1. Introduction
Clinical cancer trials establish the standard of care in a field that is marked by constant innovation and novel therapeutics. Clinical trial participation has also been shown to directly benefit participants’ overall health and healthcare outcomes. In a 2017 meta-analysis by Nijjar et al. evaluating 21 studies, it was shown that trial participation significantly improved the survival rate of women with ovarian cancer compared to non-participants [
1]. Cultivating a tailored and robust clinical trial menu to maximize enrollment has the potential to improve outcomes for individual patients and the broader population and should be a priority in cancer care.
However, the success of clinical trials is limited by recruitment and participation of patients. Over 80% of clinical trials do not enroll enough patients, and roughly 19% of trials end due to insufficient enrollment [
2]. Of the identified barriers to trial participation, the majority disproportionately impact minority and underserved populations. Although the 1993 Revitalization Act of the National Institutes of Health adopted a policy mandating the inclusion of minorities and women, there is still an underrepresentation of various ethnic and racial minorities, women, individuals of lower socioeconomic status, elderly, and rural patient populations in clinical trials [
1,
3,
4,
5,
6,
7,
8,
9,
10]. To promote trial enrollment, it is critical that we understand our patient population, their barriers and preferences, and what makes a trial appealing to those that are eligible [
7,
11,
12,
13,
14,
15,
16,
17,
18,
19,
20,
21,
22,
23,
24,
25,
26].
At our medium-sized, mixed academic–community Gynecologic Oncology practice, we offer a wide variety of gynecologic cancer clinical trials: cancer care delivery and therapeutic trials, phase I–IV. Our gynecologic oncologists administer chemotherapy and so have continuity with patients throughout the cancer journey. We serve a diverse, mixed urban–rural catchment. We have had success bolstering trial enrollment to over 20% of our gynecologic cancer patients, with intentional emphasis on trials and care delivery that would be appealing to those in our catchment that are typically marginalized (such as black women and rural patients). However, there has been no formal evaluation of which specific patient or trial factors impact eventual trial enrollment versus enrollment failure. We felt it would be beneficial to plainly describe specific trial design elements that are associated with trial enrollment. Our objective is to evaluate trial enrollment trends according to both patient and trial demographics/features for those who have sought gynecologic cancer care with our team.
2. Methods
An IRB-approved, retrospective cohort study was completed to evaluate all patients who screened positive for a clinical trial through the Gynecologic Oncology practice’s manual screening process. Screening events were defined as: (1) new neoplasms, (2) new cancer diagnosis for established patients, (3) recurrences or progressions of known disease, (4) changes in patient’s treatment regimen, or (5) discussion of a patient’s case at bi-monthly Gyn Onc tumor boards. Definitions of these events are detailed in
Table 1. Statistical analyses were then performed as described below.
Data was collected from July 2022 to December 2023. This included demographic information for each patient, trials for which each patient was considered, trials patients were enrolled on, and each trial’s individual characteristics, including disease site, funding source, biopsy requirements, and specific inclusion and exclusion criteria. The timeline was also considered, including when new trials were opened, so that the heterogeneity of trial characteristics was fully appreciated. A list of all trials that were available at our practice and descriptions of each trial are presented in
Supplementary Table S1.
Patient data was obtained retrospectively from the electronic medical records of the LSU Health Gynecologic Oncology Division, encompassing the University Medical Center New Orleans (UMCNO), East Jefferson General Hospital (EJGH), and West Jefferson Medical Center (WJMC) sites. Data were collected using a standardized abstraction form approved under the institutional IRB protocol, which outlined the primary data sources, extraction process, and verification steps. Study personnel recorded demographics, cancer characteristics, treatment history, and clinical trial enrollment data. All entries were verified for accuracy by cross-referencing source documentation, and a random 10% of records underwent secondary review by the principal investigator for quality control. Data was de-identified prior to analysis, with linkage files stored separately on a secure, password-protected LSU Health network drive in accordance with IRB-approved procedures.
3. Statistical Analysis
Categorical variables were summarized using counts and percentages, while continuous variables were summarized as means with standard deviations. Group comparisons were conducted using Fisher’s exact tests for categorical variables and Wilcoxon rank-sum tests for continuous variables. Multivariable logistic regression was performed with enrollment status as the dependent variable. For patient-level analyses, covariates included age, race, ethnicity, insurance status, marital status, cancer stage, and distance from the trial site. For trial-level analyses, covariates included prior cancer history and prior treatment requirements, with adjustment for patient-level factors such as distance, insurance, race, and marital status. Trial-level variables (e.g., prior cancer exclusion, prior treatment requirement) were evaluated separately using logistic regression models adjusted for patient-level covariates such as distance from site, insurance status, race, and marital status. Given the exploratory intent of the study and the modest sample size, variable selection procedures were not performed. All analyses were conducted using R (version 4.3.1; R Foundation for Statistical Computing, Vienna, Austria).
4. Results
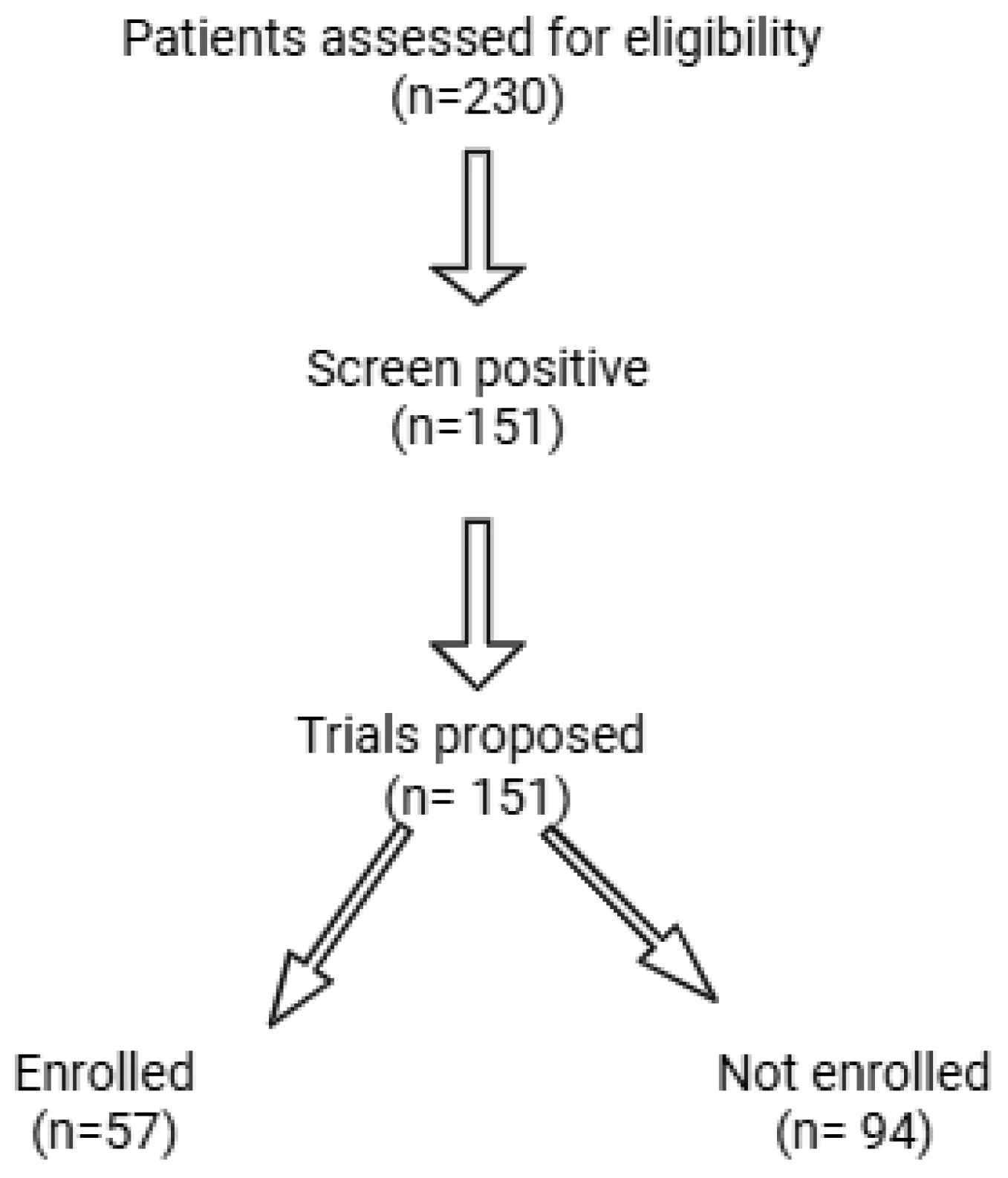
A total of 151 patients were included in the final analysis. The flow of patients through screening and enrollment is shown in
Figure 1. Of 230 patients assessed for eligibility, 151 screened positive for at least one trial, and 57 of these were ultimately enrolled. The average age of participants was 60.7 years old, and most patients studied presented with endometrial cancer (61.6%). There were a total of 72 Black patients (47.7%) and 63 White patients (41.7%) included, with respective enrollment rates of 31.9% and 34.9%. The rest of the demographic information is presented in
Table 2.
The inclusion and exclusion criteria requirements, trial stage, cancer type, and other characteristics of available trials were considered and reported on. In our portfolio, there were eight trials open for endometrial cancer (42.1%), five for cervical cancer (26.3%), and six for ovarian cancer (31.6%). Of these, n = 11 trials required a biopsy (57.9%), and n = 18 required in-person follow-up visits (94.7%). Most studies were interventional based (78.9%), as opposed to observational. Also, there are more therapeutic drug trials (73.7%), as opposed to other interventional/surgical trials. These data are presented in
Table 3. The enrollment rates of each individual trial and enrollment rates based on trial characteristics were calculated, as shown in
Supplementary Tables S2 and S3. These supplementary tables provide expanded breakdowns of individual trial characteristics and enrollment rates for completeness. When looking at trial characteristics compared to enrollment rates, there was a significant increase in enrollment likelihood if the trial did not exclude subjects with prior cancer (50.0% vs. 33.3%,
p = 0.046). Clinical trials that required patients to have no prior chemotherapy demonstrated a trend for less successful enrollment compared to those trials which permitted prior chemotherapy 27.3% vs. 44.1%, (
p = 0.07).
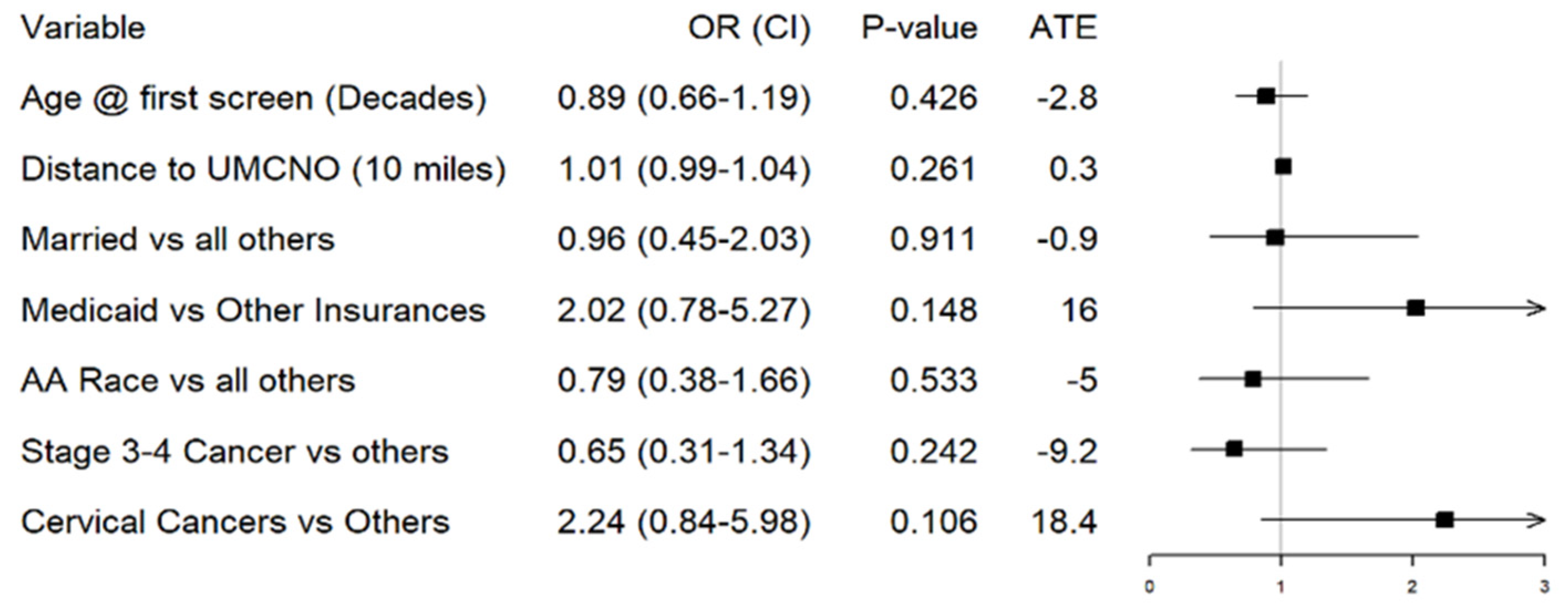
After adjustment, there were no significant differences between patient factors (age, marital status, race, ethnicity, and number of screening events) and enrollment status (
Figure 2,
Table S4). Enrollment status was defined as a dichotomous variable, with yes/no answer options. The “yes” category indicated that patients were screened, found to be candidates (“screen positive”), and subsequently enrolled on trial. The “no” category included all patients who screened positive at one of the screening events identified by the Gyn Onc team (detailed in
Table 1) but ultimately did not enroll on trial. It did not include patients who screened negative, only those who were classified as “screen positive, enrollment failure” events.
There was a drastic, unadjusted estimated increase in enrollment for Medicaid patients compared to patients with private insurance, Medicare, and free care (55.2% vs. 32%,
p = 0.031). However, a multivariable logistic regression evaluating patient enrollment based on demographic and cancer characteristics showed that insurance status (Medicaid vs. others) lost significance,
p = 0.148. Marital status was also analyzed across the clinical trials but there was no significant difference for those who enrolled compared to those who did not enroll (
p = 0.288). There were higher rates of enrollment seen among Asian patients and patients whose race was reported as “unknown” (>50%,
p = 0.125). These groups also had smaller sample sizes (n = 16) compared to White/Black patients. The aforementioned multivariable logistic regression calculated a
p-value of 0.533 for the variable of Black race vs. all others. When considering the primary gynecologic cancer sites of patients enrolled in clinical trials, there was no statistically significant association with enrollment and cervical cancer as the primary disease site (15.2% of patients enrolled,
p = 0.106), compared to other primary disease sites. In our population, we found that patients with Stage I cancer accounted for 42.1% of patients enrolled, which trended higher than the enrollment rates for other cancer stages, though the trend was not found to be statistically significant (
p = 0.087). Trials that these patients were able to enroll in included SISTER, NRG CC010, FIERCE, NERG GY006, and NRG GY026, which are further described in
Supplementary Table S1.
There was also a significant relationship between the distance traveled to the clinical trial site and clinical trial enrollment. Patients who lived within 20 miles of the clinical trial site, enrolled 30.1% of the time (n = 93), compared to 46.6% for those who lived more than 20 miles away (n = 58) (p = 0.032). The overall logistic regression model was statistically significant (likelihood-ratio test p = 0.004) with a Nagelkerke R2 ≈ 0.21, indicating moderate model fit. Although several univariate associations were significant (e.g., Medicaid insurance status, p = 0.031; distance from site, p = 0.032), no individual covariate retained significance after multivariable adjustment.
5. Discussion
Unlike prior reports, we aimed to look at not just how patient characteristics impact clinical trial enrollment, but also how the actual trial characteristics, such as inclusion and exclusion criteria, trial design, or funding source affect enrollment success. Our findings showed equitable trial enrollment across different races and insurance statuses. This suggests that following not only patient demographics, but also trial criteria, is important for tailoring a targeted portfolio to a specific patient catchment.
Furthermore, we showed robust enrollment for Medicaid patients and patients with cervical cancer. While this may reflect our unique patient population, these two characteristics are likely confounding. There is a well-documented higher incidence of cervical cancer in patients who are underinsured, likely due to limited preventative care and screening [
27]. This population is often considered hard to engage, especially due to the barriers to care these patients disproportionately face. However, this study shows that these patients, if offered the necessary resources, will participate in clinical research. We were able to identify ways to fill care gaps by evaluating trial demographics and comparing them directly to patient characteristics and will help guide future trial openings to continue to endorse equity.
As an example, we found that patients were more likely to enroll if they lived farther from the site of clinical trials. This is initially counterintuitive because distance is classically thought of as a barrier to care. While it is true that our robust trial portfolio likely attracts participants willing to travel for advanced care. However, in a less resourced population, such as is often seen with cervical cancer and Medicaid, it is crucial to highlight the work we have put into designing a system that helps to overcome these barriers. We have active clinical trial navigation, specifically with virtual options such as a virtual remote nursing program, which provides more opportunities for follow-ups closer to home. We have mechanisms in place to facilitate virtual trial enrollment and follow up activities for patients at remote sites. This has been particularly useful for some of our earlier stage endometrial cancer trials like NRG CC010 and SISTER. Collaboration with community clinics and offering innovative solutions to the barrier created by distance has made this practice a large referral site for trial enrollment, enhancing and expanding our patient catchment area.
Regarding patient presentation, in evaluating our “screen no trial available” population we identified a gap in trial availability for early-stage disease, and we quickly addressed this by opening trials that would include these early-stage frontline patients, like NRG CC010, SISTER, FIERCE, and ROCC. Our center has developed a robust manual clinical trial screening process, which has allowed for efficient trials opening to meet current patient needs [
28]. Subsequently, many patients we enrolled in had Stage 1 disease. Of available trials, 42.1% of them targeted primary disease presentation. Patients who had more advanced disease stages and comorbidities are often less likely to participate in clinical trials compared to their counterparts with earlier stage disease [
2]. Experimental therapeutics for patients with advanced disease with few treatment options are important to explore. However, this patient population comes with comorbidities and exhaustion from the burdens of what they have been through. Furthermore, early phase investigational therapeutic trials are often high-stakes, costly for both the patient and the institution, and more exclusive to those in excellent health. It remains an important priority, but if we focus exclusively on advanced and end-stages of disease space we risk addressing the particular needs in our specific cancer population served. Trials targeting patients who are earlier in their cancer journeys and for early-stage disease are often focused on complete remission and symptom management. These trials answer critical questions and often expose patients with fewer comorbidities and less disease burden to trials in a lower-stakes environment. Our patient population has shown that they are interested in and willing to enroll in trials in the earliest stages of cancer care. Many describe the experience as rewarding and, when need be, go on to enroll again at disease progression or recurrence.
Trials that excluded patients with a history of chemotherapy had lower rates of enrollment. This is concerning in Louisiana, where we have a “cancer alley” along the corridor from New Orleans to Baton Rouge. To be inclusive of our catchment, we realized early on that we had to be careful about opening trials that would permit patients with prior cancer and cancer therapies to join. As cancer prevalence and survivorship increases so does the likelihood of having had cancer. Accommodating patients with a history of cancer and cancer therapy will be a critical consideration in our goal to be inclusive and applicable to the real world.
Within the confines of improving our clinical trial profile, we wanted to understand both our patient population and how our portfolio is serving that population. This work helps delineate clinical trial criteria that can usher in future enrollment success. Additionally, this study analyzes features of clinical trials but also features of the enrollees. It accounts for social factors that can impact enrollment such as marital status and distance to trial site. Our study has focused on enrollment in gynecologic oncology clinical trials. Approximately 84,000 new cases are diagnosed and about 28,000 deaths occur each year from gynecologic cancer among women in the United States [
29]. Thus, it is crucial to investigate factors that can maximize patient enrollment for an area that affects a big population. Furthermore, this study did not exclude clinical trials by date. As many clinical trials as possible were included, maximizing the pool of data to extract from and learn from.
When considering limitations of this study, it is important to recognize that data for the clinical trials included were only collected from a single site, though the trials are cooperative group trials open at multiple sites across the country. Thus, for this “single institution” study, the findings may not be applicable to patient enrollment trends at other institutions or regions of the country. Additionally, due to the pilot nature of this study, we did not have previous data on the most predictive clinical trial characteristics, and how to best classify and track these criteria. Furthermore, the limited trial portfolio decreases the diversity of trials open at the time of our data analysis. This study could be enhanced by collecting patient feedback to enhance understanding of what barriers are deemed most important when considering clinical trial participation. Patient feedback can broaden our understanding of these trends and allow us to further refine clinical trials to maximize patient participation and satisfaction.
The importance of identifying trial design features and patient demographics that significantly impact enrollment can help guide researchers and physicians in developing their trial menus to maximize appeal and likelihood of patient participation in the future. Thus, clinical trials should be adjusted to meet patient needs because patients will readily engage if it is made accessible to them. After this evaluation, we have shown what patient and trial factors had a positive and/or negative impact on enrollment. When going through clinical trial feasibility questionnaires collecting this type of data will be an important resource for cancer sites and selecting the projects that best fit their clinical population and trial team structures.
6. Conclusions
In this study, we examined patient and trial level factors that impact patient participation. We found that patients with early-stage disease and those enrolled in trials with more inclusive eligibility criteria had the highest participation rates. In contrast, we observed that excluding patients with a prior history of cancer or chemotherapy limited enrollment, signifying the need for trial designs that reflect real world populations. We also noted higher participation rates among patients willing to travel farther for clinical trials, suggesting that structural support, such as a virtual navigator, plays an important role in overcoming traditional barriers. Future efforts should elaborate on these observations in a larger, multi-institutional context and engage patient perspectives to understand strategies that maximize enrollment and ensure that clinical trials remain accessible, inclusive, and meaningful for the diverse population of gynecologic oncology patients we serve.
7. Limitations
We acknowledge that this study has several limitations. First, the number of enrollment events in the logistic regression models was modest, which causes the possibility of overfitting. We limited the analysis to a small set of clinically meaningful variables, but results should be interpreted as exploratory and hypothesis-generating. Second, although non-parametric tests were applied in some comparisons, continuous variables were summarized as means with standard deviations for clinical interpretability, which may not fully capture non-normal distributions. Third, beyond insurance status, additional socioeconomic variables (e.g., income or payment method) were not consistently available and could not be analyzed. Future multi-institutional studies with larger sample sizes and richer sociodemographic data are needed to confirm and expand these results.
This study was presented in part at the Society of Gynecologic Oncology 2024 Meeting by LaMiah Hall, MPH.
Supplementary Materials
The following supporting information can be downloaded at:
https://www.mdpi.com/article/10.3390/cancers17233774/s1, Table S1. Open trials at our Gynecologic Oncology practice. Table S2. Enrollment Rates for Clinical Trials. Table S3. Enrollment Rate Based off Trial Designs. Table S4. Multivariable logistic regression for whether patient factors influence trial enrollment overall.
Author Contributions
Conceptualization, M.K., H.P. and Y.M.; methodology, M.K., H.P. and Y.M.; data collection, M.K., H.P. and Y.M.; formal analysis, L.H. and A.C.; data curation, L.H. and A.C.; writing—original draft preparation, M.K., H.P. and Y.M.; writing—review and editing, all authors; supervision, N.N., T.C. and A.J. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
The study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board of LSU Health Sciences Center–New Orleans (protocol exempt under 45 CFR 46.104(d), Category 4iii; waiver of informed consent and HIPAA authorization granted).
Informed Consent Statement
Patient consent was waived due to the study’s designation as exempt secondary research involving identifiable private information under 45 CFR 46.104(d), Category 4iii.
Data Availability Statement
De-identified data supporting the findings of this study are available from the corresponding author upon reasonable request.
Conflicts of Interest
The authors declare no conflicts of interest.
Abbreviations
| UMCNO | University Medical Center New Orleans, a primary academic affiliate of Louisiana State University (LSU) Health Sciences Center and a participating clinical site for gynecologic oncology trials. |
| EJGH | East Jefferson General Hospital, an affiliated community site contributing to patient recruitment and trial participation. |
| WJMC | West Jefferson Medical Center, an affiliated partner institution supporting clinical trial screening and data collection. |
| LSU | Louisiana State University Health Sciences Center, the coordinating academic institution for study oversight, IRB approval, and research infrastructure. |
| SISTER | Social Interventions for Support during Treatment for Endometrial Cancer and Recurrence; a multi-site randomized controlled trial evaluating the impact of virtual social and behavioral interventions on survivorship outcomes in endometrial cancer. |
| NRG CC010 | A Phase III Trial of the Impact of Sentinel Lymph Node Mapping on Patient-Reported Lower Extremity Limb Dysfunction in Endometrial Cancer; an NRG Oncology trial assessing functional outcomes following sentinel lymph node procedures. |
| NRG GY006 | Incorporation of Triapine (T) with Cisplatin Chemoradiation (CRT) for Locally Advanced Cervical and Vaginal Cancer; an NRG Oncology Phase III study investigating enhanced chemoradiation strategies for cervical malignancies. |
| NRG GY026 | A Phase II/III Study of Paclitaxel/Carboplatin Alone or Combined with Either Trastuzumab and Hyaluronidase-Oysk (Herceptin Hylecta) or Pertuzumab, Trastuzumab, and Hyaluronidase-Zzxf (Phesgo) in HER2-Positive, Stage I–IV Endometrial Serous Carcinoma or Carcinosarcoma; an NRG Oncology trial evaluating HER2-targeted therapies. |
| FIERCE | Facilitating Inclusive Enrollment in Research for Cancer Equity; a Phase Ib trial evaluating pembrolizumab com-bined with vaginal cuff brachytherapy and chemotherapy for high-intermediate–risk endometrial cancer. |
| ROCC | Robotic versus Open Radical Hysterectomy for Cervical Cancer (GOG-3043); a randomized controlled trial comparing surgical approaches to radical hysterectomy in cervical cancer. |
References
- Nijjar, S.K.; D’Amico, M.I.; Wimalaweera, N.A.; Cooper, N.; Zamora, J.; Khan, K.S. Participation in clinical trials improves outcomes in women’s health: A systematic review and meta-analysis. BJOG Int. J. Obstet. Gynaecol. 2017, 124, 863–871. [Google Scholar] [CrossRef]
- Thomas, C.; Mulnick, S.; Krucien, N.; Marsh, K. How do study design features and participant characteristics influence willingness to participate in clinical trials? Results from a choice experiment. BMC Med. Res. Methodol. 2022, 22, 323. [Google Scholar] [CrossRef]
- Mahmud, A.; Zalay, O.; Springer, A.; Arts, K.; Eisenhauer, E. Barriers to participation in clinical trials: A physician survey. Curr. Oncol. 2018, 25, 119–125. [Google Scholar] [CrossRef]
- Ford, J.G.; Howerton, M.W.; Lai, G.Y.; Gary, T.L.; Bolen, S.; Gibbons, C.; Tilburt, J.; Baffi, C.; Tanpitukpongse, T.P.; Wilson, R.F.; et al. Barriers to recruiting underrepresented populations to cancer clinical trials: A systematic review. Cancer 2008, 112, 228–242. [Google Scholar] [CrossRef]
- Jorge, S.; Masshoor, S.; Gray, H.J.; Swisher, E.M.; Doll, K.M. Participation of Patients with Limited English Proficiency in Gynecologic Oncology Clinical Trials. J. Natl. Compr. Cancer Netw. JNCCN. 2023, 21, 27–32.e2. [Google Scholar] [CrossRef]
- Mathews, C.; Restivo, A.; Raker, C.; Weitzen, S.; Disilvestro, P. Willingness of gynecologic cancer patients to participate in clinical trials. Gynecol. Oncol. 2009, 112, 161–165. [Google Scholar] [CrossRef]
- Rivers, D.; August, E.M.; Sehovic, I.; Lee Green, B.; Quinn, G.P. A systematic review of the factors influencing African Americans’ participation in cancer clinical trials. Contemp. Clin. Trials. 2013, 35, 13–32. [Google Scholar] [CrossRef]
- Scalici, J.; Finan, M.A.; Black, J.; Harmon, M.D.; Nicolson, W.; Lankes, H.A.; Brady, W.E.; Rocconi, R.P. Minority participation in Gynecologic Oncology Group (GOG) Studies. Gynecol. Oncol. 2015, 138, 441–444. [Google Scholar] [CrossRef]
- Brangman, S.A. Achieving diversity in study populations: The importance of community engagement. J. Am. Geriatr. Soc. 2022, 70, 3080–3086. [Google Scholar] [CrossRef]
- Mbuagbaw, L.; Aves, T.; Shea, B.; Jull, J.; Welch, V.; Taljaard, M.; Yoganathan, M.; Greer-Smith, R.; Wells, G.; Tugwell, P. Considerations and guidance in designing equity-relevant clinical trials. Int. J. Equity Health 2017, 16, 93. [Google Scholar]
- Bois, A.D.; Rochon, J.; Lamparter, C.; Pfisterer, J. Pattern of care and impact of participation in clinical studies on the outcome in ovarian cancer. Int. J. Gynecol. Cancer. 2005, 15, 183–191. [Google Scholar] [CrossRef]
- Bouzalmate-Hajjaj, A.; Massó Guijarro, P.; Khan, K.S.; Bueno-Cavanillas, A.; Cano-Ibáñez, N. Benefits of participation in clinical trials: An umbrella review. Int. J. Environ. Res. Public. Health. 2022, 19, 15368. [Google Scholar] [CrossRef]
- Chaudhari, N.; Ravi, R.; Gogtay, N.J.; Thatte, U.M. Recruitment and retention of the participants in clinical trials: Challenges and solutions. Perspect. Clin. Res. 2020, 11, 64–69. [Google Scholar] [CrossRef]
- Gregg, J.R.; Horn, L.; Davidson, M.A.; Gilbert, J. Patient enrollment onto clinical trials: The role of physician knowledge. J. Cancer Educ. Off. J. Am. Assoc. Cancer Educ. 2014, 29, 74–79. [Google Scholar] [CrossRef]
- Chhatre, S.; Jefferson, A.; Cook, R.; Meeker, C.R.; Kim, J.H.; Hartz, K.M.; Wong, Y.N.; Caruso, A.; Newman, D.; Morales, K.; et al. Patient-centered recruitment and retention for a randomized controlled study. Trials 2018, 19, 205. [Google Scholar] [CrossRef]
- Sood, A.; Prasad, K.; Chhatwani, L.; Shinozaki, E.; Cha, S.S.; Loehrer, L.L.; Wahner-Roedler, D.L. Patients’ attitudes and preferences about participation and recruitment strategies in clinical trials. Mayo Clin. Proc. 2009, 84, 243–247. [Google Scholar] [CrossRef]
- Newington, L.; Metcalfe, A. Factors influencing recruitment to research: Qualitative study of the experiences and perceptions of research teams. BMC Med. Res. Methodol. 2014, 14, 10. [Google Scholar] [CrossRef]
- Nasser, N.; Grady, D.; Balke, C.W. Commentary: Improving participant recruitment in clinical and translational research. Acad. Med. 2011, 86, 1334–1335. [Google Scholar] [CrossRef]
- Kaplan, P.C.; Nápoles, A.M.; Dohan, D.; Hwang, E.S.; Melisko, M.; Nickleach, D.; Quinn, J.A.; Haas, J. Clinical trial discussion, referral, and recruitment: Physician, patient, and system factors. Cancer Causes Control. 2013, 24, 979–988. [Google Scholar] [CrossRef]
- James, A.S.; Daley, C.M.; Engelman, K.; Greiner, K.A.; Ellerbeck, E. Process evaluation of recruitment for a cancer screening trial in primary care. Health Promot. Pract. 2011, 12, 696–703. [Google Scholar] [CrossRef]
- Sully, B.G.; Julious, S.A.; Nicholl, J. A reinvestigation of recruitment to randomised, controlled, multicenter trials: A review of trials funded by two UK funding agencies. Trials 2013, 14, 166–167. [Google Scholar] [CrossRef]
- Mullins, C.D.; Abdulhalim, A.M.; Lavallee, D.C. Continuous patient engagement in comparative effectiveness research. JAMA 2012, 307, 1587–1588. [Google Scholar] [CrossRef]
- Schuster, A.L.R.; Hampel, H.; Paskett, E.D.; Bridges, J.F.P. Rethinking Patient Engagement in Cancer Research. Patient 2023, 16, 89–93. [Google Scholar] [CrossRef]
- Pittell, H.; Calip, G.S.; Pierre, A.; Ryals, C.A.; Altomare, I.; Royce, T.J.; Guadamuz, J.S. Racial and Ethnic Inequities in US Oncology Clinical Trial Participation from 2017 to 2022. JAMA Netw. Open 2023, 6, e2322515. [Google Scholar] [CrossRef]
- Duma, N.; Vera Aguilera, J.; Paludo, J.; Haddox, C.; Gonzalez, M.; Wang, Y.; Leventakos, K.; Hubbard, J.; Mansfield, A.; Go, R.; et al. Representation of minorities and women in oncology clinical trials: Review of the past 14 years. J. Oncol. Pract. 2018, 14, e1–e10. [Google Scholar] [CrossRef]
- Guerra, C.E.; Fleury, M.E.; Byatt, L.P.; Lian, T.; Pierce, L. Strategies to advance equity in cancer clinical trials. Am. Soc. Clin. Oncol. Educ. Book. 2022, 42, 1–11. [Google Scholar] [CrossRef]
- Saraiya, M.; Cheung, L.C.; Soman, A.; Mix, J.; Kennery, K.; Chen, X.; Perkins, R.B.; Schiffman, M.; Wentzensen, N.; Miller, J. Risk of cervical precancer and cancer among uninsured and underserved women from 2009 to 2017. Am. J. Obstet. Gynecol. 2021, 224, e1–e366. [Google Scholar] [CrossRef]
- Klein, M.; Pirzadah, H.; Magharehabed, Y.; Chapple, A.; Nair, N.; Jernigan, A.; Castellano, T. Developing a manual clinical trials screening process in a diverse southern gynecologic oncology practice. Gynecol. Oncol. Rep. 2025, 57, 101549. [Google Scholar] [CrossRef]
- Stewart, S.L.; Lakhani, N.; Brown, P.M.; Larkin, O.A.; Moore, A.R.; Hayes, N.S. Gynecologic Cancer Prevention and Control in the National Comprehensive Cancer Control Program: Progress, Current Activities, and Future Directions. J. Womens Health. 2013, 22, 651–657. [Google Scholar] [CrossRef]
| Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).