Nanosized Cu-SSZ-13 and Its Application in NH3-SCR
Abstract
1. Introduction
2. Results and Discussion
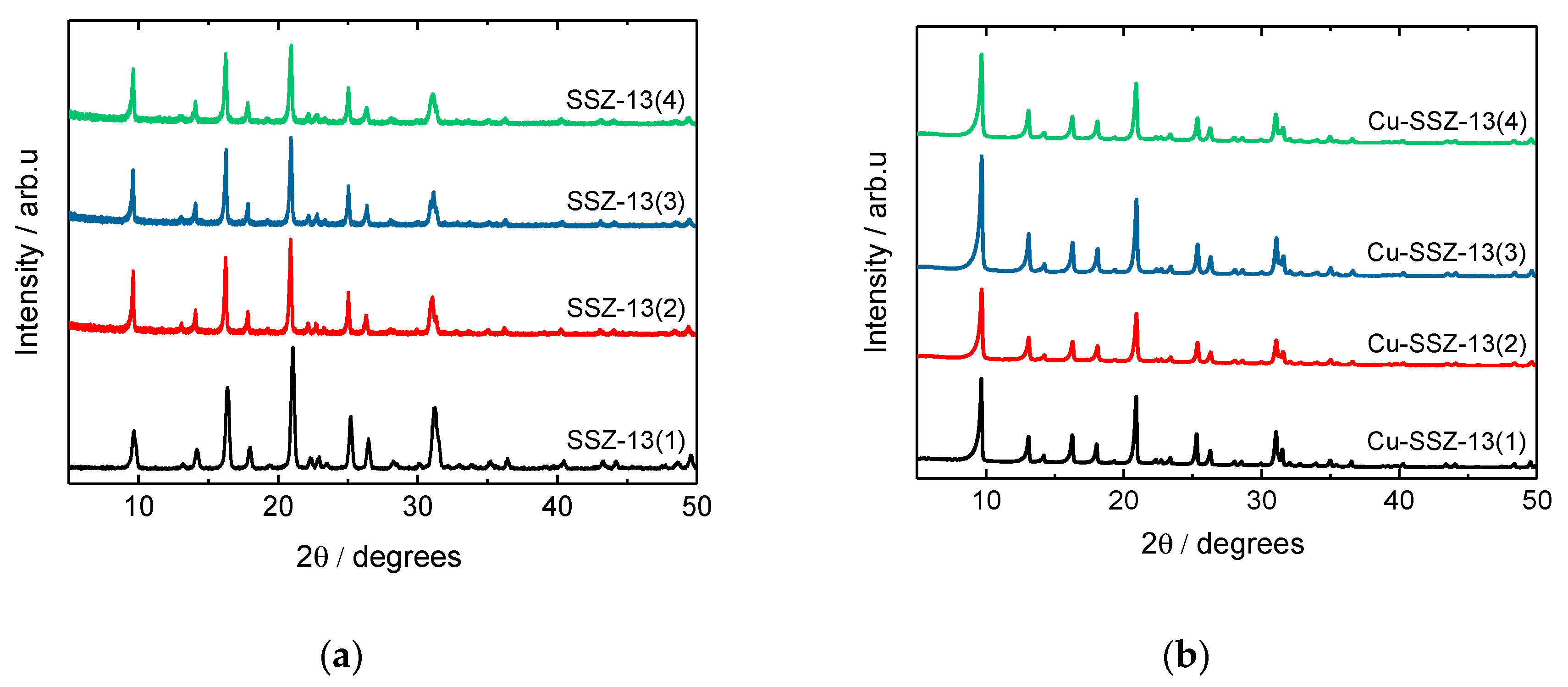
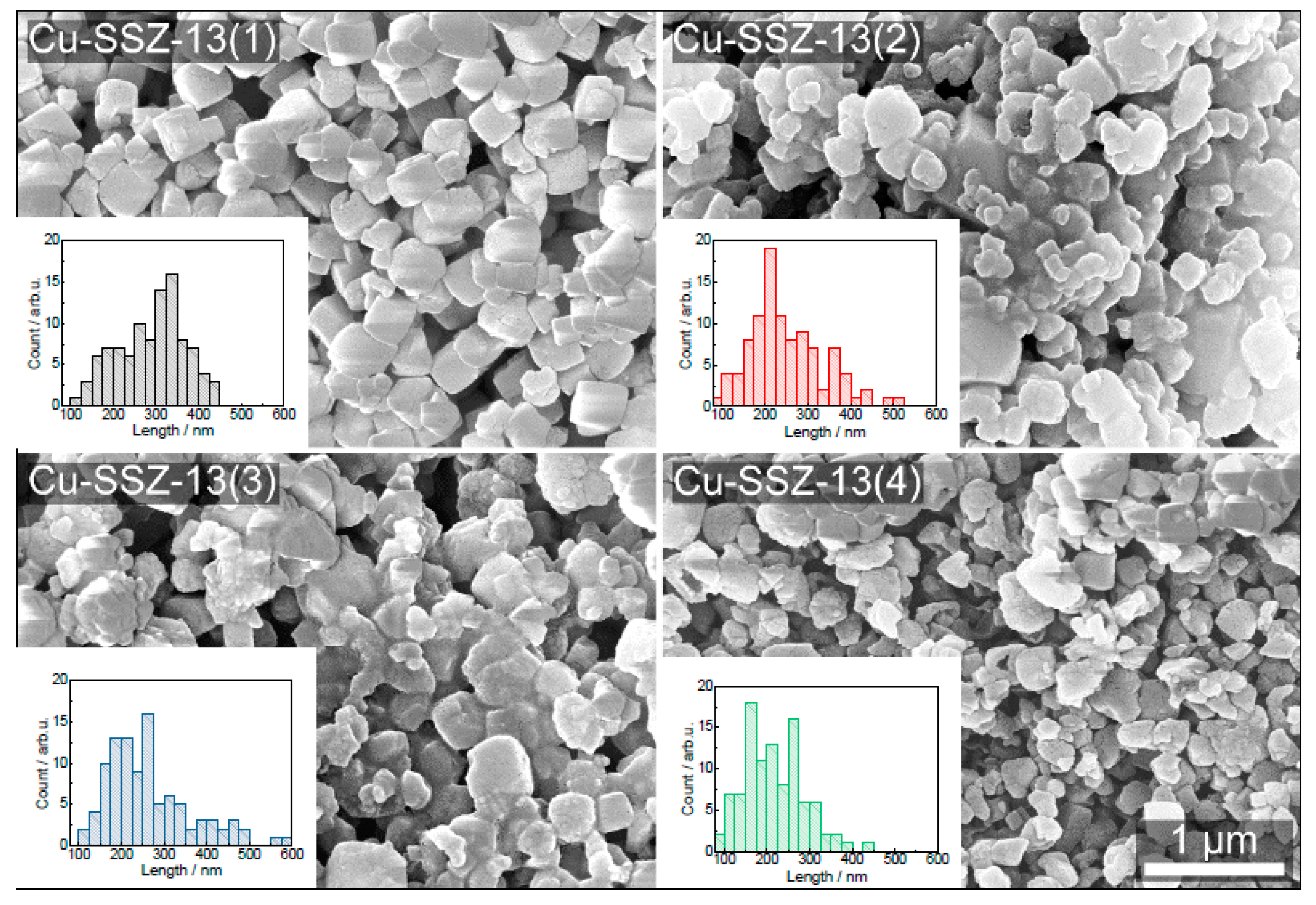
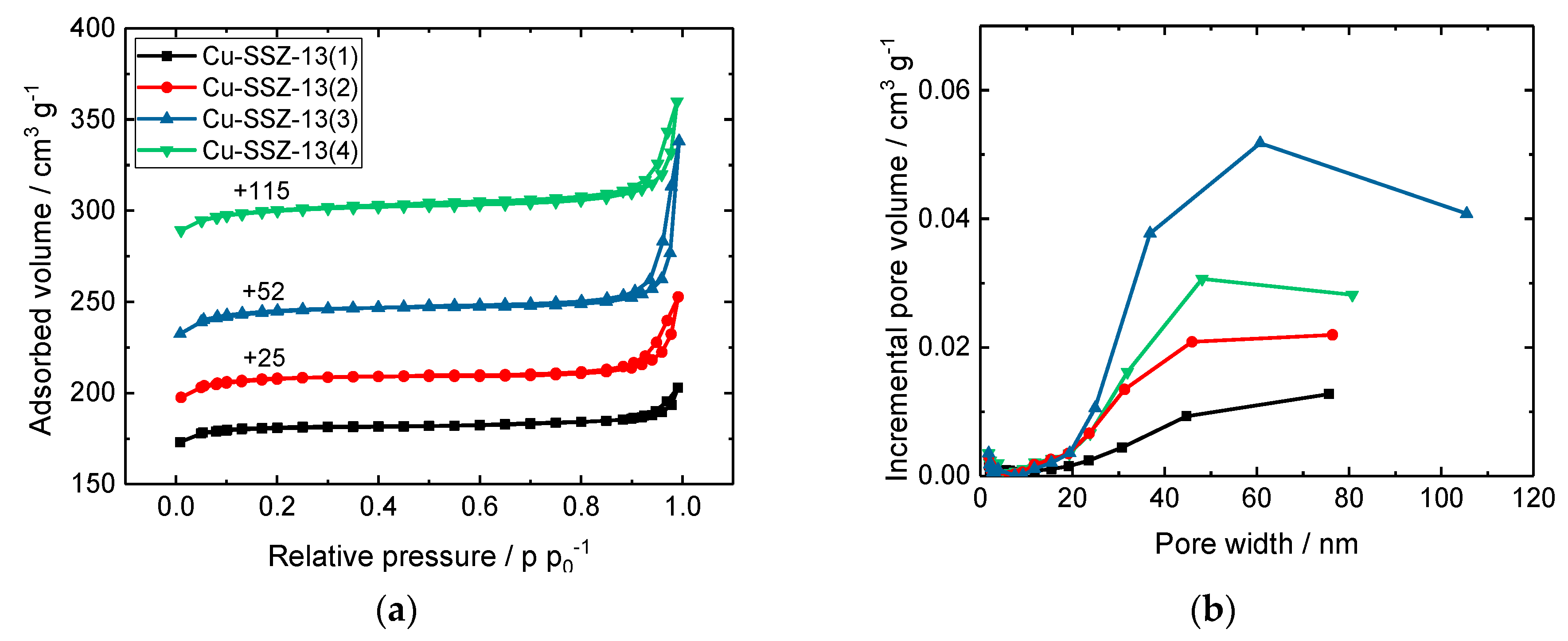
2.1. Structural and Textural Properties, Morphology of Cu-SSZ-13
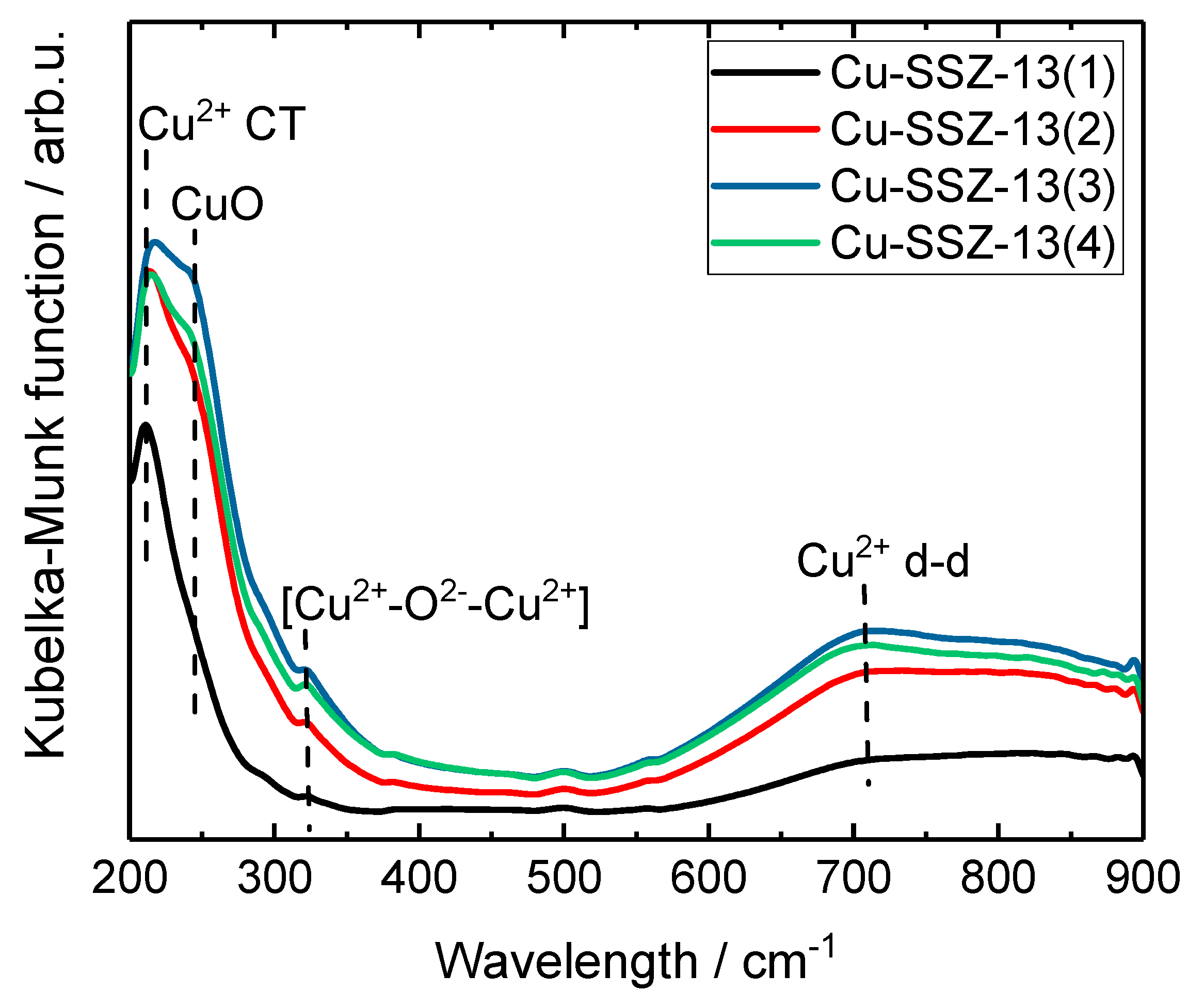
2.2. Status of Copper Oxide Species
2.3. Catalytic SCR Studies
3. Materials and Methods
3.1. Catalyst Preparation
3.2. Catalyst Characterization
3.3. Catalyst Experiments
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Baerlocher, C.; McCusker, L.B.; Olson, D.H. Atlas of Zeolite Framework Types; Elsevier: Amsterdam, The Netherlands, 2007. [Google Scholar]
- Gao, F.; Walter, E.D.; Washton, N.M.; Szanyi, J.; Peden, C.H.F. Synthesis and evaluation of Cu-SAPO-34 catalysts for ammonia selective catalytic reduction. 1. Aqueous solution ion exchange. ACS Catal. 2013, 3, 2083–2093. [Google Scholar] [CrossRef]
- Deka, U.; Lezcano-Gonzalez, I.; Warrender, S.J.; Lorena, A.; Paul, P.; Wright, A.; Weckhuysen, B.M.; Beale, A.M. Changing active sites in Cu–CHA catalysts: deNOx selectivity as a function of the preparation method. Micro. Meso. Mater. 2013, 166, 144–152. [Google Scholar] [CrossRef]
- Wang, D.; Gao, F.; Peden, C.H.F.; Li, J.; Kamasamudram, K.; Epling, W.S. Selective catalytic reduction of NOx with NH3 over a Cu-SSZ-13 catalyst prepared by a solid-state ion-exchange method. ChemCatChem 2014, 6, 1579–1583. [Google Scholar] [CrossRef]
- Gao, F.; Walter, E.D.; Washton, N.M.; Szanyi, J.; Peden, C.H.F. Synthesis and evaluation of Cu/SAPO-34 catalysts for NH3-SCR 2: Solid-state ion exchange and one-pot synthesis. Appl. Catal. B Environ. 2015, 162, 501–514. [Google Scholar] [CrossRef]
- Yan, C.; Cheng, H.; Yuan, Z.; Wang, S. The role of isolated Cu2+ location in structural stability of Cu-modified SAPO-34 in NH3-SCR of NO. Environ. Technol. 2015, 36, 169–177. [Google Scholar] [CrossRef]
- Xue, J.; Wang, X.; Qi, G.; Wang, J.; Shen, M.; Li, W. Characterization of copper species over Cu/SAPO-34 in selective catalytic reduction of NOx with ammonia: Relationships between active Cu sites and de-NOx performance at low temperature. J. Catal. 2013, 297, 56–64. [Google Scholar] [CrossRef]
- Wang, L.; Li, W.; Qi, G.; Weng, D. Location and nature of Cu species in Cu/SAPO-34 for selective catalytic reduction of NO with NH3. J. Catal. 2012, 289, 21–29. [Google Scholar] [CrossRef]
- Deka, U.; Juhin, A.; Eilertsen, E.A.; Emerich, H.; Green, M.A.; Korhonen, S.T.; Weckhuysen, B.M.; Beale, A.M. Confirmation of Isolated Cu2+ Ions in SSZ-13 Zeolite as Active Sites in NH3-Selective Catalytic Reduction. J. Phys. Chem. C 2012, 116, 4809–4818. [Google Scholar] [CrossRef]
- Fickel, D.W.; Lobo, R.F. Copper coordination in Cu-SSZ-13 and Cu-SSZ-16 investigated by variable-temperature XRD. J. Phys. Chem. C 2010, 114, 1633–1640. [Google Scholar] [CrossRef]
- Kwak, J.H.; Zhu, H.; Lee, J.H.; Peden, C.H.F.; Szanyi, J. Two different cationic positions in Cu-SSZ-13? Chem. Commun. 2012, 48, 4758–4760. [Google Scholar] [CrossRef]
- McEwen, J.-S.; Anggara, T.; Schneider, W.F.; Kispersky, V.F.; Miller, J.T.; Delgass, W.N.; Ribeiro, F.H. Integrated operando X-ray absorption and DFT characterization of Cu-SSZ-13 exchange sites during the selective catalytic reduction of NOx with NH3. Catal. Today 2012, 184, 129–144. [Google Scholar] [CrossRef]
- Xie, L.; Liu, F.; Ren, L.; Shi, X.; Xiao, F.-S.; He, H. Excellent performance of one-pot synthesized Cu-SSZ-13 catalyst for the selective catalytic reduction of NOx with NH3. Environ. Sci. Technol. 2014, 48, 566–572. [Google Scholar] [CrossRef]
- Ren, L.; Zhu, L.; Yang, C.; Chen, Y.; Sun, Q.; Zhang, H.; Li, C.; Nawaz, F.; Meng, X.; Xiao, F.-S. Designed copper–amine complex as an efficient template for one-pot synthesis of Cu-SSZ-13 zeolite with excellent activity for selective catalytic reduction of NOx by NH3. Chem. Commun. 2011, 47, 9789–9791. [Google Scholar] [CrossRef]
- Valtchev, V.; Lubomira Tosheva, L. Porous nanosized particles: Preparation, properties, and applications. Chem. Rev. 2013, 113, 6734–6760. [Google Scholar] [CrossRef]
- Gao, F.; Walter, E.D.; Karp, E.M.; Luo, J.; Tonkyn, R.G.; Kwak, J.H.; Szanyi, J.; Peden, C.H.F. Structure-activity relationships in NH3-SCR over Cu-SSZ-13 as probed by reaction kinetics and EPR studies. J. Catal. 2013, 300, 20–29. [Google Scholar] [CrossRef]
- Wang, J.; Shao, L.; Wang, C.; Wang, J.; Shen, M.; Li, W. Controllable preparation of various crystal size and nature of intra-crystalline diffusion in Cu/SSZ-13 NH3-SCR catalysts. J. Catal. 2018, 367, 221–228. [Google Scholar] [CrossRef]
- Bates, S.A.; Verma, A.A.; Paolucci, C.; Parekh, A.A.; Anggara, T.; Yezerets, A.; Schneider, W.F.; Miller, J.T.; Delgass, W.N.; Ribeiro, F.H. Identification of the active Cu site in standard selective catalytic reduction with ammonia on Cu-SSZ-13. J. Catal. 2014, 312, 87–97. [Google Scholar] [CrossRef]
- Prodinger, S.; Derewinski, M.A.; Wang, Y.; Washton, N.M.; Walter, E.D.; Szanyi, J.; Gao, F.; Wang, Y.; Peden, C.H.F. Sub-micron Cu/SSZ-13: Synthesis and application as selective catalytic reduction (SCR) catalysts. Appl. Catal. B Environ. 2017, 201, 461–469. [Google Scholar] [CrossRef]
- Wang, D.; Zhang, L.; Li, J.; Kamasamudram, K.; Epling, W.S. NH3-SCR over Cu/SAPO-34–Zeolite acidity and Cu structure changes as a function of Cu loading. Catal. Today 2014, 231, 64–74. [Google Scholar] [CrossRef]
- Fan, S.; Xue, J.; Yu, T.; Fan, D.; Hao, T.; Shen, M.; Li, W. The effect of synthesis methods on Cu species and active sites over Cu/SAPO-34 for NH3-SCR reaction. Catal. Sci. Technol. 2013, 3, 2357–2364. [Google Scholar] [CrossRef]
- Di Iorio, J.R.; Gounder, R. Controlling the isolation and pairing of aluminum in chabazite zeolites using mixtures of organic and inorganic structure-directing agents. Chem. Mater. 2016, 28, 2236–2247. [Google Scholar] [CrossRef]
- Sierka, M.; Eichler, U.; Datka, J.; Sauer, J. Heterogeneity of Brønsted acidic sites in faujasite type zeolites due to aluminum content and framework structure. J. Phys. Chem. B 1998, 102, 6397–6404. [Google Scholar] [CrossRef]
- Li, S.; Li, H.; Gounder, R.; Debellis, A.; Müller, I.B.; Prasad, S.; Moini, A.; Schneider, W.F. First-principles comparison of proton and divalent copper cation exchange energy landscapes in SSZ-13 zeolite. J. Phys. Chem. C 2018, 122, 23564–23573. [Google Scholar] [CrossRef]
- Van Oers, C.J.; Góra-Marek, K.; Prelot, B.; Datka, J.; Meynen, V.; Cool, P. Demonstrating the benefits and pitfalls of various acidity characterization techniques by a case study on bimodal aluminosilicates. Langmuir 2014, 30, 1880–1887. [Google Scholar] [CrossRef]
- Mapes, J.E.; Eischens, R.P. The infrared spectra of ammonia chemisorbed on cracking catalysts. J. Phys. Chem. 1954, 78, 1059–1062. [Google Scholar] [CrossRef]
- Palčić, A.; Ordomsky, V.V.; Qin, Z.; Georgieva, V.; Valtchev, V. Tuning zeolite properties for a highly efficient synthesis of propylene from methanol. Chem. Eur. J. 2018, 24, 13136–13149. [Google Scholar] [CrossRef]
- Thommes, M.; Kaneko, K.; Neimark, A.V.; Olivier, J.P.; Rodriguez-Reinoso, F.; Rouquerol, J.; Sing, K.S.W. Physisorption of gases, with special reference to the evaluation of surface area and pore size distribution (IUPAC Technical Report). Pure Appl. Chem. 2015, 87, 1051–1069. [Google Scholar] [CrossRef]
- Usui, T.; Liu, Z.; Ibe, S.; Zhu, J.; Anand, C.; Igarashi, H.; Onaya, N.; Sasaki, Y.; Shiramata, Y.; Kusamoto, T.; et al. Improve the Hydrothermal Stability of Cu-SSZ-13 Zeolite Catalyst by Loading a Small Amount of Ce. ACS Catal. 2018, 8, 9165–9173. [Google Scholar] [CrossRef]
- Leistner, K.; Mihai, O.; Wijayanti, K.; Kumar, A.; Kamasamudram, K.; Currier, N.W.; Yezerets, A.; Olsson, L. Comparison of Cu/BEA, Cu/SSZ-13 and Cu/SAPO-34 for ammonia-SCR reactions. Catal. Today 2015, 258, 49–55. [Google Scholar] [CrossRef]
- Gomes, S.R.; Bion, N.; Blanchard, G.; Rousseau, S.; Bellière-Baca, V.; Harlé, V.; Duprez, D.; Epron, F. Thermodynamic and experimental studies of catalytic reforming of exhaust gas recirculation in gasoline engines. Appl. Catal. B Environ. 2011, 102, 44–53. [Google Scholar] [CrossRef]
- Park, J.-H.; Park, H.J.; Baik, J.H.; Nam, I.-S.; Shin, C.-H.; Lee, J.-H.; Cho, B.K.; Oh, S.H. Hydrothermal stability of CuZSM5 catalyst in reducing NO by NH3 for the urea selective catalytic reduction process. J. Catal. 2006, 240, 47–57. [Google Scholar] [CrossRef]
- Bin, F.; Song, C.; Lv, G.; Song, J.; Wu, S.; Li, X. Selective catalytic reduction of nitric oxide with ammonia over zirconium-doped copper/ZSM-5 catalysts. Appl. Catal. B Environ. 2014, 150–151, 532–543. [Google Scholar] [CrossRef]
- Lezcano-Gonzalez, I.; Deka, U.; van der Bij, H.E.; Paalanen, P.; Weckhuysen, B.M.; Beale, A.M. Chemical deactivation of Cu-SSZ-13 ammonia selective catalytic reduction (NH3-SCR) systems. Appl. Catal. B Environ. 2014, 154–155, 339–349. [Google Scholar] [CrossRef]
- Li, L.; Zhang, F.; Guan, N.; Richter, M.; Fricke, R. Selective catalytic reduction of NO by propane in excess oxygen over IrCu-ZSM-5 catalyst. Catal. Comm. 2007, 8, 583–588. [Google Scholar] [CrossRef]
- Han, M.-J.; Jiao, Y.-L.; Zhou, C.-H.; Guo, Y.-L.; Guo, Y.; Lu, G.-Z.; Wang, L.; Zhan, W.-C. Catalytic activity of Cu–SSZ-13 prepared with different methods for NH3-SCR reaction. Rare Metals 2019, 38, 210–220. [Google Scholar] [CrossRef]
- Yashnik, S.; Ismagilov, Z. Cu-substituted ZSM-5 catalyst: Controlling of DeNOx reactivity via ion-exchange mode with copper–ammonia solution. Appl. Catal. B Environ. 2015, 170, 241–254. [Google Scholar] [CrossRef]
- Deka, U.; Lezcano-Gonzalez, I.; Weckhuysen, B.M.; Beale, A.M. Local environment and nature of Cu active sites in zeolite-based catalysts for the selective catalytic reduction of NOx. ACS Catal. 2013, 3, 413–427. [Google Scholar] [CrossRef]
- Verma, A.; Bates, S.A.; Anggara, T.; Paolucci, C.; Parekh, A.A.; Kamasamudram, K.; Yezerets, A.; Miller, J.T.; Delgass, W.N.; Schneider, W.F.; et al. NO oxidation: A probe reaction on Cu-SSZ-13. J. Catal. 2014, 312, 179–190. [Google Scholar] [CrossRef]
- Hadjiivanov, K.; Dimitrov, L. IR spectroscopy study of CO and NOx adsorption on a Cu/Zr-HMS catalyst. Micro. Meso. Mater. 1999, 27, 49–56. [Google Scholar] [CrossRef]
- Padley, M.B.; Rochester, C.H.; Hutchings, G.J.; King, F. FTIR spectroscopic study of thiophene, SO2, and CO adsorption on Cu/Al2O3 catalysts. J. Catal. 1994, 148, 438–452. [Google Scholar] [CrossRef]
- Góra-Marek, K.; Palomares, A.E.; Glanowska, A.; Sadowska, K.; Datka, J. Copper sites in zeolites-quantitative IR studies. Micropor. Mesopor. Mater. 2012, 162, 175–180. [Google Scholar] [CrossRef]
- Bordiga, S.; Lamberti, C.; Bonino, F.; Travert, A.; Thibault-Starzyk, F. Probing zeolites by vibrational spectroscopies. Chem. Soc. Rev. 2015, 44, 7262–7341. [Google Scholar] [CrossRef] [PubMed]
- Zamadics, M.; Chen, X.; Kevan, L. Study of copper(II) location and adsorbate interaction in CuH-SAPO-34 molecular sieve by electron spin resonance and electron spin echo modulation spectroscopies. J. Phys. Chem. 1992, 96, 2652–2657. [Google Scholar] [CrossRef]
- Godiksen, A.; Vennestrøm, P.N.R.; Rasmussen, S.B.; Mossin, S. Identification and quantification of copper sites in zeolites by electron paramagnetic resonance spectroscopy. Top. Catal. 2017, 60, 13–29. [Google Scholar] [CrossRef]
- Giordanino, F.; Vennestrøm, P.N.R.; Lundegaard, L.F.; Stappen, F.N.; Mossin, S.; Beato, P.; Bordiga, S.; Lamberti, C. Characterization of Cu-exchanged SSZ-13: A comparative FTIR, UV-Vis, and EPR study with Cu-ZSM-5 and Cu-β with similar Si/Al and Cu/Al ratios. Dalton Trans. 2013, 42, 12741–12761. [Google Scholar] [CrossRef] [PubMed]
- Conesa, J.C.; Soria, J. Electron spin resonance of copper-exchanged Y zeolites. Part 1.-Behaviour of the cation during dehydration. J. Chem. Soc. Faraday Trans. 1979, 75, 406–422. [Google Scholar] [CrossRef]
- Godiksen, A.; Stappen, F.N.; Vennestrøm, P.N.R.; Giordanino, F.; Rasmussen, S.B.; Lundegaard, L.F.; Mossin, S. Coordination environment of copper sites in Cu-CHA zeolite investigated by electron paramagnetic resonance. J. Phys. Chem. C 2014, 118, 23126–23138. [Google Scholar] [CrossRef]
- Fernández, E.; Moreno-González, M.; Moliner, M.; Blasco, T.; Boronat, M.; Corma, A. Modeling of EPR parameters for Cu(II): Application to the selective reduction of NOx catalyzed by Cu-zeolites. Top. Catal. 2018, 61, 810–832. [Google Scholar] [CrossRef]
- Kucherov, A.V.; Slinkin, A.A.; Kondrat’ev, D.A.; Bondarenko, T.N.; Rubinstein, A.M.; Minachev, K.M. Cu2+-cation location and reactivity in mordenite and ZSM-5: e.s.r.-study. Zeolites 1985, 5, 320–324. [Google Scholar] [CrossRef]
- Carl, P.J.; Larsen, S.C. Variable-temperature electron paramagnetic resonance studies of copper-exchanged zeolites. J. Catal. 1999, 182, 208–218. [Google Scholar] [CrossRef]
- Jabłońska, M.; Beale, A.M.; Nocuń, M.; Palkovits, R. Ag-Cu based catalysts for the selective ammonia oxidation into nitrogen and water vapour. Appl. Catal. B Environ. 2018, 232, 275–287. [Google Scholar] [CrossRef]
- Góra-Marek, K.; Tarach, K.A.; Piwowarska, Z.; Łaniecki, M.; Chmielarz, L. Ag-loaded zeolites Y and USY as catalysts for selective ammonia oxidation. Catal. Sci. Technol. 2016, 6, 1651–1660. [Google Scholar] [CrossRef]
- Wang, H.; Xu, R.; Jin, Y.; Zhang, R. Zeolite structure effects on Cu active center, SCR performance and stability of Cu-zeolite catalysts. Catal. Today 2019, 327, 295–308. [Google Scholar] [CrossRef]
- Kwak, J.H.; Tonkyn, R.G.; Kim, D.H.; Szanyi, J.; Peden, C.H. Excellent activity and selectivity of Cu-SSZ-13 in the selective catalytic reduction of NOx with NH3. J. Catal. 2010, 275, 187–190. [Google Scholar] [CrossRef]
- Fickel, D.W.; D’Addio, E.; Lauterbach, J.A.; Lobo, R.F. The ammonia selective catalytic reduction activity of copper-exchanged small-pore zeolites. Appl. Catal. B Environ. 2011, 102, 441–448. [Google Scholar] [CrossRef]
- Shan, Y.; Shi, X.; Du, J.; Yan, Z.; Yu, Y.; He, H. SSZ-13 synthesized by solvent-free method: A potential candidate for NH3-SCR catalyst with high activity and hydrothermal stability. Ind. Eng. Chem. Res. 2019, 58, 5397–5403. [Google Scholar] [CrossRef]
- He, D.; Wang, Z.; Deng, D.; Deng, S.; He, H.; Liu, L. Synthesis of Cu-SSZ-13 catalyst by using different silica sources for NO-SCR by NH3. Mol. Catal. 2020, 484, 110738. [Google Scholar] [CrossRef]
- Han, J.; Jin, X.; Song, C.; Bi, Y.; Liu, O.; Liu, C.; Ji, N.; Lu, X.; Ma, D.; Li, Z. Rapid synthesis and NH3-SCR activity of SSZ-13 zeolite via coal gangue. Green. Chem. 2020, 22, 219–229. [Google Scholar] [CrossRef]
- Jabri, H.A.; Miyake, K.; Ono, K.; Nakai, M.; Inoue, R.; Hirota, Y.; Uchida, Y.; Wang, Y.; Nishitoba, T.; Yokoi, T.; et al. Dry gel conversion synthesis of Cu/SSZ-13 as a catalyst with high performance for NH3-SCR. Micropor. Mesopor. Mater. 2020, 297, 109780. [Google Scholar] [CrossRef]
- Fahami, A.R.; Günter, T.; Doronkin, D.E.; Casapu, M.; Zengel, D.; Vuong, T.H.; Simon, M.; Breher, F.; Kucherov, A.V.; Brückner, A.; et al. The dynamic nature of Cu sites in Cu-SSZ-13 and the origin of the seagull NOx conversion profile during NH3-SCR. React. Chem. Eng. 2019, 4, 1000–1018. [Google Scholar] [CrossRef]
- Guo, J.; Yang, W.; Zhang, Y.; Gan, L.; Fan, C.; Chen, J.; Peng, Y.; Li, J. A multiple-active-site Cu/SSZ-13 for NH3-SCO: Influence of Si/Al ratio on the catalytic performance. Catal. Commun. 2020, 135, 105751. [Google Scholar] [CrossRef]
- Yu, T.; Wang, J.; Yu Huang, Y.; Shen, M.; Li, W.; Wang, J. NH3 oxidation mechanism over Cu/SAPO-34 catalysts prepared by different methods. ChemCatChem 2014, 6, 2074–2083. [Google Scholar] [CrossRef]
- Stoll, S.; Schweiger, A. EasySpin, a comprehensive software package for spectral simulation and analysis in EPR. J. Magn. Reson. 2006, 178, 42–55. [Google Scholar] [CrossRef] [PubMed]



| Sample | Molar Batch Composition | Time/d | Product |
|---|---|---|---|
| SSZ-13(1) | 1 SiO2:0.0167 Al2O3:0.25 NaOH:0.25 TMAdaOH:15 H2O | 6 | CHA |
| SSZ-13(2) | 1 SiO2:0.0167 Al2O3:0.25 NaOH:0.125 TMAdaOH:15 H2O | 6 | CHA |
| SSZ-13(3) | 1 SiO2:0.0167 Al2O3:0.2 NaOH:0.125 TMAdaOH:15 H2O | 10 | CHA |
| SSZ-13(4) | 1 SiO2:0.0167 Al2O3:0.3 NaOH:0.125 TMAdaOH:15 H2O | 5 | CHA |
| Sample | Si; Al; Cu Content/wt. % | n(Si)/n(Al); n(Cu)/n(Al) | aS(BET)/m2 g−1 | VMIC/cm3 g−1 | VTOT/cm3 g−1 | H2 Consumption /mmol H2 g−1 |
|---|---|---|---|---|---|---|
| Cu-SSZ-13(1) | 37.6; 2.6; 4.4 | 13.9; 0.72 | 575 | 0.268 | 0.314 | 1.08 |
| Cu-SSZ-13(2) | 38.6; 2.2; 4.9 | 16.9; 0.95 | 582 | 0.265 | 0.352 | 1.16 |
| Cu-SSZ-13(3) | 37.0; 2.0; 5.2 | 17.8; 1.10 | 615 | 0.275 | 0.442 | 1.25 |
| Cu-SSZ-13(4) | 34.6; 2.4; 5.1 | 13.8; 0.90 | 591 | 0.262 | 0.379 | 1.18 |
| Sample | CBrönsted /μmol g−1 | CLewis /μmol g−1 | Rel. Int. | TOF (150 °C) 103/s−1 |
|---|---|---|---|---|
| Cu-SSZ-13(1) | 180 | 1150 | 6.57 | 0.55 |
| Cu-SSZ-13(2) | 0 | 1395 | 1.00 | 0.52 |
| Cu-SSZ-13(3) | 20 | 880 | 1.27 | 0.46 |
| Cu-SSZ-13(4) | 35 | 825 | 1.69 | 0.42 |
| Cu Site | gꓕ | g// | Aꓕ/MHz | A///MHz | lwpp/mT |
|---|---|---|---|---|---|
| A | 2.055 ± 0.003 | 2.347 ± 0.002 | 45 ± 8 | 459 ± 10 | 2.8 ± 0.3 |
| B | 2.058 ± 0.003 | 2.316 ± 0.004 | 38 ± 8 | 488 ± 7 | 2.8 ± 0.3 |
| C | 2.063 ± 0.003 | 2.252 ± 0.003 | 44 ± 5 | 490 ± 8 | 2.8 ± 0.3 |
| Sample | A/% | B/% | C/% |
|---|---|---|---|
| Cu-SSZ-13(1) | 80 | 10 | 10 |
| Cu-SSZ-13(2) | 64 | 24 | 12 |
| Cu-SSZ-13(3) | 64 | 24 | 12 |
| Cu-SSZ-13(4) | 70 | 15 | 15 |
| Sample | Reaction Conditions | Operation Temperature for Achieving > 80% NO Conversion | Formation of By-Products | Ref. |
|---|---|---|---|---|
| Cu-SSZ-13(1) | 500 ppm NO, 575 ppm NH3, 4 Vol. % O2 and balance He, GHSV = 60,000 h−1 | 175–450 °C | <6% N2O yield; <45 ppm N2O | This work |
| Cu-SSZ-13(2–4) Ion-exchange; 4.4–5.3 wt. % of Cu | 175–400 °C | <10% N2O yield; <55 ppm N2O | ||
| Cu-SSZ-13 Ion-exchange; 2.9 wt. % of Cu | 500 ppm NO, 500 ppm NH3, 10 Vol.-% O2 and balance N2, GHSV not provided | 200–500 °C | not shown | [56] |
| Cu-SSZ-13 Ion-exchange; 2.2–2.8 wt. % of Cu | 500 ppm NO, 500 ppm NH3, 5 Vol.-% O2, 5 Vol. % H2O and balance N2, GHSV = 400,000 h−1 | 225–525 °C | <10 ppm N2O | [57] |
| Cu-SSZ-13 Ion-exchange; 4.76 wt. % of Cu | 1000 ppm NO, 1000 ppm NH3, 6 Vol. % O2, 5 Vol. % H2O and balance He, GHSV = 300,000 h−1 | 175–500 °C | <3% N2O yield | [54] |
| Cu-SSZ-13 Ion-exchange; 4.0–4.9 wt. % of Cu | 500 ppm NO, 500 ppm NH3, 10 Vol. % O2 and balance N2, GHSV = 30,000 h−1 | 200–500 °C | <25 ppm N2O | [58] |
| Cu-SSZ-13 Ion-exchange; 3.97 wt. % of Cu | 500 ppm NO, 500 ppm NH3, 10 Vol. % O2 and balance N2, GHSV = 100,000 h−1 | 175–450 °C | >95% N2 selectivity | [59] |
| Cu-SSZ-13 Ion-exchange; 3.97 wt. % of Cu | 300 ppm NO, 300 ppm NH3, 5 Vol. % O2, 3 Vol. % H2O and balance N2, GHSV not shown | 200–550 °C | not shown | [60] |
| Cu-SSZ-13 Ion-exchange; 1.00–1.17 wt. % of Cu | 500 ppm NO, 500 ppm NH3, 10 Vol. % O2, 3 Vol. % H2O and balance N2, GHSV = 40,000 h−1 | 250–550 °C | <6 ppm N2O | [17] |
| Cu-SSZ-13 Ion-exchange; 1.45–1.62 wt. % of Cu | 350 ppm NO, 350 ppm NH3, 14 Vol. % O2, 2.5 Vol. % H2O and balance N2, GHSV = 200,000 h−1 | 200–500 °C | not shown | [19] |
| Cu-SSZ-13 Hydrothermal synthesis; 2.5 wt. % of Cu | 500 ppm NO, 500 ppm NH3, 5 Vol. % O2 and balance Ar, GHSV = 180,000 h−1 | 200–600 °C | not shown | [36] |
| Cu-SSZ-13 One-pot synthesis; 3.8 wt. % of Cu | 500 ppm NO, 500 ppm NH3, 5 Vol. % O2 and balance N2, GHSV = 200,000 h−1 | 175–550 °C | >90% N2 selectivity | [13] |
| Cu-SSZ-13 Ion-exchange; 3.1 wt. % of Cu monolith, aged | 400 ppm NO, 500 ppm NH3, 8 Vol. % O2, 5 Vol. % H2O and balance Ar, GHSV = 30,300 h−1 | 200–600 °C | <10 ppm N2O | [30] |
| Cu-SSZ-13 Ion-exchange; Content of copper not shown | 350 ppm NO, 350 ppm NH3, 14 Vol. % O2, 2 Vol. % H2O and balance N2, GHSV = 30,000 h−1 | 200–550 °C | <30 ppm NO2 <30 ppm N2O | [55] |
| Cu-SSZ-13 Ion-exchange; 1.2 wt. % of Cu | 1000 ppm NO, 1000 ppm NH3, 10 Vol. % O2, 5 Vol. % H2O and balance N2, GHSV = 200,000 h−1 | 300–550 °C | <10 ppm N2O | [61] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Palčić, A.; Bruzzese, P.C.; Pyra, K.; Bertmer, M.; Góra-Marek, K.; Poppitz, D.; Pöppl, A.; Gläser, R.; Jabłońska, M. Nanosized Cu-SSZ-13 and Its Application in NH3-SCR. Catalysts 2020, 10, 506. https://doi.org/10.3390/catal10050506
Palčić A, Bruzzese PC, Pyra K, Bertmer M, Góra-Marek K, Poppitz D, Pöppl A, Gläser R, Jabłońska M. Nanosized Cu-SSZ-13 and Its Application in NH3-SCR. Catalysts. 2020; 10(5):506. https://doi.org/10.3390/catal10050506
Chicago/Turabian StylePalčić, Ana, Paolo Cleto Bruzzese, Kamila Pyra, Marko Bertmer, Kinga Góra-Marek, David Poppitz, Andreas Pöppl, Roger Gläser, and Magdalena Jabłońska. 2020. "Nanosized Cu-SSZ-13 and Its Application in NH3-SCR" Catalysts 10, no. 5: 506. https://doi.org/10.3390/catal10050506
APA StylePalčić, A., Bruzzese, P. C., Pyra, K., Bertmer, M., Góra-Marek, K., Poppitz, D., Pöppl, A., Gläser, R., & Jabłońska, M. (2020). Nanosized Cu-SSZ-13 and Its Application in NH3-SCR. Catalysts, 10(5), 506. https://doi.org/10.3390/catal10050506







