Systematic Review and Network Meta-Analysis of Immune Checkpoint Inhibitors in Combination with Chemotherapy as a First-Line Therapy for Extensive-Stage Small Cell Carcinoma
Abstract
Simple Summary
Abstract
1. Introduction
2. Results
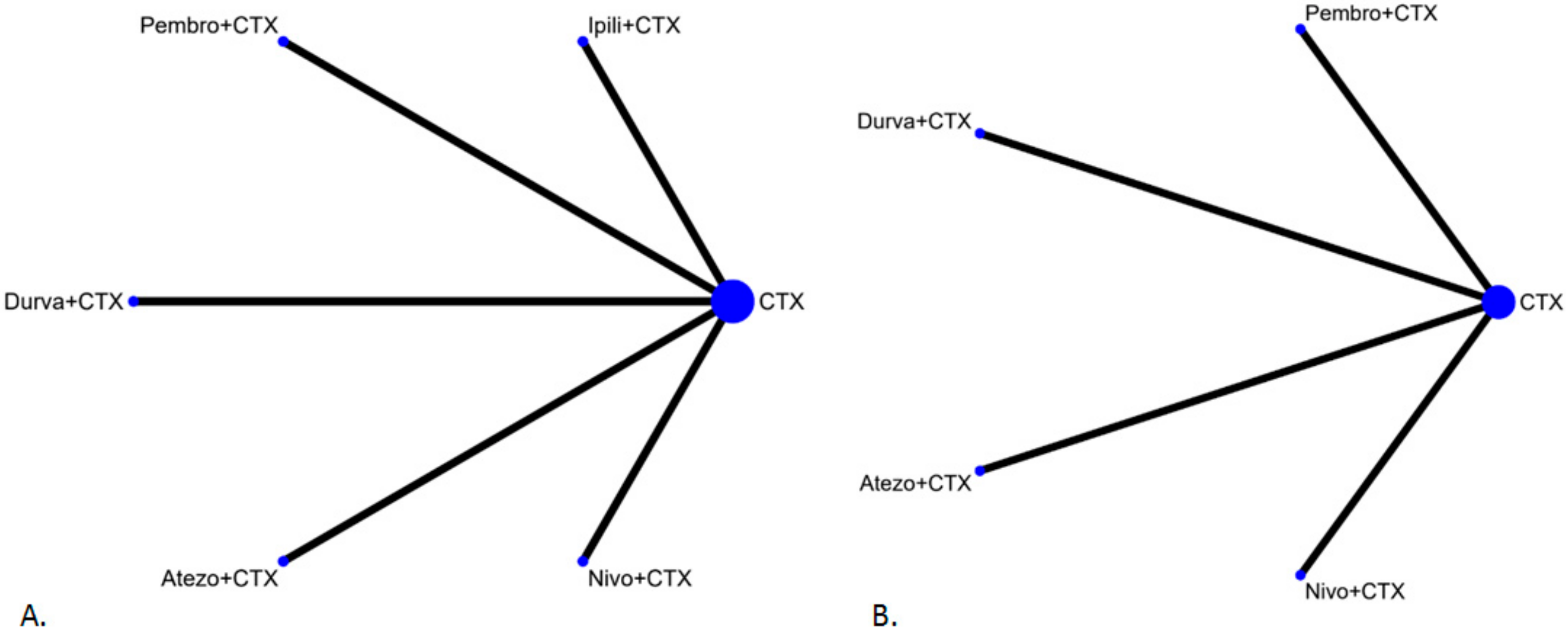
2.1. Literature Search
Study Characteristics and Quality Evaluation
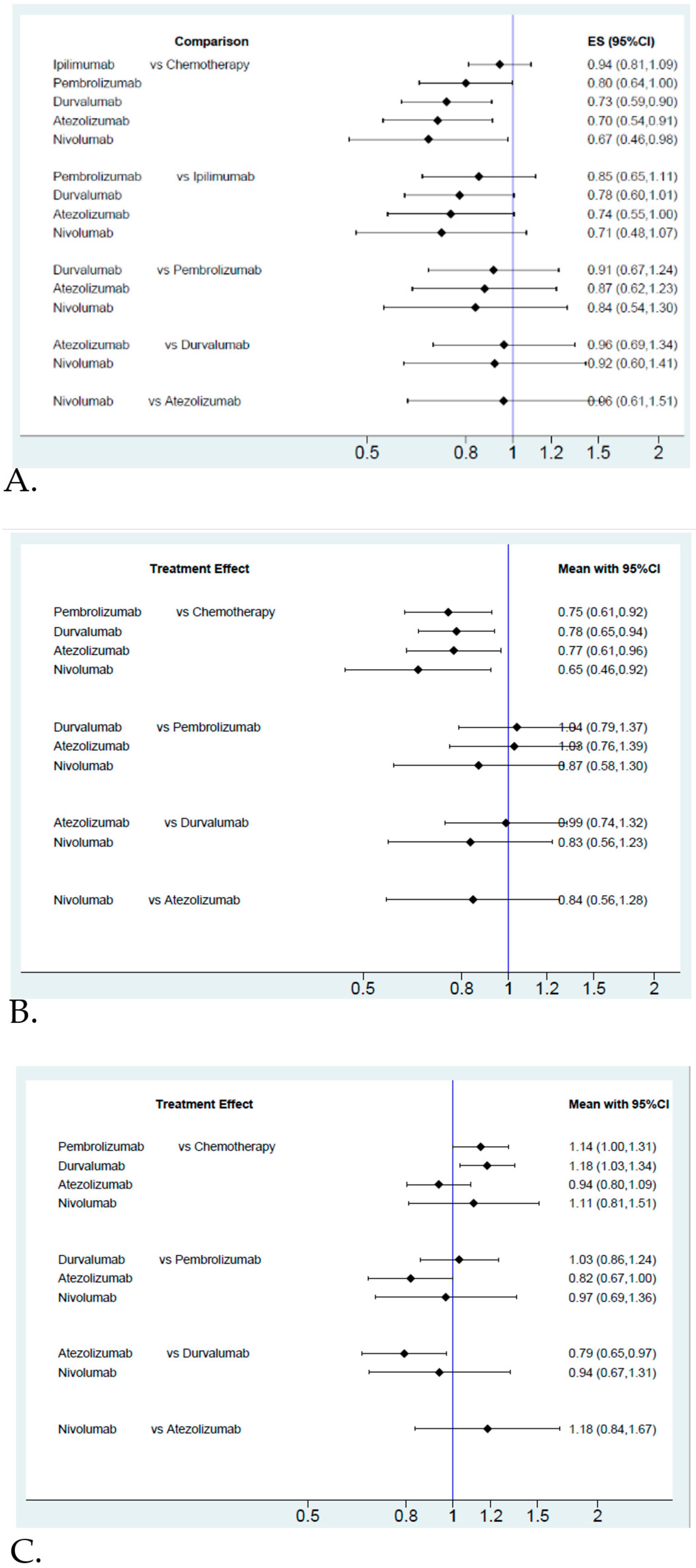
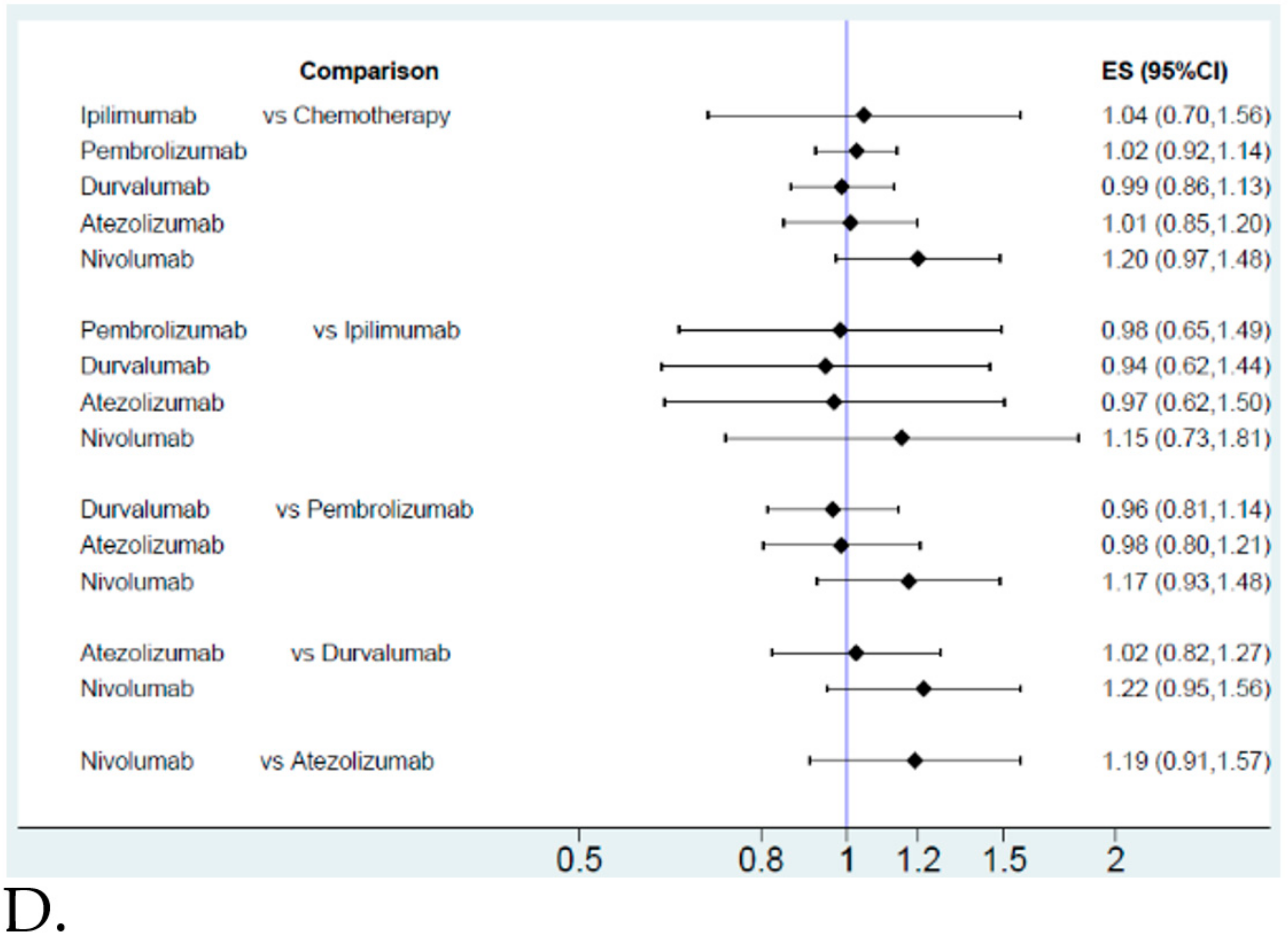
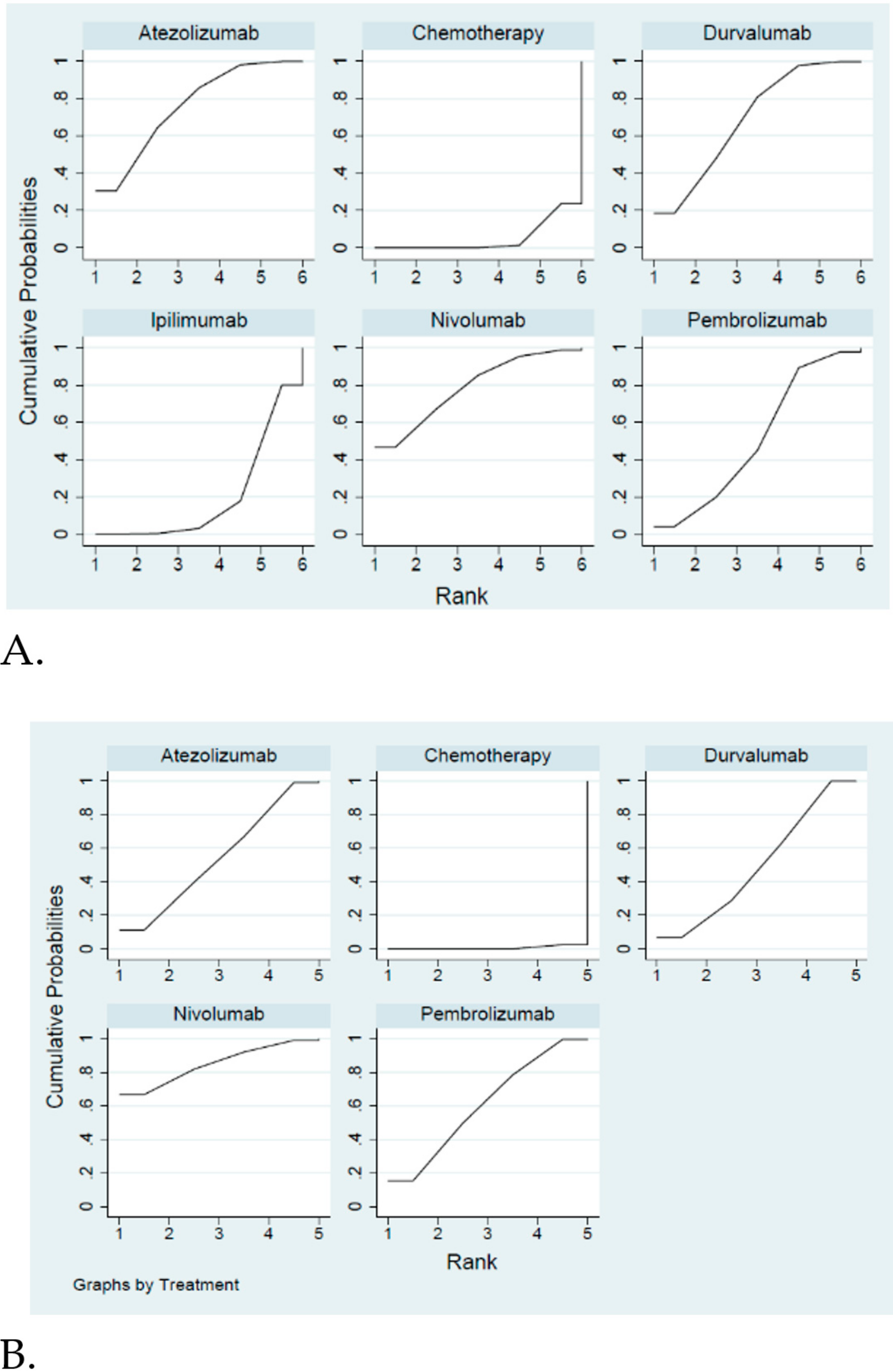
2.2. Pooled Results for ICIs and Their Effect on Efficacy and Safety
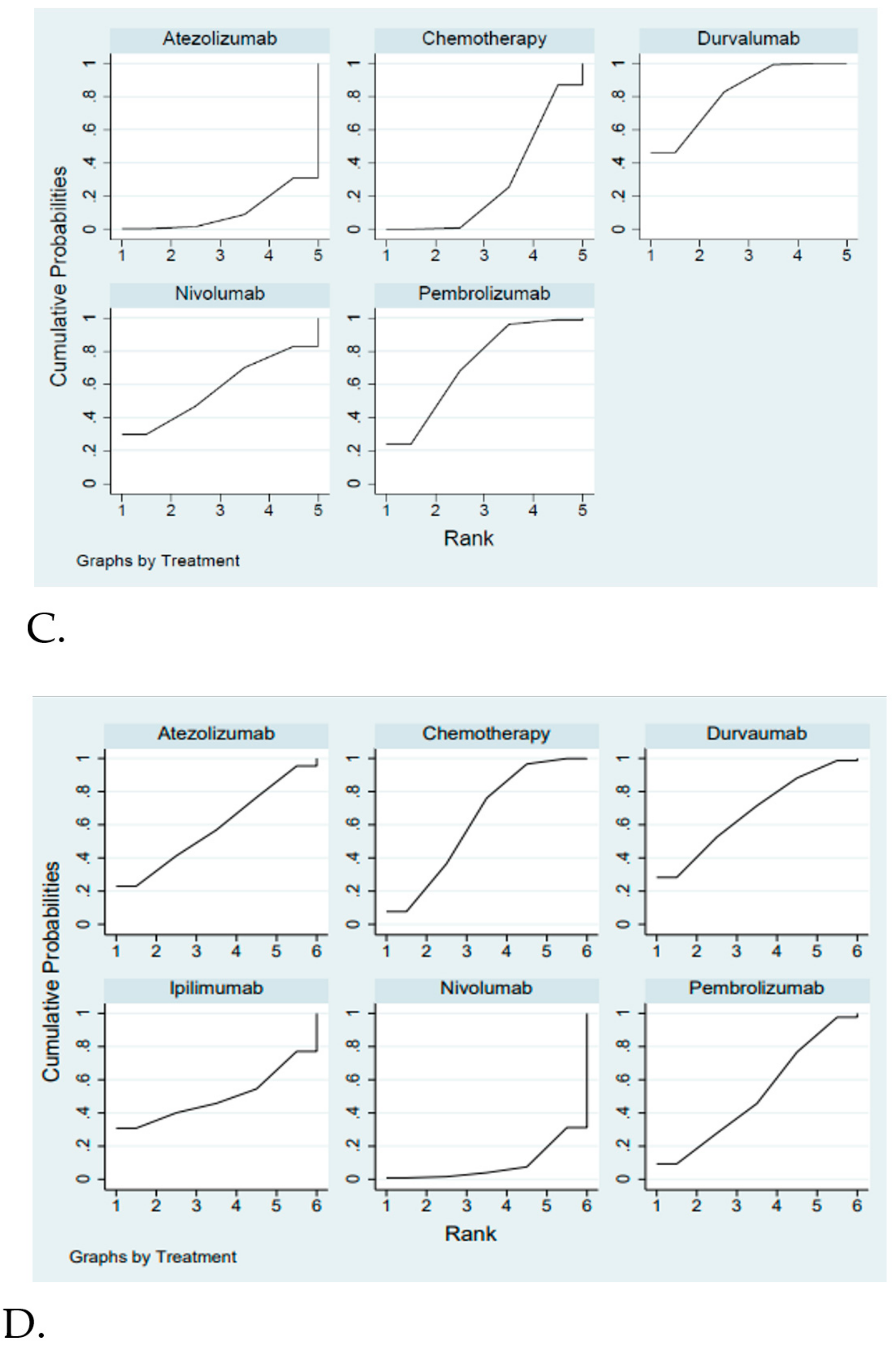
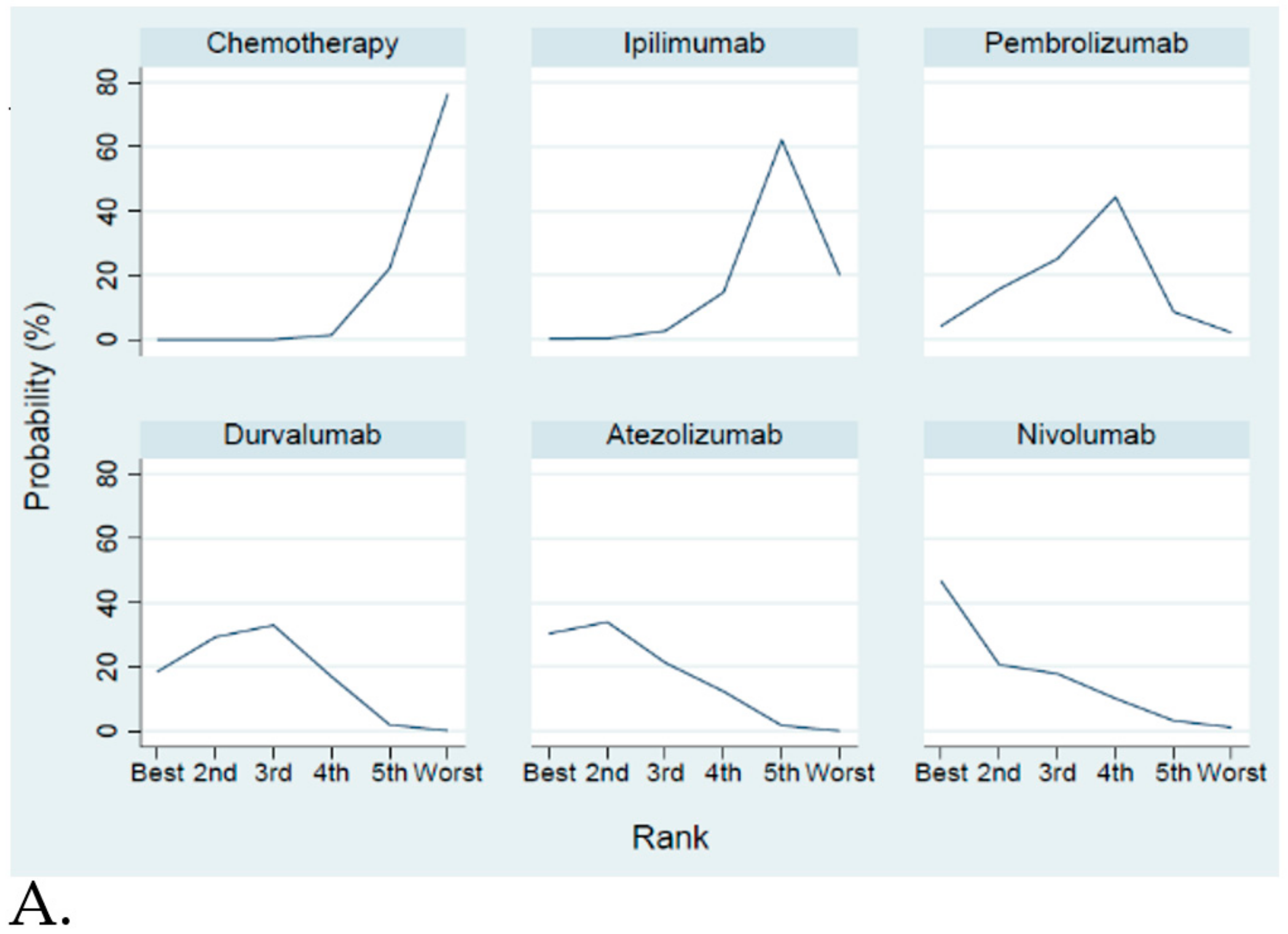
Efficacy and Safety Evaluation from the Network Meta-Analysis
2.3. Efficacy and Safety Evaluation
3. Discussion
4. Methods
4.1. Search Strategy and Study Selection
4.2. Data Extraction and Quality Assessment
4.3. Data Synthesis and Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Sabari, J.K.; Lok, B.H.; Laird, J.H.; Poirier, J.T.; Rudin, C.M. Unravelling the biology of sclc: Implications for therapy. Nat. Rev. Clin. Oncol. 2017, 14, 549–561. [Google Scholar] [CrossRef] [PubMed]
- Subbiah, S.; Nam, A.; Garg, N.; Behal, A.; Kulkarni, P.; Salgia, R. Small cell lung cancer from traditional to innovative therapeutics: Building a comprehensive network to optimize clinical and translational research. J. Clin. Med. 2020, 9, 2433. [Google Scholar] [CrossRef] [PubMed]
- George, J.; Lim, J.S.; Jang, S.J.; Cun, Y.; Ozretic, L.; Kong, G.; Leenders, F.; Lu, X.; Fernandez-Cuesta, L.; Bosco, G.; et al. Comprehensive genomic profiles of small cell lung cancer. Nature 2015, 524, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Peifer, M.; Fernandez-Cuesta, L.; Sos, M.L.; George, J.; Seidel, D.; Kasper, L.H.; Plenker, D.; Leenders, F.; Sun, R.; Zander, T.; et al. Integrative genome analyses identify key somatic driver mutations of small-cell lung cancer. Nat. Genet. 2012, 44, 1104–1110. [Google Scholar] [CrossRef]
- Dong, Z.; Bing, X.; Yong, Y.; Peng, Y.; Sheng-Nan, L.; Qiang, L. Progress in immunotherapy for small cell lung cancer. World J. Clin. Oncol. 2020, 11, 370–377. [Google Scholar]
- Oze, I.; Hotta, K.; Kiura, K.; Ochi, N.; Takigawa, N.; Fujiwara, Y.; Tabata, M.; Tanimoto, M. Twenty-seven years of phase iii trials for patients with extensive disease small-cell lung cancer: Disappointing results. PLoS ONE 2009, 4, e7835. [Google Scholar] [CrossRef]
- Rudin, C.M.; Ismaila, N.; Hann, C.L.; Malhotra, N.; Movsas, B.; Norris, K.; Pietanza, M.C.; Ramalingam, S.S.; Turrisi, A.T., 3rd; Giaccone, G. Treatment of small-cell lung cancer: American society of clinical oncology endorsement of the american college of chest physicians guideline. J. Clin. Oncol. 2015, 33, 4106–4111. [Google Scholar] [CrossRef]
- Nicholson, A.G.; Chansky, K.; Crowley, J.; Beyruti, R.; Kubota, K.; Turrisi, A.; Eberhardt, W.E.; van Meerbeeck, J.; Rami-Porta, R.; Goldstraw, P.; et al. The international association for the study of lung cancer lung cancer staging project: Proposals for the revision of the clinical and pathologic staging of small cell lung cancer in the forthcoming eighth edition of the tnm classification for lung cancer. J. Thorac. Oncol. 2016, 11, 300–311. [Google Scholar] [CrossRef]
- Almutairi, A.R.; Alkhatib, N.; Martin, J.; Babiker, H.M.; Garland, L.L.; McBride, A.; Abraham, I. Comparative efficacy and safety of immunotherapies targeting the pd-1/pd-l1 pathway for previously treated advanced non-small cell lung cancer: A bayesian network meta-analysis. Crit. Rev. Oncol. Hematol. 2019, 142, 16–25. [Google Scholar] [CrossRef]
- Ando, K.; Kishino, Y.; Homma, T.; Kusumoto, S.; Yamaoka, T.; Tanaka, A.; Ohmori, T.; Ohnishi, T.; Sagara, H. Nivolumab plus ipilimumab versus existing immunotherapies in patients with pd-l1-positive advanced non-small cell lung cancer: A systematic review and network meta-analysis. Cancers 2020, 12, 1905. [Google Scholar] [CrossRef]
- Franek, J.; Cappelleri, J.C.; Larkin-Kaiser, K.A.; Wilner, K.D.; Sandin, R. Systematic review and network meta-analysis of first-line therapy for advanced egfr-positive non-small-cell lung cancer. Future Oncol. 2019, 15, 2857–2871. [Google Scholar] [CrossRef] [PubMed]
- Calles, A.; Aguado, G.; Sandoval, C.; Alvarez, R. The role of immunotherapy in small cell lung cancer. Clin. Transl. Oncol. 2019, 21, 961–976. [Google Scholar] [CrossRef] [PubMed]
- Horn, L.; Mansfield, A.S.; Szczesna, A.; Havel, L.; Krzakowski, M.; Hochmair, M.J.; Huemer, F.; Losonczy, G.; Johnson, M.L.; Nishio, M.; et al. First-line atezolizumab plus chemotherapy in extensive-stage small-cell lung cancer. N. Engl. J. Med. 2018, 379, 2220–2229. [Google Scholar] [CrossRef] [PubMed]
- Leal, T.; Wang, Y.; Dowlati, A.; Chen, Y.; Ramesh, A.; Razaq, M.; Liu, J.; Joseph, C.; King, D.M.; Ahuja, H.G.; et al. Randomized phase II clinical trial of cisplatin/carboplatin and etoposide (CE) alone or in combination with nivolumab as frontline therapy for extensivestage small cell lung cancer (ESSCLC): ECOG-ACRIN EA5161. J. Clin. Oncol. 2020, 38, 9000. [Google Scholar] [CrossRef]
- Paz-Ares, L.; Dvorkin, M.; Chen, Y.; Reinmuth, N.; Hotta, K.; Trukhin, D.; Statsenko, G.; Hochmair, M.J.; Ozguroglu, M.; Ji, J.H.; et al. Durvalumab plus platinum-etoposide versus platinum-etoposide in first-line treatment of extensive-stage small-cell lung cancer (caspian): A randomised, controlled, open-label, phase 3 trial. Lancet 2019, 394, 1929–1939. [Google Scholar] [CrossRef]
- Reck, M.; Bondarenko, I.; Luft, A.; Serwatowski, P.; Barlesi, F.; Chacko, R.; Sebastian, M.; Lu, H.; Cuillerot, J.M.; Lynch, T.J. Ipilimumab in combination with paclitaxel and carboplatin as first-line therapy in extensive-disease-small-cell lung cancer: Results from a randomized, double-blind, multicenter phase 2 trial. Ann. Oncol. 2013, 24, 75–83. [Google Scholar] [CrossRef]
- Reck, M.; Luft, A.; Szczesna, A.; Havel, L.; Kim, S.W.; Akerley, W.; Pietanza, M.C.; Wu, Y.L.; Zielinski, C.; Thomas, M.; et al. Phase iii randomized trial of ipilimumab plus etoposide and platinum versus placebo plus etoposide and platinum in extensive-stage small-cell lung cancer. J. Clin. Oncol. 2016, 34, 3740–3748. [Google Scholar] [CrossRef]
- Rudin, C.M.; Awad, M.M.; Navarro, A.; Gottfried, M.; Peters, S.; Csoszi, T.; Cheema, P.K.; Rodriguez-Abreu, D.; Wollner, M.; Yang, J.C.; et al. Pembrolizumab or placebo plus etoposide and platinum as first-line therapy for extensive-stage small-cell lung cancer: Randomized, double-blind, phase iii keynote-604 study. J. Clin. Oncol. 2020, 38, 2369–2379. [Google Scholar] [CrossRef]
- Antonia, S.J.; Lopez-Martin, J.A.; Bendell, J.; Ott, P.A.; Taylor, M.; Eder, J.P.; Jager, D.; Pietanza, M.C.; Le, D.T.; de Braud, F.; et al. Nivolumab alone and nivolumab plus ipilimumab in recurrent small-cell lung cancer (checkmate 032): A multicentre, open-label, phase 1/2 trial. Lancet Oncol. 2016, 17, 883–895. [Google Scholar] [CrossRef]
- Iams, W.T.; Porter, J.; Horn, L. Immunotherapeutic approaches for small-cell lung cancer. Nat. Rev. Clin. Oncol. 2020, 17, 300–312. [Google Scholar] [CrossRef]
- Chung, H.C.; Piha-Paul, S.A.; Lopez-Martin, J.; Schellens, J.H.M.; Kao, S.; Miller, W.H., Jr.; Delord, J.P.; Gao, B.; Planchard, D.; Gottfried, M.; et al. Pembrolizumab after two or more lines of previous therapy in patients with recurrent or metastatic sclc: Results from the keynote-028 and keynote-158 studies. J. Thorac. Oncol. 2020, 15, 618–627. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Wang, S.; Zhang, Y.; Wei, H.; Yu, J. Efficacy and safety of immune checkpoint inhibitors (icis) in extensive-stage small cell lung cancer (sclc). J. Cancer Res. Clin. Oncol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Jones, G.S.; Elimian, K.; Baldwin, D.R.; Hubbard, R.B.; McKeever, T.M. A systematic review of survival following anti-cancer treatment for small cell lung cancer. Lung Cancer 2020, 141, 44–55. [Google Scholar] [CrossRef] [PubMed]
- Facchinetti, F.; Di Maio, M.; Tiseo, M. Adding pd-1/pd-l1 inhibitors to chemotherapy for the first-line treatment of extensive stage small cell lung cancer (sclc): A meta-analysis of randomized trials. Cancers 2020, 12, 2645. [Google Scholar] [CrossRef]
- Landre, T.; Chouahnia, A.K.; Des Guetz, G.; Assié, J.B.; Chouaid, C. 1799P Immune checkpoint inhibitor plus chemotherapy versus chemotherapy alone as first-line for extensive stage small cell lung cancer: A meta-analysis. Ann. Oncol. 2020, 31 (Suppl. S4), S1041. [Google Scholar] [CrossRef]
- Villaruz, L.C.; Socinski, M.A. The Clinical Viewpoint: Definitions, Limitations of RECIST, Practical Considerations of Measurement. Clin. Cancer Res. 2013, 19, 2629–2636. [Google Scholar] [CrossRef]
- Systematic Review and Network Meta-Analysis of First-Line Therapy Combination of Immune Checkpoint Inhibitors and Chemotherapy for Extensive-Stage Small Cell Carcinoma (ID: CRD42020215762). Available online: https://www.crd.york.ac.uk/prospero/ (accessed on 26 November 2020).
- U.S. Department of Health and Human Services. Common Terminology Criteria for Adverse Events (CTCAE) Version 5.0. Available online: https://ctep.cancer.gov/protocolDevelopment/electronicapplications/docs/ (accessed on 9 October 2020).
- The Nordic Cochrane Centre, The Cochrane Collaboration. Review Manager (RevMan); Version 5.3; The Nordic Cochrane Centre, The Cochrane Collaboration: Copenhagen, Denmark, 2014. [Google Scholar]
- Stata Corp. Stata Statistical Software: Release 12, Version; StataCorp LP: College Station, TX, USA, 2011. [Google Scholar]


| Trial Name | ICI-Based Treatment | Year | Phase | Age ≥ 65 (%) | Males (%) | Brain or CNS * Meta (%) | Experimental Arm 1 (Number) | Experimental Arm 2 (Number) | Control Arm (Number) | Criteria for Progression or Response | Criteria for AE * |
|---|---|---|---|---|---|---|---|---|---|---|---|
| PD-1 inhibitors | |||||||||||
| Keynote-604 | pembrolizumab | 2020 | 3 | 52.3% | 64.9% | 12.14% | (n = 228) | (n = 225) | RECIST * | CTCAE * | |
| [18] | Induction: 4 × 21 days cycle | EP alone | version 1.1 | ||||||||
| Pembrolizumab + EP * | 4 × 21 days cycle | ||||||||||
| maintenance * | |||||||||||
| pembrolizumab every 3 weeks | |||||||||||
| EA5161 | nivolumab | 2020 | 2 | unknown | 44.4% | 11.25% | (n = 80) | (n = 80) | RECIST | CTCAE | |
| [14] | median | induction: 4 × 21 days cycle | EP alone | version 1.1 | |||||||
| Age = 65 | Nivolumab + EP | 4 × 21 days cycle | |||||||||
| maintenance * | |||||||||||
| nivolumab every 2 weeks | |||||||||||
| PD-L1 inhibitors | |||||||||||
| IMpower133 | atezolizumab | 2018 | 3 | 46.2% | 64.8% | 8.68% | (n = 201) | (n = 202) | RECIST | CTCAE | |
| [13] | induction: 4 × 21 days cycle | EP alone | version 1.1 | ||||||||
| atezolizumab + EP | 4×21 days cycle | ||||||||||
| maintenance * | |||||||||||
| atezolizumab every 3 weeks | |||||||||||
| CASPIAN | durvalumab | 2019 | 3 | 39.7% | 69.6% | 10.24% | (n = 268) | (n = 268) | (n = 269) | RECIST | CTCAE |
| [15] | induction: 4 × 21 days cycle | induction: 4 × 21 days cycle | EP alone | version 1.1 | |||||||
| durvalumab + EP | durvalumab + EP + tremelimumab | 4 × 21 days cycle | |||||||||
| Maintenance * | Maintenance * | ||||||||||
| durvalumab every 4 weeks | durvalumab every 4 weeks | ||||||||||
| CTLA4 inhibitors | |||||||||||
| CA184-041 | ipilimumab | 2013 | 2 | 23.1% | 78.4% | unknown | (n = 42) | (n = 42) | (n = 45) | 1. mWHO * | CTCAE |
| [16] | phase regman 6 × 21 days cycle | concurrent regman 6 × 21 days cycle | paclitaxel + carboplatin 4 × 21 days cycle | ||||||||
| paclitaxel + carboplatin at cycle 1–2 | ipilimumab +paclitaxel + | 2. irRC * | |||||||||
| ipilimumab +paclitaxel + carboplatin at cycle 3–6 | carboplatin at cycle 1–4 | ||||||||||
| paclitaxel + carboplatin at cycle 5–6 | |||||||||||
| maintenance * | maintenance * | ||||||||||
| ipilimumab every 12 weeks | ipilimumab every 12 weeks | ||||||||||
| CA184-156 | ipilimumab | 2016 | 3 | 39.6% | 56.9% | 10.48% | (n = 478) | (n = 476) | 1. mWHO | CTCAE | |
| [17] | phase regman: 6 × 21 days cycle | EP alone 4 × 21 days cycle | |||||||||
| EP at cycle 1–2 | 2. irRC | ||||||||||
| ipilimumab + EP at cycle 3–6 | |||||||||||
| maintenance * | |||||||||||
| Ipilimumab every 12 weeks | |||||||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, H.-L.; Tu, Y.-K.; Chang, H.-M.; Lee, T.-H.; Wu, K.-L.; Tsai, Y.-C.; Lee, M.-H.; Yang, C.-J.; Hung, J.-Y.; Chong, I.-W. Systematic Review and Network Meta-Analysis of Immune Checkpoint Inhibitors in Combination with Chemotherapy as a First-Line Therapy for Extensive-Stage Small Cell Carcinoma. Cancers 2020, 12, 3629. https://doi.org/10.3390/cancers12123629
Chen H-L, Tu Y-K, Chang H-M, Lee T-H, Wu K-L, Tsai Y-C, Lee M-H, Yang C-J, Hung J-Y, Chong I-W. Systematic Review and Network Meta-Analysis of Immune Checkpoint Inhibitors in Combination with Chemotherapy as a First-Line Therapy for Extensive-Stage Small Cell Carcinoma. Cancers. 2020; 12(12):3629. https://doi.org/10.3390/cancers12123629
Chicago/Turabian StyleChen, Hsiao-Ling, Yu-Kang Tu, Hsiu-Mei Chang, Tai-Huang Lee, Kuan-Li Wu, Yu-Chen Tsai, Mei-Hsuan Lee, Chih-Jen Yang, Jen-Yu Hung, and Inn-Wen Chong. 2020. "Systematic Review and Network Meta-Analysis of Immune Checkpoint Inhibitors in Combination with Chemotherapy as a First-Line Therapy for Extensive-Stage Small Cell Carcinoma" Cancers 12, no. 12: 3629. https://doi.org/10.3390/cancers12123629
APA StyleChen, H.-L., Tu, Y.-K., Chang, H.-M., Lee, T.-H., Wu, K.-L., Tsai, Y.-C., Lee, M.-H., Yang, C.-J., Hung, J.-Y., & Chong, I.-W. (2020). Systematic Review and Network Meta-Analysis of Immune Checkpoint Inhibitors in Combination with Chemotherapy as a First-Line Therapy for Extensive-Stage Small Cell Carcinoma. Cancers, 12(12), 3629. https://doi.org/10.3390/cancers12123629






