inbred lines population. Theor. Appl. Genet. 2004, 110, 106–115. [Google Scholar] [CrossRef] [PubMed]
- Baga, M.; Fowler, D.B.; Chibbar, R.N. Identification of genomic regions determining the phenological development leading to floral transition in wheat (Triticum aestivum L.). J. Exp. Bot. 2009, 60, 3575–3585. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.P.; Tian, J.C.; Zhao, L.; Liu, B.; Chen, G.F. Detection of quantitative trait loci for heading date based on the doubled haploid progeny of two elite Chinese wheat cultivars. Genetica 2009, 135, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Kuchel, H.; Hollamby, G.; Langridge, P.; Williams, K.; Jefferies, S.P. Identification of genetic loci associated with ear-emergence in bread wheat. Theor. Appl. Genet. 2006, 113, 1103–1112. [Google Scholar] [CrossRef] [PubMed]
- Wurschum, T.; Langer, S.M.; Longin, C.F.H.; Tucker, M.R.; Leiser, W.L. A three-component system incorporating Ppd-D1, copy number variation at Ppd-B1, and numerous small-effect quantitative trait loci facilitates adaptation of heading time in winter wheat cultivars of worldwide origin. Plant Cell Environ. 2018, 41, 1407–1416. [Google Scholar] [CrossRef] [PubMed]
- Hanocq, E.; Laperche, A.; Jaminon, O.; Laine, A.L.; Le Gouis, J. Most significant genome regions involved in the control of earliness traits in bread wheat, as revealed by QTL meta-analysis. Theor. Appl. Genet. 2007, 114, 569–584. [Google Scholar] [CrossRef] [PubMed]
- Ellis, M.H.; Rebetzke, G.J.; Chandler, P.; Bonnett, D.; Spielmeyer, W.; Richards, R.A. The effect of mdifferent height reducing genes on the early growth of wheat. Funct. Plant Biol. 2004, 31, 583–589. [Google Scholar] [CrossRef]
- Ma, Z.Q.; Zhao, D.M.; Zhang, C.Q.; Zhang, Z.Z.; Xue, S.L.; Lin, F.; Kong, Z.X.; Tian, D.G.; Luo, Q.Y. Molecular genetic analysis of five spike-related traits in wheat using RIL and immortalized F-2 populations. Mol. Genet. Genom. 2007, 277, 31–42. [Google Scholar] [CrossRef] [PubMed]
- Huang, X.Q.; Coster, H.; Ganal, M.W.; Roder, M.S. Advanced backcross QTL analysis for the identification of quantitative trait loci alleles from wild relatives of wheat (Triticum aestivum L.). Theor. Appl. Genet. 2003, 106, 1379–1389. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.; Kulwal, P.L.; Balyan, H.S.; Gupta, P.K. QTL mapping for yield and yield contributing traits in two mapping populations of bread wheat. Mol. Breed. 2007, 19, 163–177. [Google Scholar] [CrossRef]
- Anderson, J.A.; Sorrells, M.E.; Tanksley, S.D. RFLP analysis of genomic regions associated with resistance to pre-harvest sprouting in wheat. Crop Sci. 1993, 33, 453–459. [Google Scholar] [CrossRef]
- Mares, D.J.; Mrva, K. Mapping quantitative trait loci associated with variation in grain dormancy in Australian wheat. Aust. J. Agric. Res. 2001, 52, 1257–1265. [Google Scholar] [CrossRef]
- Flintham, J.; Adlam, R.; Bassoi, M.; Holdsworth, M.; Gale, M. Mapping genes for resistance to sprouting damage in wheat. Euphytica 2002, 126, 39–45. [Google Scholar] [CrossRef]
- Ogbonnaya, F.C.; Imtiaz, M.; Ye, G.; Hearnden, P.R.; Hernandez, E.; Eastwood, R.F.; Van Ginkel, M.; Shorter, S.C.; Winchester, J.M. Genetic and QTL analyses of seed dormancy and preharvest sprouting resistance in the wheat germplasm CN10955. Theor. Appl. Genet. 2008, 116, 891–902. [Google Scholar] [CrossRef] [PubMed]
- Tan, M.K.; Sharp, P.J.; Lu, M.Q.; Howes, N. Genetics of grain dormancy in a white wheat. Aust. J. Agric. Res. 2006, 57, 1157–1165. [Google Scholar]
- Noda, K.; Matsuura, T.; Maekawa, M.; Taketa, S. Chromosomes responsible for sensitivity of embryo to abscisic acid and dormancy in wheat. Euphytica 2002, 123, 203–209. [Google Scholar] [CrossRef]
- Cabral, A.; Jordan, M.; McCartney, C.; You, F.; Humphreys, D.; MacLachlan, R.; Pozniak, C. Identification of candidate genes, regions and markers for pre-harvest sprouting resistance in wheat (Triticum aestivum L.). Plant Biol. 2014, 14, 340–352. [Google Scholar] [CrossRef] [PubMed]
- Bennett, D.; Reynolds, M.; Mullan, D.; Izanloo, A.; Kuchel, H.; Langridge, P.; Schnurbusch, T. Detection of two major grain yield QTL in bread wheat (Triticum aestivum L.) under heat, drought and high yield potential environments. Theor. Appl. Genet. 2012, 125, 1473–1485. [Google Scholar] [CrossRef] [PubMed]
- Edae, E.A.; Byrne, P.F.; Haley, S.D.; Lopes, M.S.; Reynolds, M.P. Genome-wide association mapping of yield and yield components of spring wheat under contrasting moisture regimes. Theor. Appl. Genet. 2014, 4, 791–807. [Google Scholar] [CrossRef] [PubMed]
- Pinto, R.S.; Reynolds, M.P.; Mathews, K.L. Heat and drought adaptive QTL in a wheat population designed to minimize confounding agronomic effects. Theor. Appl. Genet. 2010, 121, 1001–1021. [Google Scholar] [CrossRef] [PubMed]
- Sukumaran, S.S.; Dreisigacker, S.; Lopes, M.; Chavez, P.; Reynolds, M.P. Genome-wide association study for grain yield and related traits in an elite spring wheat population grown in temperate irrigated environments. Theor. Appl. Genet. 2015, 2, 353–363. [Google Scholar] [CrossRef] [PubMed]
- Araus, J.L.; Casadesus, J.; Bort, J. Recent tools for the screening of physiological traits determining yield. In Application of Physiology in Wheat Breeding; Reynolds, M.P., Ortiz-Monasterio, J.I., McNab, A., Eds.; CIMMYT: Mexico City, Mexico, 2001; pp. 59–77. [Google Scholar]
- Kirigwi, F.M.; Van Ginkel, M.; Brown-Guedira, G.; Gill, B.S.; Paulsen, G.M.; Fritz, A.K. Markers associated with a QTL for grain yield in wheat under drought. Mol. Breed. 2007, 20, 401–413. [Google Scholar] [CrossRef]
- Dao, H.Q.; Byrne, P.F.; Reid, S.D.; Haley, S.D. Validation of quantitative trait loci for grain quality-related traits in a winter wheat mapping population. Euphytica 2017, 213, 5. [Google Scholar] [CrossRef]
| Fort Collins 2007–2008 | Greeley 2008–2009 | H2 (90% CI) § | |||||||
|---|---|---|---|---|---|---|---|---|---|
| Full Irrigation (08FW) | Limited Irrigation (08FD) | Full Irrigation (09GW) | Limited Irrigation (09GD) | ||||||
| Trait † | Mean ± SE ‡ | Min–Max | Mean ± SE | Min–Max | Mean ± SE | Min–Max | Mean ± SE | Min–Max | |
| Plht (cm) | 79.4 ± 0.81 | 64.9–90.8 | 67.9 ± 0. 92 | 52.2–79.3 | 91.1 ± 0.72 | 74.9–104.3 | 74.6 ± 0.75 | 65.7–82.1 | 0.82 (0.78–0.85) |
| Ll (cm) | 13.9 ± 0.29 | 11.1–16.6 | 10.7 ± 0.39 | 9.4–12.3 | 17.3 ± 0.30 | 16.2–18.2 | 16.1 ± 0.23 | 14.3–17.5 | 0.60 (0.51–0.67) |
| Lw (cm) | 1.24 ± 0.01 | 1.09–1.67 | 1.08 ± 0.02 | 0.93–1.27 | 1.42 ± 0.02 | 1.32–1.53 | 1.29 ± 0.03 | 1.12–1.44 | 0.74 (0.68–0.79) |
| Sl (cm) | 8.6 ± 0.08 | 7.0–10.4 | 8.6 ± 0.10 | 7.2–9.8 | 9.4 ± 0.08 | 8.1–11.2 | 9.7 ± 0.13 | 8.7–10.8 | 0.84 (0.81–0.87) |
| Dth (d) | 156.3 ± 0.31 | 151.0–164.7 | 152.5 ± 0.16 | 149.5–157.9 | 144.7 ± 0.23 | 139.5–154.5 | 143.2 ± 0.19 | 138.7–152.4 | 0.96 (0.96–0.98) |
| Dpm (d) | 191.2 ± 0.30 | 189.5–195.5 | 186.7 ± 0.50 | 185.3–189.0 | 184.2 ± 0.38 | 180.1–190.9 | 181.5 ± 0.31 | 178.9–189.9 | 0.78 (0.61–0.79) |
| Gfd (d) | 34.8 ± 0.32 | 30.9–38.6 | 33.9 ± 0.38 | 31.1–37.1 | 39.4 ± 0.35 | 37.4–41.7 | 38.2 ± 0.22 | 36.3–41.3 | 0.84 (0.81–0.89) |
| Gfr (g d−1) | 4.39 ± 0.09 | 3.44–5.17 | 3.20 ± 0.20 | 2.66–3.81 | 4.25 ± 0.09 | 2.79–4.85 | 3.51 ± 0.10 | 1.90–4.37 | 0.60 (0.51–0.67) |
| Agb (g) | 366.3 ± 9.73 | 343.6–690.6 | 296.9 ± 22.7 | 278.3–321.8 | 463.5 ± 12.6 | 394.6–507.0 | 401.5 ± 13.0 | 319.3–470.9 | 0.43 (0.29–0.53) |
| Hi | 0.42 ± 0.01 | 0.39–0.48 | 0.37 ± 0.01 | 0.31–0.44 | 0.36 ± 0.01 | 0.19–0.43 | 0.34 ± 0.01 | 0.18–0.46 | 0.62 (0.54–0.69) |
| 200 Kwt (g) | 7.13 ± 0.08 | 5.31–10.53 | 7.24 ± 0.08 | 5.14–9.28 | 7.30 ± 0.09 | 4.60–8.44 | 7.22 ± 0.08 | 5.12–8.43 | 0.89 (0.88–0.92) |
| Gy (kg ha −1) | 3719.7 ± 42.1 | 2091.9–4609.6 | 2923.9 ± 40.82 | 1571.6–3832.9 | 3760.9 ± 34.2 | 837.1–4676.6 | 3056.9 ± 33.3 | 823.9–4040.1 | 0.63 (0.57–0.68) |
| Phs | 0.65 ± 0.01 | 0.31–0.81 | -------- | ------- | 0.46 ± 0.02 | 0.33–0.56 | -------- | ------- | 0.29 (0.14–0.42) |
| NDVI | -------- | -------- | -------- | ------- | 0.71 ± 0.01 | 0.66–0.73 | 0.71 ± 0.02 | 0.67–0.73 | 0.29 (0.13–0.42) |
| Peak Position | |||||||
|---|---|---|---|---|---|---|---|
| Trait † | QTL ‡ | Environment § | Marker Interval ¶ | cM | LOD | A # | % R2 †† |
| Plht | QPlht.cob-2B.1 | 08FD | wPt-9402–Xbarc200 | 72.6 | 3.86 | −1.12 | 6.3 |
| QPlht.cob-3A | 08FW, 09GW, 08FD, 09GD | Xwmc50–Xbarc356 | 72.2–76.2 | 3.03–6.79 | −1.59–−0.7 | 11.5–15.3 | |
| QPlht.cob-4D.1 | 08FD | Xwmc720–wPt-0941 | 16.1 | 5.02 | −1.23 | 7.53 | |
| QPlht.cob-5A | 09GD | Xgwm443-Xwmc713 | 6.4 | 4.21 | −0.85 | 7.7 | |
| QPlht.cob-5B.1a | 08FW, 09GW, 09GD | wPt-1475–Xgdm62 | 52.5–79.0 | 2.67–4.83 | −1.37–−0.69 | 4.9–7.8 | |
| QPlht.cob-5B.1b | 08FD | Xgdm62–wPt-3661 | 89.5 | 5.21 | −1.28 | 8.18 | |
| QPlht.cob-6A.1 | 08FW, 09GW, 08FD, 09GD | Xcfd-1–Xwmc256 | 61.0–71.0 | 3.94–14.02 | −2.49–−0.94 | 9.4–17.3 | |
| QPlht.cob-7B | 08FD, 09GW | wPt-8920–wPt-7602 | 29.7–44.1 | 3.64–4.13 | −1.18–−1.15 | 6.09–7.72 | |
| QPlht.cob-7D.2 | 08FW, 09GW, 08FD, 09GD | Vrn-D3–wPt-0789 | 0.0–12.0 | 6.05–12.26 | 1.91–2.26 | 11.0–20.4 | |
| Ll | QLl.cob-1B.1 | 09GW, 08FD, 09GD | Sr24-Glu-B3 | 1.0–2.0 | 3.57–5.64 | −0.15–−0.14 | 7.3–11.5 |
| QLl.cob-2B.1a | 08FW | wPt-3561–Xbarc200 | 78.8 | 4.81 | −0.14 | 11.3 | |
| QLl.cob-2B.1b | 09GW | Xbarc55–Xgwm374 | 106.0 | 2.77 | −0.12 | 5.1 | |
| QLl.cob-3A | 09GW | Xwmc11-wPt-1688 | 19.3 | 2.96 | 0.13 | 6.0 | |
| QLl.cob-6B.1 | 08FW, 08FD | wPt-5596–wPt-2587 | 21.9–28.5 | 3.57–4.03 | 0.16–0.25 | 7.7–8.7 | |
| QLl.cob-7D.2 | 09GD | wPt-0789–Xcfd14 | 16.8 | 3.22 | −0.19 | 6.3 | |
| Lw | QLw.cob-1B.1 | 09GW, 08FD | Xbarc8–Glu-B1 | 29.3–45.9 | 4.43–4.65 | −0.02 | 9.5–10.2 |
| QLw.cob-2D.2 | 09GW | Xgdm35–Xgwm261 | 7.3 | 5.16 | −0.02 | 10.7 | |
| QLw.cob-4A.1 | 08FD | Xgwm610–Xwmc48 | 8.4 | 3.37 | 0.02 | 6.8 | |
| Sl | QSl.cob-1A.1 | 08FW, 09GW, 08FD, 09GD | Xbarc83-Glu-A1 | 85.4–96.4 | 2.89–8.34 | 0.11–0.23 | 5.9–15.2 |
| QSl.cob-1D | 08FW | Xbarc169-Glu-D1 | 87.9 | 2.71 | 0.12 | 4.4 | |
| QSl.cob-2B.1 | 08FW, 09GW, 09GD | Xbarc200–Xwmc770 | 82.8–93.4 | 2.97–7.42 | −0.22–−0.14 | 6.3–13.5 | |
| QSl.cob-3B.2 | 08FW | Xgwm533–Xgwm493 | 2.0 | 2.79 | −0.12 | 4.2 | |
| QSl.cob-3D.1 | 09GW | Xgwm314–Xgwm341 | 6.0 | 4.68 | −0.19 | 10.6 | |
| QSl.cob-4D.2 | 09GD | Xwmc622–Xcfd54a | 0.0 | 3.25 | 0.10 | 5.6 | |
| QSl.cob-6B.1 | 08FW, 09GD | Xwmc397–Xbarc198 | 55.5 | 3.0–4.37 | 0.11–0.13 | 4.9–7.7 | |
| QSl.cob-7A.2 | 08FW, 09GW | Xgwm60–wPt-3393 | 4.0–28.4 | 3.06–6.31 | 0.12–0.21 | 4.6–13.4 | |
| QSl.cob-7B | 09GD | Xbarc278–wPt-3873 | 88.3 | 3.24 | 0.10 | 5.5 | |
| QSl.cob-7D.2 | 08FD | Vrn-D3–Xbarc126 | 4.0 | 2.95 | 0.13 | 4.8 | |
| Dth | QDth.cob-1B.1 | 08FW, O8FD | Glu-B3–wPt-3927 | 4.2–8.5 | 2.76–4.55 | 0.36–0.57 | 4.3–5.3 |
| QDth.cob-2B.1 | 08FW, 08FD, 09GW, 09GD | Xbarc200–Xgwm429 | 76.8–83.4 | 13.79–17.16 | −1.17–−0.77 | 20.0–23.0 | |
| QDth.cob-3A | 08FW, 08FD, 09GD | Xwmc532–Xwmc50 | 46.2–55.2 | 2.72–3.28 | −0.52–−0.43 | 4.3–7.8 | |
| QDth.cob-7A.1 | 09GD | Xgwm635–Xbarc70 | 6.9 | 3.2 | 0.39 | 3.4 | |
| QDth.cob-7B | 08FW, 09GW | wPt-8920–Xgwm569 | 31.1 | 2.89–3.46 | −0.51–−0.46 | 3.6–4.5 | |
| QDth.cob-7D.2 | 08FW, 08FD, 09GW, 09GD | Vrn-D3–Xbarc126 | 0.0-6.1 | 13.78–25.88 | 0.87–1.43 | 25.9–38.8 | |
| Dpm | QDpm.cob-1B.1 | 09GW | Xbarc181–Xcfd20a | 40.5 | 3.55 | −0.39 | 5.3 |
| QDpm.cob-2B.1 | 08FW, 08FD, 09GW, 09GD | Xbarc200-Xgwm429 | 77.8–81.8 | 7.27–8.87 | −0.64–−0.26 | 14.3–17.5 | |
| QDpm.cob-3A | 08FD | Xwmc532–Xwmc50 | 59.2 | 3.71 | −0.21 | 9.9 | |
| QDpm.cob-7A.2 | 08FD | Xgwm60–Xbarc219 | 2.0 | 6.22 | 0.46 | 13.6 | |
| QDpm.cob-7B | 09GW | Xgwm569–wPt-7602 | 44.1 | 4.18 | −0.51 | 10.9 | |
| QDpm.cob-7D.2 | 08FW, 09GW, 09GD | Vrn-D3-Xbarc126 | 0.0–2.0 | 2.68–8.29 | 0.14–0.58 | 4.7–14.7 | |
| Gfd | QGfd.cob-1B.1 | 08FD, 08FW | Sr24-Glu-B3 | 4.2 | 3.99–5.0 | −0.38–−0.31 | 4.9–6.8 |
| QGfd.cob-2B.1 | 08FW, 08FD, 09GD | wPt-3561-Xbarc55 | 73.6–99.6 | 6.55–12.16 | 0.36–0.62 | 11.4–18.5 | |
| QGfd.cob-3A | 08FW | Xwmc50–Xgwm674 | 66.2 | 2.93 | 0.34 | 4.1 | |
| QGfd.cob-7A.2 | 08FW, 09GD | Xgwm60–wPt-1928 | 5.9–8.6 | 3.46–3.57 | −0.35–−0.29 | 4.1–5.6 | |
| QGfd.cob-7B | 09GW | wPt-7602–Xwmc76 | 68.4 | 2.86 | −0.19 | 5.3 | |
| QGfd.cob-7D.2 | 08FW, 08FD, 09GW, 09GD | Vrn-D3-Xbarc126 | 1.0–2.0 | 8.77–22.14 | −0.99–−0.37 | 17.1–35.3 | |
| Gfr | QGfr.cob-2D.2 | 09GD | wPt-4413-wPt-2544 | 53.3 | 4.36 | −0.13 | 9.5 |
| QGfr.cob-5A | 08FW | Xbarc165–Xbarc360 | 53.0 | 4.19 | 0.08 | 8.0 | |
| QGfr.cob-5B.1 | 09GD | Xbarc74–Xgwm540 | 59.6 | 3.44 | −0.11 | 6.6 | |
| QGfr.cob-7B | 09GD | Xgwm333–Xbarc278 | 82.8 | 3.79 | −0.12 | 8.4 | |
| QGfr.cob-7D.2 | 08FW | Xbarc126-wPt-0789 | 11.1 | 5.13 | 0.09 | 10.3 | |
| Agb | QAgb.cob-2D.2 | 08FW,09GD | wPt-0298-wPt-4413 | 33.7–52.3 | 3.49–3.65 | −7.79–−2.78 | 8.3–9.7 |
| QAgb.cob-4D.2 | 08FD | Xwmc622–Xcfd54a | 0.0 | 3.41 | −2.16 | 6.9 | |
| QAgb.cob-5B.1 | 09GD | Xgwm499–Xgdm62 | 84.0 | 3.0 | −6.8 | 6.1 | |
| QAgb.cob-6A.1 | 09GW | Xcfd1–Xwmc256 | 71.0 | 3.39 | −4.54 | 7.4 | |
| QAgb.cob-7D.2 | 08FW | Xbarc126-wPt-0789 | 12.0 | 2.88 | 2.16 | 5.9 | |
| Hi | QHi.cob-1B.1 | 09GD | wPt-5562–Xbarc8 | 25.3 | 2.75 | −0.01 | 6.8 |
| QHi.cob-2B.1 | 09GD | Xbarc55–Xbarc349a | 110.1 | 4.29 | 0.01 | 9.1 | |
| QHi.cob-6B.1 | 08FW, 08FD | wPt-2218–Xwmc397 | 28.5–52.3 | 2.89–4.74 | 0.01 | 6.4–10.4 | |
| 200 Kwt | Q200Kwt.cob-1A.1 | 09GW, 09GD | wPt-3904-wPt-9757 | 73.4–85.4 | 3.78–4.32 | 0.14–0.17 | 7.7–8.1 |
| Q200Kwt.cob-1B.1a | 08FW | Sr24-Glu-B3 | 1.0 | 4.56 | −0.16 | 6.8 | |
| Q200Kwt.cob-1B.1b | 08FD, 09GD | Xcfd20a-wPt-0705 | 46.9–50.6 | 4.85–7.44 | −0.19–−0.15 | 10.3–12.3 | |
| Q200Kwt.cob-2B.1 | 08FW, 09GW | wPt-3561–Xwmc770 | 79.8–86.8 | 6.26–7.81 | 0.21–0.24 | 12.0–14.5 | |
| Q200Kwt.cob-2D.1 | 08FD | Xbarc95–Xgdm35 | 3.6 | 2.79 | −0.11 | 4.3 | |
| Q200Kwt.cob-2D.2 | 08FW, 08FD | wPt-0298-wPt-4413 | 29.7–39.7 | 2.87–3.91 | −0.18–−0.13 | 5.5–7.7 | |
| Q200Kwt.cob-3B.1 | 08FD, 09GD | Xwmc231-Xgwm285 | 10.8–12.8 | 4.88–6.31 | 0.16–0.18 | 9.9–10.1 | |
| Q200Kwt.cob-6A.1 | 08FW, 08FD, 09GW, 09GD | Xcfd1-Xwmc256 | 69.0–71.0 | 3.60–7.59 | −0.23–−0.13 | 7.7–14.5 | |
| Q200Kwt.cob-7D.2 | 08FW | Vrn-D3-Xbarc126 | 0.0 | 5.95 | −0.21 | 10.1 | |
| Gy | QGy.cob-2D.2 | 08FD | wPt-4413-wPt-2544 | 52.3 | 4.04 | −107.83 | 10.1 |
| QGy.cob-5A | 08FD, 09GW, 09GD | Xbarc165–Xbarc360 | 51.0–53.0 | 2.98–5.35 | 84.27–159.6 | 6.2–11.6 | |
| QGy.cob-5B.1 | 09GD | Xbarc4–Xgwm540 | 59.6 | 7.33 | −185.16 | 13.7 | |
| QGy-cob-7B | 08FW, 09GD | wPt-7602–Xwmc76 | 59.1–65.4 | 2.70–4.96 | −102.9–−162.9 | 6.9–10.6 | |
| Phs | QPhs.cob-2B.1 | 09GW | Xgwm374–Xbarc349a | 121.4 | 3.4 | 0.01 | 9.6 |
| QPhs.cob-3D.1 | 08FW | Xgwm341–Xbarc125 | 24.9 | 2.76 | 0.02 | 5.8 | |
| QPhs.cob-4A.1 | 08FW | Xwmc48–Xwmc420 | 11.6 | 5.12 | 0.03 | 10.4 | |
| QPhs.cob-4D.1 | 08FW | wPt-0941-wPt-5809 | 22.1 | 4.08 | 0.03 | 10.3 | |
| NDVI | QNDVI.cob-4B.1 | GD09 | wPt-0391–Xcfd54b | 41.2 | 2.69 | 0.01 | 5.9 |
| QNDVI.cob-6A.1 | GW09 | Xcfd1–Xwmc256 | 70.0 | 3.44 | −0.01 | 7.6 | |
| Dsi | QDsi.cob-5B.1 | G09 | Xbarc216–Xgwm540 | 59.6 | 6.40 | 0.18 | 11.9 |
| QDsi.cob-7B | G09 | wPt-7602–Xgwm573 | 67.4 | 6.02 | 0.18 | 11.5 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
El-Feki, W.M.; Byrne, P.F.; Reid, S.D.; Haley, S.D. Mapping Quantitative Trait Loci for Agronomic Traits in Winter Wheat under Different Soil Moisture Levels. Agronomy 2018, 8, 133. https://doi.org/10.3390/agronomy8080133
El-Feki WM, Byrne PF, Reid SD, Haley SD. Mapping Quantitative Trait Loci for Agronomic Traits in Winter Wheat under Different Soil Moisture Levels. Agronomy. 2018; 8(8):133. https://doi.org/10.3390/agronomy8080133
Chicago/Turabian StyleEl-Feki, Walid M., Patrick F. Byrne, Scott D. Reid, and Scott D. Haley. 2018. "Mapping Quantitative Trait Loci for Agronomic Traits in Winter Wheat under Different Soil Moisture Levels" Agronomy 8, no. 8: 133. https://doi.org/10.3390/agronomy8080133
APA StyleEl-Feki, W. M., Byrne, P. F., Reid, S. D., & Haley, S. D. (2018). Mapping Quantitative Trait Loci for Agronomic Traits in Winter Wheat under Different Soil Moisture Levels. Agronomy, 8(8), 133. https://doi.org/10.3390/agronomy8080133





