Recent Advances in Carbon-Silica Composites: Preparation, Properties, and Applications
Abstract
:1. Introduction
1.1. Carbon Material
1.1.1. Activated Carbon
1.1.2. Carbon Nanotubes
1.1.3. Graphene
1.2. Silica Material
1.2.1. Silica Gel
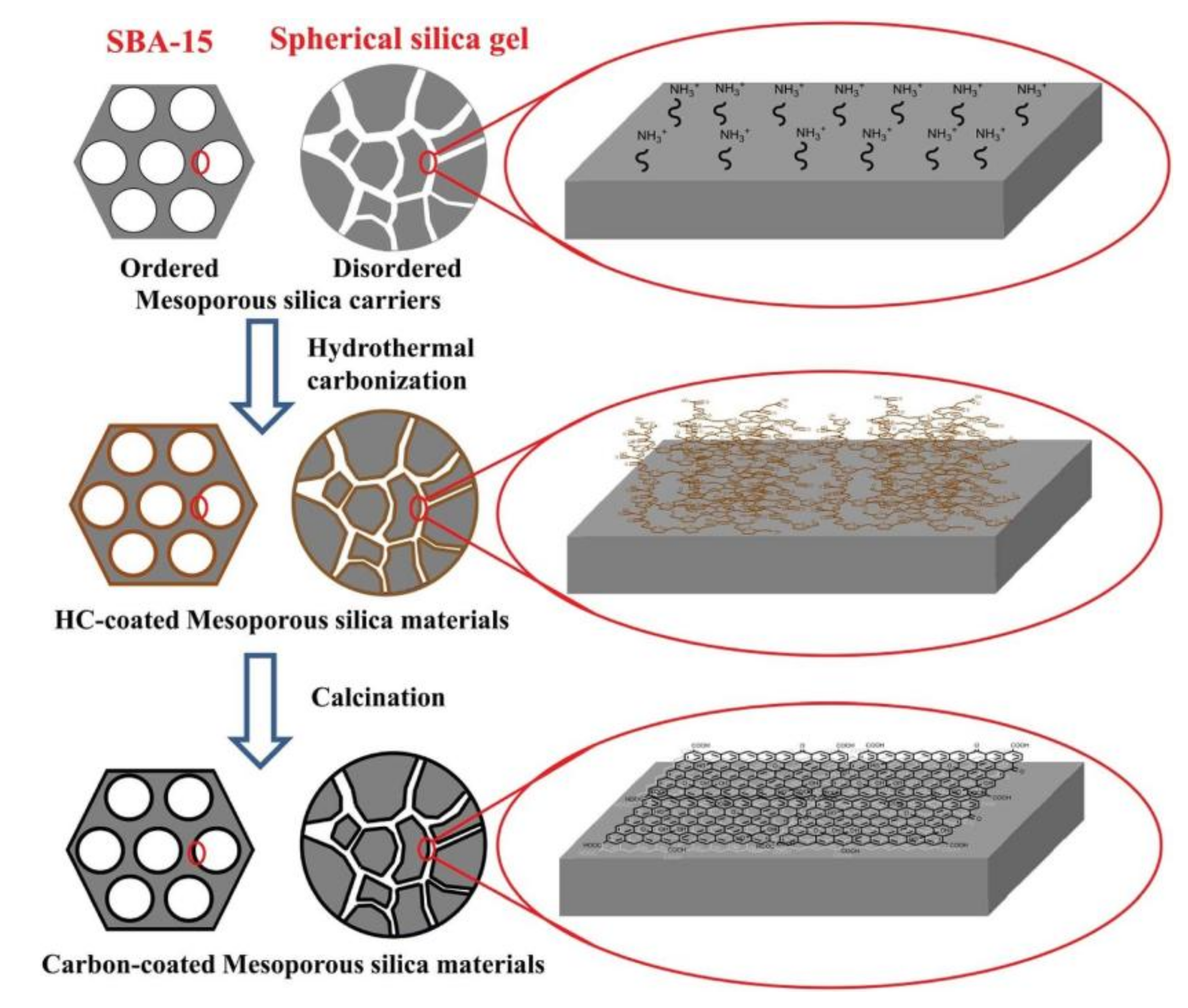
1.2.2. Mesoporous Silica
1.2.3. Molecular Sieve
1.3. Research Hotspots in Porous Composites
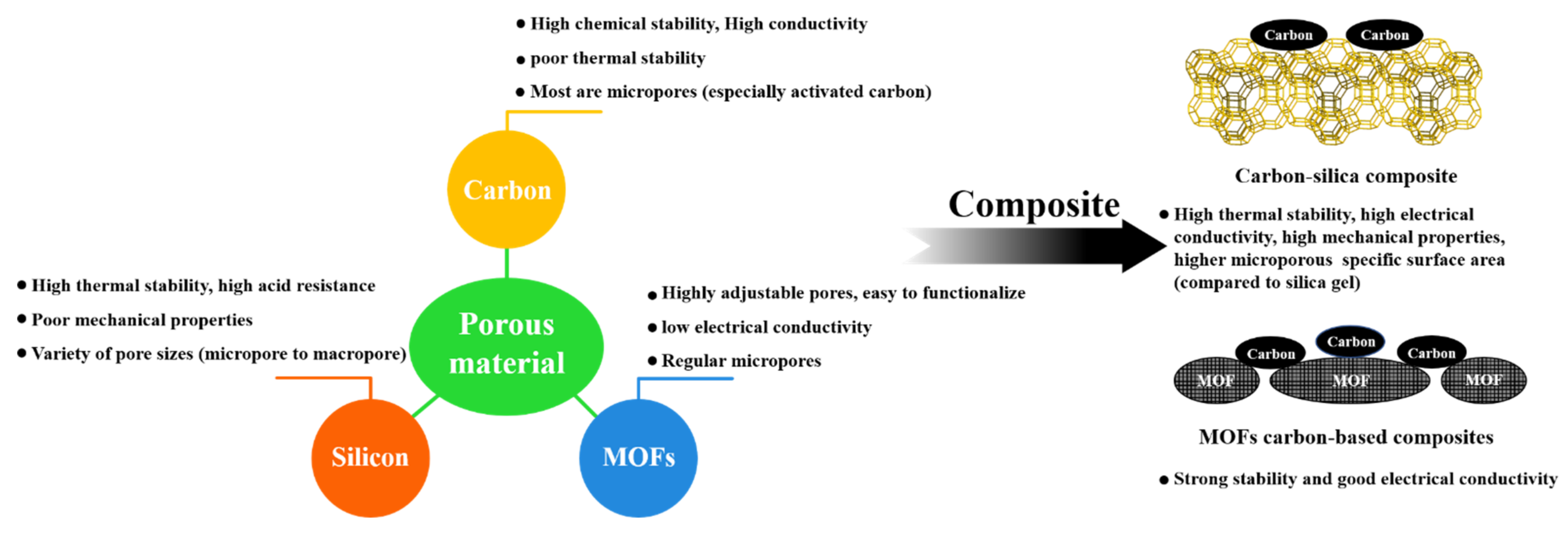
1.4. Definition of Carbon-Silica Composites and Their Advantages
1.4.1. Definition of Carbon-Silica Composites
1.4.2. Comparison of Carbon-Silica Composite with Other Porous Materials
2. Synthetic Method
2.1. Hybrid Pyrolysis Carbonization
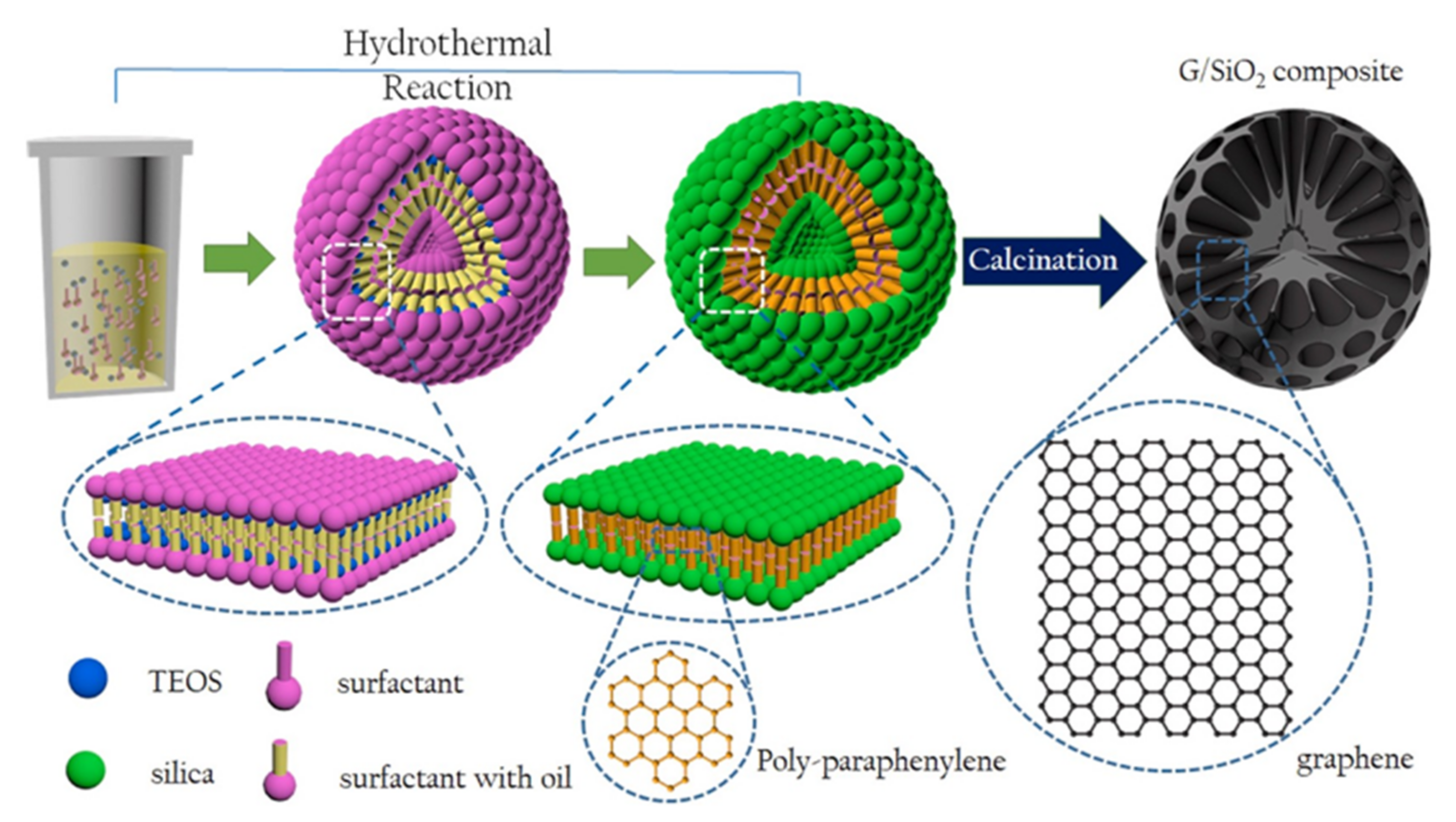
2.2. Hydrothermal Carbonization
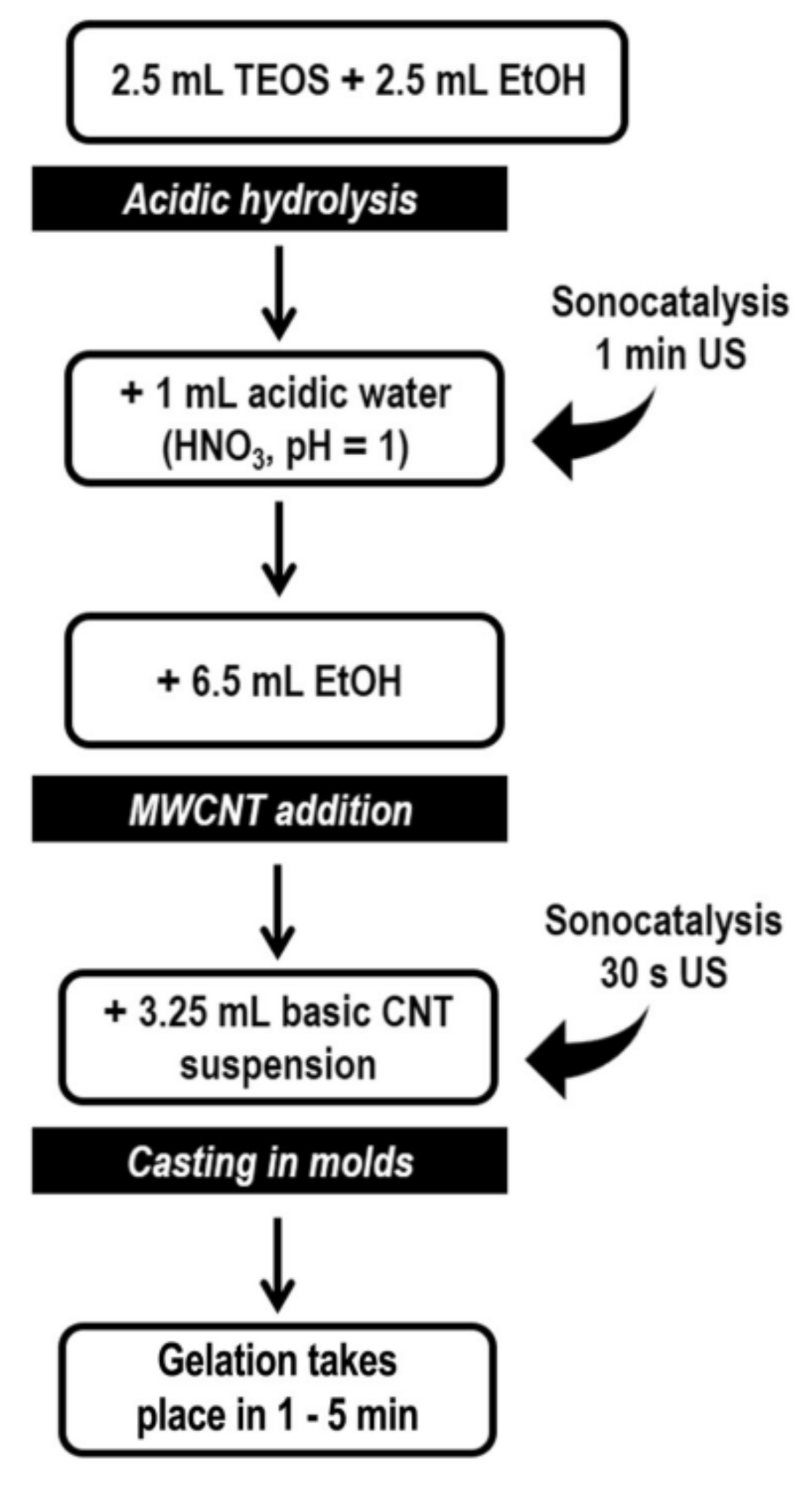
2.3. Sol-Gel Method
3. Application
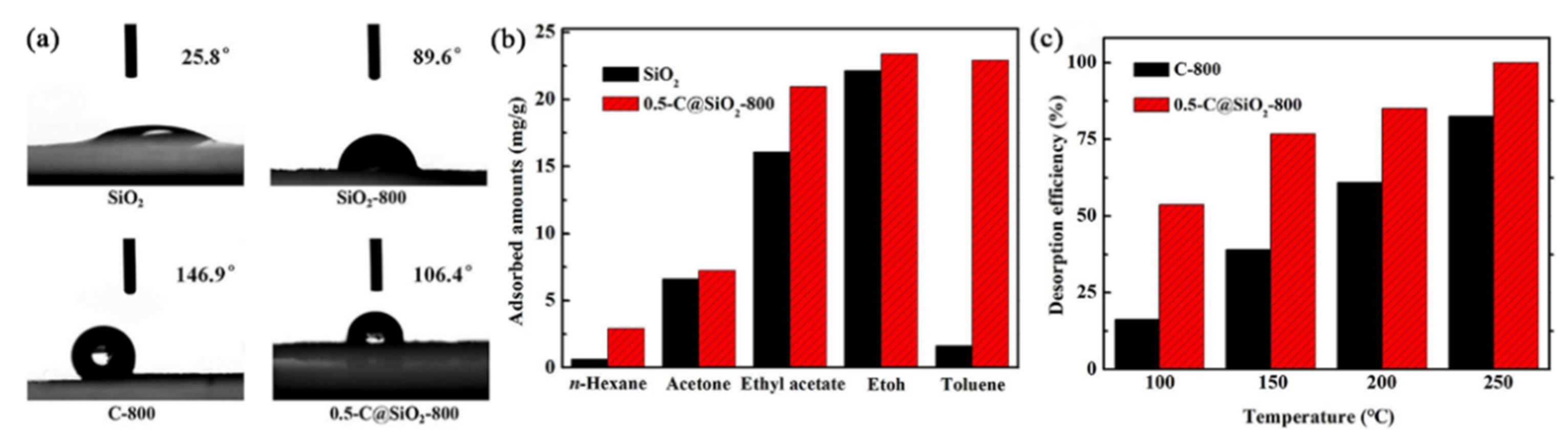
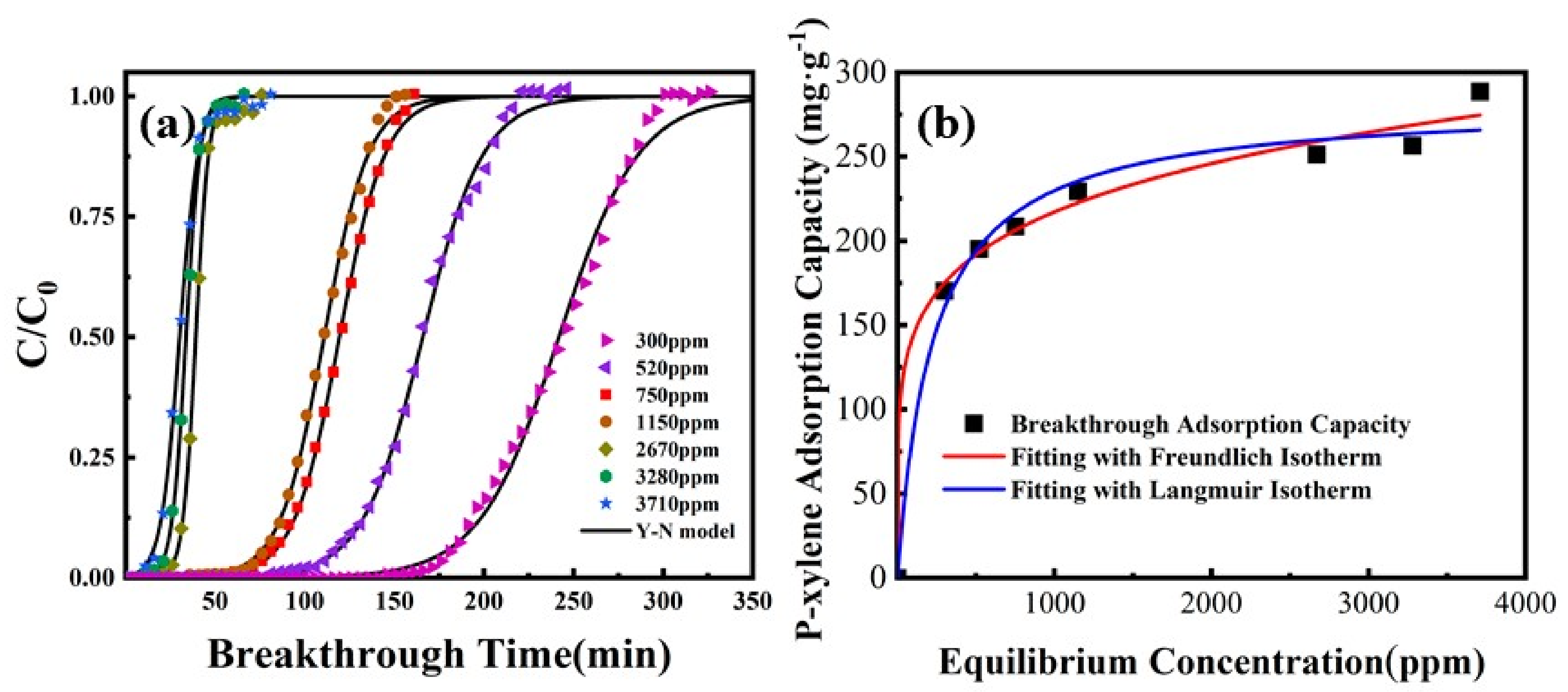
3.1. Adsorbent
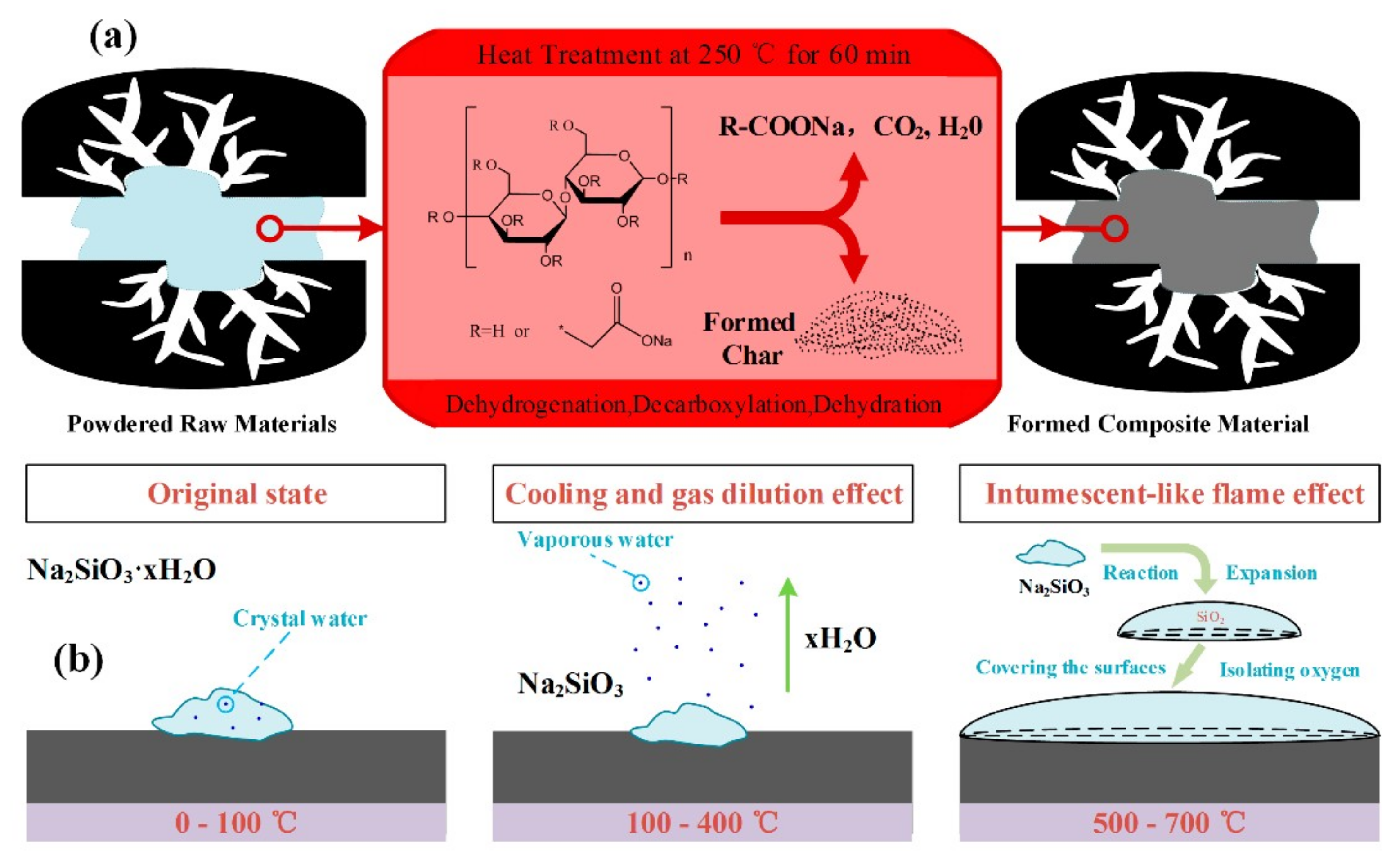
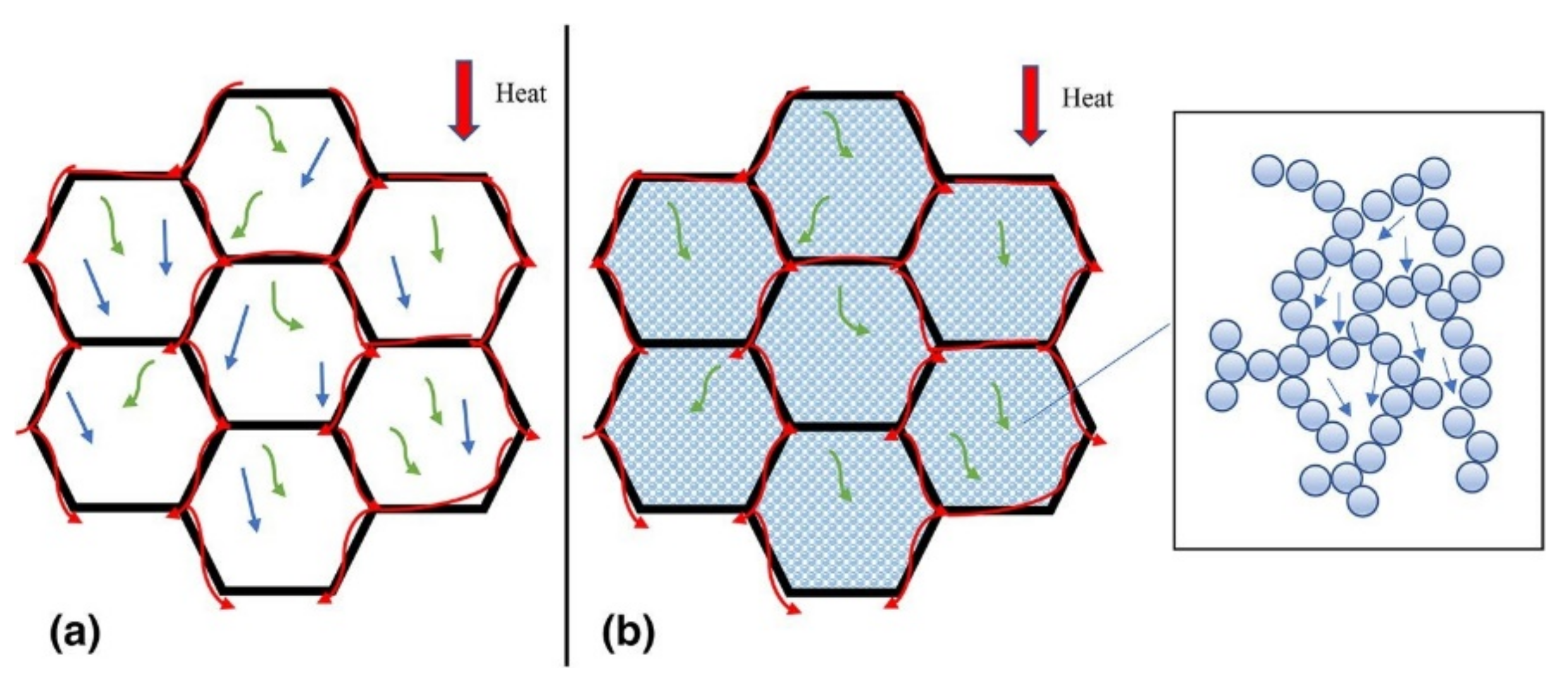
3.2. Insulation Materials
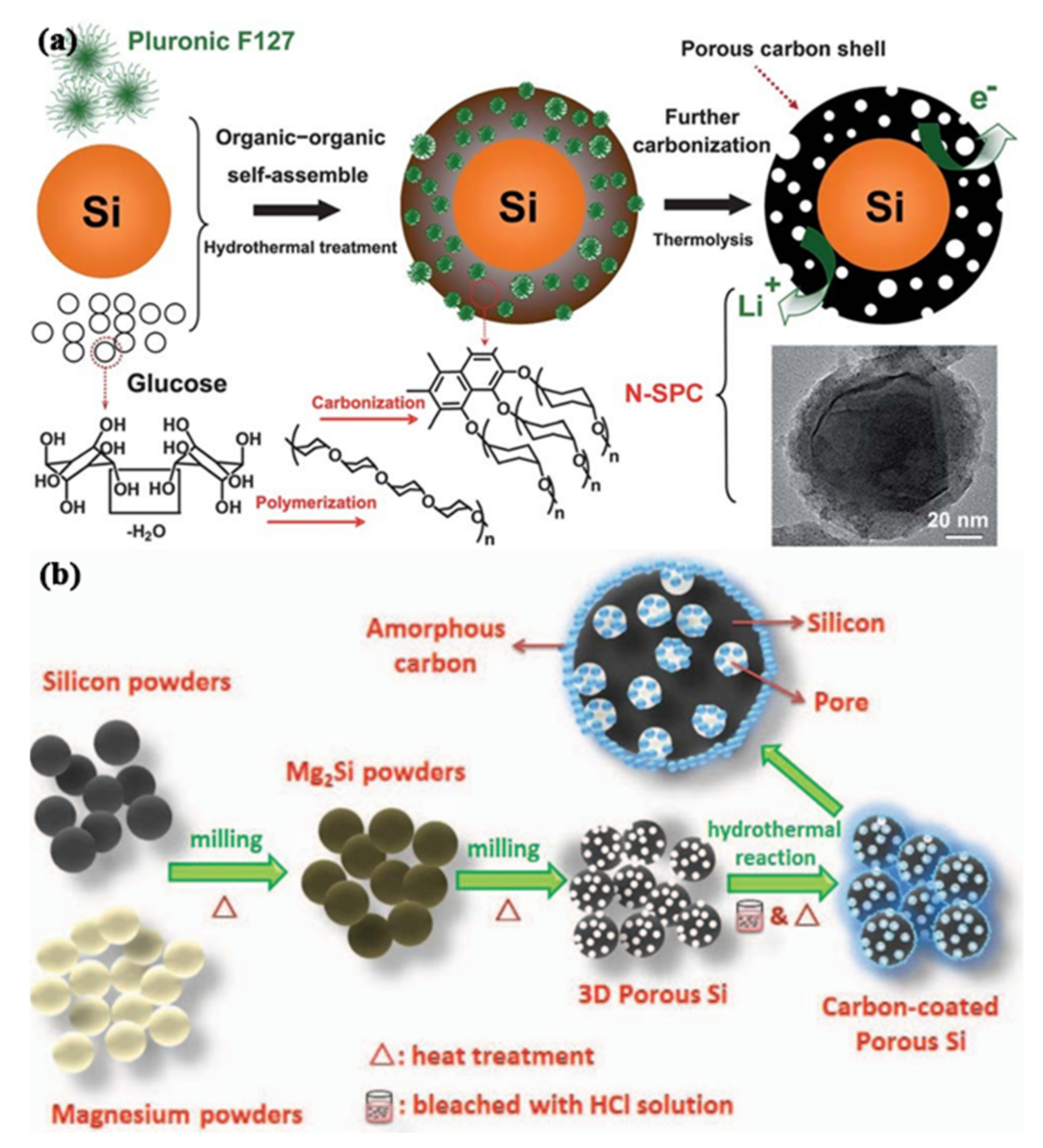
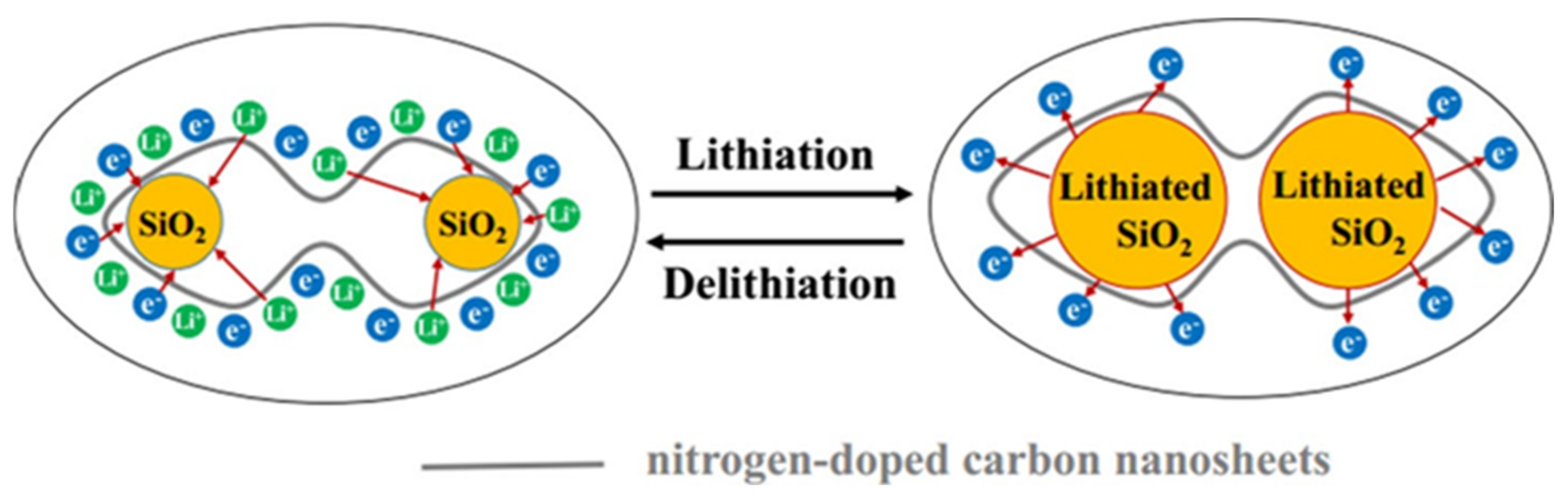
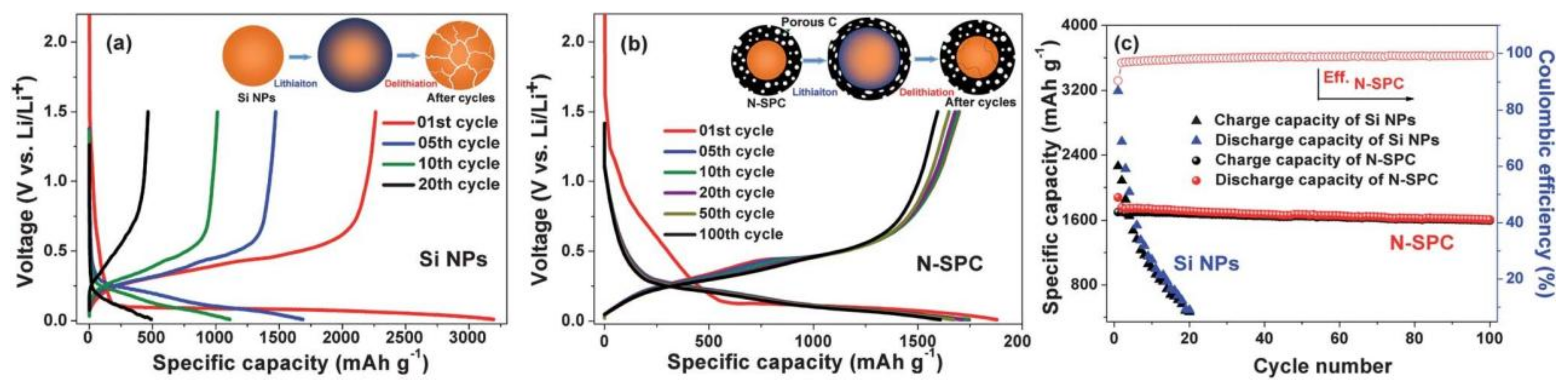
3.3. Lithium-Ion Battery Anode Material
3.4. Biosensors
4. Conclusions and Perspective
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Zaporotskova, I.V.; Boroznina, N.P.; Parkhomenko, Y.N.; Kozhitov, L.V. Carbon nanotubes: Sensor properties. A review. Mod. Electron. Mater. 2016, 2, 95–105. [Google Scholar] [CrossRef]
- Moreno-Castilla, C. Adsorption of organic molecules from aqueous solutions on carbon materials. Carbon 2004, 42, 83–94. [Google Scholar] [CrossRef] [Green Version]
- He, L.; Weniger, F.; Neumann, H.; Beller, M. Synthesis, Characterization, and Application of Metal Nanoparticles Supported on Nitrogen-Doped Carbon: Catalysis beyond Electrochemistry. Angew. Chem. Int. Ed. 2016, 55, 12582–12594. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Liao, J.; Yu, L.; Lv, R.; Li, P.; Sun, W.; Tan, R.; Duan, X.; Zhang, L.; Li, F.; et al. 2020 Roadmap on Carbon Materials for Energy Storage and Conversion. Chem.-Asian J. 2020, 15, 995–1013. [Google Scholar] [CrossRef] [PubMed]
- Zhai, Y.; Dou, Y.; Zhao, D.; Fulvio, P.F.; Mayes, R.; Dai, S. Carbon Materials for Chemical Capacitive Energy Storage. Adv. Mater. 2011, 23, 4828–4850. [Google Scholar] [CrossRef] [PubMed]
- Titirici, M.-M.; White, R.J.; Brun, N.; Budarin, V.L.; Su, D.S.; del Monte, F.; Clark, J.H.; MacLachlan, M.J. Sustainable carbon materials. Chem. Soc. Rev. 2014, 44, 250–290. [Google Scholar] [CrossRef]
- Danish, M.; Ahmad, T. A review on utilization of wood biomass as a sustainable precursor for activated carbon production and application. Renew. Sustain. Energy Rev. 2018, 87, 1–21. [Google Scholar] [CrossRef]
- Ao, W.; Fu, J.; Mao, X.; Kang, Q.; Ran, C.; Liu, Y.; Zhang, H.; Gao, Z.; Li, J.; Liu, G.; et al. Microwave assisted preparation of activated carbon from biomass: A review. Renew. Sustain. Energy Rev. 2018, 92, 958–979. [Google Scholar] [CrossRef]
- Reza, M.S.; Yun, C.S.; Afroze, S.; Radenahmad, N.; Abu Bakar, M.S.; Saidur, R.; Taweekun, J.; Azad, A.K. Preparation of activated carbon from biomass and its’ applications in water and gas purification, a review. Arab J. Basic Appl. Sci. 2020, 27, 208–238. [Google Scholar] [CrossRef]
- Fiyadh, S.S.; AlSaadi, M.A.; Jaafar, W.Z.; AlOmar, M.K.; Fayaed, S.S.; Mohd, N.S.; Hin, L.S.; El-Shafie, A. Review on heavy metal adsorption processes by carbon nanotubes. J. Clean. Prod. 2019, 230, 783–793. [Google Scholar] [CrossRef]
- Kinloch, I.A.; Suhr, J.; Lou, J.; Young, R.J.; Ajayan, P.M. Composites with carbon nanotubes and graphene: An outlook. Science 2018, 362, 547–553. [Google Scholar] [CrossRef] [Green Version]
- Norizan, M.N.; Moklis, M.H.; Demon, S.Z.N.; Halim, N.A.; Samsuri, A.; Mohamad, I.S.; Knight, V.F.; Abdullah, N. Carbon nanotubes: Functionalisation and their application in chemical sensors. RSC Adv. 2020, 10, 43704–43732. [Google Scholar] [CrossRef]
- Tiwari, S.K.; Sahoo, S.; Wang, N.; Huczko, A. Graphene research and their outputs: Status and prospect. J. Sci. Adv. Mater. Devices 2020, 5, 10–29. [Google Scholar] [CrossRef]
- Mohan, V.B.; Lau, K.-T.; Hui, D.; Bhattacharyya, D. Graphene-based materials and their composites: A review on production, applications and product limitations. Compos. Part B Eng. 2018, 142, 200–220. [Google Scholar] [CrossRef]
- Yu, W.; Sisi, L.; Haiyan, Y.; Jie, L. Progress in the functional modification of graphene/graphene oxide: A review. RSC Adv. 2020, 10, 15328–15345. [Google Scholar] [CrossRef]
- Yang, G.; Li, L.; Lee, W.B.; Cheung, N.M. Structure of graphene and its disorders: A review. Sci. Technol. Adv. Mater. 2018, 19, 613–648. [Google Scholar] [CrossRef] [Green Version]
- Huang, H.; Shi, H.; Das, P.; Qin, J.; Li, Y.; Wang, X.; Su, F.; Wen, P.; Li, S.; Lu, P.; et al. The Chemistry and Promising Applications of Graphene and Porous Graphene Materials. Adv. Funct. Mater. 2020, 30, 1909035. [Google Scholar] [CrossRef]
- Gu, W.; Yushin, G. Review of nanostructured carbon materials for electrochemical capacitor applications: Advantages and limitations of activated carbon, carbide-derived carbon, zeolite-templated carbon, carbon aerogels, carbon nanotubes, onion-like carbon, and graphene. WIREs Energy Environ. 2013, 3, 424–473. [Google Scholar] [CrossRef]
- Allen, S.J.; Whitten, L.; McKay, G. The Production and Characterisation of Activated Carbons: A Review. Dev. Chem. Eng. Miner. Process. 2008, 6, 231–261. [Google Scholar] [CrossRef]
- Yuen, F.K.; Hameed, B. Recent developments in the preparation and regeneration of activated carbons by microwaves. Adv. Colloid Interface Sci. 2009, 149, 19–27. [Google Scholar] [CrossRef]
- Xiang, Y.; Xu, Z.; Wei, Y.; Zhou, Y.; Yang, X.; Yang, Y.; Yang, J.; Zhang, J.; Luo, L.; Zhou, Z. Carbon-based materials as adsorbent for antibiotics removal: Mechanisms and influencing factors. J. Environ. Manag. 2019, 237, 128–138. [Google Scholar] [CrossRef]
- Pallarés, J.; González-Cencerrado, A.; Arauzo, I. Production and characterization of activated carbon from barley straw by physical activation with carbon dioxide and steam. Biomass Bioenergy 2018, 115, 64–73. [Google Scholar] [CrossRef] [Green Version]
- Baughman, R.H.; Zakhidov, A.A.; de Heer, W.A. Carbon Nanotubes—The Route Toward Applications. Science 2002, 297, 787–792. [Google Scholar] [CrossRef] [Green Version]
- De Volder, M.F.; Tawfick, S.H.; Baughman, R.H.; Hart, A.J. Carbon nanotubes: Present and future commercial applications. Science 2013, 339, 535–539. [Google Scholar] [CrossRef] [Green Version]
- Dai, H. Carbon nanotubes: Opportunities and challenges. Surf. Sci. 2002, 500, 218–241. [Google Scholar] [CrossRef]
- Geim, A.K.; Novoselov, K.S. The rise of graphene. In Nanoscience and Technology; World Scientific: Singapore, 2009; pp. 11–19. [Google Scholar]
- Li, X.; Yu, J.; Swelm, W.; Al-Ghamdi, A.A.; Xie, J. Graphene in Photocatalysis: A Review. Small 2016, 12, 6640–6696. [Google Scholar] [CrossRef]
- Berry, V. Impermeability of graphene and its applications. Carbon 2013, 62, 1–10. [Google Scholar] [CrossRef]
- Geim, A.K. Graphene: Status and prospects. Science 2009, 324, 1530–1534. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, X.; Qi, X.; Boey, F.; Zhang, H. Graphene-based composites. Chem. Soc. Rev. 2012, 41, 666–686. [Google Scholar] [CrossRef] [PubMed]
- Carlisle, E.M. Silicon. In Biochemistry of the Essential Ultratrace Elements; Frieden, E., Ed.; Biochemistry of the Elements; Springer: Boston, MA, USA, 1984; Volume 3. [Google Scholar] [CrossRef]
- Zougagh, M.; De Torres, A.G. Chelating sorbents based on silica gel and their application in atomic spectrometry. Anal. Bioanal. Chem. 2005, 381, 1103–1113. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, A.K.; Laya, M.S.; Vera, W.J. Silica gel in organic synthesis. Russ. Chem. Rev. 2001, 70, 971–990. [Google Scholar] [CrossRef]
- Narayan, R.; Nayak, U.Y.; Raichur, A.M.; Garg, S. Mesoporous Silica Nanoparticles: A Comprehensive Review on Synthesis and Recent Advances. Pharmaceutics 2018, 10, 118. [Google Scholar] [CrossRef] [Green Version]
- Vinu, A.; Hossain, K.Z.; Ariga, K. Recent Advances in Functionalization of Mesoporous Silica. J. Nanosci. Nanotechnol. 2005, 5, 347–371. [Google Scholar] [CrossRef]
- Kerstens, D.; Smeyers, B.; Van Waeyenberg, J.; Zhang, Q.; Yu, J.; Sels, B.F. State of the Art and Perspectives of Hierarchical Zeolites: Practical Overview of Synthesis Methods and Use in Catalysis. Adv. Mater. 2020, 32, 2004690. [Google Scholar] [CrossRef]
- Hu, G.; Yang, J.; Duan, X.; Farnood, R.; Yang, C.; Yang, J.; Liu, W.; Liu, Q. Recent developments and challenges in zeolite-based composite photocatalysts for environmental applications. Chem. Eng. J. 2021, 417, 129209. [Google Scholar] [CrossRef]
- Leboda, R.; Mendyk, E.; Gierak, A.; Tertykh, V.A. Hydrothermal modification of silica gels (xerogels). Colloids Surf. 1995, 105, 181–189. [Google Scholar] [CrossRef]
- Zhao, T.; Chen, L.; Lin, R.; Zhang, P.; Lan, K.; Zhang, W.; Li, X.; Zhao, D. Interfacial Assembly Directed Unique Mesoporous Architectures: From Symmetric to Asymmetric. Acc. Mater. Res. 2020, 1, 100–114. [Google Scholar] [CrossRef]
- Cecilia, J.A.; García-Sancho, C.; Jiménez-Gómez, C.P.; Moreno-Tost, R.; Maireles-Torres, P. Porous Silicon-Based Catalysts for the Dehydration of Glycerol to High Value-Added Products. Materials 2018, 11, 1569. [Google Scholar] [CrossRef] [Green Version]
- Deneyer, A.; Ke, Q.; Devos, J.; Dusselier, M. Zeolite Synthesis under Nonconventional Conditions: Reagents, Reactors, and Modi Operandi. Chem. Mater. 2020, 32, 4884–4919. [Google Scholar] [CrossRef]
- Dusselier, M.; Davis, M.E. Small-Pore Zeolites: Synthesis and Catalysis. Chem. Rev. 2018, 118, 5265–5329. [Google Scholar] [CrossRef]
- Qin, Z.; Cychosz, K.A.; Melinte, G.; El Siblani, H.; Gilson, J.-P.; Thommes, M.; Fernandez, C.; Mintova, S.; Ersen, O.; Valtchev, V. Opening the Cages of Faujasite-Type Zeolite. J. Am. Chem. Soc. 2017, 139, 17273–17276. [Google Scholar] [CrossRef]
- Qin, Z.; Melinte, G.; Gilson, J.-P.; Jaber, M.; Bozhilov, K.; Boullay, P.; Mintova, S.; Ersen, O.; Valtchev, V. The Mosaic Structure of Zeolite Crystals. Angew. Chem. Int. Ed. 2016, 55, 15049–15052. [Google Scholar] [CrossRef]
- Verboekend, D.; Nuttens, N.; Locus, R.; Van Aelst, J.; Verolme, P.; Groen, J.C.; Pérez-Ramírez, J.; Sels, B.F. Synthesis, characterisation, and catalytic evaluation of hierarchical faujasite zeolites: Milestones, challenges, and future directions. Chem. Soc. Rev. 2015, 45, 3331–3352. [Google Scholar] [CrossRef] [Green Version]
- Verboekend, D.; Vilé, G.; Pérez-Ramírez, J. Hierarchical Y and USY Zeolites Designed by Post-Synthetic Strategies. Adv. Funct. Mater. 2011, 22, 916–928. [Google Scholar] [CrossRef]
- Lin, C.C.H.; Dambrowitz, K.A.; Kuznicki, S.M. Evolving applications of zeolite molecular sieves. Can. J. Chem. Eng. 2011, 90, 207–216. [Google Scholar] [CrossRef]
- Hasan, Z.; Jhung, S.H. Removal of hazardous organics from water using metal-organic frameworks (MOFs): Plausible mechanisms for selective adsorptions. J. Hazard. Mater. 2015, 283, 329–339. [Google Scholar] [CrossRef]
- Liu, X.-W.; Sun, T.-J.; Hu, J.-L.; Wang, S.-D. Composites of metal–organic frameworks and carbon-based materials: Preparations, functionalities and applications. J. Mater. Chem. A 2016, 4, 3584–3616. [Google Scholar] [CrossRef]
- Hardman, J.S.; Street, P.J.; Twamley, C.S. Studies of spontaneous combustion in beds of activated carbon. Fuel 1980, 59, 151–156. [Google Scholar] [CrossRef]
- Abdulsalam, J.; Onifade, M.; Bada, S.; Mulopo, J.; Genc, B. The Spontaneous Combustion of Chemically Activated Carbons from South African Coal Waste. Combust. Sci. Technol. 2020, 1–17. [Google Scholar] [CrossRef]
- Li, X.; Wang, B.; Cao, Y.; Zhao, S.; Wang, H.; Feng, X.; Zhou, J.; Ma, X. Water Contaminant Elimination Based on Metal–Organic Frameworks and Perspective on Their Industrial Applications. ACS Sustain. Chem. Eng. 2019, 7, 4548–4563. [Google Scholar] [CrossRef]
- Zhao, R.; Shi, X.; Ma, T.; Rong, H.; Wang, Z.; Cui, F.; Zhu, G.; Wang, C. Constructing Mesoporous Adsorption Channels and MOF–Polymer Interfaces in Electrospun Composite Fibers for Effective Removal of Emerging Organic Contaminants. ACS Appl. Mater. Interfaces 2020, 13, 755–764. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Huang, R.; Zou, J.; Liao, D.; Yu, J.; Jiang, X. A sensitive sensor based on MOFs derived nanoporous carbons for electrochemical detection of 4-aminophenol. Ecotoxicol. Environ. Saf. 2020, 191, 110194. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Liu, Z.; Jiang, S.; Hou, H. Carbonization: A feasible route for reutilization of plastic wastes. Sci. Total Environ. 2019, 710, 136250. [Google Scholar] [CrossRef] [PubMed]
- Nizamuddin, S.; Siddiqui, M.; Mubarak, N.; Baloch, H.A.; Mazari, S.; Tunio, M.; Griffin, G.; Srinivasan, M.; Tanksale, A.; Riaz, S. Advanced Nanomaterials Synthesis from Pyrolysis and Hydrothermal Carbonization: A Review. Curr. Org. Chem. 2018, 22, 446–461. [Google Scholar] [CrossRef]
- Libra, A.J.; Ro, K.S.; Kammann, C.; Funke, A.; Berge, N.D.; Neubauer, Y.; Titirici, M.-M.; Fühner, C.; Bens, O.; Kern, J.; et al. Hydrothermal carbonization of biomass residuals: A comparative review of the chemistry, processes and applications of wet and dry pyrolysis. Biofuels 2011, 2, 71–106. [Google Scholar] [CrossRef] [Green Version]
- Inagaki, M.; Park, K.C.; Endo, M. Carbonization under pressure. New Carbon Mater. 2010, 25, 409–420. [Google Scholar] [CrossRef]
- Alotaibi, N.; Hammud, H.H.; Karnati, R.K.; Hussain, S.G.; Mazher, J.; Prakasam, T. Cobalt–carbon/silica nanocomposites prepared by pyrolysis of a cobalt 2,2′-bipyridine terephthalate complex for remediation of cationic dyes. RSC Adv. 2020, 10, 17660–17672. [Google Scholar] [CrossRef]
- Song, L.; Feng, D.; Campbell, C.G.; Gu, D.; Forster, A.M.; Yager, K.G.; Fredin, N.; Lee, H.-J.; Jones, R.L.; Zhao, D.; et al. Robust conductive mesoporous carbon-silica composite films with highly ordered and oriented orthorhombic structures from triblock-copolymer template co-assembly. J. Mater. Chem. 2010, 20, 1691–1701. [Google Scholar] [CrossRef]
- Sotiriou, K.; Supanchaiyamat, N.; Jiang, T.; Janekarn, I.; García, A.M.; Budarin, V.L.; MacQuarrie, D.J.; Hunt, A.J. Synthesis and application of tuneable carbon-silica composites from the microwave pyrolysis of waste paper for selective recovery of gold from acidic solutions. RSC Adv. 2020, 10, 25228–25238. [Google Scholar] [CrossRef]
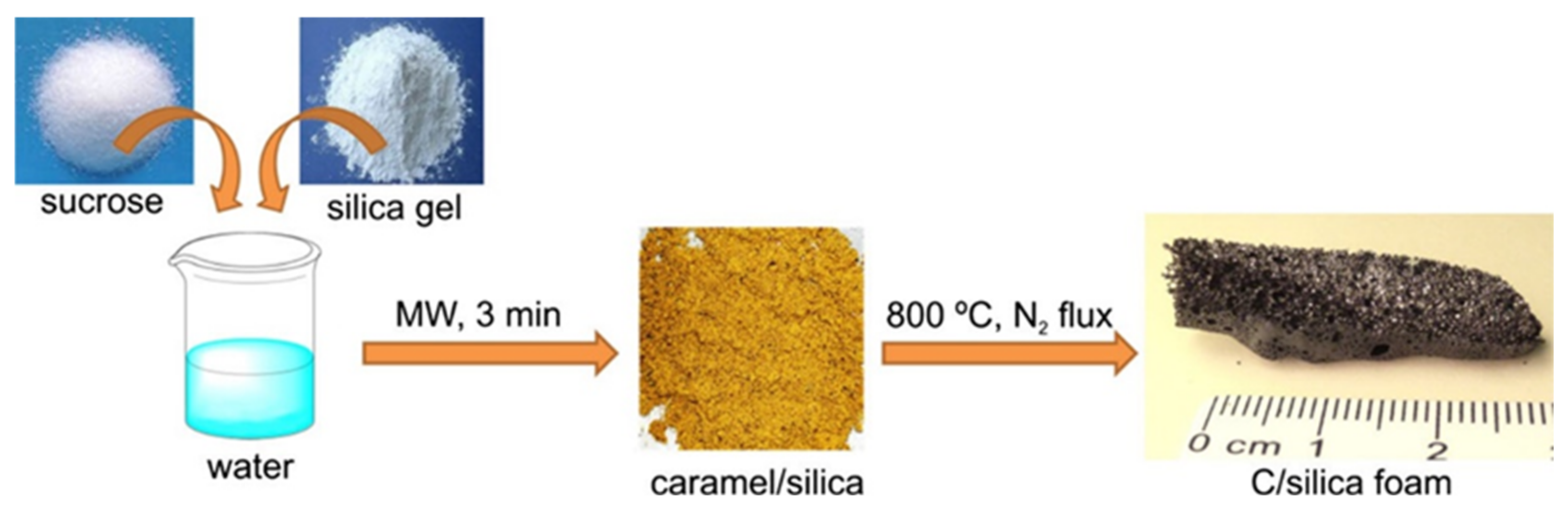
- Canencia, F.; Darder, M.; Aranda, P.; Fernandes, F.; Gouveia, R.; Ruiz-Hitzky, E. Conducting macroporous carbon foams derived from microwave-generated caramel/silica gel intermediates. J. Mater. Sci. 2017, 52, 11269–11281. [Google Scholar] [CrossRef]
- Tomaszewski, W.; Gun’Ko, V.; Skubiszewska-Zięba, J.; Charmas, B.; Leboda, R. Influence of carbon deposits and subsequent silylation of silica gel on sorption efficiency of explosive nitramines. Colloids Surf. A Physicochem. Eng. Asp. 2014, 468, 76–86. [Google Scholar] [CrossRef]
- Zhang, S.; Gao, Y.; Dan, H.; Xu, X.; Yue, Q.; Yan, J.; Wang, W.; Gao, B. Effect of washing conditions on adsorptive properties of mesoporous silica carbon composites by in-situ carbothermal treatment. Sci. Total Environ. 2020, 716, 136770. [Google Scholar] [CrossRef]
- Mao, J.; Chen, M.; Deng, Y.; Liu, H.; Ju, Z.; Xing, Z.; Cao, X. Synthesis of uniform silica nanospheres wrapped in nitrogen-doped carbon nanosheets with stable lithium-ion storage properties. J. Mater. Sci. 2019, 54, 12767–12781. [Google Scholar] [CrossRef]
- Han, Z.; Kong, S.; Sui, H.; Li, X.; Zhang, Z. Preparation of Carbon-Silicon Doping Composite Adsorbent Material for Removal of VOCs. Materials 2019, 12, 2438. [Google Scholar] [CrossRef] [Green Version]
- Wang, K.; Pei, S.; He, Z.; Huang, L.-A.; Zhu, S.; Guo, J.; Shao, H.; Wang, J. Synthesis of a novel porous silicon microsphere@carbon core-shell composite via in situ MOF coating for lithium ion battery anodes. Chem. Eng. J. 2019, 356, 272–281. [Google Scholar] [CrossRef]
- Wang, T.; Zhai, Y.; Zhu, Y.; Li, C.; Zeng, G. A review of the hydrothermal carbonization of biomass waste for hydrochar formation: Process conditions, fundamentals, and physicochemical properties. Renew. Sustain. Energy Rev. 2018, 90, 223–247. [Google Scholar] [CrossRef]
- Nizamuddin, S.; Baloch, H.A.; Griffin, G.; Mubarak, N.; Bhutto, A.W.; Abro, R.; Mazari, S.; Ali, B.S. An overview of effect of process parameters on hydrothermal carbonization of biomass. Renew. Sustain. Energy Rev. 2017, 73, 1289–1299. [Google Scholar] [CrossRef]
- Khan, T.A.; Saud, A.S.; Jamari, S.S.; Ab Rahim, M.H.; Park, J.-W.; Kim, H.-J. Hydrothermal carbonization of lignocellulosic biomass for carbon rich material preparation: A review. Biomass Bioenergy 2019, 130, 105384. [Google Scholar] [CrossRef]
- Shen, Y. A review on hydrothermal carbonization of biomass and plastic wastes to energy products. Biomass Bioenergy 2020, 134, 105479. [Google Scholar] [CrossRef]
- Titirici, M.-M.; Antonietti, M. Chemistry and materials options of sustainable carbon materials made by hydrothermal carbonization. Chem. Soc. Rev. 2009, 39, 103–116. [Google Scholar] [CrossRef]
- Román, S.; Libra, J.; Berge, N.; Sabio, E.; Ro, K.; Li, L.; Ledesma, B.; Álvarez, A.; Bae, S. Hydrothermal Carbonization: Modeling, Final Properties Design and Applications: A Review. Energies 2018, 11, 216. [Google Scholar] [CrossRef] [Green Version]
- Bie, X.; Xiong, M.; Wang, B.; Dong, Y.; Chen, Z.; Huang, R. Glucose hydrothermal encapsulation of carbonized silicone polyester to prepare anode materials for lithium batteries with improved cycle stability. RSC Adv. 2022, 12, 9238–9248. [Google Scholar] [CrossRef]
- Ruttert, M.; Holtstiege, F.; Hüsker, J.; Börner, M.; Winter, M.; Placke, T. Hydrothermal-derived carbon as a stabilizing matrix for improved cycling performance of silicon-based anodes for lithium-ion full cells. Beilstein J. Nanotechnol. 2018, 9, 2381–2395. [Google Scholar] [CrossRef] [PubMed]
- Lu, X.; He, J.; Xie, J.; Zhou, Y.; Liu, S.; Zhu, Q.; Lu, H. Preparation of hydrophobic hierarchical pore carbon-silica composite and its adsorption performance toward volatile organic compounds. J. Environ. Sci. 2019, 87, 39–48. [Google Scholar] [CrossRef]
- Zhao, X.; Liu, S.; Peng, J.; Li, X.; Niu, H.; Zhang, H.; Wang, L.; Wu, R. Facile one-pot synthesized hydrothermal carbon from cyclodextrin: A stationary phase for hydrophilic interaction liquid chromatography. J. Chromatogr. A 2018, 1585, 144–151. [Google Scholar] [CrossRef] [PubMed]
- Shui, Z.; Yao, L.; Pu, X.; Yang, L.; Jiang, W.; Jiang, X. Synthesis of a Novel Zeolite–Activated Carbon Composite Using Lithium–Silicon-Powder Waste for Ammonia-Nitrogen and Methylene Blue Removal. Ind. Eng. Chem. Res. 2020, 59, 14616–14624. [Google Scholar] [CrossRef]
- Sun, X.; Yu, W.; Yan, J.; Li, J.; Jin, G.; Feng, J.; Guo, Z.; Liang, X. Mesoporous silica–carbon composites fabricated by a universal strategy of hydrothermal carbonization: Controllable synthesis and applications. RSC Adv. 2018, 8, 27207–27215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Saraji, M.; Jafari, M.T.; Mossaddegh, M. Carbon nanotubes@silicon dioxide nanohybrids coating for solid-phase micro-extraction of organophosphorus pesticides followed by gas chromatography-corona discharge ion mobility spectrometric detection. J. Chromatogr. A 2016, 1429, 30–39. [Google Scholar] [CrossRef] [PubMed]
- Qian, H.; Li, W.; Wang, X.; Xie, F.; Li, W.; Qu, Q. Simultaneous growth of graphene/mesoporous silica composites using liquid precursor for HPLC separations. Appl. Surf. Sci. 2020, 537, 148101. [Google Scholar] [CrossRef]
- Shao, D.; Tang, D.; Mai, Y.; Zhang, L. Nanostructured silicon/porous carbon spherical composite as a high capacity anode for Li-ion batteries. J. Mater. Chem. A 2013, 1, 15068–15075. [Google Scholar] [CrossRef]
- Yang, X.; Shi, C.; Zhang, L.; Liang, G.; Ni, S.; Wen, Z. Preparation of Three Dimensional Porous Silicon with Fluoride-Free Method and Its Application in Lithium Ion Batteries. ECS Solid State Lett. 2013, 2, M53–M56. [Google Scholar] [CrossRef]
- Tseng, T.K.; Lin, Y.S.; Chen, Y.J.; Chu, H. A Review of Photocatalysts Prepared by Sol-Gel Method for VOCs Removal. Int. J. Mol. Sci. 2010, 11, 2336–2361. [Google Scholar] [CrossRef]
- Parashar, M.; Shukla, V.K.; Singh, R. Metal oxides nanoparticles via sol–gel method: A review on synthesis, characterization and applications. J. Mater. Sci. Mater. Electron. 2020, 31, 3729–3749. [Google Scholar] [CrossRef]
- Amiri, S.; Rahimi, A. Hybrid nanocomposite coating by sol–gel method: A review. Iran. Polym. J. 2016, 25, 559–577. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Ishizaka, T.; Kurokawa, Y. KOBAYASHI (2005) Preparation of alumina films by the sol-gel method.pdf. J. Mater. Sci. 2005, 40, 263–283. [Google Scholar] [CrossRef]
- Abdollahi, A.; Roghani-Mamaqani, H.; Salami-Kalajahi, M.; Mousavi, A.; Razavi, B.; Shahi, S. Preparation of organic-inorganic hybrid nanocomposites from chemically modified epoxy and novolac resins and silica-attached carbon nanotubes by sol-gel process: Investigation of thermal degradation and stability. Prog. Org. Coat. 2018, 117, 154–165. [Google Scholar] [CrossRef]
- Fu, L.; Zhu, J.; Huang, W.; Fang, J.; Sun, X.; Wang, X.; Liao, K. Preparation of Nano-Porous Carbon-Silica Composites and Its Adsorption Capacity to Volatile Organic Compounds. Processes 2020, 8, 372. [Google Scholar] [CrossRef] [Green Version]
- Piñero, M.; Mesa-Díaz, M.D.M.; Santos, D.d.L.; Reyes-Peces, M.V.; Díaz-Fraile, J.A.; de la Rosa-Fox, N.; Esquivias, L.; Morales-Florez, V. Reinforced silica-carbon nanotube monolithic aerogels synthesised by rapid controlled gelation. J. Sol-Gel Sci. Technol. 2018, 86, 391–399. [Google Scholar] [CrossRef]
- Yang, W.-J.; Yuen, A.C.Y.; Li, A.; Lin, B.; Chen, T.B.Y.; Yang, W.; Lu, H.-D.; Yeoh, G.H. Recent progress in bio-based aerogel absorbents for oil/water separation. Cellulose 2019, 26, 6449–6476. [Google Scholar] [CrossRef]
- Du, A.; Zhou, B.; Zhang, Z.; Shen, J. A Special Material or a New State of Matter: A Review and Reconsideration of the Aerogel. Materials 2013, 6, 941–968. [Google Scholar] [CrossRef] [Green Version]
- Lamy-Mendes, A.; Silva, R.F.; Durães, L. Advances in carbon nanostructure–silica aerogel composites: A review. J. Mater. Chem. A 2017, 6, 1340–1369. [Google Scholar] [CrossRef]
- Liu, H.; Li, T.; Shi, Y.; Zhao, X. Thermal Insulation Composite Prepared from Carbon Foam and Silica Aerogel Under Ambient Pressure. J. Mater. Eng. Perform. 2015, 24, 4054–4059. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, Z.; Zhang, J.; Ai, S.; Tang, H. Ultralight and thermal insulation carbon foam/SiO2 aerogel composites. J. Porous Mater. 2019, 26, 1305–1312. [Google Scholar] [CrossRef]
- Lee, K.Y.; Nah, H.Y.; Choi, H.; Parale, V.G.; Park, H.H. Methyltrimethoxysilane silica aerogel composite with carboxyl-functionalised multi-wall carbon nanotubes. Int. J. Nanotechnol. 2018, 15, 587–597. [Google Scholar] [CrossRef]
- Ślosarczyk, A.; Wojciech, S.; Piotr, Z.; Paulina, J. Synthesis and characterization of carbon fiber/silica aerogel nanocomposites. J. Non-Cryst. Solids 2015, 416, 1–3. [Google Scholar] [CrossRef]
- Xie, H.; Ye, Q.; Liu, J.; Zhou, G.; Liu, W.; Lu, H.; Yang, W. Study on thermal properties of poly(vinyl alcohol) and polyurethane composites with multiwalled carbon nanotubes/silica nanohybrids. J. Thermoplast. Compos. Mater. 2016, 30, 404–415. [Google Scholar] [CrossRef]
- Menshutina, N.; Tsygankov, P.; Ivanov, S. Synthesis and Properties of Silica and Alginate Hybrid Aerogel Particles with Embedded Carbon Nanotubes (CNTs) for Selective Sorption. Materials 2018, 12, 52. [Google Scholar] [CrossRef] [Green Version]
- Carbonaro, C.M.; Thakkar, S.V.; Ludmerczki, R.; Olla, C.; Pinna, A.; Loche, D.; Malfatti, L.; Marincola, F.C.; Casula, M.F. How porosity affects the emission of fluorescent carbon dot-silica porous composites. Microporous Mesoporous Mater. 2020, 305, 110302. [Google Scholar] [CrossRef]
- Vassilakopoulou, A.; Georgakilas, V.; Koutselas, I. Encapsulation and protection of carbon dots within MCM-41 material. J. Sol-Gel Sci. Technol. 2017, 82, 795–800. [Google Scholar] [CrossRef]
- Haldoupis, E.; Nair, S.; Sholl, D.S. Efficient Calculation of Diffusion Limitations in Metal Organic Framework Materials: A Tool for Identifying Materials for Kinetic Separations. J. Am. Chem. Soc. 2010, 132, 7528–7539. [Google Scholar] [CrossRef]
- Choi, S.; Drese, J.H.; Jones, C.W. Adsorbent Materials for Carbon Dioxide Capture from Large Anthropogenic Point Sources. Chem. Sus. Energy Chem. 2009, 2, 796–854. [Google Scholar] [CrossRef] [PubMed]
- Kyzas, G.Z.; Deliyanni, E.A.; Matis, K.A. Graphene oxide and its application as an adsorbent for wastewater treatment. J. Chem. Technol. Biotechnol. 2013, 89, 196–205. [Google Scholar] [CrossRef]
- Liu, W.; Li, Z.; Zhang, S.-P.; Jian, W.-W.; Ma, D.-Z. Adsorption performance of multi-walled carbon nanotube-SiO2 adsorbent for toluene. J. Fuel Chem. Technol. 2021, 49, 861–872. [Google Scholar] [CrossRef]
- Gan, F.; Wang, B.; Jin, Z.; Xie, L.; Dai, Z.; Zhou, T.; Jiang, X. From typical silicon-rich biomass to porous carbon-zeolite composite: A sustainable approach for efficient adsorption of CO2. Sci. Total Environ. 2021, 768, 144529. [Google Scholar] [CrossRef]
- Qu, G.; Han, Y.; Qi, J.; Xing, X.; Hou, M.; Sun, Y.; Wang, X.; Sun, G. Rapid iodine capture from radioactive wastewater by green and low-cost biomass waste derived porous silicon–carbon composite. RSC Adv. 2021, 11, 5268–5275. [Google Scholar] [CrossRef]
- Kaushika, N.; Sumathy, K. Solar transparent insulation materials: A review. Renew. Sustain. Energy Rev. 2003, 7, 317–351. [Google Scholar] [CrossRef]
- Aditya, L.; Mahlia, T.; Rismanchi, B.; Ng, H.; Hasan, M.; Metselaar, H.; Muraza, O.; Aditiya, H. A review on insulation materials for energy conservation in buildings. Renew. Sustain. Energy Rev. 2017, 73, 1352–1365. [Google Scholar] [CrossRef]
- De las Casas, C.; Li, W. A review of application of carbon nanotubes for lithium ion battery anode material. J. Power Sources 2012, 208, 74–85. [Google Scholar] [CrossRef]
- Kucinskis, G.; Bajars, G.; Kleperis, J. Graphene in lithium ion battery cathode materials: A review. J. Power Sources 2013, 240, 66–79. [Google Scholar] [CrossRef]
- Fergus, J.W. Recent developments in cathode materials for lithium ion batteries. J. Power Sources 2010, 195, 939–954. [Google Scholar] [CrossRef]
- Mu, T.; Zuo, P.; Lou, S.; Pan, Q.; Li, Q.; Du, C.; Gao, Y.; Cheng, X.; Ma, Y.; Yin, G. A two-dimensional nitrogen-rich carbon/silicon composite as high performance anode material for lithium ion batteries. Chem. Eng. J. 2018, 341, 37–46. [Google Scholar] [CrossRef]
- Ma, T.; Xu, H.; Yu, X.; Li, H.; Zhang, W.; Cheng, X.; Zhu, W.; Qiu, X. Lithiation Behavior of Coaxial Hollow Nanocables of Carbon–Silicon Composite. ACS Nano 2019, 13, 2274–2280. [Google Scholar] [CrossRef]
- Kwon, H.J.; Hwang, J.-Y.; Shin, H.-J.; Jeong, M.-G.; Chung, K.Y.; Sun, Y.-K.; Jung, H.-G. Nano/Microstructured Silicon–Carbon Hybrid Composite Particles Fabricated with Corn Starch Biowaste as Anode Materials for Li-Ion Batteries. Nano Lett. 2019, 20, 625–635. [Google Scholar] [CrossRef]
- Yang, X.; Zhang, P.; Wen, Z.; Zhang, L. High performance silicon/carbon composite prepared by in situ carbon-thermal reduction for lithium ion batteries. J. Alloys Compd. 2010, 496, 403–406. [Google Scholar] [CrossRef]
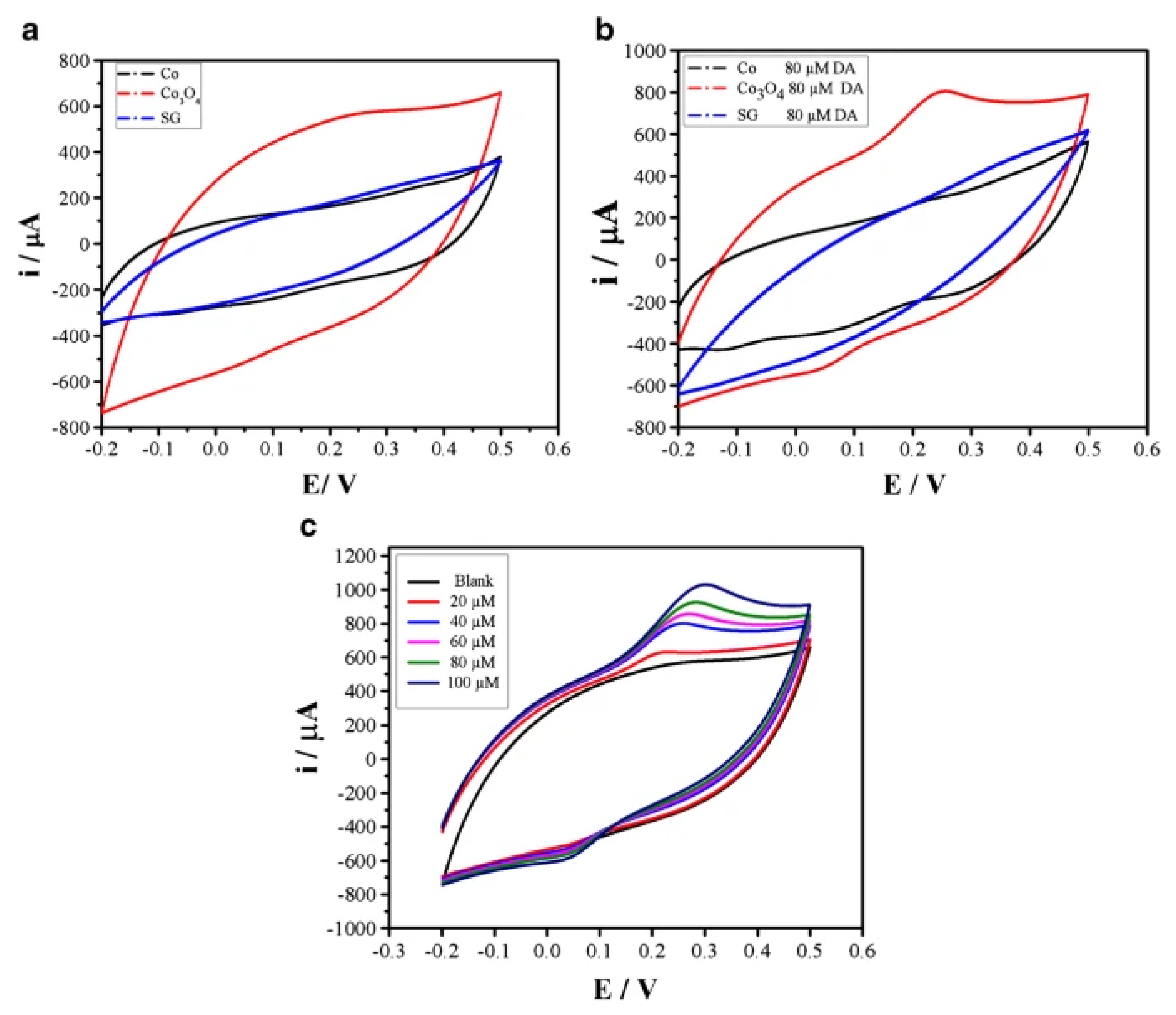
- Younus, A.R.; Iqbal, J.; Muhammad, N.; Rehman, F.; Tariq, M.; Niaz, A.; Badshah, S.; Saleh, T.A.; Rahim, A. Nonenzymatic amperometric dopamine sensor based on a carbon ceramic electrode of type SiO2/C modified with Co3O4 nanoparticles. Mikrochim. Acta 2019, 186, 471. [Google Scholar] [CrossRef]



| Material | Advantages | Disadvantages | Refs. |
|---|---|---|---|
| Activated carbon | Wide range of precursors, high specific surface, low acid-base reactivity | Difficult to regenerate | [7,8,9] |
| Carbon nanotubes | High mechanical strength, excellent electrical and thermal performance | Difficult to disperse in aqueous and organic solvents | [10,11,12] |
| Graphene | Outstanding optical and mechanical properties | Difficult to disperse in aqueous and organic solvents, complicated synthesis | [13,14,15,16,17] |
| Material | Advantages | Disadvantages | Refs. |
|---|---|---|---|
| Silica gel | Excellent mechanical properties, good adsorption capacity | Easily hydrolyzed in alkaline environments, poor adsorption selectivity | [32,33] |
| Mesoporous silica | Well-organized and controllable pores, easy functionalization, high thermal stability | Difficult to synthesize on a large scale | [34,35] |
| Molecular sieve | High specific surface, strong acidity | High energy consumption during desorption, high cost | [36,37] |
| Carbon Source | Silica Source | Pyrolysis Temperature (°C) | Pyrolysis Time (min) | Atmosphere | Benefits/Remarks | Ref. |
|---|---|---|---|---|---|---|
| Co(terephthalate) (2,2′-bipyridine) | Silica | 850 | 600 | Nitrogen | BJH surface area was 55.4 m2/g | [59] |
| Phenolic resin | Tetraethyl orthosilicate | 800 | 240 | Nitrogen | Uniformly ordered mesopores | [60] |
| Bio-oil | Amorphous silica gel | 800 | 10 | Nitrogen | BET surface area was 1056 m2/g, pore diameter was 4.8 nm | [61] |
| Carbon Source | Silica Source | Hydrothermal Temperature (°C) | Hydrothermal Time (min) | Benefits/Remarks | Ref. |
|---|---|---|---|---|---|
| Glucose, graphene oxide | Silicon-containing polyester | 160 | 360 | BET surface area was 12.7 m2/g, thermogravimetric temperature was 550 °C | [74] |
| Glucose | Silicon nanoparticles | 180 | 150 | Thermogravimetric temperature was 525 °C | [75] |
| Sucrose | Mesoporous spherical silica gel | 150 | 1200 | BET surface area was 416.0 m2/g, pore size was 6.64 nm, thermogravimetric temperature was 550 °C | [76] |
| Carbon Source | Silica Source | Catalyst for the Hydrolysis Reaction | Catalyst for the Polycondensation Reaction | Benefits/Remarks | Ref. |
|---|---|---|---|---|---|
| Multi-walled carbon nanotubes | Tetraethyl orthosilicate | Formic acid | Ethylenediamine | Thermogravimetric temperature was 500 °C | [88] |
| Activated carbon powder | Tetraethyl orthosilicate | Hydrochloric acid | Ammonia water | BET surface area was 725.0 m2/g, average pore size was 3.5 nm | [89] |
| Multi-walled carbon nanotubes | Tetraethyl orthosilicate | Nitric acid | Ammonium hydroxide | BET surface area was 650.0 m2/g, pore size was 17 nm | [90] |
| Carbon Source | Silica Source | Adsorbate | Adsorption Capacity | Benefits/Remarks | Ref. |
|---|---|---|---|---|---|
| Multi-walled carbon nanotubes | Tetraethyl silicate | Toluene | 50.3 mg/g | BET surface area was 964.1 m2/g, micropore surface area was 465.3 m2/g, average pore diameter was 0.492 nm | [105] |
| Rice husk | NaX zeolite | CO2 | 79.6 mg/g | BET surface area was 980.8 m2/g, ultra-micropore volume was 0.181 cm3/g, 99% regeneration efficiency after 10 cycles | [106] |
| Paper mill sludge | Paper mill sludge | Iodine | 248.07 mg/g | BET surface area was 518.6 m2/g, micropore pore volume was 0.109 cm3/g, mean pore diameter was 5.7 nm | [107] |
| Carbon Source | Silica Source | Reversible Capacity after Cycling | Benefits/Remarks | Ref. |
|---|---|---|---|---|
| Citric acid and melamine | Nano-sized silicon powder | 1000 mAh/g after 300 cycles at 2 A/g | BET surface area was 107.0 m2/g, pore size was 4 nm | [113] |
| Multiwall carbon nanotube | Tetraethyl orthosilicate | 1150 mAh/g over 500 cycles | Average coulombic efficiency was up to 99.9% | [114] |
| Corn starch | Si nanoparticles | 1440 mAh/g over 500 cycles | Fast charge and discharge capability of 12 min | [115] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wu, T.; Ke, Q.; Lu, M.; Pan, P.; Zhou, Y.; Gu, Z.; Cui, G.; Lu, H. Recent Advances in Carbon-Silica Composites: Preparation, Properties, and Applications. Catalysts 2022, 12, 573. https://doi.org/10.3390/catal12050573
Wu T, Ke Q, Lu M, Pan P, Zhou Y, Gu Z, Cui G, Lu H. Recent Advances in Carbon-Silica Composites: Preparation, Properties, and Applications. Catalysts. 2022; 12(5):573. https://doi.org/10.3390/catal12050573
Chicago/Turabian StyleWu, Tianhao, Quanli Ke, Mei Lu, Pengyun Pan, Ying Zhou, Zhenyu Gu, Guokai Cui, and Hanfeng Lu. 2022. "Recent Advances in Carbon-Silica Composites: Preparation, Properties, and Applications" Catalysts 12, no. 5: 573. https://doi.org/10.3390/catal12050573
APA StyleWu, T., Ke, Q., Lu, M., Pan, P., Zhou, Y., Gu, Z., Cui, G., & Lu, H. (2022). Recent Advances in Carbon-Silica Composites: Preparation, Properties, and Applications. Catalysts, 12(5), 573. https://doi.org/10.3390/catal12050573








