Single Crystal X-Ray Structure for the Disordered Two Independent Molecules of Novel Isoflavone: Synthesis, Hirshfeld Surface Analysis, Inhibition and Docking Studies on IKKβ of 3-(2,3-dihydrobenzo[b][1,4]dioxin-6-yl)-6,7-dimethoxy-4H-chromen-4-one
Abstract
1. Introduction
2. Materials and Methods
2.1. General
2.2. Crystal Structure Determination
2.3. Hirshfeld Surfaces
2.4. In Silico Docking with IκB Kinaseβ (IKKβ)
2.5. Cells and Cell Culture
2.6. Western Blot Analysis
2.7. Clonogenic Assay
3. Results and Discussion
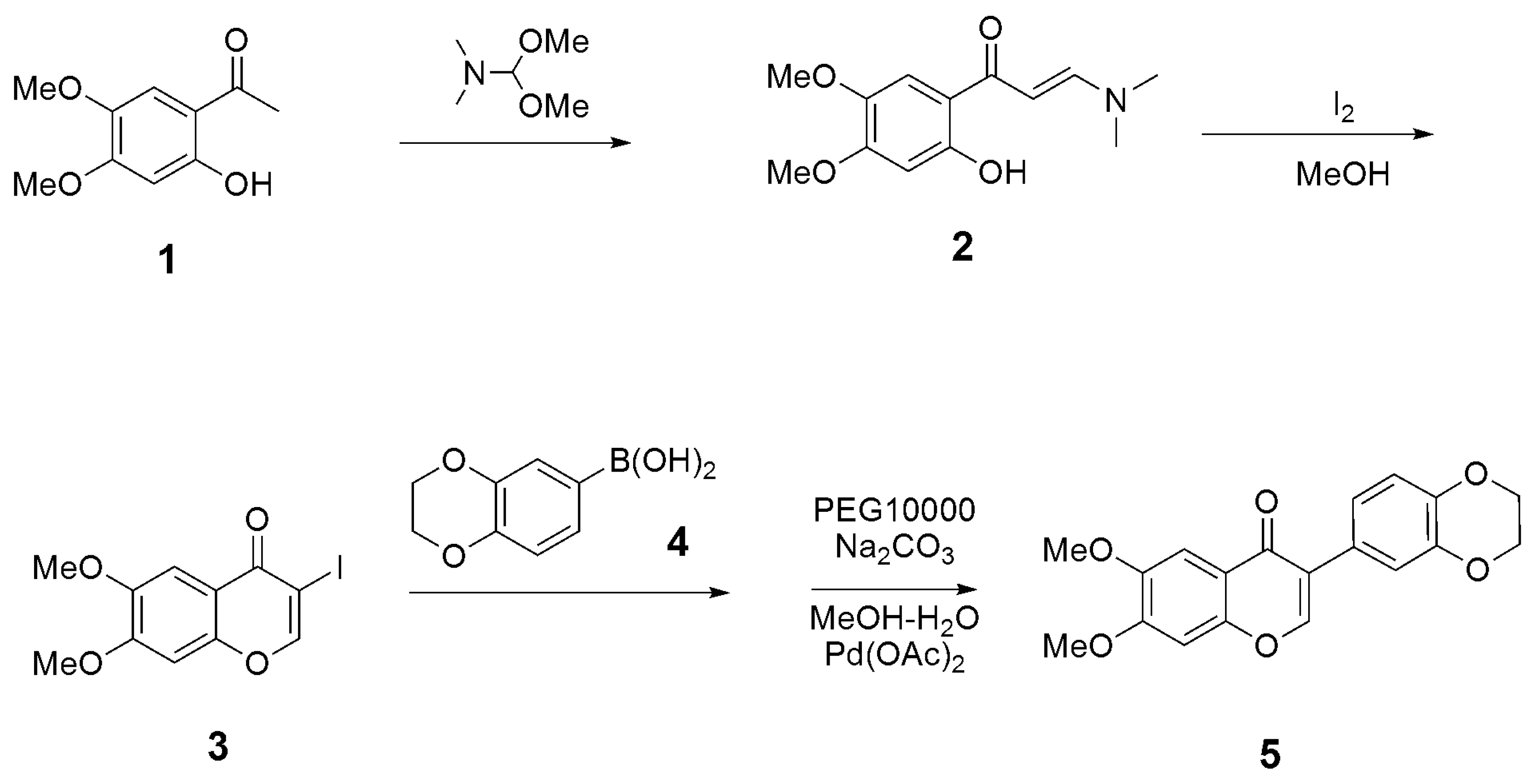
3.1. Synthesis
3.1.1. Synthesis of (E)-3-(dimethylamino)-1-(2-hydroxy-4,5-dimethoxyphenyl)prop-2-en-1-one (2)
3.1.2. Synthesis of 3-iodo-6,7-dimethoxy-4H-chromen-4-one (3)
3.1.3. Synthesis of 3-(2,3-dihydrobenzo[b][1,4]dioxin-6-yl)-6,7-dimethoxy-4H-chromen-4-one (5)
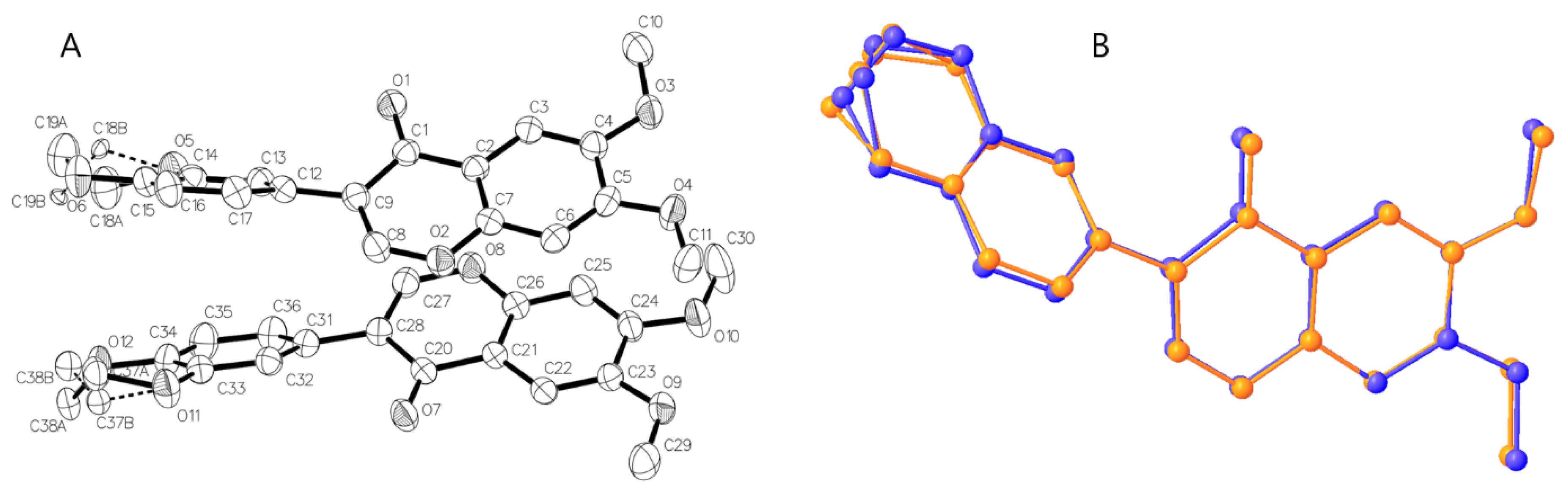
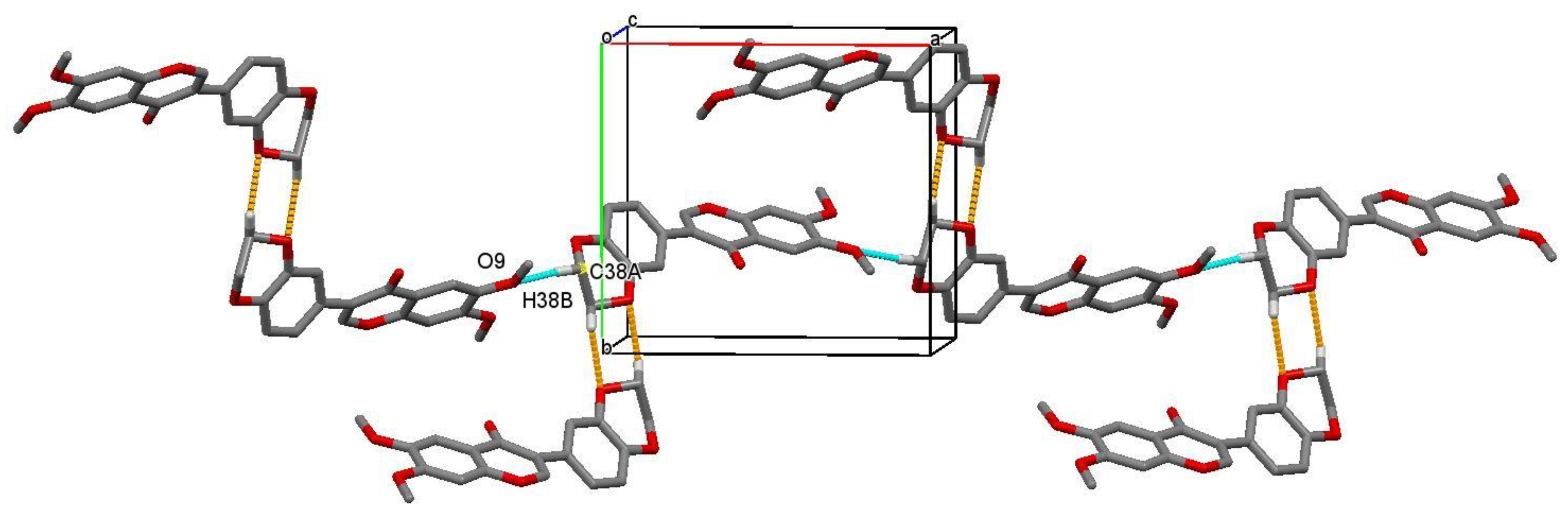
3.2. Crystal Structure of Isoflavone Compound 5
3.3. Hirshfeld Surface analysis of Compound 5
3.4. In Silico Docking with IKKβ
3.5. Effect of Compound 5 on the Inhibition of IKK Pathway
3.6. Effect of Compound 5 on the Inhibition of Clonogenicity of HCT116 Cells
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Luo, J.-L.; Kamata, H.; Karin, M. IKK/NF-κB signaling: Balancing life and death—A new approach to cancer therapy. J. Clin. Investig. 2005, 115, 2625–2632. [Google Scholar] [CrossRef] [PubMed]
- Tang, E.D.; Inohara, N.; Wang, C.-Y.; Nuñez, G.; Guan, K.-L. Roles for homotypic interactions and transautophosphorylation in IκB kinase beta (IKKβ) activation. J. Biol. Chem. 2003, 278, 38566–38570. [Google Scholar] [CrossRef] [PubMed]
- Chen, A.; Koehler, A.N. Transcription Factor Inhibition: Lessons Learned and Emerging Targets. Trends Mol. Med. 2020, 26, 508–518. [Google Scholar] [CrossRef]
- Karin, M.; Cao, Y.; Greten, F.R.; Li, Z.W. NF-kappaB in cancer: From innocent bystander to major culprit. Nat. Rev. Cancer 2002, 2, 301–310. [Google Scholar] [CrossRef]
- Balkwill, F. Tumour necrosis factor and cancer. Nat. Rev. Cancer 2009, 9, 361–371. [Google Scholar] [CrossRef]
- Gaur, U.; Aggarwal, B.B. Regulation of proliferation, survival and apoptosis by members of the TNF superfamily. Biochem. Pharmacol. 2003, 66, 1403–1408. [Google Scholar] [CrossRef]
- Wilson, G.L.; Lill, M.A. Integrating structure-based and ligand-based approaches for computational drug design. Future Med. Chem. 2011, 3, 735–750. [Google Scholar] [CrossRef]
- Batool, M.; Ahmad, B.; Choi, S. A Structure-Based Drug Discovery Paradigm. Int. J. Mol. Sci. 2019, 20, 2783. [Google Scholar] [CrossRef]
- Rushe, M.; Silvian, L.; Bixler, S.; Chen, L.L.; Cheung, A.; Bowes, S.; Cuervo, H.; Berkowitz, S.; Zheng, T.; Guckian, K.; et al. Structure of a NEMO/IKK—Associating Domain Reveals Architecture of the Interaction Site. Structure 2008, 16, 798–808. [Google Scholar] [CrossRef]
- Xu, G.; Lo, Y.-C.; Li, Q.; Napolitano, G.; Wu, X.; Jiang, X.; Dreano, M.; Karin, M.; Wu, H. Crystal structure of inhibitor of κB kinase β. Nat. Cell Biol. 2011, 472, 325–330. [Google Scholar] [CrossRef]
- Dong, T.; Li, C.; Wang, X.; Dian, L.; Zhang, X.; Li, L.; Chen, S.; Cao, R.; Li, L.; Huang, N.; et al. Ainsliadimer A selectively inhibits IKKα/β by covalently binding a conserved cysteine. Nat. Commun. 2015, 6, 6522. [Google Scholar] [CrossRef] [PubMed]
- Elkamhawy, A.; Kim, N.Y.; Hassan, A.H.; Park, J.-E.; Paik, S.; Yang, J.-E.; Oh, K.-S.; Lee, B.H.; Lee, M.Y.; Shin, K.J.; et al. Thiazolidine-2,4-dione-based irreversible allosteric IKK-β kinase inhibitors: Optimization into in vivo active anti-inflammatory agents. Eur. J. Med. Chem. 2020, 188, 111955. [Google Scholar] [CrossRef] [PubMed]
- Heyninck, K.; Lahtela-Kakkonen, M.; Van der Veken, P.; Haegeman, G.; Vanden Berghe, W. Withaferin a inhibits NK-κB activation by targeting cysteine 179 in IKKβ. Biochem. Pharmacol. 2014, 91, 501–509. [Google Scholar] [CrossRef] [PubMed]
- Sordi, R.; Chiazza, F.; Johnson, F.L.; Patel, N.S.A.; Brohi, K.; Collino, M.; Thiemermann, C. Inhibition of IκB Kinase Attenuates the Organ Injury and Dysfunction Associated with Hemorrhagic Shock. Mol. Med. 2015, 21, 563–575. [Google Scholar] [CrossRef] [PubMed]
- Uota, S.; Dewan, Z.; Saitoh, Y.; Muto, S.; Itai, A.; Utsunomiya, A.; Watanabe, T.; Yamamoto, N.; Yamaoka, S. An IκB kinase 2 inhibitor IMD-0354 suppresses the survival of adult T-cell leukemia cells. Cancer Sci. 2011, 103, 100–106. [Google Scholar] [CrossRef]
- Choi, S.I.; Lee, S.Y.; Jung, W.J.; Lee, S.H.; Lee, E.J.; Min, K.H.; Hur, G.Y.; Lee, S.H.; Lee, S.Y.; Kim, J.H.; et al. The effect of an IκB-kinase-beta (IKKβ) inhibitor on tobacco smoke-induced pulmonary inflammation. Exp. Lung Res. 2016, 42, 182–189. [Google Scholar] [CrossRef]
- Bassères, D.S.; Ebbs, A.; Cogswell, P.C.; Baldwin, A.S. IKK is a therapeutic target in KRSA-induced lung cancer with disrupted p53 activity. Genes Cancer 2014, 5, 41–55. [Google Scholar] [CrossRef]
- Nan, J.; Du, Y.; Chen, X.; Bai, Q.; Wang, Y.; Zhang, X.; Zhu, N.; Zhang, J.; Hou, J.; Wang, Q.; et al. TPCA-1 is a direct dual inhibitor of STAT3 and NK-κB and regresses mutant EGFR-associated human non-small cell lung cancers. Mol. Cancer Ther. 2014, 13, 617–629. [Google Scholar] [CrossRef]
- Liu, Q.; Wu, H.; Chim, S.M.; Zhou, L.; Zhao, J.; Feng, H.; Wei, Q.; Wang, Q.; Zheng, M.H.; Tan, R.X.; et al. SC-514, a selective inhibitor of IKKβ attenuates RANKL-induced osteoclastogenesis and NK-κB activation. Biochem. Pharmacol. 2013, 86, 1775–1783. [Google Scholar] [CrossRef]
- Deng, C.; Lipstein, M.; Rodriguez, R.; Serrano, X.O.; McIntosh, C.; Tsai, W.Y.; Wasmuth, A.S.; Jaken, S.; O’Connor, O.A. The novel IKK2 inhibitor LY2409881 potently synergizes with histone deacetylase inhibitors in preclinical models of lymphoma through the downregulation of NK-κB. Clin. Cancer Res. 2015, 21, 134–145. [Google Scholar] [CrossRef]
- Ping, H.; Yang, F.; Wang, M.; Niu, Y.; Xing, N. IKK inhibitor suppresses epithelial-mesenchymal transition and induces cell death in prostate cancer. Oncol. Rep. 2016, 36, 1658–1664. [Google Scholar] [CrossRef] [PubMed]
- Yan, F.; Yang, F.; Wang, R.; Yao, X.J.; Bai, L.; Zeng, X.; Huang, J.; Wong, V.K.W.; Lam, C.W.K.; Zhou, H.; et al. Isoliquiritigenin suppresses human T Lymphocyte activation via covalently binding cysteine 46 of IκB kinase. Oncotarget 2016, 8, 34223–34235. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.Y.; Woo, Y.; Hyun, J.; Yong, Y.; Koh, D.; Lee, Y.H.; Lim, Y. Relationship between the structures of flavonoids and their NF-κB-dependent transcriptional activities. Bioorg. Med. Chem. Lett. 2011, 21, 6036–6041. [Google Scholar] [CrossRef] [PubMed]
- Jeandet, P. Phytolexins: Current progress and future prospects. Molecules 2015, 20, 2770–2774. [Google Scholar] [CrossRef]
- Guven, H.; Arici, A.; Simsek, O. Flavonoids in Our Foods: A Short Review. J. Basic Clin. Health Sci. 2019, 3, 96–106. [Google Scholar] [CrossRef]
- Zhao, L.; Yuan, X.; Wang, J.; Feng, Y.; Ji, F.; Li, Z.; Bian, J. A review on flavones targeting serine/threonine protein kinases for potential anticancer drugs. Bioorg. Med. Chem. 2019, 27, 677–685. [Google Scholar] [CrossRef]
- Zhuang, C.; Zhang, W.; Sheng, C.; Zhang, W.; Xing, C.; Miao, Z. Chalcone: A Privileged Structure in Medicinal Chemistry. Chem. Rev. 2017, 117, 7762–7810. [Google Scholar] [CrossRef]
- Singh, M.; Kaur, M.; Silakari, O. Flavones: An important scaffold for medicinal chemistry. Eur. J. Med. Chem. 2014, 84, 206–239. [Google Scholar] [CrossRef]
- Seo, G.; Hyun, C.; Koh, D.; Park, S.; Lim, Y.; Kim, Y.M.; Cho, M. A Novel Synthetic Material, BMM, Accelerates Wound Repair by Stimulating Re-Epithelialization and Fibroblast Activation. Int. J. Mol. Sci. 2018, 19, 1164. [Google Scholar] [CrossRef]
- Sophors, P.; Kim, Y.M.; Seo, G.; Huh, J.-S.; Lim, Y.; Koh, D.S.; Cho, M. A synthetic isoflavone, DCMF, promotes human keratinocyte migration by activating Src/FAK signaling pathway. Biochem. Biophys. Res. Commun. 2016, 472, 332–338. [Google Scholar] [CrossRef]
- Shin, S.Y.; Lee, J.M.; Lee, M.S.; Koh, D.; Jung, H.; Lim, Y.; Lee, Y.H. Targeting Cancer Cells via the Reactive Oxygen Species-Mediated Unfolded Protein Response with a Novel Synthetic Polyphenol Conjugate. Clin. Cancer Res. 2014, 20, 4302–4313. [Google Scholar] [CrossRef]
- Shin, S.; Yoon, H.; Ahn, S.; Kim, D.-W.; Bae, D.-H.; Koh, D.; Lee, Y.H.; Lim, Y. Structural Properties of Polyphenols Causing Cell Cycle Arrest at G1 Phase in HCT116 Human Colorectal Cancer Cell Lines. Int. J. Mol. Sci. 2013, 14, 16970–16985. [Google Scholar] [CrossRef]
- Jo, G.; Ahn, S.; Kim, B.-G.; Park, H.R.; Kim, Y.H.; Choo, H.A.; Koh, D.; Chong, Y.; Ahn, J.-H.; Lim, Y. Chromenylchalcones with inhibitory effects on monoamine oxidase B. Bioorg. Med. Chem. 2013, 21, 7890–7897. [Google Scholar] [CrossRef]
- Ahn, S.; Sung, J.; Lee, J.H.; Yoo, M.; Lim, Y.; Shin, S.Y.; Koh, D. Synthesis, Single Crystal X-Ray Structure, Hirshfeld Surface Analysis, DFT Computations, Docking Studies on Aurora Kinases and an Anticancer Property of 3-(2,3-Dihydrobenzo[b][1,4]dioxin-6-yl)-6-methoxy-4H-chromen-4-one. Crystals 2020, 10, 413. [Google Scholar] [CrossRef]
- Hwang, D.; Hyun, J.; Jo, G.; Koh, D.; Lim, Y. Synthesis and complete assignment of NMR data of 20 chalcones. Magn. Reson. Chem. 2010, 49, 41–45. [Google Scholar] [CrossRef]
- Bruker. APEX2, SAINT and SADABS; Bruker AXS Inc.: Madison, WI, USA, 2012. [Google Scholar]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Turner, M.J.; McKinnon, J.J.; Wolff, S.K.; Grimwood, D.J.; Spackman, P.R.; Jayatilaka, D.; Spackman, M.A. CrystalExplorer17; University of Western Australia: Crawley, Australia, 2017. [Google Scholar]
- McKinnon, J.J.; Jayatilaka, D.; Spackman, M.A. Towards quantitative analysis of intermolecular interactions with Hirshfeld surfaces. Chem. Commun. 2007, 37, 3814–3816. [Google Scholar] [CrossRef]
- Liu, S.; Misquitta, Y.R.; Olland, A.; Johnson, M.A.; Kelleher, K.S.; Kriz, R.; Lin, L.L.; Stahl, M.; Mosyak, L. Crystal Structure of a Human IκB Kinase β Asymmetric Dimer. J. Biol. Chem. 2013, 288, 22758–22767. [Google Scholar] [CrossRef]
- Shin, S.Y.; Lee, Y.; Kim, B.S.; Lee, J.; Ahn, S.; Koh, D.; Lim, Y.; Lee, Y.H. Inhibitory Effect of Synthetic Flavone Derivatives on Pan-Aurora Kinases: Induction of G2/M Cell-Cycle Arrest and Apoptosis in HCT116 Human Colon Cancer Cells. Int. J. Mol. Sci. 2018, 19, 4086. [Google Scholar] [CrossRef]
- Franken, N.A.P.; Rodermond, H.M.; Stap, J.; Haveman, J.; Van Bree, C. Clonogenic assay of cells in vitro. Nat. Protoc. 2006, 1, 2315–2319. [Google Scholar] [CrossRef]
- Kim, B.S.; Shin, S.Y.; Ahn, S.; Koh, D.; Lee, Y.H.; Lim, Y. Biological evaluation of 2-pyrazolinyl-1-carbothioamide derivatives against HCT116 human colorectal cancer cell lines and elucidation on QSAR and molecular binding modes. Bioorg. Med. Chem. 2017, 25, 5423–5432. [Google Scholar] [CrossRef] [PubMed]
- Biegasiewicz, K.F.; Gordon, J.S.; Rodriguez, D.A.; Priefer, R. Development of a general approach to the synthesis of a library of isoflavonoid derivatives. Tetrahedron Lett. 2014, 55, 5210–5212. [Google Scholar] [CrossRef]
- Liu, L.; Zhang, Y.; Wang, Y. Phosphine-free palladium acetate catalyzed Suzuki reaction in water. J. Org. Chem. 2005, 70, 6122–6125. [Google Scholar] [CrossRef] [PubMed]
- Bisseyou, Y.B.M.; Ouattara, M.; Soro, P.A.; Kakou-Yao, R.; Tenon, A.J. Crystal structure, Hirshfeld surface analysis and contact enrichment ratios of 1-(2,7-dimethylimidazo[1,2-a]pyridin-3-yl)-2-(1,3-dithiolan-2-ylidene)ethanone monohydrate. Acta Crystallogr. Sect. E Crystallogr. Commun. 2019, 75, 1934–1939. [Google Scholar] [CrossRef] [PubMed]
- Spackman, M.A.; Jayatilaka, D. Hirshfeld surface analysis. CrystEngComm 2009, 11, 19–32. [Google Scholar] [CrossRef]
- Cardoso, M.V.D.O.; Moreira, D.R.M.; Filho, G.B.O.; Cavalcanti, S.M.T.; Coelho, L.C.D.; Espíndola, J.W.P.; Gonzalez, L.R.; Rabello, M.M.; Hernandes, M.Z.; Ferreira, P.M.P.; et al. Design, synthesis and structure–activity relationship of phthalimides endowed with dual antiproliferative and immunomodulatory activities. Eur. J. Med. Chem. 2015, 96, 491–503. [Google Scholar] [CrossRef]
- Hu, M.C.-T.; Lee, D.-F.; Xia, W.; Golfman, L.S.; Ou-Yang, F.; Yang, J.-Y.; Zou, Y.; Bao, S.; Hanada, N.; Saso, H.; et al. IκB Kinase Promotes Tumorigenesis through Inhibition of Forkhead FOXO3a. Cell 2004, 117, 225–237. [Google Scholar] [CrossRef]
- Gao, Z.; Hwang, D.; Bataille, F.; Lefevre, M.; York, D.; Quon, M.J.; Ye, J. Serine Phosphorylation of Insulin Receptor Substrate 1 by Inhibitor κB Kinase Complex. J. Biol. Chem. 2002, 277, 48115–48121. [Google Scholar] [CrossRef]
- Karin, M.; Yamamoto, Y.; Wang, Q.M. The IKK NF-κB system: A treasure trove for drug development. Nat. Rev. Drug Discov. 2004, 3, 17–26. [Google Scholar] [CrossRef]









| CCDC deposit number | 2027508 |
| Empirical formula | C19 H16 O6 |
| Formula weight | 340.32 |
| Temperature | 223(2) K |
| Wavelength | 0.71073 Å |
| Crystal system | Monoclinic |
| Space group | P21/c |
| Unit cell dimensions | a = 12.0654(5) Å b = 11.0666(5) Å c = 123.9550(11) Å β = 101.3757(16)°. |
| Volume | 3135.7(2) Å3 |
| Z | 8 |
| Density (calculated) | 1.442 Mg/m3 |
| Absorption coefficient | 0.108 mm−1 |
| F(000) | 1424 |
| Crystal size | 0.393 × 0.305 × 0.134 mm3 |
| Theta range for data collection | 1.722 to 28.347° |
| Index ranges | −16 ≤ h ≤ 16, −14 ≤ k ≤ 14, −31 ≤ l ≤ 31 |
| Reflections collected | 102751 |
| Independent reflections | 7798 [R(int) = 0.0604] |
| Completeness to theta = 25.242° | 99.7 % |
| Max. and min. transmission | 0.7457 and 0.6970 |
| Refinement method | Full-matrix least-squares on F2 |
| Data/restraints/parameters | 7798/12/492 |
| Goodness-of-fit on F2 | 1.063 |
| Final R indices [I>2sigma(I)] | R1 = 0.0430, wR2 = 0.1004 |
| R indices (all data) | R1 = 0.0666, wR2 = 0.1179 |
| Largest diff. peak and hole | 0.226 and −0.206 e.Å−3 |
| I | II | ||
|---|---|---|---|
| O(5)–C(18A) | 1.412(4) | O(11)–C(37A) | 1.447(3) |
| O(5)–C(18B) | 1.513(7) | O(11)–C(37B) | 1.422(14) |
| C(18A)–C(19A) | 1.506(8) | C(37A)–C(38A) | 1.507(6) |
| C(18B)–C(19B) | 1.458(13) | C(37B)–C(38B) | 1.45(3) |
| C(19A)–O(6) | 1.495(4) | C(38A)–O(12) | 1.452(3) |
| C(19B)–O(6) | 1.402(6) | C(38B)–O(12) | 1.449(15) |
| O(5)–C(18A)–C(19A) | 107.5(4) | O(11)–C(37A)–C(38A) | 109.2(3) |
| C(18A)–C(19A)–O(6) | 109.5(4) | O(12)–C(38A)–C(37A) | 108.8(3) |
| O(5)–C(18B)–C(19B) | 113.2(7) | C(38B)–C(37B)–O(11) | 110.9(15) |
| O(6)–C(19B)–C(18B) | 106.5(8) | C(37B)–C(38B)–O(12) | 105.2(16) |
| C(15)–O(6)–C(19A) | 111.59(18) | C(34)–O(12)–C(38A) | 113.50(15) |
| C(15)–O(6)–C(19B) | 113.9(3) | C(34)–O(12)–C(38B) | 108.9(5) |
| C(3)–C(4)–O(3)–C(10) | 10.6(3) | C(22)–C(23)–O(9)–C(29) | 2.6(2) |
| C(6)–C(5)–O(4)–C(11) | −5.5(2) | C(25)–C(24)–O(10)–C(30) | −1.5(3) |
| D–H…A | d(D–H) | d(H…A) | d(D…A) | <(DHA) |
|---|---|---|---|---|
| C(29)–H(29B)…O(1)#1 | 0.97 | 2.43 | 3.396(2) | 172.2 |
| C(38A)–H(38B)…O(9)#2 | 0.98 | 2.46 | 3.396(3) | 157.8 |
| C(10)–H(10B)…O(7)#3 | 0.97 | 2.48 | 3.424(2) | 165.6 |
| C(37A)–H(37B)…O(11)#4 | 0.98 | 2.59 | 3.071(4) | 110.7 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shin, S.Y.; Lee, Y.H.; Lim, Y.; Lee, H.J.; Lee, J.H.; Yoo, M.; Ahn, S.; Koh, D. Single Crystal X-Ray Structure for the Disordered Two Independent Molecules of Novel Isoflavone: Synthesis, Hirshfeld Surface Analysis, Inhibition and Docking Studies on IKKβ of 3-(2,3-dihydrobenzo[b][1,4]dioxin-6-yl)-6,7-dimethoxy-4H-chromen-4-one. Crystals 2020, 10, 911. https://doi.org/10.3390/cryst10100911
Shin SY, Lee YH, Lim Y, Lee HJ, Lee JH, Yoo M, Ahn S, Koh D. Single Crystal X-Ray Structure for the Disordered Two Independent Molecules of Novel Isoflavone: Synthesis, Hirshfeld Surface Analysis, Inhibition and Docking Studies on IKKβ of 3-(2,3-dihydrobenzo[b][1,4]dioxin-6-yl)-6,7-dimethoxy-4H-chromen-4-one. Crystals. 2020; 10(10):911. https://doi.org/10.3390/cryst10100911
Chicago/Turabian StyleShin, Soon Young, Young Han Lee, Yoongho Lim, Ha Jin Lee, Ji Hye Lee, Miri Yoo, Seunghyun Ahn, and Dongsoo Koh. 2020. "Single Crystal X-Ray Structure for the Disordered Two Independent Molecules of Novel Isoflavone: Synthesis, Hirshfeld Surface Analysis, Inhibition and Docking Studies on IKKβ of 3-(2,3-dihydrobenzo[b][1,4]dioxin-6-yl)-6,7-dimethoxy-4H-chromen-4-one" Crystals 10, no. 10: 911. https://doi.org/10.3390/cryst10100911
APA StyleShin, S. Y., Lee, Y. H., Lim, Y., Lee, H. J., Lee, J. H., Yoo, M., Ahn, S., & Koh, D. (2020). Single Crystal X-Ray Structure for the Disordered Two Independent Molecules of Novel Isoflavone: Synthesis, Hirshfeld Surface Analysis, Inhibition and Docking Studies on IKKβ of 3-(2,3-dihydrobenzo[b][1,4]dioxin-6-yl)-6,7-dimethoxy-4H-chromen-4-one. Crystals, 10(10), 911. https://doi.org/10.3390/cryst10100911





