Simultaneous Recovery of Precious and Heavy Metal Ions from Waste Electrical and Electronic Equipment (WEEE) Using Polymer Films Containing Cyphos IL 101
Abstract
1. Introduction
2. Materials and Methods
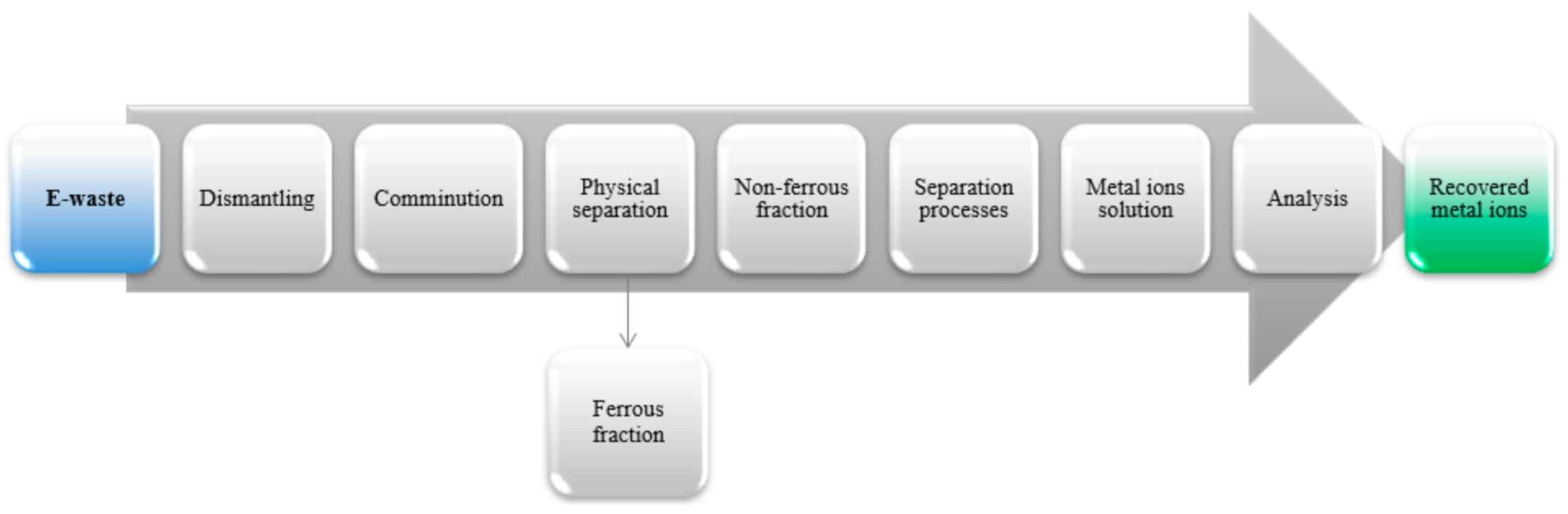
2.1. E-Waste
2.2. Reagents
2.3. Preparation of Polymer Films Containing Cyphos IL 101
2.4. Preparation of Polymer Films Containing Cyphos IL 101
- The computer pins were qualitatively analyzed for the presence of metals. This study was performed using a Panalytical XRF Minipal X-ray fluorescence spectrometer.
- Computer pin samples (0.5 g) were flooded with 50 mL of concentrated (65%) nitric(V) acid and allowed to stand for 24 h. After some time, gold was released on the surface of the solution (Figure 4a). At the bottom, however, a certain amount of undissolved precipitate remained. The gold flakes were separated from the solution through the decantation of the solution from over the undissolved precipitate, and then this solution was centrifugated. In this way, 0.36 mg of gold was obtained.
- The undissolved precipitate (Figure 4b) mentioned in point 2, together with the small amount of remaining solution, was separated by filtration.
- To the filter containing precipitate mentioned in point 3, 50 mL of aqua regia was added (Figure 4c). After some time, the filter with the precipitate dissolved completely.
2.5. Recovery of Metal Ions on Polymer Films Containing Cyphos IL 101
- A polymer film (Figure 5) was placed in 45 mL of nitric acid(V) solution (A) for 24 h to bind the metal ions present in the solution on its surface. Samples of the solution were taken at set intervals.
- At the same time, a polymer film was put into 38 mL of aqua regia solution (B) for 24 h to bind the metal ions present in the solution on its surface. Samples of the solution were taken at set intervals.
- After removal from the above solutions (Figure 6a,b), the polymer films were air-dried and placed in 10 mL of 5 M nitric acid for 24 h to desorb the metal ions bound on the film surface. This step was intended to allow the films to be used in subsequent sorption and desorption cycles.
3. Results and Discussion
3.1. Results of Leaching of E-Waste
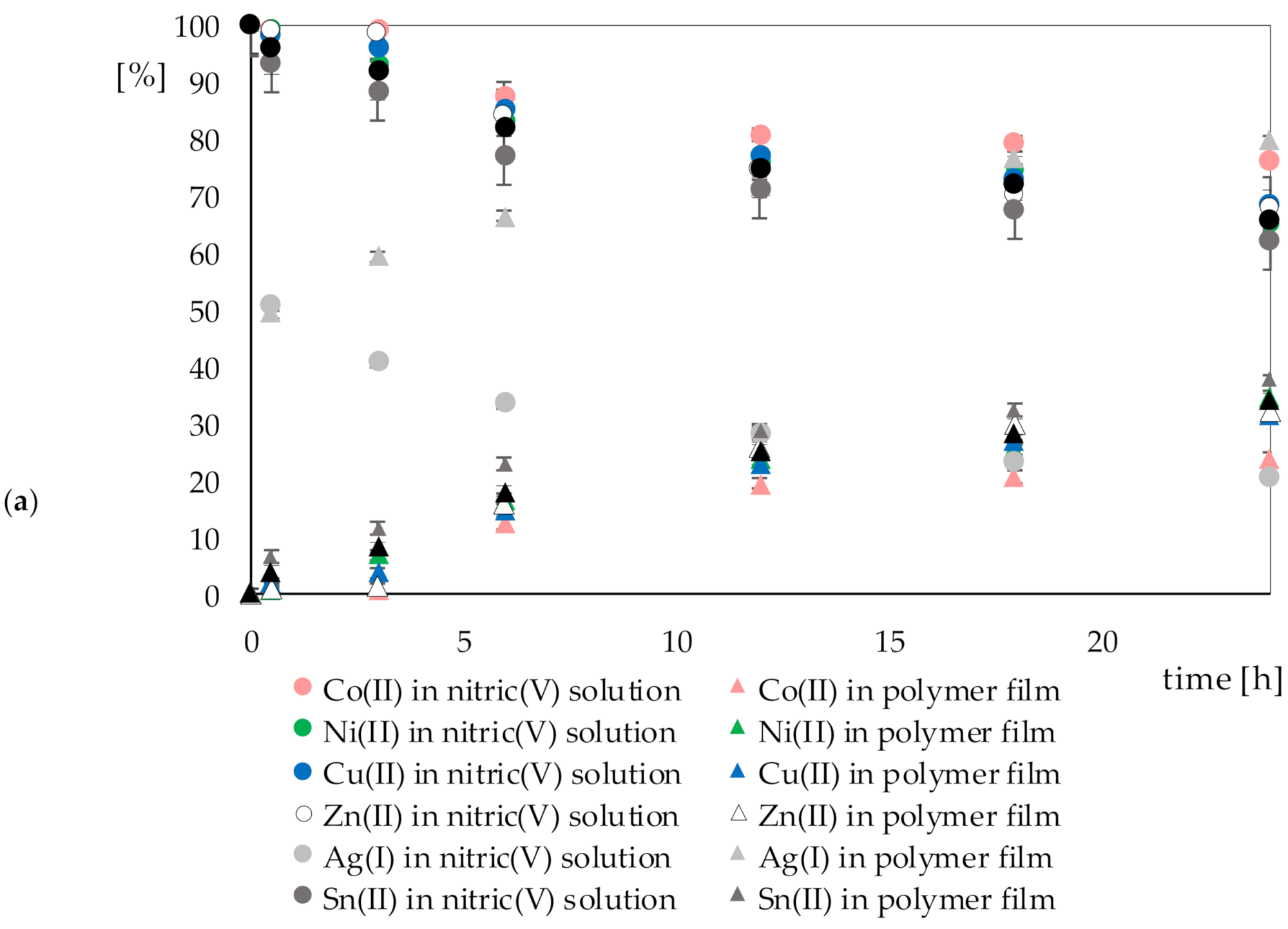
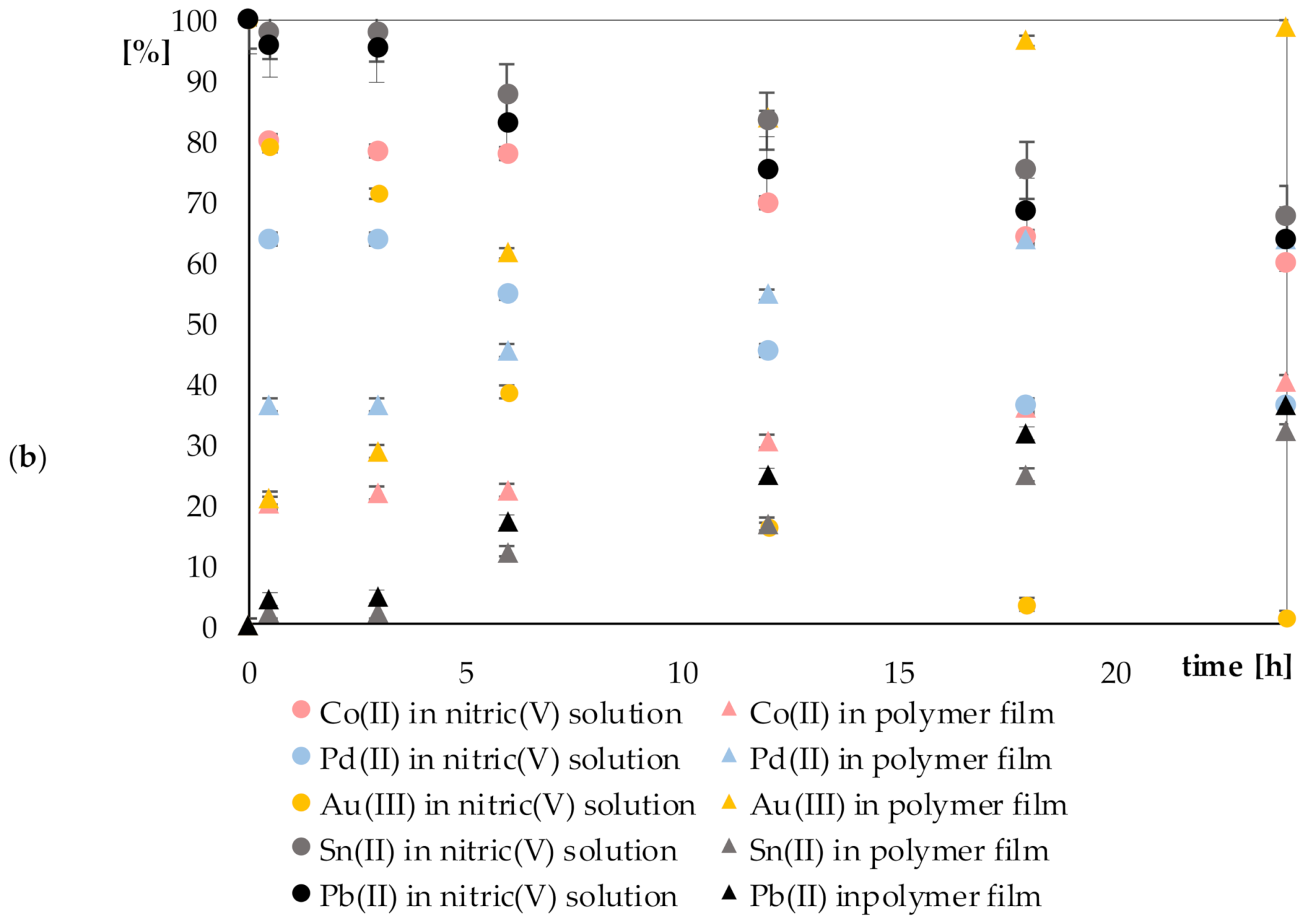
3.2. Results of Recovery of Metal Ions on Polymer Films Containing Cyphos IL 101
3.3. Evaluation of the Long-Term Suitability of Polymer Films Containing Cyphos IL101
4. Summary
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zeng, X.; Gong, R.; Chen, W.-Q.; Li, J. Uncovering the Recycling Potential of “New” WEEE in China. Environ. Sci. Technol. 2016, 50, 1347–1358. [Google Scholar] [CrossRef]
- Hamdan, S.; Saidan, M.N. Estimation of E-waste Generation, Residential Behavior, and Disposal Practices from Major Governorates in Jordan. Environ. Manag. 2020, 66, 884–898. [Google Scholar] [CrossRef] [PubMed]
- Shaikh, S.; Thomas, K.; Zuhair, S. An exploratory study of e-waste creation and disposal: Upstream considerations. Resour. Conserv. Recycl. 2020, 155, 104662. [Google Scholar] [CrossRef]
- Directive 2002/96/EC of the European Parliament and of the Council of 27 January 2003 on Waste Electrical and Electronic Equipment (WEEE). Available online: https://eur-lex.europa.eu/legal-content/EN/TXT/?uri=celex%3A32002L0096 (accessed on 31 February 2021).
- Sethurajan, M.; van Hullebusch, E.D.; Fontana, D.; Akcil, A.; Deveci, H.; Batinic, B.; Leal, J.P.; Gasche, T.A.; Kucuker, M.A.; Kuchta, K.; et al. Recent advances on hydrometallurgical recovery of critical and precious elements from end of life electronic wastes—A review. Crit. Rev. Environ. Sci. Technol. 2019, 49, 212–275. [Google Scholar] [CrossRef]
- Akcil, A.; Agcasulu, I.; Swain, B. Valorization of waste LCD and recovery of critical raw material for circular economy: A review. Resour. Conserv. Recycl. 2019, 149, 622–637. [Google Scholar] [CrossRef]
- Anand, A.; Jha, K.J.; Kumar, V.; Sahu, R. Recycling of precious metal gold from waste electrical and electronic equipments (WEEE): A review. In Proceedings of the XIII International Seminar on Mineral Processing Technology, Bhubaneswar, India, 10–12 December 2013; pp. 916–923. [Google Scholar]
- Li, W.; Achal, V. Environmental and health impacts due to e-waste disposal in China—A review. Sci. Total. Environ. 2020, 737, 139745. [Google Scholar] [CrossRef] [PubMed]
- Sun, Z.; Cao, H.; Xiao, Y.; Sietsma, J.; Jin, W.; Agterhuis, H.; Yang, Y. Toward Sustainability for Recovery of Critical Metals from Electronic Waste: The Hydrochemistry Processes. ACS Sustain. Chem. Eng. 2017, 5, 21–40. [Google Scholar] [CrossRef]
- Hagelüken, C.; Corti, C.W. Recycling of gold from electronics: Cost-effective use through ‘Design for Recycling’. Gold Bull. 2010, 43, 209–220. [Google Scholar] [CrossRef]
- Pasiecznik, I.; Banaszkiewicz, K.; Syska, Ł. Local community e-waste awareness and behavior. Polish case study. Environ. Prot. Eng. 2017, 43, 287–289. [Google Scholar] [CrossRef]
- Kornacki, W. Recykling płytek drukowanych. In II Krajowa Konferencja Naukowo-Techniczna “Ekologia w Elektronice”; Przemysłowy Instytut Elektroniki: Warsaw, Poland, 2002. [Google Scholar]
- Ibanescu, D.; Gavrilescu, D.C.; Teodosiu, C.; Fiore, S. Assessment of the waste electrical and electronic equipment management systems profile and sustainability in developed and developing European Union countries. Waste Manag. 2018, 73, 39–53. [Google Scholar] [CrossRef]
- Fizaine, F. The economics of recycling rate: New insights from waste electrical and electronic equipment. Resour. Policy 2020, 67, 101675. [Google Scholar] [CrossRef]
- Bahubalendruni, M.V.A.R.; Varupala, V.P. Disassembly Sequence Planning for Safe Disposal of End-of-Life Waste Electric and Electronic Equipment. Natl. Acad. Sci. Lett. 2020, 1–5. [Google Scholar] [CrossRef]
- Tanskanen, P. Management and recycling of electronic waste. Acta Mater. 2013, 61, 1001–1011. [Google Scholar] [CrossRef]
- Doidge, E.D.; Carson, I.; Tasker, P.A.; Ellis, R.J.; Morrison, C.A.; Love, J.B. A Simple Primary Amide for the Selective Recovery of Gold from Secondary Resources. Angew. Chem. Int. Ed. 2016, 55, 12436–12439. [Google Scholar] [CrossRef]
- Kaya, M. Recovery of metals and nonmetals from electronic waste by physical and chemical recycling processes. Waste Manag. 2016, 57, 64–90. [Google Scholar] [CrossRef] [PubMed]
- Rao, M.D.; Singh, K.K.; Morrison, C.A.; Love, J.B. Challenges and opportunities in the recovery of gold from electronic waste. RSC Adv. 2020, 10, 4300–4309. [Google Scholar] [CrossRef]
- Assadian, M.; Idris, M.H.; Shahri, S.M.G.; Gholampour, B. Gold Recovery from WEEE by Chlorine System. Appl. Mech. Mater. 2013, 330, 123–125. [Google Scholar] [CrossRef]
- Rohit, I.; Dhanunjaya, M.; Arunabh, R.; Himanshu, M.; Verma, R.; Singh, K.K. Potential of polymer inclusion membrane process for selective recovery of metal values from waste printed circuit boards: A review. J. Clean. Prod. 2020, 265, 121621. [Google Scholar] [CrossRef]
- Kavitha, N.; Palanivelu, K. Recovery of copper(II) through polymer inclusion membrane with di(2-ethylhexyl) phosphoric acid as carrier from e-waste. J. Membr. Sci. 2012, 415-416, 663–669. [Google Scholar] [CrossRef]
- Kubota, F.; Kono, R.; Yoshida, W.; Sharaf, M.; Kolev, S.D.; Goto, M. Recovery of gold ions from discarded mobile phone leachate by solvent extraction and polymer inclusion membrane (PIM) based separation using an amic acid extractant. Sep. Purif. Technol. 2019, 214, 156–161. [Google Scholar] [CrossRef]
- Miguel, E.R.D.S.; Garduño-García, A.V.; Aguilar, J.C.; de Gyves, J. Gold(III) Transport through Polymer Inclusion Membranes: Efficiency Factors and Pertraction Mechanism Using Kelex 100 as Carrier. Ind. Eng. Chem. Res. 2007, 46, 2861–2869. [Google Scholar] [CrossRef]
- Bonggotgetsakul, Y.Y.N.; Cattrall, R.W.; Kolev, S.D. Recovery of gold from aqua regia digested electronic scrap using a poly(vinylidene fluoride-co-hexafluoropropene) (PVDF-HFP) based polymer inclusion membrane (PIM) containing Cyphos® IL 104. J. Membr. Sci. 2016, 514, 274–281. [Google Scholar] [CrossRef]
- Nowik-Zajac, A.; Zawierucha, I.; Kozlowski, C.; Nowik-Zając, A. Selective removal of silver(I) using polymer inclusion membranes containing calixpyrroles. RSC Adv. 2019, 9, 31122–31132. [Google Scholar] [CrossRef]
- Bahrami, S.; Yaftian, M.R.; Najvak, P.; Dolatyari, L.; Shayani-Jam, H.; Kolev, S.D. PVDF-HFP based polymer inclusion membranes containing Cyphos® IL 101 and Aliquat® 336 for the removal of Cr(VI) from sulfate solutions. Sep. Purif. Technol. 2020, 250, 117251. [Google Scholar] [CrossRef]
- Campos, K.; Vincent, T.; Bunio, P.; Trochimczuk, A.; Guibal, E. Gold Recovery from HCl Solutions using Cyphos IL-101 (a Quaternary Phosphonium Ionic Liquid) Immobilized in Biopolymer Capsules. Solvent Extr. Ion Exch. 2008, 26, 570–601. [Google Scholar] [CrossRef]
- Vincent, T.; Parodi, A.; Guibal, E. Pt recovery using Cyphos IL-101 immobilized in biopolymer capsules. Sep. Purif. Technol. 2008, 62, 470–479. [Google Scholar] [CrossRef]
- Regel-Rosocka, M.; Rzelewska, M.; Baczynska, M.; Janus, M.; Wisniewski, M. Removal of palladium(II) from aqueous chloride solutions with Cyphos phosphoniumionic liquids as metal ion carriers for liquid-liquid extraction and transport across polymer inclusion membranes. Physicochem. Probl. Miner. Process. 2015, 51, 621–631. [Google Scholar] [CrossRef]
- Pospiech, B. Application of Phosphonium Ionic Liquids as Ion Carriers in Polymer Inclusion Membranes (PIMs) for Separation of Cadmium(II) and Copper(II) from Aqueous Solutions. J. Solut. Chem. 2015, 44, 2431–2447. [Google Scholar] [CrossRef]
- Nowik-Zajac, A.; Zawierucha, I.; Kozlowski, C. Selective Transport of Ag(I) through a Polymer Inclusion Membrane Containing a Calix[4]pyrrole Derivative from Nitrate Aqueous Solutions. Int. J. Mol. Sci. 2020, 21, 5348. [Google Scholar] [CrossRef]
- Radzyminska-Lenarcik, E.; Ulewicz, M.; Pyszka, I. Application of Polymer Inclusion Membranes Doped with Alkylimidazole to Separation of Silver and Zinc Ions from Model Solutions and after Battery Leaching. Materials 2020, 13, 3103. [Google Scholar] [CrossRef]
- Yaftian, M.R.; Almeida, M.I.G.; Cattrall, R.W.; Kolev, S.D. Selective extraction of vanadium(V) from sulfate solutions into a polymer inclusion membrane composed of poly(vinylidenefluoride-co-hexafluoropropylene) and Cyphos® IL 101. J. Membr. Sci. 2018, 545, 57–65. [Google Scholar] [CrossRef]
- Burczyk, B. Green Chemistry Outline; Publishing House of the Wrocław; University of Technology: Wroclaw, Poland, 2014. [Google Scholar]
- Earle, M.J.; Esperança, J.M.; Gilea, M.A.; Lopes, J.N.C.; Rebelo, L.P.; Magee, J.W.; Seddon, K.R.; Widegren, J.A. The distillation and volatility of ionic liquids. Nat. Cell Biol. 2006, 439, 831–834. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, R. Water: From Clusters to the Bulk. Angew. Chem. Int. Ed. 2001, 40, 1808–1827. [Google Scholar] [CrossRef]
- Cieszyńska, A.; Regel-Rosocka, M.; Wiśniewski, M. Extraction of Palladium(II) Ions from Chloride Solutions with Phosphonium Ionic Liquid Cyphos® IL101. Pol. J. Chem. Technol. 2007, 9, 99–101. [Google Scholar] [CrossRef]
- Negrea, A.; Lupa, L.; Ciopec, M.; Negrea, P. Characterization of Strontium Adsorption from Aqueous Solutions Using Inorganic Materials Impregnated with Ionic Liquid. Int. J. Chem. Eng. Appl. 2013, 4, 326–331. [Google Scholar] [CrossRef]
- Navarro, R.; Saucedo, I.; Gonzalez, C.; Guibal, E. Amberlite XAD-7 impregnated with Cyphos IL-101 (tetraalkylphosphonium ionic liquid) for Pd(II) recovery from HCl solutions. Chem. Eng. J. 2012, 185, 226–235. [Google Scholar] [CrossRef]
- Maj, R. Analiza Niemieckiego System Recyklingowego Elektroodpadów Oraz Uwarunkowania Prawno-Gospodarcze Wprowadzenia Go w Polsce; Pracodawcy Rzeczypospolitej Polskiej: Warsaw, Poland, 2015; pp. 25–28. ISBN 978-83-942639-1-1. [Google Scholar]
- Juarez, C.; Dutra, A. Gold electrowinning from thiourea solutions. Miner. Eng. 2000, 13, 1083–1096. [Google Scholar] [CrossRef]
- Ippolito, N.M.; Medici, F.; Pietrelli, L.; Piga, L. Effect of Acid Leaching Pre-Treatment on Gold Extraction from Printed Circuit Boards of Spent Mobile Phones. Materials 2021, 14, 362. [Google Scholar] [CrossRef] [PubMed]






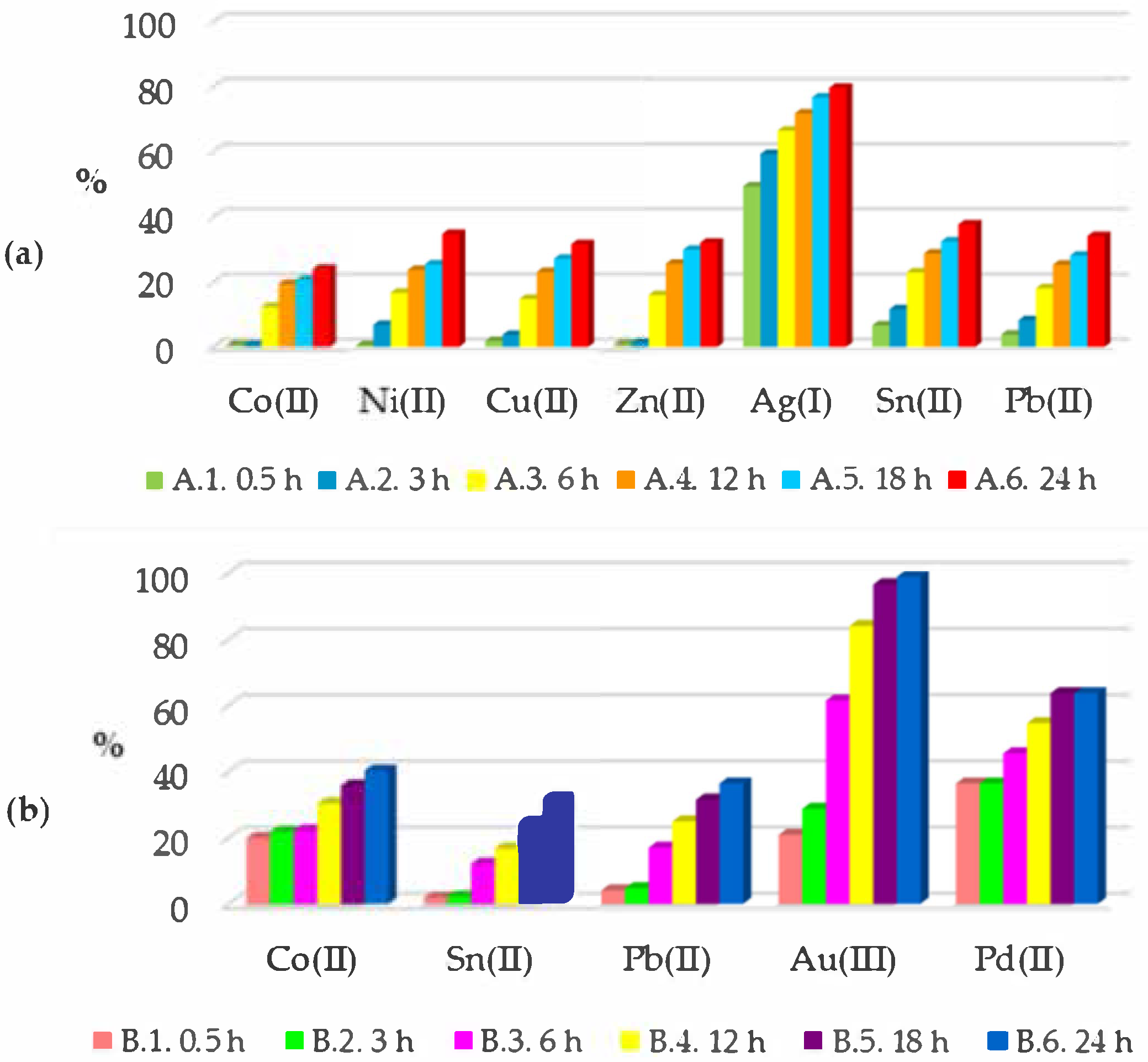
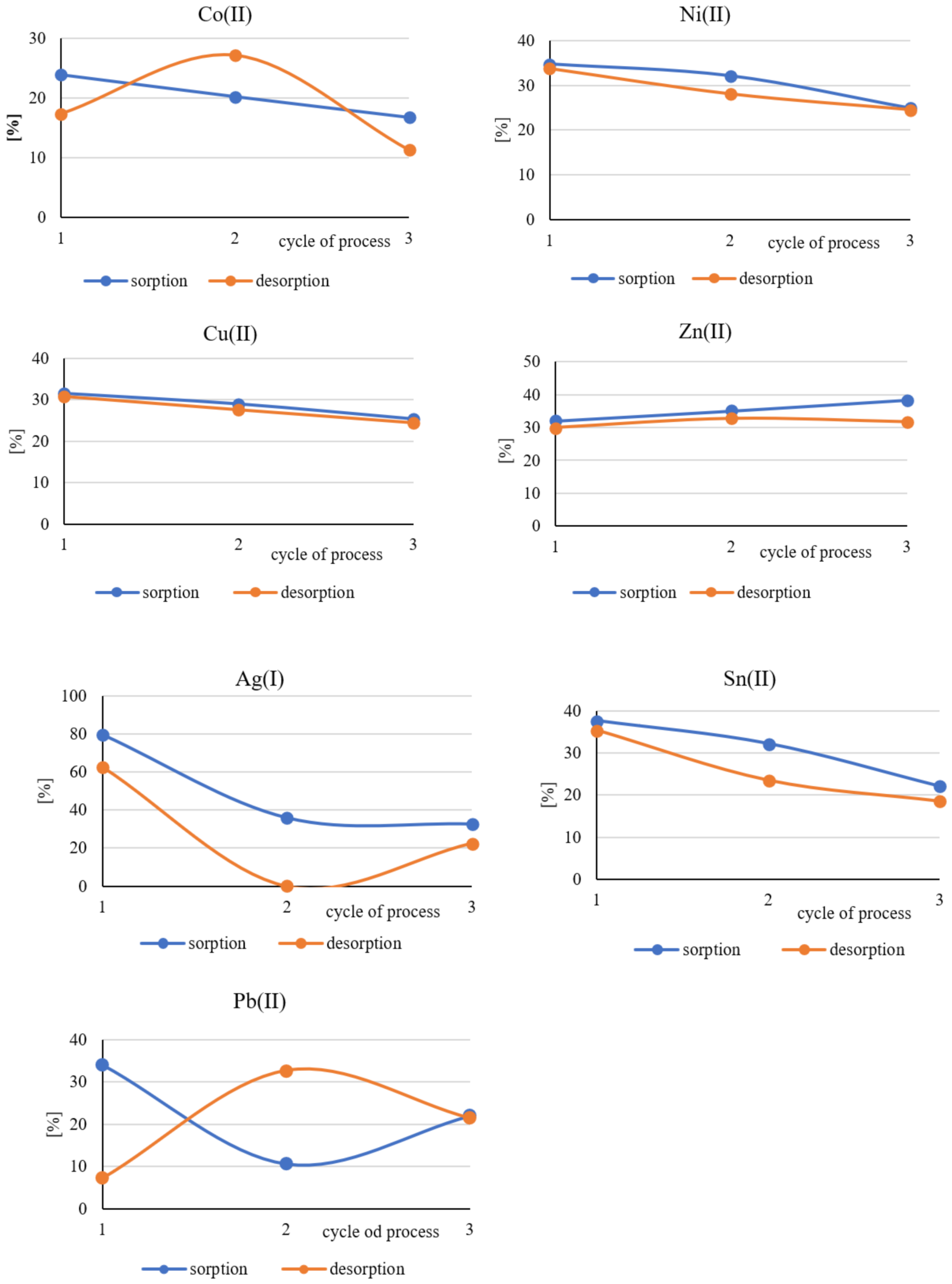
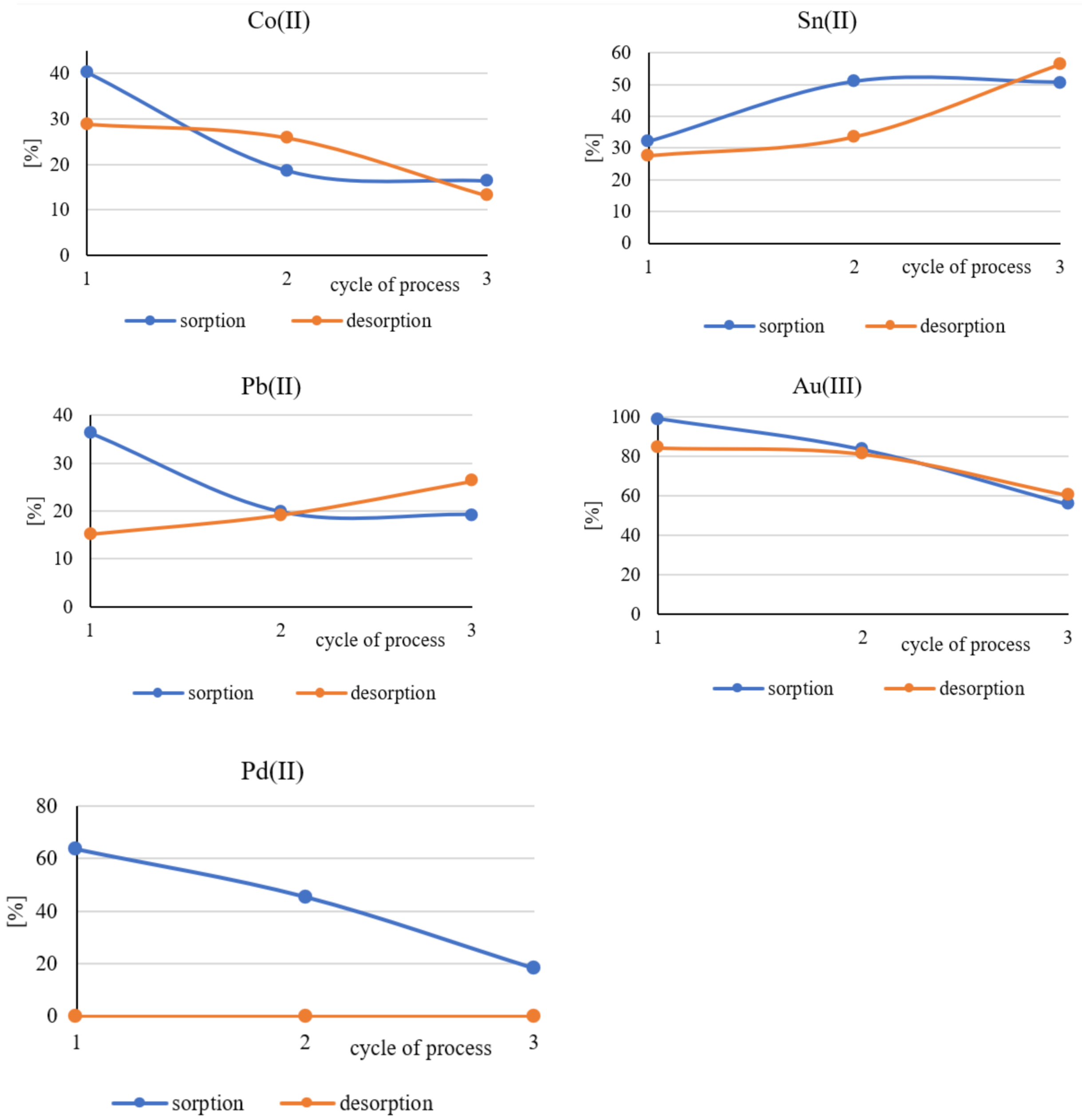
| Symbol of the Sample | Co(II) | Ni(II) | Cu(II) | Zn(II) | Ag(I) | Sn(II) | Pb(II) | Au(III) | Pd(II) | Ta(V) | |
|---|---|---|---|---|---|---|---|---|---|---|---|
| (ppb) | |||||||||||
| A.0 | 35.1 | 239,656.29 | 301,789.35 | 102,075.88 | 4.32 | 3065.40 | 3262.64 | - | - | BLQ | |
| B.0 | 22.1 | - | - | - | - | 15,111.88 | 2309.07 | 47.63 | 0.11 | BLQ | |
| Symbol of the Sample | Time (h) | Co(II) | Ni(II) | Cu(II) | Zn(II) | Ag(I) | Sn(II) | Pb(II) |
|---|---|---|---|---|---|---|---|---|
| (ppb) | ||||||||
| A.1 | 0.50 | 34.92 | 238,563.29 | 296,386.02 | 101,145.38 | 2.19 | 2863.98 | 3139.67 |
| A.2 | 1.50 | 34.94 | 223,442.34 | 290,767.62 | 100,853.95 | 1.76 | 2712.10 | 2996.41 |
| A.3 | 3.00 | 30.83 | 199,932.97 | 257,349.24 | 85,830.85 | 1.45 | 2363.87 | 2676.53 |
| A.4 | 10.00 | 28.31 | 183,021.32 | 232,285.68 | 75,987.33 | 1.22 | 2185.95 | 2438.11 |
| A.5 | 23.00 | 27.91 | 178,906.54 | 220,067.10 | 71,645.99 | 1.01 | 2072.34 | 2347.74 |
| A.6 | 24.00 | 26.72 | 156,570.95 | 206,577.27 | 69,413.90 | 0.88 | 1911.74 | 2152.41 |
| 76.07% | 65.33% | 68.45% | 68.00% | 20.37% | 62.36% | 65.97% | ||
| Symbol of the Sample | Time (h) | Co(II) | Sn(II) | Pb(II) | Au(III) | Pd(II) |
|---|---|---|---|---|---|---|
| (ppb) | ||||||
| B.1 | 0.50 | 1.77 | 14,823.34 | 2215.05 | 37.70 | 0.07 |
| B.2 | 1.50 | 1.73 | 14,783.23 | 2200.64 | 33.95 | 0.07 |
| B.3 | 3.00 | 1.72 | 13,281.13 | 1917.68 | 18.37 | 0.06 |
| B.4 | 10.00 | 1.54 | 12,586.45 | 1734.79 | 7.64 | 0.05 |
| B.5 | 23.00 | 1.42 | 11,367.46 | 1582.00 | 1.65 | 0.04 |
| B.6 | 24.00 | 1.32 | 10,241.67 | 1469.91 | 0.54 | 0.04 |
| 59.73% | 67.77% | 63.66% | 1.13% | 36.36% | ||
| Metal Ions | qt (mg/g) in A Solution | qt (mmol/g) in A Solution | qt (mg/g) in B Solution | qt (mmol/g) in B Solution |
|---|---|---|---|---|
| Co(II) | 4.33 × 10−4 | 7.35 × 10−7 | 3.87 × 10−5 | 6.57 × 10−7 |
| Ni(II) | 4.28 | 7.30 × 10−2 | - | - |
| Cu(II) | 4.91 | 7.72 × 10−2 | - | - |
| Zn(II) | 1.68 | 2.57 × 10−2 | - | - |
| Ag(I) | 1.77 × 10−4 | 1.64 × 10−6 | - | - |
| Sn(II) | 5.95 × 10−2 | 5.01 × 10−4 | 2.12 × 10−2 | 1.78 × 10−3 |
| Pb(II) | 5.72 × 10−2 | 2.76 × 10−4 | 3.65 × 10−2 | 1.76 × 10−4 |
| Au(III) | - | - | 2.05 × 10−3 | 1.04 × 10−5 |
| Pd(II) | - | - | 3.05 × 10−5 | 2.86 × 10−8 |
| Co(II) | Ni(II) | Cu(II) | Zn(II) | Ag(I) | Sn(II) | Pb(II) | Au(III) | Pd(II) | |
|---|---|---|---|---|---|---|---|---|---|
| mg of Metal Ions/0.5 g Electro-Scrap | 0.00038 | 3.73884 | 4.28454 | 1.46979 | 0.00015 | 0.23698 | 0.08185 | 0.00179 | 0.000002 |
| Co(II) | Ni(II) | Cu(II) | Zn(II) | Ag(I) | Sn(II) | Pb(II) | Au(III) | Pd(II) | |
|---|---|---|---|---|---|---|---|---|---|
| mg of Metal Ions/0.5 g Electro-Scrap | 0.00076 | 7.47768 | 8.56909 | 2.93958 | 0.00031 | 0.47397 | 0.16369 | 0.72358 * | 0.000005 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Witt, K.; Urbaniak, W.; Kaczorowska, M.A.; Bożejewicz, D. Simultaneous Recovery of Precious and Heavy Metal Ions from Waste Electrical and Electronic Equipment (WEEE) Using Polymer Films Containing Cyphos IL 101. Polymers 2021, 13, 1454. https://doi.org/10.3390/polym13091454
Witt K, Urbaniak W, Kaczorowska MA, Bożejewicz D. Simultaneous Recovery of Precious and Heavy Metal Ions from Waste Electrical and Electronic Equipment (WEEE) Using Polymer Films Containing Cyphos IL 101. Polymers. 2021; 13(9):1454. https://doi.org/10.3390/polym13091454
Chicago/Turabian StyleWitt, Katarzyna, Włodzimierz Urbaniak, Małgorzata A. Kaczorowska, and Daria Bożejewicz. 2021. "Simultaneous Recovery of Precious and Heavy Metal Ions from Waste Electrical and Electronic Equipment (WEEE) Using Polymer Films Containing Cyphos IL 101" Polymers 13, no. 9: 1454. https://doi.org/10.3390/polym13091454
APA StyleWitt, K., Urbaniak, W., Kaczorowska, M. A., & Bożejewicz, D. (2021). Simultaneous Recovery of Precious and Heavy Metal Ions from Waste Electrical and Electronic Equipment (WEEE) Using Polymer Films Containing Cyphos IL 101. Polymers, 13(9), 1454. https://doi.org/10.3390/polym13091454






