Effects of Root Temperature on the Plant Growth and Food Quality of Chinese Broccoli (Brassica oleracea var. alboglabra Bailey)
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials and Growth Conditions
2.2. Root Temperature Setup
2.3. Harvest and Sample Preparation
2.4. Elemental Analysis
2.5. Soluble Sugars, Total Chlorophyll and Starch Quantification
2.6. Glucosinolates Analysis
2.7. Statistical Analysis
3. Results
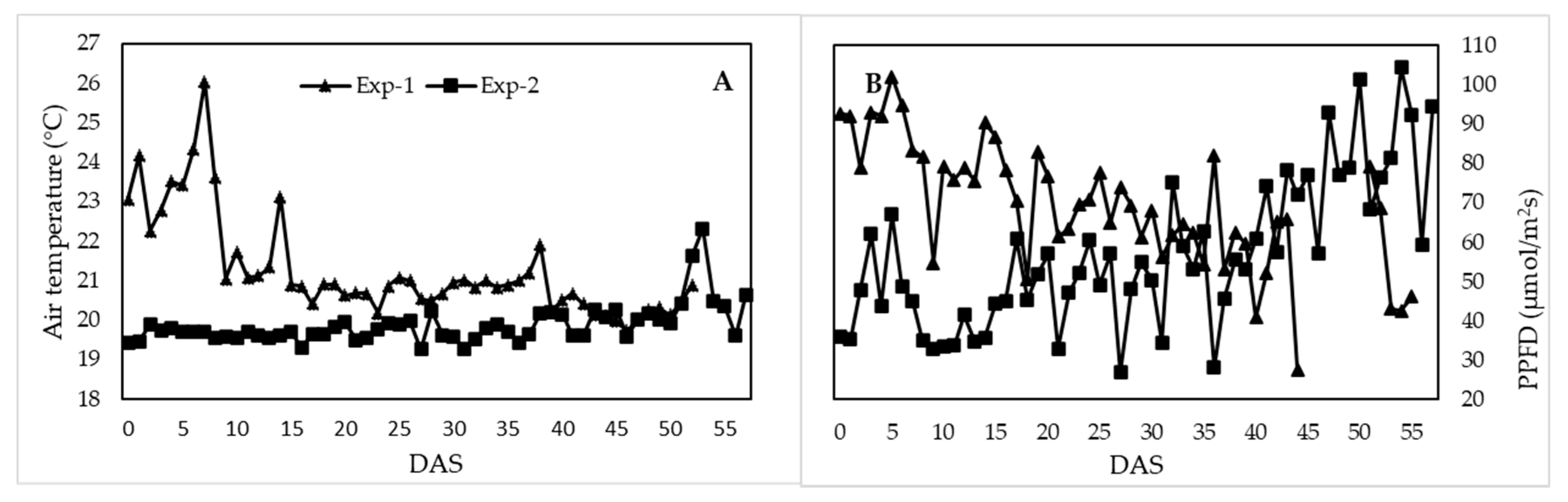
3.1. Greenhouse Climate Conditions
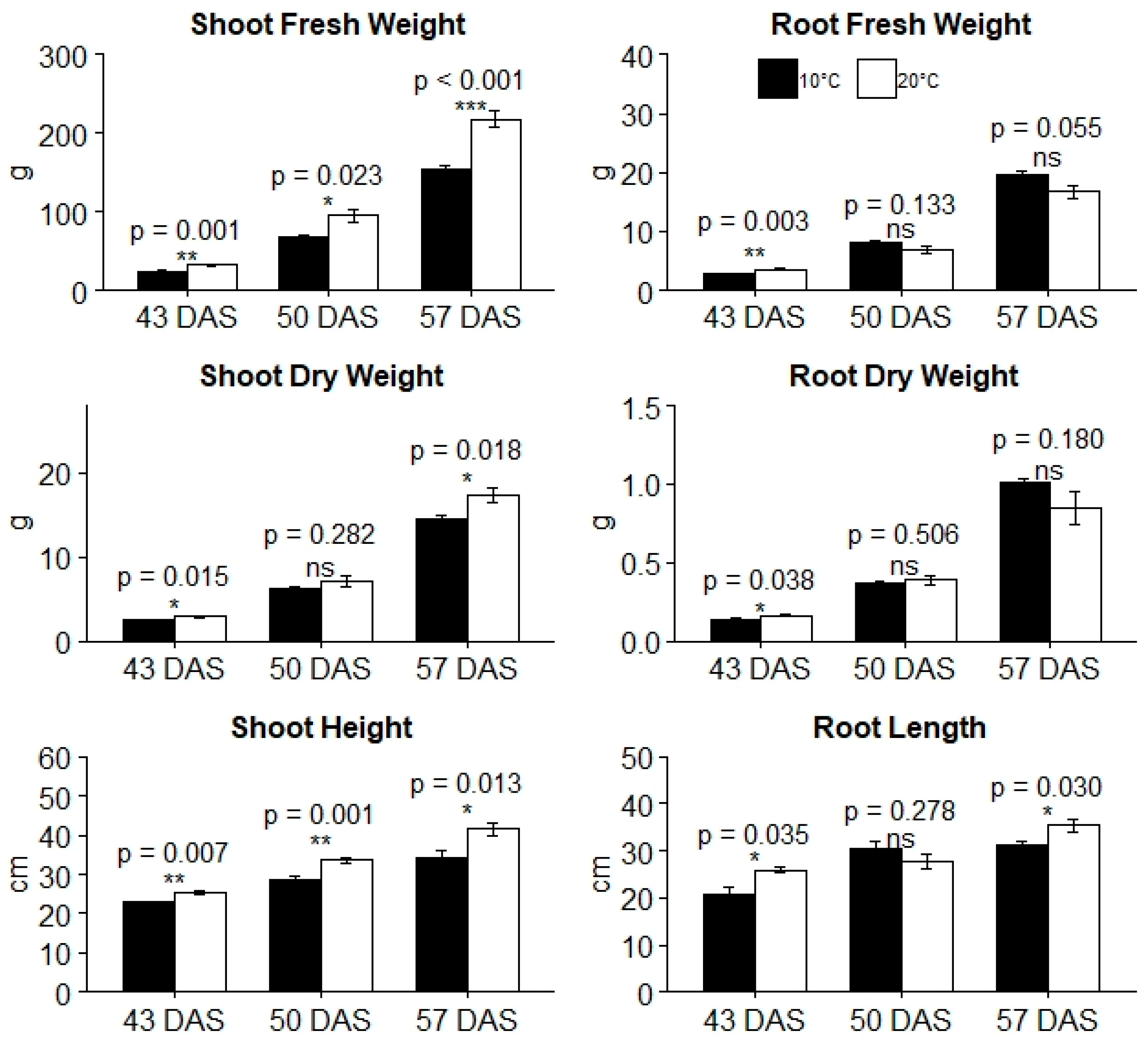
3.2. Effects of Root Temperature on Plant Growth
3.3. Effects of Root Temperature on Elemental Composition
3.4. Effects of Root Temperature on Soluble Sugars, Total Chlorophyll and Starch
3.5. Effects of Root Temperature on Glucosinolates
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chapin, F.S. Phosphate absorption capacity and acclimation potential in plants along a latitudinal gradient. Science 1974, 183, 521–523. [Google Scholar] [CrossRef]
- Sun, J.; Lu, N.; Xu, H.; Maruo, T.; Guo, S. Root zone cooling and exogenous spermidine root-pretreatment promoting Lactuca sativa L. growth and photosynthesis in the high-temperature season. Front. Plant Sci. 2016, 7, 368. [Google Scholar] [CrossRef]
- Setter, T.L.; Greenway, H. Growth reductions of rice at low root temperature: Decreases in nutrient uptake and development of chlorosis. J. Exp. Bot. 1988, 39, 811–829. [Google Scholar] [CrossRef]
- Díaz-Pérez, J.C. Root zone temperature, plant growth and yield of broccoli [Brassica oleracea (Plenck) var. italica] as affected by plastic film mulches. Sci. Hortic. 2009, 123, 156–163. [Google Scholar] [CrossRef]
- Adebooye, O.C.; Schmitz-Eiberger, M.; Lankes, C.; Noga, G.J. Inhibitory effects of sub-optimal root zone temperature on leaf bioactive components, photosystem II (PS II) and mineral uptake in Trichosanthes cucumerina L. Cucurbitaceae. Acta Physiol. Plant. 2010, 32, 67–73. [Google Scholar] [CrossRef]
- Aidoo, M.K.; Sherman, T.; Lazarovitch, N.; Fait, A.; Rachmilevitch, S. Physiology and metabolism of grafted bell pepper in response to low root-zone temperature. Funct. Plant. Biol. 2019, 46, 339–349. [Google Scholar] [CrossRef]
- Yan, Q.Y.; Duan, Z.Q.; Mao, J.D.; Xun, L.I.; Fei, D.O.N.G. Low root zone temperature limits nutrient effects on cucumber seedling growth and induces adversity physiological response. J. Integr. Agric. 2013, 12, 1450–1460. [Google Scholar] [CrossRef]
- Sakamoto, M.; Suzuki, T. Effect of root-zone temperature on growth and quality of hydroponically grown red leaf lettuce (Lactuca sativa L. cv. Red Wave). Am. J. Plant. Sci. 2015, 6, 2350. [Google Scholar] [CrossRef]
- Sakamoto, M.; Suzuki, T. Elevated root-zone temperature modulates growth and quality of hydroponically grown carrots. Agric. Sci. 2015, 6, 749. [Google Scholar] [CrossRef]
- Sakamoto, M.; Uenishi, M.; Miyamoto, K.; Suzuki, T. Effect of root-zone temperature on the growth and fruit quality of hydroponically grown strawberry plants. J. Agric. Sci. 2016, 8, 122–131. [Google Scholar] [CrossRef]
- Rosa, E.; David, M.; Gomes, M.H. Glucose, fructose and sucrose content in broccoli, white cabbage and Portuguese cabbage grown in early and late seasons. J. Sci. Food Agric. 2001, 81, 1145–1149. [Google Scholar] [CrossRef]
- Chen, X.; Wu, J.; Zhou, S.; Yang, Y.; Ni, X.; Yang, J.; Zhu, Z.; Shi, C. Application of near-infrared reflectance spectroscopy to evaluate the lutein and β-carotene in Chinese kale. J. Food Compos. Anal. 2009, 22, 148–153. [Google Scholar] [CrossRef]
- Qian, H.; Liu, T.; Deng, M.; Miao, H.; Cai, C.; Shen, W.; Wang, Q. Effects of light quality on main health-promoting compounds and antioxidant capacity of Chinese kale sprouts. Food Chem. 2016, 196, 1232–1238. [Google Scholar] [CrossRef] [PubMed]
- Deng, M.; Qian, H.; Chen, L.; Sun, B.; Chang, J.; Miao, H.; Wang, Q. Influence of pre-harvest red light irradiation on main phytochemicals and antioxidant activity of Chinese kale sprouts. Food Chem. 2017, 222, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Sun, B.; Yan, H.; Liu, N.; Wei, J.; Wang, Q. Effect of 1-MCP treatment on postharvest quality characters, antioxidants and glucosinolates of Chinese kale. Food Chem. 2012, 131, 519–526. [Google Scholar] [CrossRef]
- Fuentes, F.; Paredes-Gonzalez, X.; Kong, A.-N.T. Dietary glucosinolates sulforaphane, phenethyl isothiocyanate, indole-3-carbinol/3,3′-diindolylmethane: Antioxidative stress/inflammation, Nrf2, epigenetics/epigenomics and in vivo cancer chemopreventive efficacy. Curr. Pharm. 2015, 1, 179–196. [Google Scholar] [CrossRef]
- Wieczorek, M.N.; Walczak, M.; Skrzypczak-Zielińska, M.; Jeleń, H.H. Bitter taste of Brassica vegetables: The role of genetic factors, receptors, isothiocyanates, glucosinolates, and flavor context. Crit. Rev. Food Sci. Nutr. 2018, 58, 3130–3140. [Google Scholar] [CrossRef]
- Kawashima, L.M.; Valente Soares, L.M. Mineral profile of raw and cooked leafy vegetables consumed in Southern Brazil. J. Food Compos. Anal. 2003, 16, 605–611. [Google Scholar] [CrossRef]
- Steindal, A.L.H.; Rødven, R.; Hansen, E.; Mølmann, J. Effects of photoperiod, growth temperature and cold acclimatisation on glucosinolates, sugars and fatty acids in kale. Food Chem. 2015, 174, 44–51. [Google Scholar] [CrossRef]
- Justen, V.L.; Fritz, V.A. Temperature-induced glucosinolate accumulation is associated with expression of BrMYB transcription factors. HortScience 2013, 48, 47–52. [Google Scholar] [CrossRef]
- Cumbus, I.P.; Nye, P.H. Root zone temperature effects on growth and nitrate absorption in rape (Brassica napus cv. Emerald). J. Exp. Bot. 1982, 33, 1138–1146. [Google Scholar] [CrossRef]
- Cumbus, I.P.; Nye, P.H. Root zone temperature effects on growth and phosphate absorption in rape Brassica napus cv. Emerald. J. Exp. Bot. 1985, 36, 219–227. [Google Scholar] [CrossRef]
- Macduff, J.H.; Hopper, M.J.; Wild, A. The effect of root temperature on growth and uptake of ammonium and nitrate by Brassica napus L. in flowing solution culture. J. Exp. Bot. 1987, 38, 42–52. [Google Scholar] [CrossRef]
- Kleier, C.; Farnsworth, B.; Winner, W. Biomass, reproductive output, and physiological responses of rapid-cycling Brassica (Brassica rapa) to ozone and modified root temperature. New Phytol. 1998, 139, 657–664. [Google Scholar] [CrossRef]
- Sun, B.; Liu, N.; Zhao, Y.; Yan, H.; Wang, Q. Variation of glucosinolates in three edible parts of Chinese kale (Brassica alboglabra Bailey) varieties. Food Chem. 2011, 124, 941–947. [Google Scholar] [CrossRef]
- He, F.; Thiele, B.; Watt, M.; Kraska, T.; Ulbrich, A.; Kuhn, A.J. Effects of root cooling on plant growth and fruit quality of cocktail tomato during two consecutive seasons. J. Food Qual. 2019. [Google Scholar] [CrossRef]
- Viola, R.; Davies, H.V. A microplate reader assay for rapid enzymatic quantification of sugars in potato tubers. Potato Res. 1992, 35, 55–58. [Google Scholar] [CrossRef]
- Volden, J.; Bengtsson, G.B.; Wicklund, T. Glucosinolates, l-ascorbic acid, total phenols, anthocyanins, antioxidant capacities and colour in cauliflower (Brassica oleracea L. ssp. botrytis); effects of long-term freezer storage. Food Chem. 2009, 112, 967–976. [Google Scholar] [CrossRef]
- Doheny-Adams, T.; Redeker, K.; Kittipol, V.; Bancroft, I.; Hartley, S.E. Development of an efficient glucosinolate extraction method. Plant Methods 2017, 13, 17. [Google Scholar] [CrossRef]
- Cao, H. Practical Cultivation Technique of Chinese Kale, Choy Sum, Wild Cabbage and Leaf Mustard; China Agricultural Press: Beijing, China, 2004; pp. 1–11. [Google Scholar]
- Yang, X.; Yang, Y.Y. The effects of temperature on the growth and development and flower stalk formation of Chinese kale (Brassica alboglabra Bailey). China Veg. 2002, 4, 33–34. (In Chinese) [Google Scholar] [CrossRef]
- De Cnodder, T.; Vissenberg, K.; Van Der Straeten, D.; Verbelen, J.-P. Regulation of cell length in the Arabidopsis thaliana root by the ethylene precursor 1-aminocyclopropane-1-carboxylic acid: A matter of apoplastic reactions. New Phytol. 2005, 168, 541–550. [Google Scholar] [CrossRef]
- He, J.; Tan, L.P.; Lee, S.K. Root-zone temperature effects on photosynthesis, 14C-photoassimilate partitioning and growth of temperate lettuce (Lactuca sativa cv. “Panama”) in the tropics. Photosynthetica 2009, 47, 95–103. [Google Scholar] [CrossRef]
- Zhang, Y.P.; Qiao, Y.X.; Zhang, Y.L.; Zhou, Y.H.; Yu, J.Q. Effects of root temperature on leaf gas exchange and xylem sap abscisic acid concentrations in six Cucurbitaceae species. Photosynthetica 2008, 46, 356. [Google Scholar] [CrossRef]
- Bumgarner, N.R.; Scheerens, J.C.; Mullen, R.W.; Bennett, M.A.; Ling, P.P.; Kleinhenz, M.D. Root-zone temperature and nitrogen affect the yield and secondary metabolite concentration of fall-and spring-grown, high-density leaf lettuce. J. Sci. Food Agric. 2012, 92, 116–124. [Google Scholar] [CrossRef]
- Dalla Costa, L.; Tomasi, N.; Gottardi, S.; Iacuzzo, F.; Cortella, G.; Manzocco, L.; Pinton, R.; Mimmo, T.; Cesco, S. The effect of growth medium temperature on corn salad [Valerianella locusta (L.) Laterr] baby leaf yield and quality. HortScience 2011, 46, 1619–1625. [Google Scholar] [CrossRef]
- Aroca, R.; Tognoni, F.; Irigoyen, J.J.; Sánchez-Díaz, M.; Pardossi, A. Different root low temperature response of two maize genotypes differing in chilling sensitivity. Plant. Physiol. Biochem. 2001, 39, 1067–1073. [Google Scholar] [CrossRef]
- Schwarz, D.; Rouphael, Y.; Colla, G.; Venema, J.H. Grafting as a tool to improve tolerance of vegetables to abiotic stresses: Thermal stress, water stress and organic pollutants. Sci. Hortic. 2010, 127, 162–171. [Google Scholar] [CrossRef]
- Aloni, B.; Cohen, R.; Karni, L.; Aktas, H.; Edelstein, M. Hormonal signaling in rootstock–scion interactions. Sci. Hortic. 2010, 127, 119–126. [Google Scholar] [CrossRef]
- Paul, M.J.; Foyer, C.H. Sink regulation of photosynthesis. J. Exp. Bot. 2001, 52, 1383–1400. [Google Scholar] [CrossRef] [PubMed]
- Equiza, M. Morphological, anatomical and physiological responses related to differential shoot vs. root growth inhibition at low temperature in spring and winter wheat. Ann. Bot. 2001, 87, 67–76. [Google Scholar] [CrossRef]
- Starck, Z.; Niemyska, B.; Bogdan, J.; Tawalbeh, R.N.A. Response of tomato plants to chilling stress in association with nutrient or phosphorus starvation. Plant Soil 2000, 226, 99–106. [Google Scholar] [CrossRef]
- Pettersson, S. Low root zone temperature effects on net mineral nutrient uptake and distribution in barley (Hordeum vulgare). J. Plant Physiol. 1995, 145, 459–464. [Google Scholar] [CrossRef]
- Olle, M.; Bender, I. Causes and control of calcium deficiency disorders in vegetables: A review. J. Hortic. Sci. Biotechnol. 2009, 84, 577–584. [Google Scholar] [CrossRef]
- Baghour, M.; Moreno, D.A.; Víllora, G.; López-Cantarero, I.; Hernández, J.; Castilla, N.; Romero, L. Root-zone temperature influences the distribution of Cu and Zn in potato-plant organs. J. Agric. Food Chem. 2002, 50, 140–146. [Google Scholar] [CrossRef] [PubMed]
- Chadirin, Y.; Hidaka, K.; Takahashi, T.; Sago, Y.; Wajima, T.; Kitano, M. Application of temperature stress to roots of spinach I. Effect of the low temperature stress on quality. Environ. Control Biol. 2011, 49, 133–139. [Google Scholar] [CrossRef][Green Version]
- Sami, F.; Yusuf, M.; Faizan, M.; Faraz, A.; Hayat, S. Role of sugars under abiotic stress. Plant. Physiol. Biochem. 2016, 109, 54–61. [Google Scholar] [CrossRef]
- León-Chan, R.G.; López-Meyer, M.; Osuna-Enciso, T.; Sañudo-Barajas, J.A.; Heredia, J.B.; León-Félix, J. Low temperature and ultraviolet-B radiation affect chlorophyll content and induce the accumulation of UV-B-absorbing and antioxidant compounds in bell pepper (Capsicum annuum) plants. Environ. Exp. Bot. 2017, 139, 143–151. [Google Scholar] [CrossRef]
- Guy, C.; Kaplan, F.; Kopka, J.; Selbig, J.; Hincha, D.K. Metabolomics of temperature stress. Physiol. Plant. 2008, 132, 220–235. [Google Scholar] [CrossRef]
- Ensminger, I.; Busch, F.; Huner, N.P.A. Photostasis and cold acclimation: Sensing low temperature through photosynthesis. Physiol. Plant. 2006, 126, 28–44. [Google Scholar] [CrossRef]
- Gazula, A.; Kleinhenz, M.D.; Streeter, J.G.; Miller, A.R. Temperature and cultivar effects on anthocyanin and chlorophyll b concentrations in three related Lollo Rosso lettuce cultivars. HortScience 2005, 40, 1731–1733. [Google Scholar] [CrossRef]
- Kalisz, A.; Jezdinský, A.; Pokluda, R.; Sękara, A.; Grabowska, A.; Gil, J. Impacts of chilling on photosynthesis and chlorophyll pigment content in juvenile basil cultivars. Hortic. Environ. Biotechnol. 2016, 57, 330–339. [Google Scholar] [CrossRef]
- Nguyen, D.T.; Lu, N.; Kagawa, N.; Kitayama, M.; Takagaki, M. Short-term root-zone temperature treatment enhanced the accumulation of secondary metabolites of hydroponic coriander (Coriandrum sativum L.) grown in a plant factory. Agronomy 2020, 10, 413. [Google Scholar] [CrossRef]
- La, G.X.; Fang, P.; Li, Y.; Wang, Y. Determination of desulpho-glucosinolates in bolting stems of Chinese kale by liquid chromatography-mass spectrometry. J. Zhejiang Univ. (Agric. Life Sci.) 2008, 34, 557–563. (In Chinese) [Google Scholar] [CrossRef]
- La, G.X.; Fang, P.; Teng, Y.B.; Li, Y.J.; Lin, X.Y. Effect of CO2 enrichment on the glucosinolate contents under different nitrogen levels in bolting stem of Chinese kale (Brassica alboglabra L.). J. Zhejiang Univ. Sci. B 2009, 10, 454–464. [Google Scholar] [CrossRef] [PubMed]
- Del Carmen Martínez-Ballesta, M.; Moreno, D.A.; Carvajal, M. The physiological importance of glucosinolates on plant response to abiotic stress in Brassica. Int. J. Mol. Sci. 2013, 14, 11607–11625. [Google Scholar] [CrossRef] [PubMed]
- Soydam Aydin, S.; Büyük, I.; Aras, S. Relationships among lipid peroxidation, SOD enzyme activity, and SOD gene expression profile in Lycopersicum esculentum L. exposed to cold stress. Genet. Mol. Res. 2013, 12, 3220–3229. [Google Scholar] [CrossRef]
- Schulz, E.; Tohge, T.; Zuther, E.; Fernie, A.R.; Hincha, D.K. Flavonoids are determinants of freezing tolerance and cold acclimation in Arabidopsis Thaliana. Sci. Rep. 2016, 6, 34027. [Google Scholar] [CrossRef]
- Engelen-Eigles, G.; Holden, G.; Cohen, J.D.; Gardner, G. The effect of temperature, photoperiod, and light quality on gluconasturtiin concentration in Watercress (Nasturtium officinale R. Br.). J. Agric. Food Chem. 2006, 54, 328–334. [Google Scholar] [CrossRef]
- Kissen, R.; Eberl, F.; Winge, P.; Uleberg, E.; Martinussen, I.; Bones, A.M. Effect of growth temperature on glucosinolate profiles in Arabidopsis thaliana accessions. Phytochemistry 2016, 130, 106–118. [Google Scholar] [CrossRef]
- Charron, C.S.; Sams, C.E. Glucosinolate content and myrosinase activity in rapid-cycling Brassica oleracea grown in a controlled environment. J. Am. Soc. Hortic. Sci. 2004, 129, 321–330. [Google Scholar] [CrossRef]
- Guo, L.; Yang, R.; Wang, Z.; Guo, Q.; Gu, Z. Glucoraphanin, sulforaphane and myrosinase activity in germinating broccoli sprouts as affected by growth temperature and plant organs. J. Funct. Foods 2014, 9, 70–77. [Google Scholar] [CrossRef]
- Rosa, E.A.; Rodrigues, P.M. The effect of light and temperature on glucosinolate concentration in the leaves and roots of cabbage seedlings. J. Sci. Food Agric. 1998, 78, 208–212. [Google Scholar] [CrossRef]
- Cocetta, G.; Mishra, S.; Raffaelli, A.; Ferrante, A. Effect of heat root stress and high salinity on glucosinolate metabolism in wild rocket. J. Plant. Physiol. 2018, 231, 261–270. [Google Scholar] [CrossRef] [PubMed]
- Kask, K.; Kännaste, A.; Talts, E.; Copolovici, L.; Niinemets, Ü. How specialized volatiles respond to chronic and short-term physiological and shock heat stress in Brassica nigra. Plant Cell Environ. 2016, 39, 2027–2042. [Google Scholar] [CrossRef] [PubMed]
- Farnham, M.W.; Wilson, P.E.; Stephenson, K.K.; Fahey, J.W. Genetic and environmental effects on glucosinolate content and chemoprotective potency of broccoli. Plant. Breed. 2004, 123, 60–65. [Google Scholar] [CrossRef]
- Barrero-Gil, J.; Huertas, R.; Rambla, J.L.; Granell, A.; Salinas, J. Tomato plants increase their tolerance to low temperature in a chilling acclimation process entailing comprehensive transcriptional and metabolic adjustments. Plant Cell Environ. 2016, 39, 2303–2318. [Google Scholar] [CrossRef]
- Ntatsi, G.; Savvas, D.; Papasotiropoulos, V.; Katsileros, A.; Zrenner, R.M.; Hincha, D.K.; Zuther, E.; Schwarz, D. Rootstock sub-optimal temperature tolerance determines transcriptomic responses after long-term root cooling in rootstocks and scions of grafted tomato plants. Front. Plant Sci. 2017, 8, 911. [Google Scholar] [CrossRef]
- Yang, Y.-W.; Chen, H.-C.; Jen, W.-F.; Liu, L.-Y.; Chang, M.-C. Comparative transcriptome analysis of shoots and roots of TNG67 and TCN1 rice seedlings under cold stress and following subsequent recovery: Insights into metabolic pathways, phytohormones, and transcription factors. PLoS ONE 2015, 10, e0131391. [Google Scholar] [CrossRef]
- Ruelland, E.; Vaultier, M.-N.; Zachowski, A.; Hurry, V. Cold signalling and cold acclimation in plants. Adv. Bot. Res. 2009, 49, 35–150. [Google Scholar] [CrossRef]
| Experiments | Date | Treatment | ||
|---|---|---|---|---|
| Root Temperature | Harvest | Duration | ||
| Exp-1 | 20 Aug–11 Oct, 2017 | 15 vs. 20 °C | 11 October, 2017 | 24 Sep–11 Oct, 2017 |
| Exp-2 | 13 Feb–12 Apr, 2018 | 10 vs. 20 °C | 29 Mar 5 Apr 12 Apr, 2018 | 21 Mar–12 Apr, 2018 |
| Compound | Precursor Ion [m/z] | Product Ion [m/z] | Cone Voltage [V] | Collision Energy [V] | |
|---|---|---|---|---|---|
| Aliphatic | Sinigrin | 358.1 | 195 | 100 | 20 |
| Progoitrin | 388 | 195 | 100 | 20 | |
| Glucoraphanin | 436.1 | 372.1 | 100 | 22 | |
| Gluconapin | 372.1 | 359 | 100 | 20 | |
| Glucoiberin | 422.1 | 358.1 | 100 | 22 | |
| Glucoalyssin | 450.2 | 386.2 | 100 | 22 | |
| Indolic | Glucobrassicin | 447.1 | 259.1 | 100 | 22 |
| 4-Methoxyglucobrassicin | 477.1 | 195 | 100 | 23 | |
| Neoglucobrassicin | 477.1 | 446.1 | 100 | 13 | |
| 4-Hydroxyglucobrassicin | 463.1 | 267 | 100 | 18 |
| Exp-1 | Exp-2 | ||||||
|---|---|---|---|---|---|---|---|
| Treatment | 15 °C | 20 °C | p-Value | 10 °C | 20 °C | p-Value | |
| Shoot | Yield (g) | 153.5 ± 21.2 | 154.7 ± 22.2 | 0.878 | 137.2 ± 9.0 | 172.9 ± 32.2 | 0.041 |
| FW (g) | 170.4 ± 21.6 | 175.5 ± 21.6 | 0.511 | 153.4 ± 13.7 | 218.0 ± 24.7 | <0.001 | |
| DW (g) | 14.83 ± 2.16 | 15.38 ± 2.37 | 0.499 | 14.52 ± 0.99 | 17.40 ± 2.09 | 0.018 | |
| Height (cm) | 35.30 ± 5.84 | 35.77 ± 4.53 | 0.802 | 34.35 ± 3.82 | 41.55 ± 4.35 | 0.013 | |
| Shoot ratio (%) | 92.94 ± 0.61 | 94.37 ± 0.86 | <0.001 | 93.47 ± 0.34 | 95.37 ± 1.22 | 0.011 | |
| Root | FW (g) | 15.32 ± 2.37 | 11.28 ± 1.63 | <0.001 | 19.58 ± 1.23 | 16.67 ± 2.83 | 0.055 |
| DW (g) | 1.12 ± 0.16 | 0.91 ± 0.15 | <0.001 | 1.01 ± 0.06 | 0.85 ± 0.26 | 0.180 | |
| Length (cm) | 29.43 ± 5.07 | 26.73 ± 5.73 | 0.177 | 31.30 ± 2.12 | 35.52 ± 3.35 | 0.029 | |
| (% DW) | Exp-1 | Exp-2 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 53 DAS | 43 DAS | 50 DAS | 57 DAS | ||||||||||
| 15 °C | 20 °C | p-Value | 10 °C | 20 °C | p-Value | 10 °C | 20 °C | p-Value | 10 °C | 20 °C | p-Value | ||
| Leaves | C | 38.75 ± 0.58 | 38.63 ± 0.63 | 0.583 | 39.89 ± 0.47 | 36.55 ± 0.62 | <0.001 | 37.91 ± 0.89 | 34.86 ± 0.56 | <0.001 | 38.28 ± 0.84 | 36.81 ± 0.99 | 0.021 |
| N | 6.35 ± 0.15 | 6.39 ± 0.11 | 0.361 | 5.32 ± 0.09 | 6.34 ± 0.12 | <0.001 | 5.06 ± 0.21 | 6.19 ± 0.12 | <0.001 | 5.4 ± 0.41 | 5.93 ± 0.29 | 0.028 | |
| P | 0.63 ± 0.07 | 0.62 ± 0.04 | 0.887 | 0.48 ± 0.04 | 0.66 ± 0.06 | 0.001 | 0.52 ± 0.07 | 0.62 ± 0.05 | 0.032 | 0.69 ± 0.04 | 0.78 ± 0.06 | 0.011 | |
| K | 2.89 ± 0.29 | 2.85 ± 0.27 | 0.682 | 5.01 ± 0.45 | 6.14 ± 0.21 | 0.003 | 6.55 ± 1.15 | 6.18 ± 0.82 | 0.582 | 5.69 ± 0.84 | 5.31 ± 0.62 | 0.400 | |
| Ca | 3.62 ± 0.50 | 3.38 ± 0.42 | 0.159 | 4.07 ± 0.46 | 5.94 ± 0.50 | <0.001 | 4.41 ± 0.81 | 5.63 ± 0.74 | 0.038 | 4.35 ± 1.12 | 5.05 ± 0.95 | 0.270 | |
| Mg | 0.55 ± 0.06 | 0.46 ± 0.04 | <0.001 | 0.62 ± 0.07 | 0.91 ± 0.06 | <0.001 | 0.68 ± 0.12 | 0.79 ± 0.11 | 0.157 | 0.73 ± 0.15 | 0.74 ± 0.12 | 0.889 | |
| Stems | C | 38.54 ± 0.97 | 38.09 ± 1.13 | 0.235 | 37.45 ± 0.63 | 35.57 ± 0.58 | 0.001 | 39.7 ± 0.95 | 36.86 ± 0.87 | 0.001 | 38.96 ± 0.69 | 37.2 ± 0.88 | 0.004 |
| N | 4.53 ± 0.35 | 4.35 ± 0.18 | 0.094 | 2.94 ± 0.22 | 4.01 ± 0.18 | <0.001 | 3.88 ± 0.17 | 4.32 ± 0.23 | 0.010 | 3.69 ± 0.16 | 3.94 ± 0.14 | 0.016 | |
| P | 0.49 ± 0.04 | 0.51 ± 0.05 | 0.293 | 0.38 ± 0.06 | 0.53 ± 0.05 | 0.003 | 0.61 ± 0.03 | 0.65 ± 0.04 | 0.125 | 0.56 ± 0.02 | 0.57 ± 0.06 | 0.130 | |
| K | 5.52 ± 0.49 | 5.52 ± 0.59 | 0.992 | 7.45 ± 1.30 | 8.12 ± 0.77 | 0.360 | 7.73 ± 1.05 | 10.58 ± 1.08 | 0.003 | 7.4 ± 0.44 | 8.28 ± 1.23 | 0.146 | |
| Ca | 0.86 ± 0.17 | 0.84 ± 0.19 | 0.663 | 1.13 ± 0.26 | 1.12 ± 0.08 | 0.990 | 1.13 ± 0.12 | 1.69 ± 0.32 | 0.014 | 1.17 ± 0.17 | 1.48 ± 0.22 | 0.020 | |
| Mg | 0.29 ± 0.03 | 0.26 ± 0.03 | 0.048 | 0.39 ± 0.07 | 0.40 ± 0.04 | 0.773 | 0.45 ± 0.06 | 0.55 ± 0.04 | 0.014 | 0.44 ± 0.06 | 0.52 ± 0.09 | 0.122 | |
| Roots | C | 37.30 ± 0.90 | 37.18 ± 1.86 | 0.826 | 44.9 ± 0.29 | 45.15 ± 0.35 | 0.267 | 45.38 ± 0.37 | 43.5 ± 2.32 | 0.146 | 45.90 ± 0.32 | 45.51 ± 0.41 | 0.098 |
| N | 5.44 ± 0.22 | 4.97 ± 0.28 | <0.001 | 5.4 ± 0.44 | 4.46 ± 0.26 | 0.006 | 5.17 ± 0.15 | 3.82 ± 0.55 | 0.004 | 4.44 ± 0.37 | 3.82 ± 0.54 | 0.045 | |
| P | 0.95 ± 0.14 | 0.96 ± 0.11 | 0.69 | 0.69 ± 0.16 | 0.46 ± 0.07 | 0.029 | 0.45 ± 0.05 | 0.38 ± 0.11 | 0.220 | 0.45 ± 0.04 | 0.36 ± 0.05 | 0.009 | |
| K | 0.43 ± 0.05 | 0.37 ± 0.04 | <0.001 | 0.30 ± 0.07 | 0.19 ± 0.03 | 0.015 | 0.26 ± 0.15 | 0.34 ± 0.11 | 0.384 | 0.30 ± 0.14 | 0.25 ± 0.13 | 0.454 | |
| Ca | 0.63 ± 0.08 | 0.73 ± 0.11 | 0.003 | 0.78 ± 0.09 | 0.81 ± 0.07 | 0.636 | 0.64 ± 0.07 | 0.64 ± 0.11 | 0.957 | 0.69 ± 0.09 | 0.72 ± 0.13 | 0.624 | |
| Mg | 0.38 ± 0.05 | 0.26 ± 0.03 | <0.001 | 0.35 ± 0.05 | 0.22 ± 0.02 | 0.002 | 0.26 ± 0.05 | 0.15 ± 0.02 | 0.008 | 0.27 ± 0.03 | 0.18 ± 0.03 | <0.001 | |
| (mg/g FW) | Exp-1 | Exp-2 | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 53 DAS | 43 DAS | 50 DAS | 57 DAS | ||||||||||
| 15 °C | 20 °C | p-Value | 10 °C | 20 °C | p-Value | 10 °C | 20 °C | p-Value | 10 °C | 20 °C | p-Value | ||
| Leaves | Glucose | 1.18 ± 0.35 | 1.44 ± 0.45 | 0.083 | 1.87 ± 0.52 | 0.52 ± 0.12 | 0.004 | 2.83 ± 1.22 | 0.82 ± 0.08 | 0.021 | 3.50 ± 1.43 | 1.41 ± 0.46 | 0.014 |
| Fructose | 1.78 ± 0.65 | 2.10 ± 0.47 | 0.133 | 1.00 ± 0.52 | 0.27 ± 0.01 | 0.034 | 2.25 ± 1.20 | 0.48 ± 0.21 | 0.028 | 3.51 ± 1.18 | 1.43 ± 0.34 | 0.006 | |
| Sucrose | 1.38 ± 0.19 | 0.99 ± 0.20 | <0.001 | 2.20 ± 0.58 | 0.58 ± 0.17 | 0.002 | 0.71 ± 0.14 | 0.62 ± 0.30 | 0.570 | 0.91 ± 0.38 | 1.00 ± 0.21 | 0.626 | |
| Chlorophyll | 4.74 ± 1.10 | 3.97 ± 0.80 | 0.038 | 2.84 ± 0.13 | 2.30 ± 0.10 | <0.001 | 5.84 ± 0.30 | 4.43 ± 0.57 | 0.003 | 5.37 ± 0.97 | 5.41 ± 0.76 | 0.937 | |
| Starch | 2.55 ± 1.22 | 2.15 ± 1.07 | 0.341 | 2.10 ± 0.65 | 0.95 ± 0.56 | 0.017 | 1.31 ± 0.49 | 0.55 ± 0.24 | 0.011 | ||||
| Stems | Glucose | 7.81 ± 0.99 | 7.64 ± 1.00 | 0.649 | 8.27 ± 1.41 | 3.78 ± 0.53 | 0.001 | 7.43 ± 0.90 | 5.83 ± 1.25 | 0.052 | 9.37 ± 1.46 | 7.21 ± 0.77 | 0.013 |
| Fructose | 6.57 ± 0.82 | 6.49 ± 0.80 | 0.788 | 3.40 ± 1.45 | 1.04 ± 0.27 | 0.020 | 6.86 ± 0.72 | 4.62 ± 0.77 | 0.001 | 8.06 ± 0.88 | 6.29 ± 0.76 | 0.004 | |
| Sucrose | 1.91 ± 0.56 | 1.98 ± 0.53 | 0.697 | 7.50 ± 1.18 | 4.14 ± 0.80 | 0.001 | 3.77 ± 1.44 | 2.60 ± 0.59 | 0.150 | 3.41 ± 1.58 | 3.29 ± 1.32 | 0.886 | |
| Chlorophyll | 0.48 ± 0.19 | 0.19 ± 0.06 | <0.001 | 0.31 ± 0.11 | 0.23 ± 0.08 | 0.237 | 0.33 ± 0.10 | 0.28 ± 0.05 | 0.321 | 0.38 ± 0.09 | 0.29 ± 0.07 | 0.080 | |
| Starch | 0.23 ± 0.09 | 0.24 ± 0.05 | 0.607 | 0.35 ± 0.23 | 0.37 ± 0.21 | 0.915 | 0.29 ± 0.08 | 0.21 ± 0.05 | 0.069 | ||||
| (µmol/100g FW) | Exp-1 | Exp-2 | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| 53 DAS | 50 DAS | 57 DAS | |||||||||
| 15 °C | 20 °C | p-Value | 10 °C | 20 °C | p-Value | 10 °C | 20 °C | p-Value | |||
| Leaves | Aliphatic | Sinigrin | 14.66 ± 7.02 | 12.71 ± 7.15 | 0.506 | 15.35 ± 3.22 | 6.37 ± 1.91 | 0.002 | 18.11 ± 4.98 | 10.49 ± 3.65 | 0.014 |
| Progoitrin | 1.39 ± 0.55 | 1.88 ± 1.54 | 0.322 | 1.46 ± 0.18 | 0.57 ± 0.06 | <0.001 | 1.74 ± 0.69 | 0.98 ± 0.52 | 0.059 | ||
| Glucoraphanin | 2.21 ± 1.76 | 1.35 ± 0.94 | 0.152 | 1.18 ± 0.31 | 0.30 ± 0.15 | 0.001 | 3.93 ± 1.80 | 2.92 ± 1.54 | 0.322 | ||
| Gluconapin | 31.75 ± 16.74 | 34.76 ± 25.24 | 0.735 | 46.67 ± 10.77 | 19.07 ± 5.95 | 0.002 | 57.05 ± 20.01 | 41.17 ± 18.87 | 0.188 | ||
| Glucoiberin | 1.77 ± 0.86 | 0.82 ± 0.56 | 0.005 | 0.43 ± 0.12 | 0.18 ± 0.08 | 0.009 | 1.49 ± 0.62 | 1.32 ± 0.54 | 0.626 | ||
| Glucoalyssin | 0.05 ± 0.04 | 0.05 ± 0.03 | 0.710 | 0.10 ± 0.02 | 0.06 ± 0.01 | 0.012 | 0.19 ± 0.07 | 0.13 ± 0.07 | 0.135 | ||
| Total | 51.83 ± 26.60 | 51.57 ± 33.14 | 0.983 | 65.61 ± 14.51 | 26.72 ± 8.03 | 0.002 | 84.00 ± 26.66 | 58.33 ± 24.98 | 0.116 | ||
| Indolic | Glucobrassicin | 5.12 ± 2.80 | 4.66 ± 2.51 | 0.678 | 16.36 ± 7.97 | 5.79 ± 1.62 | 0.040 | 10.41 ± 3.33 | 6.96 ± 2.40 | 0.069 | |
| 4-Methoxyglucobrassicin | 0.53 ± 0.11 | 0.50 ± 0.14 | 0.557 | 0.52 ± 0.14 | 0.34 ± 0.03 | 0.036 | 0.58 ± 0.15 | 0.49 ± 0.07 | 0.201 | ||
| Neoglucobrassicin | 0.21 ± 0.14 | 0.09 ± 0.09 | 0.024 | 0.79 ± 0.31 | 0.27 ± 0.12 | 0.017 | 0.42 ± 0.18 | 0.23 ± 0.05 | 0.044 | ||
| 4-Hydroxyglucobrassicin | 1.15 ± 0.26 | 1.08 ± 0.27 | 0.502 | 0.97 ± 0.27 | 0.50 ± 0.07 | 0.017 | 1.49 ± 0.44 | 0.77 ± 0.23 | 0.008 | ||
| Total | 7.01 ± 3.07 | 6.33 ± 2.69 | 0.570 | 18.64 ± 8.50 | 6.90 ± 1.70 | 0.035 | 12.90 ± 3.98 | 8.44 ± 2.65 | 0.049 | ||
| Total | 58.84 ± 29.33 | 57.89 ± 35.68 | 0.944 | 84.25 ± 22.02 | 33.63 ± 9.44 | 0.004 | 96.90 ± 30.10 | 66.78 ± 27.55 | 0.101 | ||
| Stems | Aliphatic | Sinigrin | 14.28 ± 3.29 | 12.17 ± 4.98 | 0.236 | 27.00 ± 4.82 | 22.14 ± 6.28 | 0.210 | 14.81 ± 6.67 | 15.41 ± 4.26 | 0.856 |
| Progoitrin | 2.88 ± 0.99 | 2.68 ± 0.78 | 0.591 | 5.70 ± 1.21 | 3.59 ± 1.02 | 0.018 | 2.92 ± 1.66 | 2.55 ± 0.87 | 0.645 | ||
| Glucoraphanin | 6.35 ± 2.17 | 5.37 ± 1.24 | 0.191 | 6.97 ± 1.51 | 6.15 ± 2.42 | 0.543 | 6.46 ± 2.31 | 8.56 ± 2.73 | 0.181 | ||
| Gluconapin | 33.75 ± 10.92 | 29.56 ± 7.61 | 0.289 | 71.72 ± 15.94 | 56.72 ± 22.02 | 0.256 | 31.96 ± 18.86 | 38.34 ± 13.58 | 0.518 | ||
| Glucoiberin | 2.55 ± 1.00 | 1.64 ± 0.84 | 0.025 | 2.30 ± 0.71 | 2.06 ± 0.84 | 0.640 | 1.80 ± 1.11 | 2.28 ± 0.67 | 0.388 | ||
| Glucoalyssin | 0.07 ± 0.03 | 0.07 ± 0.02 | 0.618 | 0.24 ± 0.04 | 0.23 ± 0.12 | 0.875 | 0.21 ± 0.06 | 0.23 ± 0.07 | 0.633 | ||
| Total | 59.89 ± 17.14 | 51.49 ± 12.08 | 0.181 | 113.92 ± 23.31 | 90.89 ± 31.26 | 0.232 | 58.16 ± 30.26 | 67.38 ± 20.93 | 0.546 | ||
| Indolic | Glucobrassicin | 2.87 ± 1.12 | 2.33 ± 1.08 | 0.238 | 9.27 ± 2.97 | 8.62 ± 4.14 | 0.782 | 2.83 ± 1.81 | 4.00 ± 1.60 | 0.263 | |
| 4-Methoxyglucobrassicin | 1.41 ± 0.21 | 1.51 ± 0.24 | 0.341 | 2.45 ± 0.47 | 2.31 ± 0.74 | 0.727 | 1.99 ± 0.37 | 2.34 ± 0.27 | 0.100 | ||
| Neoglucobrassicin | 0.54 ± 0.26 | 0.36 ± 0.14 | 0.051 | 4.21 ± 1.54 | 2.59 ± 1.26 | 0.107 | 1.55 ± 1.04 | 1.47 ± 0.55 | 0.872 | ||
| 4-Hydroxyglucobrassicin | 2.51 ± 0.73 | 2.37 ± 0.43 | 0.577 | 1.94 ± 0.38 | 1.27 ± 0.42 | 0.031 | 2.45 ± 0.87 | 2.58 ± 0.39 | 0.753 | ||
| Total | 9.57 ± 2.50 | 8.89 ± 2.54 | 0.514 | 17.87 ± 5.00 | 14.79 ± 6.02 | 0.406 | 8.83 ± 3.93 | 10.39 ± 2.41 | 0.429 | ||
| Total | 60.46 ± 17.21 | 60.37 ± 13.24 | 0.162 | 131.78 ± 27.87 | 105.68 ± 36.95 | 0.247 | 66.99 ± 34.01 | 77.77 ± 23.15 | 0.533 | ||
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, F.; Thiele, B.; Santhiraraja-Abresch, S.; Watt, M.; Kraska, T.; Ulbrich, A.; Kuhn, A.J. Effects of Root Temperature on the Plant Growth and Food Quality of Chinese Broccoli (Brassica oleracea var. alboglabra Bailey). Agronomy 2020, 10, 702. https://doi.org/10.3390/agronomy10050702
He F, Thiele B, Santhiraraja-Abresch S, Watt M, Kraska T, Ulbrich A, Kuhn AJ. Effects of Root Temperature on the Plant Growth and Food Quality of Chinese Broccoli (Brassica oleracea var. alboglabra Bailey). Agronomy. 2020; 10(5):702. https://doi.org/10.3390/agronomy10050702
Chicago/Turabian StyleHe, Fang, Björn Thiele, Sharin Santhiraraja-Abresch, Michelle Watt, Thorsten Kraska, Andreas Ulbrich, and Arnd J. Kuhn. 2020. "Effects of Root Temperature on the Plant Growth and Food Quality of Chinese Broccoli (Brassica oleracea var. alboglabra Bailey)" Agronomy 10, no. 5: 702. https://doi.org/10.3390/agronomy10050702
APA StyleHe, F., Thiele, B., Santhiraraja-Abresch, S., Watt, M., Kraska, T., Ulbrich, A., & Kuhn, A. J. (2020). Effects of Root Temperature on the Plant Growth and Food Quality of Chinese Broccoli (Brassica oleracea var. alboglabra Bailey). Agronomy, 10(5), 702. https://doi.org/10.3390/agronomy10050702







