An Overview on Wood Waste Valorization as Biopolymers and Biocomposites: Definition, Classification, Production, Properties and Applications
Abstract
1. Introduction
2. General Definition of Biopolymers and Biocomposites
2.1. Biopolymers: Difference between Biodegradable and Bio-Based
2.1.1. Biodegradable Polymers
2.1.2. Bio-Based Polymers
2.2. Biocomposites
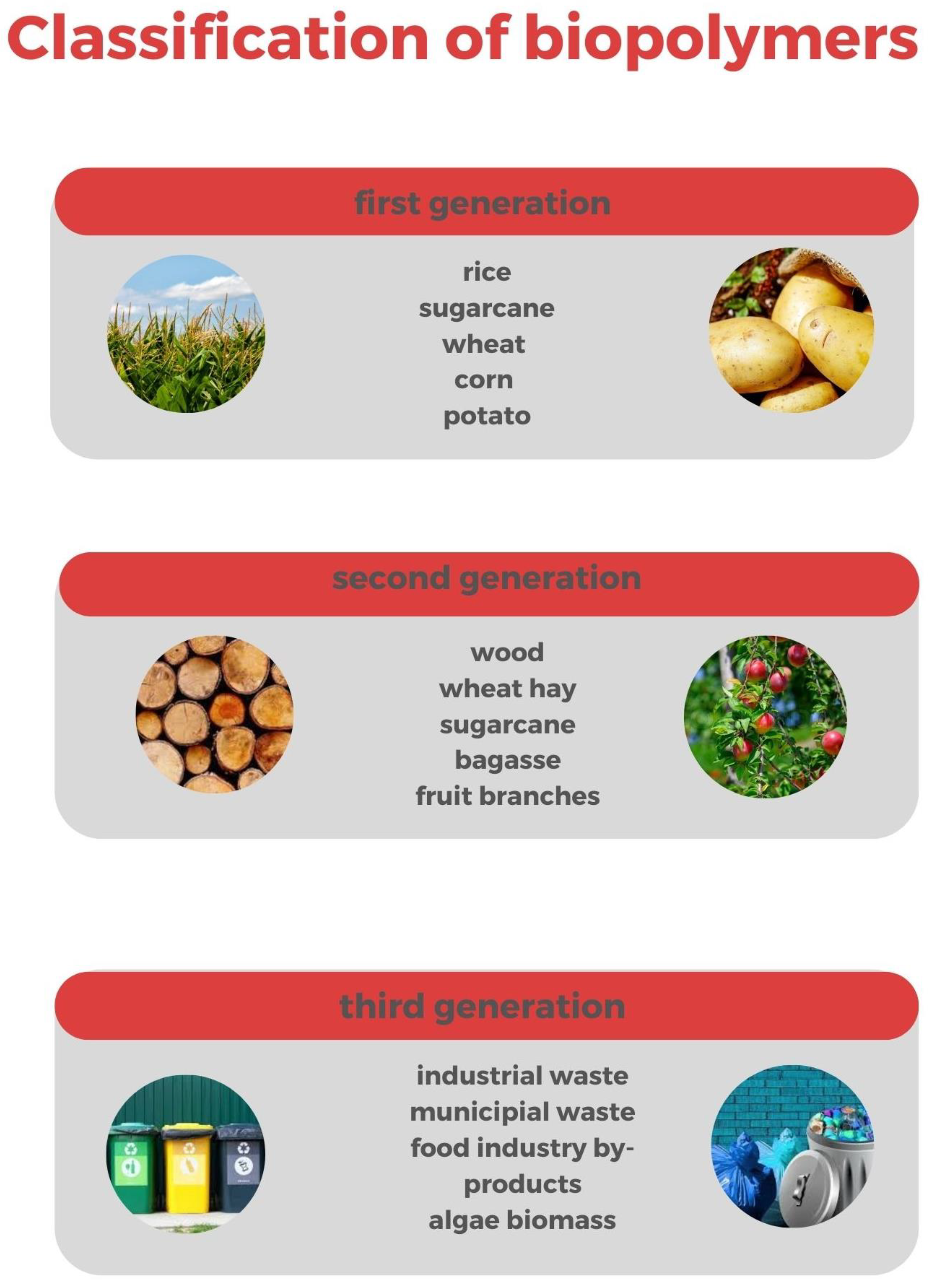
3. Production of the Main Biopolymers from Bio-Based Sources
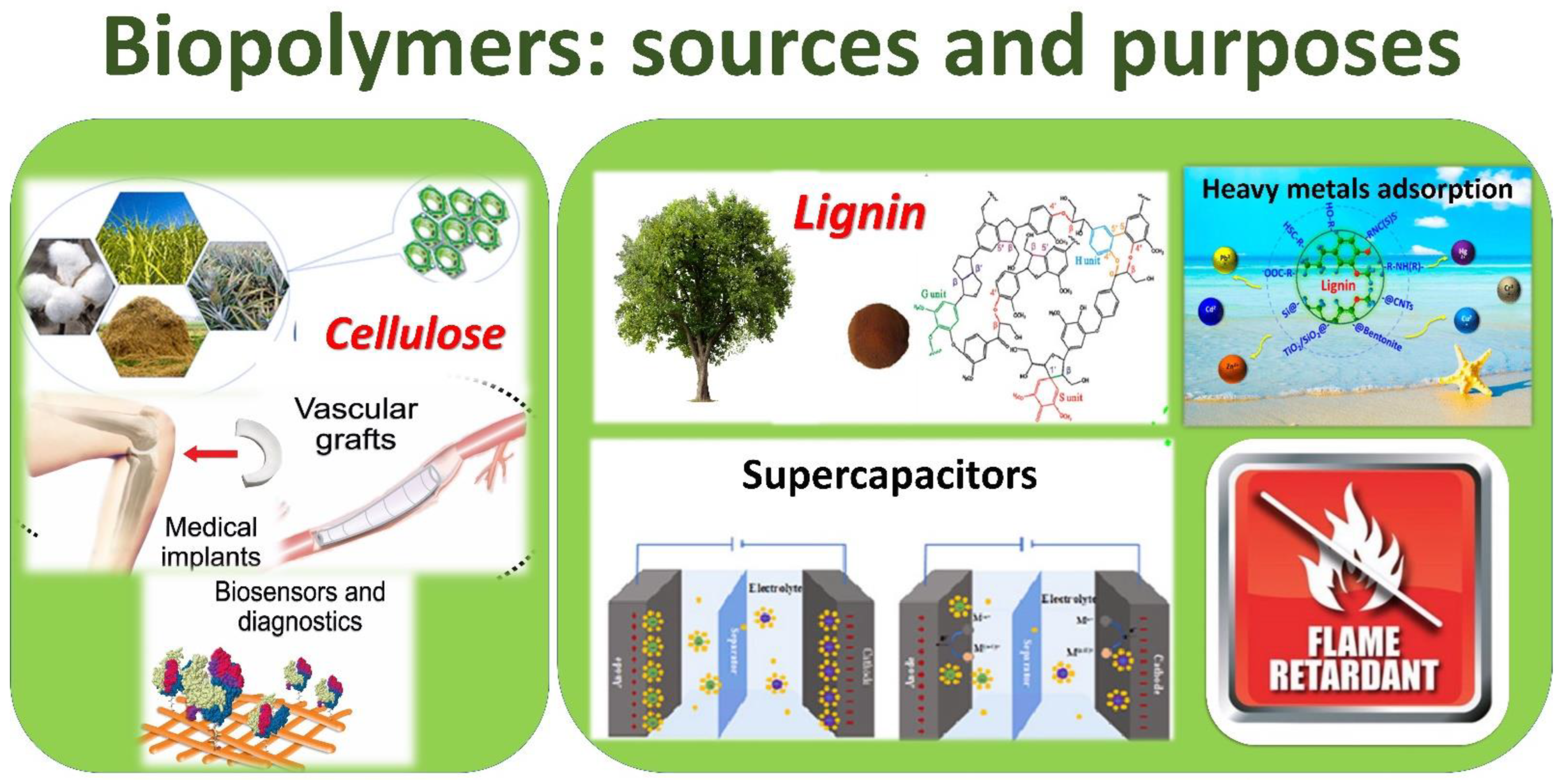
3.1. Cellulose Traditional Sources
3.2. Lignin Traditional Sources
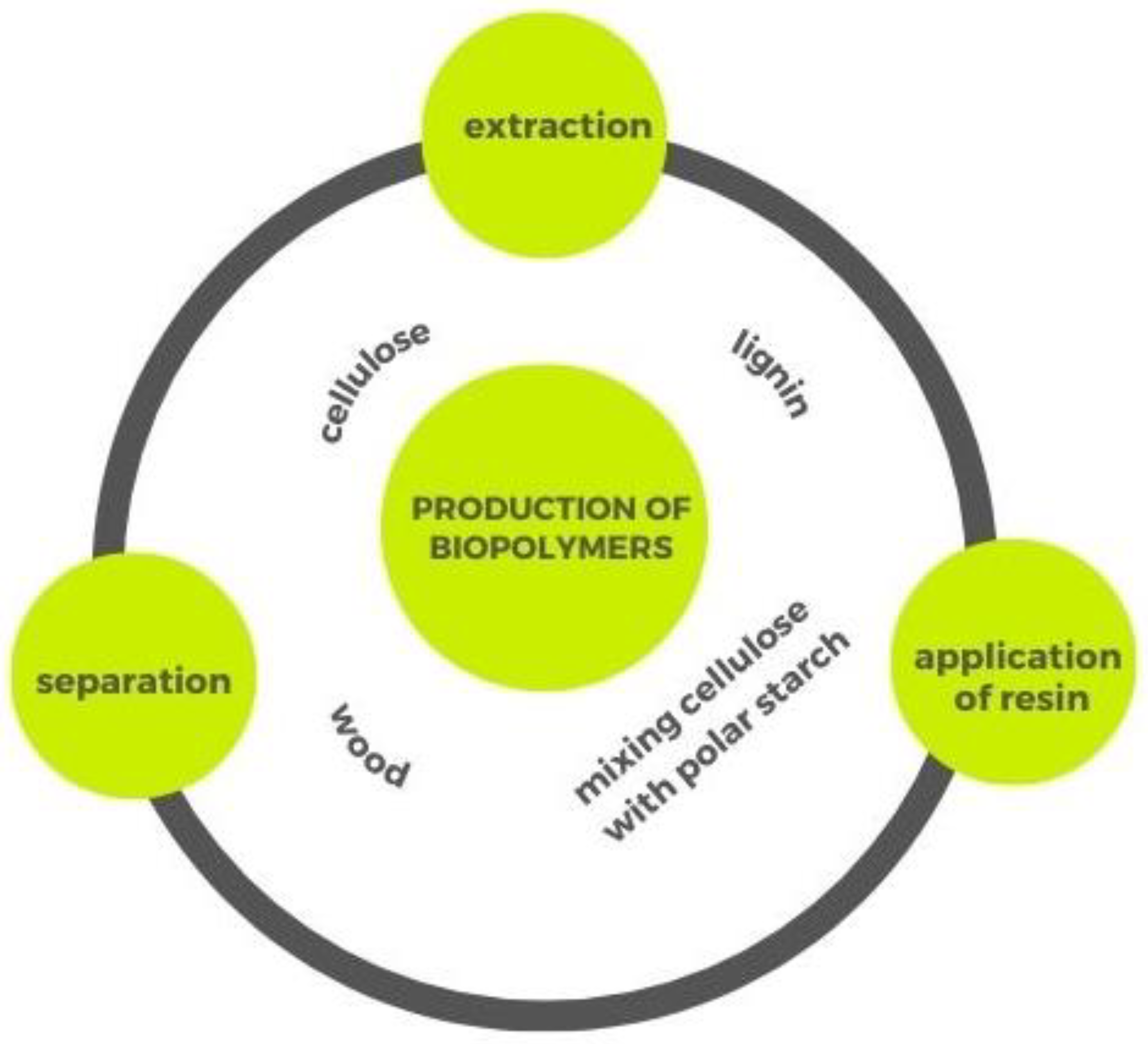
3.3. Cellulose and Lignin Biopolymers from Wood Waste
3.4. PHAs’ Traditional Sources
- First, starting with sugar, two molecules of acetyl-CoA are combined with 3-ketothiolase (PhaA) and acetoacetyl-CoA is obtained.
- In the second step, acetoacetyl-CoA is reduced using Acetoacethyl-CoA reductase (PhaB) to obtain 3-hydroxybutyryl-CoA.
- In the third step, PHB is obtained after the conversion of 3-hydroxybutyryl-CoA using PHB synthase (PhaC).
3.5. PHA from Wood Wastes
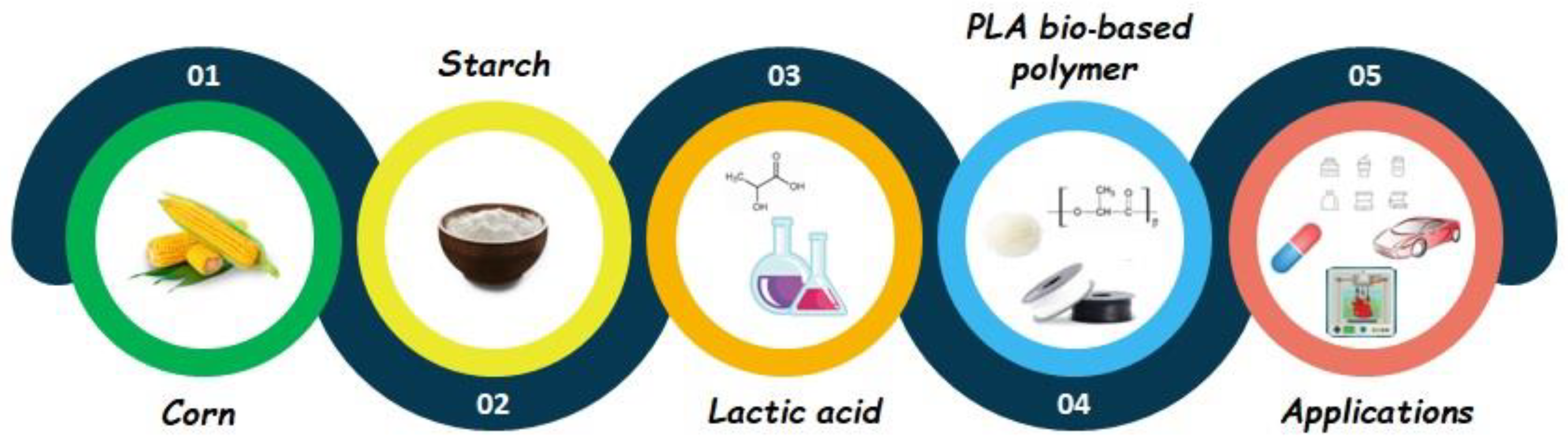
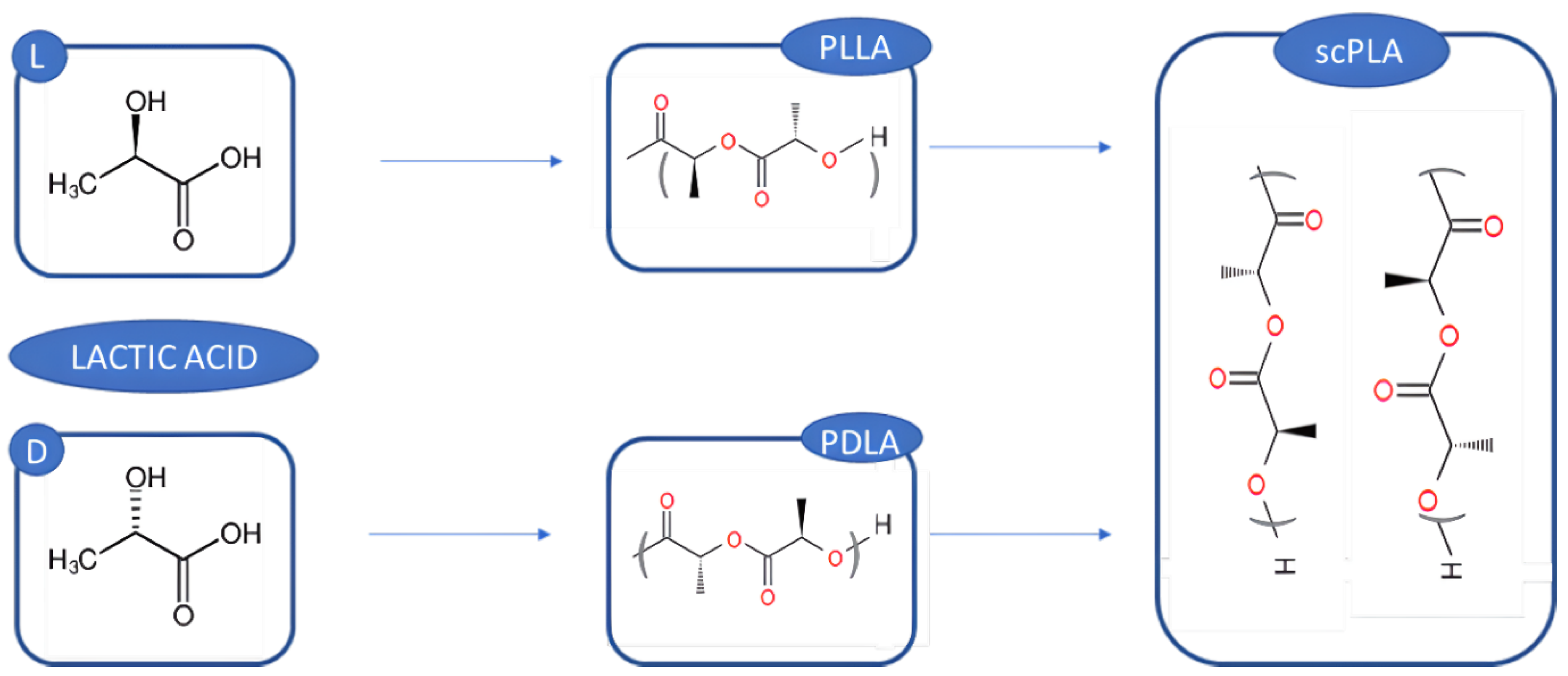
3.6. PLA’s Traditional Sources
- In the first one, mono- and disaccharides are produced by the hydrolysis of carbohydrate sources.
- Then, lactic acid is attained by the fermentation of saccharides through lactic acid bacteria (LAB).
- Finally, the purity of lactic acid is obtained using further purification processes.
- −
- Glucose and glucose syrups deriving from the conversion of starch with enzymes such as glucoamylases;
- −
- Maltose, derived from the starch enzymatic conversion with amylases of malt;
- −
- Sucrose, obtained as a byproduct or intermediate of cane sugar;
- −
- Lactose, derived from mil whey, a natural substrate of several lactic acid bacteria.
3.7. PLA from Wood Wastes Sources
4. Main Properties of Bio-Based Polymers and Biocomposites
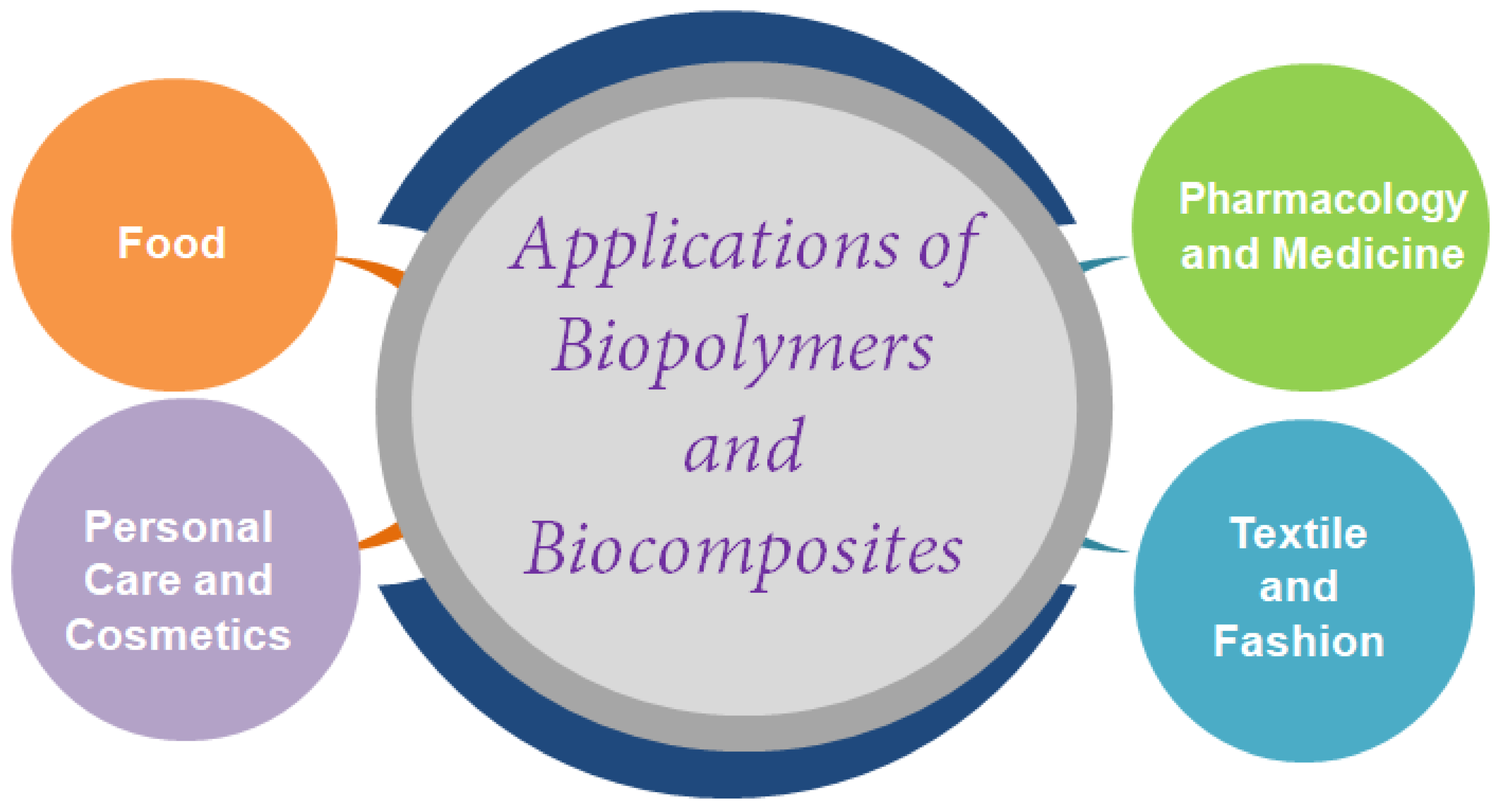
5. Market Scenarios and Applications of Biopolymers and Biocomposites
5.1. Biopolymers in Food Industry
5.2. Biopolymers in Pharmacology and Medicine
5.3. Biopolymers in Personal Care and Cosmetics
5.4. Biopolymers in Textile and Fashion
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sikorska, W.; Musioł, M.; Zawidlak-Węgrzyńska, B.; Rydz, J. End-of-Life Options for (Bio)Degradable Polymers in the Circular Economy. Adv. Polym. Technol. 2021, 2021, 6695140. [Google Scholar] [CrossRef]
- Lionetto, F.; Esposito Corcione, C. An Overview of the Sorption Studies of Contaminants on Poly(Ethylene Terephthalate) Microplastics in the Marine Environment. J. Mar. Sci. Eng. 2021, 9, 445. [Google Scholar] [CrossRef]
- Storz, H.; Vorlop, K.D. Bio-Based Plastics: Status, Challenges and Trends. Landbauforsch. Volkenrode 2013, 63, 321–332. [Google Scholar] [CrossRef]
- González-Barreiro, C.; Rial-Otero, R.; Simal-Gándara, J.; Astray, G.; Cid, A.; Mejuto, J.C.; Morales, J. Starch-Derived Cyclodextrins and Their Future in the Food Biopolymer Industry. In Starch-Based Polymeric Materials and Nanocomposites Chemistry, Processing, and Applications; CRC Press: Boca Raton, FL, USA, 2012; pp. 167–182. [Google Scholar]
- Striani, R.; Stasi, E.; Giuri, A.; Seiti, M.; Ferraris, E.; Esposito Corcione, C. Development of an Innovative and Green Method to Obtain Nanoparticles from Carbon-Based Waste Ashes. Nanomaterials 2021, 11, 577. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, F.; Esposito Corcione, C.; Montagna, F.; Maffezzoli, A. 3D Printing of Polymer Waste for Improving People’s Awareness about Marine Litter. Polymers 2020, 12, 1738. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, F.; Striani, R.; Minosi, S.; de Fazio, R.; Visconti, P.; Patrono, L.; Catarinucci, L.; Esposito Corcione, C.; Greco, A. An Innovative IoT-Oriented Prototype Platform for the Management and Valorization of the Organic Fraction of Municipal Solid Waste. J. Clean. Prod. 2020, 247, 119618. [Google Scholar] [CrossRef]
- Sánchez-Gutiérrez, M.; Espinosa, E.; Bascón-Villegas, I.; Pérez-Rodríguez, F.; Carrasco, E.; Rodríguez, A. Production of Cellulose Nanofibers from Olive Tree Harvest—A Residue with Wide Applications. Agronomy 2020, 10, 696. [Google Scholar] [CrossRef]
- Esposito Corcione, C.; Ferrari, F.; Striani, R.; Visconti, P.; Greco, A. Recycling of Organic Fraction of Municipal Solid Waste as an Innovative Precursor for the Production of Bio-Based Epoxy Monomers. Waste Manag. 2020, 109, 212–221. [Google Scholar] [CrossRef]
- Ballinas-Casarrubias, L.; Camacho-Davila, A.; Gutierrez-Méndez, N.; Ramos-Sánchez, V.H.; Chávez-Flores, D.; Manjarrez-Nevárez, L.; González-Sanchez, G. Biopolymers from Waste Biomass—Extraction, Modification and Ulterior Uses. In Recent Advances in Biopolymers; Intechopen: London, UK, 2016; pp. 3–18. [Google Scholar]
- Ibrahim, N.I.; Shahar, F.S.; Sultan, M.T.H.; Md Shan, A.U.; Safri, S.N.A.; Yazik, M.H.M. Overview of Bioplastic Introduction and Its Applications in Product Packaging. Coatings 2021, 11, 1423. [Google Scholar] [CrossRef]
- Fico, D.; Rizzo, D.; Casciaro, R.; Esposito Corcione, C. A Review of Polymer-Based Materials for Fused Filament Fabrication (FFF): Focus on Sustainability and Recycled Materials. Polymers 2022, 14, 465. [Google Scholar] [CrossRef]
- Chan, J.X.; Wong, J.F.; Hassan, A.; Zakaria, Z. Bioplastics from Agricultural Waste; Matthew Deans: Oxford, UK, 2021. [Google Scholar]
- Elsawy, M.A.; Kim, K.H.; Park, J.W.; Deep, A. Hydrolytic Degradation of Polylactic Acid (PLA) and Its Composites. Renew. Sustain. Energy Rev. 2017, 79, 1346–1352. [Google Scholar] [CrossRef]
- Reichert, C.L.; Bugnicourt, E.; Coltelli, M.B.; Cinelli, P.; Lazzeri, A.; Canesi, I.; Braca, F.; Martínez, B.M.; Alonso, R.; Agostinis, L.; et al. Bio-Based Packaging: Materials, Modifications, Industrial Applications and Sustainability. Polymers 2020, 12, 1558. [Google Scholar] [CrossRef] [PubMed]
- Gontard, N.; Sonesson, U.; Birkved, M.; Majone, M.; Bolzonella, D.; Celli, A.; Angellier-Coussy, H.; Jang, G.W.; Verniquet, A.; Broeze, J.; et al. A Research Challenge Vision Regarding Management of Agricultural Waste in a Circular Bio-Based Economy. Crit. Rev. Environ. Sci. Technol. 2018, 48, 614–654. [Google Scholar] [CrossRef]
- Ghanbarzadeh, B.; Almasi, H. Biodegradable Polymers. In Biodegradation-Life of Science; Intechopen: London, UK, 2013; pp. 141–185. [Google Scholar]
- Raza, S.; Zhang, J.; Ali, I.; Li, X.; Liu, C. Recent Trends in the Development of Biomass-Based Polymers from Renewable Resources and Their Environmental Applications. J. Taiwan Inst. Chem. Eng. 2020, 115, 293–303. [Google Scholar] [CrossRef]
- Shah, T.V.; Vasava, D.V. A Glimpse of Biodegradable Polymers and Their Biomedical Applications. E-Polymers 2019, 19, 385–410. [Google Scholar] [CrossRef]
- Karande, R.D.; Abitha, V.K.; Rane, A.V.; Mishra, R.K. Preparation of Polylactide From Synthesized Lactic Acid and Preparation of Polylactide From Synthesized Lactic Acid and Effect of Reaction Parameters on Conversion. J. Mater. Sci. Eng. Adv. Technol. 2015, 12, 1–37. [Google Scholar] [CrossRef]
- Hernández-García, E.; Vargas, M.; González-Martínez, C.; Chiralt, A. Biodegradable Antimicrobial Films for Food Packaging: Effect of Antimicrobials on Degradation. Foods 2021, 10, 1256. [Google Scholar] [CrossRef]
- Tan, B.H.; Muiruri, J.K.; Li, Z.; He, C. Recent Progress in Using Stereocomplexation for Enhancement of Thermal and Mechanical Property of Polylactide. ACS Sustain. Chem. Eng. 2016, 4, 5370–5391. [Google Scholar] [CrossRef]
- Rosen, T.; Goldberg, I.; Venditto, V.; Kol, M. Tailor-Made Stereoblock Copolymers of Poly(Lactic Acid) by a Truly Living Polymerization Catalyst. J. Am. Chem. Soc. 2016, 138, 12041–12044. [Google Scholar] [CrossRef]
- Nakajima, H.; Dijkstra, P.; Loos, K. The Recent Developments in Biobased Polymers toward General and Engineering Applications: Polymers That Are Upgraded from Biodegradable Polymers, Analogous to Petroleum-Derived Polymers, and Newly Developed. Polymers 2017, 9, 523. [Google Scholar] [CrossRef] [PubMed]
- Brizga, J.; Hubacek, K.; Feng, K. The Unintended Side Effects of Bioplastics: Carbon, Land, and Water Footprints. One Earth 2020, 3, 45–53. [Google Scholar] [CrossRef]
- A Straightforward Explanation of Biodegradable vs. Compostable vs. Oxo-Degradable Plastics. Available online: https://www.greendotbioplastics.com/biodegradable-vs-compostable-vs-oxo-1479degradable-plastics-a-straightforward-explanation/ (accessed on 10 October 2022).
- Zhao, X.; Cornish, K.; Vodovotz, Y. Narrowing the Gap for Bioplastic Use in Food Packaging: An Update. Environ. Sci. Technol. 2020, 54, 4712–4732. [Google Scholar] [CrossRef]
- Emadian, S.M.; Onay, T.T.; Demirel, B. Biodegradation of Bioplastics in Natural Environments. Waste Manag. 2017, 59, 526–536. [Google Scholar] [CrossRef] [PubMed]
- Imam, S.H.; Gordon, S.H.; Shogren, R.L.; Tosteson, T.R.; Govind, N.S.; Greene, R.V. Degradation of starch-poly(beta-hydroxybutyrate-co-beta-hydroxyvalerate) bioplastic in tropical coastal waters. Appl. Environ. Microbiol. 1999, 65, 431–437. [Google Scholar] [CrossRef]
- Patti, A.; Acierno, D. Towards the Sustainability of the Plastic Industry through Biopolymers: Properties and Potential Applications to the Textiles World. Polymers 2022, 14, 692. [Google Scholar] [CrossRef] [PubMed]
- Corcione, C.E.; Striani, R.; Ferrari, F.; Visconti, P.; Rizzo, D.; Greco, A. An Innovative Method for the Recycling of Waste Carbohydrate-Based Flours. Polymers 2020, 12, 1414. [Google Scholar] [CrossRef] [PubMed]
- Rangappa, S.M.; Siengchin, S.; Parameswaranpillai, J.; Jawaid, M.; Ozbakkaloglu, T. Lignocellulosic Fiber Reinforced Composites: Progress, Performance, Properties, Applications, and Future Perspectives. Polym. Compos. 2022, 43, 645–691. [Google Scholar] [CrossRef]
- Lomelí-Ramírez, M.G.; Kestur, S.G.; Manríquez-González, R.; Iwakiri, S.; De Muniz, G.B.; Flores-Sahagun, T.S. Bio-Composites of Cassava Starch-Green Coconut Fiber: Part II—Structure and Properties. Carbohydr. Polym. 2014, 102, 576–583. [Google Scholar] [CrossRef]
- Cheung, H.Y.; Ho, M.P.; Lau, K.T.; Cardona, F.; Hui, D. Natural Fibre-Reinforced Composites for Bioengineering and Environmental Engineering Applications. Compos. Part B Eng. 2009, 40, 655–663. [Google Scholar] [CrossRef]
- Jawaid, M.; Abdul Khalil, H.P.S. Cellulosic/Synthetic Fibre Reinforced Polymer Hybrid Composites: A Review. Carbohydr. Polym. 2011, 86, 1–18. [Google Scholar] [CrossRef]
- Thakur, V.K.; Thakur, M.K.; Gupta, R.K. Review: Raw Natural Fiber–Based Polymer Composites. Int. J. Polym. Anal. Charact. 2014, 19, 256–271. [Google Scholar] [CrossRef]
- Couret, L.; Irle, M.; Belloncle, C.; Cathala, B. Extraction and Characterization of Cellulose Nanocrystals from Post-Consumer Wood Fiberboard Waste. Cellulose 2017, 24, 2125–2137. [Google Scholar] [CrossRef]
- Hassan, I.F. Nanocellulose for Sustainable Future Applications. In Handbook of Nanomaterials and Nanocomposites for Energy and Environmental Applications; Springer: Cham, Switzerland, 2020; pp. 1–12. ISBN 9783030111557. [Google Scholar]
- Seddiqi, H.; Oliaei, E.; Honarkar, H.; Jin, J.; Geonzon, L.C.; Bacabac, R.G.; Klein-Nulend, J. Cellulose and Its Derivatives: Towards Biomedical Applications; Springer: Dordrecht, The Netherlands, 2021; Volume 28, ISBN 1057002003. [Google Scholar]
- Balart, R.; Garcia-Garcia, D.; Fombuena, V.; Quiles-Carrillo, L.; Arrieta, M.P. Biopolymers from Natural Resources. Polymers 2021, 13, 2532. [Google Scholar] [CrossRef] [PubMed]
- Poletto, M.; Ornaghi Júnior, H.L.; Zattera, A.J. Native Cellulose: Structure, Characterization and Thermal Properties. Materials 2014, 7, 6105–6119. [Google Scholar] [CrossRef] [PubMed]
- Darie, R.N.; Lack, E.; Lang Jr, F.; Sova, M.; Nistor, A.; Spiridon, I. Wastes from Wood Extraction Used in Composite Materials: Behavior after Accelerated Weathering. Int. J. Polym. Anal. Charact. 2014, 19, 453–467. [Google Scholar] [CrossRef]
- Yokoi, H.; Nakase, T.; Goto, K.; Ishida, Y.; Ohtani, H.; Tsuge, S.; Sonoda, T.; Ona, T. Rapid Characterization of Wood Extractives in Wood by Thermal Desorption-Gas Chromatography in the Presence of Tetramethylammonium Acetate. J. Anal. Appl. Pyrolysis 2003, 67, 191–200. [Google Scholar] [CrossRef]
- Ferreira, A.R.V.; Alves, V.D.; Coelhoso, I.M. Polysaccharide-Based Membranes in Food Packaging Applications. Membranes 2016, 6, 22. [Google Scholar] [CrossRef]
- Shaghaleh, H.; Xu, X.; Wang, S. Current Progress in Production of Biopolymeric Materials Based on Cellulose, Cellulose Nanofibers, and Cellulose Derivatives. RSC Adv. 2018, 8, 825–842. [Google Scholar] [CrossRef]
- Li, Y.Y.; Wang, B.; Ma, M.G.; Wang, B. Review of Recent Development on Preparation, Properties, and Applications of Cellulose-Based Functional Materials. Int. J. Polym. Sci. 2018, 2018, 8973643. [Google Scholar] [CrossRef]
- Kalidhasan, S.; Santhana KrishnaKumar, A.; Rajesh, V.; Rajesh, N. Ultrasound-Assisted Preparation and Characterization of Crystalline Cellulose-Ionic Liquid Blend Polymeric Material: A Prelude to the Study of Its Application toward the Effective Adsorption of Chromium. J. Colloid Interface Sci. 2012, 367, 398–408. [Google Scholar] [CrossRef]
- Wan, C.; Li, J. Facile Synthesis of Well-Dispersed Superparamagnetic γ-Fe2O3 Nanoparticles Encapsulated in Three-Dimensional Architectures of Cellulose Aerogels and Their Applications for Cr(VI) Removal from Contaminated Water. ACS Sustain. Chem. Eng. 2015, 3, 2142–2152. [Google Scholar] [CrossRef]
- Teodoro, F.S.; Ramos, S.N.D.C.; Elias, M.M.C.; Mageste, A.B.; Ferreira, G.M.D.; da Silva, L.H.M.; Gil, L.F.; Gurgel, L.V.A. Synthesis and Application of a New Carboxylated Cellulose Derivative. Part I: Removal of Co2+, Cu2+ and Ni2+ from Monocomponent Spiked Aqueous Solution. J. Colloid Interface Sci. 2016, 483, 185–200. [Google Scholar] [CrossRef]
- Dassanayake, R.S.; Acharya, S.; Abidi, N. Biopolymer-Based Materials from Polysaccharides: Properties, Processing, Characterization and Sorption Applications. In Advanced Sorption Process Applications; Intech Open: London, UK, 2018; pp. 1–24. [Google Scholar]
- Akhlaghi, S.P.; Berry, R.C.; Tam, K.C. Surface Modification of Cellulose Nanocrystal with Chitosan Oligosaccharide for Drug Delivery Applications. Cellulose 2013, 20, 1747–1764. [Google Scholar] [CrossRef]
- Brown, E.E.; Hu, D.; Abu Lail, N.; Zhang, X. Potential of Nanocrystalline Cellulose-Fibrin Nanocomposites for Artificial Vascular Graft Applications. Biomacromolecules 2013, 14, 1063–1071. [Google Scholar] [CrossRef]
- Dammak, A.; Moreau, C.; Beury, N.; Schwikal, K.; Winter, H.; Bonnin, E.; Saake, B.; Cathala, B. Elaboration of Multilayered Thin Films Based on Cellulose Nanocrystals and Cationic Xylans: Application to Xylanase Activity Detection. Holzforschung 2013, 67, 579–586. [Google Scholar] [CrossRef]
- Jian, C.; Gong, C.; Wang, S.; Wang, S.; Xie, X.; Wei, Y.; Yuan, J. Multifunctional Comb Copolymer Ethyl Cellulose-g-Poly(ε-Caprolactone)- Rhodamine B/Folate: Synthesis, Characterization and Targeted Bonding Application. Eur. Polym. J. 2014, 55, 235–244. [Google Scholar] [CrossRef]
- Voicu, S.I.; Condruz, R.M.; Mitran, V.; Cimpean, A.; Miculescu, F.; Andronescu, C.; Miculescu, M.; Thakur, V.K. Sericin Covalent Immobilization onto Cellulose Acetate Membrane for Biomedical Applications. ACS Sustain. Chem. Eng. 2016, 4, 1765–1774. [Google Scholar] [CrossRef]
- He, J.X.; Tan, W.L.; Han, Q.M.; Cui, S.Z.; Shao, W.; Sang, F. Fabrication of Silk Fibroin/Cellulose Whiskers–Chitosan Composite Porous Scaffolds by Layer-by-Layer Assembly for Application in Bone Tissue Engineering. J. Mater. Sci. 2016, 51, 4399–4410. [Google Scholar] [CrossRef]
- Liu, S.; Jin, M.; Chen, Y.; Gao, H.; Shi, X.; Cheng, W.; Wang, Y. High Internal Phase Emulsions Stabilised by Supramolecular Cellulose Nanocrystals and Their Application as Cell-Adhesive Macroporous Hydrogel Monoliths. J. Mater. Chem. B 2017, 5, 2671–2678. [Google Scholar] [CrossRef]
- Demitri, C.; Giuri, A.; Raucci, M.G.; Giugliano, D.; Madaghiele, M.; Sannino, A.; Ambrosio, L. Preparation and Characterization of Cellulose-Based Foams via Microwave Curing. Interface Focus 2014, 4, 20130053. [Google Scholar] [CrossRef]
- Fu, J.; Pang, Z.; Yang, J.; Huang, F.; Cai, Y.; Wei, Q. Fabrication of Polyaniline/Carboxymethyl Cellulose/Cellulose Nanofibrous Mats and Their Biosensing Application. Appl. Surf. Sci. 2015, 349, 35–42. [Google Scholar] [CrossRef]
- Xu, D.; Fan, L.; Gao, L.; Xiong, Y.; Wang, Y.; Ye, Q.; Yu, A.; Dai, H.; Yin, Y.; Cai, J.; et al. Micro-Nanostructured Polyaniline Assembled in Cellulose Matrix via Interfacial Polymerization for Applications in Nerve Regeneration. ACS Appl. Mater. Interfaces 2016, 8, 17090–17097. [Google Scholar] [CrossRef] [PubMed]
- Shahbazi, M.; Ahmadi, S.J.; Seif, A.; Rajabzadeh, G. Carboxymethyl Cellulose Film Modification through Surface Photo-Crosslinking and Chemical Crosslinking for Food Packaging Applications. Food Hydrocoll. 2016, 61, 378–389. [Google Scholar] [CrossRef]
- Hu, C.; Bai, X.; Wang, Y.; Jin, W.; Zhang, X.; Hu, S. Inkjet Printing of Nanoporous Gold Electrode Arrays on Cellulose Membranes for High-Sensitive Paper-like Electrochemical Oxygen Sensors Using Ionic Liquid Electrolytes. Anal. Chem. 2012, 84, 3745–3750. [Google Scholar] [CrossRef]
- Han, J.W.; Kim, B.; Li, J.; Meyyappan, M. A Carbon Nanotube Based Ammonia Sensor on Cellulose Paper. RSC Adv. 2014, 4, 549–553. [Google Scholar] [CrossRef]
- Shi, X.; Zheng, Y.; Wang, G.; Lin, Q.; Fan, J. PH- and Electro-Response Characteristics of Bacterial Cellulose Nanofiber/Sodium Alginate Hybrid Hydrogels for Dual Controlled Drug Delivery. RSC Adv. 2014, 4, 47056–47065. [Google Scholar] [CrossRef]
- Giese, M.; Blusch, L.K.; Khan, M.K.; Hamad, W.Y.; MacLachlan, M.J. Responsive Mesoporous Photonic Cellulose Films by Supramolecular Cotemplating. Angew. Chem. 2014, 126, 9026–9030. [Google Scholar] [CrossRef]
- Fox, D.M.; Rodriguez, R.S.; Devilbiss, M.N.; Woodcock, J.; Davis, C.S.; Sinko, R.; Keten, S.; Gilman, J.W. Simultaneously Tailoring Surface Energies and Thermal Stabilities of Cellulose Nanocrystals Using Ion Exchange: Effects on Polymer Composite Properties for Transportation, Infrastructure, and Renewable Energy Applications. ACS Appl. Mater. Interfaces 2016, 8, 27270–27281. [Google Scholar] [CrossRef]
- Ouyang, W.; Sun, J.; Memon, J.; Wang, C.; Geng, J.; Huang, Y. Scalable Preparation of Three-Dimensional Porous Structures of Reduced Graphene Oxide/Cellulose Composites and Their Application in Supercapacitors. Carbon N. Y. 2013, 62, 501–509. [Google Scholar] [CrossRef]
- Bisconti, F.; Giuri, A.; Dominici, L.; Carallo, S.; Quadrivi, E.; Po’, R.; Biagini, P.; Listorti, A.; Corcione, C.E.; Colella, S.; et al. Managing Transparency through Polymer/Perovskite Blending: A Route toward Thermostable and Highly Efficient, Semi-Transparent Solar Cells. Nano Energy 2021, 89, 106406. [Google Scholar] [CrossRef]
- Tanase-Opedal, M.; Espinosa, E.; Rodríguez, A.; Chinga-Carrasco, G. Lignin: A Biopolymer from Forestry Biomass for Biocomposites and 3D Printing. Materials 2019, 12, 3006. [Google Scholar] [CrossRef] [PubMed]
- Tribot, A.; Amer, G.; Abdou Alio, M.; de Baynast, H.; Delattre, C.; Pons, A.; Mathias, J.D.; Callois, J.M.; Vial, C.; Michaud, P.; et al. Wood-Lignin: Supply, Extraction Processes and Use as Bio-Based Material. Eur. Polym. J. 2019, 112, 228–240. [Google Scholar] [CrossRef]
- Mazurchevici, S.N.; Vaideanu, D.; Rapp, D.; Varganici, C.D.; Cărăușu, C.; Boca, M.; Nedelcu, D. Dynamic Mechanical Analysis and Thermal Expansion of Lignin-Based Biopolymers. Polymers 2021, 13, 2953. [Google Scholar] [CrossRef]
- Kun, D.; Pukánszky, B. Polymer/Lignin Blends: Interactions, Properties, Applications. Eur. Polym. J. 2017, 93, 618–641. [Google Scholar] [CrossRef]
- Borrero-l, A.M.; Valencia, C.; Franco, J.M. Lignocellulosic Materials for the Production of Biofuels, Biochemicals and Biomaterials and Applications of of Lignocellulose-Based Polyurethanes: A Review. Polymers 2022, 14, 881. [Google Scholar] [CrossRef] [PubMed]
- Vásquez-Garay, F.; Carrillo-Varela, I.; Vidal, C.; Reyes-Contreras, P.; Faccini, M.; Mendonça, R.T. A Review on the Lignin Biopolymer and Its Integration in the Elaboration of Sustainable Materials. Sustainability 2021, 13, 2697. [Google Scholar] [CrossRef]
- Sharma, S.; Sharma, A.; Mulla, S.I.; Pant, D.; Sharma, T.; Kumar, A. Lignin as Potent Industrial Biopolymer: An Introduction. In Lignin; Springer: Cham, Switzerland, 2020; pp. 1–15. [Google Scholar]
- Aro, T.; Fatehi, P. Production and Application of Lignosulfonates and Sulfonated Lignin. ChemSusChem 2017, 10, 1861–1877. [Google Scholar] [CrossRef]
- Silva, N.; Blumberga, D. Why Biopolymer Packaging Materials Are Better. Environ. Clim. Technol. 2019, 23, 366–384. [Google Scholar] [CrossRef]
- Rajesh Banu, J.; Kavitha, S.; Yukesh Kannah, R.; Poornima Devi, T.; Gunasekaran, M.; Kim, S.H.; Kumar, G. A Review on Biopolymer Production via Lignin Valorization. Bioresour. Technol. 2019, 290, 121790. [Google Scholar] [CrossRef]
- Gilca, I.A.; Popa, V.I.; Crestini, C. Obtaining Lignin Nanoparticles by Sonication. Ultrason. Sonochem 2015, 23, 369–375. [Google Scholar] [CrossRef]
- Li, H.; Deng, Y.; Liang, J.; Dai, Y.; Li, B.; Ren, Y.; Li, C. Direct Preparation of Hollow Nanospheres with Kraft Lignin: A Facile Strategy for Effective Utilization of Biomass Waste. BioResources 2016, 11, 3073–3083. [Google Scholar] [CrossRef]
- Lievonen, M.; Valle-Delgado, J.J.; Mattinen, M.L.; Hult, E.L.; Lintinen, K.; Kostiainen, M.A.; Paananen, A.; Szilvay, G.R.; Setälä, H.; Österberg, M. A Simple Process for Lignin Nanoparticle Preparation. Green Chem. 2016, 18, 1416–1422. [Google Scholar] [CrossRef]
- Bertini, F.; Canetti, M.; Cacciamani, A.; Elegir, G.; Orlandi, M.; Zoia, L. Effect of Ligno-Derivatives on Thermal Properties and Degradation Behavior of Poly(3-Hydroxybutyrate)-Based Biocomposites. Polym. Degrad. Stab. 2012, 97, 1979–1987. [Google Scholar] [CrossRef]
- Mousavioun, P.; George, G.A.; Doherty, W.O.S. Environmental Degradation of Lignin/Poly(Hydroxybutyrate) Blends. Polym. Degrad. Stab. 2012, 97, 1114–1122. [Google Scholar] [CrossRef]
- Kaneko, T.; Thi, T.H.; Shi, D.J.; Akashi, M. Environmentally Degradable, High-Performance Thermoplastics from Phenolic Phytomonomers. Nat. Mater. 2006, 5, 966–970. [Google Scholar] [CrossRef] [PubMed]
- Ouyang, W.; Huang, Y.; Luo, H.; Wang, D. Poly(Lactic Acid) Blended with Cellulolytic Enzyme Lignin: Mechanical and Thermal Properties and Morphology Evaluation. J. Polym. Environ. 2012, 20, 1–9. [Google Scholar] [CrossRef]
- Cateto, C.A.; Barreiro, M.F.; Rodrigues, A.E. Monitoring of Lignin-Based Polyurethane Synthesis by FTIR-ATR. Ind. Crop. Prod. 2008, 27, 168–174. [Google Scholar] [CrossRef]
- Mahmood, N.; Yuan, Z.; Schmidt, J.; Xu, C. Depolymerization of Lignins and Their Applications for the Preparation of Polyols and Rigid Polyurethane Foams: A Review. Renew. Sustain. Energy Rev. 2016, 60, 317–329. [Google Scholar] [CrossRef]
- Lin, Y.; Tanaka, S. Ethanol Fermentation from Biomass Resources: Current State and Prospects. Appl. Microbiol. Biotechnol. 2006, 69, 627–642. [Google Scholar] [CrossRef]
- Balat, M. Production of Bioethanol from Lignocellulosic Materials via the Biochemical Pathway: A Review. Energy Convers. Manag. 2011, 52, 858–875. [Google Scholar] [CrossRef]
- John, R.P.; Nampoothiri, K.M.; Pandey, A. Fermentative Production of Lactic Acid from Biomass: An Overview on Process Developments and Future Perspectives. Appl. Microbiol. Biotechnol. 2007, 74, 524–534. [Google Scholar] [CrossRef] [PubMed]
- Demirbas, A. Potential Applications of Renewable Energy Sources, Biomass Combustion Problems in Boiler Power Systems and Combustion Related Environmental Issues. Prog. Energy Combust. Sci. 2005, 31, 171–192. [Google Scholar] [CrossRef]
- Ten, E.; Vermerris, W.J.P. Functionalized Polymers from Lignocellulosic Biomass: State of the Art. Polymers 2013, 5, 600–642. [Google Scholar] [CrossRef]
- Diacono, M.; Persiani, A.; Testani, E.; Montemurro, F.; Ciaccia, C. Recycling Agricultural Wastes and By-Products in Organic Farming: Biofertilizer Production, Yield Performance and Carbon Footprint Analysis. Sustainability 2019, 11, 3824. [Google Scholar] [CrossRef]
- Motaung, T.E.; Linganiso, L.Z. Critical Review on Agrowaste Cellulose Applications for Biopolymers; Springer: New Delhi, India, 2018; Volume 22, ISBN 1258801892. [Google Scholar]
- Sundarraj, A.A.; Ranganathan, T.V. A Review on Cellulose and Its Utilization from Agro-Industrial Waste. Drug Invent. Today 2018, 10, 89–94. [Google Scholar]
- Urbina, L.; Corcuera, M.Á.; Gabilondo, N.; Eceiza, A.; Retegi, A. A Review of Bacterial Cellulose: Sustainable Production from Agricultural Waste and Applications in Various Fields. Cellulose 2021, 28, 8229–8253. [Google Scholar] [CrossRef]
- El Achaby, M.; Kassab, Z.; Aboulkas, A.; Gaillard, C.; Barakat, A. Reuse of Red Algae Waste for the Production of Cellulose Nanocrystals and Its Application in Polymer Nanocomposites. Int. J. Biol. Macromol. 2018, 106, 681–691. [Google Scholar] [CrossRef] [PubMed]
- Zikeli, F.; Vinciguerra, V.; D’Annibale, A.; Capitani, D.; Romagnoli, M.; Mugnozza, G.S. Preparation of Lignin Nanoparticles from Wood Waste for Wood Surface Treatment. Nanomaterials 2019, 9, 281. [Google Scholar] [CrossRef]
- Parvathy, G.; Sethulekshmi, A.S.; Jayan, J.S.; Raman, A.; Saritha, A. Lignin Based Nano-Composites: Synthesis and Applications. Process Saf. Environ. Prot. 2021, 145, 395–410. [Google Scholar] [CrossRef]
- Verlinden, R.A.J.; Hill, D.J.; Kenward, M.A.; Williams, C.D.; Radecka, I. Bacterial Synthesis of Biodegradable Polyhydroxyalkanoates. J. Appl. Microbiol. 2007, 102, 1437–1449. [Google Scholar] [CrossRef]
- Bohmert, K.; Balbo, I.; Steinbüchel, A.; Tischendorf, G.; Willmitzer, L. Constitutive Expression of the β-Ketothiolase Gene in Transgenic Plants. A Major Obstacle for Obtaining Polyhydroxybutyrate-Producing Plants. Plant Physiol. 2002, 128, 1282–1290. [Google Scholar] [CrossRef] [PubMed]
- Steinbüchel, A.; Lütke-Eversloh, T. Metabolic Engineering and Pathway Construction for Biotechnological Production of Relevant Polyhydroxyalkanoates in Microorganisms. Biochem. Eng. J. 2003, 16, 81–96. [Google Scholar] [CrossRef]
- Shang, L.; Jiang, M.; Chang, H.N. Poly(3-Hydroxybutyrate) Synthesis in Fed-Batch Culture of Ralstonia Eutropha with Phosphate Limitation under Different Glucose Concentrations. Biotechnol. Lett. 2003, 25, 1415–1419. [Google Scholar] [CrossRef] [PubMed]
- Peters, V.; Rehm, B.H.A. In Vivo Monitoring of PHA Granule Formation Using GFP-Labeled PHA Synthases. FEMS Microbiol. Lett. 2005, 248, 93–100. [Google Scholar] [CrossRef]
- Pötter, M.; Steinbüchel, A. Poly(3-Hydroxybutyrate) Granule-Associated Proteins: Impacts on Poly(3-Hydroxybutyrate) Synthesis and Degradation. Biomacromolecules 2005, 6, 552–560. [Google Scholar] [CrossRef]
- Tsuge, T. Metabolic Improvements and Use of Inexpensive Carbon Sources in Microbial Production of Polyhydroxyalkanoates. J. Biosci. Bioeng. 2002, 94, 579–584. [Google Scholar] [CrossRef]
- Stasi, E.; Giuri, A.; Ferrari, F.; Armenise, V.; Colella, S.; Listorti, A.; Rizzo, A.; Ferraris, E.; Corcione, C.E. Biodegradable Carbon-Based Ashes/Maize Starch Composite Films for Agricultural Applications. Polymers 2020, 12, 524. [Google Scholar] [CrossRef]
- Katiyar, V.; Gaur, S.S.; Pal, A.K.; Kumar, A. Properties of Plastics for Packaging Applications. In Polymers for Packaging Applications; CRC Press: Boca Raton, FL, USA, 2014; pp. 3–39. [Google Scholar]
- Bugnicourt, E.; Cinelli, P.; Lazzeri, A.; Alvarez, V.A. Polyhydroxyalkanoate (PHA): Review of Synthesis, Characteristics, Processing and Potential Applications in Packaging. eXPRESS Polym. Lett. 2014, 8, 791–808. [Google Scholar] [CrossRef]
- Sánchez-Safont, E.L.; Aldureid, A.; Lagarón, J.M.; Gámez-Pérez, J.; Cabedo, L. Biocomposites of Different Lignocellulosic Wastes for Sustainable Food Packaging Applications. Compos. Part B Eng. 2018, 145, 215–225. [Google Scholar] [CrossRef]
- Chen, G.Q.; Zhang, G.; Park, S.J.; Lee, S.Y. Industrial Scale Production of Poly(3-Hydroxybutyrate-Co-3-Hydroxyhexanoate). Appl. Microbiol. Biotechnol. 2001, 57, 50–55. [Google Scholar] [CrossRef]
- Godbole, S.; Gote, S.; Latkar, M.; Chakrabarti, T. Preparation and Characterization of Biodegradable Poly-3-Hydroxybutyrate-Starch Blend Films. Bioresour. Technol. 2003, 86, 33–37. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.M. Wood Waste—Carbon Source for Polyhydroxyalkanoates (PHAs) Production. Int. J. For. Wood Sci. 2017, 4, 36–40. [Google Scholar]
- Vaidya, A.N.; Pandey, R.A.; Mudliar, S.; Kumar, M.S.; Chakrabarti, T.; Devotta, S. Production and Recovery of Lactic Acid for Polylactide—An Overview. Crit. Rev. Environ. Sci. Technol. 2005, 35, 429–467. [Google Scholar] [CrossRef]
- Anjum, A.; Zuber, M.; Zia, K.M.; Noreen, A.; Anjum, M.N.; Tabasum, S. Microbial Production of Polyhydroxyalkanoates (PHAs) and Its Copolymers: A Review of Recent Advancements. Int. J. Biol. Macromol. 2016, 89, 161–174. [Google Scholar] [CrossRef] [PubMed]
- Ntaikou, I.; Valencia Peroni, C.; Kourmentza, C.; Ilieva, V.I.; Morelli, A.; Chiellini, E.; Lyberatos, G. Microbial Bio-Based Plastics from Olive-Mill Wastewater: Generation and Properties of Polyhydroxyalkanoates from Mixed Cultures in a Two-Stage Pilot Scale System. J. Biotechnol. 2014, 188, 138–147. [Google Scholar] [CrossRef] [PubMed]
- Hinchliffe, J.D.; Madappura, A.P.; Mohammad, S.; Syed, D.; Roy, I. Biomedical Applications of Bacteria-Derived Polymers. Polymers 2021, 13, 1081. [Google Scholar] [CrossRef]
- Khosravi-Darani, K.; Bucci, D.Z. Application of Poly (Hydroxyalkanoate) in Food Packaging: Improvements by Nanotechnology. Chem. Biochem. Eng. Q. 2015, 29, 275–285. [Google Scholar] [CrossRef]
- Ilyas, R.A.; Zuhri, M.Y.M.; Norrrahim, M.N.F.; Misenan, M.S.M.; Jenol, M.A.; Samsudin, S.A.; Nurazzi, N.M.; Asyraf, M.R.M.; Supian, A.B.M.; Bangar, S.P.; et al. Natural Fiber-Reinforced Polycaprolactone Green and Hybrid Biocomposites for Various Advanced Applications. Polymers 2022, 14, 182. [Google Scholar] [CrossRef]
- Lim, J.; You, M.; Li, J.; Li, Z. Emerging Bone Tissue Engineering via Polyhydroxyalkanoate (PHA)-Based Scaffolds. Mater. Sci. Eng. C 2017, 79, 917–929. [Google Scholar] [CrossRef]
- Ortega, F.; Versino, F.; López, O.V.; García, M.A. Biobased Composites from Agro-Industrial Wastes and by-Products. In Emergent Materials; Springer International Publishing: New York, NY, USA, 2022; Volume 5, pp. 873–921. ISBN 0123456789. [Google Scholar]
- Zheng, Y.; Chen, J.C.; Ma, Y.M.; Chen, G.Q. Engineering Biosynthesis of Polyhydroxyalkanoates (PHA) for Diversity and Cost Reduction. Metab. Eng. 2020, 58, 82–93. [Google Scholar] [CrossRef]
- Singh, A.K.; Srivastava, J.K.; Chandel, A.K.; Sharma, L.; Mallick, N.; Singh, S.P. Biomedical Applications of Microbially Engineered Polyhydroxyalkanoates: An Insight into Recent Advances, Bottlenecks, and Solutions. Appl. Microbiol. Biotechnol. 2019, 103, 2007–2032. [Google Scholar] [CrossRef]
- Lamberti, F.M.; Román-Ramírez, L.A.; Wood, J. Recycling of Bioplastics: Routes and Benefits. J. Polym. Environ. 2020, 28, 2551–2571. [Google Scholar] [CrossRef]
- Rivas, L.F.; Casarin, S.A.; Nepomuceno, N.C.; Alencar, M.I.; Agnelli, J.A.M.; De Medeiros, E.S.; De Oliveira Wanderley Neto, A.; De Oliveira, M.P.; De Medeiros, A.M.; Ferreira Santos, A.S. Reprocessability of PHB in Extrusion: ATR-FTIR, Tensile Tests and Thermal Studies. Polimeros 2017, 27, 122–128. [Google Scholar] [CrossRef]
- Alavi, S.; Thomas, S.; Sandeep, K.P.; Kalarikkal, N.; Varghese, J.; Yaragalla, S. Polymers for Packaging Applications; CRC Press: Boca Raton, FL, USA, 2014. [Google Scholar]
- Chiarini, L.; Mara, L.; Tabacchioni, S. Influence of Growth Supplements on Lactic Acid Production in Whey Ultrafiltrate by Lactobacillus Helveticus. Appl. Microbiol. Biotechnol. 1992, 36, 461–464. [Google Scholar] [CrossRef]
- Boergardts, P.; Krischke, W.; Chmiel, H.; Troesch, W. Development of an Integrated Process for the Production of Lactic Acid from Whey Permeate. Prog. Biotechnol. 1994, 9, 905–908. [Google Scholar]
- Abdel-Rahman, M.A.; Tashiro, Y.; Sonomoto, K. Recent Advances in Lactic Acid Production by Microbial Fermentation Processes. Biotechnol. Adv. 2013, 31, 877–902. [Google Scholar] [CrossRef]
- Cubas-Cano, E.; Venus, J.; González-Fernández, C.; Tomás-Pejó, E. Assessment of Different Bacillus Coagulans Strains for L-Lactic Acid Production from Defined Media and Gardening Hydrolysates: Effect of Lignocellulosic Inhibitors. J. Biotechnol. 2020, 323, 9–16. [Google Scholar] [CrossRef]
- Tan, J.; Abdel-Rahman, M.A.; Sonomoto, K. Biorefinery-Based Lactic Acid Fermentation: Microbial Production of Pure Monomer Product. Adv. Polym. Sci. 2018, 279, 27–66. [Google Scholar] [CrossRef]
- Chai, C.Y.; Tan, I.S.; Foo, H.C.Y.; Lam, M.K.; Tong, K.T.X.; Lee, K.T. Sustainable and Green Pretreatment Strategy of Eucheuma Denticulatum Residues for Third-Generation L-Lactic Acid Production. Bioresour. Technol. 2021, 330, 124930. [Google Scholar] [CrossRef]
- Wang, Y.; Cao, W.; Luo, J.; Qi, B.; Wan, Y. One Step Open Fermentation for Lactic Acid Production from Inedible Starchy Biomass by Thermophilic Bacillus Coagulans IPE22. Bioresour. Technol. 2019, 272, 398–406. [Google Scholar] [CrossRef]
- Sawai, H.; Na, K.; Sasaki, N.; Mimitsuka, T.; Minegishi, S.I.; Henmi, M.; Yamada, K.; Shimizu, Y.S.; Yonehara, T. Membrane-Integrated Fermentation System for Improving the Optical Purity of D-Lactic Acid Produced during Continuous Fermentation. Biosci. Biotechnol. Biochem. 2011, 75, 2326–2332. [Google Scholar] [CrossRef] [PubMed]
- Ikada, Y.; Jamshidi, K.; Tsuji, H.; Hyon, S.H. Stereocomplex Formation between Enantiomeric Poly(Lactides). Macromolecules 1987, 20, 904–906. [Google Scholar] [CrossRef]
- de la Torre, I.; Ladero, M.; Santos, V.E. Production of D-Lactic Acid by Lactobacillus Delbrueckii Ssp. Delbrueckii from Orange Peel Waste: Techno-Economical Assessment of Nitrogen Sources. Appl. Microbiol. Biotechnol. 2018, 102, 10511–10521. [Google Scholar] [CrossRef]
- Ma, K.; Hu, G.; Pan, L.; Wang, Z.; Zhou, Y.; Wang, Y.; Ruan, Z.; He, M. Highly Efficient Production of Optically Pure L-Lactic Acid from Corn Stover Hydrolysate by Thermophilic Bacillus Coagulans. Bioresour. Technol. 2016, 219, 114–122. [Google Scholar] [CrossRef] [PubMed]
- Nwamba, M.C.; Sun, F.; Mukasekuru, M.R.; Song, G.; Harindintwali, J.D.; Boyi, S.A.; Sun, H. Trends and Hassles in the Microbial Production of Lactic Acid from Lignocellulosic Biomass. Environ. Technol. Innov. 2021, 21, 101337. [Google Scholar] [CrossRef]
- Abdel-Rahman, M.A.; Tashiro, Y.; Sonomoto, K. Lactic Acid Production from Lignocellulose-Derived Sugars Using Lactic Acid Bacteria: Overview and Limits. J. Biotechnol. 2011, 156, 286–301. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Cao, W.; Luo, J.; Wan, Y. Exploring the Potential of Lactic Acid Production from Lignocellulosic Hydrolysates with Various Ratios of Hexose versus Pentose by Bacillus Coagulans IPE22. Bioresour. Technol. 2018, 261, 342–349. [Google Scholar] [CrossRef]
- Yadav, A.K.; Chaudhari, A.B.; Kothari, R.M. Bioconversion of Renewable Resources into Lactic Acid: An Industrial View. Crit. Rev. Biotechnol. 2011, 31, 1–19. [Google Scholar] [CrossRef]
- Mussatto, S.I.; Roberto, I.C. Alternatives for Detoxification of Diluted-Acid Lignocellulosic Hydrolyzates for Use in Fermentative Processes: A Review. Bioresour. Technol. 2004, 93, 1–10. [Google Scholar] [CrossRef]
- Vijayakumar, J.; Aravindan, R.; Viruthagiri, T. Recent Trends in the Production, Purification and Application of Lactic Acid. Chem. Biochem. Eng. Q. 2008, 22, 245–264. [Google Scholar]
- Zhou, S.; Causey, T.B.; Hasona, A.; Shanmugam, K.T.; Ingram, L.O. Production of Optically Pure D-Lactic Acid in Mineral Salts Medium by Metabolically Engineered Escherichia Coli W3110. Appl. Environ. Microbiol. 2003, 69, 399–407. [Google Scholar] [CrossRef] [PubMed]
- Baek, S.H.; Kwon, E.Y.; Kim, Y.H.; Hahn, J.S. Metabolic Engineering and Adaptive Evolution for Efficient Production of D-Lactic Acid in Saccharomyces Cerevisiae. Appl. Microbiol. Biotechnol. 2016, 100, 2737–2748. [Google Scholar] [CrossRef] [PubMed]
- Eş, I.; Mousavi Khaneghah, A.; Barba, F.J.; Saraiva, J.A.; Sant’Ana, A.S.; Hashemi, S.M.B. Recent Advancements in Lactic Acid Production—A Review. Food Res. Int. 2018, 107, 763–770. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, A.S.; Thomsen, A.B. Optimization of Wet Oxidation Pretreatment of Wheat Straw. Bioresour. Technol. 1998, 64, 139–151. [Google Scholar] [CrossRef]
- Chandel, A.K.; ES, C.; Rudravaram, R.; Narasu, L.; Rao, V.; Ravindra, P. Economics and Environmental Impact of Bioethanol Production Technologies: An Appraisal. Biotechnol. Mol. Biol. Rev. 2007, 2, 14–32. [Google Scholar]
- Kodali, B.; Pogaku, R. Pretreatment Studies of Ricebran for the Effective Production of Cellulose. Electr. J. Environ. Agric. Food Chem. 2006, 5, 1253–1264. [Google Scholar]
- Mohammed, A.A.B.A.; Omran, A.A.B.; Hasan, Z.; Ilyas, R.A.; Sapuan, S.M. Wheat Biocomposite Extraction, Structure, Properties and Characterization: A Review. Polymers 2021, 13, 3624. [Google Scholar] [CrossRef]
- Shahavi, M.H.; Selakjani, P.P.; Abatari, M.N.; Antov, P.; Savov, V. Novel Biodegradable Poly (Lactic Acid)/Wood Leachate Composites: Investigation of Antibacterial, Mechanical, Morphological, and Thermal Properties. Polymers 2022, 14, 1227. [Google Scholar] [CrossRef]
- Khan, M.Z.R.; Srivastava, S.K.; Gupta, M.K. A State-of-the-Art Review on Particulate Wood Polymer Composites: Processing, Properties and Applications. Polym. Test. 2020, 89, 106721. [Google Scholar] [CrossRef]
- Udayakumar, G.P.; Muthusamy, S.; Selvaganesh, B.; Sivarajasekar, N.; Rambabu, K.; Banat, F.; Sivamani, S.; Sivakumar, N.; Hosseini-Bandegharaei, A.; Show, P.L. Biopolymers and Composites: Properties, Characterization and Their Applications in Food, Medical and Pharmaceutical Industries. J. Environ. Chem. Eng. 2021, 9, 105322. [Google Scholar] [CrossRef]
- Fico, D.; Rizzo, D.; De Carolis, V.; Montagna, F.; Palumbo, E.; Corcione, C.E. Development and Characterization of Sustainable PLA/Olive Wood Waste Composites for Rehabilitation Applications Using Fused Filament Fabrication (FFF). J. Build. Eng. 2022, 56, 104673. [Google Scholar] [CrossRef]
- Greco, A.; Ferrari, F. Thermal Behavior of PLA Plasticized by Commercial and Cardanol-Derived Plasticizers and the Effect on the Mechanical Properties. J. Therm. Anal. Calorim. 2021, 146, 131–141. [Google Scholar] [CrossRef]
- Reddy, M.S.B.; Ponnamma, D.; Choudhary, R.; Sadasivuni, K.K. A Comparative Review of Natural and Synthetic Biopolymer Composite Scaffolds. Polymers 2021, 13, 1105. [Google Scholar] [CrossRef] [PubMed]
- Imre, B.; Pukánszky, B. Compatibilization in Bio-Based and Biodegradable Polymer Blends. Eur. Polym. J. 2013, 49, 1215–1233. [Google Scholar] [CrossRef]
- Shesan, O.J.; Stephen, A.C.; Chioma, A.G.; Neerish, R.; Rotimi, S.E. Improving the Mechanical Properties of Natural Fiber Composites for Structural and Biomedical Applications. In Renewable and Sustainable Composites; IntechOpen: London, UK, 2019; pp. 1–27. [Google Scholar]
- Meng, L.; Xie, F.; Zhang, B.; Wang, D.K.; Yu, L. Natural Biopolymer Alloys with Superior Mechanical Properties. ACS Sustain. Chem. Eng. 2019, 7, 2792–2802. [Google Scholar] [CrossRef]
- George, A.; Sanjay, M.R.; Srisuk, R.; Parameswaranpillai, J.; Siengchin, S. A Comprehensive Review on Chemical Properties and Applications of Biopolymers and Their Composites. Int. J. Biol. Macromol. 2020, 154, 329–338. [Google Scholar] [CrossRef]
- Chan, C.M.; Vandi, L.J.; Pratt, S.; Halley, P.; Richardson, D.; Werker, A.; Laycock, B. Composites of Wood and Biodegradable Thermoplastics: A Review. Polym. Rev. 2018, 58, 444–494. [Google Scholar] [CrossRef]
- Liu, R.; Chen, Y.; Cao, J. Effects of Modifier Type on Properties of in Situ Organo-Montmorillonite Modified Wood Flour/Poly(Lactic Acid) Composites. ACS Appl. Mater. Interfaces 2016, 8, 161–168. [Google Scholar] [CrossRef]
- Febrianto, F.; Yoshioka, M.; Nagai, Y.; Tahir, P.M.D.; Syafii, W.; Shiraishi, N. The Morphological, Mechanical and Physical Properties of Wood Flour-Poly Lactic Acid Composites under Various Filler Types. J. Biol. Sci. 2006, 6, 555–563. [Google Scholar]
- Tao, Y.; Wang, H.; Li, Z.; Li, P.; Shi, S.Q. Development and Application Ofwood Flour-Filled Polylactic Acid Composite Filament for 3d Printing. Materials 2017, 10, 339. [Google Scholar] [CrossRef]
- Le Duigou, A.; Castro, M.; Bevan, R.; Martin, N. 3D Printing of Wood Fibre Biocomposites: From Mechanical to Actuation Functionality. Mater. Des. 2016, 96, 106–114. [Google Scholar] [CrossRef]
- Das, A.K.; Agar, D.A.; Rudolfsson, M.; Larsson, S.H. A Review on Wood Powders in 3D Printing: Processes, Properties and Potential Applications. J. Mater. Res. Technol. 2021, 15, 241–255. [Google Scholar] [CrossRef]
- Xie, G.; Zhang, Y.; Lin, W. Plasticizer Combinations and Performance of Wood Flour-Poly(Lactic Acid) 3D Printing Filaments. BioResources 2017, 12, 6736–6748. [Google Scholar] [CrossRef]
- Zhang, Q.; Ma, R.; Ma, L.; Zhang, L.; Fan, Y.; Wang, Z. Contribution of Lignin in Esterified Lignocellulose Nanofibers (LCNFs) Prepared by Deep Eutectic Solvent Treatment to the Interface Compatibility of LCNF/PLA Composites. Ind. Crop. Prod. 2021, 166, 113460. [Google Scholar] [CrossRef]
- Harussani, M.M.; Sapuan, S.M.; Firdaus, A.H.M.; El-Badry, Y.A.; Hussein, E.E.; El-Bahy, Z.M. Determination of the Tensile Properties and Biodegradability of Cornstarch-Based Biopolymers Plasticized with Sorbitol and Glycerol. Polymers 2021, 13, 3709. [Google Scholar] [CrossRef]
- Vandi, L.J.; Chan, C.M.; Werker, A.; Richardson, D.; Laycock, B.; Pratt, S. Wood-PHA Composites: Mapping Opportunities. Polymers 2018, 10, 751. [Google Scholar] [CrossRef]
- Sliwa, F.; El Bounia, N.E.; Charrier, F.; Marin, G.; Malet, F. Mechanical and Interfacial Properties of Wood and Bio-Based Thermoplastic Composite. Compos. Sci. Technol. 2012, 72, 1733–1740. [Google Scholar] [CrossRef]
- Wu, C.S.; Liao, H.T.; Cai, Y.X. Characterisation, Biodegradability and Application of Palm Fibre-Reinforced Polyhydroxyalkanoate Composites. Polym. Degrad. Stab. 2017, 140, 55–63. [Google Scholar] [CrossRef]
- Peidayesh, H.; Mosnáčková, K.; Špitalský, Z.; Heydari, A.; Šišková, A.O.; Chodák, I. Thermoplastic Starch–Based Composite Reinforced by Conductive Filler Networks: Physical Properties and Electrical Conductivity Changes during Cyclic Deformation. Polymers 2021, 13, 3819. [Google Scholar] [CrossRef]
- Zeng, G.S.; Hu, C.; Zou, S.; Zhang, L.; Sun, G. BP Neural Network Model for Predicting the Mechanical Performance of a Foamed Wood-Fiber Reinforced Thermoplastic Starch Composite. Polym. Compos. 2019, 40, 3923–3928. [Google Scholar] [CrossRef]
- Dorigato, A.; Fredi, G.; Pegoretti, A. Thermo-Mechanical Behavior of Novel Wood Laminae-Thermoplastic Starch Biodegradable Composites with Thermal Energy Storage/Release Capability. Front. Mater. 2019, 6, 76. [Google Scholar] [CrossRef]
- Curvelo, A.A.S.; De Carvalho, A.J.F.; Agnelli, J.A.M. Thermoplastic Starch-Cellulosic Fibers Composites: Preliminary Results. Carbohydr. Polym. 2001, 45, 183–188. [Google Scholar] [CrossRef]
- Miranda, C.S.; Ferreira, M.S.; Magalhães, M.T.; Santos, W.J.; Oliveira, J.C.; Silva, J.B.A.; José, N.M. Mechanical, Thermal and Barrier Properties of Starch-Based Films Plasticized with Glycerol and Lignin and Reinforced with Cellulose Nanocrystals. Mater. Today Proc. 2015, 2, 63–69. [Google Scholar] [CrossRef]
- Da Silva, V.; Batista, K.C.; Zattera, A.J.; Silva, D.; Pezzin, A. Poly(3-Hydroxybutyrate-Co-3-Hydroxyvalerate)/Wood Powder Biocomposites: Thermal and Mechanical Properties and Water Absorption Profile. J. Reinf. Plast. Compos. 2014, 33, 741–748. [Google Scholar] [CrossRef]
- Song, Z.; Xiao, H.; Zhao, Y. Hydrophobic-Modified Nano-Cellulose Fiber/PLA Biodegradable Composites for Lowering Water Vapor Transmission Rate (WVTR) of Paper. Carbohydr. Polym. 2014, 111, 442–448. [Google Scholar] [CrossRef]
- Souza, A.G.; Ferreira, R.R.; Paula, L.C.; Mitra, S.K.; Rosa, D.S. Starch-Based Films Enriched with Nanocellulose-Stabilized Pickering Emulsions Containing Different Essential Oils for Possible Applications in Food Packaging. Food Packag. Shelf Life 2021, 27, 100615. [Google Scholar] [CrossRef]
- Nazrin, A.; Sapuan, S.M.; Zuhri, M.Y.M. Mechanical, Physical and Thermal Properties of Sugar Palm Nanocellulose Reinforced Thermoplastic Starch (TPS)/Poly (Lactic Acid) (PLA) Blend Bionanocomposites. Polymers 2020, 12, 2216. [Google Scholar] [CrossRef]
- Pérez-Pacheco, E.; Canto-Pinto, J.C.; Moo-Huchin, V.M.; Estrada-Mota, I.A.; Estrada-León, R.J.; Chel-Guerrero, L. Thermoplastic Starch (TPS)-Cellulosic Fibers Composites: Mechanical Properties and Water Vapor Barrier: A Review. In Composites from Renewable and Sustainable Materials; InTechOpen: London, UK, 2016. [Google Scholar]
- Aila-Suárez, S.; Palma-Rodríguez, H.M.; Rodríguez-Hernández, A.I.; Hernández-Uribe, J.P.; Bello-Pérez, L.A.; Vargas-Torres, A. Characterization of Films Made with Chayote Tuber and Potato Starches Blending with Cellulose Nanoparticles. Carbohydr. Polym. 2013, 98, 102–107. [Google Scholar] [CrossRef]
- Li, M.; Jia, Y.; Shen, X.; Shen, T.; Tan, Z.; Zhuang, W.; Zhao, G.; Zhu, C.; Ying, H. Investigation into Lignin Modified PBAT/Thermoplastic Starch Composites: Thermal, Mechanical, Rheological and Water Absorption Properties. Ind. Crop. Prod. 2021, 171, 113916. [Google Scholar] [CrossRef]
- Karkhanis, S.S.; Stark, N.M.; Sabo, R.C.; Matuana, L.M. Water Vapor and Oxygen Barrier Properties of Extrusion-Blown Poly(Lactic Acid)/Cellulose Nanocrystals Nanocomposite Films. Compos. Part A Appl. Sci. Manuf. 2018, 114, 204–211. [Google Scholar] [CrossRef]
- Harmaen, A.S.; Khalina, A.; Azowa, I.; Hassan, M.A.; Tarmian, A. Thermal and Biodegradation Properties of Poly (Lactic Acid)/Fertilizer/Oil Palm Fibers Blends Biocomposites. Polym. Compos. 2014, 36, 576–583. [Google Scholar] [CrossRef]
- Ruz-Cruz, M.A.; Herrera-Franco, P.J.; Flores-Johnson, E.A.; Moreno-Chulim, M.V.; Galera-Manzano, L.M.; Valadez-González, A. Thermal and Mechanical Properties of PLA-Based Multiscale Cellulosic Biocomposites. J. Mater. Res. Technol. 2022, 18, 485–495. [Google Scholar] [CrossRef]
- Sanchez-Garcia, M.D.; Gimenez, E.; Lagaron, J.M. Morphology and Barrier Properties of Solvent Cast Composites of Thermoplastic Biopolymers and Purified Cellulose Fibers. Carbohydr. Polym. 2008, 71, 235–244. [Google Scholar] [CrossRef]
- Espino-Pérez, E.; Bras, J.; Ducruet, V.; Guinault, A.; Dufresne, A.; Domenek, S. Influence of Chemical Surface Modification of Cellulose Nanowhiskers on Thermal, Mechanical, and Barrier Properties of Poly(Lactide) Based Bionanocomposites. Eur. Polym. J. 2013, 49, 3144–3154. [Google Scholar] [CrossRef]
- Martínez-Sanz, M.; Villano, M.; Oliveira, C.; Albuquerque, M.G.E.; Majone, M.; Reis, M.; Lopez-Rubio, A.; Lagaron, J.M. Characterization of Polyhydroxyalkanoates Synthesized from Microbial Mixed Cultures and of Their Nanobiocomposites with Bacterial Cellulose Nanowhiskers. N. Biotechnol. 2014, 31, 364–376. [Google Scholar] [CrossRef]
- Torres-Giner, A.S. Development of PHA/Fiber-Based Composites with Antimicrobial Performance for Active Food Packaging Applications; University of Minho: Braga, Portugal, 2017. [Google Scholar]
- Döhler, N.; Wellenreuther, C.; Wolf, A. Market Dynamics of Biodegradable Bio-Based Plastics: Projections and Linkages to European Policies. EFB Bioecon. J. 2022, 2, 100028. [Google Scholar] [CrossRef]
- ICIS Independent Commodity Intelligence Service. Available online: https://www.icis.com/explore/ (accessed on 16 September 2022).
- Nanda, S.; Patra, B.R.; Patel, R.; Bakos, J.; Dalai, A.K. Innovations in Applications and Prospects of Bioplastics and Biopolymers: A Review. Environ. Chem. Lett. 2022, 20, 379–395. [Google Scholar] [CrossRef]
- van den Oever, M.; Molenveld, K.; van der Zee, M.; Bos, H. Biobased and Biodegradable Plastics-–Facts and Figures. Focus on Food Packaging in the Netherlands; Springer: Berlin/Heidelberg, Germany, 2017; Volume 4, ISBN 9783319682556. [Google Scholar]
- Fico, D.; Rizzo, D.; De Carolis, V.; Montagna, F.; Esposito Corcione, C. Sustainable Polymer Composites Manufacturing through 3D Printing Technologies by Using Recycled Polymer and Filler. Polymers 2022, 14, 3756. [Google Scholar] [CrossRef]
- Díez-Pascual, A.M. Synthesis and Applications of Biopolymer Composites. Int. J. Mol. Sci. 2019, 20, 2321. [Google Scholar] [CrossRef]
- Plastic Finder. Available online: https://www.plasticfinder.it/ (accessed on 16 September 2022).
- Dawson, P.L. Packaging. In Poultry Meat Processing; CRC Press: Boca Raton, FL, USA, 2000; pp. 83–106. ISBN 9781420042177. [Google Scholar]
- Wang, Q.; Chen, W.; Zhu, W.; McClements, D.J.; Liu, X.; Liu, F. A Review of Multilayer and Composite Films and Coatings for Active Biodegradable Packaging. npj Sci. Food 2022, 6, 18. [Google Scholar] [CrossRef]
- Provin, A.P.; de Aguiar Dutra, A.R.; de Sousa e Silva Gouveia, I.C.A.; Cubas, e.A.L.V. Circular Economy for Fashion Industry: Use of Waste from the Food Industry for the Production of Biotextiles. Technol. Forecast. Soc. Chang. 2021, 169, 120858. [Google Scholar] [CrossRef]
- Kumar, S.S. Biopolymers in Medical Applications. In Technical Textile; CRC Press: Boca Raton, FL, USA, 2007; pp. 1–9. [Google Scholar]
- Bayón, B.; Berti, I.R.; Gagneten, A.M.; Castro, G.R. Biopolymers from Wastes to High-Value Products in Biomedicine. In Waste to Wealth. Energy, Environment, and Sustainability; Springer Nature: Singapore; Singapore Pte Ltd.: Singapore, 2018; pp. 1–44. ISBN 9789811074318. [Google Scholar]
- Jacob, J.; Haponiuk, J.T.; Thomas, S.; Gopi, S. Biopolymer Based Nanomaterials in Drug Delivery Systems: A Review. Mater. Today Chem. 2018, 9, 43–55. [Google Scholar] [CrossRef]
- Abdelhak, M. A Review: Application of Biopolymers in the Pharmaceutical Formulation. J. Adv. Bio-Pharm. 2019, 1, 15–25. [Google Scholar] [CrossRef]
- Mitura, S.; Sionkowska, A.; Jaiswal, A. Biopolymers for Hydrogels in Cosmetics: Review. J. Mater. Sci. Mater. Med. 2020, 31, 50. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Sharma, S.; Kumar Nadda, A.; Saad Bala Husain, M.; Gupta, A. Biopolymers from Waste Biomass and Its Applications in the Cosmetic Industry: A Review. Mater. Today Proc. 2022, 68, 873–879. [Google Scholar] [CrossRef]
- Barbulova, A.; Colucci, G.; Apone, F. New Trends in Cosmetics: By-Products of Plant Origin and Their Potential Use as Cosmetic Active Ingredients. Cosmetics 2015, 2, 82–92. [Google Scholar] [CrossRef]
- Younes, B. Classification, Characterizatio, and the Production Processes of Biopolymers Used in the Textiles Industry. J. Text. Inst. 2016, 108, 674–682. [Google Scholar] [CrossRef]
- de Silva, C.J.G., Jr.; de Medeiros, A.D.M.; de Amorim, J.D.P.; do Nascimento, H.A.; Converti, A.; Costa, A.F.S.; Sarubbo, L.A. Bacterial Cellulose Biotextiles for the Future of Sustainable Fashion: A Review. Environ. Chem. Lett. 2021, 19, 2967–2980. [Google Scholar] [CrossRef]
- Sivakanthan, S.; Rajendran, S.; Gamage, A.; Madhujith, T.; Mani, S. Antioxidant and Antimicrobial Applications of Biopolymers: A Review. Food Res. Int. 2020, 136, 109327. [Google Scholar] [CrossRef]
- Kumar, L.; Ramakanth, D.; Akhila, K.; Gaikwad, K.K. Edible Films and Coatings for Food Packaging Applications: A Review. Environ. Chem. Lett. 2022, 20, 875–900. [Google Scholar] [CrossRef]
- Khalid, M.Y.; Arif, Z.U. Novel Biopolymer-Based Sustainable Composites for Food Packaging Applications: A Narrative Review. Food Packag. Shelf Life 2022, 33, 100892. [Google Scholar] [CrossRef]
- Das, D.; Panesar, P.S.; Saini, C.S.; Kennedy, J.F. Improvement in Properties of Edible Film through Non-Thermal Treatments and Nanocomposite Materials: A Review. Food Packag. Shelf Life 2022, 32, 100843. [Google Scholar] [CrossRef]
- Feng, M.; Yu, L.; Zhu, P.; Zhou, X.; Liu, H.; Yang, Y.; Zhou, J.; Gao, C.; Bao, X.; Chen, P. Development and Preparation of Active Starch Films Carrying Tea Polyphenol. Carbohydr. Polym. 2018, 196, 162–167. [Google Scholar] [CrossRef]
- Galus, S.; Kibar, E.A.A.; Gniewosz, M.; Kraśniewska, K. Novel Materials in the Preparation of Edible Films and Coatings—A Review. Coatings 2020, 10, 674. [Google Scholar] [CrossRef]
- Perumal, A.B.; Huang, L.; Nambiar, R.B.; He, Y.; Li, X.; Sellamuthu, P.S. Application of Essential Oils in Packaging Films for the Preservation of Fruits and Vegetables: A Review. Food Chem. 2022, 375, 131810. [Google Scholar] [CrossRef] [PubMed]
- Bayram, B.; Ozkan, G.; Kostka, T.; Capanoglu, E.; Esatbeyoglu, T. Valorization and Application of Fruit and Vegetable Wastes and By-Products for Food Packaging Materials. Molecules 2021, 26, 4031. [Google Scholar] [CrossRef] [PubMed]
- Lionetto, F.; Esposito Corcione, C. Recent Applications of Biopolymers Derived from Fish Industry Waste in Food Packaging. Polymers 2021, 13, 2337. [Google Scholar] [CrossRef]
- Elhussieny, A.; Faisal, M.; D’Angelo, G.; Aboulkhair, N.T.; Everitt, N.M.; Fahim, I.S. Valorisation of Shrimp and Rice Straw Waste into Food Packaging Applications. Ain Shams Eng. J. 2020, 11, 1219–1226. [Google Scholar] [CrossRef]
- Baysal, G.; Olcay, H.S.; Keresteci, B.; Özpinar, H. The Antioxidant and Antibacterial Properties of Chitosan Encapsulated with the Bee Pollen and the Apple Cider Vinegar. J. Biomater. Sci. 2022, 33, 995–1011. [Google Scholar] [CrossRef]
- Torres-giner, S.; Figueroa-lopez, K.J.; Melendez-rodriguez, B.; Prieto, C.; Pardo-figuerez, M.; Lagaron, J.M. Emerging Trends in Biopolymers for Food Packaging 1. 1 Introduction to Polymers in Packaging. In Emerging Trends in Biopolymers for Food Packaging; Woodhead Publishing: Sawston, UK, 2021. [Google Scholar]
- Schmidt, J.; Grau, L.; Auer, M.; Maletz, R.; Woidasky, J. Multilayer Packaging in a Circular Economy. Polymers 2022, 14, 1825. [Google Scholar] [CrossRef]
- Omerović, N.; Djisalov, M.; Živojević, K.; Mladenović, M.; Vunduk, J.; Milenković, I.; Knežević, N.; Gadjanski, I.; Vidić, J. Antimicrobial Nanoparticles and Biodegradable Polymer Composites for Active Food Packaging Applications. Compr. Rev. Food Sci. Food Saf. 2021, 20, 2428–2454. [Google Scholar] [CrossRef] [PubMed]
- Nikolic, M.V.; Vasiljevic, Z.Z.; Auger, S.; Vidic, J. Metal Oxide Nanoparticles for Safe Active and Intelligent Food Packaging. Trends Food Sci. Technol. 2021, 116, 655–668. [Google Scholar] [CrossRef]
- Shershneva, E.G. Biodegradable Food Packaging: Benefits and Adverse Effects. In IOP Conference Series: Earth and Environmental Science; IOP Publishing: Bristol, UK, 2022; Volume 988. [Google Scholar]
- Bhat, R.; Abdullah, N.; Din, R.H.; Tay, G.S. Producing Novel Sago Starch Based Food Packaging Films by Incorporating Lignin Isolated from Oil Palm Black Liquor Waste. J. Food Eng. 2013, 119, 707–713. [Google Scholar] [CrossRef]
- Tumwesigye, K.S.; Oliveira, J.C.; Sousa-Gallagher, M.J. New Sustainable Approach to Reduce Cassava Borne Environmental Waste and Develop Biodegradable Materials for Food Packaging Applications. Food Packag. Shelf Life 2016, 7, 8–19. [Google Scholar] [CrossRef]
- Gheorghita, R.; Anchidin-Norocel, L.; Filip, R.; Dimian, M.; Covasa, M. Applications of Biopolymers for Drugs and Probiotics Delivery. Polymers 2021, 13, 2729. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.S.; Rodrigues, L.C.; Fernandes, E.M.; Reis, R.L. Biopolymer Membranes in Tissue Engineering. In Biopolymer Membranes and Films; Elsevier: Amsterdam, The Netherlands, 2020; pp. 141–163. ISBN 9780128181348. [Google Scholar]
- Xu, W.; Wang, X.; Sandler, N.; Willför, S.; Xu, C. Three-Dimensional Printing of Wood-Derived Biopolymers: A Review Focused on Biomedical Applications. ACS Sustain. Chem. Eng. 2018, 6, 5663–5680. [Google Scholar] [CrossRef] [PubMed]
- de Souza, R.F.B.; de Souza, F.C.B.; Bierhalz, A.C.K.; Pires, A.L.R.; Moraes, A.M. Biopolymer-Based Films and Membranes as Wound Dressings Renata. In Biopolymers Membranes and Films; Elsevier: Amsterdam, The Netherlands, 2020; pp. 165–194. ISBN 9780128181348. [Google Scholar]
- Bibire, T.; Yilmaz, O.; Ghiciuc, C.M.; Bibire, N.; Radu, D. Biopolymers for Surgical Applications. Coatings 2022, 12, 211. [Google Scholar] [CrossRef]
- Haleem, N.; Arshad, M.; Shahid, M.; Tahir, M.A. Synthesis of Carboxymethyl Cellulose from Waste of Cotton Ginning Industry. Carbohydr. Polym. 2014, 113, 249–255. [Google Scholar] [CrossRef]
- Zainal, S.H.; Mohd, N.H.; Suhaili, N.; Anuar, F.H.; Lazim, A.M.; Othaman, R. Preparation of Cellulose-Based Hydrogel: A Review. J. Mater. Res. Technol. 2021, 10, 935–952. [Google Scholar] [CrossRef]
- Cui, X.; Lee, J.; Ng, K.R.; Chen, W.N. Food Waste Durian Rind-Derived Cellulose Organohydrogels: Toward Anti-Freezing and Antimicrobial Wound Dressing. ACS Sustain. Chem. Eng. 2021, 9, 1304–1312. [Google Scholar] [CrossRef]
- Amores-Monge, V.; Goyanes, S.; Ribba, L.; Lopretti, M.; Sandoval-Barrantes, M.; Camacho, M.; Corrales-Ureña, Y.; Vega-Baudrit, J.R. Pineapple Agro-Industrial Wastes to Produce Biomedical Applications Post-COVID-19 Pandemic: Biorefinery and the Circular Economy. Preprints 2022, 2022040192. [Google Scholar] [CrossRef]
- Rebelo, R.; Fernandes, M.; Fangueiro, R. Biopolymers in Medical Implants: A Brief Review. Procedia Eng. 2017, 200, 236–243. [Google Scholar] [CrossRef]
- Shirvan, A.R.; Nouri, A. Medical Textiles. In Advances in Functional and Protective Textiles; Butola, B.S., Ed.; Elsevier: Chennai, India, 2020; pp. 291–333. ISBN 9780128202579. [Google Scholar]
- Calcagnile, P.; Cacciatore, G.; Demitri, C.; Montagna, F.; Corcione, C.E. A Feasibility Study of Processing Polydimethylsiloxane-Sodium Carboxymethylcellulose Composites by a Low-Cost Fused Deposition Modeling 3D Printer. Materials 2018, 11, 1578. [Google Scholar] [CrossRef] [PubMed]
- Corcione, C.E.; Gervaso, F.; Scalera, F.; Montagna, F.; Maiullaro, T.; Sannino, A.; Maffezzoli, A. 3D Printing of Hydroxyapatite Polymer-Based Composites for Bone Tissue Engineering. J. Polym. Eng. 2017, 37, 741–746. [Google Scholar] [CrossRef]
- Esposito Corcione, C.; Scalera, F.; Gervaso, F.; Montagna, F.; Sannino, A.; Maffezzoli, A. One-Step Solvent-Free Process for the Fabrication of High Loaded PLA/HA Composite Filament for 3D Printing. J. Therm. Anal. Calorim. 2018, 134, 575–582. [Google Scholar] [CrossRef]
- Esposito Corcione, C.; Gervaso, F.; Scalera, F.; Padmanabhan, S.K.; Madaghiele, M.; Montagna, F.; Sannino, A.; Licciulli, A.; Maffezzoli, A. Highly Loaded Hydroxyapatite Microsphere/PLA Porous Scaffolds Obtained by Fused Deposition Modelling. Ceram. Int. 2019, 45, 2803–2810. [Google Scholar] [CrossRef]
- Veeman, D.; Sai, M.S.; Sureshkumar, P.; Jagadeesha, T.; Natrayan, L.; Ravichandran, M.; Mammo, W.D. Additive Manufacturing of Biopolymers for Tissue Engineering and Regenerative Medicine: An Overview, Potential Applications, Advancements, and Trends. Int. J. Polym. Sci. 2021, 2021, 4907027. [Google Scholar] [CrossRef]
- Bartlett, S. Printing Organs on Demand. Lancet Respir. Med. 2013, 1, 684. [Google Scholar] [CrossRef]
- Gross, B.C.; Erkal, J.L.; Lockwood, S.Y.; Chen, C.; Spence, D.M. Evaluation of 3D Printing and Its Potential Impact on Biotechnology and the Chemical Sciences. Anal. Chem. 2014, 86, 3240–3253. [Google Scholar] [CrossRef]
- Esposito Corcione, C.; Gervaso, F.; Scalera, F.; Montagna, F.; Sannino, A.; Maffezzoli, A. The Feasibility of Printing Polylactic Acid–Nanohydroxyapatite Composites Using a Low-Cost Fused Deposition Modeling 3D Printer. J. Appl. Polym. Sci. 2017, 134, 44656. [Google Scholar] [CrossRef]
- Bonferoni, M.C.; Caramella, C.; Catenacci, L.; Conti, B.; Dorati, R.; Ferrari, F.; Genta, I.; Modena, T.; Perteghella, S.; Rossi, S.; et al. Biomaterials for Soft Tissue Repair and Regeneration: A Focus on Italian Research in the Field. Pharmaceutics 2021, 13, 1341. [Google Scholar] [CrossRef] [PubMed]
- Gregor, A.; Filová, E.; Novák, M.; Kronek, J.; Chlup, H.; Buzgo, M.; Blahnová, V.; Lukášová, V.; Bartoš, M.; Nečas, A.; et al. Designing of PLA Scaffolds for Bone Tissue Replacement Fabricated by Ordinary Commercial 3D Printer. J. Biol. Eng. 2017, 11, 31. [Google Scholar] [CrossRef] [PubMed]
- Bashari, A.; Rouhani Shirvan, A.; Shakeri, M. Cellulose-Based Hydrogels for Personal Care Products. Polym. Adv. Technol. 2018, 29, 2853–2867. [Google Scholar] [CrossRef]
- SANICOT. Available online: https://www.sanicot.com/contatti-assorbenti-cotone/ (accessed on 10 September 2022).
- Intima Luna. Available online: https://www.intimaluna.it/ecoluna/ (accessed on 10 September 2022).
- Lacoste, C.; Lopez-Cuesta, J.M.; Bergeret, A. Development of a Biobased Superabsorbent Polymer from Recycled Cellulose for Diapers Applications. Eur. Polym. J. 2019, 116, 38–44. [Google Scholar] [CrossRef]
- Bhattacharya, P. Aa Review on the Impacts of Microplastic Beads Used in Cosmetics. Acta Biomed. Sci. 2016, 3, 47–52. [Google Scholar]
- Jang, J.H.; So, B.R.; Yeo, H.J.; Kang, H.J.; Kim, M.J.; Lee, J.J.; Jung, S.K.; Jung, Y.H. Preparation of Cellulose Microfibril (CMF) from Gelidium Amansii and Feasibility of CMF as a Cosmetic Ingredient. Carbohydr. Polym. 2021, 257, 117569. [Google Scholar] [CrossRef]
- Meyabadi, T.F.; Dadashian, F.; Sadeghi, G.M.M.; Asl, H.E.Z. Spherical Cellulose Nanoparticles Preparation from Waste Cotton Using a Green Method. Powder Technol. 2014, 261, 232–240. [Google Scholar] [CrossRef]
- Bongao, H.; Gabatino, R.; Arias, C.F.H.; Magdaluyo, E.R., Jr. Micro/Nanocellulose from Waste Pili (Canarium Ovatum) Pulp as a Potential Anti-Ageing Ingredient for Cosmetic Formulations. Mater. Today Proc. 2020, 22, 275–280. [Google Scholar] [CrossRef]
- ANOMERA. Available online: https://www.anomera.ca/ (accessed on 10 September 2022).
- Gabryś, T.; Fryczkowska, B.; Grzybowska-Pietras, J.; Biniaś, D. Modification and Properties of Cellulose Nonwoven Fabric—Multifunctional Mulching Material for Agricultural Applications. Materials 2021, 14, 4335. [Google Scholar] [CrossRef]
- Tesfaye, T.; Ayele, M.; Ferede, E.; Gibril, M.; Kong, F.; Sithole, B. A Techno_economic Feasibility of a Process for Extraction of Starch from Waste Avocado Seeds. Clean Technol. Environ. Policy 2020, 23, 581–595. [Google Scholar] [CrossRef]
- Tesfaye, T.; Gibril, M.; Sithole, B.; Ramjugernath, D.; Chavan, R.; Chunilall, V. Valorisation of Avocado Seeds: Extraction and Characterisation of Starch for Textile Applications. Clean Technol. Environ. Policy 2018, 20, 2135–2154. [Google Scholar] [CrossRef]
- Cano-Vicent, A.; Tambuwala, M.M.; Hassan, S.S.; Barh, D.; Aljabali, A.A.A.; Birkett, M.; Arjunan, A.; Serrano-Aroca, Á. Fused Deposition Modelling: Current Status, Methodology, Applications and Future Prospects. Addit. Manuf. 2021, 47, 102378. [Google Scholar] [CrossRef]
- Biswas, M.C. Fused Deposition Modeling 3D Printing Technology in Textile and Fashion Industry: Materials and Innovation. Mod. Concepts Mater. Sci. 2019, 2, 1–5. [Google Scholar] [CrossRef]
- Loh, G.H.; Sotayo, A.; Pei, E. Development and Testing of Material Extrusion Additive Manufactured Polymer–Textile Composites. Fash. Text. 2021, 8, 2. [Google Scholar] [CrossRef]
- Jiang, T.; Duan, Q.; Zhu, J.; Liu, H.; Yu, L. Starch-Based Biodegradable Materials: Challenges and Opportunities. Adv. Ind. Eng. Polym. Res. 2018, 39, 245–258. [Google Scholar] [CrossRef]
- Kenawy, E.R.; Kamoun, E.A.; Mohy Eldin, M.S.; El-Meligy, M.A. Physically Crosslinked Poly(Vinyl Alcohol)-Hydroxyethyl Starch Blend Hydrogel Membranes: Synthesis and Characterization for Biomedical Applications. Arab. J. Chem. 2014, 7, 372–380. [Google Scholar] [CrossRef]
- Encalada, K.; Aldás, M.B.; Proaño, E.; Valle, V. An Overview of Starch-Based Biopolymers and Their Biodegradability; Ciencia e Ingeniería, Universidad de los Andes: Mérida, Venezuela, 2018; Volume 39. [Google Scholar]
- Solihat, N.N.; Sari, F.P.; Falah, F.; Ismayati, M.; Lubis, M.A.R.; Fatriasari, W.; Santoso, E.B.; Syafii, W. Lignin as an Active Biomaterial: A Review. J. Sylva Lestari 2021, 9, 1. [Google Scholar] [CrossRef]
- Ilyas, R.A.; Sapuan, S.M.; Harussani, M.M.; Hakimi, M.Y.A.Y.; Haziq, M.M.; Atikah, M.S.N.; Asyraf, M.R.M.; Ishak, M.R.; Razman, M.R.; Nurazzi, N.M.; et al. Polylactic Acid (Pla) Biocomposite: Processing, Additive Manufacturing and Advanced Applications. Polymers 2021, 13, 1326. [Google Scholar] [CrossRef]
- Ilyas, R.A.; Sapuan, S.M.; Kadier, A.; Kalil, M.S.; Ibrahim, R.; Atikah, M.S.N.; Nurazzi, N.M.; Nazrin, A.; Lee, C.H.; Faiz Norrrahim, M.N.; et al. Properties and Characterization of PLA, PHA, and Other Types of Biopolymer Composites; Elsevier Inc.: Amsterdam, The Netherlands, 2020; ISBN 9780128196618. [Google Scholar]
- Rodriguez-Contreras, A. Recent Advances in the Use of Polyhydroyalkanoates in Biomedicine. Bioengineering 2019, 6, 82. [Google Scholar] [CrossRef]


| Types | Chemical Group | Example | References |
|---|---|---|---|
| Biomass-based polymers | Polysaccharides Proteins and lipids |
| [16,17,18] |
| Polymers derived via microbial fermentation | Polyhydroxyalkanoates (PHA) |
| [19,20,21] |
| Agro-resource monomers are used to chemically manufacture polymers | Poly(hydroxyacid) |
| [22,23,24] |
| Bioplastic | Biodegradability | Ref. | ||||
|---|---|---|---|---|---|---|
| Environment | Condition | Biodegradability (%) | Biodegradability/ Degradability Method | Testing Period (Day) | ||
| Starch-based | Compost (starch, thermoplastic) | 58 °C | 73.1 | CO2 produced | 56 | [11,27,28,29] |
| Soil (wheat, starch-derived plastic) | 20 °C, 60% RH | 14.2 | CO2 produced | 110 | ||
| Marine (neat starch) | 26 °C | 100 | Weight loss | 50 | ||
| Cellulose-based | Compost (cellulose acetate) | 53 °C | 100 | CO2 produced | 18 | [11,27,28] |
| Soil (bacterial and vegetable cellulose) | 25 °C | 100 | Weight loss | 180 | ||
| Simulated marine environment (neat cellulose) | Room temperature | 75 | Oxygen consumed | 150 | ||
| PLA | Compost | 58 °C, 60% RH | 60–70 | CO2 produced | 30 | [11,12,27,30] |
| Soil | 10–25 °C | 0 | CO2 produced | 120 | ||
| Simulated marine environment | 25 °C | 3–4 | CO2 produced | 180 | ||
| PHB | Compost | 55 °C, 70% RH | 80 | CO2 produced | 28 | [11,27,28] |
| Soil | 20 °C, 60% RH | 48.5 | CO2 produced | 280 | ||
| Simulated marine environment | 25 °C | 38–45 | CO2 produced | 180 | ||
| Young’s Modulus | Amorphous Plasticized PLA (MPa) | Semicrystalline Plasticized PLA (MPa) | |
|---|---|---|---|
| Plasticizer | |||
| PEG 400 | 581 | 962 | |
| Cardanol | 691 | 1156 | |
| Epoxidized cardanol | 353 | 961 | |
| Bio-Based Polymers | Density (g/cm3) | Tensile Strength (MPa) | Young’s Modulus (GPa) | Elongation at Break (%) | Tg (°C) | Tm (°C) | Ref. |
|---|---|---|---|---|---|---|---|
| Wood–starch | 1.29–1.41 | 14–36 | 0.7–4.8 | 1.1–2.9 | −33–−26 | 83.1–130.3 | [161,169,176,177,180,181,182,183,184] |
| Wood–PLA | 1.26–1.41 | 30–71 | 1.2–8.9 | 1.0–3.1 | 52.0–60.8 | 143.4–169.0 | [12,154,161,162,163,181,185,186,187,188,189] |
| Wood–PHA | 1.23 | 13–65 | 0.4–6.1 | 0.5–7 | −2.2–−6.0 | 56.8–158.7 | [161,170,172,190,191] |
| Bio-based Polymers and Biocomposites | Properties | Applications | Source | Ref. |
|---|---|---|---|---|
| Starch-based | Low toxicity, biocompatibility and equivalent mechanical and degrading qualities. | Packaging applications, wound-healing materials, drug delivery system, agricultural foils, textiles, automobiles and transportation, construction and building materials, etc. | Plants | [215,261,265,266,267] |
| Cellulose-based | Microbial characteristics, exceptionally crystalline, chemically and thermally stable. | Packaging applications, edible films and coatings, hydrogels for personal care products, medical device, biosensors, drug delivery system, electronic and energy devices, cosmetics, textiles and nonwovens etc. | Plants | [45,250,256,259] |
| Lignin-based | Biodegradability, hydrophilicity, low-cost, nontoxicity, thermal and mechanical stability | Food packaging, applications in biocide systems, adhesive resins and foams, filling materials, construction and building materials, biomedical applications | Plants | [154,205,227,231,268] |
| PLA-based | Higher mechanical strength, degradation in nature either through reduction or by M, excellent barrier and permeability properties. | Packaging applications, 3D printing, biomedical applications, scaffolds and medical implants, textiles and nonwoven fabrics, agricultural applications, etc. | Fermentation/conventional chemistry followed by polymerization | [12,226,238,264,269] |
| PHA-based | Biocompatible, biodegradable, considerable elastomeric with excellent elongation at break. | Food packaging and coatings, scaffolds and medical implants, textile industry, etc. | Bacterial fermentation | [205,207,270,271] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ferrari, F.; Striani, R.; Fico, D.; Alam, M.M.; Greco, A.; Esposito Corcione, C. An Overview on Wood Waste Valorization as Biopolymers and Biocomposites: Definition, Classification, Production, Properties and Applications. Polymers 2022, 14, 5519. https://doi.org/10.3390/polym14245519
Ferrari F, Striani R, Fico D, Alam MM, Greco A, Esposito Corcione C. An Overview on Wood Waste Valorization as Biopolymers and Biocomposites: Definition, Classification, Production, Properties and Applications. Polymers. 2022; 14(24):5519. https://doi.org/10.3390/polym14245519
Chicago/Turabian StyleFerrari, Francesca, Raffaella Striani, Daniela Fico, Mohammad Mahbubul Alam, Antonio Greco, and Carola Esposito Corcione. 2022. "An Overview on Wood Waste Valorization as Biopolymers and Biocomposites: Definition, Classification, Production, Properties and Applications" Polymers 14, no. 24: 5519. https://doi.org/10.3390/polym14245519
APA StyleFerrari, F., Striani, R., Fico, D., Alam, M. M., Greco, A., & Esposito Corcione, C. (2022). An Overview on Wood Waste Valorization as Biopolymers and Biocomposites: Definition, Classification, Production, Properties and Applications. Polymers, 14(24), 5519. https://doi.org/10.3390/polym14245519










