Disease Severity, Resistance Analysis, and Expression Profiling of Pathogenesis-Related Protein Genes after the Inoculation of Fusarium equiseti in Wheat
Abstract
:1. Introduction
2. Materials and Methods
2.1. Fungus Culture and Inoculum Preparation
2.2. Seed Sterilization and Soil Inoculation
2.3. Disease Severity Analysis
2.4. Biochemical Parameters
2.5. Physiological Parameters
2.6. RNA Extraction, Quantification, and cDNA Synthesis
2.7. Primer Designing, RT-PCR, and qRT-PCR Analysis
2.8. Statistical Data Analysis
3. Results
3.1. Disease Severity Profiling
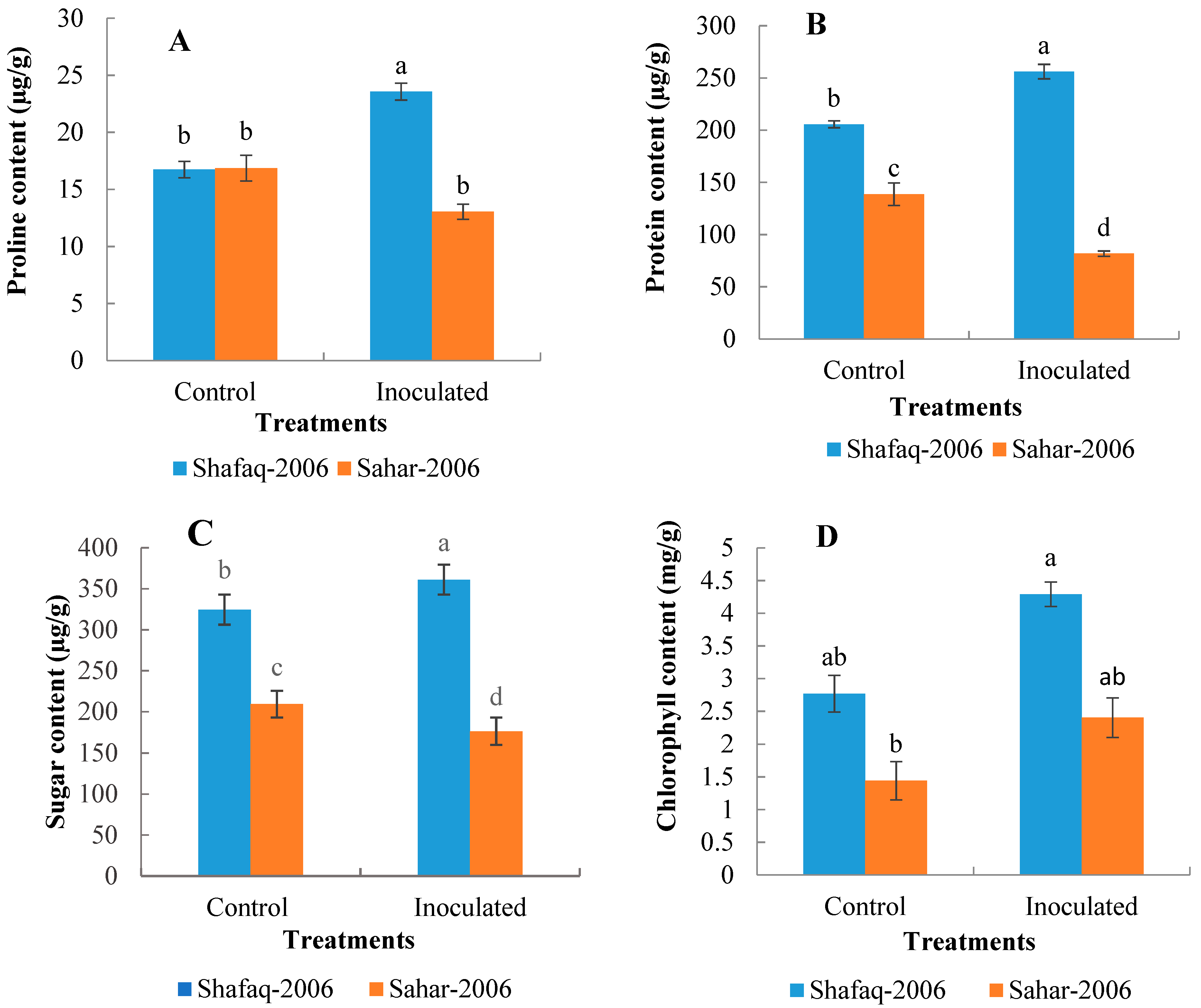
3.2. Biochemical Parameters
3.3. Physiological Parameters
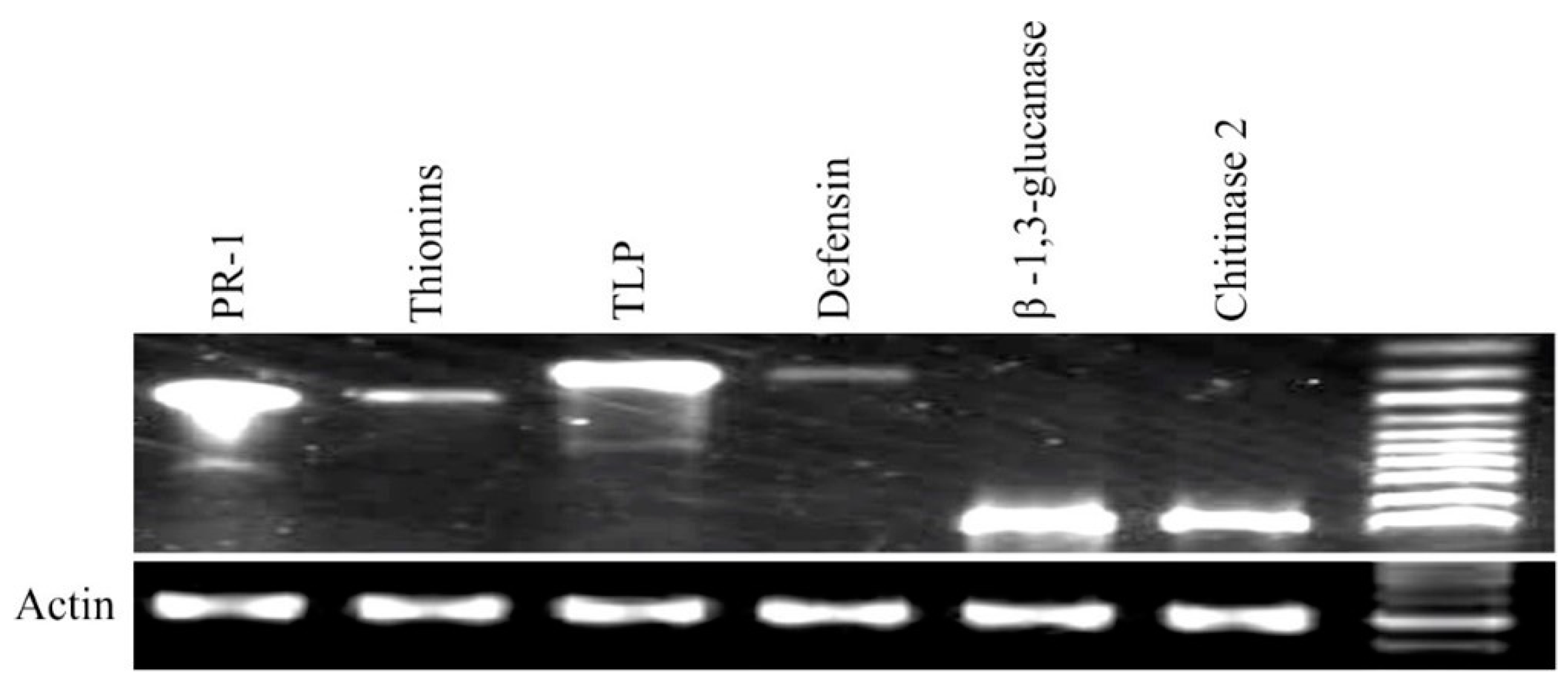
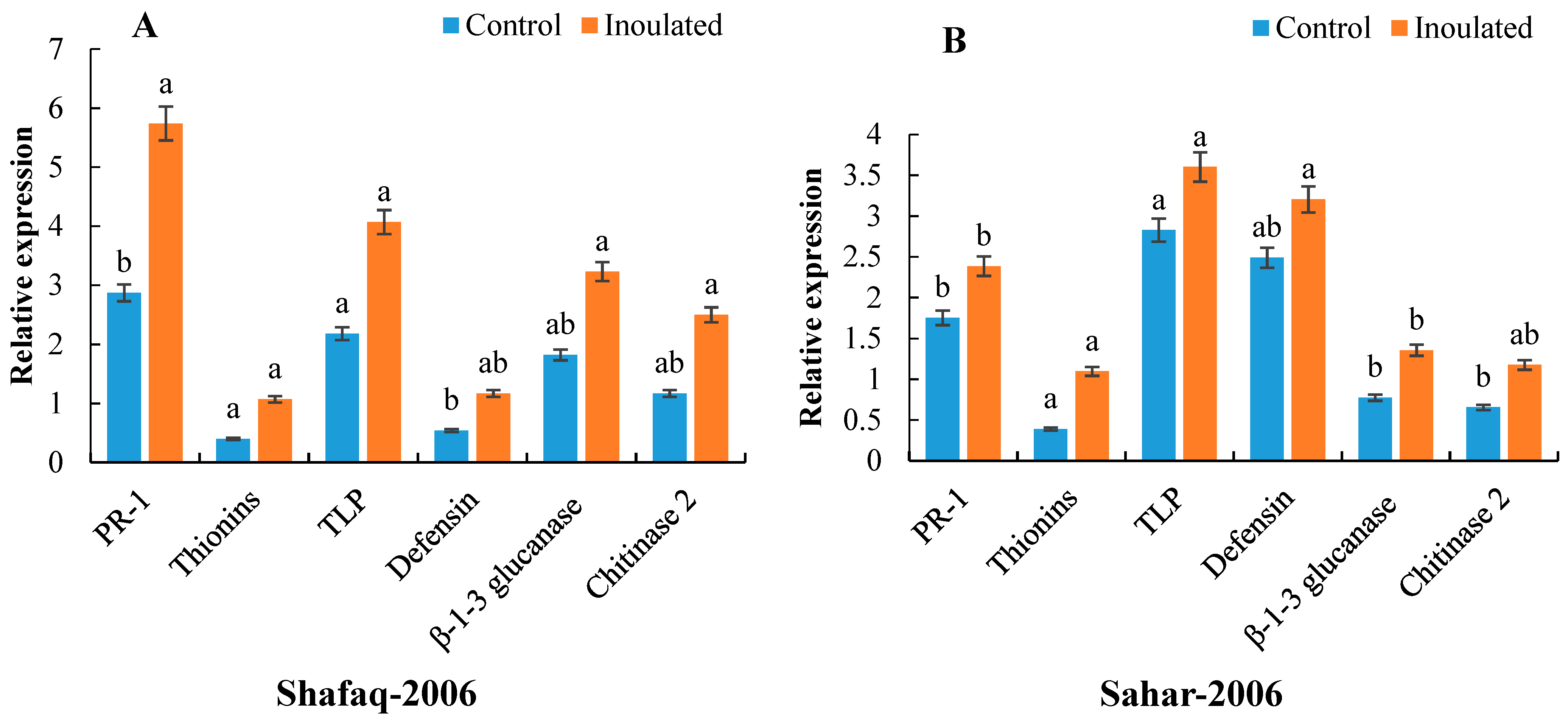
3.4. Expression Analysis by RT-PCR and qRT-PCR
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Panhwar, Q.A.; Ali, A.; Depar, N.; Shah, J.A. Screening of plant growth promoting rhizobacteria for sustainable wheat (Triticum aestivum L.) crop production. Pak. J. Bot. 2020, 52, 345–353. [Google Scholar] [CrossRef]
- Kong, H.; Zhang, Z.; Qin, J.; Akram, N.A. Interactive effects of abscisic acid (ABA) and drought stress on the physiological responses of winter wheat (Triticum aestivum L.). Pak. J. Bot. 2021, 53, 1545–1551. [Google Scholar] [CrossRef]
- Naz, S.; Perveen, S. Response of wheat (Triticum aestivum L. var. galaxy-2013) to pre-sowing seed treatment with thiourea under drought stress. Pak. J. Bot. 2021, 53, 1209–1217. [Google Scholar] [CrossRef]
- Messiaen, C.; Cassini, R. Recherches sur les Fusarioses. IV. La systématique des Fusarium. Ann. Epiphyt. 1968, 19, 387–454. [Google Scholar]
- Booth, C. The genus fusarium. Genus Fusarium, 1971; 237p. [Google Scholar] [CrossRef]
- Shaner, G. Epidemiology of Fusarium head blight of small grain cereals in North America. Fusarium Head Blight Wheat Barley 2003, 84–119. [Google Scholar]
- Gale, L. Population biology of Fusarium species causing head blight of grain crops. Fusarium Head Blight Wheat Barley 2003, 120–143. [Google Scholar]
- Palmero, D.; Iglesias, C.; De Cara, M.; Lomas, T.; Santos, M.; Tello, J. Species of Fusarium isolated from river and sea water of southeastern Spain and pathogenicity on four plant species. Plant Dis. 2009, 93, 377–385. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Munis, M.F.H.; Tu, L.; Deng, F.; Tan, J.; Xu, L.; Xu, S.; Long, L.; Zhang, X. A thaumatin-like protein gene involved in cotton fiber secondary cell wall development enhances resistance against Verticillium dahliae and other stresses in transgenic tobacco. Biochem. Biophys. Res. Commun. 2010, 393, 38–44. [Google Scholar] [CrossRef]
- Van Loon, L. Induced resistance in plants and the role of pathogenesis-related proteins. Eur. J. Plant Pathol. 1997, 103, 753–765. [Google Scholar] [CrossRef]
- Xu, L.; Zhu, L.; Tu, L.; Guo, X.; Long, L.; Sun, L.; Gao, W.; Zhang, X. Differential gene expression in cotton defence response to Verticillium dahliae by SSH. J. Phytopathol. 2011, 159, 606–615. [Google Scholar] [CrossRef]
- Veronese, P.; Ruiz, M.T.; Coca, M.A.; Hernandez-Lopez, A.; Lee, H.; Ibeas, J.I.; Damsz, B.; Pardo, J.M.; Hasegawa, P.M.; Bressan, R.A. In defense against pathogens. Both plant sentinels and foot soldiers need to know the enemy. Plant Physiol. 2003, 131, 1580–1590. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mohamed, I.A.; Shalby, N.; MA El-Badri, A.; Saleem, M.H.; Khan, M.N.; Nawaz, M.A.; Qin, M.; Agami, R.A.; Kuai, J.; Wang, B. Stomata and xylem vessels traits improved by melatonin application contribute to enhancing salt tolerance and fatty acid composition of Brassica napus L. plants. Agronomy 2020, 10, 1186. [Google Scholar] [CrossRef]
- Hameed, A.; Akram, N.A.; Saleem, M.H.; Ashraf, M.; Ahmed, S.; Ali, S.; Abdullah Alsahli, A.; Alyemeni, M.N. Seed treatment with α-tocopherol regulates growth and key physio-biochemical attributes in carrot (Daucus carota L.) plants under water limited regimes. Agronomy 2021, 11, 469. [Google Scholar] [CrossRef]
- Nawaz, M.; Wang, X.; Saleem, M.H.; Khan, M.H.U.; Afzal, J.; Fiaz, S.; Ali, S.; Ishaq, H.; Khan, A.H.; Rehman, N.; et al. Deciphering plantago ovata forsk leaf extract mediated distinct germination, growth and physio-biochemical improvements under water stress in maize (Zea mays L.) at early growth stage. Agronomy 2021, 11, 1404. [Google Scholar] [CrossRef]
- Borad, V.; Sriram, S. Pathogenesis-related proteins for the plant protection. Asian J Exp Sci 2008, 22, 189–196. [Google Scholar]
- Sudisha, J.; Sharathchandra, R.; Amruthesh, K.; Kumar, A.; Shetty, H.S. Pathogenesis related proteins in plant defense response. In Plant Defence: Biological Control; Springer: Berlin/Heidelberg, Germany, 2012; pp. 379–403. [Google Scholar]
- Bohlmann, H.; Clausen, S.; Behnke, S.; Giese, H.; Hiller, C.; Reimann-Philipp, U.; Schrader, G.; Barkholt, V.; Apel, K. Leaf-specific thionins of barley—A novel class of cell wall proteins toxic to plant-pathogenic fungi and possibly involved in the defence mechanism of plants. EMBO J. 1988, 7, 1559–1565. [Google Scholar] [CrossRef]
- Kang, Z.; Buchenauer, H. Immunocytochemical localization of cell wall-bound thionins and hydroxyproline-rich glycoproteins in Fusarium culmorum-infected wheat spikes. J. Phytopathol. 2003, 151, 120–129. [Google Scholar] [CrossRef]
- Bryngelsson, T.; Green, B. Characterization of a pathogenesis-related, thaumatin-like protein isolated from barley challenged with an incompatible race of mildew. Physiol. Mol. Plant Pathol. 1989, 35, 45–52. [Google Scholar] [CrossRef]
- Pierpoint, W.; Jackson, P.; Evans, R.M. The presence of a thaumatin-like protein, a chitinase and a glucanase among the pathogenesis-related proteins of potato (Solanum tuberosum). Physiol. Mol. Plant Pathol. 1990, 36, 325–338. [Google Scholar] [CrossRef]
- Ebrahim, S.; Usha, K.; Singh, B. Pathogenesis related (PR) proteins in plant defense mechanism. Sci. Against Microb. Pathog. 2011, 2, 1043–1054. [Google Scholar]
- Anguelova-Merhar, V.; VanDer Westhuizen, A.; Pretorius, Z. β-1, 3-glucanase and chitinase activities and the resistance response of wheat to leaf rust. J. Phytopathol. 2001, 149, 381–384. [Google Scholar] [CrossRef]
- Nielsen, K.K.; Mikkelsen, J.D.; Kragh, K.M.; Bojsen, K. An acidic class III chitinase in sugar beet: Induction by Cercospora beticola, characterization, and expression in transgenic tobacco plants. Mol. Plant Microb. Interact. 1993, 6, 495. [Google Scholar] [CrossRef]
- Lawrence, C.B.; Singh, N.P.; Qiu, J.; Gardner, R.G.; Tuzun, S. Constitutive hydrolytic enzymes are associated with polygenic resistance of tomato to Alternaria solani and may function as an elicitor release mechanism. Physiol. Mol. Plant Pathol. 2000, 57, 211–220. [Google Scholar] [CrossRef]
- Caruso, C.; Chilosi, G.; Caporale, C.; Leonardi, L.; Bertini, L.; Magro, P.; Buonocore, V. Induction of pathogenesis-related proteins in germinating wheat seeds infected with Fusarium culmorum. Plant Sci. 1999, 140, 87–97. [Google Scholar] [CrossRef]
- Mauch, F.; Staehelin, L.A. Functional Implications of the Subcellular Localization of Ethylene-Induced Chitinase and [beta]-1, 3-Glucanase in Bean Leaves. Plant Cell 1989, 1, 447–457. [Google Scholar] [CrossRef]
- Kombrink, E.; Somssich, I. Pathogenesis-related proteins and plant defense. In Plant Relationships; Springer: Berlin/Heidelberg, Germany, 1997; pp. 107–128. [Google Scholar]
- Broekaert, W.F.; Cammue, B.P.; De Bolle, M.F.; Thevissen, K.; De Samblanx, G.W.; Osborn, R.W.; Nielson, K. Antimicrobial peptides from plants. Crit. Rev. Plant Sci. 1997, 16, 297–323. [Google Scholar] [CrossRef]
- De Oliveira Carvalho, A.; Moreira Gomes, V. Plant defensins and defensin-like peptides-biological activities and biotechnological applications. Curr. Pharm. Design 2011, 17, 4270–4293. [Google Scholar] [CrossRef] [PubMed]
- Atiq-ur-Rehman, R.; Shahzad, A.; Muhammad, F.; Muhammad, Z.; Shamim, I.; Yasmin, A. Status of foliar diseases of wheat in Punjab, Pakistan. Mycopath 2011, 9, 39–42. [Google Scholar]
- Wains, M.S.; Ali, M.A.; Hussain, M.; Anwar, J.; Zulkiffal, M.; Sabir, W. Aphid dynamics in relation to meteorological factors and various management practices in bread wheat. J. Plant Protect. Res. 2010, 50, 385–392. [Google Scholar] [CrossRef]
- Beniwal, M.; Chawla, P.; Singh, R. Comparison of different methods of inoculation and evaluation of wheat material against Neovossia indica (Mitra) Mundkur. Crop Res. (Hisar) 2001, 21, 105–108. [Google Scholar]
- Ullah, H.Z.; Haque, M.; Rauf, C.; Akhtar, L.; Munir, M. Comparative virulence in isolates of Tilletia indica and host resistance against Karnal bunt of wheat. J. Animal Plant Sci. 2012, 22, 467–472. [Google Scholar]
- Ibrahim, T.; Bano, A.; Chaudhary, H.J.; Imran, M.; Mehmood, Z.; Hassan, S.W.; Muhammad, N.; Naqvi, S.A.H.; Munis, M.F.H. Evaluation of different inoculation methods for the induction of spot blotch caused by Helminthosporium sativum in wheat. Philipp. J. Crop Sci. 2014, 39, 27–33. [Google Scholar]
- Ahmed, B.; Sharif, F.; Sarhan, A. Effect of certain micronutrients on Fusarium wilt of tomato. J. Agric. Water Res. Plant Protec. 1987, 6, 13–28. [Google Scholar]
- Dubois, M.; Gilles, K.A.; Hamilton, J.K.; Rebers, P.T.; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem. 1956, 28, 350–356. [Google Scholar] [CrossRef]
- Johnson, R.R.; Balwani, T.L.; Johnson, L.; McClure, K.; Dehority, B. Corn plant maturity. II. Effect on in vitro cellulose digestibility and soluble carbohydrate content. J. Animal Sci. 1966, 25, 617–623. [Google Scholar] [CrossRef]
- Bates, L.S.; Waldren, R.P.; Teare, I. Rapid determination of free proline for water-stress studies. Plant Soil 1973, 39, 205–207. [Google Scholar] [CrossRef]
- Lowry, O.H.; Rosebrough, N.J.; Farr, A.L.; Randall, R.J. Protein measurement with the Folin phenol reagent. J. Biol. Chem. 1951, 193, 265–275. [Google Scholar] [CrossRef]
- Arnon, D.I. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in Beta vulgaris. Plant Physiol. 1949, 24, 1. [Google Scholar] [CrossRef] [Green Version]
- Wheatherley, P. Studies in the water relations of cotton plants. I. The field measurement of water deficit in leaves. New Phytol. 1950, 49, 81–87. [Google Scholar] [CrossRef]
- Claussen, W. Proline as a measure of stress in tomato plants. Plant Sci. 2005, 168, 241–248. [Google Scholar] [CrossRef]
- Mattioli, R.; Costantino, P.; Trovato, M. Proline accumulation in plants: Not only stress. Plant Signal. Behav. 2009, 4, 1016–1018. [Google Scholar] [CrossRef] [PubMed]
- Vendruscolo, E.C.G.; Schuster, I.; Pileggi, M.; Scapim, C.A.; Molinari, H.B.C.; Marur, C.J.; Vieira, L.G.E. Stress-induced synthesis of proline confers tolerance to water deficit in transgenic wheat. J. Plant Physiol. 2007, 164, 1367–1376. [Google Scholar] [CrossRef]
- Qayyum, A.; Razzaq, A.; Ahmad, M.; Jenks, M.A. Water stress causes differential effects on germination indices, total soluble sugar and proline content in wheat (Triticum aestivum L.) genotypes. Afr. J. Biotechnol. 2011, 10, 14038–14045. [Google Scholar]
- Amirjani, M.R.; Mahdiyeh, M. Antioxidative and biochemical responses of wheat to drought stress. J. Agric. Biol. Sci. 2013, 8, 291–301. [Google Scholar]
- Arjunan, G.; Vidhyasekaran, P.; Kandaswamy, T.K. Changes in amino acids and amides content in jowar leaves infected by Helminthosporium turcicum. Curr. Sci. (India) 1976, 45, 229–230. [Google Scholar]
- Mohammadkhani, N.; Heidari, R. Water Stress Induced by Polyethylene Glycol 6000 and Sodium. Pak. J. Biol. Sci. 2008, 2, 92–97. [Google Scholar]
- Nagaveni, T. Resistance Factors for Leaf Blight of Barley Caused by Helminthosporium Sativum Pam, King and Bakke; University of Agricultural Sciences, Bangalore: Bengaluru, India, 2005. [Google Scholar]
- Klement, Z.; Goodman, R. The hypersensitive reaction to infection by bacterial plant pathogens. Annu. Rev. Phytopathol. 1967, 5, 17–44. [Google Scholar] [CrossRef]
- Manghwar, H.; Hussain, A.; Ullah, A.; Gul, S.; Shaban, M.; Khan, A.H.; Ali, M.; Sani, S.G.A.S.; Chaudhary, H.J.; Munis, M.F.H. Expression analysis of defense related genes in wheat and maize against Bipolaris sorokiniana. Physiol. Mol. Plant Pathol. 2018, 103, 36–46. [Google Scholar] [CrossRef]
- Krishna, K.; Bagyaraj, D. Growth and nutrient uptake of peanut inoculated with the mycorrhizal fungus Glomus fasciculatum compared with non-inoculated ones. Plant Soil 1984, 77, 405–408. [Google Scholar] [CrossRef]
- Anjum, S.A.; Xie, X.-Y.; Wang, L.-C.; Saleem, M.F.; Man, C.; Lei, W. Morphological, physiological and biochemical responses of plants to drought stress. Afr. J. Agric. Res. 2011, 6, 2026–2032. [Google Scholar]
- Devi, M.C.; Reddy, M. Growth response of groundnut to VAM fungus and Rhizobium inoculation. Plant Pathol. Bull. 2001, 10, 71–78. [Google Scholar]
- Rajasekaran, S.; Nagarajan, S. Effect of dual inoculation (AM Fungi and Rhizobium) on chlorophyll content of Vigna unguiculata (L.) Walp. Var. Pusa 151. Mycorrhiza News 2005, 17, 10–11. [Google Scholar]
- Aroca, R.; Ruiz-Lozano, J.M.; Zamarreño, Á.M.; Paz, J.A.; García-Mina, J.M.; Pozo, M.J.; López-Ráez, J.A. Arbuscular mycorrhizal symbiosis influences strigolactone production under salinity and alleviates salt stress in lettuce plants. J. Plant Physiol. 2013, 170, 47–55. [Google Scholar] [CrossRef]
- Wang, J.; Fu, Z.; Ren, Q.; Zhu, L.; Lin, J.; Zhang, J.; Cheng, X.; Ma, J.; Yue, J. Effects of arbuscular mycorrhizal fungi on growth, photosynthesis, and nutrient uptake of Zelkova serrata (Thunb.) Makino seedlings under salt stress. Forests 2019, 10, 186. [Google Scholar] [CrossRef] [Green Version]
- Niakan, M.; Darvishkhezri, M.; Iranbakhsh, A.; Barzegar, A. Changes of sorghum growth in response to drought and allelopathy stresses. Annals Biol. Res. 2013, 4, 18–22. [Google Scholar]
- Younis, M.; El-Shahaby, O.; Abo-Hamed, S.; Ibrahim, A. Effects of water stress on growth, pigments and 14CO2 assimilation in three sorghum cultivars. J. Agron. Crop Sci. 2000, 185, 73–82. [Google Scholar] [CrossRef]
- Al-Abdalall, A.H.A. Assessment of yield loss caused by root rots in wheat and barley. J. Food Agric. Environ. 2010, 8, 638–641. [Google Scholar]
- Jafar, M.S.; Nourmohammadi, G.; Maleki, A. Effect of water deficit on seedling, plantlets and compatible solutes of forage Sorghum cv. Speedfeed. In Proceedings of the 4th International Crop Science Congress, Brisbane, Austrialia, 26 September–1 October 2004. [Google Scholar]
- Eyal, Y.; Sagee, O.; Fluhr, R. Dark-induced accumulation of a basic pathogenesis-related (PR-1) transcript and a light requirement for its induction by ethylene. Plant Mol. Biol. 1992, 19, 589–599. [Google Scholar] [CrossRef]
- Velazhahan, R.; Muthukrishnan, S. Transgenic tobacco plants constitutively overexpressing a rice thaumatin-like protein (PR-5) show enhanced resistance to Alternaria alternata. Biol. Plant. 2003, 47, 347–354. [Google Scholar] [CrossRef]
- Datta, K.; Velazhahan, R.; Oliva, N.; Ona, I.; Mew, T.; Khush, G.; Muthukrishnan, S.; Datta, S. Over-expression of the cloned rice thaumatin-like protein (PR-5) gene in transgenic rice plants enhances environmental friendly resistance to Rhizoctonia solani causing sheath blight disease. Theor. Appl. Genet. 1999, 98, 1138–1145. [Google Scholar] [CrossRef]
- Monteiro, S.; Barakat, M.; Piçarra-Pereira, M.A.; Teixeira, A.R.; Ferreira, R.B. Osmotin and thaumatin from grape: A putative general defense mechanism against pathogenic fungi. Phytopathology 2003, 93, 1505–1512. [Google Scholar] [CrossRef] [Green Version]
- Brogue, K.; Chet, I.; Holliday, M.; Cressman, R.; Biddle, P.; Knowlton, S.; Mauvais, C.J.; Broglie, R. Transgenic plants with enhanced resistance to the fungal pathogen Rhizoctonia solani. Science 1991, 254, 1194–1197. [Google Scholar] [CrossRef] [PubMed]
- Okay, S.; ÖZCENGİZ, G. Molecular cloning, characterization, and homologous expression of an endochitinase gene from Bacillus thuringiensis serovar morrisoni. Turk. J. Biol. 2011, 35, 1–7. [Google Scholar]
- Tripathi, A.; Aggarwal, R.; Yadav, A. Differential expression analysis of defense-related genes responsive to Tilletia indica infection in wheat. Turk. J. Biol. 2013, 37, 606–613. [Google Scholar] [CrossRef]



| Gene | Accession Number | Forward Primer (5′ → 3′) | Reverse Primer (5′ → 3′) | Product Size (bp) |
|---|---|---|---|---|
| PR-1 | HM489878.1 | GCCAGCTACTACTCTCTCCG | AGGTATCCCATGCACGACTC | 175 |
| Thionins | AY253444.1 | AAGCACTTCTGGATTTCGCC | CATCCTGTTCATCGCTGCAG | 168 |
| TLP | KJ764822.1 | TTCCTCCTCCTGGCTGTTTT | ATATCCTCCCGGCTTTGGTG | 175 |
| Chitinase 2 | AB029935.1 | ACGGCGATATGGTTCTGGAT | TAGCGCTTGTAGAACCCGAT | 209 |
| β-1,3-glucanase | DQ090946.1 | CTACAGGTCCAAGGGCATCA | GCGGCGATGTACTTGATGTT | 210 |
| Defensin | KJ551546.1 | TGTCCAATAAGAACTGCGCG | TGGTTCCATGGGCTAGCTAG | 161 |
| Actin | GQ339780.1 | GAGAAGCTCGCATATGTGGC | TCCAGCAGCTTCCATACCA | 180 |
| Variety | Treatment | Mean Number of Spots per Leaf | Diseased Plants (%) |
|---|---|---|---|
| Shafaq-2006 | Control | 0 | 0 |
| Inoculated | 4 | 2 | |
| Sahar-2006 | Control | 0 | 0 |
| Inoculated | 16.60 | 18.80 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manghwar, H.; Hussain, A.; Ali, Q.; Saleem, M.H.; Abualreesh, M.H.; Alatawi, A.; Ali, S.; Munis, M.F.H. Disease Severity, Resistance Analysis, and Expression Profiling of Pathogenesis-Related Protein Genes after the Inoculation of Fusarium equiseti in Wheat. Agronomy 2021, 11, 2124. https://doi.org/10.3390/agronomy11112124
Manghwar H, Hussain A, Ali Q, Saleem MH, Abualreesh MH, Alatawi A, Ali S, Munis MFH. Disease Severity, Resistance Analysis, and Expression Profiling of Pathogenesis-Related Protein Genes after the Inoculation of Fusarium equiseti in Wheat. Agronomy. 2021; 11(11):2124. https://doi.org/10.3390/agronomy11112124
Chicago/Turabian StyleManghwar, Hakim, Amjad Hussain, Qurban Ali, Muhammad Hamzah Saleem, Muyassar H. Abualreesh, Aishah Alatawi, Shafaqat Ali, and Muhammad Farooq Hussain Munis. 2021. "Disease Severity, Resistance Analysis, and Expression Profiling of Pathogenesis-Related Protein Genes after the Inoculation of Fusarium equiseti in Wheat" Agronomy 11, no. 11: 2124. https://doi.org/10.3390/agronomy11112124
APA StyleManghwar, H., Hussain, A., Ali, Q., Saleem, M. H., Abualreesh, M. H., Alatawi, A., Ali, S., & Munis, M. F. H. (2021). Disease Severity, Resistance Analysis, and Expression Profiling of Pathogenesis-Related Protein Genes after the Inoculation of Fusarium equiseti in Wheat. Agronomy, 11(11), 2124. https://doi.org/10.3390/agronomy11112124











