Liquid Biopsy Serial Monitoring of Treatment Responses and Relapse in Advanced Esophageal Squamous Cell Carcinoma
Abstract
1. Introduction
2. Results
2.1. Patient Characteristics
2.2. Role of CTC Enumeration for Prediction and Prognosis of Interim Reassessment, Progression-Free and Overall- Survival
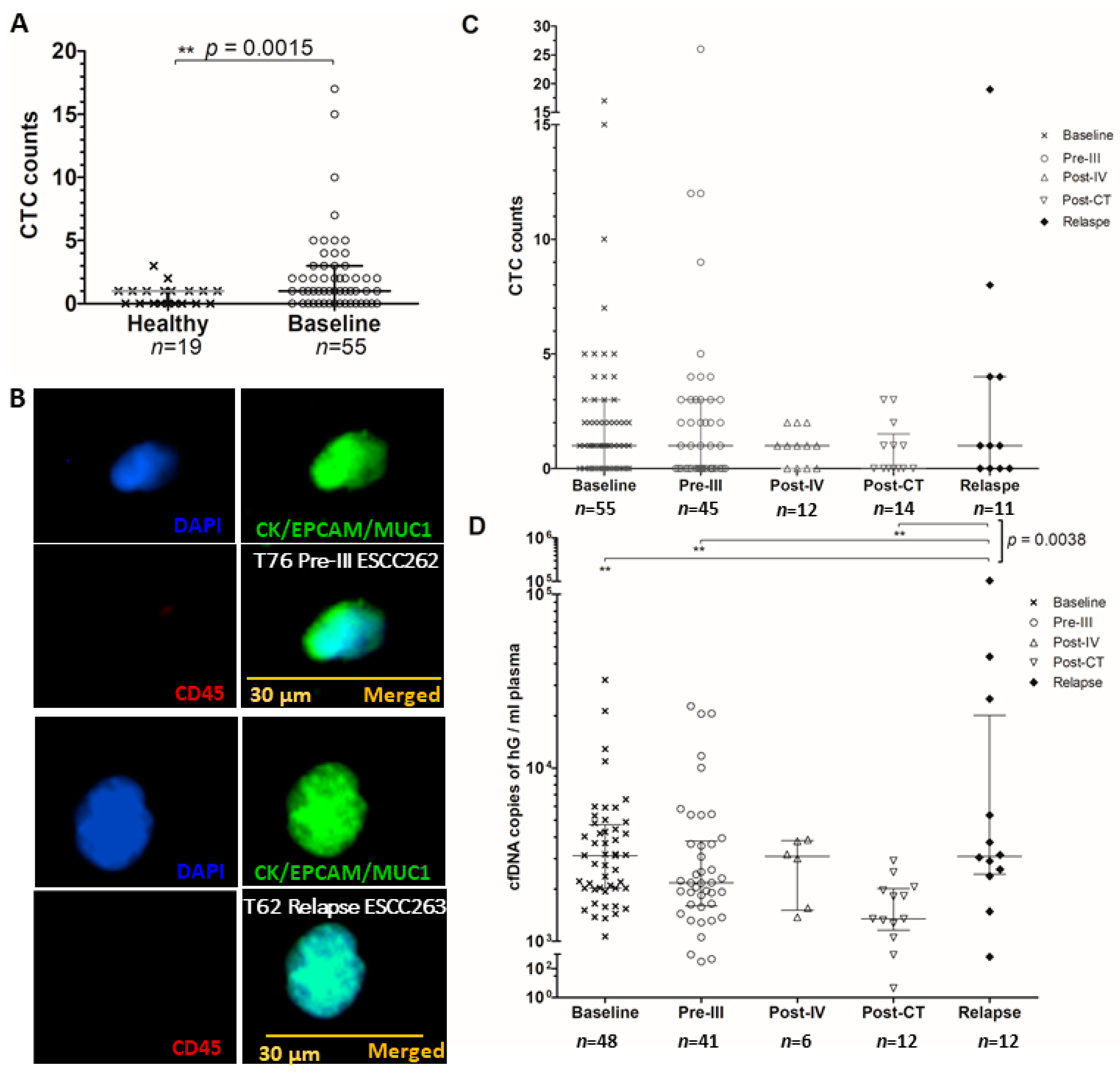
2.2.1. Serial CTC Enumeration
2.2.2. Baseline CTC Enumeration Is Not Associated with Interim Reassessment, Progression-Free and Overall- Survival
2.2.3. CTC Enumeration at Pre-Cycle III Chemotherapy Is Associated with Adverse Outcome
2.3. Role of cfDNA Levels for Prediction and Prognosis of Interim Reassessment, Progression-Free and Overall- Survival
2.3.1. Serial cfDNA Levels
2.3.2. Baseline and CT Pre-Cycle III cfDNA Levels Are Associated with Overall Survival
2.4. Early Prediction of Disease Progression for Patients with Unfavorable Changes of CTC Counts from Baseline to Pre-Cycle III
2.5. Early Prediction for Disease Relapse for Patients with Integration of Unfavorable Changes of CTC Counts and cfDNAs from Baseline to Pre-Cycle III
2.6. COX Regression Analysis of Independent Prognostic Role of CTC and cfDNA Values
3. Discussion
4. Materials and Methods
4.1. Study Population
4.2. CTC Enrichment and Enumeration
4.3. cfDNA Isolation and Quantification
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Survival Rates for Esophageal Cancer. Available online: https://www.cancer.org/cancer/esophagus-cancer/detection-diagnosis-staging/survival-rates.html (accessed on 23 February 2020).
- Hong Kong Cancer Registry 2017. Available online: http://www3.ha.org.hk/cancereg (accessed on 23 February 2020).
- World Health Organizatin (WHO). Available online: http://globocan.iarc.fr/Pages/fact_sheets_cancer.aspx (accessed on 25 March 2020).
- Law, S.; Kwong, D.L.; Kwok, K.F.; Wong, K.H.; Chu, K.M.; Sham, J.S.; Wong, J. Improvement in treatment results and long-term survival of patients with esophageal cancer: Impact of chemoradiation and change in treatment strategy. Ann. Surg. 2003, 238, 339–347. [Google Scholar] [CrossRef] [PubMed]
- Baba, Y.; Watanabe, M.; Yoshida, N.; Baba, H. Neoadjuvant treatment for esophageal squamous cell carcinoma. World J. Gastrointest. Oncol. 2014, 6, 121–128. [Google Scholar] [CrossRef] [PubMed]
- Shapiro, J.; van Lanschot, J.J.B.; Hulshof, M.; van Hagen, P.; van Berge Henegouwen, M.I.; Wijnhoven, B.P.L.; van Laarhoven, H.W.M.; Nieuwenhuijzen, G.A.P.; Hospers, G.A.P.; Bonenkamp, J.J.; et al. Neoadjuvant chemoradiotherapy plus surgery versus surgery alone for oesophageal or junctional cancer (CROSS): Long-term results of a randomised controlled trial. Lancet Oncol. 2015, 16, 1090–1098. [Google Scholar] [CrossRef]
- van Hagen, P.; Hulshof, M.C.; van Lanschot, J.J.; Steyerberg, E.W.; van Berge Henegouwen, M.I.; Wijnhoven, B.P.; Richel, D.J.; Nieuwenhuijzen, G.A.; Hospers, G.A.; Bonenkamp, J.J.; et al. Preoperative chemoradiotherapy for esophageal or junctional cancer. N. Engl. J. Med. 2012, 366, 2074–2084. [Google Scholar] [CrossRef] [PubMed]
- Thiery, J.P.; Lim, C.T. Tumor dissemination: An EMT affair. Cancer Cell 2013, 23, 272–273. [Google Scholar] [CrossRef] [PubMed]
- Haber, D.A.; Velculescu, V.E. Blood-based analyses of cancer: Circulating tumor cells and circulating tumor DNA. Cancer Discov. 2014, 4, 650–661. [Google Scholar] [CrossRef]
- Lorente, D.; Olmos, D.; Mateo, J.; Dolling, D.; Bianchini, D.; Seed, G.; Flohr, P.; Crespo, M.; Figueiredo, I.; Miranda, S.; et al. Circulating tumour cell increase as a biomarker of disease progression in metastatic castration-resistant prostate cancer patients with low baseline CTC counts. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. ESMO 2018, 29, 1554–1560. [Google Scholar] [CrossRef]
- Poveda, A.; Kaye, S.B.; McCormack, R.; Wang, S.; Parekh, T.; Ricci, D.; Lebedinsky, C.A.; Tercero, J.C.; Zintl, P.; Monk, B.J. Circulating tumor cells predict progression free survival and overall survival in patients with relapsed/recurrent advanced ovarian cancer. Gynecol. Oncol. 2011, 122, 567–572. [Google Scholar] [CrossRef]
- Budd, G.T.; Cristofanilli, M.; Ellis, M.J.; Stopeck, A.; Borden, E.; Miller, M.C.; Matera, J.; Repollet, M.; Doyle, G.V.; Terstappen, L.W.; et al. Circulating tumor cells versus imaging--predicting overall survival in metastatic breast cancer. Clin. Cancer Res. Off. J. Am. Assoc. Cancer Res. 2006, 12, 6403–6409. [Google Scholar] [CrossRef]
- Smerage, J.B.; Barlow, W.E.; Hortobagyi, G.N.; Winer, E.P.; Leyland-Jones, B.; Srkalovic, G.; Tejwani, S.; Schott, A.F.; O’Rourke, M.A.; Lew, D.L.; et al. Circulating tumor cells and response to chemotherapy in metastatic breast cancer: SWOG S0500. J. Clin. Oncol. 2014, 32, 3483–3489. [Google Scholar] [CrossRef]
- Carter, L.; Rothwell, D.G.; Mesquita, B.; Smowton, C.; Leong, H.S.; Fernandez-Gutierrez, F.; Li, Y.; Burt, D.J.; Antonello, J.; Morrow, C.J.; et al. Molecular analysis of circulating tumor cells identifies distinct copy-number profiles in patients with chemosensitive and chemorefractory small-cell lung cancer. Nat. Med. 2017, 23, 114–119. [Google Scholar] [CrossRef]
- Alix-Panabieres, C.; Pantel, K. Clinical Applications of Circulating Tumor Cells and Circulating Tumor DNA as Liquid Biopsy. Cancer Discov. 2016. [Google Scholar] [CrossRef]
- Wicha, M.S.; Hayes, D.F. Circulating tumor cells: Not all detected cells are bad and not all bad cells are detected. J. Clin. Oncol. 2011, 29, 1508–1511. [Google Scholar] [CrossRef]
- Grover, P.K.; Cummins, A.G.; Price, T.J.; Roberts-Thomson, I.C.; Hardingham, J.E. Circulating tumour cells: The evolving concept and the inadequacy of their enrichment by EpCAM-based methodology for basic and clinical cancer research. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. ESMO 2014, 25, 1506–1516. [Google Scholar] [CrossRef] [PubMed]
- Agnoletto, C.; Corra, F.; Minotti, L.; Baldassari, F.; Crudele, F.; Cook, W.J.J.; Di Leva, G.; d’Adamo, A.P.; Gasparini, P.; Volinia, S. Heterogeneity in Circulating Tumor Cells: The Relevance of the Stem-Cell Subset. Cancers 2019, 11, 483. [Google Scholar] [CrossRef]
- Matsushita, D.; Uenosono, Y.; Arigami, T.; Yanagita, S.; Nishizono, Y.; Hagihara, T.; Hirata, M.; Haraguchi, N.; Arima, H.; Kijima, Y.; et al. Clinical Significance of Circulating Tumor Cells in Peripheral Blood of Patients with Esophageal Squamous Cell Carcinoma. Ann. Surg. Oncol. 2015, 22, 3674–3680. [Google Scholar] [CrossRef] [PubMed]
- Khoo, B.L.; Warkiani, M.E.; Tan, D.S.; Bhagat, A.A.; Irwin, D.; Lau, D.P.; Lim, A.S.; Lim, K.H.; Krisna, S.S.; Lim, W.T.; et al. Clinical validation of an ultra high-throughput spiral microfluidics for the detection and enrichment of viable circulating tumor cells. PLoS ONE 2014, 9, e99409. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.D.; Yuan, X.; Xue, J.J.; Wang, R.; Zhang, Z.R.; Tong, J.D. Clinical significance of carcinoembryonic antigen-, cytokeratin 19-, or survivin-positive circulating tumor cells in the peripheral blood of esophageal squamous cell carcinoma patients treated with radiotherapy. Dis. Esophagus. 2012, 25, 750–756. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Han, L.; Zhang, W.; Shan, L.; Wang, Y.; Song, P.; Peng, C.; Zhao, X. Preoperative chemotherapy compared with postoperative adjuvant chemotherapy for squamous cell carcinoma of the thoracic oesophagus with the detection of circulating tumour cells randomized controlled trial. Int. J. Surg. 2019, 73, 1–8. [Google Scholar] [CrossRef]
- Hou, H.W.; Warkiani, M.E.; Khoo, B.L.; Li, Z.R.; Soo, R.A.; Tan, D.S.; Lim, W.T.; Han, J.; Bhagat, A.A.; Lim, C.T. Isolation and retrieval of circulating tumor cells using centrifugal forces. Sci. Rep. 2013, 3, 1259. [Google Scholar] [CrossRef]
- Warkiani, M.E.; Khoo, B.L.; Wu, L.; Tay, A.K.; Bhagat, A.A.; Han, J.; Lim, C.T. Ultra-fast, label-free isolation of circulating tumor cells from blood using spiral microfluidics. Nat. Protoc. 2016, 11, 134–148. [Google Scholar] [CrossRef] [PubMed]
- Azad, T.D.; Chaudhuri, A.A.; Fang, P.; Qiao, Y.; Esfahani, M.S.; Chabon, J.J.; Hamilton, E.G.; Yang, Y.D.; Lovejoy, A.; Newman, A.M.; et al. Circulating Tumor DNA Analysis for Detection of Minimal Residual Disease After Chemoradiotherapy for Localized Esophageal Cancer. Gastroenterology 2020, 158, 494–505. [Google Scholar] [CrossRef] [PubMed]
- Khan, K.H.; Cunningham, D.; Werner, B.; Vlachogiannis, G.; Spiteri, I.; Heide, T.; Mateos, J.F.; Vatsiou, A.; Lampis, A.; Damavandi, M.D.; et al. Longitudinal Liquid Biopsy and Mathematical Modeling of Clonal Evolution Forecast Time to Treatment Failure in the PROSPECT-C Phase II Colorectal Cancer Clinical Trial. Cancer Discov. 2018, 8, 1270–1285. [Google Scholar] [CrossRef] [PubMed]
- Creemers, A.; Krausz, S.; Strijker, M.; van der Wel, M.J.; Soer, E.C.; Reinten, R.J.; Besselink, M.G.; Wilmink, J.W.; van de Vijver, M.J.; van Noesel, C.J.M.; et al. Clinical value of ctDNA in upper-GI cancers: A systematic review and meta-analysis. Biochim. Biophys. Acta Rev. Cancer 2017, 1868, 394–403. [Google Scholar] [CrossRef]
- Ko, J.M.; Vardhanabhuti, V.; Ng, W.T.; Lam, K.O.; Ngan, R.K.; Kwong, D.L.; Lee, V.H.; Lui, Y.H.; Yau, C.C.; Kwan, C.K.; et al. Clinical utility of serial analysis of circulating tumour cells for detection of minimal residual disease of metastatic nasopharyngeal carcinoma. Br. J. Cancer 2020. [Google Scholar] [CrossRef]
- Muro, K.; Lordick, F.; Tsushima, T.; Pentheroudakis, G.; Baba, E.; Lu, Z.; Cho, B.C.; Nor, I.M.; Ng, M.; Chen, L.T.; et al. Pan-Asian adapted ESMO Clinical Practice Guidelines for the management of patients with metastatic oesophageal cancer: A JSMO-ESMO initiative endorsed by CSCO, KSMO, MOS, SSO and TOS. Ann. Oncol. Off. J. Eur. Soc. Med. Oncol. ESMO 2019, 30, 34–43. [Google Scholar] [CrossRef]
- Nakashima, S.; Natsugoe, S.; Matsumoto, M.; Miyazono, F.; Nakajo, A.; Uchikura, K.; Tokuda, K.; Ishigami, S.; Baba, M.; Takao, S.; et al. Clinical significance of circulating tumor cells in blood by molecular detection and tumor markers in esophageal cancer. Surgery 2003, 133, 162–169. [Google Scholar] [CrossRef]
- Qiao, Y.; Li, J.; Shi, C.; Wang, W.; Qu, X.; Xiong, M.; Sun, Y.; Li, D.; Zhao, X.; Zhang, D. Prognostic value of circulating tumor cells in the peripheral blood of patients with esophageal squamous cell carcinoma. OncoTargets Ther. 2017, 10, 1363–1373. [Google Scholar] [CrossRef]
- Hsieh, C.C.; Hsu, H.S.; Chang, S.C.; Chen, Y.J. Circulating Cell-Free DNA Levels Could Predict Oncological Outcomes of Patients Undergoing Esophagectomy for Esophageal Squamous Cell Carcinoma. Int. J. Mol. Sci. 2016, 17, 2131. [Google Scholar] [CrossRef]
- Luo, H.; Li, H.; Hu, Z.; Wu, H.; Liu, C.; Li, Y.; Zhang, X.; Lin, P.; Hou, Q.; Ding, G.; et al. Noninvasive diagnosis and monitoring of mutations by deep sequencing of circulating tumor DNA in esophageal squamous cell carcinoma. Biochem. Biophys. Res. Commun. 2016, 471, 596–602. [Google Scholar] [CrossRef] [PubMed]
- Openshaw, M.R.; Richards, C.J.; Guttery, D.S.; Shaw, J.A.; Thomas, A.L. The genetics of gastroesophageal adenocarcinoma and the use of circulating cell free DNA for disease detection and monitoring. Expert Rev. Mol. Diagn. 2017, 17, 459–470. [Google Scholar] [CrossRef] [PubMed]
- Ueda, M.; Iguchi, T.; Masuda, T.; Nakahara, Y.; Hirata, H.; Uchi, R.; Niida, A.; Momose, K.; Sakimura, S.; Chiba, K.; et al. Somatic mutations in plasma cell-free DNA are diagnostic markers for esophageal squamous cell carcinoma recurrence. Oncotarget 2016, 7, 62280–62291. [Google Scholar] [CrossRef] [PubMed]
- Andolfo, I.; Petrosino, G.; Vecchione, L.; De Antonellis, P.; Capasso, M.; Montanaro, D.; Gemei, M.; Troncone, G.; Iolascon, A.; Orditura, M.; et al. Detection of erbB2 copy number variations in plasma of patients with esophageal carcinoma. BMC Cancer 2011, 11, 126. [Google Scholar] [CrossRef]
- Neumann, M.H.D.; Bender, S.; Krahn, T.; Schlange, T. ctDNA and CTCs in Liquid Biopsy—Current Status and Where We Need to Progress. Comput. Struct. Biotechnol. J. 2018, 16, 190–195. [Google Scholar] [CrossRef]
- Brabletz, T.; Kalluri, R.; Nieto, M.A.; Weinberg, R.A. EMT in cancer. Nat. Rev. Cancer 2018, 18, 128–134. [Google Scholar] [CrossRef]
- Yu, M.; Bardia, A.; Wittner, B.S.; Stott, S.L.; Smas, M.E.; Ting, D.T.; Isakoff, S.J.; Ciciliano, J.C.; Wells, M.N.; Shah, A.M.; et al. Circulating breast tumor cells exhibit dynamic changes in epithelial and mesenchymal composition. Science 2013, 339, 580–584. [Google Scholar] [CrossRef]
- Wong, V.C.; Ko, J.M.; Lam, C.T.; Lung, M.L. Succinct workflows for circulating tumor cells after enrichment: From systematic counting to mutational profiling. PLoS ONE 2017, 12, e0177276. [Google Scholar] [CrossRef]


| Parameters | Patients (n = 57) | CTC1 | CTC2 | cfDNA1 d Median ± SD | cfDNA2 d Median ± SD | ||
|---|---|---|---|---|---|---|---|
| ≥3 | 0–2 | ≥3 | 0–2 | ||||
| Median age (range) | 63 (49–76) | ||||||
| <63 | 28 (49%) | 6 | 22 | 7 | 15 | 3425 ± 2496 | 2311 ± 3231 |
| ≥63 | 29 (51%) | 9 | 18 | 7 | 16 | 2577 ± 6914 | 2171 ± 6482 |
| Sex | |||||||
| Male | 49 (86%) | 13 | 34 | 12 | 27 | 3116 ± 3690 | 2176 ± 4665 |
| Female | 8 (14%) | 2 | 6 | 2 | 4 | 4186 ± 11004 | 2755 ± 8598 |
| G category a | |||||||
| GX | 1 (1.8%) | - | - | - | - | ||
| G2 | 34 (59.6%) | 8 | 24 | 8 | 20 | 2663 ± 3975 | 2176 ± 5316 |
| G3 | 22 (38.6%) | 6 | 16 | 6 | 11 | 3682 ± 7054 | 2065 ± 5616 |
| Tumor location | |||||||
| Upper | 9 (27.3%) | 4 | 4 | 3 | 5 | 2580 ± 1719 | 2447 ± 1326 |
| Middle | 26 (41.8%) | 5 | 21 | 6 | 12 | 3171 ± 2201 | 2167 ± 3283 |
| Lower | 19 (30.9%) | 5 | 13 | 5 | 13 | 2220 ± 8927 | 1939 ± 6789 |
| Unknown | 3 (5.3%) | - | - | - | - | ||
| Stage b | |||||||
| III | 14 (24.6%) | 5 | 9 | 4 | 5 | 2111 ± 6785 | 3598 ± 7188 |
| IV | 42 (73.7%) | 10 | 30 | 10 | 25 | 3137 ± 5113 | 2167 ± 5054 |
| Unknown | 1 (1.8%) | - | - | - | - | ||
| Previous treatment | |||||||
| No | 27 (47.4%) | 7 | 20 | 6 | 12 | 3171 ± 4662 | 3069 ± 6395 |
| Yes | 30 (52.6%) | 8 | 20 | 8 | 19 | 3116 ± 5911 | 1954 ± 4513 |
| Distant Metastasis | |||||||
| No | 15 (26.3%) | 5 | 10 | 4 | 6 | 2111 ± 6785 | 3598 ± 7188 |
| Yes | 42 (73.7%) | 10 | 30 | 10 | 25 | 3137 ± 5113 | 2167 ± 5054 |
| Synchronous metastasis | 15 (35.7%) | 4 | 11 | 3 | 8 | 3679 ± 3077 | 2429 ± 6126 |
| Metachronous metastasis | 27 (64.3%) | 6 | 19 | 7 | 17 | 2954 ± 6018 | 2048 ± 4650 |
| Metastasis to one site | 25 (43.9%) | 8 | 16 | 4 | 14 | 3130 ± 6484 | 1954 ± 6432 |
| Metastasis to ≥2 sites | 17 (29.8%) | 2 | 14 | 6 | 11 | 3143 ± 2142 | 2171 ± 2581 |
| Site of Metastasis | |||||||
| Liver | 12 (21.1%) | 1 | 10 | 3 | 9 | 3130 ± 2515 | 2176 ± 3063 |
| Lung | 13 (22.8%) | 4 | 8 | 5 | 5 | 2791 ± 8211 | 2167 ± 8013 |
| Lymph node | 33 (57.9%) | 6 | 25 | 7 | 22 | 3123 ± 1997 | 2048 ± 2550 |
| Others c | 11 (19.4%) | - | - | - | - | ||
| Variables | Univariate Analysis | Multivariate Analysis b | ||
|---|---|---|---|---|
| HR (95% CI) | p-Value | HR (95% CI) | p-Value | |
| (A) Interim Reassessment (n = 52) | ||||
| Age | 1.003 (0.95–1.06) | 0.906 | - | - |
| Gender (F vs. M ref) | 1.553 (0.52–4.62) | 0.428 | - | - |
| L cat (Others vs. Lower ref) | 0.886 (0.40–1.96) | 0.766 | - | - |
| G cat (G3 vs. G2 ref) | 0.615 (0.29–1.41) | 0.251 | - | - |
| Previous treatment (Yes vs. no ref) | 0.882 (0.40–1.93) | 0.753 | - | - |
| Stage at CTC blood collection (IV vs. III ref) | 0.262 (0.11–0.62) | 0.002 | - | - |
| Primary tumor resected at CTC blood collection (n = 50) (Yes vs. no ref) | 0.548 (0.22–1.34) | 0.201 | - | - |
| Metastasis Meta vs. Syn ref (n = 38) | 1.310 (0.48–3.55) | 0.596 | - | - |
| LN metastasis (Yes vs. no ref) | 0.392 (0.18–0.84) | 0.016 | - | - |
| Lung metastasis (Yes vs. no ref) | 1.333 (0.54–3.32) | 0.537 | - | - |
| Liver metastasis (Yes vs. no ref) | 0.715 (0.24–2.09) | 0.541 | - | - |
| Baseline CTC count (3 vs. 0–2 ref) (n = 50) | 2.072 (0.87–4.95) | 0.101 | - | - |
| Pre-cycle III CTC count (3 vs. 0–2 ref) (n = 42) | 3.426 (1.32–8.87) | 0.011 | 3.426 (1.32–8.87) | 0.011 |
| Baseline log cfDNA * (n = 39) | 4.980 (0.91–27.22) | 0.064 | - | - |
| Pre-cycle III log cfDNA * (n = 35) | 2.241 (0.77–6.53) | 0.139 | - | - |
| (B) Progression-free survival (PFS) (n = 54) | ||||
| Age | 0.956 (0.91–1.00) | 0.062 | - | - |
| Gender (F vs. M ref) | 0.969 (0.45–2.08) | 0.935 | - | - |
| L cat (Others vs. Lower ref) (n = 53) | 1.286 (0.70–2.35) | 0.414 | - | - |
| G cat (G3 vs. G2 ref) (n = 53) | 0.867 (0.48–1.56) | 0.633 | - | - |
| Previous Treatment (Yes vs. no ref) | 0.561 (0.30–1.05) | 0.071 | - | - |
| Stage at CTC blood collection (IV vs. III ref) | 0.430 (0.22–0.85) | 0.016 | - | - |
| Primary tumor resected at CTC blood collection (n = 51) (Yes vs. no ref) | 0.373 (0.19–0.75) | 0.005 | 0.402 (0.19–0.87) | 0.02 |
| Metastasis (Meta vs. Syn ref) (n = 40) | 0.517 (0.25–1.08) | 0.080 | - | - |
| LN metastasis (Yes vs. no ref) | 0.618 (0.35–1.11) | 0.106 | - | - |
| Lung metastasis (Yes vs. no ref)) | 1.167 (0.60–2.25) | 0.646 | - | - |
| Liver metastasis (Yes vs. no ref) | 0.449 (0.21–0.98) | 0.043 | - | - |
| Baseline CTC count (≥3 vs. 0–2 CTCs ref) (n = 52) | 1.349 (0.69–2.63) | 0.380 | - | - |
| Pre-cycle III CTC count (≥3 vs. 0–2 CTCs ref) (n = 43) | 3.680 (1.73–7.81) | 0.001 | 4.014 (1.81–8.88) | 0.001 |
| Baseline log cfDNA * (n = 47) | 1.959 (0.67–5.76) | 0.222 | - | - |
| Pre-cycle III log cfDNA * (n = 40) | 1.681 (0.70–4.06) | 0.249 | - | - |
| (C) Overall survival (OS) (n = 57) | ||||
| Age | 0.940 (0.89–0.99) | 0.027 | 0.932 (0.87–0.99) | 0.032 |
| Gender (F vs. M ref) | 0.886 (0.38–2.04) | 0.776 | - | - |
| L cat (Others vs. Lower ref) (n = 56) | 1.884 (0.89–3.99) | 0.097 | - | - |
| G cat (G3 vs. G2 ref) (n = 53) | 0.821 (0.43–1.57) | 0.552 | - | - |
| Previous Treatment (Yes vs. no ref) | 0.534 (0.28–1.02) | 0.056 | - | - |
| Stage at CTC blood collection (IV vs. III ref) (n = 56) | 0.620 (0.30–1.27) | 0.192 | - | - |
| Primary tumor resected at CTC blood drawn (n = 53) (Yes vs. no ref) | 0.584 (0.29–1.17) | 0.127 | - | - |
| Metastasis (Meta vs. Syn ref) (n = 42) | 0.565 (0.27–1.21) | 0.140 | - | - |
| LN metastasis (Yes vs. no ref) | 0.926 (0.48–1.78) | 0.821 | - | - |
| Lung metastasis (Yes vs. no ref) | 1.002 (0.46–2.20) | 0.997 | - | - |
| Liver metastasis (Yes vs. no ref) | 0.784 (0.37–1.67) | 0.529 | - | - |
| Baseline CTC count (≥3 vs. 0–2 CTCs ref) (n = 55) | 0.973 (0.44–2.14) | 0.946 | - | - |
| Pre-cycle III CTC count (≥3 vs. 0–2 CTCs ref) (n = 45) | 3.576 (1.63–7.84) | 0.001 | - | - |
| Baseline log cfDNA * (n = 48) | 8.338 (2.42–28.7) | 0.001 | - | - |
| Pre-cycle III log cfDNA * (n = 41) | 5.451 (1.74–17.1) | 0.004 | - | - |
| Change of cfDNA1/2 (n = 40) | - | - | ||
| Group 0 (Other combinations, n = 21) | Reference | |||
| Group 1 (High cfDNA1 & cfDNA2, n = 19) | 4.444 (1.85–10.68) | 0.001 | ||
| Change of CTC1/2 (n = 43) | - | - | ||
| Group 0 (Low CTC1 & CTC2, n = 22) | Reference | 0.012 | ||
| Group 1 (Other combinations, n = 16) | 3.103 (1.11–8.65) | 0.089 | ||
| Group 2 (High CTC1 & CTC2, n = 5) | 6.178 (1.87–20.4) | 0.004 | ||
| Combined changes of CTC and cfDNA a | ||||
| (n = 44) | ||||
| Group 0 (n = 12) | Reference | 0.001 | 0.002 | |
| Group 1 (n = 22) | 3.103 (1.11–8.65) | 0.030 | 6.008 (1.27–28.5) | 0.008 |
| Group 2 (n = 8) | 6.178 (1.87–20.4) | 0.030 | 9.520 (1.81–50.0) | 0.008 |
| Group 3 (n = 2) | 53.07 (7.02–401) | 1.2 × 10−4 | 81.958 (7.95–845) | 2.2 × 10−4 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ko, J.M.Y.; Ng, H.Y.; Lam, K.O.; Chiu, K.W.H.; Kwong, D.L.W.; Lo, A.W.I.; Wong, J.C.; Lin, R.C.W.; Fong, H.C.H.; Li, J.Y.K.; et al. Liquid Biopsy Serial Monitoring of Treatment Responses and Relapse in Advanced Esophageal Squamous Cell Carcinoma. Cancers 2020, 12, 1352. https://doi.org/10.3390/cancers12061352
Ko JMY, Ng HY, Lam KO, Chiu KWH, Kwong DLW, Lo AWI, Wong JC, Lin RCW, Fong HCH, Li JYK, et al. Liquid Biopsy Serial Monitoring of Treatment Responses and Relapse in Advanced Esophageal Squamous Cell Carcinoma. Cancers. 2020; 12(6):1352. https://doi.org/10.3390/cancers12061352
Chicago/Turabian StyleKo, Josephine Mun Yee, Hoi Yan Ng, Ka On Lam, Keith Wan Hang Chiu, Dora Lai Wan Kwong, Anthony Wing Ip Lo, Jean Chrysei Wong, Roger Chia Wei Lin, Henry Chun Hung Fong, Jason Ying Ki Li, and et al. 2020. "Liquid Biopsy Serial Monitoring of Treatment Responses and Relapse in Advanced Esophageal Squamous Cell Carcinoma" Cancers 12, no. 6: 1352. https://doi.org/10.3390/cancers12061352
APA StyleKo, J. M. Y., Ng, H. Y., Lam, K. O., Chiu, K. W. H., Kwong, D. L. W., Lo, A. W. I., Wong, J. C., Lin, R. C. W., Fong, H. C. H., Li, J. Y. K., Dai, W., Law, S., & Lung, M. L. (2020). Liquid Biopsy Serial Monitoring of Treatment Responses and Relapse in Advanced Esophageal Squamous Cell Carcinoma. Cancers, 12(6), 1352. https://doi.org/10.3390/cancers12061352





