1. Introduction
The catalytic activity of a gold-based heterogeneous catalyst depends on the nature of the support and in particular on the size of the gold nanoparticles deposited on it, which in turn depends on the synthetic methodology. The size of the nanoparticles is of significant importance, since in many studies it was evidenced a correlation of inverse proportionality between the activity of the catalyst and the size of the gold nanoparticles [
1]. The techniques for the preparation of gold nanoparticles are various. Almost all use chloroauric acid (HAuCl
4∙3H
2O) as the metal precursor. The most used techniques in order to obtain an efficient heterogeneous catalyst are: impregnation (IMP), deposition-precipitation (DP) and colloidal methods. As regard as gold samples, dry impregnation is the first method historically used. The procedure involves filling the pores of the support by adding an aqueous HAuCl
4 solution to form a wet paste, which is then dried and calcined. However, this technique does not allow obtaining high gold dispersion. In fact, it leads to nanoparticles of size above 30 nm, due to sintering during the heat treatment, promoted by the Cl
− ions still present on the sample [
2]. Therefore, the gold-based catalysts prepared by this technique have a low catalytic activity [
3]. An alternative synthetic approach for Au loading has been recently reported [
4,
5], namely a liquid phase reductive deposition (LPRD). The method consists of mixing a solution of HAuCl
4 with a solution of NaOH. The resulting solution was aged for 24 h, in the dark, to complete the hydroxylation of Au
3+ ions. Then the appropriate amount of support was added to the solution. In LPRD, unlike IMP, a washing procedure is carried out, in order to eliminate residual chloride. DP is a widely used procedure for the preparation of gold-based catalysts supported on metal oxides. In fact, this method works very well with support whose isoelectric point is greater than 5, such as magnesia, alumina, titania, zirconia and ceria. In a typical procedure, after the addition of the support to an aqueous solution of HAuCl
4∙3H
2O, the pH of the suspension is brought to a fixed value by the addition of NaOH. The whole is heated under stirring, before washing to remove the Na
+ and Cl
− ions, drying and calcining in air. This basic approach has many variables such as for example the synthesis temperature and the pH. In general the pH is set between 7 and 9, since previous studies have shown that this value corresponds to the proper compromise between the loading of high gold amount on the support and the achievement of particles with the proper size (1–10 nm) [
6].
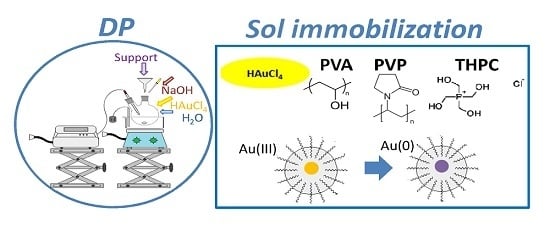
The colloidal methods involve the use of a surfactant, which has the fundamental role of limiting the growth and aggregation of the particles, forming a monolayer on the surface of the metal particles; in this way also the long-term stability of the catalyst increases. Such a stage of formation of micelles by the protective agent is followed by a reduction stage from Au
3+ to Au
0. Historically, the first colloidal method involves the reduction of gold ions by citrate [
7]. Methods that are much more efficient use the NaOH/tetrakis (hydroxymethyl) phosphonium chloride (THPC) system or organic polymers using NaBH
4 as a reducing agent. In the first case the NaOH/THPC system protects the gold particles, to prevent aggregation, and it also works as
in situ generator of the reducing agent [
8]. In the second case, polymers such as polyvinyl alcohol (PVA) or polyvinylpyrrolidone (PVP) may be used as stabilizing agents. They are commercially available, relatively inexpensive, soluble in water, non-toxic and both have a long chain of carbon atoms functionalized, respectively, with OH groups and pyrrolidone groups, as reported in
Scheme 1.
As the choice of the support plays a vital role in determining the activity and selectivity of a catalyst, in order to assess the effect of the support we have investigated three different metal oxides. In particular, we have chosen TiO
2, as it is inexpensive and has high chemical stability; ZrO
2, for its thermal and chemical stability; CeO
2, because it has a high ability to store and deliver oxygen (this is a very important point for oxidation reactions), due to its ability to change the oxidation state from +3 to +4 [
9]. Moreover, essential characteristics for a support are its stability in the reaction media, a low cost, adequate porous structure, acid-base properties [
10] and high surface area. In fact, a good support must be able to allow a high dispersion of the active phase with a high degree of thermal stability of the catalyst.
The aim of the work is the synthesis of new gold catalysts active in oxidation reactions. As catalytic test we have investigated the oxidation of glucose, which is a product derivable from biomass and exploited for the future bio-refinery. The biomass conversion processes must be competitive from an economic point of view but must be also environmentally sustainable. Therefore, we need the development of specific technologies. Particular attention is paid to the fraction of lignocellulosic biomass (LCF) that is the indigestible part of plants; in fact, it is not only the most abundant fraction in nature, but it is completely made up of carbohydrates, in particular glucose, which are a rich source of carbon. In fact, they can be then converted into a set of “bio-platform” molecules (BPM) which can be used as starting materials for the production of high commercial value products. In the literature there are few studies on the oxidation of glucose to glucaric acid [
11], while great interest is placed at the oxidation to gluconic acid, because of its wide field of industrial applications (the annual world production of gluconic acid is of 100,000 tonnes [
12]). Currently, at industrial level, the gluconic acid is produced by enzymatic oxidation of glucose, using biotechnological processes involving bacteria such as
Aspergillus niger and
Gluconobacter suboxydans [
13]. However, this process presents considerable disadvantages, both from the practical point of view and from the economic point of view, as the removal of waste water; the low rate of reaction, the death of the bacteria and the accumulation of the substances secreted by these [
14]. The existence of such problems using enzymatic oxidation for the production of gluconic acid, has led the scientific community to look for an alternative view by green chemistry. A sustainable method is the oxidation with oxygen in mild reaction conditions of an aqueous glucose solution using a heterogeneous catalyst.
The first reaction of carbohydrate oxidation mediated by catalysts was conducted in 1861 by von Gorup-Besanez [
15], using a Pt-based catalyst. In the last two decades studies have been conducted on the use of catalysts based on Pt and Pd for the oxidation reaction of
d-glucose to gluconic acid, which, however, have the disadvantages of not being selective against oxidation of the group aldehyde [
16] and undergo deactivation in basic and acid environments. This last point is crucial: the rate of the oxidation reaction of a carbohydrate mainly depends on the metal and on the pH, while the selectivity depends primarily on pH and temperature [
17]. The dependence of the reaction by the pH rate is determined by the fact that in basic conditions, the carbohydrate is present in the open linear form, in which it is present an aldehyde group, which is much more reactive towards oxidation than hydroxyl. The selectivity problems can be overcome by promoting catalysis, by the Pt or Pd [
18], with metals such as Bi or Pb, but these catalysts are not stable since they suffer from leaching.
After the discovery of the ability of gold nanoparticles to catalyze oxidation reactions, the interest in gold as a catalyst in selective oxidation of carbohydrates increased. In fact, gold is more resistant to deactivation than Pt and Pd. Moreover, due to this characteristic, the use of gold-based catalysts takes the advantage of being able to work in a wide range of pH, which is not possible with the biotechnological process. In 2002, Biella
et al. [
19] published studies on glucose oxidation reaction using Au/C catalyst and obtaining a selectivity of 100% with respect to gluconic acid.
An effect of the support for the reaction was found by Ishida
et al. [
20] comparing catalysts supported on metal oxides (TiO
2, CeO
2, ZrO
2 and Al
2O
3) and one supported on C. The stability of the catalyst was proven by Baatz
et al. [
21] who recycled 20 times at 40 °C an Au/Al
2O
3 catalyst and from Mirescu
et al. [
22] who recycled 17 times at 40 °C an Au/TiO
2 catalyst. In the present paper it was investigated the oxidation of glucose. The aim of the work is the synthesis of new gold catalysts active in oxidation reactions. The catalysts must have very small gold particles and must be well dispersed on the support. For this reason, we have compared the two synthetic approaches of deposition precipitation (DP) and colloidal methods. It was also rated as the different surfactants used in the colloidal method affect the behavior of the catalyst.
3. Materials and Methods
3.1. Materials
(NH4)2Ce(NO3)6 (≥98.5%, Sigma-Aldrich, St Louis, MO, USA); ZrOCl2·8H2O (≥99.5%, Sigma-Aldrich, Sigma Aldrich Chemie, GMBH, Riedstr, Germany); TiOSO4·H2SO4·H2O (Aldrich Chemistry, Sigma-Aldrich, St Louis, MO, USA); PVA (98% P.M. 13.000–23.000 d, Sigma-Aldrich); PVP (P.M. 40.000 d, Sigma-Aldrich); THPC (80% in water, Aldrich Chemistry).
3.2. Synthesis of Supports
Ceria support was synthesized by precipitation from (NH
4)
2Ce(NO
3)
6 by urea at 100 °C in aqueous solution [
24,
25]. The solution was mixed and boiled for 6 h at 100 °C, the precipitate was washed twice in boiling deionized water and dried at 110 °C overnight. The material was then calcined in flowing air (50 mL/min) at 500 °C for 3 h.
Zr(OH)
4 was prepared by precipitation from ZrOCl
2·8H
2O at constant pH = 8.6 and then aged for 20 h at 90 °C [
26,
27]. Then zirconium hydroxide was calcined in air (30 mL/min STP) at 650 °C for 3 h.
Titanium hydroxide was precipitated at pH = 8.6 from 0.5 M titanyl sulphate aqueous solution [
28,
29]. In particular, 40 g of TiOSO
4 xH
2SO
4 yH
2O (Aldrich) were dissolved in 300 mL of distilled water at room temperature under vigorous stirring. The Ti(OH)
4 precipitation was obtained by the drop wise addition of 9 M ammonia solution under vigorous stirring. The suspension was magnetically stirred at 60 °C for 20 h. Then the precipitate was filtered, washed with distilled water in order to remove SO
42− ions and dried at 110 °C for 18 h. The absence of sulphates in the material was verified by IEC analysis. Finally the hydroxide was calcined in air flow at 300 °C for 3 h.
3.3. Synthesis of Catalysts
In all cases the amount of support was calculated as having a final gold loading of 2 wt. %.
3.3.1. Synthesis by Deposition Precipitation (DP)
Gold was added by DP method at pH = 8.6. The oxide supports were suspended in an aqueous solution of HAuCl4·3H2O for 3 h and the pH was controlled by the addition of NaOH (0.5 M). After filtration the samples were dried at 35 °C overnight and finally calcined in air for 1 h at 300 °C. Samples where denoted as ACeDP300, AZrDP300 and ATiDP300.
3.3.2. Synthesis by PVA Sol-Imobilization
A 1 wt. % polyvinyl alcohol (PVA) solution was added to an aqueous HAuCl
4 solution under vigorous stirring at 0 °C (Au/PVA (
w/
w) = 2) [
30]. Then a 0.1 M of freshly prepared solution of NaBH
4 (NaBH
4/Au (mol/mol) = 4) was added, to form a ruby-red metallic sol. Within a few minutes of sol generation, the sol was immobilized by adding the support (CeO
2, ZrO
2 or TiO
2. As regard as TiO
2, this has been previously acidified with H
2SO
4 at pH = 1 [
20]) under vigorous stirring and aged for 12 h. After filtration, the samples were washed for several times with distilled water. The samples were dried at 110 °C overnight. Samples where denoted as ACePVA, AZrPVA and ATiPVA. Part of the final catalysts was finally calcined in air (30 mL/min) for 1 h at different temperatures.
3.3.3. Synthesis by PVP Sol-Imobilization
A 2 wt. % polyvinylpyrrolidone (PVP) solution was added to an aqueous HAuCl
4 solution under vigorous stirring at 0 °C (Au/PVP (
w/
w) = 0.25) [
31]. Then a 0.1 M of freshly prepared solution of NaBH
4 (NaBH
4/Au (mol/mol) = 4) was added, to form a ruby-red metallic sol. Within a few minutes of sol generation, the sol was immobilized by adding the support (CeO
2, ZrO
2 or TiO
2. As regard as TiO
2, this has been previously acidified with H
2SO
4 at pH = 1) under vigorous stirring and aged for 20 h. After filtration, the samples were washed for several times with distilled water. The samples were dried at 110 °C overnight. Samples where denoted as ACePVP, AZrPVP and ATiPVP. Part of the final catalysts was finally calcined in air (30 mL/min) for 1 h at different temperatures.
3.3.4. Synthesis by THPC Sol-Imobilization
A 0.05 M solution of tetrakis (hydroxymethyl)phosphonium chloride (THPC) was added to a 10
−3 M solution of NaOH (NaOH/THPC (mol/mol) = 0.042). Then, an aqueous HAuCl
4 solution was added dropwise under vigorous stirring at room temperature (Au/THPC (
w/
w) = 1) [
27]. The sol was immobilized by adding the support (CeO
2, ZrO
2 or TiO
2 previously acidified with H
2SO
4 at pH = 1) under vigorous stirring and aged for 5 h. After filtration, the samples were washed for several times with distilled water. The samples were dried at 110 °C overnight. Samples where denoted as ACeTHPC, AZrTHPC and ATiTHPC. Part of the final catalysts was finally calcined in air (30 mL/min) for 1 h at different temperatures.
3.4. Methods
Surface areas and pore size distributions were obtained from N
2 adsorption/desorption isotherms at −196 °C (using a Micromeritics ASAP 2000 analyser, Micromeritics, Milan, Italy). Surface area was calculated from the N
2 adsorption isotherm by the BET equation, and pore size distribution was determined by the BJH method [
32]. Total pore volume was taken at
p/
p0 = 0.99.
The sulphate content was determined by ion chromatography (IC). Sulphate concentration was calculated as the average of two independent analyses, each including two chromatographic determinations.
The gold amount for both fresh and exhausted catalysts was determined by atomic absorption spectroscopy (AAS) after microwave disaggregation of the samples (100 mg) using a Perkin-Elmer Analyst 100 (Perkin Elmer, Waltham, MA, USA).
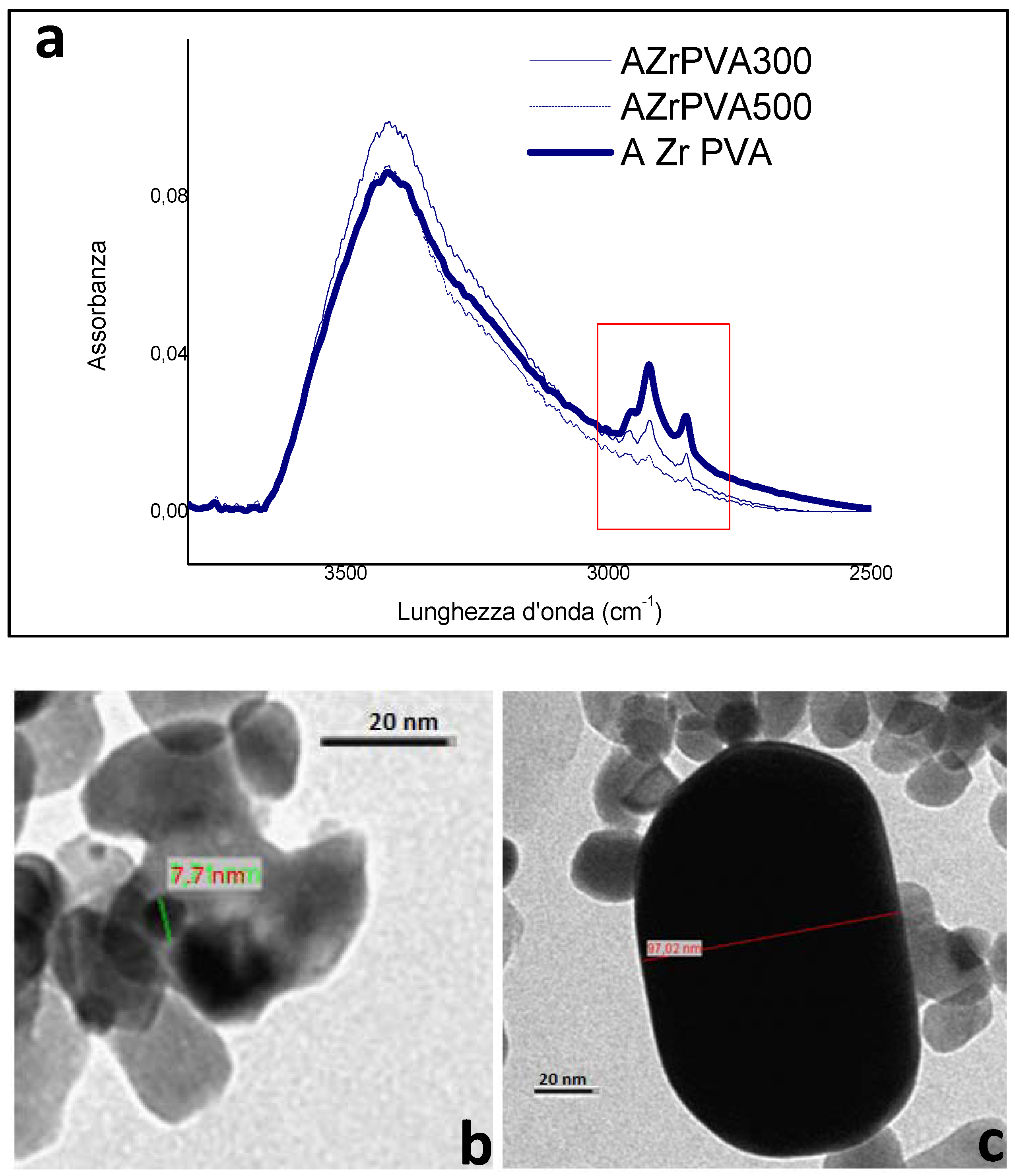
TEM images have been obtained using a Philips 208 Transmission Electron Microscope (FEI, Hillsboro, OR, USA). The samples were prepared by putting one drop of an ethanol dispersion of the catalysts on a copper grid pre-coated with a Formvar film and dried in air.
SEM images have been obtained using a Field Emission Gun Electron Scanning Microscopy LEO 1525 (Carl Zeiss Microscopy, Jena, Germany), after metallization with graphite. The images were acquired by AsB (angular selective BSE) detector (Carl Zeiss Microscopy, Jena, Germany) while elemental composition was determined using Bruker Quantax EDS (Bruker Nano GmbH, Berlin, Germany).
FTIR spectra were obtained on a Bruker spectrophotometer (Bruker Tensor 27, Bruker Optik GmbH, Ettlingen, Germany) (4 cm−1 resolution, MCT detector). Reduced samples were inspected in the form of self-supporting tablets (~25 mg·cm−2). Quartz cells (equipped with KBr windows) connected to a gas vacuum line equipped with rotary and turbomolecular pumps were used.
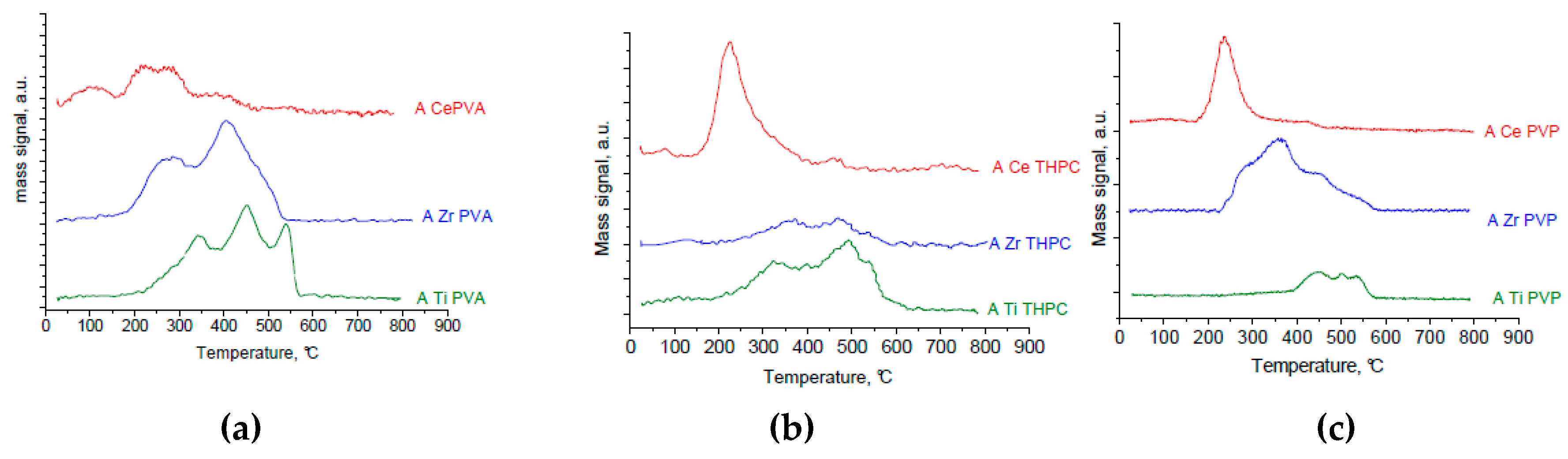
Thermal analyses (TG/DTA) were performed on a STA 409 PC/PG instrument (NETZSCH, Selb, Germany) in flowing air (20 mL/min) with temperature rate set at 5 °C/min in the 25–800 °C temperature range.
TPO measurements were carried out in a lab-made equipment: samples (100 mg) were heated with a temperature rate of 10 °C/min from 25 °C to 600 °C in a 5% O2/He flow (40 mL/min). The effluent gases were analyzed by a TCD detector (GOW-MAC Instrument Co., Shannon, Ireland) and by a Genesys 422 quadrupole mass analyzer (QMS) (EES Genesys House Northwich, Cheshire, UK).
3.5. Catalytic Reactions
Commercial Dextrose (100% pure, PURE Dextro, distributed by Pro Nutrition) was used as substrate. Its oxidation to (
Scheme 2) gluconic acid was carried out in a batch reactor, typically at atmospheric pressure and 60 °C. In particular, 1 g of
d-glucose in 50 mL of distilled H
2O was brought to pH = 9.5 by adding a solution of NaOH 0.1 M. An O
2 flow (100 mL/min) was bubbled in the solution through a porous septum. Separately, 130 mg of catalyst were suspended in 20 mL of distilled H
2O, brought to pH = 9.5 with a solution 0.1 M of NaOH and then charged into the reactor. To monitor the conversion of
d-glucose to gluconic acid, the latter was titrated with NaOH 0.1 M, while keeping the pH constant at 9.5 during the reaction.
The results obtained with the titration were compared with those obtained by means of ion chromatography (IC). A precolumn Dionex AG11, a column Dionex AS11, a suppressor Dionex ASRS-300, a TCD Dionex ED40 (100 mA) were used. In all cases IC results were comparable with those of titration.
4. Conclusions
Heterogeneous catalysis plays a key role in the future biorefinery and there is great interest in the development of new catalysts. Focusing on the transformation of platform molecules through oxidation reactions, the use of nanostructured catalysts based on gold seems to be the most promising alternative. The work was focused on the synthesis of catalysts based on gold nanoparticles on three different supports (CeO2, ZrO2 and TiO2) and two different techniques: deposition-precipitation and colloidal method. Moreover, in the latter case the role of the surfactant (PVA, PVP, THPC) was also investigated. The catalysts were tested in a model reaction: the oxidation of glucose to gluconic acid, which takes place in an aqueous environment and under mild conditions (60 °C and atmospheric pressure).
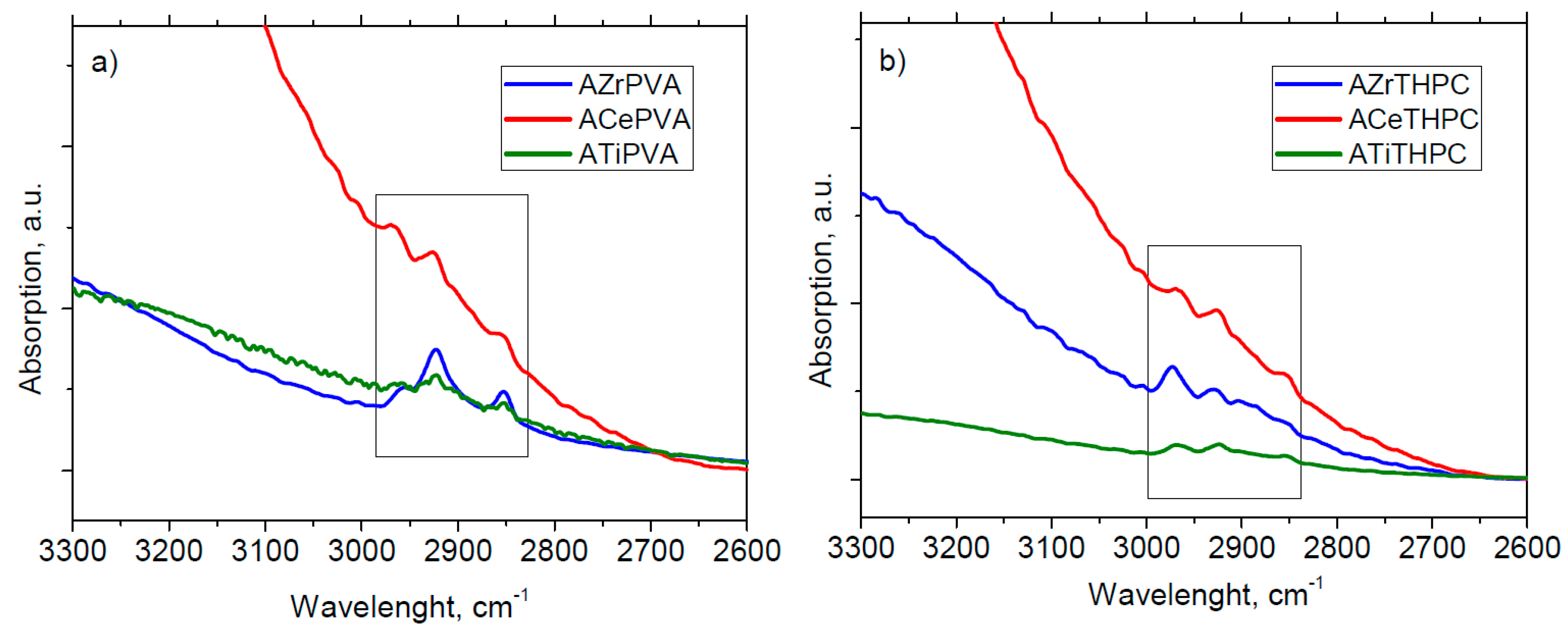
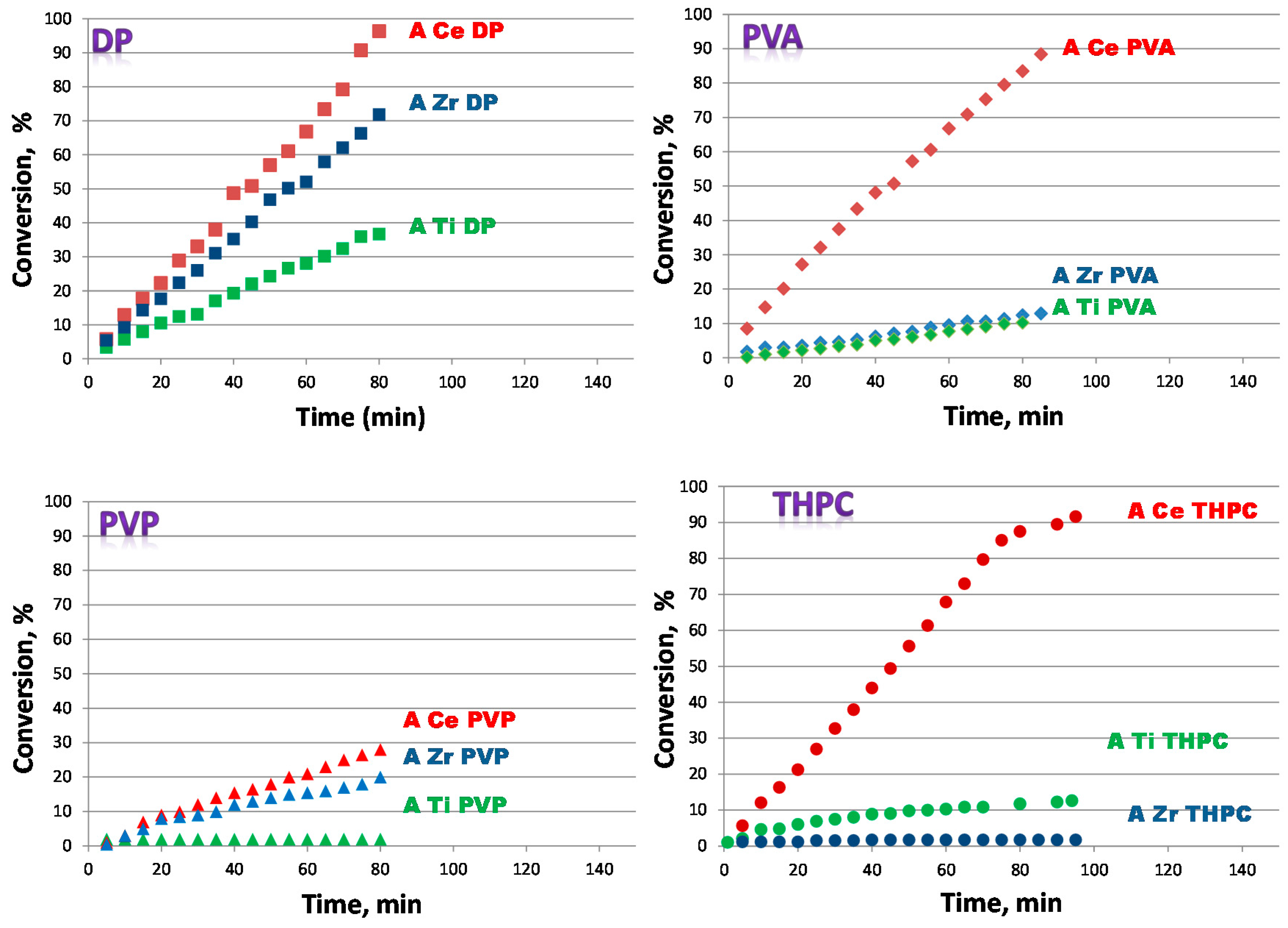
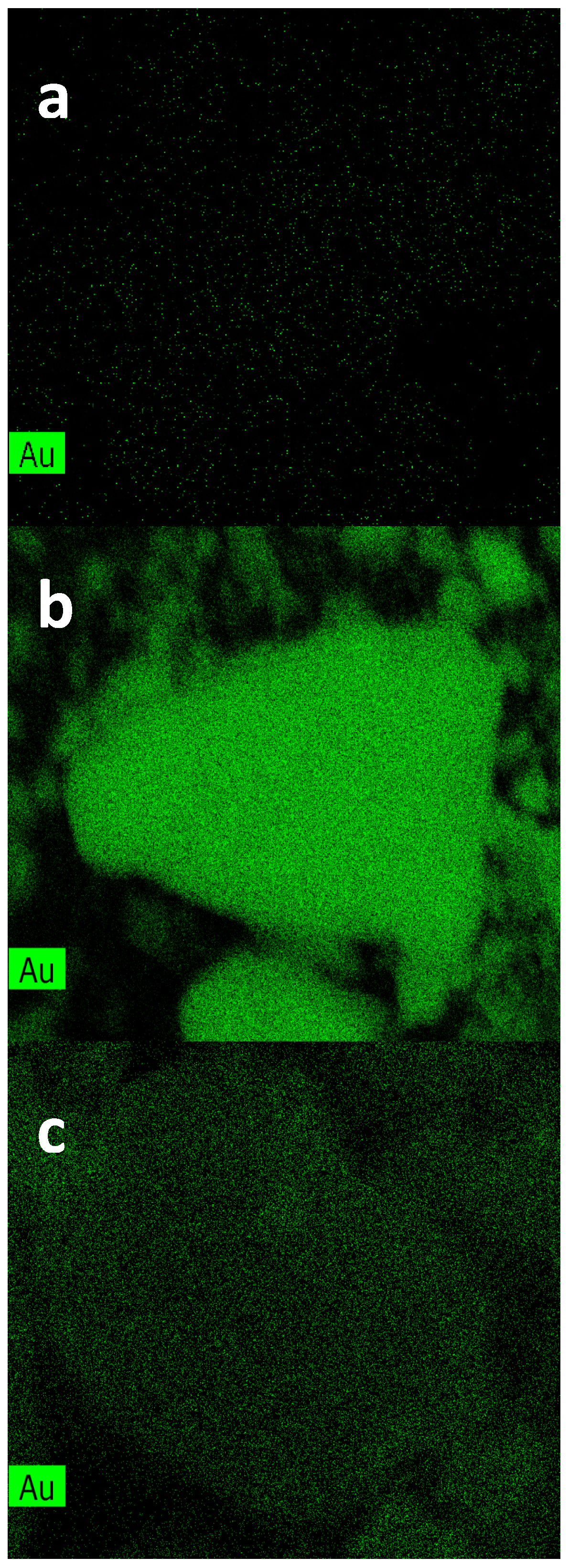
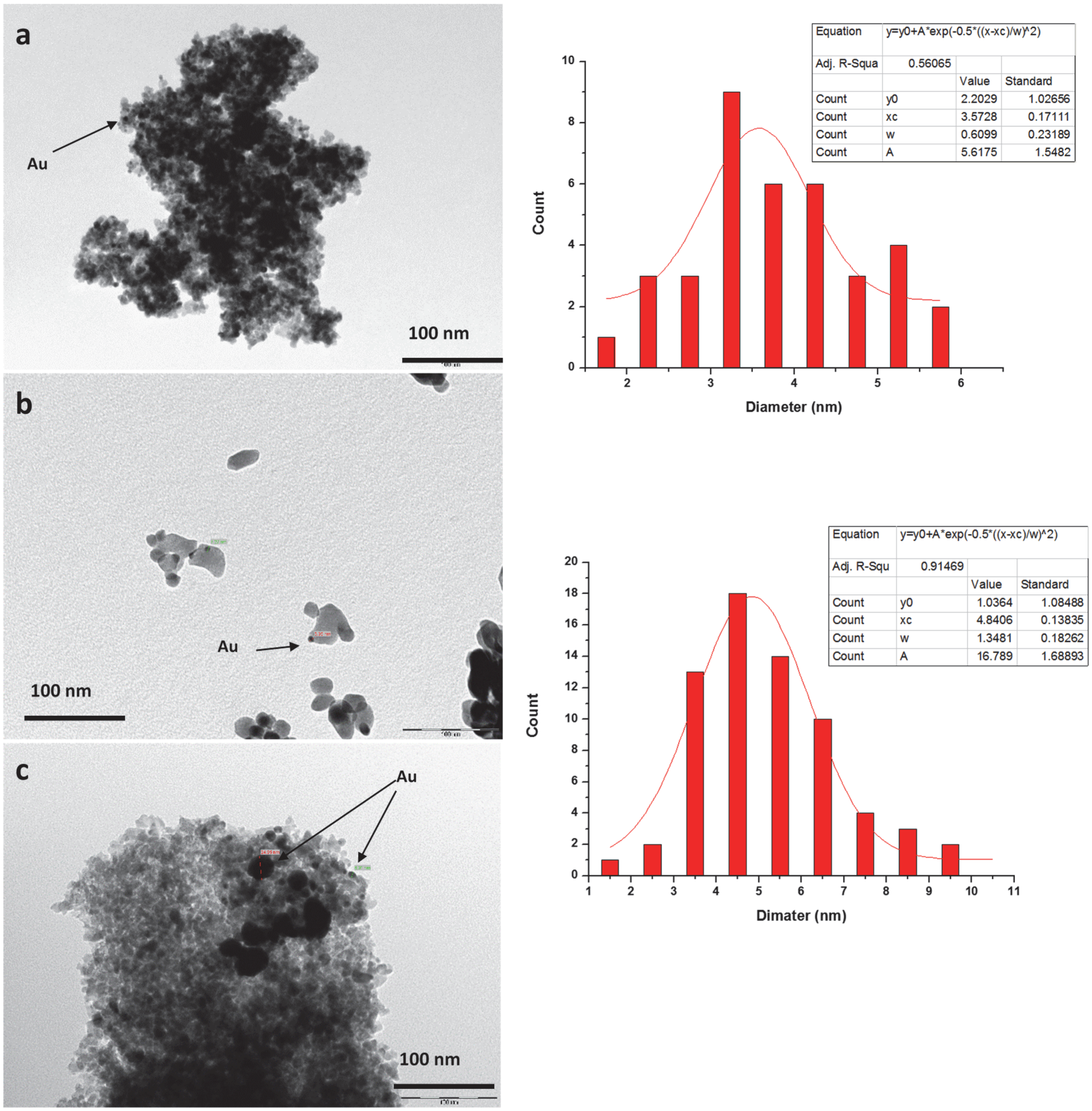
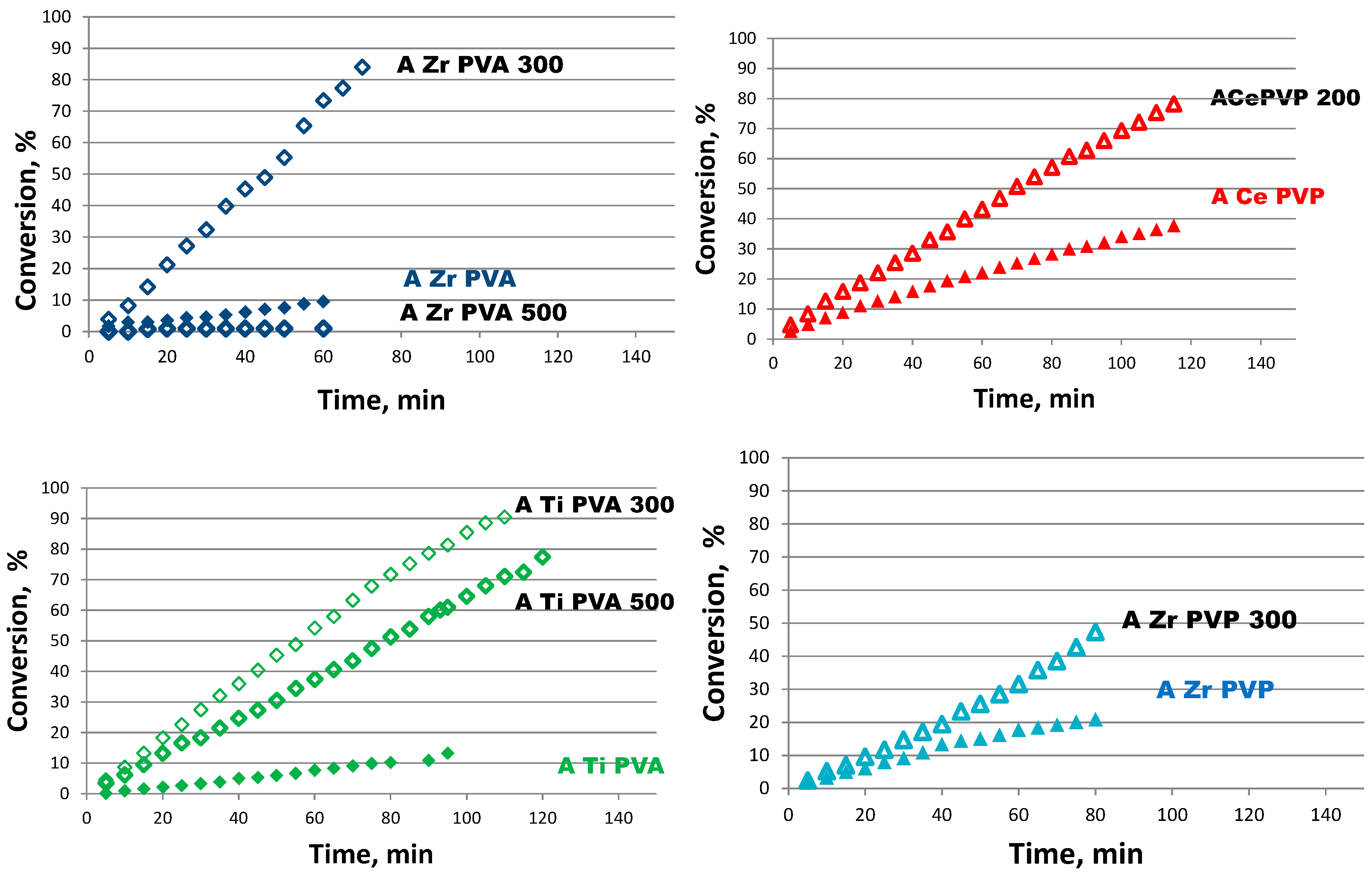
TEM and SEM analyses have shown as for catalysts prepared by colloidal method with PVA small gold nanoparticles are obtained. The latter is a necessary condition, but not sufficient for a good conversion. In fact, for an active sample, the excess of surfactant must be removed because it would coat the surface of the catalyst. The surfactant, however, should not be completely eliminated, since it has the fundamental role of stabilizing the sample preventing nanoparticles from aggregation.
As for the catalysts prepared by colloidal method with THPC, only the sample supported on ceria has sufficiently small nanoparticles. This is a clear indication that the size of the particles not only depends on the synthetic procedure but also on the kind of support.
As regard as catalysts prepared by DP, their activity is lower than samples prepared by PVA. Moreover it is observed a minor effect of the support: catalysts prepared with DP technique do not show a big difference in activity. Samples prepared by colloidal method with PVP gave the worst performance.
It was evidenced that both the synthetic approach and the kind of support affect the catalysts’ activity. In fact, by focusing on the three different supports, with all the preparation methods, the ceria has proved to be the best support. This is due to its ability to obtain small gold nanoparticles and to its ability to accumulate oxygen.
The most appropriate synthesis methodology proved to be the colloidal method with PVA.
Combining the effect of the synthetic method and the effect of the support, ACePVA resulted the best performing catalyst. In fact, it is the most active and is completely recyclable after a simple washing with water.