Copper–Cerium–Tin Oxide Catalysts for Preferential Oxidation of CO in Hydrogen: Effects of Synthesis Method and Copper Content
Abstract
1. Introduction
2. Results
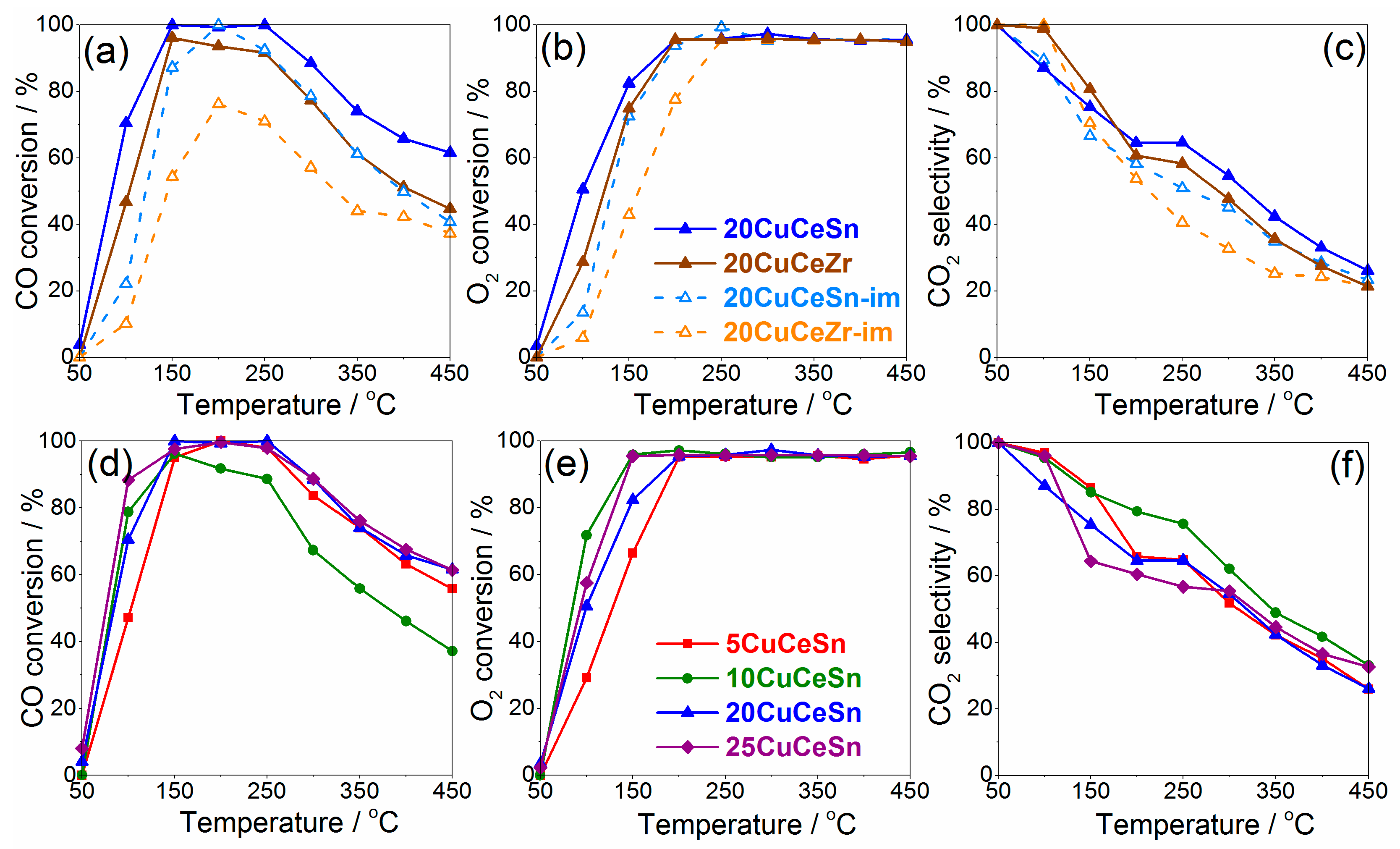
2.1. Catalytic Performance in CO-PROX
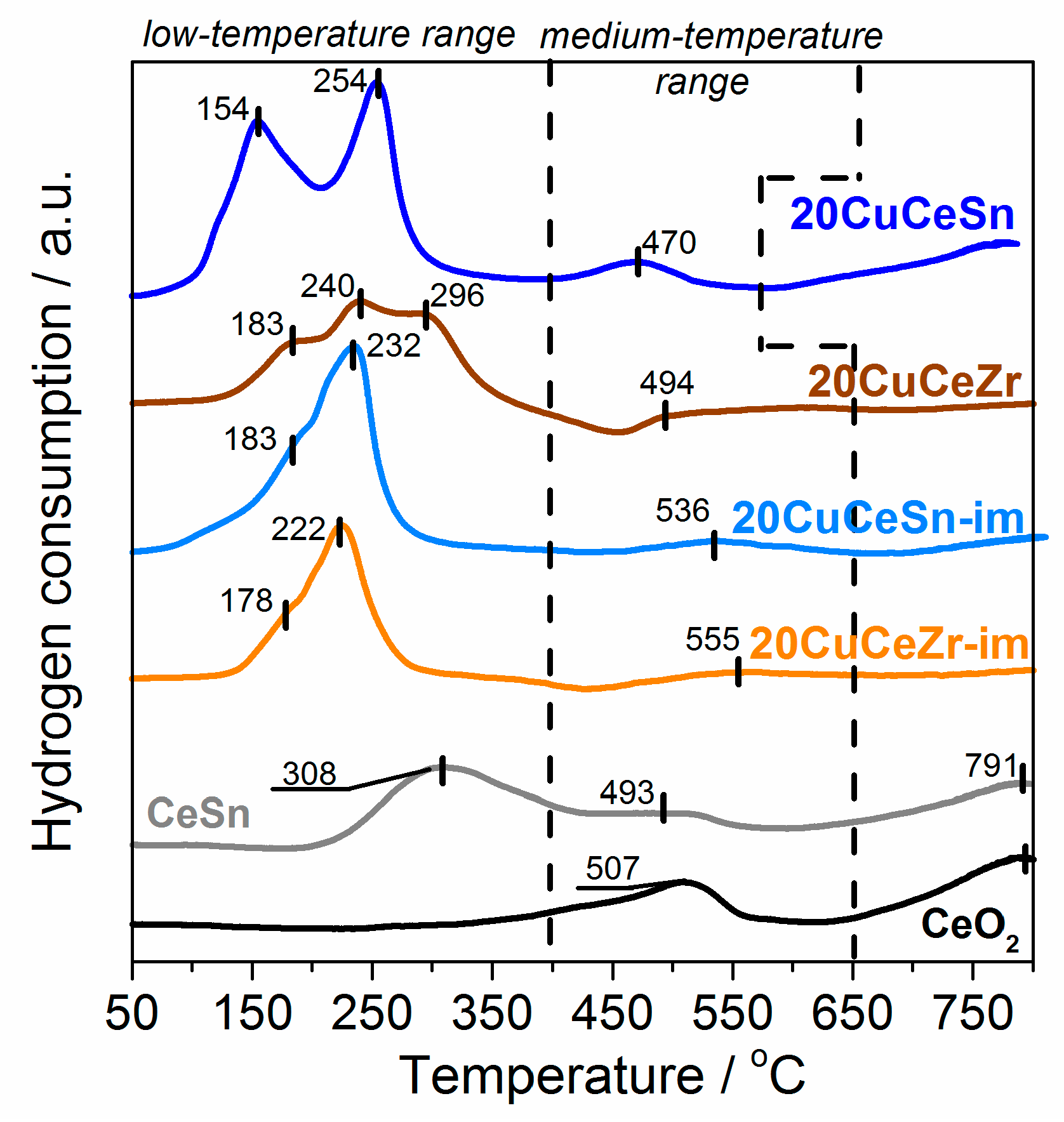
2.2. Physicochemical Characterization of Catalysts Synthesized by Different Techniques
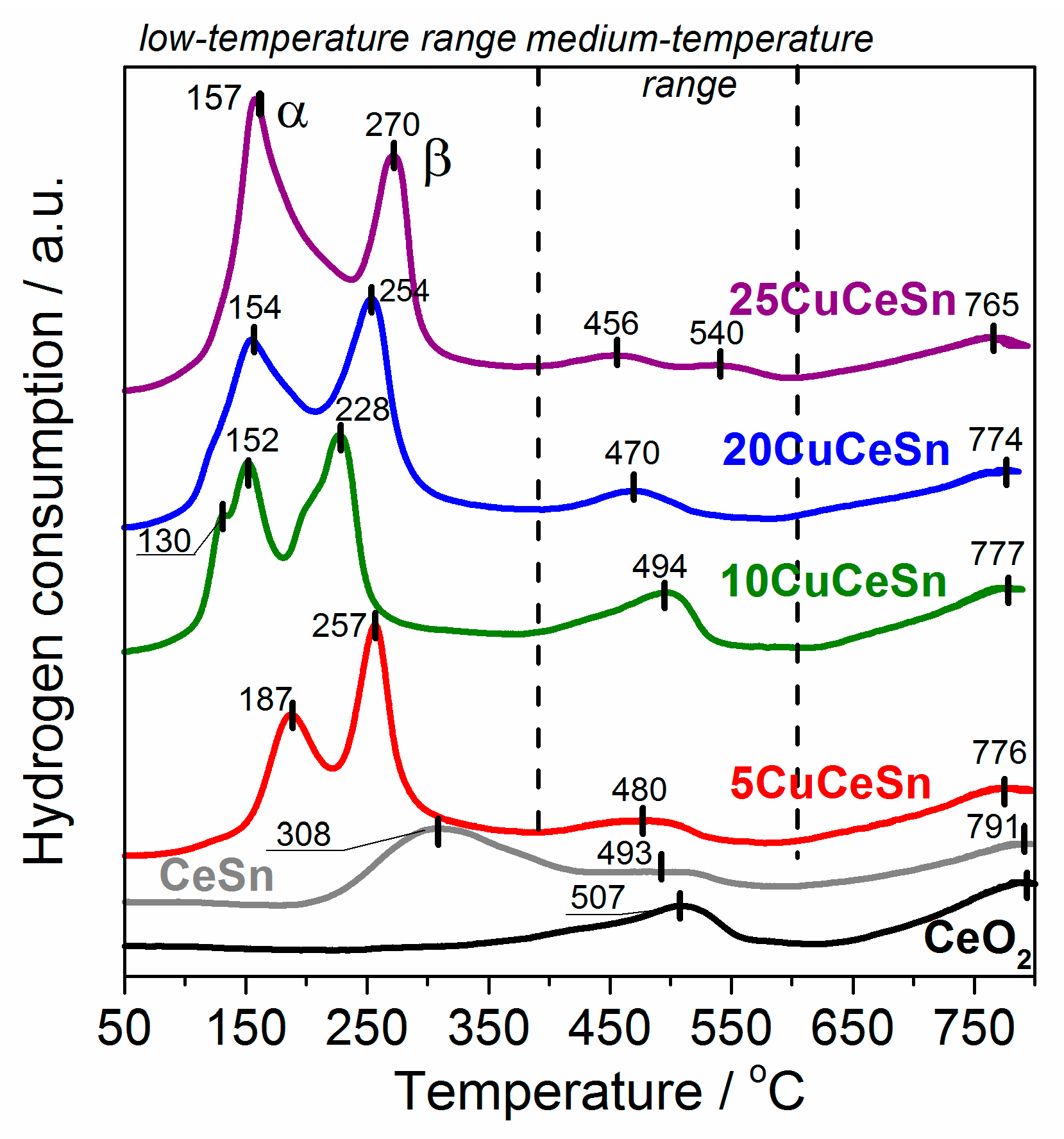
2.3. Effect of Copper Content on Physicochemical Properties of Catalysts
3. Discussion
4. Materials and Methods
4.1. Catalysts Preparation
4.2. Catalysts Characterization
4.3. Catalytic Tests
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Jing, P.; Gong, X.; Liu, B.; Zhang, J. Recent Advances in Synergistic Effect Promoted Catalysts for Preferential Oxidation of Carbon Monoxide. Catal. Sci. Technol. 2020, 10, 919–934. [Google Scholar] [CrossRef]
- Sahebdelfar, S.; Ravanchi, M.T. Carbon Monoxide Clean-up of the Reformate Gas for PEM Fuel Cell Applications: A Conceptual Review. Int. J. Hydrogen Energy, 2022; in press. [Google Scholar] [CrossRef]
- Gu, D.; Jia, C.J.; Bongard, H.; Spliethoff, B.; Weidenthaler, C.; Schmidt, W.; Schüth, F. Ordered Mesoporous Cu–Ce–O Catalysts for CO Preferential Oxidation in H2-Rich Gases: Influence of Copper Content and Pretreatment Conditions. Appl. Catal. B Environ. 2014, 152–153, 11–18. [Google Scholar] [CrossRef]
- Ayastuy, J.L.; Gurbani, A.; González-Marcos, M.P.; Gutiérrez-Ortiz, M.A. Selective CO Oxidation in H2 Streams on CuO/CexZr1−xO2 Catalysts: Correlation between Activity and Low Temperature Reducibility. Int. J. Hydrogen Energy 2012, 37, 1993–2006. [Google Scholar] [CrossRef]
- Chee, S.W.; Arce-Ramos, J.M.; Li, W.; Genest, A.; Mirsaidov, U. Structural Changes in Noble Metal Nanoparticles during CO Oxidation and Their Impact on Catalyst Activity. Nat. Commun. 2020 111 2020, 11, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Altass, H.M.; Ahmed, S.A.; Salama, R.S.; Moussa, Z.; Jassas, R.S.; Alsantali, R.I.; Al-Rooqi, M.M.; Ibrahim, A.A.; Khder, M.A.; Morad, M.; et al. Low Temperature CO Oxidation Over Highly Active Gold Nanoparticles Supported on Reduced Graphene Oxide@Mg-BTC Nanocomposite. Catal. Letters 2022, 1, 1–11. [Google Scholar]
- Kim, Y.H.; Park, E.D.; Lee, H.C.; Lee, D.; Lee, K.H. Preferential CO Oxidation over Supported Noble Metal Catalysts. Catal. Today 2009, 146, 253–259. [Google Scholar] [CrossRef]
- Martínez-Munuera, J.C.; Giménez-Mañogil, J.; Yeste, M.P.; Hungría, A.B.; Cauqui, M.A.; García-García, A.; Calvino, J.J. New Findings Regarding the Role of Copper Entity Particle Size on the Performance of Cu/Ceria-Based Catalysts in the CO-PROX Reaction. Appl. Surf. Sci. 2022, 575, 151717. [Google Scholar] [CrossRef]
- Wu, Z.; Zhu, H.; Qin, Z.; Wang, H.; Ding, J.; Huang, L.; Wang, J. CO Preferential Oxidation in H2-Rich Stream over a CuO/CeO2 Catalyst with High H2O and CO2 Tolerance. Fuel 2013, 104, 41–45. [Google Scholar] [CrossRef]
- Chagas, C.A.; Schmal, M. The Effect of Copper Oxide on the CuO–NiO/CeO2 Structure and Its Influence on the CO-PROX Reaction. Int. J. Hydrogen Energy 2022, 47, 8858–8866. [Google Scholar] [CrossRef]
- Araújo, V.D.; Bellido, J.D.A.; Bernardi, M.I.B.; Assaf, J.M.; Assaf, E.M. CuO–CeO2 Catalysts Synthesized in One-Step: Characterization and PROX Performance. Int. J. Hydrogen Energy 2012, 37, 5498–5507. [Google Scholar] [CrossRef]
- Silva, R.B.M.; de Oliveira, C.S.; Teixeira-Neto, É.; Sigoli, F.A.; Mazali, I.O. Improvement of PROX-CO Catalytical Performance by Modulation of the Pore Structure of CeO2 Nanorods Decorated with Au Nanoparticles. Microporous Mesoporous Mater. 2022, 330, 111574. [Google Scholar] [CrossRef]
- Konsolakis, M. The Role of Copper–Ceria Interactions in Catalysis Science: Recent Theoretical and Experimental Advances. Appl. Catal. B Environ. 2016, 198, 49–66. [Google Scholar] [CrossRef]
- Martínez-Arias, A.; Hungría, A.B.; Fernández-García, M.; Conesa, J.C.; Munuera, G. Preferential Oxidation of CO in a H2-Rich Stream over CuO/CeO2 and CuO/(Ce,M)Ox (M = Zr, Tb) Catalysts. J. Power Sources 2005, 151, 32–42. [Google Scholar] [CrossRef]
- Hermes, E.D.; Jenness, G.R.; Schmidt, J.R. Decoupling the Electronic, Geometric and Interfacial Contributions to Support Effects in Heterogeneous Catalysis. Mol. Simul. 2015, 41, 123–133. [Google Scholar] [CrossRef]
- Colón, G.; Valdivieso, F.; Pijolat, M.; Baker, R.T.; Calvino, J.J.; Bernal, S. Textural and Phase Stability of CexZr1−xO2 Mixed Oxides under High Temperature Oxidising Conditions. Catal. Today 1999, 50, 271–284. [Google Scholar] [CrossRef]
- Janvier, C.; Pijolat, M.; Valdivieso, F.; Soustelle, M.; Zing, C. Thermal Stability of Ce1−xZrxO2 Solid Solution Powders. J. Eur. Ceram. Soc. 1998, 18, 1331–1337. [Google Scholar] [CrossRef]
- Fornasiero, P.; Balducci, G.; Di Monte, R.; Kašpar, J.; Sergo, V.; Gubitosa, G.; Ferrero, A.; Graziani, M. Modification of the Redox Behaviour of CeO2 Induced by Structural Doping with ZrO2. J. Catal. 1996, 164, 173–183. [Google Scholar] [CrossRef]
- Di Monte, R.; Fornasiero, P.; Kaspar, J.; Graziani, M.; Gatica, J.M.; Bernal, S.; Gomez-Herrero, A. Stabilisation of Nanostructured Ce0.2Zr0.8O2 Solid Solution by Impregnation on Al2O3: A Suitable Method for the Production of Thermally Stable Oxygen Storage/Release Promoters for Three-Way Catalysts. Chem. Commun. 2000, 21, 2167–2168. [Google Scholar] [CrossRef]
- Ozawa, M.; Kimura, M.; Isogai, A. The Application of Ce-Zr Oxide Solid Solution to Oxygen Storage Promoters in Automotive Catalysts. J. Alloys Compd. 1993, 193, 1–2. [Google Scholar] [CrossRef]
- Kozlov, A.I.; Do, H.K.; Yezerets, A.; Andersen, P.; Kung, H.H.; Kung, M.C. Effect of Preparation Method and Redox Treatment on the Reducibility and Structure of Supported Ceria–Zirconia Mixed Oxide. J. Catal. 2002, 209, 417–426. [Google Scholar] [CrossRef]
- Qiu, Z.; Guo, X.; Mao, J.; Zhou, R. Elucidating the Structure, Redox Properties and Active Entities of High-Temperature Thermally Aged CuOx–CeO2 Catalysts for CO-PROX. Phys. Chem. Chem. Phys. 2021, 23, 15582–15590. [Google Scholar] [CrossRef] [PubMed]
- Qiu, Z.; Guo, X.; Mao, J.; Zhou, R. Insights into the Structure-Performance Relationship of CuOx-CeO2 Catalysts for Preferential Oxidation of CO: Investigation on Thermally Induced Copper Migration Process. Appl. Surf. Sci. 2022, 600, 154100. [Google Scholar] [CrossRef]
- Chen, Y.-Z.; Liaw, B.-J.; Chen, H.-C. Selective Oxidation of CO in Excess Hydrogen over CuO/CexZr1-xO2 Catalysts. Int. J. Hydrogen Energy 2006, 31, 427–435. [Google Scholar] [CrossRef]
- Chen, Y.Z.; Liaw, B.J.; Huang, C.W. Selective Oxidation of CO in Excess Hydrogen over CuO/CexSn1-xO2 Catalysts. Appl. Catal. A Gen. 2006, 302, 168–176. [Google Scholar] [CrossRef]
- Iglesias-González, A.; Ayastuy, J.L.; González-Marcos, M.P.; Gutiérrez-Ortiz, M.A. CuO/CexSn1-xO2 Catalysts with Low Tin Content for CO Removal from H2-Rich Streams. Int. J. Hydrogen Energy 2014, 39, 5213–5224. [Google Scholar] [CrossRef]
- Ayastuy, J.L.; Iglesias-González, A.; Gutiérrez-Ortiz, M.A. Synthesis and Characterization of Low Amount Tin-Doped Ceria (CexSn1-xO2−δ) for Catalytic CO Oxidation. Chem. Eng. J. 2014, 244, 372–381. [Google Scholar] [CrossRef]
- Kaplin, I.Y.; Lokteva, E.S.; Tikhonov, A.V.; Zhilyaev, K.A.; Golubina, E.V.; Maslakov, K.I.; Kamaev, A.O.; Isaikina, O.Y. Templated Synthesis of Copper Modified Tin-Doped Ceria for Catalytic CO Oxidation. Top. Catal. 2020, 63, 86–98. [Google Scholar] [CrossRef]
- Maciel, C.G.; Silva, T.D.F.; Hirooka, M.I.; Belgacem, M.N.; Assaf, J.M. Effect of Nature of Ceria Support in CuO/CeO2 Catalyst for PROX-CO Reaction. Fuel 2012, 97, 245–252. [Google Scholar] [CrossRef]
- Jayakumar, G.; Albert Irudayaraj, A.; Dhayal Raj, A. A Comprehensive Investigation on the Properties of Nanostructured Cerium Oxide. Opt. Quantum Electron. 2019, 51, 1–15. [Google Scholar] [CrossRef]
- Loridant, S. Raman Spectroscopy as a Powerful Tool to Characterize Ceria-Based Catalysts. Catal. Today 2021, 373, 98–111. [Google Scholar] [CrossRef]
- Gouadec, G.; Colomban, P. Raman Spectroscopy of Nanomaterials: How Spectra Relate to Disorder, Particle Size and Mechanical Properties. Prog. Cryst. Growth Charact. Mater. 2007, 53, 1–56. [Google Scholar] [CrossRef]
- Sudarsanam, P.; Hillary, B.; Amin, M.H.; Rockstroh, N.; Bentrup, U.; Brückner, A.; Bhargava, S.K. Heterostructured Copper-Ceria and Iron-Ceria Nanorods: Role of Morphology, Redox, and Acid Properties in Catalytic Diesel Soot Combustion. Langmuir 2018, 34, 2663–2673. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Li, M.; Howe, J.; Meyer, H.M.; Overbury, S.H. Probing Defect Sites on CeO2 Nanocrystals with Well-Defined Surface Planes by Raman Spectroscopy and O2 Adsorption. Langmuir 2010, 26, 16595–16606. [Google Scholar] [CrossRef]
- Choudhury, B.; Choudhury, A. Ce3+ and Oxygen Vacancy Mediated Tuning of Structural and Optical Properties of CeO2 Nanoparticles. Mater. Chem. Phys. 2012, 131, 666–671. [Google Scholar] [CrossRef]
- Kaplin, I.Y.; Lokteva, E.S.; Maslakov, K.I.; Tikhonov, A.V.; Kharlanov, A.N.; Fionov, A.V.; Kamaev, A.O.; Isaikina, O.Y.; Maksimov, S.V.; Golubina, E.V. Ceria-Silica Mesoporous Catalysts for CO Preferential Oxidation in H2-Rich Stream: The Effect of Ce:Si Ratio and Copper Modification. Appl. Surf. Sci. 2022, 594, 153473. [Google Scholar] [CrossRef]
- Sedmak, G.; Hočevar, S.; Levec, J. Transient Kinetic Model of CO Oxidation over a Nanostructured Cu0.1Ce0.9O2−y Catalyst. J. Catal. 2004, 222, 87–99. [Google Scholar] [CrossRef]
- Liu, W.; Flytzani-Stephanopoulos, M. Total Oxidation of Carbon-Monoxide and Methane over Transition Metal Fluorite Oxide Composite Catalysts. II. Catalyst Characterization and Reaction-Kinetics. J. Catal. 1995, 153, 317–332. [Google Scholar] [CrossRef]
- Miranda Cruz, A.R.; Assaf, E.M.; Gomes, J.F.; Assaf, J.M. Active Copper Species of Co-Precipitated Copper-Ceria Catalysts in the CO-PROX Reaction: An in Situ XANES and DRIFTS Study. Catal. Today 2020, 381, 42–49. [Google Scholar] [CrossRef]
- Wang, W.-W.; Du, P.-P.; Zou, S.-H.; He, H.-Y.; Wang, R.-X.; Jin, Z.; Shi, S.; Huang, Y.-Y.; Si, R.; Song, Q.-S.; et al. Highly Dispersed Copper Oxide Clusters as Active Species in Copper-Ceria Catalyst for Preferential Oxidation of Carbon Monoxide. ACS Catal. 2015, 5, 2088–2099. [Google Scholar] [CrossRef]
- Wang, J.; Chen, H.; Hu, Z.; Yao, M.; Li, Y. A Review on the Pd-Based Three-Way Catalyst. Catal. Rev.-Sci. Eng. 2015, 57, 79–144. [Google Scholar] [CrossRef]
- Martínez-Arias, A.; Gamarra, D.; Fernández-García, M.; Hornés, A.; Bera, P.; Koppány, Z.; Schay, Z. Redox-Catalytic Correlations in Oxidised Copper-Ceria CO-PROX Catalysts. Catal. Today 2009, 143, 211–217. [Google Scholar] [CrossRef]
- Wang, W.W.; Yu, W.Z.; Du, P.P.; Xu, H.; Jin, Z.; Si, R.; Ma, C.; Shi, S.; Jia, C.J.; Yan, C.H. Crystal Plane Effect of Ceria on Supported Copper Oxide Cluster Catalyst for CO Oxidation: Importance of Metal-Support Interaction. ACS Catal. 2017, 7, 1313–1329. [Google Scholar] [CrossRef]
- Tang, X.; Zhang, B.; Li, Y.; Xu, Y.; Xin, Q.; Shen, W. Carbon Monoxide Oxidation over CuO/CeO2 Catalysts. Catal. Today 2004, 93–95, 191–198. [Google Scholar] [CrossRef]
- Holgado, J.P.; Munuera, G.; Espinós, J.P.; González-Elipe, A.R. XPS Study of Oxidation Processes of CeOx Defective Layers. Appl. Surf. Sci. 2000, 158, 164–171. [Google Scholar] [CrossRef]
- Liu, W.; Sarofim, A.F.; Flytzani-Stephanopoulosi, M. Complete Oxidation of Carbon Monoxide and Methane over Metal-Promoted Fluorite Oxide Catalysts. Chem. Eng. Sci. 1994, 49, 4871–4888. [Google Scholar] [CrossRef]
- He, D.; Liu, L.S.; Ren, J.; Hu, T.P. Catalytic Combustion of Volatile Organic Compounds over CuO-CeO2 Supported on SiO2-Al2O3 Modified Glass-Fiber Honeycomb. J. Fuel Chem. Technol. 2017, 45, 354–361. [Google Scholar] [CrossRef]
- Yu, Q.; Liu, L.; Dong, L.; Li, D.; Liu, B.; Gao, F.; Sun, K.; Dong, L.; Chen, Y. Effects of Ce/Zr Ratio on the Reducibility, Adsorption and Catalytic Activity of CuO/CexZr1−xO2/Al2O3 Catalysts for NO Reduction by CO. Appl. Catal. B Environ. 2010, 96, 350–360. [Google Scholar] [CrossRef]
- Lan, F.; Wang, X.; Xu, X.; Zhang, R.; Zhang, N. Preparation and Characterization of SnO2 Catalysts for CO and CH4 Oxidation. React. Kinet. Mech. Catal. 2012, 106, 113–125. [Google Scholar] [CrossRef]
- Wang, H.; Wang, H.; Li, X.; Li, C. Nature of Active Tin Species and Promoting Effect of Nickle in Silica Supported Tin Oxide for Dehydrogenation of Propane. Appl. Surf. Sci. 2017, 407, 456–462. [Google Scholar] [CrossRef]
- Nyathi, T.M.; Fischer, N.; York, A.P.E.; Claeys, M. Environment-Dependent Catalytic Performance and Phase Stability of Co3O4 in the Preferential Oxidation of Carbon Monoxide Studied in-situ. ACS Catal. 2020, 10, 11892–11911. [Google Scholar] [CrossRef]
- Davó-Quiñonero, A.; Bailón-García, E.; López-Rodríguez, S.; Juan-Juan, J.; Lozano-Castelló, D.; García-Melchor, M.; Herrera, F.C.; Pellegrin, E.; Escudero, C.; Bueno-López, A. Insights into the Oxygen Vacancy Filling Mechanism in CuO/CeO2 Catalysts: A Key Step toward High Selectivity in Preferential CO Oxidation. ACS Catal. 2020, 10, 6532–6545. [Google Scholar] [CrossRef]
- Slavinskaya, E.M.; Zadesenets, A.V.; Stonkus, O.A.; Stadnichenko, A.I.; Shchukarev, A.V.; Shubin, Y.V.; Korenev, S.V.; Boronin, A.I. Thermal Activation of Pd/CeO2-SnO2 Catalysts for Low-Temperature CO Oxidation. Appl. Catal. B Environ. 2020, 277, 119275. [Google Scholar] [CrossRef]
- Kaplin, I.Y.; Lokteva, E.S.; Golubina, E.V.; Shishova, V.V.; Maslakov, K.I.; Fionov, A.V.; Isaikina, O.Y.; Lunin, V.V. Efficiency of Manganese Modified CTAB-Templated Ceria-Zirconia Catalysts in Total CO Oxidation. Appl. Surf. Sci. 2019, 485, 432–440. [Google Scholar] [CrossRef]




| Sample | SBET, m2/g | Average CeO2 Crystallite Size, nm | XPS Atomic and Ion Ratios | H2 Uptake, μmol/g | ||||
|---|---|---|---|---|---|---|---|---|
| Ce/Cu | Ce/Sn(Zr) | Ce3+/Ce4+ | Cu+/Cu2+ | Low T Range (80–400 °C) | Medium T Range (400–650 °C) | |||
| 20CuCeSn | 149 ± 15 | 5 ± 1 | 5.8 | 6.3 | 0.11 | 0.70 | 1479 | 100 |
| 20CuCeZr | 28 ± 3 | 10 ± 1 | 4.0 | 9.7 | 0.11 | 0.20 | 1417 | 159 |
| 20CuCeSn-im | 81 ± 8 | 12 ± 1 | 2.9 | 20.0 | 0.04 | 0.18 | 1248 | 85 |
| 20CuCeZr-im | 27 ± 3 | 15 ± 1 | 2.2 | 10.3 | 0.07 | 0.39 | 933 | 136 |
| Sample | AAS Copper Content, wt.% | SBET, m2/g | SEM-EDX Atomic Ratio | XPS Atomic and Ion Ratios | ||
|---|---|---|---|---|---|---|
| Ce/Cu | Ce/Cu | Ce3+/Ce4+ | Cu+/Cu2+ | |||
| 5CuCeSn | 1.8 | 145 ± 15 | 24.0 | 20.6 | 0.27 | 0.72 |
| 10CuCeSn | 3.4 | 183 ± 18 | 12.0 | 13.0 | 0.22 | 1.08 |
| 20CuCeSn | 6.1 | 149 ± 15 | 5.6 | 5.8 | 0.11 | 0.70 |
| 25CuCeSn | 7.5 | 155 ± 16 | 5.2 | 4.5 | 0.11 | 0.81 |
| Sample | Hydrogen Uptake, μmol/g | ∆Tαβ 1, °C | H2/Cu, mol. Ratio (from AAS) 2 | ||
|---|---|---|---|---|---|
| Low-Temperature Range (80−400 °C) | Medium-Temperature Range (400−650 °C) | Total | |||
| 5CuCeSn | 977 | 92 | 1069 | 70 | 3.7 |
| 10CuCeSn | 1213 | 242 | 1455 | 76 | 2.8 |
| 20CuCeSn | 1479 | 100 | 1579 | 100 | 1.9 |
| 25CuCeSn | 1697 | 105 | 1802 | 113 | 1.8 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kaplin, I.Y.; Lokteva, E.S.; Tikhonov, A.V.; Maslakov, K.I.; Isaikina, O.Y.; Golubina, E.V. Copper–Cerium–Tin Oxide Catalysts for Preferential Oxidation of CO in Hydrogen: Effects of Synthesis Method and Copper Content. Catalysts 2022, 12, 1575. https://doi.org/10.3390/catal12121575
Kaplin IY, Lokteva ES, Tikhonov AV, Maslakov KI, Isaikina OY, Golubina EV. Copper–Cerium–Tin Oxide Catalysts for Preferential Oxidation of CO in Hydrogen: Effects of Synthesis Method and Copper Content. Catalysts. 2022; 12(12):1575. https://doi.org/10.3390/catal12121575
Chicago/Turabian StyleKaplin, Igor Yu., Ekaterina S. Lokteva, Artem V. Tikhonov, Konstantin I. Maslakov, Oksana Ya. Isaikina, and Elena V. Golubina. 2022. "Copper–Cerium–Tin Oxide Catalysts for Preferential Oxidation of CO in Hydrogen: Effects of Synthesis Method and Copper Content" Catalysts 12, no. 12: 1575. https://doi.org/10.3390/catal12121575
APA StyleKaplin, I. Y., Lokteva, E. S., Tikhonov, A. V., Maslakov, K. I., Isaikina, O. Y., & Golubina, E. V. (2022). Copper–Cerium–Tin Oxide Catalysts for Preferential Oxidation of CO in Hydrogen: Effects of Synthesis Method and Copper Content. Catalysts, 12(12), 1575. https://doi.org/10.3390/catal12121575








