Legume Biofortification and the Role of Plant Growth-Promoting Bacteria in a Sustainable Agricultural Era
Abstract
1. Introduction
2. The Importance of Legumes in the Accomplishment of the Sustainable Development Goals
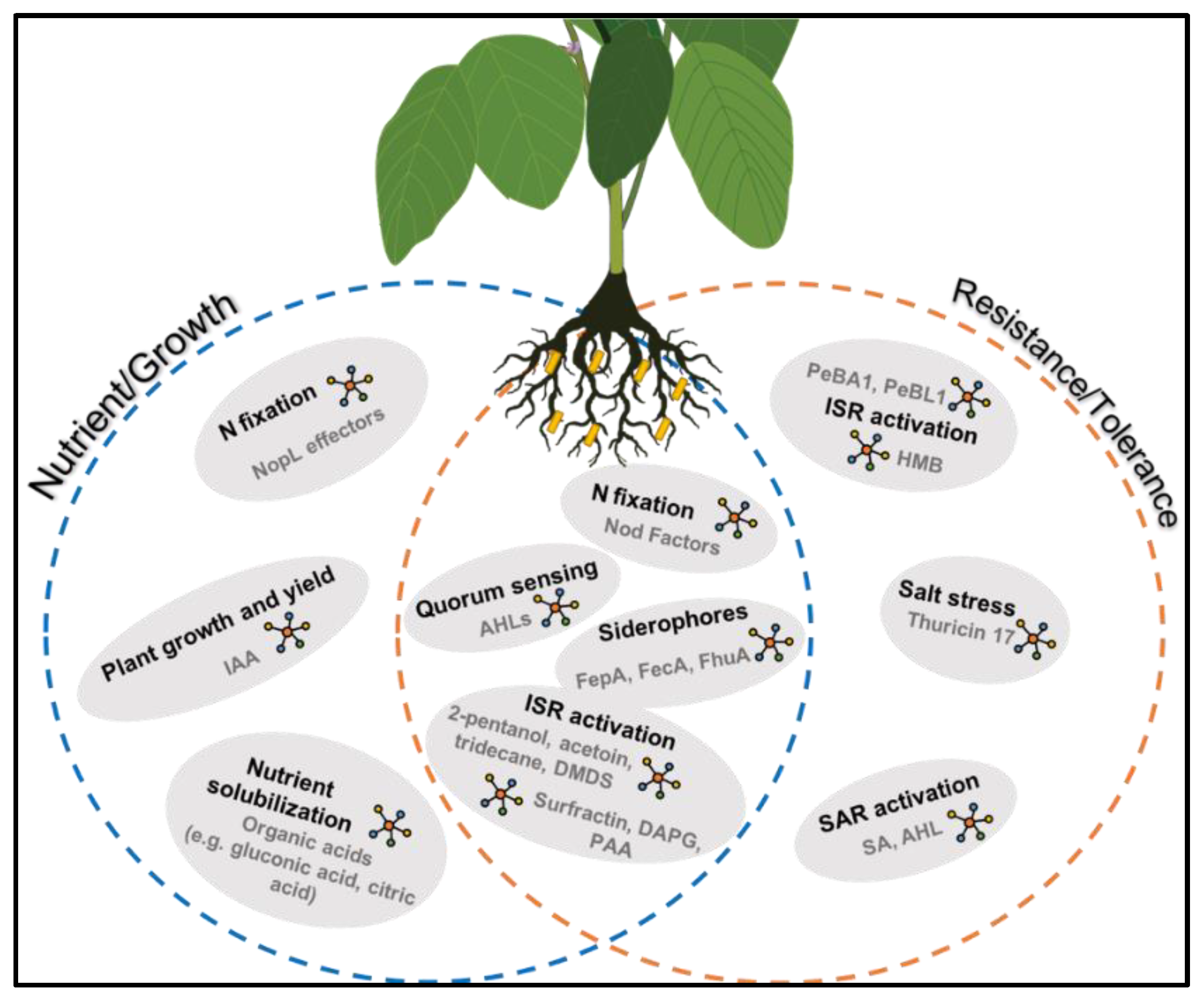
3. The Role of PGPB in Legume Nutrition
4. Iron Biofortification of Legumes with PGPB: A Case Study
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- UNDESA. World Population Prospects: The 2012 Revision, United Nations Department of Economic and Social Affairs, New York. 2013. Available online: https://population.un.org/wpp/Publications/Files/WPP2012_HIGHLIGHTS.pdf (accessed on 6 February 2019).
- Jewell, M.C.; Campbell, B.C.; Godwin, I.D. Transgenic Plants for Abiotic Stress Resistance. In Transgenic Crop Plants; Springer: Berlin/Heidelberg, Germany, 2010. [Google Scholar]
- Meena, V.S.; Meena, S.K.; Verma, J.P.; Kumar, A.; Aeron, A.; Mishra, P.K.; Bisht, J.K.; Pattanayak, A.; Naveed, M.; Dotaniya, M.L. Plant Beneficial Rhizospheric Microorganism (PBRM) Strategies to Improve Nutrients Use Efficiency: A Review. Ecol. Eng. 2017, 107, 8–32. [Google Scholar] [CrossRef]
- Godfray HC, J.; Beddington, J.R.; Crute, I.R.; Haddad, L.; Lawrence, D.; Muir, J.F.; Pretty, J.; Robinson, S.; Thomas, S.M.; Toulmin, C. Food Security: The Challenge of Feeding 9 Billion People. Science 2010, 327, 812–818. [Google Scholar] [CrossRef]
- Kumar, A.; Maurya, B.R.; Raghuwanshi, R.; Meena, V.S.; Tofazzal Islam, M. Co-Inoculation with Enterobacter and Rhizobacteria on Yield and Nutrient Uptake by Wheat (Triticum Aestivum L.) in the Alluvial Soil Under Indo-Gangetic Plain of India. J. Plant Growth Regul. 2017, 36, 608–617. [Google Scholar] [CrossRef]
- Kumar, A.; Meena, V.S.; Maurya, B.R.; Raghuwanshi, R.; Bisht, J.K.; Pattanayak, A. Book Review. Appl. Soil Ecol. 2017, 117–118, 221–222. [Google Scholar] [CrossRef]
- Nath, D.; Maurya, B.R.; Meena, V.S. Documentation of Five Potassium- and Phosphorus-Solubilizing Bacteria for Their K and P-Solubilization Ability from Various Minerals. Biocatal. Agric. Biotechnol. 2017, 10, 174–181. [Google Scholar] [CrossRef]
- Finn, S. Nutrition Insecurity and Malnutrition in Developed Countries. In Addressing Malnutrition to Improve Global Health; Science/AAAS: Washington, DC, USA, 2014. [Google Scholar]
- WHO. Biofortication of Staple Crops. Available online: http://www.who.int/elena/titles/biofortification/en/ (accessed on 23 September 2019).
- White, P.J.; Broadley, M.R. Biofortification of Crops with Seven Mineral Elements Often Lacking in Human Diets - Iron, Zinc, Copper, Calcium, Magnesium, Selenium and Iodine. New Phytol. 2009, 182, 49–84. [Google Scholar] [CrossRef]
- Carvalho, S.M.P.; Vasconcelos, M.W. Producing More with Less: Strategies and Novel Technologies for Plant-Based Food Biofortification. Food Res. Int. 2013, 54, 961–971. [Google Scholar] [CrossRef]
- Golden Rice. Available online: http://www.goldenrice.org/ (accessed on 23 September 2019).
- International Rice Research Institute. Available online: http://irri.org/golden-rice/faqs (accessed on 23 September 2019).
- Garg, M.; Sharma, N.; Sharma, S.; Kapoor, P.; Kumar, A.; Chunduri, V.; Arora, P. Biofortified Crops Generated by Breeding, Agronomy, and Transgenic Approaches Are Improving Lives of Millions of People around the World. Front. Nutr. 2018, 5, 12. [Google Scholar] [CrossRef]
- Bouis, H.; Saltzman, A.; Birol, E. Improving Nutrition through Biofortification. In Agriculture for improved nutrition: Seizing the momentum; CAB International: Wallingford, UK, 2019. [Google Scholar]
- Fuentes-Ramirez, L.E.; Caballero-Mellado, J. Bacterial Biofertilizers. In PGPR: Biocontrol and Biofertilization; Springer: Berlin/Heidelberg, Germany, 2005. [Google Scholar]
- Ku, Y.S.; Rehman, H.M.; Lam, H.-M. Possible Roles of Rhizospheric and Endophytic Microbes to Provide a Safe and Affordable Means of Crop Biofortification. Agronomy 2019, 9, 764. [Google Scholar] [CrossRef]
- Glick, B. Bacteria with ACC Deaminase Can Promote Plant Growth and Help to Feed the World. Microbiol. Res. 2014, 169, 30–39. [Google Scholar] [CrossRef]
- Pereira, S.I.A.; Castro, P.M.L. Phosphate-Solubilizing Rhizobacteria Enhance Zea Mays Growth in Agricultural P-Deficient Soils. Ecol. Eng. 2014, 73, 526–535. [Google Scholar] [CrossRef]
- Bahadur, I.; Maurya, B.R.; Kumar, A.; Meena, V.S.; Raghuwanshi, R. Towards the Soil Sustainability and Potassium-Solubilizing Microorganisms. In Potassium Solubilizing Microorganisms for Sustainable Agriculture; Springer India: New Delhi, India, 2016. [Google Scholar]
- Etesami, H.; Beattie, G.A. Plant-Microbe Interactions in Adaptation of Agricultural Crops to Abiotic Stress Conditions. In Probiotics and Plant Health; Springer Singapore: Singapore, 2017. [Google Scholar]
- İpek, M.; Eşitken, A. The Actions of PGPR on Micronutrient Availability in Soil and Plant Under Calcareous Soil Conditions: An Evaluation over Fe Nutrition. In Plant-Microbe Interactions in Agro-Ecological Perspectives; Springer Singapore: Singapore, 2017. [Google Scholar]
- Karnwal, A. Isolation and Identification of Plant Growth Promoting Rhizobacteria from Maize (Zea Mays L.) Rhizosphere and Their Plant Growth Promoting Effect on Rice (Oryza Sativa L.). J. Plant Prot. Res. 2017, 57, 144–151. [Google Scholar] [CrossRef]
- Moustaine, M.; Elkahkahi, R.; Benbouazza, A.; Benkirane, R.; EH, A. Effect of Plant Growth Promoting Rhizobacterial (PGPR) Inoculation on Growth in Tomato (Solanum Lycopersicum L.) and Characterization for Direct PGP Abilities in Morocco. Int. J. Environ. Agric. Biotechnol. 2017, 2, 590–596. [Google Scholar]
- Sharifi, P. The Effect of Plant Growth Promoting Rhizobacteria (PGPR), Salicylic Acid and Drought Stress on Growth Indices, the Chlorophyll and Essential Oil of Hyssop (Hyssopus Officinalis). Biosci. Biotechnol. Res. Asia 2017, 14, 1033–1042. [Google Scholar] [CrossRef]
- Kumari, M.; Gopal, A.; Lakshmipathy, R. Effect of Stress Tolerant Plant Growth Promoting Rhizobacteria on Growth of Blackgram under Stress Condition. Int. J. Curr. Microbiol. Appl. Sci. 2018, 7, 1479–1487. [Google Scholar] [CrossRef]
- Soares, J.C.; Santos, C.S.; Carvalho SM, P.; Pintado, M.M.; Vasconcelos, M.W. Preserving the Nutritional Quality of Crop Plants under a Changing Climate: Importance and Strategies. Plant Soil 2019, 443, 1–26. [Google Scholar] [CrossRef]
- Campbell, B.M.; Thornton, P.; Zougmoré, R.; van Asten, P.; Lipper, L. Sustainable Intensification: What Is Its Role in Climate Smart Agriculture? Curr. Opin. Environ. Sustain. 2014, 8, 39–43. [Google Scholar] [CrossRef]
- Khan, A.; Singh, J.; Upadhayay, V.K.; Singh, A.V.; Shah, S. Microbial Biofortification: A Green Technology Through Plant Growth Promoting Microorganisms. In Sustainable Green Technologies for Environmental Management; Springer Singapore: Singapore, 2019. [Google Scholar]
- United Nations. Sustainable Development Knowledge Platform. Available online: http://sustainabledevelopment.un.org/about (accessed on 21 September 2019).
- Daly, D.H.; Velivelli, S.L.S.; Prestwich, B.D. The Role of Soil Microbes in Crop Biofortification. In Agriculturally Important Microbes for Sustainable Agriculture; Springer Singapore: Singapore, 2017. [Google Scholar]
- Rehman, H.M.; Cooper, J.W.; Lam, H.-M.; Yang, S.H. Legume Biofortification Is an Underexploited Strategy for Combatting Hidden Hunger. Plant. Cell Environ. 2019, 42, 52–70. [Google Scholar] [CrossRef]
- Jha, A.B.; Warkentin, T.D. Biofortification of Pulse Crops: Status and Future Perspectives. Plants 2020, 9, 73. [Google Scholar] [CrossRef]
- Myers, S.S.; Zanobetti, A.; Kloog, I.; Huybers, P.; Leakey, A.D.B.; Bloom, A.J.; Carlisle, E.; Dietterich, L.H.; Fitzgerald, G.; Hasegawa, T.; et al. Increasing CO2 Threatens Human Nutrition. Nature 2014, 510, 139–142. [Google Scholar] [CrossRef]
- Martín-Cabrejas, M. Legumes: Nutritional Quality, Processing and Potential Health Benefits. In Legumes and Their Associated Health Benefits; Royal Society of Chemistry: London, UK, 2019. [Google Scholar]
- Ferreira, H.; Vasconcelos, M.; Gil, A.M.; Pinto, E. Benefits of Pulse Consumption on Metabolism and Health: A Systematic Review of Randomized Controlled Trials. Crit. Rev. Food Sci. Nutr. 2020, 1–12. [Google Scholar] [CrossRef]
- Karkanis, A.; Ntatsi, G.; Lepse, L.; Fernández, J.A.; Vågen, I.M.; Rewald, B.; Alsiņa, I.; Kronberga, A.; Balliu, A.; Olle, M.; et al. Faba Bean Cultivation—Revealing Novel Managing Practices for More Sustainable and Competitive European Cropping Systems. Front. Plant Sci. 2018, 9, 1115. [Google Scholar] [CrossRef]
- Atienza, S.G.; Rubiales, D. Legumes in Sustainable Agriculture. Crop Pasture Sci. 2017, 68, i–ii. [Google Scholar] [CrossRef]
- Ma, Y.; Schwenke, G.; Sun, L.; Liu, D.L.; Wang, B.; Yang, B. Modeling the Impact of Crop Rotation with Legume on Nitrous Oxide Emissions from Rain-Fed Agricultural Systems in Australia under Alternative Future Climate Scenarios. Sci. Total Environ. 2018, 630, 1544–1552. [Google Scholar] [CrossRef]
- Vaz Patto, M.C.; Amarowicz, R.; Aryee AN, A.; Boye, J.I.; Chung, H.-J.; Martín-Cabrejas, M.A.; Domoney, C. Achievements and Challenges in Improving the Nutritional Quality of Food Legumes. CRC. Crit. Rev. Plant Sci. 2015, 34, 105–143. [Google Scholar] [CrossRef]
- Herman, E.M.; Schmidt, M.A. The Potential for Engineering Enhanced Functional-Feed Soybeans for Sustainable Aquaculture Feed. Front. Plant Sci. 2016, 7, 440. [Google Scholar] [CrossRef]
- Xu, J.G.; Tian, C.R.; Hu, Q.P.; Luo, J.Y.; Wang, X.D.; Tian, X.D. Dynamic Changes in Phenolic Compounds and Antioxidant Activity in Oats ( Avena Nuda L.) during Steeping and Germination. J. Agric. Food Chem. 2009, 57, 10392–10398. [Google Scholar] [CrossRef]
- Tajaddini, A.; Pourzand, A.; Sanaat, Z.; Pirouzpanah, S. Dietary Resistant Starch Contained Foods and Breast Cancer Risk: A Case-Control Study in Northwest of Iran. Asian Pac. J. Cancer Prev. 2015, 16, 4185–4192. [Google Scholar] [CrossRef]
- Zhu, B.; Sun, Y.; Qi, L.; Zhong, R.; Miao, X. Dietary Legume Consumption Reduces Risk of Colorectal Cancer: Evidence from a Meta-Analysis of Cohort Studies. Sci. Rep. 2015, 5, 8797. [Google Scholar] [CrossRef]
- Li, J.; Mao, Q.-Q. Legume Intake and Risk of Prostate Cancer: A Meta-Analysis of Prospective Cohort Studies. Oncotarget 2017, 8, 44776–44784. [Google Scholar] [CrossRef]
- Nderitu, A.M.; Dykes, L.; Awika, J.M.; Minnaar, A.; Duodu, K.G. Phenolic Composition and Inhibitory Effect against Oxidative DNA Damage of Cooked Cowpeas as Affected by Simulated in Vitro Gastrointestinal Digestion. Food Chem. 2013, 141, 1763–1771. [Google Scholar] [CrossRef] [PubMed]
- Papanikolaou, Y.; Fulgoni, V.L. Bean Consumption Is Associated with Greater Nutrient Intake, Reduced Systolic Blood Pressure, Lower Body Weight, and a Smaller Waist Circumference in Adults: Results from the National Health and Nutrition Examination Survey 1999-2002. J. Am. Coll. Nutr. 2008, 27, 569–576. [Google Scholar] [CrossRef] [PubMed]
- Ha, V.; Sievenpiper, J.L.; de Souza, R.J.; Jayalath, V.H.; Mirrahimi, A.; Agarwal, A.; Chiavaroli, L.; Mejia, S.B.; Sacks, F.M.; Di Buono, M.; et al. Effect of Dietary Pulse Intake on Established Therapeutic Lipid Targets for Cardiovascular Risk Reduction: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Can. Med. Assoc. J. 2014, 186, E252–E262. [Google Scholar] [CrossRef]
- Rebello, C.J.; Greenway, F.L.; Finley, J.W. A Review of the Nutritional Value of Legumes and Their Effects on Obesity and Its Related Co-Morbidities. Obes. Rev. 2014, 15, 392–407. [Google Scholar] [CrossRef] [PubMed]
- Miller, V.; Mente, A.; Dehghan, M.; Rangarajan, S.; Zhang, X.; Swaminathan, S.; Dagenais, G.; Gupta, R.; Mohan, V.; Lear, S.; et al. Fruit, Vegetable, and Legume Intake, and Cardiovascular Disease and Deaths in 18 Countries (PURE): A Prospective Cohort Study. Lancet 2017, 390, 2037–2049. [Google Scholar] [CrossRef]
- Lopes, L.; Martins, M.; Farias, L.; Brito, A.; Lima, G.; Carvalho, V.; Pereira, C.; Conde Júnior, A.; Saldanha, T.; Arêas, J.; et al. Cholesterol-Lowering and Liver-Protective Effects of Cooked and Germinated Mung Beans (Vigna Radiata L.). Nutrients 2018, 10, 821. [Google Scholar] [CrossRef]
- Yanai, H.; Tada, N. Effects of Intake of Soy and Non-Soy Legume on Serum HDL-Cholesterol Levels. J. Endocrinol. Metab. 2018, 8, 83–86. [Google Scholar] [CrossRef]
- Singhal, P.; Kaushik, G.; Mathur, P. Antidiabetic Potential of Commonly Consumed Legumes: A Review. Crit. Rev. Food Sci. Nutr. 2014, 54, 655–672. [Google Scholar] [CrossRef]
- Dhillon, P.K.; Bowen, L.; Kinra, S.; Bharathi, A.V.; Agrawal, S.; Prabhakaran, D.; Reddy, K.S.; Ebrahim, S. Legume Consumption and Its Association with Fasting Glucose, Insulin Resistance and Type 2 Diabetes in the Indian Migration Study. Public Health Nutr. 2016, 19, 3017–3026. [Google Scholar] [CrossRef]
- Becerra-Tomás, N.; Díaz-López, A.; Rosique-Esteban, N.; Ros, E.; Buil-Cosiales, P.; Corella, D.; Estruch, R.; Fitó, M.; Serra-Majem, L.; Arós, F.; et al. Legume Consumption Is Inversely Associated with Type 2 Diabetes Incidence in Adults: A Prospective Assessment from the PREDIMED Study. Clin. Nutr. 2018, 37, 906–913. [Google Scholar] [CrossRef]
- Gebrelibanos, M.; Tesfaye, D.; Raghavendra, Y.; Sintayeyu, B. Nutritional and Health Implications of Legumes. Int. J. Pharm. Sci. Res. 2013, 4, 1269–1279. [Google Scholar]
- Opie, R.S.; Itsiopoulos, C.; Parletta, N.; Sanchez-Villegas, A.; Akbaraly, T.N.; Ruusunen, A.; Jacka, F.N. Dietary Recommendations for the Prevention of Depression. Nutr. Neurosci. 2017, 20, 161–171. [Google Scholar] [CrossRef] [PubMed]
- Zhong, X.; Ge, J.; Chen, S.; Xiong, Y.; Ma, S.; Chen, Q. Association between Dietary Isoflavones in Soy and Legumes and Endometrial Cancer: A Systematic Review and Meta-Analysis. J. Acad. Nutr. Diet. 2018, 118, 637–651. [Google Scholar] [CrossRef]
- Gómez de Cedrón, M.; Ramírez de Molina, A.; Reglero, G. Legumes and Cancer. Available online: https://www.glnc.org.au/legumes-2/legumes-and-health/legumes-and-cancer/ (accessed on 9 December 2019).
- Ghosh, A.; Hasim Reja, M.; Nalia, A.; Kanthal, S.; Maji, S.; Venugopalan, V.K.; Nath, R. Micronutrient Biofortification in Pulses: An Agricultural Approach. Curr. J. Appl. Sci. Technol. 2019, 35, 1–12. [Google Scholar] [CrossRef]
- Foyer, C.H.; Nguyen, H.; Lam, H.-M. Legumes-The Art and Science of Environmentally Sustainable Agriculture. Plant. Cell Environ. 2019, 42, 1–5. [Google Scholar] [CrossRef]
- Zamioudis, C.; Pieterse, C.M.J. Modulation of Host Immunity by Beneficial Microbes. Mol. Plant Microbe Interact. 2012, 25, 139–150. [Google Scholar] [CrossRef]
- Sattiraju, K.S.; Kotiyal, S.; Arora, A.; Maheshwari, M. Plant Growth-Promoting Microbes: Contribution to Stress Management in Plant Hosts. In Environmental Biotechnology: For Sustainable Future; Springer Singapore: Singapore, 2019. [Google Scholar]
- Glick, B.R. Plant Growth-Promoting Bacteria: Mechanisms and Applications. Scientifica 2012, 2012, 1–15. [Google Scholar] [CrossRef]
- Islam, M.R.; Sultana, T.; Joe, M.M.; Yim, W.; Cho, J.-C.; Sa, T. Nitrogen-Fixing Bacteria with Multiple Plant Growth-Promoting Activities Enhance Growth of Tomato and Red Pepper. J. Basic Microbiol. 2013, 53, 1004–1015. [Google Scholar] [CrossRef]
- Rodríguez, H.; Fraga, R.; Gonzalez, T.; Bashan, Y. Genetics of Phosphate Solubilization and Its Potential Applications for Improving Plant Growth-Promoting Bacteria. In First International Meeting on Microbial Phosphate Solubilization; Springer Netherlands: Dordrecht, The Netherlands, 2007. [Google Scholar]
- Delvasto, P.; Ballester, A.; Muñoz, J.A.; González, F.; Blázquez, M.L.; Igual, J.M.; Valverde, A.; García-Balboa, C. Mobilization of Phosphorus from Iron Ore by the Bacterium Burkholderia Caribensis FeGL03. Miner. Eng. 2009, 22, 1–9. [Google Scholar] [CrossRef]
- Glick, B.R.; Karaturovíc, D.M.; Newell, P.C. A Novel Procedure for Rapid Isolation of Plant Growth Promoting Pseudomonads. Can. J. Microbiol. 1995, 41, 533–536. [Google Scholar] [CrossRef]
- Vacheron, J.; Desbrosses, G.; Bouffaud, M.-L.; Touraine, B.; Moënne-Loccoz, Y.; Muller, D.; Legendre, L.; Wisniewski-Dyé, F.; Prigent-Combaret, C. Plant Growth-Promoting Rhizobacteria and Root System Functioning. Front. Plant Sci. 2013, 4, 356. [Google Scholar] [CrossRef]
- Maheshwari, D.K.; Dheeman, S.; Agarwal, M. Phytohormone-Producing PGPR for Sustainable Agriculture. In Bacterial Metabolites in Sustainable Development and Biodiversity; Springer: Cham, Switzerland, 2015. [Google Scholar]
- Sayyed, R.; Badgujar, M.; Sonawane, H.; Mhaske, M.; Chincholkar, S. Production of Microbial Iron Chelators (Siderophores) by Fluorescent Pseudomonads. Indian J. Biotechnol. 2005, 4, 484–490. [Google Scholar]
- Ahemad, M.; Kibret, M. Mechanisms and Applications of Plant Growth Promoting Rhizobacteria: Current Perspective. J. King Saud Univ. Sci. 2014, 26, 1–20. [Google Scholar] [CrossRef]
- Beneduzi, A.; Ambrosini, A.; Passaglia, L. Plant Growth-Promoting Rhizobacteria (PGPR): Their Potential as Antagonists and Biocontrol Agents. Genet. Mol. Biol. 2012, 35, 1044–1051. [Google Scholar] [CrossRef] [PubMed]
- Elshahat, M.R.; Ahmed, A.A.; Enas, A.H.; Fekria, M.S. Plant Growth Promoting Rhizobacteria and Their Potential for Biocontrol of Phytopathogens. Afr. J. Microbiol. Res. 2016, 10, 486–504. [Google Scholar] [CrossRef]
- Premachandra, D.; Hudek, L.; Brau, L. Bacterial Modes of Action for Enhancing of Plant Growth. J. Biotechnol. Biomater. 2016, 6, 236. [Google Scholar]
- Rosier, A.; Medeiros, F.H.V.; Bais, H.P. Defining Plant Growth Promoting Rhizobacteria Molecular and Biochemical Networks in Beneficial Plant-Microbe Interactions. Plant Soil 2018, 428, 35–55. [Google Scholar] [CrossRef]
- Benidire, L.; Pereira, S.I.A.; Castro, P.M.L.; Boularbah, A. Assessment of Plant Growth Promoting Bacterial Populations in the Rhizosphere of Metallophytes from the Kettara Mine, Marrakech. Environ. Sci. Pollut. Res. 2016, 23, 21751–21765. [Google Scholar] [CrossRef]
- Moreira, H.; Pereira, S.I.A.; Marques, A.P.G.C.; Rangel, A.O.S.S.; Castro, P.M.L. Mine Land Valorization through Energy Maize Production Enhanced by the Application of Plant Growth-Promoting Rhizobacteria and Arbuscular Mycorrhizal Fungi. Environ. Sci. Pollut. Res. 2016, 23, 6940–6950. [Google Scholar] [CrossRef]
- Verma, M.; Mishra, J.; Arora, N. Plant Growth-Promoting Rhizobacteria: Diversity and Applications. In Environmental Biotechnology: For Sustainable Future; Springer Singapore: Singapore, 2019. [Google Scholar]
- Malusá, E.; Vassilev, N. A Contribution to Set a Legal Framework for Biofertilisers. Appl. Microbiol. Biotechnol. 2014, 98, 6599–6607. [Google Scholar] [CrossRef]
- Rajkumar, M.; Ae, N.; Prasad, M.N.V.; Freitas, H. Potential of Siderophore-Producing Bacteria for Improving Heavy Metal Phytoextraction. Trends Biotechnol. 2010, 28, 142–149. [Google Scholar] [CrossRef]
- Egamberdieva, D.; Berg, G.; Lindström, K.; Räsänen, L.A. Co-Inoculation of Pseudomonas Spp. with Rhizobium Improves Growth and Symbiotic Performance of Fodder Galega (Galega Orientalis Lam.). Eur. J. Soil Biol. 2010, 46, 269–272. [Google Scholar] [CrossRef]
- Mishra, P.K.; Bisht, S.C.; Ruwari, P.; Joshi, G.K.; Singh, G.; Bisht, J.K.; Bhatt, J.C. Bioassociative Effect of Cold Tolerant Pseudomonas Spp. and Rhizobium Leguminosarum-PR1 on Iron Acquisition, Nutrient Uptake and Growth of Lentil (Lens Culinaris L.). Eur. J. Soil Biol. 2011, 47, 35–43. [Google Scholar] [CrossRef]
- Mishra, P.K.; Bisht, S.C.; Mishra, S.; Selvakumar, G.; Bisht, J.K.; Gupta, H.S. Coinoculation of Rhizobium Leguminosarum-PR1 with a Cold Tolerant Pseudomonas Sp. Improves Iron Acquisition, Nutrient Uptake and Growth of Field Pea (Pisum Sativum L.). J. Plant Nutr. 2012, 35, 243–256. [Google Scholar] [CrossRef]
- Stajkovic, O.; Delic, D.; Josic, D.; Kuzmanovic, D.; Rasulic, N.; Knezevic-Vukcevic, J. Improvement of Common Bean Growth by Co-Inoculation with Rhizobium and Plant Growth-Promoting Bacteria. Rom. Biotechnol. Lett. 2011, 16, 5919–5926. [Google Scholar]
- Ramesh, A.; Sharma, S.K.; Sharma, M.P.; Yadav, N.; Joshi, O.P. Inoculation of Zinc Solubilizing Bacillus Aryabhattai Strains for Improved Growth, Mobilization and Biofortification of Zinc in Soybean and Wheat Cultivated in Vertisols of Central India. Appl. Soil Ecol. 2014, 73, 87–96. [Google Scholar] [CrossRef]
- Gopalakrishnan, S.; Vadlamudi, S.; Samineni, S.; Sameer Kumar, C.V. Plant Growth-Promotion and Biofortification of Chickpea and Pigeonpea through Inoculation of Biocontrol Potential Bacteria, Isolated from Organic Soils. Springerplus 2016, 5, 1882. [Google Scholar] [CrossRef] [PubMed]
- Patel, P.; Trivedi, G.; Saraf, M. Iron Biofortification in Mungbean Using Siderophore Producing Plant Growth Promoting Bacteria. Environ. Sustain. 2018, 1, 357–365. [Google Scholar] [CrossRef]
- Htwe, A.Z.; Moh, S.M.; Moe, K.; Yamakawa, T. Effects of Co-Inoculation of Bradyrhizobium Japonicum SAY3-7 and Streptomyces Griseoflavus P4 on Plant Growth, Nodulation, Nitrogen Fixation, Nutrient Uptake, and Yield of Soybean in a Field Condition. Soil Sci. Plant Nutr. 2018, 64, 222–229. [Google Scholar] [CrossRef]
- Ullah, A.; Farooq, M.; Hussain, M. Improving the Productivity, Profitability and Grain Quality of Kabuli Chickpea with Co-Application of Zinc and Endophyte Bacteria Enterobacter Sp. MN17. Arch. Agron. Soil Sci. 2019, 1–16. [Google Scholar] [CrossRef]
- Ahmad, M.; Adil, Z.; Hussain, A.; Mumtaz, M.; Nafees, M.; Ahmad, I.; Jamil, M. Potential of Phosphate Solubilizing Bacillus Strains for Improving Growth and Nutrient Uptake in Mungbean and Maize Crops. Pak. J. Agric. Sci. 2019, 56, 283–289. [Google Scholar]
- Dogra, N.; Yadav, R.; Kaur, M.; Adhikary, A.; Kumar, S.; Ramakrishna, W. Nutrient Enhancement of Chickpea Grown with Plant Growth Promoting Bacteria in Local Soil of Bathinda, Northwestern India. Physiol. Mol. Biol. Plants 2019, 25, 1251–1259. [Google Scholar] [CrossRef] [PubMed]
- du Jardin, P. Plant Biostimulants: Definition, Concept, Main Categories and Regulation. Sci. Hortic. 2015, 196, 3–14. [Google Scholar] [CrossRef]
- Vejan, P.; Abdullah, R.; Khadiran, T.; Ismail, S.; Nasrulhaq Boyce, A. Role of Plant Growth Promoting Rhizobacteria in Agricultural Sustainability—A Review. Molecules 2016, 21, 573. [Google Scholar] [CrossRef] [PubMed]
- BCC Research. Biopesticides: The Global Market. Available online: https://www.mordorintelligence.com/industry-reports/global-biopesticides-market-industry (accessed on 6 February 2019).
- Arora, N.K.; Verma, M.; Mishra, J. Rhizobial Bioformulations: Past, Present and Future. In Rhizotrophs: Plant Growth Promotion to Bioremediation; Springer Singapore: Singapore, 2017. [Google Scholar]
- Mishra, P.; Das, D. Rejuvenation of Biofertilizer for Sustainable Agriculture and Economic Development. Cons. J Sustain. Dev. 2014, 11, 41–61. [Google Scholar]
- García-Fraile, P.; Menéndez, E.; Rivas, R. Role of Bacterial Biofertilizers in Agriculture and Forestry. AIMS Bioeng. 2015, 2, 183–205. [Google Scholar] [CrossRef]
- Thomas, L.; Singh, I. Microbial Biofertilizers: Types and Applications. In Biofertilizers for Sustainable Agriculture and Environment; Springer: Cham, Switzerland, 2019. [Google Scholar]
- Bhardwaj, D.; Ansari, M.; Sahoo, R.; Tuteja, N. Biofertilizers Function as Key Player in Sustainable Agriculture by Improving Soil Fertility, Plant Tolerance and Crop Productivity. Microb. Cell Fact. 2014, 13, 66. [Google Scholar] [CrossRef]
- WHO. Micronutrient Deficiencies: Iron Deficiency Anaemia. Available online: https://www.nhs.uk/conditions/iron-deficiency-anaemia/ (accessed on 24 September 2019).
- Marschner, P. Marschner’s Mineral Nutrition of Higher Plants; Elsevier: Amsterdam, The Netherlands, 2012. [Google Scholar]
- del Campillo, M.C.; Torrent, J. Predicting the Incidence of Iron Chlorosis in Calcareous Soils of Southern Spain. Commun. Soil Sci. Plant Anal. 1992, 23, 399–416. [Google Scholar] [CrossRef]
- Miethke, M.; Marahiel, M.A. Siderophore-Based Iron Acquisition and Pathogen Control. Microbiol. Mol. Biol. Rev. 2007, 71, 413–451. [Google Scholar] [CrossRef]
- Li, M.; Ahammed, G.J.; Li, C.; Bao, X.; Yu, J.; Huang, C.; Yin, H.; Zhou, J. Brassinosteroid Ameliorates Zinc Oxide Nanoparticles-Induced Oxidative Stress by Improving Antioxidant Potential and Redox Homeostasis in Tomato Seedling. Front. Plant Sci. 2016, 7, 615. [Google Scholar] [CrossRef]
- Gendre, D.; Czernic, P.; Conéjéro, G.; Pianelli, K.; Briat, J.-F.; Lebrun, M.; Mari, S. TcYSL3, a Member of the YSL Gene Family from the Hyper-Accumulator Thlaspi Caerulescens, Encodes a Nicotianamine-Ni/Fe Transporter. Plant J. 2006, 49, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Payne, S.M. Detection, Isolation, and Characterization of Siderophores. Methods Enzymol. 1994, 235, 329–344. [Google Scholar]
- Boukhalfa, H.; Crumbliss, A.L. Chemical Aspects of Siderophore Mediated Iron Transport. Biometals 2002, 15, 325–339. [Google Scholar] [CrossRef] [PubMed]
- Hider, R.C.; Kong, X. Chemistry and Biology of Siderophores. Nat. Prod. Rep. 2010, 27, 637. [Google Scholar] [CrossRef] [PubMed]
- Crowley, D.E.; Reid, C.P.P.; Szaniszlo, P.J. Utilization of Microbial Siderophores in Iron Acquisition by Oat. Plant Physiol. 1988, 87, 680–685. [Google Scholar] [CrossRef]
- Lucena, C.; Waters, B.M.; Romera, F.J.; Garcia, M.J.; Morales, M.; Alcantara, E.; Perez-Vicente, R. Ethylene Could Influence Ferric Reductase, Iron Transporter, and H+-ATPase Gene Expression by Affecting FER (or FER-like) Gene Activity. J. Exp. Bot. 2006, 57, 4145–4154. [Google Scholar] [CrossRef]
- Bacaicoa, E.; Mora, V.; Zamarreño, Á.M.; Fuentes, M.; Casanova, E.; García-Mina, J.M. Auxin: A Major Player in the Shoot-to-Root Regulation of Root Fe-Stress Physiological Responses to Fe Deficiency in Cucumber Plants. Plant Physiol. Biochem. 2011, 49, 545–556. [Google Scholar] [CrossRef]
- Tian, F.; Ding, Y.; Zhu, H.; Yao, L.; Du, B. Genetic Diversity of Siderophore-Producing Bacteria of Tobacco Rhizosphere. Braz. J. Microbiol. 2009, 40, 276–284. [Google Scholar] [CrossRef]
- Saharan, B.; Nehra, V. Plant Growth Promoting Rhizobacteria: A Critical Review. Life Sci. Med. Res. 2011, 2011, LSMR-21. [Google Scholar]
- Esitken, A. Use of Plant Growth Promoting Rhizobacteria in Horticultural Crops. In Bacteria in Agrobiology: Crop Ecosystems; Springer: Berlin/Heidelberg, Germany, 2011. [Google Scholar]
- Khalid, S.; Asghar, H.; Akhtar, M.; Aslam, A.; Zahir, Z. Biofortification of Iron in Chickpea by Plant Growth Promoting Rhizobacteria. Pak. J. Bot. 2015, 47, 1191–1194. [Google Scholar]
- Sathya, A.; Vijayabharathi, R.; Srinivas, V.; Gopalakrishnan, S. Plant Growth-Promoting Actinobacteria on Chickpea Seed Mineral Density: An Upcoming Complementary Tool for Sustainable Biofortification Strategy. 3 Biotech 2016, 6, 138. [Google Scholar] [CrossRef] [PubMed]
| Potential Beneficial Health Effects | References |
|---|---|
| Increased immunological, metabolic, and hormonal regulation | [40,41] |
| Anticarcinogenic (breast, colorectal, endometrium, and prostate cancers) and anti-inflammatory effects | [42,43,44,45,46] |
| Reduced risk of cardiovascular and obesity-related diseases, and metabolic syndrome | [47,48,49,50] |
| Reduced cholesterol levels | [51,52] |
| Reduced risk of type 2 diabetes mellitus | [53,54,55] |
| Reduced risk of osteoporosis and depression | [56,57,58,59] |
| Bacterial Genera | Crops | Contribution to Biofortification | References |
|---|---|---|---|
| Rhizobium galegae bv. orientalis HAMBI 540 + Pseudomonas trivialis 3Re27 | Fodder galega | Increase N content | [82] |
| Rhizobium leguminosarum-PR1 + Pseudomonas sp. NARs1/Pseudomonas sp. PGERs17 | Lentil, Pea | Increase Fe, N, and P uptake | [83,84] |
| Pseudomonas sp. LG + Rhizobium phaseoli strain 123 | Common bean | Increase P and N uptake | [85] |
| Bacillus aryabhattai MDSR7 and MDSR14, Enterobacter sp. MN17. | Chickpea, Soybean, Wheat | Increase Zn uptake | [86,90] |
| Acinetobacter tandoii SRI-305, Enterobacter ludwigii SRI-229 | Chickpea, Pigeonpea | Increase Fe, Zn, Cu, Mn, and Ca uptake | [87] |
| Bradyrhizobium japonicum SAY3-7 and Streptomyces griseoflavus P4 | Soybean | Increase N, P, K, Ca, and Mg uptake | [89] |
| Pantoea dispersa MPJ9, Pseudomonas putida MPJ6 | Mung bean | Increase Fe uptake | [88] |
| Bacillus aryabhattai S10 + B. subtilis ZM63 | Mung bean | Increase N, P, and K uptake | [91] |
| Symbion-K (Frauteria aurantia), Pseudomonas sp. RA6, P. citronellis (PC), Serratia sp. S2, Serratia marcescens CDP-13 | Chickpea | Increase macro- and micronutrient uptake | [92] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Roriz, M.; Carvalho, S.M.P.; Castro, P.M.L.; Vasconcelos, M.W. Legume Biofortification and the Role of Plant Growth-Promoting Bacteria in a Sustainable Agricultural Era. Agronomy 2020, 10, 435. https://doi.org/10.3390/agronomy10030435
Roriz M, Carvalho SMP, Castro PML, Vasconcelos MW. Legume Biofortification and the Role of Plant Growth-Promoting Bacteria in a Sustainable Agricultural Era. Agronomy. 2020; 10(3):435. https://doi.org/10.3390/agronomy10030435
Chicago/Turabian StyleRoriz, Mariana, Susana M. P. Carvalho, Paula M. L. Castro, and Marta W. Vasconcelos. 2020. "Legume Biofortification and the Role of Plant Growth-Promoting Bacteria in a Sustainable Agricultural Era" Agronomy 10, no. 3: 435. https://doi.org/10.3390/agronomy10030435
APA StyleRoriz, M., Carvalho, S. M. P., Castro, P. M. L., & Vasconcelos, M. W. (2020). Legume Biofortification and the Role of Plant Growth-Promoting Bacteria in a Sustainable Agricultural Era. Agronomy, 10(3), 435. https://doi.org/10.3390/agronomy10030435






