Effects of Light on Adventitious Rooting In Vitro
Abstract
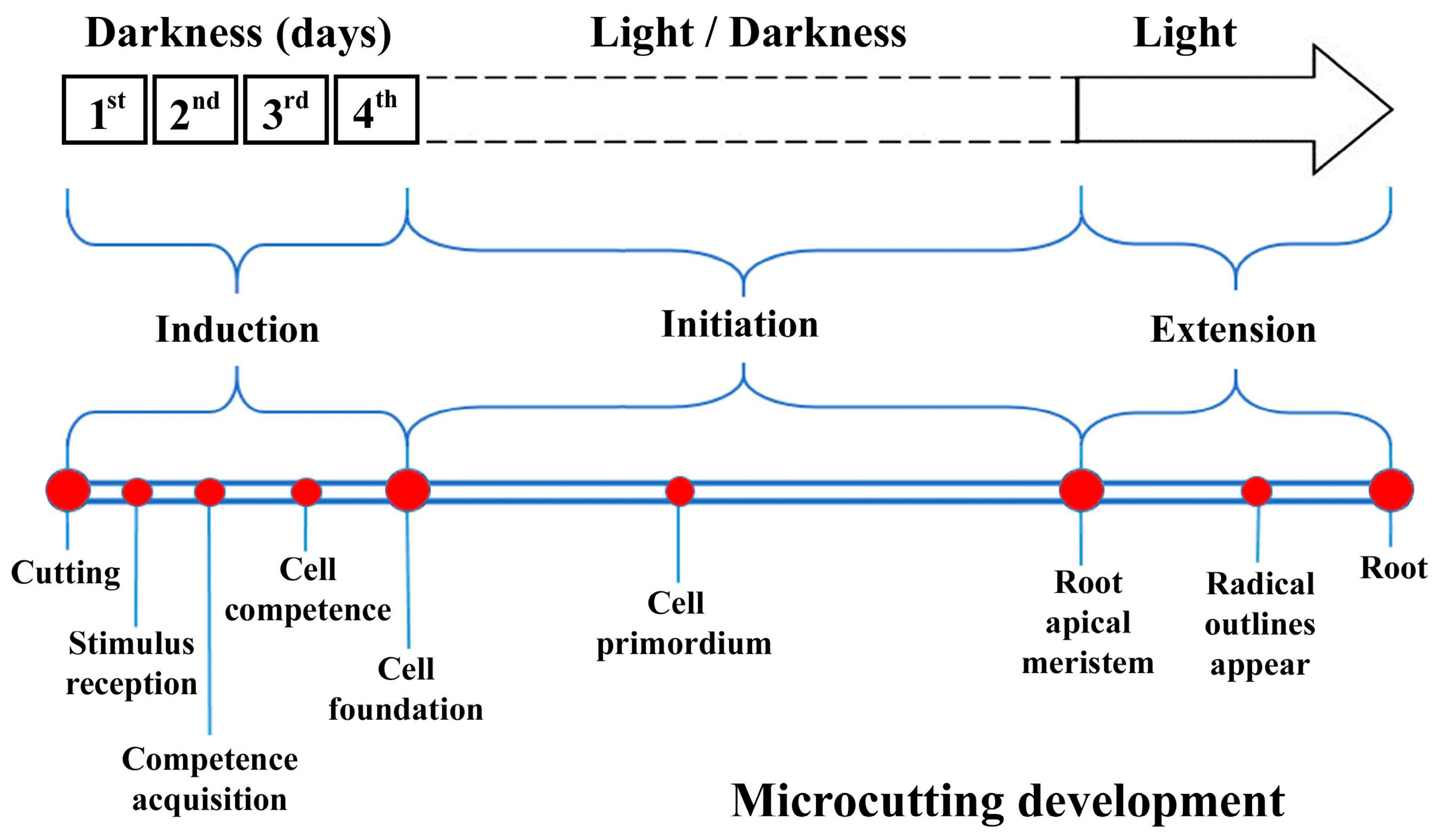
1. Introduction
2. Light and In Vitro Rooting Process
2.1. Light and Hormonal Component
2.2. Light and Carbohydrates
2.3. Light and Antioxidants
2.4. Light Quality and Adventitious Rooting
2.4.1. Effects of Red (RL) and Far-Red Light (FRL)
2.4.2. Combined Effects of Red Light (RL) and Blue Light (BL)
2.4.3. Effects of Blue Light (BL)
2.4.4. Effects of Green Light (GL)
2.4.5. Effects of White Light (WL)
2.4.6. Effects of YL
2.4.7. Effects of Ultraviolet Light (UVL)
2.5. Effects of Light Intensity
2.6. Effects of Darkness (D)
2.7. Effects of Light During In Vitro Rooting on Acclimatization
3. Concluding Remarks
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Díaz-Sala, C.A. Perspective on adventitious root formation in tree species. Plants 2020, 9, 1789. [Google Scholar] [CrossRef] [PubMed]
- Bellini, C.; Pacurar, D.I.; Perrone, I. Adventitious roots and lateral roots: Similarities and differences. Annu. Rev. Plant Biol. 2014, 65, 639–666. [Google Scholar] [CrossRef] [PubMed]
- Steffens, B.; Rasmussen, A. The physiology of adventitious roots. Plant Physiol. 2016, 170, 603–617. [Google Scholar] [CrossRef] [PubMed]
- Lahti, M.; Aphalo, P.J.; Finér, L.; Ryyppö, A.; Lehto, T.; Mannerkoski, H. Effects of soil temperature on shoot and root growth and nutrient uptake of 5-year-old Norway spruce seedlings. Tree Physiol. 2005, 25, 115–122. [Google Scholar] [CrossRef]
- Kurepin, L.; Walton, L.; Hayward, A.; Emery, R.; Pharis, R.; Reid, D. Interactions between plant hormones and light quality signaling in regulating the shoot growth of Arabidopsis thaliana seedlings. Botany 2012, 90, 237–246. [Google Scholar] [CrossRef]
- Luziatelli, F.; Gatti, L.; Ficca, A.G.; Medori, G.; Silvestri, C.; Melini, F.; Muleo, R.; Ruzzi, M. Metabolites secreted by a plant-growth-promoting Pantoea agglomerans strain improved rooting of Pyrus communis L. cv Dar Gazi cuttings. Front. Microbiol. 2020, 11, 539359. [Google Scholar] [CrossRef]
- Valerio, C.; Medori, G.; Luziatelli, F.; Melini, F.; Gatti, L.; Ruzzi, M.; Muleo, R.; Forgione, I. Bioregulation of adventitious root induction by metabolites secreted from plant growth promoting Pantoea agglomerans strains. Acta Hortic. 2023, 1359, 33–42. [Google Scholar] [CrossRef]
- Ma, Y.; Zhang, Y.; Xu, J.; Qi, J.; Liu, X.; Guo, L.; Zhang, H. Research on the mechanisms of phytohormone signaling in regulating root development. Plants 2024, 13, 3051. [Google Scholar] [CrossRef]
- Pacifici, E.; Polverari, L.; Sabatini, S. Plant hormone cross-talk: The pivot of root growth. J. Exp. Bot. 2015, 66, 1113–1121. [Google Scholar] [CrossRef]
- Bustillo-Avendaño, E.; Ibanez, S.; Sanz, O.; Sousa Barros, J.A.; Gude, I.; Perianez-Rodriguez, J.; Micol, J.L.; Del Pozo, C.J.; Moreno-Risueno, A.M.; Perez-Perez, M.J. Regulation of hormonal control, cell reprogramming, and patterning during de novo root organogenesis. Plant Physiol. 2018, 176, 1709–1727. [Google Scholar] [CrossRef]
- Altamura, M.M. Root histogenesis in herbaceous and woody explants cultured in vitro. A critical review. Agronomie 1996, 16, 589–602. [Google Scholar] [CrossRef]
- Porfírio, S.; Gomes da Silva, M.D.R.; Cabrita, M.J.; Azadi, P.; Peixe, A. Reviewing current knowledge on olive (Olea europaea L.) adventitious root formation. Sci. Hortic. 2016, 198, 207–226. [Google Scholar] [CrossRef]
- Muleo, R.; Morini, S. Physiological dissection of blue and red light regulation of apical dominance and branching in M9 apple rootstock growing in vitro. J. Plant Physiol. 2008, 165, 1838–1846. [Google Scholar] [CrossRef] [PubMed]
- Cavallaro, V.; Pellegrino, A.; Muleo, R.; Forgione, I. Light and plant growth regulators on in vitro proliferation. Plants 2022, 11, 844. [Google Scholar] [CrossRef]
- Brini, F.; Mseddi, K.; Brestic, M.; Landi, M. Hormone-mediated plant responses to light quality and quantity. Environ. Exp. Bot. 2022, 202, 105026. [Google Scholar] [CrossRef]
- Lau, O.S.; Deng, X.W. Plant hormone signaling lightens up: Integrators of light and hormones. Curr. Opin. Plant Biol. 2010, 13, 571–577. [Google Scholar] [CrossRef]
- Sassi, M.; Wang, J.; Ruberti, I.; Vernoux, T.; Xu, J. Shedding light on auxin movement: Light-regulation of polar auxin transport in the photocontrol of plant development. Plant Signal Behav. 2013, 8, e23355. [Google Scholar] [CrossRef]
- Iacona, C.; Medori, G.; Thomas, B.; Roncasaglia, R.; Dradi, G.; Radicetti, E.; Mancinelli, R.; Muleo, R.; Forgione, I. Apical dominance and branching in plantlets of Colt cherry lines expressing different light and auxin signalling sensitivities. Agronomy 2023, 13, 2018. [Google Scholar] [CrossRef]
- Thomas, B.; Dickinson, H. Evidence for two photoreceptors controlling growth in de-etiolated seedlings. Planta 1979, 146, 545–550. [Google Scholar] [CrossRef]
- Reed, J.W.; Nagatani, A.; Elich, T.D.; Fagan, M.; Chory, J. Phytochrome A and phytochrome B have overlapping but distinct functions in Arabidopsis development. Plant Physiol. 1994, 104, 1139–1149. [Google Scholar] [CrossRef]
- Poudel, P.R.; Kataoka, I.; Mochioka, R. Effect of red and blue-light-emitting diodes on growth and morphogenesis of grapes. Plant Cell Tissue Organ Cult. 2008, 92, 147–153. [Google Scholar] [CrossRef]
- Lin, K.H.; Huang, M.Y.; Huang, W.D.; Hsu, M.H.; Yang, Z.W.; Yang, C.M. The effects of red, blue, and white light-emitting diodes on the growth, development, and edible quality of hydroponically grown lettuce (Lactuca sativa L. var. capitata). Sci. Hortic. 2013, 150, 86–91. [Google Scholar] [CrossRef]
- Chen, M.; Chory, J.; Fankhauser, C. Light signal transduction in higher plants. Annu. Rev. Genet. 2004, 38, 87–117. [Google Scholar] [CrossRef]
- Christie, J.M. Phototropin blue-light receptors. Annu. Rev. Plant Biol. 2007, 58, 21–45. [Google Scholar] [CrossRef] [PubMed]
- Paradiso, R.; Proietti, S. Light-quality manipulation to control plant growth and photomorphogenesis in greenhouse horticulture: The state of the art and the opportunities of modern LED systems. J. Plant Growth Reg. 2022, 41, 742–780. [Google Scholar] [CrossRef]
- Lee, J.H.; Nam, S.Y. Vegetative propagation of six Pachyphytum species as influenced by different LED light qualities. Hortic. Sci. Technol. 2023, 41, 237–249. [Google Scholar] [CrossRef]
- Lee, J.H.; Jang, I.T.; Kim, E.A.; Shin, E.J.; Lee, S.; Lee, M.; Nam, S.Y. Evaluating the influence of various light spectra on the growth and morphological responses of air plant Tillandsia ionantha planch. Grown under non-substrate and restricted irrigation conditions in a controlled environment facility. J. Agri. Life Environ. Sci. 2024, 36, 546–561. [Google Scholar] [CrossRef]
- Dutta Gupta, S.; Agarwal, A. Artificial lighting system for plant growth and development: Chronological advancement, working principles, and comparative assessment. In Light Emitting Diodes for Agriculture; Springer: Singapore, 2017; pp. 1–25. [Google Scholar] [CrossRef]
- Bidabadi, S.S.; Jain, S.M. Cellular, Molecular, and physiological aspects of in vitro plant regeneration. Plants 2020, 9, 702. [Google Scholar] [CrossRef]
- Velada, I.; Cardoso, H.; Porfirio, S.; Peixe, A. Expression profile of PIN-formed auxin efflux carrier genes during IBA-Induced in vitro adventitious rooting in Olea europaea L. Plants 2020, 9, 185. [Google Scholar] [CrossRef]
- Devi, J.; Kaur, E.; Swarnkar, M.K.; Acharya, V. De novo transcriptome analysis provides insights into formation of in vitro adventitious root from leaf explants of Arnebia euchroma. BMC Plant Biol. 2021, 21, 414. [Google Scholar] [CrossRef]
- Li, S.W. Molecular bases for the regulation of adventitious root generation in plants. Front. Plant Sci. 2021, 12, 614072. [Google Scholar] [CrossRef] [PubMed]
- Zhi, L.; Hu, X. Adventitious root regeneration: Molecular basis and influencing factors. Phyton 2023, 2815, 2840. [Google Scholar] [CrossRef]
- Ai, Y.; Qian, X.; Wang, X.; Chen, Y.; Zhang, T.; Chao, Y.; Zhao, Y. Uncovering early transcriptional regulation during adventitious root formation in Medicago sativa. BMC Plant Biol. 2023, 23, 176. [Google Scholar] [CrossRef] [PubMed]
- Ljung, K.; Bhalerao, R.P.; Sandberg, G. Sites and homeostatic control of auxin biosynthesis in Arabidopsis during vegetative growth. Plant J. 2021, 28, 465–474. [Google Scholar] [CrossRef]
- Friml, J. Auxin transport—Shaping the plant. Curr. Opin. Plant Biol. 2003, 6, 7–12. [Google Scholar] [CrossRef]
- Blakeslee, J.J.; Peer, W.A.; Murphy, A.S. Auxin transport. Curr. Opin. Plant Biol. 2005, 8, 494–500. [Google Scholar] [CrossRef]
- Peret, B.; Swarup, K.; Ferguson, A.; Seth, M.; Yang, Y.; Dhondt, S.; James, N.; Casimiro, I.; Perry, P.; Syed, A. AUX/LAX genes encode a family of auxin influx transporters that perform distinct functions during Arabidopsis development. Plant Cell 2012, 24, 2874–2885. [Google Scholar] [CrossRef]
- Wisniewska, J.; Xu, J.; Seifertova, D.; Brewer, P.B.; Ruzicka, K.; Blilou, I.; Rouquie, D.; Benkova, E.; Scheres, B.; Friml, J. Polar PIN localization directs auxin flow in plants. Science 2006, 312, 883. [Google Scholar] [CrossRef]
- Forde, B.G. Local and long-range signaling pathways regulating plant responses to nitrate. Annu. Rev. Plant Biol. 2002, 53, 203–224. [Google Scholar] [CrossRef]
- De Smet, I.; Vanneste, S.; Inze, D.; Beeckman, T. Lateral root initiation or the birth of a new meristem. Plant Mol. Biol. 2006, 60, 871–887. [Google Scholar] [CrossRef]
- Bohn-Courseau, I. Auxin: A major regulator of organogenesis. CR Biol. 2010, 333, 290–296. [Google Scholar] [CrossRef] [PubMed]
- Adem, M.; Sharma, L.; Shekhawat, G.S.; Šafranek, M.; Jásik, J. Auxin signaling, transport, and regulation during adventitious root formation. Curr. Plant Biol. 2024, 40, 100385. [Google Scholar] [CrossRef]
- Vanneste, S.; Friml, J. Auxin: A trigger for change in plant development. Cell 2009, 136, 1005–1016. [Google Scholar] [CrossRef] [PubMed]
- Lavenus, J.; Goh, T.; Roberts, I.; Guyomarc’h, S.; Lucas, M.; De Smet, I.; Fukaki, H.; Beeckman, T.; Bennett, M.; Laplaze, L. Lateral root development in Arabidopsis: Fifty shades of auxin. Trends Plant Sci. 2013, 18, 450–458. [Google Scholar] [CrossRef]
- Christiaens, A.; Gobin, B.; Van Labeke, M. Light quality and adventitious rooting: A mini-review. Acta Hortic. 2016, 1134, 385–394. [Google Scholar] [CrossRef]
- Yun, F.; Liu, H.; Deng, Y.; Hou, X.; Liao, W. The Role of light-regulated auxin signaling in root development. Int. J. Mol. Sci. 2023, 24, 5253. [Google Scholar] [CrossRef]
- Fasano, R.; Gonzalez, N.; Tosco, A.; Dal Piaz, F.; Docimo, T.; Serrano, R.; Grillo, S.; Leone, A.; Inzé, D. Role of Arabidopsis UV RESISTANCE LOCUS 8 in plant growth reduction under osmotic stress and low levels of UV-B. Mol. Plant 2014, 7, 773–791. [Google Scholar] [CrossRef]
- Kumari, S.; Panigrahi, K.C.S. Light and auxin signaling cross-talk programme root development in plants. J. Biosci. 2019, 44, 26. [Google Scholar] [CrossRef]
- Alallaq, S.; Ranjan, A.; Brunoni, F.; Novák, O.; Lakehal, A.; Bellini, C. Red light controls adventitious root regeneration by modulating hormone homeostasis in Picea abies seedlings. Front. Plant Sci. 2020, 11, 586140. [Google Scholar] [CrossRef]
- Sauer, M.; Robert, S.; Kleine-Vehn, J. Auxin: Simply complicated. J. Exp. Bot. 2013, 64, 2565–2577. [Google Scholar] [CrossRef]
- Kurepin, L.V.; Emery, R.J.N.; Pharis, R.P.; Reid, D.M. Uncoupling light quality from light irradiance effects in Helianthus annuus shoots: Putative roles for plant hormones in leaf and internode growth. J. Exp. Bot. 2007, 58, 2145–2157. [Google Scholar] [CrossRef] [PubMed]
- Iacona, C.; Muleo, R. Light quality affects in vitro adventitious rooting and ex vitro performance of cherry rootstock Colt. Sci. Hortic. 2010, 125, 630–636. [Google Scholar] [CrossRef]
- Daud, N.; Faizal, A.; Geelen, D. Adventitious rooting of Jatropha curcas L. is stimulated by phloroglucinol and by red LED light. Vitr. Cell. Dev. Biol. Plant 2013, 49, 183–190. [Google Scholar] [CrossRef]
- George, E.F.; Hall, M.A.; De Klerk, G.-J. Effects of the physical environment. In Plant Propagation by Tissue Culture 3rd Edition. Volume 1. The Background; George, E.F., Davies, W., Eds.; Springer: Dordrecht, The Netherlands, 2008; pp. 423–464. ISBN 9781402050046. [Google Scholar] [CrossRef]
- Bhatt, B.B.; Tomar, Y.K. Effect of IBA on rooting performance of Citrus auriantifolia swingle (Kagzi–lime) in different growing conditions. Nat. Sci. 2010, 8, 8–11. [Google Scholar]
- Zimmerman, R.H. Rooting apple cultivars in vitro: Interactions among light, temperature, phloroglucinol and auxin. Plant Cell Tiss. Org. Cult. 1984, 3, 301–311. [Google Scholar] [CrossRef]
- Dubranszki, J.; da Silva, J.A.T. Micropropagation of apple—A review. Biotechnol. Adv. 2010, 28, 462–488. [Google Scholar] [CrossRef]
- Hammatt, N.; Grant, N.J. Micropropagation of mature British wild cherry. Plant Cell Tiss. Organ Cult. 1997, 47, 103–110. [Google Scholar] [CrossRef]
- De Klerk, G.J.; Guan, H.Y.; Huisman, P.; Marinova, S. Effects of phenolic compounds on adventitious root formation and oxidative decarboxylation of applied indoleacetic acid in Malus ‘Jork 9’. Plant Growth Regul. 2011, 63, 175–185. [Google Scholar] [CrossRef]
- Lotfi, M.; Mars, M.; Werbrouck, S. Optimizing pear micropropagation and rooting with light emitting diodes and trans-cinnamic acid. Plant Growth Regul. 2019, 88, 173–180. [Google Scholar] [CrossRef]
- Steenackers, W.; Klíma, P.; Quareshy, M.; Cesarino, I.; Kumpf, R.P.; Corneillie, S.; Araújo, P.; Viaene, T.; Goeminne, G.; Nowack, M.K.; et al. Cis-cinnamic acid is a novel, natural auxin efflux inhibitor that promotes lateral root formation. Plant Physiol. 2017, 173, 552–565. [Google Scholar] [CrossRef]
- Shin, K.S.; Murthy, H.N.; Heo, J.W.; Hahn, E.J.; Paek, K.Y. The effect of light quality on the growth and development of in vitro cultured Doritaenopsis plants. Acta Physiol. Plant. 2008, 30, 339–343. [Google Scholar] [CrossRef]
- Gu, A.; Liu, W.; Ma, C.; Cui, J.; Henny, R.J.; Chen, J. Regeneration of Anthurium andraeanum from leaf explants and evaluation of microcutting rooting and growth under different light qualities. Hort. Sci. 2012, 47, 88–92. [Google Scholar] [CrossRef]
- Shohael, A.M.; Ali, M.B.; Yu, K.W.; Hahn, E.J.; Islam, R.; Paek, K.Y. Effect of light on oxidative stress, secondary metabolites and induction of antioxidant enzymes in Eleutherococcus senticosus somatic embryos in bioreactor. Process Biochem. 2006, 41, 1179–1185. [Google Scholar] [CrossRef]
- Yamakawa, T.; Kurahashi, O.; Ishida, K.; Kato, S.; Kodama, T.; Minoda, Y. Stability of indole-3-acetic-acid to autoclaving, aeration and light illumination. Agric Biol Chem. 1979, 43, 879–880. [Google Scholar]
- Peer, W.A.; Murphy, A.S. Flavonoids and auxin transport: Modulators or regulators? Trends Plant Sci. 2007, 12, 556–563. [Google Scholar] [CrossRef] [PubMed]
- Buer, C.S.; Imin, N.; Djordjevic, M.A. Flavonoids: New roles for old molecules. J. Integr. Plant Biol. 2010, 52, 98–111. [Google Scholar] [CrossRef] [PubMed]
- Ghimire, B.K.; Lee, J.G.; Yoo, J.H.; Kim, J.K.; Yu, C.Y. The Influence of Light-Emitting Diodes (LEDs) on the growth, antioxidant activities, and metabolites in adventitious root of Panax ginseng CA Meyer. In Light Emitting Diodes for Agriculture, 1st ed.; Springer: Singapore, 2017; pp. 259–272. [Google Scholar] [CrossRef]
- Wu, H.-C.; Lin, C.-C. Red light-emitting diode light irradiation improves root and leaf formation in difficult to propagate Protea cynaroides L. plantlets in vitro. HortScience 2012, 47, 1490–1494. [Google Scholar] [CrossRef]
- Ebisawa, M.; Shaoji, K.; Mieko, K.; Shimomura, K.; Yoshihara, T. Supplementary ultraviolet radiation B together with blue light at night increased quercetin content and flavonol synthase gene expression in leaf lettuce (Lactuca sativa L.). Environ. Cont. Biol. 2008, 46, 1–11. [Google Scholar] [CrossRef]
- Manivannan, A.; Soundararajan, P.; Halimah, N.; Ko, C.H. Blue LED light enhances growth, phytochemical contents, and antioxidant enzyme activities of Rehmannia glutinosa cultured in vitro. Hort. Environ. Biotechnol. 2015, 56, 105–113. [Google Scholar] [CrossRef]
- Mucciarelli, M.; Gallino, M.; Maffei, M.; Scannerini, S. Effects of 3,4-dihydroxybenzoic acid on tobacco (Nicotiana tabacum L.) cultured in vitro. Growth regulation in callus and organ cultures. Plant Biosyst. 2000, 134, 185–192. [Google Scholar] [CrossRef]
- Fletcher, R.; Peterson, R.; Zalik, S. Effect of light quality on elongation, adventitious root production and the relation of cell number and cell size to bean seedling elongation. Plant Physiol. 1965, 40, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Fuernkranz, H.A.; Nowak, C.A.; Maynard, C.A. Light effects on in vitro adventitious root formation in axillary shoots of mature Prunus serotina. Physiol. Plant 1990, 80, 337–341. [Google Scholar] [CrossRef]
- Gabryszewska, E.; Rudnicki, R.M. The effects of light quality on the growth and development of shoots and roots of Ficus benjamina in vitro. Acta Hortic. 1997, 418, 163–168. [Google Scholar] [CrossRef]
- Niemi, K.; Julkunen-Tiitto, R.; Tegelberg, R.; Häggman, H. Light sources with different spectra affect root and mycorrhiza formation in Scots pine in vitro. Tree Physiol. 2005, 25, 123–128. [Google Scholar] [CrossRef][Green Version]
- Ruedell, C.M.; de Almeida, M.R.; Schwambach, J.; Posenato, C.F.; FettNeto, A.G. Pre and post-severance effects of light quality on carbohydrate dynamics and microcutting adventitious rooting of two Eucalyptus species of contrasting recalcitrance. Plant Growth Regul. 2013, 69, 235–245. [Google Scholar] [CrossRef]
- Moon, H.K.; Park, S.Y.; Kim, Y.W.; Kim, C.S. Growth of Tsuru-rindo (Tripterospermum japonicum) cultured in vitro under various sources of light-emitting diode (LED) irradiation. J. Plant Biol. 2006, 49, 174–179. [Google Scholar] [CrossRef]
- Manivannan, A.; Soundararajan, P.; Park, Y.G.; Wei, H.; Kim, S.H.; Jeong, B.R. Blue and red light-emitting diodes improve the growth and physiology of in vitro-grown carnations ‘Green Beauty’ and ‘Purple Beauty’. Hortic. Environ. Biotechnol. 2017, 58, 12–20. [Google Scholar] [CrossRef]
- Rossi, F.; Baraldi, R.; Facini, O.; Lercari, B. Photomorphogenic effects on in vitro rooting of Prunus roostock GF 655-2. Plant Cell Tissue Organ. Cult. 1993, 32, 145–151. [Google Scholar] [CrossRef]
- Bertazza, G.; Baraldi, R.; Predieri, S. Light effects on in vitro rooting of pear cultivars of different rhizogenic ability. Plant Cell Tissue Organ Cult. 1995, 41, 139–143. [Google Scholar] [CrossRef]
- Gabryszewska, E.; Rudnicki, R. The influence of light quality on the shoot proliferation and rooting of Gerbera jamesonii in vitro. Acta Agrobot. 1995, 48, 105–111. [Google Scholar] [CrossRef][Green Version]
- Kromer, K.; Kukulczarika, K.; Strzelecka, K. The effect of visible radiation waves of different length and growth regulators on the growth and development of bromelias. Acta Univ. Wratislay 1996, 61, 37–53. [Google Scholar]
- Baque, M.A.; Hahn, E.-J.; Paek, K.-Y. Induction mechanism of adventitious root from leaf explants of Morinda citrifolia as affected by auxin and light quality. Vitr. Cell. Dev. Biol. Plant 2010, 46, 71–80. [Google Scholar] [CrossRef]
- Park, S.Y.; Kim, M.J. Development of zygotic embryos and seedlings is affected by radiation spectral compositions from light emitting diode (LED) system in Chestnut (Castanea crenata S. et Z.). J. Korean For. Soc. 2010, 99, 750–754. [Google Scholar]
- Li, H.M.; Xu, Z.G.; Tang, C.M. Effect of light-emitting diodes on growth and morphogenesis of upland cotton (Gossypium hirsutum L.) plantlets in vitro. Plant Cell Tissue Org. Cult. 2010, 103, 155–163. [Google Scholar] [CrossRef]
- Budiarto, K. Spectral quality affects morphogenesis on Anthurium plantlet during in vitro culture. AJAS 2010, 32, 234–240. [Google Scholar] [CrossRef]
- Cho, K.H.; Laux, V.Y.; Wallace-Springer, N.; Clark, D.G.; Folta, K.M.; Colquhoun, T.A. Effects of light quality on vegetative cutting and in vitro propagation of coleus (Plectranthus scutellarioides). Hort. Sci. 2019, 54, 926–935. [Google Scholar] [CrossRef]
- Kurilcik, A.; Canova, M.R.; Dapkuniene, S.; Zilinskaite, S.; Kurilcik, G. In vitro culture of Chrysanthemum plantlets using light emitting diodes. Cent. Eur. J. Biol. 2008, 2, 161–167. [Google Scholar] [CrossRef]
- Nacheva, L.; Dimitrova, N.; Koleva-Valkova, L.; Stefanova, M.; Ganeva, T.; Nesheva, M.; Tarakanov, I.; Vassilev, A. In vitro multiplication and rooting of Plum rootstock ‘Saint Julien’ (Prunus domestica Subsp. Insititia) under fluorescent light and different LED Spectra. Plants 2023, 12, 2125. [Google Scholar] [CrossRef]
- Gabarkiewicz, B.; Gabryszewska, E.; Rudnicki, R.; Goszczyńska, D. Effect of light quality on in vitro growing of Dieffenbachia cv. Compacta. Acta Hort. 1997, 418, 159–161. [Google Scholar] [CrossRef]
- Kozak, D. The influence of light quality and BA on in vitro growth and development of Gardenia jasminoides Ellis. Acta Sci. Pol-hortoru. 2011, 10, 65–73. [Google Scholar]
- Lee, N.N.; Choi, Y.E.; Moon, H.K. Effect of LEDs on shoot multiplication and rooting of rare plant Abeliophyllum distichum Nakai. J. Plant Biochem. Biotechnol. 2014, 41, 94–99. [Google Scholar] [CrossRef]
- Mengxi, L.; Zhigang, X.; Yang, Y.; Yijie, F. Effects of different spectral lights on Oncidium PLBs induction, proliferation, and plant regeneration. Plant Cell Tissue Organ Cult. 2011, 106, 1–10. [Google Scholar] [CrossRef]
- Salisbury, F.J.; Hall, A.; Grierson, C.S.; Halliday, K.J. Phytochrome coordinates Arabidopsis shoot and root development. Plant J. 2007, 50, 429–438. [Google Scholar] [CrossRef]
- Canamero, R.; Bakrim, N.; Bouly, J.P.; Garay, A.; Dudkin, E.; Habricot, Y.; Ahmad, M. Cryptochromes photoreceptors cry1 and cry2 antagonistically regulate primary root elongation in Arabidopsis thaliana. Planta 2006, 224, 995–1003. [Google Scholar] [CrossRef]
- Galen, C.; Rabenold, J.J.; Liscum, E. Functional ecology of a blue light photoreceptor: Effects of phototropin-1 on root growth enhance drought tolerance in Arabidopsis thaliana. New Phytol. 2007, 173, 91–99. [Google Scholar] [CrossRef]
- Meng, L.; Song, W.; Liu, S.; Dong, J.; Zhang, Y.; Wang, C.; Xu, Y.; Wang, S. Light quality regulates lateral root development in tobacco seedlings by shifting auxin distributions. J. Plant Growth Regul. 2015, 34, 574–583. [Google Scholar] [CrossRef]
- Zeng, J.; Wang, Q.; Lin, J.; Deng, K.; Zhao, X.; Tang, D.; Liu, X. Arabidopsis cryptochrome-1 restrains lateral roots growth by inhibiting auxin transport. J. Plant Physiol. 2010, 167, 670–673. [Google Scholar] [CrossRef]
- Moni, A.; Lee, A.Y.; Briggs, W.R.; Han, I.S. The blue light receptor Phototropin 1 suppresses lateral root growth by controlling cell elongation. Plant Biology (Stuttg) 2015, 17, 34–40. [Google Scholar] [CrossRef]
- Gutierrez, L.; Mongelard, G.; Floková, K.; Pacurar, D.I.; Novák, O.; Staswick, P. Auxin controls Arabidopsis adventitious root initiation by regulating jasmonic acid homeostasis. Plant Cell 2012, 24, 2515–2527. [Google Scholar] [CrossRef]
- Chung, J.-P.; Huang, C.-Y.; Dai, T.-E. Spectral effects on embryogenesis and plantlet growth of Oncidium ‘Gower Ramsey’. Sci. Hortic. 2010, 124, 511–516. [Google Scholar] [CrossRef]
- Darouez, H.; Werbrouck, S.P.O. Red and far-red light combined with trans-cinnamic acid enhances in vitro rooting and reduces callus formation in lavender. Horticulturae 2024, 10, 954. [Google Scholar] [CrossRef]
- Nhut, D.T.; Hong, L.; Watanabe, H.; Goi, M.; Tanaka, M. Growth of banana plantlets cultured in vitro under red and blue light-emitting diode (LED) irradiation source. Acta Hortic. 2002, 575, 117–124. [Google Scholar] [CrossRef]
- Jiang, L.; Wang, Z.; Jin, G.; Lu, D.; Li, X. Responses of Favorita potato plantlets cultured in vitro under fluorescent and light-emitting diode (LED) light sources. Am. J. Potato Res. 2019, 96, 396–402. [Google Scholar] [CrossRef]
- Ferreira, L.T.; de Araújo Silva, M.M.; Ulisses, C.; Camara, T.R.; Willadino, L. Using LED lighting in somatic embryogenesis and micropropagation of an elite sugarcane variety and its effect on redox metabolism during acclimatization. Plant Cell Tissue Organ Cult. 2017, 128, 211–221. [Google Scholar] [CrossRef]
- Hung, C.D.; Hong, C.-H.; Jung, H.-B.; Kim, S.-K.; Van Ket, N.; Nam, M.-W.; Choi, D.-H.; Lee, H.-I. Growth and morphogenesis of encapsulated strawberry shoot tips under mixed LEDs. Sci. Hortic. 2015, 194, 194–200. [Google Scholar] [CrossRef]
- Huang, W.; Yang, G.; Li, F.; Zheng, L.; Ma, J. Effects of different light qualities-LED on apple rootstock JM7 tissue culture seedling. Am. J. Plant Biol. 2018, 3, 17–20. [Google Scholar] [CrossRef]
- Lian, M.L.; Murthy, H.N.; Paek, K.Y. Effects of light emitting diodes (LEDs) on the in vitro induction and growth of bulblets of Lilium oriental hybrid “Pesaro”. Sci. Hortic. 2002, 94, 365–370. [Google Scholar] [CrossRef]
- Verstraeten, I.; Geelen, D. Adventitious rooting and browning are differentially controlled by auxin in rooting-recalcitrant Elegia capensis (Burm. f.) Schelpe. J. Plant Growth Regul. [CrossRef]
- Pawłowska, B.; Szewczyk-Taranek, B.; Dziedzic, E.; Żupnik, M. Rooting response under LED systems in Rosa canina in vitro cultures. Acta Hortic. 2017, 1155, 519–524. [Google Scholar] [CrossRef]
- Li, H.; Tang, C.; Xu, Z. The effects of different light qualities on rapeseed (Brassica napus L.) plantlet growth and morphogenesis in vitro. Sci. Hortic. 2013, 150, 117–124. [Google Scholar] [CrossRef]
- Kwon, A.R.; Cui, H.Y.; Lee, H.; Shin, H.; Kang, K.S.; Park, S.Y. Light quality affects shoot regeneration, cell division, and wood formation in elite clones of Populus euramericana. Acta Physiol. Plant. 2015, 37, 65. [Google Scholar] [CrossRef]
- Chée, R. In vitro culture of Vitis: The effects of light spectrum, manganese sulfate and potassium iodide on morphogenesis. Plant Cell Tissue Organ Cult. 1986, 7, 121–134. [Google Scholar] [CrossRef]
- Zhai, S.W.; Xiang, Z.X.; Chen, C.Y.; Lu, Y.T.; Yuan, T.T. PIN3-mediated auxin transport contributes to blue light-induced adventitious root formation in Arabidopsis. Plant Sci. 2021, 312, 111044. [Google Scholar] [CrossRef]
- Tanaka, M.; Takamura, T.; Watanabe, H.; Endo, M.; Yanagi, T.; Okamoto, K. In vitro growth of Cymbidium plantlets cultured under superbright red and blue light-emitting diodes (LEDs). J. Hortic. Sci. Biotechnol. 1998, 73, 39–44. [Google Scholar] [CrossRef]
- Nhut, D.T.; Takamura, T.; Watanabe, H.; Okamoto, K.; Tanaka, M. Responses of strawberry plantlets cultured in vitro under superbright red and blue light-emitting diodes (LEDs). Plant Cell Tissue Organ Cult. 2003, 73, 43–52. [Google Scholar] [CrossRef]
- Li, C.-X.; Xu, Z.-G.; Dong, R.-Q.; Chang, S.-X.; Wang, L.-Z.; Khalil-Ur-Rehman, M.; Tao, J.-M. An RNA-Seq analysis of grape plantlets grown in vitro reveals different responses to blue, green, red LED light, and white fluorescent light. Front. Plant Sci. 2017, 8, 78. [Google Scholar] [CrossRef] [PubMed]
- Alvarenga, I.C.A.; Pacheco, F.V.; Silva, S.T.; Bertolucci, S.K.V.; Pinto, J.E.B.P. In vitro culture of Achillea millefolium L.: Quality and intensity of light on growth and production of volatiles. Plant Cell Tissue Organ Cult. 2015, 122, 299–308. [Google Scholar] [CrossRef]
- Xu, Y.; Liang, Y.; Yang, M. Effects of composite LED light on root growth and antioxidant capacity of Cunninghamia lanceolata tissue culture seedlings. Sci. Rep. 2019, 9, 9766. [Google Scholar] [CrossRef] [PubMed]
- Miler, N.; Zalewska, M. The influence of light color on micropropagation of chrysanthemum. Acta Hort. 2006, 725, 347–350. [Google Scholar] [CrossRef]
- Antonopoulou, K.; Dimassi, K.; Therios, I.; Chatzissavvidis, C. The influence of radiation quality on the in vitro rooting and nutrient concentrations of peach rootstock. Biol. Plant 2004, 48, 549–553. [Google Scholar] [CrossRef]
- Wilken, D.; Jiménez Gonzalez, E.; Gerth, A.; Gómez-Kosky, R.; Schumann, A.; Claus, D. Effect of immersion systems, lighting, and TIS designs on biomass increase in micropropagating banana (Musa Spp. Cv.’Grande Naine’AAA). Vitr. Cell. Dev. Biol. Plant. 2014, 50, 582–589. [Google Scholar] [CrossRef]
- Ramírez-Mosqueda, M.; Iglesias-Andreu, L.; Luna-Sánchez, I. Light quality affects growth and development of in vitro plantlet of Vanilla planifolia Jacks. S. Afr. J. Bot. 2017, 109, 288–293. [Google Scholar] [CrossRef]
- Latkowska, M.; Chmiel, H. The influence of light quality and growth regulators on shoot regeneration and rooting in vitro of chrysanthemum (Dendranthema grandiflora) cv. Escort). Akad. Tech.-Rol. W Bydg. Zesz. Nauk. 1996, 197, 129–136. [Google Scholar]
- Śmigielska, M.; Jerzy, M. Adventitious roots and bulbs formation on Hyacinthus orientalis leaf cuttings under different colours of artificial light. Acta Sci. Pol. 2013, 12, 157–164. [Google Scholar]
- Gonçalves, D.S.; Souza, D.M.S.C.; Fernandes, S.B.; Molinari, L.V.; Dorigan, A.F.; Konzen, E.R.; Teixeira, G.L.; Brondani, G.E. Effect of light spectra on in vitro multiplication, elongation and adventitious rooting stages of Bambusa vulgaris Schrad. ex J. C. Wendl. Adv. Bamboo Sci. 2023, 4, 100035. [Google Scholar] [CrossRef]
- Yang, Y.; Zhang, L.; Chen, P.; Liang, T.; Li, X.; Liu, H. UV-B photoreceptor UVR8 interacts with MYB73/MYB77 to regulate auxin responses and lateral root development. EMBO J. 2020, 39, e101928. [Google Scholar] [CrossRef] [PubMed]
- Walker, N.; Jacques, R.; Miginiac, E. Action of light on rooting in vitro and acclimatization of Sequoia sempervirens to soil. Acta Hort. 1987, 212, 289–301. [Google Scholar] [CrossRef]
- Kepenek, K. Photosynthetic effects of light-emitting diode (LED) on in vitro-derived strawberry (Fragaria x Ananassa cv. Festival) plants under in vitro conditions. Erwerbs-Obstbau 2019, 61, 179–187. [Google Scholar] [CrossRef]
- Noè, N.; Eccher, T. Influence of irradiance on in vitro growth and proliferation of Vaccinium corymbosum (highbush blueberry) and subsequent rooting in vivo. Physiol. Plant. 1994, 91, 273–275. [Google Scholar] [CrossRef]
- Li, S.; Zhou, L.; Wu, S.; Liu, L.; Huang, M.; Lin, S.; Ding, G. Effects of LED light on Acacia melanoxylon bud proliferation in vitro and root growth ex vitro. Open Life Sci. 2019, 14, 349–357. [Google Scholar] [CrossRef]
- Mencuccini, M. Effect of medium darkening on in vitro rooting capability and rooting seasonality of olive (Olea europaea L.) cultivars. Sci. Hortic. 2003, 97, 129–139. [Google Scholar] [CrossRef]
- Pan, M.; Van Staden, J. The use of charcoal in in vitro culture-A review. Plant Growth Regul. 2003, 26, 155–163. [Google Scholar] [CrossRef]
- Hammerschlag, A.G. Factors influencing in vitro multiplication and rooting of the plum rootstock ‘Myrobalan’ (Prunus cerasifera Ehrh.). J. Am. Soc. Hortic. Sci. 1982, 107, 44–47. [Google Scholar] [CrossRef]
- Channuntapipat, C.; Sedgley, M.; Collins, G. Micropropagation of almond cultivars Nonpareil and Ne Plus Ultra and the hybrid rootstock Titan x Nemaguard. Sci. Hortic. 2003, 98, 473–484. [Google Scholar] [CrossRef]
- Rugini, E.; Fedeli, E. Olive (Olea europaea L.) as an oilseed crop. In Legumes and 1887 Oilseed Crops I, Biotechnology in Agriculture and Forestry; Bajaj, Y.P.S., Ed.; Springer: Berlin/Heidelberg, Germany, 1990; pp. 593–641. [Google Scholar] [CrossRef]
- Bassuk, N.; Maynard, B. Stock plant etiolation. Hort.Sci. 1987, 22, 749–750. [Google Scholar] [CrossRef]
- Druart, P.; Kevers, C.; Boxus, P.; Gaspar, T. In vitro promotion of root formation by apple shoots through darkness effect on endogenous phenols and peroxidases. Z. Für Pflanzenphysiol. 1982, 108, 429–436. [Google Scholar] [CrossRef]
- Seon, J.H.; Cui, Y.Y.; Kozai, T.; Paek, K.Y. Influence of in vitro growth conditions on photosynthetic competence and survival rate of Rehmannia glutinosa plantlets during acclimatization period. Plant Cell Tiss. Organ Cult. 2000, 61, 135–142. [Google Scholar] [CrossRef]
- Fan, C.; Manivannan, A.; Wei, H. Light quality-mediated influence of morphogenesis in micropropagated horticultural crops: A comprehensive overview. Biomed Res Int. 2022, 2022, 4615079. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, P.H.V.; Arruda, F.; Forti, V.A. Slow-grown in vitro conservation of Heliconia champneiana cv. Splash under different light spectra. Sci. Agric. 2018, 75, 163–166. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muleo, R.; Hassan, M.I.; Pellegrino, A.; Cavallaro, V. Effects of Light on Adventitious Rooting In Vitro. Agronomy 2025, 15, 2597. https://doi.org/10.3390/agronomy15112597
Muleo R, Hassan MI, Pellegrino A, Cavallaro V. Effects of Light on Adventitious Rooting In Vitro. Agronomy. 2025; 15(11):2597. https://doi.org/10.3390/agronomy15112597
Chicago/Turabian StyleMuleo, Rosario, Mohamed I. Hassan, Alessandra Pellegrino, and Valeria Cavallaro. 2025. "Effects of Light on Adventitious Rooting In Vitro" Agronomy 15, no. 11: 2597. https://doi.org/10.3390/agronomy15112597
APA StyleMuleo, R., Hassan, M. I., Pellegrino, A., & Cavallaro, V. (2025). Effects of Light on Adventitious Rooting In Vitro. Agronomy, 15(11), 2597. https://doi.org/10.3390/agronomy15112597







