Platinum-Containing Nanoparticles on N-Doped Carbon Supports as an Advanced Electrocatalyst for the Oxygen Reduction Reaction
Abstract
:1. Introduction
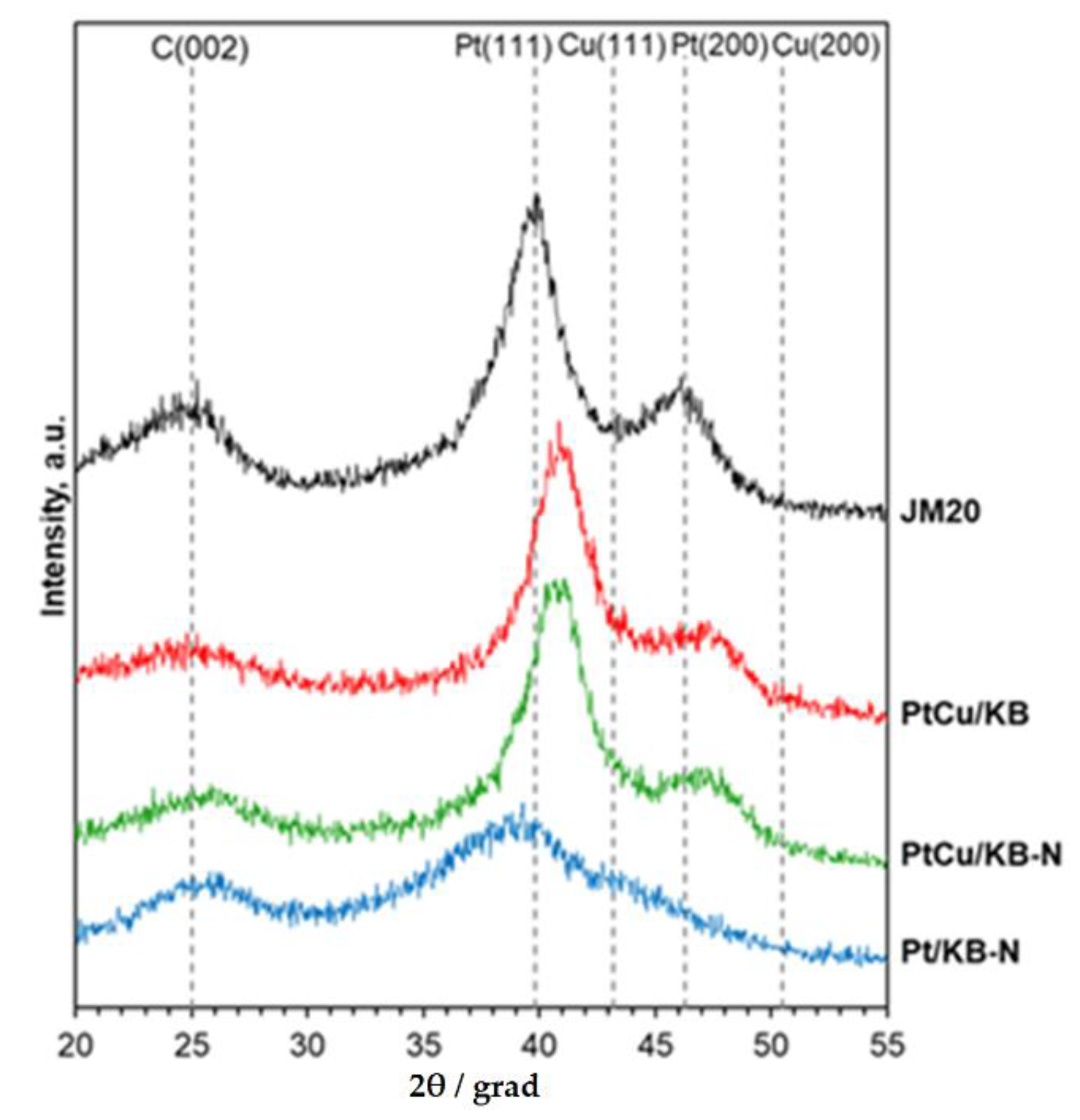
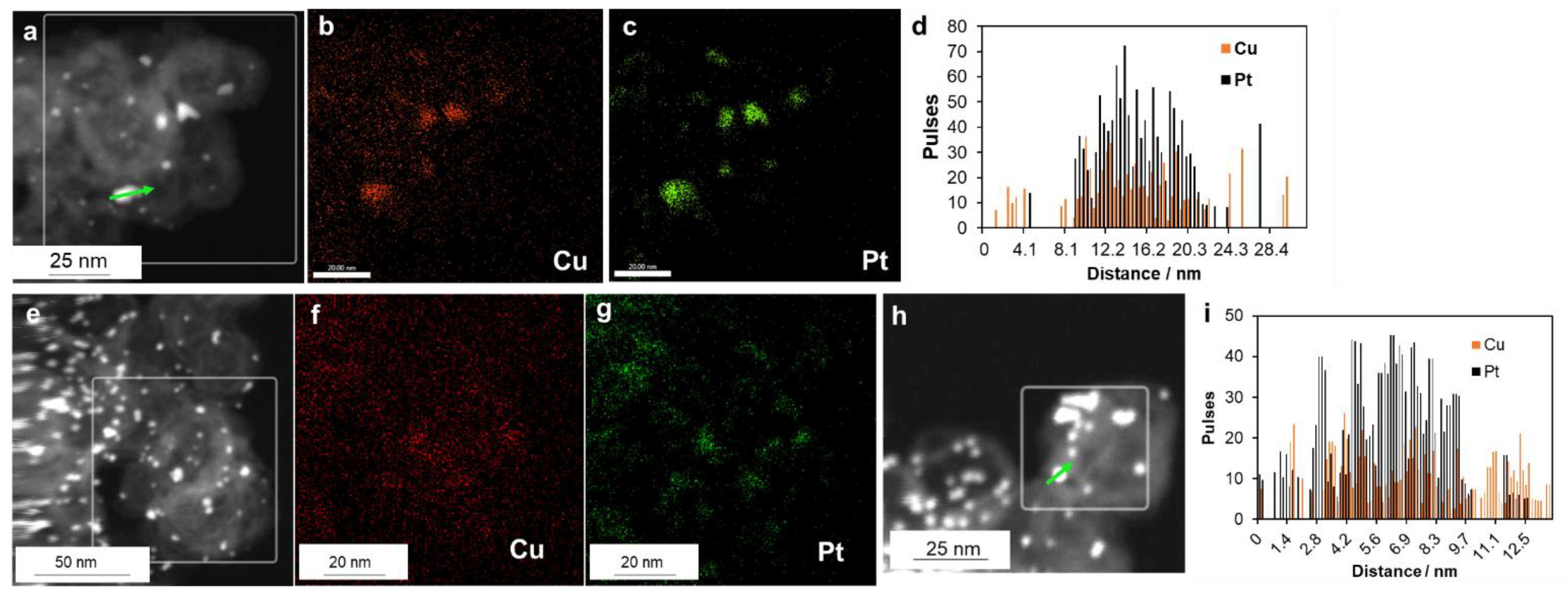
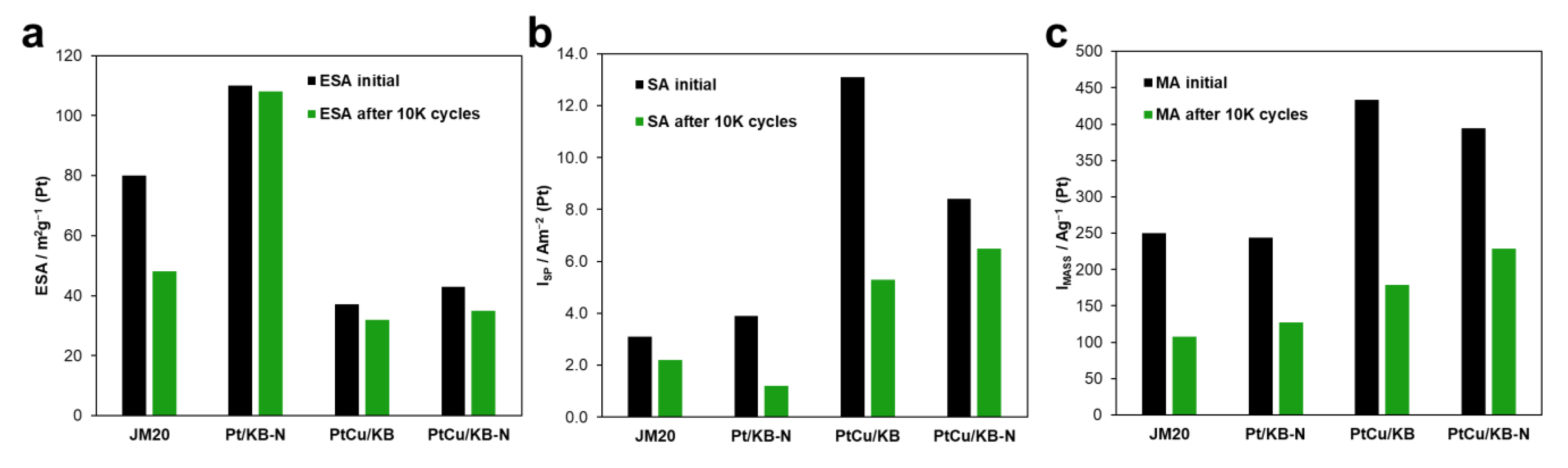
2. Results
3. Materials and Methods
3.1. Synthesis of the Modified Support
3.2. Synthesis of Catalysts
3.3. Attestation of Catalysts’ Structural and Morphological Characteristics
3.4. Attestation of the Electrochemical Characteristics of the Catalysts
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PEMFC | Proton exchange membrane fuel cells |
| NPs | Nanoparticles |
| MEA | Membrane–electrode assembly |
| XRD | X-ray diffraction |
| TEM | Transmission electron microscopy |
| EDX | Energy-dispersive X-ray spectroscopy |
| TGA | Thermogravimetric analysis |
| DSC | Differential scanning calorimetry |
| ESA | Electrometrically active surface area |
| CV | Cyclic voltammetry |
| ORR | Oxygen reduction reaction |
| STEM | Scanning transmission electron microscopy |
| RDE | Rotating disk electrode |
| RHE | Reversible hydrogen electrode |
| XRF | X-ray fluorescence analysis |
References
- Daud, W.R.W.; Rosli, R.E.; Majlan, E.H.; Hamid, S.A.A.; Mohamed, R.; Husaini, T. PEM fuel cell system control: A review. Renew. Energy 2017, 2, 629–638. [Google Scholar] [CrossRef]
- Filippov, S.P.; Yaroslavtsev, A.B. Hydrogen energy: Development prospects and materials. Russ. Chem. Rev. 2021, 90, 627–643. [Google Scholar] [CrossRef]
- Owejan, J.P.; Owejan, J.E.; Gu, W. Impact of Platinum Loading and Catalyst Layer Structure on PEMFC Performance. J. Electrochem. Soc. 2013, 160, F824–F833. [Google Scholar] [CrossRef]
- Yaroslavtsev, A.B.; Dobrovolsckiy, Y.A.; Shaglaeve, N.S.; Frolova, L.A.; Gerasimova, E.V.; Sanginov, E.A. Nanostructured materials for low-temperature fuel cells. Russ. Chem. Rev. 2012, 81, 191–201. [Google Scholar] [CrossRef]
- Li, C.; Tan, H.; Lin, J.; Luo, X.; Wang, S.; You, J.; Kim, J. Emerging Pt-based electrocatalysts with highly open nanoarchitectures for boosting oxygen reduction reaction. Nano Today 2018, 21, 91–105. [Google Scholar] [CrossRef]
- Banham, D.W.H.; Ye, S. Current Status and Future Development of Catalyst Materials and Catalyst Layers for Proton Exchange Membrane Fuel Cells: An Industrial Perspective. ACS Energy Lett. 2017, 2, 629–638. [Google Scholar] [CrossRef]
- Kuna, E.; Mrdenovic, D.; Jönsson-Niedziółka, M.; Pieta, P.; Pieta, I.S. Bimetallic nanocatalysts supported on graphitic carbon nitride for sustainable energy development: The shape-structure–activity relation. Nanoscale Adv. 2021, 3, 1342–1351. [Google Scholar] [CrossRef]
- Xiao, Z.; Wu, H.; Zhong, H.; Abdelhafiz, A.; Zeng, J. De-alloyed PtCu/C catalysts with enhanced electrocatalytic performance for the oxygen reduction reaction. Nanoscale 2021, 13, 13896–13904. [Google Scholar] [CrossRef]
- Guo, N.; Xue, H.; Bao, A.; Wang, Z.; Sun, J.; Song, T.; Ge, X.; Zhang, W.; Huang, K.; He, F.; et al. Achieve Superior Electrocatalytic Performance by Surface Copper Vacancy Defects during Electrochemical Etching Process. Angew. Chem. Int. Ed. 2020, 59, 13778–13784. [Google Scholar] [CrossRef]
- Chen, A.; Holt-Hindle, P. Platinum-Based Nanostructured Materials: Synthesis, Properties, and Applications. Chem. Rev. 2010, 110, 3767–3804. [Google Scholar] [CrossRef]
- Kodama, K.; Nagai, T.; Kuwaki, A.; Jinnouchi, R.; Morimoto, Y. Challenges in applying highly active Pt-based nanostructured catalysts for oxygen reduction reactions to fuel cell vehicles. Nat. Nanotechnol. 2021, 16, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Moriau, L.J.; Hrnjić, A.; Pavlišič, A.; Kamšek, A.R.; Petek, U.; Ruiz-Zepeda, F.; Sala, M.; Pavko, L.; Selih, V.S.; Bele, M.; et al. Resolving the nanoparticles’ structure-property relationships at the atomic level: A study of Pt-based electrocatalysts. iScience 2021, 24, 102102. [Google Scholar] [CrossRef] [PubMed]
- Strasser, P.; Koh, S.; Greeley, J. Voltammetric surface dealloying of Pt bimetallic nanoparticles: An experimental and DFT computational analysis. Phys. Chem. Chem. Phys. 2008, 10, 3670–3683. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.; Yu, Y.; Xin, H.L.; Hovden, R.; Ercius, P.; Mundy, J.A.; Chen, H.; Richard, J.H.; Muller, D.A.; DiSalvo, F.J.; et al. Tuning Oxygen Reduction Reaction Activity via Controllable Dealloying: A Model Study of Ordered Cu3Pt/C Intermetallic Nanocatalysts. Nano Lett. 2012, 12, 5230–5238. [Google Scholar] [CrossRef] [PubMed]
- Oezaslan, M.; Strasser, P. Activity of dealloyed PtCo3 and PtCu3 nanoparticle electrocatalyst for oxygen reduction reaction in polymer electrolyte membrane fuel cell. J. Power Sources 2011, 196, 5240–5249. [Google Scholar] [CrossRef]
- Neyerlin, K.C.; Srivastava, R.; Yu, C.; Strasser, P. Electrochemical activity and stability of dealloyed Pt–Cu and Pt–Cu–Co electrocatalysts for the oxygen reduction reaction (ORR). J. Power Sources 2009, 186, 261–267. [Google Scholar] [CrossRef]
- Pavlets, A.; Alekseenko, A.; Menshchikov, V.; Belenov, S.; Volochaev, V.; Pankov, I.; Safronenko, O.; Guterman, V. Influence of Electrochemical Pretreatment Conditions of PtCu/C Alloy Electrocatalyst on Its Activity. Nanomaterials 2021, 11, 1499. [Google Scholar] [CrossRef]
- Menshchikov, V.; Alekseenko, A.; Guterman, V.; Nechitailov, A.; Glebova, N.; Tomasov, A.; Spiridonova, O.; Belenov, S.; Zelenina, N.; Safronenko, O. Effective Platinum-Copper Catalysts for Methanol Oxidation and Oxygen Reduction in Proton-Exchange Membrane Fuel Cell. Nanomaterials 2020, 10, 742. [Google Scholar] [CrossRef] [Green Version]
- Jung, N.; Chung, D.Y.; Ryu, J.; Yoo, S.J.; Sung, Y.-E. Pt-based nanoarchitecture and catalyst design for fuel cell applications. Nano Today 2014, 9, 433–456. [Google Scholar] [CrossRef]
- Pavlov, V.I.; Gerasimova, E.V.; Zolotukhina, E.V.; Dobrovolsky, Y.A.; Don, G.M.; Yaroslavtsev, A.B. Degradation of Pt/C electrocatalysts having different morphology in low-temperature PEM fuel cells. Nanotechnol. Russ. 2016, 11, 743–750. [Google Scholar] [CrossRef]
- Guterman, V.E.; Belenov, S.V.; Alekseenko, A.A.; Volochaev, V.A.; Tabachkova, N.Y. The relationship between activity and stability of deposited platinum-carbon electrocatalysts. Russ. J. Electrochem. 2017, 53, 531–539. [Google Scholar] [CrossRef]
- Yano, H.; Watanabea, M.; Iiyamaa, A.; Uchida, H. Particle-size effect of Pt cathode catalysts on durability in fuel cells. Nano Energy 2016, 29, 323–333. [Google Scholar] [CrossRef]
- Park, Y.-C.; Kakinuma, K.; Uchida, M.; Uchida, H.; Watanabe, M. Deleterious effects of interim cyclic voltammetry on Pt/carbon black catalyst degradation during start-up/shutdown cycling evaluation. Electrochim. Acta 2014, 123, 82–92. [Google Scholar]
- Meier, J.C.; Galeano, C.; Katsounaros, I.; Witte, J.; Bongard, H.J.; Topalov, A.A.; Baldizzone, C.; Mezzavilla, S.; Schüth, F.; Mayrhofer, K.J.J. Design criteria for stable Pt/C fuel cell catalysts. Beilstein J. Nanotechnol. 2014, 5, 44–67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Holby, E.F.; Sheng, W.; Shao-Horn, Y.; Morgan, D. Pt nanoparticle stability in PEM fuel cells: Influence of particle size distribution and crossover hydrogen. Energy Environ. Sci. 2009, 2, 865–871. [Google Scholar] [CrossRef]
- Hasché, F.; Oezaslan, M.; Strasser, P. Activity, stability and degradation of multi walled carbon nanotube (MWCNT) supported Pt fuel cell electrocatalysts. Phys. Chem. Chem. Phys. 2010, 12, 15251. [Google Scholar] [CrossRef]
- Capelo, A.; Esteves, M.A.; Sa, A.I.; Silva, R.A.; Cangueiro, L.; Almeida, A.; Vilar, R.; Rangel, C.M. Stability and durability under potential cycling of Pt/C catalyst with new surface-functionalized carbon support. Int. J. Hydrogen Energy 2016, 41, 12962–12975. [Google Scholar] [CrossRef]
- Baschuk, J.J.; Li, X. Carbon monoxide poisoning of proton exchange membrane fuel cells. Int. J. Energy Res. 2001, 25, 695–713. [Google Scholar] [CrossRef]
- Yan, W.-M.; Chu, H.; Lu, M.-X.; Weng, F.-B.; Jung, G.-B.; Lee, C. Degradation of proton exchange membrane fuel cells due to CO and CO2 poisoning. J. Power Sources 2009, 188, 141–147. [Google Scholar] [CrossRef]
- Khan, I.A.; Qian, Y.; Badshah, A.; Zhao, D.; Nadeem, M.A. Fabrication of Highly Stable and Efficient PtCu Alloy Nanoparticles on Highly Porous Carbon for Direct Methanol Fuel Cells. ACS Appl. Mater. Interfaces 2016, 8, 20793–20801. [Google Scholar] [CrossRef]
- Kim, J.; Hong, Y.; Lee, K.; Kim, J.Y. Highly Stable Pt-Based Ternary Systems for Oxygen Reduction Reaction in Acidic Electrolytes. Adv. Energy Mater. 2020, 10, 2002049. [Google Scholar] [CrossRef]
- Dicks, A.L. The role of carbon in fuel cells. J. Power Sources 2006, 156, 128–141. [Google Scholar] [CrossRef]
- Wang, X.; Li, W.; Chen, Z.; Waje, M.; Yan, Y. Durability investigation of carbon nanotube as catalyst support for proton exchange membrane fuel cell. J. Power Sources 2006, 158, 154–159. [Google Scholar] [CrossRef]
- Gribov, E.N.; Kuznetsov, A.N.; Voropaev, I.N.; Golovin, V.A.; Simonov, P.A.; Romanenko, A.V.; Okunev, A.G. Analysis of the Corrosion Kinetic of Pt/C Catalysts Prepared on Different Carbon Supports Under the “Start-Stop” Cycling. Electrocatalysis 2016, 7, 159–173. [Google Scholar] [CrossRef]
- Zheng, X.; Wu, J.; Cao, X.; Abbott, J.; Jin, C.; Wang, H.; Strasser, P.; Yang, R.; Chen, X.; Wu, G. N-, P-, and S-doped graphene-like carbon catalysts derived from onium salts with enhanced oxygen chemisorption for Zn-air battery cathodes. Appl. Catal. B 2019, 241, 442–451. [Google Scholar] [CrossRef]
- Imran Jafri, R.; Rajalakshmi, N.; Ramaprabhu, S. Nitrogen doped graphene nanoplatelets as catalyst support for oxygen reduction reaction in proton exchange membrane fuel cell. J. Mater. Chem. 2010, 20, 7114–7117. [Google Scholar] [CrossRef]
- Wang, Y.-Z.; Ko, T.-H.; Huang, W.-Y.; Hsieh, T.-H.; Ho, K.-S.; Chen, Y.-Y.; Hsieh, S.-J. Preparation of Pt-Catalyst by Poly(p-phenylenediamine) Nanocomposites Assisted by Microwave Radiation for Proton Exchange Membrane Fuel Cell. Polymers 2018, 10, 1388. [Google Scholar] [CrossRef] [Green Version]
- Chen, Y.; Wang, J.; Liu, H.; Li, R.; Sun, X.; Ye, S.; Knights, S. Enhanced stability of Pt electrocatalysts by nitrogen doping in CNTs for PEM fuel cells. Electrochem. Commun. 2009, 11, 2071–2076. [Google Scholar] [CrossRef]
- Moguchikh, E.A.; Paperzh, K.O.; Alekseenko, A.A.; Gribov, E.N.; Tabachkova, N.Y.; Maltseva, N.V.; Tkachev, A.G.; Neskoromnaya, E.A.; Melezhik, A.V.; Butova, V.V.; et al. Platinum nanoparticles supported on nitrogen-doped carbons as electrocatalysts for oxygen reduction reaction. J. Appl. Electroc. 2021, 52, 231–246. [Google Scholar] [CrossRef]
- Zahoor, A.; Christy, M.; Hwang, Y.J.; Lim, Y.R.; Kim, P.; Nahm, K.S. Improved electrocatalytic activity of carbon materials by nitrogen doping. Appl. Catal. B Environ. 2014, 147, 633–641. [Google Scholar] [CrossRef]
- Zafar, Z.; Ni, Z.H.; Wu, X.; Shi, Z.X.; Nan, H.Y.; Bai, J.; Sun, L.T. Evolution of Raman spectra in nitrogen doped graphene. Carbon 2013, 61, 57–62. [Google Scholar] [CrossRef]
- Ott, S.; Orfanidi, A.; Schmies, H.; Anke, B.; Nong, H.N.; Hbner, J.; Gernert, U.; Gliech, M.; Lerch, M.; Strasser, P. Ionomer distribution control in porous carbon-supported catalyst layers for high-power and low Pt-loaded proton exchange membrane fuel cells. Nat. Mater. 2020, 19, 77–85. [Google Scholar] [CrossRef]
- Leontyev, I.N.; Belenov, S.V.; Guterman, V.E.; Haghi-Ashtiani, P.; Shaganov, A.P.; Dkhil, B. Catalytic Activity of Carbon-Supported Pt Nanoelectrocatalysts. Why Reducing the Size of Pt Nanoparticles is Not Always Beneficial. J. Phys. Chem. C 2011, 115, 5429–5434. [Google Scholar] [CrossRef]
- Guterman, V.E.; Belenov, S.V.; Krikov, V.V.; Vysochina, L.L.; Yohannes, W.; Tabachkova, N.Y.; Balakshina, E.N. Reasons for the differences in the kinetics of thermal oxidation of Pt/C nanostructured materials. J. Phys. Chem. C 2014, 118, 23835–23844. [Google Scholar] [CrossRef]
- Nagai, T.; Jahn, C.; Jia, H. Improved Accelerated Stress Tests for ORR Catalysts Using a Rotating Disk Electrode. J. Electrochem. Soc. 2019, 166, F3111–F3115. [Google Scholar] [CrossRef]
- Zalitis, C.M.; Kucernak, A.R.; Sharman, J.; Wright, E. Design principles for platinum nanoparticles catalysing electrochemical hydrogen evolution and oxidation reactions: Edges are much more active than facets. J. Mater. Chem. 2017, 5, 23328–23338. [Google Scholar] [CrossRef] [Green Version]
- Pavlets, A.S.; Alekseenko, A.A.; Tabachkova, N.Y.; Safronenko, O.I.; Nikulin, A.Y.; Alekseenko, D.V.; Guterman, V.E. A novel strategy for the synthesis of Pt–Cu uneven nanoparticles as an efficient electrocatalyst toward oxygen reduction. Int. J. Hydrogen Energy 2021, 46, 5255–5368. [Google Scholar] [CrossRef]
- Gao, J.; Mao, M.; Li, P.; Liu, R.; Song, H.; Sun, K.; Zhang, S. Segmentation and Re-encapsulation of Porous PtCu Nanoparticles by Generated Carbon Shell for Enhanced Ethylene-glycol Oxidation and Oxygen-reduction Reaction. ACS Appl. Mater. Interfaces 2020, 12, 6298−6308. [Google Scholar] [CrossRef] [PubMed]
- Shinozaki, K.; Zack, J.W.; Pylypenko, S.; Pivovar, B.S.; Kocha, S.S. Oxygen reduction reaction measurements on platinum electrocatalysts utilizing rotating disk electrode technique: II. Influence of ink formulation, catalyst layer uniformity and thickness. J. Electrochem. Soc. 2015, 162, 1384–1396. [Google Scholar] [CrossRef]
- Khudhayer, W.J.; Kariuki, N.N.; Wang, X.; Myers, D.J.; Shaikh, A.U.; Karabacak, T. Oxygen Reduction Reaction Electrocatalytic Activity of Glancing Angle Deposited Platinum Nanorod. Arrays. J. Electrochem. Soc. 2011, 158, 1029–1041. [Google Scholar] [CrossRef] [Green Version]





| Sample | Mass Fraction of Pt, % | Mass Fraction of Pt + M, % | Dav, nm (XRD) | Dav, nm (TEM) |
|---|---|---|---|---|
| Pt/KB-N | 22.0 | 22.0 | <1.0 | 2.0 ± 0.1 |
| PtCu/KB | 19.6 | 21.4 | 2.9 ± 0.1 | 3.6 ± 0.2 |
| PtCu/KB-N | 22.0 | 23.7 | 2.7 ± 0.1 | 2.9 ± 0.1 |
| JM20 | 20.0 | 20.0 | 2.4 ± 0.1 | 2.7 ± 0.1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Alekseenko, A.; Pavlets, A.; Moguchikh, E.; Tolstunov, M.; Gribov, E.; Belenov, S.; Guterman, V. Platinum-Containing Nanoparticles on N-Doped Carbon Supports as an Advanced Electrocatalyst for the Oxygen Reduction Reaction. Catalysts 2022, 12, 414. https://doi.org/10.3390/catal12040414
Alekseenko A, Pavlets A, Moguchikh E, Tolstunov M, Gribov E, Belenov S, Guterman V. Platinum-Containing Nanoparticles on N-Doped Carbon Supports as an Advanced Electrocatalyst for the Oxygen Reduction Reaction. Catalysts. 2022; 12(4):414. https://doi.org/10.3390/catal12040414
Chicago/Turabian StyleAlekseenko, Anastasia, Angelina Pavlets, Elizaveta Moguchikh, Mikhail Tolstunov, Evgeny Gribov, Sergey Belenov, and Vladimir Guterman. 2022. "Platinum-Containing Nanoparticles on N-Doped Carbon Supports as an Advanced Electrocatalyst for the Oxygen Reduction Reaction" Catalysts 12, no. 4: 414. https://doi.org/10.3390/catal12040414
APA StyleAlekseenko, A., Pavlets, A., Moguchikh, E., Tolstunov, M., Gribov, E., Belenov, S., & Guterman, V. (2022). Platinum-Containing Nanoparticles on N-Doped Carbon Supports as an Advanced Electrocatalyst for the Oxygen Reduction Reaction. Catalysts, 12(4), 414. https://doi.org/10.3390/catal12040414










