Abstract
Energy production, distribution, and storage remain paramount to a variety of applications that reflect on our daily lives, from renewable energy systems, to electric vehicles and consumer electronics. Hydrogen is the sole element promising high energy, emission-free, and sustainable energy, and metal hydrides in particular have been investigated as promising materials for this purpose. While offering the highest gravimetric and volumetric hydrogen storage capacity of all known materials, metal hydrides are plagued by some serious deficiencies, such as poor kinetics, high activation energies that lead to high operating temperatures, poor recyclability, and/or stability, while environmental considerations related to the treatment of end-of-life fuel disposal are also of concern. A strategy to overcome these limitations is offered by nanotechnology, namely embedding reactive hydride compounds in nanosized supports such as graphene. Graphene is a 2D carbon material featuring unique mechanical, thermal, and electronic properties, which all recommend its use as the support for metal hydrides. With its high surface area, excellent mechanical strength, and thermal conductivity parameters, graphene can serve as the support for simple and complex hydrides as well as RHC (reactive hydride composites), producing nanocomposites with very attractive hydrogen storage properties.
Keywords:
graphene; energy storage; hydrogen; metal hydrides; composites; nanoconfinement; supercapacitor; batteries 1. Introduction
Energy storage is a key driver and supporter of the everyday needs of society. Within this context, metal hydrides are promising systems with the ability to store and release hydrogen gas, the sole element promising a sustainable, emission-free future [1,2,3,4,5,6,7,8,9]. While there are many binary and complex hydrides known, only those belonging to lightweight metals have practical implications due to their high gravimetric and volumetric H2 content, in line with DOE’s current targets. However, their slow reaction kinetics, possible side reactions, and poor recyclability have prompted researchers to find mitigation strategies to overcome these shortcomings. The recent advances in SSHS (solid-state hydrogen storage) materials have been summarized, and they entail both physisorption and chemisorption of hydrogen [10]. In contrast to conventional systems based on high-pressure gas phase or low-temperature liquid-state hydrogen, SSHS systems have the tangible potential to offer high hydrogen storage capacity, sustainable performance, recyclability with little loss in storage capacity, as well as manageable production costs [1,2,3,4,5,6,7,8,9,10].
Among recent advances, nanoconfinement in a properly chosen support has shown important improvements regarding stability, thermodynamic parameters, and overall kinetics, oftentimes affording reversible systems where pristine hydrides showed little or no reversibility. For instance, MgH2, which arguably is the metal hydride most investigated to date, features too high energy barriers (ΔH = −75 kJ/mol, Tdes = 573 K) to be considered in its neat form in hydrogen storage tanks as such. Additionally, the dehydrogenated material (Mg) is a very reactive metal (εox = +2.37 V), susceptible to oxidation by even minute oxygen traces, which would lead to MgO and, upon moisture exposure, to Mg(OH)2; this way, the reversibility of the cycle Mg/MgH2 would be lost. Additionally, bare MgH2 is also plagued by particle agglomeration during cycling, which can lead to a continuous decrease in H2 storage capacity and even slower kinetics. These downsides observed for the model system Mg/MgH2 have prompted scientists to further tune the thermodynamic parameters by altering the morphological features of MgH2. Nanostructuring, alloying, and compounding with additives or catalysts are among the most utilized mitigating strategies.
Confinement in nano-sized supports would afford better particle size control upon absorption/desorption a/d cycling, along with increased stability due to the confinement matrix (typically silica or carbon). Furthermore, additional hydride may support interactions that further decrease total system energy. While many types of carbo materials have been investigated as supports (OMC-ordered mesoporous carbon, AC activated carbon, carbon nanotubes CNTs, etc.), graphene has emerged as a two-dimensional carbon material with unique electronic, mechanical, and thermal properties that can act as an efficient support material for metal hydrides. Several properties are essential to accomplishing this task: its large surface area (in excess of 2500 m2/g), the layered structure forbidding hydride NP from agglomeration, high electrical conductivity, and excellent mechanical strength. The inclusion of hydride materials into nanoporous scaffolds is also an important aspect; while ball milling (BM) seems to be the prevalent method, the incipient wetness/infiltration method and in situ generation of hydride materials inside the matrix are gaining momentum, especially due to the better dispersion and polydispersity of NPs produced by the latter methods. With a higher surface-to-volume ratio, hydride NPs are expected to behave differently in hydrogenation studies, with lower enthalpies compared to bulk and enhanced kinetics due in part to more efficient H2 diffusion in the thin hydride layer of a few nm [1,2,3]. Solvent infiltration/evaporation is more commonly used in the case of complex hydride solutions, given their higher solubility in ethereal solutions such as MTBE and similar inert ethers [2,3,4,5,6].
The current review aims to discuss the recent advances in graphene-supported metal hydrides for energy storage applications, covering the main features of composite systems based on graphene (G, GO, and related) and metal hydrides (LiH, MgH2, LiBH4, LiAlH4, NaBH4, NaAlH4, Mg(BH4)2, Mg2NiH4, Mn(BH4)2, and others) [11]. The applications of the generated nanocomposites have also been briefly described, with an emphasis on the energy storage properties of said composites. The conclusions and future outlook directions identify some areas where improvement strategies may yield enhanced energy storage properties of hydride/graphene composites, as well as raising awareness regarding the proper handling and processing of end-of-life composite energy materials, a prerequisite for a sustainable future built around a hydrogen economy.
2. Overview of Metal Hydrides and Graphene Supports
In an effort to reduce CO2 emissions and the subsequent greenhouse effect, H2 production can be achieved from renewable sources (biomass, water) by electrolysis, thermolysis, or photocatalytic splitting [11]. However, the lightweight and limited space requirements for a viable vehicular fuel have shifted attention towards solid-state hydrogen materials, and metal hydrides are an excellent example of such materials. However, metal hydrides do not cumulatively meet the requirements for high gravimetric content (~6.5 wt.%, ideally), fast and reliable recycling behavior (over 1000 cycles without significant degradation), and moderate thermodynamic parameters (enthalpies around 30 kJ/mol) that would allow operation under the expected range (−40…100 °C). Some of these shortcomings can be alleviated by the inclusion of the active hydrogen-storing compound in a nanoporous matrix such as graphene [12]. Graphene is a 2D material with very appealing properties, highlighting its potential use as support for various reactive species, including metals and metal hydrides [13]. By embedding hydride species into graphene supports, valuable nanocomposites can be obtained with direct use for energy storage applications (Figure 1) [11,12,13].
Figure 1.
Graphene sits at the core of various improvement strategies for better and more effective embedding of active hydrogen storage materials.
Engineering MHx@G composites has been no easy task, but a vast majority of researchers have turned their attention towards the Mg/MgH2 system, in part due to its favorable thermodynamic properties (ΔH = 65.8–75.2 kJ/mol), high H2 storage capacity (7.7 wt.%, 109 g H2/L), and most important, due to the reversible nature of the hydrogenation/dehydrogenation process [14,15,16]. Assessing the support contribution to H2 storage is equally importantly, and functionalization strategies have yielded interesting results in ball-milled Mg-B-electrochemically synthesized reduced graphene oxide (erGO) with enhanced H-uptake kinetics [17]. B-doped graphene has also been investigated by density-functional theory DFT studies [18]. Tan et al. have shown, using principal calculations, that the adsorption energy of H2…B-doped G can be tuned, enhancing the interaction by positively charging the support. BC5, for instance, can store up to 5.3 wt.% H2. More significantly, the study sheds light on the heteroatom doping of graphene; B-doped G forms easier than N-doped G (5.6 eV/atom for the former vs. 8.0 eV/atom for the latter) [18]. Combining metal doping (Ni) with B-doped carbon-based materials, Wang et al. studied and compared various forms of carbon with those of 3D graphene-based materials, obtaining composites with surfaces functionalized with O-containing groups that further enhanced the spillover effect with a positive effect on H2 storage properties [19]. Edge-functionalization of G for kinetic control of HSM (hydrogen storage materials) has also been reported [20].
Among various strategies to improve the behavior of hydrogen storage materials at the nanoscale, nanoconfinement and nanostructuring were shown to be very effective [2,21,22]. Nanostructured metal hydrides MHx showed a tangible improvement in H2 storage properties [2,21], while nanostructures of type hydride @ C in particular were proven to be a model system for the future solid-state hydrogen storage fuel, as evidenced by DFT computation by Shevlin and Guo in 2008 [22]. DFT simulations have played a pivotal role in the expansion of the hydrogen storage field, the above-mentioned resource comparing a few of the most important hydrides: MgH2, LiNH2, LiBH4, and derived reactive hydride composites RHCs, as well as their interaction with various carbon systems [22].
2.1. Metal-Decorated Graphene
An important strategy for tuning graphene for energy storage applications has been to decorate the support with metals or clusters of metals. Among the investigated materials are metallized siligraphene nanosheets (SiG) with varying light metal decorations (Li, Na, K, Mg, Ca, Sc, Ti) [23], metal-decorated graphene (Li, Na, Mg)/G, DFT study [24], or K in K @ B-substituted G [25]. Given the potential of AlH3 for hydrogen storage, Al-doping has also been explored in Al/G composites [26,27], Aln clusters supported by coronene and graphene G (DFT study) [27], and Al/Si –SLG (SLG, single layer graphene + Si +Al) [28].
Palladium is well known for its high affinity for H2, and many studies have been devoted to the theoretical modelling of this interaction; Pd-decorated N-doped G, DFT study [29]; Pdn @ G (n = 1–4) in BC3 variant [30], and Cu- and Pd-decorated G, DFT study [31]. Titanium and its clusters have also been investigated: Ti3 clusters [32]; Ti4—decorated B/N-doped G [33]; Ti4 & Ni4–doped G nanoplatelets [34]; and Ti–Al subnanoclusters on G [35].
Most reports, however, focus on Mg-doped porous carbonaceous materials, such as Mg@ G flake nanocomposites (H2 generation from H2O) [36], Mg@graphite for comparison purposes [37], Mg@rGO layers [38,39], Mg@Heteroatom–doped G [40], Mg@B–doped G [41], and Mg/defected GO [42]. Additionally, various alloys have also been studied for graphene supports: Mg alloy @rGO–V2O3 [43], rGO–EC@AB5 hybrid material (EC = ethyl cellulose, AB5 = La(Ni0.95Fe0.05)5) (LNF) [44], or MmNi3.55Co0.75Mn0.4Al0.3/G nanoplatelets (Mm denotes mischmetals) [45].
2.2. Mechanistic Insight and Kinetics of H2…Support Interaction
Pristine graphene can chemically absorb H2 and its theoretical storage capacity is 7.7 wt.%; the hydrogenated graphene (graphane, (CH)n, a sp3 hybridized analog of graphene) releases H2(g) at ~400 °C, with an Ea = 158 kJ/mol (1.64 eV) [46]. While an intriguing material in its pristine form, its thermodynamic parameters make it less feasible for scaling up processes aimed at vehicular applications; however, it is worth noting the similarity of the activation energy deduced for graphane and that of metal hydrides.
The fundamental understanding of the adsorption/desorption mechanism of H2 in graphene is paramount to developing new materials aimed at this task; a pertinent comparison between physisorption and chemisorption on graphene was reported in 2011, where the physical limitations of G (5 wt.% H2 storage) were correlated to the entropy contribution TΔS and the large van der Waals distance between two H2 molecules (0.3 nm), further preventing the increase in gravimetric storage capacity of pristine graphene [7]. The interaction H2…G was studied by DFT in single- and double-vacancy graphene by Wu et al., with direct implications for the behavior of defected graphene during hydrogenation studies [47]. The mechanism of H2 interaction with Al-doped porous graphene has been reported by Ao et al., showing by DFT that Al/G can store up to 10.5 wt.% H2, with a relatively low H2 adsorption energy of −1.11 to −0.41 eV/H2, which would potentially allow hydrogen absorption/desorption near room temperature conditions, in agreement with the findings from the analysis of atomic charges, electronic distribution, and density of states (DOS) of the system [26]. The enhanced interaction was potentially due to the polarization of the adsorbed H2 molecules.
Akilan et al. have studied by DFT the adsorption of H2 molecules on B/N-doped defected (5-8-5, 55-77, 555-777 and 5555-6-7777 defects) graphene sheets [48]. The N-atom addition (donor behavior, n-type semiconductor) increases the delocalized electrons, while the B atoms (acceptor, p-type semiconductor) increase the localized electrons in the considered system. The most efficient adsorption was modelled when the H2 molecule approached the sheet in a perpendicular direction (−80 meV), while the least efficient interaction was observed in a parallel orientation (−9 meV), while the delocalized electron density was higher on the fusion points of the pentagonal and hexagonal rings and would therefore enhance H2 adsorption [48]. Another supporting DFT study of H2 storage on TM-doped defected graphene (TM = transition metal) revealed that in the case of TM = Sc, the 555-777/Sc structure doped with Sc showed the maximum H2 capacity, with H2 binding energies in the range 0.2–0.4 eV/H2 [49].
The advances regarding TM-loaded Mg-based alloys/G have been reviewed recently [8]. A few important points are attributed to graphene: It can inhibit grain growth, thus aiding the overall cyclability of the composite, and it can (co-)catalyze the hydrogenation process, in which the electron transfer between Mg and C plays a key role [8].
The cyclic behavior of metal hydrides can be affected by issues related to grain growth. This can be partly overcome with the formation of G layers encapsulating MgH2 to prevent grain growth [50]. In this report, Lototskyy et al. used various carbon sources (graphite, AC, MWCNTs, etc.) and showed that the formation of graphene sheets during high-energy reactive ball milling in hydrogen (HRBM) is responsible for the encapsulation of MgH2, noting an increase in a/d cycling behavior along with a more reduced size of the MgH2 crystallites (40–125 nm vs. 180 nm in pristine form) [50]. The catalytic role of graphene nanoplatelets (GNP) over H2 storage kinetics in Mg has been studied by Ruse et al. [51]. The enhancement of more than an order of magnitude was attributed to GNP properties (size, thickness, defect density, and specific surface area), and these can be further tuned to alter H2 storage kinetics in Mg–GNP nanocomposites [51]. A carbon-neutral, reversible, and sustainable process that produces H2 is the formate-bicarbonate system, where graphene has also served as a support of Pd and Ru metals [52].
2.3. Manufacturing Techniques
Several techniques have been utilized to introduce metal catalysts into graphene, synthesizing (nano)composites containing graphene and carbonaceous materials [9]. While ball milling and its variants remain a key technique, other options have been explored: Electrostatic layer-by-layer self-assembled G/MWCNTs [53], Uranium U-decorated G (H2 and D2 adsorption) [54], and plasma-assisted milling in Mg@FLG composites (few-layer graphene nanosheets) [55]. Given the remarkable properties of 2D graphene on hydride storing materials, the synthesis of 2D MgH2 has also been proposed in DFT studies [56].
3. Applications of Graphene-Based Hydride Nanocomposites
Graphene-supported metal hydrides have potential applications in hydrogen storage, battery electrodes, and fuel cells, among others. These are mainly the contributions of the embedded metal hydrides that have been utilized in energy storage devices such as batteries and fuel cells. By embedding metal hydrides in high-surface-area graphene, the obtained nanocomposites can further enhance the performance and efficiency of pristine hydrides, potentially yielding competitive novel energy storage devices with improved cycling stability and durability [2,21].
3.1. Batteries—Battery Electrodes
Graphene-supported metal hydrides have also been studied as electrode materials in batteries and fuel cells. The addition of graphene to metal hydride electrodes can improve their cycling stability and reduce their electrode polarization. This improves the efficiency and lifespan of the batteries and fuel cells. The high surface area of graphene also enhances the electrochemical performance of the electrodes by providing more active sites for electrochemical reactions.
Composites such as MHx@G have been utilized for batteries [57,58,59]. Mixed-valence oxides (Co3O4) and hydroxides (Ni(OH)2) have also been explored in Co3O4 nanocubes on N-doped G for Ni-metal hydride batteries [60] and Ni(OH)2-coated N-doped G aerogel as replacement NiCd and NiMH batteries [61]. Various anode materials based on graphene have recently emerged, such as MgH2 self-assembled on G as anode material for Li-ion batteries [62].
3.2. (Super)capacitors, Electrochemical Storage
Supercapacitors are another energy storage device that can benefit from the addition of graphene-supported metal hydrides in MHx@G composites [57,58]. Supercapacitors are electrochemical devices that can store energy by electrostatically adsorbing ions at the surface of the electrode [53]. The improvement of using Li-ion (120–170 Wh/Kg) vs. Ni-metal hydride (40–100 Wh/Kg) has to be considered when designing new storage devices [58]. The high surface area and conductivity of graphene can enhance the electrochemical performance of supercapacitors. The addition of metal hydride nanoparticles to the graphene electrodes can further enhance the energy storage capacity and stability of supercapacitors. Other materials based on graphene have also been investigated, for instance Fe3O4 @ G composite for high-performance Li-ion hybrid capacitors [63]. Electrochemical storage properties have also been studied on heteroatom-substituted materials, such as N-doped C [64], but also on La0.7Mg0.3(Ni0.85Co0.15)3.5@Ni/Graphene [65].
3.3. Solar Cells and Portable Electronic Devices
Graphene-supported metal hydrides have also been studied for their application in solar cells. The addition of metal hydrides to the graphene electrodes in solar cells can improve their photovoltaic performance by enhancing charge separation and transport properties. The high conductivity and large surface area of graphene also facilitate electron transfer and reduce recombination losses in the solar cells. In addition to the above applications, graphene-supported metal hydrides have also been studied for their potential use in energy storage for portable electronic devices, power grid stabilization, and energy harvesting. The unique properties of graphene and metal hydrides can be tailored to meet the specific requirements of these applications and enhance their performance [2].
3.4. Energy Storage
One of the most promising applications of graphene-supported metal hydrides is in hydrogen storage for fuel cell vehicles. Fuel cell vehicles are gaining popularity due to their high efficiency and low emissions. However, the limited availability of hydrogen storage materials has hindered their widespread adoption. This aspect will be discussed in more detail in the following section (Section 4) [2,21].
4. Hydrogen Storage Properties of Composites MHx@G
Metal hydrides have shown promise as hydrogen storage materials, but their low hydrogen storage capacity and slow kinetics have limited their application. The addition of graphene as a support material can enhance the hydrogen storage capacity and kinetics of metal hydrides, making them a promising candidate for hydrogen storage in fuel cell vehicles.
4.1. Binary Hydrides—The Case of MgH2
While light metal hydrides show great promise as H2-storing materials, the literature and scientific research are still focused on MgH2 due to its advantageous gravimetric content, activation energies for desorption/adsorption, and multiple synthesis methods. The following subsections explore the H2 storage properties of non-catalyzed and catalyzed nanocomposites such as MgH2@G.
4.1.1. Non-Catalyzed Support
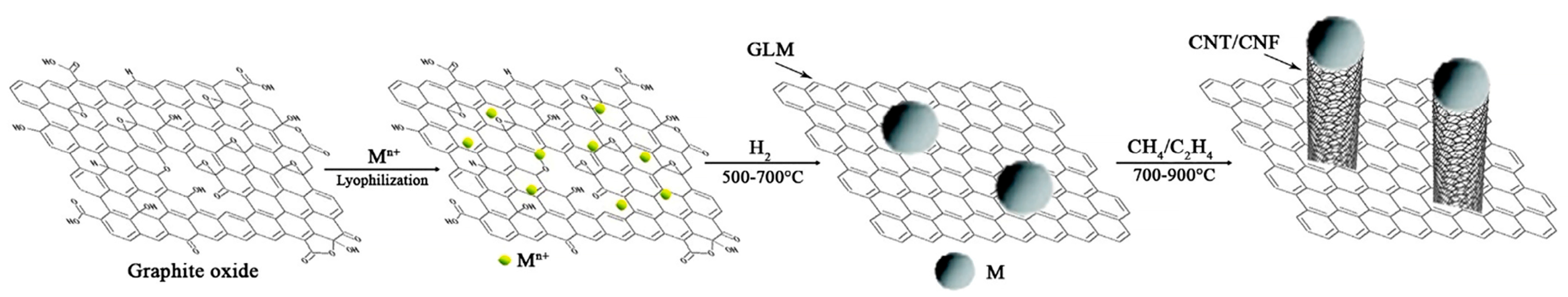
A great variety of reports in the literature focus on the Mg/MgH2 system embedded in various supports, including graphene [1] and GLM (graphene-like materials) (Figure 2) [16], although some other binary hydrides (layer-by-layer lithiated composites such as LiH/G for instance) have also been reported [66]. For instance, MgH2 NPs nanoparticles have been embedded in GNS (graphene nanosheets) to yield MgH2 NPs@GNS composites [67], MgH2 NPs nanoparticles uniformly-grown@G [68].
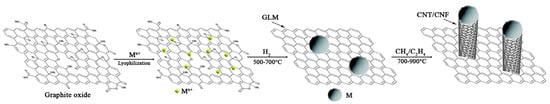
Figure 2.
Synthesis of metal-carbon-graphene composites. Reprinted from ref. [16] under Creative Commons Attribution (CC BY) license.
The mechanism of MgH2@G composite synthesis by reactive mechanical grinding (under 2 MPa H2) has been studied by Jang et al., who concluded that G played a catalytic role in H2 absorption, considering the absorption kinetics and PCT (pressure-composition-temperature) curves [69]. The composite MgH2–5 wt.% graphene could store 3.69 wt.% H2 at 423 K, while increasing the graphene amount in MgH2–5 wt.% graphene allowed 5.08 wt.% at the same temperature (423 K) [69]. Comparing the PCI isotherms for the two samples, it seemed that MgH2–10 wt.% graphene displays a lower activation energy Ea and better cyclic stability compared to MgH2–5 wt.% graphene [69].
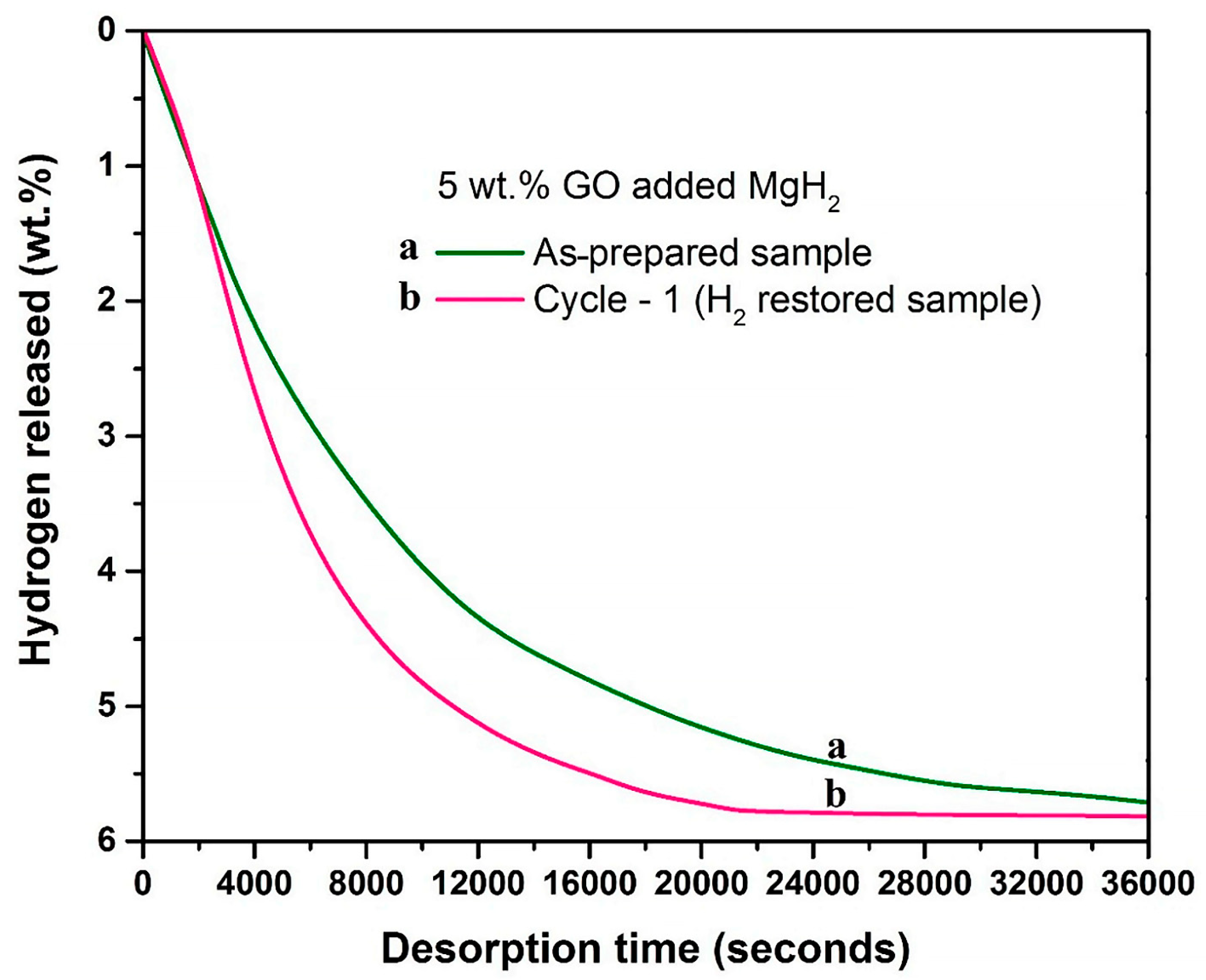
Another strategy to enhance the stability of MgH2 during cycling was to create Mg/rGO multilaminates to produce Mg/MgH2@rGO during cycling. These composites exhibited a very high 6.5 wt.% reversible H2 storage and a capacity of 0.105 Kg H2/L [70]. When MgH2 interacts with 5 wt.% GO incorporated in MgH2 during hydrogenation studies, it also causes a reduction in the porous support (GO→rGO), as highlighted by a study by Pukazhselvan et al. (Figure 3) [71].
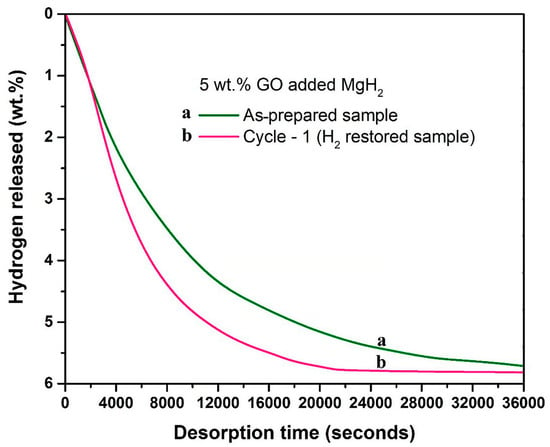
Figure 3.
Dehydrogenation kinetics profiles obtained for 5 wt.% GO incorporated MgH2. Profile “a’’ corresponds to the as-prepared sample, and profile “b” corresponds to the hydrogen-restored sample. Dehydrogenation temperature/pressure: 315 °C/1 bar. Reprinted from reference [71] under Creative Commons Attribution (CC BY) license.
The reduction occurs even during composite preparation, namely during mechanical milling, and it affects the dehydrogenation temperature of MgH2 (a reduction of 60 °C vs. pristine) by lowering the dehydrogenation activation energy (by 20 kJ/mol). Considering the affinity of Mg for oxygen, this could also be used as a method to obtain MgO-decked rGO composites [71]. Various carbon scaffolds have been used for MgH2, including but not limited to graphite, graphene derivatives, and carbon fiber [72]. Combined experimental and DFT studies on the helical form of graphene nanofibers (HGNF) by Singh et al. [73] revealed that the extra carbon sheet edges exposed would further improve MgH2 dehydrogenation compared to G-catalyzed MgH2, and the experimental results showed a 45 °C improvement due to HGNF use in MgH2 + 7 wt.% HGNF nanocomposites. The DFT results supported this finding by revealing a weakening of Mg-H bonds by interaction with zigzag and armchair type HGNF, forming C–Mg–H and C–H bonds [73]. Combined DFT and experimental computations have also been performed on other MgH2@G variants, which confirmed once again the catalytic role of G in reducing the dehydrogenation enthalpy and dehydrogenation activation energy of MgH2 [74].
Other approaches tackle graphene support functionalization, as is the case of MgH2–FG (FG = fluorographene nanosheets). Using fluorographene, the nanocomposite MgH2–FG absorbed 6.0 wt.% within 5 min, and desorbed 5.9 wt.% within 50 min, at 300 °C [75].
4.1.2. Catalyzed Support
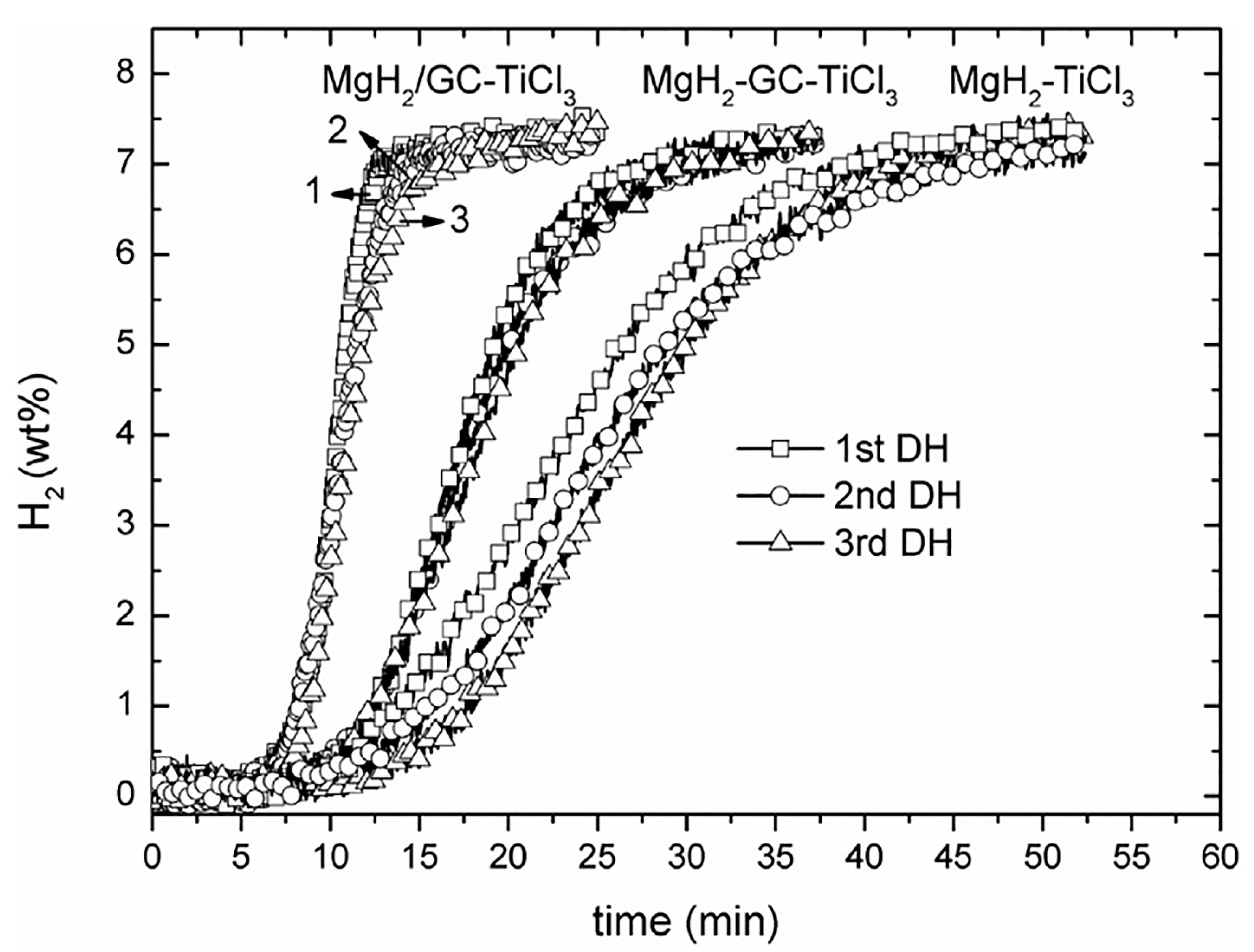
Various catalysts have been utilized to improve the behavior of hydride@graphene composites. For instance, MgH2 catalyzed by TiH2@Gr (desorption onset at 204 °C), with comparative improvements relative to the catalytic activity of Ti@Gr, TiO2@Gr, and TiH2@Gr [76]. Other Ti-based salts such as TiCl3 have been used in MgH2 catalyzed by TiCl3 and GO-based porous carbon (GC) in composites such as MgH2/GC-TiCl3, obtained by isothermal hydrogenation of MgBu2/GC, first to yield MgH2/GC and then ball milling with TiCl3 to form the final composite with about 78% MgH2 [77]. The MgH2/GC-TiCl3 composite can release reversibly up to 7.6 wt.% H2 within 9 min at 300 °C, a result superior to even the MgH2-GC-TiCl3 mixture (weight ratio of 1:1:0.08, produced by direct ball milling for 5 h) (Figure 4) [77]. This highlights the advantages of the stepwise synthesis procedure outlined above.
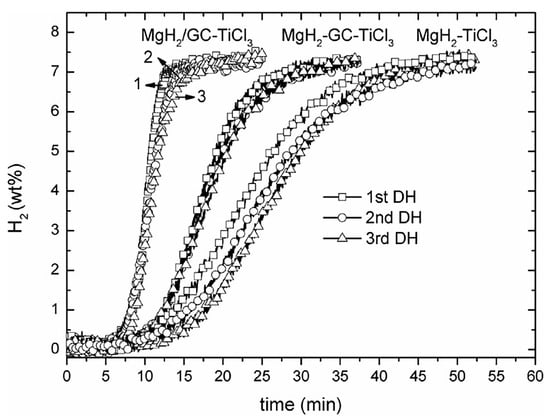
Figure 4.
Cyclic dehydriding (DH) curves of the MgH2-TiCl3, MgH2/GC-TiCl3 and MgH2-GC-TiCl3 composite samples at 300 C under 0.1 bar H2. Reprinted with permission from Ref. [77].
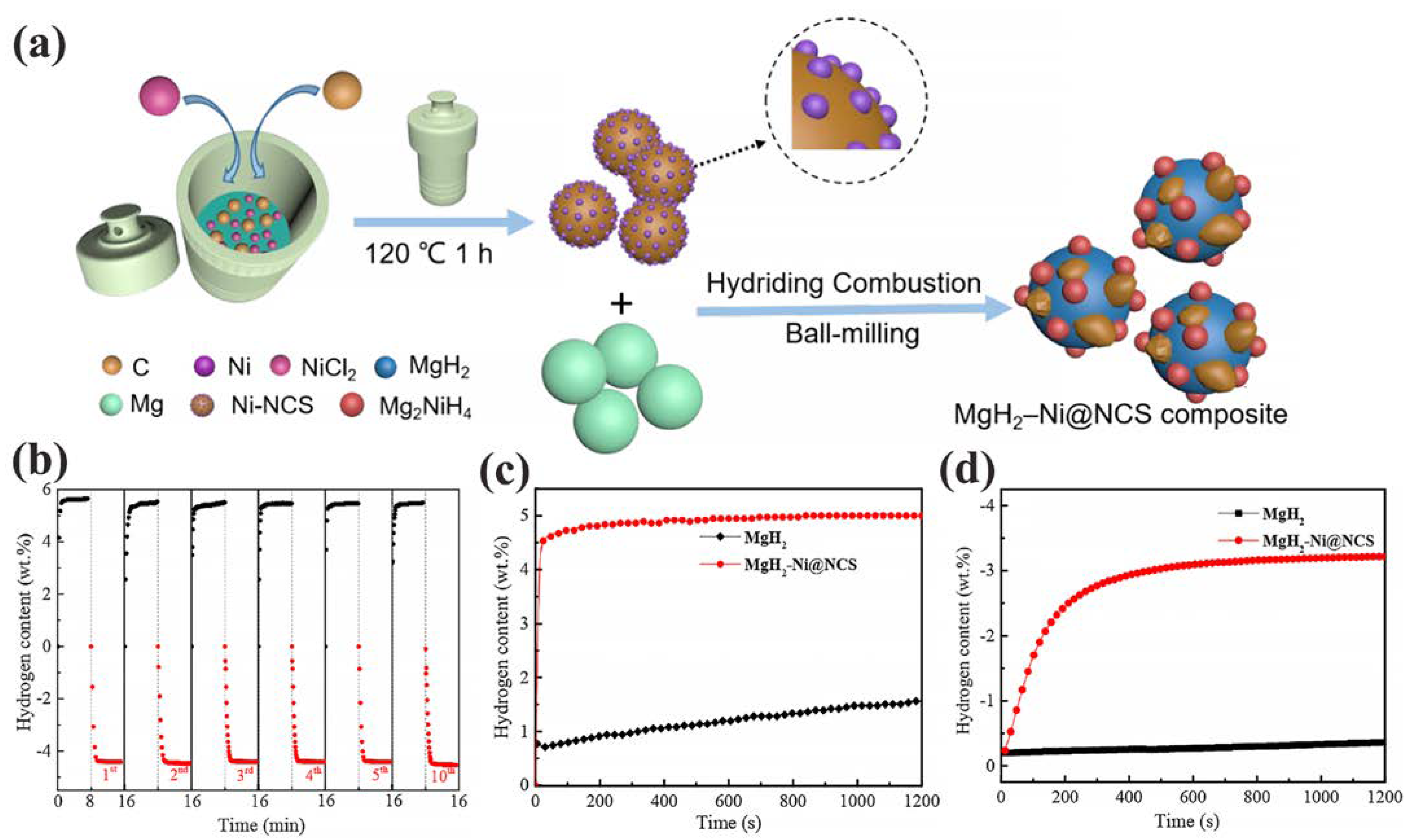
Among the metals showing the most striking improvements in hydrogenation studies, Ni has a distinct role. For instance, MgH2 has shown improvements in dehydrogenation behavior when catalyzed by Ni@N-doped C spheres [78], or by NiCo–functionalized G [79]. MgH2–Ni@NCS composites produced by Wang et al. were obtained by hydriding combustion and high energy ball milling Ni@NCS into Mg [78]. These composites showed good stability even after 10 a/d cycles, and moderate H2 storage parameters (a: 5. wt.%, d: 4.3 wt.% H2 within 8 min at 623 K; a: 4.2 wt.% in 60 min at 373 K (Figure 5)) [78].
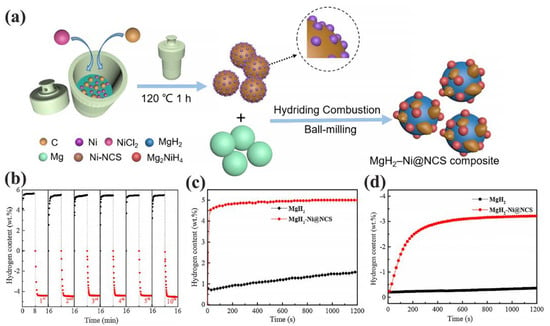
Figure 5.
(a) Preparation process of the MgH2–Ni@NCS composite. (b) Absorption and desorption cycling of MgH2–Ni@NCS composite at 623 K; (c) Isothermal hydrogenation curves of the MgH2–Ni@NCS composite and milled MgH2 at 423 K; (d) Isothermal dehydrogenation curves of the MgH2–Ni@NCS composite and milled MgH2 at 598 K. Reprinted with permission from Ref. [78].
When NiCo bimetallic NPs (4–6 nm) were used to functionalize graphene oxide supports, NiCo/rGO were obtained via a one-pot synthesis and produced energy storing nanocomposites MgH2–NiCo/rGO with excellent cycling stability, featuring reduced Ea = 105 kJ/mol, absorbing 6.1 wt.% H2 within 350 s at 300 °C under 0.9 MPa H2 [79]. The metal sources were the corresponding metal chlorides (NiCl2.6H2O and CoCl2.6H2O) that underwent sonication with GO/EG, reflux, and reduction with N2H4.H2O/NaOH [79].
Other support functions have also been pursued, such as MgH2@PTh core-shell NPs on G (PTh = polythiophene) for use in Li-ion batteries [59], or MgH2@NiPc (NiPc = nickel phthalocyanine)/G [80].
A summary of the main parameters in other doped-graphene supports for binary hydrides or alloys is given in Table 1.

Table 1.
Doped-graphene supports for Mg/MgH2 and related alloys.
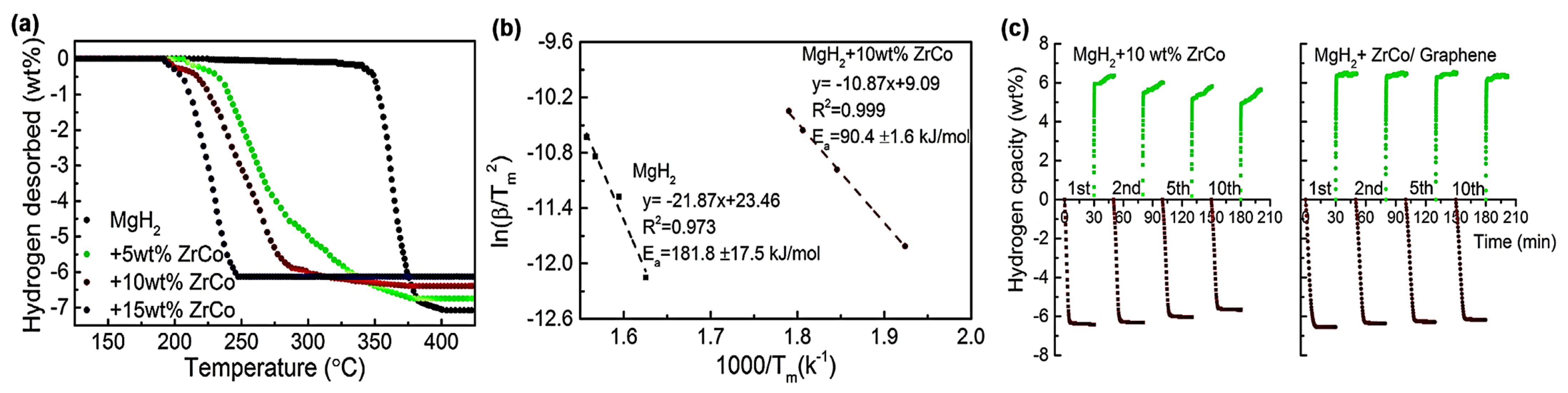
For instance, Zhang et al. used 2D ZrCo nanosheets to catalyze MgH2 decomposition [84]. When 10 wt.% ZrCo was added, the composite MgH2@10 wt.% ZrCo/G released 6.3 wt.% H2 at 300 °C in 5 min. (Figure 6). When bimetallic catalysts such as ZrCo are homogeneously dispersed on G, they play a role similar to that of Mg2Ni/Mg2NiH4 (as a “hydrogen pump”), easing the dissociation of H2 as well as its recombination, along with weakening the Mg–H bonds, further improving the hydrogen storage properties of MgH2 [84].
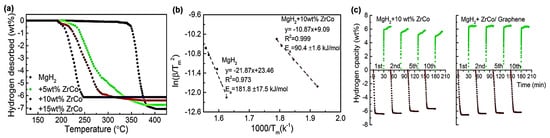
Figure 6.
(a) DSC curves of MgH2 + x (x = 5, 10, 15) wt.% ZrCo composites; (b) Kissinger’s plots (b) of the MgH2 with and without 10 wt.% ZrCo composites; (c) Isothermal dehydrogenation/hydrogenation curves of the MgH2 + 10 wt.% ZrCo and MgH2 + 10 wt.% ZrCo/5 wt.% Graphene composites as a function of cycling. Reproduced with permission from ref. [84].
4.2. Complex Hydrides Embedded in Graphene Supports: M(BH4)n, M(AlH4)n
Nanoconfined complex hydrides have emerged as prime candidates in energy storage applications. They possess the highest H2 gravimetric storage capacity, and careful tuning of their composition, additives, and morphology could yield highly reversible systems with real prospects in vehicular applications [2]. Graphene (non-catalyzed or catalyzed) has served as the support for manufacturing such stable, high-performance nanocomposites of complex hydride@G. A summary of more representative examples of such composites is given in Table 2.

Table 2.
Doped-graphene supports for complex hydrides M(BH4)n, M(AlH4)n and related hydrides.
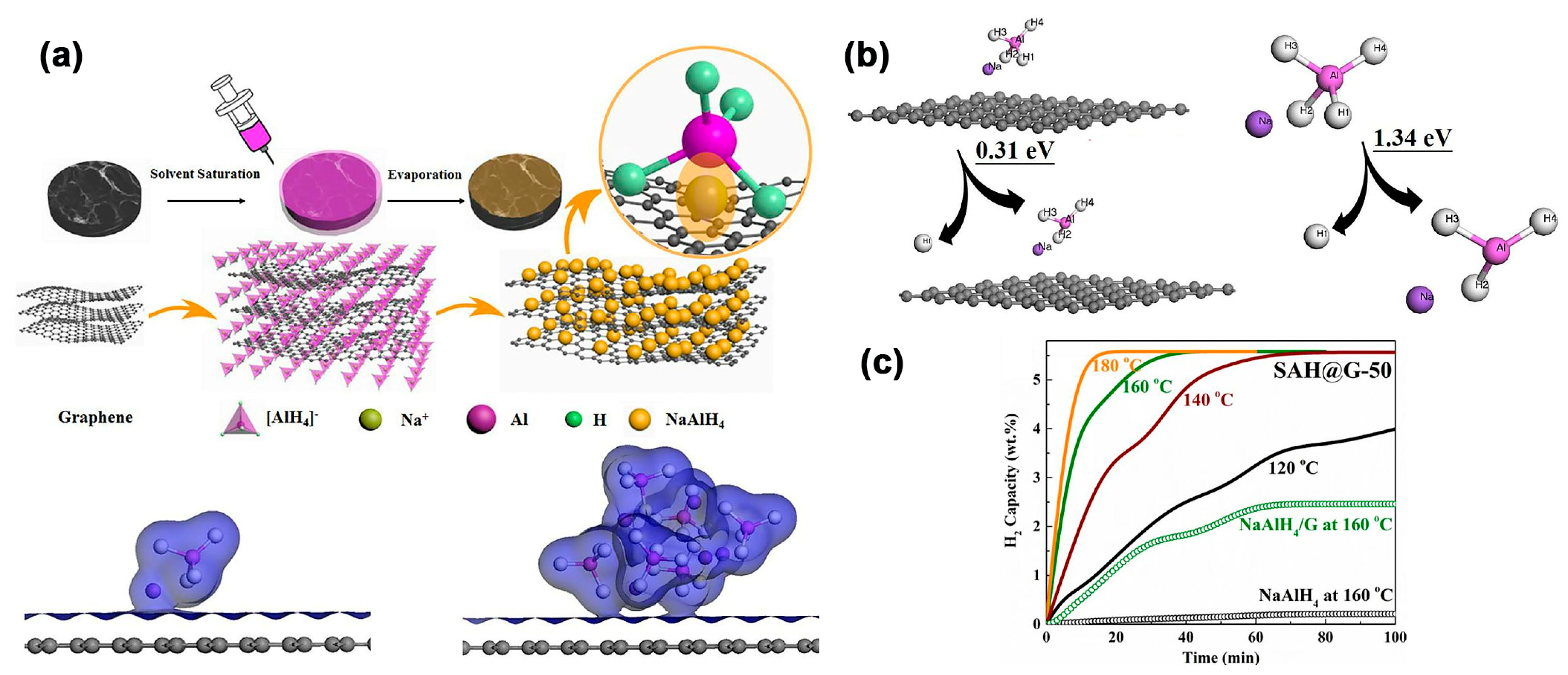
Huang et al. have described an interesting method to prepare NaAlH4-based composites with an average hydride size of 12.4 nm, while highlighting the advantage of supporting the complex hydride on graphene (0.31 eV vs. 1.34 eV) (Figure 7) [114]. With high theoretical storage capacities (7.5 wt.% gravimetric, 94 g H2/L volumetric), NaAlH4 could desorb H2 in three steps (via Na3AlH6—3.7 wt.%, further decomposition to NaH—1.9 wt.% and the often overlooked third step to Na metal); however, due to thermodynamic considerations, only the first two steps are achievable within reasonable temperature ranges.
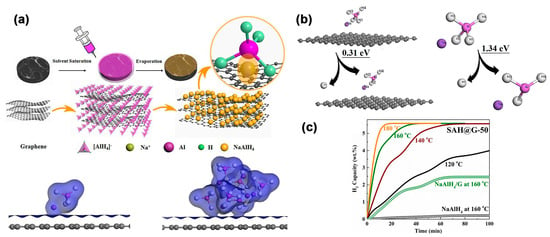
Figure 7.
(a) Schematic illustration of the synthesis of SAH@G-50. Isosurface of electron density of the NaAlH4 on graphene and (NaAlH4)6 on graphene; (b) Energies required for the removal of hydrogen from NaAlH4 with (0.31 eV) and without the presence of graphene (1.34 eV); (c) Isothermal dehydrogenation of SAH@G-50 in comparison with NaAlH4 and the ball-milled NaAlH4/G composite at various temperatures. Reprinted with permission from Ref. [114].
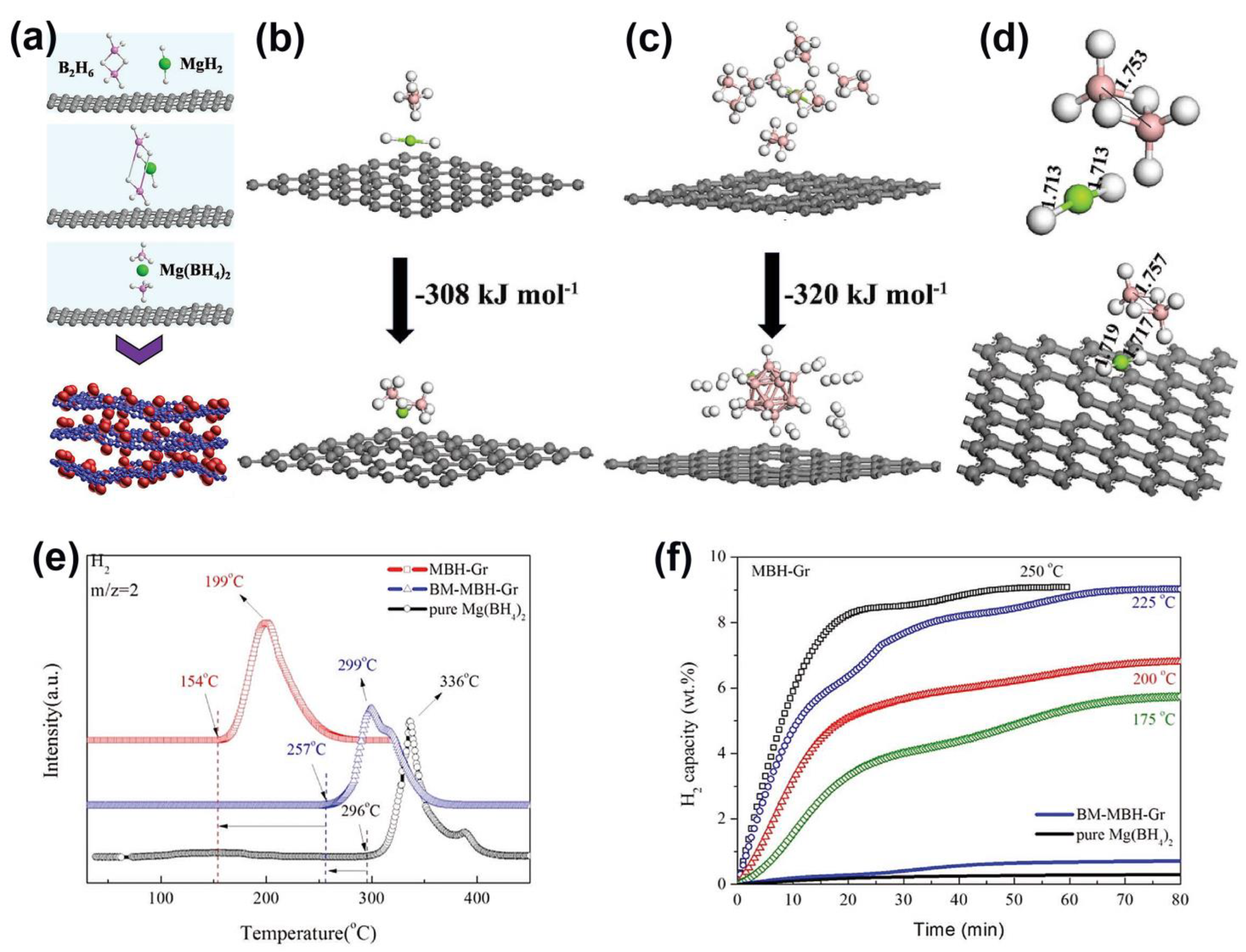
While mechanochemistry or solvent infiltration are typically utilized to confine metal hydrides in nanoporous supports, it is widely accepted that the in situ generation of hydride species can provide more intimate contact between the active hydrogen storage species and the support, resulting in enhanced hydrogenation parameters. Zhang et al., for instance, synthesized the Mg(BH4)2@G (MBH–Gr) composite, which has the ability to start releasing H2 at 154 °C vs. mechanochemically milled BM–MBH–Gr (257 °C) or pristine Mg(BH4)2 (296 °C), showing a tangible ~6 wt.% storage around 200 °C (Figure 8). The synthesis proceeded via the hydrogenation of MgBu2, which shows favorable adsorption of graphene, and β–MgH2 NPs of ~8 nm are formed highly dispersed on the substrate (“MH-Gr”) as a result of this interaction. The large spacing between graphene layers could be responsible for the lack of agglomeration during the solid–gas reaction with B2H6 in order to generate Mg(BH4)2 NPs (reaction (1)) [124].
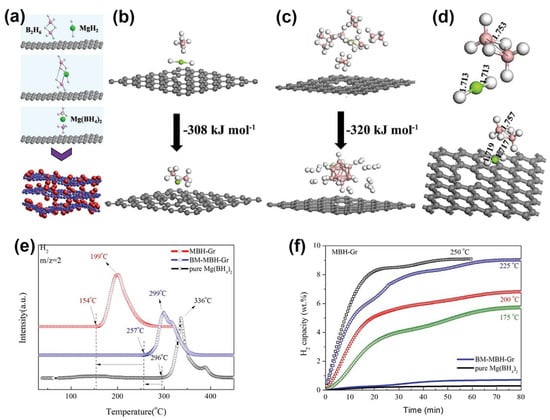
Figure 8.
(a) Schematic illustration of the fabrication of MBH-Gr via a space-confined solid–gas reaction: the graphene-tailored solid–gas reaction between MH-Gr and B2H6 to synthesize Mg(BH4)2 NPs that are uniformly distributed on graphene (MBH-Gr) under the protection of hydrogen; (b–d): Relaxed atomic configurations of MgH2 and B2H6, and calculated reaction enthalpies for the formation of (b) Mg(BH4)2 and (c) MgB12H12 from the reaction between MgH2 and B2H6 with the support of graphene. (d) Relaxed atomic configurations of MgH2 and B2H6 without and with the support of graphene. Gray, white, green, and pink spheres are C, H, Mg, and B atoms, respectively; (e) Mass spectra of the as-synthesized MBH-Gr compared with BM-MBH-Gr and pure Mg(BH4)2; (f) Isothermal dehydrogenation of the as-synthesized MBH-Gr at various temperatures, with BM-MBH-Gr and pure Mg(BH4)2 at 175 °C included for comparison. Reprinted with permission from Ref. [124].
However, the authors also noted simultaneous formation of MgB12H12 (2478 cm−1 in FTIR spectra), and DFT data showed that the synthesis of MgB12H12 (∆H = −320 kJ/mol) and Mg(BH4)2 ((∆H = −308 kJ/mol) is fairly similar. These values suggest that the presence of graphene favors the formation of Mg(BH4)2. The optimized geometries (Figure 8d) also reveal that graphene weakens Mg–H and B–B bonds (they are longer in the presence of graphene vs. pristine), congruent with the easier formation of Mg(BH4)2 [124].
4.3. Reactive Hydride Composites (RHCs) in G Hosts
There are only a few examples of MgH2-based nancomposites; for instance, the MgH2–LiBH4 RHC [133] and 2 LiBH4–MgH2 @ G (80 wt.% loading capacity) [134]. In the case of the latter composite, the synthesis method started from MgH2 @ G [134]. Not surprisingly, the MgH2 was compounded with LiBH4, another key component and widely–explored hydrogen storage complex hydride (18.5 wt.%, 121 kg H2/m3), with the positive results making use of LiBH4 destabilization by binary metal hydrides such as MgH2, in addition to the advantageous nanoconfinement effect in the porous graphene support [133]. The mutual destabilization of the system 2LIBH4–MgH2 is depicted in reaction (2), where the final decomposition products are LiH and MgB2. Even without graphene supports, the RHC system 2LIBH4–MgH2 showed reversibility under moderate conditions (50 bar H2, 300 °C), considerably lower than the harsh rehydrogenation requirements from spent LiBH4 (200 bar H2, 700 °C).
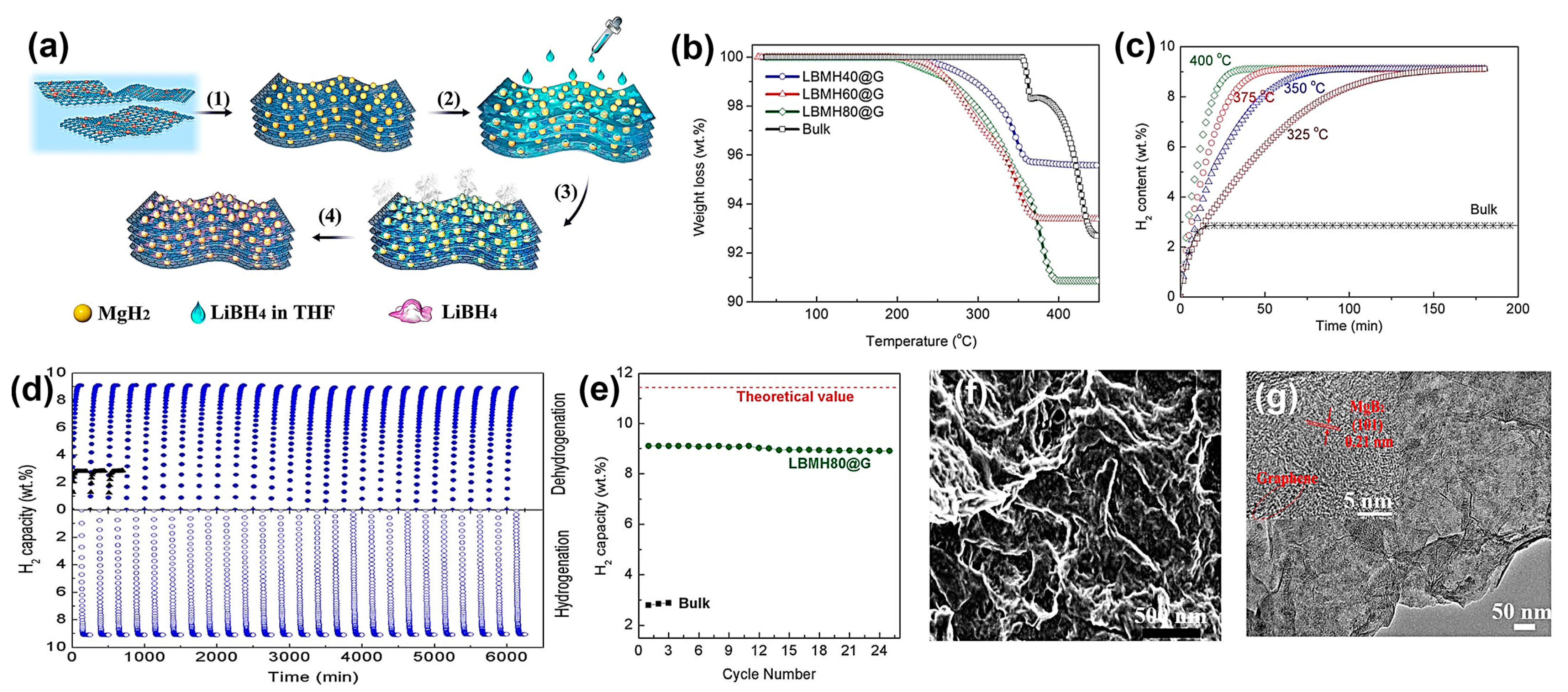
Xia et al. have demonstrated the solvothermal procedure to impregnate graphene with RHC precursors that finally yielded the binary composite 2LiBH4–MgH2, with homogeneous particle size (10.5 nm) uniformly dispersed on graphene support even at very high loadings, which afforded a reversible 9.1 wt.% hydrogen capacity that did not degrade below 8.9 wt.% after 25 a/d cycles (Figure 9) [134].
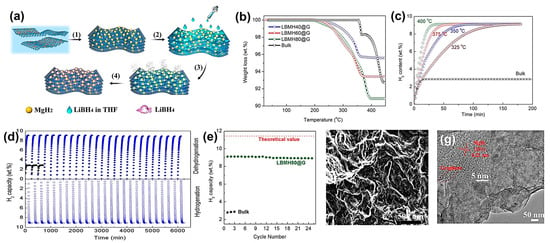
Figure 9.
(a) Schematic illustration of the fabrication of graphene-supported 2LiBH4-MgH2 nanocomposite: (1) self-assembly of uniform MgH2 NPs on graphene via solvothermal treatment; (2) infiltration of LiBH4 solution; (3, 4) removal of solvent and heterogeneous nucleation of LiBH4 on MgH2 NPs.; (b) Thermogravimetric analysis curves of the as-prepared 2LiBH4-MgH2 nanocomposites anchored on graphene with various loadings compared with the ball-milled composite (bulk); (c) isothermal dehydrogenation of LBMH80@G at various temperatures under a back pressure of 0.3 MPa.; (d) Long-term cycling performance of the dehydrogenation (under a back pressure of 0.3 MPa) and hydrogenation for LBMH80@G and bulk 2LiBH4-MgH2 composite at 350 °C; (e) normalized H2 capacity as a function of cycle number, where the hydrogen capacities are normalized to the theoretical value of 2LiBH4-MgH2 composite; (f) SEM and (g) TEM images of LBMH80@G after 15 cycles of dehydrogenation. Reprinted with permission from Ref. [134].
While other C-based materials supporting RHCs are rather common, the same cannot be said about G-supported RHCs. The improvements observed in lowered dehydrogenation temperature and parameters can be also traced back to the thermal conductivity of the graphene support, which enables the effective dissipation of heat and uniform heating throughout the composite [133]. Interestingly, nanoconfinement might not be solely responsible for the observed effects, as XRD data showed characteristic hydride peaks; hence, these were not fully confined, and the main role might actually be reserved to the highly specific surface area of graphene as well as to the nanosized metal hydrides.
4.4. Other Hydride Systems Confined in 2D-Graphene
Among hydrogen storing materials, ammonia borane (AB) plays a special role. While it has been investigated thoroughly, the mechanism of dehydrogenation is still not very clear, presumably due to polymerization processes occurring during thermal decomposition, and, perhaps more importantly, the system is not reversible. However, there have been attempts to incorporate AB into the porosity of catalyzed graphene supports, which yielded composites such as AB–Ni-Co/rGO catalyst [135], AB/Cu@CuCoOx supported on G [136], or AB/rGO (or GO) produced by ice-templating sheets of GO, serving as supports for the one-step integration of AB. The AB@rGO nanocomposite released H2 as low as 70 °C [137]. A closely-related lithiated species of AB, namely lithium amidoborane LiNH2BH3, has been utilized to produce LiNH2BH3/G nanocomposites that released 10.9 wt.% H2 without (BN)x byproducts such as borazine [138].
Other 2D materials such as MXenes have also been incorporated in hydrogen storing materials such as hydrides, providing excellent improvements, presumably due to the presence of catalytic Ti sites and superior morphological features such as its highly-specific surface area [139].
Liquid organic hydrides can also release H2 under G–CNT composite catalysts [140]. Substituted alkyl (R = Me) and aryl (R = Ph) silanes RxSiH4−x (R = Ph, Me) have also been employed in H2 generation from MeOH. In this instance, however, the Ru-based catalyst was immobilized in the rGO support, producing the highly-active catalyst [Ru]–rGO (0.05 mol%), where [Ru] = [Ru(p-cym)Cl2(NHCl)] complex [141]. To sum up, there are various approaches spanning metal salt catalysts [142], heterostructured supports [143], Ni-based catalysts supported by DFT computations [144], trimetallic catalysts FeCoNi NPs [145], or bimetallic PdRu [146] that have all played an essential role in the development of efficient catalysts and supports for metal hydrides and magnesium hydride in particular.
5. Conclusions
Featuring high hydrogen storage capacity and electrochemical properties, metal hydrides embedded in graphene supports have emerged as a new class of nanocomposite systems with promising features for energy storage systems. However, their development and integration in practical systems still face challenges to be overcome for their widespread adoption and long-term viability. The cost-effectiveness of these composite systems is an essential factor in their widespread adoption and increased lifespan viability. The cost of raw materials as well as production and energy costs are still too high, prompting the development of cost-effective and scalable fabrication processes.
Regarding the environmental impacts and ways to limit their availability, the use of CRM (critical raw materials) such as Mg, Ni, and Ti and their continuing depletion worldwide are of concern. The development of alternative materials that do not rely on CRMs is crucial for the sustainability of this technology. The long-term stability and durability of metal hydrides embedded in graphene supports are crucial for their sustainability, as frequent replacements can result in the generation of more waste and have a significant environmental impact. The optimization of the synthesis process and the development of durable and stable materials can improve the lifespan and performance of metal hydrides embedded in graphene supports, making them more sustainable. The disposal of metal hydrides embedded in graphene supports at the end of their lifecycle is also a concern. The development of recycling processes for metal hydrides and graphene supports can reduce the environmental impact of their disposal.
6. Current Challenges and Future Outlook
Energy storage systems are today in need of technological breakthroughs to revolutionize the way energy is produced, distributed, and stored. Various reviews have presented the critical shortcomings of current technology [3]. Nanocomposites based on metal hydrides and graphene currently feature hydrogen storage capacities lower than the targets set by the Department of Energy (DOE) for practical hydrogen storage (6.5 wt.% as the ultimate goal). Moreover, the cycling stability of metal hydrides needs to be improved to ensure the long-term performance of the composite material, while the potential side reactions occurring at the nanoscale must be further tuned and reduced. The scalability and cost-effectiveness of the synthesis methods used to prepare graphene-metal hydride composites need further refinement. Current methods such as ball milling (most commonly used) and in situ growth are time-consuming and require expensive equipment with high associated energy bills, which limits their practical application for large-scale production, which is compulsory for industrial adoption of this technology.
Future research efforts addressing these challenges ought to focus on developing new metal hydrides with higher hydrogen storage capacities and improved cycling stability. In addition, new synthesis methods should be explored to improve the scalability and cost-effectiveness of the process. Advances in computational modeling and simulation can also be used to predict the performance of new metal hydride candidates and optimize their synthesis routes. In this regard, thermodynamic considerations are an essential starting point for the successful selection of future hydride-based materials.
Additionally, while graphene does feature many desirable properties, other carbon-based materials can be explored for the development of new hybrid materials with enhanced properties for energy storage applications, such as carbon nanotubes (MWCNTs) or other forms of porous carbon (OMC, CMK-3, etc.). Carbon materials can be further activated to enhance hydrogen-support interactions, which in turn will lead to a more effective H2 storage material. Hence, current strategies need to focus on enhancing H2-support interactions [4] by any of the following approaches: heteroatom-doping of porous G/other nanoporous materials; coordinatively unsaturated metal sites in MOFs; nanoconfinement of H2 into nanopores (<1 nm); and enhanced orbital interactions H2-adsorbent [4].
As the global demand for clean energy solutions continues to grow, the development of efficient energy storage systems such as graphene-metal hydride composites can play a crucial role in accelerating the transition towards a sustainable energy future.
Funding
This work was supported by the Romanian Ministry of Research and Innovation through the Project No. PN-III-P1-1.1-TE-2021-1657 (TE 84/2022) and by the Core Program of the National Institute of Materials Physics, granted by the Romanian Ministry of Research, Innovation and Digitization through the Project PC3-PN23080303.
Data Availability Statement
Not applicable.
Acknowledgments
This work was supported by the Romanian Ministry of Research and Innovation through the Project No. PN-III-P1-1.1-TE-2021-1657 (TE 84/2022) and Project PC3-PN23080303.
Conflicts of Interest
The authors declare no conflict of interest. The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript; or in the decision to publish the results.
References
- Zhang, J.; Yan, S.; Qu, H. Recent progress in magnesium hydride modified through catalysis and nanoconfinement. Int. J. Hydrogen Energy 2018, 43, 1545–1565. [Google Scholar] [CrossRef]
- Comanescu, C. Recent Development in Nanoconfined Hydrides for Energy Storage. Int. J. Mol. Sci. 2022, 23, 7111. [Google Scholar] [CrossRef] [PubMed]
- Sreedhar, I.; Kamani, K.M.; Kamani, B.M.; Reddy, B.M.; Venugopal, A. A Bird’s Eye view on process and engineering aspects of hydrogen storage. Renew. Sustain. Energy Rev. 2018, 91, 838–860. [Google Scholar] [CrossRef]
- So, S.H.; Sung, S.J.; Yang, S.J.; Park, C.R. Where to go for the Development of High-Performance H-2 Storage Materials at Ambient Conditions? Electron. Mater. Lett. 2023, 19, 1–18. [Google Scholar] [CrossRef]
- Comanescu, C. Paving the Way to the Fuel of the Future—Nanostructured Complex Hydrides. Int. J. Mol. Sci. 2023, 24, 143. [Google Scholar] [CrossRef]
- Comanescu, C. Complex Metal Borohydrides: From Laboratory Oddities to Prime Candidates in Energy Storage Applications. Materials 2022, 15, 2286. [Google Scholar] [CrossRef]
- Ding, F.; Yakobson, B.I. Challenges in hydrogen adsorptions: From physisorption to chemisorption. Front. Phys. 2011, 6, 142–150. [Google Scholar] [CrossRef]
- Feng, D.C.; Zhou, D.S.; Zhao, Z.Y.; Zhai, T.T.; Yuan, Z.M.; Sun, H.; Ren, H.P.; Zhang, Y.H. Progress of graphene and loaded transition metals on Mg-based hydrogen storage alloys. Int. J. Hydrogen Energy 2021, 46, 33468–33485. [Google Scholar] [CrossRef]
- Free, Z.; Hernandez, M.; Mashal, M.; Mondal, K. A Review on Advanced Manufacturing for Hydrogen Storage Applications. Energies 2021, 14, 8513. [Google Scholar] [CrossRef]
- Boateng, E.; Chen, A.C. Recent advances in nanomaterial-based solid-state hydrogen storage. Mater. Today Adv. 2020, 6, 100022. [Google Scholar] [CrossRef]
- Jawhari, A.H. Novel Nanomaterials for Hydrogen Production and Storage: Evaluating the Futurity of Graphene/Graphene Composites in Hydrogen Energy. Energies 2022, 15, 9085. [Google Scholar] [CrossRef]
- Arbuzov, A.A.; Mozhzhukhin, S.A.; Volodin, A.A.; Fursikov, P.V.; Tarasov, B.P. Graphene-like nanostructures: Synthesis and use for preparation of catalysts and hydrogen storage composites. Russ. Chem. B+ 2016, 65, 1893–1901. [Google Scholar] [CrossRef]
- Xia, G.; Zhang, B.; Chen, X.; Sun, D.; Guo, Z.; Liang, F.; Yu, X. Graphene for Energy Storage and Conversion: Synthesis and Interdisciplinary Applications. Electrochem. Energy Rev. 2020, 3, 395–430. [Google Scholar] [CrossRef]
- Liu, W.; Setijadi, E.; Crema, L.; Bartali, R.; Laidani, N.; Aguey-Zinsou, K.F.; Speranza, G. Carbon nanostructures/Mg hybrid materials for hydrogen storage. Diam. Relat. Mater. 2018, 82, 19–24. [Google Scholar] [CrossRef]
- Yao, Q.L.; Ding, Y.Y.; Lu, Z.H. Noble-metal-free nanocatalysts for hydrogen generation from boron- and nitrogen-based hydrides. Inorg. Chem. Front. 2020, 7, 3837–3874. [Google Scholar] [CrossRef]
- Tarasov, B.P.; Arbuzov, A.A.; Volodin, A.A.; Fursikov, P.V.; Mozhzhuhin, S.A.; Lototskyy, M.V.; Yartys, V.A. Metal hydride–Graphene composites for hydrogen based Energy storage. J. Alloys Compd. 2022, 896, 162881. [Google Scholar] [CrossRef]
- Aditya, M.V.V.S.; Panda, S.; Tatiparti, S.S.V. Boron from net charge acceptor to donor and its effect on hydrogen uptake by novel Mg-B-electrochemically synthesized reduced graphene oxide. Sci. Rep. 2021, 11, 10995. [Google Scholar] [CrossRef]
- Tan, X.; Tahini, H.A.; Smith, S.C. Conductive Boron-Doped Graphene as an Ideal Material for Electrocatalytically Switchable and High-Capacity Hydrogen Storage. ACS Appl. Mater. Interfaces 2016, 8, 32815–32822. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, K.; Guan, C.; He, Z.M.; Lu, Z.S.; Chen, T.; Liu, J.; Tan, X.; Tan, T.T.Y.; Li, C.M. Surface functionalization-enhanced spillover effect on hydrogen storage of Ni-B nanoalloy-doped activated carbon. Int. J. Hydrogen Energy 2011, 36, 13663–13668. [Google Scholar] [CrossRef]
- Wan, L.F.; Cho, E.S.; Marangoni, T.; Shea, P.; Kang, S.Y.; Rogers, C.; Zaia, E.; Cloke, R.R.; Wood, B.C.; Fischer, F.R.; et al. EdgeFunctionalized Graphene Nanoribbon Encapsulation to Enhance Stability and Control Kinetics of Hydrogen Storage Materials. Chem. Mater. 2019, 31, 2960–2970. [Google Scholar] [CrossRef]
- Asefa, T.; Koh, K.; Yoon, C.W. CO2-Mediated H-2 Storage-Release with Nanostructured Catalysts: Recent Progresses, Challenges, and Perspectives. Adv. Energy Mater. 2019, 9, 1158. [Google Scholar] [CrossRef]
- Shevlin, S.A.; Guo, Z.X. Density functional theory simulations of complex hydride and carbon-based hydrogen storage materials. Chem. Soc. Rev. 2009, 38, 211–225. [Google Scholar] [CrossRef] [PubMed]
- Naqvi, S.R.; Hussain, T.; Luo, W.; Ahuja, R. Metallized siligraphene nanosheets (SiC7) as high capacity hydrogen storage materials. Nano Res. 2018, 11, 3802–3813. [Google Scholar] [CrossRef]
- Aydin, S.; Simsek, M. The enhancement of hydrogen storage capacity in Li, Na and Mg-decorated BC3 graphene by CLICH and RICH algorithms. Int. J. Hydrogen Energy 2019, 44, 7354–7370. [Google Scholar] [CrossRef]
- Tokarev, A.; Avdeenkov, A.V.; Langmi, H.; Bessarabov, D.G. Modeling hydrogen storage in boron-substituted graphene decorated with potassium metal atoms. Int. J. Energy Res. 2015, 39, 524–528. [Google Scholar] [CrossRef]
- Ao, Z.M.; Dou, S.X.; Xu, Z.M.; Jiang, Q.G.; Wang, G.X. Hydrogen storage in porous graphene with Al decoration. Int. J. Hydrogen Energy 2014, 39, 16244–16251. [Google Scholar] [CrossRef]
- Costanzo, F.; van Hemert, M.C.; Kroes, G.J. Promoting Effect of Carbon Surfaces on H-2 Dissociation on Al-n Clusters by First Principles Calculations. J. Phys. Chem. C 2014, 118, 513–522. [Google Scholar] [CrossRef]
- Zhang, Y.; Cui, H.; Tian, W.Z.; Liu, T.; Wang, Y.Z. Effect of hydrogen adsorption Energy on the electronic and optical properties of Si-modified single-layer graphene with an Al decoration. AIP Adv. 2020, 10, 045012. [Google Scholar] [CrossRef]
- Bakhshi, F.; Farhadian, N. Improvement of hydrogen storage capacity on the palladium-decorated N-doped graphene sheets as a novel adsorbent: A hybrid MD-GCMC simulation study. Int. J. Hydrogen Energy 2019, 44, 13655–13665. [Google Scholar] [CrossRef]
- Ramos-Castillo, C.M.; Reveles, J.U.; Zope, R.R.; de Coss, R. Palladium Clusters Supported on Graphene Monovacancies for Hydrogen Storage. J. Phys. Chem. C 2015, 119, 8402–8409. [Google Scholar] [CrossRef]
- Choudhary, A.; Malakkal, L.; Siripurapu, R.K.; Szpunar, B.; Szpunar, J. First principles calculations of hydrogen storage on Cu and Pd-decorated graphene. Int. J. Hydrogen Energy 2016, 41, 17652–17656. [Google Scholar] [CrossRef]
- Bora, P.L.; Ahmad, R.; Singh, A.K. Remarkable enhancement in hydrogen storage on free-standing Ti3B and BC3 supported Ti-3 clusters. Int. J. Hydrogen Energy 2015, 40, 1054–1061. [Google Scholar] [CrossRef]
- Intayot, R.; Rungnim, C.; Namuangruk, S.; Yodsin, N.; Jungsuttiwong, S. Ti-4-Decorated B/N-doped graphene as a high-capacity hydrogen storage material: A DFT study. Dalton Trans. 2021, 50, 11398–11411. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Castillo, C.M.; Reveles, J.U.; Cifuentes-Quintal, M.E.; Zope, R.R.; de Coss, R. Ti-4- and Ni-4-Doped Defective Graphene Nanoplatelets as Efficient Materials for Hydrogen Storage. J. Phys. Chem. C 2016, 120, 5001–5009. [Google Scholar] [CrossRef]
- Ramos-Castillo, C.M.; Reveles, J.U.; Cifuentes-Quintal, M.E.; Zope, R.R.; de Coss, R. Hydrogen storage in bimetallic Ti-Al sub-nanoclusters supported on graphene. Phys. Chem. Chem. Phys. 2017, 19, 21174–21184. [Google Scholar] [CrossRef]
- Bartali, R.; Speranza, G.; Aguey-Zinsou, K.F.; Testi, M.; Micheli, V.; Canteri, R.; Fedrizzi, M.; Gottardi, G.; Coser, G.; Crema, L.; et al. Efficient hydrogen generation from water using nanocomposite flakes based on graphene and magnesium. Sustain. Energy Fuels 2018, 2, 2516–2525. [Google Scholar] [CrossRef]
- Bouaricha, S.; Dodelet, J.P.; Guay, D.; Huot, J.; Schulz, R. Study of the activation process of Mg-based hydrogen storage materials modified by graphite and other carbonaceous compounds. J. Mater. Res. 2001, 16, 2893–2905. [Google Scholar] [CrossRef]
- Cho, E.S.; Ruminski, A.M.; Liu, Y.S.; Shea, P.T.; Kang, S.Y.; Zaia, E.W.; Park, J.Y.; Chuang, Y.D.; Yuk, J.M.; Zhou, X.W.; et al. Hierarchically Controlled Inside-Out Doping of Mg Nanocomposites for Moderate Temperature Hydrogen Storage. Adv. Funct. Mater. 2017, 27, 1704316. [Google Scholar] [CrossRef]
- Dun, C.; Jeong, S.; Kwon, D.H.; Kang, S.; Stavila, V.; Zhang, Z.L.; Lee, J.W.; Mattox, T.M.; Heo, T.W.; Wood, B.C.; et al. Hydrogen Storage Performance of Preferentially Oriented Mg/rGO Hybrids. Chem. Mater. 2022, 34, 2963–2971. [Google Scholar] [CrossRef]
- Cho, Y.; Kang, S.; Wood, B.C.; Cho, E.S. Heteroatom-Doped Graphenes as Actively Interacting 2D Encapsulation Media for Mg-Based Hydrogen Storage. ACS Appl. Mater. Interfaces 2022, 14, 20823–20834. [Google Scholar] [CrossRef]
- Dong, S.; Lv, E.F.; Wang, J.H.; Li, C.Q.; Ma, K.; Gao, Z.Y.; Yang, W.J.; Ding, Z.; Wu, C.C.; Gates, I.D. Construction of transition metal-decorated boron doped twin-graphene for hydrogen storage: A theoretical prediction. Fuel 2021, 304, 121351. [Google Scholar] [CrossRef]
- Han, D.J.; Kim, S.; Cho, E.S. Revealing the role of defects in graphene oxide in the evolution of magnesium nanocrystals and the resulting effects on hydrogen storage. J. Mater. Chem. A 2021, 9, 9875–9881. [Google Scholar] [CrossRef]
- Du, J.Q.; Lan, Z.Q.; Zhang, H.; Lu, S.X.; Liu, H.Z.; Guo, J. Catalytic enhanced hydrogen storage properties of Mg-based alloy by the addition of reduced graphene oxide supported V2O3 nanocomposite. J. Alloys Compd. 2019, 802, 660–667. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Gupta, B.K.; Tripathi, P.; Veziroglu, A.; Hudson, M.S.L.; Abu Shaz, M.; Srivastava, O.N. Development and Demonstration of Air Stable rGO-EC@AB(5) Type Hydrogenated Intermetallic Hybrid for Hydrogen Fuelled Devices. Adv. Sustain. Syst. 2017, 1, 1700087. [Google Scholar] [CrossRef]
- Cui, R.C.; Yang, C.C.; Li, M.M.; Jin, B.; Ding, X.D.; Jiang, Q. Enhanced high-rate performance of ball-milled MmNi(3.55)Co(0.75)Mn(0.4)Al(0.3) hydrogen storage alloys with graphene nanoplatelets. J. Alloys Compd. 2017, 693, 126–131. [Google Scholar] [CrossRef]
- Morse, J.R.; Zugell, D.A.; Patterson, E.; Baldwin, J.W.; Willauer, H.D. Hydrogenated graphene: Important material properties regarding its application for hydrogen storage. J. Power Sources 2021, 494, 229734. [Google Scholar] [CrossRef]
- Wu, X.J.; Fei, Z.J.; Liu, W.G.; Tan, J.; Wang, G.H.; Xia, D.Q.; Deng, K.; Chen, X.K.; Xiao, D.T.; Wu, S.W.; et al. Adsorption and desorption of hydrogen on/from single-vacancy and double-vacancy graphenes. Nucl. Sci. Tech. 2019, 30, 69. [Google Scholar] [CrossRef]
- Akilan, R.; Vinnarasi, S.; Mohanapriya, S.; Shankar, R. Adsorption of H(2)molecules on B/N-doped defected graphene sheets—A DFT study. Struct. Chem. 2020, 31, 2413–2434. [Google Scholar] [CrossRef]
- Lotfi, R.; Saboohi, Y. A comparative study on hydrogen interaction with defective graphene structures doped by transition metals. Phys. E 2014, 60, 104–111. [Google Scholar] [CrossRef]
- Lototskyy, M.; Sibanyoni, J.M.; Denys, R.V.; Williams, M.; Pollet, B.G.; Yartys, V.A. Magnesium-carbon hydrogen storage hybrid materials produced by reactive ball milling in hydrogen. Carbon 2013, 57, 146–160. [Google Scholar] [CrossRef]
- Ruse, E.; Buzaglo, M.; Pri-Bar, I.; Shunak, L.; Nadiv, R.; Pevzner, S.; Siton-Mendelson, O.; Skripnyuk, V.M.; Rabkin, E.; Regev, O. Hydrogen storage kinetics: The graphene nanoplatelet size effect. Carbon 2018, 130, 369–376. [Google Scholar] [CrossRef]
- Bahuguna, A.; Sasson, Y. Formate-Bicarbonate Cycle as a Vehicle for Hydrogen and Energy Storage. Chemsuschem 2021, 14, 1258–1283. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Heintzman, E.; Price, C. Electrostatic Layer-By-Layer Self-Assembled Graphene/Multi-Walled Carbon Nanotubes Hybrid Multilayers as Efficient ‘All Carbon’ Supercapacitors. J. Nanosci. Nanotechnol. 2016, 16, 4771–4782. [Google Scholar] [CrossRef] [PubMed]
- Ghalami, Z.; Ghoulipour, V.; Khanchi, A. Hydrogen and deuterium adsorption on uranium decorated graphene nanosheets: A combined molecular dynamics and density functional theory study. Curr. Appl. Phys. 2019, 19, 536–541. [Google Scholar] [CrossRef]
- Lang, C.G.; Ouyang, L.Z.; Yang, L.L.; Dai, L.Y.; Wu, D.F.; Shao, H.Y.; Zhu, M. Enhanced hydrogen storage kinetics in Mg@FLG composite synthesized by plasma assisted milling. Int. J. Hydrogen Energy 2018, 43, 17346–17352. [Google Scholar] [CrossRef]
- Lee, J.; Sung, D.; Chung, Y.K.; Bin Song, S.; Huh, J. Unveiling two-dimensional magnesium hydride as a hydrogen storage material via a generative adversarial network. Nanoscale Adv. 2022, 4, 2332–2338. [Google Scholar] [CrossRef]
- Ates, M.; Chebil, A.; Yoruk, O.; Dridi, C.; Turkyilmaz, M. Reliability of electrode materials for supercapacitors and batteries in Energy storage applications: A review. Ionics 2022, 28, 27–52. [Google Scholar] [CrossRef]
- Chaichi, A.; Wang, Y.; Gartia, M.R. Substrate Engineered Interconnected Graphene Electrodes with Ultrahigh Energy and Power Densities for Energy Storage Applications. ACS Appl. Mater. Interfaces 2018, 10, 21235–21245. [Google Scholar] [CrossRef]
- Xia, G.; Zhang, B.; Chen, X.; Sun, D.; Guo, Z.; Liang, F.; Yu, X. Molecular-Scale Functionality on Graphene To Unlock the Energy Capabilities of Metal Hydrides for High-Capacity Lithium-Ion Batteries. ACS Nano 2018, 12, 8177–8186. [Google Scholar] [CrossRef]
- Li, M.M.; Wang, Y.; Yang, C.C.; Jiang, Q. In situ grown Co3O4 nanocubes on N-doped graphene as a synergistic hybrid for applications in nickel metal hydride batteries. Int. J. Hydrogen Energy 2018, 43, 18421–18435. [Google Scholar] [CrossRef]
- Sirisinudomkit, P.; Iamprasertkun, P.; Krittayavathananon, A.; Pettong, T.; Dittanet, P.; Sawangphruk, M. Hybrid Energy Storage of Ni(OH)(2)-coated N-doped Graphene Aerogel//N-doped Graphene Aerogel for the Replacement of NiCd and NiMH Batteries. Sci. Rep. 2017, 7, 1124. [Google Scholar] [CrossRef] [PubMed]
- Zhang, B.; Xia, G.; Sun, D.; Fang, F.; Yu, X. Magnesium Hydride Nanoparticles Self-Assembled on Graphene as Anode Material for High-Performance Lithium-Ion Batteries. ACS Nano 2018, 12, 3816–3824. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.J.; Li, C.; Zhang, X.; Sun, X.Z.; Wang, K.; Ma, Y.W. High Performance Lithium-Ion Hybrid Capacitors Employing Fe3O4-Graphene Composite Anode and Activated Carbon Cathode. ACS Appl. Mater. Interfaces 2017, 9, 17137–17145. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Xu, X.D.; Liu, D.; Li, J.; Wei, Y.H.; Tang, H.L.; Li, J.S.; Li, X.; Xie, Z.Z.; Qu, D.Y. The impacts of nitrogen doping on the electrochemical hydrogen storage in a carbon. Int. J. Energy Res. 2021, 45, 9326–9339. [Google Scholar] [CrossRef]
- Lan, Z.Q.; Zeng, K.; Wei, B.; Li, G.X.; Ning, H.; Guo, J. Nickel-graphene nanocomposite with improved electrochemical performance for La0.7Mg0.3(Ni0.85Co0.15)(3.5) electrode. Int. J. Hydrogen Energy 2017, 42, 12458–12466. [Google Scholar] [CrossRef]
- Xia, G.; Tan, Y.; Chen, X.; Fang, F.; Sun, D.; Li, X.; Guo, Z.; Yu, X. Oxygen-free Layer-by-Layer Assembly of Lithiated Composites on Graphene for Advanced Hydrogen Storage. Adv. Sci. 2016, 4, 1600257. [Google Scholar] [CrossRef]
- Zhang, Q.; Huang, Y.; Xu, L.; Zang, L.; Guo, H.; Jiao, L.; Yuan, H.; Wang, Y. Highly Dispersed MgH2 Nanoparticle–Graphene Nanosheet Composites for Hydrogen Storage. ACS Appl. Nano Mater. 2019, 2, 3828–3835. [Google Scholar] [CrossRef]
- Huang, Y.Q.; Xia, G.L.; Chen, J.; Zhang, B.P.; Li, Q.; Yu, X.B. One-step uniform growth of magnesium hydride nanoparticles on graphene. Prog. Nat. Sci.-Mater. 2017, 27, 87–93. [Google Scholar] [CrossRef]
- Jang, M.H.; Park, S.H.; Hong, T.W. Hydrogenation Behaviors of MgHx-Graphene Composites by Reactive Mechanical Grinding. Korean J. Met. Mater. 2016, 54, 288–294. [Google Scholar] [CrossRef]
- Cho, E.S.; Ruminski, A.M.; Aloni, S.; Liu, Y.-S.; Guo, J.; Urban, J.J. Graphene oxide/metal nanocrystal multilaminates as the atomic limit for safe and selective hydrogen storage. Nat. Commun. 2016, 7, 10804. [Google Scholar] [CrossRef]
- Pukazhselvan, D.; Shaula, A.L.; Mikhalev, S.M.; Bdikin, I.; Fagg, D.P. Elucidating Evidence for the In Situ Reduction of Graphene Oxide by Magnesium Hydride and the Consequence of Reduction on Hydrogen Storage. Catalysts 2022, 12, 735. [Google Scholar] [CrossRef]
- Han, D.J.; Bang, K.R.; Cho, H.; Cho, E.S. Effect of carbon nanoscaffolds on hydrogen storage performance of magnesium hydride. Korean J. Chem. Eng. 2020, 37, 1306–1316. [Google Scholar] [CrossRef]
- Singh, M.K.; Bhatnagar, A.; Pandey, S.K.; Mishra, P.C.; Srivastava, O.N. Experimental and first principle studies on hydrogen desorption behavior of graphene nanofibre catalyzed MgH2. Int. J. Hydrogen Energy 2017, 42, 960–968. [Google Scholar] [CrossRef]
- Zhang, J.; Yu, X.F.; Mao, C.; Long, C.G.; Chen, J.; Zhou, D.W. Influences and mechanisms of graphene-doping on dehydrogenation properties of MgH2: Experimental and first-principles studies. Energy 2015, 89, 957–964. [Google Scholar] [CrossRef]
- Zhang, L.T.; Chen, L.X.; Xiao, X.Z.; Fan, X.L.; Shao, J.; Li, S.Q.; Ge, H.W.; Wang, Q.D. Fluorographene nanosheets enhanced hydrogen absorption and desorption performances of magnesium hydride. Int. J. Hydrogen Energy 2014, 39, 12715–12726. [Google Scholar] [CrossRef]
- Verma, S.K.; Bhatnagar, A.; Shukla, V.; Soni, P.K.; Pandey, A.P.; Yadav, T.P.; Srivastava, O.N. Multiple improvements of hydrogen sorption and their mechanism for MgH2 catalyzed through TiH2@Gr. Int. J. Hydrogen Energy 2020, 45, 19516–19530. [Google Scholar] [CrossRef]
- Wang, K.K.; Wu, G.L.; Cao, H.J.; Li, H.L.; Zhao, X.S. Improved reversible dehydrogenation properties of MgH2 by the synergetic effects of graphene oxide-based porous carbon and TiCl3. Int. J. Hydrogen Energy 2018, 43, 7440–7446. [Google Scholar] [CrossRef]
- Wang, Y.; Ding, Z.M.; Li, X.J.; Ren, S.Q.; Zhou, S.H.; Zhang, H.M.; Li, Y.; Han, S.M. Improved hydrogen storage properties of MgH2 by nickel@nitrogen-doped carbon spheres. Dalton Trans. 2020, 49, 3495–3502. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, G.; An, C.H.; Li, L.; Qiu, F.Y.; Wang, Y.J.; Jiao, L.F.; Yuan, H.T. Bimetallic NiCo Functional Graphene: An Efficient Catalyst for Hydrogen-Storage Properties of MgH2. Chem.-Asian J. 2014, 9, 2576–2583. [Google Scholar] [CrossRef]
- Zhang, J.; Qu, H.; Yan, S.; Wu, G.; Yu, X.F.; Zhou, D.W. Catalytic effect of nickel phthalocyanine on hydrogen storage properties of magnesium hydride: Experimental and first-principles studies. Int. J. Hydrogen Energy 2017, 42, 28485–28497. [Google Scholar] [CrossRef]
- Xie, X.B.; Chen, M.; Liu, P.; Shang, J.X.; Liu, T. High hydrogen desorption properties of Mg-based nanocomposite at moderate temperatures: The effects of multiple catalysts in situ formed by adding nickel sulfides/graphene. J. Power Sources 2017, 371, 112–118. [Google Scholar] [CrossRef]
- Zhang, J.; Qu, H.; Wu, G.; Song, L.B.; Yu, X.F.; Zhou, D.W. Remarkably enhanced dehydrogenation properties and mechanisms of MgH2 by sequential-doping of nickel and graphene. Int. J. Hydrogen Energy 2016, 41, 17433–17441. [Google Scholar] [CrossRef]
- Zhang, J.G.; Zhu, Y.F.; Zang, X.X.; Huan, Q.Q.; Su, W.; Zhu, D.L.; Li, L.Q. Nickel-decorated graphene nanoplates for enhanced H-2 sorption properties of magnesium hydride at moderate temperatures. J. Mater. Chem. A 2016, 4, 2560–2570. [Google Scholar] [CrossRef]
- Zhang, L.T.; Cai, Z.L.; Zhu, X.Q.; Yao, Z.D.; Sun, Z.; Ji, L.; Yan, N.H.; Xiao, B.B.; Chen, L.X. Two-dimensional ZrCo nanosheets as highly effective catalyst for hydrogen storage in MgH2. J. Alloys Compd. 2019, 805, 295–302. [Google Scholar] [CrossRef]
- Leng, H.Y.; Miao, N.; Li, Q. Improved hydrogen storage properties of MgH2 by the addition of KOH and graphene. Int. J. Hydrogen Energy 2020, 45, 28183–28189. [Google Scholar] [CrossRef]
- Li, M.M.; Yang, C.C.; Chen, L.X.; Jiang, Q. Hydrogen storage alloys/reduced graphite oxide: An efficient hybrid electrode with enhanced high-rate dischargeability. Electrochim. Acta 2016, 200, 59–65. [Google Scholar] [CrossRef]
- Huang, H.X.; Yuan, J.G.; Zhang, B.; Zhang, J.G.; Zhu, Y.F.; Li, L.Q.; Wu, Y.; Zhou, S.X. Improvement in the hydrogenation-dehydrogenation performance of a Mg-Al alloy by graphene supported Ni. Int. J. Hydrogen Energy 2020, 45, 798–808. [Google Scholar] [CrossRef]
- Huang, X.T.; Tao, A.P.; Guo, J.; Wei, W.L.; Guo, J.; Lan, Z.Q. Synergistic effect of TiF3@ graphene on the hydrogen storage properties of Mg-Al alloy. Int. J. Hydrogen Energy 2018, 43, 1651–1657. [Google Scholar] [CrossRef]
- Lan, Z.Q.; Sun, Z.Z.; Ding, Y.C.; Ning, H.; Wei, W.L.; Guo, J. Catalytic action of Y2O3@graphene nanocomposites on the hydrogen-storage properties of Mg-Al alloys. J. Mater. Chem. A 2017, 5, 15200–15207. [Google Scholar] [CrossRef]
- Ji, L.; Zhang, L.T.; Yang, X.L.; Zhu, X.Q.; Chen, L.X. The remarkably improved hydrogen storage performance of MgH2 by the synergetic effect of an FeNi/rGO nanocomposite. Dalton Trans. 2020, 49, 4146–4154. [Google Scholar] [CrossRef]
- Liu, G.; Wang, K.F.; Li, J.P.; Wang, Y.J.; Yuan, H.T. Enhancement of hydrogen desorption in magnesium hydride catalyzed by graphene nanosheets supported Ni-CeOx hybrid nanocatalyst. Int. J. Hydrogen Energy 2016, 41, 10786–10794. [Google Scholar] [CrossRef]
- Liu, G.; Wang, Y.J.; Jiao, L.F.; Yuan, H.T. Solid-state synthesis of amorphous TiB2 nanoparticles on graphene nanosheets with enhanced catalytic dehydrogenation of MgH2. Int. J. Hydrogen Energy 2014, 39, 3822–3829. [Google Scholar] [CrossRef]
- Liu, J.C.; Liu, Y.N.; Liu, Z.B.; Ma, Z.L.; Ding, Y.J.; Zhu, Y.F.; Zhang, Y.; Zhang, J.G.; Li, L.Q. Effect of rGO supported NiCu derived from layered double hydroxide on hydrogen sorption kinetics of MgH2. J. Alloys Compd. 2019, 789, 768–776. [Google Scholar] [CrossRef]
- Zhou, D.M.; Cui, K.X.; Zhou, Z.W.; Liu, C.R.; Zhao, W.; Li, P.; Qu, X.H. Enhanced hydrogen-storage properties of MgH2 by Fe-Ni catalyst modified three-dimensional graphene. Int. J. Hydrogen Energy 2021, 46, 34369–34380. [Google Scholar] [CrossRef]
- Liu, J.C.; Ma, Z.L.; Liu, Z.B.; Tang, Q.K.; Zhu, Y.F.; Lin, H.J.; Zhang, Y.; Zhang, J.G.; Liu, Y.; Li, L.Q. Synergistic effect of rGO supported Ni3Fe on hydrogen storage performance of MgH2. Int. J. Hydrogen Energy 2020, 45, 16622–16633. [Google Scholar] [CrossRef]
- Luo, S.C.; Li, S.J.; Liu, Y.N.; Zhang, J.G.; Zhu, Y.F.; Zhang, Y.; Lin, H.J.; Li, L.Q. Synergistically tuned hydrogen storage thermodynamics and kinetics of Mg-Al alloys by Cu formed in situ mechanochemically. J. Alloys Compd. 2019, 806, 370–377. [Google Scholar] [CrossRef]
- Peng, D.D.; Ding, Z.M.; Zhang, L.; Fu, Y.K.; Wang, J.S.; Li, Y.; Han, S.M. Remarkable hydrogen storage properties and mechanisms of the shell-core MgH2@carbon aerogel microspheres. Int. J. Hydrogen Energy 2018, 43, 3731–3740. [Google Scholar] [CrossRef]
- Tian, M.; Shang, C.X. Mg-based composites for enhanced hydrogen storage performance. Int. J. Hydrogen Energy 2019, 44, 338–344. [Google Scholar] [CrossRef]
- Shukla, V.; Bhatnagar, A.; Verma, S.K.; Pandey, A.P.; Vishwakarma, A.K.; Srivastava, P.; Yadav, T.P.; Srivastava, O.N. Simultaneous improvement of kinetics and thermodynamics based on SrF2 and SrF2@Gr additives on hydrogen sorption in MgH2. Mater. Adv. 2021, 2, 4277–4290. [Google Scholar] [CrossRef]
- Song, M.C.; Zhang, L.T.; Zheng, J.G.; Yu, Z.D.; Wang, S.N. Constructing graphene nanosheet-supported FeOOH nanodots for hydrogen storage of MgH2. Int. J. Miner. Met. Mater. 2022, 29, 1464–1473. [Google Scholar] [CrossRef]
- Tarasov, B.P.; Arbuzov, A.A.; Mozhzhuhin, S.A.; Volodin, A.A.; Fursikov, P.V.; Lototskyy, M.V.; Yartys, V.A. Hydrogen storage behavior of magnesium catalyzed by nickel-graphene nanocomposites. Int. J. Hydrogen Energy 2019, 44, 29212–29223. [Google Scholar] [CrossRef]
- Zhang, B.P.; Xia, G.L.; Chen, W.; Gu, Q.F.; Sun, D.L.; Yu, X.B. Controlled-Size Hollow Magnesium Sulfide Nanocrystals Anchored on Graphene for Advanced Lithium Storage. ACS Nano 2018, 12, 12741–12750. [Google Scholar] [CrossRef] [PubMed]
- Zhao, X.D.; Ma, Z.N.; Wu, D.H.; Zhang, X.; Jing, Y.; Zhou, Z. Computational study of catalytic effect of C3N4 on H-2 release from complex hydrides. Int. J. Hydrogen Energy 2015, 40, 8897–8902. [Google Scholar] [CrossRef]
- Yu, H.Z.; Du, A.J.; Song, Y.; Searles, D.J. Graphyne and Graphdiyne: Versatile Catalysts for Dehydrogenation of Light Metal Complex Hydrides. J. Phys. Chem. C 2013, 117, 21643–21650. [Google Scholar] [CrossRef]
- Gasnier, A.; Luguet, M.; Pereira, A.G.; Troiani, H.; Zampieri, G.; Gennari, F.C. Entanglement of N-doped graphene in resorcinolformaldehyde: Effect over nanoconfined LiBH4 for hydrogen storage. Carbon 2019, 147, 284–294. [Google Scholar] [CrossRef]
- Gasnier, A.; Amica, G.; Juan, J.; Troiani, H.; Gennari, F.C. N-Doped Graphene-Rich Aerogels Decorated with Nickel and Cobalt Nanoparticles: Effect on Hydrogen Storage Properties of Nanoconfined LiBH4. J. Phys. Chem. C 2020, 124, 115–125. [Google Scholar] [CrossRef]
- Ye, J.K.; Xia, G.L.; Yu, X.B. In-situ constructed destabilization reaction of LiBH4 wrapped with graphene toward stable hydrogen storage reversibility. Mater. Today Energy 2021, 22, 100885. [Google Scholar] [CrossRef]
- Xu, G.; Zhang, W.; Zhang, Y.; Zhao, X.; Wen, P.; Ma, D. Fe3O4 Nanoclusters Highly Dispersed on a Porous Graphene Support as an Additive for Improving the Hydrogen Storage Properties of LiBH4. RSC Adv. 2018, 8, 19353–19361. [Google Scholar] [CrossRef]
- Gasnier, A.; Gennari, F.C. Graphene Entanglement in a Mesoporous Resorcinol-Formaldehyde Matrix Applied to the Nanoconfinement of LiBH4 for Hydrogen Storage. RSC Adv. 2017, 7, 27905–27912. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, L.; Zhang, W.; Ren, Z.; Huang, Z.; Hu, J.; Gao, M.; Pan, H.; Liu, Y. Nano-synergy enables highly reversible storage of 9.2 wt% hydrogen at mild conditions with lithium borohydride. Nano Energy 2021, 83, 105839. [Google Scholar] [CrossRef]
- Xia, Y.P.; Wei, S.; Huang, Q.; Li, J.Q.; Cen, X.H.; Zhang, H.Z.; Chu, H.L.; Sun, L.X.; Xu, F.; Huang, P.R. Facile synthesis of NiCo2O4-anchored reduced graphene oxide nanocomposites as efficient additives for improving the dehydrogenation behavior of lithium alanate. Inorg. Chem. Front. 2020, 7, 1257–1272. [Google Scholar] [CrossRef]
- Palade, P.; Comanescu, C.; Radu, C. Synthesis of Nickel and Cobalt Ferrite-Doped Graphene as Efficient Catalysts for Improving the Hydrogen Storage Kinetics of Lithium Borohydride. Materials 2023, 16, 427. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.T.; Ding, X.L.; Zhang, Q.G. Self-Printing on Graphitic Nanosheets with Metal Borohydride Nanodots for Hydrogen Storage. Sci. Rep. 2016, 6, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.Q.; Xia, G.L.; Zhang, J.; Guo, Z.P.; Yu, X.B. Graphene-tailored molecular bonds for advanced hydrogen and lithium storage performance. Energy Storage Mater. 2019, 17, 178–185. [Google Scholar] [CrossRef]
- Ren, Z.; Zhang, X.; Li, H.-W.; Huang, Z.; Hu, J.; Gao, M.; Pan, H.; Liu, Y. Titanium Hydride Nanoplates Enable 5 Wt% of Reversible Hydrogen Storage by Sodium Alanate below 80 °C. Research 2021, 2021, 9819176. [Google Scholar] [CrossRef]
- Kumar, L.H.; Rao, C.V.; Viswanathan, B. Catalytic effects of nitrogen-doped graphene and carbon nanotube additives on hydrogen storage properties of sodium alanate. J. Mater. Chem. A 2013, 1, 3355–3361. [Google Scholar] [CrossRef]
- Li, Y.T.; Fang, F.; Fu, H.L.; Qiu, J.M.; Song, Y.; Li, Y.S.; Sun, D.L.; Zhang, Q.G.; Ouyang, L.Z.; Zhu, M. Carbon nanomaterial-assisted morphological tuning for thermodynamic and kinetic destabilization in sodium alanates. J. Mater. Chem. A 2013, 1, 5238–5246. [Google Scholar] [CrossRef]
- Do, H.W.; Kim, H.; Cho, E.S. Enhanced hydrogen storage kinetics and air stability of nanoconfined NaAlH4 in graphene oxide framework. RSC Adv. 2021, 11, 32533–32540. [Google Scholar] [CrossRef]
- Huang, Y.K.; Shao, H.X.; Zhang, Q.Y.; Zang, L.; Guo, H.N.; Liu, Y.F.; Jiao, L.F.; Yuan, H.T.; Wang, Y.J. Layer-by-layer uniformly confined Graphene-NaAlH4 composites and hydrogen storage performance. Int. J. Hydrogen Energy 2020, 45, 28116–28122. [Google Scholar] [CrossRef]
- Qian, Z.; Hudson, M.S.L.; Raghubanshi, H.; Scheicher, R.H.; Pathak, B.; Araujo, C.M.; Blomqvist, A.; Johansson, B.; Srivastava, O.N.; Ahuja, R. Excellent Catalytic Effects of Graphene Nanofibers on Hydrogen Release of Sodium alanate. J. Phys. Chem. C 2012, 116, 10861–10866. [Google Scholar] [CrossRef]
- Wang, J.; Ebner, A.D.; Prozorov, T.; Zidan, R.; Ritter, J.A. Effect of graphite as a co-dopant on the dehydrogenation and hydrogenation kinetics of Ti-doped sodium aluminum hydride. J. Alloys Compd. 2005, 395, 252–262. [Google Scholar] [CrossRef]
- Zheng, H.Y.; Ding, Z.Q.; Xie, Y.J.; Li, J.F.; Huang, C.K.; Cai, W.T.; Liu, H.Z.; Guo, J. Cerium hydride generated during ball milling and enhanced by graphene for tailoring hydrogen sorption properties of sodium alanate. Int. J. Hydrogen Energy 2021, 46, 4168–4180. [Google Scholar] [CrossRef]
- Xu, L.Y.; Ge, Q.F. Effect of defects and dopants in graphene on hydrogen interaction in graphene-supported NaAlH4. Int. J. Hydrogen Energy 2013, 38, 3670–3680. [Google Scholar] [CrossRef]
- Zhang, H.Y.; Xia, G.L.; Zhang, J.; Sun, D.L.; Guo, Z.P.; Yu, X.B. Graphene-Tailored Thermodynamics and Kinetics to Fabricate Metal Borohydride Nanoparticles with High Purity and Enhanced Reversibility. Adv. Energy Mater. 2018, 8, 1702975. [Google Scholar] [CrossRef]
- Jeong, S.; Heo, T.W.; Oktawiec, J.; Shi, R.; Kang, S.Y.; White, J.L.; Schneemann, A.; Zaia, E.W.; Wan, L.F.; Ray, K.G.; et al. A Mechanistic Analysis of Phase Evolution and Hydrogen Storage Behavior in Nanocrystalline Mg(BH4)2 within Reduced Graphene Oxide. ACS Nano 2020, 14, 1745–1756. [Google Scholar] [CrossRef] [PubMed]
- White, J.L.; Strange, N.A.; Sugar, J.D.; Snider, J.L.; Schneemann, A.; Lipton, A.S.; Toney, M.F.; Allendorf, M.D.; Stavila, V. Melting of Magnesium Borohydride under High Hydrogen Pressure: Thermodynamic Stability and Effects of Nanoconfinement. Chem. Mater. 2020, 32, 5604–5615. [Google Scholar] [CrossRef]
- Wang, Z.M.; Tao, S.; Li, J.J.; Deng, J.Q.; Zhou, H.Y.; Yao, Q.R. The Improvement of Dehydriding the Kinetics of NaMgH3 Hydride via Doping with Carbon Nanomaterials. Metal 2017, 7, 9. [Google Scholar] [CrossRef]
- Bhatnagar, A.; Pandey, S.K.; Shahi, R.R.; Hudson, M.S.L.; Shaz, M.A.; Srivastava, O.N. Synthesis, characterization and hydrogen sorption studies of mixed sodium-potassium alanate. Cryst. Res. Technol. 2013, 48, 520–531. [Google Scholar] [CrossRef]
- Varin, R.A.; Bidabadi, A.S.; Polanski, M.; Biglari, M.; Stobinski, L. The effects of filamentary Ni, graphene and lithium amide (LiNH2) additives on the dehydrogenation behavior of mechano-chemically synthesized crystalline manganese borohydride (Mn(BH4)(2)) and its solvent filtration/extraction. Mater. Res. Bull. 2018, 100, 394–406. [Google Scholar] [CrossRef]
- Li, N.; Du, Y.; Feng, Q.P.; Huang, G.W.; Xiao, H.M.; Fu, S.Y. A Novel Type of Battery-Supercapacitor Hybrid Device with Highly Switchable Dual Performances Based on a Carbon Skeleton/Mg2Ni Free-Standing Hydrogen Storage Electrode. ACS Appl. Mater. Interfaces 2017, 9, 44828–44838. [Google Scholar] [CrossRef]
- Du, Y.; Li, N.; Zhang, T.L.; Feng, Q.P.; Du, Q.; Wu, X.H.; Huang, G.W. Reduced Graphene Oxide Coating with Anticorrosion and Electrochemical Property-Enhancing Effects Applied in Hydrogen Storage System. ACS Appl. Mater. Interfaces 2017, 9, 28980–28989. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.G.; Zhu, Y.F.; Lin, H.J.; Liu, Y.N.; Zhang, Y.; Li, S.Y.; Ma, Z.L.; Li, L.Q. Metal Hydride Nanoparticles with Ultrahigh Structural Stability and Hydrogen Storage Activity Derived from Microencapsulated Nanoconfinement. Adv. Mater. 2017, 29, 1700760. [Google Scholar] [CrossRef] [PubMed]
- Puszkiel, J.; Gasnier, A.; Amica, G.; Gennari, F. Tuning LiBH4 for Hydrogen Storage: Destabilization, Additive, and Nanoconfinement Approaches. Molecules 2020, 25, 163. [Google Scholar] [CrossRef] [PubMed]
- Xia, G.L.; Tan, Y.B.; Wu, F.L.; Fang, F.; Sun, D.L.; Guo, Z.P.; Huang, Z.G.; Yu, X.B. Graphene-wrapped reversible reaction for advanced hydrogen storage. Nano Energy 2016, 26, 488–495. [Google Scholar] [CrossRef]
- Chou, C.C.; Chen, B.H.; Lee, D.J. Hydrogen storage in a chemical hydride fuel system containing ammonia borane and Ni-Co/r-GO catalyst. Enregy Procedia 2014, 61, 142–145. [Google Scholar] [CrossRef]
- Li, J.L.; Ren, X.Y.; Lv, H.; Wang, Y.Y.; Li, Y.F.; Liu, B. Highly efficient hydrogen production from hydrolysis of ammonia borane over nanostructured Cu@CuCoOx supported on graphene oxide. J. Hazard. Mater. 2020, 391, 122199. [Google Scholar] [CrossRef]
- Champet, S.; van den Berg, J.; Szczesny, R.; Godula-Jopek, A.; Gregory, D.H. Nano-inclusion in one step: Spontaneous icetemplating of porous hierarchical nanocomposites for selective hydrogen release. Sustain. Energy Fuels 2019, 3, 396–400. [Google Scholar] [CrossRef]
- Liu, X.R.; Wu, Y.F.; Wang, S.M.; Li, Z.N.; Guo, X.M.; Ye, J.H.; Jiang, L.J. Current progress and research trends on lithium amidoborane for hydrogen storage. J. Mater. Sci. 2020, 55, 2645–2660. [Google Scholar] [CrossRef]
- Kumar, P.; Singh, S.; Hashmi, S.A.R.; Kim, K.H. MXenes: Emerging 2D materials for hydrogen storage. Nano Energy 2021, 85, 105989. [Google Scholar] [CrossRef]
- Shi, L.J.; Liu, X.J.; Tuo, Y.X.; Xu, J.; Li, P.; Han, Y.F. Graphene CNT composite as catalyst support for microwave-assisted hydrogen releasing from liquid organic hydride. Int. J. Hydrogen Energy 2017, 42, 17403–17413. [Google Scholar] [CrossRef]
- Ventura-Espinosa, D.; Carretero-Cerdan, A.; Baya, M.; Garcia, H.; Mata, J.A. Catalytic Dehydrogenative Coupling of Hydrosilanes with Alcohols for the Production of Hydrogen On-demand: Application of a Silane/Alcohol Pair as a Liquid Organic Hydrogen Carrier. Chem.-Eur. J. 2017, 23, 10815–10821. [Google Scholar] [CrossRef] [PubMed]
- He, T.; Wang, X.H.; Liu, H.Z.; Gao, S.C.; Wang, Y.Y.; Li, S.Q.; Yan, M. Enhanced hydrogen desorption/absorption properties of magnesium hydride with CeF3@Gn. Int. J. Hydrogen Energy 2020, 45, 4754–4764. [Google Scholar] [CrossRef]
- Li, Q.Y.; Qiu, S.Y.; Wu, C.Z.; Lau, T.; Sun, C.H.; Jia, B.H. Computational Investigation of MgH2/Graphene Heterojunctions for Hydrogen Storage. J. Phys. Chem. C 2021, 125, 2357–2363. [Google Scholar] [CrossRef]
- Sigal, A.; Rojas, M.I.; Leiva, E.P.M. Interferents for hydrogen storage on a graphene sheet decorated with nickel: A DFT study. Int. J. Hydrogen Energy 2011, 36, 3537–3546. [Google Scholar] [CrossRef]
- Singh, S.; Bhatnagar, A.; Shukla, V.; Vishwakarma, A.K.; Soni, P.K.; Verma, S.K.; Shaz, M.A.; Sinha, A.S.K.; Srivastava, O.N. Ternary transition metal alloy FeCoNi nanoparticles on graphene as new catalyst for hydrogen sorption in MgH2. Int. J. Hydrogen Energy 2020, 45, 774–786. [Google Scholar] [CrossRef]
- Al-Msrhad, T.M.H.; Devrim, Y.; Uzundurukan, A.; Budak, Y. Investigation of hydrogen production from sodium borohydride by carbon nano tube-graphene supported PdRu bimetallic catalyst for PEM fuel cell application. Int. J. Energy Res. 2021, 46, 4156–4173. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).