Biofumigation for Fighting Replant Disease- A Review
Abstract
1. The Replant Disease Syndrome
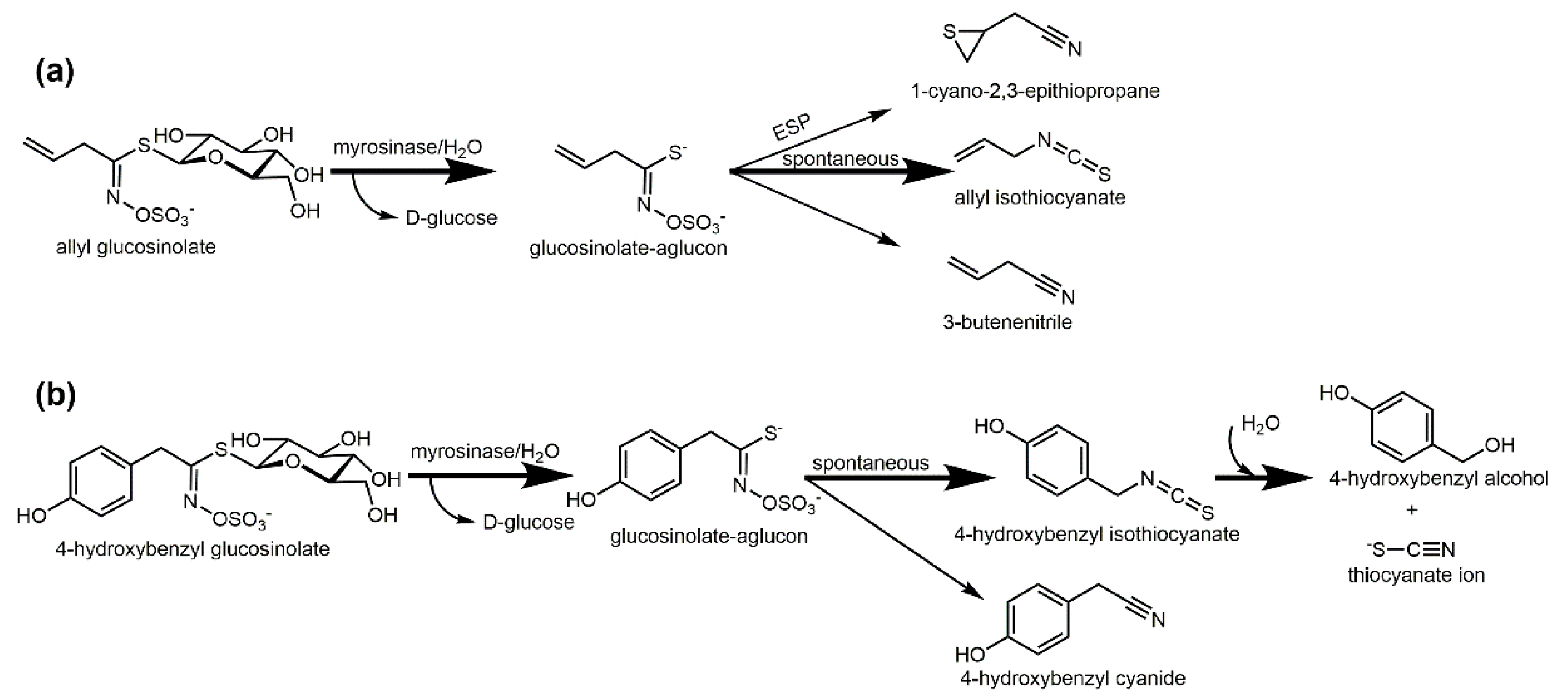
2. Biofumigation
3. Effects of Biofumigation on the Soil Biota
4. Efficacy of Biofumigation on Replant Disease
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Utkhede, R.S. Soil sickness, replant problem or replant disease and its integrated control. Allelopath. J. 2006, 18, 23–38. [Google Scholar]
- Winkelmann, T.; Smalla, K.; Amelung, W.; Baab, G.; Grunewaldt-Stöcker, G.; Kanfra, X.; Meyhöfer, R.; Reim, S.; Schmitz, M.; Vetterlein, D. Apple replant disease: Causes and mitigation strategies. Curr. Issues Mol. Biol. 2019, 30, 89–106. [Google Scholar] [CrossRef]
- Huang, L.-F.; Song, L.-X.; Xia, X.-J.; Mao, W.-H.; Shi, K.; Zhou, Y.-H.; Yu, J.-Q. Plant-soil feedbacks and soil sickness: From mechanisms to application in agriculture. J. Chem. Ecol. 2013, 39, 232–242. [Google Scholar] [CrossRef]
- Mazzola, M.; Manici, L.M. Apple replant disease: Role of microbial ecology in cause and control. Annu. Rev. Phytopathol. 2012, 50, 45–65. [Google Scholar] [CrossRef]
- Israel, D.W.; Giddens, J.E.; Powell, W.W. The toxicity of peach tree roots. Plant Soil 1973, 39, 103–112. [Google Scholar] [CrossRef]
- Benizri, E.; Piutti, S.; Verger, S.; Pagès, L.; Vercambre, G.; Poessel, J.L.; Michelot, P. Replant diseases: Bacterial community structure and diversity in peach rhizosphere as determined by metabolic and genetic fingerprinting. Soil Biol. Biochem. 2005, 37, 1738–1746. [Google Scholar] [CrossRef]
- Mai, W.F.; Abawi, G.S. Determining the cause and extent of apple, cherry and pear replant diseases under controlled conditions. Phytopathology 1978, 68, 1540–1544. [Google Scholar] [CrossRef]
- Reginato, G.; Córdova, C.; Mauro, C. Evaluation of rootstock and management practices to avoid cherry replant diesease in Chile. Acta Hortic. 2008, 795, 357–362. [Google Scholar] [CrossRef]
- Spethmann, W.; Otto, G. Replant problems and soil sickness. In Encyclopedia of Rose Science; Roberts, A.V., Debener, T., Gudin, S., Eds.; Elsevier: Oxford, UK, 2003; pp. 169–180. [Google Scholar]
- Baumann, A.; Yim, B.; Grunewaldt-Stöcker, G.; Liu, B.; Beerhues, L.; Sapp, M.; Nesme, J.; Sørensen, S.J.; Smalla, K.; Winkelmann, T. Rose replant disease: Detailed analyses of plant reactions, root endophytes and rhizosphere microbial communities. Acta Hortic. 2020, in press. [Google Scholar]
- Deal, D.; Mail, W.; Boothroyd, C. A survey of biotic relationships in grape replant situations. Phytopathology 1972, 62, 503–507. [Google Scholar] [CrossRef]
- Westphal, A.; Browne, G.T.; Schneider, S. Evidence for biological nature of the grape replant problem in California. Plant Soil 2002, 242, 197–203. [Google Scholar] [CrossRef]
- Elmer, W. Asparagus decline and replant problem: A look back and a look forward at strategies for mitigating losses. Acta Hortic. 2018, 1223, 195–204. [Google Scholar] [CrossRef]
- Wu, L.; Wang, J.; Huang, W.; Wu, H.; Chen, J.; Yang, Y.; Zhang, Z.; Lin, W. Plant-microbe rhizosphere interactions mediated by Rehmannia glutinosa root exudates under consecutive monoculture. Sci. Rep. 2015, 5, 15871. [Google Scholar] [CrossRef]
- Long, F.; Lin, Y.M.; Hong, T.; Wu, C.Z.; Li, J. Soil sickness in horticulture and forestry: A review. Allelopathy J. 2019, 47, 57–72. [Google Scholar] [CrossRef]
- Singh, H.P.; Batish, D.R.; Kohli, R.K. Autotoxicity: Concept, organisms, and ecological significance. Crit. Rev. Plant Sci. 1999, 18, 757–772. [Google Scholar] [CrossRef]
- Kaur, H.; Kaur, R.; Kaur, S.; Baldwin, I.T.; Inderjit. Taking ecological function seriously: Soil microbial communities can obviate allelopathic effects of released metabolites. PLoS ONE 2009, 4, e4700. [Google Scholar] [CrossRef]
- Mazzola, M. Identification and pathogenicity of Rhizoctonia spp. isolated from apple roots and orchard soils. Phytopathology 1997, 87, 582–587. [Google Scholar] [CrossRef]
- Mazzola, M. Elucidation of the microbial complex having a causal role in the development of apple replant disease in Washington. Phytopathology 1998, 88, 930–938. [Google Scholar] [CrossRef]
- Manici, L.M.; Kelderer, M.; Caputo, F.; Saccà, M.L.; Nicoletti, F.; Topp, A.R.; Mazzola, M. Involvement of Dactylonectria and Ilyonectria spp. in tree decline affecting multi-generation apple orchards. Plant Soil 2018, 425, 217–230. [Google Scholar] [CrossRef]
- Tewoldemedhin, Y.T.; Mazzola, M.; Labuschagne, I.; McLeod, A. A multi-phasic approach reveals that apple replant disease is caused by multiple biological agents, with some agents acting synergistically. Soil Biol. Biochem. 2011, 43, 1917–1927. [Google Scholar] [CrossRef]
- Franke-Whittle, I.H.; Manici, L.M.; Insam, H.; Stres, B. Rhizosphere bacteria and fungi associated with plant growth in soils of three replanted apple orchards. Plant Soil 2015, 395, 317–333. [Google Scholar] [CrossRef]
- Yim, B.; Winkelmann, T.; Ding, G.-C.; Smalla, K. Different bacterial communities in heat and gamma irradiation treated replant disease soils revealed by 16S rRNA gene analysis—Contribution to improved aboveground apple plant growth? Front. Microbiol. 2015, 6. [Google Scholar] [CrossRef] [PubMed]
- Radl, V.; Winkler, J.B.; Kublik, S.; Yang, L.; Winkelmann, T.; Vestergaard, G.; Schröder, P.; Schloter, M. Reduced microbial potential for the degradation of phenolic compounds in the rhizosphere of apple plantlets grown in soils affected by replant disease. Environ. Microbiome 2019, 14, 8. [Google Scholar] [CrossRef]
- Jaffee, B.A.; Abawi, G.S.; Mai, W.F. Role of soil microflora and Pratylenchus penetrans in an apple replant disease. Phytopathology 1982, 72, 247–251. [Google Scholar] [CrossRef]
- Kanfra, X.; Liu, B.; Beerhues, L.; Sørensen, S.J.; Heuer, H. Free-living nematodes together with associated microbes play an essential role in apple replant disease. Front. Plant. Sci. 2018, 9, 1666. [Google Scholar] [CrossRef] [PubMed]
- Yim, B.; Nitt, H.; Wrede, A.; Jacquiod, S.; Sørensen, S.J.; Winkelmann, T.; Smalla, K. Effects of soil pre-teatment with Basamid® ganules, Brassica juncea, Raphanus sativus, and Tagetes patula on bacterial and fungal communities at two apple replant disease sites. Front. Microbiol. 2017, 8. [Google Scholar] [CrossRef]
- Forge, T.; Neilsen, G.; Neilsen, D. Organically acceptable practices to improve replant success of temperate tree-fruit crops. Sci. Hort. 2016, 200, 205–214. [Google Scholar] [CrossRef]
- Franke-Whittle, I.H.; Juárez, M.F.-D.; Insam, H.; Schweizer, S.; Naef, A.; Topp, A.-R.; Kelderer, M.; Rühmer, T.; Baab, G.; Henfrey, J.; et al. Performance evaluation of locally available composts to reduce replant disease in apple orchards of central Europe. Renew. Agric. Food Syst. 2018, 34, 543–557. [Google Scholar] [CrossRef]
- De Corato, U. Disease-suppressive compost enhances natural soil suppressiveness against soil-borne plant pathogens: A critical review. Rhizosphere 2020, 13, 100192. [Google Scholar] [CrossRef]
- Hewavitharana, S.S.; Mazzola, M. Carbon source-dependent efects of aaerobic soil disinfestation on soil microbiome and suppression of Rhizoctonia solani AG-5 and Pratylenchus penetrans. Phytopathology 2016, 106, 1015–1028. [Google Scholar] [CrossRef]
- Browne, G.; Ott, N.; Poret-Peterson, A.; Gouran, H.; Lampinen, B. Efficacy of anaerobic soil disinfestation for control of Prunus replant disease. Plant Dis. 2018, 102, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Dorcas, K.I.; Ian, A.M. Malus germplasm varies in resistance or tolerance to apple replant disease in a mixture of New York orchard soils. HortScience 2000, 35, 262–268. [Google Scholar] [CrossRef]
- Reim, S.; Siewert, C.; Winkelmann, T.; Wöhner, T.; Hanke, M.-V.; Flachowsky, H. Evaluation of Malus genetic resources for tolerance to apple replant disease (ARD). Sci. Hort. 2019, 256, 108517. [Google Scholar] [CrossRef]
- Nicola, L.; Turco, E.; Albanese, D.; Donati, C.; Thalheimer, M.; Pindo, M.; Insam, H.; Cavalieri, D.; Pertot, I. Fumigation with dazomet modifies soil microbiota in apple orchards affected by replant disease. Appl. Soil Ecol. 2017, 113, 71–79. [Google Scholar] [CrossRef]
- Nyoni, M.; Mazzola, M.; Wessels, J.P.B.; McLeod, A. The efficacy of semiselective chemicals and chloropicrin/1,3-dichloropropene–containing fumigants in managing apple replant disease in South Africa. Plant Dis. 2019, 103, 1363–1373. [Google Scholar] [CrossRef]
- Rudolph, R.E.; Zasada, I.A.; Hesse, C.; DeVetter, L.W. Brassicaceous seed meal, root removal, and chemical fumigation vary in their effects on soil quality parameters and Pratylenchus penetrans in a replanted floricane raspberry production system. Appl. Soil Ecol. 2019, 133, 44–51. [Google Scholar] [CrossRef]
- Gimsing, A.; Kirkegaard, J. Glucosinolates and biofumigation: Fate of glucosinolates and their hydrolysis products in soil. Phytochem. Rev. 2009, 8, 299–310. [Google Scholar] [CrossRef]
- Edwards, S.; Ploeg, A. Evaluation of 31 potential biofumigant Brassicaceous plants as hosts for three meloiodogyne species. J. Nematol. 2014, 46, 287–295. [Google Scholar]
- Ntalli, N.; Caboni, P. A review of isothiocyanates biofumigation activity on plant parasitic nematodes. Phytochem. Rev. 2017, 16, 827–834. [Google Scholar] [CrossRef]
- Kruger, D.H.M.; Fourie, J.C.; Malan, A.P. Control potential of Brassicaceae cover crops as green manure and their host status for Meloidogyne javanica and Criconemoides xenoplax. South Afr. J. Enol. Vitic. 2015, 36, 165–174. [Google Scholar] [CrossRef][Green Version]
- Neubauer, C.; Heitmann, B.; Müller, C. Biofumigation potential of Brassicaceae cultivars to Verticillium dahliae. Eur. J. Plant Pathol. 2014, 140, 341–352. [Google Scholar] [CrossRef]
- Ríos, P.; Obregón, S.; González, M.; de Haro, A.; Sánchez, M.E. Screening brassicaceous plants as biofumigants for management of Phytophthora cinnamomi oak disease. For. Pathol. 2016, 46, 652–659. [Google Scholar] [CrossRef]
- Matthiessen, J.N.; Kirkegaard, J.A. Biofumigation and enhanced biodegradation: Opportunity and challenge in soilborne pest and disease management. Crit. Rev. Plant Sci. 2006, 25, 235–265. [Google Scholar] [CrossRef]
- Brown, P.D.; Morra, M.J. Glucosinolate-containing plant tissues as bioherbicides. J. Agric. Food Chem. 1995, 43, 3070–3074. [Google Scholar] [CrossRef]
- Hanschen, F.S.; Lamy, E.; Schreiner, M.; Rohn, S. Reactivity and stability of glucosinolates and their breakdown products in foods. Angew. Chem. Int. Ed. 2014, 53, 11430–11450. [Google Scholar] [CrossRef] [PubMed]
- Blažević, I.; Montaut, S.; Burčul, F.; Olsen, C.E.; Burow, M.; Rollin, P.; Agerbirk, N. Glucosinolate structural diversity, identification, chemical synthesis and metabolism in plants. Phytochemistry 2020, 169, 112100. [Google Scholar] [CrossRef] [PubMed]
- Bellostas, N.; Sørensen, J.C.; Sørensen, H. Profiling glucosinolates in vegetative and reproductive tissues of four Brassica species of the U-triangle for their biofumigation potential. J. Sci. Food Agric. 2007, 87, 1586–1594. [Google Scholar] [CrossRef]
- Kirkegaard, J.A.; Sarwar, M. Biofumigation potential of brassicas I. Plant Soil 1998, 201, 71–89. [Google Scholar] [CrossRef]
- Sarwar, M.; Kirkegaard, J.A. Biofumigation potential of brassicas II. Plant Soil 1998, 201, 91–101. [Google Scholar] [CrossRef]
- Witzel, K.; Abu Risha, M.; Albers, P.; Börnke, F.; Hanschen, F.S. Identification and characterization of three epithiospecifier protein isoforms in Brassica oleracea. Front. Plant. Sci. 2019, 10. [Google Scholar] [CrossRef]
- Wittstock, U.; Burow, M. Glucosinolate breakdown in Arabidopsis: Mechanism, regulation and biological significance. Arab. Book 2010, 8, e0134. [Google Scholar] [CrossRef] [PubMed]
- Hanschen, F.S.; Yim, B.; Winkelmann, T.; Smalla, K.; Schreiner, M. Degradation of biofumigant isothiocyanates and allyl glucosinolate in soil and their effects on the microbial community composition. PLoS ONE 2015, 10, e0132931. [Google Scholar] [CrossRef] [PubMed]
- Hansson, D.; Morra, M.J.; Borek, V.; Snyder, A.J.; Johnson-Maynard, J.L.; Thill, D.C. Ionic thiocyanate (SCN−) production, fate, and phytotoxicity in soil amended with Brassicaceae seed meals. J. Agric. Food Chem. 2008, 56, 3912–3917. [Google Scholar] [CrossRef] [PubMed]
- Borek, V.; Morra, M.J. Ionic thiocyanate (SCN-) production from 4-hydroxybenzyl glucosinolate contained in Sinapis alba seed meal. J. Agric. Food Chem. 2005, 53, 8650–8654. [Google Scholar] [CrossRef]
- Dufour, V.; Stahl, M.; Baysse, C. The antibacterial properties of isothiocyanates. Microbiology 2015, 161, 229–243. [Google Scholar] [CrossRef] [PubMed]
- Fahey, J.W.; Haristoy, X.; Dolan, P.M.; Kensler, T.W.; Scholtus, I.; Stephenson, K.K.; Talalay, P.; Lozniewski, A. Sulforaphane inhibits extracellular, intracellular, and antibiotic-resistant strains of Helicobacter pylori and prevents benzo[a]pyrene-induced stomach tumors. Proc. Natl. Acad. Sci. USA 2002, 99, 7610–7615. [Google Scholar] [CrossRef]
- Hu, P.; Hollister, E.B.; Somenahally, A.C.; Hons, F.M.; Gentry, T.J. Soil bacterial and fungal communities respond differently to various isothiocyanates added for biofumigation. Front. Microbiol. 2015, 5. [Google Scholar] [CrossRef]
- Azaiez, I.; Meca, G.; Manyes, L.; Fernández-Franzón, M. Antifungal activity of gaseous allyl, benzyl and phenyl isothiocyanate in vitro and their use for fumonisins reduction in bread. Food Control 2013, 32, 428–434. [Google Scholar] [CrossRef]
- Aissani, N.; Tedeschi, P.; Maietti, A.; Brandolini, V.; Garau, V.L.; Caboni, P. Nematicidal activity of allylisothiocyanate from horseradish (Armoracia rusticana) roots against Meloidogyne incognita. J. Agric. Food Chem. 2013, 61, 4723–4727. [Google Scholar] [CrossRef]
- Gabler, F.M.; Fassel, R.; Mercier, J.; Smilanick, J.L. Influence of temperature, inoculation interval, and dosage on biofumigation with muscodor albus to control postharvest gray mold on grapes. Plant Dis. 2006, 90, 1019–1025. [Google Scholar] [CrossRef]
- Morra, M.J.; Kirkegaard, J.A. Isothiocyanate release from soil-incorporated Brassica tissues. Soil Biol. Biochem. 2002, 34, 1683–1690. [Google Scholar] [CrossRef]
- Brown, P.D.; Morra, M.J.; McCaffrey, J.P.; Auld, D.L.; Williams, L. Allelochemicals produced during glucosinolate degradation in soil. J. Chem. Ecol. 1991, 17, 2021–2034. [Google Scholar] [CrossRef] [PubMed]
- Gardiner, J.B.; Morra, M.J.; Eberlein, C.V.; Brown, P.D.; Borek, V. Allelochemicals released in soil following incorporation of rapeseed (Brassica napus) green manures. J. Agric. Food Chem. 1999, 47, 3837–3842. [Google Scholar] [CrossRef]
- Bangarwa, S.K.; Norsworthy, J.K.; Mattice, J.D.; Gbur, E.E. Glucosinolate and isothiocyanate production from Brassicaceae cover crops in a plasticulture production system. Weed Sci. 2011, 59, 247–254. [Google Scholar] [CrossRef]
- Gimsing, A.L.; Kirkegaard, J.A. Glucosinolate and isothiocyanate concentration in soil following incorporation of Brassica biofumigants. Soil Biol. Biochem. 2006, 38, 2255–2264. [Google Scholar] [CrossRef]
- Wang, L.; Mazzola, M. Field evaluation of reduced rate Brassicaceae seed meal amendment and rootstock genotype on the microbiome and control of apple replant disease. Phytopathology 2019, 109, 1378–1391. [Google Scholar] [CrossRef]
- Mazzola, M.; Brown, J. Efficacy of brassicaceous seed meal formulations for the control of apple replant disease in conventional and organic production systems. Plant Dis. 2010, 94, 835–842. [Google Scholar] [CrossRef]
- Mazzola, M.; Zhao, X. Brassica juncea seed meal particle size influences chemistry but not soil biology-based suppression of individual agents inciting apple replant disease. Plant Soil 2010, 337, 313–324. [Google Scholar] [CrossRef]
- Mazzola, M.; Brown, J.; Izzo, A.D.; Cohen, M.F. Mechanism of action and efficacy of seed meal-induced pathogen suppression differ in a Brassicaceae species and time-dependent manner. Phytopathology 2007, 97, 454–460. [Google Scholar] [CrossRef]
- Yim, B.; Hanschen, F.S.; Wrede, A.; Nitt, H.; Schreiner, M.; Smalla, K.; Winkelmann, T. Effects of biofumigation using Brassica juncea and Raphanus sativus in comparison to disinfection using Basamid on apple plant growth and soil microbial communities at three field sites with replant disease. Plant Soil 2016, 406, 389–408. [Google Scholar] [CrossRef]
- Gimsing, A.L.; Sørensen, J.C.; Strobel, B.W.; Hansen, H.C.B. Adsorption of glucosinolates to metal oxides, clay minerals and humic acid. Appl. Clay Sci. 2007, 35, 212–217. [Google Scholar] [CrossRef]
- Gimsing, A.L.; Strobel, B.W.; Hansen, H.C.B. Degradation and sorption of 2-propenyl and benzyl isothiocyanate in soil. Environ. Toxicol. Chem. 2009, 28, 1178–1184. [Google Scholar] [CrossRef] [PubMed]
- Borek, V.; Morra, M.J.; Brown, P.D.; McCaffrey, J.P. Transformation of the glucosinolate-derived allelochemicals allyl isothiocyanate and allylnitrile in soil. J. Agric. Food Chem. 1995, 43, 1935–1940. [Google Scholar] [CrossRef]
- Price, A.J.; Charron, C.S.; Saxton, A.M.; Sams, C.E. Allyl isothiocyanate and carbon dioxide produced during degradation of Brassica juncea tissue in different soil conditions. HortScience 2005, 40, 1734–1739. [Google Scholar] [CrossRef]
- Matthiessen, J.N.; Desmarchelier, J.M.; Vu, L.T.; Shackleton, M.A. Comparative efficacy of fumigants against hatchling whitefringed beetle (Coleoptera: Curculionidae) larvae and their sorption by soil. J. Econ. Entomol. 1996, 89, 1372–1378. [Google Scholar] [CrossRef]
- Matthiessen, J.N.; Shackleton, M.A. Biofumigation: Environmental impacts on the biological activity of diverse pure and plant-derived isothiocyanates. Pest Manag. Sci. 2005, 61, 1043–1051. [Google Scholar] [CrossRef]
- Dungan, R.S.; Gan, J.; Yates, S.R. Accelerated degradation of methyl isothiocyanate in soil. Water, Air, Soil Pollut. 2003, 142, 299–310. [Google Scholar] [CrossRef]
- Warton, B.; Matthiessen, J.N.; Shackleton, M.A. Cross-enhancement: Enhanced biodegradation of isothiocyanates in soils previously treated with metham sodium. Soil Biol. Biochem. 2003, 35, 1123–1127. [Google Scholar] [CrossRef]
- Cohen, M.F.; Yamasaki, H.; Mazzola, M. Brassica napus seed meal soil amendment modifies microbial community structure, nitric oxide production and incidence of Rhizoctonia root rot. Soil Biol. Biochem. 2005, 37, 1215–1227. [Google Scholar] [CrossRef]
- Mazzola, M.; Granatstein, D.M.; Elfving, D.C.; Mullinix, K. Suppression of specific apple root pathogens by Brassica napus seed meal amendment regardless of glucosinolate content. Phytopathology 2001, 91, 673–679. [Google Scholar] [CrossRef]
- Omirou, M.; Rousidou, C.; Bekris, F.; Papadopoulou, K.; Menkissoglou-Spiroudi, U.; Ehaliotis, C.; Karpouzas, D. The impact of biofumigation and chemical fumigation methods on the structure and function of the soil microbial community. Microb. Ecol. 2011, 61, 201–213. [Google Scholar] [CrossRef] [PubMed]
- Vervoort, M.T.W.; Vonk, J.A.; Brolsma, K.M.; Schütze, W.; Quist, C.W.; de Goede, R.G.M.; Hoffland, E.; Bakker, J.; Mulder, C.; Hallmann, J.; et al. Release of isothiocyanates does not explain the effects of biofumigation with Indian mustard cultivars on nematode assemblages. Soil Biol. Biochem. 2014, 68, 200–207. [Google Scholar] [CrossRef]
- Cohen, M.F.; Mazzola, M. Resident bacteria, nitric oxide emission and particle size modulate the effect of Brassica napus seed meal on disease incited by Rhizoctonia solani and Pythium spp. Plant Soil 2006, 286, 75–86. [Google Scholar] [CrossRef]
- Thorup-Kristensen, K.; Magid, J.; Jensen, L.S. Catch crops and green manures as biological tools in nitrogen management in temperate zones. In Advances in Agronomy; Academic Press: Cambridge, MA, USA, 2003; Volume 79, pp. 227–302. [Google Scholar]
- Wang, D.; Rosen, C.; Kinkel, L.; Cao, A.; Tharayil, N.; Gerik, J. Production of methyl sulfide and dimethyl disulfide from soil-incorporated plant materials and implications for controlling soilborne pathogens. Plant Soil 2009, 324, 185–197. [Google Scholar] [CrossRef]
- Bending, G.D.; Lincoln, S.D. Characterisation of volatile sulphur-containing compounds produced during decomposition of Brassica juncea tissues in soil. Soil Biol. Biochem. 1999, 31, 695–703. [Google Scholar] [CrossRef]
- Lewis, J.; Papavizas, G. Evolution of volatile sulfur-containing compounds from decomposition of crucifers in soil. Soil Biol. Biochem. 1970, 2, 239–246. [Google Scholar] [CrossRef]
- Bremner, J.; Bundy, L. Inhibition of nitrification in soils by volatile sulfur compounds. Soil Biol. Biochem. 1974, 6, 161–165. [Google Scholar] [CrossRef]
- Canessa, E.F.; Morrell, J.J. Effect of mixtures of carbon disulfide and methylisothiocyanate on survival of wood-colonizing fungi. Wood Fiber Sci. 1995, 27, 207–224. [Google Scholar]
- Gurtler, J.B. Pathogen decontamination of food crop soil: A review. J. Food Prot. 2017, 80, 1461–1470. [Google Scholar] [CrossRef]
- Marks, H.S.; Hilson, J.A.; Leichtweis, H.C.; Stoewsand, G.S. S-Methylcysteine sulfoxide in Brassica vegetables and formation of methyl methanethiosulfinate from Brussels sprouts. J. Agric. Food Chem. 1992, 40, 2098–2101. [Google Scholar] [CrossRef]
- Morris, C.J.; Thompson, J.F. The identification of (+)S-methyl-L-cysteine sulfoxide in plants. J. Am. Chem. Soc. 1956, 78, 1605–1608. [Google Scholar] [CrossRef]
- De Corato, U.; Pane, C.; Bruno, G.L.; Cancellara, F.A.; Zaccardelli, M. Co-products from a biofuel production chain in crop disease management: A review. Crop. Prot. 2015, 68, 12–26. [Google Scholar] [CrossRef]
- De Corato, U.; De Bari, I.; Viola, E.; Pugliese, M. Assessing the main opportunities of integrated biorefining from agro-bioenergy co/by-products and agroindustrial residues into high-value added products associated to some emerging markets: A review. Renew. Sustain. Energy Rev. 2018, 88, 326–346. [Google Scholar] [CrossRef]
- Van Ommen Kloeke, A.E.E.; van Gestel, C.M.; Styrishave, B.; Hansen, M.; Ellers, J.; Roelofs, D. Molecular and life-history effects of a natural toxin on herbivorous and non-target soil arthropods. Ecotoxicology 2012, 21, 1084–1093. [Google Scholar] [CrossRef] [PubMed]
- Van Ommen Kloeke, A.E.E.; Jager, T.; van Gestel, C.A.M.; Ellers, J.; Pomeren, M.v.; Krommenhoek, T.; Styrishave, B.; Hansen, M.; Roelofs, D. Time-related survival effects of two gluconasturtiin hydrolysis products on the terrestrial isopod Porcellio Scaber. Chemosphere 2012, 89, 1084–1090. [Google Scholar] [CrossRef] [PubMed]
- Van Ommen Kloeke, A.E.E.; Gong, P.; Ellers, J.; Roelofs, D. Effects of a natural toxin on life history and gene expression of Eisenia andrei. Environ. Toxicol. Chem. 2014, 33, 412–420. [Google Scholar] [CrossRef] [PubMed]
- Fouché, T.; Maboeta, M.; Claassens, S. Effect of biofumigants on soil microbial communities and ecotoxicology of earthworms (Eisenia andrei). Water Air Soil Pollut. 2016, 227, 256. [Google Scholar] [CrossRef]
- Dutta, T.K.; Khan, M.R.; Phani, V. Plant-parasitic nematode management via biofumigation using brassica and non-brassica plants: Current status and future prospects. Curr. Plant Biol. 2019, 17, 17–32. [Google Scholar] [CrossRef]
- Vukicevich, E.; Lowery, T.; Bowen, P.; Úrbez-Torres, J.R.; Hart, M. Cover crops to increase soil microbial diversity and mitigate decline in perennial agriculture. A review. Agron. Sustain. Dev. 2016, 36, 48. [Google Scholar] [CrossRef]
- Bending, G.D.; Lincoln, S.D. Inhibition of soil nitrifying bacteria communities and their activities by glucosinolate hydrolysis products. Soil Biol. Biochem. 2000, 32, 1261–1269. [Google Scholar] [CrossRef]
- Siebers, M.; Rohr, T.; Ventura, M.; Schütz, V.; Thies, S.; Kovacic, F.; Jaeger, K.-E.; Berg, M.; Dörmann, P.; Schulz, M. Disruption of microbial community composition and identification of plant growth promoting microorganisms after exposure of soil to rapeseed-derived glucosinolates. PLoS ONE 2018, 13, e0200160. [Google Scholar] [CrossRef]
- Wang, Q.; Ma, Y.; Yang, H.; Chang, Z. Effect of biofumigation and chemical fumigation on soil microbial community structure and control of pepper Phytophthora blight. World J. Microbiol. Biotechnol. 2014, 30, 507–518. [Google Scholar] [CrossRef] [PubMed]
- Zaccardelli, M.; Villecco, D.; Celano, G.; Scotti, R. Soil amendment with seed meals: Short term effects on soil respiration and biochemical properties. Appl. Soil Ecol. 2013, 72, 225–231. [Google Scholar] [CrossRef]
- Meng, L.; Yao, X.; Yang, Z.; Zhang, R.; Zhang, C.; Wang, X.; Xu, N.; Li, S.; Liu, T.; Zheng, C. Changes in soil microbial diversity and control of Fusarium oxysporum in continuous cropping cucumber greenhouses following biofumigation. Emir. J. Food Agric. 2018, 30, 644–653. [Google Scholar] [CrossRef]
- Wang, Q.; Ma, Y.; Wang, G.; Gu, Z.; Sun, D.; An, X.; Chang, Z. Integration of biofumigation with antagonistic microorganism can control Phytophthora blight of pepper plants by regulating soil bacterial community structure. Eur. J. Soil Biol. 2014, 61, 58–67. [Google Scholar] [CrossRef]
- Nuñez-Zofío, M.; Larregla del Palacio, S.; Garbisu, C. Repeated biodisinfection controls the incidence of Phytophthora root and crown rot of pepper while improving soil quality. Span. J. Agric. Res. 2012, 10, 12. [Google Scholar] [CrossRef]
- Mowlick, S.; Yasukawa, H.; Inoue, T.; Takehara, T.; Kaku, N.; Ueki, K.; Ueki, A. Suppression of spinach wilt disease by biological soil disinfestation incorporated with Brassica juncea plants in association with changes in soil bacterial communities. Crop. Prot. 2013, 54, 185–193. [Google Scholar] [CrossRef]
- Behera, B.C.; Patra, M.; Dutta, S.K.; Thatoi, H.N. Isolation and characterisation of sulphur oxidising Bacteria from mangrove soil of Mahanadi river delta and their sulphur oxidising ability. J. Appl. Environ. Microbiol. 2014, 2, 1–5. [Google Scholar] [CrossRef]
- Zaccardelli, M.; De Nicola, F.; Villecco, D.; Scotti, R. The development and suppressive activity of soil microbial communities under compost amendment. J. Soil Sci. Plant Nutr. 2013, 13, 730–742. [Google Scholar] [CrossRef]
- Palaniyandi, S.A.; Yang, S.H.; Zhang, L.; Suh, J.-W. Effects of actinobacteria on plant disease suppression and growth promotion. Appl. Microbiol. Biotechnol. 2013, 97, 9621–9636. [Google Scholar] [CrossRef]
- Grunewaldt-Stöcker, G.; Mahnkopp, F.; Popp, C.; Maiss, E.; Winkelmann, T. Diagnosis of apple replant disease (ARD): Microscopic evidence of early symptoms in fine roots of different apple rootstock genotypes. Sci. Hort. 2019, 243, 583–594. [Google Scholar] [CrossRef]
- Edwards, L.; Utkhede, R.S.; Vrain, T. Effect of antagonistic plants on apple replant disease. Acta Hortic. 1994, 363, 135–140. [Google Scholar] [CrossRef]
- Mazzola, M.; Mullinix, K. Comparative field efficacy of management strategies containing Brassica napus seed meal or green manure for the control of apple replant disease. Plant Dis. 2005, 89, 1207–1213. [Google Scholar] [CrossRef] [PubMed]
- Mazzola, M.; Brown, J.; Izzo, A.; Ghanem, R.A.; Cohen, M. Progress towards development of biologically-based strategies for the management of apple replant disease. Phytopathol. Pol. 2006, 39, 11–18. [Google Scholar]
- Seigies, A.T.; Pritts, M. Cover crop rotations alter soil microbiology and reduce replant disorders in strawberry. HortScience 2006, 41, 1303. [Google Scholar] [CrossRef]
- Mazzola, M.; Brown, J.; Zhao, X.; Izzo, A.D.; Fazio, G. Interaction of brassicaceous seed meal and apple rootstock on recovery of Pythium spp. and Pratylenchus penetrans from roots grown in replant soils. Plant Dis. 2009, 93, 51–57. [Google Scholar] [CrossRef]
- Mazzola, M.; Reardon, C.L.; Brown, J. Initial Pythium species composition and Brassicaceae seed meal type influence extent of Pythium-induced plant growth suppression in soil. Soil Biol. Biochem. 2012, 48, 20–27. [Google Scholar] [CrossRef]
- Weerakoon, D.M.N.; Reardon, C.L.; Paulitz, T.C.; Izzo, A.D.; Mazzola, M. Long-term suppression of Pythium abappressorium induced by Brassica juncea seed meal amendment is biologically mediated. Soil Biol. Biochem. 2012, 51, 44–52. [Google Scholar] [CrossRef]
- Mazzola, M.; Hewavitharana, S.S.; Strauss, S.L. Brassica seed meal soil amendments transform the rhizosphere microbiome and improve apple production through resistance to pathogen reinfestation. Phytopathology 2015, 105, 460–469. [Google Scholar] [CrossRef]
- Pokharel, R.R.; Reighard, G.L. Evaluation of biofumigation, soil solarization and rootstock on peach replant disease. Acta Hortic. 2015, 1084, 577–584. [Google Scholar] [CrossRef]
- Wang, L.; Mazzola, M. Interaction of Brassicaceae seed meal soil amendment and apple rootstock genotype on microbiome structure and replant disease suppression. Phytopathology 2019, 109, 607–614. [Google Scholar] [CrossRef] [PubMed]
| Kind of Replant Disease | Bio-Fumigation Treatment | Environmental Conditions | Measurement of Efficacy by | Efficacy | Observations | Ref. |
|---|---|---|---|---|---|---|
| Apple replant disease | Brassica napus as cover crop | No information provided | Field trials, counts of Pratylenchus penetrans and recovery of Pythium from soil | No positive effects | No reduction, but rather an increase in Pratylenchus penetrans and Pythium abundance | [114] |
| Apple replant disease | B. napus seed meal 0.1–2.0% | Incubation in the greenhouse (20 °C), no information on soil moisture etc. | Greenhouse pot trials | Increased plant growth, but toxic effects at high concentration. | No consistent reduction in Pythium infections, suppression of Rhizoctonia and Pratylenchus penetrans at 0.1% and increased abundance of fluorescent Pseudomonas spp. at 0.1 and 1.0% | [81] |
| Apple replant disease | B. napus seed meal 8.5 t ha−1 and green manure (for one-three years) | Seed meal incorporation in May 2001, some variants covered by plastic foil (no information on soil temperature/moisture) | Field trial with tree growth and yield measurements | Growth and yield improvement by both, B. napus green manure and seed meal treatments, especially when combined with fungicide treatment. | Reduction of ARD associated pathogens, i.e., Pratylenchus penetrans, Pythium, Cylindrocarpon, Rhizoctonia, but not Fusarium by the combined treatment of B. napus seed meal and fungicide, not by green manure. | [115] |
| Apple replant disease | Brassica juncea plant material (1–3 years) and B. napus seed meal combined with other treatments | No information provided | Field test and greenhouse bio-test of plant growth and yield (field) | Cumulative yield increase in a site-dependent way, mainly by seed meal treatments | Control of Cylindrocarpon, Rhizoctonia and Pythium ultimum by seed meal treatments, best in combination with a fungicide treatment, lower effect on Pratylenchus penetrans | [116] |
| Straw-berry replant disease | B. juncea cover crop incorpo-rated into the soil | Incorporation of plant residues in April 2002, no further information on soil temperature or moisture | Pot trial and field experiment | Fruit yield as well as vegetative growth parameters increased in the pot and the field trial | Rhizoctonia abundance was reduced by mustard treatment, but causes for this kind of replant disease is not clear. | [117] |
| Apple replant disease | Seed meals of B. juncea, Sinapis alba and B. napus; 0.5% (wt/wt) | Eight weeks of incubation at 22 ± 3 °C, no information on soil moisture | Greenhouse bio-test in pots | Seed meal improved apple seedling growth, seed meal reduced Rhizoctonia solani infection in native but not in pasteurized soil, while Streptomyces ssp. increased it | B. juncea seed meal was most effective in Pratylenchus penetrans suppression and the only seed meal that did not increase Phytium populations | [70] |
| Apple replant disease | Seed meals of B. juncea, S. alba and B. napus; 0.5% (vol/vol) | Blending and sieving (< 1 mm) of seed meals, 8 weeks of incubation at 22 ± 3 °C, no information on soil moisture | Greenhouse bio-test in pots | Seed meal-specific effects on Pythium and Pratylenchus penetrans numbers and infections. | B. juncea seed meal suppressed Pythium and P. penetrans populations. | [118] |
| Apple replant disease | Seed meal of B. juncea; 0.3% (wt/wt) = 4.5 t ha−1 | Fine (<1 mm) and coarse (2–4 mm) seed meal particles incorporated, no further information on soil temperature or moisture | Bio-test in greenhouse, variation of particle sizes of seed meal | Suppression of Rhizoctonia solani SG5 (for fine seed meal), Pratylenchus penetrans and Pythium spp. infections | Biological and chemical effects of the seed meal, increased population densities of Streptomyces and more free-living nematodes | [69] |
| Apple replant disease | Seed meals of B. juncea, S. alba and B. napus; 4.5 kg m−1 tree row | Incorporation in April 2005, May 2006, April 2007, respectively, tarped with plastic foil for 1 week, no further information on soil temperature or moisture | Field trial with measures of tree diameter and cumulative yield | Significant improvement of tree growth and cumulative fruit yield when seed meals (except for B. napus) were combined with fungicide soil drench | Seed meal specific effects, B. napus resulted in increased Pythium and Pratylenchus penetrans densities, whereas B. juncea reduced both pathogens as well as Cylindrocarpon infections but only when combined with fungicide drench. Without fungicide treatment, B. napus and S. alba seed meal amendments caused Pythium and B. juncea caused Phytophtora infections. | [68] |
| Apple replant disease | Seed meals of B. juncea, S. alba and B. napus; 0.3% (wt/wt) | Blending and sieving (<1mm) of seed meals, 48 h incubation in plastic bags, no information on soil temperature or moisture | Bio-test in greenhouse | Reduction of apple seedling mortality after B. juncea seed meal application in one soil. | Soil-dependent and seed meal-dependent shifts in Pythium communities, S. alba led to increased P. ultimum levels. | [119] |
| Apple replant disease | Seed meal of B. juncea; 0.3% (wt/wt) = 4.5 t ha−1 | Fine (<1 mm) and coarse (2–4 mm) seed meal particles incorporated, bagged or non-bagged incubation for 48 h, no further information on soil temperature or moisture | Bio-test in greenhouse | Reduction of Pythium abappressorium infections, especially in the bagged variants | Suppressiveness of soil was achieved, possibly due to long-term changes in fungal communities, especially promotion of Trichoderma spp. | [120] |
| Apple replant disease | Seed meal blends of B. juncea, S. alba and B. napus; 6.7 t ha−1 | Incorporation of seed meals once in March 2010 or twice in September 2009 and April 2010, tarped for 1 week, no further information on soil temperature or moisture | Field test of plant growth and yield | Significant increase in tree growth of B. juncea + S. alba, positive long-term effect (4 years), but mortality if applied few weeks prior to planting. Efficacy superior to chemical fumigation | Effective reduction of Pratylenchus penetrans mainly in the first year, Pythium infections enduringly reduced. Resilient changes in rhizosphere microbial communities. | [121] |
| Peach replant disease | B. juncea plant biomass and canola seed meal cake in a field experiment | Watering before incorporation in June 20, 1 day later tarping, recording of soil temperature during the 2-months treatment (26–34 °C) | Field test of plant growth | Significantly improved tree growth | Better plant health, lower mortality | [122] |
| Apple replant disease | Incorpora-tion of plant material of B. juncea and Raphanus sativus in the field | Incorporation in May and August 2012 and 2013, no further information on soil temperature or moisture | Greenhouse bio-test of plant growth and field test | Site specific increase in biomass production after biofumigation. | Nutrient effect and stronger shifts in fungal than in bacterial community composition | [71] |
| Apple replant disease | Incorpora-tion of plant material of B. juncea and R. sativus in the field | Incorporation in May and August 2012 and 2013, no further information on soil temperature or moisture | Field test of plant growth | Site specific effects (only in the tested sandy soil about 150% increase in growth, no significant change in the second soil). | Bacterial genera with increased abundance: Arthrobacter (R. sativus), Ferruginibacter (B. juncea, R. sativus). Fungal genera of higher abundance: Podospora, Monographella and Mucor (B. juncea, R. sativus) | [27] |
| Apple replant disease | Incorpora-tion of seed meal formulation of B. juncea and S. alba 1:1 in the field, 2.2, 4.4, 6.6 t ha−1 | Incorporation in April 2016, tarping for 2 weeks, soil temperature: 12−14 °C, no information on soil moisture | Field test of plant growth | Significantly improved tree growth, 4.4 t ha−1 was optimal | Soil fumigation and seed meal amendments suppressed Pythium infection in rootstook-specific way. Long-term effect on soil microbial communities. Beneficial microbes increased due to biofumigation | [67] |
| Apple replant disease | Incorpora-tion of seed meal formulation of B. juncea and S. alba 1:1, dosage, 2.2, 4.4, 6.6 t ha−1 | Incorporation into moist soil (−63 to −92 hPa), incubation in bags for 48 h under greenhouse conditions | Greenhouse bio-test in pots | Significantly improved tree growth at all dosages; no difference between 4.4 and 6.6 t ha−1, high efficacy in P. penetrans and Pythium ssp. control | Geneva rootstocks had less colonization by Pythium ssp. or P. penetrans compared to Mailling rootstocks; both rootstock genotype and soil treatment affected soil microbiom | [123] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hanschen, F.S.; Winkelmann, T. Biofumigation for Fighting Replant Disease- A Review. Agronomy 2020, 10, 425. https://doi.org/10.3390/agronomy10030425
Hanschen FS, Winkelmann T. Biofumigation for Fighting Replant Disease- A Review. Agronomy. 2020; 10(3):425. https://doi.org/10.3390/agronomy10030425
Chicago/Turabian StyleHanschen, Franziska S., and Traud Winkelmann. 2020. "Biofumigation for Fighting Replant Disease- A Review" Agronomy 10, no. 3: 425. https://doi.org/10.3390/agronomy10030425
APA StyleHanschen, F. S., & Winkelmann, T. (2020). Biofumigation for Fighting Replant Disease- A Review. Agronomy, 10(3), 425. https://doi.org/10.3390/agronomy10030425






