The Effect of Microbial Inoculation under Various Nitrogen Regimes on the Uptake of Nutrients by Apple Trees
Abstract
:1. Introduction
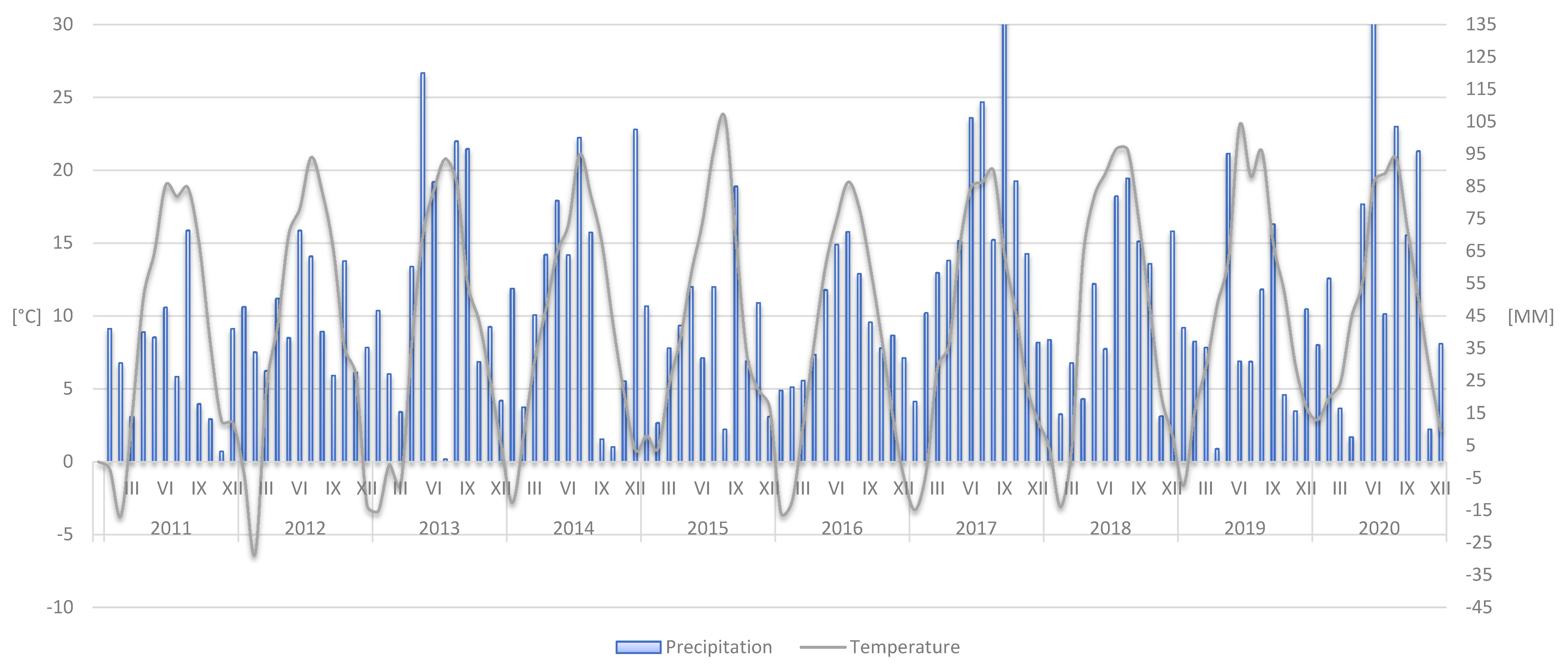
2. Materials and Methods
2.1. Nutritional Status of Leaves and Fruits
2.2. Mycorrhizal Colonization
2.3. Statistical Analysis
3. Results
3.1. Mycorrhizal Parameters
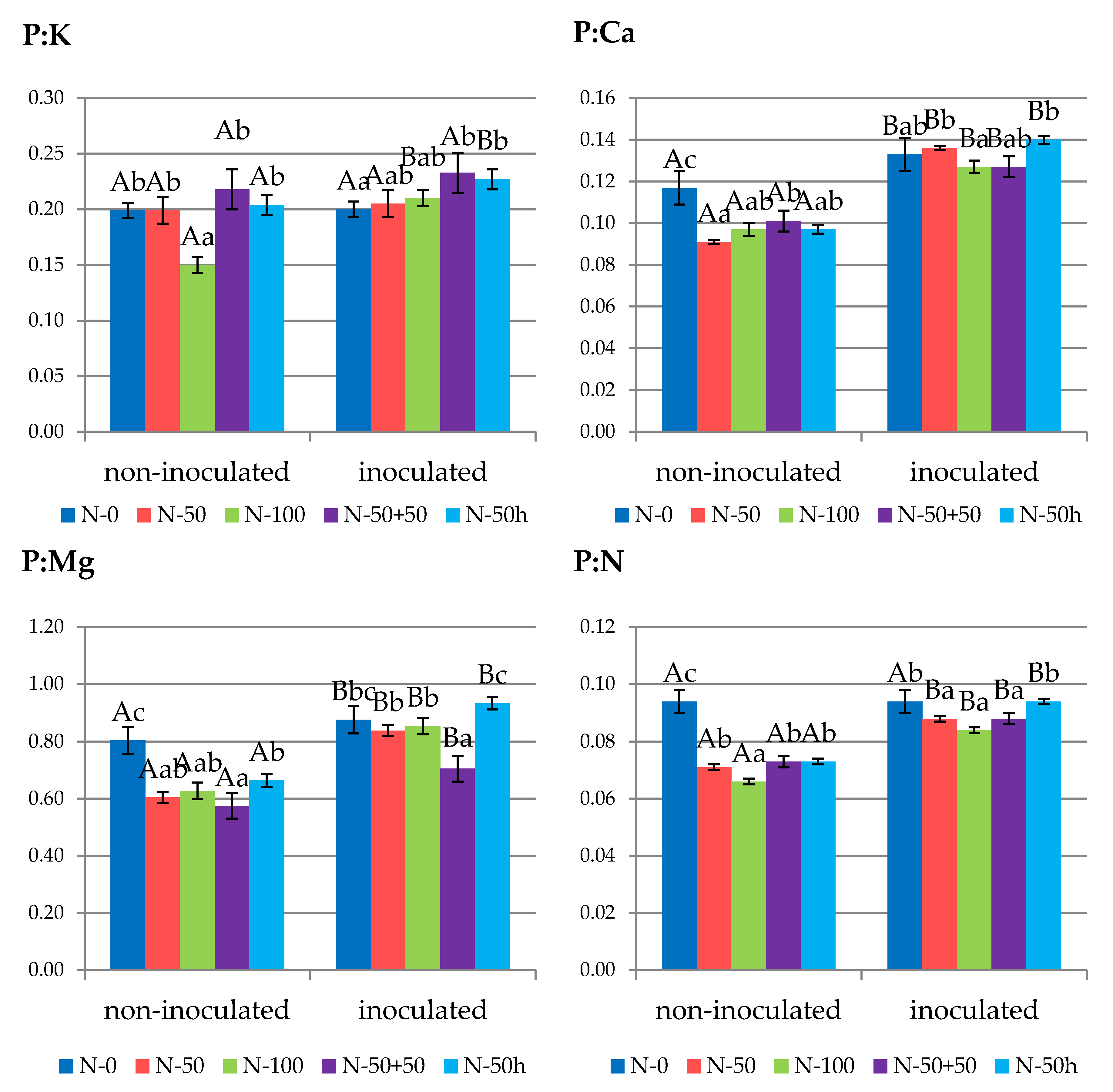
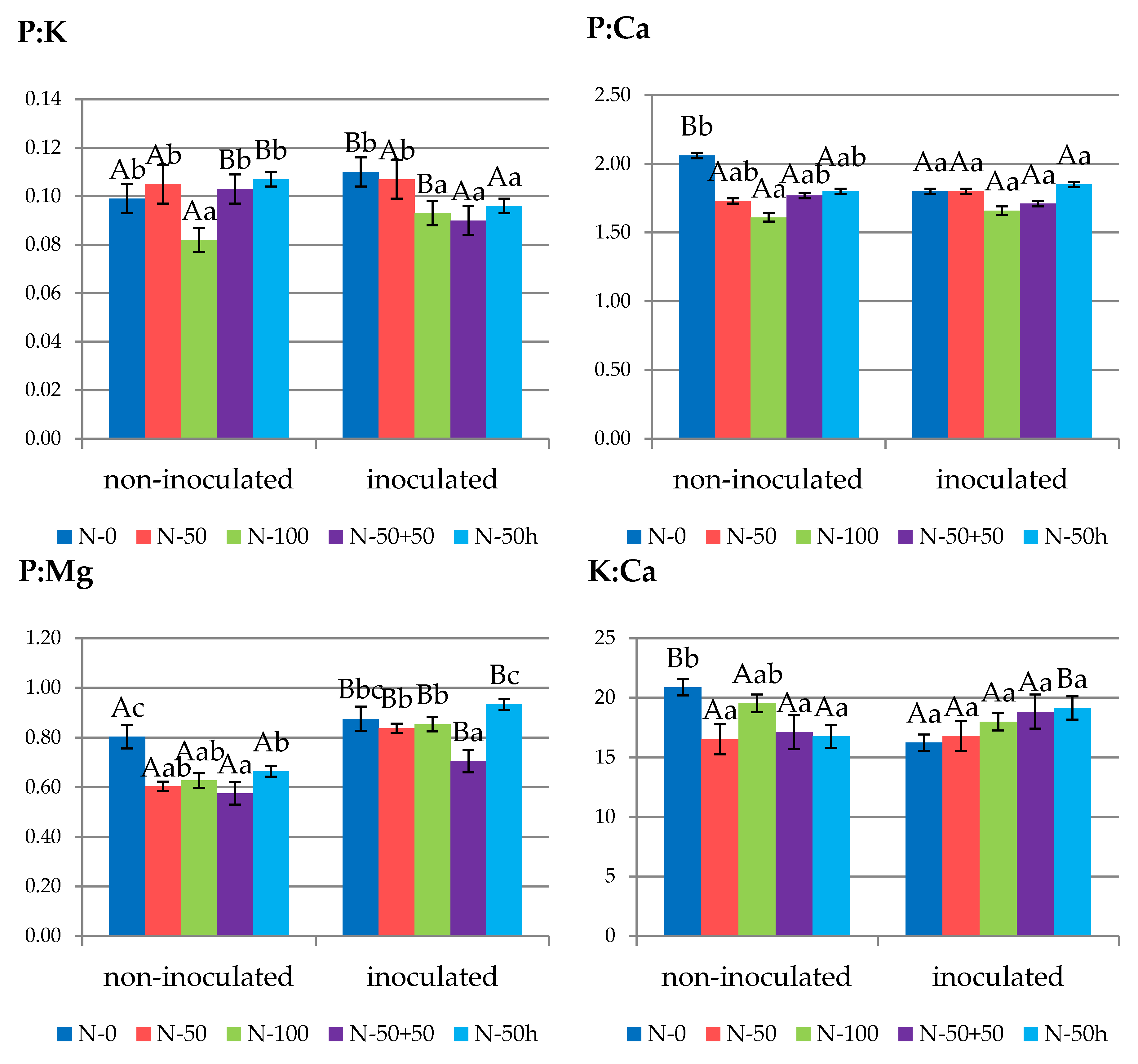
3.2. Leaf Nutrient Status
3.3. Fruit Nutrient Status
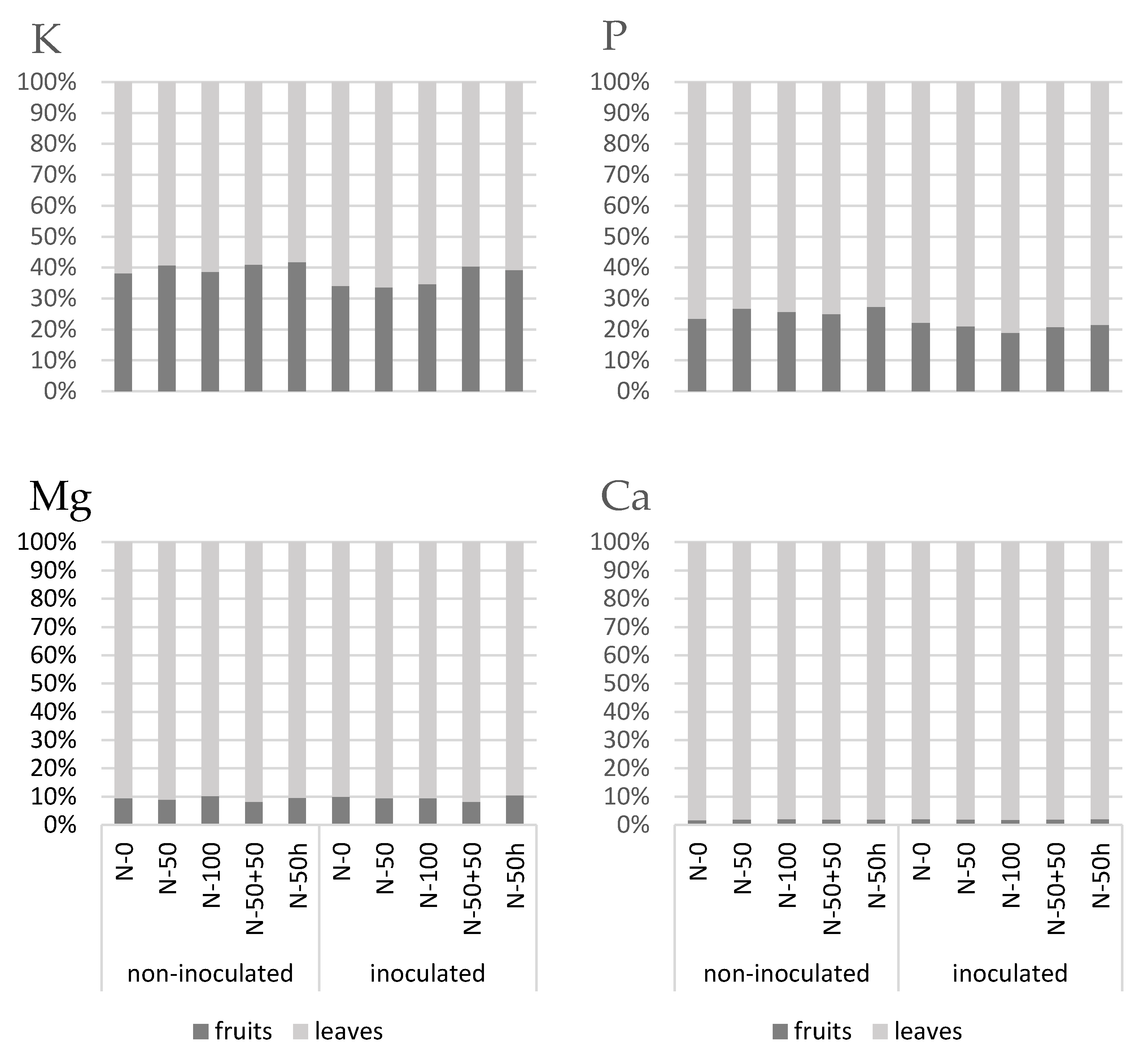
3.4. Macroelement Partitioning of Leaves and Fruits
3.4.1. Leaves
3.4.2. Fruits
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Martin, M.H.; Marschner, H. The Mineral Nutrition of Higher Plants. J. Ecol. 1988, 76, 1250. [Google Scholar] [CrossRef] [Green Version]
- Epstein, E.; Bloom, A. Mineral Nutrition of Plants: Principles and Perspectives, 2nd ed.; Sinauer Associates, Inc.: Sunderland, MA, USA, 2005. [Google Scholar]
- Galloway, J.N.; Cowling, E.B. Reactive nitrogen and the world: 200 years of change. Ambio 2002, 31, 64–71. [Google Scholar] [CrossRef]
- Fernandez, M.S.; Rosiello, R.O.P. Mineral nitrogen in plant physiology and plant nutrition. Crit. Rev. Plant Sci. 1995, 14, 111–148. [Google Scholar] [CrossRef]
- Galloway, J.N.; Aber, J.D.; Erisman, J.W.; Seitzinger, S.P.; Howarth, R.; Cowling, E.B.; Cosby, B.J. The Nitrogen Cascade. BioScience 2003, 53, 341–356. [Google Scholar] [CrossRef]
- Tilman, D.; Cassman, K.G.; Matson, P.A.; Naylor, R.; Polasky, S. Agricultural sustainability and intensive production practices. Nature 2002, 418, 671–677. [Google Scholar] [CrossRef] [PubMed]
- Environmental Defense Fund. The Case for Reforming Farm Programs to Preserve the Environment and Help Family Farmers, Ranchers, and Foresters; Environmental Defense Fund: Washington, DC, USA, 2001. [Google Scholar]
- Elser, J.J.; Bennett, E.M.B.M. A broken biogeochemical cycle. Nat. Cell Biol. 2011, 478, 29–31. [Google Scholar] [CrossRef] [PubMed]
- Tubiello, F.N.; Salvatore, M.; Ferrara, A.F.; House, J.; Federici, S.; Rossi, S.; Biancalani, R.; Golec, R.D.C.; Jacobs, H.; Flammini, A.; et al. The Contribution of Agriculture, Forestry and other Land Use activities to Global Warming, 1990. Glob. Chang. Biol. 2015, 21, 2655–2660. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bauer, S.E.; Tsigaridis, K.; Miller, R. Significant atmospheric aerosol pollution caused by world food cultivation. Geophys. Res. Lett. 2016, 43, 5394–5400. [Google Scholar] [CrossRef] [Green Version]
- Tilman, D.; Clark, M.; Williams, D.; Kimmel, K.; Polasky, S.; Packer, D.T.K.K.S.P.C. Future threats to biodiversity and pathways to their prevention. Nat. Cell Biol. 2017, 546, 73–81. [Google Scholar] [CrossRef]
- Kramer, S.B.; Reganold, J.P.; Glover, J.D.; Bohannan, B.J.M.; Mooney, H.A. Reduced nitrate leaching and enhanced denitrifier activity and efficiency in organically fertilized soils. Proc. Natl. Acad. Sci. USA 2006, 103, 4522–4527. [Google Scholar] [CrossRef] [Green Version]
- Mulvaney, R.L.; Khan, S.A.; Ellsworth, T.R. Synthetic Nitrogen Fertilizers Deplete Soil Nitrogen: A Global Dilemma for Sustainable Cereal Production. J. Environ. Qual. 2009, 38, 2295–2314. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Russell, A.E.; Cambardella, C.A.; Laird, D.A.; Jaynes, D.B.; Meek, D.W. Nitrogen fertilizer effects on soil carbon balances in Midwestern U.S. agricultural systems. Ecol. Appl. 2009, 19, 1102–1113. [Google Scholar] [CrossRef] [Green Version]
- Robertson, G.P.; Bruulsema, T.W.; Gehl, R.J.; Kanter, D.; Mauzerall, D.L.; Rotz, C.A.; Williams, C.O. Nitrogen–climate interactions in US agriculture. Biogeochemistry 2013, 114, 41–70. [Google Scholar] [CrossRef] [Green Version]
- Collins, H.P.; Rasmussen, P.E.; Douglas, C.L., Jr. Croprotation and residue management effect on soil carbon andmicrobial dynamics. Soil Sci. Soc. Am. J. 1992, 56, 783–788. [Google Scholar] [CrossRef]
- Lovell, R.D.; Jarvis, S.C.; Bardgett, R.S. Soil microbialbiomass and activity in long-term grassland: Effects of man-agement changes. Soil Biol. Biochem. 1995, 27, 969–975. [Google Scholar] [CrossRef]
- Schnurer, J.; Clarholm, M.; Rosswall, T. Microbial bio-mass and activity in agricultural soil with different organicmatter contents. Soil Biol. Biochem. 1985, 17, 611–618. [Google Scholar] [CrossRef]
- Anderson, T.; Domsch, K.H. Ratios of microbial biom-ass carbon to total organic carbon in arable soils. Soil Biol. Biochem. 1989, 21, 471–479. [Google Scholar] [CrossRef]
- Ross, D.J.; Tate, K.R. Microbial C and N, and respira-tory activity in litter and soil of southern Beech (Nothofa-gus) forest: Distribution and properties. Soil Biol. Biochem. 1993, 25, 477–483. [Google Scholar] [CrossRef]
- Raj, H.; Sharma, S. Integration of soil solarization and chemical sterilization with beneficial microorganisms for the control of white root rot and growth of nursery apple. Sci. Hortic. 2009, 119, 126–131. [Google Scholar] [CrossRef]
- Krishna, H.; Das, B.; Attri, B.L.; Grover, M.; Ahmed, N. Suppression of Botryosphaeria canker of apple by arbuscular mycorrhizal fungi. Crop. Prot. 2010, 29, 1049–1054. [Google Scholar] [CrossRef]
- Miller, D.D.; Domoto, P.A.; Walker, C. Mycorrhizal Fungi at Eighteen Apple Rootstock Plantings in The United States. New Phytol. 1985, 100, 379–391. [Google Scholar] [CrossRef]
- An, Z.; Shen, T.; Wang, H. Mycorrhizal fungi in relation to growth and mineral nutrition of apple seedlings. Sci. Hortic. 1993, 54, 275–285. [Google Scholar] [CrossRef]
- Matsubara, Y.-I.; Karikomi, T.; Ikuta, M.; Hori, H.; Ishikawa, S.; Harada, T. Effect of Arbuscular Mycorrhizal Fungus Inoculation on Growth of Apple (Malus ssp.) Seedlings. J. Jpn. Soc. Hortic. Sci. 1996, 65, 297–302. [Google Scholar] [CrossRef]
- Forge, T.; Muehlchen, A.; Hackenberg, C.; Neilsen, G.; Vrain, T. Effects of preplant inoculation of apple (Malus domestica Borkh.) with arbuscular mycorrhizal fungi on population growth of the root-lesion nematode, Pratylenchus penetrans. Plant Soil 2001, 236, 185–196. [Google Scholar] [CrossRef]
- Berdeni, D.; Cotton, T.E.A.; Daniell, T.J.; Bidartondo, M.I.; Cameron, D.; Evans, K.L. The Effects of Arbuscular Mycorrhizal Fungal Colonisation on Nutrient Status, Growth, Productivity, and Canker Resistance of Apple (Malus pumila). Front. Microbiol. 2018, 9, 1461. [Google Scholar] [CrossRef]
- Öpik, M.; Moora, M.; Liira, J.; Zobel, M. Composition of root-colonizing arbuscular mycorrhizal fungal communities in different ecosystems around the globe. J. Ecol. 2006, 94, 778–790. [Google Scholar] [CrossRef]
- Smith, S.E.; Read, D. Mycorrhizal Symbiosis; Academic Press: Cambridge, MA, USA, 2008. [Google Scholar]
- Thirkell, T.J.; Charters, M.D.; Elliott, A.J.; Sait, S.M.; Field, K.J. Are mycorrhizal fungi our sustainable saviours? Considerations for achieving food security. J. Ecol. 2017, 105, 921–929. [Google Scholar] [CrossRef] [Green Version]
- Fester, T.; Sawers, R. Progress and Challenges in Agricultural Applications of Arbuscular Mycorrhizal Fungi. Crit. Rev. Plant Sci. 2011, 30, 459–470. [Google Scholar] [CrossRef]
- Miozzi, L.; Vaira, A.M.; Catoni, M.; Fiorilli, V.; Accotto, G.P.; Lanfranco, L. Arbuscular Mycorrhizal Symbiosis: Plant Friend or Foe in the Fight Against Viruses? Front. Microbiol. 2019, 10, 1238. [Google Scholar] [CrossRef] [PubMed]
- Bahadur, A.; Batool, A.; Nasir, F.; Jiang, S.; Mingsen, Q.; Zhang, Q.; Pan, J.; Liu, Y.; Feng, H. Mechanistic Insights into Arbuscular Mycorrhizal Fungi-Mediated Drought Stress Tolerance in Plants. Int. J. Mol. Sci. 2019, 20, 4199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Daei, G.; Ardekani, M.; Rejali, F.; Teimuri, S.; Miransari, M. Alleviation of salinity stress on wheat yield, yield components, and nutrient uptake using arbuscular mycorrhizal fungi under field conditions. J. Plant Physiol. 2009, 166, 617–625. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Li, X.; Gao, Y.; Liu, H.; Gao, Y.; van der Heijden, M.G.A.; Ren, A. Plant endophytes and arbuscular mycorrhizal fungi alter plant competition. Funct. Ecol. 2018, 32, 1168–1179. [Google Scholar] [CrossRef]
- Zhang, Y.-C.; Wang, P.; Wu, Q.-H.; Zou, Y.-N.; Bao, Q.; Wu, Q.-S. Arbuscular mycorrhizas improve plant growth and soil structure in trifoliate orange under salt stress. Arch. Agron. Soil Sci. 2016, 63, 491–500. [Google Scholar] [CrossRef]
- De Novais, C.B.; Sbrana, C.; Jesus, E.D.C.; Rouws, L.F.M.; Giovannetti, M.; Avio, L.; Siqueira, J.O.; Júnior, O.J.S.; Da Silva, E.M.R.; De Faria, S.M. Mycorrhizal networks facilitate the colonization of legume roots by a symbiotic nitrogen-fixing bacterium. Mycorrhiza 2020, 30, 389–396. [Google Scholar] [CrossRef]
- Piazza, G.; Ercoli, L.; Nuti, M.; Pellegrino, E. Interaction Between Conservation Tillage and Nitrogen Fertilization Shapes Prokaryotic and Fungal Diversity at Different Soil Depths: Evidence From a 23-Year Field Experiment in the Mediterranean Area. Front. Microbiol. 2019, 10, 2047. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Treseder, K.K. A meta-analysis of mycorrhizal responses to nitrogen, phosphorus, and atmospheric CO2 in field studies. New Phytol. 2004, 164, 347–355. [Google Scholar] [CrossRef] [Green Version]
- Egerton-Warburton, L.M.; Johnson, N.C.; Allen, E.B. Mycorrhizal Community Dynamics Following Nitrogen Fertilization: A Cross-Site Test in Five Grasslands. Ecol. Monogr. 2007, 77, 527–544. [Google Scholar] [CrossRef] [Green Version]
- Marklein, A.R.; Houlton, B.Z. Nitrogen inputs accelerate phosphorus cycling rates across a wide variety of terrestrial ecosystems. New Phytol. 2012, 193, 696–704. [Google Scholar] [CrossRef]
- Ortas, I. Mycorrhizal species significantly increase citrus yield and nutrient concentration under field conditions. Acta Hortic. 2018, 1217, 171–178. [Google Scholar] [CrossRef]
- Ortas, I. Role of mycorrhizae on mineral nutrition of fruit trees. Acta Hortic. 2018, 1217, 271–284. [Google Scholar] [CrossRef]
- Wrona, D. Effect of nitrogen fertilization on growth, cropping and fruit quality of ‘Šampion’ apple trees during 9 years after planting. Folia Hortic. 2004, 16, 55–60. [Google Scholar]
- Kjeldahl, J. Neue Methode zur Bestimmung des Stickstoffs in organischen Körpern. Anal. Bioanal. Chem. 1883, 22, 366–382. [Google Scholar] [CrossRef] [Green Version]
- Derkowska, E.; Paszt, L.S.; Sumorok, B.; Dyki, B. Colonisation of apple and blackcurrant roots by arbuscular mycorrhizal fungi following mycorrhisation and the use of organic mulches. Folia Hortic. 2013, 25, 117–122. [Google Scholar] [CrossRef] [Green Version]
- Derkowska, E.; Paszt, L.S.; Dyki, B.; Sumorok, B. Assessment of Mycorrhizal Frequency in the Roots of Fruit Plants Using Different Dyes. Adv. Microbiol. 2015, 5, 54–64. [Google Scholar] [CrossRef] [Green Version]
- Trouvelot, A.; Kough, J.L.; Gianinazzi-Pearson, V. Mesure du Taux de Mycorhization VA d’un Systemeradiculaire. Recherche de Methods D’estimation Avant une Signification Fonctionnelle. In Physiological and Genetical Aspects of Mycorrhizae, 1st ed.; Gia-ninazzi-Pearson, V., Gianinazzi, S., Eds.; INRA: Paris, France, 1986; pp. 217–221. [Google Scholar]
- Przybyłko, S.; Kowalczyk, W.; Wrona, D. The Effect of Mycorrhizal Fungi and PGPR on Tree Nutritional Status and Growth in Organic Apple Production. Agronomy 2021, 11, 1402. [Google Scholar] [CrossRef]
- Balota, E.L.; Machineski, O.; Truber, P.V.; Scherer, A.; De Souza, F.S. Physic nut plants present high mycorrhizal dependency under conditions of low phosphate availability. Braz. J. Plant Physiol. 2011, 23, 33–44. [Google Scholar] [CrossRef]
- Nouri, E.; Breuillin-Sessoms, F.; Feller, U.; Reinhardt, D. Phosphorus and Nitrogen Regulate Arbuscular Mycorrhizal Symbiosis in Petunia hybrida. PLoS ONE 2014, 9, e90841. [Google Scholar] [CrossRef]
- Mei, L.; Yang, X.; Zhang, S.; Zhang, T.; Guo, J. Arbuscular mycorrhizal fungi alleviate phosphorus limitation by reducing plant N:P ratios under warming and nitrogen addition in a temperate meadow ecosystem. Sci. Total. Environ. 2019, 686, 1129–1139. [Google Scholar] [CrossRef]
- Gao, X.; Guo, H.; Zhang, Q.; Guo, H.; Zhang, L.; Zhang, C.; Gou, Z.; Liu, Y.; Wei, J.; Chen, A.; et al. Arbuscular mycorrhizal fungi (AMF) enhanced the growth, yield, fiber quality and phosphorus regulation in upland cotton (Gossypium hirsutum L.). Sci. Rep. 2020, 10, 2084. [Google Scholar] [CrossRef] [Green Version]
- Pan, S.; Wang, Y.; Qiu, Y.; Chen, D.; Zhang, L.; Ye, C.; Guo, H.; Zhu, W.; Chen, A.; Xu, G.; et al. Nitrogen-induced acidification, not N-nutrient, dominates suppressive N effects on arbuscular mycorrhizal fungi. Glob. Chang. Biol. 2020, 26, 6568–6580. [Google Scholar] [CrossRef]
- Gyuricza, V.; De Boulois, H.D.; Declerck, S. Effect of potassium and phosphorus on the transport of radiocesium by arbuscular mycorrhizal fungi. J. Environ. Radioact. 2010, 101, 482–487. [Google Scholar] [CrossRef] [PubMed]
- Priyadharsini, P.; Muthukumar, T. Interactions between arbuscular mycorrhizal fungi and potassium-solubilizing microorganisms on agricultural productivity. In Potassium Solubilizing Microorganisms for Sustainable Agriculture; Meena, V.S., Maurya, B.R., Verma, J.P., Meena, R.S., Eds.; Springer: New Delhi, India, 2016; pp. 111–125. ISBN 978-81-322-2774-8. [Google Scholar] [CrossRef]
| Year | Treatment | Nitrogen Fertilization | Mycorrhizal Parameters (%) | ||
|---|---|---|---|---|---|
| F | AMI | RMI | |||
| 2012 | M0 | N-0 | 23.33 ± 8.82 | 0.093 ± 0.05 | 0.378 ± 0.11 |
| N-50 | 20.33 ± 6.69 | 0.079 ± 0.04 | 0.375 ± 0.11 | ||
| N-100 | 40.00 ± 9.96 | 0.133 ± 0.05 | 0.350 ± 0.13 | ||
| N-50+50 | 27.67 ± 4.04 | 0.084 ± 0.07 | 0.308 ± 0.06 | ||
| N-50h | 24.67 ± 7.04 | 0.089 ± 0.04 | 0.350 ± 0.13 | ||
| M | N-0 | 17.67 ± 6.94 | 0.055 ± 0.03 | 0.198 ± 0.03 | |
| N-50 | 12.38 ± 5.18 | 0.067 ± 0.08 | 0.243 ± 0.07 | ||
| N-100 | 27.67 ± 8.57 | 0.081 ± 0.02 | 0.267 ± 0.03 | ||
| N-50+50 | 32.00 ± 6.57 | 0.100 ± 0.05 | 0.294 ± 0.04 | ||
| N-50h | 14.67 ± 7.81 | 0.073 ± 0.02 | 0.238 ± 0.02 | ||
| 2016 | M0 | N-0 | 27.78 ± 7.91 | 0.278 ± 0.10 | 1.000 ± 0.12 |
| N-50 | 28.89 ± 9.21 | 0.644 ± 0.25 | 2.222 ± 0.19 | ||
| N-100 | 31.11 ± 6.98 | 1.400 ± 0.68 | 4.033 ± 2.72 | ||
| N-50+50 | 33.33 ± 6.67 | 0.511 ± 0.10 | 1.600 ± 0.53 | ||
| N-50h | 31.11 ± 8.11 | 0.489 ± 0.25 | 1.489 ± 0.43 | ||
| M | N-0 | 51.11 ± 7.70 | 5.822 ± 1.32 | 11.64 ± 3.62 | |
| N-50 | 44.05 ± 14.37 | 3.878 ± 0.49 | 7.197.637± | ||
| N-100 | 37.78 ± 8.85 | 2.022 ± 0.96 | 5.294 ± 2.21 | ||
| N-50+50 | 34.44 ± 5.08 | 0.967 ± 0.32 | 2.822 ± 0.96 | ||
| N-50h | 42.23 ± 9.52 | 5.044 ± 1.86 | 12.13 ± 5.12 | ||
| 2020 | M0 | N-0 | 70.00 ± 8.82 | 1.100 ± 0.21 | 1.564 ± 0.15 |
| N-50 | 64.44 ± 7.69 | 0.822 ± 0.20 | 1.269 ± 0.24 | ||
| N-100 | 77.78 ± 18.36 | 4.511 ± 3.25 | 5.086 ± 2.06 | ||
| N-50+50 | 65.00 ± 8.34 | 1.002 ± 0.29 | 1.521 ± 0.26 | ||
| N-50h | 72.22 ± 11.71 | 1.922 ± 1.05 | 2.547 ± 1.06 | ||
| M | N-0 | 100.0 ± 0.00 | 23.74 ± 6.82 | 23.74 ± 6.82 | |
| N-50 | 100.0 ± 0.00 | 34.33 ± 4.26 | 34.33 ± 4.26 | ||
| N-100 | 100.0 ± 0.00 | 24.84 ± 5.96 | 24.84 ± 5.96 | ||
| N-50+50 | 100.0 ± 0.00 | 36.56 ± 2.60 | 36.56 ± 2.60 | ||
| N-50h | 100.0 ± 0.00 | 34.56 ± 8.47 | 34.56 ± 8.47 | ||
| Year | <0.0001 | <0.0001 | <0.0001 | ||
| Treatment | <0.0001 | <0.0001 | <0.0001 | ||
| Nitrogen fertilization | 0.5147 | 0.2396 | 0.3096 | ||
| Year × Treatment | <0.0001 | <0.0001 | <0.0001 | ||
| Year × Nitrogen fertilization | 0.5921 | 0.02899 | 0.0256 | ||
| Treatment × Nitrogen fertilization | 0.1889 | 0.0197 | 0.0204 | ||
| Year × Treatment × Nitrogen fertilization | 0.7641 | 0.0027 | 0.0026 | ||
| Treatment | Nitrogen Fertilization | Macronutrient (% d.m.) | ||||
|---|---|---|---|---|---|---|
| N | P | K | Mg | Ca | ||
| M0 | N-0 | A 2.17 ± 0.05 a | A 0.204 ± 0.007 d | A 1.02 ± 0.06 b | A 0.25 ± 0.01 ab | B 1.74 ± 0.07 b |
| N-50 | A 2.33 ± 0.03 b | A 0.164 ± 0.004 b | A 0.83 ± 0.04 a | B 0.27 ± 0.01 b | B 1.81 ± 0.05 b | |
| N-100 | A 2.24 ± 0.02 bc | A 0.156 ± 0.003 a | B 1.04 ± 0.05 b | B 0.25 ± 0.01 a | A 1.60 ± 0.04 a | |
| N-50+50 | A 2.40 ± 0.02 c | A 0.176 ± 0.006 c | A 0.82 ± 0.07 a | A 0.31 ± 0.02 c | A 1.73 ± 0.05 b | |
| N-50h | A 2.41 ± 0.06 c | A 0.169 ± 0.003 bc | A 0.83 ± 0.03 a | B 0.26 ± 0.01 ab | B 1.76 ± 0.01 b | |
| M | N-0 | B 2.28 ± 0.02 a | B 0.215 ± 0.004 bc | A 1.08 ± 0.03 b | A 0.25 ± 0.01 a | A 1.62 ± 0.06 bc |
| N-50 | B 2.40 ± 0.03 c | B 0.210 ± 0.005 b | B 1.03 ± 0.08 b | A 0.25 ± 0.01 a | A 1.54 ± 0.04 c | |
| N-100 | A 2.35 ± 0.04 bc | B 0.198 ± 0.007 a | A 0.95 ± 0.03 ab | A 0.23 ± 0.01 a | A 1.57 ± 0.06 b | |
| N-50+50 | A 2.41 ± 0.02 c | B 0.212 ± 0.005 bc | B 0.92 ± 0.05 a | A 0.30 ± 0.02 b | A 1.68 ± 0.04 a | |
| N-50h | A 2.34 ± 0.03 b | B 0.219 ± 0.006 c | B 0.97 ± 0.06 ab | A 0.24 ± 0.01 a | A 1.57 ± 0.07 b | |
| Treatment | Nitrogen Fertilization | Macronutrient (% d.m.) | |||
|---|---|---|---|---|---|
| P | K | Mg | Ca | ||
| M0 | N-0 | A 0.063 ± 0.004 b | B 0.633 ± 0.007 b | A 0.027 ± 0.001 a | A 0.031 ± 0.001 a |
| N-50 | B 0.060 ± 0.004 b | B 0.571 ± 0.019 a | A 0.027 ± 0.001 a | A 0.035 ± 0.002 a | |
| N-100 | B 0.054 ± 0.003 a | B 0.655 ± 0.010 b | B 0.028 ± 0.002 a | B 0.034 ± 0.004 a | |
| N-50+50 | A 0.058 ± 0.003 ab | A 0.566 ± 0.010 a | A 0.027 ± 0.001 a | A 0.033 ± 0.002 a | |
| N-50h | A 0.064 ± 0.003 b | A 0.597 ± 0.007 a | A 0.027 ± 0.003 a | A 0.036 ± 0.002 a | |
| M | N-0 | A 0.061 ± 0.004 b | A 0.556 ± 0.010 b | A 0.027 ± 0.001 b | A 0.035 ± 0.003 b |
| N-50 | A 0.056 ± 0.004 b | A 0.521 ± 0.017 a | A 0.026 ± 0.001 ab | A 0.031 ± 0.001 ab | |
| N-100 | A 0.047 ± 0.003 a | A 0.502 ± 0.023 a | A 0.024 ± 0.001 a | A 0.028 ± 0.002 a | |
| N-50+50 | A 0.056 ± 0.003 b | B 0.618 ± 0.009 c | A 0.027 ± 0.001 b | A 0.033 ± 0.003 ab | |
| N-50h | A 0.060 ± 0.001 b | B 0.624 ± 0.021 c | A 0.027 ± 0.001 b | A 0.034 ± 0.006 ab | |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bokszczanin, K.Ł.; Wrona, D.; Przybyłko, S. The Effect of Microbial Inoculation under Various Nitrogen Regimes on the Uptake of Nutrients by Apple Trees. Agronomy 2021, 11, 2348. https://doi.org/10.3390/agronomy11112348
Bokszczanin KŁ, Wrona D, Przybyłko S. The Effect of Microbial Inoculation under Various Nitrogen Regimes on the Uptake of Nutrients by Apple Trees. Agronomy. 2021; 11(11):2348. https://doi.org/10.3390/agronomy11112348
Chicago/Turabian StyleBokszczanin, Kamila Łucja, Dariusz Wrona, and Sebastian Przybyłko. 2021. "The Effect of Microbial Inoculation under Various Nitrogen Regimes on the Uptake of Nutrients by Apple Trees" Agronomy 11, no. 11: 2348. https://doi.org/10.3390/agronomy11112348
APA StyleBokszczanin, K. Ł., Wrona, D., & Przybyłko, S. (2021). The Effect of Microbial Inoculation under Various Nitrogen Regimes on the Uptake of Nutrients by Apple Trees. Agronomy, 11(11), 2348. https://doi.org/10.3390/agronomy11112348






