Venous Thromboembolism and Its Risk Factors in Children with Acute Lymphoblastic Leukemia in Israel: A Population-Based Study
Simple Summary
Abstract
1. Introduction
2. Results
2.1. Patient Characteristics
2.2. Prevalence and Characteristics of VTE Events
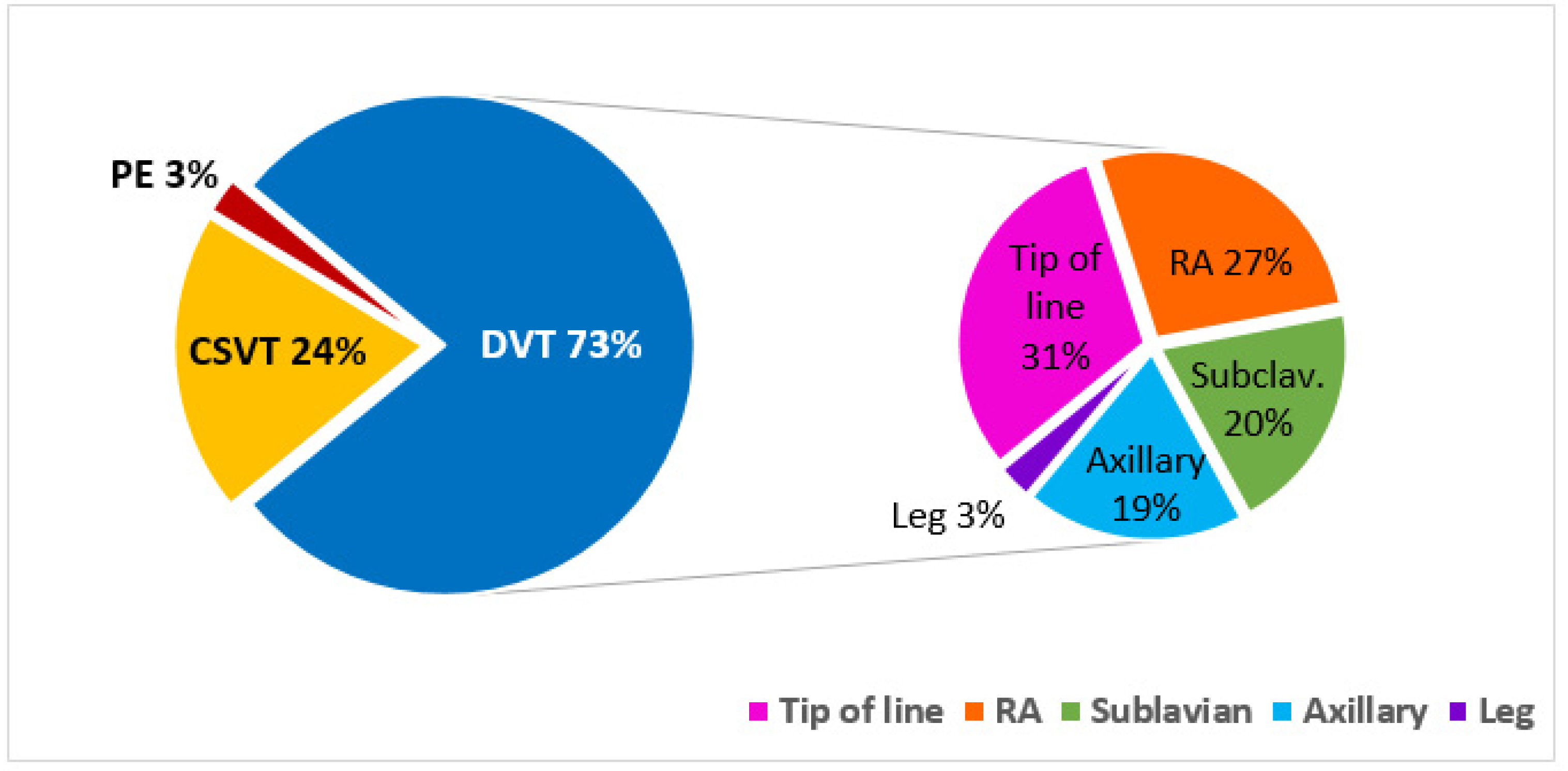
2.3. Sites of VTE Events
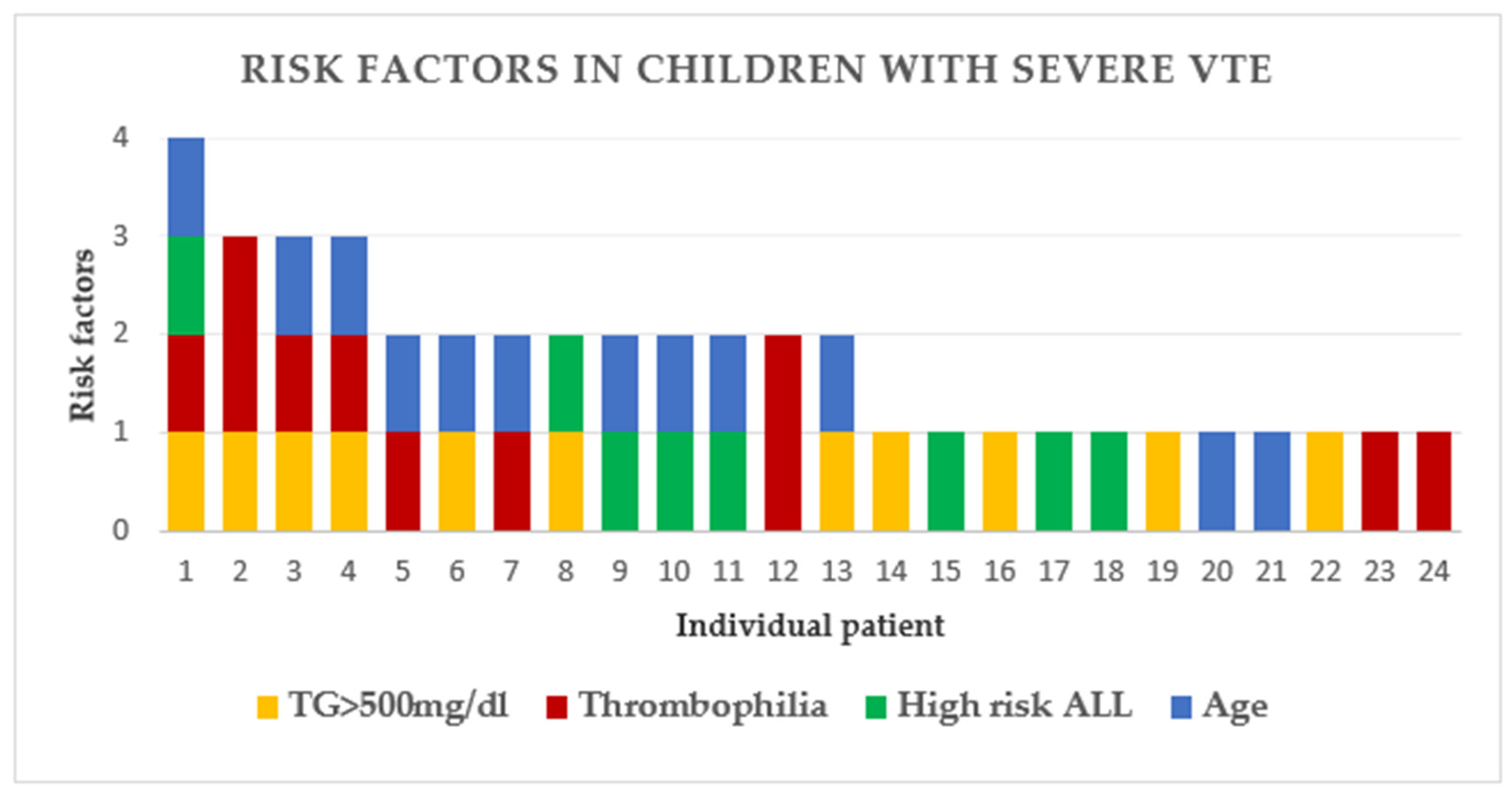
2.4. Risk Factors for VTE; Univariate Analysis
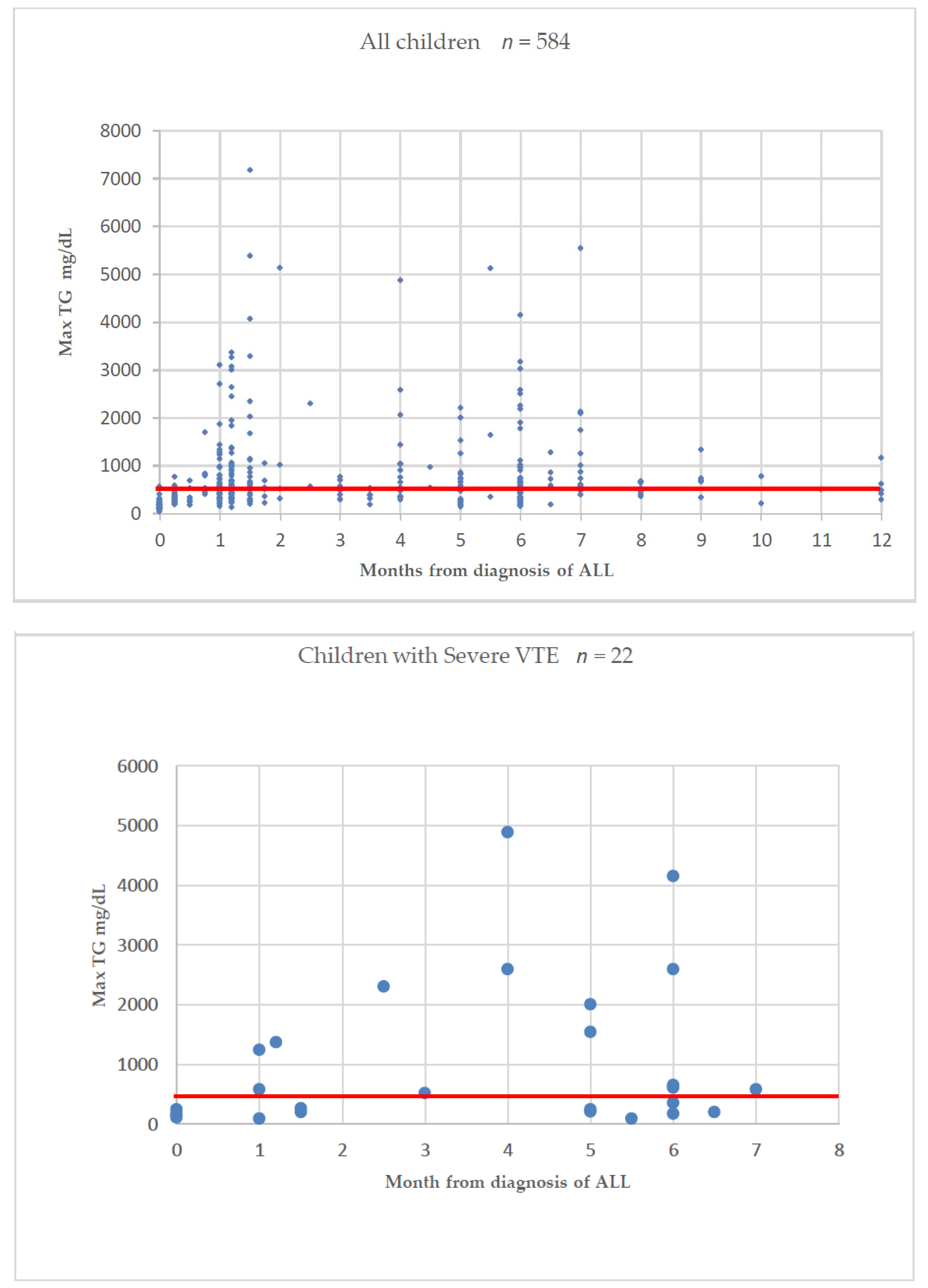
Hypertriglyceridemia in Children with ALL
2.5. Risk Factors for VTE; Multivariate Analysis
2.6. Outcome of VTE
3. Discussion
4. Conclusions
5. Methods
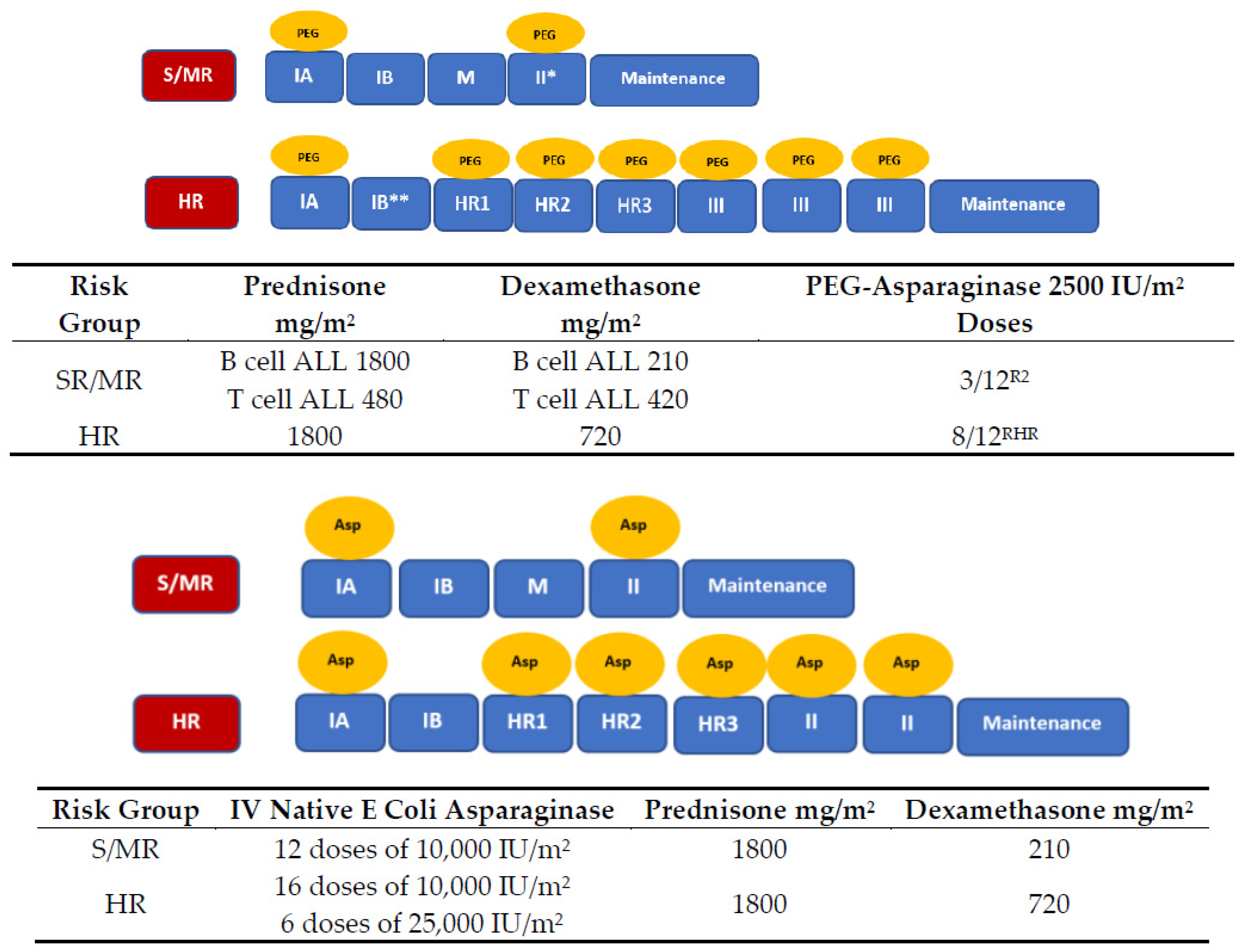
5.1. Patients and Setting
5.2. Data Collection
5.3. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Caruso, V.; Iacoviello, L.; Di Castelnuovo, A.; Storti, S.; Mariani, G.; de Gaetano, D.; Donati, M.B. Thrombotic complications in childhood acute lymphoblastic leukemia: A meta-analysis of 17 prospective studies comprising 1752 pediatric patients. Blood 2006, 108, 2216–2222. [Google Scholar] [CrossRef]
- Priest, J.R.; Ramsay, N.K.; Bennett, A.J.; Krivit, W.; Edson, J.R. The effect of L-asparaginase on antithrombin, plasminogen, and plasma coagulation during therapy for acute lymphoblastic leukemia. J. Pediatr. 1982, 100, 990–995. [Google Scholar] [CrossRef]
- Rodeghiero, F.; Castaman, G.; Dini, E. Fibrinopeptide A changes during remission induction treatment with L-asparaginase in acute lymphoblastic leukemia: Evidence for activation of blood coagulation. Thromb. Res. 1990, 57, 31–38. [Google Scholar] [CrossRef]
- Mitchell, L.; Hoogendoorn, H.; Giles, A.R.; Vegh, P.; Andrew, M. Increased endogenous thrombin generation in children with acute lymphoblastic leukemia: Risk of thrombotic complications in L’Asparaginase induced antithrombin III deficiency. Blood 1994, 83, 386–391. [Google Scholar] [CrossRef]
- Oner, A.F.; Gürgey, A.; Kirazli, S.; Okur, H.; Tunç, B. Changes of hemostatic factors in children with acute lymphoblastic leukemia receiving combined chemotherapy including high dose methylprednisolone and L-asparaginase. Leuk. Lymphoma 1999, 33, 361–364. [Google Scholar] [CrossRef]
- Giordano, P.; Del Vecchio, G.C.; Santoro, N.; Arcamone, G.; Coppola, B.; Altomare, M.; Schettini, F.; Iolascon, A.; De Mattia, D. Thrombin generation in children with acute lymphoblastic leukemia: Effect of leukemia immunophenotypic subgroups. Pediatr. Hematol. Oncol. 2000, 17, 667–672. [Google Scholar] [CrossRef]
- Uszyński, M.; Osińska, M.; Zekanowska, E.; Ziółkowska, E. Children with acute lymphoblastic leukemia: Is there any subgroup of children without elevated thrombin generation? A preliminary study utilizing measurements of thrombin-antithrombin III complexes. Med. Sci. Monit. 2000, 6, 108–111. [Google Scholar]
- Rickles, F.R.; Falanga, A. Molecular basis for the relationship between thrombosis and cancer. Thromb. Res. 2001, 102, V215–V224. [Google Scholar] [CrossRef]
- Sutherland, D.E.; Weitz, I.C.; Liebman, H.A. Thromboembolic complications of cancer: Epidemiology, pathogenesis, diagnosis, and treatment. Am. J. Hematol. 2003, 72, 43–52. [Google Scholar] [CrossRef]
- Mondelaers, V.; Bauters, T.; De Moerloose, B.; Benoit, Y. PEG-asparaginase in the treatment of childhood acute lymphoblastic leukaemia. Belg. J. Hematol. 2013, 4, 138–143. [Google Scholar]
- Bushman, J.E.; Palmieri, D.; Whinna, H.C.; Church, F.C. Insight into the mechanism of asparaginase-induced depletion of antithrombin III in treatment of childhood acute lymphoblastic leukemia. Leuk. Res. 2000, 24, 559–565. [Google Scholar] [CrossRef]
- Pui, C.H.; Chesney, C.M.; Weed, J.; Jackson, C.W. Altered von Willebrand factor molecule in children with thrombosis following asparaginase-prednisone-vincristine therapy for leukemia. J. Clin. Oncol. 1985, 3, 1266–1272. [Google Scholar] [CrossRef]
- Shapiro, A.D.; Clarke, S.L.; Christian, J.M.; Odom, L.F.; Hathaway, W.E. Thrombosis in children receiving L-asparaginase. Determining patients at risk. Am. J. Pediatr. Hematol. Oncol. 1993, 15, 400–405. [Google Scholar]
- Mitchell, L.G.; Sutor, A.H.; Andrew, M. Hemostasis in childhood acute lymphoblastic leukemia: Coagulopathy induced by disease and treatment. Semin. Thromb. Hemost. 1995, 21, 390–401. [Google Scholar] [CrossRef]
- Leone, G.; Gugliotta, L.; Mazzucconi, M.G.; De Stefano, V.; Belmonte, M.M.; Dragoni, F.; Specchia, G.; Centra, A.; Gamba, G.; Camera, A.; et al. Evidence of a hypercoagulable state in patients with acute lymphoblastic leukemia treated with low dose of E. coli L-asparaginase: A GIMEMA study. Thromb. Haemost. 1993, 69, 12–15. [Google Scholar]
- Payne, J.H.; Vora, A.J. Thrombosis and acute lymphoblastic leukaemia. Br. J. Haematol. 2007, 138, 430–445. [Google Scholar] [CrossRef]
- Male, C.; Chait, P.; Andrew, M.; Hanna, K.; Julian, J.; Mitchell, L. PARKAA Investigators. Central venous line-related thrombosis in children: Association with central venous line location and insertion technique. Blood 2003, 101, 4273–4278. [Google Scholar] [CrossRef]
- Wermes, C.; von Depka Prondzinski, M.; Lichtinghagen, R.; Barthels, M.; Welte, K.; Sykora, K.W. Clinical relevance of genetic risk factors for thrombosis in paediatric oncology patients with central venous catheters. Eur. J. Pediatr. 1999, 158, S143–S146. [Google Scholar] [CrossRef]
- Athale, U.H.; Chan, A.K. Thrombosis in children with acute lymphoblastic leukemia: Part I. Epidemiology of thrombosis in children with acute lymphoblastic leukemia. Thromb. Res. 2003, 111, 125–131. [Google Scholar] [CrossRef]
- Silverman, L.B.; Gelber, R.D.; Dalton, V.K.; Asselin, B.L.; Barr, R.D.; Clavell, L.A.; Hurwitz, C.A.; Moghrabi, A.; Samson, Y.; Schorin, M.A.; et al. Improved outcome of children with acute lymphoblastic leukemia: Results of Dana-Farber Consortium Protocol 91-01. Blood 2001, 97, 1211–1217. [Google Scholar] [CrossRef]
- Athale, U.H.; Siciliano, S.A.; Crowther, M.; Barr, R.D.; Chan, A.K. Thromboembolism in children with acute lymphoblastic leukaemia treated on Dana-Farber Cancer Institute protocols: Effect of age and risk stratification of disease. Br. J. Haematol. 2005, 129, 803–810. [Google Scholar] [CrossRef]
- Rank, C.U.; Toft, N.; Tuckuviene, R.; Grell, K.; Nielsen, O.J.; Frandsen, T.L.; Marquart, H.V.H.; Albertsen, B.K.; Ulf Tedgard, U.; Helene Hallbook, H.; et al. Thromboembolism in acute lymphoblastic leukemia: Results of NOPHO ALL2008 protocol treatment in patients aged 1 to 45 years. Blood 2018, 131, 2475–2484. [Google Scholar] [CrossRef]
- Wermes, C.; Fleischhacck, G.; Junker, R.; Schobess, R.; Schawabe, D.; Sykora, K.W.; Nowak-Gottl, U. Cerebral venous sinus thrombosis in children with acute lymphoblastic leukemia carrying the MTHFR TT677 genotype and further prothrombotic risk factors. Klin. Padiatr. 1999, 211, 211–214. [Google Scholar] [CrossRef]
- Ott, N.; Ramsay, N.K.C.; Priest, J.R.; Lipton, M.; Pui, C.H.; Steinherz, P.; Nesbit, M.E. Sequelae of thrombotic or hemorrhagic complications following L-asparaginase therapy for childhood lymphoblastic leukemia. Am. J. Pediatr. Hematol. Oncol. 1988, 10, 191–195. [Google Scholar] [CrossRef]
- Mitchell, L.G.; Lambers, M.; Flege, S.; Kenet, G.; Li-Thiao-Te, V.; Holzhauer, S.; Bidlingmaier, C.; Fruhwald, M.C.; Heller, C.; Schmidt, W.; et al. Validation of a predictive model for identifying an increased risk for thromboembolism in children with acute lymphoblastic leukemia: Results of a multicenter cohort study. Blood 2010, 115, 4999–5004. [Google Scholar] [CrossRef]
- Bhojwani, D.; Darbandi, R.; Pei, D. Severe hypertriglyceridaemia during therapy for childhood acute lymphoblastic leukaemia. Eur. J. Cancer 2014, 50, 2685–2694. [Google Scholar] [CrossRef]
- Finch, E.R.; Smith, C.A.; Yang, W.; Liu, Y.; Kornegay, N.M.; Panetta, J.C.; Crews, K.R.; Molinelli, A.R.; Cheng, C.; Pei, D.; et al. Asparaginase formulation impacts hypertriglyceridemia during therapy for acute lymphoblastic leukemia. Pediatr. Blood Cancer 2020, 1, e28040. [Google Scholar] [CrossRef]
- Cohen, H.; Bielorai, B.; Harats, D.; Toren, A.; Pinhas-Hamiel, O. Conservative treatment of L-asparaginase-associated lipid abnormalities in children with acute lymphoblastic leukemia. Pediatr. Blood Cancer 2010, 54, 703–706. [Google Scholar] [CrossRef]
- Stock, W.; Douer, D.; DeAngelo, D.J.; Arellano, M.; Advani, A.; Damon, L.; Kovacsovics, T.; Litzow, M.; Rytting, M.; Borthakur, G.; et al. Prevention and management of asparaginase/ pegasparaginase-associated toxicities in adults and older adolescents: Recommendations of an expert panel. Leuk. Lymphoma 2011, 52, 2237–2253. [Google Scholar] [CrossRef]
- Howard, S.C.; Pui, C.H. Endocrine complications in pediatric patients with acute lymphoblastic leukemia. Blood Rev. 2002, 16, 224–234. [Google Scholar] [CrossRef]
- Steinherz, P.G. Transient, severe hyperlipidemia in patients with acute lymphoblastic leukemia treated with prednisone and asparaginase. Cancer 1994, 74, 3234–3239. [Google Scholar] [CrossRef]
- Place, A.E.; Stevenson, K.E.; Vrooman, L.M. Intravenous pegylated asparaginase versus intramuscular native Escherichia coli L-asparaginase in newly diagnosed childhood acute lymphoblastic leukaemia (DFCI 05-001): A randomised, open-label phase 3 trial. Lancet Oncol. 2015, 16, 1677–1690. [Google Scholar] [CrossRef]
- Franchini, M.; Targher, G.; Montagnana, M.; Lippi, G. The metabolic syndrome and the risk of arterial and venous thrombosis. Thromb. Res. 2008, 122, 727–735. [Google Scholar] [CrossRef] [PubMed]
- Ageno, W.; Prandoni, P.; Romualdi, E.; Ghirarduzzi, A.; Dentali, F.; Pesavento, R.; Crowther, M.; Venco, A. The metabolic syndrome and the risk of venous thrombosis: A case–control study. J. Thromb. Headmost. 2006, 4, 1914–1918. [Google Scholar] [CrossRef]
- Doggen, C.J.; Smith, N.L.; Lemaitre, R.N.; Heckbert, S.R.; Rosendaal, F.R.; Psaty, B.M. Serum lipid levels and the risk of venous thrombosis. Arter. Thromb. Vasc. Biol. 2004, 24, 1970–1975. [Google Scholar] [CrossRef]
- Vaya, A.; Mira, Y.; Ferrando, F.; Contreras, M.T.; Estelles, A.; Espana, F.; Corella, D.; Aznar, J. Hyperlipidemia and venous thromboembolism in patients lacking thrombophilic risk factors. Br. J. Haematol. 2002, 118, 255–259. [Google Scholar] [CrossRef]
- Barzilai-Birenboim, S.; Arad-Cohen, N.; Nirel, R.; Avrahami, G.; Harlev, D.; Gilad, G.; Elhasid, R.; Shai Izraeli, S.; Litichever, N.; Elitzur, S. Thrombophilia screening and thromboprophylaxis may benefit specific ethnic subgroups with paediatric acute lymphoblastic leukaemia. Br. J. Haematol. 2019, 184, 994–998. [Google Scholar] [CrossRef]
- Elhasid, R.; Lanir, N.; Sharon, R.; Levin, C.; Postovsky, S.; Ben Barak, A.; Brenner, B. Prophylactic therapy with enoxaparin during L-asparaginase treatment in children with acute lymphoblastic leukemia. Blood Coagul. Fibrinolysis 2001, 12, 367–370. [Google Scholar] [CrossRef]
- Hunault-Berger, M.; Chevallier, P.; DeLain, M.; Bulabois, C.-E.; Bologna, S.; Bernard, M.; Lafon, I.; Cornillon, J.; Maakaroun, A.; Tizon, A.; et al. GOELAMS (Groupe Ouest-Est des Leucémies Aiguës et Maladies du Sang). Changes in antithrombin and fibrinogen levels during induction chemotherapy with L-asparaginase in adult patients with acute lymphoblastic leukemia or lymphoblastic lymphoma. Use of supportive coagulation therapy and clinical outcome: The CAPELAL study. Haematologica 2008, 93, 1488–1494. [Google Scholar] [CrossRef]
- Meister, B.; Kropshofer, G.; Klein-Franke, A.; Strasak, A.M.; Hager, J.; Streif, W. Comparison of low-molecular-weight heparin and antithrombin versus antithrombin alone for the prevention of symptomatic venous thromboembolism in children with acute lymphoblastic leukemia. Pediatr. Blood Cancer 2008, 50, 298–303. [Google Scholar] [CrossRef]
- Greiner, J.; Schrappe, M.; Claviez, A.; Zimmermann, M.; Niemeyer, C.; Kolb, R.; Eberl, W.; Berthold, F.; Bergsträsser, E.; Gnekow, A.; et al. THROMBOTECT—A randomized study comparing low molecular weight heparin, antithrombin and unfractionated heparin for thromboprophylaxis during induction therapy of acute lymphoblastic leukemia in children and adolescents. Haematologica 2019, 104, 756–765. [Google Scholar] [CrossRef] [PubMed]
- U.S. Department of Health and Human Services. National Institutes of Health and National Cancer Institute. Common Terminology Criteria for Adverse Events (CTCAE) Version 4.0. (Published 28 May 2009; v4.03: 14 June 2010). Available online: https://evs.nci.nih.gov/ftp1/CTCAE/CTCAE_4.03/Archive/CTCAE_4.0_2009-05-29_QuickReference_8.5x11.pdf (accessed on 22 September 2020).
- Schmiegelow, K.; Attarbaschi, A.; Barzilai, S.; Escherich, G.; Frandsen, T.L.; Halsey, C.; Hough, R.; Jeha, S.; Kato, M.; Liang, D.C.; et al. Ponte di Legno toxicity working group. Consensus definitions of 14 severe acute toxic effects for childhood lymphoblastic leukaemia treatment: A Delphi consensus. Lancet Oncol. 2016, 17, e231–e239. [Google Scholar] [CrossRef]
- Warrens, M. On Association Coefficients for 2x2 Tables and Properties That Do Not Depend on the Marginal Distributions. Psychometrika 2008, 73, 777–789. [Google Scholar] [CrossRef] [PubMed]
- Hanley, J.A.; McNeil, B.J. The meaning and use of the area under a receiver operating characteristic (ROC) curve. Radiology 1982, 143, 29–36. [Google Scholar] [CrossRef]

| Characteristic | Value |
|---|---|
| Sex | |
| Female | 501 (42.1%) |
| Male | 690 (57.9%) |
| Age (year), median (range) | 6 (1.02–19.30) |
| ALL treatment risk group | |
| High risk | 262 (22.0%) |
| Non-high risk | 929 (78.0%) |
| Lineage | |
| T | 190 (16.0%) |
| B | 1001 (84.0%) |
| Treatment protocol * | |
| 2002 | 591 (49.6%) |
| 2009 | 600 (50.4%) |
| Risk Factors | Children n (%) | No VTE | VTE | p-Value * VTE vs. No VTE | Severe VTE | p-Value † Severe VTE vs. No VTE | Mild VTE | p-Value † Mild VTE vs. No VTE |
|---|---|---|---|---|---|---|---|---|
| All Cohort | n = 1191 | n = 1102 | n = 89 | n = 24 | n = 65 | |||
| Protocol | ||||||||
| BFM 2002 BFM 2009 | 591 (49.6%) 600 (50.4%) | 563 539 | 28 (4.7%) 61 (10.2%) | 0.001 | 6 (1.0%) 18 (3.0%) | 0.010 | 22 (3.7%) 43 (7.2%) | 0.007 |
| Age group | ||||||||
| <10 years ≥10 years | 840 (70.5%) 351 (29.5%) | 789 313 | 51 (57.3%) 38 (42.7%) | 0.015 | 12 (50%) 12 (50%) | 0.037 | 39 (60%) 26 (40%) | 0.050 |
| Risk group | ||||||||
| HR Non-HR | 262 (22.0%) 929 (78.0%) | 233 869 | 29 (32.6%) 60 (67.4%) | 0.039 | 8 (33.3%) 16 (66.7%) | 0.202 | 21 (32.3%) 44 (67.7%) | 0.042 |
| Lineage | ||||||||
| T B | 190 (16.0%) 100 (84.0%) | 175 927 | 15 (16.8%) 74 (83.2%) | 0.921 | 4 (16.7%) 20 (83.3%) | 11 (16.9%) 54 (83.1%) | ||
| Sex | ||||||||
| Female Male | 501 (42.1%) 690 (57.9%) | 470 632 | 31 (34.8%) 58 (65.2%) | 0.179 | 6 (25%) 18 (75%) | 25 (38.5%) 40 (61.5%) | ||
| SCMCI γ | n = 388 | n = 31 | n = 15 | n = 16 | ||||
| TG | ||||||||
| ≤500 mg/dL ψ > 500 mg/dL | 285 (73.5%) 103 (26.5%) | 270 87 | 15 (48.4%) 16 (51.6%) | 0.005 | 7 (46.7%) 8 (53.3%) | 0.029 | 8 (50%) 8 (50%) | 0.035 |
| Thrombophilia | ||||||||
| Yes No | 52 (13.4%) 336 (86.6%) | 37 320 | 15 (48.4%) 16 (51.6%) | <0.001 | 9 (60%) 6 (40%) | <0.001 | 6 (37.5%) 10 (62.5%) | 0.005 |
| BMI% | 46.7 ± 32.4 | 45.1 ± 33.6 | 0.613 | 50.7 ± 32.8 | 39.5 ± 34.6 |
| Pt. | Age (year) | CL | Phase | Event | TG Diag | Max TG | Time Max TG | RF + | Symptoms | Risk Group | Thrombophilia | ASP. Formula | Modifications | Status | Relapse |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 1 | 12.1 | Port | IA | CSVT | 275 | 526 | 3 m | 4 | Coma | B-HR | FII | PEG | Stop PEG | Alive | yes |
| 2 | 5.4 | Port | HDMTX | CSVT | 259 | 1554 | 5 m | 3 | Mild headaches | B-S/MR | FVL + FVL | PEG | No | Alive | No |
| 3 | 13.9 | Port | IA | CSVT | 261 | 2600 | 6 m | 3 | Hemiparesis & dysarthria | T-nHR | Lp(a) | PEG | Stop PEG | Alive | No |
| 4 | 18.5 | Port | IB | CSVT | 179 | 662 | 6 m | 3 | Coma | T-nHR | Homocy | PEG | Stop PEG | Alive | No |
| 5 | 13.7 | Port | IB | CSVT | 113 | 267 | 6 w | 2 | Hemiparesis & dysarthria | B-S/MR | APLA | E coli | No | Alive | No |
| 6 | 13 | Pick | IA | CSVT | 210 | 4163 | 6 m | 2 | SZ & hemiparesis | B-S/MR | no | E coli | Stop ASP | Alive | No |
| 7 | 15.4 | Port | IB | CSVT | 61 | 360 | 6 m | 2 | Severe headaches | B-S/MR | FVL | PEG | No | Alive | No |
| 8 | 9 | Port | IB | CSVT | 204 | 2309 | 5 m | 2 | Earaches | B-HR | no | PEG | No | Alive | No |
| 9 | 15 | Port | IIB | CSVT | 117 | 148 | 3 w | 2 | Hemiparesis & facialis | B-HR | no | E coli | No | Alive | No |
| 10 | 15.5 | Port | IIIA | PE | NA | 208 | NA | 2 | Chest pain & dyspnea | B-HR | no | E coli | No | Alive | No |
| 11 | 16 | Pick | IA | CSVT | NA | NA | NA | 2 | SZ | B-HR | NA | E coli | Stop PEG | Alive | No |
| 12 | 4.1 | Port | IB | CSVT | 127 | 127 | 1 | 2 | Mild headaches & fatigue | B-S/MR | FII + FVL | PEG | No | Alive | No |
| 13 | 10.9 | Port | IB | CSVT | 113 | 610 | 6 m | 2 | SZ | B-S/MR | no | PEG | Stop PEG | Alive | No |
| 14 | 1.7 | Port | IA | CSVT | 87 | 1247 | 4 w | 1 | Severe headaches & earaches | B-S/MR | no | PEG | No | Alive | No |
| 15 | 6.8 | Port | IA | PE | 72 | 250 | 4 w | 1 | Tachycardia | T-HR | no | PEG | No | Alive | No |
| 16 | 7 | Port | IB | CSVT | 168 | 1370 | 5 w | 1 | SZ | B-S/MR | no | PEG | No | Alive | No |
| 17 | 7 | Pick | HR3 | CSVT | NA | 211 | NA | 1 | SZ | B-HR | no | PEG | No | Dead | Yes |
| 18 | 7.2 | Port | IA | CSVT | NA | 100 | NA | 1 | SZ | B-HR | no | PEG | Stop PEG | Alive | No |
| 19 | 9.2 | Port | IA | CSVT | 228 | 2013 | 5 m | 1 | Status epilepticus | B-S/MR | no | PEG | Postpone IT | Alive | yes |
| 20 | 13.2 | Pick | IIB | CSVT | NA | NA | NA | 1 | Hemiparesis | T-nHR | NA | E coli | No | Alive | No |
| 21 | 17.6 | Port | IA | CSVT | NA | 98 | NA | 1 | SZ and syncope | B-S/MR | NA | PEG | Postpone IB | Alive | No |
| 22 | 4.9 | Port | IB | CSVT | 256 | 582 | 4 w | 1 | Severer headaches | B-S/MR | no | PEG | No | Alive | No |
| 23 | 7.2 | Port | IIA | PE | 95 | 175 | 6 m | 1 | Tachycardia & tachypnea | B-S/MR | Lp(a) | PEG | No | Alive | No |
| 24 | 2.4 | Port | IB | CSVT | 108 | 206 | 5 m | 1 | SZ | B-S/MR | FII | PEG | Stop PEG | Alive | No |
| Covariate | Reference | OR | 95% CI | p-Value |
|---|---|---|---|---|
| Age | <10 years | 0.95 | 0.36, 2.35 | 0.91 |
| Gender | Female | 1.46 | 0.61, 3.74 | 0.41 |
| BMI percentile | 0.99 | 0.98, 1.01 | 0.30 | |
| Protocol | ALL-IC BFM 2002 | 2.30 | 0.97, 5.83 | 0.07 |
| Thrombophilia | No | 10.58 | 4.41, 26.28 | <0.001 |
| Lineage | Non-T | 0.80 | 0.23, 2.40 | 0.71 |
| Risk group | Non-HR | 0.63 | 0.20, 1.75 | 0.40 |
| Severe hyper-TG ≥ 500 mg/dL | Non-severe | 3.90 | 1.59, 9.78 | 0.003 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Barzilai-Birenboim, S.; Nirel, R.; Arad-Cohen, N.; Avrahami, G.; Ben Harush, M.; Barg, A.A.; Bielorai, B.; Elhasid, R.; Gilad, G.; Toren, A.; et al. Venous Thromboembolism and Its Risk Factors in Children with Acute Lymphoblastic Leukemia in Israel: A Population-Based Study. Cancers 2020, 12, 2759. https://doi.org/10.3390/cancers12102759
Barzilai-Birenboim S, Nirel R, Arad-Cohen N, Avrahami G, Ben Harush M, Barg AA, Bielorai B, Elhasid R, Gilad G, Toren A, et al. Venous Thromboembolism and Its Risk Factors in Children with Acute Lymphoblastic Leukemia in Israel: A Population-Based Study. Cancers. 2020; 12(10):2759. https://doi.org/10.3390/cancers12102759
Chicago/Turabian StyleBarzilai-Birenboim, Shlomit, Ronit Nirel, Nira Arad-Cohen, Galia Avrahami, Miri Ben Harush, Assaf Arie Barg, Bella Bielorai, Ronit Elhasid, Gil Gilad, Amos Toren, and et al. 2020. "Venous Thromboembolism and Its Risk Factors in Children with Acute Lymphoblastic Leukemia in Israel: A Population-Based Study" Cancers 12, no. 10: 2759. https://doi.org/10.3390/cancers12102759
APA StyleBarzilai-Birenboim, S., Nirel, R., Arad-Cohen, N., Avrahami, G., Ben Harush, M., Barg, A. A., Bielorai, B., Elhasid, R., Gilad, G., Toren, A., Weinreb, S., Izraeli, S., & Elitzur, S. (2020). Venous Thromboembolism and Its Risk Factors in Children with Acute Lymphoblastic Leukemia in Israel: A Population-Based Study. Cancers, 12(10), 2759. https://doi.org/10.3390/cancers12102759






