Perspectives of Molecularly Imprinted Polymer-Based Drug Delivery Systems in Cancer Therapy
Abstract
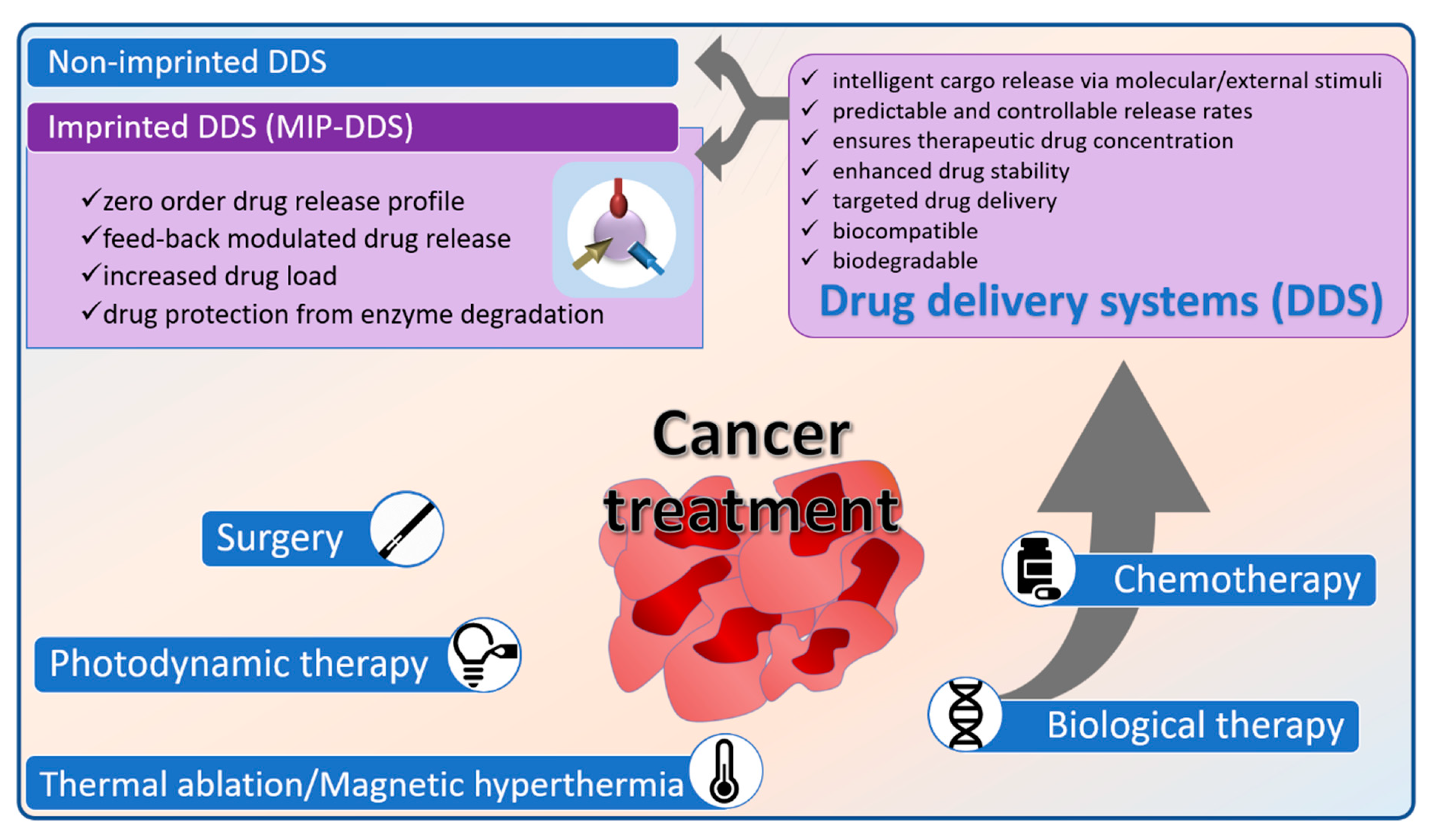
1. Therapeutic Approaches in Cancer Therapy
2. Non-Imprinted DDS for Cancer Treatment
3. Molecular Imprinting
4. Molecular Imprinting Approaches in MIP-DDS Development for Cancer Therapy
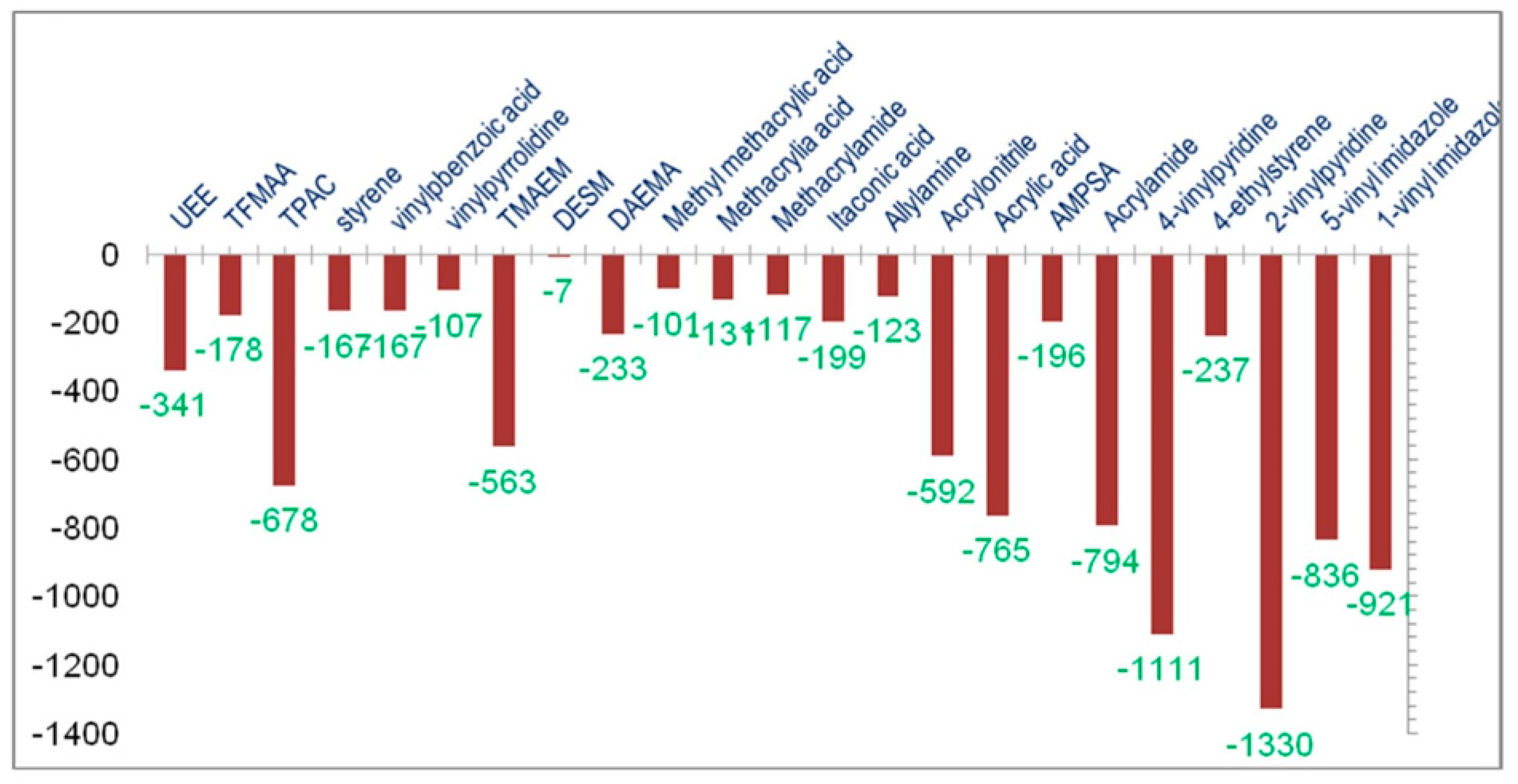
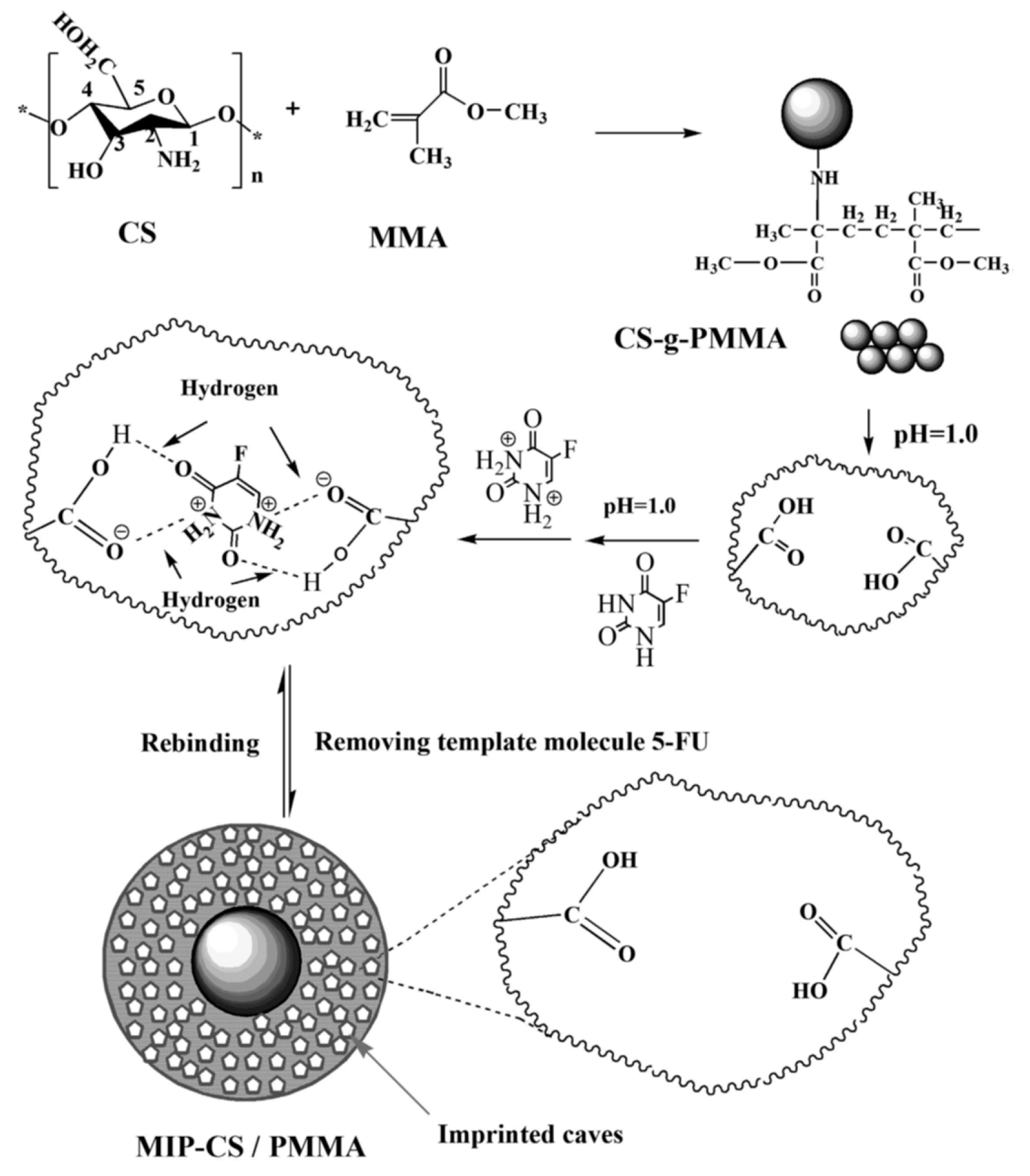
4.1. Non-Covalent Imprinting
4.2. Covalent Imprinting
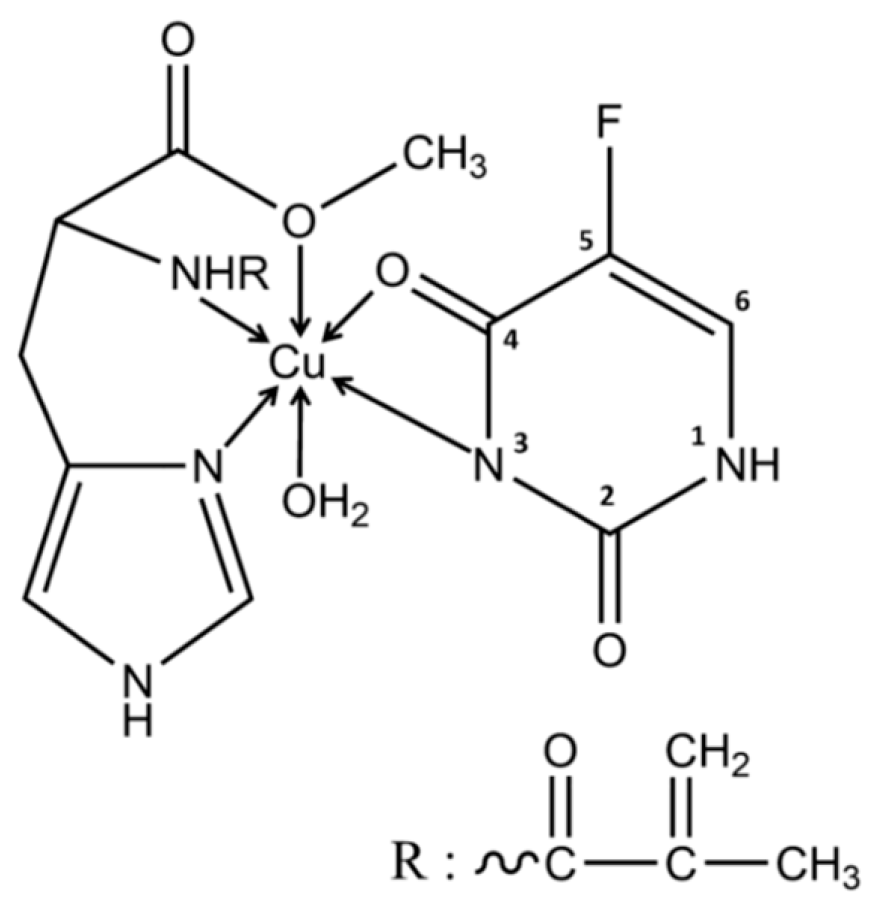
4.3. Metal Ion-Mediated Imprinting
5. Stimuli Responsive Imprinted DDS for Cancer Treatment
6. Active Targeting
7. Biocompatibility and Biodegradability of MIP-DDS
8. Disambiguation of Modified-Release DDS Types
9. In Vivo Evaluated Imprinted DDS
9.1. Cancer Therapy
9.2. Other Therapeutic Applications
10. Conclusions and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Pérez-Herrero, E.; Fernández-Medarde, A. Advanced targeted therapies in cancer: Drug nanocarriers, the future of chemotherapy. Eur. J. Pharm. Biopharm. 2015, 93, 52–79. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Li, L. Photodynamic combinational therapy in cancer treatment. J. BUON. Off. J. Balk. Union Oncol. 2018, 23, 561–567. [Google Scholar]
- Hanahan, D.; Weinberg, R.A. The hallmarks of cancer. Cell 2000, 100, 57–70. [Google Scholar] [CrossRef]
- Chabner, B.A.; Roberts, T.G., Jr. Timeline: Chemotherapy and the war on cancer. Nat. Rev. Cancer 2005, 5, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Mujokoro, B.; Adabi, M.; Sadroddiny, E.; Adabi, M.; Khosravani, M. Nano-structures mediated co-delivery of therapeutic agents for glioblastoma treatment: A review. Mater. Sci. Eng. C 2016, 69, 1092–1102. [Google Scholar] [CrossRef]
- Din, F.U.; Aman, W.; Ullah, I.; Qureshi, O.S.; Mustapha, O.; Shafique, S.; Zeb, A. Effective use of nanocarriers as drug delivery systems for the treatment of selected tumors. Int. J. Nanomed. 2017, 12, 7291–7309. [Google Scholar] [CrossRef]
- Kryscio, D.R.; Peppas, N.A. Mimicking biological delivery through feedback-controlled drug release systems based on molecular imprinting. AIChE J. 2009, 55, 1311–1324. [Google Scholar] [CrossRef]
- Wen, H.; Jung, H.; Li, X. Drug delivery approaches in addressing clinical pharmacology-related issues: Opportunities and challenges. AAPS J. 2015, 17, 1327–1340. [Google Scholar] [CrossRef]
- Kamaly, N.; Yameen, B.; Wu, J.; Farokhzad, O.C. Degradable controlled-release polymers and polymeric nanoparticles: Mechanisms of controlling drug release. Chem. Rev. 2016, 116, 2602–2663. [Google Scholar] [CrossRef]
- Silva, M.S.d.; Casimiro, T. High affinity polymers by molecular imprinting for drug delivery. In Polymerization; Gomes, A.D.S., Ed.; InTech: Rijeka, Croatia, 2012; p. 7. [Google Scholar]
- Luliński, P. Molecularly imprinted polymers based drug delivery devices: A way to application in modern pharmacotherapy. A review. Mater. Sci. Eng. C 2017, 76, 1344–1353. [Google Scholar] [CrossRef] [PubMed]
- Tuwahatu, C.A.; Yeung, C.C.; Lam, Y.W.; Roy, V.A.L. The molecularly imprinted polymer essentials: Curration of anticancer, ophtalmic, and projected gene therapy drug delivery systems. J. Control. Release 2018, 287, 24–34. [Google Scholar] [CrossRef] [PubMed]
- Zaidi, S.A. Molecular imprinted polymers as drug delivery vehicles. Drug Deliv. 2016, 23, 2262–2271. [Google Scholar] [CrossRef] [PubMed]
- Puoci, F.; Cirillo, G.; Curcio, M.; Parisi, O.I.; Iemma, F.; Picci, N. Molecularly imprinted polymers in drug delivery: State of art and future perspectives. Expert Opin. Drug Deliv. 2011, 8, 1379–1393. [Google Scholar] [CrossRef] [PubMed]
- Bobo, D.; Robinson, K.J.; Islam, J.; Thurecht, K.J.; Corrie, S.R. Nanoparticle-based medicines: A review of fda-approved materials and clinical trials to date. Pharm. Res. 2016, 33, 2373–2387. [Google Scholar] [CrossRef] [PubMed]
- Finn, M. Emerging methods for the rapid determination of enantiomeric excess. Chirality 2002, 14, 534–540. [Google Scholar] [CrossRef]
- U.S. Food and Drug Administration (FDA). Hematology/Oncology (Cancer) Approvals and Safty Notifications; U.S. Department of Health and Human Services: Washington, DC, USA, 2019.
- Wang, X.; Wang, Y.; Chen, Z.G.; Shin, D.M. Advances of cancer therapy by nanotechnology. Cancer Res. Treat. 2009, 41, 1–11. [Google Scholar] [CrossRef]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; Rodriguez-Torres, M.D.P.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S.; et al. Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef]
- Kakkar, A.; Traverso, G.; Farokhzad, O.C.; Weissleder, R.; Langer, R. Evolution of macromolecular complexity in drug delivery systems. Nat. Rev. Chem. 2017, 1, 0063. [Google Scholar] [CrossRef]
- Merkle, H.P. Drug delivery’s quest for polymers: Where are the frontiers? Eur. J. Pharm. Biopharm. 2015, 97, 293–303. [Google Scholar] [CrossRef]
- Fuhrmann, K.; Fuhrmann, G. Recent advances in oral delivery of macromolecular drugs and benefits of polymer conjugation. Curr. Opin. Colloid Interface Sci. 2017, 31, 67–74. [Google Scholar] [CrossRef]
- Vogus, D.R.; Krishnan, V.; Mitragotri, S. A review on engineering polymer drug conjugates to improve combination chemotherapy. Curr. Opin. Colloid Interface Sci. 2017, 31, 75–85. [Google Scholar] [CrossRef]
- Liu, Z.; Jiao, Y.; Wang, Y.; Zhou, C.; Zhang, Z. Polysaccharides-based nanoparticles as drug delivery systems. Adv. Drug Deliv. Rev. 2008, 60, 1650–1662. [Google Scholar] [CrossRef] [PubMed]
- Liechty, W.B.; Kryscio, D.R.; Slaughter, B.V.; Peppas, N.A. Polymers for drug delivery systems. Annu. Rev. Chem. Biomol. Eng. 2010, 1, 149–173. [Google Scholar] [CrossRef]
- BelBruno, J.J. Molecularly imprinted polymers. Chem. Rev. 2019, 119, 94–119. [Google Scholar] [CrossRef]
- Beyazit, S.; Tse Sum Bui, B.; Haupt, K.; Gonzato, C. Molecularly imprinted polymer nanomaterials and nanocomposites by controlled/living radical polymerization. Prog. Polym. Sci. 2016, 62, 1–21. [Google Scholar] [CrossRef]
- Sobiech, M.; Bujak, P.; Lulinski, P.; Pron, A. Semiconductor nanocrystal-polymer hybrid nanomaterials and their application in molecular imprinting. Nanoscale 2019, 11, 12030–12074. [Google Scholar] [CrossRef]
- Huynh, T.-P.; Sharma, P.S.; Sosnowska, M.; D’Souza, F.; Kutner, W. Functionalized polythiophenes: Recognition materials for chemosensors and biosensors of superior sensitivity, selectivity, and detectability. Prog. Polym. Sci. 2015, 47, 1–25. [Google Scholar] [CrossRef]
- Chen, L.; Wang, X.; Lu, W.; Wu, X.; Li, J. Molecular imprinting: Perspectives and applications. Chem. Soc. Rev. 2016, 45, 2137–2211. [Google Scholar] [CrossRef]
- Alvarez-Lorenzo, C.; Concheiro, A. Handbook of Molecularly Imprinted Polymers; Smithers Rapra: Shawbury, UK, 2013. [Google Scholar]
- Kumar, N.; Narayanan, N.; Gupta, S. Application of magnetic molecularly imprinted polymers for extraction of imidacloprid from eggplant and honey. Food Chem. 2018, 255, 81–88. [Google Scholar] [CrossRef]
- Bates, F.; Busato, M.; Piletska, E.; Whitcombe, M.J.; Karim, K.; Guerreiro, A.; del Valle, M.; Giorgetti, A.; Piletsky, S. Computational design of molecularly imprinted polymer for direct detection of melamine in milk. Sep. Sci. Technol. 2017, 52, 1441–1453. [Google Scholar] [CrossRef]
- Hu, M.; Huang, P.; Suo, L.; Wu, F. Polydopamine-based molecularly imprinting polymers on magnetic nanoparticles for recognition and enrichment of ochratoxins prior to their determination by hplc. Mikrochim. Acta 2018, 185, 300. [Google Scholar] [CrossRef] [PubMed]
- Luliński, P.; Giebułtowicz, J.; Wroczyński, P.; Maciejewska, D. A highly selective molecularly imprinted sorbent for extraction of 2-aminothiazoline-4-carboxylic acid—Synthesis, characterization and application in post-mortem whole blood analysis. J. Chromatogr. A 2015, 1420, 16–25. [Google Scholar] [CrossRef] [PubMed]
- Marć, M.; Wieczorek, P.P. Application potential of dummy molecularly imprinted polymers as solid-phase extraction sorbents for determination of low-mass polybrominated diphenyl ethers in soil and sediment samples. Microchem. J. 2019, 144, 461–468. [Google Scholar] [CrossRef]
- Piletsky, S.A.; Turner, N.W.; Laitenberger, P. Molecularly imprinted polymers in clinical diagnostics—Future potential and existing problems. Med. Eng. Phys. 2006, 28, 971–977. [Google Scholar] [CrossRef] [PubMed]
- Ansari, S.; Karimi, M. Recent configurations and progressive uses of magnetic molecularly imprinted polymers for drug analysis. Talanta 2017, 167, 470–485. [Google Scholar] [CrossRef]
- Saylan, Y.; Akgonullu, S.; Yavuz, H.; Unal, S.; Denizli, A. Molecularly imprinted polymer based sensors for medical applications. Sensors (Basel) 2019, 19, 1279. [Google Scholar] [CrossRef]
- Smolinska-Kempisty, K.; Ahmad, O.S.; Guerreiro, A.; Karim, K.; Piletska, E.; Piletsky, S. New potentiometric sensor based on molecularly imprinted nanoparticles for cocaine detection. Biosens. Bioelectron. 2017, 96, 49–54. [Google Scholar] [CrossRef]
- Menon, S.; Jesny, S.; Girish Kumar, K. A voltammetric sensor for acetaminophen based on electropolymerized-molecularly imprinted poly(o-aminophenol) modified gold electrode. Talanta 2018, 179, 668–675. [Google Scholar] [CrossRef]
- Iacob, B.-C.; Bodoki, E.; Oprean, R. Recent advances in capillary electrochromatography using molecularly imprinted polymers. Electrophoresis 2014, 35, 2722–2732. [Google Scholar] [CrossRef]
- Iacob, B.-C.; Bodoki, A.E.; Oprean, L.; Bodoki, E. Metal—Ligand interactions in molecular imprinting. In Ligand; Chandraleka, S., Biswas, B., Eds.; Intech Open: Rijeka, Croatia, 2018. [Google Scholar]
- Pan, J.; Chen, W.; Ma, Y.; Pan, G. Molecularly imprinted polymers as receptor mimics for selective cell recognition. Chem. Soc. Rev. 2018, 15, 5574–5587. [Google Scholar] [CrossRef] [PubMed]
- Cowen, T.; Karim, K.; Piletsky, S. Computational approaches in the design of synthetic receptors—A review. Anal. Chim. Acta 2016, 936, 62–74. [Google Scholar] [CrossRef] [PubMed]
- Iskierko, Z.; Sharma, P.S.; Bartold, K.; Pietrzyk-Le, A.; Noworyta, K.; Kutner, W. Molecularly imprinted polymers for separating and sensing of macromolecular compounds and microorganisms. Biotechnol. Adv. 2016, 34, 30–46. [Google Scholar] [CrossRef] [PubMed]
- Couvreur, P. Nanoparticles in drug delivery: Past, present and future. Adv. Drug Deliv. Rev. 2013, 65, 21–23. [Google Scholar] [CrossRef]
- Yokoyama, M.; Fukushima, S.; Uehara, R.; Okamoto, K.; Kataoka, K.; Sakurai, Y.; Okano, T. Characterization of physical entrapment and chemical conjugation of adriamycin in polymeric micelles and their design for in vivo delivery to a solid tumor. J. Control. Release 1998, 50, 79–92. [Google Scholar] [CrossRef]
- Iacob, B.-C.; Bodoki, E.; Oprean, R. Chiral electrochemical sensors based on molecularly imprinted polymers with pharmaceutical applications. In Handbook of Sustainable Polymers; Pan Stanford: Singapore, 2015; pp. 587–614. [Google Scholar]
- Oliveira, D.; Gomes, C.P.; Dias, R.C.S.; Costa, M.R.P.F.N. Molecular imprinting of 5-fluorouracil in particles with surface raft grafted functional brushes. React. Funct. Polym. 2016, 107, 35–45. [Google Scholar] [CrossRef]
- Kan, W.; Li, X. Mathematical modeling and sustained release property of a 5-fluorouracil imprinted vehicle. Eur. Polym. J. 2013, 49, 4167–4175. [Google Scholar] [CrossRef]
- Madadian-Bozorg, N.; Zahedi, P.; Shamsi, M.; Safarian, S. Poly (methacrylic acid)-based molecularly imprinted polymer nanoparticles containing 5-fluourouracil used in colon cancer therapy potentially. Polym. Adv. Technol. 2018, 29, 2401–2409. [Google Scholar] [CrossRef]
- Mo, C.E.; Chai, M.H.; Zhang, L.P.; Ran, R.X.; Huang, Y.P.; Liu, Z.S. Floating molecularly imprinted polymers based on liquid crystalline and polyhedral oligomeric silsesquioxanes for capecitabine sustained release. Int. J. Pharm. 2019, 557, 293–303. [Google Scholar] [CrossRef]
- Cegłowski, M.; Kurczewska, J.; Ruszkowski, P.; Schroeder, G. Application of paclitaxel-imprinted microparticles obtained using two different cross-linkers for prolonged drug delivery. Eur. Polym. J. 2019, 118, 328–336. [Google Scholar] [CrossRef]
- Zhang, L.-P.; Tang, S.-H.; Mo, C.-E.; Wang, C.; Huang, Y.-P.; Liu, Z.-S. Synergistic effect of liquid crystal and polyhedral oligomeric silsesquioxane to prepare molecularly imprinted polymer for paclitaxel delivery. Eur. Polym. J. 2018, 98, 226–236. [Google Scholar] [CrossRef]
- Esfandyari-Manesh, M.; Darvishi, B.; Ishkuh, F.A.; Shahmoradi, E.; Mohammadi, A.; Javanbakht, M.; Dinarvand, R.; Atyabi, F. Paclitaxel molecularly imprinted polymer-peg-folate nanoparticles for targeting anticancer delivery: Characterization and cellular cytotoxicity. Mater. Sci. Eng. C 2016, 62, 626–633. [Google Scholar] [CrossRef] [PubMed]
- Bai, J.; Zhang, Y.; Chen, L.; Yan, H.; Zhang, C.; Liu, L.; Xu, X. Synthesis and characterization of paclitaxel-imprinted microparticles for controlled release of an anticancer drug. Mater. Sci. Eng. C 2018, 92, 338–348. [Google Scholar] [CrossRef] [PubMed]
- Azizi, I.F.; Mehran, J.; Mehdi, E.-M.; Rassoul, D.; Fatemeh, A. Synthesis and characterization of paclitaxel-imprinted nanoparticles for recognition and controlled release of an anticancer drug. J. Mater. Sci. 2014, 49, 6343–6352. [Google Scholar]
- Suksuwan, A.; Lomlim, L.; Rungrotmongkol, T.; Nakpheng, T.; Dickert, F.L.; Suedee, R. The composite nanomaterials containing (r)-thalidomide-molecularly imprinted polymers as a recognition system for enantioselective-controlled release and targeted drug delivery. J. Appl. Polym. Sci. 2015, 132. [Google Scholar] [CrossRef]
- Jaiswal, L.; Rakkit, S.; Pochin, K.; Jaisamut, P.; Tanthana, C.; Tanmanee, N.; Srichana, T.; Suedee, R. A thalidomide templated molecularly imprinted polymer that promotes a biologically active chiral entity tagged in colon carcinoma cells and protein-related immune activation. Process Biochem. 2015, 50, 2035–2050. [Google Scholar] [CrossRef]
- Ichikawa, M.; Akimoto, J.; Miki, Y.; Maeda, J.; Takahashi, T.; Fujiwara, Y.; Kohno, M. Photodynamic therapy with talaporfin sodium induces dose- and time-dependent apoptotic cell death in malignant meningioma hkbmm cells. Photodiagnosis Photodyn. Ther. 2019, 25, 29–34. [Google Scholar] [CrossRef]
- Scrivano, L.; Parisi, O.I.; Iacopetta, D.; Ruffo, M.; Ceramella, J.; Sinicropi, M.S.; Puoci, F. Molecularly imprinted hydrogels for sustained release of sunitinib in breast cancer therapy. Polym. Adv. Technol. 2019, 30, 743–748. [Google Scholar] [CrossRef]
- Hashemi-Moghaddam, H.; Kazemi-Bagsangani, S.; Jamili, M.; Zavareh, S. Evaluation of magnetic nanoparticles coated by 5-fluorouracil imprinted polymer for controlled drug delivery in mouse breast cancer model. Int. J. Pharm. 2016, 497, 228–238. [Google Scholar] [CrossRef]
- Puoci, F.; Cirillo, G.; Curcio, M.; Iemma, F.; Spizzirri, U.G.; Picci, N. Molecularly imprinted solid phase extraction for the selective hplc determination of alpha-tocopherol in bay leaves. Anal. Chim. Acta 2007, 593, 164–170. [Google Scholar] [CrossRef]
- Meng, A.C.; LeJeune, J.; Spivak, D.A. Multi-analyte imprinting capability of omnimips versus traditional molecularly imprinted polymers. J. Mol. Recognit. JMR 2009, 22, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Gardouh, A.R.; Barakat, B.M.; Qushawy, M.K.E.; El-Kazzaz, A.Y.; Sami, M.M.; Zaitone, S.A. Antitumor activity of a molecularly imprinted nanopreparation of 5-flurouracil against ehrlich’s carcinoma solid tumors grown in mice: Comparison to free 5-flurouracil. Chem. Biol. Interact. 2018, 295, 52–63. [Google Scholar] [CrossRef] [PubMed]
- Griffete, N.; Fresnais, J.; Espinosa, A.; Wilhelm, C.; Bee, A.; Menager, C. Design of magnetic molecularly imprinted polymer nanoparticles for controlled release of doxorubicin under an alternative magnetic field in athermal conditions. Nanoscale 2015, 7, 18891–18896. [Google Scholar] [CrossRef] [PubMed]
- Hassanpour, A.; Irandoust, M.; Soleimani, E.; Zhaleh, H. Increasing the anticancer activity of azidothymidine toward the breast cancer via rational design of magnetic drug carrier based on molecular imprinting technology. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 103, 109771. [Google Scholar] [CrossRef]
- Kempe, M. Antibody-mimicking polymers as chiral stationary phases in hplc. Anal. Chem. 1996, 68, 1948–1953. [Google Scholar] [CrossRef]
- Talavat, L.; Güner, A. Thermodynamic computational calculations for preparation 5-fluorouracil magnetic moleculary imprinted polymers and their application in controlled drug release. Inorg. Chem. Commun. 2019, 103, 119–127. [Google Scholar] [CrossRef]
- Korde, B.A.; Mankar, J.S.; Phule, S.; Krupadam, R.J. Nanoporous imprinted polymers (nanomips) for controlled release of cancer drug. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 99, 222–230. [Google Scholar] [CrossRef]
- Kloskowski, A.; Pilarczyk, M.; Przyjazny, A.; Namieśnik, J. Progress in development of molecularly imprinted polymers as sorbents for sample preparation. Crit. Rev. Anal. Chem. 2009, 39, 43–58. [Google Scholar] [CrossRef]
- Qu, G.; Zheng, S.; Liu, Y.; Xie, W.; Wu, A.; Zhang, D. Metal ion mediated synthesis of molecularly imprinted polymers targeting tetracyclines in aqueous samples. J. Chromatogr. B 2009, 877, 3187–3193. [Google Scholar] [CrossRef]
- Ulbrich, K.; Hola, K.; Subr, V.; Bakandritsos, A.; Tucek, J.; Zboril, R. Targeted drug delivery with polymers and magnetic nanoparticles: Covalent and noncovalent approaches, release control, and clinical studies. Chem. Rev. 2016, 116, 5338–5431. [Google Scholar] [CrossRef]
- Liu, T.; Qiao, Z.; Wang, J.; Zhang, P.; Zhang, Z.; Guo, D.S.; Yang, X. Molecular imprinted s-nitrosothiols nanoparticles for nitric oxide control release as cancer target chemotherapy. Colloids Surf. B Biointerfaces 2019, 173, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Bai, L.H.; Chen, X.X.; Huang, Y.P.; Zhang, Q.W.; Liu, Z.S. Chiral separation of racemic mandelic acids by use of an ionic liquid-mediated imprinted monolith with a metal ion as self-assembly pivot. Anal. Bioanal. Chem. 2013, 405, 8935–8943. [Google Scholar] [CrossRef] [PubMed]
- Çetin, K.; Denizli, A. 5-fluorouracil delivery from metal-ion mediated molecularly imprinted cryogel discs. Colloids Surf. B Biointerfaces 2015, 126, 401–406. [Google Scholar] [CrossRef] [PubMed]
- Zhou, S.; Luoma, S.E.; St Armour, G.E.; Thakkar, E.; Mackay, T.F.C.; Anholt, R.R.H. A drosophila model for toxicogenomics: Genetic variation in susceptibility to heavy metal exposure. PLoS Genet 2017, 13, e1006907. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Zhang, L.; Wang, P.; Du, S. Coordinate bonding strategy for molecularly imprinted hydrogels: Toward ph-responsive doxorubicin delivery. J. Pharm. Sci. 2014, 103, 643–651. [Google Scholar] [CrossRef]
- Bakhshpour, M.; Yavuz, H.; Denizli, A. Controlled release of mitomycin c from phemah-cu(ii) cryogel membranes. Artif. Cells Nanomed. Biotechnol. 2018, 46, 946–954. [Google Scholar] [CrossRef]
- Trumbo, P.; Yates, A.A.; Schlicker, S.; Poos, M. Dietary reference intakes: Vitamin a, vitamin k, arsenic, boron, chromium, copper, iodine, iron, manganese, molybdenum, nickel, silicon, vanadium, and zinc. J. Am. Diet. Assoc. 2001, 101, 294–301. [Google Scholar] [CrossRef]
- Mura, S.; Nicolas, J.; Couvreur, P. Stimuli-responsive nanocarriers for drug delivery. Nat. Mater. 2013, 12, 991–1003. [Google Scholar] [CrossRef]
- Taresco, V.; Alexander, C.; Singh, N.; Pearce, A.K. Stimuli-responsive prodrug chemistries for drug delivery. Adv. Ther. 2018, 1, 1800030. [Google Scholar] [CrossRef]
- Zhang, K.; Guan, X.; Qiu, Y.; Wang, D.; Zhang, X.; Zhang, H. A ph/glutathione double responsive drug delivery system using molecular imprint technique for drug loading. Appl. Surf. Sci. 2016, 389, 1208–1213. [Google Scholar] [CrossRef]
- Kazemi, S.; Sarabi, A.A.; Abdouss, M. Synthesis and characterization of magnetic molecularly imprinted polymer nanoparticles for controlled release of letrozole. Korean J. Chem. Eng. 2016, 33, 3289–3297. [Google Scholar] [CrossRef]
- Cazares-Cortes, E.; Nerantzaki, M.; Fresnais, J.; Wilhelm, C.; Griffete, N.; Menager, C. Magnetic nanoparticles create hot spots in polymer matrix for controlled drug release. Nanomaterials (Basel) 2018, 8, 850. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Chen, L.; Zhang, H.; Yang, Y.; Liu, X.; Chen, Y. Temperature and magnetism bi-responsive molecularly imprinted polymers: Preparation, adsorption mechanism and properties as drug delivery system for sustained release of 5-fluorouracil. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 61, 158–168. [Google Scholar] [CrossRef] [PubMed]
- Sedghi, R.; Yassari, M.; Heidari, B. Thermo-responsive molecularly imprinted polymer containing magnetic nanoparticles: Synthesis, characterization and adsorption properties for curcumin. Colloids Surf. B Biointerfaces 2018, 162, 154–162. [Google Scholar] [CrossRef]
- Bakhshizadeh, M.; Mohajeri, S.A.; Esmaily, H.; Aledavood, S.A.; Varshoei Tabrizi, F.; Seifi, M.; Hadizadeh, F.; Sazgarnia, A. Utilizing photosensitizing and radiosensitizing properties of tio2-based mitoxantrone imprinted nanopolymer in fibrosarcoma and melanoma cells. Photodiagnosis Photodyn. Ther. 2019, 25, 472–479. [Google Scholar] [CrossRef]
- Ekpenyong-Akiba, A.E.; Canfarotta, F.; Abd H, B.; Poblocka, M.; Casulleras, M.; Castilla-Vallmanya, L.; Kocsis-Fodor, G.; Kelly, M.E.; Janus, J.; Althubiti, M.; et al. Detecting and targeting senescent cells using molecularly imprinted nanoparticles. Nanoscale Horiz. 2019, 4, 757–768. [Google Scholar] [CrossRef]
- Asadi, E.; Abdouss, M.; Leblanc, R.M.; Ezzati, N.; Wilson, J.N.; Azodi-Deilami, S. In vitro/in vivo study of novel anti-cancer, biodegradable cross-linked tannic acid for fabrication of 5-fluorouracil-targeting drug delivery nano-device based on a molecular imprinted polymer. RSC Adv. 2016, 6, 37308–37318. [Google Scholar] [CrossRef]
- Hashemi-Moghaddam, H.; Zavareh, S.; Karimpour, S.; Madanchi, H. Evaluation of molecularly imprinted polymer based on her2 epitope for targeted drug delivery in ovarian cancer mouse model. React. Functional Polym. 2017, 121, 82–90. [Google Scholar] [CrossRef]
- Canfarotta, F.; Lezina, L.; Guerreiro, A.; Czulak, J.; Petukhov, A.; Daks, A.; Smolinska-Kempisty, K.; Poma, A.; Piletsky, S.; Barlev, N.A. Specific drug delivery to cancer cells with double-imprinted nanoparticles against epidermal growth factor receptor. Nano Lett. 2018, 18, 4641–4646. [Google Scholar] [CrossRef]
- Zhang, L.; Qi, Z.; Huang, Q.; Zeng, K.; Sun, X.; Li, J.; Liu, Y.-N. Imprinted-like biopolymeric micelles as efficient nanovehicles for curcumin delivery. Colloids Surf. B Biointerfaces 2014, 123, 15–22. [Google Scholar] [CrossRef]
- Zheng, X.F.; Lian, Q.; Yang, H.; Wang, X. Surface molecularly imprinted polymer of chitosan grafted poly(methyl methacrylate) for 5-fluorouracil and controlled release. Sci. Rep. 2016, 6, 21409. [Google Scholar] [CrossRef] [PubMed]
- Ulery, B.D.; Nair, L.S.; Laurencin, C.T. Biomedical applications of biodegradable polymers. J. Polym. Sci. B Polym. Phys. 2011, 49, 832–864. [Google Scholar] [CrossRef] [PubMed]
- Li, H.; Jin, Z.; Cho, S.; Jeon, M.J.; Nguyen, V.D.; Park, J.O.; Park, S. Folate-receptor-targeted nir-sensitive polydopamine nanoparticles for chemo-photothermal cancer therapy. Nanotechnology 2017, 28, 425101. [Google Scholar] [CrossRef] [PubMed]
- Amin, D.R.; Higginson, C.J.; Korpusik, A.B.; Gonthier, A.R.; Messersmith, P.B. Untemplated resveratrol-mediated polydopamine nanocapsule formation. ACS Appl. Mater. Interfaces 2018, 10, 34792–34801. [Google Scholar] [CrossRef] [PubMed]
- Choi, C.K.; Li, J.; Wei, K.; Xu, Y.J.; Ho, L.W.; Zhu, M.; To, K.K.; Choi, C.H.; Bian, L. A gold@polydopamine core-shell nanoprobe for long-term intracellular detection of micrornas in differentiating stem cells. J. Am. Chem. Soc. 2015, 137, 7337–7346. [Google Scholar] [CrossRef] [PubMed]
- Zhang, M.; Zhang, X.; He, X.; Chen, L.; Zhang, Y. A self-assembled polydopamine film on the surface of magnetic nanoparticles for specific capture of protein. Nanoscale 2012, 4, 3141–3147. [Google Scholar] [CrossRef] [PubMed]
- Park, J.; Brust, T.F.; Lee, H.J.; Lee, S.C.; Watts, V.J.; Yeo, Y. Polydopamine-based simple and versatile surface modification of polymeric nano drug carriers. ACS Nano 2014, 8, 3347–3356. [Google Scholar] [CrossRef]
- Zavareh, S.; Mahdi, M.; Erfanian, S.; Hashemi-Moghaddam, H. Synthesis of polydopamine as a new and biocompatible coating of magnetic nanoparticles for delivery of doxorubicin in mouse breast adenocarcinoma. Cancer Chemother. Pharmacol. 2016, 78, 1073–1084. [Google Scholar] [CrossRef]
- Turkmen, D.; Bereli, N.; Corman, M.E.; Shaikh, H.; Akgol, S.; Denizli, A. Molecular imprinted magnetic nanoparticles for controlled delivery of mitomycin c. Artif. Cells Nanomed. Biotechnol. 2014, 42, 316–322. [Google Scholar] [CrossRef]
- Singh, B.; Chauhan, N. Preliminary evaluation of molecular imprinting of 5-fluorouracil within hydrogels for use as drug delivery systems. Acta Biomater. 2008, 4, 1244–1254. [Google Scholar] [CrossRef]
- Vasapollo, G.; Del Sole, R.; Mergola, L.; Lazzoi, M.R.; Scardino, A.; Scorrano, S.; Mele, G. Molecularly imprinted polymers: Present and future prospective. Int. J. Mol. Sci. 2011, 12, 5908–5945. [Google Scholar] [CrossRef] [PubMed]
- Figuerola, A.; Di Corato, R.; Manna, L.; Pellegrino, T. From iron oxide nanoparticles towards advanced iron-based inorganic materials designed for biomedical applications. Pharmacol. Res. 2010, 62, 126–143. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, M.; Simchi, A.; Imani, M.; Milani, A.S.; Stroeve, P. An in vitro study of bare and poly(ethylene glycol)-co-fumarate-coated superparamagnetic iron oxide nanoparticles: A new toxicity identification procedure. Nanotechnology 2009, 20, 225104. [Google Scholar] [CrossRef] [PubMed]
- Perrie, Y.; Rades, T. Controlling drug delivery. In Pharmaceutics: Drug Delivery and Targeting, 2nd ed.; Pharmaceutical Press: London, UK, 2012. [Google Scholar]
- Kaamyabi, S.; Habibi, D.; Amini, M.M. Preparation and characterization of the ph and thermosensitive magnetic molecular imprinted nanoparticle polymer for the cancer drug delivery. Bioorg. Med. Chem. Lett. 2016, 26, 2349–2354. [Google Scholar] [CrossRef] [PubMed]
- Yarong, X.; Xiaoling, H.; Ping, G.; Chunbao, D.; Yuan, T.; Shichao, D.; Zhiling, L.; Chaoren, Y. A novel controllable molecularly imprinted drug delivery system based on the photothermal effect of graphene oxide quantum dots. J. Mater. Sci. 2019, 54, 9124–9139. [Google Scholar]
- Duarte, A.R.C.; Casimiro, T.; Aguiar-Ricardo, A.; Simplício, A.L.; Duarte, C.M.M. Supercritical fluid polymerisation and impregnation of molecularly imprinted polymers for drug delivery. J. Supercrit. Fluids 2006, 39, 102–106. [Google Scholar] [CrossRef]
- Hemmati, K.; Sahraei, R.; Ghaemy, M. Synthesis and characterization of a novel magnetic molecularly imprinted polymer with incorporated graphene oxide for drug delivery. Polymer 2016, 101, 257–268. [Google Scholar] [CrossRef]
- Asadi, E.; Abdouss, M.; Leblanc, R.M.; Ezzati, N.; Wilson, J.N.; Kordestani, D. Synthesis, characterization and in vivo drug delivery study of a biodegradable nano-structured molecularly imprinted polymer based on cross-linker of fructose. Polymer 2016, 97, 226–237. [Google Scholar] [CrossRef]
- Ruela, A.L.M.; Figueiredo, E.C.; Pereira, G.R. Molecularly imprinted polymers as nicotine transdermal delivery systems. Chem. Eng. J. 2014, 248, 1–8. [Google Scholar] [CrossRef]
- Ruela, A.L.M.; de Figueiredo, E.C.; Carvalho, F.C.; de Araújo, M.B.; Pereira, G.R. Adsorption and release of nicotine from imprinted particles synthesised by precipitation polymerisation: Optimising transdermal formulations. Eur. Polym. J. 2018, 100, 67–76. [Google Scholar] [CrossRef]
- Hiratani, H.; Fujiwara, A.; Tamiya, Y.; Mizutani, Y.; Alvarez-Lorenzo, C. Ocular release of timolol from molecularly imprinted soft contact lenses. Biomaterials 2005, 26, 1293–1298. [Google Scholar] [CrossRef] [PubMed]
- Tieppo, A.; White, C.J.; Paine, A.C.; Voyles, M.L.; McBride, M.K.; Byrne, M.E. Sustained in vivo release from imprinted therapeutic contact lenses. J. Control. Release 2012, 157, 391–397. [Google Scholar] [CrossRef] [PubMed]
- Hui, A.; Willcox, M.; Jones, L. In vitro and in vivo evaluation of novel ciprofloxacin-releasing silicone hydrogel contact lensesciprofloxacin-releasing contact lenses. Investig. Ophthalmol. Vis. Sci. 2014, 55, 4896–4904. [Google Scholar] [CrossRef] [PubMed]
- Tashakori-Sabzevar, F.; Mohajeri, S.A. Development of ocular drug delivery systems using molecularly imprinted soft contact lenses. Drug Dev. Ind. Pharm. 2015, 41, 703–713. [Google Scholar] [CrossRef] [PubMed]
| Choices in Molecular Imprinting/Expected Features | MIPs in Analytical Sciences | MIPs as DDS |
|---|---|---|
| Imprinting technique (template/monomer interaction) | VARIABLE non-covalent>>covalent, pivot based | VARIABLE non-covalent>>pivot based |
| Monomer selection | NO CONSTRAINS | LIMITATIONS Biocompatible, biodegradable, Particular functionalities for stimuli-responsive MIPs |
| Cross-linker selection | NO CONSTRAINS | LIMITATIONS Biocompatible and biodegradable with impact on MIP performance |
| Polymerization initiation/MIP morphology | NO CONSTRAINS Photo-, thermal initiation, electropolymerization/Bulk polymer; NPs; films | CONSTRAINS Photo-, thermal initiation/Bulk polymer (hydrogels); NPs |
| Template removal | CRITICAL sometimes tedious | NO NEED |
| Well-defined, homogenous binding sites | HIGH High IF for specific re-binding—improved selectivity | HIGH/VARIABLE Improved drug loading/Combined release profiles |
| Degree of cross-linking | HIGH Polymer rigidity affecting selectivity | VARIABLE Adjustable release kinetics |
| Solvent (porogen) | VARIABLE (aprotic favored in non-covalent imprinting) Porosity important | IDEALLY WATER Traces of non-aqueous solvents may be toxic Porosity affecting release kinetics |
| Active Drug (T) | Imprinting Approach Polymerization Mixture (M/C/I/S) | DDS Type/Targeted Delivery | Release Mechanism Drug Load; Drug Release | Biocompatibility/Biodegradability | Development Stage | Ref. |
|---|---|---|---|---|---|---|
| 5-Fluorouracil | Cu(II) mediated imprinting MAH, HEMA/MBA/APS, TEMED/water | Implantable cryogel discs/- | Swelling-controlled drug release Drug release—80%/9 h (pH 7.4, 37 °C) | Yes/No (acrylic Ms, high Cu(II) load) | Material characterization In vitro release studies | [78] |
| 5-Fluorouracil | Non-covalent imprinting AM/EDMA/AIBN/ACN + methanol | Nanospheres/- | Release mechanism—diffusion/erosion Drug release—60%/10 h (pH 1.2–6.8, 37 °C) | Yes/No (acrylic Ms) | Material characterization In vitro release studies In vivo studies: Female Swiss albino mice inoculated with human Earlich ascites carcinoma cells xenograft | [67] |
| 5-Fluorouracil | Non-covalent imprinting Dopamine/water | MIP-coated Fe3O4 NPs/Magnetically assisted DD | Release mechanism—diffusion/erosion Drug release—80%/4 h (pH 7.4, 37 °C) | Yes/Yes | Material characterization In vitro release, cytotoxicity studies In vivo studies: Breast adenocarcinoma in Balb/c mice | [64] |
| 5-Fluorouracil | Non-covalent imprinting Crosslinked tannic acid/AIBN/hexadecane + SDS in water | MIP-coated Fe3O4@SiO2@FITC-MPS NPs/Magnetically assisted DD | Release mechanism—diffusion/erosion Drug release—70%/80 h (pH 7.4, 37 °C) | Yes/Yes | Material characterization In vitro release, cytotoxicity studies In vivo pharmacokinetic studies: Healthy male Wistar rats | [92] |
| 5-Fluorouracil | Non-covalent imprinting MAA or HEMA/EDMA /AIBN-ACN | Microparticles with polymer functional brushes (FB) (PMAA, PHEMA, PNIPA)/- | Stimuli responsive drug release (pH—MIP with PMAA FB; temperature—MIP with PNIPA FB) Drug load—32.3 μmol·g−1 for MAA MIP with PMAA FB; 28.8 μmol·g−1 for HEMA MIP with PHEMA FB; 27.6 μmol·g−1 for MAA MIP with PNIPA FB Drug release—82%/24 h (pH 10) vs. 41%/24 h (pH 2) for MAA MIP with PMAA FB; 96%/24 h (pH 10) vs. 68.5%/24 h (pH 2) vs. for HEMA MIP with PHEMA FB; 50%/24 h (20 °C) vs. 21%/24 h (40 °C) for MAA MIP with PNIPA FB | Yes/No (acrylic Ms) | Material characterization In vitro release studies | [51] |
| 5-Fluorouracil | Non-covalent imprinting Chitosan, MMA/APS/water | Microspheres/- | Stimuli responsive release (pH) Drug load—96 mg/g (pH = 1.2) Drug release—48%/2 h and 95%/30 h (pH 7.4, 20 °C) vs. 2.5%/30 h (pH 1.2, 20 °C), 30% (pH 4, 20 °C), 62% (pH 6.8, 20 °C) | Yes/No (acrylic Ms) | Material characterization In vitro release studies | [96] |
| 5-Fluorouracil | Non-covalent imprinting NIPA/MBA/APS/water | MIP-coated Fe3O4@CSi nanospheres/Magnetically assisted DD | Stimuli responsive release (temperature); Drug load—96.53 mg/g Drug release—70% (25 °C)/1.6 h, 91.2%/1.6 h (45 °C); | Yes/No (acrylic Ms) | Material characterization In vitro release studies | [88] |
| 5-Fluorouracil | Non-covalent imprinting AA or 4-Vpy/EDMA/AIBN/ACN + methanol | MIP-coated Fe3O4@SiO2@FITC-MPS NPs/Magnetically assisted DD | Stimuli responsive release (pH) Drug release—90%/30 days (pH = 5.8, 37 °C) vs. 70%/30 days (pH = 7.4, 37 °C) for 4-Vpy MIP | Yes/No (acrylic Ms) | Material characterization In vitro release studies | [71] |
| 5-Fluorouracil | Non-covalent imprinting MAA, HEMA/EDMA/AIBN/- | Hydrogel/- | Swelling-controlled drug release Drug load—0.0914 mg/g Drug release—30%/5 h (pH 6.8, 25 °C) | Yes/No (acrylic Ms) | Material characterization In vitro release studies | [52] |
| 5-Fluorouracil | MAA/EDMA/AIBN/ACN | Nanospheres | Mechanism of release—diffusion/erosion Drug load Drug release—40%/10 h (pH 7.4, 37 °C) (burst release); 80%/96 h (pH 7.4, 37 °C) | Yes/No (acrylic Ms) | Material characterization In vitro release, cytotoxicity studies | [53] |
| 5-Fluorouracil | Non-covalent imprinting AA, HEMA/MBA/APS, TEMED/water | Hydrogel/- | Swelling-controlled drug release Drug load—0.875 mg/g Drug release—45%/5 h (37 °C) | Yes/No (acrylic Ms) | Material characterization In vitro release studies | [105] |
| Capecitabine | Non-covalent imprinting MPDE (LC), POSS, MAA/EDMA/AIBN/toluene + ACN | POSS-LC nanocomposite (floating oral DDS)/- | Mechanism of release—diffusion/erosion Drug load—164.21 mg/g Drug release—80%/12 h | Yes/No (acrylic Ms) | Material characterization In vitro release, cytotoxicity studies In vivo pharmacokinetic studies: Healthy male Wistar rats | [54] |
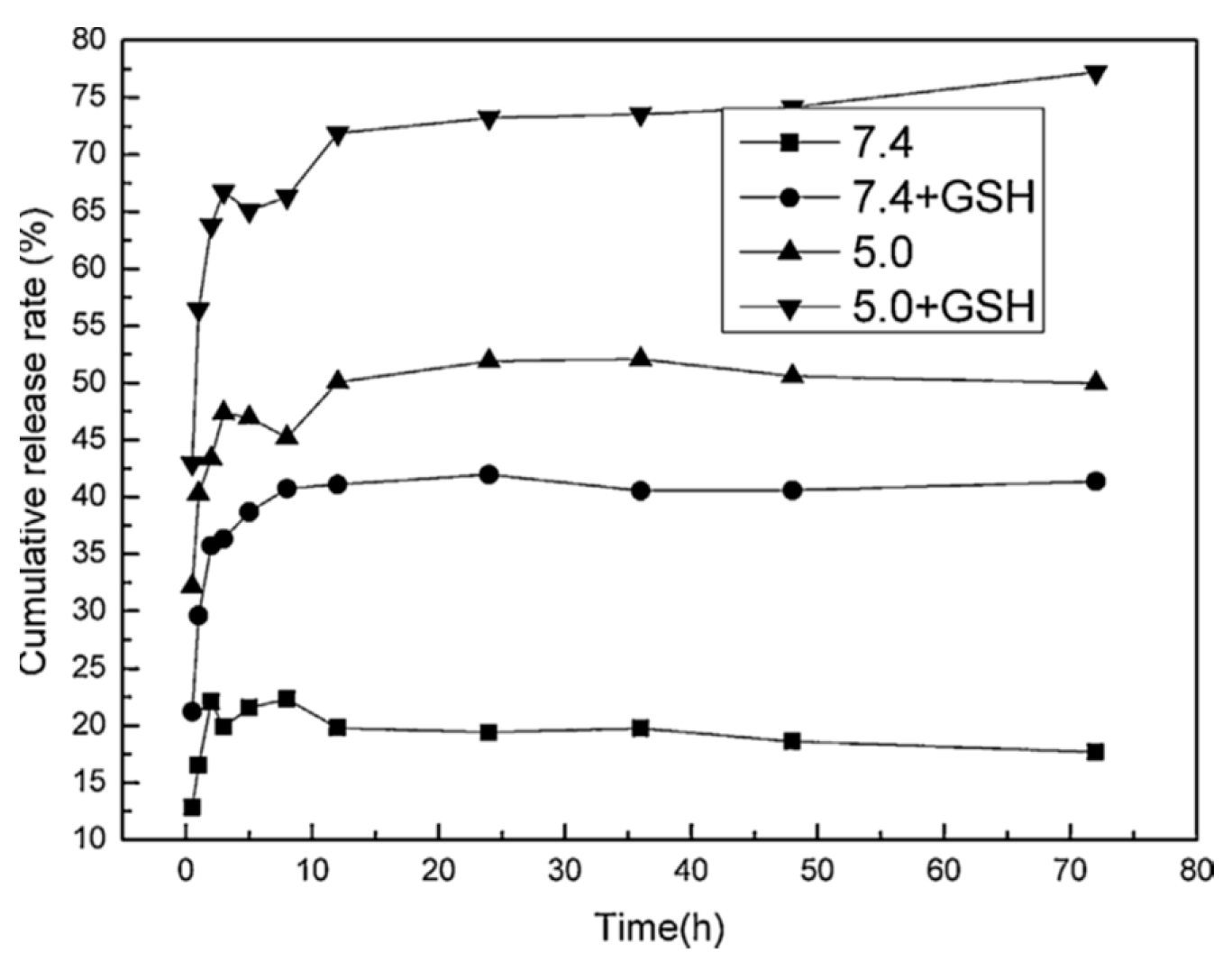
| Doxorubicin | Non-covalent imprinting MABHD, EDMA/DMAP, AIBN/ethanol | MIP-coated mesoporous silica NPs/- | Stimuli responsive release (pH, GSH) Drug load—10.5 ± 0.2 wt.% Drug release—72%/12 h (GSH 10 mM, pH 5, 37 °C) vs. 22% (no GSH, pH 7.4, 37 °C) and 42% (GSH, pH 7.4, 37 °C) | Yes/No | Material characterization In vitro release, cellular uptake cytotoxicity studies | [85] |
| Doxorubicin | Cu(II) mediated imprinting 4-Vpy, HEMA/MBA/APS, SBS/water | Hydrogel | Stimuli responsive release (pH) Drug load—6.74 μmol·g−1 Drug release: 10%/7 days (pH 7.2, 37 °C), 60%/7 days (pH 5, 37 °C) | Yes/No (acrylic Ms, high Cu(II) load) | Material characterization In vitro release studies | [80] |
| Doxorubicin and epitope of HER2 protein (Human epidermal growth factor) | Non-covalent imprinting Dopamine/water | Double imprinted MIP-coated mesoporous silica nanospheres/Targeted delivery of DOX (specific target—HER2) | Mechanism of release—diffusion/erosion Drug load Drug release—most of drug within 4 h, reaching a maximum after 24 h (pH 7.4, 37 °C) | Yes/Yes | Material characterization In vitro release studies In vivo studies: Female C57BL/6 nude mice—SKOV3 human ovarian cancer cells xenograft | [93] |
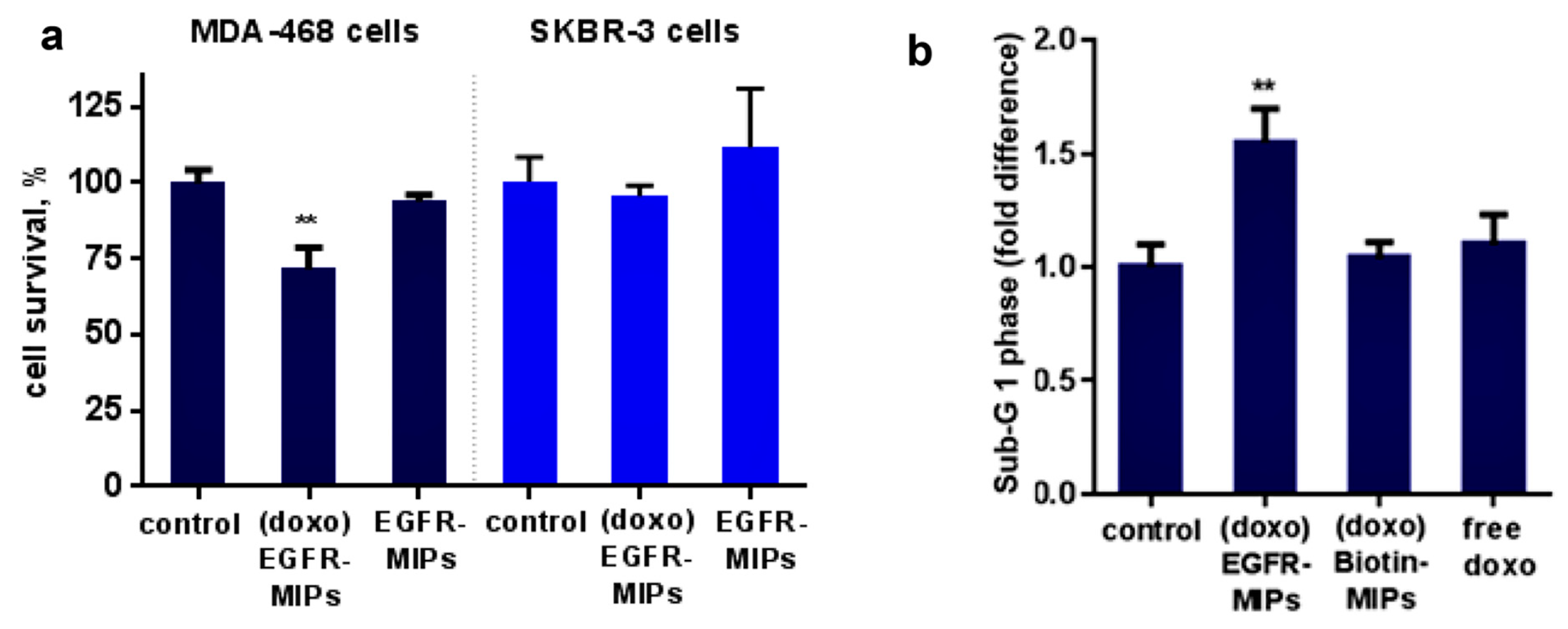
| Doxorubicin and epitope of EGFR (Epidermal growth factor receptor) | Non-covalent imprinting NIPA, N-tert-butylacrylamide, AA, N-(3-aminopropyl)methacrylamide/MBA/APS, TEMED/water | Double imprinted nanospheres Targeted delivery of DOX (specific target—EGFR) | Mechanism of release—diffusion/erosion Drug load Drug release | Yes/No (acrylic Ms) | Material characterization In vitro cellular uptake and cytotoxicity studies | [94] |
| Doxorubicin | Non-covalent imprinting NIPA/EDMA/AIBN/water + ethanol | MIP-coated Fe3O4 NPs/Magnetically assisted DD | Stimuli responsive release (pH) Drug release—70%/144 h (pH 5.8, 37 °C) vs. 12% (pH 7.4, 37 °C) | Yes/No (acrylic Ms) | Material characterization In vitro release studies | [110] |
| Doxorubicin | Non-covalent imprinting Dopamine/water | MIP-coated Fe3O4 NPs/Magnetically assisted DD | Mechanism of release—diffusion/erosion Drug release—90%/8 h (pH 7.4, 37 °C) | Yes/Yes | Material characterization In vitro release studies In vivo studies: BALB/C inbred female mice—papillary breast adenocarcinoma mammary tumor | [103] |
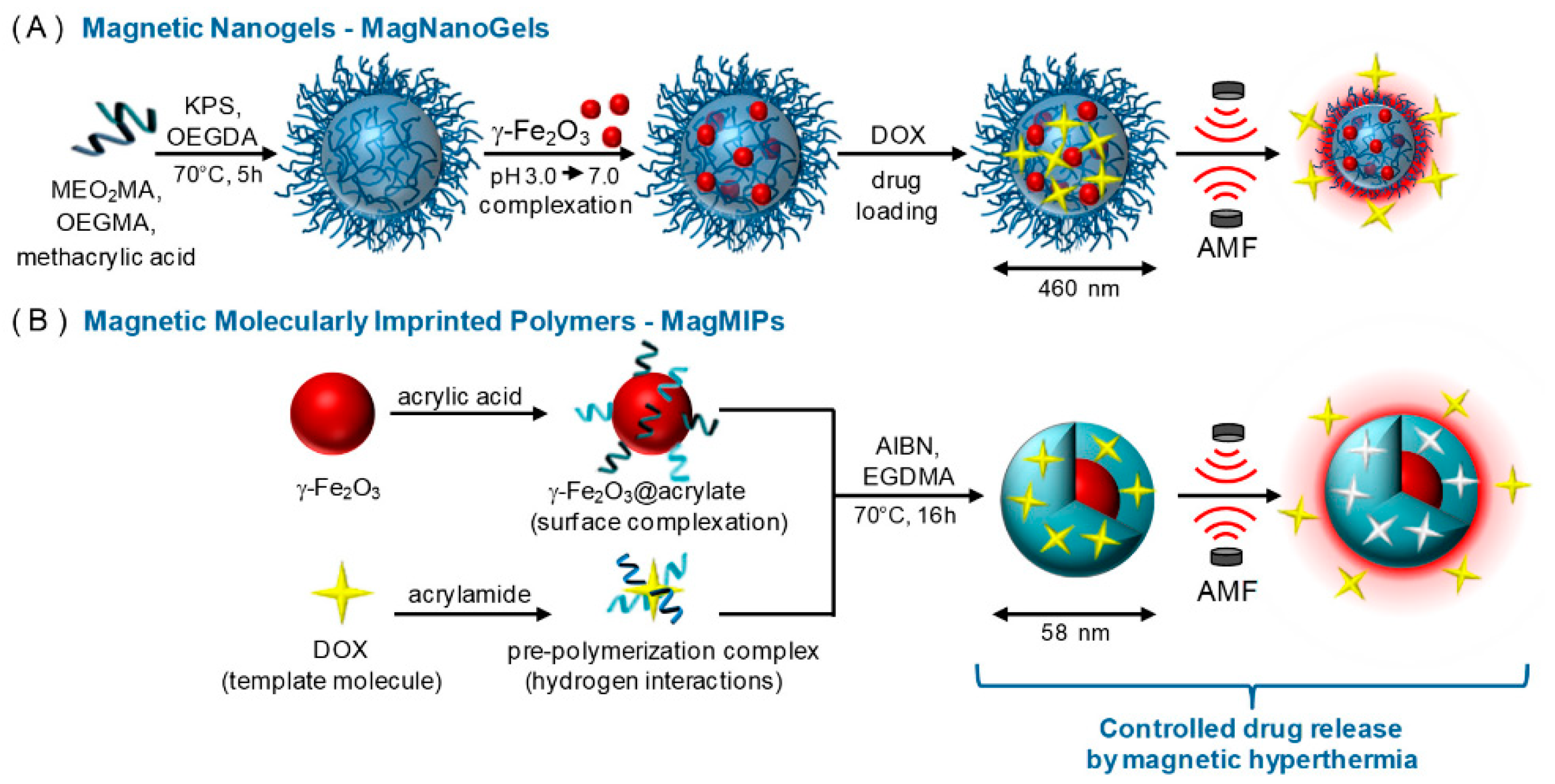
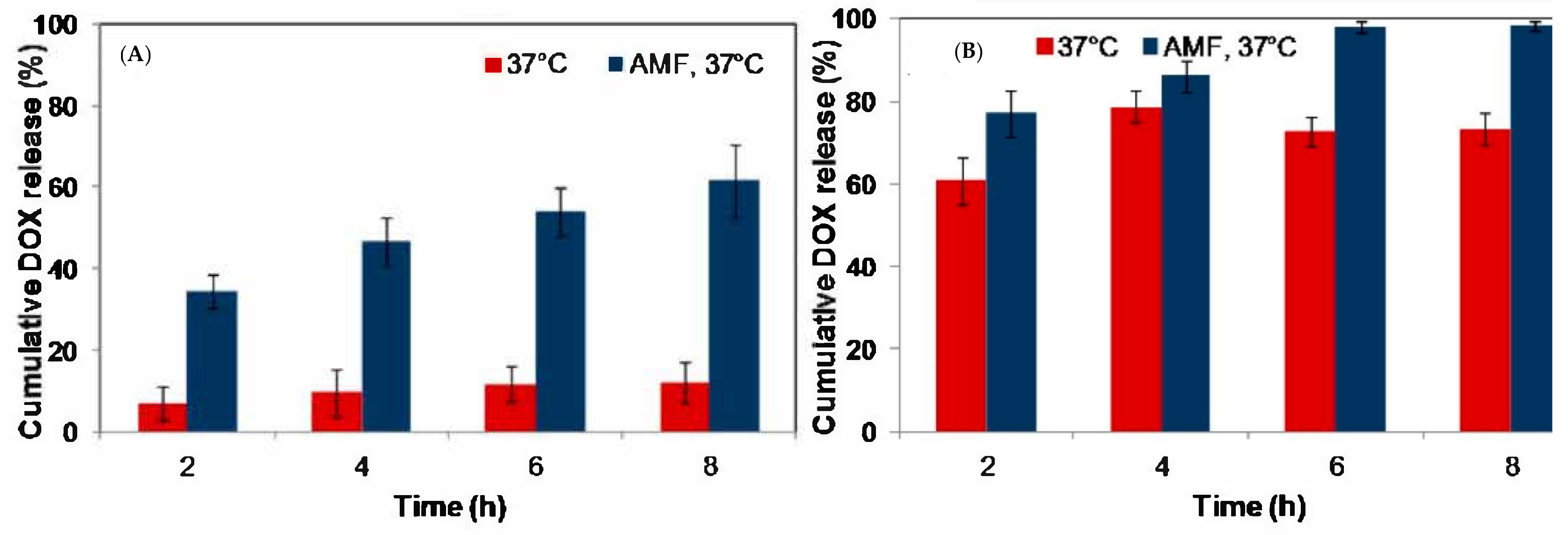
| Doxorubicin | Non-covalent imprinting AM, AA/EDMA/AIBN/ethanol | MIP-coated Fe3O4 NPs/Magnetically assisted DD | Alternative magnetic field (AMF)—controlled Drug release Drug load—35.6 μmol·g−1 Drug release—60%/8 h (AMF, 37 °C), vs. 12%/8 h (no AMF, 37 °C) | Yes/No (acrylic Ms) | Material characterization In vitro release studies In vitro cellular uptake and intracellular drug release studies | [68,87] |
| Doxorubicin | Non-covalent imprinting MMA/EDMA/AIBN/water-oil (cetyl alcohol) | MIP doped graphene oxide quantum dots (GQDs) microspheres/- | NIR radiation—controlled release (inductive NIR heating) Drug load—7.08 wt.% Drug release—36.54%/3 h (NIR radiation, pH 7.4) vs. 12%/3 h (NIR radiation, pH 7.4) | No/No (vinylic surfactant, (acrylic Ms) | Material characterization In vitro release studies | [111] |
| Paclitaxel | Non-covalent imprinting MAA, HEMA/EDMA or TRIM/AIBN/toluene | Microparticles/- | Mechanism of release—diffusion/erosion Drug load—13.32 mg/g (MIPTRIM); 9.86 mg/g (MIPEDMA) Drug release—85%/50 h (MIPTRIM, pH 7.4, 37 °C) and 40%/50 h (MIPEDMA, pH 7.4, 37 °C) | Yes/No (acrylic Ms) | Material characterization In vitro release, cytotoxicity studies | [55] |
| Paclitaxel | Non-covalent imprinting MPDE (LC), POSS, MAA/EDMA/AIBN/toluene and ACN | POSS-MPDE (LC) nanocomposite/- | Release mechanism—diffusion/erosion Drug load—106.93 μmol·g−1 Drug release—rate 4.6 μg/mL/15 h | Yes/No (acrylic Ms) | Material characterization In vitro release, cytotoxicity studies In vivo pharmacokinetic studies: Healthy male Wistar rats | [56] |
| Paclitaxel | Non-covalent imprinting MAA, MMA/EDMA/AIBN/hexadecane + chloroform and water + SDS | MIP NPs conjugated to PEG-FA (MIP-PEG-FA)/Targeted delivery of paclitaxel (specific target—the folate receptor) | Mechanism of release—diffusion/erosion Drug load—13.1 wt.% Drug release—11.2%/24 h (pH 5, 37 °C), 15%/100 h (pH 5, 37 °C) | Yes/No (acrylic Ms) | Material characterization In vitro release, cytotoxicity studies | [57] |
| Paclitaxel | Non-covalent imprinting M-POSS, MAA/EDMA /AIBN/ACN | M-POSS microparticles/- | Release mechanism—diffusion/erosion Drug load—17.1 wt.% Drug release—burst release: 10.7%/5 h (pH 5, 37 °C); 12%/100 h (pH 5, 37 °C) | Yes/No (acrylic Ms) | Material characterization In vitro release, cytotoxicity studies | [58] |
| Paclitaxel | Non-covalent imprinting MAA, MMA/EDMA, TRIM/AIBN/hexadecane, chloroform + water, SDS | NPs/- | Release mechanism—diffusion/erosion Drug load—17.8 wt.% Drug release—37.7%/48 h (pH 5, 37 °C) | Yes/No (acrylic Ms) | Material characterization In vitro release studies | [59] |
| Curcumin | Non-covalent imprinting (Dummy T: tea polyphenol) Gelatin-dextran conjugates/genipin/water | Polymeric micelles/- | Release mechanism—diffusion/erosion Drug load—100 mg/g Drug release: 54%/72 h (pH 2, 37 °C), 47%/72 h (pH 5, 37 °C), 60%/72 h (pH 6.8, 37 °C) | Yes/Yes | Material characterization In vitro release, cytotoxicity studies In vivo pharmacokinetic studies: Healthy male Sprague–Dawley rats | [95] |
| Curcumin | Non-covalent imprinting Acryl functionalized β-CD, NIPA/MBA/AIBN/ACN-free radical polymerization | MIP-coated Fe3O4@SiO2@MPS nanocomposite/Magnetically assisted DD | Stimuli responsive release (temperature); Drug load—77 mg/g Drug release—burst release 45%/7 h (25 °C and 38 °C); 62%/3 days (25 °C), 86%/3 days (38 °C) | Yes/No (acrylic Ms) | Material characterization In vitro release studies | [89] |
| Azidothymidine | Non-covalent imprinting ITC/EDMA/AIBN/ACN | MIP-coated Fe3O4@SiO2—MPS NPs/Magnetically assisted DD | Stimuli responsive release (pH) Drug load—170.75 mg/g Drug release—burst release 80%/10 h (pH 5, 37 °C), 90%/75 h (pH 5, 37 °C); 15%/75 h (pH 7.4, 37 °C) | Yes/No (acrylic C)) | Material characterization In vitro release, cytotoxicity studies | [69] |
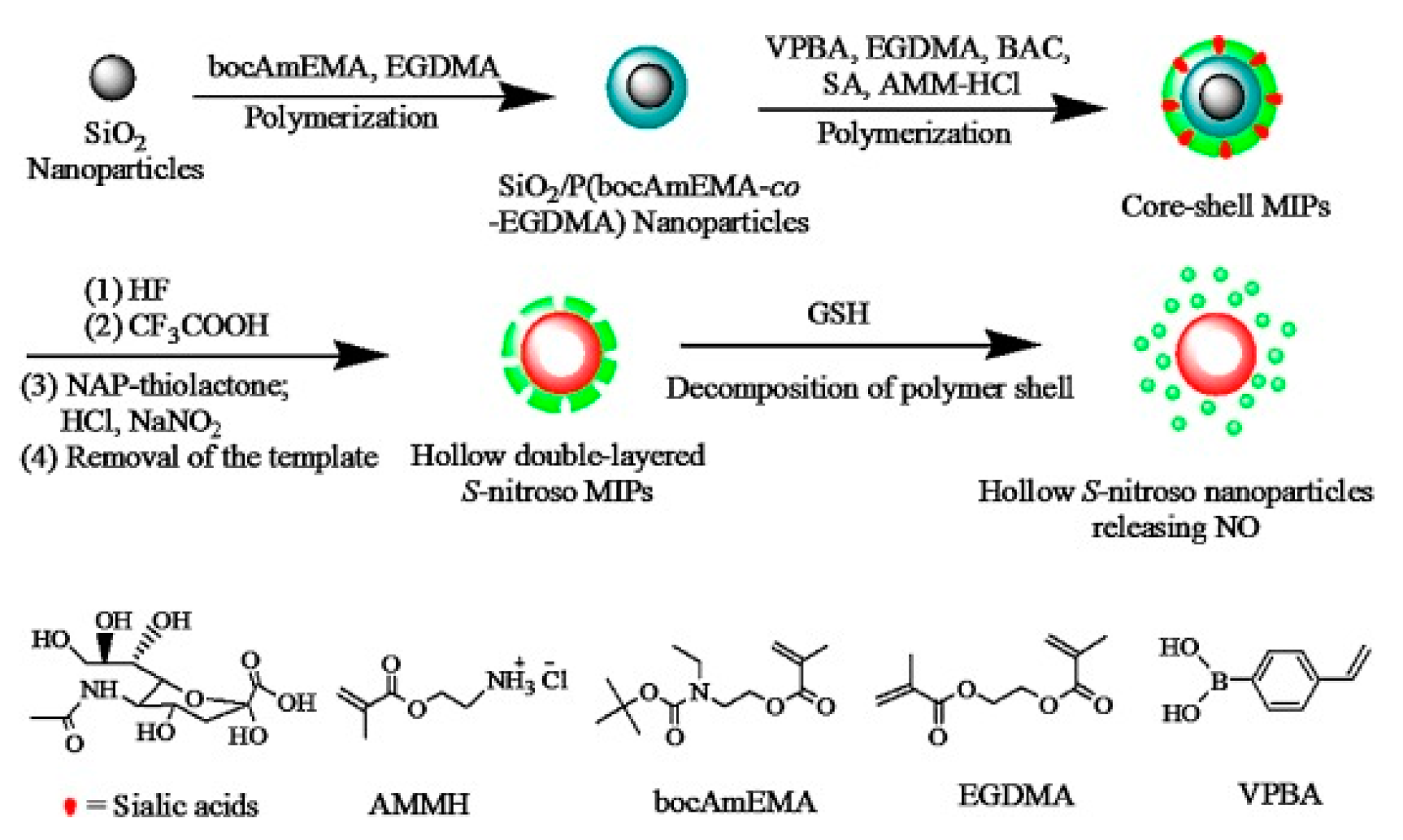
| Sialic acid/S-nitrosothiols | Non-covalent imprinting VPBA, AMMH/BAC, EDMA/BPO/ACN | MIP-coated SiO2/P(EDMA-co-bocAmEMA) NPs/Targeted delivery of S-nitrosothiols (thiol mediated cell uptake, specific target—sialic acid over-expressed on cancer cell membrane) | Stimuli responsive release (GSH or Cu(I) triggered release of nitrous oxide, NO) Drug load—2.1 μmol·g−1 Drug release—1.8 μmol·mg−1, t1/2 = 220 min, (200 µM Cu(I), pH 7.4, 37 °C); 1.7 μmol·mg−1, t1/2 = 145 min, (5 mM GSH, pH 7.4, 37 °C); 0.3 μmol·mg−1, t1/2 = 445 min, (3 mM GSH, pH 7.4, 37 °C) | Yes/No (acrylic Ms) | Material characterization In vitro release, cytotoxicity studies | [76] |
| R-(+)-thalidomide S-(-)-thalidomide S,R-(±)-thalidomide | Non-covalent imprinting NVP, MAA, AA/EDMA, TRIM/AIBN/chloroform | Microspheres/- | Release mechanism—diffusion/erosion Drug load Drug release—15%/75 h (R-(+)-thalidomide MIP, pH 5.5), 17%/75 h (S-(-)-thalidomide and S,R-(±)-thalidomide, pH 5.5) | Yes/No (acrylic Ms) | Material characterization In vitro release, cytotoxicity studies | [61] |
| R-(+)-thalidomide | Non-covalent imprinting MAA, 2,6-bis(acrylamido)Py/MBA/AIBN/methanol | MIP-Poloxamer NPs (Physically deposited MIP-Poloxamer 407, or chemically grafted MIP-acrylate-derived Poloxamer)/- | Stimuli responsive release (temperature); Drug load—3.1 μmol·g−1 Drug release | Yes/No (acrylic Ms) | Material characterization In vitro release, cytotoxicity studies | [60] |
| Mitoxantron | Non-covalent imprinting MAA/polycaprolactone diacylate/AIBN/DMSO + chloroform | MIP-coated TiO2 NPs/- | Photodynamic effect-based release Drug load Drug release | Yes/No (acrylic Ms) | Material characterization In vitro cytotoxicity studies | [90] |
| Sunitinib | Non-covalent imprinting MAA/EDMA/AIBN/chloroform | Hydrogel/- | Swelling-controlled drug release Drug load Drug release—58%/6 h; 76%/24 h | Yes/No (acrylic Ms) | Material characterization In vitro release, cytotoxicity studies | [63] |
| Mitomycin C | Non-covalent imprinting MAH, HEMA/EDMA/KPS/water + PVA | MIP-coated Fe3O4 NPs/Magnetically assisted DD | Swelling-controlled drug release Drug load—24 μmol·g−1 Drug release—90%/5 h (pH 6, 25 °C, mitomycin C load 8 mg/g) | Yes/No (acrylic Ms) | Material characterization In vitro release studies | [104] |
| Mitomycin C | Cu(II) mediated imprinting MAH, HEMA/MBA/APS, TEMED/phosphate-buffered saline | Implantable cryogel membranes | Swelling-controlled drug release Drug load—8 mg·g−1 Drug release—92.5%/5 h (pH 7.4, 37 °C) | Yes/No (acrylic Ms, high Cu(II) load) | Material characterization In vitro release, cytotoxicity studies | [81] |
| Amygdalin | Non-covalent imprinting 4-Vpy/ EDMA/BPO/ACN | NPs/- | Swelling-controlled drug release Drug load—0.98 mg/g Drug release—50%/24 h (pH 7, 37 °C), 35%/24 h (pH 2, 37 °C) | Yes/No (acrylic Ms) | Material characterization In vitro release, cytotoxicity studies | [72] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bodoki, A.E.; Iacob, B.-C.; Bodoki, E. Perspectives of Molecularly Imprinted Polymer-Based Drug Delivery Systems in Cancer Therapy. Polymers 2019, 11, 2085. https://doi.org/10.3390/polym11122085
Bodoki AE, Iacob B-C, Bodoki E. Perspectives of Molecularly Imprinted Polymer-Based Drug Delivery Systems in Cancer Therapy. Polymers. 2019; 11(12):2085. https://doi.org/10.3390/polym11122085
Chicago/Turabian StyleBodoki, Andreea Elena, Bogdan-Cezar Iacob, and Ede Bodoki. 2019. "Perspectives of Molecularly Imprinted Polymer-Based Drug Delivery Systems in Cancer Therapy" Polymers 11, no. 12: 2085. https://doi.org/10.3390/polym11122085
APA StyleBodoki, A. E., Iacob, B.-C., & Bodoki, E. (2019). Perspectives of Molecularly Imprinted Polymer-Based Drug Delivery Systems in Cancer Therapy. Polymers, 11(12), 2085. https://doi.org/10.3390/polym11122085








