Co(II/III) Complexes with Benzoxazole and Benzothiazole Ligands as Efficient Heterogenous Photocatalysts for Organic Dyes Degradation
Abstract
1. Introduction
2. Results and Discussion
2.1. Synthesis and Characterization
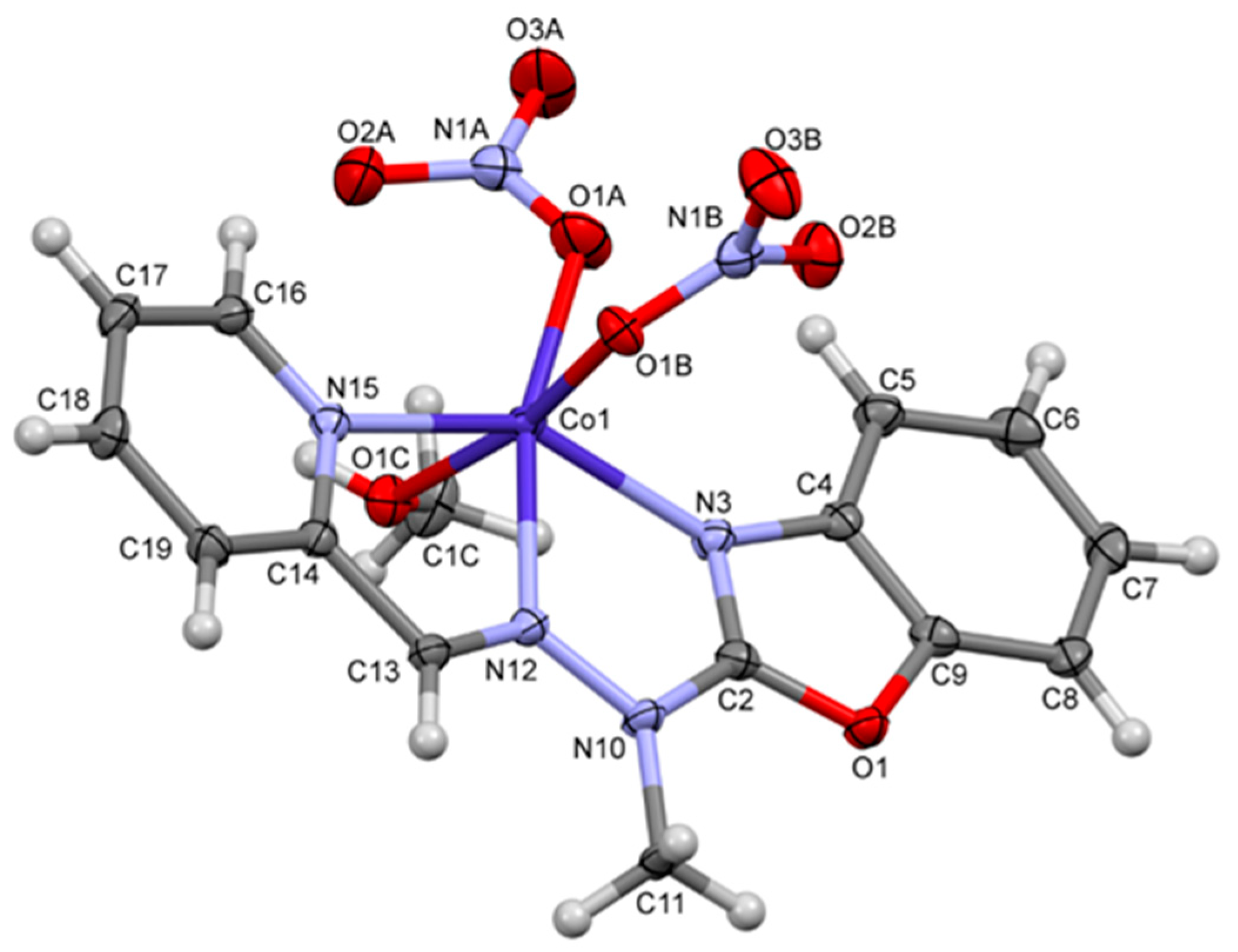
2.2. Description of the Structures
2.3. Cyclic Voltammetry
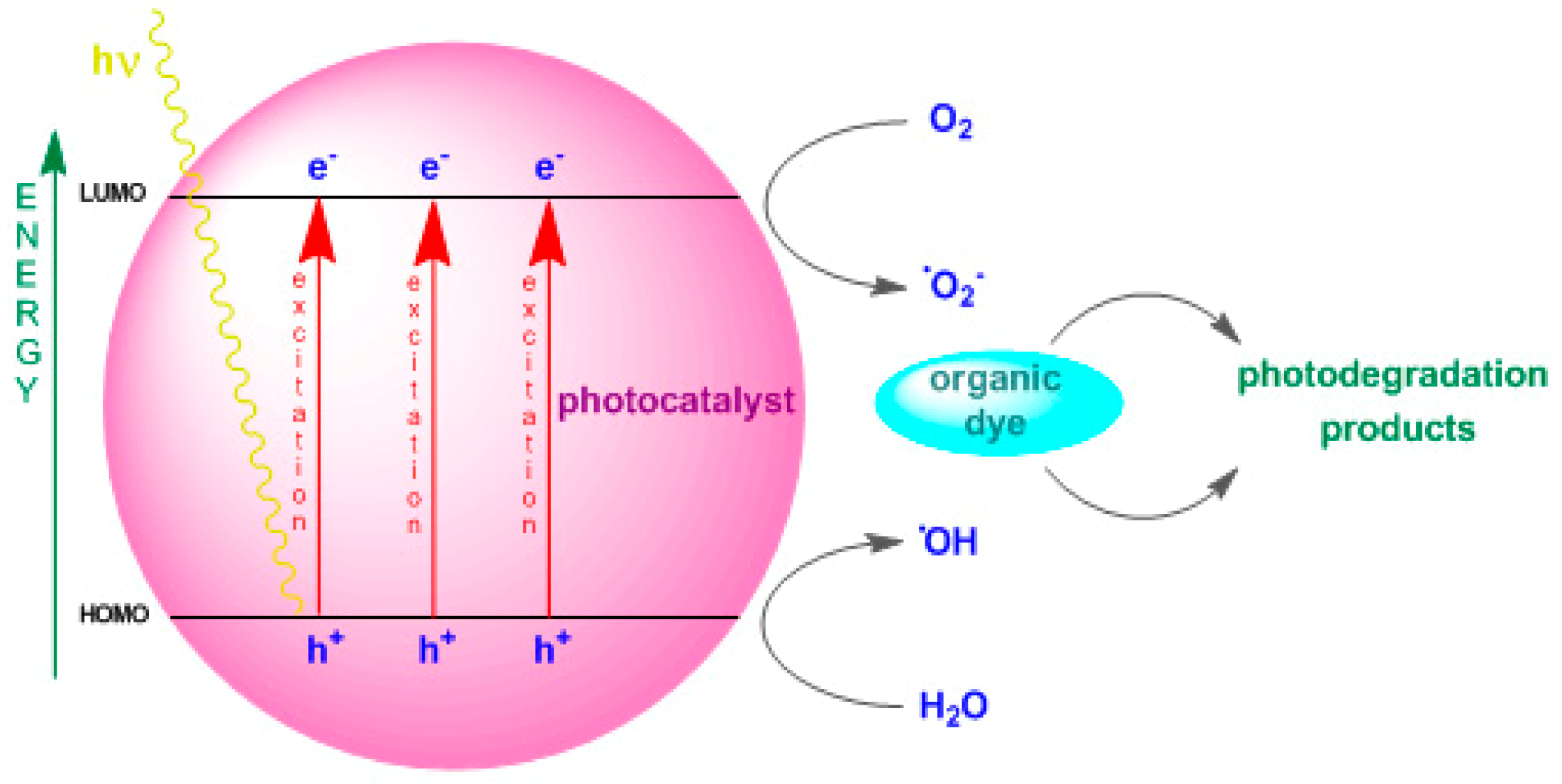
2.4. Photostability and Photodegradation Studies
2.5. Optical Properties of Compounds
3. Materials and Methods
3.1. Materials and Physical Measurements
3.2. Synthesis of Ligands
3.3. Synthesis of Complexes (1–3)
3.3.1. Complex 1 [CoIII(L1)2]
3.3.2. Complex 2 [CoIIL2](O2-NO)2
3.3.3. Complex 3 [CoIIL3(O-NO2)2(CH3OH)]
3.4. X-ray Crystallography
3.5. Cyclic Voltammetry
3.5.1. Reagents and Solutions
3.5.2. Electrodes
3.6. Photocatalytic Studies
3.6.1. Photocatalytic and Degradation Experiments
3.6.2. Photostability Experiments
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AR18 | Acid Red 18 |
| CV | cyclic voltammetry |
| DMF | dimethylformamide |
| DR | diffuse-reflectance |
| RB21 | Reactive Blue 21 |
| SCE | saturated calomel electrode |
| TBAP | tetrabutylammonium perchlorate |
References
- Han, X.; Han, Z.; Li, J.; Zhao, J.; Zhao, X. Coordinative integration of copper (II) and iron (II) phthalocyanine into amidoximated PAN fiber for enhanced photocatalytic activity under visible light irradiation. J. Colloid Interface Sci. 2019, 533, 333–343. [Google Scholar] [CrossRef]
- Ghoreishi, S.M.; Haghighi, R. Chemical catalytic reaction and biological oxidation for treatment of non-biodegradable textile effluent. Chem. Eng. J. 2003, 95, 163–169. [Google Scholar] [CrossRef]
- Zollinger, H. Color Chemistry. Synthesis, Properties and Applications of Organic Dyes and Pigments; VCH Publishers: New York, NY, USA, 1987; pp. 92–100. [Google Scholar]
- Silva, M.C.; Corrêa, A.D.; Amorim, M.T.S.P.; Parpot, P.; Torres, J.A.; Chagas, P.M.B. Decolorization of the phthalocyanine dye reactive blue 21 by turnip peroxidase and assessment of its oxidation products. J. Mol. Catal. B Enzym. 2012, 77, 9–14. [Google Scholar] [CrossRef]
- Zhang, L.; Zhao, Y.; Zhong, L.; Wang, Y.; Chai, S.; Yang, T.; Han, X. Cu2S-Cu-TiO2 mesoporous carbon composites for the degradation of high concentration of methyl orange under visible light. Appl. Surf. Sci. 2017, 422, 1093–1101. [Google Scholar] [CrossRef]
- Matthews, R.D.; Bottomley, L.A.; Pavlostathis, S.G. Palladium-catalyzed hydrogen reduction and decolorization of reactive phthalocyanine dyes. Desalination 2009, 248, 816–825. [Google Scholar] [CrossRef]
- Alves de Lima, R.O.; Bazo, A.P.; Salvadori, D.M.F.; Rech, C.M.; de Palma Oliveira, D.; de Aragão Umbuzeiro, G. Mutagenic and carcinogenic potential of a textile azo dye processing plant effluent that impacts a drinking water source. Mutat. Res. Genet. Toxicol. Environ. Mutagen. 2007, 626, 53–60. [Google Scholar] [CrossRef] [PubMed]
- Brüschweiler, B.J.; Merlot, C. Azo dyes in clothing textiles can be cleaved into a series of mutagenic aromatic amines which are not regulated yet. Regul. Toxicol. Pharmacol. 2017, 88, 214–226. [Google Scholar] [CrossRef] [PubMed]
- Katheresan, V.; Kansedo, J.; Lau, S.Y. Efficiency of various recent wastewater dye removal methods: A review. J. Environ. Chem. Eng. 2018, 6, 4676–4697. [Google Scholar] [CrossRef]
- Xia, S.-J.; Liu, F.-X.; Ni, Z.-M.; Shi, W.; Xue, J.-L.; Qian, P.-P. Ti-based layered double hydroxides: Efficient photocatalysts for azo dyes degradation under visible light. Appl. Catal. B 2014, 144, 570–579. [Google Scholar] [CrossRef]
- Chen, Z.; Xu, Y.-J. Ultrathin TiO2 layer coated-CdS spheres core–shell nanocomposite with enhanced visible-light photoactivity. ACS Appl. Mater. Interfaces 2013, 5, 13353–13363. [Google Scholar] [CrossRef] [PubMed]
- Deng, Y.; Chen, Y.; Chen, B.; Ma, J. Preparation, characterization and photocatalytic activity of CuBi2O4/NaTaO3 coupled photocatalysts. J. Alloys Compd. 2013, 559, 116–122. [Google Scholar] [CrossRef]
- Di Paola, A.; García-López, E.; Marcì, G.; Palmisano, L. A survey of photocatalytic materials for environmental remediation. J. Hazard. Mater. 2012, 211–212, 3–29. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, R.; Li, J.; Li, L.; Lin, S. First-principles study on transition metal-doped anatase TiO2. Nanoscale Res. Lett. 2014, 9, 46. [Google Scholar] [CrossRef] [PubMed]
- Dostani, M.; Kianfar, A.H.; Momeni, M.M. Visible light photocatalytic activity of novel Ni2+, Cu2+ and VO2 complexes derived from vanillin bidentate Schiff base ligand doped on TiO2 nanoparticles. J. Mater. Sci. Mater. Electron. 2017, 28, 633–640. [Google Scholar] [CrossRef]
- Eshwar Rao, S.; Gayathri, V. Poly(styrene–divinyl benzene)-immobilized Fe(III) complex of 1,3-bis(benzimidazolyl)benzene: Efficient catalyst for the photocatalytic degradation of xylenol orange. J. Appl. Polym. Sci. 2018, 135, 46480. [Google Scholar] [CrossRef]
- Qin, L.; Hu, Q.; Wu, Y.; Cai, J.-L.; Li, Y.-Y. Three novel Co(ii)/Ni(ii)-based coordination polymers as efficient heterogeneous catalysts for dye degradation. CrystEngComm 2018, 20, 4042–4048. [Google Scholar] [CrossRef]
- Qiao, Y.; Ren, S.-S.; Liu, L.-H.; Guan, W.-S.; Li, Z.-M.; Che, G.-B.; Liu, C.-B.; Wang, Y.-Y.; Wang, Q.-W.; Li, X.-Y.; et al. Adsorption and photocatalytic properties of transition metal Zinc(II) complex based on 5-(4-(tetrazol-5-yl)phenyl)isophthalic acid. J. Mol. Struct. 2018, 1161, 238–245. [Google Scholar] [CrossRef]
- Xu, Z.; Li, Q.; He, X.; Li, H.; Wang, Y.; Cao, J. Construction of mixed-valence Cu(I)/Cu(II) 3-D framework and its photocatalytic activities. Polyhedron 2018, 151, 478–482. [Google Scholar] [CrossRef]
- Malato, S.; Blanco, J.; Vidal, A.; Richter, C. Photocatalysis with solar energy at a pilot-plant scale: An overview. Appl. Catal. B 2002, 37, 1–15. [Google Scholar] [CrossRef]
- Casero, I.; Sicilia, D.; Rubio, S.; Pérez-Bendito, D. Chemical degradation of aromatic amines by Fenton’s reagent. Water Res. 1997, 31, 1985–1995. [Google Scholar] [CrossRef]
- Perkowski, J.; Jóźwiak, W.; Kos, L.; Stajszczyk, P. Application of fenton’s reagent in detergent separation in highly concentrated water solutions. Fibres Text. East. Eur. 2006, 14, 114–119. [Google Scholar]
- Barbusiński, K. The modified fenton process for decolorization of dye wastewater. Polish J. Environ. Stud. 2005, 14, 281–285. [Google Scholar]
- Li, X.; Xiong, Z.; Ruan, X.; Xia, D.; Zeng, Q.; Xu, A. Kinetics and mechanism of organic pollutants degradation with cobalt–bicarbonate–hydrogen peroxide system: Investigation of the role of substrates. Appl. Catal. A 2012, 411–412, 24–30. [Google Scholar] [CrossRef]
- Xu, A.; Li, X.; Ye, S.; Yin, G.; Zeng, Q. Catalyzed oxidative degradation of methylene blue by in situ generated cobalt (II)-bicarbonate complexes with hydrogen peroxide. Appl. Catal. B 2011, 102, 37–43. [Google Scholar] [CrossRef]
- Spier, E.; Neuenschwander, U.; Hermans, I. Insights into the Cobalt(II)-Catalyzed Decomposition of Peroxide. Angew. Chem. Int. Ed. 2013, 52, 1581–1585. [Google Scholar] [CrossRef] [PubMed]
- Song, Q.; Jia, M.; Ma, W.; Fang, Y.; Huang, Y. Heterogeneous degradation of toxic organic pollutants by hydrophobic copper Schiff-base complex under visible irradiation. Sci. China Chem. 2013, 56, 1775–1782. [Google Scholar] [CrossRef]
- Fik, M.A.; Kubicki, M.; Odachowska, A.E.; Karpińska, J.; Patroniak, V. Helicity in photocatalysis—advantage or obstacle? Studies on silver complexes—synthesis, structure, and photocatalytic Activity. Eur. J. Inorg. Chem. 2016, 2016, 5530–5538. [Google Scholar] [CrossRef]
- Fik, M.A.; Czepa, W.; Kubicki, M.; Patroniak, V. Formation of non-covalent porous framework with Fe ions and N2O Schiff base ligand: Structural and thermal studies. Supramol. Chem. 2017, 29, 643–650. [Google Scholar] [CrossRef]
- Fik, M.A.; Löffler, M.; Weselski, M.; Kubicki, M.; Korabik, M.J.; Patroniak, V. New Fe(II) complexes with Schiff base ligand: Synthesis, spectral characterization, magnetic studies and thermal stability. Polyhedron 2015, 102, 609–614. [Google Scholar] [CrossRef]
- Gorczyński, A.; Pakulski, D.; Szymańska, M.; Kubicki, M.; Bułat, K.; Łuczak, T.; Patroniak, V. Electrochemical deposition of the new manganese(II) Schiff-base complex on a gold template and its application for dopamine sensing in the presence of interfering biogenic compounds. Talanta 2016, 149, 347–355. [Google Scholar] [CrossRef]
- Newman, R.C.; Burstein, G.T. Reactions of scratched gold electrodes in alkaline solution. J. Electroanal. Chem. 1981, 129, 343–348. [Google Scholar] [CrossRef]
- Marcinkowski, D.; Fik, M.A.; Łuczak, T.; Kubicki, M.; Patroniak, V. New Mn(II) complexes with benzoxazole-based ligands: Synthesis, structure and their electrochemical behavior. Polyhedron 2018, 141, 125–132. [Google Scholar] [CrossRef]
- Bard, A.J.; Faulkner, L.R. Electrochemical Methods: Fundamentals and Applications; Wiley: New York, NY, USA, 2001. [Google Scholar]
- Garrell, R.L.; Chadwick, J.E.; Severance, D.L.; McDonald, N.A.; Myles, D.C. Adsorption of sulfur containing molecules on gold: The effect of oxidation on monolayer formation and stability characterized by experiments and theory. J. Am. Chem. Soc. 1995, 117, 11563–11571. [Google Scholar] [CrossRef]
- Xu, S.; Podoprygorina, G.; Böhmer, V.; Ding, Z.; Rooney, P.; Rangan, C.; Mittler, S. Tetraurea calix[4]arenes with sulfur functions: Synthesis, dimerization to capsules, and self-assembly on gold. Org. Biomol. Chem. 2007, 5, 558–568. [Google Scholar] [CrossRef]
- Savarino, P.; Viscardi, G.; Quagliotto, P.; Perracino, P.; Barni, E. Voltammetric behaviour of heterocyclic systems. Pyridyl-substituted benzimidazoles, benzoxazoles and benzothiazoles. J. Heterocyclic Chem. 1997, 34, 1479–1485. [Google Scholar] [CrossRef]
- Komissarov, A.N.; Makarov, D.A.; Yuzhakova, O.A.; Savvina, L.P.; Kuznetsova, N.A.; Kaliya, O.L.; Lukyanets, E.А.; Negrimovsky, V.M. Synthesis and some properties of phosphonomethyl substituted phthalocyanines. Macroheterocycles 2012, 5, 169–174. [Google Scholar] [CrossRef][Green Version]
- Khataee, A.R.; Kasiri, M.B. Photocatalytic degradation of organic dyes in the presence of nanostructured titanium dioxide: Influence of the chemical structure of dyes. J. Mol. Catal. A Chem. 2010, 328, 8–26. [Google Scholar] [CrossRef]
- Iasco, O.; Novitchi, G.; Jeanneau, E.; Tommasino, J.B.; Roques, N.; Luneau, D. Versatile chemical transformations of benzoxazole based ligands on complexation with 3d-metal ions. Inorg. Chem. 2012, 51, 2588–2596. [Google Scholar] [CrossRef]
- Ananthakrishnan, S.J.; Kumar, B.S.; Somanathan, N.; Mandal, A.B. Supramolecular assembly in side-chain conjugated thiophene copolymers. RSC Adv. 2013, 3, 8331–8340. [Google Scholar] [CrossRef]
- Qu, Y.; Duan, X. Progress, challenge and perspective of heterogeneous photocatalysts. Chem. Soc. Rev. 2013, 42, 2568–2580. [Google Scholar] [CrossRef]
- Almeida Guerra, W.N.; Teixeira Santos, J.M.; Raddi de Araujo, L.R. Decolorization and mineralization of reactive dyes by a photocatalytic process using ZnO and UV radiation. Water Sci. Technol. 2012, 66, 158–164. [Google Scholar] [CrossRef] [PubMed]
- Miklos, D.B.; Remy, C.; Jekel, M.; Linden, K.G.; Drewes, J.E.; Hübner, U. Evaluation of advanced oxidation processes for water and wastewater treatment—A critical review. Water Res. 2018, 139, 118–131. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, P.; Goyal, S.; Patnala, P.K. Degradation of reactive, acid and basic textile dyes in the presence of ultrasound and rare earths [Lanthanum and Praseodymium]. Ultrason. Sonochem. 2014, 21, 1994–2009. [Google Scholar] [CrossRef] [PubMed]
- Olya, M.E.; Pirkarami, A. On the positive role of doping Cu and N2 on TiO2 in improving dye degradation efficiency: Providing reaction mechanisms. Korean J. Chem. Eng. 2015, 32, 1586–1597. [Google Scholar] [CrossRef]
- Wałęsa-Chorab, M.; Patroniak, V.; Kubicki, M.; Kądziołka, G.; Przepiórski, J.; Michalkiewicz, B. Synthesis, structure, and photocatalytic properties of new dinuclear helical complex of silver(I) ions. J. Catal. 2012, 291, 1–8. [Google Scholar] [CrossRef]
- Marcinkowski, D.; Wałęsa-Chorab, M.; Patroniak, V.; Kubicki, M.; Kądziołka, G.; Michalkiewicz, B. A new polymeric complex of silver(i) with a hybrid pyrazine–bipyridine ligand—synthesis, crystal structure and its photocatalytic activity. New J. Chem. 2014, 38, 604–610. [Google Scholar] [CrossRef]
- Rigaku Oxford Diffraction. CrysAlis PRO (Version 1.171.38.41); Rigaku Oxford Diffraction: Yarnton, UK, 2015. [Google Scholar]
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Cryst. A 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Cryst. C 2015, 71, 3–8. [Google Scholar] [CrossRef]
- Łuczak, T. Adsorption of methylamine at a polycrystalline gold/solution interface. Coll. Surf. A 2006, 280, 125–129. [Google Scholar] [CrossRef]
- Available online: www.reaxys.com (accessed on 2 October 2019).





| Entry | Dye | Source of Irradiation | Type of Photolysis | Kinetic Constant k [min−1] |
|---|---|---|---|---|
| 1 | Reactive Blue 21 | Sunlight | self-decomposition | 7 × 10−4 |
| 2 | 254 nm | 8 × 10−4 | ||
| 3 | Sunlight | TiO2-catalyzed | 4.9 × 10−3 | |
| 4 | 254 nm | 4.1 × 10−3 | ||
| 5 | Sunlight | 1-catalyzed | 7 × 10−4 | |
| 6 | 254 nm | 2.5 × 10−3 | ||
| 7 | Sunlight | 2-catalyzed | 2.6 × 10−3 | |
| 8 | 254 nm | 1.2 × 10−3 | ||
| 9 | Sunlight | 3-catalyzed | 5 × 10−4 | |
| 10 | 254 nm | na * | ||
| 11 | Acid Red 18 | Sunlight | self-decomposition | 1.5 × 10−3 |
| 12 | 254 nm | 0.4 × 10−3 | ||
| 13 | Sunlight | TiO2-catalyzed | 1.6 × 10−3 | |
| 14 | 254 nm | 0.3 × 10−3 | ||
| 15 | Sunlight | 1-catalyzed | 6 × 10−4 | |
| 16 | 254 nm | na * | ||
| 17 | Sunlight | 2-catalyzed | 2.3 × 10−3 | |
| 18 | 254 nm | 2.5 × 10−3 | ||
| 19 | Sunlight | 3-catalyzed | 0–45 min induction period 45–120 min K = 8 × 10−4 | |
| 20 | 254 nm | na * |
| HL1 | L2 | L3 | 1 | 2 | 3 | |
|---|---|---|---|---|---|---|
| Energy gap [eV] | 1.550 | 2.205 | 1.625 | 1.774 | 1.774 | 1.621 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szymańska, M.; Czepa, W.; Hołubowicz, C.; Świsłocka, R.; Łuczak, T.; Kubicki, M.; Karpińska, J.; Fik-Jaskółka, M.A.; Patroniak, V. Co(II/III) Complexes with Benzoxazole and Benzothiazole Ligands as Efficient Heterogenous Photocatalysts for Organic Dyes Degradation. Catalysts 2019, 9, 913. https://doi.org/10.3390/catal9110913
Szymańska M, Czepa W, Hołubowicz C, Świsłocka R, Łuczak T, Kubicki M, Karpińska J, Fik-Jaskółka MA, Patroniak V. Co(II/III) Complexes with Benzoxazole and Benzothiazole Ligands as Efficient Heterogenous Photocatalysts for Organic Dyes Degradation. Catalysts. 2019; 9(11):913. https://doi.org/10.3390/catal9110913
Chicago/Turabian StyleSzymańska, Martyna, Włodzimierz Czepa, Cezary Hołubowicz, Renata Świsłocka, Teresa Łuczak, Maciej Kubicki, Joanna Karpińska, Marta A. Fik-Jaskółka, and Violetta Patroniak. 2019. "Co(II/III) Complexes with Benzoxazole and Benzothiazole Ligands as Efficient Heterogenous Photocatalysts for Organic Dyes Degradation" Catalysts 9, no. 11: 913. https://doi.org/10.3390/catal9110913
APA StyleSzymańska, M., Czepa, W., Hołubowicz, C., Świsłocka, R., Łuczak, T., Kubicki, M., Karpińska, J., Fik-Jaskółka, M. A., & Patroniak, V. (2019). Co(II/III) Complexes with Benzoxazole and Benzothiazole Ligands as Efficient Heterogenous Photocatalysts for Organic Dyes Degradation. Catalysts, 9(11), 913. https://doi.org/10.3390/catal9110913







