Cutaneous Metastases in Ovarian Cancer
Abstract
1. Introduction
2. Epidemiology
3. Patterns
3.1. Sister Joseph Nodules
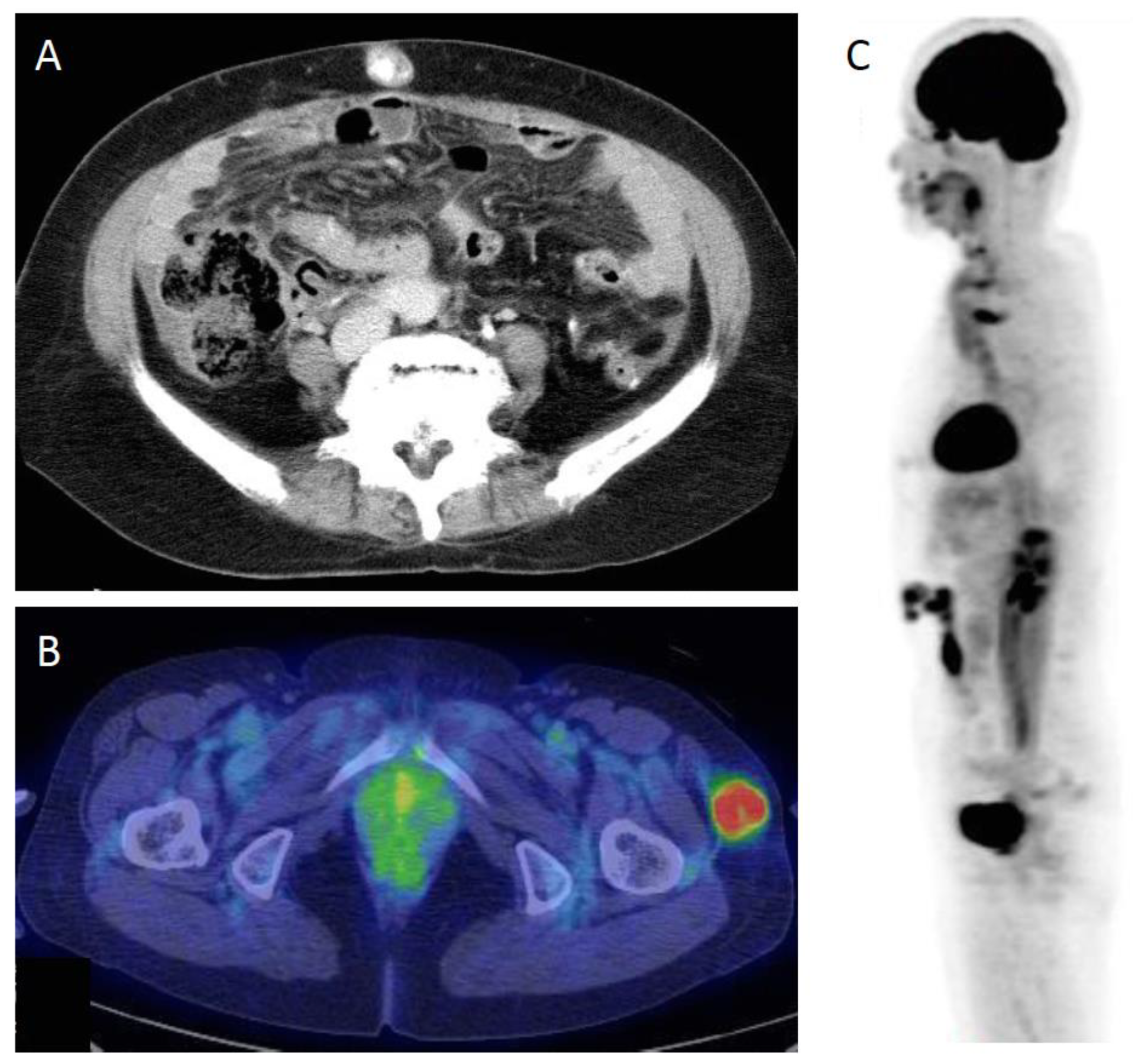
3.2. Non-SJN Metastasis
4. Possible Mechanisms
4.1. Preceding Conditions
4.1.1. Intraperitoneal Metastasis
4.1.2. Surgery and Chemotherapy
4.1.3. Superficial Lymphadenopathy
4.2. Mode of Spread
4.2.1. Contiguous Spread
4.2.2. Direct Implantation
4.2.3. Lymphatic Spread and Extranodular Extension
4.2.4. Hematogenous Spread
4.3. Factors Affecting Tumor Growth
4.3.1. Wound Healing and Inflammation
4.3.2. Adipose Tissue
5. Risk Factors
5.1. Tumor Factors
5.2. Treatment Factors
5.3. Patient Factors
6. Diagnosis
6.1. Sister Joseph Nodule
6.2. Non-SJN Metastases
6.3. Diagnostic Tests
7. Prognostic Factors
8. Treatment
9. Conclusions
Supplementary Materials
Funding
Conflicts of Interest
References
- Dauplat, J.; Hacker, N.F.; Nieverg, R.K.; Berek, J.S.; Rose, T.P.; Sagae, S. Distant metastases in epithelial ovarian carcinoma. Cancer 1987, 60, 1561–1566. [Google Scholar] [CrossRef]
- Otsuka, I.; Matsuura, T. Skin metastases in epithelial ovarian and fallopian tube carcinoma. Medicine 2017, 96, e7798. [Google Scholar] [CrossRef]
- Patsner, B.; Mann, W.J.; Chumas, J.; Loesch, M. Herpetiform cutaneous metastases following negative second look laparotomy for ovarian adenocarcinoma. Arch. Gynecol. Obstet. 1988, 244, 63–67. [Google Scholar] [CrossRef]
- Traiman, P.; De Luca, L.A.; Bacchi, C.E. An extremely large, cauliflower-type, cutaneous metastasis of ovarian cancer associated with good prognosis. Gynecol. Oncol. 1994, 53, 239–241. [Google Scholar] [CrossRef]
- Cowan, L.J.; Roller, J.I.; Connelly, P.J.; Nahhas, W.A. Extraovarian stage IV peritoneal serous papillary carcinoma presenting as an asymptomatic skin lesion—A case report and literature review. Gynecol. Oncol. 1995, 57, 433–435. [Google Scholar] [CrossRef]
- Leminen, A.; Lehtovirta, P. Spread of ovarian cancer after laparoscopic surgery: Report of eight cases. Gynecol. Oncol. 1999, 75, 387–390. [Google Scholar] [CrossRef]
- Cormio, G.; Capotorto, M.; Di Vagno, G.; Cazzolla, A.; Carriero, C.; Selvaggi, L. Skin metastases in ovarian carcinoma: A report of nine cases and a review of the literature. Gynecol. Oncol. 2003, 90, 682–685. [Google Scholar] [CrossRef]
- Demirci, S.; Yavas, F.; Ozsaran, Z.; Ozsaran, A.; Dikmen, Y.; Zekioglu, O.; Karabulut, B.; Aras, A.B. Palliative radiotherapy for the skin metastasis of ovarian cancer: A case report and review of the literature. Med. Oncol. 2010, 27, 628–631. [Google Scholar] [CrossRef]
- Gupta, P.; Kumari, A.; Kriplani, A.; Bhatia, N. Sister Mary Joseph’s nodule as the first and only sign of recurrence in a case of stage Ia carcinoma ovary. BMJ Case Rep. 2014, 2014, bcr2013201779. [Google Scholar] [CrossRef]
- Cheng, H.; Gao, C.; Zhang, R.; Yang, Z.; Zhang, G. Two independent incidences of skin metastases in the umbilicus and abdominal wall in ovarian serous adenocarcinoma. A case report and review of the literature. Medicine 2017, 96, 49. [Google Scholar] [CrossRef]
- Kim, M.K.; Kim, S.H.; Lee, Y.Y.; Choi, C.H.; Kim, T.J.; Lee, J.W.; Lee, J.H.; Bae, D.S.; Kim, B.G. Metastatic skin lesion of lower extremities in a patient recurrent serous papillary ovarian carcinoma: A case report and literature review. Cancer Res. Treat. 2012, 44, 142–145. [Google Scholar] [CrossRef]
- Cheng, B.; Lu, W.; Xiaonyun, W.; Yaxia, C.; Xie, X. Extra-abdominal metastases from epithelial ovarian carcinoma: An analysis of 20 cases. Int. J. Gynecol. Cancer 2009, 19, 611–614. [Google Scholar] [CrossRef]
- Brownstein, M.H.; Helwig, E.B. Metastatic tumors of the skin. Cancer 1972, 29, 1298–1307. [Google Scholar] [CrossRef]
- Lookingbill, D.P.; Spangler, N.; Helm, K.F. Cutaneous metastases in patients with metastatic carcinoma: A retrospective study of 4020 patients. J. Am. Acad. Dermatol. 1993, 29, 228–236. [Google Scholar] [CrossRef]
- Sariya, D.; Rugh, K.; Adams-McDonnell, R.; Cusack, C.; Xu, X.W.; Elenitsas, R.; Seykora, J.; Pasha, T.; Zhang, P.; Baldassano, M.; et al. Clinicopathologic correlation of cutaneous metastases: Experience form a cancer center. Arch. Dermatol. 2007, 143, 613–620. [Google Scholar] [CrossRef]
- Tharakaram, S. Metastases to the skin. Int. J. Dermatol. 1988, 27, 240–242. [Google Scholar] [CrossRef]
- Brustman, L.; Seltzer, V. Sister Joseph’s nodule: Seven cases of umbilical metastases from gynecologic malignancies. Gynecol. Oncol. 1984, 19, 155–162. [Google Scholar] [CrossRef]
- Kato, K.; Koshiba, H.; Kikuchi, N.; Ogino, Y.; Fujita, H.; Okumura, J.; Murakami, A. Sister Mary Joseph’s nodule: Report of a case of umbilical metastasis from the primary tubal cancer. Kyoto Second Red Cross Hosp. Med. J. 2000, 21, 90–97. (In Japanese) [Google Scholar]
- Hashimoto, T.; Ezaki, K.; Yamada, K.; Isonishi, S.; Sasaki, H.; Ochiai, K.; Tanaka, T. Six cases of ovarian carcinoma with umbilical metastasis. Sanfujinka No Jissai 2000, 49, 2063–2067. (In Japanese) [Google Scholar]
- Yu, S.; Kurimasa, E.; Ohyagi, C.; Isobe, A.; Miyanishi, K.; Boku, K.; Yamamoto, T. Five cases of Sister Mary Joseph’s nodule treated at Sakai Municipal Hospital. Sakai Munic. Hosp. J. 2008, 11, 23–27. (In Japanese) [Google Scholar]
- Kurt, S.; Camuzcuoglu, H.; Camuzcuoglu, A.; Dicle, N.; Tinar, S. Umbilical metastasis of serous component as a first sign of mixed type epithelial ovarian cancer. Eur. J. Gynaecol. Oncol. 2009, 30, 455–457. [Google Scholar]
- Fukushima, J.; Shinohara, S.; Tagaya, H.; Fukazawa, H.; Omori, M.; Hasi, A.; Hirata, S. Two cases of advanced adnexal malignant tumor detected with Sister Mary Joseph’s Nodule. Yamanashi Obstet. Gynecol. J. 2014, 4, 53–59. (In Japanese) [Google Scholar]
- Kirshtein, B.; Meirovitz, M.; Okon, E.; Piura, B. Sister Mary Joseph’s nodule as the first presenting sign of primary fallopian tube adenocarcinoma. J. Minim. Invasive Gynecol. 2006, 13, 234–236. [Google Scholar] [CrossRef]
- Calista, D.; Florentini, C.; Landi, G. Umbilical metastasis from ovarian carcinoma: Sister Mary Joseph’s nodule. J. Eur. Acad. Dermatol. Venereol. 2002, 16, 84–85. [Google Scholar] [CrossRef]
- Matsubara, M.; Osada, K.; Ohara, M.; Hondo, T.; Kimura, K. A case of ovarian carcinoma presenting as an umbilical metastasis. Kanto J. Obstet. Gynecol. 2004, 41, 377–382. (In Japanese) [Google Scholar]
- Mizuno, M.; Sumida, K.; Kuno, T.; Miyamoto, O.; Takahashi, Y.; Hirose, S.; Iida, K. A case of ovarian carcinoma with Sister Mary Joseph’s nodule. A case report. Inazawa Munic. Hosp. J. 2006, 10, 74–78. (In Japanese) [Google Scholar]
- Haneda, T.; Kimura, T.; Sarukawa, M.; Horiguchi, D.; Kohda, H.; Takigawa, M. Ovarian carcinoma with Sister Mary Joseph’s nodule. Hifu Rinsho 2001, 42, 342–343. (In Japanese) [Google Scholar]
- Yokota, Y.; Suzuki, K.; Honda, K.; Kase, H.; Kato, M.; Igarashi, T. Ovarian cancer with umbilical metastasis detected by scraping cytology. Niigata Obstet. Gynecol. J. 2014, 109, 68–71. (In Japanese) [Google Scholar]
- Ohta, S.; Yamakawa, Y.; Hasegawa, T.; Tateno, M.; Matsui, K. Advanced ovarian cancer with Sister Mary Joseph’s nodule: A case report. Gan to Kagaku Ryoho 2007, 34, 117–119. (In Japanese) [Google Scholar]
- Kolwijck, E.; Boss, E.A.; van Altena, A.M.; Beex, L.V.; Massuger, L.F. Stage IV epithelial ovarian carcinoma in 18 year old patient presenting with a Sister Mary Joseph’s nodule and metastasis in both breasts: A case report and review of the literature. Gynecol. Oncol. 2007, 107, 583–585. [Google Scholar] [CrossRef]
- Mimura, T.; Yagi, Y.; Asami, Y.; Miyamoto, S.; Iitsuka, C.; Onuki, M.; Ishikawa, T.; Kushima, M.; Matsumoto, K.; Sekizawa, A. Sister Mary Joseph nodule in ovarian cancer: A case report and literature review. Kanto J. Obstet. Gynaecol. 2018, 55, 479–484. (In Japanese) [Google Scholar]
- Haraguchi, K.; Sekine, M.; Aoyama, R.; Hirano, T. Sister Mary Joseph’s nodule presented 10 months before ovarian cancer detection. Hifubyo Rinsho 2007, 29, 837–840. (In Japanese) [Google Scholar]
- Waraich, N.; Mitchell, J.; Holliday, H.; Iftikhar, S.Y. Ovarian cancer presenting as chest wall subcutaneous nodule: A case report and a literature review. Gynecol. Oncol. 2006, 100, 430–432. [Google Scholar] [CrossRef]
- Antonio, A.M.; Alves, J.V.; Goulao, J.; Bartolo, E. Ovarian carcinoma presenting as cutaneous nasal metastasis. An. Bras. Dermatol. 2016, 91, 101–104. [Google Scholar] [CrossRef]
- Rieger, N.; McIntosh, N. Port site metastasis from synchronous primaries of the colon and ovary following laparoscopic cholecystectomy. Eur. J. Surg. Oncol. 1998, 24, 144–154. [Google Scholar] [CrossRef]
- Tsai, H.W.; Yuan, C.C.; Wang, P.H. Umbilicus as the only site of metastasis in recurrent ovarian cancer. J. Chin. Med. Assoc. 2006, 69, 233–235. [Google Scholar] [CrossRef]
- Nikolaou, M.; Zyli, P.; Stamenkovic, S.; Koniari, K.; Psachoulia, C.; Kalofonos, H.P. Sister Mary Joseph’s nodule in advanced ovarian cancer. J. Obstet. Gynaecol. 2013, 33, 214–215. [Google Scholar] [CrossRef]
- Husein-ElAhmed, H.; Aneiros-Fernandez, J.; Arias-Santiago, S.; Naranjo-Sintes, R. Sister Mary Joseph’s nodule as a metastasis of ovarian adenocarcinoma. Int. J. Dermatol. 2010, 49, 1045–1046. [Google Scholar] [CrossRef]
- Kohler, M.F.; Soper, J.T.; Tucker, J.A., Jr.; Clarke-Pearson, D.L. Isolated incisional metastases after intraperitoneal radioactive chromic phosphate therapy for ovarian carcinoma. Cancer 1991, 68, 1380–1383. [Google Scholar] [CrossRef]
- Carlson, N.L.; Krivak, T.C.; Winter, W.E., III; Macri, C.I. Port Site Metastasis of Ovarian Carcinoma Remote from Laparoscopic Surgery for Benign Disease. Gynecol. Oncol. 2002, 85, 529–531. [Google Scholar] [CrossRef]
- Woopen, H.; Pietzner, K.; Darb-Esfahani, S.; Oskay-Oezcelik, G.; Sehouli, J. Extraperitoneal response to intraperitoneal immunotherapy with catumaxomab in a patient with cutaneous lymphangiosis carcinomatosa from ovarian cancer: A case report and review of the literature. Med. Oncol. 2012, 29, 3416–3420. [Google Scholar] [CrossRef]
- Charalampidis, C.; Lampaki, S.; Zarogoulidis, P.; Lazaridis, G.; Mpaka, S.; Kosmidis, C.; Tsakiridis, K.; Kioumis, I.; Pavlidis, P.; Karapantzzos, I.; et al. Fine-needle aspiration of skin metastasis in ovarian cancer-report of two cases and review of the literature. Ann. Transl. Med. 2016, 4, 447. [Google Scholar] [CrossRef]
- Kayikcioglu, F.; Boran, N.; Ayhan, A.; Güler, N. Inflammatory Breast Metastases of Ovarian Cancer: A Case Report. Gynecol. Oncol. 2001, 83, 613–616. [Google Scholar] [CrossRef]
- Yilmaz, Z.; Bese, T.; Demirkiran, F.; Ilvan, S.; Sanioglu, C.; Arvas, M.; Kosebay, D. Skin metastasis in ovarian carcinoma. Int. J. Gynecol. Cancer 2006, 16 (Suppl. 1), 414–418. [Google Scholar] [CrossRef]
- Wiechert, A.C.; Garrett, L.A.; Lin, G.; Goodman, A. Management of a skin metastasis in a patient with advanced ovarian cancer. Gynecol. Oncol. Rep. 2012, 2, 124–126. [Google Scholar] [CrossRef][Green Version]
- Achimas-Cadariu, P.; Vlad, C.; Fetica, B.; Zgaia, A.; Cainap, C.N. Unusual skin metastasis in a patient with recurrent micropapillary serous ovarian carcinoma—A case report and review of the literature. Clujul Med. 2015, 88, 237–240. [Google Scholar] [CrossRef]
- Schonmann, R.; Altaras, M.; Biron, T.; Bernheim, J.; Fishman, A. Inflammatory skin metastases from ovarian carcinoma: A case report and review of the literature. Gynecol. Oncol. 2003, 90, 670–672. [Google Scholar] [CrossRef]
- Yokoyama, T.; Nakanishi, Y.; Yorishima, M.; Tanaka, M.; Chiba, T.; Hiura, M.; Mandai, K.; Moriwaki, S.; Fukui, K. A case of postoperative cutaneous metastasis from ovarian carcinoma with long term survival. Sanka to Fujinka 1989, 56, 491–495. (In Japanese) [Google Scholar]
- Matsui, H.; Suzuka, K.; Yamazawa, K.; Tanaka, N.; Mitsuhashi, A.; Seki, K.; Sekiya, S. Scalp metastasis of a serous ovarian cancer. Acta Obstet. Gynecol. Scand. 2002, 81, 577–578. [Google Scholar] [CrossRef]
- Wuntkal, R.; Maheshwari, A.; Gupta, S.; Ramadwar, M.; Tongaonkar, H.B. Cutaneous metastases in a case of fallopian-tube carcinoma. Lancet Oncol. 2004, 5, 663. [Google Scholar] [CrossRef]
- Lalich, D.; Tawfik, O.; Chapman, J.; Fraga, G. Cutaneous metastasis of ovarian carcinoma with shadow cells mimicking a primary pilomatrical neoplasm. Am. J. Dermatopathol. 2010, 32, 500–504. [Google Scholar] [CrossRef]
- Nakamura, S.; Hashimoto, Y.; Nishi, K.; Mizumoto, T.; Takahashi, H.; Iizuka, H. Cutaneous lymphangitis carcinomatosis metastasis of extra-ovarian primary peritoneal carcinoma. Acta Derm. Venereol. 2012, 92, 640–641. [Google Scholar] [CrossRef]
- Cokmert, S.; Demir, L.; Doganay, L.; Demir, N.; Kocacelebi, K.; Unek, I.T.; Gezer, E.; Kilic, K.; Alakavuklar, M. Large cell neuroendocrine carcinoma of the ovary and its skin metastases: A case report and review of the literature. West Indian Med. J. 2014, 63, 667–672. [Google Scholar]
- Hsiu, J.G.; Given, F.T., Jr.; Kemp, G.M. Tumor implantation after diagnostic laparoscopic biopsy of serous ovarian tumors of low malignant potential. Obstet. Gynecol. 1986, 68, 90S–93S. [Google Scholar]
- Gleeson, N.C.; Nicosia, S.V.; Mark, J.E.; Hoffman, M.S.; Cavanagh, D. Abdominal wall metastases from ovarian cancer after laparoscopy. Am. J. Obstet. Gynecol. 1993, 169, 522–523. [Google Scholar] [CrossRef]
- Shepherd, J.H.; Carter, P.G.; Lower, D. Wound recurrence by implantation of a borderline ovarian tumour following laparoscopic removal. Br. J. Obstet. Gynaecol. 1994, 101, 265–266. [Google Scholar] [CrossRef]
- Morice, P.; Camatte, S.; Larregain-Fournier, D.; Thoury, A.; Duvillard, P.; Castaigne, D. Port-site implantation after laparoscopic treatment of borderline ovarian tumors. Obstet. Gynecol. 2004, 104, 1167–1170. [Google Scholar] [CrossRef]
- Furukawa, N.; Nishioka, K.; Noguchi, T.; Kajihara, H.; Horie, K. Port-Site Metastasis of Mucinous Borderline Ovarian Tumor after Laparoscopy. Case Rep. Oncol. 2014, 7, 804–809. [Google Scholar] [CrossRef]
- Majmudar, B.; Wiskind, A.K.; Croft, B.N.; Dudley, A.G. The Sister (Mary) Joseph nodule: Its significance in gynecology. Gynecol. Oncol. 1991, 40, 152–159. [Google Scholar] [CrossRef]
- Otsuka, I. Cutaneous metastasis after surgery, injury, lymphadenopathy, and peritonitis: Possible mechanisms. Int. J. Mol. Sci. 2019, 20, 3286. [Google Scholar] [CrossRef]
- Childers, J.M.; Aqua, K.A.; Surwit, E.A.; Hallum, A.V.; Hatch, K.D. Abdominal wall tumor implantation following laparoscopy for malignant conditions. Obstet. Gynecol. 1994, 84, 765–769. [Google Scholar]
- Van Dam, P.A.; DeCloedt, J.; Tjalma, W.A.A.; Buytaert, P.; Becquart, D.; Vergote, I.B. Trocar implantation metastasis after laparoscopy in patients with advanced ovarian cancer: Can the risk be reduced? Am. J. Obstet. Gynecol. 1999, 181, 536–541. [Google Scholar] [CrossRef]
- Huang, K.G.; Wang, C.J.; Chang, T.C.; Liou, J.D.; Hsueh, S.; Lai, C.H.; Huang, L.W. Management of port-site metastasis after laparoscopic surgery for ovarian cancer. Am. J. Obstet. Gynecol. 2003, 189, 16–21. [Google Scholar] [CrossRef]
- Ramirez, P.T.; Frumovitz, M.; Wolf, J.K.; Levenback, C. Laparoscopic port-site metastases in patients with gynaecological malignancies. Int. J. Gynecol. Cancer 2004, 14, 1070–1077. [Google Scholar] [CrossRef]
- Vergote, I.; Marquette, S.; Amant, F.; Berteloot, P.; Neven, P. Port-site metastases after open laparoscopy: A study in 173 patients with advanced ovarian carcinoma. Int. J. Gynecol. Cancer 2005, 15, 776–779. [Google Scholar] [CrossRef]
- Ramirez, P.T.; Wolf, J.K.; Levenback, C. Laparoscopic port-site metastases: Etiology and prevention. Gynecol. Oncol. 2003, 91, 179–189. [Google Scholar] [CrossRef]
- Kadar, N. Port-site recurrences following laparoscopic operations for gynaecological malignancies. Br. J. Obstet. Gynaecol. 1997, 104, 1308–1313. [Google Scholar] [CrossRef]
- Plentl, A.A.; Friedman, E.A. Lymphatics System of the Female Genitalia; W.B. Saunders: Philadelphia, PA, USA, 1971; pp. 173–195. [Google Scholar]
- Agrawal, A.; Yau, A.; Magliocco, A.; Chu, P. Cutaneous metastatic disease in cervical cancer: A case report. J. Obstet. Gynaecol. Can. 2010, 32, 467–472. [Google Scholar] [CrossRef]
- Fan, T.; Zhao, Q.; Chen, J.J.; Chen, W.T.; Pearl, M.L. Clinical significance of circulating tumor cells detected by an invasion assay in peripheral blood of patients with ovarian cancer. Gynecol. Oncol. 2009, 112, 185–191. [Google Scholar] [CrossRef]
- Sundaram, G.M.; Quah, S.; Sampath, P. Cancer: The dark side of wound healing. FEBS J. 2018, 285, 4516–4534. [Google Scholar] [CrossRef]
- Qian, B.Z. Inflammation fires up cancer metastasis. Semin. Cancer Biol. 2017, 47, 170–176. [Google Scholar] [CrossRef]
- Mantovani, A.; Marchesi, F.; Malesci, A.; Laghi, L.; Allavena, P. Tumor-Associated Macrophages as Treatment Targets in Oncology. Nat. Rev. Clin. Oncol. 2017, 14, 399–416. [Google Scholar] [CrossRef]
- Yang, L.; Zhang, Y. Tumor-associated macrophages: From basic research to clinical application. J. Hematol. Oncol. 2017, 10, 58. [Google Scholar] [CrossRef]
- Chanmee, T.; Ontong, P.; Konno, K.; Itano, N. Tumor-Associated Macrophages as Major Players in the Tumor Microenvironment. Cancers 2014, 6, 1670–1680. [Google Scholar] [CrossRef]
- Walter, N.D.; Rice, P.L.; Redente, E.F.; Kauvar, E.F.; Lemond, L.; Aly, T.; Wanebo, K.; Chan, E.D. Wound Healing after Trauma May Predispose to Lung Cancer Metastasis. Review of Potential Mechanisms. Am. J. Respir. Cell Mol. Biol. 2011, 44, 591–596. [Google Scholar] [CrossRef]
- Keskin, D.; Kim, J.; Cooke, V.G.; Wu, C.C.; Sugimoto, H.; Gu, C.; De Palma, M.; Kalluri, R.; LeBleu, V.S. Targeting vascular pericytes in hypoxic tumors increases lung metastasis via angiopoietin-2. Cell Rep. 2015, 10, 1066–1081. [Google Scholar] [CrossRef]
- Browning, L.; Patel, M.R.; Horvath, E.B.; Tawara, K.; Jorcyk, C.L. IL-6 and ovarian cancer: Inflammatory cytokines in promotion of metastasis. Cancer Manag. Res. 2018, 10, 6685–6693. [Google Scholar] [CrossRef]
- Bai, W.K.; Zhang, W.; Hu, B. Vascular endothelial growth factor suppresses dendritic cells function of human prostate cancer. Onco Targets Ther. 2018, 11, 1267–1274. [Google Scholar] [CrossRef]
- Terme, M.; Pernot, S.; Marcheteau, E.; Sandoval, F.; Benhamouda, N.; Colussi, O.; Dubreuil, O.; Carpentier, A.F.; Tartour, E.; Taieb, J. VEGFA-VEGFR Pathway Blockade Inhibits Tumor-Induced Regulatory T-cell Proliferation in Colorectal Cancer. Cancer Res. 2013, 73, 539–549. [Google Scholar] [CrossRef]
- Smith, H.A.; Kang, Y. The metastasis-promoting roles of tumor-associated immune cells. J. Mol. Med. 2013, 91, 411–429. [Google Scholar] [CrossRef]
- Nieman, K.M.; Romero, I.L.; Van Houten, B.; Lengyel, E. Adipose tissue and adipocytes supports tumorigenesis and metastasis. Biochim. Biophys. Acta 2013, 181, 1533–1541. [Google Scholar] [CrossRef]
- Varricchi, G.; Ganata, F.; Loffredo, S.; Genovese, A.; Maone, G. Angiogenesis and lymphangiogenesis in inflammatory skin disorders. J. Am. Acad. Dermatol. 2015, 73, 144–153. [Google Scholar] [CrossRef]
- Kurman, R.J.; Shih, I.M. Pathogenesis of ovarian cancer: Lessons from morphology and molecular biology and their clinical implications. Int. J. Gynecol. Pathol. 2008, 27, 151–160. [Google Scholar] [CrossRef]
- Prat, J.; D’Angelo, E.; Espinosa, I. Ovarian carcinomas: At least five different diseases with distinct histological features and molecular genetics. Hum. Pathol. 2018, 80, 11–27. [Google Scholar] [CrossRef]
- Powless, C.A.; Aletti, G.D.; Bakkum-Gamez, J.N.; Cliby, W.A. Risk factors for lymph node metastasis in apparent early-stage epithelial ovarian cancer: Implications for surgical staging. Gynecol. Oncol. 2011, 122, 536–540. [Google Scholar] [CrossRef]
- Vang, R.; Shih, I.M.; Kurman, R.J. Fallopian tube precursors of ovarian low- and high-grade serous neoplasms. Histopathology 2013, 62, 44–58. [Google Scholar] [CrossRef]
- Gershenson, D.M. Low-grade serous carcinoma of the ovary or peritoneum. Ann. Oncol. 2016, 27 (Suppl. 1), i45–i49. [Google Scholar] [CrossRef]
- Sugarbaker, P.H. Sister Mary Joseph’s sign. J. Am. Coll. Surg. 2001, 193, 339–340. [Google Scholar] [CrossRef]
- Wang, P.H.; Yuan, C.C.; Lin, G.; Ng, H.T.; Chao, H.T. Risk factors contributing to early occurrence of port site metastases of laparoscopic surgery for malignancy. Gynecol. Oncol. 1999, 72, 38–44. [Google Scholar] [CrossRef]
- Berek, J.S. Ovarian cancer spread: Is laparoscopy to blame? Lancet 1995, 346, 200. [Google Scholar] [CrossRef]
- Paolucci, V.; Schaeff, B.; Schneider, M.; Gutt, C. Tumor seeding following laparoscopy: International survey. World J. Surg. 1999, 23, 989–997. [Google Scholar] [CrossRef]
- Abu-Rustum, N.R.; Rhee, E.H.; Chi, D.S.; Sonoda, Y.; Gemignani, M.; Barakar, R.R. Subcutaneous Tumor Implantation after Laparoscopic Procedures in Women with Malignant Disease. Obstet. Gynecol. 2004, 103, 480–487. [Google Scholar] [CrossRef]
- Nodofor, B.T.; Soliman, P.T.; Schmeler, K.M.; Nick, A.M.; Frumovitz, M.; Ramirez, P.T. Rate of port-site metastasis is uncommon in patients undergoing robotic surgery for gynecological malignancies. Int. J. Gynecol. Cancer 2011, 21, 936–940. [Google Scholar] [CrossRef]
- Robinson, W.R.; Beyer, J.; Griffin, S.; Kanjanavaikoon, P. Extraperitoneal metastases from recurrent ovarian cancer. Int. J. Gynecol. Cancer 2012, 22, 43–46. [Google Scholar] [CrossRef]
- Pavlidis, E.T.; Pavlidis, T.E. Role of bevacizumab in colorectal cancer growth and its adverse effects: A review. World J. Gastroenterol. 2013, 19, 5051–5060. [Google Scholar] [CrossRef]
- Paez-Ribes, M.; Allen, E.; Hudock, J.; Takeda, T.; Okuyama, H.; Vinals, F.; Inoue, M.; Bergers, G.; Hanahan, D.; Casanovas, O. Antiangiogenic therapy elicits malignant progression of tumors to increased local invasion and distant metastasis. Cancer Cell 2009, 15, 220–224. [Google Scholar] [CrossRef]
- Dalton, H.J.; Pradeep, S.; McGuire, M.; Hailemichael, Y.; Shaolin, M.A.; Lyons, Y.; Armaiz-Pena, G.N.; Previs, R.A.; Hansen, J.M.; Rupaimoole, R.; et al. Macrophages Facilitate Resistance to Anti-VEGF Therapy by Altered VEGFR Expression. Clin. Cancer Res. 2017, 23, 7034–7046. [Google Scholar] [CrossRef]
- Cho, U.; Kim, B.; Kim, S.; Han, Y.; Song, Y.S. Pro-inflammatory M1 macrophage enhances metastatic potential of ovarian cancer cells through NF-κB activation. Mol. Carcinog. 2018, 57, 235–242. [Google Scholar] [CrossRef]
- Iyengar, N.M.; Gucalp, A.; Dammenberg, A.J.; Hudis, C.A. Obesity and Cancer Mechanisms: Tumor Microenvironment and Inflammation. J. Clin. Oncol. 2016, 34, 4270–4276. [Google Scholar] [CrossRef]
- Shaw, A.C.; Goldstein, D.R.; Montgomery, R.R. Age-dependent dysregulation of innate immunity. Nat. Rev. Immunol. 2013, 13, 875–887. [Google Scholar] [CrossRef]
- Jackaman, C.; Tomay, F.; Duong, L.; Razak, N.B.A.; Pixley, F.J.; Metharom, P.; Nelson, D.J. Aging and cancer: The role of macrophages and neutrophils. Ageing Res. Rev. 2017, 36, 105–116. [Google Scholar] [CrossRef]
- Pawelec, G. Does patient age influence anti-cancer immunity? Semin. Immunopathol. 2019, 41, 125–131. [Google Scholar] [CrossRef]
- Weyand, C.M.; Goronzy, J.J. Aging of the Immune System. Mechanisms and Therapeutic Targets. Ann. Am. Thorac. Soc. 2016, 13, S422–S428. [Google Scholar] [CrossRef]
- Schwartz, R.A. Cutaneous metastatic disease. J. Am. Acad. Dermatol. 1995, 33, 161–185. [Google Scholar] [CrossRef]
- Lookingbill, D.P.; Spangler, N.; Sexton, F.M. Skin involvement as the presenting sign of internal carcinoma. A retrospective study of 7316 cancer patients. J. Am. Acad. Dermatol. 1990, 22, 19–26. [Google Scholar] [CrossRef]
- Galvan, V.G. Sister Mary Joseph’s nodule. Ann. Intern. Med. 1998, 128, 410. [Google Scholar] [CrossRef]
- Papalas, J.A.; Selim, M.A. Metastatic vs primary malignant neoplasms affecting the umbilicus: Clinicopathologic features of 77 tumors. Ann. Diagn. Pathol. 2011, 15, 237–242. [Google Scholar] [CrossRef]
- Steck, W.D.; Helwig, E.B. Tumors of the umbilicus. Cancer 1965, 18, 907–911. [Google Scholar] [CrossRef]
- Chaffins, M.L.; Altman, D.A.; Balle, M.R.; Lowe, L.; Rodman, O.G. Metastatic umbilical carcinoma. Cutis 1993, 52, 37–39. [Google Scholar]
- Wronski, M.; Klucinski, A.; Krasnodebski, I.W. Sister Mary Joseph nodule:a tip of an iceberg. J. Ultrasound Med. 2014, 33, 531–534. [Google Scholar] [CrossRef]
- Poparic, S.; Prohic, A.; Kuskunovic-Vlahovljak, S.; Guso, E.; Kapetanovic, A. Sister Mary Joseph Nodule in an Ovary Adenocarcinoma. Med. Arch. 2017, 71, 154–157. [Google Scholar] [CrossRef]
- Chernoff, K.A.; Marghoob, A.A.; Larouture, M.E.; Deng, L.; Busam, K.J.; Myskowski, P.L. Dermoscopic findings in cutaneous metastases. JAMA Dermatol. 2014, 150, 429–433. [Google Scholar] [CrossRef]
- Psarras, K.; Symeonidis, N.; Baltatzis, M.; Notopoulos, A.; Nikolaidou, C. Umbilical metastasis as primary manifestation of cancer: A small series and review of the literature. J. Clin. Diagn. Res. 2014, 8, 17–19. [Google Scholar] [CrossRef]
- Bamias, A.; Efstathiou, E.; Vassilakopoulou, M.; Koutsoukou, V.; Papademitrioou, C.; Rodolakis, A.; Mitsibounas, D.; Vlahos, G.; Dimopoulos, M.A. Late relapse of epithelial ovarian cancer: A single institution experience. Eur. J. Gynaecol. Oncol. 2005, 26, 439–442. [Google Scholar]
- Zylberberg, B.; Dormont, D.; Madelenat, P.; Darai, E. Relapse after more than 20 years of follow-up for epithelial ovarian carcinoma. Obstet. Gynecol. 2004, 103, 1082–1084. [Google Scholar] [CrossRef]
- Trope, C.G.; Elstrand, M.B.; Sandstad, B.; Davidson, B.; Oksefjell, H. Neoadjuvant chemotherapy, interval debulking surgery or primary surgery in ovarian carcinoma FIGO stage IV? Eur. J. Cancer 2012, 48, 2146–2154. [Google Scholar] [CrossRef]
- Lee, S.W.; Park, S.M.; Kim, Y.M.; Kim, Y.S.; Choi, E.K.; Kim, D.Y.; Kim, J.H.; Nam, J.H.; Kim, Y.T. Radiation therapy is a treatment to be considered for recurrent epithelial ovarian cancer after chemotherapy. Tumori 2011, 97, 590–595. [Google Scholar] [CrossRef]
- Fujiwara, K.; Suzuki, S.; Yoden, E.; Ishikawa, H.; Imajo, Y.; Kohno, I. Local radiation therapy for localized relapsed or refractory ovarian cancer patients with or without symptoms after chemotherapy. Int. J. Gynecol. Cancer 2002, 12, 250–256. [Google Scholar] [CrossRef]
- Reinhold, R.G.; Lokich, J.J. Electrocoagulation: Palliative surgery to control metastatic cutaneous malignancy. J. Surg. Oncol. 1979, 11, 207–211. [Google Scholar] [CrossRef]
- Nakai, N.; Okuzawa, Y.; Katoh, N. Clinical usefulness of Mohs’ chemosurgery for palliative purposes in patients with cutaneous squamous cell carcinoma with risk factors or without indication for surgery: Three case reports. J. Dermatol. 2015, 42, 405–407. [Google Scholar] [CrossRef]
- Brix, N.; Tiefenthaller, A.; Anders, H.; Belka, C.; Lauber, K. Abscopal, immunological effects of radiotherapy: Narrowing the gap between clinical and preclinical experiences. Immunol. Rev. 2017, 280, 249–279. [Google Scholar] [CrossRef]
- Henriques, L.; Palumbo, M.; Guay, M.P.; Bahoric, B.; Baik, M.; Kavan, P.; Batist, G. Imiquimod in the Treatment of Breast Cancer Skin Metastasis. J. Clin. Oncol. 2014, 32, e22–e25. [Google Scholar] [CrossRef]
- Fossati, M.; Buzzonetti, A.; Monego, G.; Catzola, V.; Scambia, G.; Fattorossi, A.; Battaglia, A. Immunological changes in the ascites of cancer patients after intraperitoneal administration of the bispecific antibody catumaxomab (anti-EpCAM × anti-CD3). Gynecol. Oncol. 2015, 138, 343–351. [Google Scholar] [CrossRef]
- Hamanishi, J.; Mandai, M.; Ikeda, T.; Minami, M.; Kawaguchi, A.; Murayama, T.; Kanai, M.; Mori, Y.; Matsumoto, S.; Chikuma, S.; et al. Safety and Antitumor Activity of Anti–PD-1 Antibody, Nivolumab, in Patients with Platinum-Resistant Ovarian Cancer. J. Clin. Oncol. 2015, 33, 4015–4022. [Google Scholar] [CrossRef]
© 2019 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Otsuka, I. Cutaneous Metastases in Ovarian Cancer. Cancers 2019, 11, 1292. https://doi.org/10.3390/cancers11091292
Otsuka I. Cutaneous Metastases in Ovarian Cancer. Cancers. 2019; 11(9):1292. https://doi.org/10.3390/cancers11091292
Chicago/Turabian StyleOtsuka, Isao. 2019. "Cutaneous Metastases in Ovarian Cancer" Cancers 11, no. 9: 1292. https://doi.org/10.3390/cancers11091292
APA StyleOtsuka, I. (2019). Cutaneous Metastases in Ovarian Cancer. Cancers, 11(9), 1292. https://doi.org/10.3390/cancers11091292



