Surface Modification of Nanocrystalline TiO2 Materials with Sulfonated Porphyrins for Visible Light Antimicrobial Therapy
Abstract
1. Introduction
2. Results
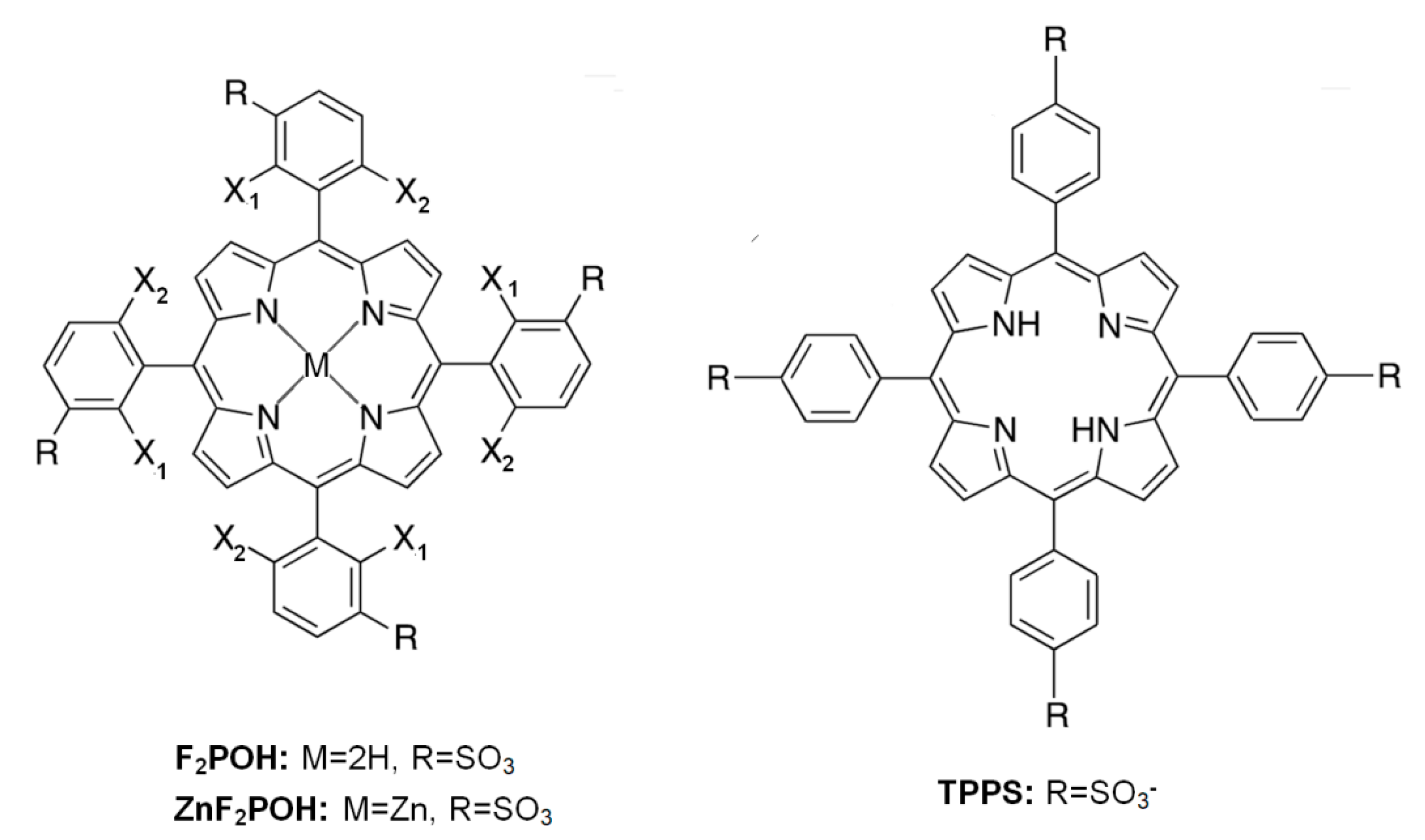
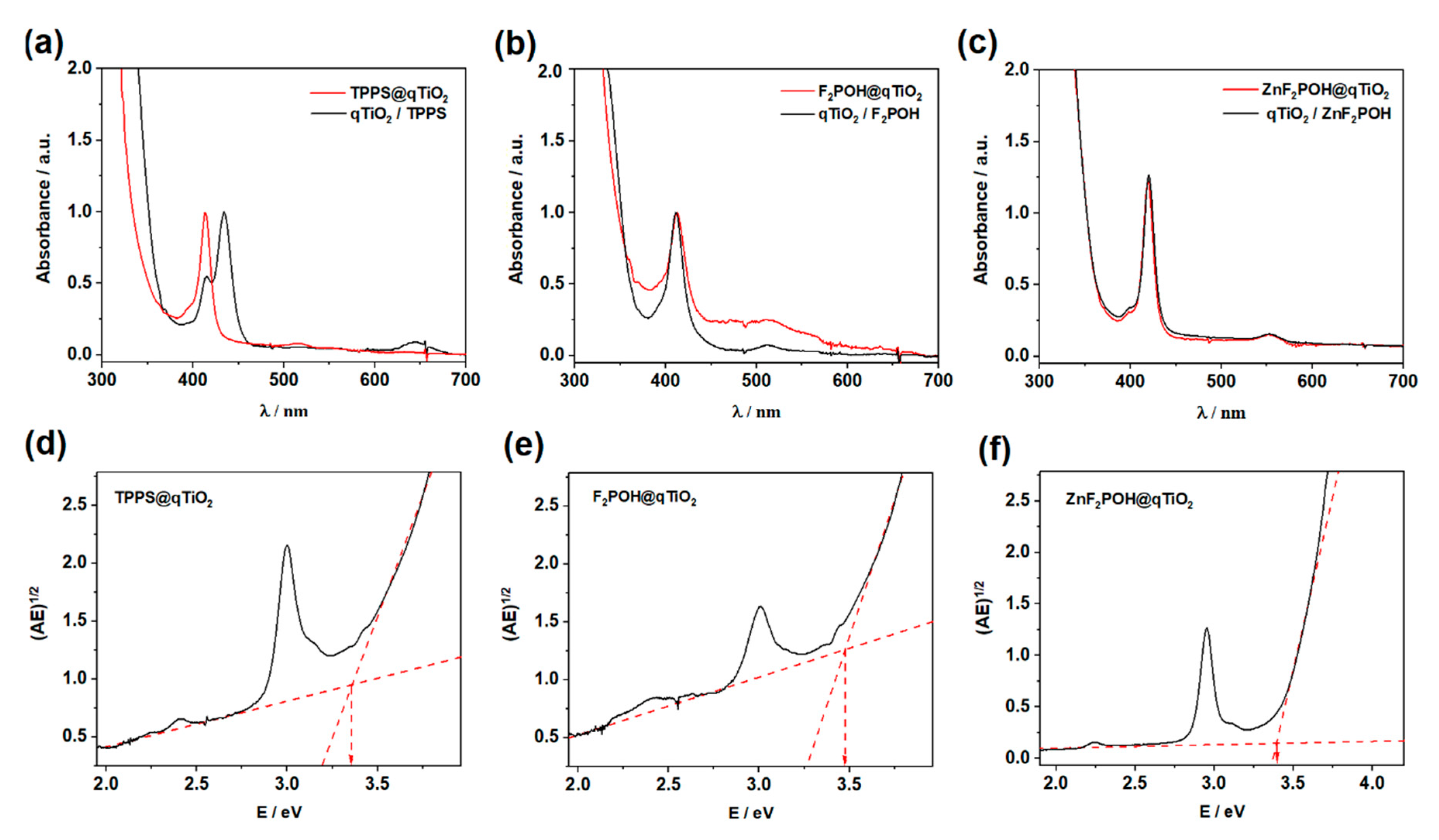
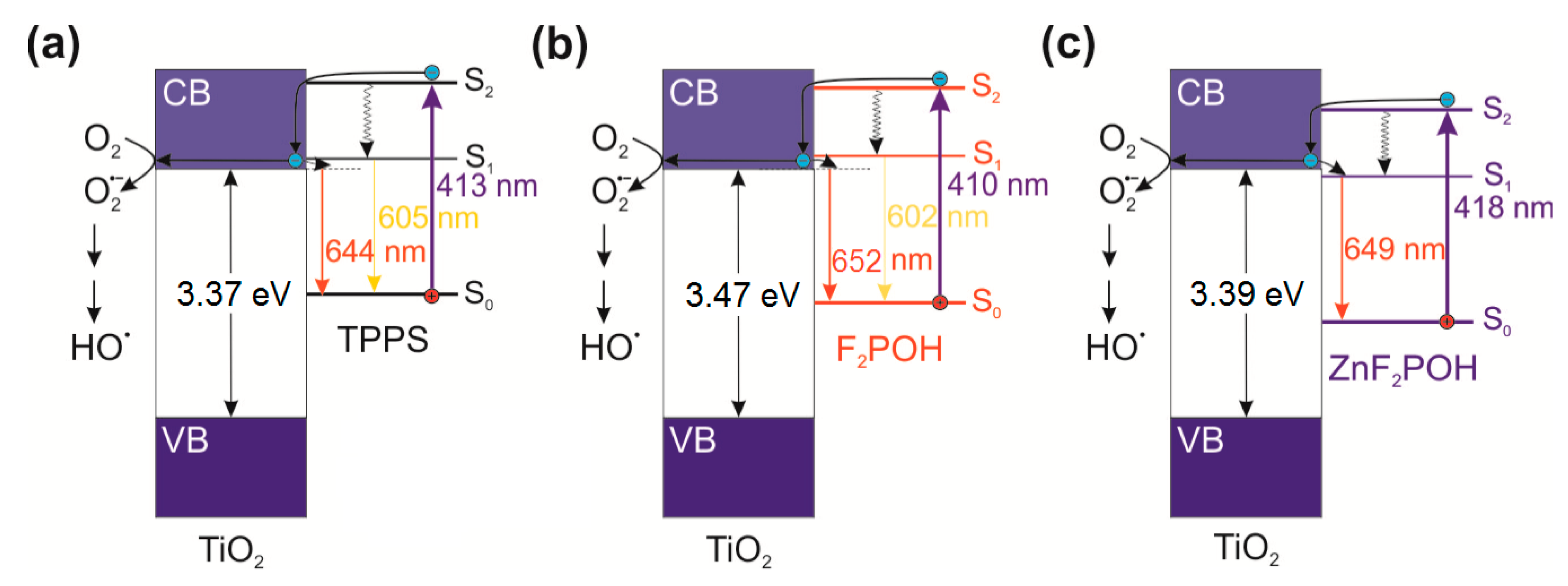
2.1. Synthesis, Optical Properties, and Characterization of the Materials
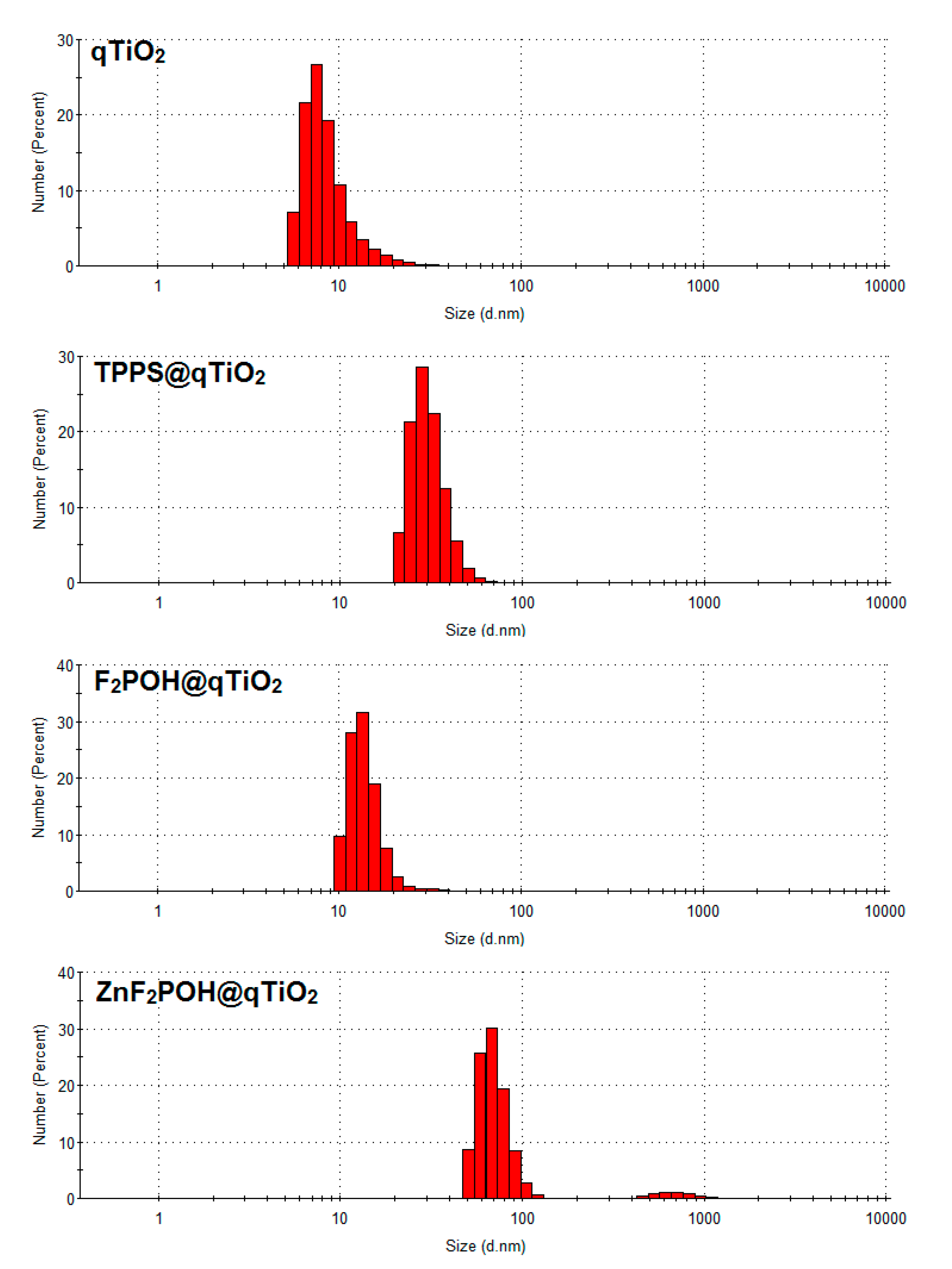
Scanning Electron Microscopy (SEM) Analysis of Prepared Nanomaterials
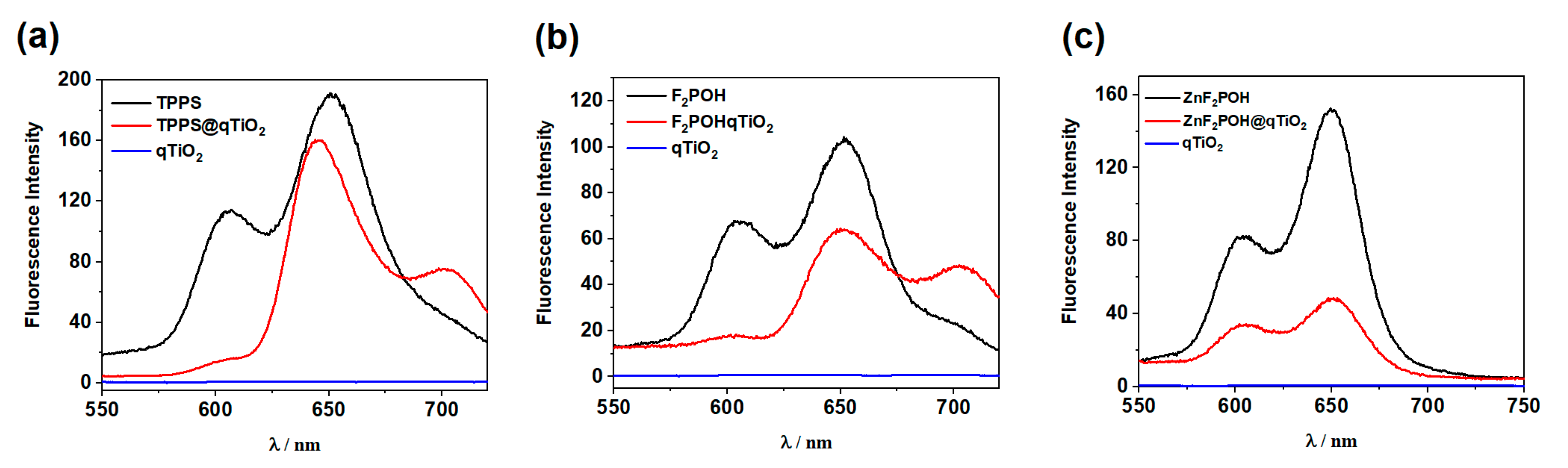
2.2. Fluorescence Quenching by Colloidal qTiO2 Nanoparticles
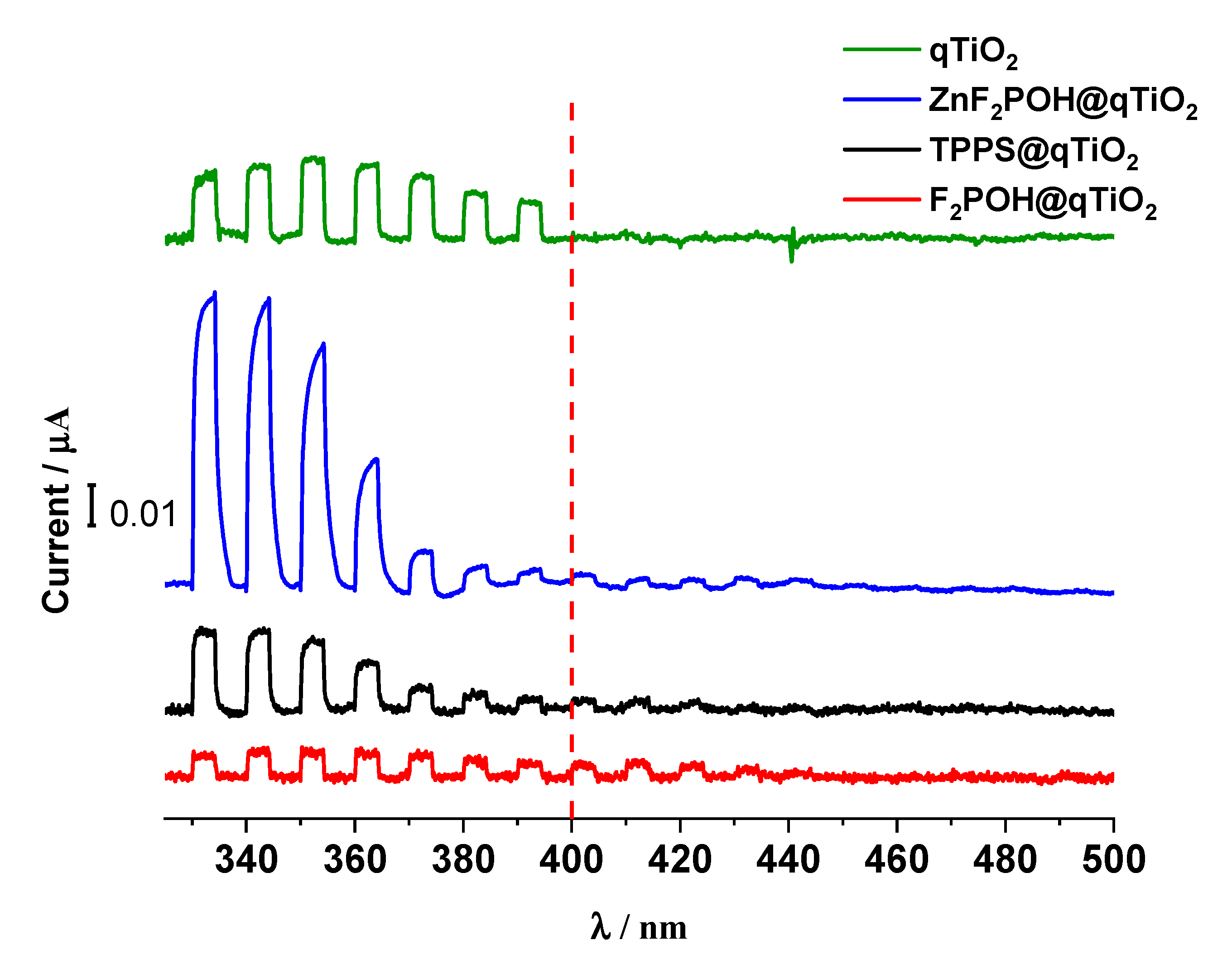
2.3. Photoelectrochemical Properties of qTiO2 Material
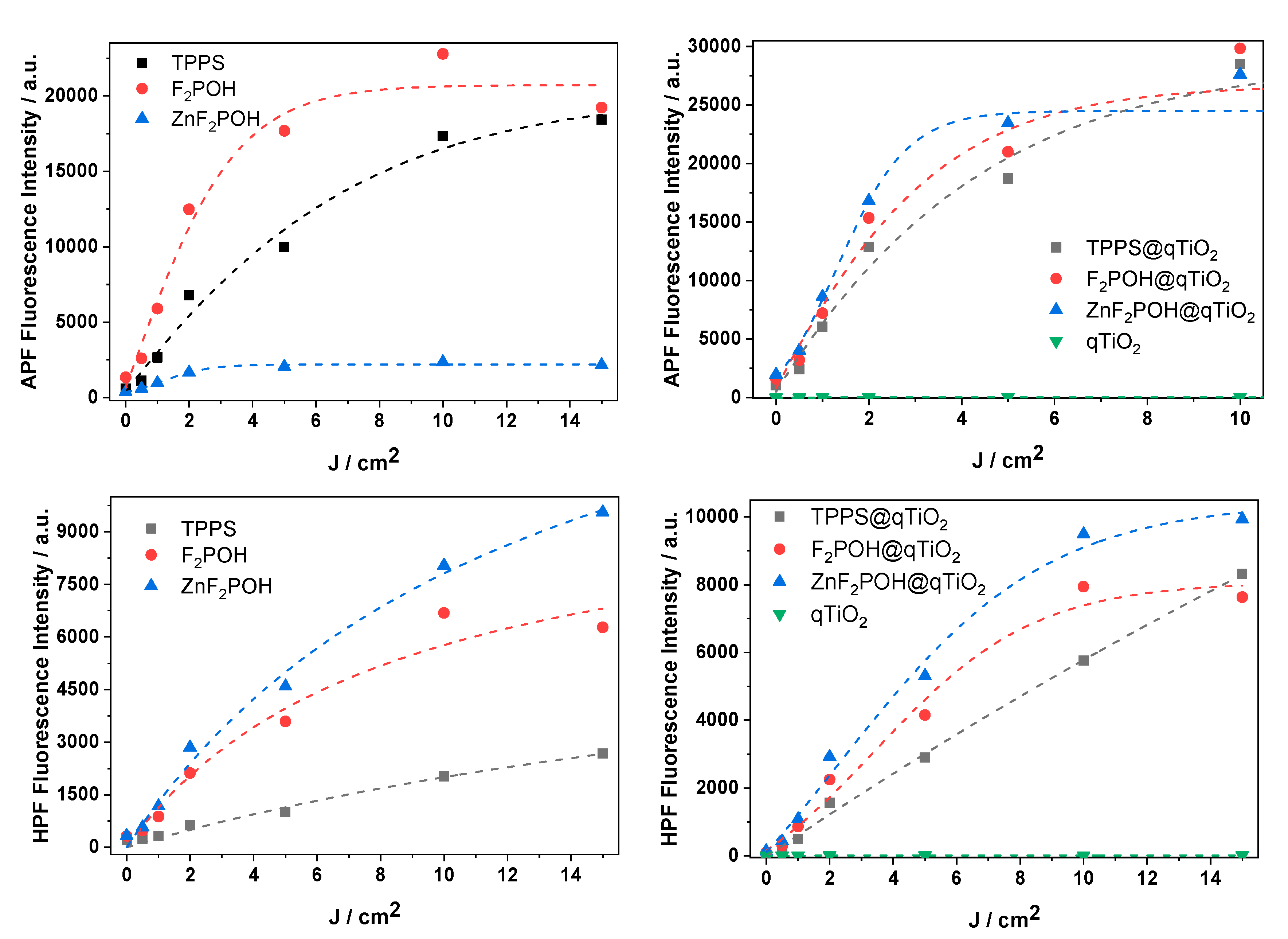
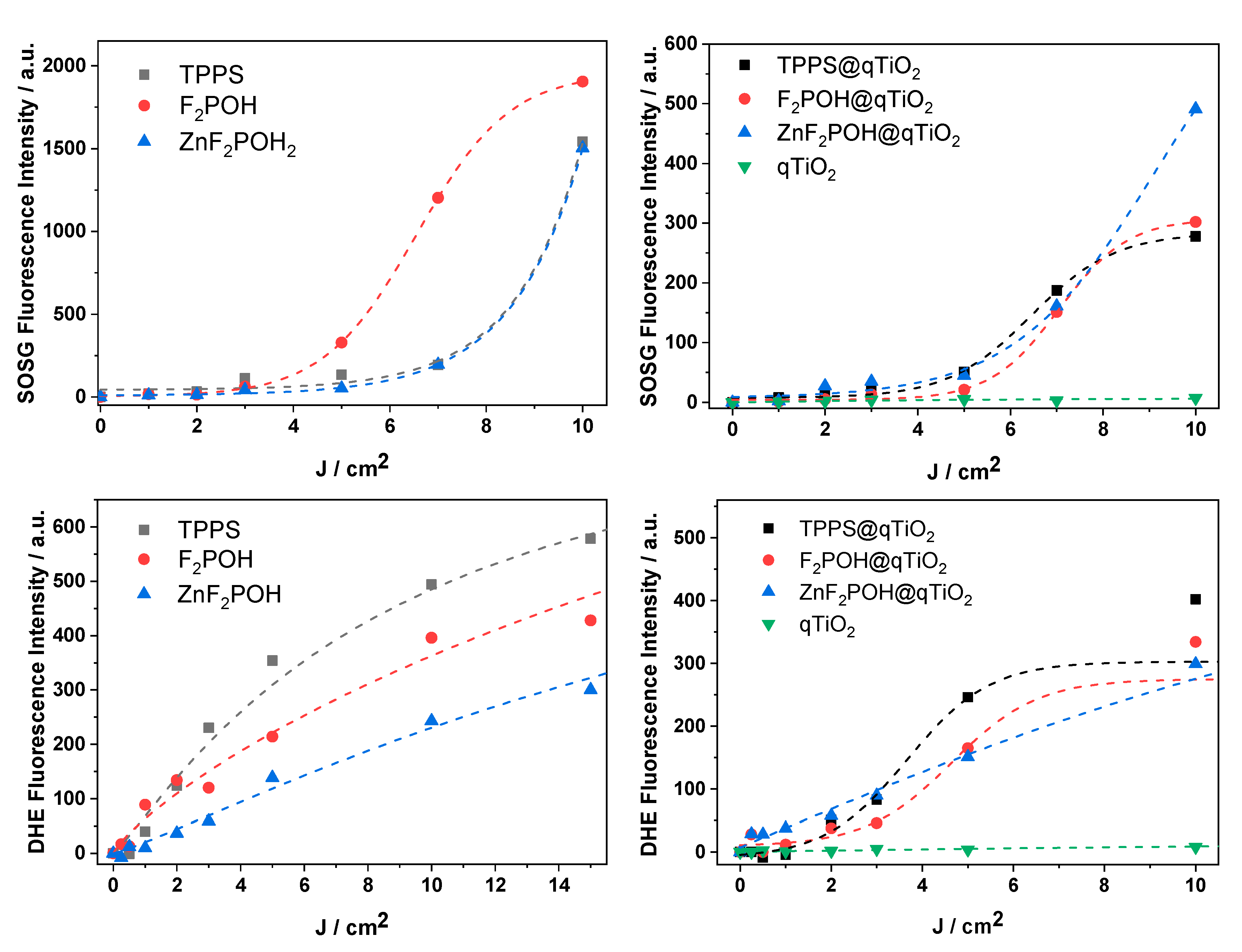
2.4. ROS Detection by Molecular Fluorescent Probes
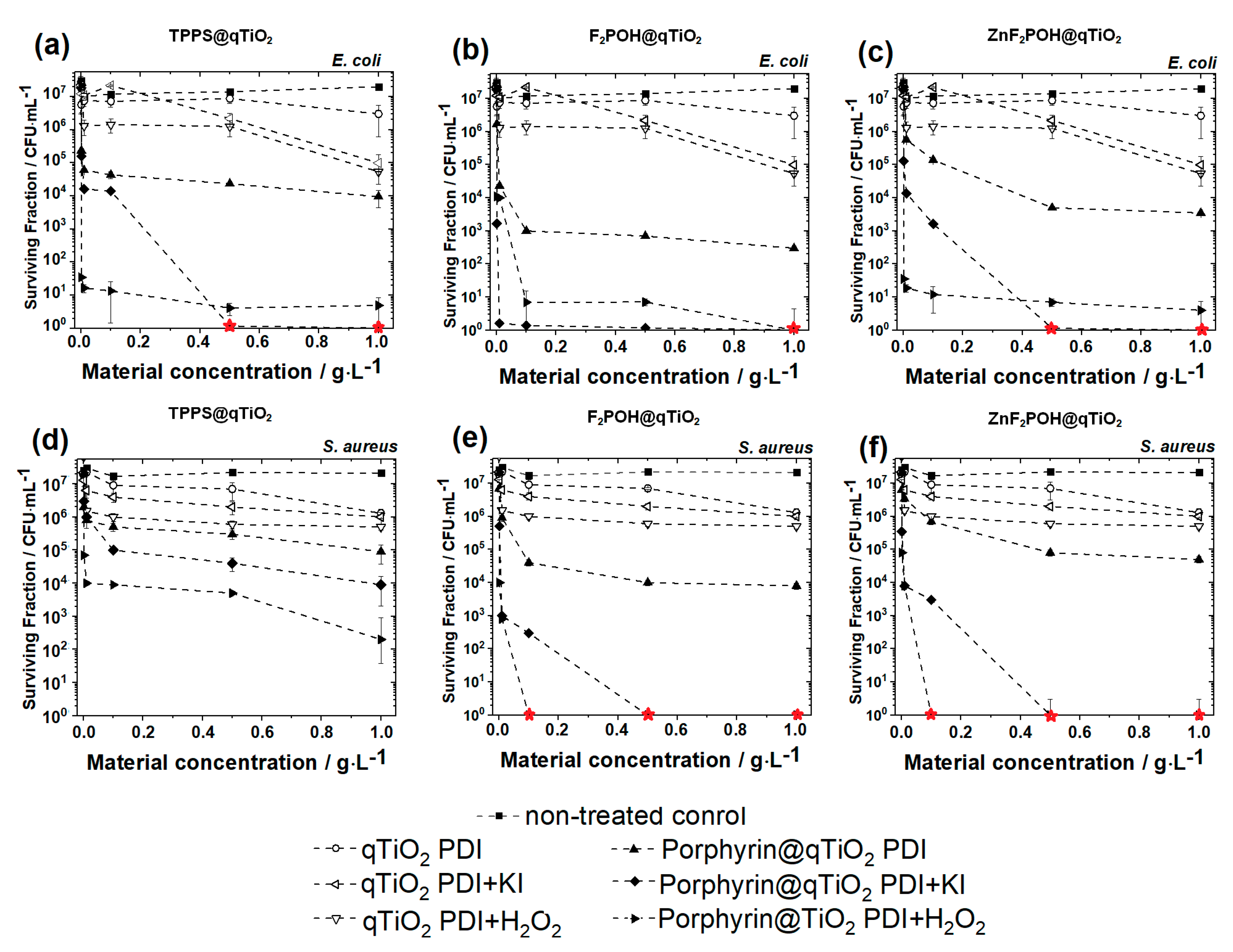
2.5. Antimicrobial Studies
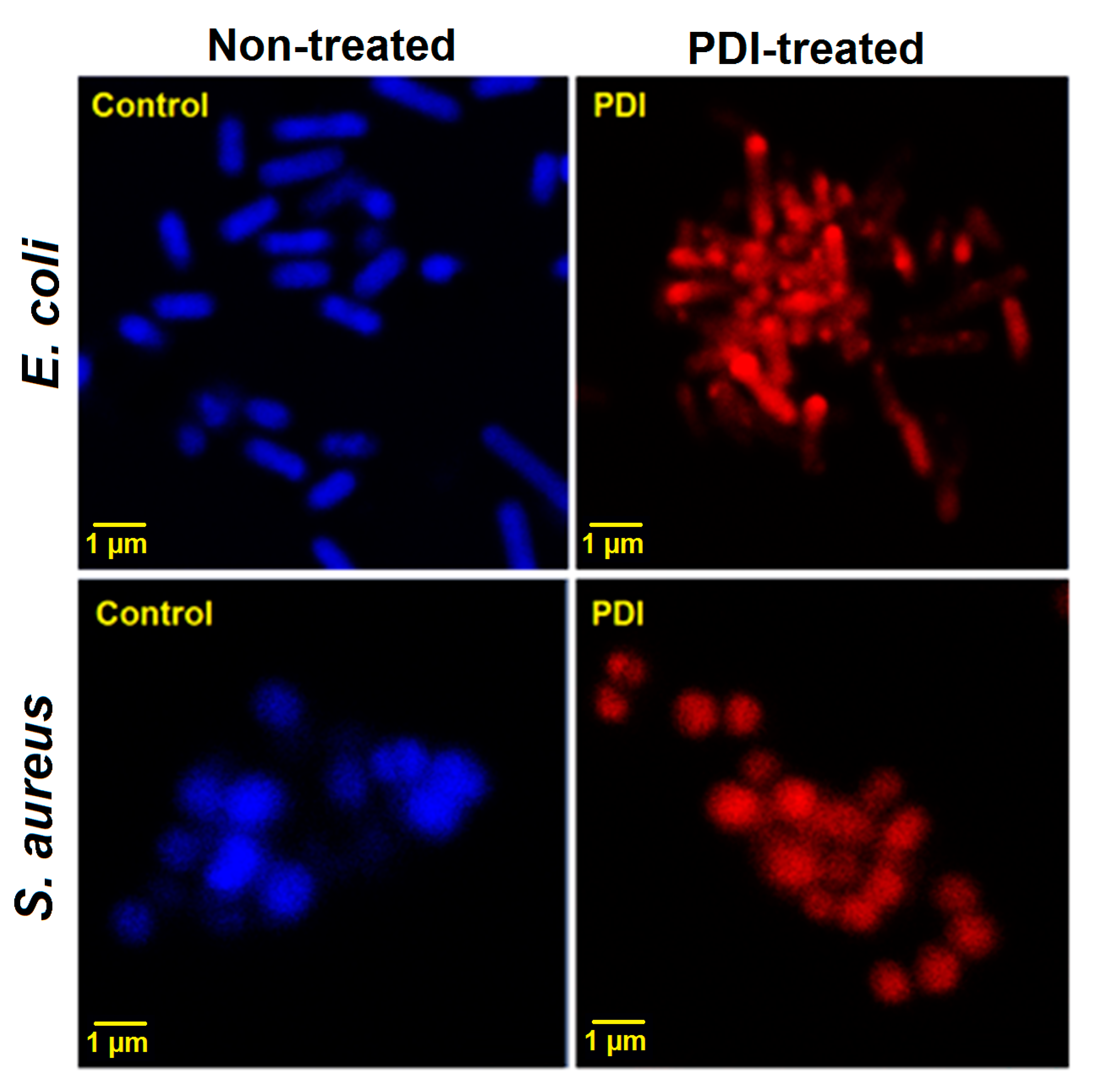
2.6. Bacteria Imaging by Scanning Electron Microscopy (SEM) and Confocal Laser Scanning Microscopy (CLSM)
3. Discussion and Conclusions
4. Materials and Methods
4.1. Chemicals
4.2. Porphyrins@qTiO2 Materials Preparation
4.3. UV/VIS Electronic Absorption and Emission Spectra Measurements
4.4. Dynamic Light Scattering (DLS) and Zeta Potential Measurements
4.5. Photoelectrochemical Measurements
4.6. Characterization of Porphyrin@qTiO2 Materials Using Scanning Electron Microscopy (SEM)
4.7. Detection of Reactive Oxygen Species Using Fluorescent Probes
4.8. Photodynamic Inactivation (PDI) of Bacteria
4.9. CLSM Fluorescence Imaging of Bacteria
4.10. Statistics
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zouzelka, R.; Remzova, M.; Plsek, J.; Brabec, L.; Rathousky, J. Immobilized rGO/TiO2 Photocatalyst for Decontamination of Water. Catalysts 2019, 9, 708. [Google Scholar] [CrossRef]
- Oszajca, M.; Brindell, M.; Orzeł, Ł.; Dąbrowski, J.M.; Śpiewak, K.; Łabuz, P.; Pacia, M.; Stochel-Gaudyn, A.; Macyk, W.; van Eldik, R. Mechanistic studies on versatile metal-assisted hydrogen peroxide activation processes for biomedical and environmental incentives. Coord. Chem. Rev. 2016, 327, 143–165. [Google Scholar] [CrossRef]
- Kočí, K.; Reli, M.; Troppová, I.; Šihor, M.; Kupková, J.; Kustrowski, P.; Praus, P. Photocatalytic decomposition of N2O over TiO2/g-C3N4 photocatalysts heterojunction. Appl. Surf. Sci. 2017, 396, 1685–1695. [Google Scholar] [CrossRef]
- Falkowski, M.; Rebis, T.; Kryjewski, M.; Popenda, L.; Lijewski, S.; Jurga, S.; Mielcarek, J.; Milczarek, G.; Goslinski, T. An enhanced electrochemical nanohybrid sensing platform consisting of reduced graphene oxide and sulfanyl metalloporphyrazines for sensitive determination of hydrogen peroxide and L-cysteine. Dyes Pigments 2017, 138, 190–203. [Google Scholar] [CrossRef]
- Sadowski, R.; Strus, M.; Buchalska, M.; Heczko, P.B.; Macyk, W. Visible light induced photocatalytic inactivation of bacteria by modified titanium dioxide films on organic polymers. Photochem. Photobiol. Sci. 2015, 14, 514–519. [Google Scholar] [CrossRef] [PubMed]
- Lee, C.M.; Palaniandy, P.; Dahlan, I. Pharmaceutical residues in aquatic environment and water remediation by TiO2 heterogeneous photocatalysis: A review. Environ. Earth Sci. 2017, 76, 611. [Google Scholar] [CrossRef]
- Fagan, R.; McCormack, D.E.; Dionysiou, D.D.; Pillai, S.C. A review of solar and visible light active TiO2 photocatalysis for treating bacteria, cyanotoxins and contaminants of emerging concern. Mater. Sci. Semicond. Process. 2016, 42, 2–14. [Google Scholar] [CrossRef]
- Byrne, J.; Dunlop, P.; Hamilton, J.; Fernández-Ibáñez, P.; Polo-López, I.; Sharma, P.; Vennard, A. A review of heterogeneous photocatalysis for water and surface disinfection. Molecules 2015, 20, 5574–5615. [Google Scholar] [CrossRef]
- Sułek, A.; Pucelik, B.; Kuncewicz, J.; Dubin, G.; Dąbrowski, J.M. Sensitization of TiO2 by halogenated porphyrin derivatives for visible light biomedical and environmental photocatalysis. Catal. Today 2019, 335, 538–549. [Google Scholar] [CrossRef]
- Regiel-Futyra, A.; Dąbrowski, J.M.; Mazuryk, O.; Śpiewak, K.; Kyzioł, A.; Pucelik, B.; Brindell, M.; Stochel, G. Bioinorganic antimicrobial strategies in the resistance era. Coord. Chem. Rev. 2017, 351, 76–117. [Google Scholar] [CrossRef]
- Sobotta, L.; Skupin-Mrugalska, P.; Mielcarek, J.; Goslinski, T.; Balzarini, J. Photosensitizers mediated photodynamic inactivation against virus particles. Mini Rev. Med. Chem. 2015, 15, 503–521. [Google Scholar] [CrossRef] [PubMed]
- Jori, G.; Brown, S.B. Photosensitized inactivation of microorganisms. Photochem. Photobiol. Sci. 2004, 3, 403–405. [Google Scholar] [CrossRef] [PubMed]
- Donnelly, R.F.; McCarron, P.A.; Tunney, M.M. Antifungal photodynamic therapy. Microbiol. Res. 2008, 163, 1–12. [Google Scholar] [CrossRef]
- Costerton, J.W.; Stewart, P.S.; Greenberg, E.P. Bacterial biofilms: A common cause of persistent infections. Science 1999, 284, 1318–1322. [Google Scholar] [CrossRef] [PubMed]
- Maisch, T. Anti-microbial photodynamic therapy: Useful in the future? Lasers Med. Sci. 2007, 22, 83–91. [Google Scholar] [CrossRef]
- Maisch, T.; Szeimies, R.-M.; Jori, G.; Abels, C. Antibacterial photodynamic therapy in dermatology. Photochem. Photobiol. Sci. 2004, 3, 907–917. [Google Scholar] [CrossRef] [PubMed]
- Kawczyk-Krupka, A.; Pucelik, B.; Międzybrodzka, A.; Sieroń, A.R.; Dąbrowski, J.M. Photodynamic therapy as an alternative to antibiotic therapy for the treatment of infected leg ulcers. Photodiagnosis Photodyn. Ther. 2018, 23, 132–143. [Google Scholar] [CrossRef] [PubMed]
- Dlugaszewska, J.; Szczolko, W.; Koczorowski, T.; Skupin-Mrugalska, P.; Teubert, A.; Konopka, K.; Kucinska, M.; Murias, M.; Düzgüneş, N.; Mielcarek, J. Antimicrobial and anticancer photodynamic activity of a phthalocyanine photosensitizer with N-methyl morpholiniumethoxy substituents in non-peripheral positions. J. Inorg. Biochem. 2017, 172, 67–79. [Google Scholar] [CrossRef]
- Huang, L.; Xuan, Y.; Koide, Y.; Zhiyentayev, T.; Tanaka, M.; Hamblin, M.R. Type I and Type II mechanisms of antimicrobial photodynamic therapy: An in vitro study on gram-negative and gram-positive bacteria. Lasers Surg. Med. 2012, 44, 490–499. [Google Scholar] [CrossRef]
- Dąbrowski, J.M. Reactive oxygen species in photodynamic therapy: Mechanisms of their generation and potentiation. In Advances in Inorganic Chemistry; Elsevier: Amsterdam, The Netherlands, 2017; Volume 70, pp. 343–394. [Google Scholar]
- Martin, J.P.; Logsdon, N. The role of oxygen radicals in dye-mediated photodynamic effects in Escherichia coli B. J. Biol. Chem. 1987, 262, 7213–7219. [Google Scholar]
- Alves, E.; Faustino, M.A.; Neves, M.G.; Cunha, Â.; Nadais, H.; Almeida, A. Potential applications of porphyrins in photodynamic inactivation beyond the medical scope. J. Photochem. Photobiol. C Photochem. Rev. 2015, 22, 34–57. [Google Scholar] [CrossRef]
- Lopes, L.Q.S.; Ramos, A.P.; Copetti, P.M.; Acunha, T.V.; Iglesias, B.A.; Santos, R.C.V.; Machado, A.K.; Sagrillo, M.R. Antimicrobial activity and safety applications of meso-tetra (4-pyridyl) platinum (II) porphyrin. Microb. Pathog. 2019, 128, 47–54. [Google Scholar] [CrossRef] [PubMed]
- Castro, K.A.; Moura, N.M.; Figueira, F.; Ferreira, R.I.; Simões, M.M.; Cavaleiro, J.A.; Faustino, M.A.F.; Silvestre, A.J.; Freire, C.S.; Tomé, J.P. New Materials Based on Cationic Porphyrins Conjugated to Chitosan or Titanium Dioxide: Synthesis, Characterization and Antimicrobial Efficacy. Int. J. Mol. Sci. 2019, 20, 2522. [Google Scholar] [CrossRef] [PubMed]
- Pucelik, B.; Paczyński, R.; Dubin, G.; Pereira, M.M.; Arnaut, L.G.; Dąbrowski, J.M. Properties of halogenated and sulfonated porphyrins relevant for the selection of photosensitizers in anticancer and antimicrobial therapies. PLoS ONE 2017, 12, e0185984. [Google Scholar] [CrossRef] [PubMed]
- Jiang, D.; Xu, Y.; Wu, D.; Sun, Y. Visible-light responsive dye-modified TiO2 photocatalyst. J. Solid State Chem. 2008, 181, 593–602. [Google Scholar] [CrossRef]
- Li, H.; Li, M.; Zhang, L.; Zhang, X.; Ma, Y.; Yu, B.; Wei, Q.; Yin, S. Dipyridylbenzene as a charming sensitizer to significantly enhance the photocatalytic activity of titanium dioxide. Appl. Catal. B Environ. 2018, 232, 472–480. [Google Scholar] [CrossRef]
- Lord, M.S.; Foss, M.; Besenbacher, F. Influence of nanoscale surface topography on protein adsorption and cellular response. Nano Today 2010, 5, 66–78. [Google Scholar] [CrossRef]
- Huang, Y.-Y.; Sharma, S.K.; Dai, T.; Chung, H.; Yaroslavsky, A.; Garcia-Diaz, M.; Chang, J.; Chiang, L.Y.; Hamblin, M.R. Can nanotechnology potentiate photodynamic therapy? Nanotechnol. Rev. 2012, 1, 111–146. [Google Scholar] [CrossRef]
- Kozinska, A.; Zadlo, A.; Labuz, P.; Broniec, A.; Pabisz, P.; Sarna, T. The Ability of Functionalized Fullerenes and Surface-Modified TiO2 Nanoparticles to Photosensitize Peroxidation of Lipids in Selected Model Systems. Photochem. Photobiol. 2019, 95, 227–236. [Google Scholar]
- Mesquita, M.Q.; Dias, C.J.; Neves, M.P.M.S.; Almeida, A.; Faustino, M.F. Revisiting current photoactive materials for antimicrobial photodynamic therapy. Molecules 2018, 23, 2424. [Google Scholar] [CrossRef]
- Hajipour, M.J.; Fromm, K.M.; Ashkarran, A.A.; de Aberasturi, D.J.; de Larramendi, I.R.; Rojo, T.; Serpooshan, V.; Parak, W.J.; Mahmoudi, M. Antibacterial properties of nanoparticles. Trends Biotechnol. 2012, 30, 499–511. [Google Scholar] [CrossRef] [PubMed]
- Hamblin, M.R.; Hasan, T. Photodynamic therapy: A new antimicrobial approach to infectious disease? Photochem. Photobiol. Sci. 2004, 3, 436–450. [Google Scholar] [CrossRef]
- Daniel, M.-C.; Astruc, D. Gold nanoparticles: Assembly, supramolecular chemistry, quantum-size-related properties, and applications toward biology, catalysis, and nanotechnology. Chem. Rev. 2004, 104, 293–346. [Google Scholar] [CrossRef] [PubMed]
- Demidova, T.N.; Hamblin, M.R. Effect of cell-photosensitizer binding and cell density on microbial photoinactivation. Antimicrob. Agents Chemother. 2005, 49, 2329–2335. [Google Scholar] [CrossRef] [PubMed]
- Hamblin, M.R. Potentiation of antimicrobial photodynamic inactivation by inorganic salts. Expert Rev. Anti-Infect. Ther. 2017, 15, 1059–1069. [Google Scholar] [CrossRef] [PubMed]
- Łabuz, P.; Sadowski, R.; Stochel, G.; Macyk, W. Visible light photoactive titanium dioxide aqueous colloids and coatings. Chem. Eng. J. 2013, 230, 188–194. [Google Scholar] [CrossRef]
- Arce-Sarria, A.; Machuca-Martínez, F.; Bustillo-Lecompte, C.; Hernández-Ramírez, A.; Colina-Márquez, J. Degradation and Loss of Antibacterial Activity of Commercial Amoxicillin with TiO2/WO3-Assisted Solar Photocatalysis. Catalysts 2018, 8, 222. [Google Scholar] [CrossRef]
- Sakar, M.; Prakash, R.M.; Do, T.-O. Insights into the TiO2-Based Photocatalytic Systems and Their Mechanisms. Catalysts 2019, 9, 680. [Google Scholar] [CrossRef]
- Buchalska, M.; Łabuz, P.; Kuncewicz, J.; Macyk, W. Mechanism of the photocatalytic processes on titanium (IV) titanium (IV)-catecholate catecholate complexes complexes upon two colors irradiation. In Proceedings of the 7th European Meeting on Solar Chemistry and Photocatalysis: Environmental Applications—SPEA7, Oporto, Portugal, 17–20 June 2012. [Google Scholar]
- Jańczyk, A.; Wolnicka-Głubisz, A.; Urbanska, K.; Kisch, H.; Stochel, G.; Macyk, W. Photodynamic activity of platinum (IV) chloride surface-modified TiO2 irradiated with visible light. Free Radic. Biol. Med. 2008, 44, 1120–1130. [Google Scholar] [CrossRef] [PubMed]
- Macyk, W.; Szaciłowski, K.; Stochel, G.; Buchalska, M.; Kuncewicz, J.; Łabuz, P. Titanium (IV) complexes as direct TiO2 photosensitizers. Coord. Chem. Rev. 2010, 254, 2687–2701. [Google Scholar] [CrossRef]
- Dąbrowski, J.M.; Pucelik, B.; Regiel-Futyra, A.; Brindell, M.; Mazuryk, O.; Kyzioł, A.; Stochel, G.; Macyk, W.; Arnaut, L.G. Engineering of relevant photodynamic processes through structural modifications of metallotetrapyrrolic photosensitizers. Coord. Chem. Rev. 2016, 325, 67–101. [Google Scholar] [CrossRef]
- Pucelik, B.; Gürol, I.; Ahsen, V.; Dumoulin, F.; Dąbrowski, J.M. Fluorination of phthalocyanine substituents: Improved photoproperties and enhanced photodynamic efficacy after optimal micellar formulations. Eur. J. Med. Chem. 2016, 124, 284–298. [Google Scholar] [CrossRef]
- Dąbrowski, J.M.; Pucelik, B.; Pereira, M.M.; Arnaut, L.G.; Stochel, G. Towards tuning PDT relevant photosensitizer properties: Comparative study for the free and Zn2+ coordinated meso-tetrakis [2,6-difluoro-5-(N-methylsulfamylo) phenyl] porphyrin. J. Coord. Chem. 2015, 68, 3116–3134. [Google Scholar] [CrossRef]
- Regulska, E.; Rivera-Nazario, D.M.; Karpinska, J.; Plonska-Brzezinska, M.E.; Echegoyen, L. Zinc Porphyrin-Functionalized Fullerenes for the Sensitization of Titania as a Visible-Light Active Photocatalyst: River Waters and Wastewaters Remediation. Molecules 2019, 24, 1118. [Google Scholar] [CrossRef] [PubMed]
- Stangel, C.; Schubert, C.; Kuhri, S.; Rotas, G.; Margraf, J.T.; Regulska, E.; Clark, T.; Torres, T.; Tagmatarchis, N.; Coutsolelos, A.G. Tuning the reorganization energy of electron transfer in supramolecular ensembles–metalloporphyrin, oligophenylenevinylenes, and fullerene–and the impact on electron transfer kinetics. Nanoscale 2015, 7, 2597–2608. [Google Scholar] [CrossRef]
- Regulska, E.; Rivera-Nazario, D.M.; Karpinska, J.; Plonska-Brzezinska, M.E.; Echegoyen, L. Enhanced Photocatalytic Performance of Porphyrin/Phthalocyanine and Bis (4-pyridyl) pyrrolidinofullerene modified Titania. ChemistrySelect 2017, 2, 2462–2470. [Google Scholar] [CrossRef]
- Carvalho, C.M.; Alves, E.; Costa, L.; Tomé, J.P.; Faustino, M.A.; Neves, M.G.; Tomé, A.C.; Cavaleiro, J.A.; Almeida, A.; Cunha, A.N. Functional cationic nanomagnet-porphyrin hybrids for the photoinactivation of microorganisms. ACS Nano 2010, 4, 7133–7140. [Google Scholar] [CrossRef] [PubMed]
- Lauceri, R.; Gurrieri, S.; Bellacchio, E.; Contino, A.; Monsu’scolaro, L.; Romeo, A.; Toscano, A.; Purrello, R. J-Type Aggregates of the Anionic Meso-Tetrakis (4-Sulfonatophenyl) Porphine Induced by ‚Hindered’Cationic Porphyrins. Supramol. Chem. 2000, 12, 193–202. [Google Scholar] [CrossRef]
- Dąbrowski, J.M.; Pucelik, B.; Pereira, M.M.; Arnaut, L.G.; Macyk, W.; Stochel, G. New hybrid materials based on halogenated metalloporphyrins for enhanced visible light photocatalysis. RSC Adv. 2015, 5, 93252–93261. [Google Scholar] [CrossRef]
- Satoh, N.; Nakashima, T.; Kamikura, K.; Yamamoto, K. Quantum size effect in TiO2 nanoparticles prepared by finely controlled metal assembly on dendrimer templates. Nat. Nanotechnol. 2008, 3, 106. [Google Scholar] [CrossRef]
- Reddy, K.M.; Manorama, S.V.; Reddy, A.R. Bandgap studies on anatase titanium dioxide nanoparticles. Mater. Chem. Phys. 2003, 78, 239–245. [Google Scholar] [CrossRef]
- Tachibana, Y.; Haque, S.A.; Mercer, I.P.; Durrant, J.R.; Klug, D.R. Electron injection and recombination in dye sensitized nanocrystalline titanium dioxide films: A comparison of ruthenium bipyridyl and porphyrin sensitizer dyes. J. Phys. Chem. B 2000, 104, 1198–1205. [Google Scholar] [CrossRef]
- Nazeeruddin, M.K.; Péchy, P.; Renouard, T.; Zakeeruddin, S.M.; Humphry-Baker, R.; Cointe, P.; Liska, P.; Cevey, L.; Costa, E.; Shklover, V.; et al. Engineering of efficient panchromatic sensitizers for nanocrystalline TiO2-based solar cells. J. Am. Chem. Soc. 2001, 123, 1613–1624. [Google Scholar] [CrossRef] [PubMed]
- Qi, K.; Zasada, F.; Piskorz, W.; Indyka, P.; Grybos, J.; Trochowski, M.; Buchalska, M.; Kobielusz, M.; Macyk, W.; Sojka, Z. Self-Sensitized Photocatalytic Degradation of Colorless Organic Pollutants Attached to Rutile Nanorods Experimental and Theoretical DFT+ D Studies. J. Phys. Chem. C 2016, 120, 5442–5456. [Google Scholar] [CrossRef]
- Sadowski, R.; Wach, A.; Buchalska, M.; Kuśtrowski, P.; Macyk, W. Photosensitized TiO2 films on polymers–Titania-polymer interactions and visible light induced photoactivity. Appl. Surf. Sci. 2019, 475, 710–719. [Google Scholar] [CrossRef]
- Kobielusz, M.; Pilarczyk, K.; Świętek, E.; Szaciłowski, K.; Macyk, W. Spectroelectrochemical analysis of TiO2 electronic states–Implications for the photocatalytic activity of anatase and rutile. Catal. Today 2018, 309, 35–42. [Google Scholar] [CrossRef]
- Buchalska, M.; Kobielusz, M.; Matuszek, A.; Pacia, M.; Wojtyła, S.; Macyk, W. On oxygen activation at rutile-and anatase-TiO2. ACS Catal. 2015, 5, 7424–7431. [Google Scholar] [CrossRef]
- Buchalska, M.; Łabuz, P.; Bujak, Ł.; Szewczyk, G.; Sarna, T.; Maćkowski, S.; Macyk, W. New insight into singlet oxygen generation at surface modified nanocrystalline TiO2–the effect of near-infrared irradiation. Dalton Trans. 2013, 42, 9468–9475. [Google Scholar] [CrossRef]
- Yang, S.M.; Lee, D.W.; Park, H.J.; Kwak, M.H.; Park, J.M.; Choi, M.G. Hydrogen Peroxide Enhances the Antibacterial Effect of Methylene Blue-based Photodynamic Therapy on Biofilm-forming Bacteria. Photochem. Photobiol. 2019, 95, 833–838. [Google Scholar] [CrossRef]
- Garcez, A.S.; Núñez, S.C.; Baptista, M.S.; Daghastanli, N.A.; Itri, R.; Hamblin, M.R.; Ribeiro, M.S. Antimicrobial mechanisms behind photodynamic effect in the presence of hydrogen peroxide. Photochem. Photobiol. Sci. 2011, 10, 483–490. [Google Scholar] [CrossRef]
- McCullagh, C.; Robertson, P.K. Photo-dynamic biocidal action of methylene blue and hydrogen peroxide on the cyanobacterium Synechococcus leopoliensis under visible light irradiation. J. Photochem. Photobiol. B Biol. 2006, 83, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Funk, R.S.; Krise, J.P. Exposure of cells to hydrogen peroxide can increase the intracellular accumulation of drugs. Mol. Pharm. 2007, 4, 154–159. [Google Scholar] [CrossRef]
- Huang, Y.-Y.; Choi, H.; Kushida, Y.; Bhayana, B.; Wang, Y.; Hamblin, M.R. Broad-spectrum antimicrobial effects of photocatalysis using titanium dioxide nanoparticles are strongly potentiated by addition of potassium iodide. Antimicrob. Agents Chemother. 2016, 60, 5445–5453. [Google Scholar] [CrossRef]
- Wen, X.; Zhang, X.; Szewczyk, G.; El-Hussein, A.; Huang, Y.-Y.; Sarna, T.; Hamblin, M.R. Potassium iodide potentiates antimicrobial photodynamic inactivation mediated by rose bengal in in vitro and in vivo studies. Antimicrob. Agents Chemother. 2017, 61, e00467-17. [Google Scholar] [CrossRef]
- Huang, L.; Szewczyk, G.; Sarna, T.; Hamblin, M.R. Potassium iodide potentiates broad-spectrum antimicrobial photodynamic inactivation using Photofrin. ACS Infect. Dis. 2017, 3, 320–328. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.; El-Hussein, A.; Xuan, W.; Hamblin, M.R. Potentiation by potassium iodide reveals that the anionic porphyrin TPPS4 is a surprisingly effective photosensitizer for antimicrobial photodynamic inactivation. J. Photochem. Photobiol. B Biol. 2018, 178, 277–286. [Google Scholar] [CrossRef] [PubMed]
- Pinto, S.M.; Henriques, C.A.; Tome, V.A.; Vinagreiro, C.S.; Calvete, M.J.; Dąbrowski, J.M.; Pineiro, M.; Arnaut, L.G.; Pereira, M.M. Synthesis of meso-substituted porphyrins using sustainable chemical processes. J. Porphyr. Phthalocyanines 2016, 20, 45–60. [Google Scholar] [CrossRef]
- Johnstone, R.A.; Nunes, M.L.P.; Pereira, M.M.; Gonsalves, A.M.d.A.R.; Serra, A.C. Improved syntheses of 5, 10, 15, 20-tetrakisaryl-and tetrakisalkylporphyrins. Heterocycles 1996, 7, 1423–1437. [Google Scholar]



| Homogeneous | Heterogeneous (after Modification with qTiO2) | |||||
|---|---|---|---|---|---|---|
| Sensitizer | λmax/nm | λmax/nm with qTiO2 | Eg/eV | Size/nm DLS | Size/nm SEM (Average ± SD) | ζ/mV |
| TPPS | 413, 518, 555, 580, 640 | 413,516, 556, 645 | 3.37 | 13 | 38 ± 6 | −27 |
| F2POH | 410, 509, 541, 575, 640 | 413, 512, 644 | 3.47 | 28 | 55 ± 11 | −26 |
| ZnF2POH | 418, 553 | 419, 552 | 3.39 | 68 | 41 ± 9 | −20 |
| qTiO2 | 3.50 | 7 | 56 ± 24 | −33 | ||
| Sensitizer | λexp/nm | ΦΔa | τ0 | λexp + qTiO2 | τfl + qTiO2 |
|---|---|---|---|---|---|
| TPPS | 605, 650 | 0.64 | 3.41 | 605 *, 645, 705 | 5.71 |
| F2POH | 602, 651 | 0.65 | 5.04 | 602 *, 652, 703 | 7.25 |
| ZnF2POH | 603, 649 | 0.85 | 2.28 | 603, 649 | 4.38 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sułek, A.; Pucelik, B.; Kobielusz, M.; Łabuz, P.; Dubin, G.; Dąbrowski, J.M. Surface Modification of Nanocrystalline TiO2 Materials with Sulfonated Porphyrins for Visible Light Antimicrobial Therapy. Catalysts 2019, 9, 821. https://doi.org/10.3390/catal9100821
Sułek A, Pucelik B, Kobielusz M, Łabuz P, Dubin G, Dąbrowski JM. Surface Modification of Nanocrystalline TiO2 Materials with Sulfonated Porphyrins for Visible Light Antimicrobial Therapy. Catalysts. 2019; 9(10):821. https://doi.org/10.3390/catal9100821
Chicago/Turabian StyleSułek, Adam, Barbara Pucelik, Marcin Kobielusz, Przemysław Łabuz, Grzegorz Dubin, and Janusz M. Dąbrowski. 2019. "Surface Modification of Nanocrystalline TiO2 Materials with Sulfonated Porphyrins for Visible Light Antimicrobial Therapy" Catalysts 9, no. 10: 821. https://doi.org/10.3390/catal9100821
APA StyleSułek, A., Pucelik, B., Kobielusz, M., Łabuz, P., Dubin, G., & Dąbrowski, J. M. (2019). Surface Modification of Nanocrystalline TiO2 Materials with Sulfonated Porphyrins for Visible Light Antimicrobial Therapy. Catalysts, 9(10), 821. https://doi.org/10.3390/catal9100821








