1. Introduction
The vascular response to hypoxia is an important mechanism that maintains organ function under stress, which is particularly important for vital organs. The cellular response to hypoxia is regulated by the hypoxia-inducible factor-1 (HIF-1) transcription factor. Hypoxia stabilizes the HIF-1 subunit HIF-1α allowing it to activate transcription and mediate an adaptive response. In endothelial cells, HIF-1 regulates the transcription of vascular endothelial growth factor (VEGF), and autocrine VEGF signaling is essential for endothelial cell survival and vascular homeostasis [
1,
2]. VEGF stimulates cellular responses by binding to the cell surface receptors, VEGFR1 and VEGFR2, on the vascular endothelium. VEGFR2 appears to mediate almost all of the known cellular responses to VEGF [
3], whereas VEGFR1 is considered to be inhibitory by acting as a decoy receptor, competing with VEGFR2 for binding to VEGF [
3].
Angiogenesis, the formation of blood vessels, is an adaptive response of tissues to hypoxia. The transcription factor hypoxia-inducible factor-1 (HIF-1) is considered a central regulator of hypoxia. HIF-1α is stabilized under hypoxia, and thereby mediates adaptive responses to hypoxia by activating transcription of numerous angiogenic genes, such as VEGF, and inducing angiogenesis. Likewise, loss of HIF-1α in endothelial cells disrupts a hypoxia-driven VEGF autocrine loop necessary for tumorigenesis [
2]. Endothelial expression of HIF-1 regulates endogenous VEGF expression and autocrine VEGF signaling is essential for endothelial cell survival [
1]. As a result, HIF-1 induces angiogenesis and enhances endothelial survival, in so doing restoring tissue oxygen homeostasis under ischemic conditions.
VEGF is an important signaling protein involved in various activities in vascular biology [
3]. It stimulates cellular responses by binding to cell surface receptors, VEGFR1 and VEGFR2, on vascular endothelium. VEGFR2 appears to mediate almost all of the known cellular responses to VEGF [
3]. The function of VEGFR1 is less well defined, although it is thought to modulate VEGFR2 signaling. The deletion of VEGFR1 in mice results in embryonic lethality due to overgrowth of endothelial cells, leading to disorganization and dysfunction of the vasculature [
3]. VEGF-A, the major factor for angiogenesis, binds to two receptor tyrosine kinases, VEGFR-1 and VEGFR-2, and regulates endothelial cell proliferation, migration, vascular permeability, secretion and other endothelial functions. VEGFR-2 exhibits a strong RTK activity towards pro-angiogenic signals, whereas the soluble VEGFR-1 functions as an endogenous VEGF inhibitor [
4,
5]. However, not much is known about the regulatory mechanisms controlling the differential expression of VEGFR1. Perhaps a net balance in the production of VEGFR1 isoforms determines vessel growth and regression. These findings suggest the possibility that the primary function of VEGFR1 is as a negative regulator of vascular development. The other possible mechanism may be that VEGFR1 acts as a decoy receptor, competing with VEGFR2 for binding to VEGF.
The levels of proteins within cells are determined not only by synthesis, but also by degradation. Many rapidly degraded proteins function as regulatory molecules, such as transcription factors. The rapid turnover of these proteins is necessary to allow their levels to change quickly in response to external stimuli. In eukaryotic cells, two major pathways—the ubiquitin-proteasome pathway and lysosomal proteolysis—mediate protein degradation [
6]. The first pathway of selective protein degradation in eukaryotic cells uses proteasome that targets cytosolic and nuclear proteins for rapid proteolysis [
7]. The other pathway of protein degradation in eukaryotic cells involves the uptake of proteins by lysosomes. Lysosomes are membrane-enclosed organelles that contain digestive enzymes, including several proteases [
8]. They have several roles in cell metabolism, including the digestion of extracellular proteins taken up by endocytosis as well as the gradual turnover of cytoplasmic organelles and cytosolic proteins [
6,
9].
Vav1 (also known as Vav) is a guanine nucleotide exchange factor (GEF) that activates small Rho GTPase. Rho GTPase is well known for its functions in the regulation of cytoskeleton arrangement, cell motility and cell-cell adhesion. The Vav family has three members in vertebrates with Vav1 mostly restricted to hematopoietic cells, being found from the pluripotent stem cells to most mature stages of the lymphoid and myeloid-erythroid lineages [
10,
11]. Vav1 was initially identified as an oncogene capable of transforming NIH3T3 cells, and subsequently, it was found to be an important signal transducer with a pivotal role in hematopoietic cell activation, cell growth and differentiation [
10,
11,
12]. Mice without Vav1 are viable, fertile and grossly normal except with partial blockade in lymphocyte development [
13,
14,
15]. Since hematopoietic cells and endothelial cells share a common progenitor, it is no surprise that Vav1 is also detected in vascular endothelium using Vav1 promoter-driven Cre reporter mice [
16]. However, its function in the vascular system is completely unknown.
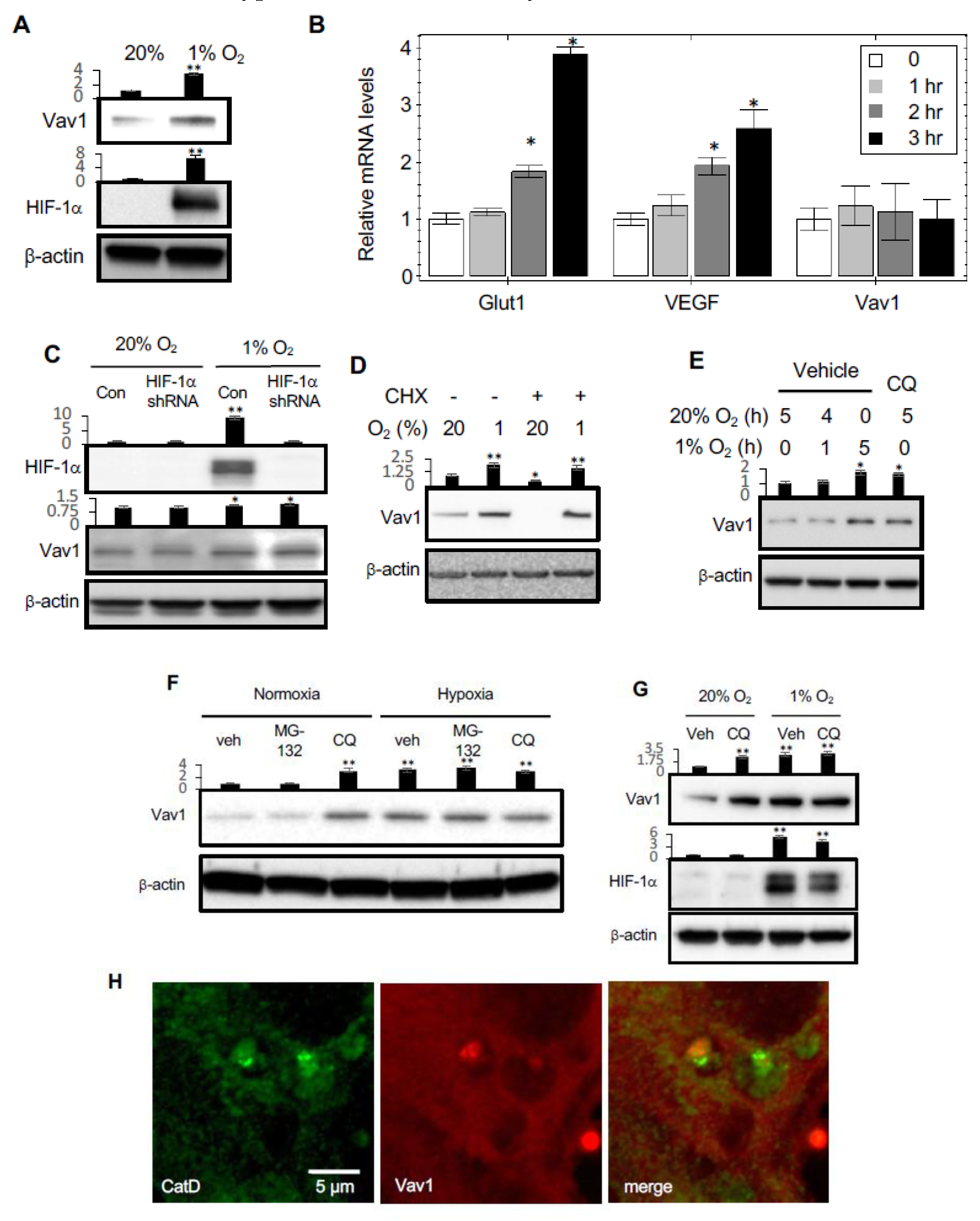
In this study, we hypothesized that Vav1 protein is upregulated in hypoxia, which may contribute to the stress mechanism in hypoxia by HIF-1 regulation. We found the mechanism of how Vav1 is controlled in hypoxia, which may contribute to the understanding of tumor progressions in hypoxia, such as aggressive hypoxic tumors.
3. Materials and Methods
3.1. Experimental Designs
In order to determine which level of Vav1 is controlled, we measured the protein and mRNA level; the protein level was measured when the nascent protein synthesis was blocked by cycloheximide. The pathway of Vav1 proteolysis was determined by chloroquine for lysosomal inhibition and MG-132 for proteasomal inhibition. The location of the Vav1 molecule was determined by confocal microscopy. Following the analysis of the protein sequence of Vav1, the interaction of Vav1 and VEGFR1 was determined by immunoprecipitation as well as the mutant study of the candidate interaction site from VEGFR1. Since we hypothesized that VEGFR1 is the carrier of Vav1 for the lysosomal degradation pathway, we triggered VEGFR1 using PlGF, which led to the degradation of VEGFR1 and Vav1. Since we observed that Vav1 controls HIF1α, we studied the mechanism. Since p38 MAPK is affected by hypoxia and HIF1α, we measured the p32/Siah2/PHD3 mechanism after controlling Vav1 genetically. Along with our previous finding, that lysosomal activity is controlled by hypoxia through the mTORc1/TFEB/V-ATPase pathway [
19], we validated that Vav1 degradation in hypoxia is affected by this pathway. In order to prove this, we measured the level of V-ATPase component molecules in HUVECs incubated in hypoxia.
3.2. Cell Culture and Reagents
HUVECs (Lonza, Walkersville, MD, USA) were cultured with EGM-2 medium (Lonza) and maintained at 37 °C with 5% CO2. Hypoxia was performed by incubating cells in an incubator with 1% O2 (Thermo, Middletown, VA, USA). Human PlGF and VEGF-E were purchased from ProSpec (East Brunswick, NJ, USA). Cycloheximide and chloroquine were purchased from Tocris (Bristol, UK), bafilomycin-A (BafA) was from Sigma (St. Louis, MO, USA). HeLa cells were obtained from ATCC (Manassas, VA, USA) and cultured in 10% FBS (ThermoFisher Scientific, Waltham, MA, USA) containing DMEM (ThermoFisher Scientific).
3.3. Transfection and Lentiviral Transduction
Lentiviral control, Vav1 shRNA, VEGFR1 shRNA, and ATP6v1b2 constructs were obtained from Sigma (St. Louis, MO, USA). Constructs were prepared as lentivirus to transduce knockdown or overexpression. Briefly, lentiviral vectors were co-transfected with VSV.G and envelope vectors into 60% confluent 293T cells in 10-cm culture plates using Fugene HD (Promega, Madison, WI, USA). After three days of incubation, the culture supernatant was collected and concentrated to 500 μL using Lenti-X concentrator (Takara, Kusatsu, Japan). 8 μg/mL of polybrene and 10 μL of the concentrated virus was added to HUVEC and then incubated for 48 h or more for transduction.
3.4. Western Blot and Immunoprecipitation
For hypoxic treatment, HUVECs were incubated under either 20% O2 or 1% O2 for 24 h. The levels of HIF-1α, Siah2, pSiah2, PHD3, p38 and phospho-p38 were analyzed by Western blot using specific antibodies (Cell Signaling, Danvers, MA, USA). SB 203580 (Cell Signaling) at 10 μM was used to inhibit p38 phosphorylation.
For immunoprecipitation, HeLa cells were lysed with lysis buffer (Cell Signaling) and immunoprecipitated with antibodies against Flag (Sigma, St. Louis, MO, USA) or VEGFR1 (Genetex, Irvine, CA, USA) overnight, followed by protein A/G magnetic beads (Pierce, Waltham, MA, USA). The membranes were probed with antibodies against Vav1 (EMD Millipore, Billerica, MA, USA), Flag, HIF-1α (BD Biosciences, San Jose, CA, USA), VEGFR1 (Abcam, Cambridge, UK) and Ubiquitin (Cell Signaling). Western blot images were quantified by using densitometry [
23].
3.5. Immunofluorescent Staining
Cells were stained with 4% paraformaldehyde for 30 min. Cells were probed with 1:200 diluted primary antibodies and 1:500 diluted fluorescent secondary antibodies. Vav1 antibody was obtained from Genetex (Irvine, CA, USA), VEGFR1 and Cathepsin D antibodies were obtained from ThermoFisher (Waltham, MA, USA), Lamp2 antibody was purchased from DHSB (Iowa City, IA, USA). At least ten images were analyzed from each group with a LSM780 confocal microscope (Zeiss, Oberkochen, Germany) and the representative images were selected.
3.6. Real-Time RT-PCR Analysis
Real-time RT-PCR was performed using total RNA isolated on RNeasy Quick spin columns (QIAGEN, CA). One μg of total RNA was used to perform a reverse transcriptase-polymerase chain reaction (RT-PCR) using iScript supermix (Biorad, Hercules, CA, USA). The sequence of PCR primers used are: Vav1, 5′-CAACCTGCGTGAGGTCAAC-3′ and 5′-ACCTTGCCAAAATCCTGCACA-3′; VEGF, 5′-TGTACCTCCACCATGCCAAGT-3′ and, 5′-CGCTGGTAGACGTCCATGAA-3′; PDK1, 5′-ACCAGGACAGCCAATACAAG-3′, and 5′-CCTCGGTCACTCATCTTCAC-3′; Glut1, 5′-ACGCTCTGATCCCTCTCAGT-3′ and 5′-GCAGTACACACCGATGATGAAG-3′; EPO, 5′-ACCAACATTGCTTGTGCCAC-3′ and 5′-TCTGAATGCTTCCTGCTCTGG-3′. Values are expressed as fold increases relative to the reference sample (untreated control) and analyzed with CFX manager (Biorad). All primers were purchased from Sigma.
3.7. Statistical Analysis
All statistical analyses were carried out using Prism 6 (La Jolla, CA, USA). Quantitative variables were analyzed by t-test, one-way ANOVA test. All statistical analysis was two-sided, and p < 0.05 was considered statistically significant.
4. Conclusions
The vascular response to hypoxia is a powerful mechanism to maintain organ function by reducing the negative effects of oxygen deprivation. Thus, the identification of molecular mediators that regulate vascular homeostasis is of great importance. This study identifies Vav1 as a key regulator of the vascular response to hypoxia. We find that Vav1 is continually produced and degraded in normoxic conditions by interaction with VEGFR1 and trafficking to the lysosomes. Hypoxia somehow blocks this degradation mechanism, leading to Vav1 accumulation, which subsequently leads to HIF-1α accumulation.
Vav1 is largely considered to be a hematopoietic-specific protein [
10,
11,
12]. The Vav1 promoter-driven Cre mice are commonly used for specific gene deletion in hematopoietic cells. However, the current study confirms expression of Vav1 in endothelial cells, which is in agreement with a genetic tracing study indicating Vav1 in endothelium [
16], as well as the notion that endothelial cells and blood cells are derived from a common progenitor, and they often share common mediators and pathways. This finding raises a concern regarding specificity when using Vav1-Cre mice for gene deletion, specifically in hematopoietic cells.
Endothelial-specific deletion of HIF-1 disrupts a hypoxia-driven VEGF autocrine loop [
2]. The endogenous production of VEGF in endothelial cells and cell-autonomous activity is crucial for vascular homeostasis. In the absence of autocrine VEGF signaling, endothelial cells undergo apoptosis [
1]. This phenotype is manifested without detectable changes in the total levels of VEGF and cannot be rescued by exogenous VEGF [
1]. Our data demonstrate that Vav1 is essential for HIF-1α accumulation in hypoxia in endothelial cells. Thus, in the absence of Vav1, the endothelium is unable to achieve HIF-1 activation and induction of VEGF, likely leading to a significant increase in endothelial apoptosis under stress.
Hypoxia activates p38 MAPK [
20], and p38 stabilizes HIF-1α [
21]. In T cells, Vav1 acts as a point of integration of signal transduction for receptor-mediated p38 activation [
24]. Consistent with these findings, we show that hypoxia induces p38 phosphorylation and HIF-1α accumulation in endothelial cells, which is dependent on Vav1. Without Vav1, hypoxia fails to activate p38, thereby interrupting the pathway of HIF-1α accumulation. p38 phosphorylates Siah2, which increases Siah2-mediated degradation of PHD3, thus preventing HIF-1α degradation [
25]. Our data suggest that Vav1 is upstream of p38 and is essential for hypoxia-mediated activation of p38. Without Vav1, hypoxia is unable to activate p38, preventing the subsequent activation of Siah2 and PHD3 degradation, necessary for HIF-1α accumulation.
Vav1 is continually produced in endothelial cells and has a high turnover rate due to lysosomal-mediated degradation. These findings reveal that the regulation of Vav1 is analogous to HIF-1α regulation. Both proteins are constitutively produced, and both are continually degraded via lysosomal (Vav1) and proteasomal (HIF-1α) pathways under normoxia. Hypoxia stabilizes Vav1, and Vav1 is essential for HIF-1α accumulation. Together, these two proteins are key mediators of the vascular response to hypoxia.
It has been reported that Vav1 is targeted to lysosomes by interaction with the cytoplasmic chaperone Hsc70 for degradation in pancreatic tumor cells [
26]. In this study, we found that Vav1 is transported to lysosomes by another carrier protein, VEGFR1, in endothelial cells. VEGFR1 is a receptor tyrosine kinase molecule that is known to be degraded by lysosomal proteolysis [
27]. Vav1 binds at the binding motif of Y-K-E-P to VEGFR1, and knockdown of VEGFR1 inhibits Vav1 degradation, and conversely activation of VEGFR1 increases Vav1 degradation. VEGFR1 is known as an inhibitory receptor for VEGF, which promotes angiogenesis. Thus, our findings suggest a potential new mechanism by which VEGFR1 inhibits VEGF-mediated angiogenesis: activation of this receptor induces Vav1 degradation, a GEF protein for small RhoGTPase, and thus plays a negative role in cell motility and angiogenesis.
Hypoxia is a condition in which the body or a region of the body is deprived of adequate oxygen supply at the tissue levels [
28]. It is a common stress associated with various pathological disorders such as cancer and affects many aspects of cellular and molecular activities, as well as therapeutic responses. Hypoxic tumor cells have invasive and migratory behavior [
28]. Furthermore, hypoxic tumor cells are less responsive to chemotherapy and not easy to treat in clinical data. Our recent finding proved that hypoxia downregulates lysosomal activity [
19]. Furthermore, through this study, we report why HIF-1 control is not simply controlled by hypoxia by itself. These findings may provide a molecular explanation for the poor therapeutic targeting of HIF in several clinical trials, and for the observation that hypoxic tumors are often aggressive and resistant to therapy. Hopefully, controlling HIF-1 through Vav1 and lysosomal activity may suggest a new therapeutic approach to hypoxic tumors.
In summary, this study reports a protective role of Vav1 in vascular biology. Vav1 controls HIF-1α stabilization through the p38/Siah2/PHD3 pathway. In normoxic condition, Vav1 binds to VEGFR1, which carries Vav1 and donates to lysosomes for proteolysis. In contrast, hypoxia upregulates Vav1 protein by inhibiting lysosomes, which is analogous to HIF-1α regulation by hypoxia: both proteins are constitutively produced and degraded in normoxia, allowing homeostasis. Consequently, hypoxia rapidly stabilizes Vav1, which is required for HIF-1α accumulation. Our study shows that Vav1 is the key mediator controlling the stabilization of HIF1α in hypoxia. With this finding, we report a novel pathway to stabilize HIF-1, which explains why clinical trials targeting HIF-1 have not been successful in the past. Keeping this finding in mind, targeting Vav1 may be the new approach to overcome hypoxic tumors.