Direct Z-Scheme Heterojunction α-MnO2/BiOI with Oxygen-Rich Vacancies Enhanced Photoelectrocatalytic Degradation of Organic Pollutants under Visible Light
Abstract
:1. Introduction
2. Results and Discussion
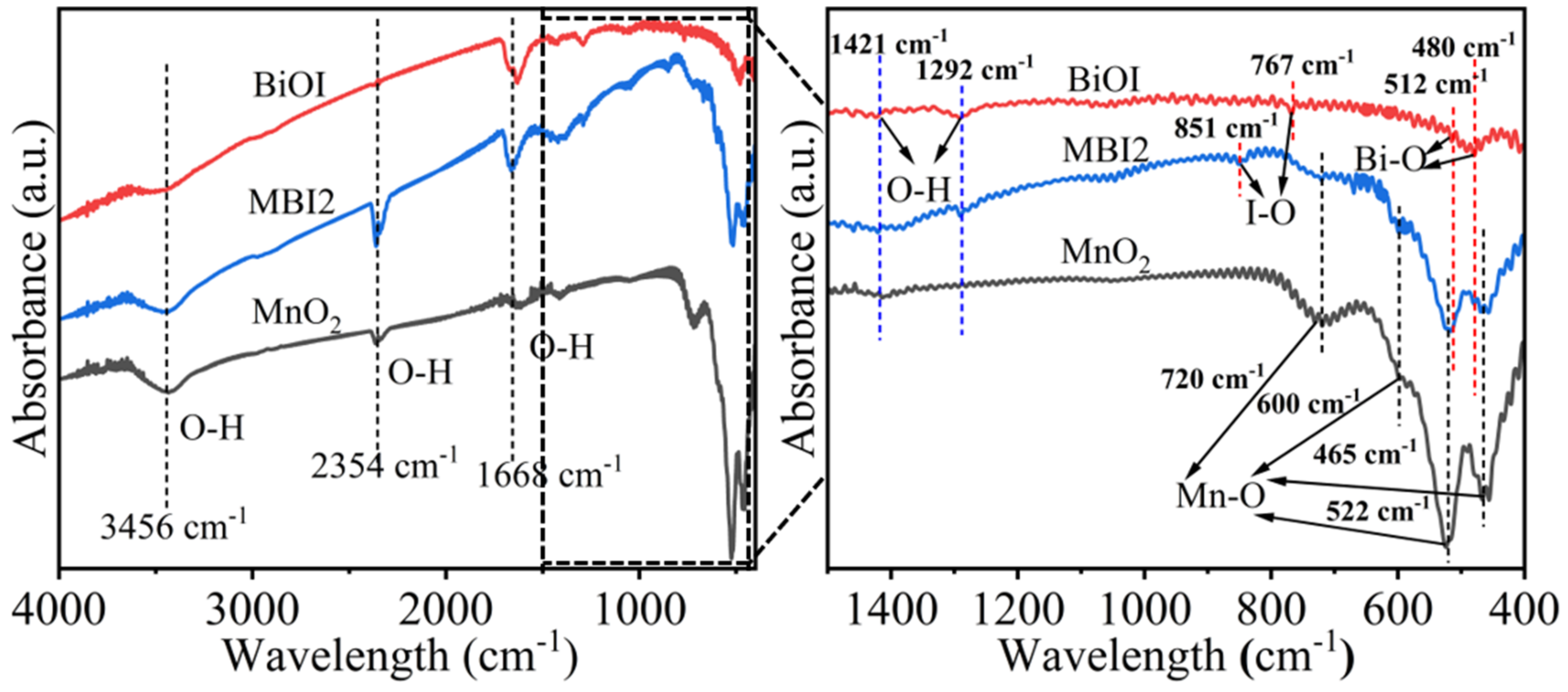
2.1. Photoelectric Properties of Different Photoelectrocatalysts
2.2. PEC Performances and Recyclability Test
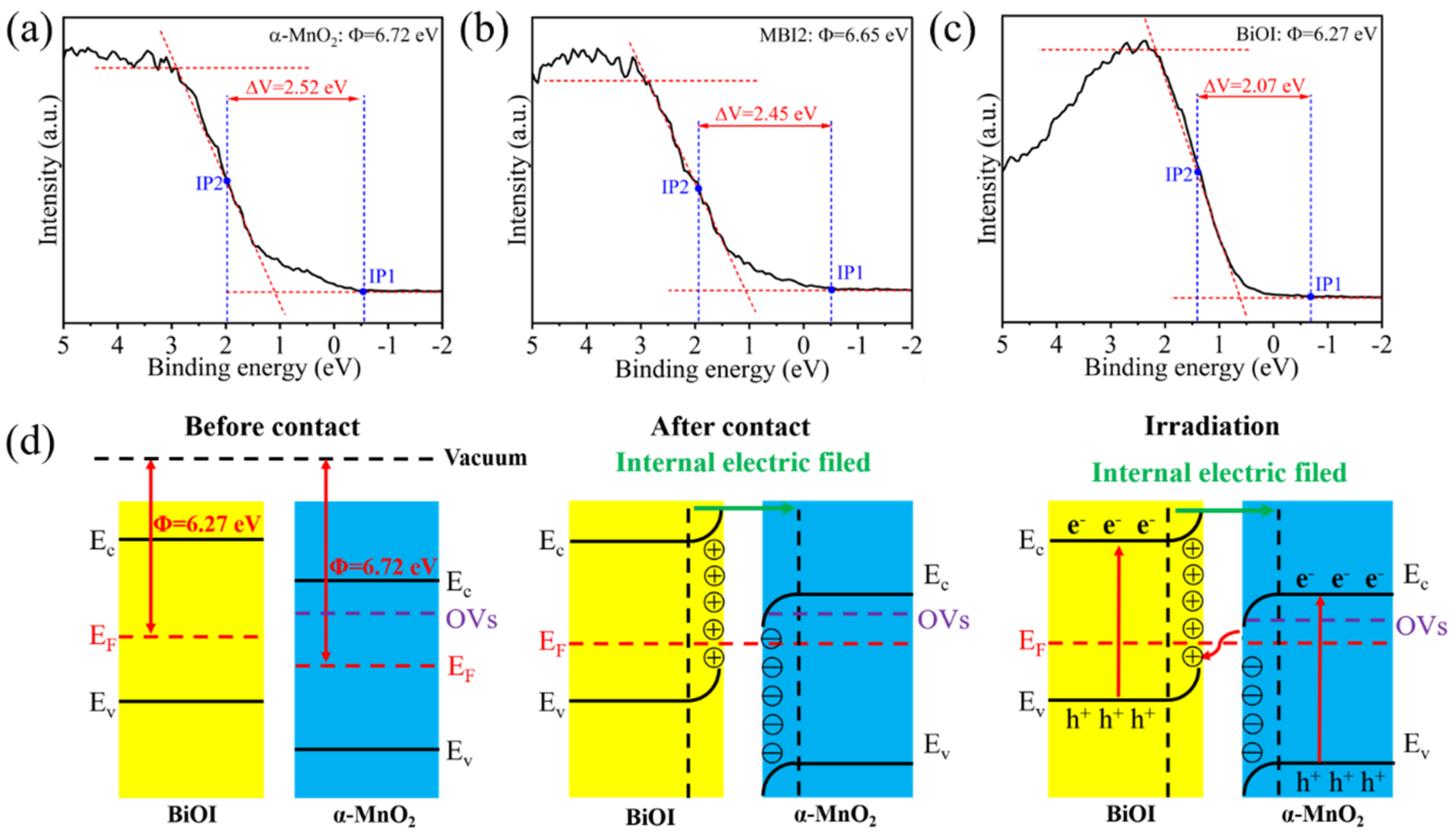
2.3. PEC Degradation Mechanism Study
3. Experimental Section
3.1. Chemicals and Reagents
3.2. Preparation of 1D α-MnO2 Nanorods
3.3. Preparation of α-MnO2/BiOI
3.4. Characterizations
3.5. PEC Degradation Experiment
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Bhoyar, T.; Vidyasagar, D.; Umare, S.S. Mitigating phytotoxicity of tetracycline by metal-free 8-hydroxyquinoline functionalized carbon nitride photocatalyst. J. Environ. Sci. 2023, 125, 37–46. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Dong, N.; Zhang, J.; Wang, W.; Tan, F.; Wang, X.; Qiao, X.; Keung Wong, P. Investigation on visible-light photocatalytic performance and mechanism of zinc peroxide for tetracycline degradation and Escherichia coli inactivation. J. Colloid Interface Sci. 2022, 624, 137–149. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Cai, M.; Wang, C.; Liu, Y.; Li, N.; Zhang, P.; Li, X. Rationally designed Ta3N5/BiOCl S-scheme heterojunction with oxygen vacancies for elimination of tetracycline antibiotic and Cr(VI): Performance, toxicity evaluation and mechanism insight. J. Mater. Sci. Technol. 2022, 123, 177–190. [Google Scholar] [CrossRef]
- Li, S.; Wang, C.; Cai, M.; Liu, Y.; Dong, K.; Zhang, J. Designing oxygen vacancy mediated bismuth molybdate (Bi2MoO6)/N-rich carbon nitride (C3N5) S-scheme heterojunctions for boosted photocatalytic removal of tetracycline antibiotic and Cr(VI): Intermediate toxicity and mechanism insight. J. Colloid Interface Sci. 2022, 624, 219–232. [Google Scholar] [CrossRef] [PubMed]
- Oluwole, A.O.; Olatunji, O.S. Photocatalytic degradation of tetracycline in aqueous systems under visible light irridiation using needle-like SnO2 nanoparticles anchored on exfoliated g-C3N4. Environ. Sci. Eur. 2022, 34, 5–12. [Google Scholar] [CrossRef]
- Perumal, K.; Shanavas, S.; Ahamad, T.; Karthigeyan, A.; Murugakoothan, P. Construction of Ag2CO3/BiOBr/CdS ternary composite photocatalyst with improved visible-light photocatalytic activity on tetracycline molecule degradation. J. Environ. Sci. 2023, 125, 47–60. [Google Scholar] [CrossRef]
- Zheng, J.; Fan, C.; Li, X.; Yang, Q.; Wang, D.; Duan, A.; Ding, J.; Rong, S.; Chen, Z.; Luo, J.; et al. Enhanced photodegradation of tetracycline hydrochloride by hexameric AgBr/Zn-Al MMO S-scheme heterojunction photocatalysts: Low metal leaching, degradation mechanism and intermediates. Chem. Eng. J. 2022, 446, 137371. [Google Scholar] [CrossRef]
- Niu, J.; Song, Z.; Gao, X.; Ji, Y.; Zhang, Y. Construction of Bi2WO6 composites with carbon-coated Cu2O for effective degradation of tetracycline. J. Alloys Compd. 2021, 884, 161292. [Google Scholar] [CrossRef]
- Wang, T.; Zheng, J.; Cai, J.; Liu, Q.; Zhang, X. Visible-light-driven photocatalytic degradation of dye and antibiotics by activated biochar composited with K+ doped g-C3N4: Effects, mechanisms, actual wastewater treatment and disinfection. Sci. Total Environ. 2022, 839, 155955. [Google Scholar] [CrossRef]
- Li, H.; Xu, X.; Wang, J.; Han, X.; Xu, Z. A Robust PVDF-Assisted Composite Membrane for Tetracycline Degradation in Emulsion and Oil-Water Separation. Nanomaterials 2021, 11, 3201. [Google Scholar] [CrossRef]
- Cheng, S.; Zhang, C.; Li, J.; Pan, X.; Zhai, X.; Jiao, Y.; Li, Y.; Dong, W.; Qi, X. Highly efficient removal of antibiotic from biomedical wastewater using Fenton-like catalyst magnetic pullulan hydrogels. Carbohydr. Polym. 2021, 262, 117951. [Google Scholar] [CrossRef]
- Li, Y.; Fu, M.; Wang, R.; Wu, S.; Tan, X. Efficient removal TC by Zn@SnO2/PI via the synergy of adsorption and photocatalysis under visible light. Chem. Eng. J. 2022, 444, 136567. [Google Scholar] [CrossRef]
- Su, P.; Fu, W.; Hu, Z.; Jing, J.; Zhou, M. Insights into transition metal encapsulated N-doped CNTs cathode for self-sufficient electrocatalytic degradation. Appl. Catal. B Environ. 2022, 313, 121457. [Google Scholar] [CrossRef]
- Ma, E.; Sun, G.; Duan, F.; Wang, H.; Wang, H. Visible-light-responsive Z-scheme heterojunction MoS2 NTs/CuInS2 QDs photoanode for enhanced photoelectrocatalytic degradation of tetracycline. Appl. Mater. Today 2022, 28, 101504. [Google Scholar] [CrossRef]
- Deng, W.; Xiong, K.; Ge, N.; Yu, M.; Chen, L.; Wang, J. Cobalt and titanium co-doped zinc ferrite film photoanode with boosted interfacial photoelectrocatalytic activity for efficient degradation of tetracycline via the covalency competition in the Zn-O-Fe backbone. Chem. Eng. J. 2022, 433, 134456. [Google Scholar] [CrossRef]
- Ancy, K.; Bindhu, M.R.; Bai, J.S.; Gatasheh, M.K.; Hatamleh, A.A.; Ilavenil, S. Photocatalytic degradation of organic synthetic dyes and textile dyeing waste water by Al and Fe co-doped TiO2 nanoparticles. Environ. Res. 2022, 206, 112492. [Google Scholar] [CrossRef]
- Zabelin, D.; Zabelina, A.; Miliutina, E.; Trelin, A.; Elashnikov, R.; Nazarov, D.; Maximov, M.; Kalachyova, Y.; Sajdl, P.; Lancok, J.; et al. Design of hybrid Au grating/TiO2 structure for NIR enhanced photo-electrochemical water splitting. Chem. Eng. J. 2022, 443, 136440. [Google Scholar] [CrossRef]
- Yan, L.; Ni, C.; Duan, Y.; Tian, Z.; Zhang, K.; Li, B.; Liang, J.; Feng, H.; Deng, X.; Zhang, H.; et al. Surface plasmon resonance and oxygen vacancy on Bi/BiO1−y ClxBr1−x synergistically boost high-efficiently photodegradation acetaminophen in waste water. Colloids Surf. A Physicochem. Eng. Asp. 2022, 648, 129310. [Google Scholar] [CrossRef]
- Jiang, G.; Zhu, B.; Sun, J.; Liu, F.; Wang, Y.; Zhao, C. Enhanced activity of ZnS (111) by N/Cu co-doping: Accelerated degradation of organic pollutants under visible light. J. Environ. Sci. 2023, 125, 244–257. [Google Scholar] [CrossRef]
- Liu, X.; Li, F.; Liu, Y.; Li, P.; Chen, L.; Li, B.; Qian, T.; Liu, W. Degradation of diclofenac in a photosensitization-like photocatalysis process using palladium quantum dots deposited graphite carbon nitride under solar light. J. Environ. Chem. Eng. 2022, 10, 107545. [Google Scholar] [CrossRef]
- Ye, X.; Wu, J.; Chen, F. Bifunctional metastable LaOCl@hcp-Ni nanocomposite via Mott-Schottky effect for improved photoelectrocatalytic and photocatalytic activities. J. Alloys Compd. 2022, 912, 165032. [Google Scholar] [CrossRef]
- Feng, X.; Yu, Z.; Shan, M.; Long, R.; Li, X.; Liao, K. Z-type ZnAl-LDO/Ag2S heterojunction activated peroxysulfate to degrade tetracycline hydrochloride under visible light efficiently. Chem. Eng. J. 2022, 443, 136422. [Google Scholar] [CrossRef]
- Li, M.; Xing, Z.; Zhang, Z.; Wang, Y.; Liu, M.; Li, Z.; Wang, N.; Zhou, W. Hollow core-shell Z-scheme heterojunction on self-floating carbon fiber cloth with robust photocatalytic-photothermal performance. J. Clean. Prod. 2022, 360, 132166. [Google Scholar] [CrossRef]
- Xiong, J.; Di, J.; Xia, J.; Zhu, W.; Li, H. Surface Defect Engineering in 2D Nanomaterials for Photocatalysis. Adv. Funct. Mater. 2018, 28, 1801983. [Google Scholar] [CrossRef]
- Shaddad, M.N.; Arunachalam, P.; Hezam, M.; Al-Saeedan, N.M.; Gimenez, S.; Bisquert, J.; Al-Mayouf, A.M. Unprecedented solar water splitting of dendritic nanostructured Bi2O3 films by combined oxygen vacancy formation and Na2MoO4 doping. Int. J. Hydrogen Energy 2021, 46, 23702–23714. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, J.; Balogun, M.S.; Tong, Y.; Huang, Y. Oxygen vacancy–based metal oxides photoanodes in photoelectrochemical water splitting. Mater. Today Sustain. 2022, 18, 100118. [Google Scholar] [CrossRef]
- Sayed, M.; Ren, B.; Ali, A.M.; Al-Anazi, A.; Nadagouda, M.N.; Ismail, A.A.; Dionysiou, D.D. Solar light induced photocatalytic activation of peroxymonosulfate by ultra-thin Ti3+ self-doped Fe2O3/TiO2 nanoflakes for the degradation of naphthalene. Appl. Catal. B Environ. 2022, 315, 121532. [Google Scholar] [CrossRef]
- Yang, J.; Su, H.; Wu, Y.; Li, D.; Zhang, D.; Sun, H.; Yin, S. Facile synthesis of kermesinus BiOI with oxygen vacancy for efficient hydrogen generation. Chem. Eng. J. 2021, 420, 127607. [Google Scholar] [CrossRef]
- Liu, X.; Sui, B.; Camargo, P.H.C.; Wang, J.; Sun, J. Tuning band gap of MnO2 nanoflowers by Alkali metal doping for enhanced Ferroptosis/phototherapy synergism in Cancer. Appl. Mater. Today 2021, 23, 101027. [Google Scholar] [CrossRef]
- Salari, H.; Yaghmaei, H. Z-scheme 3D Bi2WO6/MnO2 heterojunction for increased photoinduced charge separation and enhanced photocatalytic activity. Appl. Surf. Sci. 2020, 532, 147413. [Google Scholar] [CrossRef]
- Jing, Y.; Fan, A.; Guo, J.; Shen, T.; Yuan, S.; Chu, Y. Synthesis of an ultrathin MnO2 nanosheet-coated Bi2WO6 nanosheet as a heterojunction photocatalyst with enhanced photocatalytic activity. Chem. Eng. J. 2022, 429, 132193. [Google Scholar] [CrossRef]
- Liang, Z.; Wang, H.; Zhang, K.; Ma, G.; Zhu, L.; Zhou, L.; Yan, B. Oxygen-defective MnO2/ZIF-8 nanorods with enhanced antibacterial activity under solar light. Chem. Eng. J. 2022, 428, 131349. [Google Scholar] [CrossRef]
- Ma, S.; Luo, X.; Ran, G.; Li, Y.; Cao, Z.; Liu, X.; Chen, G.; Yan, J.; Wang, L. Defect engineering of ultrathin 2D nanosheet BiOI/Bi for enhanced photothermal-catalytic synergistic bacteria-killing. Chem. Eng. J. 2022, 435, 134810. [Google Scholar] [CrossRef]
- Zhang, L.; Wu, X.; Li, J. Sn-doped BiOI assisted the excellent photocatalytic performance of multi-shelled ZnO microspheres under simulated sunlight. Chemosphere 2022, 290, 133309. [Google Scholar] [CrossRef]
- Du, C.; Zhang, Z.; Tan, S.; Yu, G.; Chen, H.; Zhou, L.; Yu, L.; Su, Y.; Zhang, Y.; Deng, F. Construction of Z-scheme g-C3N4/MnO2/GO ternary photocatalyst with enhanced photodegradation ability of tetracycline hydrochloride under visible light radiation. Environ. Res. 2021, 200, 111427. [Google Scholar] [CrossRef]
- Shaheen, S.; Iqbal, A.; Ikram, M.; Ul-Ain, K.; Naz, S.; Ul-Hamid, A.; Shahzadi, A.; Haider, A.; Nabgan, W.; Haider, J. Effective Disposal of Methylene Blue and Bactericidal Benefits of Using GO-Doped MnO2 Nanorods Synthesized through One-Pot Synthesis. ACS Omega 2021, 6, 24866–24878. [Google Scholar] [CrossRef]
- Zheng, X.; Chu, Y.; Miao, B.; Fan, J. Ag-doped Bi2WO6/BiOI heterojunction used as photocatalyst for the enhanced degradation of tetracycline under visible-light and biodegradability improvement. J. Alloys Compd. 2022, 893, 162382. [Google Scholar] [CrossRef]
- Ding, D.; Zhou, Y.; He, T.; Rong, S. Facet selectively exposed alpha-MnO2 for complete photocatalytic oxidation of carcinogenic HCHO at ambient temperature. Chem. Eng. J. 2022, 431, 133737. [Google Scholar] [CrossRef]
- Sarker, S.; Macharia, D.K.; Zhang, Y.; Zhu, Y.; Li, X.; Wen, M.; Meng, R.; Yu, N.; Chen, Z.; Zhu, M. Synthesis of MnO2-Ag Nanojunctions with Plasmon-Enhanced Photocatalytic and Photothermal Effects for Constructing Rewritable Mono-/Multi-Color Fabrics. ACS Appl. Mater. Interfaces 2022, 14, 5545–5557. [Google Scholar] [CrossRef]
- Bao, X.; Zhang, M.; Wang, Z.; Dai, D.; Wang, P.; Cheng, H.; Liu, Y.; Zheng, Z.; Dai, Y.; Huang, B. Molten-salt assisted synthesis of Cu clusters modified TiO2 with oxygen vacancies for efficient photocatalytic reduction of CO2 to CO. Chem. Eng. J. 2022, 445, 136718. [Google Scholar] [CrossRef]
- Yang, G.; Xiong, J.; Lu, M.; Wang, W.; Li, W.; Wen, Z.; Li, S.; Li, W.; Chen, R.; Cheng, G. Co-embedding oxygen vacancy and copper particles into titanium-based oxides (TiO2, BaTiO3, and SrTiO3) nanoassembly for enhanced CO2 photoreduction through surface/interface synergy. J. Colloid Interface Sci. 2022, 624, 348–361. [Google Scholar] [CrossRef]
- Huang, Y.; Lai, J.; Huang, Y.; Luo, X.; Yang, X.; Liu, Z.; Duan, Y.; Li, C. Response mechanism of Chlamydomonas reinhardtii to nanoscale bismuth oxyiodide (nano-BiOI): Integrating analysis of mineral nutrient metabolism and metabolomics. J. Environ. Sci. 2022, 121, 13–24. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.; Zou, X.; Chen, Q.; Fan, W.; Gong, Z. Pumice-loaded rGO@MnO2 nanomesh photocatalyst with visible light response for rapid degradation of ciprofloxacin. Sep. Purif. Technol. 2022, 297, 121502. [Google Scholar] [CrossRef]
- Zhang, K.; Chen, H.; Liu, Y.; Deng, J.; Jing, L.; Rastegarpanah, A.; Pei, W.; Han, Z.; Dai, H. Two-dimensional Bi2WxMo1−xO6 solid solution nanosheets for enhanced photocatalytic toluene oxidation to benzaldehyde. Appl. Catal. B Environ. 2022, 315, 121545. [Google Scholar] [CrossRef]
- Wang, X.; Sun, K.; Gu, S.; Zhang, Y.; Wu, D.; Zhou, X.; Gao, K.; Ding, Y. Construction of a novel electron transfer pathway by modifying ZnIn2S4 with α-MnO2 and Ag for promoting solar H2 generation. Appl. Surf. Sci. 2021, 549, 149341. [Google Scholar] [CrossRef]
- Zhao, F.; Song, Y.; Liu, X.; Zhang, X.; Ma, C.; Wang, G.; Fu, Y.; Ma, H. Unraveling Electron-deficient Setaria-viridis-like Co3O4@MnO2 heterostructure with superior photoelectrocatalytic efficiency for water remediation. Appl. Surf. Sci. 2022, 573, 151473. [Google Scholar] [CrossRef]
- Sukidpaneenid, S.; Chawengkijwanich, C.; Pokhum, C.; Isobe, T.; Opaprakasit, P.; Sreearunothai, P. Multi-function adsorbent-photocatalyst MXene-TiO2 composites for removal of enrofloxacin antibiotic from water. J. Environ. Sci. 2023, 124, 414–428. [Google Scholar] [CrossRef]
- Dong, H.; Zuo, Y.; Song, N.; Hong, S.; Xiao, M.; Zhu, D.; Sun, J.; Chen, G.; Li, C. Bimetallic synergetic regulating effect on electronic structure in cobalt/vanadium co-doped carbon nitride for boosting photocatalytic performance. Appl. Catal. B Environ. 2021, 287, 119954. [Google Scholar] [CrossRef]
- Ouyang, C.; Quan, X.; Zhang, C.; Pan, Y.; Li, X.; Hong, Z.; Zhi, M. Direct Z-scheme ZnIn2S4@MoO3 heterojunction for efficient photodegradation of tetracycline hydrochloride under visible light irradiation. Chem. Eng. J. 2021, 424, 130510. [Google Scholar] [CrossRef]
- Xiong, J.; Li, X.; Huang, J.; Gao, X.; Chen, Z.; Liu, J.; Li, H.; Kang, B.; Yao, W.; Zhu, Y. CN/rGO@BPQDs high-low junctions with stretching spatial charge separation ability for photocatalytic degradation and H2O2 production. Appl. Catal. B Environ. 2020, 266, 118602. [Google Scholar] [CrossRef]
- Huang, W.; Gao, Y.; Wang, J.; Ding, P.; Yan, M.; Wu, F.; Liu, J.; Liu, D.; Guo, C.; Yang, B.; et al. NIR Photocatalysis: Plasmonic Enhanced Reactive Oxygen Species Activation on Low-Work-Function Tungsten Nitride for Direct Near-Infrared Driven Photocatalysis. Small 2020, 16, 2070247. [Google Scholar] [CrossRef]
- Shen, S.; Zhang, H.; Xu, A.; Zhao, Y.; Lin, Z.; Wang, Z.; Zhong, W.; Feng, S. Construction of NiSe2/BiVO4 Schottky junction derived from work function discrepancy for boosting photocatalytic activity. J. Alloys Compd. 2021, 875, 160071. [Google Scholar] [CrossRef]
- Zeng, Y.; Xu, Y.; Zhong, D.; Yao, H.; Zhong, N. Peroxymonosulfate activated by photocatalytic fuel cell with g-C3N4/BiOI/Ti photoanode to enhance rhodamine B degradation and electricity generation. J. Hazard. Mater. 2022, 425, 127967. [Google Scholar] [CrossRef]


















Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jia, L.; Li, F.; Yang, C.; Yang, X.; Kou, B.; Xing, Y.; Peng, J.; Ni, G.; Cao, Z.; Zhang, S.; et al. Direct Z-Scheme Heterojunction α-MnO2/BiOI with Oxygen-Rich Vacancies Enhanced Photoelectrocatalytic Degradation of Organic Pollutants under Visible Light. Catalysts 2022, 12, 1596. https://doi.org/10.3390/catal12121596
Jia L, Li F, Yang C, Yang X, Kou B, Xing Y, Peng J, Ni G, Cao Z, Zhang S, et al. Direct Z-Scheme Heterojunction α-MnO2/BiOI with Oxygen-Rich Vacancies Enhanced Photoelectrocatalytic Degradation of Organic Pollutants under Visible Light. Catalysts. 2022; 12(12):1596. https://doi.org/10.3390/catal12121596
Chicago/Turabian StyleJia, Litao, Fanghua Li, Chenjia Yang, Xiaonan Yang, Beibei Kou, Yonglei Xing, Juan Peng, Gang Ni, Zhong Cao, Shiyu Zhang, and et al. 2022. "Direct Z-Scheme Heterojunction α-MnO2/BiOI with Oxygen-Rich Vacancies Enhanced Photoelectrocatalytic Degradation of Organic Pollutants under Visible Light" Catalysts 12, no. 12: 1596. https://doi.org/10.3390/catal12121596
APA StyleJia, L., Li, F., Yang, C., Yang, X., Kou, B., Xing, Y., Peng, J., Ni, G., Cao, Z., Zhang, S., Zhao, T., & Jin, X. (2022). Direct Z-Scheme Heterojunction α-MnO2/BiOI with Oxygen-Rich Vacancies Enhanced Photoelectrocatalytic Degradation of Organic Pollutants under Visible Light. Catalysts, 12(12), 1596. https://doi.org/10.3390/catal12121596







