Carotenoids in Cancer Apoptosis—The Road from Bench to Bedside and Back
Abstract
1. Introduction
1.1. Aim of the Study
1.2. Source of the Data
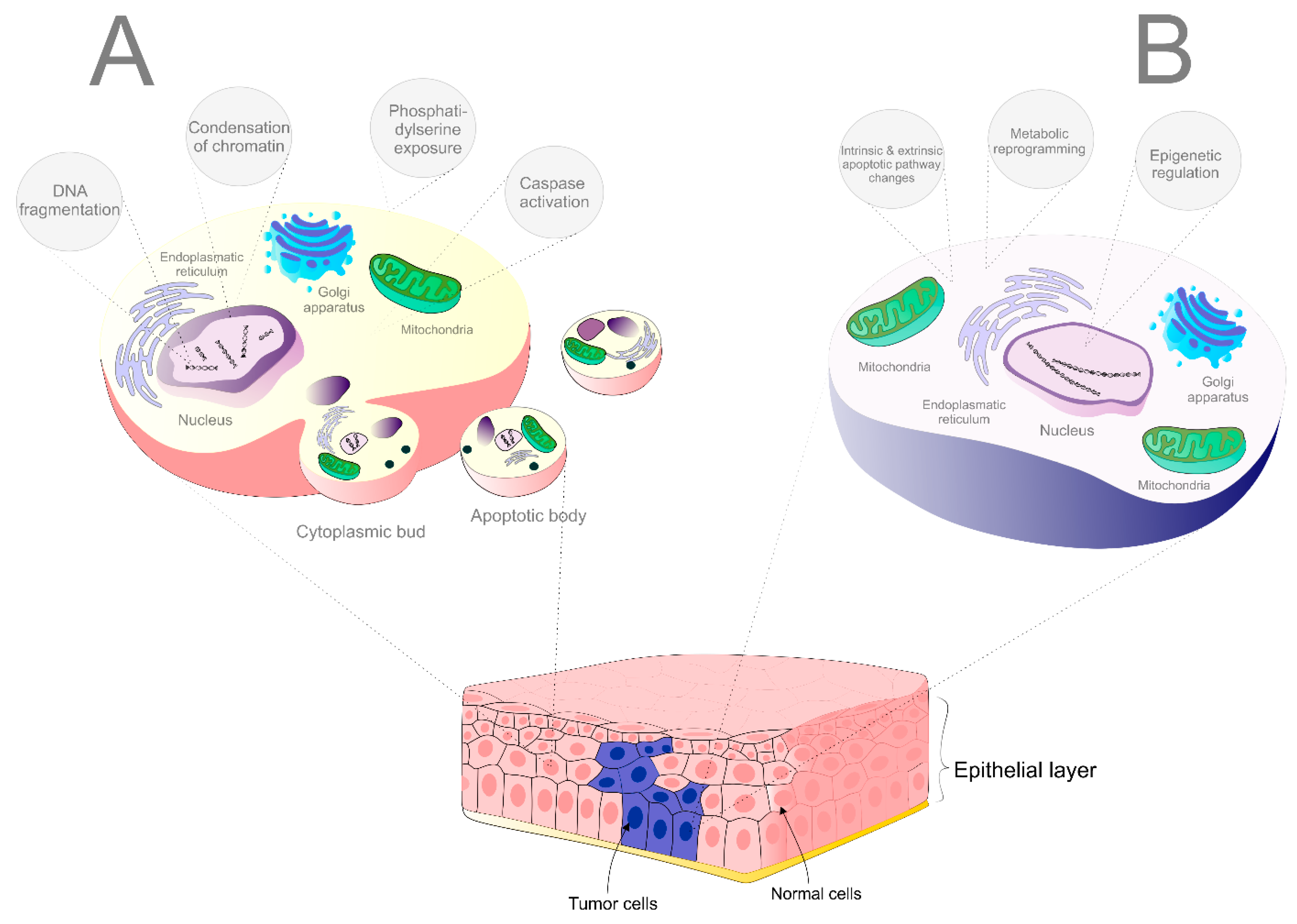
2. Insights into the Cell Death Mechanisms
3. A Molecular View and Pathways Involved in Apoptosis
3.1. Extrinsic Pathway
3.2. Intrinsic Pathway
3.3. Other Mechanisms Involved in Regulation of Apoptosis
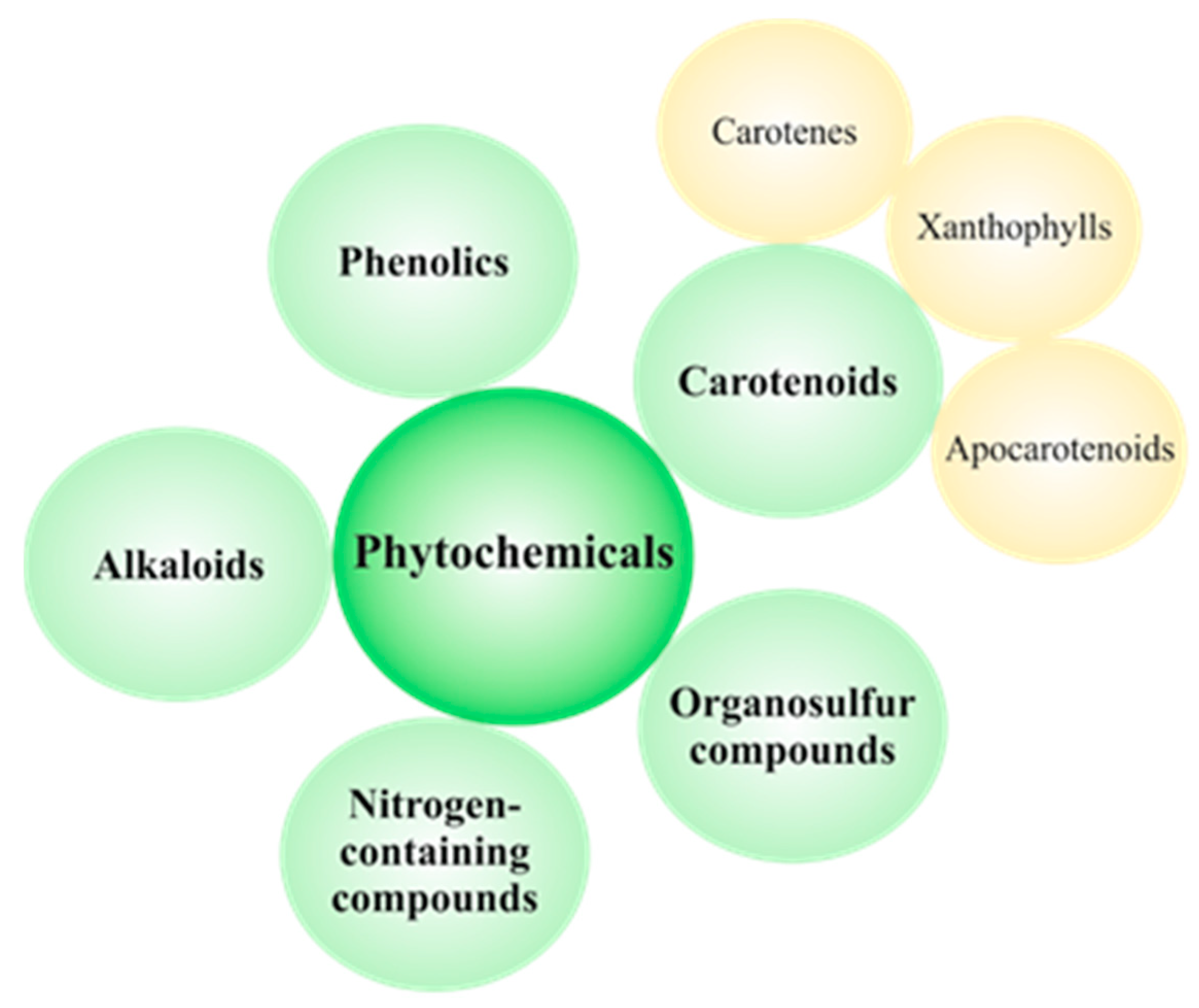
4. Carotenoids
5. The Use of Carotenoids in Preclinical Cancer Research
5.1. Carotenoids-Induced Apoptosis in In Vitro and In Vivo Studies
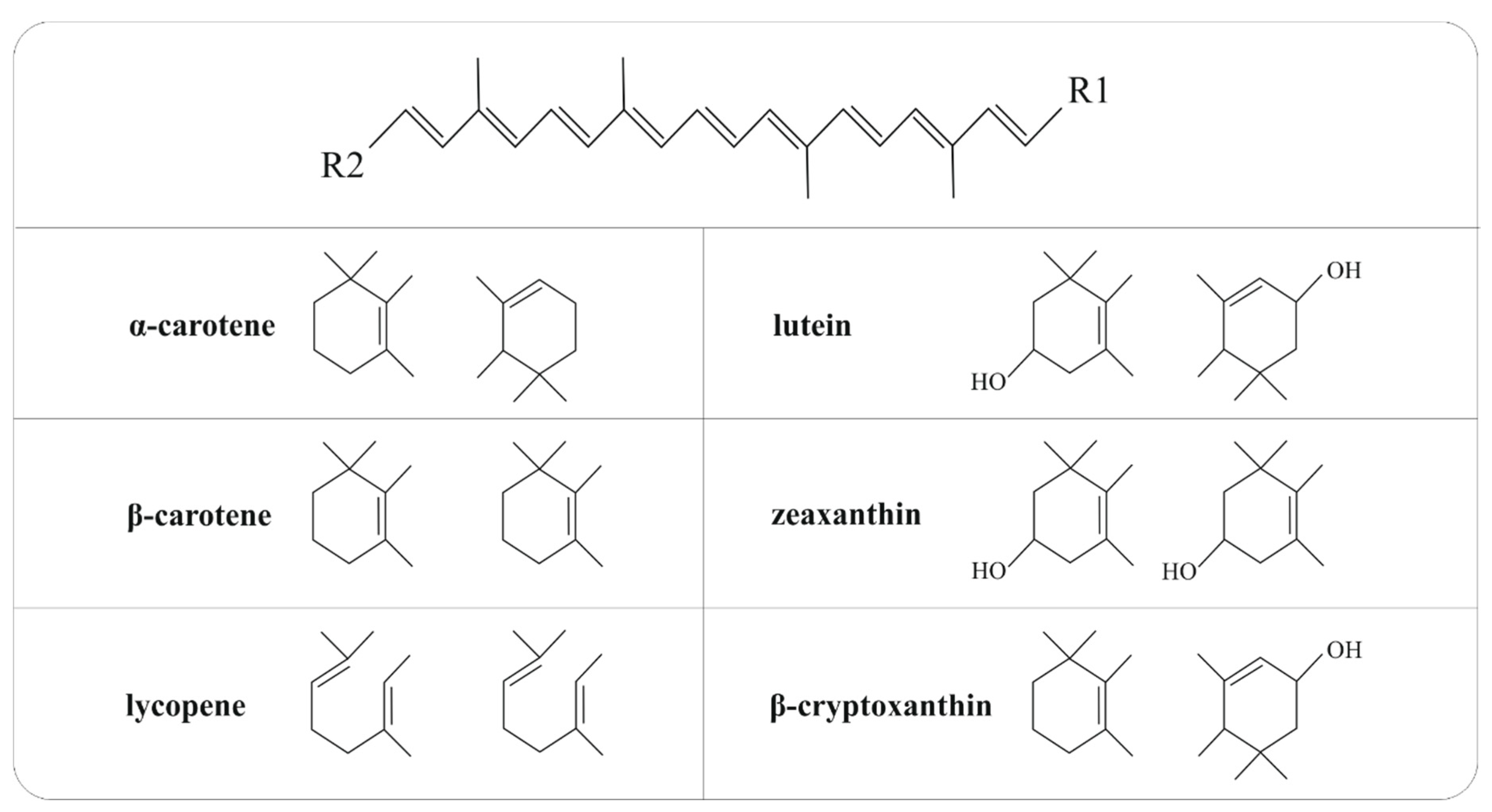
5.1.1. Carotenes
β-Carotene
Lycopene
5.1.2. Xanthophylls
Lutein
β-Cryptoxanthin
Astaxanthin
Fucoxanthin
Fucoxanthinol
Deinoxanthin
5.1.3. Apocarotenoids
β-Ionone
Crocetin
Crocin
Picrocrocin
Bixin
Retinoids
5.2. Nanotechnology and Carotenoids
6. Carotenoids in Clinical Research
Carotenoids in Clinical Research Targeting Apoptosis
7. The Content and Bioavailability of Carotenoids
7.1. The Effects of Cooking Method on the Content of Carotenoids
7.2. Current Trends in the Bioavailability of Carotenoids
7.3. The Effects of Dietary Constituents on Carotenoid Bioavailability
7.4. Biofortification as a Way to Increase the Content and Bioavailability of Carotenoids
7.5. Solid Dispersion and Microemulsions as a Mean of Increased Solubility and Bioavailability of Carotenoids
8. The Utility Potential of Carotenoids in the Framework of 3P Medicine
8.1. The Level of General Cancer Prevention
8.2. The Level of Cancer Prevention in Stratified Groups at Increased Cancer Risk
8.3. Cancer Prevention in Relevant Syndromes and Comorbid Conditions
8.4. Secondary Chemoprevention of Metastatic Disease
9. Conclusions and Expert Recommendations
Author Contributions
Funding
Conflicts of Interest
References
- Safarzadeh, E.; Sandoghchian Shotorbani, S.; Baradaran, B. Herbal Medicine as Inducers of Apoptosis in Cancer Treatment. Adv. Pharm. Bull. 2014, 4, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Baba, A.I.; Câtoi, C. Carcinogenesis. In Comparative Oncology; The Publishing House of the Romanian Academy: Bucharest, Romania, 2007. [Google Scholar]
- Koklesova, L.; Liskova, A.; Samec, M.; Qaradakhi, T.; Zulli, A.; Smejkal, K.; Kajo, K.; Jakubikova, J.; Behzadi, P.; Pec, M.; et al. Genoprotective activities of plant natural substances in cancer and chemopreventive strategies in the context of 3P medicine. EPMA J. 2020, 11, 261–287. [Google Scholar] [CrossRef] [PubMed]
- Prakash, O.; Kumar, A.; Kumar, P.; Ajeet, A. Anticancer Potential of Plants and Natural Products: A Review. Am. J. Pharmacol. Sci. 2013, 1, 104–115. [Google Scholar] [CrossRef]
- Yang, H.-L.; Chen, C.-S.; Chang, W.-H.; Lu, F.-J.; Lai, Y.-C.; Chen, C.-C.; Hseu, T.-H.; Kuo, C.-T.; Hseu, Y.-C. Growth inhibition and induction of apoptosis in MCF-7 breast cancer cells by Antrodia camphorata. Cancer Lett. 2006, 231, 215–227. [Google Scholar] [CrossRef]
- Sarkar, S.; Horn, G.; Moulton, K.; Oza, A.; Byler, S.; Kokolus, S.; Longacre, M. Cancer Development, Progression, and Therapy: An Epigenetic Overview. Int. J. Mol. Sci. 2013, 14, 21087–21113. [Google Scholar] [CrossRef]
- Pfeffer, C.M.; Singh, A.T.K. Apoptosis: A Target for Anticancer Therapy. Int. J. Mol. Sci. 2018, 19, 448. [Google Scholar] [CrossRef]
- Green, D.R.; Galluzzi, L.; Kroemer, G. Metabolic control of cell death. Science 2014, 345, 1250256. [Google Scholar] [CrossRef]
- Jasek, K.; Kubatka, P.; Samec, M.; Liskova, A.; Smejkal, K.; Vybohova, D.; Bugos, O.; Biskupska-Bodova, K.; Bielik, T.; Zubor, P.; et al. DNA Methylation Status in Cancer Disease: Modulations by Plant-Derived Natural Compounds and Dietary Interventions. Biomolecules 2019, 9, 289. [Google Scholar] [CrossRef]
- Carneiro, B.A.; El-Deiry, W.S. Targeting apoptosis in cancer therapy. Nat. Rev. Clin. Oncol. 2020, 17, 395–407. [Google Scholar] [CrossRef]
- Gatenby, R.; Brown, J. The Evolution and Ecology of Resistance in Cancer Therapy. Cold Spring Harb. Perspect. Med. 2018, 8, a033415. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Lin, D. 25–Molecular Aspects of Carotenoid Metabolizing Enzymes and Implications for Ophthalmology. In Handbook of Nutrition, Diet, and the Eye, 2nd ed.; Preedy, V.R., Watson, R.R., Eds.; Academic Press: Cambridge, MA, USA, 2019; pp. 415–424. ISBN 978-0-12-815245-4. [Google Scholar]
- Langi, P.; Kiokias, S.; Varzakas, T.; Proestos, C. Carotenoids: From Plants to Food and Feed Industries. Methods Mol. Biol. Clifton NJ 2018, 1852, 57–71. [Google Scholar] [CrossRef]
- Davinelli, S.; Nielsen, M.E.; Scapagnini, G. Astaxanthin in Skin Health, Repair, and Disease: A Comprehensive Review. Nutrients 2018, 10, 522. [Google Scholar] [CrossRef] [PubMed]
- Rao, A.; Rao, L. Carotenoids and human health. Pharmacol. Res. 2007, 55, 207–216. [Google Scholar] [CrossRef]
- Johnson, E.J. The role of carotenoids in human health. Nutr. Clin. Care Off. Publ. Tufts Univ. 2002, 5, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Olson, J.A. Benefits and liabilities of vitamin A and carotenoids. J. Nutr. 1996, 126, 1208S–1212S. [Google Scholar] [CrossRef]
- Xavier, A.A.O.; Pérez-Gálvez, A. Carotenoids as a Source of Antioxidants in the Diet. In Carotenoids in Nature; Stange, C., Ed.; Subcellular Biochemistry; Springer International Publishing: Cham, Switzerland, 2016; Volume 79, pp. 359–375. ISBN 978-3-319-39124-3. [Google Scholar]
- Chew, B.P.; Park, J.S.; Wong, M.W.; Wong, T.S. A comparison of the anticancer activities of dietary beta-carotene, canthaxanthin and astaxanthin in mice in vivo. Anticancer Res. 1999, 19, 1849–1853. [Google Scholar]
- Devasagayam, T.P.A.; Tilak, J.C.; Boloor, K.K.; Sane, K.S.; Ghaskadbi, S.S.; Lele, R.D. Free radicals and antioxidants in human health: Current status and future prospects. J. Assoc. Physicians India 2004, 52, 794–804. [Google Scholar]
- Abidov, M.; Ramazanov, Z.; Seifulla, R.; Grachev, S. The effects of Xanthigen in the weight management of obese premenopausal women with non-alcoholic fatty liver disease and normal liver fat. Diabetes Obes. Metab. 2010, 12, 72–81. [Google Scholar] [CrossRef]
- Nishino, H.; Murakosh, M.; Ii, T.; Takemura, M.; Kuchide, M.; Kanazawa, M.; Mou, X.Y.; Wada, S.; Masuda, M.; Ohsaka, Y.; et al. Carotenoids in cancer chemoprevention. Cancer Metastasis Rev. 2002, 21, 257–264. [Google Scholar] [CrossRef]
- Chatterjee, M.; Roy, K.; Janarthan, M.; Das, S.; Chatterjee, M. Biological activity of carotenoids: Its implications in cancer risk and prevention. Curr. Pharm. Biotechnol. 2012, 13, 180–190. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Dang, F.; Deng, C. β-Cryptoxanthin induced anti-proliferation and apoptosis by G0/G1 arrest and AMPK signal inactivation in gastric cancer. Eur. J. Pharmacol. 2019, 859, 172528. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.-S.; Lee, H.-A.; Lim, J.Y.; Kim, Y.; Jung, C.-H.; Yoo, S.-H.; Kim, Y. β-Carotene inhibits neuroblastoma cell invasion and metastasis in vitro and in vivo by decreasing level of hypoxia-inducible factor-1α. J. Nutr. Biochem. 2014, 25, 655–664. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.-Y.; Huang, S.-M.; Yang, C.-M.; Hu, M.-L. Diverse Effects of β-Carotene on Secretion and Expression of VEGF in Human Hepatocarcinoma and Prostate Tumor Cells. Molecules 2012, 17, 3981–3988. [Google Scholar] [CrossRef] [PubMed]
- Chryssanthi, D.G.; Lamari, F.N.; Iatrou, G.; Pylara, A.; Karamanos, N.K.; Cordopatis, P. Inhibition of breast cancer cell proliferation by style constituents of different Crocus species. Anticancer Res. 2007, 27, 357–362. [Google Scholar] [PubMed]
- Tang, L.; Jin, T.; Zeng, X.; Wang, J.-S. Lycopene inhibits the growth of human androgen-independent prostate cancer cells in vitro and in BALB/c nude mice. J. Nutr. 2005, 135, 287–290. [Google Scholar] [CrossRef]
- Guo, L.; Zhu, H.; Lin, C.; Che, J.; Tian, X.; Han, S.; Zhao, H.; Zhu, Y.; Mao, D. Associations between antioxidant vitamins and the risk of invasive cervical cancer in Chinese women: A case-control study. Sci. Rep. 2015, 5, 13607. [Google Scholar] [CrossRef]
- Jeong, Y.; Lim, J.W.; Kim, H. Lycopene Inhibits Reactive Oxygen Species-Mediated NF-κB Signaling and Induces Apoptosis in Pancreatic Cancer Cells. Nutrients 2019, 11, 762. [Google Scholar] [CrossRef]
- Gong, X.; Smith, J.; Swanson, H.; Rubin, L. Carotenoid Lutein Selectively Inhibits Breast Cancer Cell Growth and Potentiates the Effect of Chemotherapeutic Agents through ROS-Mediated Mechanisms. Molecules 2018, 23, 905. [Google Scholar] [CrossRef]
- Ishijima, N.; Kanki, K.; Shimizu, H.; Shiota, G. Activation of AMP-activated protein kinase by retinoic acid sensitizes hepatocellular carcinoma cells to apoptosis induced by sorafenib. Cancer Sci. 2015, 106, 567–575. [Google Scholar] [CrossRef]
- Mollaei, H.; Safaralizadeh, R.; Babaei, E.; Abedini, M.R.; Hoshyar, R. The anti-proliferative and apoptotic effects of crocin on chemosensitive and chemoresistant cervical cancer cells. Biomed. Pharmacother. 2017, 94, 307–316. [Google Scholar] [CrossRef] [PubMed]
- Dowlatshahi, K.; Mehta, R.G.; Thomas, C.F.; Dinger, N.M.; Moon, R.C. Therapeutic effect of N-(4-hydroxyphenyl)retinamide on N-methyl-N-nitrosourea-induced rat mammary cancer. Cancer Lett. 1989, 47, 187–192. [Google Scholar] [CrossRef]
- Green, A.; Shilkaitis, A.; Christov, K. 4-(hydroxyphenyl)retinamide selectively inhibits the development and progression of ductal hyperplastic lesions and carcinoma in situ in mammary gland. Carcinogenesis 1999, 20, 1535–1540. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Rao, R.D.; Cobleigh, M.A.; Gray, R.; Graham, M.L.; Norton, L.; Martino, S.; Budd, G.T.; Ingle, J.N.; Wood, W.C. Phase III double-blind, placebo-controlled, prospective randomized trial of adjuvant tamoxifen vs. tamoxifen and fenretinide in postmenopausal women with positive receptors (EB193): An intergroup trial coordinated by the Eastern Cooperative Oncology Group. Med. Oncol. Northwood Lond. Engl. 2011, 28, S39–S47. [Google Scholar] [CrossRef]
- Puntoni, M.; Petrera, M.; Campora, S.; Garrone, E.; Defferrari, C.; Torrisi, R.; Johansson, H.; Bruno, S.; Curotto, A.; DeCensi, A. Prognostic Significance of VEGF after Twenty-Year Follow-up in a Randomized Trial of Fenretinide in Non-Muscle-Invasive Bladder Cancer. Cancer Prev. Res. 2016, 9, 437–444. [Google Scholar] [CrossRef]
- Moore, M.M.; Stockler, M.; Lim, R.; Mok, T.S.K.; Millward, M.; Boyer, M.J. A phase II study of fenretinide in patients with hormone refractory prostate cancer: A trial of the Cancer Therapeutics Research Group. Cancer Chemother. Pharmacol. 2010, 66, 845–850. [Google Scholar] [CrossRef]
- Rodon, J.; Jacobs, C.D.; Chu, Q.; Rowinsky, E.K.; Lopez-Anaya, A.; Takimoto, C.H.; Wakelee, H.A. A phase I pharmacokinetic study of bexarotene with paclitaxel and carboplatin in patients with advanced non-small cell lung cancer (NSCLC). Cancer Chemother. Pharmacol. 2012, 69, 825–834. [Google Scholar] [CrossRef]
- Wakelee, H.A.; Middleton, G.; Dunlop, D.; Ramlau, R.; Leighl, N.; Hao, D.; Lopez-Anaya, A.; Zatloukal, P.; Jacobs, C.D. A phase I pharmacokinetic study of bexarotene with vinorelbine and cisplatin in patients with advanced non-small-cell lung cancer (NSCLC). Cancer Chemother. Pharmacol. 2012, 69, 815–824. [Google Scholar] [CrossRef]
- Ianhez, M.; Pinto, S.A.; Miot, H.A.; Bagatin, E. A randomized, open, controlled trial of tretinoin 0.05% cream vs. low-dose oral isotretinoin for the treatment of field cancerization. Int. J. Dermatol. 2019, 58, 365–373. [Google Scholar] [CrossRef]
- Jan, R.; Chaudhry, G.-S. Understanding Apoptosis and Apoptotic Pathways Targeted Cancer Therapeutics. Adv. Pharm. Bull. 2019, 9, 205–218. [Google Scholar] [CrossRef]
- Hotchkiss, R.S.; Strasser, A.; McDunn, J.E.; Swanson, P.E. Cell Death in Disease: Mechanisms and Emerging Therapeutic Concepts. N. Engl. J. Med. 2009, 361, 1570–1583. [Google Scholar] [CrossRef] [PubMed]
- D’Arcy, M.S. Cell death: A review of the major forms of apoptosis, necrosis and autophagy. Cell Biol. Int. 2019, 43, 582–592. [Google Scholar] [CrossRef] [PubMed]
- Dhuriya, Y.K.; Sharma, D. Necroptosis: A regulated inflammatory mode of cell death. J. Neuroinflamm. 2018, 15, 199. [Google Scholar] [CrossRef] [PubMed]
- Dillon, C.P.; Douglas, R.G. Molecular Cell Biology of Apoptosis and Necroptosis in Cancer. Available online: https://pubmed.ncbi.nlm.nih.gov/27558815/ (accessed on 31 July 2020).
- Tait, S.W.G.; Ichim, G.; Green, D.R. Die another way—Non-apoptotic mechanisms of cell death. J. Cell Sci. 2014, 127, 2135–2144. [Google Scholar] [CrossRef]
- Favaloro, B.; Allocati, N.; Graziano, V.; Di Ilio, C.; De Laurenzi, V. Role of Apoptosis in disease. Aging 2012, 4, 330–349. [Google Scholar] [CrossRef]
- Parrish, A.B.; Freel, C.D.; Kornbluth, S. Cellular Mechanisms Controlling Caspase Activation and Function. Cold Spring Harb. Perspect. Biol. 2013, 5, a008672. [Google Scholar] [CrossRef]
- Cerella, C.; Grandjenette, C.; Diederich, M.D.M. Roles of Apoptosis and Cellular Senescence in Cancer and Aging. Available online: http://www.eurekaselect.com/128138/article (accessed on 25 May 2020).
- Elmore, S. Apoptosis: A Review of Programmed Cell Death. Toxicol. Pathol. 2007, 35, 495–516. [Google Scholar] [CrossRef]
- Fouad, Y.A.; Aanei, C. Revisiting the hallmarks of cancer. Am. J. Cancer Res. 2017, 7, 1016–1036. [Google Scholar]
- Sharma, A.; Boise, L.H.; Shanmugam, M. Cancer Metabolism and the Evasion of Apoptotic Cell Death. Cancers 2019, 11, 44. [Google Scholar] [CrossRef]
- Pistritto, G.; Trisciuoglio, D.; Ceci, C.; Garufi, A.; D’Orazi, G. Apoptosis as anticancer mechanism: Function and dysfunction of its modulators and targeted therapeutic strategies. Aging 2016, 8, 603–619. [Google Scholar] [CrossRef]
- Hassan, M.; Watari, H.; AbuAlmaaty, A.; Ohba, Y.; Sakuragi, N. Apoptosis and molecular targeting therapy in cancer. BioMed Res. Int. 2014, 2014, 150845. [Google Scholar] [CrossRef] [PubMed]
- Baig, S.; Seevasant, I.; Mohamad, J.; Mukheem, A.; Huri, H.Z.; Kamarul, T. Potential of apoptotic pathway-targeted cancer therapeutic research: Where do we stand? Cell Death Dis. 2016, 7, e2058. [Google Scholar] [CrossRef] [PubMed]
- Shakibaei, M.; Harikumar, K.B.; Aggarwal, B.B. Resveratrol addiction: To die or not to die. Mol. Nutr. Food Res. 2009, 53, 115–128. [Google Scholar] [CrossRef] [PubMed]
- Giménez-Bonafé, P.; Tortosa, A.; Pérez-Tomás, R. Overcoming drug resistance by enhancing apoptosis of tumor cells. Curr. Cancer Drug Targets 2009, 9, 320–340. [Google Scholar] [CrossRef] [PubMed]
- Fulda, S. Targeting apoptosis for anticancer therapy. Semin. Cancer Biol. 2015, 31, 84–88. [Google Scholar] [CrossRef] [PubMed]
- Guicciardi, M.E.; Gores, G.J. Life and death by death receptors. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2009, 23, 1625–1637. [Google Scholar] [CrossRef]
- Boatright, K.M.; Salvesen, G.S. Mechanisms of caspase activation. Curr. Opin. Cell Biol. 2003, 15, 725–731. [Google Scholar] [CrossRef]
- Degterev, A.; Boyce, M.; Yuan, J. A decade of caspases. Oncogene 2003, 22, 8543–8567. [Google Scholar] [CrossRef]
- Plati, J.; Bucur, O.; Khosravi-Far, R. Dysregulation of Apoptotic Signaling in Cancer: Molecular Mechanisms and Therapeutic Opportunities. J. Cell. Biochem. 2008, 104, 1124–1149. [Google Scholar] [CrossRef]
- Green, D.R.; Kroemer, G. The pathophysiology of mitochondrial cell death. Science 2004, 305, 626–629. [Google Scholar] [CrossRef]
- Kroemer, G.; Galluzzi, L.; Brenner, C. Mitochondrial membrane permeabilization in cell death. Physiol. Rev. 2007, 87, 99–163. [Google Scholar] [CrossRef] [PubMed]
- Danial, N.N.; Korsmeyer, S.J. Cell death: Critical control points. Cell 2004, 116, 205–219. [Google Scholar] [CrossRef]
- Bratton, S.B.; Salvesen, G.S. Regulation of the Apaf-1-caspase-9 apoptosome. J. Cell Sci. 2010, 123, 3209–3214. [Google Scholar] [CrossRef] [PubMed]
- Green, D.R.; Llambi, F. Cell Death Signaling. Cold Spring Harb. Perspect. Biol. 2015, 7, a006080. [Google Scholar] [CrossRef]
- Salvesen, G.S.; Duckett, C.S. IAP proteins: Blocking the road to death’s door. Nat. Rev. Mol. Cell Biol. 2002, 3, 401–410. [Google Scholar] [CrossRef]
- LaCasse, E.C.; Mahoney, D.J.; Cheung, H.H.; Plenchette, S.; Baird, S.; Korneluk, R.G. IAP-targeted therapies for cancer. Oncogene 2008, 27, 6252–6275. [Google Scholar] [CrossRef]
- Sacconi, A.; Biagioni, F.; Canu, V.; Mori, F.; Di Benedetto, A.; Lorenzon, L.; Ercolani, C.; Di Agostino, S.; Cambria, A.M.; Germoni, S.; et al. miR-204 targets Bcl-2 expression and enhances responsiveness of gastric cancer. Cell Death Dis. 2012, 3, e423. [Google Scholar] [CrossRef]
- Ovcharenko, D.; Kelnar, K.; Johnson, C.; Leng, N.; Brown, D. Genome-scale microRNA and small interfering RNA screens identify small RNA modulators of TRAIL-induced apoptosis pathway. Cancer Res. 2007, 67, 10782–10788. [Google Scholar] [CrossRef]
- Pekarsky, Y.; Balatti, V.; Croce, C.M. BCL2 and miR-15/16: From gene discovery to treatment. Cell Death Differ. 2018, 25, 21–26. [Google Scholar] [CrossRef]
- Hervouet, E.; Cheray, M.; Vallette, F.M.; Cartron, P.-F. DNA Methylation and Apoptosis Resistance in Cancer Cells. Cells 2013, 2, 545–573. [Google Scholar] [CrossRef]
- Cheng, M.-F.; Lee, C.-H.; Hsia, K.-T.; Huang, G.-S.; Lee, H.-S. Methylation of histone H3 lysine 27 associated with apoptosis in osteosarcoma cells induced by staurosporine. Histol. Histopathol. 2009, 24, 1105. [Google Scholar] [PubMed]
- Zhao, R.; Han, C.; Eisenhauer, E.; Kroger, J.; Zhao, W.; Yu, J.; Selvendiran, K.; Liu, X.; Wani, A.A.; Wang, Q.-E. DNA Damage-Binding Complex Recruits HDAC1 to Repress Bcl-2 Transcription in Human Ovarian Cancer Cells. Mol. Cancer Res. 2014, 12, 370–380. [Google Scholar] [CrossRef] [PubMed]
- Zou, Z.-K.; Huang, Y.-Q.; Zou, Y.; Zheng, X.-K.; Ma, X.-D. Silencing of LSD1 gene modulates histone methylation and acetylation and induces the apoptosis of JeKo-1 and MOLT-4 cells. Int. J. Mol. Med. 2017, 40, 319–328. [Google Scholar] [CrossRef] [PubMed]
- Potter, M.; Newport, E.; Morten, K.J. The Warburg effect: 80 years on. Biochem. Soc. Trans. 2016, 44, 1499–1505. [Google Scholar] [CrossRef]
- Choi, C.-I. Astaxanthin as a Peroxisome Proliferator-Activated Receptor (PPAR) Modulator: Its Therapeutic Implications. Mar. Drugs 2019, 17, 242. [Google Scholar] [CrossRef]
- Lee, C.-H.; Olson, P.; Evans, R.M. Minireview: Lipid metabolism, metabolic diseases, and peroxisome proliferator-activated receptors. Endocrinology 2003, 144, 2201–2207. [Google Scholar] [CrossRef]
- Marx, N.; Duez, H.; Fruchart, J.-C.; Staels, B. Peroxisome proliferator-activated receptors and atherogenesis: Regulators of gene expression in vascular cells. Circ. Res. 2004, 94, 1168–1178. [Google Scholar] [CrossRef]
- Peters, J.M.; Shah, Y.M.; Gonzalez, F.J. The role of peroxisome proliferator-activated receptors in carcinogenesis and chemoprevention. Nat. Rev. Cancer 2012, 12, 181–195. [Google Scholar] [CrossRef]
- Mullen, T.D.; Obeid, L.M. Ceramide and apoptosis: Exploring the enigmatic connections between sphingolipid metabolism and programmed cell death. Anticancer Agents Med. Chem. 2012, 12, 340–363. [Google Scholar] [CrossRef]
- Pelicano, H.; Carney, D.; Huang, P. ROS stress in cancer cells and therapeutic implications. Drug Resist. Updat. Rev. Comment. Antimicrob. Anticancer Chemother. 2004, 7, 97–110. [Google Scholar] [CrossRef]
- Redza-Dutordoir, M.; Averill-Bates, D.A. Activation of apoptosis signalling pathways by reactive oxygen species. Biochim. Biophys. Acta 2016, 1863, 2977–2992. [Google Scholar] [CrossRef] [PubMed]
- Halliwell, B. Free radicals and antioxidants—quo vadis? Trends Pharmacol. Sci. 2011, 32, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Seshacharyulu, P.; Ponnusamy, M.P.; Haridas, D.; Jain, M.; Ganti, A.K.; Batra, S.K. Targeting the EGFR signaling pathway in cancer therapy. Expert Opin. Ther. Targets 2012, 16, 15–31. [Google Scholar] [CrossRef] [PubMed]
- Hsu, T.C.; Young, M.R.; Cmarik, J.; Colburn, N.H. Activator protein 1 (AP-1)- and nuclear factor kappaB (NF-kappaB)-dependent transcriptional events in carcinogenesis. Free Radic. Biol. Med. 2000, 28, 1338–1348. [Google Scholar] [CrossRef]
- Johnson, G.L.; Lapadat, R. Mitogen-activated protein kinase pathways mediated by ERK, JNK, and p38 protein kinases. Science 2002, 298, 1911–1912. [Google Scholar] [CrossRef]
- Sui, X.; Kong, N.; Ye, L.; Han, W.; Zhou, J.; Zhang, Q.; He, C.; Pan, H. p38 and JNK MAPK pathways control the balance of apoptosis and autophagy in response to chemotherapeutic agents. Cancer Lett. 2014, 344, 174–179. [Google Scholar] [CrossRef]
- Cuadrado, A.; Nebreda, A.R. Mechanisms and functions of p38 MAPK signalling. Biochem. J. 2010, 429, 403–417. [Google Scholar] [CrossRef]
- Shakibaei, M.; Schulze-Tanzil, G.; Takada, Y.; Aggarwal, B.B. Redox regulation of apoptosis by members of the TNF superfamily. Antioxid. Redox Signal. 2005, 7, 482–496. [Google Scholar] [CrossRef]
- Bonizzi, G.; Karin, M. The two NF-κB activation pathways and their role in innate and adaptive immunity. Trends Immunol. 2004, 25, 280–288. [Google Scholar] [CrossRef]
- Groner, B.; Hennighausen, L. The versatile regulation of cellular events by Jak-Stat signaling: From transcriptional control to microtubule dynamics and energy metabolism. Horm. Mol. Biol. Clin. Investig. 2012, 10, 193–200. [Google Scholar] [CrossRef]
- Hennighausen, L.; Robinson, G.W. Interpretation of cytokine signaling through the transcription factors STAT5A and STAT5B. Genes Dev. 2008, 22, 711–721. [Google Scholar] [CrossRef] [PubMed]
- Rani, A.; Murphy, J.J. STAT5 in Cancer and Immunity. J. Interferon Cytokine Res. Off. J. Int. Soc. Interferon Cytokine Res. 2016, 36, 226–237. [Google Scholar] [CrossRef] [PubMed]
- Cantrell, D.A. Phosphoinositide 3-kinase signalling pathways. J. Cell Sci. 2001, 114, 1439–1445. [Google Scholar]
- Zhang, J.; Wang, X.; Vikash, V.; Ye, Q.; Wu, D.; Liu, Y.; Dong, W. ROS and ROS-Mediated Cellular Signaling. Available online: https://www.hindawi.com/journals/omcl/2016/4350965/ (accessed on 3 June 2020).
- Vousden, K.H.; Lu, X. Live or let die: The cell’s response to p53. Nat. Rev. Cancer 2002, 2, 594–604. [Google Scholar] [CrossRef] [PubMed]
- El-Deiry, W.S. Regulation of p53 downstream genes. Semin. Cancer Biol. 1998, 8, 345–357. [Google Scholar] [CrossRef]
- Jaiswal, S.; Sharma, P. Role and regulation of p27 in neuronal apoptosis. J. Neurochem. 2017, 140, 576–588. [Google Scholar] [CrossRef]
- Lu, M.; Wang, Y.; Xu, F.; Xiang, J.; Chen, D. The prognostic of p27(kip1) in ovarian cancer: A meta-analysis. Arch. Gynecol. Obstet. 2016, 293, 169–176. [Google Scholar] [CrossRef]
- Zubair, H.; Azim, S.; Ahmad, A.; Khan, M.; Patel, G.; Singh, S.; Singh, A. Cancer Chemoprevention by Phytochemicals: Nature’s Healing Touch. Molecules 2017, 22, 395. [Google Scholar] [CrossRef]
- Liu, R.H. Health benefits of fruit and vegetables are from additive and synergistic combinations of phytochemicals. Am. J. Clin. Nutr. 2003, 78, 517S–520S. [Google Scholar] [CrossRef]
- Liu, R.H. Potential Synergy of Phytochemicals in Cancer Prevention: Mechanism of Action. J. Nutr. 2004, 134, 3479S–3485S. [Google Scholar] [CrossRef]
- Milani, A.; Basirnejad, M.; Shahbazi, S.; Bolhassani, A. Carotenoids: Biochemistry, pharmacology and treatment. Br. J. Pharmacol. 2017, 174, 1290–1324. [Google Scholar] [CrossRef] [PubMed]
- Stahl, W.; Sies, H. Bioactivity and protective effects of natural carotenoids. Biochim. Biophys. Acta 2005, 1740, 101–107. [Google Scholar] [CrossRef] [PubMed]
- Kiokias, S.; Proestos, C.; Varzakas, T. A Review of the Structure, Biosynthesis, Absorption of Carotenoids-Analysis and Properties of their Common Natural Extracts. Curr. Res. Nutr. Food Sci. J. 2016, 4, 25–37. [Google Scholar] [CrossRef]
- Saini, R.K.; Nile, S.H.; Park, S.W. Carotenoids from fruits and vegetables: Chemistry, analysis, occurrence, bioavailability and biological activities. Food Res. Int. 2015, 76, 735–750. [Google Scholar] [CrossRef]
- Fernández-García, E.; Carvajal-Lérida, I.; Jarén-Galán, M.; Garrido-Fernández, J.; Pérez-Gálvez, A.; Hornero-Méndez, D. Carotenoids bioavailability from foods: From plant pigments to efficient biological activities. Food Res. Int. 2012, 46, 438–450. [Google Scholar] [CrossRef]
- Wan-Loy, C.; Siew-Moi, P. Marine Algae as a Potential Source for Anti-Obesity Agents. Mar. Drugs 2016, 14, 222. [Google Scholar] [CrossRef]
- Lu, X.; Sun, H.; Zhao, W.; Cheng, K.-W.; Chen, F.; Liu, B. A Hetero-Photoautotrophic Two-Stage Cultivation Process for Production of Fucoxanthin by the Marine Diatom Nitzschia laevis. Mar. Drugs 2018, 16, 219. [Google Scholar] [CrossRef]
- Shukla, M.; Varalakshmi, K.N. Apoptosis induction in cancer cell lines by the carotenoid Fucoxanthinol from Pseudomonas stutzeri JGI 52. Indian J. Pharmacol. 2018, 50, 116–122. [Google Scholar] [CrossRef]
- Li, Y.; Liu, L.; Sun, P.; Zhang, Y.; Wu, T.; Sun, H.; Cheng, K.-W.; Chen, F. Fucoxanthinol from the Diatom Nitzschia Laevis Ameliorates Neuroinflammatory Responses in Lipopolysaccharide-Stimulated BV-2 Microglia. Mar. Drugs 2020, 18, 116. [Google Scholar] [CrossRef]
- Polcz, M.E.; Barbul, A. The Role of Vitamin A in Wound Healing. Nutr. Clin. Pract. Off. Publ. Am. Soc. Parenter. Enter. Nutr. 2019, 34, 695–700. [Google Scholar] [CrossRef]
- Maoka, T. Carotenoids as natural functional pigments. J. Nat. Med. 2020, 74, 1–16. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, M.E.; Carroll, Y.; Corridan, B.; Olmedilla, B.; Granado, F.; Blanco, I.; Van den Berg, H.; Hininger, I.; Rousell, A.-M.; Chopra, M.; et al. A European carotenoid database to assess carotenoid intakes and its use in a five-country comparative study. Br. J. Nutr. 2001, 85, 499–507. [Google Scholar] [CrossRef] [PubMed]
- Eisenhauer, B.; Natoli, S.; Liew, G.; Flood, V.M. Lutein and Zeaxanthin—Food Sources, Bioavailability and Dietary Variety in Age-Related Macular Degeneration Protection. Nutrients 2017, 9, 120. [Google Scholar] [CrossRef] [PubMed]
- Ishikawa, C.; Tafuku, S.; Kadekaru, T.; Sawada, S.; Tomita, M.; Okudaira, T.; Nakazato, T.; Toda, T.; Uchihara, J.-N.; Taira, N.; et al. Antiadult T-cell leukemia effects of brown algae fucoxanthin and its deacetylated product, fucoxanthinol. Int. J. Cancer 2008, 123, 2702–2712. [Google Scholar] [CrossRef] [PubMed]
- Wu, H.-L.; Fu, X.-Y.; Cao, W.-Q.; Xiang, W.-Z.; Hou, Y.-J.; Ma, J.-K.; Wang, Y.; Fan, C.-D. Induction of Apoptosis in Human Glioma Cells by Fucoxanthin via Triggering of ROS-Mediated Oxidative Damage and Regulation of MAPKs and PI3K-AKT Pathways. J. Agric. Food Chem. 2019, 67, 2212–2219. [Google Scholar] [CrossRef]
- Sowmya Shree, G.; Yogendra Prasad, K.; Arpitha, H.S.; Deepika, U.R.; Nawneet Kumar, K.; Mondal, P.; Ganesan, P. β-carotene at physiologically attainable concentration induces apoptosis and down-regulates cell survival and antioxidant markers in human breast cancer (MCF-7) cells. Mol. Cell. Biochem. 2017, 436, 1–12. [Google Scholar] [CrossRef]
- Pascal, J.M. The comings and goings of PARP-1 in response to DNA damage. DNA Repair 2018, 71, 177–182. [Google Scholar] [CrossRef]
- Zhu, X.; Zhang, Y.; Li, Q.; Yang, L.; Zhang, N.; Ma, S.; Zhang, K.; Song, J.; Guan, F. β-Carotene Induces Apoptosis in Human Esophageal Squamous Cell Carcinoma Cell Lines via the Cav-1/AKT/NF-κB Signaling Pathway: β-CAROTENE AND ESOPHAGEAL CARCINOMA. J. Biochem. Mol. Toxicol. 2016, 30, 148–157. [Google Scholar] [CrossRef]
- Zhang, Y.; Zhu, X.; Huang, T.; Chen, L.; Liu, Y.; Li, Q.; Song, J.; Ma, S.; Zhang, K.; Yang, B.; et al. β-Carotene synergistically enhances the anti-tumor effect of 5-fluorouracil on esophageal squamous cell carcinoma in vivo and in vitro. Toxicol. Lett. 2016, 261, 49–58. [Google Scholar] [CrossRef]
- Vijay, K.; Sowmya, P.R.-R.; Arathi, B.P.; Shilpa, S.; Shwetha, H.J.; Raju, M.; Baskaran, V.; Lakshminarayana, R. Low-dose doxorubicin with carotenoids selectively alters redox status and upregulates oxidative stress-mediated apoptosis in breast cancer cells. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2018, 118, 675–690. [Google Scholar] [CrossRef]
- Naz, H.; Khan, P.; Tarique, M.; Rahman, S.; Meena, A.; Ahamad, S.; Luqman, S.; Islam, A.; Ahmad, F.; Hassan, M.I. Binding studies and biological evaluation of β-carotene as a potential inhibitor of human calcium/calmodulin-dependent protein kinase IV. Int. J. Biol. Macromol. 2017, 96, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Choi, J.; Lim, J.W.; Kim, H. β-Carotene-induced apoptosis is mediated with loss of Ku proteins in gastric cancer AGS cells. Genes Nutr. 2015, 10, 467. [Google Scholar] [CrossRef] [PubMed]
- Akçakaya, H.; Tok, S.; Dal, F.; Cinar, S.A.; Nurten, R. β-carotene treatment alters the cellular death process in oxidative stress-induced K562 cells. Cell Biol. Int. 2017, 41, 309–319. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Li, Y.; Hu, H. Effects of lycopene on ovarian cancer cell line SKOV3 in vitro: Suppressed proliferation and enhanced apoptosis. Mol. Cell. Probes 2019, 46, 101419. [Google Scholar] [CrossRef]
- Poyton, R.O.; Ball, K.A.; Castello, P.R. Mitochondrial generation of free radicals and hypoxic signaling. Trends Endocrinol. Metab. TEM 2009, 20, 332–340. [Google Scholar] [CrossRef]
- Han, H.; Lim, J.W.; Kim, H. Lycopene Inhibits Activation of Epidermal Growth Factor Receptor and Expression of Cyclooxygenase-2 in Gastric Cancer Cells. Nutrients 2019, 11, 2113. [Google Scholar] [CrossRef]
- Dong, G.; Mao, Q.; Xia, W.; Xu, Y.; Wang, J.; Xu, L.; JIANG, F. PKM2 and cancer: The function of PKM2 beyond glycolysis. Oncol. Lett. 2016, 11, 1980–1986. [Google Scholar] [CrossRef]
- Qiu, X.; Yuan, Y.; Vaishnav, A.; Tessel, M.A.; Nonn, L.; van Breemen, R.B. Effects of Lycopene on Protein Expression in Human Primary Prostatic Epithelial Cells. Cancer Prev. Res. 2013, 6, 419–427. [Google Scholar] [CrossRef]
- Wang, R.; Lu, X.; Yu, R. Lycopene Inhibits Epithelial-Mesenchymal Transition and Promotes Apoptosis in Oral Cancer via PI3K/AKT/m-TOR Signal Pathway. Drug Des. Devel. Ther. 2020, 14, 2461–2471. [Google Scholar] [CrossRef]
- Peng, S.J.; Li, J.; Zhou, Y.; Tuo, M.; Qin, X.X.; Yu, Q.; Cheng, H.; Li, Y.M. In vitro effects and mechanisms of lycopene in MCF-7 human breast cancer cells. Genet. Mol. Res. GMR 2017, 16, 13. [Google Scholar] [CrossRef]
- Da Costa Pereira Soares, N.; Machado, C.L.; Trindade, B.B.; do Canto Lima, I.C.; Gimba, E.R.P.; Teodoro, A.J.; Takiya, C.; Borojevic, R. Lycopene Extracts from Different Tomato-Based Food Products Induce Apoptosis in Cultured Human Primary Prostate Cancer Cells and Regulate TP53, Bax and Bcl-2 Transcript Expression. Asian Pac. J. Cancer Prev. APJCP 2017, 18, 339–345. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.-L.; Zhao, Y.-N.; Shi, Z.-Z.; Cong, D.; Bai, Y.-S. Lutein Inhibits Cell Growth and Activates Apoptosis via the PI3K/AKT/mTOR Signaling Pathway in A549 Human Non-Small-Cell Lung Cancer Cells. J. Environ. Pathol. Toxicol. Oncol. Off. Organ. Int. Soc. Environ. Toxicol. Cancer 2018, 37, 341–350. [Google Scholar] [CrossRef]
- Li, Y.; Zhang, Y.; Liu, X.; Wang, M.; Wang, P.; Yang, J.; Zhang, S. Lutein inhibits proliferation, invasion and migration of hypoxic breast cancer cells via downregulation of HES1. Int. J. Oncol. 2018, 52, 2119–2129. [Google Scholar] [CrossRef] [PubMed]
- Kavalappa, Y.P.; Gopal, S.S.; Ponesakki, G. Lutein inhibits breast cancer cell growth by suppressing antioxidant and cell survival signals and induces apoptosis. J. Cell. Physiol. 2020, 1–12. [Google Scholar] [CrossRef] [PubMed]
- San Millán, C.; Soldevilla, B.; Martín, P.; Gil-Calderón, B.; Compte, M.; Pérez-Sacristán, B.; Donoso, E.; Peña, C.; Romero, J.; Granado-Lorencio, F.; et al. β-Cryptoxanthin Synergistically Enhances the Antitumoral Activity of Oxaliplatin through ΔNP73 Negative Regulation in Colon Cancer. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2015, 21, 4398–4409. [Google Scholar] [CrossRef] [PubMed]
- Hormozi, M.; Ghoreishi, S.; Baharvand, P. Astaxanthin induces apoptosis and increases activity of antioxidant enzymes in LS-180 cells. Artif. Cells Nanomedicine Biotechnol. 2019, 47, 891–895. [Google Scholar] [CrossRef] [PubMed]
- Ni, X.; Yu, H.; Wang, S.; Zhang, C.; Shen, S. Astaxanthin Inhibits PC-3 Xenograft Prostate Tumor Growth in Nude Mice. Mar. Drugs 2017, 15, 66. [Google Scholar] [CrossRef]
- Liu, X.; Song, M.; Gao, Z.; Cai, X.; Dixon, W.; Chen, X.; Cao, Y.; Xiao, H. Stereoisomers of Astaxanthin Inhibit Human Colon Cancer Cell Growth by Inducing G2/M Cell Cycle Arrest and Apoptosis. J. Agric. Food Chem. 2016, 64, 7750–7759. [Google Scholar] [CrossRef]
- Sowmya, P.R.-R.; Arathi, B.P.; Vijay, K.; Baskaran, V.; Lakshminarayana, R. Astaxanthin from shrimp efficiently modulates oxidative stress and allied cell death progression in MCF-7 cells treated synergistically with β-carotene and lutein from greens. Food Chem. Toxicol. Int. J. Publ. Br. Ind. Biol. Res. Assoc. 2017, 106, 58–69. [Google Scholar] [CrossRef]
- Shao, Y.; Ni, Y.; Yang, J.; Lin, X.; Li, J.; Zhang, L. Astaxanthin Inhibits Proliferation and Induces Apoptosis and Cell Cycle Arrest of Mice H22 Hepatoma Cells. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2016, 22, 2152–2160. [Google Scholar] [CrossRef]
- Chen, W.; Zhang, H.; Liu, Y. Anti-Inflammatory and Apoptotic Signaling Effect of Fucoxanthin on Benzo(A)Pyrene-Induced Lung Cancer in Mice. J. Environ. Pathol. Toxicol. Oncol. Off. Organ. Int. Soc. Environ. Toxicol. Cancer 2019, 38, 239–251. [Google Scholar] [CrossRef] [PubMed]
- Walczak, H.; Miller, R.E.; Ariail, K.; Gliniak, B.; Griffith, T.S.; Kubin, M.; Chin, W.; Jones, J.; Woodward, A.; Le, T.; et al. Tumoricidal activity of tumor necrosis factor-related apoptosis-inducing ligand in vivo. Nat. Med. 1999, 5, 157–163. [Google Scholar] [CrossRef] [PubMed]
- Jin, Y.; Qiu, S.; Shao, N.; Zheng, J. Fucoxanthin and Tumor Necrosis Factor-Related Apoptosis-Inducing Ligand (TRAIL) Synergistically Promotes Apoptosis of Human Cervical Cancer Cells by Targeting PI3K/Akt/NF-κB Signaling Pathway. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2018, 24, 11–18. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Cheng, J.; Min, Z.; Yin, T.; Zhang, R.; Zhang, W.; Hu, L.; Cui, Z.; Gao, C.; Xu, S.; et al. Effects of fucoxanthin on autophagy and apoptosis in SGC-7901cells and the mechanism. J. Cell. Biochem. 2018, 119, 7274–7284. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zheng, J.; Zhang, Y.; Wang, Z.; Yang, Y.; Bai, M.; Dai, Y. Fucoxanthin Activates Apoptosis via Inhibition of PI3K/Akt/mTOR Pathway and Suppresses Invasion and Migration by Restriction of p38-MMP-2/9 Pathway in Human Glioblastoma Cells. Neurochem. Res. 2016, 41, 2728–2751. [Google Scholar] [CrossRef]
- Yu, R.-X.; Yu, R.-T.; Liu, Z. Inhibition of two gastric cancer cell lines induced by fucoxanthin involves downregulation of Mcl-1 and STAT3. Hum. Cell 2018, 31, 50–63. [Google Scholar] [CrossRef]
- Terasaki, M.; Mutoh, M.; Fujii, G.; Takahashi, M.; Ishigamori, R.; Masuda, S. Potential ability of xanthophylls to prevent obesity-associated cancer. World J. Pharmacol. 2014, 3, 140–152. [Google Scholar] [CrossRef]
- Tamura, S.; Narita, T.; Fujii, G.; Miyamoto, S.; Hamoya, T.; Kurokawa, Y.; Takahashi, M.; Miki, K.; Matsuzawa, Y.; Komiya, M.; et al. Inhibition of NF-kappaB transcriptional activity enhances fucoxanthinol-induced apoptosis in colorectal cancer cells. Genes Environ. 2019, 41. [Google Scholar] [CrossRef]
- Choi, Y.-J.; Hur, J.-M.; Lim, S.; Jo, M.; Kim, D.H.; Choi, J.-I. Induction of apoptosis by deinoxanthin in human cancer cells. Anticancer Res. 2014, 34, 1829–1835. [Google Scholar]
- Abd-Elbaset, M.; Mansour, A.M.; Ahmed, O.M.; Abo-Youssef, A.M. The potential chemotherapeutic effect of β-ionone and/or sorafenib against hepatocellular carcinoma via its antioxidant effect, PPAR-γ, FOXO-1, Ki-67, Bax, and Bcl-2 signaling pathways. Naunyn. Schmiedebergs Arch. Pharmacol. 2020, 393, 1611–1624. [Google Scholar] [CrossRef]
- Yang, X.-W.; Shen, G.-Z.; Cao, L.-Q.; Jiang, X.-F.; Peng, H.-P.; Shen, G.; Chen, D.; Xue, P. MicroRNA-1269 promotes proliferation in human hepatocellular carcinoma via downregulation of FOXO1. BMC Cancer 2014, 14, 909. [Google Scholar] [CrossRef] [PubMed]
- Koeffler, H.P. Peroxisome Proliferator-activated Receptor γ and Cancers. Clin. Cancer Res. 2003, 9, 1–9. [Google Scholar] [PubMed]
- Dong, H.-W.; Wang, K.; Chang, X.-X.; Jin, F.-F.; Wang, Q.; Jiang, X.-F.; Liu, J.-R.; Wu, Y.-H.; Yang, C. Beta-ionone-inhibited proliferation of breast cancer cells by inhibited COX-2 activity. Arch. Toxicol. 2019, 93, 2993–3003. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Qu, Y.; Shen, X.-Y.; Ouyang, T.; Fu, W.-B.; Luo, T.; Wang, H.-Q. Multiple Signal Pathways Involved in Crocetin-Induced Apoptosis in KYSE-150 Cells. Pharmacology 2019, 103, 263–272. [Google Scholar] [CrossRef] [PubMed]
- Ray, P.; Guha, D.; Chakraborty, J.; Banerjee, S.; Adhikary, A.; Chakraborty, S.; Das, T.; Sa, G. Crocetin exploits p53-induced death domain (PIDD) and FAS-associated death domain (FADD) proteins to induce apoptosis in colorectal cancer. Sci. Rep. 2016, 6, 32979. [Google Scholar] [CrossRef] [PubMed]
- Wang, G.; Zhang, B.; Wang, Y.; Han, S.; Wang, C. Crocin promotes apoptosis of human skin cancer cells by inhibiting the JAK/STAT pathway. Exp. Ther. Med. 2018, 16, 5079–5084. [Google Scholar] [CrossRef]
- Deng, L.; Li, J.; Lu, S.; Su, Y. Crocin inhibits proliferation and induces apoptosis through suppressing MYCN expression in retinoblastoma. J. Biochem. Mol. Toxicol. 2019, 33, e22292. [Google Scholar] [CrossRef]
- Mostafavinia, S.E.; Khorashadizadeh, M.; Hoshyar, R. Antiproliferative and Proapoptotic Effects of Crocin Combined with Hyperthermia on Human Breast Cancer Cells. DNA Cell Biol. 2016, 35, 340–347. [Google Scholar] [CrossRef]
- Bakshi, H.A.; Hakkim, F.L.; Sam, S. Molecular Mechanism of Crocin Induced Caspase Mediated MCF-7 Cell Death: In Vivo Toxicity Profiling and Ex Vivo Macrophage Activation. Asian Pac. J. Cancer Prev. APJCP 2016, 17, 1499–1506. [Google Scholar] [CrossRef]
- Vazifedan, V.; Mousavi, S.H.; Sargolzaei, J.; Soleymanifard, S.; Fani Pakdel, A. Study of Crocin & Radiotherapy-induced Cytotoxicity and Apoptosis in the Head and Neck Cancer (HN-5) Cell Line. Iran. J. Pharm. Res. IJPR 2017, 16, 230–237. [Google Scholar]
- Diretto, G.; Ahrazem, O.; Rubio-Moraga, Á.; Fiore, A.; Sevi, F.; Argandoña, J.; Gómez-Gómez, L. UGT709G1: A novel uridine diphosphate glycosyltransferase involved in the biosynthesis of picrocrocin, the precursor of safranal in saffron (Crocus sativus). New Phytol. 2019, 224, 725–740. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Li, J.; Xiao, M. Picrocrocin exhibits growth inhibitory effects against SKMEL- 2 human malignant melanoma cells by targeting JAK/ STAT5 signaling pathway, cell cycle arrest and mitochondrial mediated apoptosis. J. BUON Off. J. Balk. Union Oncol. 2018, 23, 1163–1168. [Google Scholar]
- Rojo de la Vega, M.; Krajisnik, A.; Zhang, D.D.; Wondrak, G.T. Targeting NRF2 for Improved Skin Barrier Function and Photoprotection: Focus on the Achiote-Derived Apocarotenoid Bixin. Nutrients 2017, 9, 1371. [Google Scholar] [CrossRef] [PubMed]
- De Oliveira Júnior, R.G.; Bonnet, A.; Braconnier, E.; Groult, H.; Prunier, G.; Beaugeard, L.; Grougnet, R.; da Silva Almeida, J.R.G.; Ferraz, C.A.A.; Picot, L. Bixin, an apocarotenoid isolated from Bixa orellana L., sensitizes human melanoma cells to dacarbazine-induced apoptosis through ROS-mediated cytotoxicity. Food Chem. Toxicol. 2019, 125, 549–561. [Google Scholar] [CrossRef]
- Kumar, Y.; Phaniendra, A.; Periyasamy, L. Bixin Triggers Apoptosis of Human Hep3B Hepatocellular Carcinoma Cells: An Insight to Molecular and IN SILICO Approach. Nutr. Cancer 2018, 70, 971–983. [Google Scholar] [CrossRef]
- Jiang, L.; Pan, X.; Chen, Y.; Wang, K.; Du, Y.; Zhang, J. Preferential involvement of both ROS and ceramide in fenretinide-induced apoptosis of HL60 rather than NB4 and U937 cells. Biochem. Biophys. Res. Commun. 2011, 405, 314–318. [Google Scholar] [CrossRef]
- Darzynkiewicz, Z.; Bruno, S.; Del Bino, G.; Gorczyca, W.; Hotz, M.A.; Lassota, P.; Traganos, F. Features of apoptotic cells measured by flow cytometry. Cytometry 1992, 13, 795–808. [Google Scholar] [CrossRef]
- Barone, D.; Cito, L.; Tommonaro, G.; Abate, A.A.; Penon, D.; De Prisco, R.; Penon, A.; Forte, I.M.; Benedetti, E.; Cimini, A.; et al. Antitumoral potential, antioxidant activity and carotenoid content of two Southern Italy tomato cultivars extracts: San Marzano and Corbarino. J. Cell. Physiol. 2018, 233, 1266–1277. [Google Scholar] [CrossRef]
- Martínez-Valdivieso, D.; Font, R.; Fernández-Bedmar, Z.; Merinas-Amo, T.; Gómez, P.; Alonso-Moraga, Á.; del Río-Celestino, M. Role of Zucchini and Its Distinctive Components in the Modulation of Degenerative Processes: Genotoxicity, Anti-Genotoxicity, Cytotoxicity and Apoptotic Effects. Nutrients 2017, 9, 755. [Google Scholar] [CrossRef]
- Tran, S.; DeGiovanni, P.-J.; Piel, B.; Rai, P. Cancer nanomedicine: A review of recent success in drug delivery. Clin. Transl. Med. 2017, 6, 44. [Google Scholar] [CrossRef]
- Aghebati-Maleki, A.; Dolati, S.; Ahmadi, M.; Baghbanzhadeh, A.; Asadi, M.; Fotouhi, A.; Yousefi, M.; Aghebati-Maleki, L. Nanoparticles and cancer therapy: Perspectives for application of nanoparticles in the treatment of cancers. J. Cell. Physiol. 2020, 235, 1962–1972. [Google Scholar] [CrossRef] [PubMed]
- Awasthi, R.; Roseblade, A.; Hansbro, P.M.; Rathbone, M.J.; Dua, K.; Bebawy, M. Nanoparticles in Cancer Treatment: Opportunities and Obstacles. Curr. Drug Targets 2018, 19, 1696–1709. [Google Scholar] [CrossRef] [PubMed]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; del Pilar Rodriguez-Torres, M.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, B.; Swamy, M.K.; Sharma, S.; et al. Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.; Sharma, G.; Kushwah, V.; Ghoshal, G.; Jain, A.; Singh, B.; Shivhare, U.S.; Jain, S.; Katare, O.P. Beta carotene-loaded zein nanoparticles to improve the biopharmaceutical attributes and to abolish the toxicity of methotrexate: A preclinical study for breast cancer. Artif. Cells Nanomed. Biotechnol. 2018, 46, 402–412. [Google Scholar] [CrossRef] [PubMed]
- Langroodi, F.A.; Hafezi Ghahestani, Z.; Alibolandi, M.; Ebrahimian, M.; Hashemi, M. Evaluation of the effect of crocetin on antitumor activity of doxorubicin encapsulated in PLGA nanoparticles. Nanomed. J. 2016, 3, 23–34. [Google Scholar] [CrossRef]
- Hafezi Ghahestani, Z.; Alebooye Langroodi, F.; Mokhtarzadeh, A.; Ramezani, M.; Hashemi, M. Evaluation of anti-cancer activity of PLGA nanoparticles containing crocetin. Artif. Cells Nanomed. Biotechnol. 2017, 45, 955–960. [Google Scholar] [CrossRef]
- Mujahid, A.; Dickert, F.L. 5 - Molecularly Imprinted Polymers: Principle, Design, and Enzyme-Like Catalysis. In Molecularly Imprinted Catalysts; Li, S., Cao, S., Piletsky, S.A., Turner, A.P.F., Eds.; Elsevier: Amsterdam, The Netherlands, 2016; pp. 79–101. ISBN 978-0-12-801301-4. [Google Scholar]
- Ravi, H.; Kurrey, N.; Manabe, Y.; Sugawara, T.; Baskaran, V. Polymeric chitosan-glycolipid nanocarriers for an effective delivery of marine carotenoid fucoxanthin for induction of apoptosis in human colon cancer cells (Caco-2 cells). Mater. Sci. Eng. C Mater. Biol. Appl. 2018, 91, 785–795. [Google Scholar] [CrossRef]
- Malgarim Cordenonsi, L.; Faccendini, A.; Catanzaro, M.; Bonferoni, M.C.; Rossi, S.; Malavasi, L.; Platcheck Raffin, R.; Scherman Schapoval, E.E.; Lanni, C.; Sandri, G.; et al. The role of chitosan as coating material for nanostructured lipid carriers for skin delivery of fucoxanthin. Int. J. Pharm. 2019, 567, 118487. [Google Scholar] [CrossRef]
- Huang, R.-F.S.; Wei, Y.-J.; Inbaraj, B.S.; Chen, B.-H. Inhibition of colon cancer cell growth by nanoemulsion carrying gold nanoparticles and lycopene. Int. J. Nanomed. 2015, 10, 2823–2846. [Google Scholar] [CrossRef]
- Jain, A.; Sharma, G.; Kushwah, V.; Thakur, K.; Ghoshal, G.; Singh, B.; Jain, S.; Shivhare, U.S.; Katare, O.P. Fabrication and functional attributes of lipidic nanoconstructs of lycopene: An innovative endeavour for enhanced cytotoxicity in MCF-7 breast cancer cells. Colloids Surf. B Biointerfaces 2017, 152, 482–491. [Google Scholar] [CrossRef]
- Shanmugapriya, K.; Kim, H.; Saravana, P.S.; Chun, B.-S.; Kang, H.W. Astaxanthin-alpha tocopherol nanoemulsion formulation by emulsification methods: Investigation on anticancer, wound healing, and antibacterial effects. Colloids Surf. B Biointerfaces 2018, 172, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Shanmugapriya, K.; Kim, H.; Kang, H.W. In vitro antitumor potential of astaxanthin nanoemulsion against cancer cells via mitochondrial mediated apoptosis. Int. J. Pharm. 2019, 560, 334–346. [Google Scholar] [CrossRef] [PubMed]
- Tian, B.; Li, J.; Pang, R.; Dai, S.; Li, T.; Weng, Y.; Jin, Y.; Hua, Y. Gold Nanoparticles Biosynthesized and Functionalized Using a Hydroxylated Tetraterpenoid Trigger Gene Expression Changes and Apoptosis in Cancer Cells. ACS Appl. Mater. Interfaces 2018, 10, 37353–37363. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-F.; Huang, F.-H.; Zhang, G.-L.; Bai, D.-P.; Massimo, D.F.; Huang, Y.-F.; Gurunathan, S. Novel biomolecule lycopene-reduced graphene oxide-silver nanoparticle enhances apoptotic potential of trichostatin A in human ovarian cancer cells (SKOV3). Int. J. Nanomed. 2017, 12, 7551–7575. [Google Scholar] [CrossRef] [PubMed]
- Hennekens, C.H.; Mayrent, S.L.; Willett, W. Vitamin A, carotenoids, and retinoids. Cancer 1986, 58, 1837–1841. [Google Scholar] [CrossRef]
- Albanes, D.; Virtamo, J.; Taylor, P.R.; Rautalahti, M.; Pietinen, P.; Heinonen, O.P. Effects of supplemental beta-carotene, cigarette smoking, and alcohol consumption on serum carotenoids in the Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study. Am. J. Clin. Nutr. 1997, 66, 366–372. [Google Scholar] [CrossRef]
- Rock, C.L.; Flatt, S.W.; Wright, F.A.; Faerber, S.; Newman, V.; Kealey, S.; Pierce, J.P. Responsiveness of carotenoids to a high vegetable diet intervention designed to prevent breast cancer recurrence. Cancer Epidemiol. Biomark. Prev. Publ. Am. Assoc. Cancer Res. Cosponsored Am. Soc. Prev. Oncol. 1997, 6, 617–623. [Google Scholar]
- Antwi, S.O.; Steck, S.E.; Su, L.J.; Hebert, J.R.; Zhang, H.; Craft, N.E.; Fontham, E.T.H.; Smith, G.J.; Bensen, J.T.; Mohler, J.L.; et al. Carotenoid intake and adipose tissue carotenoid levels in relation to prostate cancer aggressiveness among African-American and European-American men in the North Carolina-Louisiana prostate cancer project (PCaP). Prostate 2016, 76, 1053–1066. [Google Scholar] [CrossRef]
- Hung, R.J.; Zhang, Z.-F.; Rao, J.Y.; Pantuck, A.; Reuter, V.E.; Heber, D.; Lu, Q.-Y. Protective effects of plasma carotenoids on the risk of bladder cancer. J. Urol. 2006, 176, 1192–1197. [Google Scholar] [CrossRef]
- Watters, J.L.; Gail, M.H.; Weinstein, S.J.; Virtamo, J.; Albanes, D. Associations between alpha-tocopherol, beta-carotene, and retinol and prostate cancer survival. Cancer Res. 2009, 69, 3833–3841. [Google Scholar] [CrossRef]
- Nishino, H. Phytochemicals in hepatocellular cancer prevention. Nutr. Cancer 2009, 61, 789–791. [Google Scholar] [CrossRef] [PubMed]
- Antwi, S.O.; Steck, S.E.; Zhang, H.; Stumm, L.; Zhang, J.; Hurley, T.G.; Hebert, J.R. Plasma carotenoids and tocopherols in relation to prostate-specific antigen (PSA) levels among men with biochemical recurrence of prostate cancer. Cancer Epidemiol. 2015, 39, 752–762. [Google Scholar] [CrossRef] [PubMed]
- Thomson, C.A.; Stendell-Hollis, N.R.; Rock, C.L.; Cussler, E.C.; Flatt, S.W.; Pierce, J.P. Plasma and Dietary Carotenoids Are Associated with Reduced Oxidative Stress in Women Previously Treated for Breast Cancer. Cancer Epidemiol. Biomark. Prev. Publ. Am. Assoc. Cancer Res. Cosponsored Am. Soc. Prev. Oncol. 2007, 16, 2008–2015. [Google Scholar] [CrossRef] [PubMed]
- Lai, G.Y.; Weinstein, S.J.; Taylor, P.R.; McGlynn, K.A.; Virtamo, J.; Gail, M.H.; Albanes, D.; Freedman, N.D. Effects of α-tocopherol and β-carotene supplementation on liver cancer incidence and chronic liver disease mortality in the ATBC study. Br. J. Cancer 2014, 111, 2220–2223. [Google Scholar] [CrossRef]
- Malila, N.; Virtamo, J.; Virtanen, M.; Pietinen, P.; Albanes, D.; Teppo, L. Dietary and serum alpha-tocopherol, beta-carotene and retinol, and risk for colorectal cancer in male smokers. Eur. J. Clin. Nutr. 2002, 56, 615–621. [Google Scholar] [CrossRef] [PubMed]
- Dorgan, J.F.; Boakye, N.A.; Fears, T.R.; Schleicher, R.L.; Helsel, W.; Anderson, C.; Robinson, J.; Guin, J.D.; Lessin, S.; Ratnasinghe, L.D.; et al. Serum carotenoids and alpha-tocopherol and risk of nonmelanoma skin cancer. Cancer Epidemiol. Biomark. Prev. Publ. Am. Assoc. Cancer Res. Cosponsored Am. Soc. Prev. Oncol. 2004, 13, 1276–1282. [Google Scholar]
- Schnäbele, K.; Briviba, K.; Bub, A.; Roser, S.; Pool-Zobel, B.L.; Rechkemmer, G. Effects of carrot and tomato juice consumption on faecal markers relevant to colon carcinogenesis in humans. Br. J. Nutr. 2008, 99, 606–613. [Google Scholar] [CrossRef]
- Omenn, G.S.; Goodman, G.E.; Thornquist, M.D.; Balmes, J.; Cullen, M.R.; Glass, A.; Keogh, J.P.; Meyskens, F.L.; Valanis, B.; Williams, J.H.; et al. Effects of a combination of beta carotene and vitamin A on lung cancer and cardiovascular disease. N. Engl. J. Med. 1996, 334, 1150–1155. [Google Scholar] [CrossRef]
- Middha, P.; Weinstein, S.J.; Männistö, S.; Albanes, D.; Mondul, A.M. β-Carotene Supplementation and Lung Cancer Incidence in the Alpha-Tocopherol, Beta-Carotene Cancer Prevention Study: The Role of Tar and Nicotine. Nicotine Tob. Res. Off. J. Soc. Res. Nicotine Tob. 2019, 21, 1045–1050. [Google Scholar] [CrossRef]
- Mondul, A.M.; Watters, J.L.; Männistö, S.; Weinstein, S.J.; Snyder, K.; Virtamo, J.; Albanes, D. Serum retinol and risk of prostate cancer. Am. J. Epidemiol. 2011, 173, 813–821. [Google Scholar] [CrossRef]
- Talvas, J.; Caris-Veyrat, C.; Guy, L.; Rambeau, M.; Lyan, B.; Minet-Quinard, R.; Lobaccaro, J.-M.A.; Vasson, M.-P.; Georgé, S.; Mazur, A.; et al. Differential effects of lycopene consumed in tomato paste and lycopene in the form of a purified extract on target genes of cancer prostatic cells. Am. J. Clin. Nutr. 2010, 91, 1716–1724. [Google Scholar] [CrossRef] [PubMed]
- Bianchi, L.; Orlandi, A.; Campione, E.; Angeloni, C.; Costanzo, A.; Spagnoli, L.G.; Chimenti, S. Topical treatment of basal cell carcinoma with tazarotene: A clinicopathological study on a large series of cases. Br. J. Dermatol. 2004, 151, 148–156. [Google Scholar] [CrossRef] [PubMed]
- Kucuk, O.; Sarkar, F.H.; Sakr, W.; Djuric, Z.; Pollak, M.N.; Khachik, F.; Li, Y.W.; Banerjee, M.; Grignon, D.; Bertram, J.S.; et al. Phase II randomized clinical trial of lycopene supplementation before radical prostatectomy. Cancer Epidemiol. Biomark. Prev. Publ. Am. Assoc. Cancer Res. Cosponsored Am. Soc. Prev. Oncol. 2001, 10, 861–868. [Google Scholar]
- Bolhassani, A. Cancer chemoprevention by natural carotenoids as an efficient strategy. Anticancer Agents Med. Chem. 2015, 15, 1026–1031. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Shnimizu, M.; Moriwaki, H. Cancer Chemoprevention by Caroteno. Molecules 2012, 17, 3202–3242. [Google Scholar] [CrossRef]
- Morifuji, M.; Ichikawa, S.; Kitade, M.; Fukasawa, T.; Asami, Y.; Manabe, Y.; Sugawara, T. Exopolysaccharides from milk fermented by lactic acid bacteria enhance dietary carotenoid bioavailability in humans in a randomized crossover trial and in rats. Am. J. Clin. Nutr. 2020, 111, 903–914. [Google Scholar] [CrossRef]
- Ellis, A.C.; Dudenbostel, T.; Crowe-White, K. Watermelon Juice: A Novel Functional Food to Increase Circulating Lycopene in Older Adult Women. Plant. Foods Hum. Nutr. Dordr. Neth. 2019, 74, 200–203. [Google Scholar] [CrossRef]
- Mapelli-Brahm, P.; Margier, M.; Desmarchelier, C.; Halimi, C.; Nowicki, M.; Borel, P.; Meléndez-Martínez, A.J.; Reboul, E. Comparison of the bioavailability and intestinal absorption sites of phytoene, phytofluene, lycopene and β-carotene. Food Chem. 2019, 300, 125232. [Google Scholar] [CrossRef]
- Zhang, S.; Ji, J.; Zhang, S.; Guan, C.; Wang, G. Effects of three cooking methods on content changes and absorption efficiencies of carotenoids in maize. Food Funct. 2020, 11, 944–954. [Google Scholar] [CrossRef]
- Raber, M.; Basen-Engquist, K.; Moran, N.E.; Chandra, J. The Healthy Cooking Index does not Predict the Carotenoid Content of Home-Cooked Meals. Nutrients 2020, 12, 524. [Google Scholar] [CrossRef]
- Rinaldi de Alvarenga, J.F.; Quifer-Rada, P.; Francetto Juliano, F.; Hurtado-Barroso, S.; Illan, M.; Torrado-Prat, X.; Lamuela-Raventós, R.M. Using Extra Virgin Olive Oil to Cook Vegetables Enhances Polyphenol and Carotenoid Extractability: A Study Applying the sofrito Technique. Molecules 2019, 24, 1555. [Google Scholar] [CrossRef] [PubMed]
- Melini, V.; Panfili, G.; Fratianni, A.; Acquistucci, R. Bioactive compounds in rice on Italian market: Pigmented varieties as a source of carotenoids, total phenolic compounds and anthocyanins, before and after cooking. Food Chem. 2019, 277, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Nie, M.; Zhang, Z.; Liu, C.; Li, D.; Huang, W.; Liu, C.; Jiang, N. Hesperetin and Hesperidin Improved β-Carotene Incorporation Efficiency, Intestinal Cell Uptake, and Retinoid Concentrations in Tissues. J. Agric. Food Chem. 2019, 67, 3363–3371. [Google Scholar] [CrossRef] [PubMed]
- Honda, M.; Kageyama, H.; Hibino, T.; Takemura, R.; Goto, M.; Fukaya, T. Enhanced Z-isomerization of tomato lycopene through the optimal combination of food ingredients. Sci. Rep. 2019, 9, 7979. [Google Scholar] [CrossRef]
- Honda, M.; Nakayama, Y.; Nishikawa, S.; Tsuda, T. Z-Isomers of lycopene exhibit greater liver accumulation than the all-E-isomer in mice. Biosci. Biotechnol. Biochem. 2020, 84, 428–431. [Google Scholar] [CrossRef]
- Watkins, J.L.; Pogson, B.J. Prospects for Carotenoid Biofortification Targeting Retention and Catabolism. Trends Plant. Sci. 2020, 25, 501–512. [Google Scholar] [CrossRef]
- Titcomb, T.J.; Sheftel, J.; Sowa, M.; Gannon, B.M.; Davis, C.R.; Palacios-Rojas, N.; Tanumihardjo, S.A. β-Cryptoxanthin and zeaxanthin are highly bioavailable from whole-grain and refined biofortified orange maize in humans with optimal vitamin A status: A randomized, crossover, placebo-controlled trial. Am. J. Clin. Nutr. 2018, 108, 793–802. [Google Scholar] [CrossRef]
- De Oliveira, C.L.; Brychkova, G.; Esteves-Ferreira, A.A.; McKeown, P.; de Souza Gomes, M.; Maluf, W.R.; Gomes, L.A.A.; Spillane, C. Thermal disruption of the food matrix of biofortified lettuce varieties modifies absorption of carotenoids by Caco-2 cells. Food Chem. 2020, 308, 125443. [Google Scholar] [CrossRef]
- Otani, S.; Miki, S.; Nakamura, Y.; Ishimoto, K.; Ago, Y.; Nakagawa, S. Improved Bioavailability of β-Carotene by Amorphous Solid Dispersion Technology in Rats. J. Nutr. Sci. Vitaminol. 2020, 66, 207–210. [Google Scholar] [CrossRef]
- Ishimoto, K.; Miki, S.; Ohno, A.; Nakamura, Y.; Otani, S.; Nakamura, M.; Nakagawa, S. β-Carotene solid dispersion prepared by hot-melt technology improves its solubility in water. J. Food Sci. Technol. 2019, 56, 3540–3546. [Google Scholar] [CrossRef]
- Berni, P.; Pinheiro, A.C.; Bourbon, A.I.; Guimarães, M.; Canniatti-Brazaca, S.G.; Vicente, A.A. Characterization of the behavior of carotenoids from pitanga (Eugenia uniflora) and buriti (Mauritia flexuosa) during microemulsion production and in a dynamic gastrointestinal system. J. Food Sci. Technol. 2020, 57, 650–662. [Google Scholar] [CrossRef] [PubMed]
- Moran, N.E.; Mohn, E.S.; Hason, N.; Erdman, J.W.; Johnson, E.J. Intrinsic and Extrinsic Factors Impacting Absorption, Metabolism, and Health Effects of Dietary Carotenoids. Adv. Nutr. 2018, 9, 465–492. [Google Scholar] [CrossRef] [PubMed]
- Uramova, S.; Kubatka, P.; Dankova, Z.; Kapinova, A.; Zolakova, B.; Samec, M.; Zubor, P.; Zulli, A.; Valentova, V.; Kwon, T.K.; et al. Plant natural modulators in breast cancer prevention: Status quo and future perspectives reinforced by predictive, preventive, and personalized medical approach. EPMA J. 2018, 9, 403–419. [Google Scholar] [CrossRef] [PubMed]
- Prasad, M.; Takkinen, H.-M.; Uusitalo, L.; Tapanainen, H.; Ovaskainen, M.-L.; Alfthan, G.; Erlund, I.; Ahonen, S.; Åkerlund, M.; Toppari, J.; et al. Carotenoid Intake and Serum Concentration in Young Finnish Children and Their Relation with Fruit and Vegetable Consumption. Nutrients 2018, 10, 1533. [Google Scholar] [CrossRef] [PubMed]
- Eggersdorfer, M.; Wyss, A. Carotenoids in human nutrition and health. Arch. Biochem. Biophys. 2018, 652, 18–26. [Google Scholar] [CrossRef]
- Aune, D.; Keum, N.; Giovannucci, E.; Fadnes, L.T.; Boffetta, P.; Greenwood, D.C.; Tonstad, S.; Vatten, L.J.; Riboli, E.; Norat, T. Dietary intake and blood concentrations of antioxidants and the risk of cardiovascular disease, total cancer, and all-cause mortality: A systematic review and dose-response meta-analysis of prospective studies. Am. J. Clin. Nutr. 2018, 108, 1069–1091. [Google Scholar] [CrossRef]
- Kucera, R.; Pecen, L.; Topolcan, O.; Dahal, A.R.; Costigliola, V.; Frank, A.; Giordano, F.A.; Golubnitschaja, O. Prostate cancer management: Long-term beliefs, epidemic developments in the early 21st century and 3PM dimensional solutions. EPMA J. 2020, 11, 399–418. [Google Scholar] [CrossRef]
- Kunin, A.; Polivka, J.; Moiseeva, N.; Golubnitschaja, O. “Dry mouth” and “Flammer” syndromes—Neglected risks in adolescents and new concepts by predictive, preventive and personalised approach. EPMA J. 2018, 9, 307–317. [Google Scholar] [CrossRef]
- Goncharenko, V.; Bubnov, R.; Polivka, J.; Zubor, P.; Biringer, K.; Bielik, T.; Kuhn, W.; Golubnitschaja, O. Vaginal dryness: Individualised patient profiles, risks and mitigating measures. EPMA J. 2019, 10, 73–79. [Google Scholar] [CrossRef]
- Qian, S.; Golubnitschaja, O.; Zhan, X. Chronic inflammation: Key player and biomarker-set to predict and prevent cancer development and progression based on individualized patient profiles. EPMA J. 2019, 10, 365–381. [Google Scholar] [CrossRef]
- Golubnitschaja, O. (Ed.) Flammer Syndrome: From Phenotype to Associated Pathologies, Prediction, Prevention and Personalisation; Advances in Predictive, Preventive and Personalised Medicine; Springer International Publishing: Cham, Switzerland, 2019; Volume 11, ISBN 978-3-030-13549-2. [Google Scholar]
- Bubnov, R.; Babenko, L.; Lazarenko, L.; Kryvtsova, M.; Shcherbakov, O.; Zholobak, N.; Golubnitschaja, O.; Spivak, M. Can tailored nanoceria act as a prebiotic? Report on improved lipid profile and gut microbiota in obese mice. EPMA J. 2019, 10, 317–335. [Google Scholar] [CrossRef] [PubMed]
- Duarte, A.A.; Mohsin, S.; Golubnitschaja, O. Diabetes care in figures: Current pitfalls and future scenario. EPMA J. 2018, 9, 125–131. [Google Scholar] [CrossRef] [PubMed]
- Avishai, E.; Yeghiazaryan, K.; Golubnitschaja, O. Impaired wound healing: Facts and hypotheses for multi-professional considerations in predictive, preventive and personalised medicine. EPMA J. 2017, 8, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Rossino, M.G.; Casini, G. Nutraceuticals for the Treatment of Diabetic Retinopathy. Nutrients 2019, 11, 771. [Google Scholar] [CrossRef] [PubMed]
- Mashhadi, N.S.; Zakerkish, M.; Mohammadiasl, J.; Zarei, M.; Mohammadshahi, M.; Haghighizadeh, M.H. Astaxanthin improves glucose metabolism and reduces blood pressure in patients with type 2 diabetes mellitus. Asia Pac. J. Clin. Nutr. 2018, 27, 341–346. [Google Scholar] [CrossRef]
- Samaha, M.M.; Said, E.; Salem, H.A. A comparative study of the role of crocin and sitagliptin in attenuation of STZ-induced diabetes mellitus and the associated inflammatory and apoptotic changes in pancreatic β-islets. Environ. Toxicol. Pharmacol. 2019, 72, 103238. [Google Scholar] [CrossRef]
- Baban, B.; Golubnitschaja, O. The potential relationship between Flammer and Sjögren syndromes: The chime of dysfunction. EPMA J. 2017, 8, 333–338. [Google Scholar] [CrossRef]
- Bubnov, R.; Polivka, J.; Zubor, P.; Konieczka, K.; Golubnitschaja, O. “Pre-metastatic niches” in breast cancer: Are they created by or prior to the tumour onset? “Flammer Syndrome” relevance to address the question. EPMA J. 2017, 8, 141–157. [Google Scholar] [CrossRef]
- Golubnitschaja, O.; Polivka, J.; Yeghiazaryan, K.; Berliner, L. Liquid biopsy and multiparametric analysis in management of liver malignancies: New concepts of the patient stratification and prognostic approach. EPMA J. 2018, 9, 271–285. [Google Scholar] [CrossRef]
- Fröhlich, H.; Patjoshi, S.; Yeghiazaryan, K.; Kehrer, C.; Kuhn, W.; Golubnitschaja, O. Premenopausal breast cancer: Potential clinical utility of a multi-omics based machine learning approach for patient stratification. EPMA J. 2018, 9, 175–186. [Google Scholar] [CrossRef]
- Golubnitschaja, O.; Flammer, J. Individualised patient profile: Clinical utility of Flammer syndrome phenotype and general lessons for predictive, preventive and personalised medicine. EPMA J. 2018, 9, 15–20. [Google Scholar] [CrossRef] [PubMed]
- Janssens, J.P.; Schuster, K.; Voss, A. Preventive, predictive, and personalized medicine for effective and affordable cancer care. EPMA J. 2018, 9, 113–123. [Google Scholar] [CrossRef] [PubMed]
- Lu, M.; Zhan, X. The crucial role of multiomic approach in cancer research and clinically relevant outcomes. golubni. EPMA J. 2018, 9, 77–102. [Google Scholar] [CrossRef] [PubMed]
- Golubnitschaja, O.; Baban, B.; Boniolo, G.; Wang, W.; Bubnov, R.; Kapalla, M.; Krapfenbauer, K.; Mozaffari, M.S.; Costigliola, V. Medicine in the early twenty-first century: Paradigm and anticipation—EPMA position paper 2016. EPMA J. 2016, 7, 23. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.A. From Bench to Bedside. J. Urol. 2016, 195, 1643–1644. [Google Scholar] [CrossRef] [PubMed]
- Means, R.T. Iron metabolism: Bedside to bench and back. Am. J. Med. Sci. 2013, 346, 402. [Google Scholar] [CrossRef]

| Carotenoids | Food Source | Reference |
|---|---|---|
| α-Carotene | carrots, coleslaw, pumpkin | [19,111] |
| β-Carotene | apricot, beans, beet, blueberry, broccoli, brussels sprouts, carrots, celery, coleslaw, courgetti, cucumber, lettuce, mango, parsley, peas, pepper, plum, pumpkin, spinach, watermelon, melon, grape, spring greens, watercress | [19,111,118] |
| Lycopene | tomatoes, watermelon | [19,111] |
| Lutein/zeaxanthin | beans, beet, broccoli, brussels sprouts, carrots, celery, coleslaw, courgetti, cucumber, kiwi, leeks, lettuce, parsley, peas, pepper, pumpkin, sweetcorn, olive oil, spinach, watercress, egg, kale, asparagus, pistachio nuts | [19,111,118,119] |
| β-Cryptoxanthin | oranges, pepper, tangerine, papaya | [14,19] |
| Carotenoids Group | Carotenoids | Study Design | Effects on Apoptosis | Reference |
|---|---|---|---|---|
| Carotenes | β-Carotene | MCF-7 human breast cancer cells | ↑ caspase-3, ↓ Bcl-2, ↓ PARP, ↓ NF-κB, ↓ BAD at Ser136, ↓ Akt and ERK1/2 activation | [122] |
| TE1, EC1, Eca109 human esophageal squamous cell carcinoma cells | ↑ caspase-3, ↓ Bcl-2, ↓ p-NF-κB, ↓ p-Akt, ↓ Akt/NF-κB pathways | [124] | ||
| Huh7 hepatoma cells | ↑ percentage of apoptotic cells with an increasement of β-carotene concentration | [127] | ||
| AGS gastric cancer cells | ↑ ROS, ↑ caspase-3, ↓ DNA repair Ku proteins | [128] | ||
| β-Carotene pretreatment | K562 human erythromyeloblastoid leukemia cells | ↑ antioxidant effects against oxidative stress, ↑ protection against the damage of oxidative stress | [129] | |
| β-Carotene, doxorubicin/luteolin, doxorubicin-treated cells | MCF-7 and MDA-MB-231 breast cancer cells | ↑ apoptosis, ↑ prooxidant action | [126] | |
| β-Carotene and 5-fluorouracil | Eca109 human esophageal squamous cell carcinoma cells and Eca109 mice xenografts | ↓ Bcl-2, ↑ caspase-3, ↑ BAX, ↓ Cav-1, ↓ p-Akt, ↓ p-NF-κB, ↓ p-mTOR, ↓ p-p70S6K | [125] | |
| Lycopene | SKOV3 ovarian cancer cells | ↓ Bcl-2, ↑ BAX | [130] | |
| PANC-1 pancreatic cancer cells | ↑ caspase-3, ↑ BAX/Bcl-2 ratio, ↓ intracellular and mitochondrial ROS, ↓ NF-κB, ↓ cIAP1, ↓ cIAP2, ↓ survivin | [31] | ||
| AGS gastric cancer cells | ↑ DNA fragmentation, ↑ caspase-3, -9 cleavage, ↑ BAX/Bcl-2 ratio, ↓ EGFR/Ras/ERK, ↓ p38 MAPK, ↓ DNA-binding activity of NF-κB p50/p50 homodimer ↓ COX-2 expression | [132] | ||
| PrE prostatic epithelial cells | ↑ PKM2, ↑ TyrRS, ↑ RPS3, ↓ CLIC1, ↓ HSP70 1A/1B, ↓ HSP27, ↓ Rho GDI 1, ↓ TCTP, ↓ lactoylglutathione lyase, ↓ Grp78, ↓ KCIP1, ↓ TXNDC17, ↑ TNF-a—induced caspases and apoptosis, ↓ Akt/mTOR, ↓ SFN, ↑ NDGR1, ↑ p53/TP53-mediated caspase and apoptosis | [134] | ||
| Oral cancer cells | ↓ PI3K/Akt/m-TOR signaling, ↑ BAX, ↓ p-PI3K, ↓ p-Akt, ↓ p-m-TOR, ↓ Bcl-2 | [135] | ||
| MCF-7 breast cancer cells | ↑ p53, ↑ BAX | [136] | ||
| Lycopene extracts from different tomato-based products | Human primary prostate cancer cells | ↑ TP53, ↑ BAX, ↓ Bcl-2 | [137] | |
| Xanthophylls | Lutein | MDA-MB-468 and MCF-7 breast cancer cells | ↑ GADD45A, ↑ BAX, ↑ caspase-3, -4, -8, ↑ TNFRSF10A, ↑ TNFRSF21, ↓ CD70, ↓ Bcl-2, ↑ p53 phosphorylation, ↑ HSP60 | [32] |
| A549 lung cancer cells | Regulation of PI3K/Akt signaling pathway | [138] | ||
| MDA-MB-157 and MCF-7 breast cancer cells | ↑ apoptosis | [139] | ||
| MDA-MB-231 and MCF-7 breast cancer cells | ↑ caspase-3, ↓ Bcl-2, ↓ poly-ADP ribose polymerase | [140] | ||
| β-Cryptoxanthin | AGS and SGC-7901 gastric cancer cells; AGS mice xenografts | ↑ caspase-3, -8, and -9, ↑ cytochrome c, ↓ PKA, ↓ pAMPK, ↓ eEF2k, ↑ AMPK signaling inactivation, ↑ G0/G1 cell cycle arrest | [25] | |
| β-Cryptoxanthin + oxaliplatin | HCT116, SW480-ADH, and SW1417 colon cancer cells | ↑ apoptosis via negative regulation of ΔNP73 | [141] | |
| Astaxanthin | LS-180 colorectal cancer cells | ↑ BAX, ↑ caspase-3, ↓ Bcl-2, ↑ antioxidant activity, ↓ malondialdehyde, ↑ superoxide dismutase, ↑ catalase, ↑ glutathione peroxidase | [142] | |
| PC-3 prostate cancer cells mice xenografts | ↑ caspase-3 | [143] | ||
| mice H22 hepatoma cells | ↑ cell cycle arrest, little impact on apoptosis | [146] | ||
| Astaxanthin + β-carotene + lutein | MCF-7 breast cancer cells | ↑ apoptosis through modulation of cyclin D1, p53, BAX, and Bcl-2 | [145] | |
| Stereoisomers of astaxanthin (S, R, S:meso:R 1:2:1) | HCT-116 and HT-29 colon cancer cells | ↑ caspase-3, ↑ PARP, ↑ G2/M cell cycle arrest, ↑ p21Cip1/Waf1, ↑ p27, ↑ p53, ↓ CDK4, ↓ CDK6 | [144] | |
| Fucoxanthin | Chemopreventive mouse model of B(a)P-induced lung cancer | ↑ antioxidant activity, ↑ caspase-9 and -3, ↓ Bcl-2 | [147] | |
| U251 human glioma cells | ↑ ROS-mediated DNA damage, ↑ PARP, ↑ caspase-3, ↑ dysfunction of MAPKs and PI3K-Akt pathways, ↑ phosphorylation of Thr183-JNK, Thr180-p38, and Thr202-ERK, ↓ phosphorylation of Ser473-Akt | [121] | ||
| SGC7901 gastric cancer cells | ↓ viability, ↑ autophagy, ↑ apoptosis, ↑ beclin-1, ↑ LC3, ↑ cleaved caspase-3, ↓ Bcl-2 | [150] | ||
| U87 and U251 human glioma cancer cells | ↓ PI3K/Akt/mTOR pathway | [151] | ||
| SGC-7901 or BGC-823 human gastric cells | ↓ Mcl-1, ↓ STAT3, ↓ p-STAT3 | [152] | ||
| Fucoxanthin + TRAIL | SiHa human cervical cancer cells | ↑ BAX, ↓ Bcl-2, ↓ PI3K/Akt/NF-κB pathway | [149] | |
| Fucoxanthinol | HCT-116 colon cancer cells | ↑ SubG1 population, ↑ NF-κB transcriptional activity | [173] | |
| Yellow pigment fraction (fucoxanthinol) | HeLa cervical cancer cells and HepG2 hepatocellular carcinoma cells | ↑ DNA fragmentation, ↑ caspase-3 | [114] | |
| Deinoxanthin | HepG2 hepatoma, HT-29 colon, and PC-3 prostate cancer cells | ↑ chromatin condensation, ↑ nuclear fragmentation, ↓ Bcl-2, ↑ BAX, ↑ caspase-3, ↑ ROS production | [155] | |
| Apocarotenoids | β-Ionone + sorafenib | Diethylnitrosamine-induced hepatocellular carcinoma of Wistar rats | ↑ BAX, ↓ Bcl-2, ↑ PPAR-γ, ↑ FOXO-1 | [156] |
| β-Ionone | MCF-7 breast cancer cells | ↑ caspase-3, ↑ cytochrome c, ↓ Bcl-2, ↑ BAX, ↑ BAX/Bcl-2 ratio, ↑ p-P38 | [159] | |
| Crocetin | KYSE-150 esophageal cancer cells | ↓ mitochondrial membrane potential, ↑ BAX, ↑ caspase-3, ↓ Bcl-2, ↓ PI3K/Akt activation, ↓ ERK1/2, ↓ p38, ↑ p53/p21 | [160] | |
| p53-expressing and p53-impaired HCT-116 colon cancer cells | p53-expressin HCT-116 cells: ↑ BAX, ↑ PIDD, ↑ BID cleavage, ↑ caspase-2, ↓ mitochondrial membrane potential, ↑ caspase-9 and -3 p53-impaired HCT-116 cells: switch-over from p53 to p73, ↑ FAS, ↑ BID, ↑ FAS-FADD-caspase-8-pathway. | [161] | ||
| Crocin | A431 and SCL-1 human skin cancer cells | ↑ G0/G1 cell cycle arrest, ↑ BID, ↑ procaspase-3, ↓ Bcl-2, ↓ JAK/STAT signaling pathway | [162] | |
| Y79 and WERI-RB-1 retinoblastoma cells | ↑ PARP, ↑ caspase-3, ↓ MYCN but the overexpression of MYCN revert the inhibitory effect of crocin | [163] | ||
| Sensitive OV2008 and chemoresistant C13 cervical cancer cells | ↑ BAX, ↑ p53, ↓ Bcl-2, ↓ miR-365 | [34] | ||
| MCF7 breast cancer cells | ↓ Bcl-2, ↑ BAX, ↓ caspase-8 and -9, ↓ cleaved caspase-3 | [165] | ||
| Crocin + hyperthermia | MDA-MB-468 breast adenocarcinoma cells | ↑ BAX/Bcl-2 ratio, ↓ apoptosis-related HSP70 and HSP90 | [164] | |
| Crocin + radiation | HN-5 head and neck cancer cells | ↑ sensitivity of radiation, ↓ cell viability, ↑ sub-G1 peak, and sensitized cancer cells to radiation-induced toxicity and apoptosis | [166] | |
| Picrocrocin | SK-MEL-2 melanoma cells | ↑ ROS production, ↓ mitochondrial membrane potential, ↓ JAK/STAT5 pathway | [168] | |
| Bixin + dacarbazine | A2058 human melanoma cells | ↑ caspase-3, ↑ ROS production, ↑ DNA damage | [170] | |
| Bixin | Hep3B human hepatocarcinoma cells | ↑ ROS level, ↓ mitochondrial membrane potential, ↑ DNA damage, ↑ BAX, ↑ FasL, ↑ caspase-9, -8 and -3, binding interaction between BAX and Fas ligand | [171] | |
| All-trans retinoic acid | HepG2 hepatocellular carcinoma cells | ↑ AMPK, ↓ intracellular ATP level, ↑ p38 MAPK, ↑ JNK, ↑ BAX, ↑ caspase-3, ↓ GLUT-1, ↓ PKM2, ↓ LDHA | [33] | |
| Fenretinide | HL60 acute promyelocytic leukemia cancer cells | Fenretinide-induced apoptosis: ↑ ROS, ↑ ceramide Fenretinide with fumonisin B1: ↓ apoptosis, reversion of the loss of mitochondrial transmembrane potential | [172] | |
| The whole plant rich in canrotenoids | Lipophilic extracts of tomatoes (San Marzano and Corbarino) | YCC-1, YCC-2 and YCC-3 gastric cancer cells | ↑ G0/G1 cell cycle arrest, ↑ RBL2/p130, ↑ PARP expression, ↑ caspase-9, ↑ p27, ↑ p21 | [174] |
| Zucchini (“Yellow” and “Light Green”) | HL60 human promyelocytic leukemia cells | ↑ DNA fragmentation and apoptosis | [175] |
| Carotenoid | Carriers System | Experimental Model | Effect | Reference |
|---|---|---|---|---|
| β-Carotene | Zein nanoparticles | MCF-7 breast cancer cells | ↑ apoptotic activity | [180] |
| Crocetin | PLGA | MCF-7 breast cancer cells | ↑ caspase-3 | [181] |
| PLGA | MCF-7 breast cancer cells | ↑ apoptosis | [182] | |
| Fucoxanthin | CS + NGs + GL | Caco-2 colorectal cancer cells | ↓ Bcl-2; ↑ BAX; ↑ caspase-3 activity | [184] |
| CS + NLCs | Psoriatic-like cellular model | ↓ Bcl-2 | [185] | |
| Lycopene | Nanoemulsion carrying gold nanoparticles | HT-29 colorectal cancer cells | ↓ procaspase -3, -8, -9; ↓ Bcl-2; ↓ PARP-1 | [186] |
| Solid lipid nanoparticles | MCF-7 breast cancer cells | ↑ greater rate of apoptosis in combination with MTX | [187] | |
| rGO-AgNPs | SKOV3 ovarian cancer ells | ↑ apoptosis | [191] | |
| Astaxanthin | Nanoemulsion | HeLa cervical cancer cells, CT26 colon cancer cells, T24 transitional cell carcinoma cells | ↑ apoptosis, ↓ cell viability | [188] |
| Nanoemulsion | HT-29 colorectal cancer cells, AGS gastric cancer cells | ↑ apoptosis, ↑ ROS generation | [189] | |
| Deinoxanthin | AuNPs | MCF-7 breast cancer cells | Regulation of genes associated with apoptosis and autophagy | [190] |
| Purpose of the Study | Study Details | Results | Reference |
|---|---|---|---|
| The effects of flavanones co-consumption (hesperetin, hesperidin, naringenin, and naringin) on β-carotene | Experimental models: in vitro digestion procedure, synthetic mixed micelles, Caco-2 cell monolayers, and gavage experiments in mice | Hesperetin (25 μM) and hesperidin (25 μM) standards: significant increase in the incorporation efficiency of the β-carotene standard to 68.7 ± 3.6 and 75.2 ± 7.5% (p < 0.05) Naringenin (25 μM) and naringin (25 μM) standards: Significant reduce in the incorporation efficiency of β-carotene by 23.8 and 26.4% (p < 0.05). | [220] |
| β-carotene cellular absorption in the Caco-2 cell model (scavenger receptor class B type I expression increase promoted by citrus flavanones) | |||
| Citrus flavanones (7.5 mg kg-1 day-1) increased the retinoid concentrations in tissues (after 3 days of gavage) Naringenin and naringin significantly decreased hepatic retinoid concentrations (p < 0.05) (after 7 days of gavage) | |||
| Enhanced Z-isomerization of tomato lycopene through food ingredients (Allium sp., Brassica sp., and Raphanus sp., Lentinus edodes, Saccharina sp. and Ecklonia sp.) | Promotion of Z-isomerization of (all-E)-lycopene in tomato puree with heating at 80 °C for 1 h | [221] | |
| Enhanced thermal Z-isomerization of (all-E)-lycopene by polysulfides, isothiocyanates, carbon disulfide, iodine (commonly contained in the above food ingredients) | |||
| Bioavailability and/or liver accumulation of lycopene (Z-isomer) | Mice | Higher bioavailability and/or liver accumulation vs. E-isomer | [222] |
| Bioavailability of β-carotene (through exopolysaccharides from milk fermented by lactic acid bacteria) | Male rats (n = 8/group) administered with β-carotene or β-carotene + fermented milk. Male rats (n = 6/group) retreated with ezetimibe (investigation of β-carotene transport mechanism) | Serum β-carotene AUC significantly higher for the β-carotene + fermented milk vs. β-carotene only | [213] |
| Significant correlation between the exopolysaccharide content of fermented milk and serum β-carotene AUC was observed | |||
| Ezetimibe treatment did not suppress elevations in serum β-carotene concentrations induced by fermented milk ingestion | |||
| Three studies using a randomized crossover method (n = 16/study) consumed a vegetable (carrot, tomato, or spinach) drink alone or with a fermented milk drink (UMIN000034838, UMIN000034839, UMIN000034840) | Significantly higher iAUC for β-carotene in plasma TRL fraction (carrot + fermented milk vs. carrot drink alone) | ||
| Significantly higher iAUC for lycopene in the plasma TRL fraction (tomato + fermented milk vs. tomato drink alone) | |||
| Significant increase in plasma lutein in all fractions after consumption of spinach + fermented milk and not with spinach drink alone |
| Purpose of the Study | Study Details | Results | Reference |
|---|---|---|---|
| Bioavailability of BCX and zeaxanthin from whole-grain and refined BCX-biofortified maize vs. white maize | Randomized, crossover, placebo-controlled trial: 9 adults (mean ± SD age: 23.4 ± 2.3 y; 5 men) were provided with muffins made from BCX-enhanced WGOM, ROM or RWM | Significantly higher BCX AUC for WGOM and ROM vs. RWM | [224] |
| Greater increase in serum BCX from WGOM muffins (131%) than from ROM muffins (108%) | |||
| Higher Zeaxanthin AUCs for WGOM and ROM vs. RWM | |||
| Biofortified lettuce varieties (effects of thermal treatment on carotenoids) | Caco-2 cells | Thermal treatment of lettuce leaves increased carotenoid availability (higher lutein and β-carotene absorption) | [225] |
| Thermal disruption of the food matrix by prior cooking reduced carotenoid levels and transfer to the micellar fraction (absorption of carotenoids from biofortified lettuce remained similar to lettuce cultivars with low carotenoid levels) |
| Purpose of the Study | Study Details | Results | Reference | |
|---|---|---|---|---|
| Solubility of β-carotene | Solid dispersion prepared by hot-melt technology with polyvinylpyrrolidone and sucrose fatty acid esters | Rats | High solubility | [226] |
| Hot-melt technology (solid dispersions technology)—the weight ratio of β-carotene:polyvinylpyrrolidone:sucrose fatty acid ester to 10%:70%:20% | Improved water solubility of β-carotene | [227] | ||
| Bioaccessibility of β-carotene, lycopene | Pitanga (E. uniflora) and buriti (M. flexuosa) microemulsions: direct processing (high-speed homogenization at 15,000 rpm and ultrasound with 20 kHz probe at 40% amplitude) of the whole pulp together with surfactant (Tween 80 or Whey Protein Isolate at 2%) and corn oil (5%) | Dynamic gastrointestinal system (simulation of human digestion) | Surfactant and oil: protection of carotenoids in fruits and microemulsions | [228] |
| Final recovery of total carotenoids, higher for microemulsions than for whole pulps | ||||
| High losses of total carotenoids in buriti and β-carotene and lycopene in pitanga during jejunum and ileum phases |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koklesova, L.; Liskova, A.; Samec, M.; Buhrmann, C.; Samuel, S.M.; Varghese, E.; Ashrafizadeh, M.; Najafi, M.; Shakibaei, M.; Büsselberg, D.; et al. Carotenoids in Cancer Apoptosis—The Road from Bench to Bedside and Back. Cancers 2020, 12, 2425. https://doi.org/10.3390/cancers12092425
Koklesova L, Liskova A, Samec M, Buhrmann C, Samuel SM, Varghese E, Ashrafizadeh M, Najafi M, Shakibaei M, Büsselberg D, et al. Carotenoids in Cancer Apoptosis—The Road from Bench to Bedside and Back. Cancers. 2020; 12(9):2425. https://doi.org/10.3390/cancers12092425
Chicago/Turabian StyleKoklesova, Lenka, Alena Liskova, Marek Samec, Constanze Buhrmann, Samson Mathews Samuel, Elizabeth Varghese, Milad Ashrafizadeh, Masoud Najafi, Mehdi Shakibaei, Dietrich Büsselberg, and et al. 2020. "Carotenoids in Cancer Apoptosis—The Road from Bench to Bedside and Back" Cancers 12, no. 9: 2425. https://doi.org/10.3390/cancers12092425
APA StyleKoklesova, L., Liskova, A., Samec, M., Buhrmann, C., Samuel, S. M., Varghese, E., Ashrafizadeh, M., Najafi, M., Shakibaei, M., Büsselberg, D., Giordano, F. A., Golubnitschaja, O., & Kubatka, P. (2020). Carotenoids in Cancer Apoptosis—The Road from Bench to Bedside and Back. Cancers, 12(9), 2425. https://doi.org/10.3390/cancers12092425








