A Review on Curability of Cancers: More Efforts for Novel Therapeutic Options Are Needed
Abstract
1. Introduction
1.1. Mortality of Cancer Cells Caused by Therapies
1.2. Cancer Staging
1.3. Concepts of Cancer Cure
1.4. Measurable for Outcomes of Cancer Patients
2. Current Cancer Therapies and Their Performances
2.1. The Evolving Role of Surgery
2.2. The Pros and Cons of Radiotherapy
2.3. The Contribution of and Concerns about Chemotherapy
2.4. The Contribution of and Concerns about Targeted Therapy
2.5. The Contribution of and Concerns about Immunotherapy
3. Curability by Cancer Type and Stage
4. Cancer Screenings and Their Pros and Cons
5. A Newly Proposed Broad-Spectrum Anti-Cancer Strategy Based on a Dual Stroma-Targeting Approach: Orchestrating with Liquid Biopsy
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ADCC | antibody-dependent cellular cytotoxicity |
| ALL | acute lymphoid leukemia |
| ALK | anaplastic lymphoma kinase |
| APC | antigen-presenting cell |
| CD | cluster of differentiation |
| CRC | colorectal cancer |
| CT | computed tomography |
| PET | positron emission tomography |
| CTLA-4 | cytotoxic T-lymphocyte-associated protein 4 |
| DFS | disease-free survival |
| DNA | deoxyribonucleic acid |
| EBRT | external beam radiotherapy |
| EGFR | epidermal growth factor receptor |
| EMA | European Medicines Agency |
| GI | gastrointestinal |
| HCC | hepatocellular carcinoma |
| HSCT | hematopoietic stem cell transplantation |
| ICIs | immune checkpoint inhibitors |
| IMRT | intensity-modulated radiation therapy |
| KRAS | Ki-ras2 Kirsten rat sarcoma viral oncogene homolog |
| MHC | major histocompatibility complex |
| MRD | minimal residual disease |
| NA | non-applicable |
| NCCN | national comprehensive cancer network |
| NP | nanoparticles |
| NPC | nasopharyngeal carcinoma |
| NSCLC | non-small cell lung cancer |
| OS | overall survival |
| PD-1 | programmed death 1 |
| PD-L1 | programmed death ligand 1 |
| PDT | photodynamic therapy |
| PFS | progression-free survival |
| PSA | prostate-specific antigen |
| RAI | radioactive iodine |
| RECIST | response evaluation criteria in solid tumours |
| R1 | rhabdomyosarcoma |
| RSR | relative survival rate |
| SBRT | stereotactic body radiation therapy |
| SCLC | small cell lung cancer |
| SEER | surveillance, epidemiology and end results |
| TACE | transcatheter arterial chemoembolization |
| TCR | T cell receptor |
| TKI | tyrosine kinase inhibitor |
| TNM | tumour-node-metastasis |
| VDAs | vascular-disrupting agents |
References
- Hanahan, D.; Weinberg, R.A. Hallmarks of cancer: The next generation. Cell 2011, 144, 646–674. [Google Scholar] [CrossRef] [PubMed]
- Arruebo, M.; Vilaboa, N.; Sáez-Gutierrez, B.; Lambea, J.; Tres, A.; Valladares, M.; González-Fernández, A. Assessment of the evolution of cancer treatment therapies. Cancers 2011, 3, 3279–3330. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.; Zheng, R.; Baade, P.D.; Zhang, S.; Zeng, H.; Bray, F.; Jemal, A.; Yu, X.Q.; He, J. Cancer statistics in China, 2015. CA Cancer J. Clin. 2016, 66, 115–132. [Google Scholar] [CrossRef] [PubMed]
- Barlas, S. The White House Launches a Cancer Moonshot: Despite Funding Questions, the Progress Appears Promising. Pharm. Ther. 2016, 41, 290–295. [Google Scholar]
- Sun, T.; Wang, S.; Sun, H.; Wen, J.; An, G.; Li, J. Improved survival in multiple myeloma, with a diminishing racial gap and a widening socioeconomic status gap over three decades. Leuk. Lymphoma 2018, 59, 49–58. [Google Scholar] [CrossRef]
- Lu, G.; Li, J.; Wang, S.; Pu, J.; Sun, H.; Wei, Z.; Ma, Y.; Wang, J.; Ma, H. The fluctuating incidence, improved survival of patients with breast cancer, and disparities by age, race, and socioeconomic status by decade, 1981-2010. Cancer Manag. Res. 2018, 10, 4899–4914. [Google Scholar] [CrossRef]
- Wang, S.; Sun, H.; Xie, Z.; Li, J.; Hong, G.; Li, D.; Mallampati, S.; Zhou, X.; Zhou, C.; Zhang, H.; et al. Improved survival of patients with hepatocellular carcinoma and disparities by age, race, and socioeconomic status by decade, 1983-2012. Oncotarget 2016, 7, 59820–59833. [Google Scholar] [CrossRef]
- Wang, S.; Tang, J.; Sun, T.; Zheng, X.; Li, J.; Sun, H.; Zhou, X.; Zhou, C.; Zhang, H.; Cheng, Z.; et al. Survival changes in patients with small cell lung cancer and disparities between different sexes, socioeconomic statuses and ages. Sci. Rep. 2017, 7, 1339. [Google Scholar] [CrossRef]
- Wang, S.; Song, R.; Sun, T.; Hou, B.; Hong, G.; Mallampati, S.; Sun, H.; Zhou, X.; Zhou, C.; Zhang, H.; et al. Survival changes in Patients with Synovial Sarcoma, 1983-2012. J. Cancer 2017, 8, 1759–1768. [Google Scholar] [CrossRef]
- Kroemer, G.; Galluzzi, L.; Vandenabeele, P.; Abrams, J.; Alnemri, E.S.; Baehrecke, E.H.; Blagosklonny, M.V.; El-Deiry, W.S.; Golstein, P.; Green, D.R.; et al. Classification of cell death: Recommendations of the Nomenclature Committee on Cell Death 2009. Cell Death Differ. 2009, 16, 3–11. [Google Scholar] [CrossRef] [PubMed]
- Makin, G.; Hickman, J.A. Apoptosis and cancer chemotherapy. Cell Tissue Res. 2000, 301, 143–152. [Google Scholar] [CrossRef] [PubMed]
- Balcer-Kubiczek, E.K. Apoptosis in radiation therapy: A double-edged sword. Exp. Oncol. 2012, 34, 277–285. [Google Scholar] [PubMed]
- Denisenko, T.V.; Sorokina, I.V.; Gogvadze, V.; Zhivotovsky, B. Mitotic catastrophe and cancer drug resistance: A link that must to be broken. Drug Resist. Updates 2016, 24, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Kaidar-Person, O.; Zagar, T.M.; Deal, A.; Moschos, S.J.; Ewend, M.G.; Sasaki-Adams, D.; Lee, C.B.; Collichio, F.A.; Fried, D.; Marks, L.B.; et al. The incidence of radiation necrosis following stereotactic radiotherapy for melanoma brain metastases: The potential impact of immunotherapy. Anti Cancer Drugs 2017, 28, 669–675. [Google Scholar] [CrossRef] [PubMed]
- Samimi, M.A.; Mirkheshti, N.; Pazouki, A. Assessing the percent of necrosis after neoadjuvant chemotherapy with 24hr infusional cisplatin/3 days Doxorubicin intermittent with Ifosfamide-Doxorubicin for osteosarcoma. Int. J. Hematol. Oncol. Stem Cell Res. 2014, 8, 5–8. [Google Scholar] [PubMed]
- Picci, P.; Bacci, G.; Campanacci, M.; Gasparini, M.; Pilotti, S.; Cerasoli, S.; Bertoni, F.; Guerra, A.; Capanna, R.; Albisinni, U.; et al. Histologic evaluation of necrosis in osteosarcoma induced by chemotherapy. Regional mapping of viable and nonviable tumor. Cancer 1985, 56, 1515–1521. [Google Scholar] [CrossRef]
- Varzaneh, F.N.; Pandey, A.; Ghasabeh, M.A.; Shao, N.; Khoshpouri, P.; Pandey, P.; Zarghampour, M.; Fouladi, D.; Liddell, R.; Anders, R.A.; et al. Prediction of post-TACE necrosis of hepatocellular carcinoma usingvolumetric enhancement on MRI and volumetric oil deposition on CT, with pathological correlation. Eur. Radiol. 2018, 28, 3032–3040. [Google Scholar] [CrossRef]
- Sellers, A.H. The clinical classification of malignant tumours: The TNM system. Can. Med. Assoc. J. 1971, 105, 836. [Google Scholar]
- Trakarnsanga, A.; Ithimakin, S.; Weiser, M.R. Treatment of locally advanced rectal cancer: Controversies and questions. World J. Gastroenterol. 2012, 18, 5521–5532. [Google Scholar] [CrossRef]
- Rosenzweig, K.E.; Gomez, J.E. Concurrent Chemotherapy and Radiation Therapy for Inoperable Locally Advanced Non-Small-Cell Lung Cancer. J. Clin. Oncol. 2017, 35, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.J.; Garcia-Aguilar, J. Advances and challenges in treatment of locally advanced rectal cancer. J. Clin. Oncol. 2015, 33, 1797–1808. [Google Scholar] [CrossRef] [PubMed]
- Kurnick, J.E.; Robinson, W.A. Cell kill hypothesis. Arch. Intern. Med. 1972, 130, 790. [Google Scholar] [CrossRef] [PubMed]
- Miller, K.; Abraham, J.H.; Rhodes, L.; Roberts, R. Use of the word ”cure” in oncology. J. Oncol. Pract. 2013, 9, 136–140. [Google Scholar] [CrossRef] [PubMed]
- Trad, D.; Bibani, N.; Sabbah, M.; Elloumi, H.; Gargouri, D.; Ouakaa, A.; Kharrat, J. Known, new and emerging risk factors of hepatocellular carcinoma (review). Presse Med. 2017, 46, 1000–1007. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2015. CA Cancer J. Clin. 2015, 65, 5–29. [Google Scholar] [CrossRef]
- Pazdur, R. Endpoints for assessing drug activity in clinical trials. Oncologist 2008, 13, 19–21. [Google Scholar] [CrossRef]
- Oba, K.; Paoletti, X.; Alberts, S.; Bang, Y.J.; Benedetti, J.; Bleiberg, H.; Catalano, P.; Lordick, F.; Michiels, S.; Morita, S.; et al. Disease-free survival as a surrogate for overall survival in adjuvant trials of gastric cancer: A meta-analysis. J. Natl. Cancer Inst. 2013, 105, 1600–1607. [Google Scholar] [CrossRef]
- Blumenthal, G.M.; Karuri, S.W.; Zhang, H.; Zhang, L.; Khozin, S.; Kazandjian, D.; Tang, S.; Sridhara, R.; Keegan, P.; Pazdur, R. Overall response rate, progression-free survival, and overall survival with targeted and standard therapies in advanced non-small-cell lung cancer: US Food and Drug Administration trial-level and patient-level analyses. J. Clin. Oncol. 2015, 33, 1008–1014. [Google Scholar] [CrossRef]
- Eisenhauer, E.A.; Therasse, P.; Bogaerts, J.; Schwartz, L.H.; Sargent, D.; Ford, R.; Dancey, J.; Arbuck, S.; Gwyther, S.; Mooney, M.; et al. New response evaluation criteria in solid tumours: Revised RECIST guideline (version 1.1). Eur. J. Cancer 2009, 45, 228–247. [Google Scholar] [CrossRef]
- Seymour, L.; Bogaerts, J.; Perrone, A.; Ford, R.; Schwartz, L.H.; Mandrekar, S.; Lin, N.U.; Litiere, S.; Dancey, J.; Chen, A.; et al. iRECIST: Guidelines for response criteria for use in trials testing immunotherapeutics. Lancet Oncol. 2017, 18, 143–152. [Google Scholar] [CrossRef]
- Mariotto, A.B.; Noone, A.-M.; Howlader, N.; Cho, H.; Keel, G.E.; Garshell, J.; Woloshin, S.; Schwartz, L.M. Cancer survival: An overview of measures, uses, and interpretation. J. Natl. Cancer Inst. Monogr. 2014, 2014, 145–186. [Google Scholar] [CrossRef] [PubMed]
- Henson, D.E.; Ries, L.A. The relative survival rate. Cancer 1995, 76, 1687–1688. [Google Scholar] [CrossRef]
- Makkar, N.; Ostrom, Q.T.; Kruchko, C.; Barnholtz-Sloan, J.S. A comparison of relative survival and cause-specific survival methods to measure net survival in cancer populations. Cancer Med. 2018, 7, 4773–4780. [Google Scholar] [CrossRef] [PubMed]
- Mok, T.S.K.; Wu, Y.-L.; Kudaba, I.; Kowalski, D.M.; Cho, B.C.; Turna, H.Z.; Castro, G.; Srimuninnimit, V.; Laktionov, K.K.; Bondarenko, I.; et al. Pembrolizumab versus chemotherapy for previously untreated, PD-L1-expressing, locally advanced or metastatic non-small-cell lung cancer (KEYNOTE-042): A randomised, open-label, controlled, phase 3 trial. Lancet 2019, 393, 1819–1830. [Google Scholar] [CrossRef]
- Armitage, J.O.; Gascoyne, R.D.; Lunning, M.A.; Cavalli, F. Non-Hodgkin lymphoma. Lancet 2017, 390, 298–310. [Google Scholar] [CrossRef]
- Shanbhag, S.; Ambinder, R.F. Hodgkin lymphoma: A review and update on recent progress. CA Cancer J. Clin. 2018, 68, 116–132. [Google Scholar] [CrossRef]
- Hamilton, J.P.; Sato, F.; Greenwald, B.D.; Suntharalingam, M.; Krasna, M.J.; Edelman, M.J.; Doyle, A.; Berki, A.T.; Abraham, J.M.; Mori, Y.; et al. Promoter methylation and response to chemotherapy and radiation in esophageal cancer. Clin. Gastroenterol. Hepatol. 2006, 4, 701–708. [Google Scholar] [CrossRef]
- Sullivan, R.; Alatise, O.I.; Anderson, B.O.; Audisio, R.; Autier, P.; Aggarwal, A.; Balch, C.; Brennan, M.F.; Dare, A.; D’Cruz, A.; et al. Global cancer surgery: Delivering safe, affordable, and timely cancer surgery. Lancet Oncol. 2015, 16, 1193–1224. [Google Scholar] [CrossRef]
- Zeng, Y.K.; Yang, Z.L.; Peng, J.S.; Lin, H.S.; Cai, L. Laparoscopy-assisted versus open distal gastrectomy for early gastric cancer: Evidence from randomized and nonrandomized clinical trials. Ann. Surg. 2012, 256, 39–52. [Google Scholar] [CrossRef]
- Choi, Y.Y.; Bae, J.M.; An, J.Y.; Hyung, W.J.; Noh, S.H. Laparoscopic gastrectomy for advanced gastric cancer: Are the long-term results comparable with conventional open gastrectomy? A systematic review and meta-analysis. J. Surg. Oncol. 2013, 108, 550–556. [Google Scholar] [CrossRef] [PubMed]
- Van Cutsem, E.; Sagaert, X.; Topal, B.; Haustermans, K.; Prenen, H. Gastric cancer. Lancet 2016, 388, 2654–2664. [Google Scholar] [CrossRef]
- Melamed, A.; Margul, D.J.; Chen, L.; Keating, N.L.; del Carmen, M.G.; Yang, J.; Seagle, B.-L.L.; Alexander, A.; Barber, E.L.; Rice, L.W.; et al. Survival after Minimally Invasive Radical Hysterectomy for Early-Stage Cervical Cancer. N. Engl. J. Med. 2018, 379, 1905–1914. [Google Scholar] [CrossRef] [PubMed]
- Strobel, O.; Büchler, M.W. Laparoscopic pancreatoduodenectomy: Safety concerns and no benefits. Lancet Gastroenterol. Hepatol. 2019, 4, 186–187. [Google Scholar] [CrossRef]
- van Hilst, J.; de Rooij, T.; Bosscha, K.; Brinkman, D.J.; van Dieren, S.; Dijkgraaf, M.G.; Gerhards, M.F.; de Hingh, I.H.; Karsten, T.M.; Lips, D.J.; et al. Laparoscopic versus open pancreatoduodenectomy for pancreatic or periampullary tumours (LEOPARD-2): A multicentre, patient-blinded, randomised controlled phase 2/3 trial. Lancet. Gastroenterol. Hepatol. 2019, 4, 199–207. [Google Scholar] [CrossRef]
- Ramirez, P.T.; Frumovitz, M.; Pareja, R.; Lopez, A.; Vieira, M.; Ribeiro, R.; Buda, A.; Yan, X.; Shuzhong, Y.; Chetty, N.; et al. Minimally Invasive versus Abdominal Radical Hysterectomy for Cervical Cancer. N. Engl. J. Med. 2018, 379, 1895–1904. [Google Scholar] [CrossRef]
- Belderbos, T.D.; van Erning, F.N.; de Hingh, I.H.; van Oijen, M.G.; Lemmens, V.E.; Siersema, P.D. Long-term Recurrence-free Survival After Standard Endoscopic Resection Versus Surgical Resection of Submucosal Invasive Colorectal Cancer: A Population-based Study. Clin. Gastroenterol. Hepatol. 2017, 15, 403–411. [Google Scholar] [CrossRef]
- Heo, J.; Jeon, S.W.; Jung, M.K.; Kim, S.K.; Kim, J.; Kim, S. Endoscopic resection as the first-line treatment for early colorectal cancer: Comparison with surgery. Surg. Endosc. 2014, 28, 3435–3442. [Google Scholar] [CrossRef]
- Pyo, J.H.; Lee, H.; Min, B.H.; Lee, J.H.; Choi, M.G.; Lee, J.H.; Sohn, T.S.; Bae, J.M.; Kim, K.M.; Ahn, J.H.; et al. Long-Term Outcome of Endoscopic Resection vs. Surgery for Early Gastric Cancer: A Non-inferiority-Matched Cohort Study. Am.J. Gastroenterol. 2016, 111, 240–249. [Google Scholar] [CrossRef]
- Das, A.; Singh, V.; Fleischer, D.E.; Sharma, V.K. A comparison of endoscopic treatment and surgery in early esophageal cancer: An analysis of surveillance epidemiology and end results data. Am.J. Gastroenterol. 2008, 103, 1340–1345. [Google Scholar] [CrossRef]
- Sheetz, K.H.; Dimick, J.B. Is It Time for Safeguards in the Adoption of Robotic Surgery. JAMA 2019, 321, 1971–1972. [Google Scholar] [CrossRef] [PubMed]
- Administration, U.F.a.D. Caution When Using Robotically-Assisted Surgical Devices in Women’s Health Including Mastectomy and Other Cancer-Related Surgeries: FDA Safety Communication. Available online: https://www.fda.gov/medicaldevices/safety/alertsandnotices/ucm632142.htm (accessed on 7 November 2019).
- Adam, M.A.; Thomas, S.; Youngwirth, L.; Pappas, T.; Roman, S.A.; Sosa, J.A. Defining a Hospital Volume Threshold for Minimally Invasive Pancreaticoduodenectomy in the United States. JAMA Surg. 2017, 152, 336–342. [Google Scholar] [CrossRef] [PubMed]
- Hayn, M.H.; Hussain, A.; Mansour, A.M.; Andrews, P.E.; Carpentier, P.; Castle, E.; Dasgupta, P.; Rimington, P.; Thomas, R.; Khan, S.; et al. The learning curve of robot-assisted radical cystectomy: Results from the International Robotic Cystectomy Consortium. Eur. Urol. 2010, 58, 197–202. [Google Scholar] [CrossRef] [PubMed]
- Bolenz, C.; Freedland, S.J.; Hollenbeck, B.K.; Lotan, Y.; Lowrance, W.T.; Nelson, J.B.; Hu, J.C. Costs of radical prostatectomy for prostate cancer: A systematic review. Eur. Urol. 2014, 65, 316–324. [Google Scholar] [CrossRef]
- Boutros, J.; Bond, M.; Beaudry, P.; Blair, G.K.; Skarsgard, E.D. Case selection in minimally invasive surgical treatment of neuroblastoma. Pediatr. Surg. Int. 2008, 24, 1177–1180. [Google Scholar] [CrossRef]
- Mulier, S.; Mulier, P.; Ni, Y.; Miao, Y.; Dupas, B.; Marchal, G.; De Wever, I.; Michel, L. Complications of radiofrequency coagulation of liver tumours. Br. J. Surg. 2002, 89, 1206–1222. [Google Scholar] [CrossRef]
- Jahangeer, S.; Forde, P.; Soden, D.; Hinchion, J. Review of current thermal ablation treatment for lung cancer and the potential of electrochemotherapy as a means for treatment of lung tumours. Cancer Treat. Rev. 2013, 39, 862–871. [Google Scholar] [CrossRef]
- Ni, Y.; Mulier, S.; Miao, Y.; Michel, L.; Marchal, G. A review of the general aspects of radiofrequency ablation. Abdom. Imaging 2005, 30, 381–400. [Google Scholar] [CrossRef]
- Finn, R.S.; Zhu, A.X.; Farah, W.; Almasri, J.; Zaiem, F.; Prokop, L.J.; Murad, M.H.; Mohammed, K. Therapies for advanced stage hepatocellular carcinoma with macrovascular invasion or metastatic disease: A systematic review and meta-analysis. Hepatology 2018, 67, 422–435. [Google Scholar] [CrossRef]
- Sieghart, W.; Hucke, F.; Peck-Radosavljevic, M. Transarterial chemoembolization: Modalities, indication, and patient selection. J. Hepatol. 2015, 62, 1187–1195. [Google Scholar] [CrossRef]
- Delaney, G.; Jacob, S.; Featherstone, C.; Barton, M. The role of radiotherapy in cancer treatment. Cancer 2005, 104, 1129–1137. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.H.W.; Kuo, M.T. Improving radiotherapy in cancer treatment: Promises and challenges. Oncotarget 2017, 8, 62742–62758. [Google Scholar] [CrossRef] [PubMed]
- Mehta, S.R.; Suhag, V.; Semwal, M.; Sharma, N. Radiotherapy: Basic Concepts and Recent Advances. Med. J. Armed Forces India 2010, 66, 158–162. [Google Scholar] [CrossRef]
- Reference, M.D. Definition for Heavy Ion. Available online: http://www.reference.md/files/D020/mD020450.html (accessed on 7 November 2019).
- Palm, A.; Johansson, K.A. A review of the impact of photon and proton external beam radiotherapy treatment modalities on the dose distribution in field and out-of-field; implications for the long-term morbidity of cancer survivors. Acta Oncol. 2007, 46, 462–473. [Google Scholar] [CrossRef]
- Kokurewicz, K.; Brunetti, E.; Welsh, G.H.; Wiggins, S.M.; Boyd, M.; Sorensen, A.; Chalmers, A.J.; Schettino, G.; Subiel, A.; DesRosiers, C.; et al. Focused very high-energy electron beams as a novel radiotherapy modality for producing high-dose volumetric elements. Sci. Rep. 2019, 9, 10837. [Google Scholar] [CrossRef]
- Higgins, K.A.; O’Connell, K.; Liu, Y.; Gillespie, T.W.; McDonald, M.W.; Pillai, R.N.; Patel, K.R.; Patel, P.R.; Robinson, C.G.; Simone, C.B., 2nd; et al. National Cancer Database Analysis of Proton Versus Photon Radiation Therapy in Non-Small Cell Lung Cancer. Int. J. Radiat. Oncol. Biol. Phys. 2017, 97, 128–137. [Google Scholar] [CrossRef]
- Jermann, M. Particle Therapy Statistics in 2014. Int. J. Part. Ther. 2015, 2, 50–54. [Google Scholar] [CrossRef]
- Group, P.T.C.-O. Particle Therapy Patient Statistics (per End of 2016). Available online: https://www.ptcog.ch/archive/patient_statistics/Patientstatistics-updateDec2016.pdf (accessed on 7 November 2019).
- Mohan, R.; Grosshans, D. Proton therapy-Present and future. Adv. Drug Deliv. Rev. 2017, 109, 26–44. [Google Scholar] [CrossRef]
- Thomas, J.; Ferme, C.; Noordijk, E.M.; Morschhauser, F.; Girinsky, T.; Gaillard, I.; Lugtenburg, P.J.; Andre, M.; Lybeert, M.L.M.; Stamatoullas, A.; et al. Comparison of 36 Gy, 20 Gy, or No Radiation Therapy After 6 Cycles of EBVP Chemotherapy and Complete Remission in Early-Stage Hodgkin Lymphoma Without Risk Factors: Results of the EORT-GELA H9-F Intergroup Randomized Trial. Int. J. Radiat. Oncol. Biol. Phys. 2018, 100, 1133–1145. [Google Scholar] [CrossRef]
- Ramroth, J.; Cutter, D.J.; Darby, S.C.; Higgins, G.S.; McGale, P.; Partridge, M.; Taylor, C.W. Dose and Fractionation in Radiation Therapy of Curative Intent for Non-Small Cell Lung Cancer: Meta-Analysis of Randomized Trials. Int. J. Radiat. Oncol. Biol. Phys. 2016, 96, 736–747. [Google Scholar] [CrossRef]
- Bradley, J.; Thorstad, W.L.; Mutic, S.; Miller, T.R.; Dehdashti, F.; Siegel, B.A.; Bosch, W.; Bertrand, R.J. Impact of FDG-PET on radiation therapy volume delineation in non–small-cell lung cancer. Int. J. Radiat. Oncol. Biol. Phys. 2004, 59, 78–86. [Google Scholar] [CrossRef] [PubMed]
- Keall, P.J.; Nguyen, D.T.; O’Brien, R.; Zhang, P.; Happersett, L.; Bertholet, J.; Poulsen, P.R. Review of Real-Time 3-Dimensional Image Guided Radiation Therapy on Standard-Equipped Cancer Radiation Therapy Systems: Are We at the Tipping Point for the Era of Real-Time Radiation Therapy. Int. J. Radiat. Oncol. Biol. Phys. 2018, 102, 922–931. [Google Scholar] [CrossRef] [PubMed]
- Jimenez, R.B.; Hickey, S.; DePauw, N.; Yeap, B.Y.; Batin, E.; Gadd, M.A.; Specht, M.; Isakoff, S.J.; Smith, B.L.; Liao, E.C.; et al. Phase II Study of Proton Beam Radiation Therapy for Patients With Breast Cancer Requiring Regional Nodal Irradiation. J. Clin. Oncol. 2019, 37, 2778–2785. [Google Scholar] [CrossRef] [PubMed]
- Pan, H.Y.; Jiang, J.; Hoffman, K.E.; Tang, C.; Choi, S.L.; Nguyen, Q.N.; Frank, S.J.; Anscher, M.S.; Shih, Y.T.; Smith, B.D. Comparative Toxicities and Cost of Intensity-Modulated Radiotherapy, Proton Radiation, and Stereotactic Body Radiotherapy Among Younger Men With Prostate Cancer. J. Clin. Oncol. 2018, 36, 1823–1830. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.M.; Carlson, D.J.; Brenner, D.J. The Tumor Radiobiology of SRS and SBRT: Are More Than the 5 Rs Involved. Int. J. Radiat. Oncol. Biol. Phys. 2014, 88, 254–262. [Google Scholar] [CrossRef] [PubMed]
- Chaudhuri, A.A.; Tang, C.; Binkley, M.S.; Jin, M.; Wynne, J.F.; von Eyben, R.; Hara, W.Y.; Trakul, N.; Loo, B.W.; Diehn, M. Stereotactic ablative radiotherapy (SABR) for treatment of central and ultra-central lung tumors. Lung Cancer 2015, 89, 50–56. [Google Scholar] [CrossRef]
- Palma, D.; Daly, M.; Urbanic, J.; Giuliani, M. Stereotactic Radiation for Ultra-Central Lung Tumors: Good Idea, or Ultra-Risky. Int. J. Radiat. Oncol. Biol. Phys. 2019, 103, 788–791. [Google Scholar] [CrossRef]
- Bezjak, A.; Paulus, R.; Gaspar, L.E.; Timmerman, R.D.; Straube, W.L.; Ryan, W.F.; Garces, Y.I.; Pu, A.T.; Singh, A.K.; Videtic, G.M.; et al. Safety and Efficacy of a Five-Fraction Stereotactic Body Radiotherapy Schedule for Centrally Located Non-Small-Cell Lung Cancer: NRG Oncology/RTOG 0813 Trial. J. Clin. Oncol. 2019, 37, 1316–1325. [Google Scholar] [CrossRef]
- Timmerman, R.; McGarry, R.; Yiannoutsos, C.; Papiez, L.; Tudor, K.; DeLuca, J.; Ewing, M.; Abdulrahman, R.; DesRosiers, C.; Williams, M.; et al. Excessive toxicity when treating central tumors in a phase II study of stereotactic body radiation therapy for medically inoperable early-stage lung cancer. J. Clin. Oncol. 2006, 24, 4833–4839. [Google Scholar] [CrossRef]
- Chicas-Sett, R.; Morales-Orue, I.; Castilla-Martinez, J.; Zafra-Martin, J.; Kannemann, A.; Blanco, J.; Lloret, M.; Lara, P.C. Stereotactic Ablative Radiotherapy Combined with Immune Checkpoint Inhibitors Reboots the Immune Response Assisted by Immunotherapy in Metastatic Lung Cancer: A Systematic Review. Int. J. Mol. Sci. 2019, 20. [Google Scholar] [CrossRef]
- Hainfeld, J.F.; Slatkin, D.N.; Smilowitz, H.M. The use of gold nanoparticles to enhance radiotherapy in mice. Phys. Med. Biol. 2004, 49, 309–315. [Google Scholar] [CrossRef] [PubMed]
- Her, S.; Jaffray, D.A.; Allen, C. Gold nanoparticles for applications in cancer radiotherapy: Mechanisms and recent advancements. Adv. Drug Deliv. Rev. 2017, 109, 84–101. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.K.; Seo, S.J.; Kim, H.T.; Kim, K.H.; Chung, M.H.; Kim, K.R.; Ye, S.J. Enhanced proton treatment in mouse tumors through proton irradiated nanoradiator effects on metallic nanoparticles. Phys. Med. Biol. 2012, 57, 8309–8323. [Google Scholar] [CrossRef] [PubMed]
- Shmatov, M.L. Importance of electric fields from ionized nanoparticles for radiation therapy. Phys. Part. Nucl. Lett. 2017, 14, 533–536. [Google Scholar] [CrossRef]
- Chen, Q.; Chen, J.; Yang, Z.; Xu, J.; Xu, L.; Liang, C.; Han, X.; Liu, Z. Nanoparticle-Enhanced Radiotherapy to Trigger Robust Cancer Immunotherapy. Adv. Mater. 2019, 31, 1802228. [Google Scholar] [CrossRef]
- Chen, J.; Luo, H.; Liu, Y.; Zhang, W.; Li, H.; Luo, T.; Zhang, K.; Zhao, Y.; Liu, J. Oxygen-Self-Produced Nanoplatform for Relieving Hypoxia and Breaking Resistance to Sonodynamic Treatment of Pancreatic Cancer. Acs Nano 2017, 11, 12849–12862. [Google Scholar] [CrossRef]
- Bonvalot, S.; Le Pechoux, C.; De Baere, T.; Kantor, G.; Buy, X.; Stoeckle, E.; Terrier, P.; Sargos, P.; Coindre, J.M.; Lassau, N.; et al. First-in-Human Study Testing a New Radioenhancer Using Nanoparticles (NBTXR3) Activated by Radiation Therapy in Patients with Locally Advanced Soft Tissue Sarcomas. Clin. Cancer Res. 2017, 23, 908–917. [Google Scholar] [CrossRef]
- Kotb, S.; Detappe, A.; Lux, F.; Appaix, F.; Barbier, E.L.; Tran, V.L.; Plissonneau, M.; Gehan, H.; Lefranc, F.; Rodriguez-Lafrasse, C.; et al. Gadolinium-Based Nanoparticles and Radiation Therapy for Multiple Brain Melanoma Metastases: Proof of Concept before Phase I Trial. Theranostics 2016, 6, 418–427. [Google Scholar] [CrossRef]
- Lux, F.; Tran, V.L.; Thomas, E.; Dufort, S.; Rossetti, F.; Martini, M.; Truillet, C.; Doussineau, T.; Bort, G.; Denat, F.; et al. AGuIX® from bench to bedside—Transfer of an ultrasmall theranostic gadolinium-based nanoparticle to clinical medicine. Br. J. Radiol. 2018, 92, 20180365. [Google Scholar] [CrossRef]
- Verry, C.; Sancey, L.; Dufort, S.; Le Duc, G.; Mendoza, C.; Lux, F.; Grand, S.; Arnaud, J.; Quesada, J.L.; Villa, J.; et al. Treatment of multiple brain metastases using gadolinium nanoparticles and radiotherapy: NANO-RAD, a phase I study protocol. BMJ Open 2019, 9, 023591. [Google Scholar] [CrossRef]
- Bonvalot, S.; Rutkowski, P.L.; Thariat, J.; Carrère, S.; Ducassou, A.; Sunyach, M.-P.; Agoston, P.; Hong, A.; Mervoyer, A.; Rastrelli, M.; et al. NBTXR3, a first-in-class radioenhancer hafnium oxide nanoparticle, plus radiotherapy versus radiotherapy alone in patients with locally advanced soft-tissue sarcoma (Act.In.Sarc): A multicentre, phase 2–3, randomised, controlled trial. Lancet Oncol. 2019, 20, 1148–1159. [Google Scholar] [CrossRef]
- Haymart, M.R.; Banerjee, M.; Stewart, A.K.; Koenig, R.J.; Birkmeyer, J.D.; Griggs, J.J. Use of radioactive iodine for thyroid cancer. JAMA 2011, 306, 721–728. [Google Scholar] [CrossRef]
- Vapiwala, N.; Glatstein, E. Fighting Prostate Cancer with Radium-223—Not Your Madame’s Isotope. N. Engl. J. Med. 2013, 369, 276–278. [Google Scholar] [CrossRef] [PubMed]
- Morschhauser, F.; Radford, J.; Van Hoof, A.; Vitolo, U.; Soubeyran, P.; Tilly, H.; Huijgens, P.C.; Kolstad, A.; d’Amore, F.; Gonzalez Diaz, M.; et al. Phase III trial of consolidation therapy with yttrium-90-ibritumomab tiuxetan compared with no additional therapy after first remission in advanced follicular lymphoma. J. Clin. Oncol. 2008, 26, 5156–5164. [Google Scholar] [CrossRef]
- Chargari, C.; Deutsch, E.; Blanchard, P.; Gouy, S.; Martelli, H.; Guerin, F.; Dumas, I.; Bossi, A.; Morice, P.; Viswanathan, A.N.; et al. Brachytherapy: An overview for clinicians. CA Cancer J. Clin. 2019, 69, 386–401. [Google Scholar] [CrossRef]
- Pouliot, J.; Beaulieu, L. 13-Modern Principles of Brachytherapy Physics: From 2-D to 3-D to Dynamic Planning and Delivery. In Leibel and Phillips Textbook of Radiation Oncology, 3rd ed.; Hoppe, R.T., Phillips, T.L., Roach, M., Eds.; W.B. Saunders: Philadelphia, PA, USA, 2010; pp. 224–244. [Google Scholar] [CrossRef]
- Harrison, L.B.; Shasha, D.; White, C.; Ramdeen, B. Radiotherapy-associated anemia: The scope of the problem. Oncologist 2000, 5, 1–7. [Google Scholar] [CrossRef]
- Stacey, R.; Green, J.T. Radiation-induced small bowel disease: Latest developments and clinical guidance. Ther. Adv. Chronic Dis. 2014, 5, 15–29. [Google Scholar] [CrossRef]
- Ali, S.Y.; Singh, G. Radiation-induced Alopecia. Int. J. Trichol. 2010, 2, 118–119. [Google Scholar] [CrossRef]
- Berrington de Gonzalez, A.; Curtis, R.E.; Kry, S.F.; Gilbert, E.; Lamart, S.; Berg, C.D.; Stovall, M.; Ron, E. Proportion of second cancers attributable to radiotherapy treatment in adults: A cohort study in the US SEER cancer registries. Lancet. Oncol. 2011, 12, 353–360. [Google Scholar] [CrossRef]
- Wissing, M.D.; Azoulay, L. Postoperative Pelvic Radiotherapy in Patients With Endometrial Cancer May Increase the Risk for Secondary Pelvic Cancers: A Post Hoc Analysis of Results From the TME, PORTEC-1, and PORTEC-2 Trials. J. Clin. Oncol. 2017, 35, 1861–1862. [Google Scholar] [CrossRef]
- Wallis, C.J.; Mahar, A.L.; Choo, R.; Herschorn, S.; Kodama, R.T.; Shah, P.S.; Danjoux, C.; Narod, S.A.; Nam, R.K. Second malignancies after radiotherapy for prostate cancer: Systematic review and meta-analysis. BMJ 2016, 352, i851. [Google Scholar] [CrossRef] [PubMed]
- Horwich, A.; Fossa, S.D.; Huddart, R.; Dearnaley, D.P.; Stenning, S.; Aresu, M.; Bliss, J.M.; Hall, E. Second cancer risk and mortality in men treated with radiotherapy for stage I seminoma. Br. J. Cancer 2014, 110, 256–263. [Google Scholar] [CrossRef] [PubMed]
- De Sanctis, V.; Alfo, M.; Di Rocco, A.; Ansuinelli, M.; Russo, E.; Osti, M.F.; Valeriani, M.; Minniti, G.; Grapulin, L.; Musio, D.; et al. Second cancer incidence in primary mediastinal B-cell lymphoma treated with methotrexate with leucovorin rescue, doxorubicin, cyclophosphamide, vincristine, prednisone, and bleomycin regimen with or without rituximab and mediastinal radiotherapy: Results from a monoinstitutional cohort analysis of long-term survivors. Hematol. Oncol. 2017, 35, 554–560. [Google Scholar] [CrossRef] [PubMed]
- Teng, C.J.; Hu, Y.W.; Chen, S.C.; Yeh, C.M.; Chiang, H.L.; Chen, T.J.; Liu, C.J. Use of Radioactive Iodine for Thyroid Cancer and Risk of Second Primary Malignancy: A Nationwide Population-Based Study. J. Natl. Cancer Inst. 2016, 108. [Google Scholar] [CrossRef] [PubMed]
- Molenaar, R.J.; Sidana, S.; Radivoyevitch, T.; Advani, A.S.; Gerds, A.T.; Carraway, H.E.; Angelini, D.; Kalaycio, M.; Nazha, A.; Adelstein, D.J.; et al. Risk of Hematologic Malignancies After Radioiodine Treatment of Well-Differentiated Thyroid Cancer. Surgery 2017, 76, 410. [Google Scholar] [CrossRef]
- Morgan, G.; Ward, R.; Barton, M. The contribution of cytotoxic chemotherapy to 5-year survival in adult malignancies. Clin. Oncol. 2004, 16, 549–560. [Google Scholar] [CrossRef]
- Lawrie, T.A.; Winter-Roach, B.A.; Heus, P.; Kitchener, H.C. Adjuvant (post-surgery) chemotherapy for early stage epithelial ovarian cancer. Cochrane Database Syst. Rev. 2015, 12. [Google Scholar] [CrossRef]
- Ando, N.; Iizuka, T.; Ide, H.; Ishida, K.; Shinoda, M.; Nishimaki, T.; Takiyama, W.; Watanabe, H.; Isono, K.; Aoyama, N.; et al. Surgery plus chemotherapy compared with surgery alone for localized squamous cell carcinoma of the thoracic esophagus: A Japan Clinical Oncology Group Study--JCOG9204. J. Clin. Oncol. 2003, 21, 4592–4596. [Google Scholar] [CrossRef]
- Hu, X.; Li, Y.Q.; Li, Q.G.; Ma, Y.L.; Peng, J.J.; Cai, S.J. Adjuvant Chemotherapy Seemed Not to Have Survival Benefit in Rectal Cancer Patients with ypTis-2N0 After Preoperative Radiotherapy and Surgery from a Population-Based Propensity Score Analysis. Oncologist 2019, 24, 803–811. [Google Scholar] [CrossRef]
- Ashdown, M.L.; Robinson, A.P.; Yatomi-Clarke, S.L.; Ashdown, M.L.; Allison, A.; Abbott, D.; Markovic, S.N.; Coventry, B.J. Chemotherapy for Late-Stage Cancer Patients: Meta-Analysis of Complete Response Rates. F1000Research 2015, 4, 232. [Google Scholar] [CrossRef]
- Macarulla, T.; Pazo-Cid, R.; Guillén-Ponce, C.; López, R.; Vera, R.; Reboredo, M.; Martin, A.M.; Rivera, F.; Beveridge, R.D.; Casta, A.L.; et al. Phase I/II Trial to Evaluate the Efficacy and Safety of Nanoparticle Albumin-Bound Paclitaxel in Combination With Gemcitabine in Patients With Pancreatic Cancer and an ECOG Performance Status of 2. J. Clin. Oncol. 2019, 37, 230–238. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, D.; Starling, N.; Rao, S.; Iveson, T.; Nicolson, M.; Coxon, F.; Middleton, G.; Daniel, F.; Oates, J.; Norman, A.R. Capecitabine and Oxaliplatin for Advanced Esophagogastric Cancer. N. Engl. J. Med. 2008, 358, 36–46. [Google Scholar] [CrossRef] [PubMed]
- Fujitani, K.; Yang, H.-K.; Mizusawa, J.; Kim, Y.-W.; Terashima, M.; Han, S.-U.; Iwasaki, Y.; Hyung, W.J.; Takagane, A.; Park, D.J.; et al. Gastrectomy plus chemotherapy versus chemotherapy alone for advanced gastric cancer with a single non-curable factor (REGATTA): A phase 3, randomised controlled trial. Lancet Oncol. 2016, 17, 309–318. [Google Scholar] [CrossRef]
- Gandhi, L.; Rodríguez-Abreu, D.; Gadgeel, S.; Esteban, E.; Felip, E.; De Angelis, F.; Domine, M.; Clingan, P.; Hochmair, M.J.; Powell, S.F.; et al. Pembrolizumab plus Chemotherapy in Metastatic Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2018, 378, 2078–2092. [Google Scholar] [CrossRef] [PubMed]
- Qin, S.; Bai, Y.; Lim, H.Y.; Thongprasert, S.; Chao, Y.; Fan, J.; Yang, T.S.; Bhudhisawasdi, V.; Kang, W.K.; Zhou, Y.; et al. Randomized, multicenter, open-label study of oxaliplatin plus fluorouracil/leucovorin versus doxorubicin as palliative chemotherapy in patients with advanced hepatocellular carcinoma from Asia. J. Clin. Oncol. 2013, 31, 3501–3508. [Google Scholar] [CrossRef]
- Primrose, J.; Falk, S.; Finch-Jones, M.; Valle, J.; O’Reilly, D.; Siriwardena, A.; Hornbuckle, J.; Peterson, M.; Rees, M.; Iveson, T.; et al. Systemic chemotherapy with or without cetuximab in patients with resectable colorectal liver metastasis: The New EPOC randomised controlled trial. Lancet Oncol. 2014, 15, 601–611. [Google Scholar] [CrossRef]
- Chera, B.S.; Amdur, R.J.; Green, R.; Shen, C.; Gupta, G.; Tan, X.; Knowles, M.; Fried, D.; Hayes, N.; Weiss, J.; et al. Phase II Trial of De-Intensified Chemoradiotherapy for Human Papillomavirus-Associated Oropharyngeal Squamous Cell Carcinoma. J. Clin. Oncol. 2019, 37, 2661–2669. [Google Scholar] [CrossRef]
- Grothey, A.; Sobrero, A.F.; Shields, A.F.; Yoshino, T.; Paul, J.; Taieb, J.; Souglakos, J.; Shi, Q.; Kerr, R.; Labianca, R.; et al. Duration of Adjuvant Chemotherapy for Stage III Colon Cancer. N. Engl. J. Med. 2018, 378, 1177–1188. [Google Scholar] [CrossRef]
- Manjelievskaia, J.; Brown, D.; McGlynn, K.A.; Anderson, W.; Shriver, C.D.; Zhu, K. Chemotherapy Use and Survival Among Young and Middle-Aged Patients With Colon Cancer. JAMA Surg. 2017, 152, 452–459. [Google Scholar] [CrossRef]
- Guarneri, V.; Dieci, M.V.; Bisagni, G.; Frassoldati, A.; Bianchi, G.V.; De Salvo, G.L.; Orvieto, E.; Urso, L.; Pascual, T.; Pare, L.; et al. De-escalated therapy for HR+/HER2+ breast cancer patients with Ki67 response after 2 weeks letrozole: Results of the PerELISA neoadjuvant study. Ann. Oncol. 2019, 30, 921–926. [Google Scholar] [CrossRef]
- Saunders, N.A.; Simpson, F.; Thompson, E.W.; Hill, M.M.; Endo-Munoz, L.; Leggatt, G.; Minchin, R.F.; Guminski, A. Role of intratumoural heterogeneity in cancer drug resistance: Molecular and clinical perspectives. EMBO Mol. Med. 2012, 4, 675–684. [Google Scholar] [CrossRef]
- Ramalingam, S.; Belani, C. Systemic chemotherapy for advanced non-small cell lung cancer: Recent advances and future directions. Oncologist 2008, 13, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Paik, S. Development and clinical utility of a 21-gene recurrence score prognostic assay in patients with early breast cancer treated with tamoxifen. Oncologist 2007, 12, 631–635. [Google Scholar] [CrossRef]
- Turkington, R.C.; Knight, L.A.; Blayney, J.K.; Secrier, M.; Douglas, R.; Parkes, E.E.; Sutton, E.K.; Stevenson, L.; McManus, D.; Halliday, S.; et al. Immune activation by DNA damage predicts response to chemotherapy and survival in oesophageal adenocarcinoma. Gut 2019. [Google Scholar] [CrossRef] [PubMed]
- Sutton, P.; Evans, J.; Jones, R.; Malik, H.; Vimalachandran, D.; Palmer, D.; Goldring, C.; Kitteringham, N. Proteomic analysis to identify biomarkers in the primary tumour that predict response to neoadjuvant chemotherapy in liver metastases. Lancet 2015, 385, 95. [Google Scholar] [CrossRef]
- Scagliotti, G.V.; Parikh, P.; von Pawel, J.; Biesma, B.; Vansteenkiste, J.; Manegold, C.; Serwatowski, P.; Gatzemeier, U.; Digumarti, R.; Zukin, M.; et al. Phase III study comparing cisplatin plus gemcitabine with cisplatin plus pemetrexed in chemotherapy-naive patients with advanced-stage non-small-cell lung cancer. J. Clin. Oncol. 2008, 26, 3543–3551. [Google Scholar] [CrossRef] [PubMed]
- Zhong, W.Z.; Wang, Q.; Mao, W.M.; Xu, S.T.; Wu, L.; Shen, Y.; Liu, Y.Y.; Chen, C.; Cheng, Y.; Xu, L.; et al. Gefitinib versus vinorelbine plus cisplatin as adjuvant treatment for stage II-IIIA (N1-N2) EGFR-mutant NSCLC (ADJUVANT/CTONG1104): A randomised, open-label, phase 3 study. Lancet Oncol. 2018, 19, 139–148. [Google Scholar] [CrossRef]
- Solomon, B.J.; Mok, T.; Kim, D.W.; Wu, Y.L.; Nakagawa, K.; Mekhail, T.; Felip, E.; Cappuzzo, F.; Paolini, J.; Usari, T.; et al. First-line crizotinib versus chemotherapy in ALK-positive lung cancer. N. Engl. J. Med. 2014, 371, 2167–2177. [Google Scholar] [CrossRef] [PubMed]
- Mok, T.S.; Wu, Y.L.; Thongprasert, S.; Yang, C.H.; Chu, D.T.; Saijo, N.; Sunpaweravong, P.; Han, B.; Margono, B.; Ichinose, Y.; et al. Gefitinib or carboplatin-Paclitaxel in pulmonary adenocarcinoma. N. Engl. J. Med. 2009, 361, 947–957. [Google Scholar] [CrossRef]
- Hellmann, M.D.; Paz-Ares, L.; Bernabe Caro, R.; Zurawski, B.; Kim, S.-W.; Carcereny Costa, E.; Park, K.; Alexandru, A.; Lupinacci, L.; de la Mora Jimenez, E.; et al. Nivolumab plus Ipilimumab in Advanced Non–Small-Cell Lung Cancer. N. Engl. J. Med. 2019. [Google Scholar] [CrossRef]
- Wong, J.R.; Morton, L.M.; Tucker, M.A.; Abramson, D.H.; Seddon, J.M.; Sampson, J.N.; Kleinerman, R.A. Risk of subsequent malignant neoplasms in long-term hereditary retinoblastoma survivors after chemotherapy and radiotherapy. J. Clin. Oncol. 2014, 32, 3284–3290. [Google Scholar] [CrossRef] [PubMed]
- Nurgali, K.; Jagoe, R.T.; Abalo, R. Editorial: Adverse Effects of Cancer Chemotherapy: Anything New to Improve Tolerance and Reduce Sequelae. Front. Pharmacol. 2018, 9, 245. [Google Scholar] [CrossRef] [PubMed]
- Coiffier, B.; Lepage, E.; Brière, J.; Herbrecht, R.; Tilly, H.; Bouabdallah, R.; Morel, P.; Van Den Neste, E.; Salles, G.; Gaulard, P.; et al. CHOP Chemotherapy plus Rituximab Compared with CHOP Alone in Elderly Patients with Diffuse Large-B-Cell Lymphoma. N. Engl. J. Med. 2002, 346, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Bergethon, K.; Shaw, A.T.; Ou, S.H.; Katayama, R.; Lovly, C.M.; McDonald, N.T.; Massion, P.P.; Siwak-Tapp, C.; Gonzalez, A.; Fang, R.; et al. ROS1 rearrangements define a unique molecular class of lung cancers. J. Clin. Oncol. 2012, 30, 863–870. [Google Scholar] [CrossRef]
- Soda, M.; Choi, Y.L.; Enomoto, M.; Takada, S.; Yamashita, Y.; Ishikawa, S.; Fujiwara, S.; Watanabe, H.; Kurashina, K.; Hatanaka, H.; et al. Identification of the transforming EML4-ALK fusion gene in non-small-cell lung cancer. Nature 2007, 448, 561–566. [Google Scholar] [CrossRef]
- Chan, B.A.; Hughes, B.G. Targeted therapy for non-small cell lung cancer: Current standards and the promise of the future. Transl. Lung Cancer Res. 2015, 4, 36–54. [Google Scholar] [CrossRef]
- Mok, T.S.; Wu, Y.L.; Ahn, M.J.; Garassino, M.C.; Kim, H.R.; Ramalingam, S.S.; Shepherd, F.A.; He, Y.; Akamatsu, H.; Theelen, W.S.; et al. Osimertinib or Platinum-Pemetrexed in EGFR T790M-Positive Lung Cancer. N. Engl. J. Med. 2017, 376, 629–640. [Google Scholar] [CrossRef]
- Carmeliet, P.; Jain, R.K. Angiogenesis in cancer and other diseases. Nature 2000, 407, 249–257. [Google Scholar] [CrossRef]
- Qin, S.; Li, A.; Yi, M.; Yu, S.; Zhang, M.; Wu, K. Recent advances on anti-angiogenesis receptor tyrosine kinase inhibitors in cancer therapy. J. Hematol. Oncol. 2019, 12, 27. [Google Scholar] [CrossRef]
- Strecker, T.E.; Odutola, S.O.; Lopez, R.; Cooper, M.S.; Tidmore, J.K.; Charlton-Sevcik, A.K.; Li, L.; MacDonough, M.T.; Hadimani, M.B.; Ghatak, A.; et al. The vascular disrupting activity of OXi8006 in endothelial cells and its phosphate prodrug OXi8007 in breast tumor xenografts. Cancer Lett. 2015, 369, 229–241. [Google Scholar] [CrossRef]
- Monk, B.J.; Sill, M.W.; Walker, J.L.; Darus, C.J.; Sutton, G.; Tewari, K.S.; Martin, L.P.; Schilder, J.M.; Coleman, R.L.; Balkissoon, J.; et al. Randomized Phase II Evaluation of Bevacizumab Versus Bevacizumab Plus Fosbretabulin in Recurrent Ovarian, Tubal, or Peritoneal Carcinoma: An NRG Oncology/Gynecologic Oncology Group Study. J. Clin. Oncol. 2016, 34, 2279–2286. [Google Scholar] [CrossRef] [PubMed]
- Lara, P.N., Jr.; Douillard, J.Y.; Nakagawa, K.; von Pawel, J.; McKeage, M.J.; Albert, I.; Losonczy, G.; Reck, M.; Heo, D.S.; Fan, X.; et al. Randomized phase III placebo-controlled trial of carboplatin and paclitaxel with or without the vascular disrupting agent vadimezan (ASA404) in advanced non-small-cell lung cancer. J. Clin. Oncol. 2011, 29, 2965–2971. [Google Scholar] [CrossRef] [PubMed]
- Rudin, C.M.; Mauer, A.; Smakal, M.; Juergens, R.; Spelda, S.; Wertheim, M.; Coates, A.; McKeegan, E.; Ansell, P.; Zhou, X.; et al. Phase I/II study of pemetrexed with or without ABT-751 in advanced or metastatic non-small-cell lung cancer. J. Clin. Oncol. 2011, 29, 1075–1082. [Google Scholar] [CrossRef]
- Hodi, F.S.; O’Day, S.J.; McDermott, D.F.; Weber, R.W.; Sosman, J.A.; Haanen, J.B.; Gonzalez, R.; Robert, C.; Schadendorf, D.; Hassel, J.C.; et al. Improved survival with ipilimumab in patients with metastatic melanoma. N. Engl. J. Med. 2010, 363, 711–723. [Google Scholar] [CrossRef] [PubMed]
- Robert, C.; Thomas, L.; Bondarenko, I.; O’Day, S.; Weber, J.; Garbe, C.; Lebbe, C.; Baurain, J.-F.; Testori, A.; Grob, J.-J.; et al. Ipilimumab plus Dacarbazine for Previously Untreated Metastatic Melanoma. N. Engl. J. Med. 2011, 364, 2517–2526. [Google Scholar] [CrossRef]
- Schadendorf, D.; Hodi, F.S.; Robert, C.; Weber, J.S.; Margolin, K.; Hamid, O.; Patt, D.; Chen, T.T.; Berman, D.M.; Wolchok, J.D. Pooled Analysis of Long-Term Survival Data From Phase II and Phase III Trials of Ipilimumab in Unresectable or Metastatic Melanoma. J. Clin. Oncol. 2015, 33, 1889–1894. [Google Scholar] [CrossRef] [PubMed]
- Eroglu, Z.; Kim, D.W.; Wang, X.; Camacho, L.H.; Chmielowski, B.; Seja, E.; Villanueva, A.; Ruchalski, K.; Glaspy, J.A.; Kim, K.B.; et al. Long term survival with cytotoxic T lymphocyte-associated antigen 4 blockade using tremelimumab. Eur. J. Cancer 2015, 51, 2689–2697. [Google Scholar] [CrossRef]
- Robert, C.; Ribas, A.; Hamid, O.; Daud, A.; Wolchok, J.D.; Joshua, A.M.; Hwu, W.J.; Weber, J.S.; Gangadhar, T.C.; Joseph, R.W.; et al. Durable Complete Response After Discontinuation of Pembrolizumab in Patients With Metastatic Melanoma. J. Clin. Oncol. 2018, 36, 1668–1674. [Google Scholar] [CrossRef]
- Gong, J.; Chehrazi-Raffle, A.; Reddi, S.; Salgia, R. Development of PD-1 and PD-L1 inhibitors as a form of cancer immunotherapy: A comprehensive review of registration trials and future considerations. J. Immunother. Cancer 2018, 6, 8. [Google Scholar] [CrossRef]
- Le, D.T.; Durham, J.N.; Smith, K.N.; Wang, H.; Bartlett, B.R.; Aulakh, L.K.; Lu, S.; Kemberling, H.; Wilt, C.; Luber, B.S.; et al. Mismatch repair deficiency predicts response of solid tumors to PD-1 blockade. Science 2017, 357, 409–413. [Google Scholar] [CrossRef]
- Larkin, J.; Chiarion-Sileni, V.; Gonzalez, R.; Grob, J.-J.; Rutkowski, P.; Lao, C.D.; Cowey, C.L.; Schadendorf, D.; Wagstaff, J.; Dummer, R.; et al. Five-Year Survival with Combined Nivolumab and Ipilimumab in Advanced Melanoma. N. Engl. J. Med. 2019, 381, 1535–1546. [Google Scholar] [CrossRef] [PubMed]
- Andrews, A. Treating with Checkpoint Inhibitors-Figure $1 Million per Patient. Am. Health Drug Benefits 2015, 8, 9. [Google Scholar] [PubMed]
- Bianco, A.; Perrotta, F.; Barra, G.; Malapelle, U.; Rocco, D.; De Palma, R. Prognostic Factors and Biomarkers of Responses to Immune Checkpoint Inhibitors in Lung Cancer. Int. J. Mol. Sci. 2019, 20, 4931. [Google Scholar] [CrossRef] [PubMed]
- Havel, J.J.; Chowell, D.; Chan, T.A. The evolving landscape of biomarkers for checkpoint inhibitor immunotherapy. Nat. Rev. Cancer 2019, 19, 133–150. [Google Scholar] [CrossRef]
- Pinato, D.J.; Howlett, S.; Ottaviani, D.; Urus, H.; Patel, A.; Mineo, T.; Brock, C.; Power, D.; Hatcher, O.; Falconer, A.; et al. Association of Prior Antibiotic Treatment With Survival and Response to Immune Checkpoint Inhibitor Therapy in Patients With Cancer. JAMA Oncol. 2019. [Google Scholar] [CrossRef]
- Champiat, S.; Dercle, L.; Ammari, S.; Massard, C.; Hollebecque, A.; Postel-Vinay, S.; Chaput, N.; Eggermont, A.; Marabelle, A.; Soria, J.C.; et al. Hyperprogressive Disease Is a New Pattern of Progression in Cancer Patients Treated by Anti-PD-1/PD-L1. Clin. Cancer Res. 2017, 23, 1920–1928. [Google Scholar] [CrossRef]
- Wang, D.Y.; Salem, J.E.; Cohen, J.V.; Chandra, S.; Menzer, C.; Ye, F.; Zhao, S.; Das, S.; Beckermann, K.E.; Ha, L.; et al. Fatal Toxic Effects Associated With Immune Checkpoint Inhibitors: A Systematic Review and Meta-analysis. JAMA Oncol. 2018, 4, 1721–1728. [Google Scholar] [CrossRef]
- Albano, D.; Bilfinger, T.; Nemesure, B. 1-, 3-, and 5-year survival among early-stage lung cancer patients treated with lobectomy vs SBRT. Lung Cancer 2018, 9, 65–71. [Google Scholar] [CrossRef]
- Ma, K.W.; Cheung, T.T. Surgical resection of localized hepatocellular carcinoma: Patient selection and special consideration. J. Hepatocell. Carcinoma 2017, 4, 1–9. [Google Scholar] [CrossRef]
- Takagi, T.; Kondo, T.; Iizuka, J.; Omae, K.; Kobayashi, H.; Yoshida, K.; Hashimoto, Y.; Tanabe, K. Comparison of survival rates in stage 1 renal cell carcinoma between partial nephrectomy and radical nephrectomy patients according to age distribution: A propensity score matching study. BJU Int. 2016, 117, 52–59. [Google Scholar] [CrossRef]
- Maca, J.; Peteja, M.; Reimer, P.; Jor, O.; Sedenkova, V.; Panackova, L.; Ihnat, P.; Burda, M.; Sevcik, P. Surgical injury: Comparing open surgery and laparoscopy by markers of tissue damage. Ther. Clin. Risk Manag. 2018, 14, 999–1006. [Google Scholar] [CrossRef] [PubMed]
- Tohme, S.; Simmons, R.L.; Tsung, A. Surgery for Cancer: A Trigger for Metastases. Cancer Res. 2017, 77, 1548–1552. [Google Scholar] [CrossRef] [PubMed]
- Lai, E.C.; Yang, G.P.; Tang, C.N. Robot-assisted laparoscopic liver resection for hepatocellular carcinoma: Short-term outcome. Am. J. Surg. 2013, 205, 697–702. [Google Scholar] [CrossRef]
- Coughlin, G.D.; Yaxley, J.W.; Chambers, S.K.; Occhipinti, S.; Samaratunga, H.; Zajdlewicz, L.; Teloken, P.; Dunglison, N.; Williams, S.; Lavin, M.F.; et al. Robot-assisted laparoscopic prostatectomy versus open radical retropubic prostatectomy: 24-month outcomes from a randomised controlled study. Lancet Oncol. 2018, 19, 1051–1060. [Google Scholar] [CrossRef]
- Uedo, N.; Takeuchi, Y.; Ishihara, R. Endoscopic management of early gastric cancer: Endoscopic mucosal resection or endoscopic submucosal dissection: Data from a Japanese high-volume center and literature review. Ann. Gastroenterol. 2012, 25, 281–290. [Google Scholar]
- Ribeiro, M.S.; Wallace, M.B. Endoscopic Treatment of Early Cancer of the Colon. Gastroenterol. Hepatol. 2015, 11, 445–452. [Google Scholar]
- Backes, Y.; de Vos Tot Nederveen Cappel, W.H.; van Bergeijk, J.; Ter Borg, F.; Schwartz, M.P.; Spanier, B.W.M.; Geesing, J.M.J.; Kessels, K.; Kerkhof, M.; Groen, J.N.; et al. Risk for Incomplete Resection after Macroscopic Radical Endoscopic Resection of T1 Colorectal Cancer: A Multicenter Cohort Study. Am. J. Gastroenterol. 2017, 112, 785–796. [Google Scholar] [CrossRef]
- Anderson, M.L.; Pasha, T.M.; Leighton, J.A. Endoscopic perforation of the colon: Lessons from a 10-year study. Am. J. Gastroenterol. 2000, 95, 3418–3422. [Google Scholar] [CrossRef]
- Chen, M.S.; Li, J.Q.; Zheng, Y.; Guo, R.P.; Liang, H.H.; Zhang, Y.Q.; Lin, X.J.; Lau, W.Y. A prospective randomized trial comparing percutaneous local ablative therapy and partial hepatectomy for small hepatocellular carcinoma. Ann. Surg. 2006, 243, 321–328. [Google Scholar] [CrossRef]
- Talenfeld, A.D.; Gennarelli, R.L.; Elkin, E.B.; Atoria, C.L.; Durack, J.C.; Huang, W.C.; Kwan, S.W. Percutaneous Ablation Versus Partial and Radical Nephrectomy for T1a Renal Cancer: A Population-Based Analysis. Ann. Intern. Med. 2018, 169, 69–77. [Google Scholar] [CrossRef]
- Kim, S.R.; Han, H.J.; Park, S.J.; Min, K.H.; Lee, M.H.; Chung, C.R.; Kim, M.H.; Jin, G.Y.; Lee, Y.C. Comparison between surgery and radiofrequency ablation for stage I non-small cell lung cancer. Eur.J. Radiol. 2012, 81, 395–399. [Google Scholar] [CrossRef] [PubMed]
- Yoon, G.S.; Wang, W.; Osunkoya, A.O.; Lane, Z.; Partin, A.W.; Epstein, J.I. Residual tumor potentially left behind after local ablation therapy in prostate adenocarcinoma. J. Urol. 2008, 179, 2203–2206. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Wei, B.; Gao, K.; Zhai, R. Biliary tract perforation following percutaneous endobiliary radiofrequency ablation: A report of two cases. Oncol. Lett. 2016, 11, 3813–3816. [Google Scholar] [CrossRef]
- Fonseca, A.Z.; Santin, S.; Gomes, L.G.; Waisberg, J.; Ribeiro, M.A., Jr. Complications of radiofrequency ablation of hepatic tumors: Frequency and risk factors. World J. Hepatol. 2014, 6, 107–113. [Google Scholar] [CrossRef]
- Lencioni, R.; Petruzzi, P.; Crocetti, L. Chemoembolization of hepatocellular carcinoma. Semin. Interv. Radiol. 2013, 30, 3–11. [Google Scholar] [CrossRef]
- Seam, P.; Janik, J.E.; Longo, D.L.; Devita, V.T., Jr. Role of chemotherapy in Hodgkin’s lymphoma. Cancer J. 2009, 15, 150–154. [Google Scholar] [CrossRef]
- Terwilliger, T.; Abdul-Hay, M. Acute lymphoblastic leukemia: A comprehensive review and 2017 update. Blood Cancer J. 2017, 7, e577. [Google Scholar] [CrossRef]
- Alexander, E.J.; White, I.M.; Horwich, A. Update on management of seminoma. Indian J. Urol. 2010, 26, 82–91. [Google Scholar] [CrossRef]
- Boffetta, P.; Kaldor, J.M. Secondary malignancies following cancer chemotherapy. Acta Oncol. 1994, 33, 591–598. [Google Scholar] [CrossRef]
- Menon, S.P.; Rajkumar, S.V.; Lacy, M.; Falco, P.; Palumbo, A. Thromboembolic events with lenalidomide-based therapy for multiple myeloma. Cancer 2008, 112, 1522–1528. [Google Scholar] [CrossRef]
- Vincent, L.; Kermani, P.; Young, L.M.; Cheng, J.; Zhang, F.; Shido, K.; Lam, G.; Bompais-Vincent, H.; Zhu, Z.; Hicklin, D.J.; et al. Combretastatin A4 phosphate induces rapid regression of tumor neovessels and growth through interference with vascular endothelial-cadherin signaling. J. Clin. Investig. 2005, 115, 2992–3006. [Google Scholar] [CrossRef] [PubMed]
- Zimmermann, M.; Oehler, C.; Mey, U.; Ghadjar, P.; Zwahlen, D.R. Radiotherapy for Non-Hodgkin’s lymphoma: Still standard practice and not an outdated treatment option. Radiat. Oncol. 2016, 11, 110. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.P.; Chan, A.T.C.; Le, Q.T.; Blanchard, P.; Sun, Y.; Ma, J. Nasopharyngeal carcinoma. Lancet 2019, 394, 64–80. [Google Scholar] [CrossRef]
- Newhauser, W.D.; Berrington de Gonzalez, A.; Schulte, R.; Lee, C. A Review of Radiotherapy-Induced Late Effects Research after Advanced Technology Treatments. Front. Oncol. 2016, 6, 13. [Google Scholar] [CrossRef]
- Mohamad, O.; Tabuchi, T.; Nitta, Y.; Nomoto, A.; Sato, A.; Kasuya, G.; Makishima, H.; Choy, H.; Yamada, S.; Morishima, T.; et al. Risk of subsequent primary cancers after carbon ion radiotherapy, photon radiotherapy, or surgery for localised prostate cancer: A propensity score-weighted, retrospective, cohort study. Lancet Oncol. 2019, 20, 674–685. [Google Scholar] [CrossRef]
- Florenzano, P.; Guarda, F.J.; Jaimovich, R.; Droppelmann, N.; González, H.; Domínguez, J.M. Radioactive Iodine Administration Is Associated with Persistent Related Symptoms in Patients with Differentiated Thyroid Cancer. Int. J. Endocrinol. 2016, 2016, 2586512. [Google Scholar] [CrossRef]
- Teunissen, J.J.; Kwekkeboom, D.J.; Krenning, E.P. Quality of life in patients with gastroenteropancreatic tumors treated with [177Lu-DOTA0,Tyr3]octreotate. J. Clin. Oncol. 2004, 22, 2724–2729. [Google Scholar] [CrossRef]
- Brabander, T.; van der Zwan, W.A.; Teunissen, J.J.M.; Kam, B.L.R.; Feelders, R.A.; de Herder, W.W.; van Eijck, C.H.J.; Franssen, G.J.H.; Krenning, E.P.; Kwekkeboom, D.J. Long-Term Efficacy, Survival, and Safety of [(177)Lu-DOTA(0),Tyr(3)]octreotate in Patients with Gastroenteropancreatic and Bronchial Neuroendocrine Tumors. Clin. Cancer Res. 2017, 23, 4617–4624. [Google Scholar] [CrossRef]
- Logghe, P.; Verlinde, R.; Bouttens, F.; Van den Broecke, C.; Deman, N.; Verboven, K.; Maes, D.; Merckx, L. Long term outcome and side effects in patients receiving low-dose I125 brachytherapy: A retrospective analysis. Int. Braz. J. Urol. 2016, 42, 906–917. [Google Scholar] [CrossRef]
- Lönn, S.; Gilbert, E.S.; Ron, E.; Smith, S.A.; Stovall, M.; Curtis, R.E. Comparison of second cancer risks from brachytherapy and external beam therapy after uterine corpus cancer. Cancer Epidemiol. Biomark. Prev. 2010, 19, 464–474. [Google Scholar] [CrossRef]
- Groenendijk, F.H.; Bernards, R. Drug resistance to targeted therapies: Deja vu all over again. Mol. Oncol. 2014, 8, 1067–1083. [Google Scholar] [CrossRef] [PubMed]
- Shih, Y.C.; Smieliauskas, F.; Geynisman, D.M.; Kelly, R.J.; Smith, T.J. Trends in the Cost and Use of Targeted Cancer Therapies for the Privately Insured Nonelderly: 2001 to 2011. J. Clin. Oncol. 2015, 33, 2190–2196. [Google Scholar] [CrossRef] [PubMed]
- Zarrin, B.; Zarifi, F.; Vaseghi, G.; Javanmard, S.H. Acquired tumor resistance to antiangiogenic therapy: Mechanisms at a glance. J. Res. Med. Sci. 2017, 22, 117. [Google Scholar] [CrossRef]
- Venook, A.P.; Niedzwiecki, D.; Lenz, H.J.; Innocenti, F.; Fruth, B.; Meyerhardt, J.A.; Schrag, D.; Greene, C.; O’Neil, B.H.; Atkins, J.N.; et al. Effect of First-Line Chemotherapy Combined With Cetuximab or Bevacizumab on Overall Survival in Patients With KRAS Wild-Type Advanced or Metastatic Colorectal Cancer: A Randomized Clinical Trial. JAMA 2017, 317, 2392–2401. [Google Scholar] [CrossRef]
- Choi, Y.I.; Lee, S.H.; Ahn, B.K.; Baek, S.U.; Park, S.J.; Kim, Y.S.; Shin, S.H. Intestinal perforation in colorectal cancers treated with bevacizumab (Avastin). Cancer Res. Treat. 2008, 40, 33–35. [Google Scholar] [CrossRef]
- Johnson, D.H.; Fehrenbacher, L.; Novotny, W.F.; Herbst, R.S.; Nemunaitis, J.J.; Jablons, D.M.; Langer, C.J.; DeVore, R.F., 3rd; Gaudreault, J.; Damico, L.A.; et al. Randomized phase II trial comparing bevacizumab plus carboplatin and paclitaxel with carboplatin and paclitaxel alone in previously untreated locally advanced or metastatic non-small-cell lung cancer. J. Clin. Oncol. 2004, 22, 2184–2191. [Google Scholar] [CrossRef]
- Haslam, A.; Prasad, V. Estimation of the Percentage of US Patients With Cancer Who Are Eligible for and Respond to Checkpoint Inhibitor Immunotherapy Drugs. JAMA Netw. Open 2019, 2, 192535. [Google Scholar] [CrossRef]
- Hartmann, J.; Schüßler-Lenz, M.; Bondanza, A.; Buchholz, C.J. Clinical development of CAR T cells-challenges and opportunities in translating innovative treatment concepts. EMBO Mol. Med. 2017, 9, 1183–1197. [Google Scholar] [CrossRef]
- Hay, A.E.; Cheung, M.C. CAR T-cells: Costs, comparisons, and commentary. J. Med. Econ. 2019, 22, 613–615. [Google Scholar] [CrossRef]
- Santomasso, B.; Bachier, C.; Westin, J.; Rezvani, K.; Shpall, E.J. The Other Side of CAR T-Cell Therapy: Cytokine Release Syndrome, Neurologic Toxicity, and Financial Burden. Am. Soc. Clin. Oncol. Educ. Book 2019, 39, 433–444. [Google Scholar] [CrossRef]
- Martin, P.J.; Counts, G.W., Jr.; Appelbaum, F.R.; Lee, S.J.; Sanders, J.E.; Deeg, H.J.; Flowers, M.E.; Syrjala, K.L.; Hansen, J.A.; Storb, R.F.; et al. Life expectancy in patients surviving more than 5 years after hematopoietic cell transplantation. J. Clin. Oncol. 2010, 28, 1011–1016. [Google Scholar] [CrossRef] [PubMed]
- Biggs, J.C.; Horowitz, M.M.; Gale, R.P.; Ash, R.C.; Atkinson, K.; Helbig, W.; Jacobsen, N.; Phillips, G.L.; Rimm, A.A.; Ringden, O.; et al. Bone marrow transplants may cure patients with acute leukemia never achieving remission with chemotherapy. Blood 1992, 80, 1090–1093. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.R.; Feng, Y.; Gribben, J.G.; Neuberg, D.; Fisher, D.C.; Mauch, P.; Nadler, L.M.; Freedman, A.S. Long-term survival after autologous bone marrow transplantation for follicular lymphoma in first remission. Biol. Blood Marrow Transplant. 2007, 13, 1057–1065. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Mateos, M.K.; O’Brien, T.A.; Oswald, C.; Gabriel, M.; Ziegler, D.S.; Cohn, R.J.; Russell, S.J.; Barbaric, D.; Marshall, G.M.; Trahair, T.N. Transplant-related mortality following allogeneic hematopoeitic stem cell transplantation for pediatric acute lymphoblastic leukemia: 25-year retrospective review. Pediatr. Blood Cancer 2013, 60, 1520–1527. [Google Scholar] [CrossRef] [PubMed]
- de Heredia, C.D.; Moreno, A.; Olive, T.; Iglesias, J.; Ortega, J.J. Role of the intensive care unit in children undergoing bone marrow transplantation with life-threatening complications. Bone Marrow Transplant. 1999, 24, 163–168. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Litwin, M.S.; Tan, H.J. The Diagnosis and Treatment of Prostate Cancer: A Review. JAMA 2017, 317, 2532–2542. [Google Scholar] [CrossRef]
- Waks, A.G.; Winer, E.P. Breast Cancer Treatment: A Review. JAMA 2019, 321, 288–300. [Google Scholar] [CrossRef]
- Marcheselli, R.; Marcheselli, L.; Cortesi, L.; Bari, A.; Cirilli, C.; Pozzi, S.; Ferri, P.; Napolitano, M.; Federico, M.; Sacchi, S. Risk of Second Primary Malignancy in Breast Cancer Survivors: A Nested Population-Based Case-Control Study. J. Breast Cancer 2015, 18, 378–385. [Google Scholar] [CrossRef]
- Surveillance, Epidemiology, and End Results (SEER) Program SEER Stat Database: Incidence-SEER 9 Regs Research Data, Nov 2018 Sub (1975-2016) (Katrina/Rita Population Adjustment) -Linked To County Attributes-Total U.S., 1969-2017 Counties, National Cancer Institute, DCCPS, Surveillance Research Program. Available online: www.seer.cancer.gov (accessed on 7 November 2019).
- Schiffman, J.D.; Fisher, P.G.; Gibbs, P. Early detection of cancer: Past, present, and future. Am. Soc. Clin. Oncol. Educ. Book. 2015, 35, 57–65. [Google Scholar] [CrossRef]
- Apolone, G.; Joppi, R.; Bertele, V.; Garattini, S. Ten years of marketing approvals of anticancer drugs in Europe: Regulatory policy and guidance documents need to find a balance between different pressures. Br. J. Cancer 2005, 93, 504–509. [Google Scholar] [CrossRef][Green Version]
- Fojo, T.; Mailankody, S.; Lo, A. Unintended consequences of expensive cancer therapeutics-the pursuit of marginal indications and a me-too mentality that stifles innovation and creativity: The John Conley Lecture. JAMA Otolaryngol. Head Neck Surg. 2014, 140, 1225–1236. [Google Scholar] [CrossRef] [PubMed]
- Hiom, S.C. Diagnosing cancer earlier: Reviewing the evidence for improving cancer survival. Br. J. Cancer 2015, 112, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Wise, P.H. Cancer drugs, survival, and ethics. BMJ 2016, 355, 5792. [Google Scholar] [CrossRef]
- Goldsmith, S.J. Radioactive Iodine Therapy of Differentiated Thyroid Carcinoma: Redesigning the Paradigm. Mol Imaging Radionucl Ther 2017, 26, 74–79. [Google Scholar] [CrossRef]
- Naci, H.; Davis, C.; Savović, J.; Higgins, J.P.T.; Sterne, J.A.C.; Gyawali, B.; Romo-Sandoval, X.; Handley, N.; Booth, C.M. Design characteristics, risk of bias, and reporting of randomised controlled trials supporting approvals of cancer drugs by European Medicines Agency, 2014–2016: Cross sectional analysis. BMJ 2019, 366, l5221. [Google Scholar] [CrossRef]
- Cramer, J.D.; Burtness, B.; Le, Q.T.; Ferris, R.L. The changing therapeutic landscape of head and neck cancer. Nat. Rev. Clin. Oncol. 2019, 16, 669–683. [Google Scholar] [CrossRef]
- Cabanillas, M.E.; McFadden, D.G.; Durante, C. Thyroid cancer. Lancet 2016, 388, 2783–2795. [Google Scholar] [CrossRef]
- Hirsch, F.R.; Scagliotti, G.V.; Mulshine, J.L.; Kwon, R.; Curran, W.J., Jr.; Wu, Y.L.; Paz-Ares, L. Lung cancer: Current therapies and new targeted treatments. Lancet 2017, 389, 299–311. [Google Scholar] [CrossRef]
- Spencer, K.L.; Kennedy, M.P.T.; Lummis, K.L.; Ellames, D.A.B.; Snee, M.; Brunelli, A.; Franks, K.; Callister, M.E.J. Surgery or radiotherapy for stage I lung cancer? An intention-to-treat analysis. Eur. Respir. J. 2019, 53, 1801568. [Google Scholar] [CrossRef]
- Gazdar, A.F.; Bunn, P.A.; Minna, J.D. Small-cell lung cancer: What we know, what we need to know and the path forward. Nat. Rev. Cancer 2017, 17, 725–737. [Google Scholar] [CrossRef]
- Lagergren, J.; Smyth, E.; Cunningham, D.; Lagergren, P. Oesophageal cancer. Lancet 2017, 390, 2383–2396. [Google Scholar] [CrossRef]
- Kojima, T.; Muro, K.; Francois, E.; Hsu, C.-H.; Moriwaki, T.; Kim, S.-B.; Lee, S.-H.; Bennouna, J.; Kato, K.; Lin, S.; et al. Pembrolizumab versus chemotherapy as second-line therapy for advanced esophageal cancer: Phase III KEYNOTE-181 study. J. Clin. Oncol. 2019, 37, 2. [Google Scholar] [CrossRef]
- Villanueva, A. Hepatocellular Carcinoma. N. Engl. J. Med. 2019, 380, 1450–1462. [Google Scholar] [CrossRef] [PubMed]
- Kamisawa, T.; Wood, L.D.; Itoi, T.; Takaori, K. Pancreatic cancer. Lancet 2016, 388, 73–85. [Google Scholar] [CrossRef]
- Brenner, H.; Kloor, M.; Pox, C.P. Colorectal cancer. Lancet 2014, 383, 1490–1502. [Google Scholar] [CrossRef]
- Shridhar, R.; Shibata, D.; Chan, E.; Thomas, C.R. Anal cancer: Current standards in care and recent changes in practice. CA Cancer J. Clin. 2015, 65, 139–162. [Google Scholar] [CrossRef]
- Capitanio, U.; Montorsi, F. Renal cancer. Lancet 2016, 387, 894–906. [Google Scholar] [CrossRef]
- Kamat, A.M.; Hahn, N.M.; Efstathiou, J.A.; Lerner, S.P.; Malmstrom, P.U.; Choi, W.; Guo, C.C.; Lotan, Y.; Kassouf, W. Bladder cancer. Lancet 2016, 388, 2796–2810. [Google Scholar] [CrossRef]
- Hanna, N.H.; Einhorn, L.H. Testicular cancer--discoveries and updates. N. Engl. J. Med. 2014, 371, 2005–2016. [Google Scholar] [CrossRef]
- Jayson, G.C.; Kohn, E.C.; Kitchener, H.C.; Ledermann, J.A. Ovarian cancer. Lancet 2014, 384, 1376–1388. [Google Scholar] [CrossRef]
- Morice, P.; Leary, A.; Creutzberg, C.; Abu-Rustum, N.; Darai, E. Endometrial cancer. Lancet 2016, 387, 1094–1108. [Google Scholar] [CrossRef]
- Cohen, P.A.; Jhingran, A.; Oaknin, A.; Denny, L. Cervical cancer. Lancet 2019, 393, 169–182. [Google Scholar] [CrossRef]
- Schadendorf, D.; van Akkooi, A.C.J.; Berking, C.; Griewank, K.G.; Gutzmer, R.; Hauschild, A.; Stang, A.; Roesch, A.; Ugurel, S. Melanoma. Lancet 2018, 392, 971–984. [Google Scholar] [CrossRef]
- Hunger, S.P.; Mullighan, C.G. Acute Lymphoblastic Leukemia in Children. N. Engl. J. Med. 2015, 373, 1541–1552. [Google Scholar] [CrossRef]
- Hucks, G.; Rheingold, S.R. The journey to CAR T cell therapy: The pediatric and young adult experience with relapsed or refractory B-ALL. Blood Cancer J. 2019, 9, 1–9. [Google Scholar] [CrossRef]
- Dohner, H.; Weisdorf, D.J.; Bloomfield, C.D. Acute Myeloid Leukemia. N. Engl. J. Med. 2015, 373, 1136–1152. [Google Scholar] [CrossRef]
- Hofmann, S.; Schubert, M.L.; Wang, L.; He, B.; Neuber, B.; Dreger, P.; Muller-Tidow, C.; Schmitt, M. Chimeric Antigen Receptor (CAR) T Cell Therapy in Acute Myeloid Leukemia (AML). J. Clin. Med. 2019, 8, 200. [Google Scholar] [CrossRef]
- Nabhan, C.; Rosen, S.T. Chronic lymphocytic leukemia: A clinical review. JAMA 2014, 312, 2265–2276. [Google Scholar] [CrossRef]
- Apperley, J.F. Chronic myeloid leukaemia. Lancet 2015, 385, 1447–1459. [Google Scholar] [CrossRef]
- Otto, S.J.; Fracheboud, J.; Looman, C.W.N.; Broeders, M.J.M.; Boer, R.; Hendriks, J.H.C.L.; Verbeek, A.L.M.; de Koning, H.J. Initiation of population-based mammography screening in Dutch municipalities and effect on breast-cancer mortality: A systematic review. Lancet 2003, 361, 1411–1417. [Google Scholar] [CrossRef]
- Davies, L.; Welch, H.G. Current thyroid cancer trends in the United States. JAMA Otolaryngol. Head Neck Surg. 2014, 140, 317–322. [Google Scholar] [CrossRef] [PubMed]
- Schoen, R.E.; Pinsky, P.F.; Weissfeld, J.L.; Yokochi, L.A.; Church, T.; Laiyemo, A.O.; Bresalier, R.; Andriole, G.L.; Buys, S.S.; Crawford, E.D.; et al. Colorectal-cancer incidence and mortality with screening flexible sigmoidoscopy. N. Engl. J. Med. 2012, 366, 2345–2357. [Google Scholar] [CrossRef] [PubMed]
- Landy, R.; Pesola, F.; Castanon, A.; Sasieni, P. Impact of cervical screening on cervical cancer mortality: Estimation using stage-specific results from a nested case-control study. Br. J. Cancer 2016, 115, 1140–1146. [Google Scholar] [CrossRef] [PubMed]
- Atkin, W.S.; Edwards, R.; Kralj-Hans, I.; Wooldrage, K.; Hart, A.R.; Northover, J.M.; Parkin, D.M.; Wardle, J.; Duffy, S.W.; Cuzick, J.; et al. Once-only flexible sigmoidoscopy screening in prevention of colorectal cancer: A multicentre randomised controlled trial. Lancet 2010, 375, 1624–1633. [Google Scholar] [CrossRef]
- Welch, H.G.; Woloshin, S.; Schwartz, L.M. Skin biopsy rates and incidence of melanoma: Population based ecological study. BMJ 2005, 331, 481. [Google Scholar] [CrossRef]
- Welch, H.G.; Prorok, P.C.; O’Malley, A.J.; Kramer, B.S. Breast-Cancer Tumor Size, Overdiagnosis, and Mammography Screening Effectiveness. N. Engl. J. Med. 2016, 375, 1438–1447. [Google Scholar] [CrossRef]
- Bleyer, A.; Welch, H.G. Effect of three decades of screening mammography on breast-cancer incidence. N. Engl. J. Med. 2012, 367, 1998–2005. [Google Scholar] [CrossRef]
- Lowenstein, L.M.; Basourakos, S.P.; Williams, M.D.; Troncoso, P.; Gregg, J.R.; Thompson, T.C.; Kim, J. Active surveillance for prostate and thyroid cancers: Evolution in clinical paradigms and lessons learned. Nat. Rev. Clin. Oncol. 2019, 16, 168–184. [Google Scholar] [CrossRef]
- Siu, A.L. Screening for Breast Cancer: U.S. Preventive Services Task Force Recommendation Statement. Ann. Intern. Med. 2016, 164, 279–296. [Google Scholar] [CrossRef]
- Welch, H.G.; Black, W.C. Overdiagnosis in cancer. J. Natl. Cancer Inst. 2010, 102, 605–613. [Google Scholar] [CrossRef]
- Bach, P.B. Reduced lung-cancer mortality with CT screening. N. Engl. J. Med. 2011, 365, 2035–2037. [Google Scholar] [CrossRef]
- Otto, S.J.; Fracheboud, J.; Verbeek, A.L.; Boer, R.; Reijerink-Verheij, J.C.; Otten, J.D.; Broeders, M.J.; de Koning, H.J. Mammography screening and risk of breast cancer death: A population-based case-control study. Cancer Epidemiol. Biomark. Prev. 2012, 21, 66–73. [Google Scholar] [CrossRef]
- Nelson, H.D.; Tyne, K.; Naik, A.; Bougatsos, C.; Chan, B.K.; Humphrey, L. Screening for breast cancer: An update for the U.S. Preventive Services Task Force. Ann. Intern. Med. 2009, 151, 727–737. [Google Scholar] [CrossRef] [PubMed]
- Porter, P.L.; El-Bastawissi, A.Y.; Mandelson, M.T.; Lin, M.G.; Khalid, N.; Watney, E.A.; Cousens, L.; White, D.; Taplin, S.; White, E. Breast tumor characteristics as predictors of mammographic detection: Comparison of interval- and screen-detected cancers. J. Natl. Cancer Inst. 1999, 91, 2020–2028. [Google Scholar] [CrossRef]
- Grossman, D.C.; Curry, S.J.; Owens, D.K.; Bibbins-Domingo, K.; Caughey, A.B.; Davidson, K.W.; Doubeni, C.A.; Ebell, M.; Epling, J.W., Jr.; Kemper, A.R.; et al. Screening for Prostate Cancer: US Preventive Services Task Force Recommendation Statement. JAMA 2018, 319, 1901–1913. [Google Scholar] [CrossRef]
- Thompson, I.M.; Pauler, D.K.; Goodman, P.J.; Tangen, C.M.; Lucia, M.S.; Parnes, H.L.; Minasian, L.M.; Ford, L.G.; Lippman, S.M.; Crawford, E.D.; et al. Prevalence of prostate cancer among men with a prostate-specific antigen level <or =4.0 ng per milliliter. N. Engl. J. Med. 2004, 350, 2239–2246. [Google Scholar] [CrossRef]
- Etzioni, R.; Penson, D.F.; Legler, J.M.; di Tommaso, D.; Boer, R.; Gann, P.H.; Feuer, E.J. Overdiagnosis due to prostate-specific antigen screening: Lessons from U.S. prostate cancer incidence trends. J. Natl. Cancer Inst. 2002, 94, 981–990. [Google Scholar] [CrossRef]
- Carter, H.B.; Piantadosi, S.; Isaacs, J.T. Clinical evidence for and implications of the multistep development of prostate cancer. J. Urol. 1990, 143, 742–746. [Google Scholar] [CrossRef]
- Etzioni, R.; Cha, R.; Feuer, E.J.; Davidov, O. Asymptomatic incidence and duration of prostate cancer. Am. J. Epidemiol. 1998, 148, 775–785. [Google Scholar] [CrossRef][Green Version]
- Pickhardt, P.J.; Kim, D.H.; Pooler, B.D.; Hinshaw, J.L.; Barlow, D.; Jensen, D.; Reichelderfer, M.; Cash, B.D. Assessment of volumetric growth rates of small colorectal polyps with CT colonography: A longitudinal study of natural history. Lancet. Oncol. 2013, 14, 711–720. [Google Scholar] [CrossRef]
- Knudsen, A.B.; Zauber, A.G.; Rutter, C.M.; Naber, S.K.; Doria-Rose, V.P.; Pabiniak, C.; Johanson, C.; Fischer, S.E.; Lansdorp-Vogelaar, I.; Kuntz, K.M. Estimation of Benefits, Burden, and Harms of Colorectal Cancer Screening Strategies: Modeling Study for the US Preventive Services Task Force. JAMA 2016, 315, 2595–2609. [Google Scholar] [CrossRef] [PubMed]
- Bretthauer, M.; Kaminski, M.F.; Loberg, M.; Zauber, A.G.; Regula, J.; Kuipers, E.J.; Hernan, M.A.; McFadden, E.; Sunde, A.; Kalager, M.; et al. Population-Based Colonoscopy Screening for Colorectal Cancer: A Randomized Clinical Trial. JAMA Intern. Med. 2016, 176, 894–902. [Google Scholar] [CrossRef] [PubMed]
- Xu, R.H.; Wei, W.; Krawczyk, M.; Wang, W.; Luo, H.; Flagg, K.; Yi, S.; Shi, W.; Quan, Q.; Li, K.; et al. Circulating tumour DNA methylation markers for diagnosis and prognosis of hepatocellular carcinoma. Nat. Mater. 2017, 16, 1155–1161. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.; Ding, J.; Ma, Z.; Sun, R.; Seoane, J.A.; Scott Shaffer, J.; Suarez, C.J.; Berghoff, A.S.; Cremolini, C.; Falcone, A.; et al. Quantitative evidence for early metastatic seeding in colorectal cancer. Nat. Genet. 2019, 51, 1113–1122. [Google Scholar] [CrossRef]
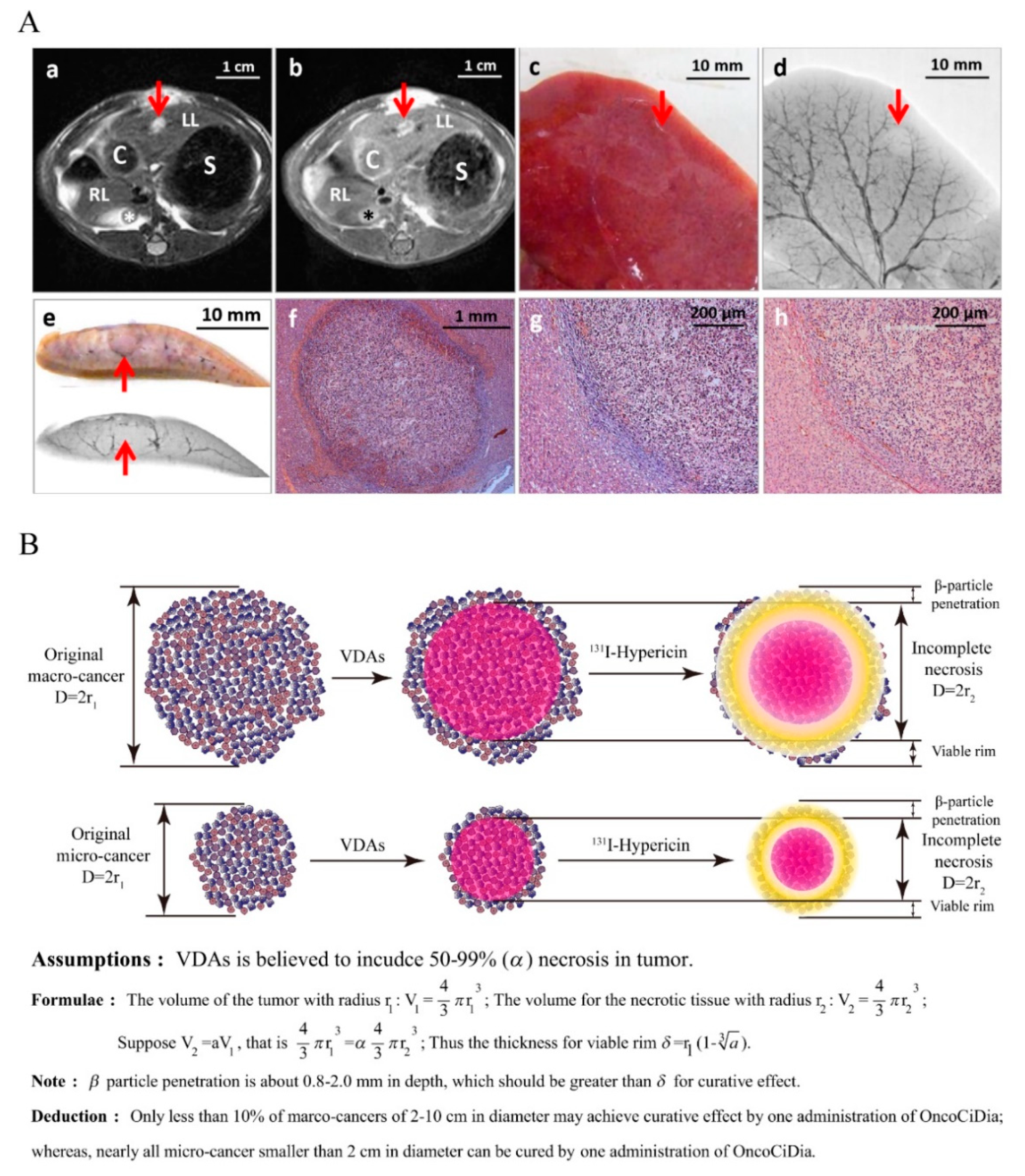
- Li, J.; Sun, Z.; Zhang, J.; Shao, H.; Cona, M.M.; Wang, H.; Marysael, T.; Chen, F.; Prinsen, K.; Zhou, L.; et al. A dual-targeting anticancer approach: Soil and seed principle. Radiology 2011, 260, 799–807. [Google Scholar] [CrossRef]
- Murray, I.; Chittenden, S.J.; Denis-Bacelar, A.M.; Hindorf, C.; Parker, C.C.; Chua, S.; Flux, G.D. The potential of (223)Ra and (18)F-fluoride imaging to predict bone lesion response to treatment with (223)Ra-dichloride in castration-resistant prostate cancer. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 1832–1844. [Google Scholar] [CrossRef]
- Werner, R.A.; Lapa, C.; Ilhan, H.; Higuchi, T.; Buck, A.K.; Lehner, S.; Bartenstein, P.; Bengel, F.; Schatka, I.; Muegge, D.O.; et al. Survival prediction in patients undergoing radionuclide therapy based on intratumoral somatostatin-receptor heterogeneity. Oncotarget 2017, 8, 7039–7049. [Google Scholar] [CrossRef]
- Chang, C.H.; Sharkey, R.M.; Rossi, E.A.; Karacay, H.; McBride, W.; Hansen, H.J.; Chatal, J.F.; Barbet, J.; Goldenberg, D.M. Molecular advances in pretargeting radioimunotherapy with bispecific antibodies. Mol. Cancer Ther. 2002, 1, 553–563. [Google Scholar]
- Milano, M.T.; Constine, L.S.; Okunieff, P. Normal tissue tolerance dose metrics for radiation therapy of major organs. Semin. Radiat. Oncol. 2007, 17, 131–140. [Google Scholar] [CrossRef]
- Michalski, J.M.; Gay, H.; Jackson, A.; Tucker, S.L.; Deasy, J.O. Radiation dose-volume effects in radiation-induced rectal injury. Int. J. Radiat. Oncol. Biol. Phys. 2010, 76, 123–129. [Google Scholar] [CrossRef]
- Dawson, L.A.; Kavanagh, B.D.; Paulino, A.C.; Das, S.K.; Miften, M.; Li, X.A.; Pan, C.; Ten Haken, R.K.; Schultheiss, T.E. Radiation-Associated Kidney Injury. Int. J. Radiat. Oncol. Biol. Phys. 2010, 76, 108–115. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Wang, H.; Jacobson, O.; Cheng, Y.; Niu, G.; Li, F.; Bai, C.; Zhu, Z.; Chen, X. Safety, Pharmacokinetics, and Dosimetry of a Long-Acting Radiolabeled Somatostatin Analog (177)Lu-DOTA-EB-TATE in Patients with Advanced Metastatic Neuroendocrine Tumors. J. Nucl. Med. 2018, 59, 1699–1705. [Google Scholar] [CrossRef] [PubMed]
- Dawson, L.A.; Ten Haken, R.K. Partial volume tolerance of the liver to radiation. Semin. Radiat. Oncol. 2005, 15, 279–283. [Google Scholar] [CrossRef] [PubMed]
- Rolleman, E.J.; Kooij, P.P.; de Herder, W.W.; Valkema, R.; Krenning, E.P.; de Jong, M. Somatostatin receptor subtype 2-mediated uptake of radiolabelled somatostatin analogues in the human kidney. Eur. J. Nucl. Med. Mol. Imaging 2007, 34, 1854–1860. [Google Scholar] [CrossRef] [PubMed]
- Reubi, J.C.; Horisberger, U.; Studer, U.E.; Waser, B.; Laissue, J.A. Human kidney as target for somatostatin: High affinity receptors in tubules and vasa recta. J. Clin. Endocrinol. Metab. 1993, 77, 1323–1328. [Google Scholar] [CrossRef] [PubMed]
- Abma, E. Ugent Is Looking For Dogs With Malignant Tumors. Available online: https://www.dierenartsenwereld.be/nl/nieuws-n2/ugent-zoekt-honden-met-kwaadaardige-tumoren-i171/ (accessed on 7 November 2019).
- OncoCiDia Phase 0 study (3M150468). Available online: https://www.kuleuven.be/onderzoek/portaal/#/projecten/3M150468?hl=en&lang=en (accessed on 7 November 2019).
- Petrelli, A.; Giordano, S. From single- to multi-target drugs in cancer therapy: When aspecificity becomes an advantage. Curr. Med. Chem. 2008, 15, 422–432. [Google Scholar]
- Bang, Y.-J.; Van Cutsem, E.; Feyereislova, A.; Chung, H.C.; Shen, L.; Sawaki, A.; Lordick, F.; Ohtsu, A.; Omuro, Y.; Satoh, T.; et al. Trastuzumab in combination with chemotherapy versus chemotherapy alone for treatment of HER2-positive advanced gastric or gastro-oesophageal junction cancer (ToGA): A phase 3, open-label, randomised controlled trial. Lancet 2010, 376, 687–697. [Google Scholar] [CrossRef]
- Liu, Y.; Yin, T.; Keyzer, F.D.; Feng, Y.; Chen, F.; Liu, J.; Song, S.; Yu, J.; Vandecaveye, V.; Swinnen, J.; et al. Micro-HCCs in rats with liver cirrhosis: Paradoxical targeting effects with vascular disrupting agent CA4P. Oncotarget 2017, 8, 55204–55215. [Google Scholar] [CrossRef]
- Pantel, K.; Alix-Panabieres, C. Liquid biopsy and minimal residual disease-latest advances and implications for cure. Nat. Rev. Clin. Oncol. 2019, 16, 409–424. [Google Scholar] [CrossRef]
- Descotes, F.; Kara, N.; Decaussin-Petrucci, M.; Piaton, E.; Geiguer, F.; Rodriguez-Lafrasse, C.; Terrier, J.E.; Lopez, J.; Ruffion, A. Non-invasive prediction of recurrence in bladder cancer by detecting somatic TERT promoter mutations in urine. Br. J. Cancer 2017, 117, 583. [Google Scholar] [CrossRef]
- Dudley, J.C.; Schroers-Martin, J.; Lazzareschi, D.V.; Shi, W.Y.; Chen, S.B.; Esfahani, M.S.; Trivedi, D.; Chabon, J.J.; Chaudhuri, A.A.; Stehr, H.; et al. Detection and Surveillance of Bladder Cancer Using Urine Tumor DNA. Cancer Discov. 2019, 9, 500–509. [Google Scholar] [CrossRef]
- Pan, H.; Gray, R.; Braybrooke, J.; Davies, C.; Taylor, C.; McGale, P.; Peto, R.; Pritchard, K.I.; Bergh, J.; Dowsett, M.; et al. 20-Year Risks of Breast-Cancer Recurrence after Stopping Endocrine Therapy at 5 Years. N. Engl. J. Med. 2017, 377, 1836–1846. [Google Scholar] [CrossRef]
- Liu, C.; Zhao, J.; Tian, F.; Cai, L.; Zhang, W.; Feng, Q.; Chang, J.; Wan, F.; Yang, Y.; Dai, B.; et al. Low-cost thermophoretic profiling of extracellular-vesicle surface proteins for the early detection and classification of cancers. Nat. Biomed. Eng. 2019, 3, 183–193. [Google Scholar] [CrossRef] [PubMed]
- Sanger, N.; Effenberger, K.E.; Riethdorf, S.; Van Haasteren, V.; Gauwerky, J.; Wiegratz, I.; Strebhardt, K.; Kaufmann, M.; Pantel, K. Disseminated tumor cells in the bone marrow of patients with ductal carcinoma in situ. Int. J. Cancer 2011, 129, 2522–2526. [Google Scholar] [CrossRef] [PubMed]
- Harper, K.L.; Sosa, M.S.; Entenberg, D.; Hosseini, H.; Cheung, J.F.; Nobre, R.; Avivar-Valderas, A.; Nagi, C.; Girnius, N.; Davis, R.J.; et al. Mechanism of early dissemination and metastasis in Her2(+) mammary cancer. Nature 2016, 540, 588–592. [Google Scholar] [CrossRef] [PubMed]
- Hosseini, H.; Obradovic, M.M.S.; Hoffmann, M.; Harper, K.L.; Sosa, M.S.; Werner-Klein, M.; Nanduri, L.K.; Werno, C.; Ehrl, C.; Maneck, M.; et al. Early dissemination seeds metastasis in breast cancer. Nature 2016, 540, 552–558. [Google Scholar] [CrossRef] [PubMed]
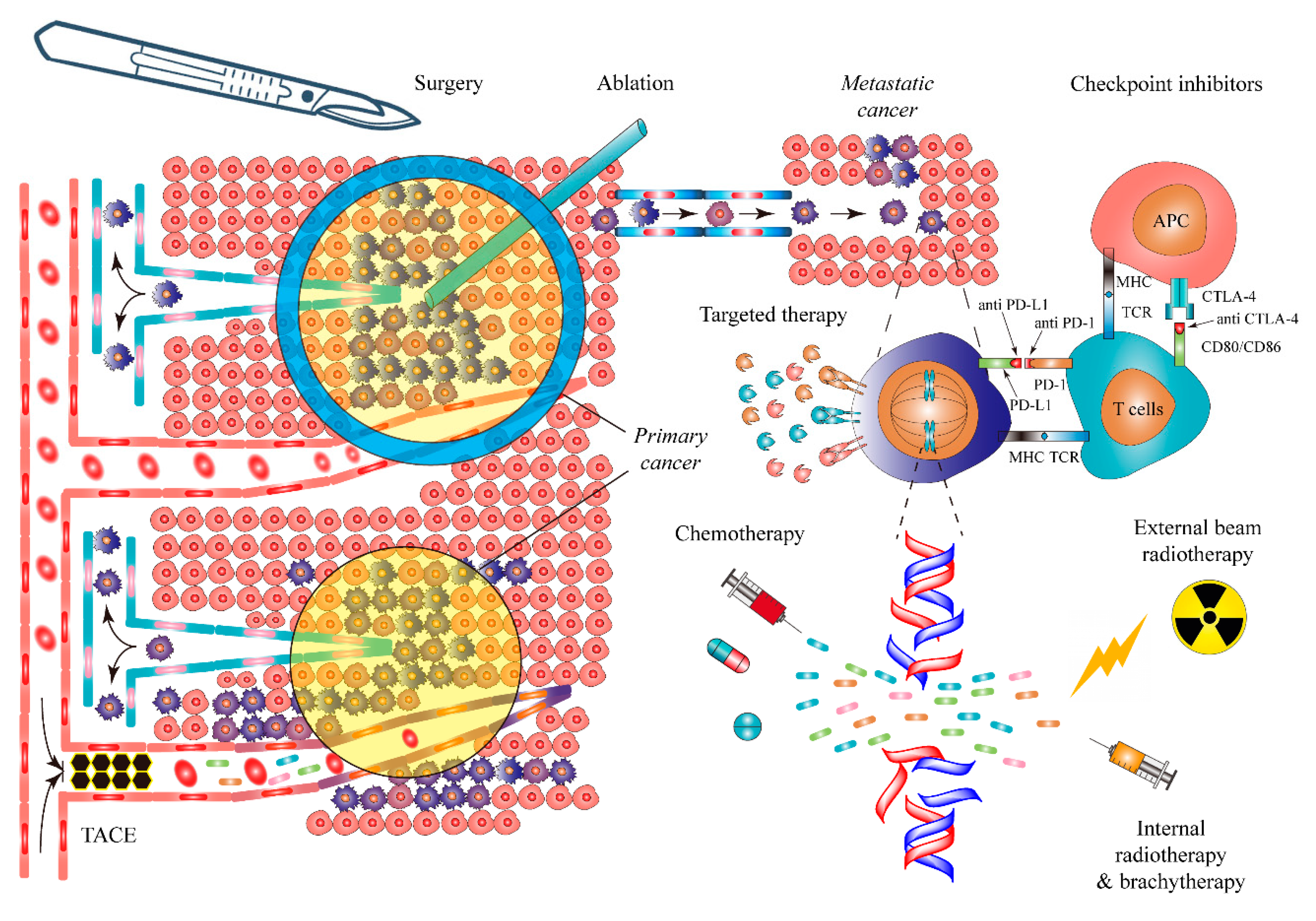

| Cancer Therapy * | Mechanism | Curative Potential | Example | Limitations |
|---|---|---|---|---|
| Surgery | ||||
| Open surgery | Physical removal of cancer, adjacent tissue, and involved lymph nodes | For early solid cancer, +++ | Early NSCLC [162], HCC [163], renal cancer [164] | Surgical injury [165], cancer dissemination [166] |
| Laparoscopic surgery | Same as above | Same as above | Same as above | Surgical injury [165], cancer dissemination [166], dependence of centralized expert surgeons [53] |
| Robotic surgery | Same as above | Same as above | HCC [167], prostate cancer [168] | Same as above, imperfectly confirmed efficacy |
| Endoscopic surgery | Same as above | Same as above | Early GI cancer [169,170] | Possible second surgery [171], perforation [172] |
| Interventions | ||||
| Ablations | In situ necrotizing cancer and adjacent tissue by local hyperthermal ablation, cryotherapy, or absolute ethanol injection. | For early eligible cancer, +++ | HCC [173], renal cancer [174], lung cancer [175] | Often incomplete ablation [176], injury to adjacent tissue [177,178] |
| TACE | Embolization of cancer supplying artery combined with local chemotherapy | −/+ | HCC [179] | Often incomplete cell death [179] |
| Chemotherapy | ||||
| Direct | Alteration of DNA synthesis and structure or cytoskeleton | For chemotherapy sensitive cancer, +++ | Early lymphoma [180], ALL [181], seminoma [182] | Pancytopenia, nausea, infertility, neuropathy [136], secondary cancer [183] |
| Indirect | Immunomodulation, vascular disrupting effect | −/+ | NA | Venous thrombosis [184], recurrence [185] |
| Radiotherapy | ||||
| External beam | Alteration of DNA structure via radicals | For radiotherapy sensitive cancer, +++ | Early lymphoma [186], NPC [187], +++ | Unintentional destruction along entrance channel [188], secondary cancer [189] |
| Radioiodine | Same as above, for thyroid cancer with iodine intake | For thyroid cancer with iodine intake, ++ | Thyroid cancer [108] | Side-effects [190], secondary cancer [109] |
| Radiopharmacy (Lutetium 177) | Same as above | −/+ | NA | Side-effects [191], secondary cancer [192] |
| Brachytherapy | Same as above | −/+ | NA | Side-effects [193], secondary cancer [194] |
| Targeted therapy | ||||
| Direct | Inhibition of signaling pathway, ADCC for monoclonal antibody | −/+ | NA | Acquired resistance [195]; narrow spectrum of optimal patients [196] |
| Indirect | Anti-angiogenesis | −/+ | NA | Acquired resistance [197], low response rate [198], rare but fatal side-effects [198,199,200] |
| Immunotherapy | ||||
| Immune checkpoint inhibitors | Restore anticancer immunity | Only possible in responded cases (about 10%) [201], + | Melanoma [148] | Unpredictability of response [158], rare but fatal side-effects [161] |
| Cellular immunotherapy | Elimination of cancer cells by immune cells with or without engineering | −/+ | Leukemia [202] | High cost [203], severe side-effects [204] |
| Bone marrow transplantation | Elimination of cancer cells by intensive chemotherapy and graft-versus-leukemia effect | Yes, for high-risk haematological cancer [205], +++ | Leukemia [206]; lymphoma [207]; | High mortality rate (5%) [208] and extensive post-transplantation care [209] |
| Endocrine therapy | Inhabitation of growth by altering hormone signaling | Unknown #, for hormone receptor positive patients, ++ | Prostate cancer [210], breast cancer [211] | Secondary cancer [212] |
| Cancer Type | Current Treatment | Curative Possibility, 5-Year RSR | Curative Methods | Stages Distribution (Localized, Regional, Distant) ‡ | 5-Year RSR by Stage ‡ |
|---|---|---|---|---|---|
| Head and Neck | Surgery, radiotherapy, chemotherapy, targeted therapy, immunotherapy [221] | ++, 68.6% | Surgery, radiotherapy | 30,2%, 52,4%, 17,4% | 85.8%, 67.3%, 40.8% |
| Thyroid | |||||
| Papillary | Surgery, radioiodine therapy (131I), targeted therapy [222] | +++, 99.7% | Surgery, radioiodine therapy (131I) | 63,6%, 33,8%, 2,7% | 100.0%, 99.4%, 82.1% |
| Follicular | Surgery, radioiodine therapy (131I), targeted therapy [222] | +++, 94.3% | Surgery, radioiodine therapy (131I) | 55,5%, 39,1%, 5,5% | 99.1%, 93.2%, 33.8% |
| Medullary | Surgery, targeted therapy [222] | +++, 80.9% | Surgery | 37,0%, 38,9%, 24,1% | 100.0%, 92.2%, 24.4% |
| Anaplastic * | Surgery, radiotherapy, targeted therapy, chemotherapy [222] | +, 4.7% | Surgery | 9,5%, 42,9%, 47,6% | 0.0%, 11.3%, 0.0% |
| Breast | |||||
| Breast | Surgery, chemotherapy, radiotherapy, targeted therapy, endocrine therapy [211] | +++, 90.8% | Surgery | 63,2%, 29,3%, 7,5% | 99.1%, 88.3%, 36.3% |
| Lung | |||||
| Non-small cell lung cancer | Surgery, radiotherapy, chemotherapy, immunotherapy, targeted therapy [223] | +, 21.0% | Surgery, radiotherapy [224] § | 18,3%, 24,1%, 57,6% | 58.3%, 32.2%, 4.7% |
| Small cell lung cancer | Surgery, radiotherapy, chemotherapy, immunotherapy [225] | +, 7.0% | Surgery | 19,3%, 80,4%, 0,3% | 23.9%, 18.4%, 3.1% |
| Gastrointestinal cancer | |||||
| Oesophagus | Surgery, radiotherapy, chemotherapy, immunotherapy [226,227] | +, 22.3% | Surgery, endoscopic resection | 21,4%, 32,6%, 45,9% | 56.0%, 28.0%, 4.2% |
| Gastric | Surgery, radiotherapy, chemotherapy, targeted therapy, immunotherapy [42] | +, 20.2% | Surgery, endoscopic resection | 22,5%, 28,9%, 48,5% | 52.8%, 28.3%, 3.1% |
| Hepatocellular carcinoma | Surgery, intervention, targeted therapy, radiotherapy, chemotherapy, immunotherapy [228] | +, 21.2% | Surgical resection, transplantation, local ablation | 53,4%, 31,6%, 15,0% | 35.1%, 9.6%, 2.1% |
| Pancreatic cancer | Surgery, targeted therapy, intervention, radiotherapy, chemotherapy [229] | +, 8.4% | Surgery | 21,0%, 78,9%, 0,1% | 32.5%, 10.8%, 3.3% |
| Colorectal | Surgery, chemotherapy, targeted therapy, radiotherapy, immunotherapy [230] | ++, 66.7% | Surgery, endoscopic resection | 42,6%, 35,6%, 21,7% | 90.2%, 75.0%, 13.6% |
| Anal | Surgery, chemotherapy, radiotherapy (EBRT, brachytherapy), targeted therapy [231] | ++, 74.3% | Surgery | 54,6%, 35,3%, 10,2% | 83.7%, 67.9%, 44.7% |
| Genitourinary cancer | |||||
| Renal | Surgery, chemotherapy, targeted therapy, immunotherapy, ablation, radiotherapy [232] | ++, 74.6% | Surgery, local ablation [174] § | 0,2%, 51,8%, 48,0% | 92.9%, 70.0%, 11.4% |
| Bladder | Surgery, chemotherapy, targeted therapy, radiotherapy, immunotherapy [233] | +++, 77.3% | Surgery | 0,3%, 77,9%, 21,8% | 90.8%, 44.9%, 3.7% |
| Prostate | Surgery, chemotherapy, radiotherapy (EBRT, brachytherapy), endocrine therapy [210] | +++, 99.5% | Surgery | 95,4% +, 4,6% | 100% +, 28.2% |
| Testicular | Surgery, chemotherapy, radiotherapy [234] | +++, 96.4% | Surgery, chemotherapy, radiotherapy | 71,5%, 17,9%, 10,6% | 99.9%, 99.1%, 73.8% |
| Gynaecological cancer | |||||
| Ovarian | Surgery, chemotherapy, radiotherapy, targeted therapy, endocrine therapy [235] | ++, 46.8% | Surgery | 64,8%, 35,0%. 0,2% | 95.0%, 62.8%, 31.7% |
| Endometrial | Surgery, chemotherapy, radiotherapy, targeted therapy, endocrine therapy, immunotherapy [236] | +++, 85.2% | Surgery | 0,3%, 63,0%, 36,7% | 96.5%, 71.6%, 32.5% |
| Cervical | Surgery, chemotherapy, radiotherapy (EBRT, brachytherapy), targeted therapy, immunotherapy [237] | ++, 69.1% | Surgery | 48,5%, 36,6%, 14,9% | 92.4%, 60.9%, 18.0% |
| Melanoma | |||||
| Melanoma | Surgery, chemotherapy, radiotherapy, targeted therapy, PDT, immunotherapy [238] | +++, 93.7% | Surgery, PDT, immunotherapy | 0,7%, 68,6%, 30,7% | 99.4%, 68.3%, 17.9% |
| Leukemia $ | |||||
| Acute lymphoid leukemia | Chemotherapy, targeted therapy, CAR-T, HSCT [239,240] | ++, 74.0% | Chemotherapy, HSCT | NA | NA |
| Acute myeloid leukemia | Chemotherapy, targeted therapy, CAR-T, HSCT [241,242] | +, 28.6% | Chemotherapy, HSCT | NA | NA |
| Chronic lymphoid leukemia | Chemotherapy, targeted therapy, HSCT, observation ¶ [243] | +++, 82.2% | Chemotherapy, HSCT | NA | NA |
| Chronic myeloid leukemia | Chemotherapy, targeted therapy, HSCT, observation ¶ [244] | ++, 70.0% | Chemotherapy, HSCT | NA | NA |
| Lymphoma £ | |||||
| Hodgkin’s | Chemotherapy, targeted therapy, HSCT, radiotherapy, immunotherapy [37] | +++, 86.7% | Chemotherapy, HSCT | 15,4%, 42,6%, 22,1%, 20,0% | 93.2%, 93.2%, 83.7%, 72.2% |
| Non-Hodgkin’s | Chemotherapy, targeted therapy, HSCT, radiotherapy, immunotherapy [36] | ++, 72.6% | Chemotherapy, HSCT | 28,1%, 16,6%, 17,5%, 37,8% | 83.6%, 77.0%, 68.3%, 66.5% |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, S.; Liu, Y.; Feng, Y.; Zhang, J.; Swinnen, J.; Li, Y.; Ni, Y. A Review on Curability of Cancers: More Efforts for Novel Therapeutic Options Are Needed. Cancers 2019, 11, 1782. https://doi.org/10.3390/cancers11111782
Wang S, Liu Y, Feng Y, Zhang J, Swinnen J, Li Y, Ni Y. A Review on Curability of Cancers: More Efforts for Novel Therapeutic Options Are Needed. Cancers. 2019; 11(11):1782. https://doi.org/10.3390/cancers11111782
Chicago/Turabian StyleWang, Shuncong, Yewei Liu, Yuanbo Feng, Jian Zhang, Johan Swinnen, Yue Li, and Yicheng Ni. 2019. "A Review on Curability of Cancers: More Efforts for Novel Therapeutic Options Are Needed" Cancers 11, no. 11: 1782. https://doi.org/10.3390/cancers11111782
APA StyleWang, S., Liu, Y., Feng, Y., Zhang, J., Swinnen, J., Li, Y., & Ni, Y. (2019). A Review on Curability of Cancers: More Efforts for Novel Therapeutic Options Are Needed. Cancers, 11(11), 1782. https://doi.org/10.3390/cancers11111782







