Effects of Epstein-Barr Virus Infection on the Risk and Prognosis of Primary Laryngeal Squamous Cell Carcinoma: A Hospital-Based Case-Control Study in Taiwan
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Populations
2.2. Clinicopathologic Evaluations
2.3. Detection of EBV DNA in Tumor Tissue and Plasma
2.4. Detection of EBV Antibody in Serum
2.5. Tissue Microarray Construction
2.6. In Situ Hybridization
2.7. Immunohistochemistry Staining
2.8. Computer-Supported Evaluation
2.9. Sample Size Calculation
2.10. Statistical Analysis
3. Results
3.1. Clinical Characteristics of Patients
3.2. EBV-Related Biomarkers in the Larynx and Peripheral Blood
3.3. Histological Factors of the Larynx
3.4. Relationship between EBV-Related Biomarkers and Clinical and Histological Characteristics in the Overall Cohort
3.5. Multi-Factor Modeling of Primary LSCC
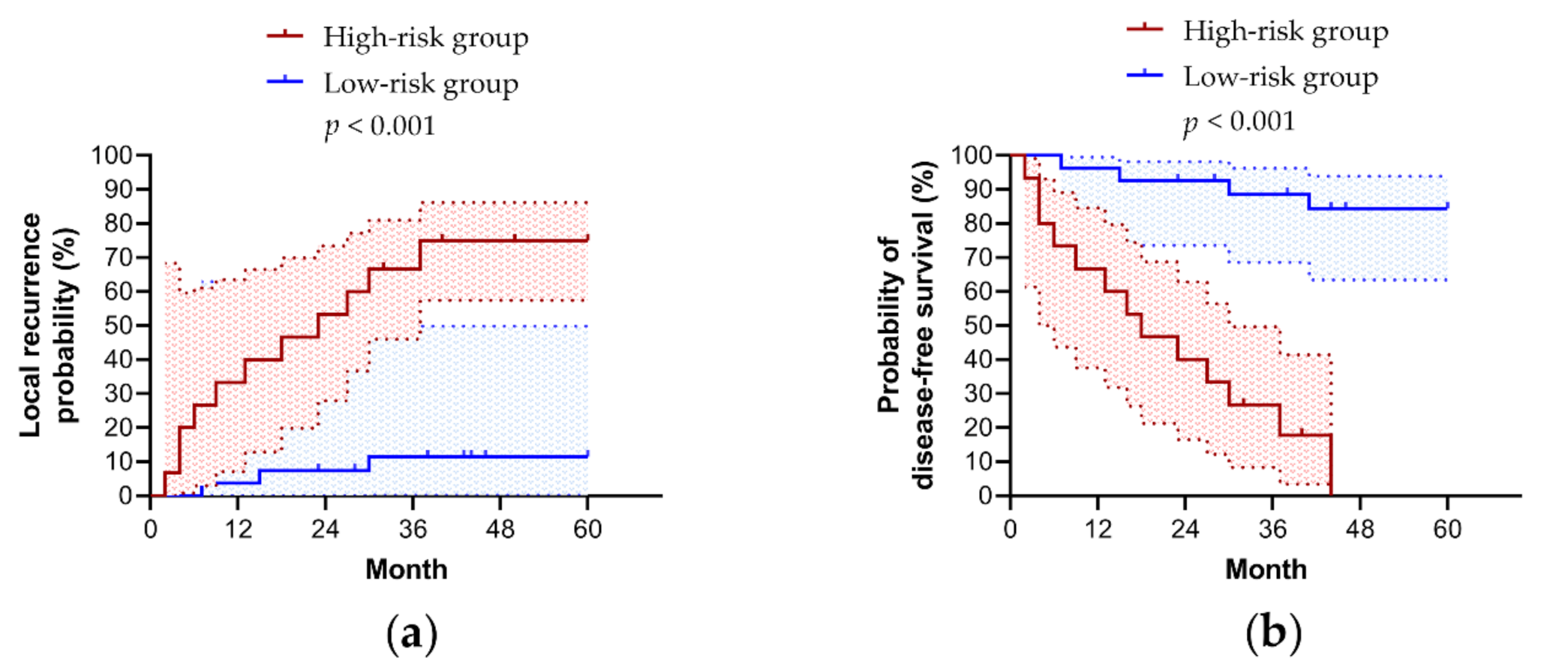
3.6. Five Year Prognoses in Patients with LSCC
3.7. Relationship between EBV-Related Biomarkers and Clinical and Histological Characteristics in Patients with LSCC
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Tai, P.; Yu, E.; Shiels, R.; Tonita, J. Long-term survival rates of laryngeal cancer patients treated by radiation and surgery, radiation alone, and surgery alone: Studied by lognormal and Kaplan-Meier survival methods. BMC Cancer 2005, 5, 13. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.; Fang, T.J.; Yung, K.; van Zante, A.; Miller, T.; Al-Jurf, S.; Schneider, S.; Courey, M. Clinical and histologic predictors of voice and disease outcome in patients with early glottic cancer. Laryngoscope 2012, 122, 2240–2247. [Google Scholar] [CrossRef] [PubMed]
- Bouvard, V.; Baan, R.; Straif, K.; Grosse, Y.; Secretan, B.; El Ghissassi, F.; Benbrahim-Tallaa, L.; Guha, N.; Freeman, C.; Galichet, L.; et al. A review of human carcinogens—Part B: Biological agents. Lancet Oncol. 2009, 10, 321–322. [Google Scholar] [CrossRef]
- de Oliveira, D.E.; Bacchi, M.M.; Macarenco, R.S.; Tagliarini, J.V.; Cordeiro, R.C.; Bacchi, C.E. Human papillomavirus and Epstein-Barr virus infection, p53 expression, and cellular proliferation in laryngeal carcinoma. Am. J. Clin. Pathol. 2006, 126, 284–293. [Google Scholar] [CrossRef]
- Ndiaye, C.; Mena, M.; Alemany, L.; Arbyn, M.; Castellsagué, X.; Laporte, L.; Bosch, F.X.; de Sanjosé, S.; Trottier, H. HPV DNA, E6/E7 mRNA, and p16INK4a detection in head and neck cancers: A systematic review and meta-analysis. Lancet Oncol. 2014, 15, 1319–1331. [Google Scholar] [CrossRef]
- Huang, C.G.; Lee, L.A.; Fang, T.J.; Li, H.Y.; Liao, C.T.; Kang, C.J.; Yen, T.C.; Tsao, K.C.; Chen, T.C.; Shih, S.R. Human papillomavirus infection is not associated with laryngeal squamous cell carcinoma in Taiwan. J. Microbiol. Immunol. Infect 2020, 53, 79–86. [Google Scholar] [CrossRef]
- Jeng, K.C.; Hsu, C.Y.; Liu, M.T.; Chung, T.T.; Liu, S.T. Prevalence of Taiwan variant of Epstein-Barr virus in throat washings from patients with head and neck tumors in Taiwan. J. Clin. Microbiol. 1994, 32, 28–31. [Google Scholar] [CrossRef]
- Zheng, Y.; Xia, P.; Zheng, H.C.; Takahashi, H.; Masuda, S.; Takano, Y. The screening of viral risk factors in tongue and pharyngolaryngeal squamous carcinoma. Anticancer. Res. 2010, 30, 1233–1238. [Google Scholar]
- Brichacek, B.; Hirsch, I.; Sibl, O.; Vilikusova, E.; Vonka, V. Association of some supraglottic laryngeal carcinomas with EB virus. Int. J. Cancer 1983, 32, 193–197. [Google Scholar] [CrossRef]
- Foltyn, S.; Strycharz-Dudziak, M.; Drop, B.; Boguszewska, A.; Polz-Dacewicz, M. Serum EBV antibodies and LMP-1 in Polish patients with oropharyngeal and laryngeal cancer. Infect Agent. Cancer 2017, 12, 31. [Google Scholar] [CrossRef]
- Callaghan, D.J.; Conner, B.R.; Strauss, M. Epstein-Barr virus antibody titers in cancer of the head and neck. Arch. Otolaryngol. 1983, 109, 781–784. [Google Scholar] [CrossRef]
- Yang, H.J.; Cho, Y.J.; Kim, H.S.; Chang, M.S.; Sung, M.W.; Kim, W.H. Association of p53 and BCL-2 expression with Epstein-Barr virus infection in the cancers of head and neck. Head Neck 2001, 23, 629–636. [Google Scholar] [CrossRef]
- Muderris, T.; Rota, S.; Muderris, T.; Inal, E.; Fidan, I. Does Epstein-Barr virus infection have an influence on the development of laryngeal carcinoma? Detection of EBV by real-time polymerase chain reaction in tumour tissues of patients with laryngeal carcinoma. Braz. J. Otorhinolaryngol. 2013, 79, 418–423. [Google Scholar] [CrossRef]
- Huang, S.C.M.; Tsao, S.W.; Tsang, C.M. Interplay of viral infection, host cell factors and tumor microenvironment in the pathogenesis of nasopharyngeal carcinoma. Cancers 2018, 10, 106. [Google Scholar] [CrossRef]
- Trask, D.K.; Wolf, G.T.; Bradford, C.R.; Fisher, S.G.; Devaney, K.; Johnson, M.; Singleton, T.; Wicha, M. Expression of Bcl-2 family proteins in advanced laryngeal squamous cell carcinoma: Correlation with response to chemotherapy and organ preservation. Laryngoscope 2002, 112, 638–644. [Google Scholar] [CrossRef]
- Li, J.; Huang, X.; Xie, X.; Wang, J.; Duan, M. Human telomerase reverse transcriptase regulates cyclin D1 and G1/S phase transition in laryngeal squamous carcinoma. Acta Otolaryngol. 2011, 131, 546–551. [Google Scholar] [CrossRef]
- Feenstra, M.; Bakema, J.; Verdaasdonk, M.; Rozemuller, E.; van den Tweel, J.; Slootweg, P.; de Weger, R.; Tilanus, M. Detection of a putative HLA-A*31012 processed (intronless) pseudogene in a laryngeal squamous cell carcinoma. Genes Chromosomes Cancer 2000, 27, 26–34. [Google Scholar] [CrossRef]
- Lagresle, C.; Gardie, B.; Eyquem, S.; Fasseu, M.; Vieville, J.C.; Pla, M.; Sigaux, F.; Bories, J.C. Transgenic expression of the p16INK4a cyclin-dependent kinase inhibitor leads to enhanced apoptosis and differentiation arrest of CD4−CD8− immature thymocytes. J. Immunol. 2002, 168, 2325–2331. [Google Scholar] [CrossRef]
- Kesselring, R.; Thiel, A.; Pries, R.; Wollenberg, B. The number of CD161 positive Th17 cells are decreased in head and neck cancer patients. Cell Immunol. 2011, 269, 74–77. [Google Scholar] [CrossRef]
- McShane, L.M.; Altman, D.G.; Sauerbrei, W.; Taube, S.E.; Gion, M.; Clark, G.M.; Statistics Subcommittee of the NCI-EORTC Working Group on Cancer Diagnostics. Reporting recommendations for tumor marker prognostic studies. J. Clin. Oncol. 2005, 23, 9067–9072. [Google Scholar] [CrossRef]
- Edge, S.B.; Compton, C.C. The American Joint Committee on Cancer: The 7th edition of the AJCC cancer staging manual and the future of TNM. Ann. Surg. Oncol. 2010, 17, 1471–1474. [Google Scholar] [CrossRef]
- Fang, T.J.; Courey, M.S.; Liao, C.T.; Yen, T.C.; Li, H.Y. Frozen margin analysis as a prognosis predictor in early glottic cancer by laser cordectomy. Laryngoscope 2013, 123, 1490–1495. [Google Scholar] [CrossRef]
- Hsin, L.J.; Fang, T.J.; Tsang, N.M.; Chin, S.C.; Yen, T.C.; Li, H.Y.; Liao, C.T.; Chen, I.H. Tumor volumetry as a prognostic factor in the management of T4a laryngeal cancer. Laryngoscope 2014, 124, 1134–1140. [Google Scholar] [CrossRef]
- Lee, L.A.; Fang, T.J.; Li, H.Y.; Huang, C.G.; Chen, T.C.; Liao, C.T.; Kang, C.J.; Chang, K.P.; Yen, T.C. Low expression of pRB predicts disease relapse in early glottic cancer treated with transoral laser microsurgery. Laryngoscope 2018, 129, E220–E226. [Google Scholar] [CrossRef]
- Fountzilas, E.; Kotoula, V.; Angouridakis, N.; Karasmanis, I.; Wirtz, R.M.; Eleftheraki, A.G.; Veltrup, E.; Markou, K.; Nikolaou, A.; Pectasides, D.; et al. Identification and validation of a multigene predictor of recurrence in primary laryngeal cancer. PLoS ONE 2013, 8, e70429. [Google Scholar] [CrossRef]
- Huang, C.G.; Lee, L.A.; Tsao, K.C.; Liao, C.T.; Yang, L.Y.; Kang, C.J.; Chang, K.P.; Huang, S.F.; Chen, I.H.; Yang, S.L.; et al. Human papillomavirus 16/18 E7 viral loads predict distant metastasis in oral cavity squamous cell carcinoma. J. Clin. Virol. 2014, 61, 230–236. [Google Scholar] [CrossRef]
- Hsu, C.L.; Chang, K.P.; Lin, C.Y.; Chang, H.K.; Wang, C.H.; Lin, T.L.; Liao, C.T.; Tsang, N.M.; Lee, L.Y.; Chan, S.C.; et al. Plasma Epstein-Barr virus DNA concentration and clearance rate as novel prognostic factors for metastatic nasopharyngeal carcinoma. Head Neck 2012, 34, 1064–1070. [Google Scholar] [CrossRef]
- Lo, Y.M.; Chan, L.Y.; Lo, K.W.; Leung, S.F.; Zhang, J.; Chan, A.T.; Lee, J.C.; Hjelm, N.M.; Johnson, P.J.; Huang, D.P. Quantitative analysis of cell-free Epstein-Barr virus DNA in plasma of patients with nasopharyngeal carcinoma. Cancer Res. 1999, 59, 1188–1191. [Google Scholar]
- Hsu, C.L.; Chan, S.C.; Chang, K.P.; Lin, T.L.; Lin, C.Y.; Hsieh, C.H.; Huang, S.F.; Tsang, N.M.; Lee, L.Y.; Ng, S.H.; et al. Clinical scenario of EBV DNA follow-up in patients of treated localized nasopharyngeal carcinoma. Oral Oncol. 2013, 49, 620–625. [Google Scholar] [CrossRef]
- Chang, K.P.; Hsu, C.L.; Chang, Y.L.; Tsang, N.M.; Chen, C.K.; Lee, T.J.; Tsao, K.C.; Huang, C.G.; Chang, Y.S.; Yu, J.S.; et al. Complementary serum test of antibodies to Epstein-Barr virus nuclear antigen-1 and early antigen: A possible alternative for primary screening of nasopharyngeal carcinoma. Oral Oncol. 2008, 44, 784–792. [Google Scholar] [CrossRef] [PubMed]
- Cho, Y.J.; Chang, M.S.; Park, S.H.; Kim, H.S.; Kim, W.H. In situ hybridization of Epstein-Barr virus in tumor cells and tumor-infiltrating lymphocytes of the gastrointestinal tract. Hum. Pathol. 2001, 32, 297–301. [Google Scholar] [CrossRef] [PubMed]
- Braun, M.; Kirsten, R.; Rupp, N.J.; Moch, H.; Fend, F.; Wernert, N.; Kristiansen, G.; Perner, S. Quantification of protein expression in cells and cellular subcompartments on immunohistochemical sections using a computer supported image analysis system. Histol. Histopathol. 2013, 28, 605–610. [Google Scholar] [CrossRef] [PubMed]
- Youden, W.J. Index for rating diagnostic tests. Cancer 1950, 3, 32–35. [Google Scholar] [CrossRef]
- Heagerty, P.J.; Lumley, T.; Pepe, M.S. Time-dependent ROC curves for censored survival data and a diagnostic marker. Biometrics 2000, 56, 337–344. [Google Scholar] [CrossRef] [PubMed]
- Steyerberg, E.W.; Eijkemans, M.J.; Harrell, F.E., Jr.; Habbema, J.D. Prognostic modeling with logistic regression analysis: In search of a sensible strategy in small data sets. Med. Decis. Mak. 2001, 21, 45–56. [Google Scholar] [CrossRef]
- Maier, H.; Gewelke, U.; Dietz, A.; Heller, W.D. Risk factors of cancer of the larynx: Results of the Heidelberg case-control study. Otolaryngol. Head Neck Surg. 1992, 107, 577–582. [Google Scholar] [CrossRef]
- Turunen, A.; Rautava, J.; Grenman, R.; Syrjanen, K.; Syrjanen, S. Epstein-Barr virus (EBV)-encoded small RNAs (EBERs) associated with poor prognosis of head and neck carcinomas. Oncotarget 2017, 8, 27328–27338. [Google Scholar] [CrossRef]
- Kelly, G.L.; Long, H.M.; Stylianou, J.; Thomas, W.A.; Leese, A.; Bell, A.I.; Bornkamm, G.W.; Mautner, J.; Rickinson, A.B.; Rowe, M. An Epstein-Barr virus anti-apoptotic protein constitutively expressed in transformed cells and implicated in burkitt lymphomagenesis: The Wp/BHRF1 link. PLoS Pathog. 2009, 5, e1000341. [Google Scholar] [CrossRef]
- Young, L.S.; Rickinson, A.B. Epstein-Barr virus: 40 years on. Nat. Rev. Cancer 2004, 4, 757–768. [Google Scholar] [CrossRef]
- Li, S.; Li, J.; Hu, T.; Zhang, C.; Lv, X.; He, S.; Yan, H.; Tan, Y.; Wen, M.; Lei, M.; et al. Bcl-2 overexpression contributes to laryngeal carcinoma cell survival by forming a complex with Hsp90β. Oncol. Rep. 2017, 37, 849–856. [Google Scholar] [CrossRef]
- Gioacchini, F.M.; Alicandri-Ciufelli, M.; Rubini, C.; Magliulo, G.; Re, M. Prognostic value of Bcl-2 expression in squamous cell carcinoma of the larynx: A systematic review. Int. J. Biol. Markers 2015, 30, e155–e160. [Google Scholar] [CrossRef]
- Douglas, C.M.; Bernstein, J.M.; Ormston, V.E.; Hall, R.C.; Merve, A.; Swindell, R.; Valentine, H.R.; Slevin, N.J.; West, C.M.; Homer, J.J. Lack of prognostic effect of carbonic anhydrase-9, hypoxia inducible factor-1alpha and bcl-2 in 286 patients with early squamous cell carcinoma of the glottic larynx treated with radiotherapy. Clin. Oncol. 2013, 25, 59–65. [Google Scholar] [CrossRef]
- McLemore, M.S.; Haigentz, M., Jr.; Smith, R.V.; Nuovo, G.J.; Alos, L.; Cardesa, A.; Brandwein-Gensler, M. Head and neck squamous cell carcinomas in HIV-positive patients: A preliminary investigation of viral associations. Head Neck Pathol. 2010, 4, 97–105. [Google Scholar] [CrossRef][Green Version]
- Yoshizaki, T.; Endo, K.; Ren, Q.; Wakisaka, N.; Murono, S.; Kondo, S.; Sato, H.; Furukawa, M. Oncogenic role of Epstein-Barr virus-encoded small RNAs (EBERs) in nasopharyngeal carcinoma. Auris Nasus Larynx 2007, 34, 73–78. [Google Scholar] [CrossRef]
- Duan, Y.; Li, Z.; Cheng, S.; Chen, Y.; Zhang, L.; He, J.; Liao, Q.; Yang, L.; Gong, Z.; Sun, L.Q. Nasopharyngeal carcinoma progression is mediated by EBER-triggered inflammation via the RIG-I pathway. Cancer Lett. 2015, 361, 67–74. [Google Scholar] [CrossRef]
- Kouvidou, C.; Rontogianni, D.; Tzardi, M.; Datseris, G.; Panayiotides, I.; Darivianaki, K.; Karidi, E.; Delides, G.; Kanavaros, P. Beta 2-microglobulin and HLA-DR expression in relation to the presence of Epstein-Barr virus in nasopharyngeal carcinomas. Pathobiology 1995, 63, 320–327. [Google Scholar] [CrossRef]
- Fitzsimmons, L.; Cartlidge, R.; Chang, C.; Sejic, N.; Galbraith, L.C.A.; Suraweera, C.D.; Croom-Carter, D.; Dewson, G.; Tierney, R.J.; Bell, A.I.; et al. EBV BCL-2 homologue BHRF1 drives chemoresistance and lymphomagenesis by inhibiting multiple cellular pro-apoptotic proteins. Cell Death Differ. 2020, 27, 1554–1568. [Google Scholar] [CrossRef]
- Lam, W.K.J.; Chan, K.C.A.; Lo, Y.M.D. Plasma Epstein-Barr virus DNA as an archetypal circulating tumour DNA marker. J. Pathol. 2019, 247, 641–649. [Google Scholar] [CrossRef]
- Yalcin, B.; Kutluk, T.; Agbaba, S.K.; Demir, C.; Talim, B. Circulating Epstein-Barr virus DNA and cell-free DNA in pediatric lymphomas. Turk. J. Pediatr. 2020, 62, 541–550. [Google Scholar] [CrossRef]
- Lo, S.; Ho, W.K.; Wei, W.I. Outcome of patients with positive Epstein-Barr virus serologic status in the absence of nasopharyngeal carcinoma in Hong Kong. Arch. Otolaryngol. Head Neck Surg. 2004, 130, 770–772. [Google Scholar] [CrossRef][Green Version]
- Morshed, K.; Polz-Dacewicz, M.; Szymanski, M.; Rajtar, B.; Ziaja-Soltys, M.; Golabek, W. Epstein-Barr virus antibodies in patients with laryngeal and hypopharyngeal cancer. Ann. Univ. Mariae Curie Sklodowska Med. 2003, 58, 227–231. [Google Scholar]
- Halmos, G.B.; Bras, L.; Siesling, S.; van der Laan, B.; Langendijk, J.A.; van Dijk, B.A.C. Age-specific incidence and treatment patterns of head and neck cancer in the Netherlands-A cohort study. Clin. Otolaryngol. 2018, 43, 317–324. [Google Scholar] [CrossRef]
- Yin, W.Z.; Yang, Q.W.; Niu, K.; Ren, M.; He, D.; Song, W.Z. Validation of reference genes for the normalization of RT-qPCR expression studies on human laryngeal cancer and hypopharyngeal cancer. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 4199–4209. [Google Scholar] [CrossRef]
- Keane, C.; Tobin, J.; Gunawardana, J.; Francis, S.; Gifford, G.; Gabrielli, S.; Gill, A.; Stevenson, W.; Talaulikar, D.; Gould, C.; et al. The tumour microenvironment is immuno-tolerogenic and a principal determinant of patient outcome in EBV-positive diffuse large B-cell lymphoma. Eur. J. Haematol. 2019, 103, 200–207. [Google Scholar] [CrossRef]
- Rammensee, H.G.; Robinson, P.J.; Crisanti, A.; Bevan, M.J. Restricted recognition of beta 2-microglobulin by cytotoxic T lymphocytes. Nature 1986, 319, 502–504. [Google Scholar] [CrossRef]
- Liu, C.; Yang, Z.; Li, D.; Liu, Z.; Miao, X.; Yang, L.; Zou, Q.; Yuan, Y. Overexpression of B2M and loss of ALK7 expression are associated with invasion, metastasis, and poor-prognosis of the pancreatic ductal adenocarcinoma. Cancer Biomark. 2015, 15, 735–743. [Google Scholar] [CrossRef]
- Zuo, J.J.; Tao, Z.Z.; Chen, C.; Hu, Z.W.; Xu, Y.X.; Zheng, A.Y.; Guo, Y. Characteristics of cigarette smoking without alcohol consumption and laryngeal cancer: Overall and time-risk relation. A meta-analysis of observational studies. Eur. Arch. Otorhinolaryngol. 2017, 274, 1617–1631. [Google Scholar] [CrossRef]
- Ramsey, T.; Guo, E.; Svider, P.F.; Lin, H.; Syeda, S.; Raza, S.N.; Fribley, A.M. Laryngeal cancer: Global socioeconomic trends in disease burden and smoking habits. Laryngoscope 2018, 128, 2039–2053. [Google Scholar] [CrossRef]
- Duncan, L.R.; Pearson, E.S.; Maddison, R. Smoking prevention in children and adolescents: A systematic review of individualized interventions. Patient Educ. Couns. 2018, 101, 375–388. [Google Scholar] [CrossRef]
- Lindson, N.; Klemperer, E.; Hong, B.; Ordonez-Mena, J.M.; Aveyard, P. Smoking reduction interventions for smoking cessation. Cochrane Database Syst. Rev. 2019, 9, CD013183. [Google Scholar] [CrossRef] [PubMed]
- Konduri, V.; Oyewole-Said, D.; Vazquez-Perez, J.; Weldon, S.A.; Halpert, M.M.; Levitt, J.M.; Decker, W.K. CD8(+)CD161(+) T-Cells: Cytotoxic me0mory cells with high therapeutic potential. Front. Immunol. 2020, 11, 613204. [Google Scholar] [CrossRef] [PubMed]
- Ostroumov, D.; Fekete-Drimusz, N.; Saborowski, M.; Kuhnel, F.; Woller, N. CD4 and CD8 T lymphocyte interplay in controlling tumor growth. Cell Mol. Life Sci. 2018, 75, 689–713. [Google Scholar] [CrossRef] [PubMed]
- Iwahori, K. Cytotoxic CD8+ lymphocytes in the tumor microenvironment. Adv. Exp. Med. Biol. 2020, 1224, 53–62. [Google Scholar] [CrossRef] [PubMed]
- Klatka, J.; Grywalska, E.; Hymos, A.; Krasowska, E.; Mielnik, M.; Siwicka-Gieroba, D.; Markowicz, J.; Trojanowski, P.; Olszanski, W.; Rolinski, J. Subpopulations of natural killer-T-like cells before and after surgical treatment of laryngeal cancer. Cent. Eur. J. Immunol. 2017, 42, 252–258. [Google Scholar] [CrossRef]
- Iliopoulou, E.G.; Karamouzis, M.V.; Missitzis, I.; Ardavanis, A.; Sotiriadou, N.N.; Baxevanis, C.N.; Rigatos, G.; Papamichail, M.; Perez, S.A. Increased frequency of CD4+ cells expressing CD161 in cancer patients. Clin. Cancer Res. 2006, 12, 6901–6909. [Google Scholar] [CrossRef]
- Turtle, C.J.; Swanson, H.M.; Fujii, N.; Estey, E.H.; Riddell, S.R. A distinct subset of self-renewing human memory CD8+ T cells survives cytotoxic chemotherapy. Immunity 2009, 31, 834–844. [Google Scholar] [CrossRef]
- Schneider, K.; Marbaix, E.; Bouzin, C.; Hamoir, M.; Mahy, P.; Bol, V.; Gregoire, V. Immune cell infiltration in head and neck squamous cell carcinoma and patient outcome: A retrospective study. Acta Oncol. 2018, 57, 1165–1172. [Google Scholar] [CrossRef]
- Lecerf, C.; Kamal, M.; Vacher, S.; Chemlali, W.; Schnitzler, A.; Morel, C.; Dubot, C.; Jeannot, E.; Meseure, D.; Klijanienko, J.; et al. Immune gene expression in head and neck squamous cell carcinoma patients. Eur. J. Cancer 2019, 121, 210–223. [Google Scholar] [CrossRef]
- Klatka, J.; Hymos, A.; Szkatula-Lupina, A.; Grywalska, E.; Klatka, B.; Terpilowski, M.; Stepulak, A. T-lymphocyte activation is correlated with the presence of anti-EBV in patients with laryngeal squamous cell carcinoma. In Vivo 2019, 33, 2007–2012. [Google Scholar] [CrossRef]
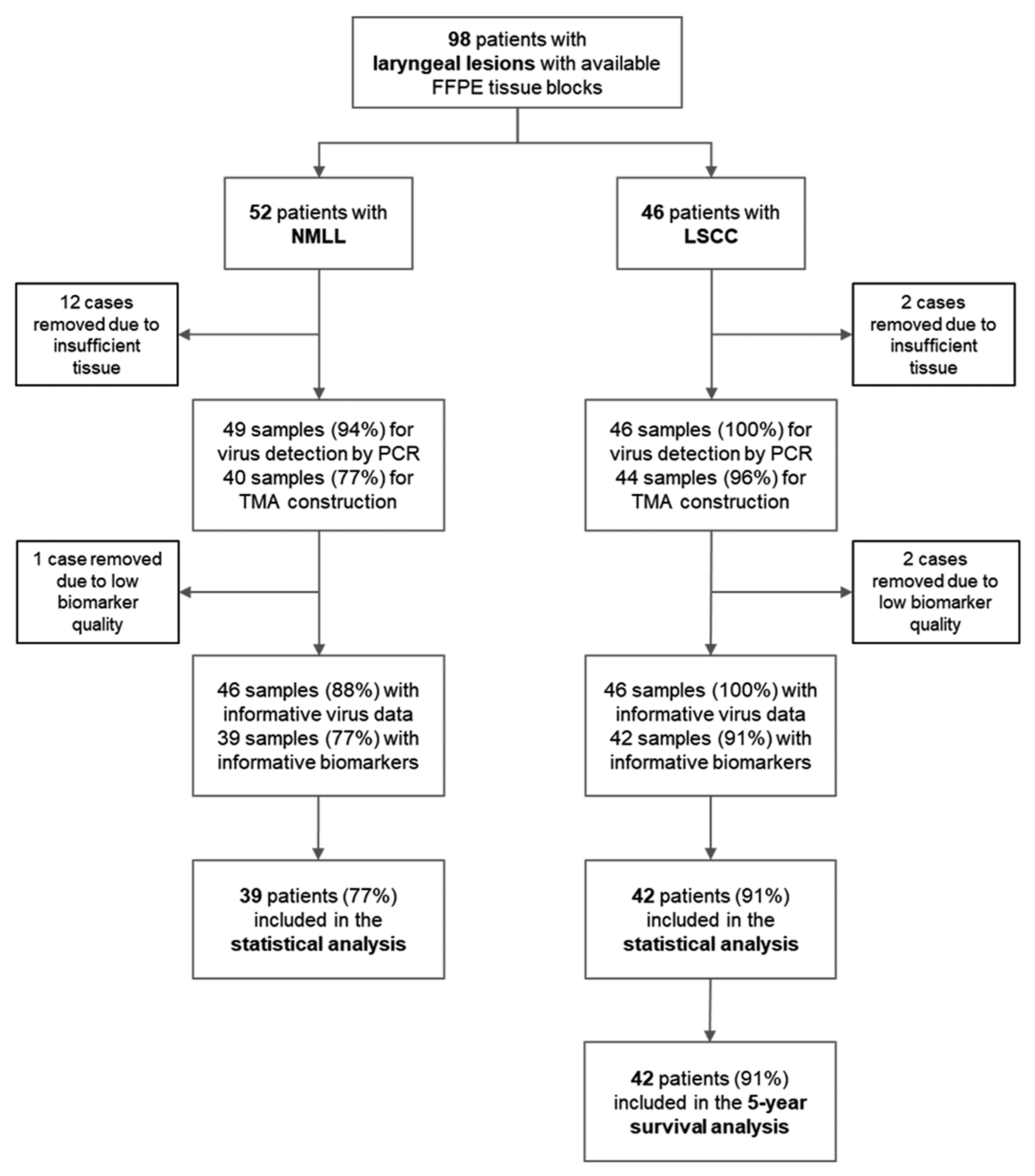
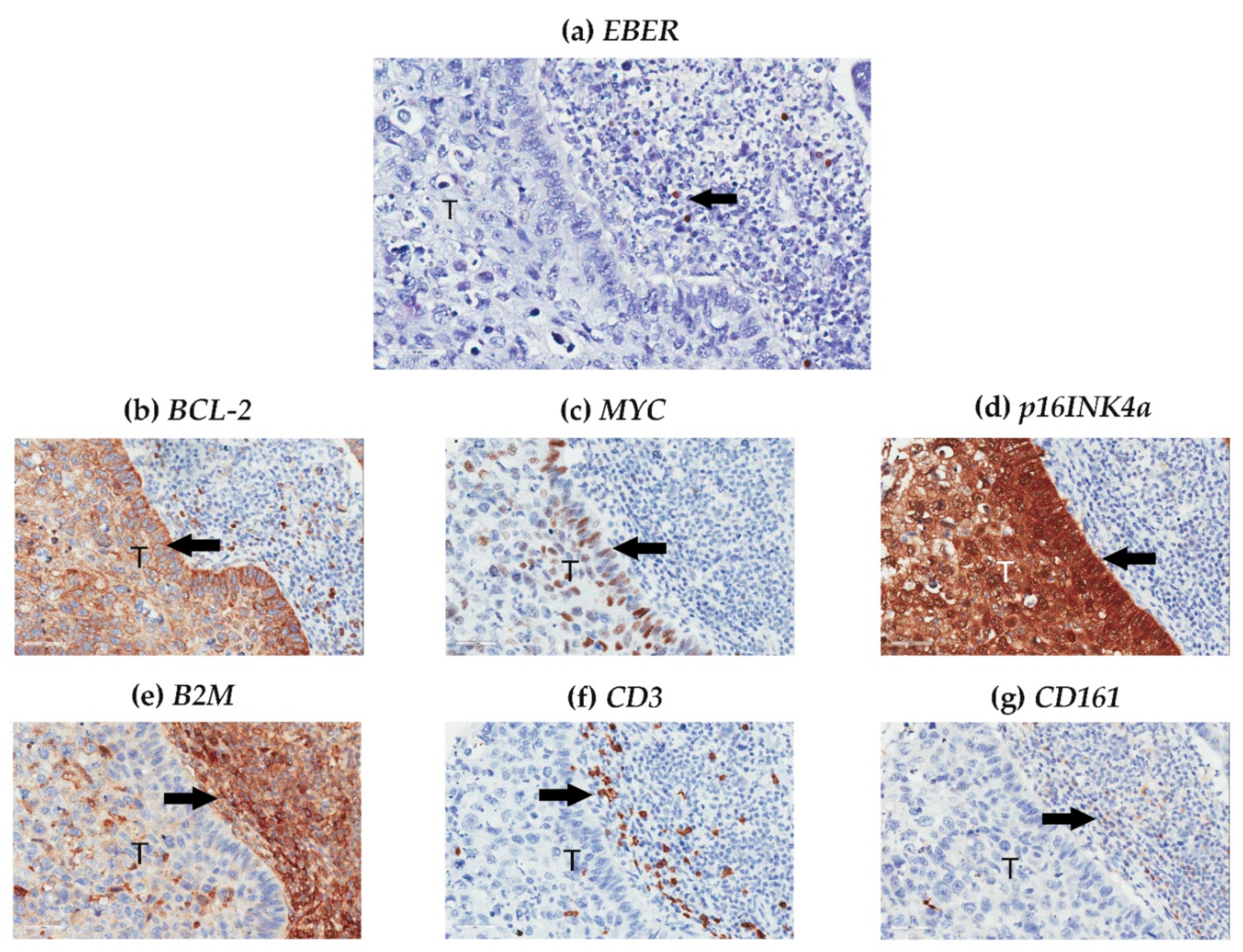
| Characteristic | Overall | Patients with LSCC | Patients with NMLL | p-Value a |
|---|---|---|---|---|
| Clinical Characteristics | ||||
| Patients | n = 81 | n = 42 | n = 39 | |
| Male, n (%) | 74 (91) | 38 (91) | 36 (92) | >0.99 |
| Female, n (%) | 7 (9) | 4 (9) | 3 (8) | |
| Age (years), median (range) | 58 (51–70) | 64 (58–74) | 52 (37–58) | <0.001 |
| Age ≥ 55 years, n (%) b | 45 (56) | 33 (79) | 12 (31) | <0.001 |
| Age < 55 years, n (%) b | 36 (44) | 9 (21) | 27 (69) | |
| Cigarette smoker, n (%) | 69 (85) | 39 (93) | 29 (74) | 0.03 |
| Never smoker, n (%) | 12 (15) | 3 (7) | 10 (26) | |
| Alcohol drinker, n (%) | 48 (59) | 27 (64) | 21 (54) | 0.37 |
| Never drinker, n (%) | 33 (41) | 15 (36) | 18 (46) | |
| Pathological Characteristics | ||||
| Polyps, n (%) | – | – | 18 (46) | – |
| Leukoplakia, n (%) | – | – | 21 (54) | – |
| T1–2, n (%) | – | 34 (81) | – | – |
| T3–4, n (%) | – | 8 (19) | – | – |
| N0, n (%) | – | 40 (95) | – | – |
| N1–2, n (%) | – | 2 (5) | – | – |
| Stage I–II, n (%) | – | 34 (81) | – | – |
| Stage III–IV, n (%) | – | 8 (19) | – | – |
| Treatment Modalities | ||||
| Transoral laser microsurgery, n (%) | – | 28 (67) | – | – |
| Total laryngectomy, n (%) | 3 (7) | – | ||
| RT, n (%) | – | 4 (10) | – | – |
| CCRT, n (%) | – | 3 (7) | – | – |
| Surgery + RT, n (%) | – | 3 (7) | – | – |
| Surgery + CCRT, n (%) | 1 (2) | – | – | |
| Single-modality treatment, n (%) | 31 (74) | – | – | |
| Multiple-modality treatment, n (%) | – | 11 (26) | – | – |
| Characteristics | Overall | Patients with LSCC | Patients with NMLL | p-Value a |
|---|---|---|---|---|
| Laryngeal Tissue | ||||
| Patients | n = 81 | n = 42 | n = 39 | |
| EBV DNA positivity, n (%) | 30 (37) | 22 (52) | 8 (20) | 0.01 |
| EBV DNA negativity, n (%) | 51 (63) | 20 (48) | 31 (80) | |
| nPI for EBER, (%), median (range) | 0.02 (0.01–0.06) | 0.03 (0.01–0.05) | 0.02 (0.01–0.07) | <0.001 |
| EBER signal ≥ 0.02%, n (%) b | 50 (62) | 28 (67) | 22 (56) | 0.37 |
| EBER signal < 0.02%, n (%) b | 31 (62) | 14 (33) | 17 (44) | |
| Peripheral Blood | ||||
| Patients | – | n = 30 | – | |
| Circulating EBV DNA positivity, n (%) | – | 2 (7) | – | – |
| Circulating EBV DNA negativity, n (%) | – | 28 (93) | – | – |
| EBV-VCA IgA positivity, n (%) | – | 4 (13) | – | – |
| EBV VCA IgA negativity, n (%) | – | 26 (87) | – | – |
| Characteristics | Overall | Patients with LSCC | Patients with NMLL | p-Value a |
|---|---|---|---|---|
| Tumor-Related Biomarkers in the Larynx | ||||
| Patients | n = 81 | n = 42 | n = 39 | |
| cPI for BCL-2, (%), median (range) | 82.9 (43.3–98.2) | 86.7 (76.8–98.7) | 61.5 (35.5–94.2) | 0.01 |
| BCL-2 expression ≥ 68.6%, n (%) b | 30 (37) | 34 (81) | 17 (44) | 0.001 |
| BCL-2 expression < 68.6%, n (%) b | 51 (63) | 8 (19) | 22 (56) | |
| nPI for MYC, (%), median (range) | 0.44 (0.24–0.56) | 0.38 (0.17–0.53) | 0.50 (0.32–0.67) | 0.03 |
| MYC expression ≤ 0.44%, n (%) b | 44 (54) | 28 (67) | 16 (41) | 0.03 |
| MYC expression > 0.44%, n (%) b | 37 (46) | 14 (33) | 23 (59) | |
| cPI for p16INK4a, (%), median (range) | 49.8 (20.3–93.8) | 45.3 (11.5–92.4) | 49.9 (25.0–96.3) | 0.21 |
| p16INK4a expression ≤ 16.8%, n (%) b | 17 (21) | 14 (33) | 3 (8) | 0.01 |
| p16INK4a expression > 16.8%, n (%) b | 64 (79) | 28 (67) | 36 (92) | |
| Host Mucosal Immune-Related Biomarkers in the Larynx | ||||
| cPI for B2M, (%), median (range) | 82.9 (68.2–96.3) | 92.5 (79.5–97.6) | 74.1 (55.9–84.0) | 0.001 |
| B2M expression ≥ 84.3%, n (%) b | 37 (46) | 29 (69) | 8 (21) | <0.001 |
| B2M expression < 84.3%, n (%) b | 44 (54) | 13 (31) | 31 (79) | |
| cPI for CD3, (%), median (range) | 11.3 (3.8–27.3) | 20.5 (9.1–32.8) | 6.3 (2.0–15.4) | 0.001 |
| CD3 expression ≥ 6.9%, n (%) b | 51 (63) | 35 (83) | 16 (41) | <0.001 |
| CD3 expression < 6.9%, n (%) b | 30 (37) | 7 (17) | 23 (59) | |
| cPI for CD161, (%), median (range) | 47.9 (14.7–76.9) | 59.8 (23.5–80.9) | 32.0 (6.7–67.3) | 0.004 |
| CD161 expression ≥ 68.8%, n (%) b | 28 (35) | 20 (48) | 8 (21) | 0.02 |
| CD161 expression < 68.8%, n (%) b | 53 (65) | 22 (52) | 31 (80) | |
| Characteristics | LSCC | Male Sex | Age ≥ 55 Years | Cigarette Smoking | Alcohol Consumption | EBV DNA Positivity | EBER Signal ≥ 0.02% | BCL-2 Expression ≥ 6 8.6% | MYC Expression ≤ 0.44% | p16INK4a Expression ≤ 16.8% | B2M Expression ≥ 84.3% | CD3 Expression ≥ 6.9% | CD161 Expression ≥ 68.8% |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| LSCC | – | ||||||||||||
| Male sex | −0.03 | – | |||||||||||
| Age ≥ 55 years | 0.48 c | −0.01 | – | ||||||||||
| Cigarette smoking | 0.25 a | 0.46 c | 0.08 | – | |||||||||
| Alcohol consumption | 0.11 | 0.28 a | −0.08 | 0.32 b | – | ||||||||
| EBV DNA positivity | 0.44 c | −0.20 | 0.18 | 0.05 | −0.04 | – | |||||||
| EBER signal ≥ 0.02% | 0.11 | 0.12 | 0.22 | 0.01 | −0.03 | −0.12 | – | ||||||
| BCL-2 expression ≥ 68.6% | 0.39 c | −0.05 | 0.09 | −0.06 | 0.04 | 0.26 a | 0.03 | – | |||||
| MYC expression ≤ 0.44% | 0.26 a | −0.19 | 0.33 b | 0.14 | −0.16 | 0.14 | 0.15 | 0.07 | – | ||||
| p16INK4a expression ≤ 16.8% | 0.32 b | −0.06 | −0.09 | 0.14 | 0.12 | 0.08 | −0.03 | 0.14 | 0.05 | – | |||
| B2M expression ≥ 84.3% | 0.49 c | 0.02 | 0.22 a | 0.06 | 0.11 | 0.14 | 0.06 | 0.24 a | 0.14 | 0.32 b | – | ||
| CD3 expression ≥ 6.9% | 0.44 c | −0.05 | 0.24 a | 0.08 | 0.15 | 0.20 | 0.13 | 0.52 c | 0.17 | 0.21 | 0.40 c | – | |
| CD161 expression ≥ 68.8% | 0.29 a | −0.05 | 0.02 | 0.11 | −0.03 | 0.06 | 0.20 | 0.24 a | 0.20 | 0.39 c | 0.17 | 0.29 b | – |
| Characteristics | 5-Year Local Recurrence | 5-Year Disease-Free Survival | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Cut-Off Value | Univariate Models | Multivariate Models | Cut-Off Value | Univariate Models | Multivariate Models | |||||
| HR (95% CI) | p-Value | HR (95% CI) | p-Value | HR (95% CI) | p-Value | HR (95% CI) | p-Value | |||
| Male sex | yes | 0.5 (0.1–2.5) | 0.43 | yes | 0.7 (0.2–3.1) | 0.65 | ||||
| Age, years | ≤63 | 3.2 (1.0–10.3) | 0.048 | – | NS | ≤63 | 1.8 (0.7–4.8) | 0.23 | ||
| Cigarette smoking | yes | 23.2 (0.1–82375.3) | 0.45 | yes | 23.6 (0.1–29976.8) | 0.39 | ||||
| Alcohol drinking | yes | 2.2 (0.6–7.9) | 0.23 | yes | 2.0 (0.6–6.1) | 0.23 | ||||
| T-stage | ≤2 | 30.7 (0.2–6190.3) | 0.21 | ≤3 | 32.8 (0.3–3521.6) | 0.14 | ||||
| N-stage | 0 | 22.0 (0–4.4 × 105) | 0.54 | ≤0 | 22.3 (0.1–1.3×105) | 0.48 | ||||
| pStage | ≤2 | 30.7 (0.2–6190.3) | 0.21 | ≤2 | 32.8 (0.3–3521.6) | 0.14 | ||||
| Treatment | TLM | 8.3 (1.1–63.8) | 0.04 | – | NS | TLM | 5.6 (1.3–24.9) | 0.02 | – | NS |
| EBV DNA positivity | yes | 0.56 (0.2–1.64) | 0.28 | yes | 0.4 (0.1–1.1) | 0.08 | ||||
| EBER signal, % | ≥0.04 | 6.2 (2.0–18.6) | 0.001 | 6.0 (1.9–18.6) | 0.002 | ≥0.04 | 7.1 (2.6–19.5) | <0.001 | 8.6 (2.9–25.3) | <0.001 |
| BCL-2 expression, % | ≤96.0 | 2.9 (0.8–10.6) | 0.10 | ≤96.0 | 3.9 (1.1–13.6) | 0.03 | – | NS | ||
| MYC expression, % | ≤0.50 | 3.1 (0.7–13.8) | 0.14 | ≤0.50 | 4.2 (1.0–18.6) | 0.06 | ||||
| p16INK4a expression, % | ≤81.6 | 1.1 (0.4–3.4) | 0.83 | ≤81.6 | 2.1 (0.7–6.0) | 0.16 | ||||
| B2M expression, % | ≤92.7 | 3.5 (1.0–12.7) | 0.05 | ≤93.8 | 2.0 (0.7–5.7) | 0.19 | ||||
| CD3 expression, % | ≤4.9 | 7.2 (2.2–1209) | 0.001 | 6.9 (1.9–24.6) | 0.003 | ≤4.9 | 6.7 (2.1–21.8) | 0.001 | 6.6 (1.9–23.6) | 0.004 |
| CD161 expression, % | ≤69.9 | 2.4 (0.7–7.5) | 0.15 | ≤73.1 | 1.8 (0.6–5.2) | 0.26 | ||||
| Characteristics | Male Sex | Age ≤ 63 years | Cigarette Smoking | Alcohol Consumption | T-Stage ≤ 2 | N-Stage = 0 | Stage ≤ 2 | Laryngeal EBV DNA Positivity | EBER Signal ≥ 0.04% | BCL-2 Expression ≤ 96.0% | MYC Expression ≤ 0.50% | p16INK4a Expression ≤ 81.6% | B2M Expression ≤ 92.7% | CD3 Expression ≤ 4.9% | CD161 Expression ≤ 69.9% |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Male sex | – | ||||||||||||||
| Age ≤63 years | −0.02 | – | |||||||||||||
| Cigarette smoking | 0.54 a | 0.08 | – | ||||||||||||
| Alcohol consumption | 0.27 | 0.31 b | 0.37 b | – | |||||||||||
| T-stage ≤ 2 | 0.13 | −0.10 | 0.25 | −0.02 | – | ||||||||||
| N-stage = 0 | −0.07 | −0.01 | −0.06 | 0.07 | 0.20 | – | |||||||||
| Stage ≤ 2 | 0.02 | −0.15 | 0.10 | 0.02 | 0.44 c | 0.46 c | – | ||||||||
| Laryngeal EBV DNA positivity | −0.34 b | −0.15 | −0.11 | 0.01 | −0.29 | −0.24 | −0.27 | – | |||||||
| EBER signal ≥ 0.04% | 0.03 | 0.24 | 0.18 | 0.14 | 0.06 | 0.14 | 0.31 b | −0.18 | – | ||||||
| BCL-2 expression ≤ 96.0% | 0.06 | 0.20 | 0.15 | 0.20 | 0.07 | −0.18 | 0.22 | −0.09 | 0.20 | – | |||||
| MYC expression ≤ 0.50% | −0.22 | −0.29 | 0.01 | −0.18 | 0.20 | 0.09 | 0.20 | 0.02 | 0.20 | 0.08 | – | ||||
| p16INK4a expression ≤ 81.6% | 0.05 | 0.44 c | 0.13 | 0.16 | 0.11 | 0.03 | 0.07 | −0.23 | 0.12 | 0.36 b | 0.25 | – | |||
| B2M expression ≤ 92.7% | 0.03 | 0.01 | −0.07 | 0.12 | 0.06 | 0.02 | 0.05 | 0.20 | 0.15 | 0.13 | 0.12 | −0.21 | – | ||
| CD3 expression ≤ 4.9% | 0.12 | 0.09 | 0.10 | −0.03 | 0.11 | 0.08 | 0.18 | −0.20 | 0.09 | 0.30 | 0.09 | 0.32 b | 0.19 | – | |
| CD161 expression ≤ 69.9% | 0.03 | 0.01 | −0.07 | 0.02 | −0.04 | 0.02 | 0.05 | 0.01 | 0.15 | 0.32 b | −0.20 | −0.30 | 0.42 c | 0.19 | – |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lee, L.-A.; Fang, T.-J.; Li, H.-Y.; Chuang, H.-H.; Kang, C.-J.; Chang, K.-P.; Liao, C.-T.; Chen, T.-C.; Huang, C.-G.; Yen, T.-C. Effects of Epstein-Barr Virus Infection on the Risk and Prognosis of Primary Laryngeal Squamous Cell Carcinoma: A Hospital-Based Case-Control Study in Taiwan. Cancers 2021, 13, 1741. https://doi.org/10.3390/cancers13071741
Lee L-A, Fang T-J, Li H-Y, Chuang H-H, Kang C-J, Chang K-P, Liao C-T, Chen T-C, Huang C-G, Yen T-C. Effects of Epstein-Barr Virus Infection on the Risk and Prognosis of Primary Laryngeal Squamous Cell Carcinoma: A Hospital-Based Case-Control Study in Taiwan. Cancers. 2021; 13(7):1741. https://doi.org/10.3390/cancers13071741
Chicago/Turabian StyleLee, Li-Ang, Tuan-Jen Fang, Hsueh-Yu Li, Hai-Hua Chuang, Chung-Jan Kang, Kai-Ping Chang, Chun-Ta Liao, Tse-Ching Chen, Chung-Guei Huang, and Tzu-Chen Yen. 2021. "Effects of Epstein-Barr Virus Infection on the Risk and Prognosis of Primary Laryngeal Squamous Cell Carcinoma: A Hospital-Based Case-Control Study in Taiwan" Cancers 13, no. 7: 1741. https://doi.org/10.3390/cancers13071741
APA StyleLee, L.-A., Fang, T.-J., Li, H.-Y., Chuang, H.-H., Kang, C.-J., Chang, K.-P., Liao, C.-T., Chen, T.-C., Huang, C.-G., & Yen, T.-C. (2021). Effects of Epstein-Barr Virus Infection on the Risk and Prognosis of Primary Laryngeal Squamous Cell Carcinoma: A Hospital-Based Case-Control Study in Taiwan. Cancers, 13(7), 1741. https://doi.org/10.3390/cancers13071741







