Peritumoral ADC Values Correlate with the MGMT Methylation Status in Patients with Glioblastoma
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
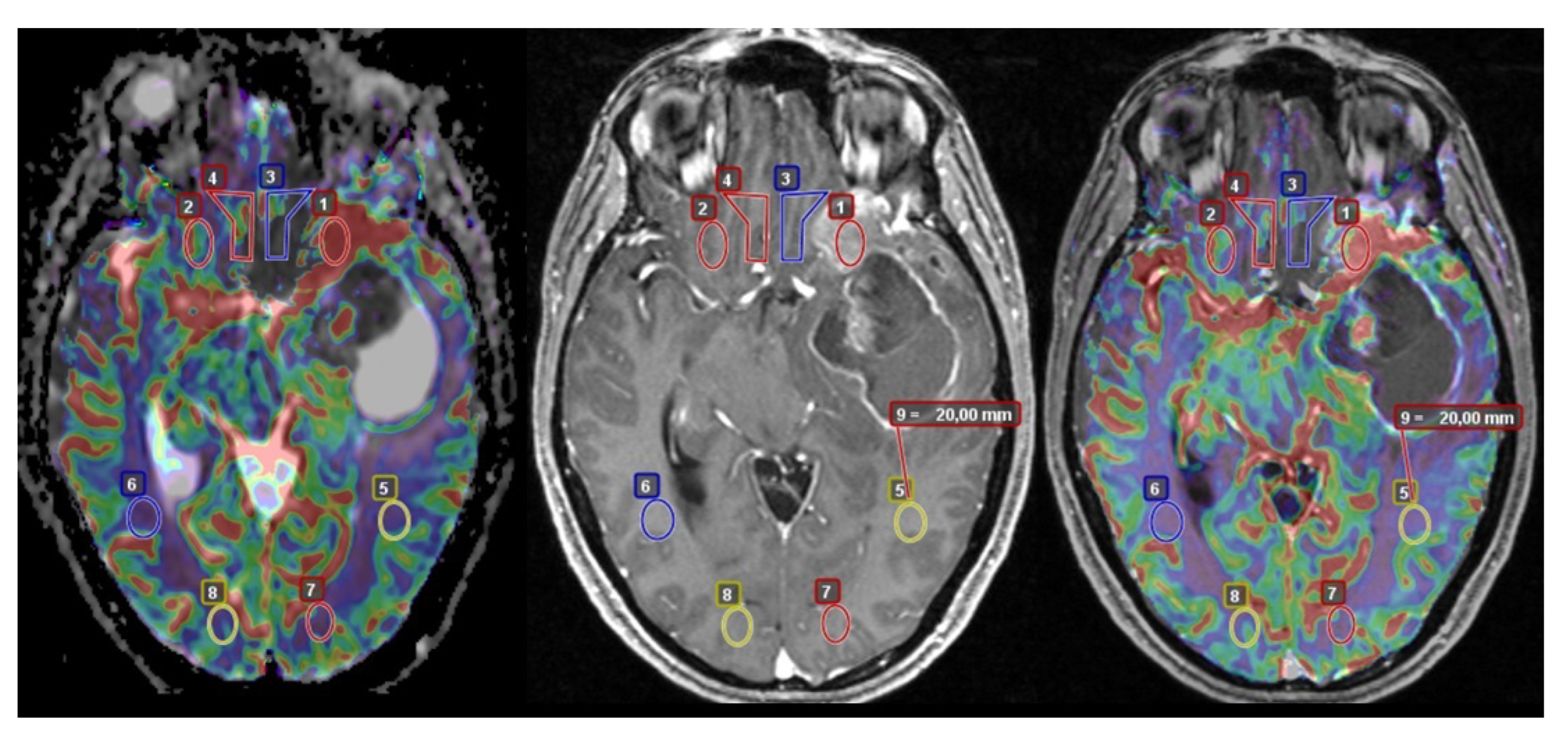
2.2. Image Acquisition and Analysis
2.3. Neuropathological Assessment
2.4. Statistical Analysis
3. Results
3.1. Descriptive Analysis
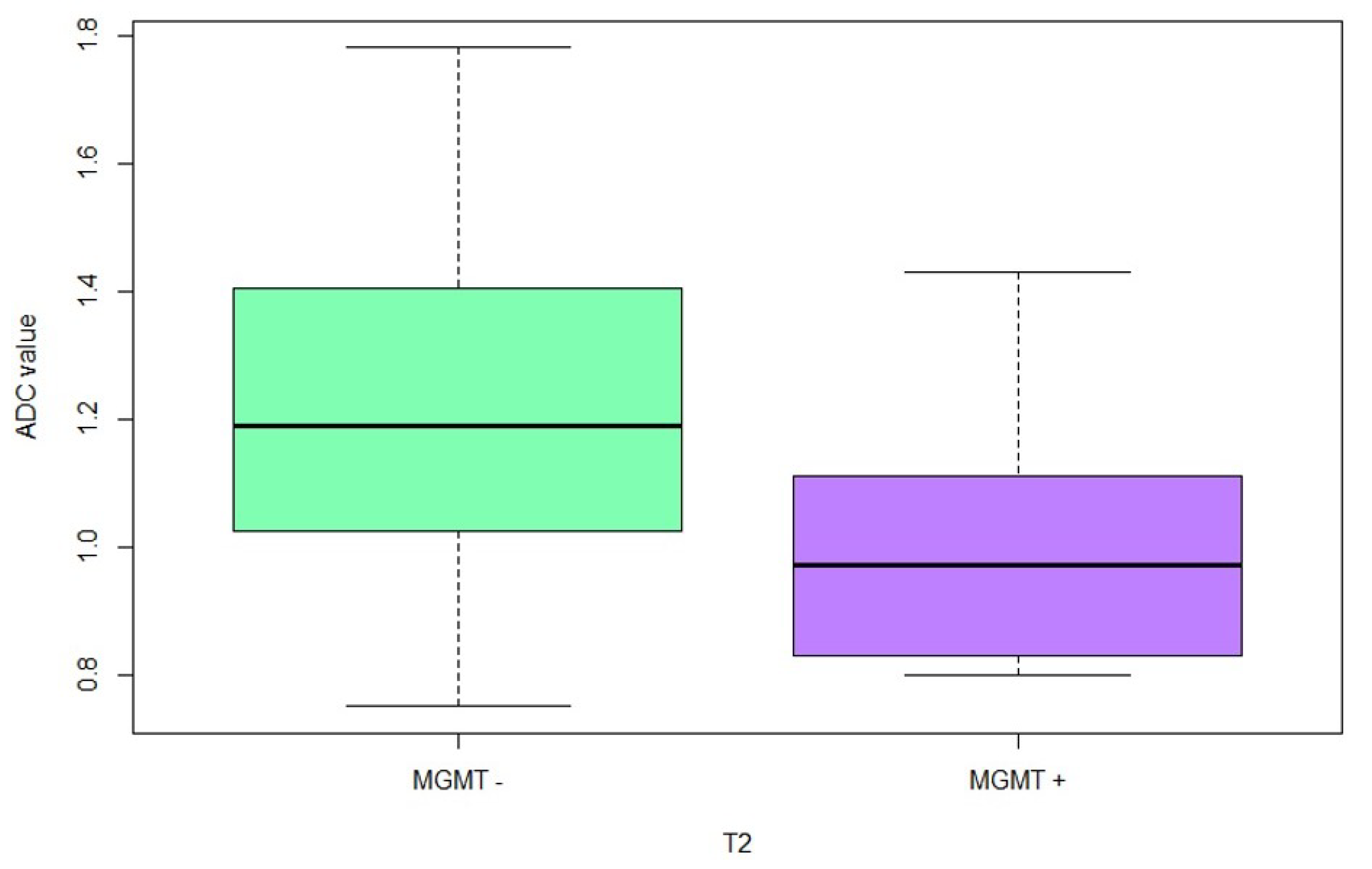
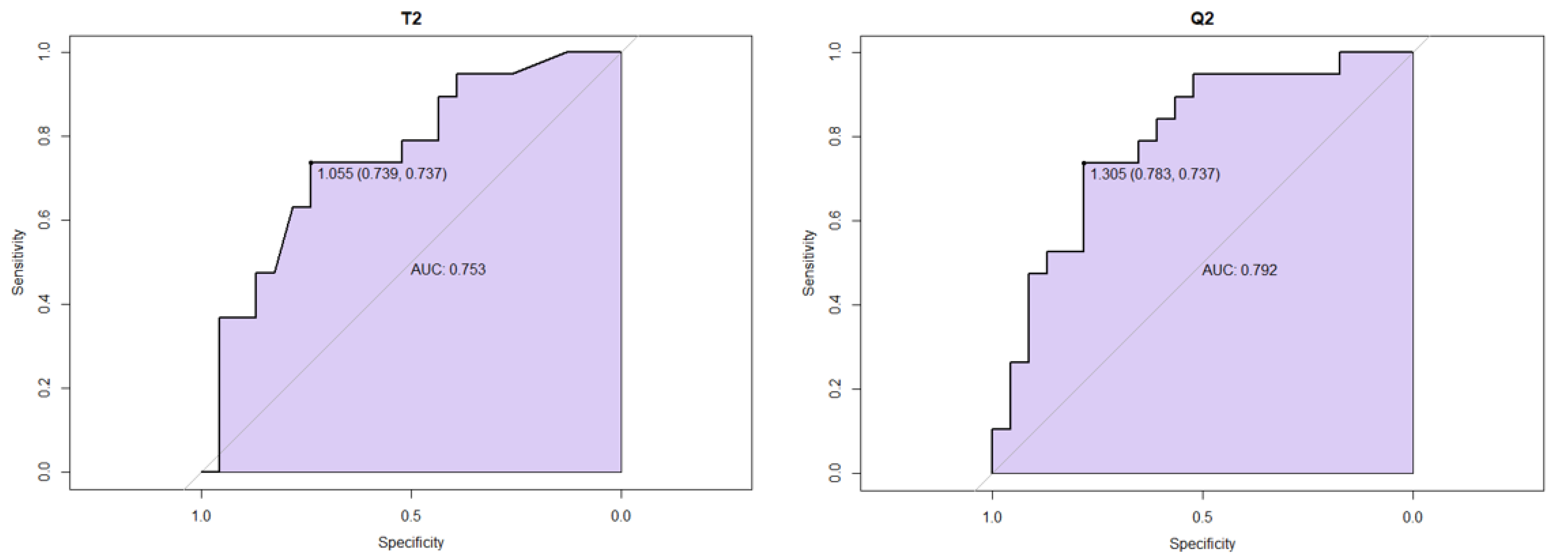
3.2. MGMT Status
3.3. Survival Analysis
4. Discussion
Limitations of the Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| ADC | apparent diffusion coefficient |
| GB | glioblastoma |
| MGMT | methylguanine-DNA 2 methyltransferase |
| MRI | magnetic resonance imaging |
| DSC | dynamic susceptibility contrast |
| ROI | region-of-interest |
| ROC | receiver-operating characteristics |
| AUC | area under the curve |
References
- Vigneswaran, K.; Neill, S.; Hadjipanayis, C. Beyond the World Health Organization grading of infiltrating gliomas: Advances in the molecular genetics of glioma classification. Ann. Transl. Med. 2015, 3, 95. [Google Scholar] [CrossRef]
- Zinn, P.; Sathyan, P.; Mahajan, B.; Bruyere, J.; Hegi, M.; Majumder, S.; Colen, R. A Novel Volume-Age-KPS (VAK) Glioblastoma Classification Identifies a Prognostic Cognate microRNA-Gene Signature. PLoS ONE 2012, 7, e41522. [Google Scholar] [CrossRef]
- Verhaak, R.; Hoadley, K.; Purdom, E.; Wang, V.; Qi, Y.; Wilkerson, M.; Miller, P.C.R.; Ding, L.; Golub, T.; Mesirov, J.; et al. Integrated Genomic Analysis Identifies Clinically Relevant Subtypes of Glioblastoma Characterized by Abnormalities in PDGFRA, IDH1, EGFR, and NF1. Cancer Cell 2010, 17, 98–110. [Google Scholar] [CrossRef]
- Klughammer, J.; Kiesel, B.; Roetzer-Pejrimovsky, T.; Fortelny, N.; Kuchler, A.; Sheffield, N.; Datlinger, P.; Peter, N.; Nenning, K.H.; Furtner, J.; et al. The DNA methylation landscape of glioblastoma disease progression shows extensive heterogeneity in time and space. bioRxiv 2017. [Google Scholar] [CrossRef]
- Sanson, M.; Marie, Y.; Paris, S.; Idbaih, A.; Laffaire, J.; Ducray, F.; Hallani, S.; Boisselier, B.; Mokhtari, K.; Hoang-Xuan, K.; et al. Isocitrate Dehydrogenase 1 Codon 132 Mutation Is an Important Prognostic Biomarker in Gliomas. J. Clin. Oncol. 2009, 27, 4150–4154. [Google Scholar] [CrossRef]
- Thon, N.; Kreth, F.; Kreth, S. Personalized treatment strategies in glioblastoma: MGMT promoter methylation status. OncoTargets Ther. 2013, 2013, 1363–1372. [Google Scholar] [CrossRef]
- Wen, P.; Macdonald, D.; Reardon, D.; Cloughesy, T.; Sorensen, A.; Galanis, E.; Degroot, J.; Wick, W.; Gilbert, M.; Lassman, A.; et al. Updated Response Assessment Criteria for High-Grade Gliomas: Response Assessment in Neuro-Oncology Working Group. J. Clin. Oncol. 2010, 28, 1963–1972. [Google Scholar] [CrossRef]
- Lilja, A.; Bergström, K.; Spännare, B.; Olsson, Y. Reliability of computed tomography in assessing histopathological features of malignant supratentorial gliomas. J. Comput. Assist. Tomogr. 1981, 5, 625–636. [Google Scholar]
- Watanabe, M.; Tanaka, R.; Takeda, N. Magnetic resonance imaging and histopathology of cerebral gliomas. Neuroradiology 1992, 34, 463–469. [Google Scholar] [CrossRef]
- Tovi, M. MR imaging in cerebral gliomas analysis of tumour tissue components. Acta Radiol. Suppl. 1993, 384, 1–24. [Google Scholar]
- Schaefer, P.W.; Grant, P.E.; Gonzalez, R.G. Diffusion-weighted MR imaging of the brain. Radiology 2000, 217, 331–345. [Google Scholar] [CrossRef]
- Yamasaki, F.; Sugiyama, K.; Ohtaki, M.; Takeshima, Y.; Abe, N.; Akiyama, Y.; Takaba, J.; Amatya, V.J.; Saito, T.; Kajiwara, Y.; et al. Glioblastoma treated with postoperative radio-chemotherapy: Prognostic value of apparent diffusion coefficient at MR imaging. Eur. J. Radiol. 2010, 73, 532–537. [Google Scholar] [CrossRef]
- Nakamura, H.; Murakami, R.; Hirai, T.; Kitajima, M.; Yamashita, Y. Can MRI-derived factors predict the survival in glioblastoma patients treated with postoperative chemoradiation therapy? Acta Radiol. 2013, 54, 214–220. [Google Scholar] [CrossRef]
- Saksena, S.; Jain, R.; Narang, J.; Scarpace, L.; Schultz, L.R.; Lehman, N.L.; Hearshen, D.; Patel, S.C.; Mikkelsen, T. Predicting survival in glioblastomas using diffusion tensor imaging metrics. J. Magn. Reson. Imaging 2010, 32, 788–795. [Google Scholar] [CrossRef]
- Qu, J.; Qin, L.; Cheng, S.; Reinshagen, K.; Li, X.; Li, H.; Dai, J.; Tao, J.; Akgoz, A.; Seethamraju, R.; et al. Residual low ADC and high FA at the resection margin correlate with poor chemoradiation response and overall survival in high-grade glioma patients. Eur. J. Radiol. 2015, 85, 657–664. [Google Scholar] [CrossRef]
- Hein, P.; Eskey, C.; Dunn, J.; Hug, E. Diffusion-Weighted Imaging in the Follow-up of Treated High-Grade Gliomas: Tumor Recurrence versus Radiation Injury. Am. J. Neuroradiol. 2004, 25, 201–209. [Google Scholar]
- Murakami, R.; Sugahara, T.; Nakamura, H.; Hirai, T.; Kitajima, M.; Hayashida, Y.; Baba, Y.; Oya, N.; Kuratsu, J.i.; Yamashita, Y. Malignant Supratentorial Astrocytoma Treated with Postoperative Radiation Therapy: Prognostic Value of Pretreatment Quantitative Diffusion-weighted MR Imaging. Radiology 2007, 243, 493–499. [Google Scholar] [CrossRef]
- Louis, D.N. The next step in brain tumor classification: “Let us now praise famous men”… or molecules? Acta Neuropathol. 2012, 124, 761–762. [Google Scholar] [CrossRef]
- Louis, D.N.; Perry, A.; Wesseling, P.; Brat, D.J.; Cree, I.A.; Figarella-Branger, D.; Hawkins, C.; Ng, H.; Pfister, S.M.; Reifenberger, G.; et al. The 2021 WHO classification of tumors of the central nervous system: A summary. Neuro-oncology 2021, 23, 1231–1251. [Google Scholar] [CrossRef]
- Han, Y.; Yan, L.F.; Wang, X.B.; Sun, Y.Z.; Zhang, X.; Liu, Z.C.; Nan, H.Y.; Hu, Y.C.; Yang, Y.; Zhang, J.; et al. Structural and advanced imaging in predicting MGMT promoter methylation of primary glioblastoma: A region-of-interest based analysis. BMC Cancer 2018, 18, 215. [Google Scholar] [CrossRef]
- Kanazawa, T.; Minami, Y.; Jinzaki, M.; Toda, M.; Yoshida, K.; Sasaki, H. Predictive markers for MGMT promoter methylation in glioblastomas. Neurosurg. Rev. 2019, 42, 867–876. [Google Scholar] [CrossRef]
- Sunwoo, L.; Choi, S.; Park, C.K.; Kim, J.; Yi, K.; Lee, W.; Yoon, T.; Song, S.; Kim, J.; Kim, J.; et al. Correlation of apparent diffusion coefficient values measured by diffusion MRI and MGMT promoter methylation semiquantitatively analyzed with MS-MLPA in patients with glioblastoma multiforme. J. Magn. Reson. Imaging 2013, 37, 351–358. [Google Scholar] [CrossRef]
- Moon, W.J.; Choi, J.W.; Roh, H.G.; Lim, S.D.; Koh, Y.C. Imaging parameters of high grade gliomas in relation to the MGMT promoter methylation status: The CT, diffusion tensor imaging, and perfusion MR imaging. Neuroradiology 2012, 54, 555–563. [Google Scholar] [CrossRef]
- Gupta, A.; Prager, A.; Young, R.; Shi, W.; Omuro, A.; Graber, J. Diffusion-weighted MR imaging and MGMT methylation status in glioblastoma: A reappraisal of the role of preoperative quantitative ADC measurements. Am. J. Neuroradiol. 2013, 34, E10. [Google Scholar] [CrossRef]
- Oh, J.; Henry, R.; Pirzkall, A.; Lu, Y.; Li, X.; Catalaa, I.; Chang, S.; Dillon, W.; Nelson, S. Survival analysis in patients with glioblastoma multiforme: Predictive value of choline-to-N-acetylaspartate index, apparent diffusion coefficient, and relative cerebral blood volume. J. Magn. Reson. Imaging 2004, 19, 546–554. [Google Scholar] [CrossRef]
- Saraswathy, S.; Crawford, F.; Lamborn, K.; Pirzkall, A.; Chang, S.; Cha, S.; Nelson, S. Evaluation of MR markers that predict survival in patients with newly diagnosed GBM prior to adjuvant therapy. J. Neuro-Oncol. 2008, 91, 69–81. [Google Scholar] [CrossRef]
- Elson, A.; Bovi, J.; Siker, M.; Schultz, C.; Paulson, E. Evaluation of absolute and normalized apparent diffusion coefficient (ADC) values within the post-operative T2/FLAIR volume as adverse prognostic indicators in glioblastoma. J. Neuro-Oncol. 2015, 122, 549–558. [Google Scholar] [CrossRef]
- Qin, L.; Li, A.; Qu, J.; Reinshagen, K.; Li, X.; Cheng, S.C.; Bryant, A.; Young, G. Normalization of ADC does not improve correlation with overall survival in patients with high-grade glioma (HGG). J. Neuro-Oncol. 2018, 137, 313–319. [Google Scholar] [CrossRef]
- Ahlhelm, F.; Hagen, T.; Schneider, G.; Dorenbeck, U.; Nabhan, A.; Reith, W. ADC mapping of normal human brain. Med. Sci. Monit. 2004, 10, MT121–MT125. [Google Scholar]
- Preusser, M.; Berghoff, A.S.; Manzl, C.; Filipits, M.; Weinhäusel, A.; Pulverer, W.; Dieckmann, K.; Widhalm, G.; Wöhrer, A.; Knosp, E.; et al. Clinical Neuropathology practice news 1-2014: Pyrosequencing meets clinical and analytical performance criteria for routine testing of MGMT promoter methylation status in glioblastoma. Clin. Neuropathol. 2014, 33, 6. [Google Scholar] [CrossRef]
- Galijašević, M.; Steiger, R.; Radović, I.; Birkl-Toeglhofer, A.M.; Birkl, C.; Deeg, L.; Mangesius, S.; Rietzler, A.; Regodić, M.; Stockhammer, G.; et al. Phosphorous magnetic resonance spectroscopy and molecular markers in IDH1 wild type glioblastoma. Cancers 2021, 13, 3569. [Google Scholar] [CrossRef]
- Ellingson, B.; Cloughesy, T.; Pope, W.; Zaw, T.; Phillips, H.; Lalezari, S.; Nghiemphu, P.; Ibrahim, H.; Naeini, K.; Harris, R.; et al. Anatomic localization of O6-methylguanine DNA methyltransferase (MGMT) promoter methylated and unmethylated tumors: A radiographic study in 358 de novo human glioblastomas. NeuroImage 2012, 59, 908–916. [Google Scholar] [CrossRef]
- Artzi, M.; Bokstein, F.; Blumenthal, D.; Aizenstein, O.; Liberman, G.; Corn, B.; Ben Bashat, D. Differentiation between vasogenic-edema versus tumor-infiltrative area in patients with glioblastoma during bevacizumab therapy: A longitudinal MRI study. Eur. J. Radiol. 2014, 83, 1250–1256. [Google Scholar] [CrossRef]
- Martín-Noguerol, T.; Mohan, S.; Santos, E.; Cabrera-Zubizarreta, A.; Luna, A. Advanced MRI assessment of non-enhancing peritumoral signal abnormality in brain lesions. Eur. J. Radiol. 2021, 143, 109900. [Google Scholar] [CrossRef]
- Wu, C.X.; Lin, G.S.; Lin, Z.X.; Zhang, J.D.; Liu, S.Y.; Zhou, C.F. Peritumoral edema shown by MRI predicts poor clinical outcome in glioblastoma. World J. Surg. Oncol. 2015, 13, 496. [Google Scholar] [CrossRef]
- Choi, Y.S.; Ahn, S.S.; Kim, D.W.; Chang, J.H.; Kang, S.G.; Kim, E.H.; Kim, S.H.; Rim, T.H.; Lee, S.K. Incremental prognostic value of ADC histogram analysis over MGMT promoter methylation status in patients with glioblastoma. Radiology 2016, 281, 175–184. [Google Scholar] [CrossRef]
- Darbar, A.; Waqas, M.; Enam, S.F.; Mahmood, S. Use of Preoperative Apparent Diffusion Coefficients to Predict Brain Tumor Grade. Cureus 2018, 10, e2284. [Google Scholar] [CrossRef]
- Wang, Q.; Zhang, J.; Xu, X.; Chen, X.; Xu, B. Diagnostic performance of apparent diffusion coefficient parameters for glioma grading. J. Neuro-Oncol. 2018, 139, 61–68. [Google Scholar] [CrossRef]
- Gates, E.; Lin, J.; Weinberg, J.; Hamilton, J.; Prabhu, S.; Hazle, J.; Fuller, G.; Baladandayuthapani, V.; Fuentes, D.; Schellingerhout, D. Guiding the first biopsy in glioma patients using estimated Ki-67 maps derived from MRI: Conventional versus advanced imaging. Neuro-Oncology 2019, 21, 527–536. [Google Scholar] [CrossRef]
- Szylberg, M.; Sokal, P.; Śledzińska, P.; Bebyn, M.; Krajewski, S.; Szylberg, Ł.; Szylberg, A.; Szylberg, T.; Krystkiewicz, K.; Birski, M.; et al. MGMT Promoter Methylation as a Prognostic Factor in Primary Glioblastoma: A Single-Institution Observational Study. Biomedicines 2022, 10, 2030. [Google Scholar] [CrossRef]
- Smrdel, U.; Popović, M.; Zwitter, M.; Bostjancic, E.; Zupan, A.; Kovac, V.; Glavac, D.; Bokal, D.; Jerebic, J. Long-term survival in glioblastoma: Methyl guanine methyl transferase (MGMT) promoter methylation as independent favourable prognostic factor. Radiol. Oncol. 2016, 50, 394–401. [Google Scholar] [CrossRef]
- Weller, M.; Stupp, R.; Reifenberger, G.; Brandes, A.; Bent, M.; Wick, W.; Hegi, M. MGMT promoter methylation in malignant gliomas: Ready for personalized medicine? Nat. Rev. Neurol. 2009, 6, 39–51. [Google Scholar] [CrossRef]
- Gupta, A.; Omuro, A.M.; Shah, A.D.; Graber, J.J.; Shi, W.; Zhang, Z.; Young, R.J. Continuing the search for MR imaging biomarkers for MGMT promoter methylation status: Conventional and perfusion MRI revisited. Neuroradiology 2012, 54, 641–643. [Google Scholar] [CrossRef]
- Huang, P.H.; Xu, A.M.; White, F.M. Oncogenic EGFR signaling networks in glioma. Sci. Signal. 2009, 2, re6. [Google Scholar] [CrossRef]

| Parameters for ADC Maps | |
|---|---|
| Echo time | 95 ms |
| Repetition time | 7500 ms |
| Matrix | 256 × 256 |
| Field of view | 230 × 230 |
| Time of acquisition | 2 min 51 s |
| Plane | axial |
| Slice thickness | 3 mm |
| Location of GBs and Frequency | |
|---|---|
| Temporal left | 11 |
| Temporal right | 4 |
| Temporoparietal left | 1 |
| Temporoparietal right | 1 |
| Frontal left | 4 |
| Frontal right | 6 |
| Parietal left | 5 |
| Parietal right | 3 |
| Basal ganglia right | 2 |
| Occipital left | 3 |
| Temporo-Occipital left | 1 |
| Temporo-Occipital right | 1 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ladenhauf, V.K.; Galijasevic, M.; Kerschbaumer, J.; Freyschlag, C.F.; Nowosielski, M.; Birkl-Toeglhofer, A.M.; Haybaeck, J.; Gizewski, E.R.; Mangesius, S.; Grams, A.E. Peritumoral ADC Values Correlate with the MGMT Methylation Status in Patients with Glioblastoma. Cancers 2023, 15, 1384. https://doi.org/10.3390/cancers15051384
Ladenhauf VK, Galijasevic M, Kerschbaumer J, Freyschlag CF, Nowosielski M, Birkl-Toeglhofer AM, Haybaeck J, Gizewski ER, Mangesius S, Grams AE. Peritumoral ADC Values Correlate with the MGMT Methylation Status in Patients with Glioblastoma. Cancers. 2023; 15(5):1384. https://doi.org/10.3390/cancers15051384
Chicago/Turabian StyleLadenhauf, Valentin Karl, Malik Galijasevic, Johannes Kerschbaumer, Christian Franz Freyschlag, Martha Nowosielski, Anna Maria Birkl-Toeglhofer, Johannes Haybaeck, Elke Ruth Gizewski, Stephanie Mangesius, and Astrid Ellen Grams. 2023. "Peritumoral ADC Values Correlate with the MGMT Methylation Status in Patients with Glioblastoma" Cancers 15, no. 5: 1384. https://doi.org/10.3390/cancers15051384
APA StyleLadenhauf, V. K., Galijasevic, M., Kerschbaumer, J., Freyschlag, C. F., Nowosielski, M., Birkl-Toeglhofer, A. M., Haybaeck, J., Gizewski, E. R., Mangesius, S., & Grams, A. E. (2023). Peritumoral ADC Values Correlate with the MGMT Methylation Status in Patients with Glioblastoma. Cancers, 15(5), 1384. https://doi.org/10.3390/cancers15051384







