Vermicompost: Enhancing Plant Growth and Combating Abiotic and Biotic Stress
Abstract
1. Introduction
2. Earthworm Impact
2.1. Soil Structure
2.2. Organic Matter Decomposition
2.3. Soil Fertility
2.4. Soil Microorganisms
3. Vermicompost and Its Derivatives as a Plant Growth Promoter
3.1. Humic Substances
3.2. Phytohormones
3.3. Microbial Activity
4. Effects of Vermicompost as Bio-Alleviator of Abiotic and Biotic Stress
4.1. Soil Salinity
4.2. Drought Stress
4.3. Insect Pests
4.4. Plant Diseases
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Erisman, J.W.; Sutton, M.A.; Galloway, J.; Klimont, Z.; Winiwarter, W. How a century of ammonia synthesis changed the world. Nat. Geosci. 2008, 1, 636–639. [Google Scholar] [CrossRef]
- Kumar, A.; Bohra, B. Green technology in relation to sustainable agriculture. In Green Technologies for Sustainable Agriculture; Daya Publishing House: Delhi, India, 2006. [Google Scholar]
- De Castro, F.; Vergaro, V.; Benedetti, M.; Baldassarre, F.; Del Coco, L.; Dell’Anna, M.M.; Mastrorilli, P.; Fanizzi, F.P.; Ciccarella, G. Visible light-activated water-soluble platicur nanocolloids: Photocytotoxicity and metabolomics studies in cancer cells. ACS Appl. 2020, 3, 6836–6851. [Google Scholar] [CrossRef] [PubMed]
- De Castro, F.; Stefàno, E.; Migoni, D.; Iaconisi, G.N.; Muscella, A.; Marsigliante, S.; Benedetti, M.; Fanizzi, F.P. Synthesis and Evaluation of the Cytotoxic Activity of Water-Soluble Cationic Organometallic Complexes of the Type [Pt (η1−C2H4OMe)(L)(Phen)]+(L = NH3, DMSO; Phen = 1,10-Phenanthroline). Pharmaceutics 2021, 13, 642. [Google Scholar] [CrossRef] [PubMed]
- Benedetti, M.; Antonucci, D.; De Castro, F.; Girelli, C.R.; Lelli, M.; Roveri, N.; Fanizzi, F.P. Metalated nucleotide chemisorption on hydroxyapatite. J. Inorg. Biochem. 2015, 153, 279–283. [Google Scholar] [CrossRef]
- Ravindran, B.; Wong, J.W.; Selvam, A.; Sekaran, G. Influence of microbial diversity and plant growth hormones in compost and vermicompost from fermented tannery waste. Bioresour. Technol. 2016, 217, 200–204. [Google Scholar] [CrossRef]
- Datta, S.; Singh, J.; Singh, S.; Singh, J. Earthworms, pesticides and sustainable agriculture: A review. Environ. Sci. Pollut. Res. 2016, 23, 8227–8243. [Google Scholar] [CrossRef]
- Tripathi, Y.; Hazaria, P.; Kaushik, P.; Kumar, A. Vermitechnology and waste management. In Verms and Vermitechnology, SB Nangia; APH Publishing Corp: New Delhi, India, 2005; pp. 9–21. [Google Scholar]
- Coria-Cayupaán, Y.S.; de Pinto, M.A.I.S.; Nazareno, M.A. Variations in bioactive substance contents and crop yields of lettuce (Lactuca sativa L.) cultivated in soils with different fertilization treatments. J. Agric. Food Chem. 2009, 57, 10122–10129. [Google Scholar] [CrossRef] [PubMed]
- Goutam, K.C.; Goutam, B.; Susanta, K.C. The effect of vermicompost and other fertilizers on cultivation of tomato plants. J. Hortic. For. 2011, 3, 42–45. [Google Scholar]
- Ruz-Jerez, B.; Ball, P.R.; Tillman, R. Laboratory assessment of nutrient release from a pasture soil receiving grass or clover residues, in the presence or absence of Lumbricus rubellus or Eisenia fetida. Soil Biol. Biochem. 1992, 24, 1529–1534. [Google Scholar] [CrossRef]
- Parkin, T.B.; Berry, E.C. Nitrogen transformations associated with earthworm casts. Soil Biol. Biochem. 1994, 26, 1233–1238. [Google Scholar] [CrossRef]
- Adhikary, S. Vermicompost, the story of organic gold: A review. Agric. Sci. 2012, 3, 905–917. [Google Scholar] [CrossRef]
- Atiyeh, R.; Arancon, N.; Edwards, C.; Metzger, J. Influence of earthworm-processed pig manure on the growth and yield of greenhouse tomatoes. Bioresour. Technol. 2000, 75, 175–180. [Google Scholar] [CrossRef]
- Yatoo, A.M.; Rasool, S.; Ali, S.; Majid, S.; Rehman, M.U.; Ali, M.; Eachkoti, R.; Rasool, S.; Rashid, S.M.; Farooq, S. Vermicomposting: An eco-friendly approach for recycling/management of organic wastes. In Bioremediation and Biotechnology; Springer: Cham, Switzerland, 2020; pp. 167–187. [Google Scholar]
- Ayyobi, H.; Hassanpour, E.; Alaqemand, S.; Fathi, S.; Olfati, J.; Peyvast, G. Vermicompost leachate and vermiwash enhance French dwarf bean yield. Int. J. Veg. Sci. 2014, 20, 21–27. [Google Scholar] [CrossRef]
- Bidabadi, S.S. Waste management using vermicompost derived liquids in sustainable horticulture. Trends Hortic. 2018, 1, 175. [Google Scholar]
- Van Oosten, M.J.; Pepe, O.; De Pascale, S.; Silletti, S.; Maggio, A. The role of biostimulants and bioeffectors as alleviators of abiotic stress in crop plants. Chem. Biol. 2017, 4, 5. [Google Scholar] [CrossRef]
- Puglisi, E.; Pascazio, S.; Suciu, N.; Cattani, I.; Fait, G.; Spaccini, R.; Crecchio, C.; Piccolo, A.; Trevisan, M. Rhizosphere microbial diversity as influenced by humic substance amendments and chemical composition of rhizodeposits. J. Geochem. Explor. 2013, 129, 82–94. [Google Scholar] [CrossRef]
- Krishnamoorthy, R.; Vajranabhaiah, S. Biological activity of earthworm casts: An assessment of plant growth promotor levels in the casts. Proc. Anim. Sci. 1986, 95, 341–351. [Google Scholar] [CrossRef]
- Amooaghaie, R.; Golmohammadi, S. Effect of vermicompost on growth, essential oil, and health of Thymus Vulgaris. Compos. Sci. Util. 2017, 25, 166–177. [Google Scholar] [CrossRef]
- Sahab, S.; Suhani, I.; Srivastava, V.; Chauhan, P.S.; Singh, R.P.; Prasad, V. Potential risk assessment of soil salinity to agroecosystem sustainability: Current status and management strategies. Sci. Total Environ. 2021, 764, 144164. [Google Scholar] [CrossRef]
- Ahmad, A.; Aslam, Z.; Hussain, D.; Bellitürk, K.; Javed, T.; Hussain, S.; Bashir, S.; Raza, A.; Alotaibi, S.; Kalaji, H.M. Rice straw vermicompost enriched with cellulolytic microbes ameliorate the negative effect of drought in wheat through modulating the morpho-physiological attributes. Front. Environ. Sci. 2022, 10, 497. [Google Scholar] [CrossRef]
- Basco, M.; Bisen, K.; Keswani, C.; Singh, H. Biological management of Fusarium wilt of tomato using biofortified vermicompost. Mycosphere 2017, 8, 467–483. [Google Scholar] [CrossRef]
- Manandhar, T.; Yami, K. Biological control of foot rot disease of rice using fermented products of compost and vermicompost. Sci. World J. 2008, 6, 52–57. [Google Scholar] [CrossRef]
- Pathma, J.; Sakthivel, N. Microbial diversity of vermicompost bacteria that exhibit useful agricultural traits and waste management potential. SpringerPlus 2012, 1, 26. [Google Scholar] [CrossRef]
- Pattnaik, S.; Reddy, M.V. Nutrient status of vermicompost of urban green waste processed by three earthworm species—Eisenia fetida, Eudrilus eugeniae, and Perionyx excavatus. Appl. Environ. Soil Sci. 2010, 2010, 967526. [Google Scholar] [CrossRef]
- Ahmed, N.; Al-Mutairi, K.A. Earthworms Effect on Microbial Population and Soil Fertility as Well as Their Interaction with Agriculture Practices. Sustainability 2022, 14, 7803. [Google Scholar] [CrossRef]
- Fragoso, C.; Kanyonyo, J.; Moreno, A.; Senapati, B.K.; Blanchart, E.; Rodriguez, C. A survey of tropical earthworms: Taxonomy, biogeography and environmental plasticity. Earthworm Manag. Trop. Agroecosyst. 1999, 1–26. [Google Scholar]
- Eggleton, P.; Inward, K.; Smith, J.; Jones, D.T.; Sherlock, E. A six year study of earthworm (Lumbricidae) populations in pasture woodland in southern England shows their responses to soil temperature and soil moisture. Soil Biol. Biochem. 2009, 41, 1857–1865. [Google Scholar] [CrossRef]
- Vivas, A.; Moreno, B.; Garcia-Rodriguez, S.; Benitez, E. Assessing the impact of composting and vermicomposting on bacterial community size and structure, and microbial functional diversity of an olive-mill waste. Bioresour. Technol. 2009, 100, 1319–1326. [Google Scholar] [CrossRef] [PubMed]
- Gopal, M.; Bhute, S.S.; Gupta, A.; Prabhu, S.; Thomas, G.V.; Whitman, W.B.; Jangid, K. Changes in structure and function of bacterial communities during coconut leaf vermicomposting. Antonie Van Leeuwenhoek 2017, 110, 1339–1355. [Google Scholar] [CrossRef]
- Edwards, C.A.; Bohlen, P.J. Biology and Ecology of Earthworms; Springer Science & Business Media: London, UK, 1996; Volume 3. [Google Scholar]
- Rabot, E.; Wiesmeier, M.; Schlüter, S.; Vogel, H.-J. Soil Structure as an Indicator of Soil Functions: A Review. Geoderma 2018, 314, 122–137. [Google Scholar] [CrossRef]
- Carpenter, D.; Hodson, M.E.; Eggleton, P.; Kirk, C. Earthworm induced mineral weathering: Preliminary results. Eur. J. Soil Biol. 2007, 43, S176–S183. [Google Scholar] [CrossRef]
- Al-Maliki, S.; Scullion, J. Interactions between earthworms and residues of differing quality affecting aggregate stability and microbial dynamics. Appl. Soil Ecol. 2013, 64, 56–62. [Google Scholar] [CrossRef]
- Ritsema, C.J.; Dekker, L. Preferential flow in water repellent sandy soils: Principles and modeling implications. J. Hydrol. 2000, 231, 308–319. [Google Scholar] [CrossRef]
- Atiyeh, R.M.; Domínguez, J.; Subler, S.; Edwards, C.A. Changes in biochemical properties of cow manure during processing by earthworms (Eisenia andrei, Bouché) and the effects on seedling growth. Pedobiologia 2000, 44, 709–724. [Google Scholar] [CrossRef]
- Lavelle, P.; Charpentier, F.; Villenave, C.; Rossi, J.-P.; Derouard, L.; Pashanasi, B.; André, J.; Ponge, J.-F.; Bernier, N. Effects of earthworms on soil organic matter and nutrient dynamics at a landscape scale over decades. Earthworm Ecol. 2004, 2, 145–160. [Google Scholar]
- Alegre, J.; Pashanasi, B.; Lavelle, P. Dynamics of soil physical properties in Amazonian agroecosystems inoculated with earthworms. Soil Sci. Soc. Am. J. 1996, 60, 1522–1529. [Google Scholar] [CrossRef]
- Ernst, G.; Henseler, I.; Felten, D.; Emmerling, C. Decomposition and mineralization of energy crop residues governed by earthworms. Soil Biol. Biochem. 2009, 41, 1548–1554. [Google Scholar] [CrossRef]
- Lubbers, I.M.; Pulleman, M.M.; Van Groenigen, J.W. Can earthworms simultaneously enhance decomposition and stabilization of plant residue carbon? Soil Biol. Biochem. 2017, 105, 12–24. [Google Scholar] [CrossRef]
- Aira, M.; Domínguez, J. Earthworm effects without earthworms: Inoculation of raw organic matter with worm-worked substrates alters microbial community functioning. PLoS ONE 2011, 6, e16354. [Google Scholar] [CrossRef] [PubMed]
- Aira, M.; Monroy, F.; Domínguez, J. Changes in bacterial numbers and microbial activity of pig slurry during gut transit of epigeic and anecic earthworms. J. Hazard. Mater. 2009, 162, 1404–1407. [Google Scholar] [CrossRef] [PubMed]
- Chapuis-Lardy, L.; Bayon, R.-C.L.; Brossard, M.; López-Hernández, D.; Blanchart, E. Role of soil macrofauna in phosphorus cycling. In Phosphorus in Action; Springer: Berlin/Heidelberg, Germany, 2011; pp. 199–213. [Google Scholar]
- Blouin, M.; Hodson, M.E.; Delgado, E.A.; Baker, G.; Brussaard, L.; Butt, K.R.; Dai, J.; Dendooven, L.; Pérès, G.; Tondoh, J. A review of earthworm impact on soil function and ecosystem services. Eur. J. Soil Sci. 2013, 64, 161–182. [Google Scholar] [CrossRef]
- Seeber, J.; Seeber, G.; Langel, R.; Scheu, S.; Meyer, E. The effect of macro-invertebrates and plant litter of different quality on the release of N from litter to plant on alpine pastureland. Biol. Fertil. Soils 2008, 44, 783–790. [Google Scholar] [CrossRef]
- Sampedro, L.; Domínguez, J. Stable isotope natural abundances (δ13C and δ15N) of the earthworm Eisenia fetida and other soil fauna living in two different vermicomposting environments. Appl. Soil Ecol. 2008, 38, 91–99. [Google Scholar] [CrossRef]
- Thakuria, D.; Schmidt, O.; Finan, D.; Egan, D.; Doohan, F.M. Gut wall bacteria of earthworms: A natural selection process. ISME J. 2010, 4, 357–366. [Google Scholar] [CrossRef]
- De Menezes, A.B.; Prendergast-Miller, M.T.; Macdonald, L.M.; Toscas, P.; Baker, G.; Farrell, M.; Wark, T.; Richardson, A.E.; Thrall, P.H. Earthworm-induced shifts in microbial diversity in soils with rare versus established invasive earthworm populations. FEMS Microbiol. Ecol. 2018, 94, fiy051. [Google Scholar] [CrossRef]
- Fujii, K.; Ikeda, K.; Yoshida, S. Isolation and characterization of aerobic microorganisms with cellulolytic activity in the gut of endogeic earthworms. Int. Microbiol. 2012, 15, 121–130. [Google Scholar] [PubMed]
- Johnsen, A.R.; Wick, L.Y.; Harms, H. Principles of microbial PAH-degradation in soil. Environ. Pollut. 2005, 133, 71–84. [Google Scholar] [CrossRef]
- Chan, K.; Baker, G.; Conyers, M.; Scott, B.; Munro, K. Complementary ability of three European earthworms (Lumbricidae) to bury lime and increase pasture production in acidic soils of south-eastern Australia. Appl. Soil Ecol. 2004, 26, 257–271. [Google Scholar] [CrossRef]
- Van Groenigen, J.W.; Van Groenigen, K.J.; Koopmans, G.F.; Stokkermans, L.; Vos, H.M.; Lubbers, I.M. How fertile are earthworm casts? A meta-analysis. Geoderma 2019, 338, 525–535. [Google Scholar] [CrossRef]
- Le Bayon, R.-C.; Milleret, R. Effects of earthworms on phosphorus dynamics–a review. Dyn. Soil Dyn. Plant 2009, 3, 21–27. [Google Scholar]
- Nuutinen, V.; Pitkänen, J.; Kuusela, E.; Widbom, T.; Lohilahti, H. Spatial variation of an earthworm community related to soil properties and yield in a grass–clover field. Appl. Soil Ecol. 1998, 8, 85–94. [Google Scholar] [CrossRef]
- Curry, J.P.; Schmidt, O. The feeding ecology of earthworms–a review. Pedobiologia 2007, 50, 463–477. [Google Scholar] [CrossRef]
- Neilson, R.; Boag, B. Feeding preferences of some earthworm species common to upland pastures in Scotland. Pedobiologia 2003, 47, 1–8. [Google Scholar] [CrossRef]
- Lowe, C.N.; Butt, K.R. Influence of food particle size on inter-and intra-specific interactions of Allolobophora chlorotica (Savigny) and Lumbricus terrestris: The 7th international symposium on earthworm ecology Cardiff Wales·2002. Pedobiologia 2003, 47, 574–577. [Google Scholar]
- Satchell, J.E. Lumbricidae. In Soil Biology; Burges, A., Raw, F., Eds.; Academic Press: London, UK, 1967; pp. 259–322. [Google Scholar]
- Moody, S.; Briones, M.; Piearce, T.; Dighton, J. Selective consumption of decomposing wheat straw by earthworms. Soil Biol. Biochem. 1995, 27, 1209–1213. [Google Scholar] [CrossRef]
- Drake, H.L.; Horn, M.A. As the worm turns: The earthworm gut as a transient habitat for soil microbial biomes. Annu. Rev. Microbiol. 2007, 61, 169–189. [Google Scholar] [CrossRef] [PubMed]
- Tiwari, S.; Tiwari, B.; Mishra, R. Microbial populations, enzyme activities and nitrogen-phosphorus-potassium enrichment in earthworm casts and in the surrounding soil of a pineapple plantation. Biol. Fertil. Soils 1989, 8, 178–182. [Google Scholar] [CrossRef]
- Decaëns, T.; Rangel, A.; Asakawa, N.; Thomas, R.J. Carbon and nitrogen dynamics in ageing earthworm casts in grasslands of the eastern plains of Colombia. Biol. Fertil. Soils 1999, 30, 20–28. [Google Scholar] [CrossRef]
- Brossard, M.; Lavelle, P.; Laurent, J.-Y. Digestion of a vertisol by the endogeic earthworm Polypheretima elongata, Megascolecidae, increases soil phosphate extractibility. Eur. J. Soil Biol. 1996, 32, 107. [Google Scholar]
- Edwards, C.; Lofty, J. Biology of Earthworms; Chapman and Hall: London, UK, 1977. [Google Scholar]
- Hong, S.W.; Lee, J.S.; Chung, K.S. Effect of enzyme producing microorganisms on the biomass of epigeic earthworms (Eisenia fetida) in vermicompost. Bioresour. Technol. 2011, 102, 6344–6347. [Google Scholar] [CrossRef]
- Schönholzer, F.; Hahn, D.; Zeyer, J. Origins and fate of fungi and bacteria in the gut of Lumbricus terrestris L. studied by image analysis. FEMS Microbiol. Ecol. 1999, 28, 235–248. [Google Scholar] [CrossRef]
- Pedersen, J.C.; Hendriksen, N.B. Effect of passage through the intestinal tract of detritivore earthworms (Lumbricus spp.) on the number of selected Gram-negative and total bacteria. Biol. Fertil. Soils 1993, 16, 227–232. [Google Scholar] [CrossRef]
- Monroy, F.; Aira, M.; Domínguez, J. Reduction of total coliform numbers during vermicomposting is caused by short-term direct effects of earthworms on microorganisms and depends on the dose of application of pig slurry. Sci. Total Environ. 2009, 407, 5411–5416. [Google Scholar] [CrossRef] [PubMed]
- Brown, G.G.; Barois, I.; Lavelle, P. Regulation of soil organic matter dynamics and microbial activityin the drilosphere and the role of interactionswith other edaphic functional domains. Eur. J. Soil Biol. 2000, 36, 177–198. [Google Scholar] [CrossRef]
- Hoeffner, K.; Monard, C.; Santonja, M.; Cluzeau, D. Feeding behaviour of epi-anecic earthworm species and their impacts on soil microbial communities. Soil Biol. Biochem. 2018, 125, 1–9. [Google Scholar] [CrossRef]
- Loera-Muro, A.; Troyo-Diéguez, E.; Murillo-Amador, B.; Barraza, A.; Caamal-Chan, G.; Lucero-Vega, G.; Nieto-Garibay, A. Effects of vermicompost leachate versus inorganic fertilizer on morphology and microbial traits in the early development growth stage in mint (Mentha spicata L.) and rosemary (Rosmarinus officinalis L.) plants under closed hydroponic system. Horticulturae 2021, 7, 100. [Google Scholar] [CrossRef]
- Bidabadi, S.S.; Dehghanipoodeh, S.; Wright, G.C. Vermicompost leachate reduces some negative effects of salt stress in pomegranate. Int. J. Recycl. Org. Waste Agric. 2017, 6, 255–263. [Google Scholar] [CrossRef]
- Suthar, S. Evidence of plant hormone like substances in vermiwash: An ecologically safe option of synthetic chemicals for sustainable farming. Ecol. Eng. 2010, 36, 1089–1092. [Google Scholar] [CrossRef]
- Kaur, A.; Pati, P.K.; Ohri, P.; Kaur, A. Effects of Vermicompost and Vermicompost Leachate on the Biochemical and Physiological Response of Withania somnifera (L.) Dunal. J. Soil Sci. Plant Nutr. 2022, 22, 3228–3242. [Google Scholar] [CrossRef]
- Aslam, Z.; Ahmad, A.; Bellitürk, K.; Iqbal, N.; Idrees, M.; Rehman, W.U.; Akbar, G.; Tariq, M.; Raza, M.; Riasat, S. 26. Effects of vermicompost, vermi-tea and chemical fertilizer on morpho-physiological characteristics of tomato (Solanum lycopersicum) in Suleymanpasa District, Tekirdag of Turkey. Pure Appl. Biol. 2020, 9, 1920–1931. [Google Scholar] [CrossRef]
- Gong, X.; Li, S.; Sun, X.; Wang, L.; Cai, L.; Zhang, J.; Wei, L. Green waste compost and vermicompost as peat substitutes in growing media for geranium (Pelargonium zonale L.) and calendula (Calendula officinalis L.). Sci. Hortic. 2018, 236, 186–191. [Google Scholar] [CrossRef]
- Yadav, A.; Garg, V. Influence of vermi-fortification on chickpea (Cicer arietinum L.) growth and photosynthetic pigments. Int. J. Recycl. Org. Waste Agric. 2015, 4, 299–305. [Google Scholar] [CrossRef]
- Rekha, G.S.; Kaleena, P.K.; Elumalai, D.; Srikumaran, M.P.; Maheswari, V.N. Effects of vermicompost and plant growth enhancers on the exo-morphological features of Capsicum annum (Linn.) Hepper. Int. J. Recycl. Org. Waste Agric. 2018, 7, 83–88. [Google Scholar] [CrossRef]
- Arancon, N.Q.; Owens, J.D.; Converse, C. The effects of vermicompost tea on the growth and yield of lettuce and tomato in a non-circulating hydroponics system. J. Plant Nutr. 2019, 42, 2447–2458. [Google Scholar] [CrossRef]
- Purwanto, A. Development of technology vermicompost production for the coffee plant Industry. J. Phys. Conf. Ser. 2021, 1876, 012020. [Google Scholar]
- Sundararasu, K.; Jeyasankar, A. Effect of vermiwash on growth and yield of brinjal, Solanum melongena (eggplant or aubergine). Asian J. Sci. Technol. 2014, 5, 171–173. [Google Scholar]
- Ravindran, B.; Lee, S.R.; Chang, S.W.; Nguyen, D.D.; Chung, W.J.; Balasubramanian, B.; Mupambwa, H.A.; Arasu, M.V.; Al-Dhabi, N.A.; Sekaran, G. Positive effects of compost and vermicompost produced from tannery waste-animal fleshing on the growth and yield of commercial crop-tomato (Lycopersicon esculentum L.) plant. J. Environ. Manag. 2019, 234, 154–158. [Google Scholar] [CrossRef]
- Mahmud, M.; Abdullah, R.; Yaacob, J.S. Effect of vermicompost on growth, plant nutrient uptake and bioactivity of ex vitro pineapple (Ananas comosus var. MD2). Agronomy 2020, 10, 1333. [Google Scholar] [CrossRef]
- Ose, A.; Andersone-Ozola, U.; Ievinsh, G. Substrate-Dependent Effect of Vermicompost on Yield and Physiological Indices of Container-Grown Dracocephalum moldavica Plants. Agriculture 2021, 11, 1231. [Google Scholar] [CrossRef]
- Aslam, Z.; Aljuaid, B.S.; Abbas, R.N.; Bashir, S.; Almas, M.H.; Awan, T.H.; Belliturk, K.; Al-Taisan, W.a.A.; Mahmoud, S.F.; Bashir, S. Reduction in the Allelopathic Potential of Conocarpus erectus L. through Vermicomposting. Sustainability 2022, 14, 12840. [Google Scholar]
- Bziouech, S.A.; Dhen, N.; Helaoui, S.; Ammar, I.B.; Dridi, B.A.M. Effect of vermicompost soil additive on growth performance, physiological and biochemical responses of tomato plants (Solanum lycopersicum L. var. Firenze) to salt stress. Emir. J. Food Agric. 2022, 34, 316–328. [Google Scholar]
- Olivares, F.L.; Busato, J.G.; de Paula, A.M.; da Silva Lima, L.; Aguiar, N.O.; Canellas, L.P. Plant growth promoting bacteria and humic substances: Crop promotion and mechanisms of action. Chem. Biol 2017, 4, 30. [Google Scholar] [CrossRef]
- Canellas, L.P.; Zandonadi, D.B.; Busato, J.G.; Baldotto, M.A.; Simões, M.L.; Martin-Neto, L.; Façanha, A.R.; Spaccini, R.; Piccolo, A. Bioactivity and chemical characteristics of humic acids from tropical soils sequence. Soil Sci. 2008, 173, 624–637. [Google Scholar] [CrossRef]
- Nardi, S.; Ertani, A.; Francioso, O. Soil–root cross-talking: The role of humic substances. J. Plant Nutr. Soil Sci. 2017, 180, 5–13. [Google Scholar] [CrossRef]
- Singh, R.; Sharma, R.; Kumar, S.; Gupta, R.; Patil, R. Vermicompost substitution influences growth, physiological disorders, fruit yield and quality of strawberry (Fragaria x ananassa Duch.). Bioresour. Technol. 2008, 99, 8507–8511. [Google Scholar] [CrossRef]
- Garcia-Mina, J.; Antolin, M.; Sanchez-Diaz, M. Metal-humic complexes and plant micronutrient uptake: A study based on different plant species cultivated in diverse soil types. Plant Soil 2004, 258, 57–68. [Google Scholar] [CrossRef]
- Zanin, L.; Tomasi, N.; Cesco, S.; Varanini, Z.; Pinton, R. Humic substances contribute to plant iron nutrition acting as chelators and biostimulants. Front. Plant Sci. 2019, 10, 675. [Google Scholar] [CrossRef]
- Aguiar, N.O.; Olivares, F.L.; Novotny, E.H.; Dobbss, L.B.; Balmori, D.M.; Santos-Júnior, L.G.; Chagas, J.G.; Façanha, A.R.; Canellas, L.P. Bioactivity of humic acids isolated from vermicomposts at different maturation stages. Plant Soil 2013, 362, 161–174. [Google Scholar] [CrossRef]
- Dobbss, L.B.; Canellas, L.P.; Olivares, F.L.; Aguiar, N.O.; Peres, L.E.P.; Azevedo, M.; Spaccini, R.; Piccolo, A.; Façanha, A.R. Bioactivity of chemically transformed humic matter from vermicompost on plant root growth. J. Agric. Food Chem. 2010, 58, 3681–3688. [Google Scholar] [CrossRef] [PubMed]
- Ievinsh, G.; Vikmane, M.; Ķirse, A.; Karlsons, A. Effect of vermicompost extract and vermicompost-derived humic acids on seed germination and seedling growth of hemp. Proc. Latv. Acad. Sci. 2017, 71, 286–292. [Google Scholar] [CrossRef]
- Gomes, G.A.; Pereira, R.A.; Sodré, G.A.; Gross, E. Humic acids from vermicompost positively influence the nutrient uptake in mangosteen seedlings. Pesqui. Agropecu. Trop. 2019, 49. [Google Scholar]
- El-Hameid, A.; Adel, S. Improving productivity and quality of mango using humic acid and vermicomposting leachate in North Sinai. Egypt. J. Desert Res. 2018, 68, 37–59. [Google Scholar] [CrossRef]
- Rosa, M.; Olimpo, A.; Fastuca, R.; Caprioglio, A. Perceptions of dental professionals and laypeople to altered dental esthetics in cases with congenitally missing maxillary lateral incisors. Prog. Orthod. 2013, 14, 34. [Google Scholar] [CrossRef] [PubMed]
- Balmori, D.; Domínguez, C.; Carreras, C.; Rebatos, S.; Farías, L.; Izquierdo, F.; Berbara, R.; García, A.C. Foliar application of humic liquid extract from vermicompost improves garlic (Allium sativum L.) production and fruit quality. Int. J. Recycl. Org. Waste Agric. 2019, 8, 103–112. [Google Scholar] [CrossRef]
- Baldotto, L.E.B.; Baldotto, M.A.; Giro, V.B.; Canellas, L.P.; Olivares, F.L.; Bressan-Smith, R. Desempenho do abacaxizeiro’Vitória’em resposta à aplicação de ácidos húmicos durante a aclimatação. Rev. Bras. Ciência Solo 2009, 33, 979–990. [Google Scholar] [CrossRef]
- Nikbakht, A.; Kafi, M.; Babalar, M.; Xia, Y.P.; Luo, A.; Etemadi, N.-a. Effect of humic acid on plant growth, nutrient uptake, and postharvest life of gerbera. J. Plant Nutr. 2008, 31, 2155–2167. [Google Scholar] [CrossRef]
- Hernández, A.; Castillo, H.; Ojeda, D.; Arras, A.; López, J.; Sánchez, E. Effect of vermicompost and compost on lettuce production. Chil. J. Agric. Res. 2010, 70, 583–589. [Google Scholar] [CrossRef]
- Subler, S.; Edwards, C.; Metzger, J. Comparing vermicomposts and composts. Biocycle 1998, 39, 63–66. [Google Scholar]
- Sinha, R.K.; Hahn, G.; Singh, P.K.; Suhane, R.K.; Allam, A. Organic farming by vermiculture: Producing safe, nutritive and protective foods by earthworms (Charles Darwin’s friends of farmers). Am. J. Exp. Agric. 2011, 1, 363–399. [Google Scholar] [CrossRef]
- Zhang, H.; Tan, S.; Wong, W.; Ng, C.; Teo, C.; Ge, L.; Chen, X.; Yong, J. Mass spectrometric evidence for the occurrence of plant growth promoting cytokinins in vermicompost tea. Biol. Fertil. Soils 2014, 50, 401–403. [Google Scholar] [CrossRef]
- Aremu, A.O.; Stirk, W.A.; Kulkarni, M.G.; Tarkowská, D.; Turečková, V.; Gruz, J.; Šubrtová, M.; Pěnčík, A.; Novák, O.; Doležal, K. Evidence of phytohormones and phenolic acids variability in garden-waste-derived vermicompost leachate, a well-known plant growth stimulant. J. Plant Growth Regul. 2015, 75, 483–492. [Google Scholar] [CrossRef]
- Li, Z.; He, Y. Roles of brassinosteroids in plant reproduction. Int. J. Mol. Sci. 2020, 21, 872. [Google Scholar] [CrossRef] [PubMed]
- Wong, W.; Tan, S.; Ge, L.; Chen, X.; Letham, D.; Yong, J. The importance of phytohormones and microbes in biostimulants: Mass spectrometric evidence and their positive effects on plant growth. In Proceedings of the II World Congress on the Use of Biostimulants in Agriculture 1148, Florence, Italy, 16–19 November 2015; pp. 49–60. [Google Scholar]
- Steffen, G.P.K.; Maldaner, J.; de Morais, R.M.; Saldanha, C.W.; Missio, E.L.; Steffen, R.B.; Osorio Filho, B.D. The vermicompost anticipates flowering and increases tomato productivity. Agrocienc. Urug. 2019, 23, e54. [Google Scholar] [CrossRef]
- Sardoei, A. Vermicompost effects on the growth and flowering of marigold (Calendula officinalis). Eur. J. Exp. Biol. 2014, 4, 651–655. [Google Scholar]
- Arora, V.; Singh, C.; Sidhu, A.; Thind, S. Irrigation, tillage and mulching effects on soybean yield and water productivity in relation to soil texture. Agric. Water Manag. 2011, 98, 563–568. [Google Scholar] [CrossRef]
- Lazcano, C.; Domínguez, J. The use of vermicompost in sustainable agriculture: Impact on plant growth and soil fertility. Soil Nutr. 2011, 10, 187. [Google Scholar]
- Ansari, A.; Ismail, S. Role of Earthworms in vermitechnology. J. Agric. Technol. 2012, 8, 405–415. [Google Scholar]
- Song, X.; Liu, M.; Wu, D.; Griffiths, B.S.; Jiao, J.; Li, H.; Hu, F. Interaction matters: Synergy between vermicompost and PGPR agents improves soil quality, crop quality and crop yield in the field. Appl. Soil Ecol. 2015, 89, 25–34. [Google Scholar] [CrossRef]
- Karnwal, A. Pseudomonas spp., a zinc-solubilizing vermicompost bacteria with plant growth-promoting activity moderates zinc biofortification in tomato. Int. J. Veg. Sci. 2021, 27, 398–412. [Google Scholar] [CrossRef]
- Ahmad, A.; Aslam, Z.; Bellitürk, K.; Hussain, S.; Bibi, I. Soil Application of Cellulolytic Microbe–Enriched Vermicompost Modulated the Morpho-physiological and Biochemical Responses of Wheat Cultivars under Different Moisture Regimes. J. Soil Sci. Plant Nutr 2022, 22, 4153–4167. [Google Scholar] [CrossRef]
- Shrimal, P.; Khan, T. Studies on the effects of vermicompost on growth parameters and chlorophyll content of bengal gram (Cicer arietinum L.) var. RSG-896. IOSR J. Environ. Sci. Toxicol. Food Technol. 2017, 11, 12–16. [Google Scholar] [CrossRef]
- Fikru, T.K.; Fikreyohannes, G. Response of garlic (Allium sativum L.) to vermicompost and mineral N fertilizer application at Haramaya, Eastern Ethiopia. Afr. J. Food Agric. Res. 2018, 13, 27–35. [Google Scholar] [CrossRef]
- Baliah, T.; Muthulakshmi, P. Effect of microbially enriched vermicompost on the growth and biochemical characteristics of Okra (Abelmoschus esculentus (L.) Moench). Adv. Plants Agric. Res 2017, 6, 00228. [Google Scholar]
- Parastesh, F.; Alikhani, H.A.; Etesami, H. Vermicompost enriched with phosphate–solubilizing bacteria provides plant with enough phosphorus in a sequential cropping under calcareous soil conditions. J. Clean. Prod. 2019, 221, 27–37. [Google Scholar] [CrossRef]
- Verma, S.; Nizam, S.; Verma, P.K. Biotic and abiotic stress signaling in plants. In Stress Signaling in Plants: Genomics and Proteomics Perspective; Springer: New York, NY, USA, 2013; Volume 1, pp. 25–49. [Google Scholar]
- Sharif, F.; Danish, M.U.; Ali, A.S.; Khan, A.U.; Shahzad, L.; Ali, H.; Ghafoor, A. Salinity tolerance of earthworms and effects of salinity and vermi amendments on growth of Sorghum bicolor. Arch. Agron. Soil Sci. 2016, 62, 1169–1181. [Google Scholar]
- Dikobe, T.B.; Mashile, B.; Sinthumule, R.R.; Ruzvidzo, O. Distinct Morpho-Physiological Responses of Maize to Salinity Stress. Am. J. Plant Sci. 2021, 12, 946–959. [Google Scholar] [CrossRef]
- Akoto-Danso, E.K.; Manka’abusi, D.; Steiner, C.; Werner, S.; Häring, V.; Nyarko, G.; Marschner, B.; Drechsel, P.; Buerkert, A. Agronomic effects of biochar and wastewater irrigation in urban crop production of Tamale, northern Ghana. Nutr. Cycl. Agroecosyst. 2019, 115, 231–247. [Google Scholar] [CrossRef]
- Alsaeedi, A.; El-Ramady, H.; Alshaal, T.; El-Garawani, M.; Elhawat, N.; Al-Otaibi, A. Exogenous nanosilica improves germination and growth of cucumber by maintaining K+/Na+ ratio under elevated Na+ stress. Plant Physiol. Biochem. 2018, 125, 164–171. [Google Scholar] [CrossRef]
- Zhang, Q.; Gu, X.; Singh, V.P.; Kong, D.; Chen, X. Spatiotemporal behavior of floods and droughts and their impacts on agriculture in China. Global Planet. Chang. 2015, 131, 63–72. [Google Scholar] [CrossRef]
- Abbott, L.; Macdonald, L.; Wong, M.; Webb, M.; Jenkins, S.; Farrell, M. Potential roles of biological amendments for profitable grain production–A review. Agric. Ecosyst. Environ. 2018, 256, 34–50. [Google Scholar] [CrossRef]
- García, A.C.; Santos, L.A.; Izquierdo, F.G.; Sperandio, M.V.L.; Castro, R.N.; Berbara, R.L.L. Vermicompost humic acids as an ecological pathway to protect rice plant against oxidative stress. Ecol. Engin. 2012, 47, 203–208. [Google Scholar] [CrossRef]
- Shilev, S. Plant-growth-promoting bacteria mitigating soil salinity stress in plants. Appl. Sci. 2020, 10, 7326. [Google Scholar] [CrossRef]
- Cicek, N.; Erdogan, M.; Yucedag, C.; Cetin, M. Improving the Detrimental Aspects of Salinity in Salinized Soils of Arid and Semi-arid Areas for Effects of Vermicompost Leachate on Salt Stress in Seedlings. Water Air Soil Pollut. 2022, 233, 197. [Google Scholar] [CrossRef]
- Hafez, E.M.; Omara, A.E.D.; Alhumaydhi, F.A.; El-Esawi, M.A. Minimizing hazard impacts of soil salinity and water stress on wheat plants by soil application of vermicompost and biochar. Physiol. Plant. 2021, 172, 587–602. [Google Scholar] [CrossRef] [PubMed]
- Sorkhi, F. Effect of vermicompost fertilizer on antioxidant enzymes and chlorophyll contents in Borago officinalis under salinity stress. Iran. J. Plant Physiol. 2021, 11, 3589–3598. [Google Scholar]
- Reyes-Perez, J.J.; Amador, B.M.; Hernández-Montiel, L.G.; Rangel, P.P.; Rueda-Puente, E.O.; Ruiz-Espinoza, F.H. Vermicompost humates as a salinity mitigator in the germination of basil. Cienc. Rural. 2021, 52. [Google Scholar] [CrossRef]
- Benazzouk, S.; Dobrev, P.I.; Djazouli, Z.-E.; Motyka, V.; Lutts, S. Positive impact of vermicompost leachate on salt stress resistance in tomato (Solanum lycopersicum L.) at the seedling stage: A phytohormonal approach. Plant Soil. 2020, 446, 145–162. [Google Scholar] [CrossRef]
- Oo, A.; Iwai, C.; Saenjan, P. Soil properties and maize growth in saline and nonsaline soils using cassava-industrial waste compost and vermicompost with or without earthworms. Land Degrad. Dev. 2015, 26, 300–310. [Google Scholar] [CrossRef]
- Demir, Z. Alleviation of adverse effects of sodium on soil physicochemical properties by application of vermicompost. Compos. Sci. Util. 2020, 28, 100–116. [Google Scholar] [CrossRef]
- Beyk-Khormizi, A.; Sarghein, S.H.; Sarafraz-Ardakani, M.R.; Moshtaghioun, S.M.; Mousavi-Kouhi, S.M.; Ganjeali, A. Ameliorating effect of vermicompost on Foeniculum vulgare under saline condition. J. Plant Nutr. 2022, 46, 1601–1615. [Google Scholar] [CrossRef]
- Alamer, K.H.; Perveen, S.; Khaliq, A.; Zia Ul Haq, M.; Ibrahim, M.U.; Ijaz, B. Mitigation of salinity stress in maize seedlings by the application of vermicompost and sorghum water extracts. Plants 2022, 11, 2548. [Google Scholar] [CrossRef] [PubMed]
- Shen, Z.; Yu, Z.; Xu, L.; Zhao, Y.; Yi, S.; Shen, C.; Wang, Y.; Li, Y.; Zuo, W.; Gu, C. Effects of vermicompost application on growth and heavy metal uptake of barley grown in mudflat salt-affected soils. Agronomy 2022, 12, 1007. [Google Scholar] [CrossRef]
- Ibrahim, H.A.; Heba, S.; Hassan, N.M. Impact on growth, yield and nutritional status of tomato plants grown in saline soil by vermicompost and ascorbic acid. Int. J. Health Sci. 2022, 6, 10134–10143. [Google Scholar] [CrossRef]
- Xiao, X.; Wu, Z.-C.; Chou, K.-C. A multi-label classifier for predicting the subcellular localization of gram-negative bacterial proteins with both single and multiple sites. PLoS ONE 2011, 6, e20592. [Google Scholar] [CrossRef]
- Kaur, H.; Manna, M.; Thakur, T.; Gautam, V.; Salvi, P. Imperative role of sugar signaling and transport during drought stress responses in plants. Physiol. Plant. 2021, 171, 833–848. [Google Scholar] [CrossRef]
- Istanbuli, T.; Baum, M.; Touchan, H.; Hamwieh, A. Evaluation of morpho-physiological traits under drought stress conditions in barley (Hordeum vulgare L.). Photosynthetica 2020, 58, 1059–1067. [Google Scholar] [CrossRef]
- Nofouzi, F. Evaluation of seed yield of durum wheat (Triticum durum) under drought stress and determining correlation among some yield components using path coefficient analysis. Cuad. Investig. UNED 2018, 10, 179–183. [Google Scholar] [CrossRef][Green Version]
- Kalagare, V.S.; Ganesan, N.M.; Iyanar, K.; Chitdeshwari, T.; Chandrasekhar, C. Strategy of multiple selection indices for discrimination of potential genotypes and associated traits for yield improvement in pearl millet [Pennisetum glaucum (L.) R. Br.]. Electron. J. Plant Breed. 2021, 12, 895–906. [Google Scholar]
- Roy, S.; Arunachalam, K.; Dutta, B.K.; Arunachalam, A. Effect of organic amendments of soil on growth and productivity of three common crops viz. Zea mays, Phaseolus vulgaris and Abelmoschus esculentus. Appl. Soil Ecol. 2010, 45, 78–84. [Google Scholar] [CrossRef]
- Chen, J.; Xu, W.; Velten, J.; Xin, Z.; Stout, J. Characterization of maize inbred lines for drought and heat tolerance. J. Soil Water Conserv. 2012, 67, 354–364. [Google Scholar] [CrossRef]
- Hosseinzadeh, S.; Amiri, H.; Ismaili, A. Effect of vermicompost fertilizer on photosynthetic characteristics of chickpea (Cicer arietinum L.) under drought stress. Photosynthetica 2016, 54, 87–92. [Google Scholar] [CrossRef]
- Dehmordy, M.; Gholami, M.; Baninasab, B. Effect of vermicompost fertilizer on growth and drought tolerance of Olive (Olea europaea L. cv. Zard). J. Plant Physiol. 2018, 7, 1–18. [Google Scholar]
- Hosseinzadeh, S.R.; Amiri, H.; Ismaili, A. Evaluation of photosynthesis, physiological, and biochemical responses of chickpea (Cicer arietinum L. cv. Pirouz) under water deficit stress and use of vermicompost fertilizer. J. Integr. Agric. 2018, 17, 2426–2437. [Google Scholar] [CrossRef]
- Hosseinzadeh, S.R.; Amiri, H.; Ismaili, A. Nutrition and biochemical responses of chickpea (Cicer arietinum L.) to vermicompost fertilizer and water deficit stress. J. Plant Nutr. 2017, 40, 2259–2268. [Google Scholar] [CrossRef]
- Özenç, D.B. Growth and transpiration of tomato seedlings grown in Hazelnut Husk compost under water-deficit stress. Compos. Sci. Util. 2008, 16, 125–131. [Google Scholar] [CrossRef]
- Atik, A. Effects of planting density and treatment with vermicompost on the morphological characteristics of oriental beech (Fagus orientalis Lipsky.). Compos. Sci. Util. 2013, 21, 87–98. [Google Scholar] [CrossRef]
- Benaffari, W.; Boutasknit, A.; Anli, M.; Ait-El-Mokhtar, M.; Ait-Rahou, Y.; Ben-Laouane, R.; Ben Ahmed, H.; Mitsui, T.; Baslam, M.; Meddich, A. The native arbuscular mycorrhizal fungi and vermicompost-based organic amendments enhance soil fertility, growth performance, and the drought stress tolerance of quinoa. Plants 2022, 11, 393. [Google Scholar] [CrossRef]
- Feizabadi, A.; Noormohammadi, G.; Fatehi, F. Changes in growth, physiology, and fatty acid profile of rapeseed cultivars treated with vermicompost under drought stress. J. Soil Sci. Plant Nutr. 2021, 21, 200–208. [Google Scholar] [CrossRef]
- Ahmad, A.; Aslam, Z.; Hussain, S.; Javed, T.; Hussain, S.; Bashir, S.; Hussain, I.; Belliturk, K.; Adamski, R.; Siuta, D. Soil application of wheat straw vermicompost enhances morpho-physiological attributes and antioxidant defense in wheat under drought stress. Front. Environ. Sci. 2022, 10, 387. [Google Scholar] [CrossRef]
- Kiran, S. Effects of vermicompost on some morphological, physiological and biochemical parameters of lettuce (Lactuca sativa var. crispa) under drought stress. Not. Bot. Horti Agrobot. Cluj-Napoca 2019, 47, 352–358. [Google Scholar] [CrossRef]
- Paymaneh, Z.; Sarcheshmehpour, M.; Mohammadi, H.; Hesni, M.A. Vermicompost and/or compost and arbuscular mycorrhizal fungi are conducive to improving the growth of pistachio seedlings to drought stress. App. Soil Ecol. 2023, 182, 104717. [Google Scholar] [CrossRef]
- Alhverdizadeh, S.; Danaee, E. Effect of Humic Acid and Vermicompost on Some Vegetative Indices and Proline Content of Catharanthus roseous under Low Water Stress. Environ. Eng. Res. 2023, 9, 141–152. [Google Scholar]
- Khosravi Shakib, A.; Rezaei Nejad, A.; Khandan Mirkohi, A.; Kalate Jari, S. Vermicompost and manure compost reduce water-deficit stress in pot marigold (Calendula officinalis L. cv. Candyman Orange). Compos. Sci. Util. 2019, 27, 61–68. [Google Scholar] [CrossRef]
- Collange, B.; Navarrete, M.; Peyre, G.; Mateille, T.; Tchamitchian, M. Root-knot nematode (Meloidogyne) management in vegetable crop production: The challenge of an agronomic system analysis. Crop Prot. 2011, 30, 1251–1262. [Google Scholar] [CrossRef]
- Doan, T.T.; Henry-des-Tureaux, T.; Rumpel, C.; Janeau, J.-L.; Jouquet, P. Impact of compost, vermicompost and biochar on soil fertility, maize yield and soil erosion in Northern Vietnam: A three year mesocosm experiment. Sci. Total Environ. 2015, 514, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Cardoza, Y.J.; Buhler, W.G. Soil organic amendment impacts on corn resistance to Helicoverpa zea: Constitutive or induced? Pedobiologia 2012, 55, 343–347. [Google Scholar] [CrossRef]
- Rostami, M.; Olia, M.; Arabi, M. Evaluation of the effects of earthworm Eisenia fetida-based products on the pathogenicity of root-knot nematode (Meloidogyne javanica) infecting cucumber. Int. J. Recycl. Org. Waste Agric. 2014, 3, 58. [Google Scholar] [CrossRef]
- Koul, O. Phytochemicals and insect control: An antifeedant approach. Crit. Rev. Plant Sci. 2008, 27, 1–24. [Google Scholar] [CrossRef]
- Mohamadi, P.; Razmjou, J.; Naseri, B.; Hassanpour, M. Humic fertilizer and vermicompost applied to the soil can positively affect population growth parameters of Trichogramma brassicae (Hymenoptera: Trichogrammatidae) on eggs of Tuta absoluta (Lepidoptera: Gelechiidae). Neotrop. Entomol. 2017, 46, 678–684. [Google Scholar] [CrossRef]
- Karthikeyan, M.; Hussain, N.; Gajalakshmi, S.; Abbasi, S. Effect of vermicast generated from an allelopathic weed lantana (Lantana camara) on seed germination, plant growth, and yield of cluster bean (Cyamopsis tetragonoloba). Environ. Sci. Pol. Res. 2014, 21, 12539–12548. [Google Scholar] [CrossRef]
- You, X.; Tojo, M.; Ching, S.; Wang, K.-H. Effects of vermicompost water extract prepared from bamboo and kudzu against and. J. Nematol. 2018, 50, 569–578. [Google Scholar] [CrossRef] [PubMed]
- Renčo, M.; Kováčik, P. Assessment of the nematicidal potential of vermicompost, vermicompost tea, and urea application on the potato-cyst nematodes Globodera rostochiensis and Globodera pallida. J. Plant Prot. Res. 2015, 55, 187–192. [Google Scholar] [CrossRef]
- Sivasabari, K.; Parthasarathy, S.; Chandran, D.; Sankaralingam, S.; Ajaykumar, R. Vermicompost and Vermi-leachate in Pest and Disease Management. In Vermicomposting for Sustainable Food Systems in Africa; Springer: Singapore, 2023; pp. 279–304. [Google Scholar]
- Huang, K.; Li, F.; Wei, Y.; Fu, X.; Chen, X. Effects of earthworms on physicochemical properties and microbial profiles during vermicomposting of fresh fruit and vegetable wastes. Bioresour. Technol. 2014, 170, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Poveda, J.; Abril-Urias, P.; Escobar, C. Biological control of plant-parasitic nematodes by filamentous fungi inducers of resistance: Trichoderma, mycorrhizal and endophytic fungi. Front. Microbiol. 2020, 11, 992. [Google Scholar] [CrossRef]
- Molinari, S.; Leonetti, P. Bio-control agents activate plant immune response and prime susceptible tomato against root-knot nematodes. PLoS ONE 2019, 14, e0213230. [Google Scholar] [CrossRef]
- Singh, A.; Jain, A.; Sarma, B.K.; Abhilash, P.; Singh, H.B. Solid waste management of temple floral offerings by vermicomposting using Eisenia fetida. Waste Manag. 2013, 33, 1113–1118. [Google Scholar] [CrossRef] [PubMed]
- Xiao, Z.; Liu, M.; Jiang, L.; Chen, X.; Griffiths, B.S.; Li, H.; Hu, F. Vermicompost increases defense against root-knot nematode (Meloidogyne incognita) in tomato plants. Appl. Soil Ecol. 2016, 105, 177–186. [Google Scholar] [CrossRef]
- Machfudz, M.; Basit, A.; Handoko, R.N.S. Effectiveness of vermicompost with additives of various botanical pesticides in controlling Plutella xylostella and their effects on the yield of cabbage (Brassica oleracea L. var. Capitata). Asian J. Agric. Biol. 2020, 8, 223–232. [Google Scholar]
- Kaur, A.; Kaur, A.; Ohri, P. Combined effects of vermicompost and vermicompost leachate on the early growth of Meloidogyne incognita stressed Withania somnifera (L.) Dunal. Environ.Sci. Pol. Res. 2022, 29, 51686–51702. [Google Scholar] [CrossRef]
- Zuhair, R.; Moustafa, Y.T.A.; Mustafa, N.S.; El-Dahshouri, M.F.; Zhang, L.; Ageba, M.F. Efficacy of amended vermicompost for bio-control of root knot nematode (RKN) Meloidogyne incognita infesting tomato in Egypt. Environ. Technol. Innov. 2022, 27, 102397. [Google Scholar] [CrossRef]
- Edwards, C.A.; Arancon, N.Q.; Vasko-Bennett, M.; Askar, A.; Keeney, G.; Little, B. Suppression of green peach aphid (Myzus persicae) (Sulz.), citrus mealybug (Planococcus citri)(Risso), and two spotted spider mite (Tetranychus urticae) (Koch.) attacks on tomatoes and cucumbers by aqueous extracts from vermicomposts. Crop Prot. 2010, 29, 80–93. [Google Scholar] [CrossRef]
- Hussain, N.; Abbasi, T.; Abbasi, S. Evaluating the fertilizer and pesticidal value of vermicompost generated from a toxic and allelopathic weed ipomoea. J. Saudi Soc. Agric. 2020, 19, 43–50. [Google Scholar] [CrossRef]
- dos Santos Pereira, T.; Macêdo, A.G.; da Silva, J.; Pinheiro, J.B.; de Paula, A.M.; Biscaia, D.; Busato, J.G. Water-extractable fraction of vermicomposts enriched with Trichoderma enhances the growth of bell pepper and tomato as well as their tolerance against Meloidogyne incognita. Sci. Horti. 2020, 272, 109536. [Google Scholar] [CrossRef]
- Razmjou, J.; Mohammadi, M.; Hassanpour, M. Effect of vermicompost and cucumber cultivar on population growth attributes of the melon aphid (Hemiptera: Aphididae). J. Econ. Entomol. 2011, 104, 1379–1383. [Google Scholar] [CrossRef]
- Ullah, M.I.; Riaz, M.; Arshad, M.; Khan, A.H.; Afzal, M.; Khalid, S.; Mehmood, N.; Ali, S.; Khan, A.M.; Zahid, S.M.A. Application of Organic Fertilizers Affect the Citrus Leafminer, Phyllocnistis citrella (Lepidoptera: Gracillariidae) Infestation and Citrus Canker Disease in Nursery Plantations. Int. J. Insect Sci. 2019, 11, 1179543319858634. [Google Scholar] [CrossRef] [PubMed]
- Arancon, N.Q.; Galvis, P.A.; Edwards, C.A. Suppression of insect pest populations and damage to plants by vermicomposts. Bioresour. Technol. 2005, 96, 1137–1142. [Google Scholar] [CrossRef]
- Gudeta, K.; Julka, J.; Kumar, A.; Bhagat, A.; Kumari, A. Vermiwash: An agent of disease and pest control in soil, a review. Heliyon 2021, 7, e06434. [Google Scholar] [CrossRef]
- Nadana, G.R.V.; Rajesh, C.; Kavitha, A.; Sivakumar, P.; Sridevi, G.; Palanichelvam, K. Induction of growth and defense mechanism in rice plants towards fungal pathogen by eco-friendly coelomic fluid of earthworm. Environ. Technol. Innov. 2020, 19, 101011. [Google Scholar] [CrossRef]
- Hakim Rabet, S.; Ketabchi, S. The effect of compost fertilizers, vermicompost and their tea on bacterial vascular wilt and growth indices in tomato seedlings. J. Appl. Res. Plant Prot. 2021, 9, 61–74. [Google Scholar]
- Kinay, P.; Mansour, M.F.; Gabler, F.M.; Margosan, D.A.; Smilanick, J.L. Characterization of fungicide-resistant isolates of Penicillium digitatum collected in California. Crop Prot. 2007, 26, 647–656. [Google Scholar] [CrossRef]
- You, X.; Wakana, D.; Ishikawa, K.; Hosoe, T.; Tojo, M. Antifungal activity of compounds isolated from bamboo vermicompost against Rhizoctonia solani AG1-IB. Adv. Microbiol. 2019, 9, 957. [Google Scholar] [CrossRef]
- Jackson, K.S.; Kumar, A.; Kumar, V. Evaluation of Organic Amendments Against Alternaria Brassicae Causing Alternaria Leaf Spot on Mustard under In Vitro and Poly House Conditions. Plant Cell Biotechnol. Mol. Biol. 2019, 20, 285–293. [Google Scholar]
- Mu, J.; Li, X.; Jiao, J.; Ji, G.; Wu, J.; Hu, F.; Li, H. Biocontrol potential of vermicompost through antifungal volatiles produced by indigenous bacteria. Biol. Control 2017, 112, 49–54. [Google Scholar] [CrossRef]
- Tian, G.-L.; Bi, Y.-M.; Jiao, X.-L.; Zhang, X.-M.; Li, J.-F.; Niu, F.-B.; Gao, W.-W. Application of vermicompost and biochar suppresses Fusarium root rot of replanted American ginseng. Appl. Microbiol. Biotechnol. 2021, 105, 6977–6991. [Google Scholar] [CrossRef] [PubMed]
- Kalantari, S.; Marefat, A.; Naseri, B.; Hemmati, R. Improvement of bean yield and Fusarium root rot biocontrol using mixtures of Bacillus, Pseudomonas and Rhizobium. Trop. Plant Pathol. 2018, 43, 499–505. [Google Scholar] [CrossRef]
- Baum, C.; Eichler-Löbermann, B.; Hrynkiewicz, K. Impact of organic amendments on the suppression of Fusarium wilt. In Organic Amendments and Soil Suppressiveness in Plant Disease Management; Springer: Cham, Switzerland, 2015; pp. 353–362. [Google Scholar]
- Srivastava, R.; Khalid, A.; Singh, U.; Sharma, A. Evaluation of arbuscular mycorrhizal fungus, fluorescent Pseudomonas and Trichoderma harzianum formulation against Fusarium oxysporum f. sp. lycopersici for the management of tomato wilt. Biol. Cont. 2010, 53, 24–31. [Google Scholar] [CrossRef]
- Litterick, A.; Wood, M. The use of composts and compost extracts in plant disease control. In Disease Control in Crops; Wiley-Blackwell: Hoboken, NJ, USA, 2009. [Google Scholar]
- Dowling, D.N.; O’Gara, F. Metabolites of Pseudomonas involved in the biocontrol of plant disease. Trends Biotechnol. 1994, 12, 133–141. [Google Scholar] [CrossRef]
- Benítez, T.; Rincón, A.M.; Limón, M.C.; Codon, A.C. Biocontrol mechanisms of Trichoderma strains. Intern. Microbiol. 2004, 7, 249–260. [Google Scholar]
- Pharand, B.; Carisse, O.; Benhamou, N. Cytological aspects of compost-mediated induced resistance against Fusarium crown and root rot in tomato. Phytopathology 2002, 92, 424–438. [Google Scholar] [CrossRef]
- Sang, M.K.; Kim, K.D. Biocontrol activity and primed systemic resistance by compost water extracts against anthracnoses of pepper and cucumber. Phytopathology 2011, 101, 732–740. [Google Scholar] [CrossRef]
- M’piga, P.; Belanger, R.; Paulitz, T.; Benhamou, N. Increased resistance to Fusarium oxysporumf. sp. radicis-lycopersiciin tomato plants treated with the endophytic bacterium Pseudomonas fluorescens strain 63-28. Physiol. Mol. Plant Pathol. 1997, 50, 301–320. [Google Scholar] [CrossRef]
- De Vleesschauwer, D.; Cornelis, P.; Höfte, M. Redox-active pyocyanin secreted by Pseudomonas aeruginosa 7NSK2 triggers systemic resistance to Magnaporthe grisea but enhances Rhizoctonia solani susceptibility in rice. Mol. Plant-Microbe Interact. 2006, 19, 1406–1419. [Google Scholar] [CrossRef] [PubMed]
- Johnson, J.M.; Ludwig, A.; Furch, A.C.; Mithöfer, A.; Scholz, S.; Reichelt, M.; Oelmüller, R. The beneficial root-colonizing fungus Mortierella hyalina promotes the aerial growth of Arabidopsis and activates calcium-dependent responses that restrict Alternaria brassicae–induced disease development in roots. Mol. Plant-Microbe Interact. 2019, 32, 351–363. [Google Scholar] [CrossRef] [PubMed]
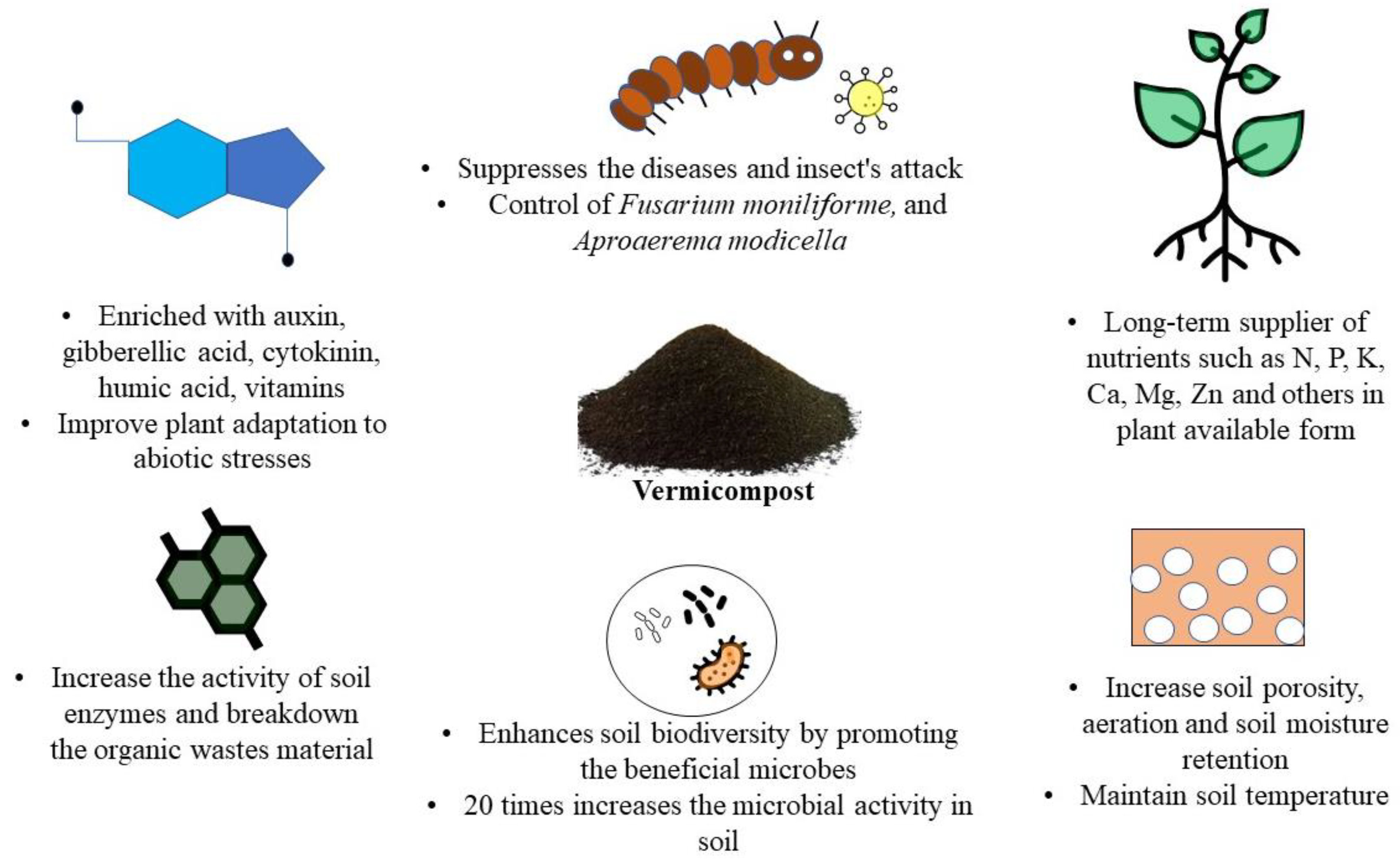
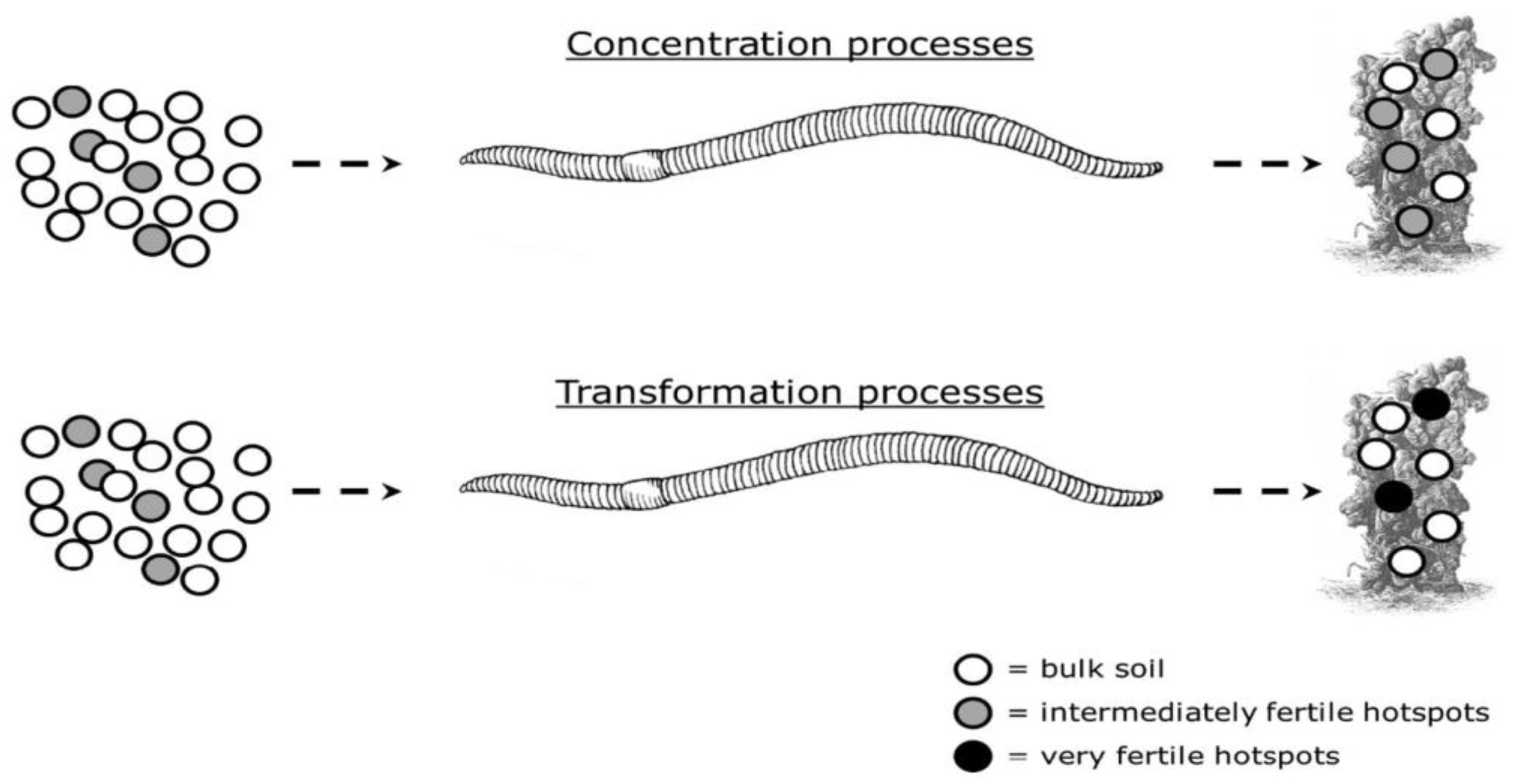
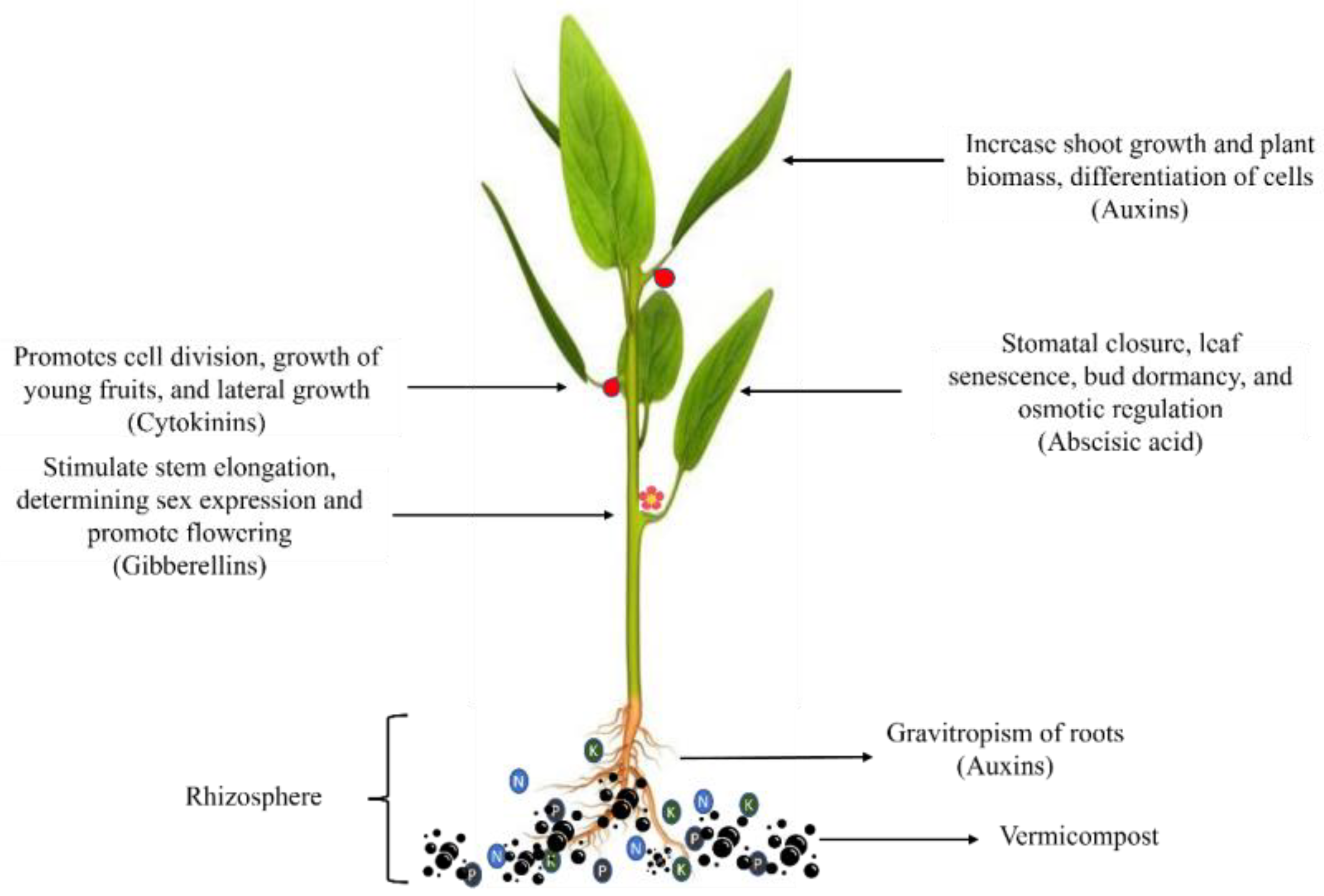
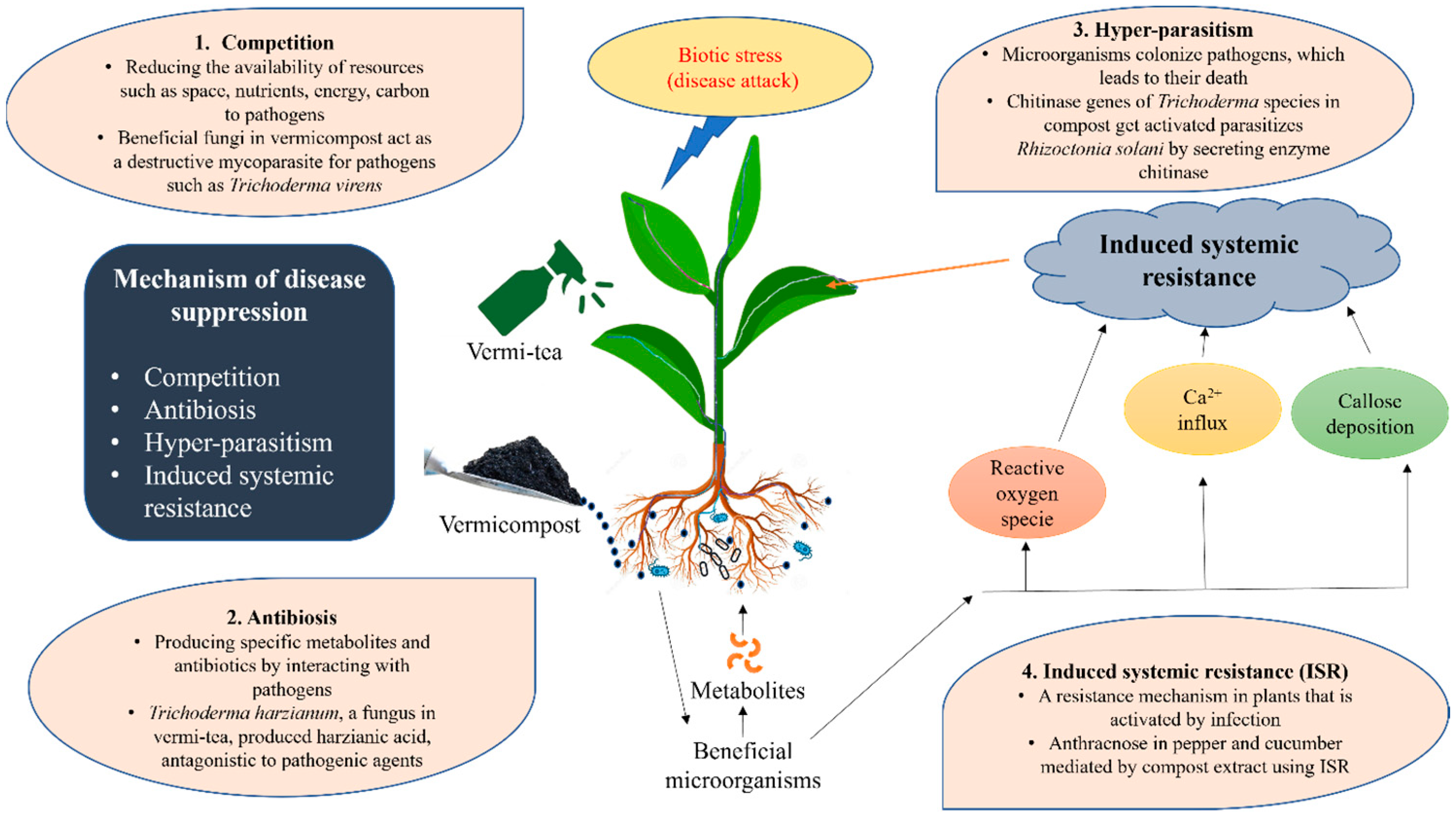
| Type of Fertilizer | Source | Biochemical and Nutritional Characteristics | Test Plant | Effect on Plant Growth and Yield | References |
|---|---|---|---|---|---|
| VC | Green wastes | pH: 7.43, EC: 1.77 dsm−1, CEC:117.44 c mol kg−1, N: 21.47 g/kg, P: 4.82 g/kg, K: 7.87 g/kg, Ca: 179.15 g/kg, Mg: 12.58 g/kg, Zn: 189.12 mg/kg | Pelargonium zonale L. and Calendula officinalis L. | Growth and flowering of both plants were higher in all vermicompost-based media. | [78] |
| VC | Cow dung, food industry sludge | EC: 1.8 dS/m, pH: 6.6, C/N ratio:11.9, TKN: 26 g/kg, TAP: 9.75 g/kg, TK: 7.6 g/kg, | Cicer arietinum L. | Total chlorophyll contents and yield increased. | [79] |
| VC | Cattle dung, leaf litter, straw | - | Capsicum annum (L.) | Vermicompost treatment (50%) revealed excellent increase in chilly growth and soil quality improvement. | [80] |
| VT | Food wastes | EC: 6.36 mS/cm, pH: 6.5 and HA: 485.12 mg/L, N: 1.3, P: 204, K: 45.4, Ca: 449 and Mg: 40.1 mg/L | Lactuca sativa, Solanum lycopersicum | Increased the yield of both test plants even at reduced concentration (25%, 50%), respectively. | [81] |
| VC | skin coffee + green waste + biochar | pH: 5.60 N: 2.76%, K: 3.84%, | Coffea | Physiological and morphological parameters of coffee plant increased. | [82] |
| VW | Cow dung | - | Solanum melongena | Plant height and number of fruits increased significantly. | [83] |
| VC | Animal fleshing, Tannery waste | pH: 6.56, EC: 1.08 dS/m, TKN: 20 g/kg, Total P: 39 g/kg, Total K: 4.4 g/kg | Lycopersicom esculentum L. | Vermicompost resulted in higher plant height (10%), stem girth (8.9%), and leaf numbers (14%). | [84] |
| VC | - | Total N: 1.54%, Total P: 0.64%, Total K: 6.31%, total Mg: 0.58%, total Ca: 1.39%, total Zn: 0.01%, total S: 0.34% | Ananas comosus (L.) Merr. | Fruits contained higher total soluble solids, titratable acidity, ascorbic acid, and total chlorophyll content. | [85] |
| VC | Cow manure and grass biomass | pH: 8.03, N: 20 mg/L, P: 1997 mg/L, K: 8300 mg/L, Mg: 3600 mg/L, Ca: 7850 mg/L | Dracocephalum moldavica L. | Increased the plant’s biomass (148%) in peat-amended vermicompost and (68%) in soil amended with vermicompost. | [86] |
| VC | C. erectus leaves + FYM | pH: 6.69, nitrogen: 1.86%, P: 0.15%, K: 0.41%, Zn: 39.4 ppm, Ca: 4085.80 ppm, Fe: 1572 ppm | Vigna radiata L. | The economical and biological yield of mung beans were boosted up to 59% and 34%, respectively. | [87] |
| VC | - | EC: 2.15 mS/cm, pH: 8.74, OM: 74%, TOC: 43.02%, Porosity: 62.5% | Solanum lycopersicum | Plants cultivated on VC substrate indicated enhanced growth and better resistance to salinity stress. | [88] |
| Amendment | Physiochemical and Nutritional Characteristics | Plant | Impact | References |
|---|---|---|---|---|
| VCL | pH: 8.32, total organic matter: 7.5%, total P: 1.2%, K: 1.5%, and N: 2.2%, and total humic + fulvic acid: 6% | Sedum album | VCL reduced effects of salinity and improved growth and quality traits of S. album. | [132] |
| VC + BC | VC had an OM content: 42%, pH: 7.4, EC: 3.8 dS/m, total N: 2.1%, total P: 7.8%, total K: 0.5%, BC had pH: 7.90, EC: 2.05 dS/m, total N: 1.9%, total P: 2.2%, total K: 2.9%, | Triticum aestivum L. | VC + BC reduced oxidative stress, proline and leaf Na+ content with increase in nutrient uptake and grain yield. Soil pH, EC, and ESP decreased. | [133] |
| VC | Organic carbon: 20.7%, EC: 5 dS/m, P: 0.7 mg/kg, K: 0.9 mg/kg, Na: 473 mg/kg | Borago officinalis | VC increased antioxidant enzymes and photosynthetic pigments activities and reduced the salinity. | [134] |
| VC | OM content: 74%, pH: 8.74, EC: 2.15 mS/cm, Salinity: 1.5 g/L, Porosity: 62.5% | Solanum lycopersicum L. | VC improved growth and better resistance to salinity stress. | [88] |
| VCH | C: 53.4%, pH: 8.7, N: 3.05%, S: 0.72%, humic acid: 4.82%, fulvic acid: 7.17% | Ocimum basilicum L. | VCH biostimulated and improved the growth and salinity tolerance of the sensitive variety. | [135] |
| VCL | - | Solanum lycopersicum L. | VCL lowers the impact of salinity on leaf senescence. | [136] |
| VW + VC | Vermicompost: pH: 7.2, EC: 2.69 mS/cm, N: 0.98%, P: 121 ppm, K: 46 ppm. Vermiwash: pH: 7.5, EC: 2.82 mS/cm, N: 0.45%, P: 41 ppm, K: 21 pmm | Sorghum bicolor L. | Vermi-amendments increased the nutritional status of soil, reduced the salt damage to plants, and improved growth. | [124] |
| VC | pH: 7.17, EC: 1.05 dS/m, TN: 4.13 g/kg, TP: 1.24 g/kg, TK: 1.70 g/kg, C:N = 15.04 | Zea mays L. | VC decreased exchangeable Na+ and improved soil microbial activities and maize growth. | [137] |
| VC | pH: 7.1, EC: 6.5 dS/m, OM: 65.5%, TN: 2.2%, TP: 7.3 mg/kg, TK: 12.8 mg/kg, Ca: 25.1 mg/kg, Zn: 216 mg/kg | VC decreased EC, bulk density, and exchangeable Na, and increased aggregate stability and total porosity, | [138] | |
| VC | - | Foeniculum vulgare | VC can limit the adverse effects of salinity stress on fennel by affecting photosynthetic pigments, membrane integrity and antioxidants activity. | [139] |
| VC + SWE | pH: 7.98, EC: 2.98 mS/cm, N: 0.07%, AP: 6.34 mg/kg, AK: 127 mg/kg, Zn: 0.97 mg/kg | Zea mays L. | VC + SWE decreased Na+ concentration, H2O2, and malondialdehyde contents at higher salinity. | [140] |
| VC | pH: 6.34, EC: 8.92 mS/cm, TN: 24.4 g/kg, TP: 15.9 g/kg, Alkaline N: 2467 mg/kg, AP: 869 mg/kg, Zn: 805 mg/kg | Hordeum vulgare L. | VC application decreased bulk density, EC, and pH of saline soil, whereas the yield of grain increased up to 512% as quantity of VC increased. | [141] |
| VC + AA | pH: 6.90, EC: 2.0 dS/m, OM: 50.3%, N: 1.20%, P: 0.50%, K: 0.80% | Lycopersicum esculentum L. cv Kasel rock | VC and ascorbic acid application decreased Na contents in leaves and increased tomato yield and quality. | [142] |
| Type of Fertilizer | Characteristics of Fertilizer | Application Rate | Drought Conditions | Test Plant | Impact | References |
|---|---|---|---|---|---|---|
| Wheat Straw VC | pH: 7.55, EC: 2.26 dS/m, N: 0.85%, P: 0.45%, K: 0.83%, Ca: 2.91%, S: 0.23% | 0, 4, 6, 8 t ha−1 | 30, 45, 70% FC | Triticum aestivum L. | VC application at 8 t ha−1 increased SOD activity (14.28%), POD (27.28%), and CAT (50%). Photosynthesis and transpiration rate increased by 27.65 and 49.25%, respectively. | [158] |
| VC | pH: 8.1, EC: 6.5 dS/m, OM: 65.5%, N: 2.2%, P: 1.7%, K: 1.5% | 0, 2.5 and 5% | No stress, moderate drought, and severe drought at 100, 50 and 25% of FC | Lactuca sativa var. crispa | VC increased SOD and CAT enzyme activities and lowered MDA content under drought stress. Chlorophyll a, chlorophyll b, and carotenoid contents increased in lettuce. | [159] |
| VC | pH: 7.1, EC: 1.5 dS/m, C/N: 15.5, N: 0.9–3%, P: 0.9–2.5%, K: 0.6–2.5%, Ca: 4.85–8%, | VC to soil ratio i.e., 0:100, 10:90, 20:80, and 30:70 | No stress, moderate drought (MS), and severe drought (SS) at 100, 75, and 25% of FC, respectively | Cicer arietinum L. | VC addition of 10 and 20% significantly enhanced the chlorophyll contents and Fv/Fm under MS and Fv/Fm, Ci, and PN under SS at the flowering stage. | [150] |
| C + VC + AMF | VC: pH: 7.4, EC: 2.2 dS/m, C/N: 25.8, TN: 1.32%, TP: 0.4%, TK: 0.4%, Fe: 1100 ppm, Zn: 50 ppm C: pH: 7.5, EC: 2 dS/m, C/N: 21.4, TN: 1.9%, TP: 0.8%, TK: 0.8%, Fe: 1000 ppm, Zn: 46 ppm | VC (4% w/w) and inoculation with and without AMF | Drought stress at three levels, i.e., well-watered: D0 (no drought), moderate stress: D1 and severe stress: D2 | Pistachio seedlings | VC and C increased plant growth (232% and 29%) and nutrient uptake (30% and 52% P uptake and 35% and 1% Zn uptake) under severe drought stress conditions. | [160] |
| VC + humic acid | - | VC (0, 25, and 50%) and humic acid (0, 50, and 100 mg/L) | 50 and 100% FC | Catharanthus roseous | VC and humic acid reduced the drought stress, improved vegetative indices, cell membrane stability index, and plant pigment content, and decreased proline content. | [161] |
| VC | pH: 7.1, EC: 1.5 dS/m, C/N: 17.5, N: 3.0%, P: 0.9%, K: 1.2%, Ca: 4.5%, Mg: 0.50% | 0, 10, 20, 30% vermicompost to soil ratio | 75, 50, and 25% FC | Cicer arietinum L. cv. Pirouz | VC at 30% increased the photosynthetic pigments, CO2 assimilation rate, transpiration, Fv/Fm, Ca, and K in root and leaf tissues, and proline and soluble protein contents in root tissues. | [152] |
| C + CV | - | C: 20 and 30% VC: 20 and 30% | 80, 60 and 40% AWC | Calendula officinalis L. cv. Candyman Orange | Total dry mass and water use efficiency was about 3-fold higher in plants grown in 30% VC or 30% manure compost/ | [162] |
| Type of Fertilizer | Parental Wastes | Earthworm | Phytopathogens | Plants | Pest Control | References |
|---|---|---|---|---|---|---|
| VC | CD | E. fetida | Meloidogyne incognita | Solanum lycopersicum L. | Root-knot nematode | [177] |
| VC, N, P, M, S | CD, VR | Lumbricus rubellus | Plutella xylostella | Brassica oleracea L. | Diamondback moth | [178] |
| VC, VL | CD | E. fetida | Meloidogyne incognita | Withania somnifera | Root parasitic nematode | [179] |
| VC | CD, TL, SD | E. fetida, Lumbricus rubellus, Perionyx ecavatus | Meloidogyne incognita | Solanum lycopersicum L. | Root parasitic nematode | [180] |
| VE | FW | - | Myzus persicae, Planococcus citri, etranychus urticae | Solanum lycopersicum L. and Cucumis sativa | Green peach aphid, citrus mealybug, two spotted spider mites | [181] |
| VC | Ipomoea leaves | E. fetida | Earias vittella | Abelmoschus esculentus | Fruit borer | [182] |
| VE | CD | E. fetida | Meloidogyne incognita | Capsicum annuum L. and Solanum lycopersicum L. | Root-knot nematode | [183] |
| VC, VE | GW | - | Globodera rostochiensis and Globodera pallida | Solanum tuberosum | Potato-cyst nematodes | [171] |
| VC | CD | - | Aphis gossypii Glover | Cucumis sativus L. | Melon aphid | [184] |
| VC | BL, CD | Pheretima posthuma | Phyllocnistis citrella | C. reticulata | Citrus leaf miner | [185] |
| VC | FW | - | Myzus persicae, Pseudococcus sp. and Pieris brassicae L. | Capsicum annuum L., Lycopersicon esculentum and Brassica oleracea L. | Aphids, mealy bugs, and cabbage white caterpillars | [186] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rehman, S.u.; De Castro, F.; Aprile, A.; Benedetti, M.; Fanizzi, F.P. Vermicompost: Enhancing Plant Growth and Combating Abiotic and Biotic Stress. Agronomy 2023, 13, 1134. https://doi.org/10.3390/agronomy13041134
Rehman Su, De Castro F, Aprile A, Benedetti M, Fanizzi FP. Vermicompost: Enhancing Plant Growth and Combating Abiotic and Biotic Stress. Agronomy. 2023; 13(4):1134. https://doi.org/10.3390/agronomy13041134
Chicago/Turabian StyleRehman, Sami ur, Federica De Castro, Alessio Aprile, Michele Benedetti, and Francesco Paolo Fanizzi. 2023. "Vermicompost: Enhancing Plant Growth and Combating Abiotic and Biotic Stress" Agronomy 13, no. 4: 1134. https://doi.org/10.3390/agronomy13041134
APA StyleRehman, S. u., De Castro, F., Aprile, A., Benedetti, M., & Fanizzi, F. P. (2023). Vermicompost: Enhancing Plant Growth and Combating Abiotic and Biotic Stress. Agronomy, 13(4), 1134. https://doi.org/10.3390/agronomy13041134










