Understanding the Interplay between COX-2 and hTERT in Colorectal Cancer Using a Multi-Omics Analysis
Abstract
1. Introduction
2. Results
2.1. Immunohistochemical Expression of COX-2
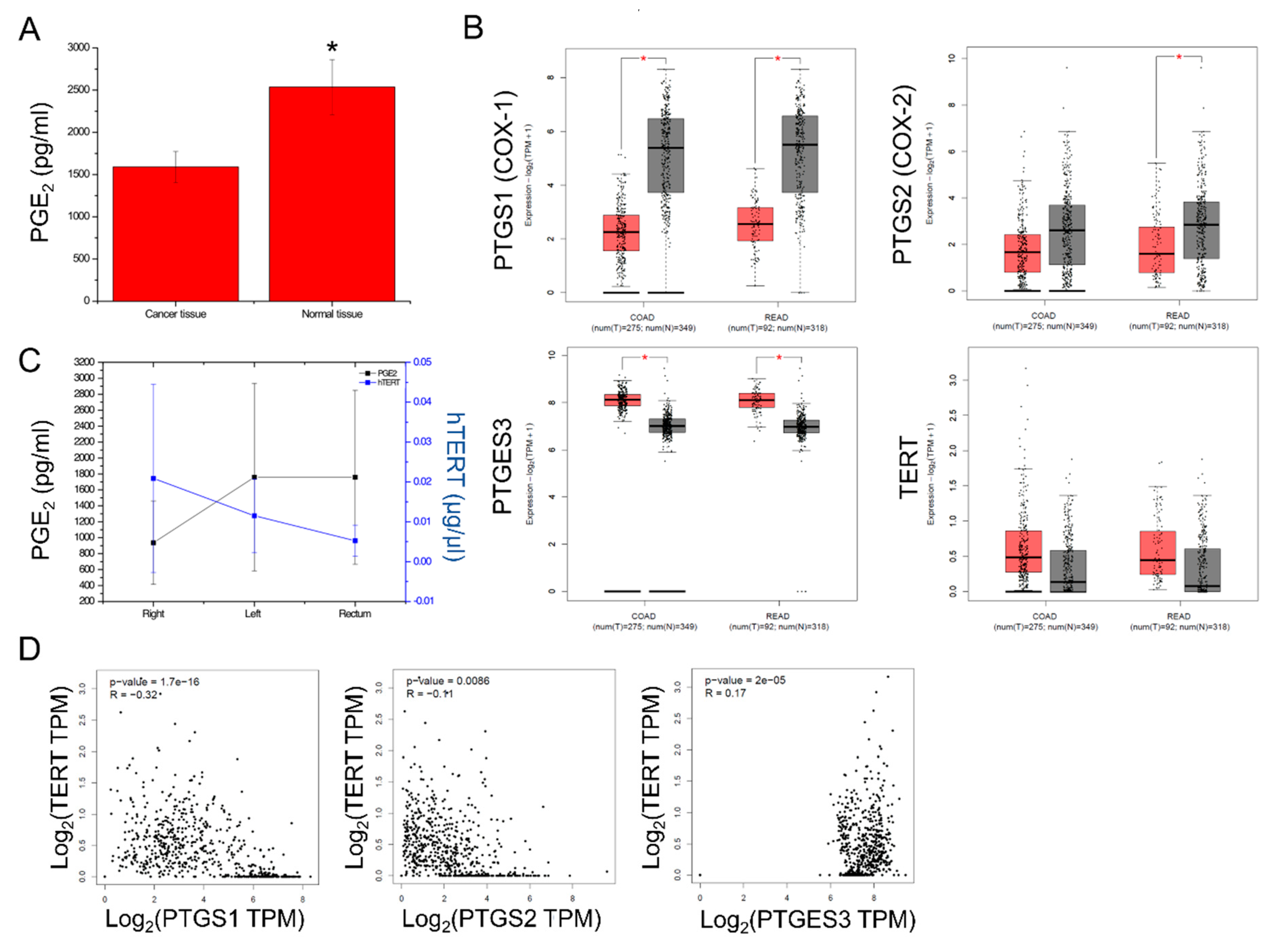
2.2. PGE2 Composition and PTGS1, PTGS2, PTGES3, TERT Expression in CRC
2.3. Isoform Gene Expression Analysis
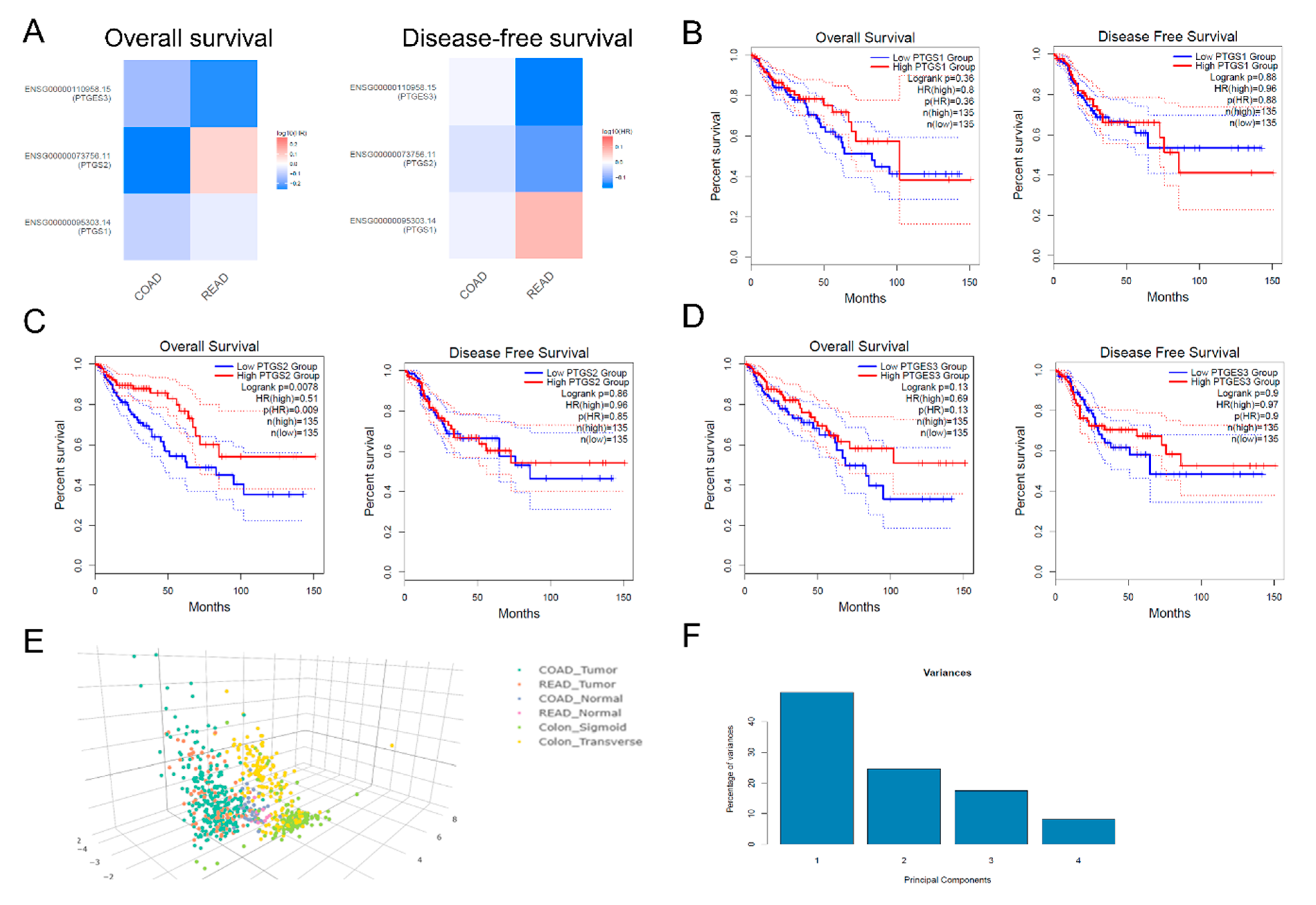
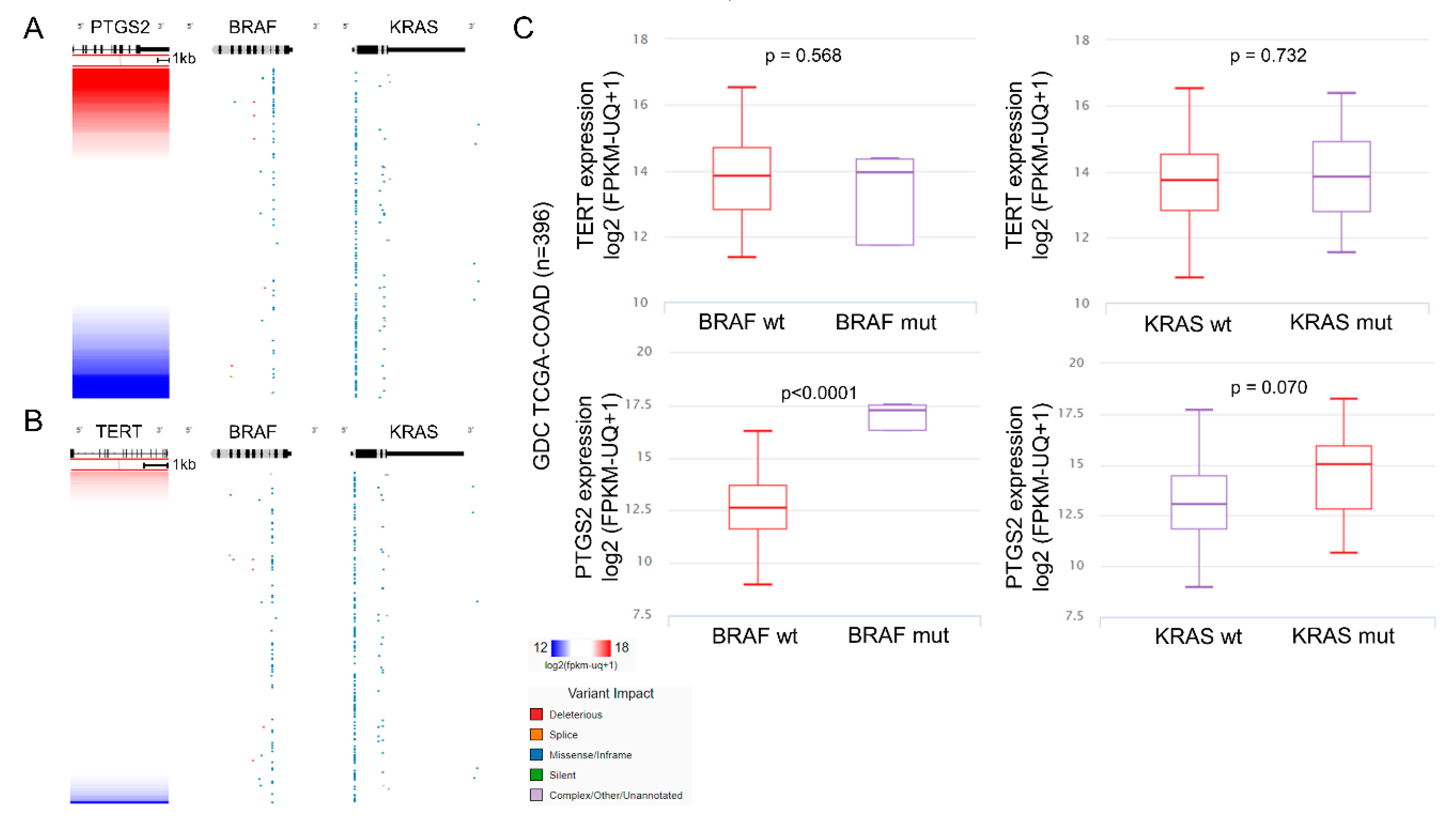
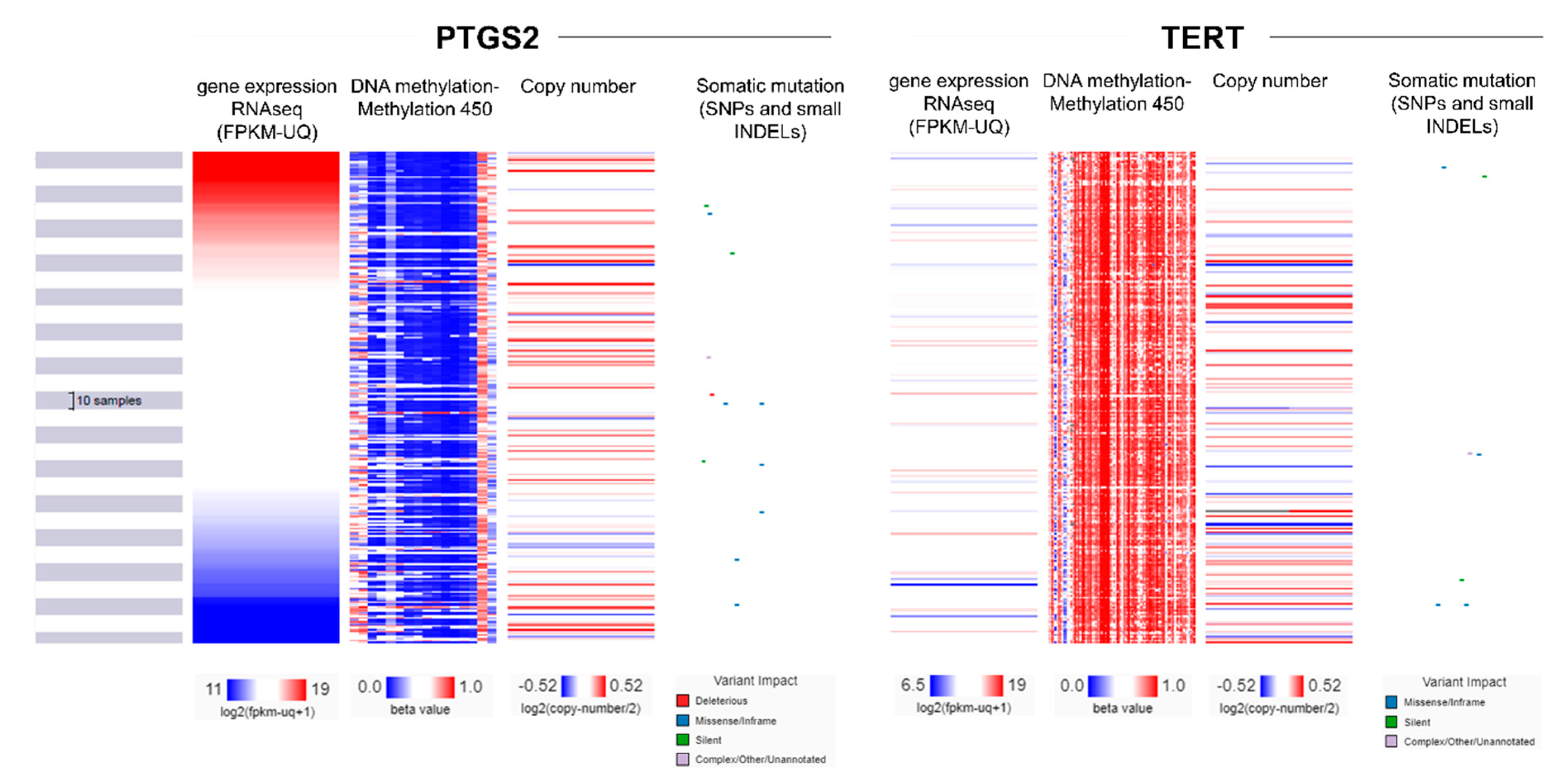
2.4. Multi-Omics Analysis
3. Discussion
4. Materials and Methods
4.1. Patients
4.2. Prostaglandin E2 Expression
4.3. Immunohistochemistry (IHC)
4.4. Telomeric Repeat Amplification Protocol (TRAP)
4.5. TCGA Data Extraction and RNA-Seq Analysis
4.6. Isoform Analysis
4.7. Survival Analysis
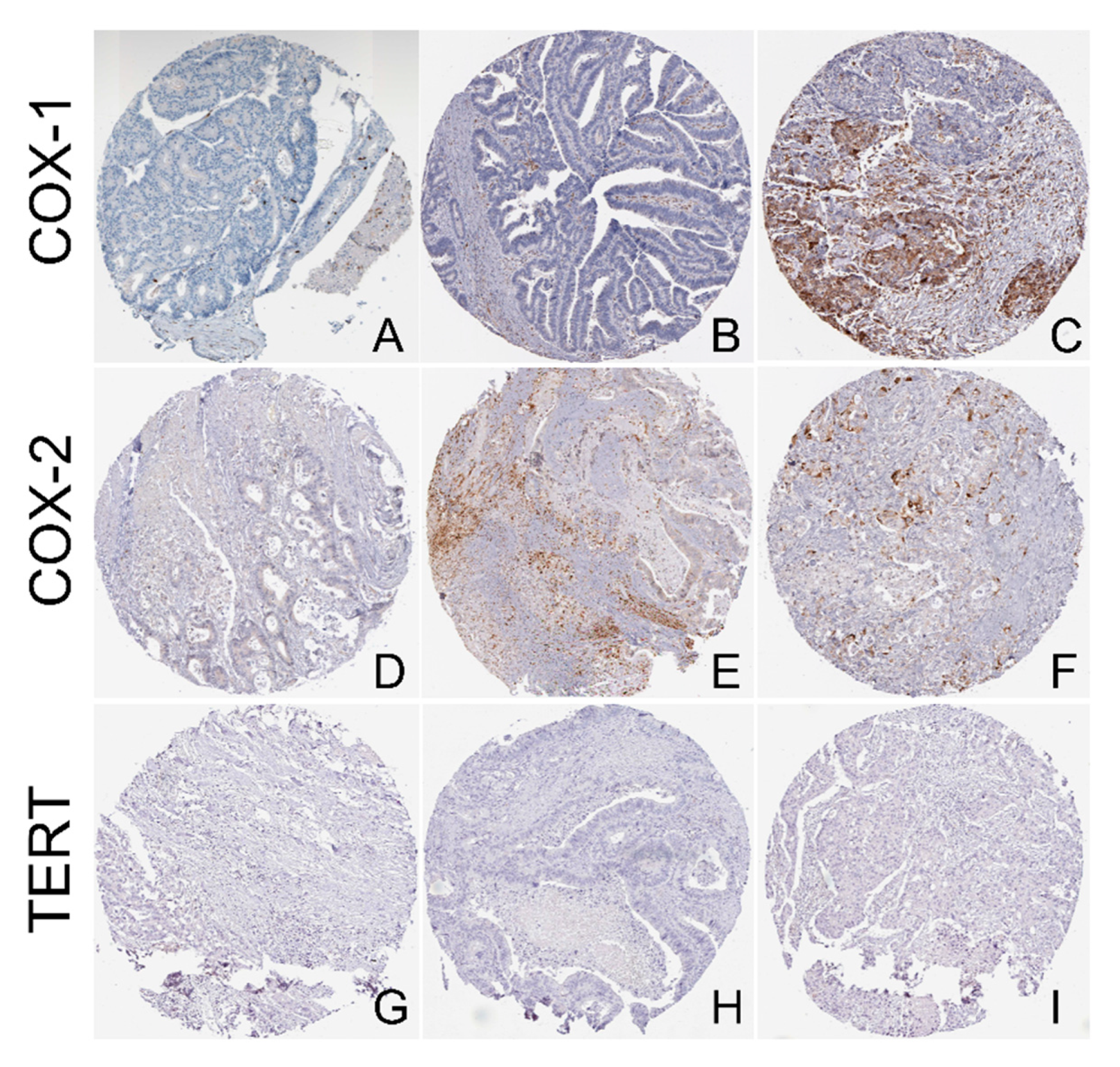
4.8. Data Extraction from the Human Protein Atlas (HPA) and Protein Expression Analysis
4.9. Statistical Analysis
4.10. Multi-Omics Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Siegel, R.L.; Miller, K.D.; Fedewa, S.A.; Ahnen, D.J.; Meester, R.G.S.; Barzi, A.; Jemal, A. Colorectal cancer statistics, 2017. CA Cancer J. Clin. 2017, 67, 177–193. [Google Scholar] [CrossRef]
- Krishnan, K.; Ruffin, M.T.; Brenner, D.E. Colon cancer chemoprevention: Clinical development of aspirin as a chemopreventive agent. J. Cell. Biochem. 1997, 67, 148–158. [Google Scholar] [CrossRef]
- Majerus, P.W. Prostaglandins: Critical roles in pregnancy and colon cancer. Curr. Boil. 1998, 8, R87–R89. [Google Scholar] [CrossRef][Green Version]
- Thun, M.J.; Namboodiri, M.M.; Heath, C.W. Aspirin Use and Reduced Risk of Fatal Colon Cancer. N. Engl. J. Med. 1991, 325, 1593–1596. [Google Scholar] [CrossRef] [PubMed]
- Giardiello, F.M.; Hamilton, S.R.; Krush, A.J.; Piantadosi, S.; Hylind, L.M.; Celano, P.; Booker, S.V.; Robinson, C.R.; Offerhaus, G.J.A. Treatment of Colonic and Rectal Adenomas with Sulindac in Familial Adenomatous Polyposis. N. Engl. J. Med. 1993, 328, 1313–1316. [Google Scholar] [CrossRef]
- Rouzer, C.A.; Marnett, L.J. Cyclooxygenases: Structural and functional insights. J. Lipid Res. 2009, 50, S29–S34. [Google Scholar] [CrossRef] [PubMed]
- Maihöfner, C.; Tegeder, I.; Euchenhofer, C.; DeWitt, D.; Brune, K.; Bang, R.; Neuhuber, W.; Geisslinger, G. Localization and regulation of cyclo-oxygenase-1 and -2 and neuronal nitric oxide synthase in mouse spinal cord. Neuroscience 2000, 101, 1093–1108. [Google Scholar] [CrossRef]
- Harris, R.C.; A McKanna, J.; Akai, Y.; Jacobson, H.R.; Dubois, R.N.; Breyer, M.D. Cyclooxygenase-2 is associated with the macula densa of rat kidney and increases with salt restriction. J. Clin. Investig. 1994, 94, 2504–2510. [Google Scholar] [CrossRef]
- Maihöfner, C.; Schlötzer-Schrehardt, U.; Gühring, H.; Zeilhofer, H.U.; O Naumann, G.; Pahl, A.; Mardin, C.; Tamm, E.R.; Brune, K. Expression of cyclooxygenase-1 and -2 in normal and glaucomatous human eyes. Investig. Ophthalmol. Vis. Sci. 2001, 42, 2616–2624. [Google Scholar]
- Vane, J.R.; Bakhle, Y.S.; Botting, R.M. Cyclooxygenases 1 and 2. Annu. Rev. Pharmacol. Toxicol. 1998, 38, 97–120. [Google Scholar] [CrossRef]
- Half, E.; Tang, X.M.; Gwyn, K.; Sahin, A.; Wathen, K.; A Sinicrope, F. Cyclooxygenase-2 expression in human breast cancers and adjacent ductal carcinoma in situ. Cancer Res. 2002, 62, 1676–1681. [Google Scholar] [PubMed]
- Ristimäki, A.; Honkanen, N.; Jänkälä, H.; Sipponen, P.; Härkönen, M. Expression of cyclooxygenase-2 in human gastric carcinoma. Cancer Res. 1997, 57, 1276–1280. [Google Scholar] [PubMed]
- Hida, T.; Yatabe, Y.; Achiwa, H.; Muramatsu, H.; Kozaki, K.; Nakamura, S.; Ogawa, M.; Mitsudomi, T.; Sugiura, T.; Takahashi, T. Increased expression of cyclooxygenase 2 occurs frequently in human lung cancers, specifically in adenocarcinomas. Cancer Res. 1998, 58, 3761–3764. [Google Scholar] [PubMed]
- Harris, R.E. Cyclooxygenase-2 (cox-2) blockade in the chemoprevention of cancers of the colon, breast, prostate, and lung. Inflammopharmacology 2009, 17, 55–67. [Google Scholar] [CrossRef] [PubMed]
- Ristimäki, A.; Nieminen, O.; Saukkonen, K.; Hotakainen, K.; Nordling, S.; Haglund, C. Expression of Cyclooxygenase-2 in Human Transitional Cell Carcinoma of the Urinary Bladder. Am. J. Pathol. 2001, 158, 849–853. [Google Scholar] [CrossRef]
- Tomozawa, S.; Tsuno, N.H.; Sunami, E.; Hatano, K.; Kitayama, J.; Osada, T.; Saito, S.; Tsuruo, T.; Shibata, Y.; Nagawa, H. Cyclooxygenase-2 overexpression correlates with tumour recurrence, especially haematogenous metastasis, of colorectal cancer. Br. J. Cancer 2002, 83, 324–328. [Google Scholar] [CrossRef]
- Crofford, L.J. COX-1 and COX-2 tissue expression: implications and predictions. J. Rheumatol. Suppl. 1997, 49, 15–19. [Google Scholar]
- Greenhough, A.; Smartt, H.J.; Moore, A.E.; Roberts, H.R.; Williams, A.C.; Paraskeva, C.; Kaidi, A. The COX-2/PGE2 pathway: key roles in the hallmarks of cancer and adaptation to the tumour microenvironment. Carcinogenesis 2009, 30, 377–386. [Google Scholar] [CrossRef]
- Tsujii, M.; Dubois, R.N. Alterations in cellular adhesion and apoptosis in epithelial cells overexpressing prostaglandin endoperoxide synthase 2. Cell 1995, 83, 493–501. [Google Scholar] [CrossRef]
- Hull, M.A.; Cuthbert, R.J.; Ko, C.W.S.; Scott, D.J.; Cartwright, E.J.; Hawcroft, G.; Perry, S.L.; Ingram, N.; Carr, I.M.; Markham, A.F.; et al. Paracrine cyclooxygenase-2 activity by macrophages drives colorectal adenoma progression in the Apc Min/+ mouse model of intestinal tumorigenesis. Sci. Rep. 2017, 7, 6074. [Google Scholar] [CrossRef]
- Shattuck-Brandt, R.L.; Varilek, G.W.; Radhika, A.; Yang, F.; Washington, M.; Dubois, R.N. Cyclooxygenase 2 expression is increased in the stroma of colon carcinomas from IL–10−/− mice. Gastroenterology 2000, 118, 337–345. [Google Scholar] [CrossRef]
- Caughey, G.E.; Cleland, L.G.; Penglis, P.S.; Gamble, J.R.; James, M.J. Roles of Cyclooxygenase (COX)-1 and COX-2 in Prostanoid Production by Human Endothelial Cells: Selective Up-Regulation of Prostacyclin Synthesis by COX-2. J. Immunol. 2001, 167, 2831–2838. [Google Scholar] [CrossRef] [PubMed]
- Harley, C.B. Telomerase and cancer therapeutics. Nat. Rev. Cancer 2008, 8, 167–179. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Liang, X.; Li, B.; Björkholm, M.; Jia, J.; Xu, D. Telomerase reverse transcriptase inhibition stimulates cyclooxygenase 2 expression in cancer cells and synergizes with celecoxib to exert anti-cancer effects. Br. J. Cancer 2013, 108, 2272–2280. [Google Scholar] [CrossRef] [PubMed]
- Zhong, Y.Q.; Xia, Z.S.; Fu, Y.R.; Zhu, Z.H. Knockdown of hTERT by SiRNA suppresses growth of Capan-2 human pancreatic cancer cell via the inhibition of expressions of Bcl-2 and COX-2. J. Dig. Dis. 2010, 11, 176–184. [Google Scholar] [CrossRef]
- Diamantopoulou, K.; Lazaris, A.; Mylona, E.; Zervas, A.; Stravodimos, K.; Nikolaou, I.; Athanassiadou, S.; Nakopoulou, L. Cyclooxygenase-2 protein expression in relation to apoptotic potential and its prognostic significance in bladder urothelial carcinoma. Anticancer. Res. 2005, 25, 4543–4549. [Google Scholar]
- Peng, L.; Zhou, Y.; Wang, Y.; Mou, H.; Zhao, Q. Prognostic Significance of COX-2 Immunohistochemical Expression in Colorectal Cancer: A Meta-Analysis of the Literature. PLoS ONE 2013, 8, e58891. [Google Scholar] [CrossRef]
- Eberhart, C.E.; Coffey, R.J.; Radhika, A.; Giardiello, F.M.; Ferrenbach, S.; Dubois, R.N. Up-regulation of cyclooxygenase 2 gene expression in human colorectal adenomas and adenocarcinomas. Gastroenterology 1994, 107, 1183–1188. [Google Scholar] [CrossRef]
- Shirasu, K.; Lahaye, T.; Tan, M.-W.; Zhou, F.; Azevedo, C.; Schulze-Lefert, P. A Novel Class of Eukaryotic Zinc-Binding Proteins Is Required for Disease Resistance Signaling in Barley and Development in C. elegans. Cell 1999, 99, 355–366. [Google Scholar] [CrossRef]
- Prieto, P.; I Jaén, R.; Calle, D.; Gómez-Serrano, M.; Núñez, E.; Fernández-Velasco, M.; Martín-Sanz, P.; Alonso, S.; Vázquez, J.; Cerdán, S.; et al. Interplay between post-translational cyclooxygenase-2 modifications and the metabolic and proteomic profile in a colorectal cancer cohort. World J. Gastroenterol. 2019, 25, 433–446. [Google Scholar] [CrossRef]
- Sheng, H.; Shao, J.; Morrow, J.D.; Beauchamp, R.D.; Dubois, R.N. Modulation of apoptosis and Bcl-2 expression by prostaglandin E2 in human colon cancer cells. Cancer Res. 1998, 58, 362–366. [Google Scholar] [PubMed]
- Cao, Y.; Pearman, A.T.; Zimmerman, G.A.; McIntyre, T.M.; Prescott, S.M. Intracellular unesterified arachidonic acid signals apoptosis. Proc. Natl. Acad. Sci. USA 2000, 97, 11280–11285. [Google Scholar] [CrossRef] [PubMed]
- Morham, S.G.; Young, D.A.; Zhang, X.; Langenbach, R. Malignant Transformation and Antineoplastic Actions of Nonsteroidal Antiinflammatory Drugs (Nsaids) on Cyclooxygenase-Null Embryo Fibroblasts. J. Exp. Med. 1999, 190, 451–460. [Google Scholar]
- Sano, H.; Kawahito, Y.; Wilder, R.L.; Hashiramoto, A.; Mukai, S.; Asai, K.; Kimura, S.; Kato, H.; Kondo, M.; Hla, T. Expression of cyclooxygenase-1 and -2 in human colorectal cancer. Cancer Res. 1995, 55, 3785–3789. [Google Scholar] [PubMed]
- Tucker, O.N.; Dannenberg, A.J.; Yang, E.K.; Zhang, F.; Teng, L.; Daly, J.M.; A Soslow, R.; Masferrer, J.L.; Woerner, B.M.; Koki, A.T.; et al. Cyclooxygenase-2 expression is up-regulated in human pancreatic cancer. Cancer Res. 1999, 59, 987–990. [Google Scholar] [PubMed]
- Zimmermann, K.C.; Sarbia, M.; A Weber, A.; Borchard, F.; Gabbert, H.E.; Schrör, K. Cyclooxygenase-2 expression in human esophageal carcinoma. Cancer Res. 1999, 59, 198–204. [Google Scholar] [PubMed]
- Yoshimura, N.; Sano, H.; Okamoto, M.; Akioka, K.; Ushigome, H.; Kadotani, Y.; Yoshimura, R.; Nobori, S.; Higuchi, A.; Ohmori, Y.; et al. Expression of Cyclooxygenase-1 and -2 in Human Breast Cancer. Surg. Today 2003, 33, 805–811. [Google Scholar] [CrossRef]
- Flossmann, E.; Rothwell, P.M. Effect of aspirin on long-term risk of colorectal cancer: Consistent evidence from randomised and observational studies. Lancet 2007, 369, 1603–1613. [Google Scholar] [CrossRef]
- Eschwège, P. A Histopathologic Investigation of PGE2 Pathways as Predictors of Proliferation and Invasion in Urothelial Carcinomas of the Bladder. Eur. Urol. 2003, 44, 435–441. [Google Scholar] [CrossRef]
- Kutchera, W.; Jones, D.A.; Matsunami, N.; Groden, J.; McIntyre, T.M.; Zimmerman, G.A.; White, R.L.; Prescott, S.M. Prostaglandin H synthase 2 is expressed abnormally in human colon cancer: evidence for a transcriptional effect. Proc. Natl. Acad. Sci. USA 1996, 93, 4816–4820. [Google Scholar] [CrossRef]
- Pisano, C.; Ottaiano, A.; Tatangelo, F.; Di Bonito, M.; Falanga, M.; Iaffaioli, V.R.; Botti, G.; Pignata, S.; Acquaviva, A.M. Cyclooxygenase-2 expression is associated with increased size in human sporadic colorectal adenomas. Anticancer. Res. 2005, 25, 2065–2068. [Google Scholar] [PubMed]
- Arnoletti, J.P.; Upson, J.; Babb, J.S.; Bellacosa, A.; Watson, J.C. Differential stromal and epithelial localization of cyclooxygenase-2 (COX-2) during colorectal tumorigenesis. J. Exp. Clin. Cancer Res. 2005, 24, 279–287. [Google Scholar] [PubMed]
- Ohta, T.; Takahashi, M.; Ochiai, A. Increased protein expression of both inducible nitric oxide synthase and cyclooxygenase-2 in human colon cancers. Cancer Lett. 2006, 239, 246–253. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, K.; Arimura, Y.; Shimizu, H.; Takahashi, H.; Endo, T.; Imai, K.; Yamano, H. Increased cyclooxygenase-2 expression in large flat colorectal tumors (laterally spreading tumors). J. Gastroenterol. 2003, 38, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Battu, S.; Beneytout, J.L.; Pairet, M.; Rigaud, M. Cyclooxygenase-2 up-regulation after FLAP transfection in human adenocarcinoma cell line HT29 cl.19A. FEBS Lett. 1998, 437, 49–55. [Google Scholar] [CrossRef]
- Mahmoud, A.S.; Umair, A.; Azzeghaiby, S.N.; Alqahtani, F.H.; Hanouneh, S.; Tarakji, B. Expression of cyclooxygenase-2 (COX-2) in colorectal adenocarcinoma: an immunohistochemical and histopathological study. Asian Pac. J. Cancer Prev. 2014, 15, 6787–6790. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Charalambous, M.P.; Maihöfner, C.; Bhambra, U.; Lightfoot, T.; Gooderham, N.J. Upregulation of cyclooxygenase-2 is accompanied by increased expression of nuclear factor-κB and IκB kinase-α in human colorectal cancer epithelial cells. Br. J. Cancer 2003, 88, 1598–1604. [Google Scholar] [CrossRef]
- Charalambous, M.P.; Lightfoot, T.; Speirs, V.; Horgan, K.; Gooderham, N.J. Expression of cox-2, nf-κb-p65, nf-κb-p50 and ikkα in malignant and adjacent normal human colorectal tissue. Br. J. Cancer 2009, 101, 106–115. [Google Scholar] [CrossRef]
- Sonoshita, M.; Takaku, K.; Oshima, M.; Sugihara, K.-I.; Taketo, M.M. Cyclooxygenase-2 expression in fibroblasts and endothelial cells of intestinal polyps. Cancer Res. 2002, 62, 6846–6849. [Google Scholar]
- Wendum, D.; Comperat, E.; Boëlle, P.Y.; Parc, R.; Masliah, J.; Trugnan, G.; Fléjou, J.F. Cytoplasmic phospholipase A2 alpha overexpression in stromal cells is correlated with angiogenesis in human colorectal cancer. Mod. Pathol. 2005, 18, 212–220. [Google Scholar] [CrossRef]
- Elder, D.J.; Halton, D.E.; Playle, L.C.; Paraskeva, C. The MEK/ERK pathway mediates COX-2-selective NSAID-induced apoptosis and induced COX-2 protein expression in colorectal carcinoma cells. Int. J. Cancer 2002, 99, 323–327. [Google Scholar] [CrossRef]
- Sheng, H.; Shao, J.; A O’Mahony, C.; Lamps, L.; Albo, D.; Isakson, P.C.; Berger, D.H.; Dubois, R.N.; Beauchamp, R.D. Transformation of intestinal epithelial cells by chronic TGF-β1 treatment results in downregulation of the type II TGF-β receptor and induction of cyclooxygenase-2. Oncogene 1999, 18, 855–867. [Google Scholar] [CrossRef] [PubMed]
- Crew, T.E.; Elder, D.J.; Paraskeva, C. A cyclooxygenase-2 (COX-2) selective non-steroidal anti-inflammatory drug enhances the growth inhibitory effect of butyrate in colorectal carcinoma cells expressing COX-2 protein: regulation of COX-2 by butyrate. Carcinogenesis 2000, 21, 69–77. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Dimberg, J.; Samuelsson, A.; Hugander, A.; Söderkvist, P. Differential expression of cyclooxygenase 2 in human colorectal cancer. Gut 1999, 45, 730–732. [Google Scholar] [CrossRef] [PubMed]
- Ayiomamitis, G.D.; Notas, G.; Zaravinos, A.; Drygiannakis, I.; Georgiadou, M.; Sfakianaki, O.; Mastrodimou, N.; Thermos, K.; Kouroumalis, E. Effects of octreotide and insulin on colon cancer cellular proliferation and correlation with hTERT activity. Oncoscience 2014, 1, 457–467. [Google Scholar] [CrossRef][Green Version]
- Roufas, C.; Chasiotis, D.; Makris, A.; Efstathiades, C.; Dimopoulos, C.; Zaravinos, A. The Expression and Prognostic Impact of Immune Cytolytic Activity-Related Markers in Human Malignancies: A Comprehensive Meta-analysis. Front. Oncol. 2018, 8, 27. [Google Scholar] [CrossRef]
- Zaravinos, A.; Roufas, C.; Nagara, M.; Moreno, B.D.L.; Oblovatskaya, M.; Efstathiades, C.; Dimopoulos, C.; Ayiomamitis, G.D. Cytolytic activity correlates with the mutational burden and deregulated expression of immune checkpoints in colorectal cancer. J. Exp. Clin. Cancer Res. 2019, 38, 1–18. [Google Scholar] [CrossRef]
- Uhlen, M.; Zhang, C.; Lee, S.; Sjöstedt, E.; Fagerberg, L.; Bidkhori, G.; Benfeitas, R.; Arif, M.; Liu, Z.; Edfors, F.; et al. A pathology atlas of the human cancer transcriptome. Science 2017, 357, eaan2507. [Google Scholar] [CrossRef]
- Goldman, M.; Craft, B.; Hastie, M.; Repečka, K.; Kamath, A.; McDade, F.; Rogers, D.; Brooks, A.N.; Zhu, J.; Haussler, D. The UCSC Xena platform for public and private cancer genomics data visualization and interpretation. bioRxiv 2019, 326470. [Google Scholar] [CrossRef]
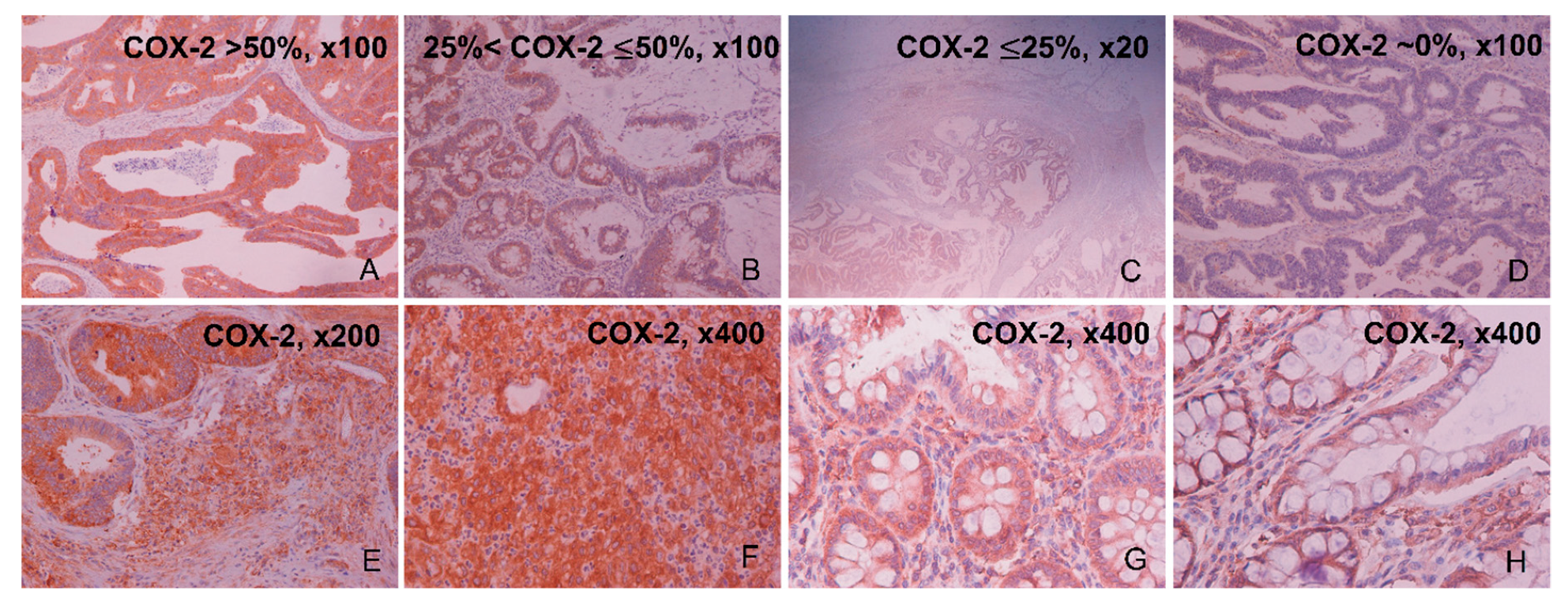

| Percentage (%) of Positive COX-2 Immunostaining by IHC | COX-2 Staining Intensity by IHC | |||||
|---|---|---|---|---|---|---|
| CRC Patient ID# | Low (≤25% of Epithelial Cancer Cells) | Medium (>25% & ≤50% of Epithelial Cancer Cells) | High (>50% of Epithelial Cancer Cells) | Weak | Moderate | Strong |
| 1405E/03 | + | + | ||||
| 2571D/03 | + | + | ||||
| 1044G/03 | + | + | ||||
| 2906E/04 | + | + | ||||
| 2908D/04 | + | + | ||||
| 3661E/03 | + | + | ||||
| 1404E/03 | + | + | ||||
| 856E/03 | + | + | ||||
| 2909D/04 | + | + | ||||
| 2831D/04 | + | + | ||||
| 975D/02 | Negative staining | |||||
| 2904Z/04 | + | + | ||||
| 948G/03 | + | + | ||||
| 348E/03 | + | + | ||||
| 5071D/03 | + | + | ||||
| 988E/04 | + | + | ||||
| 6003G/02 | + | + | ||||
| 977E/03 | + | + | ||||
| 857E/03 | Negative staining | |||||
| 304D/03 | + | + | ||||
| 3986E | + | + | ||||
| 1694G/03 | + | + | ||||
| 1096 | + | + | + | |||
| 1254G | Negative staining | |||||
| 5433G | + | + | + | |||
| 633Z | + | + | ||||
| 987E/04 | + | + | ||||
| 423D/03 | + | + | ||||
| 656D/03 | + | + | ||||
| 4961D/02 | + | + | ||||
| 5025E/02 | + | + | ||||
| 482E/03 | + | + | ||||
| 770E/03 | + | + | ||||
| 3386E | + | + | ||||
| 1403D/03 | + | + | ||||
| 5758G/02 | + | + | ||||
| 5565G | + | + | ||||
| 635D | + | + | ||||
| 3701D | + | + | ||||
| 3876D | + | + | ||||
| 8122/03 | Negative staining | |||||
| 4975E/02 | Negative staining | |||||
| 5024Z | + | + | ||||
| 5013D | + | + | ||||
| 5163Z/02 | Negative staining | |||||
| 5884E/02 | + | + | ||||
| 5024Z/02 | Negative staining | |||||
| 5019D/03 | Negative staining | |||||
| 5199G/02 | Negative staining | |||||
| Variable | Normal Mucosa | Cancer Tissue | ||||||
|---|---|---|---|---|---|---|---|---|
| No | Mean | SD | p-Value | No | Mean | SD | p-Value | |
| All cases | 35 | 2374.04 | 1695.081 | 34 | 1589.622 | 1075.305 | 0.013 | |
| Sex | ||||||||
| Male | 13 | 1976.704 | 1477.7668 | 13 | 2034.405 | 1305.7355 | ||
| Female | 22 | 2866.836 | 2109.3560 | 0.056 | 21 | 1314.280 | 822.6191 | 0.191 |
| Smoke | ||||||||
| No | 24 | 2374.842 | 1669.7945 | 24 | 1419.926 | 860.0917 | ||
| Yes | 8 | 2796.588 | 2498.2338 | 0.59 | 7 | 1667.929 | 1206.1716 | 0.545 |
| BMI | ||||||||
| <25 | 19 | 2244.542 | 1745.6862 | 19 | 1812.677 | 1191.0385 | ||
| ≥25 | 16 | 2882.578 | 2124.8944 | 0.336 | 15 | 1307.085 | 865.2102 | 0.177 |
| Duke’s stage | ||||||||
| A | 0 | 0 | ||||||
| B | 16 | 3273.561 | 2036.8244 | 15 | 1696.322 | 904.9637 | ||
| C | 15 | 1930.935 | 1782.8859 | 15 | 1423.153 | 1294.7909 | ||
| D | 4 | 1856.634 | 1055.7880 | 4 | 1813.756 | 900.6809 | ||
| A+B | 16 | 3273.561 | 2036.8244 | 0.036 | 15 | 1696.322 | 904.9637 | 0.615 |
| C+D | 19 | 1915.293 | 1630.6619 | 19 | 1505.385 | 1210.7447 | ||
| Grade | ||||||||
| low | 4 | 2617.679 | 1318.3896 | 4 | 1261.628 | 802.6919 | ||
| moderate | 28 | 2682.486 | 2039.2589 | 27 | 1700.093 | 1127.0442 | ||
| high | 3 | 1062.409 | 859.9787 | 0.393 | 3 | 1032.711 | 868.3637 | 0.495 |
| Tumor site | ||||||||
| Right | 7 | 1667.951 | 1410.0214 | 7 | 936.412 | 523.005 | ||
| Left | 7 | 2929.321 | 2674.6937 | 7 | 2077.241 | 1396.8070 | ||
| Sigmoid | 10 | 2580.901 | 1377.0804 | 10 | 1536.713 | 1007.5961 | ||
| Rectum | 11 | 2797.966 | 2169.4357 | 0.236 | 10 | 1758.445 | 1090.7798 | 0.608 |
| Right colon | 7 | 1667.951 | 1410.0214 | 7 | 936.412 | 523.0055 | ||
| Left colon | 28 | 2753.282 | 1996.2152 | 0.186 | 27 | 1758.973 | 1122.4103 | 0.071 |
| Colon | 24 | 2416.246 | 1841.0003 | 24 | 1519.279 | 1084.3813 | ||
| Rectum | 11 | 2797.966 | 2169.4357 | 0.594 | 10 | 1758.445 | 1090.7798 | 0.563 |
| Ki-67 expression | ||||||||
| negative | 3 | 3617.660 | 2180.4306 | 3 | 1245.514 | 643.9262 | ||
| positive | 32 | 2434.830 | 1907.2952 | 0.449 | 31 | 1622.923 | 1109.6303 | 0.441 |
| p53 expression | ||||||||
| negative | 12 | 3271.839 | 1679.5463 | 12 | 1526.599 | 861.8398 | ||
| positive | 23 | 2152.412 | 1967.9908 | 0.09 | 22 | 1623.998 | 1193.4976 | 0.786 |
| Characteristic | No. |
|---|---|
| Patients | |
| Cancer tissue | 49 |
| Adjacent normal tissues | 49 |
| Sex | |
| Male | 28 |
| Female | 21 |
| Smoking | |
| Yes | 12 |
| No | 34 |
| BMI | |
| <25 | 21 |
| ≥25 | 25 |
| Dukes staging system | |
| B | 25 |
| C | 19 |
| D | 5 |
| Histology grade | |
| High | 4 |
| Medium | 41 |
| Low | 4 |
| Tumor location | |
| Right colon | 8 |
| Left colon | 24 |
| Rectum | 17 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ayiomamitis, G.D.; Notas, G.; Vasilakaki, T.; Tsavari, A.; Vederaki, S.; Theodosopoulos, T.; Kouroumalis, E.; Zaravinos, A. Understanding the Interplay between COX-2 and hTERT in Colorectal Cancer Using a Multi-Omics Analysis. Cancers 2019, 11, 1536. https://doi.org/10.3390/cancers11101536
Ayiomamitis GD, Notas G, Vasilakaki T, Tsavari A, Vederaki S, Theodosopoulos T, Kouroumalis E, Zaravinos A. Understanding the Interplay between COX-2 and hTERT in Colorectal Cancer Using a Multi-Omics Analysis. Cancers. 2019; 11(10):1536. https://doi.org/10.3390/cancers11101536
Chicago/Turabian StyleAyiomamitis, Georgios D., George Notas, Thivi Vasilakaki, Aikaterini Tsavari, Styliani Vederaki, Theodosis Theodosopoulos, Elias Kouroumalis, and Apostolos Zaravinos. 2019. "Understanding the Interplay between COX-2 and hTERT in Colorectal Cancer Using a Multi-Omics Analysis" Cancers 11, no. 10: 1536. https://doi.org/10.3390/cancers11101536
APA StyleAyiomamitis, G. D., Notas, G., Vasilakaki, T., Tsavari, A., Vederaki, S., Theodosopoulos, T., Kouroumalis, E., & Zaravinos, A. (2019). Understanding the Interplay between COX-2 and hTERT in Colorectal Cancer Using a Multi-Omics Analysis. Cancers, 11(10), 1536. https://doi.org/10.3390/cancers11101536







