Variation in Leaf Morphology and Agronomic Attributes of a Naturalized Population of Medicago polymorpha L. (Burr Medic) from New South Wales, Australia, and Relationships with Climate and Soil Characteristics
Abstract
1. Introduction
2. Materials and Methods
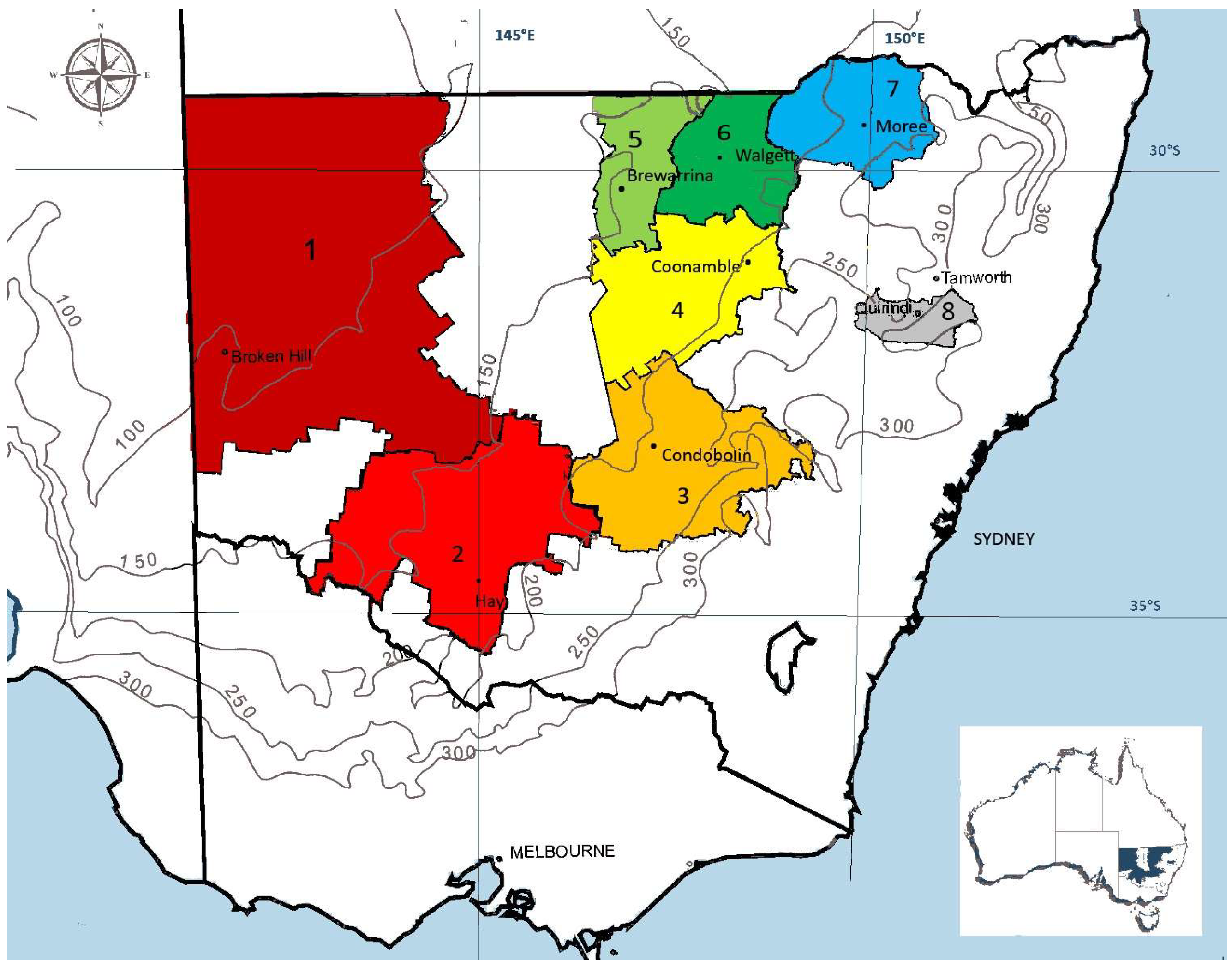
2.1. Germplasm Collection
2.2. Characterization of Germplasm
2.3. Statistical Analysis
3. Results
3.1. Plant Attributes—Days to Flower, Plant Vigor, and Pod Types
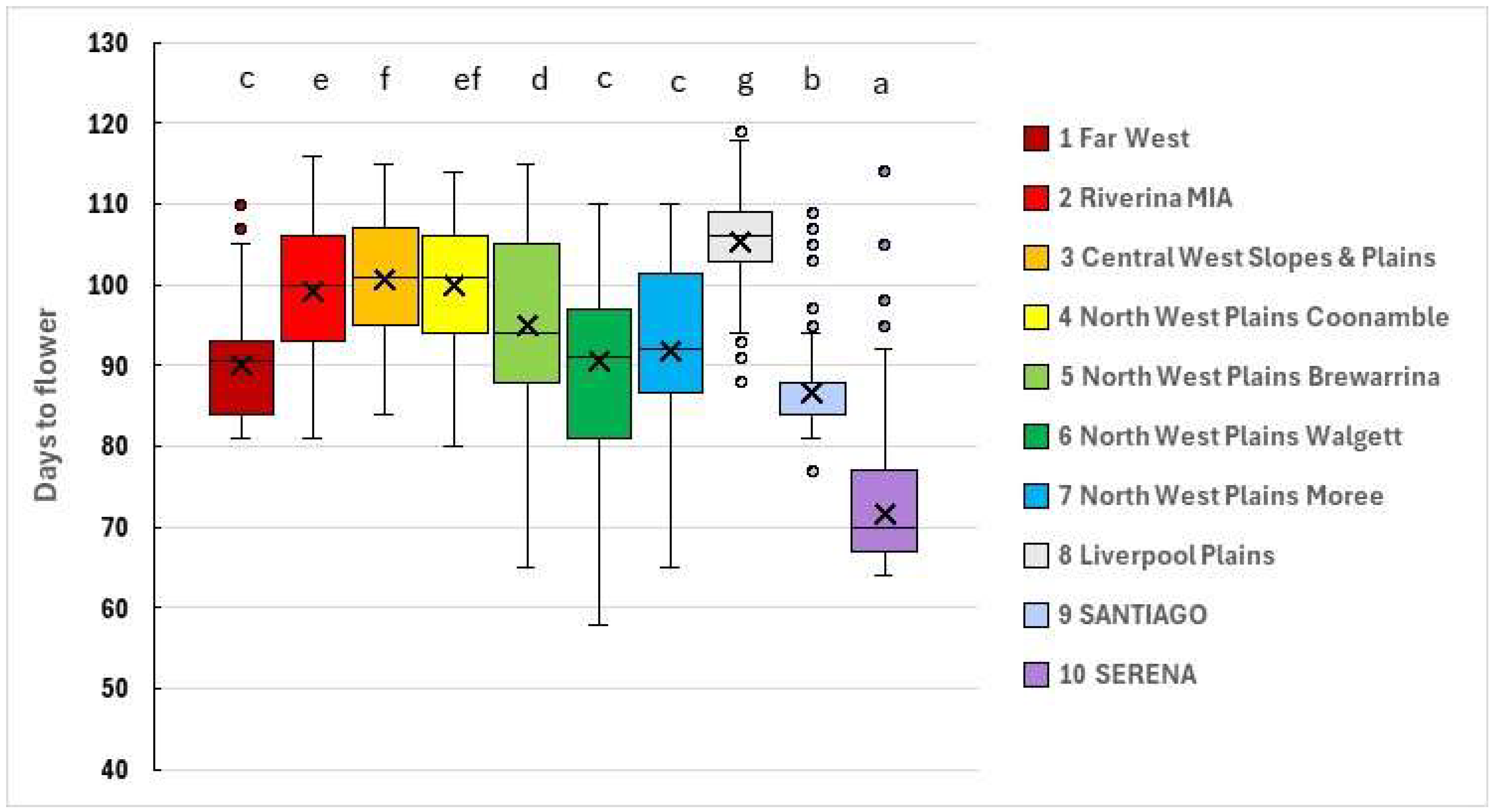
3.1.1. Days to Flower
3.1.2. Plant Vigor, Pod Coils, and Pod Spines
3.2. Correlation Analysis of Plant Attributes of Accessions with Site Characteristics
- Leaflets with no marks and slightly serrate margins on both juvenile (attribute LEAF42_1a) and adult (LEAF98_1a) plants were correlated with latitude (juvenile plants positively and adult plants negatively) and negatively correlated with minimum temperature, thus indicating a strong occurrence as adult (but not juvenile) plants among accessions collected from warmer, northern locations. This leaflet attribute was also correlated positively with soil exchangeable sodium (juvenile plants) and soil electrical conductivity (adult plants), but negatively with calcium (juvenile plants), suggesting naturalization on some sodic and slightly saline soils.
- Leaflets without marks but with distinctly dentate margins were positively correlated with winter rainfall, longitude, and soil exchangeable calcium as both juvenile (LEAF42_2) and adult plants (LEAF98_2). In juvenile plants, this attribute was also negatively correlated with latitude, and positively with minimum growing season temperature and soil exchangeable magnesium. In adult plants, this attribute was also positively correlated with annual rainfall. Plants with this attribute thus occurred more frequently in accessions from more mesic areas in the east of the collection zone.
- Leaflets with faint to obvious brown-purple proximal midrib coloration with markings more than 2 mm long with mainly serrate margins on adult plants (LEAF98_1bd) were positively correlated with altitude, longitude, and winter rainfall. These leaflet attributes were positively correlated with cation exchange capacity, soil organic C and total N. They were negatively correlated with maximum growing season temperature. Thus, plants with these markings occur mainly in fertile, mesic locations at higher altitude with lower growing season maximum temperatures.
- Leaflets with two proximal squarish smudges on either side of the midrib and slightly serrate margins on both juvenile (LEAF42_1c) and adult plants (LEAF98_1c), were positively correlated with longitude and, as adult plants, were also correlated positively with soil clay content and negatively with soil pH, indicating a strong occurrence in eastern collection regions on soils of higher clay content but with lower pH.
- Leaflets with a dark proximal inverted Y mark with slightly serrate margins as juvenile plants only (LEAF42_1d) were positively correlated with soil exchangeable calcium and were widely distributed across the collection zone.
- Leaflets with a dark or light proximal blotch with a dark band across the lower leaf on both juvenile (LEAF42_3_4) and adult (LEAF98_3_4) plants, which were mainly grey in color and with slightly serrate margins, were negatively correlated with annual rainfall, thus tending to occur in drier environments.
- Adult plants with grey colored leaflets (LEAF98 GL) were positively correlated with altitude, and those with lighter grey leaflets (LEAF98 WO), were positively correlated with soil exchangeable sodium, and negatively with annual average rainfall and soil organic C. This implies a separation in which plants with grey leaflets were strongly represented in higher altitude locations such as the Liverpool Plains, while those with light grey leaflets occurred on more sodic soils of lower fertility in drier environments.
3.3. Discriminant Analysis
4. Discussion
4.1. Time (Days) to Flower
4.2. Plant Vigor
4.3. Phenotypic Attributes—Leaflet and Pod Attributes
- Green leaflets with slightly dentate (serrate) margins and a dark, proximal inverted Y mark occurred widely on soils with high exchangeable Ca.
- Green leaflets with no leaf marks and strongly dentate margins occurred most frequently in wetter environments.
- Green leaflets with a smudge-type leaflet mark and serrate margins occurred on high clay soils of lower pH in the mesic, relatively cool, eastern Liverpool Plains region.
- Green leaflets with a brown-purple proximal midrib coloration and serrate margins occurred in cooler, higher altitude, mesic locations on fertile soils.
- Grey leaflets with serrate margins were strongly represented in higher altitude locations, particularly the Liverpool Plains.
- Green leaflets with no mark and serrate margins on adult plants occurred widely across warmer, northern regions on slightly saline soils
- Mainly grey leaflets, with proximal blotches and serrate margins occurred more frequently in drier environments.
- Light grey leaflets and serrate margins occurred on more sodic soils of lower N and P fertility in drier environments.
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AAR | Average annual rainfall |
| AHD | Australian Height Datum |
| Alt | Altitude |
| ANOVA | Analysis of variance |
| BSh | Hot Steppe. Steppe with relatively hot summers and dry winters |
| BSk | Cold Steppe. Steppe with cold winters |
| BWh | Hot Desert. Hot dry climate with little rainfall |
| CEC | Cation exchange capacity |
| Cfa | Climate with hot summers without dry season and mild wet winters |
| CWSP | Central West Slopes & Plains |
| DAS | Days after sowing |
| DTF | Days to flower |
| DF | Discriminant function |
| EC | Electrical conductivity |
| FW | Far West |
| Fl.s.d. | Fisher’s least significant difference |
| IBPGR | International Board for Plant Genetic Resources |
| Lat | Latitude |
| Long | Longitude |
| LP | Liverpool Plains |
| Max T | Maximum temperature |
| Min T | Minimum temperature |
| NWPB | North West Plains Brewarrina |
| NWPC | North West Plains Coonamble |
| NWPM | North West Plains Moree |
| NWPW | North West Plains Walgett |
| R–MIA | Riverina–Murrumbidgee Irrigation Area |
| WR | Winter rainfall |
References
- Popay, I. Medicago polymorpha (Bur Clover). CABI Compendium 33031. 2024. Available online: https://www.cabidigitallibrary.org/doi/full/10.1079/cabicompendium.33031 (accessed on 17 February 2025).
- Hely, F.W. Studies with Annual Species of Medicago in Australia. Master’s Thesis, The University of Sydney, Sydney, NSW, Australia, 1950. [Google Scholar]
- McComb, J.A. Annual Medicago species with particular reference to those occurring in Western Australia. J. R. Soc. West Aust. 1974, 57, 61–96. Available online: https://researchportal.murdoch.edu.au/esploro/outputs/journalArticle/Annual-Medicago-species-with-particular-reference/991005542397607891 (accessed on 12 July 2025).
- Lloyd, D.; O’Brien, S.; Johnson, B.; Pengelly, B.; Wurst, M. Pasture Legumes for Subtropical Grain and Pastoral Systems—The Ute Guide, 1st ed.; Primary Industries and Resources South Australia and Grains Research and Development Corporation: Adelaide, SA, Australia, 2006; ISBN 0-7590-1361-9. [Google Scholar]
- Del Pozo, A.; Ovalle, C.; Aronson, J.; Avendano, J. Ecotypic differentiation in Medicago polymorpha L., along an environmental gradient in central Chile. I. Phenology, biomass production and reproductive patterns. Plant Ecol. 2002, 159, 119–130. [Google Scholar] [CrossRef]
- Del Pozo, A.; Ovalle, C.; Aronson, J.; Avendano, J. Ecotypic differentiation in Medicago polymorpha L., along an environmental gradient in central Chile. II. Winter growth as related to phenology and temperature regime. Plant Ecol. 2002, 160, 53–59. [Google Scholar] [CrossRef]
- Clark, S. Plant Guide for Bur Clover (Medicago polymorpha L.); USDA-Natural Resources Conservation Service, Big Flats Plant Materials Center: Corning, NY, USA, 2014. Available online: https://plants.usda.gov/DocumentLibrary/plantguide/pdf/pg_mepo3.pdf (accessed on 12 July 2025).
- Andrew, W.D.; Hely, F.W. Frequency of annual species of Medicago on the major soil groups of the Macquarie Region of New South Wales. Aust. J. Agric. Res. 1960, 11, 705–713. [Google Scholar] [CrossRef]
- Hely, F.W. Frequency of annual species of Medicago on subgroups of the grey and brown soils of heavy texture of the Macquarie Region of New South Wales. Aust. J. Agric. Res. 1962, 13, 801–812. [Google Scholar] [CrossRef]
- Anon. Medicago polymorpha (FABACEAE) Burr Medic. Save Our Waterways Now. 2021. Available online: https://sown.com.au (accessed on 17 February 2025).
- Lloyd, D.L.; Thompson, J.P.; Boschma, S.P.; Young, R.R.; Johnson, B.; Teasdale, K.C. Phenotypic and agronomic variation within naturalized Medicago polymorpha L. (Burr medic) in Subtropical Queensland, Australia, and relationships with climate and soil characteristics. Agronomy 2025, 15, 139. [Google Scholar] [CrossRef]
- Cunningham, G.M.; Mulham, W.E.; Milthorpe, P.L.; Leigh, J.H. Plants of Western New South Wales. 2011. Available online: https://www.publish.csiro.au/book/6772/ (accessed on 3 July 2025).
- Taylor, G.B. Incidence and measurement of autumn seed softening within Medicago polymorpha L. Aust. J. Agric. Res. 1996, 47, 575–586. [Google Scholar] [CrossRef]
- Lloyd, D.L.; Taylor, G.B.; Johnson, B.; Teasdale, K.C. Patterns of seed softening and seedling emergence of nineteen annual medics during three years after a single seed crop in southern Queensland. Aust. J. Exp. Agric. 1997, 37, 767–778. [Google Scholar] [CrossRef]
- Doherty, M.D.; Wright, G.; McDougall, K.L. The flora of Kosciuszko National Park, New South Wales: Summary and overview. Cunninghamia A J. Plant Ecol. East. Aust. 2015, 15, 13–68. [Google Scholar]
- Benson, J.S.; Ashby, E.M. Vegetation of the Guyra 1: 100 000 map sheet New England Bioregion, New South Wales. Cunninghamia A J. Plant Ecol. East. Aust. 2000, 6, 747–872. [Google Scholar]
- Lloyd, D.L. A productive journey—Sown pastures for Southern inland Queensland: 2. Key contributions. Agric. Sci. 2021, 32, 44–50. Available online: https://search.informit.org/doi/10.3316/informit.368610402837288 (accessed on 15 July 2025).
- Puckridge, D.W.; French, R.J. The annual legume pasture in cereal—Ley farming systems of southern Australia: A review. Agric. Ecosyst. Environ. 1983, 9, 229–267. [Google Scholar] [CrossRef]
- Nichols, P.C.H.; Revell, C.K.; Humphries, A.W.; Howie, J.H.; Hall, E.J.; Sandral, G.A.; Ghamkhar, K.; Harris, C.A. Temperate pasture legumes in Australia—their history, current use, and future prospects. Crop Pasture Sci. 2012, 63, 691–725. [Google Scholar] [CrossRef]
- Skinner, D.Z.; Bauchan, G.R.; Auricht, G.; Hughes, S.A. A method for the efficient management and utilization of large germplasm collections. Crop Sci. 1999, 39, 1237–1242. [Google Scholar] [CrossRef]
- Byerlee, D. A Global North-South Mediterranean Exchange 1891–1990: Pastures for Dryland Crop-Livestock Systems1. 2023. Available online: https://www.researchgate.net/publication/370464529 (accessed on 19 February 2025).
- Mackay, J.H.E. Register of Australian Herbage Plant Cultivars. B. Legumes. 9 Annual Medics. E. L. (burr medic) cv. Serena (Reg. No. B-9e-1). J. Aust. Instit. Agric. Sci. 1976, 42, 255–256. [Google Scholar]
- Mackay, J.H.E. Register of Australian Herbage Plant Cultivars. B. Legumes. 9 Annual Medics. E. Medicago polymorpha var. brevispina (Benth.) Heyn (burr medic) cv. Circle Valley (Reg. No. B-9e-2). J. Aust. Instit. Agric. Sci. 1977, 43, 83–84. [Google Scholar]
- Ewing, M.A.; Revell, C.K.; Thorn, C.W.; Oram, R.N. Register of Australian Herbage Plant Cultivars. B. Legumes. 9. Annual Medics, (e) Medicago polymorpha L. var. brevispina (Benth.) Heyn (burr medic) cv. Santiago, Reg. No. B-9e-3, registered April 1988. Aust. J. Exp. Agric. 1989, 9, 297–298. [Google Scholar] [CrossRef]
- Young, R.R.; Lloyd, D.L.; O’Neill, M.; Teasdale, K.; Johnson, B. Variation in naturalized Medicago polymorpha collected from inland eastern Australia. In Proceedings of the 8th Australian Agronomy Conference, Toowoomba, QLD, Australia, 30 January–2 February 1996; pp. 610–612. [Google Scholar]
- Bullitta, S.; Floris, R.; Hayward, M.D.; Loi, A.; Porqueddu, C.; Veronesi, F. Morphological and biochemical variation in Sardinian populations of Medicago polymorpha L. suitable for rainfed Mediterranean conditions. Euphytica 1994, 77, 263–268. [Google Scholar] [CrossRef]
- Loi, A.; Porqueddu, C.; Veronesi, F.; Cocks, P.S. Distribution, diversity and potential agronomic value of Medicago polymorpha in Sardinia. J. Agric. Sci. 1995, 124, 419–425. [Google Scholar] [CrossRef]
- Del Pozo, A.; Ovalle, C.; Aronson, J.; Avendanoe, J. Developmental responses to temperature and photoperiod in ecotypes of Medicago polymorpha L. collected along an environmental gradient in central Chile. Ann. Bot. 2000, 85, 809–814. [Google Scholar] [CrossRef]
- Peel, M.C.; Finlayson, B.L.; McMahon, T.A. Updated world map of the Köppen-Geiger climate classification. Hydrol. Earth Syst. Sci. 2007, 11, 1633–1644. [Google Scholar] [CrossRef]
- Featherstone, W.E.; Kuhn, M. Height systems and vertical datums. A review in the Australian context. J. Geod. 2006, 80, 1–12. [Google Scholar] [CrossRef]
- Australian Government Bureau of Meteorology Data. Available online: https://www.bom.gov.au/climate/data (accessed on 3 March 2025).
- Clewett, J.F.; Clarkson, N.M.; George, D.A.; Ooi, S.; Owens, D.T.; Partridge, I.J.; Simpson, G.B. Rainman StreamFlow, version 4.3. A Comprehensive Climate and Streamflow Analysis Package on CD to Assess Seasonal Forecasts and Manage Climatic Risk; Department of Primary Industries Queensland: Brisbane, QID, Australia, 2003. [Google Scholar]
- Isbell, R.F. The Australian Soil Classification, 2nd ed.; CSIRO Australia: Collingwood, VIC, Australia, 2016. [Google Scholar]
- IBPGR. Description for Annual Medicago/Descripteurs Pour Medicago Annuelles; International Board for Plant Genetic Resources: Rome, Italy, 1991. [Google Scholar]
- VSN. International Genstat for Windows, 23rd ed.; VSN International: Hemel Hempstead, UK, 2024. [Google Scholar]
- Young, R.R.; Croft, P.H.; Sandral, G.A. Variation in flowering times and agronomic characteristics of Medicago laciniata (L.) Miller collected from diverse locations in New South Wales. Aust. J. Exp. Agric. 1992, 32, 59–63. [Google Scholar] [CrossRef]
- Graziano, D.; Di Giorgio, G.; Ruisi, P.; Amato, P.; Giambalvo, D. Variation in pheno-morphological and agronomic traits among burr medic (Medicago polymorpha L.) populations collected in Sicily, Italy. Crop Pasture Sci. 2009, 61, 59–69. [Google Scholar] [CrossRef]
- Clarkson, N.M. Adaptation and Productivity of Annual Medics; Final Report to the Australian Wool Corporation, February 1986; Department of Primary Industries: Brisbane, QLD, Australia, 1986. [Google Scholar]
- Cornish, P.S. Adaptation of Annual Medicago to a Non-Mediterranean Climate 1. The Growing Season Defined by Available Soil Water; Hochman, Z., Ed.; Department of Agriculture New South Wales Technical Bulletin 32, The Ecology and Agronomy of Annual medics: Orange, NSW, Australia, 1985; pp. 13–17. [Google Scholar]
- King, C.J. The First Fifty Years of Agriculture in New South Wales 1950; Extracts from Review of Marketing and Economics, August 1948–December 1949; Department of Agriculture Government Printer: Sydney, NSW, Australia, 1950. [Google Scholar]
- Franks, S.J.; Sim, S.; Weis, A.E. Rapid evolution of flowering time by an annual plant in response to a climate fluctuation. Proc. Natl. Acad. Sci. USA 2007, 104, 1278–1282. [Google Scholar] [CrossRef]
- Jack, C.N.; Friesen, M.L. Rapid evolution of Medicago polymorpha during invasion shifts interactions with the soybean looper. Ecol. Evol. 2019, 9, 10522–10533. [Google Scholar] [CrossRef]
- Paredes, M.; Becerra, V.; Rojo, C.; Del Pozo, A.; Ovalle, C.; Aronson, J. Ecotypic differentiation in Medicago polymorpha L. along an environmental gradient in central Chile. RAPDs studies show little genetic divergence. Euphytica 2002, 123, 431–439. [Google Scholar] [CrossRef]
- Young, R.R.; Lagudah, E.S. Ribosomal DNA, peroxidase and extension variation in genomic DNA of Medicago laciniata (L.) Miller from Australia and Africa. Aust. J. Agric. Res. 1994, 45, 1013–1023. [Google Scholar] [CrossRef]
- Zhao, D.; Sapkota, M.; Lin, M.; Beil, C.; Sheehan, M.; Greene, S.; Irish, B.M. Genetic diversity, population structure, and taxonomic confirmation in annual medic (Medicago spp.) collections from Crimea, Ukraine. Front. Plant Sci. 2024, 15, 1339298. [Google Scholar] [CrossRef]
- Izadpanah, M.; Jafari, A.A. Evaluation of Seeds and Pods Variation of 5 Annual Medic Medicago spp. J. Range Sci. 2012, 3, 71–81. Available online: https://journals.iau.ir/article_513004_ae764aaf52c36beae065420ffde2d4cb.pdf (accessed on 15 July 2025).
- Porqueddu, C.; Loi, A.; Cocks, P.S. Hardseededness and pattern of hard seed breakdown in Sardinian populations of Medicago polymorpha under field conditions. J. Agric. Sci. 1996, 126, 161–168. [Google Scholar] [CrossRef]
- Anon. Available online: https://www.barenbrug.com.au/agriculture/products/scimitar (accessed on 15 July 2025).
- Haan, R.L.; Barnes, D.K. Inheritance of Pod Type, Stem Color, and Dwarf Growth Habit in Medicago polymorpha. Crop Sci. 1998, 38, 1558–1561. [Google Scholar] [CrossRef]
- Yadav, G.; Yadav, V.; Patel, A.; Das, S.; Goyal, M.; Satpathy, S.; Patra, A. Comprehensive review on traditional uses, phytochemistry, pharmacological properties and metal nanoparticles of a leafy vegetable, Medicago polymorpha. Eur. J. Med. Chem. Rep. 2024, 11, 100164. [Google Scholar] [CrossRef]




| Attribute Designation | Description |
|---|---|
| Leaflet attributes * recorded as LEAF42 and LEAF98, at 42 and 98 days after sowing (DAS) | |
| 1a | No mark on leaflets. Leaflet margins slightly serrate. |
| 1b | Leaflets with faint to obvious brown-purple proximal midrib coloration 0.5–2 mm long. Leaflet margins slightly serrate. |
| 1c | Leaflets with two distinct proximal small squarish smudges either side of the mid-rib, sometimes merging. Leaflet margins slightly serrate. |
| 1d | Leaflets with proximal dark inverted Y-like marks. Leaflet margins slightly serrate. |
| 2 | No mark on leaflets. Leaflet margins distinctly dentate (usually occurring with 1a leaflets on the one plant). |
| 3 | Distinct proximal blotch with dark band across the base of leaflets with lighter color near leaflet bases. Leaflets usually grey. Leaflet margins slightly serrate. |
| 4 | Leaflets a larger darker form of 3. |
| Additional LEAF attributes assessed 98 DAS | |
| 1bd | 1b types with larger marks. |
| GL | Grey leaflets. |
| WO | Light grey leaflets. |
| Other plant attributes | |
| VIGOR | Plant vigor rated visually, 1 (minimum) to 6 (maximum), 107 DAS. |
| FLOWERDAY | DAS to appearance of first flower. |
| POD COILS | Number of coils on mature pods. |
| POD SPINES | Rating of mature pod spine length: (1) <1 mm; (2) 1–2 mm; (3) 2–3 mm; (4) >3 mm. |
| Region | Plant Vigor | Pod Coils | Pod Spines | |||
|---|---|---|---|---|---|---|
| Mean | SEM | Mean | SEM | Mean | SEM | |
| 1 Far West | 3.94 d | 0.036 | 3.49 bc | 0.013 | 2.24 a | 0.015 |
| 2 Riverina MIA | 3.39 a | 0.017 | 3.52 c | 0.006 | 2.85 d | 0.007 |
| 3 Central West Slopes & Plains | 3.57 c | 0.020 | 3.39 a | 0.007 | 2.82 c | 0.009 |
| 4 North West Plains Coonamble | 3.53 c | 0.015 | 3.56 d | 0.006 | 2.86 d | 0.007 |
| 5 North West Plains Brewarrina | 3.93 d | 0.023 | 3.48 b | 0.009 | 2.65 b | 0.010 |
| 6 North West Plains Walgett | 3.84 d | 0.029 | 3.88 f | 0.011 | 2.79 c | 0.013 |
| 7 North West Plains Moree | 3.52 bc | 0.027 | 3.63 e | 0.010 | 2.84 cd | 0.012 |
| 8 Liverpool Plains | 3.41 ab | 0.025 | 3.57 d | 0.009 | 3.01 e | 0.011 |
| 9 cv. SANTIAGO | 4.26 e | 0.032 | - | - | - | - |
| 10 cv. SERENA | 4.23 e | 0.033 | - | - | - | - |
| Plant Attribute | Site Characters Significantly Correlated with Plant Attributes | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| LEAF42_1a | Lat 0.412 *** | Min T −0.332 ** | Na 0.295 ** | Ca −0.292 ** | ||||||
| LEAF42_1c | Lat 0.250 * | |||||||||
| LEAF42_1d | Ca 0.394 *** | |||||||||
| LEAF42_2 | Ca 0.326 ** | Long 0.321 ** | WR 0.307 ** | Lat −0.272 * | Mg 0.252 * | Min T 0.244 * | ||||
| LEAF42_3_4 | AAR −0.284 * | |||||||||
| LEAF98_1a | Max T 0.295 ** | Lat −0.294 ** | EC 0.237 * | |||||||
| LEAF98_1c | Lat 0.246 * | pH −0.253 * | Clay 0.230 * | |||||||
| LEAF98_1bd | Alt 0.429 *** | CEC 0.305 ** | AAR 0.301 ** | Max T −0.334 ** | C 0.290 * | WR 0.282 * | Long 0.235 * | N 0.228 * | ||
| LEAF98_2 | Ca 0.315 ** | WR 0.279 * | Long 0.277 * | AAR 0.245 * | ||||||
| LEAF98_3_4 | AAR −0.237 * | |||||||||
| LEAF98_GL | Alt 0.281 * | |||||||||
| LEAF98_WO | AAR −0.254 * | C −0.244 * | Na 0.231 * | |||||||
| FLOWERDAY | Max T −0.512 *** | AAR 0.462 *** | Alt 0.394 *** | C 0.377 *** | Min T −0.367 ** | N 0.364 ** | Lat 0.350 ** | pH −0.315 ** | WR 0.295 * | Long 0.216 * |
| POD COILS | Ca 0.346 ** | |||||||||
| POD SPINES | AAR 0.273 * | WR 0.239 * | Max T −0.231 * | |||||||
| Attribute | DF1 | Attribute | DF2 |
|---|---|---|---|
| FLOWERDAY | −0.7388 | LEAF98_3_4 | −0.4096 |
| LEAF42_1a | −0.4176 | LEAF42_1a | −0.3498 |
| POD SPINES | −0.3453 | LEAF98_1a | −0.3421 |
| LEAF98_1a | −0.2624 | LEAF42_3_4 | −0.3080 |
| LEAF98_GL | −0.1388 | VIGOR | −0.2682 |
| LEAF42_1c | −0.1160 | LEAF98_WO | −0.1457 |
| LEAF98_WO | 0.1826 | LEAF42_1c | 0.1004 |
| POD COILS | 0.1845 | LEAF42_1d | 0.1052 |
| LEAF98_3_4 | 0.2715 | FLOWERDAY | 0.1256 |
| VIGOR | 0.2973 | LEAF98_1c | 0.1796 |
| LEAF42_3_4 | 0.3240 | LEAF42_1b | 0.2332 |
| POD COILS | 0.2413 | ||
| LEAF98_1b | 0.2448 | ||
| POD SPINES | 0.3536 | ||
| LEAF42_2 | 0.4484 | ||
| LEAF98_2 | 0.4761 |
| Discriminant Score | Site Characteristics | r | Probability |
|---|---|---|---|
| DS1 | pH | 0.951 | <0.001 |
| Winter rainfall | −0.742 | 0.035 | |
| Temperature min winter | 0.663 | 0.073 | |
| DS2 | Cation exchange capacity | 0.750 | 0.032 |
| Clay content | 0.723 | 0.043 | |
| Longitude | 0.701 | 0.053 | |
| Total nitrogen | 0.681 | 0.063 | |
| Annual rainfall | 0.668 | 0.070 | |
| Exchangeable magnesium | 0.641 | 0.087 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lloyd, D.L.; Thompson, J.P.; Young, R.R.; Boschma, S.P.; O’Neill, M. Variation in Leaf Morphology and Agronomic Attributes of a Naturalized Population of Medicago polymorpha L. (Burr Medic) from New South Wales, Australia, and Relationships with Climate and Soil Characteristics. Agronomy 2025, 15, 1737. https://doi.org/10.3390/agronomy15071737
Lloyd DL, Thompson JP, Young RR, Boschma SP, O’Neill M. Variation in Leaf Morphology and Agronomic Attributes of a Naturalized Population of Medicago polymorpha L. (Burr Medic) from New South Wales, Australia, and Relationships with Climate and Soil Characteristics. Agronomy. 2025; 15(7):1737. https://doi.org/10.3390/agronomy15071737
Chicago/Turabian StyleLloyd, David L., John P. Thompson, Rick R. Young, Suzanne P. Boschma, and Mark O’Neill. 2025. "Variation in Leaf Morphology and Agronomic Attributes of a Naturalized Population of Medicago polymorpha L. (Burr Medic) from New South Wales, Australia, and Relationships with Climate and Soil Characteristics" Agronomy 15, no. 7: 1737. https://doi.org/10.3390/agronomy15071737
APA StyleLloyd, D. L., Thompson, J. P., Young, R. R., Boschma, S. P., & O’Neill, M. (2025). Variation in Leaf Morphology and Agronomic Attributes of a Naturalized Population of Medicago polymorpha L. (Burr Medic) from New South Wales, Australia, and Relationships with Climate and Soil Characteristics. Agronomy, 15(7), 1737. https://doi.org/10.3390/agronomy15071737






