2.1. Capacitive MEMS Pressure Sensor Modelling
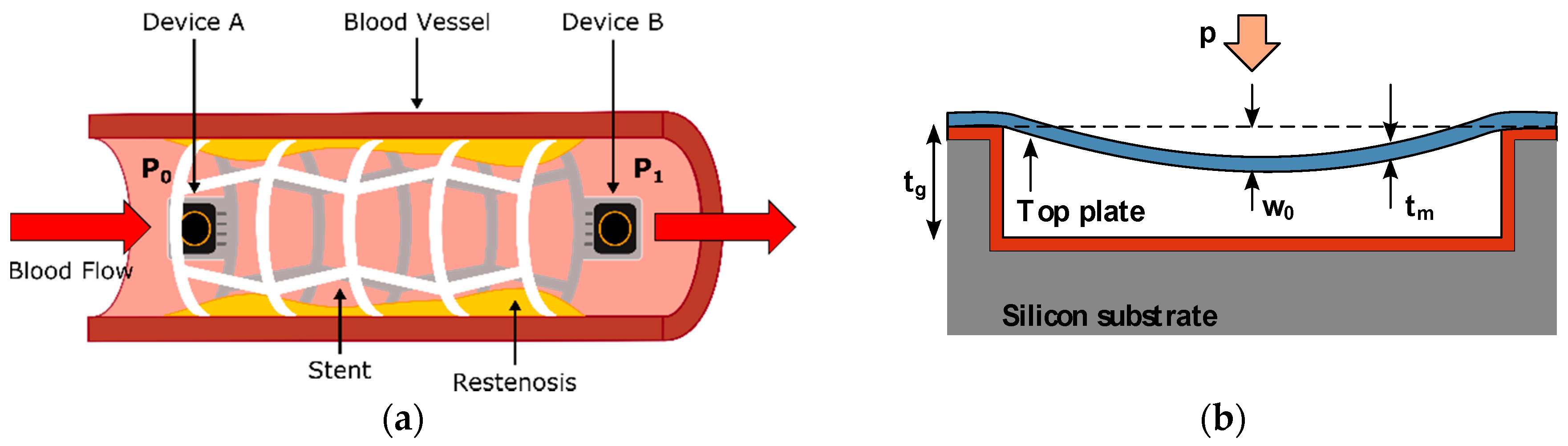
The simplest implementation of a pressure monitoring-based iStent, as show in
Figure 2a, can be built by attaching one or several MEMS capacitive pressure sensors to the longitudinal ends of a commercially available stent. The device functionality is based on the blood pressure detection performed by the sensors, reflecting on proportional changes in their equivalent capacitance [
3,
4]. Thus, MEMS sensors act as pressure-dependent capacitances, which, attached to the coil-like stent structure, form an LC-tank whose resonant frequency is modulated by the pressure inside the vessel. An external handheld device is required to perform the wireless communication with the iStent, via inductive coupling techniques, when placed close enough to the implant location.
The topology of a capacitive MEMS pressure sensor, as described in
Figure 2b, comprises a fully clamped suspended top plate with thickness
, separated a distance of
from a backplate fixed to the substrate. As can be noticed, this topology resembles the traditional parallel-plate capacitor build, so its nominal capacitance is ruled by Equation (3):
where
is the relative permittivity of the medium between the plates,
is the dielectric permittivity of vacuum, and
and
are the overlapping area and gap distance between the plates, respectively.
Once a sufficient load
is applied to the sensor, the suspended plate is forced to bend towards the backplate, in such a way that their separation is reduced and the equivalent capacitance
is increased. Hence, the resulting load-dependent capacitance can be analytically modeled by Equation (4), where parameter
refers to the local top-plate deflection.
2.1.1. Square Sensor Modelling
As extensively reported in Mechanics books and scientific papers, the governing differential equation for the deflection of a thin plate in cartesian coordinates can be expressed as follows [
10,
11]:
where
is the deflection of the square plate at any place,
is a distributed load applied to the upper surface of the plate, and
is referred as the flexural rigidity of the plate and can be defined as follows:
with
and
being the modulus of elasticity and Poisson’s ratio of the plate material, respectively.
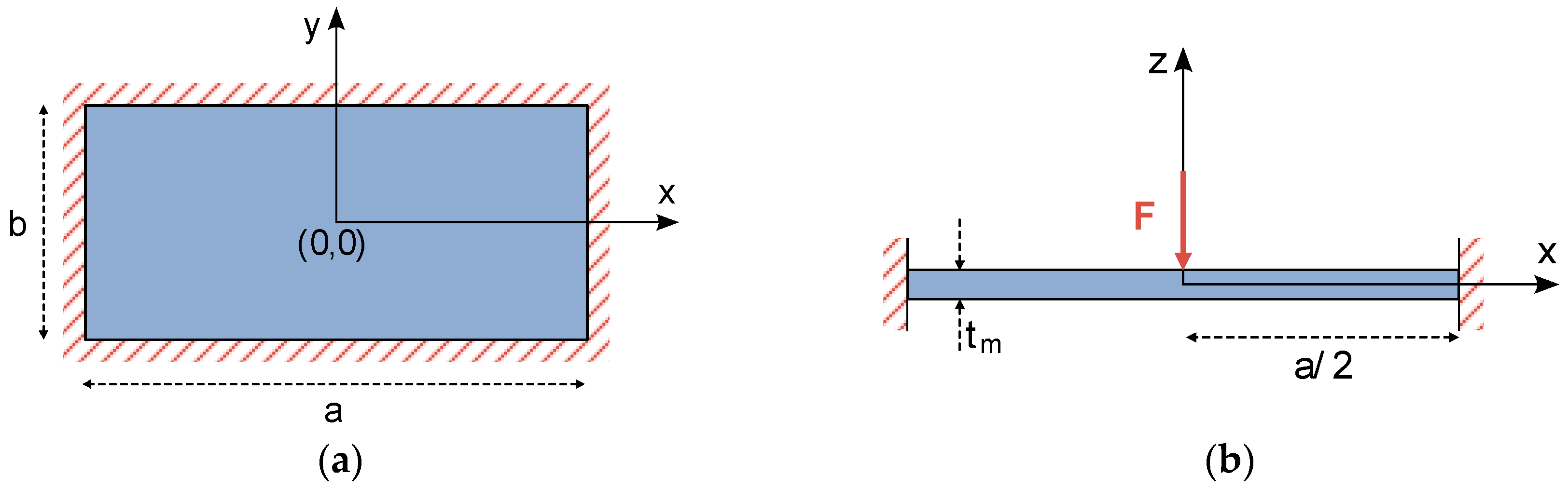
In the case of a fully-clamped square plate with side lengths
and thickness
, such as the one showed in
Figure 3a, the following set of constraints describe the plate bending behavior and can be used to solve the differential equation noted in (5).
Once a single concentrated load is applied to the center of the square plate (
Figure 3b), its deflection equation can be calculated by combining the solutions for three independent problems [
10,
11,
12]. First,
is the bending solution for a simply supported rectangular plate under a concentrated load located at its center, and can be expressed as follows:
where the geometry parameters
and
, defined as
and
, can be reduced to
for the case of a square plate with sides
.
Additionally, and are the bending solutions for a simply supported plate with distributed bending moments applied along the edges and , respectively. The applied edge moments are calculated to guarantee a slope at the boundaries equal to zero, as imposed by constraints (9) and (10).
Hence, each aforementioned solution can be defined as follows:
and
The coefficients
and
can be determined from the fully-clamped plate constraints (9) and (10), or the condition of cancelling the slope at the plate boundaries. Hence, the most significant
and
values for a square plate, given in
Table 1, can be numerically calculated by successive approximations.
To conclude, the combination of Equations (11)–(13) leads to the final bending solution:
2.1.2. Circular Sensor Modelling
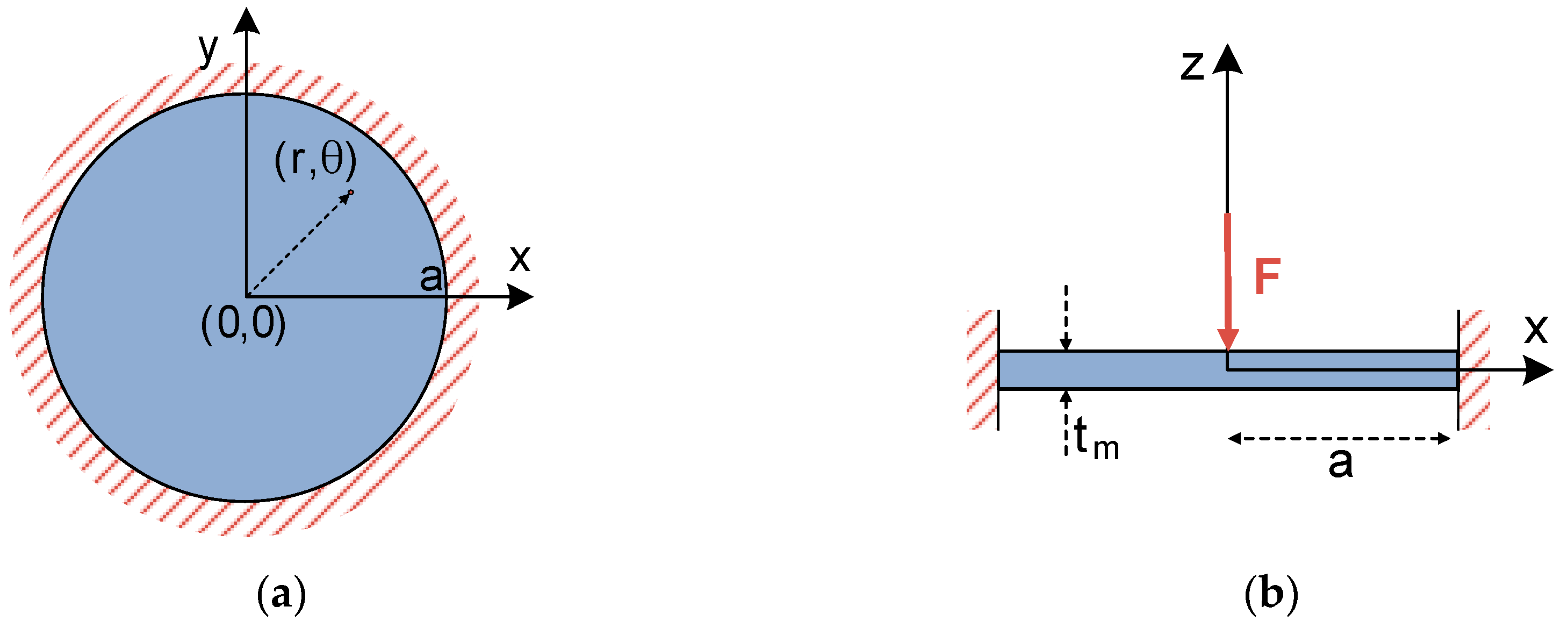
Similar to the case of a square plate, the analytical bending solution for a fully-clamped circular thin plate under a concentrated central force has been widely studied in the literature [
10,
11]. Hence, differential Equation (5) determining plate deflection can be rearranged in polar coordinates [
10,
11], and easily applied to a circular plate, such as the one included in
Figure 4.
with
being the bending of the circular plate in polar coordinates, and
the distributed load applied to its upper surface. Moreover, the boundary constraints for the plate center and perimeter
can be expressed as follows:
and
Unlike the previous case, the bending solution for the fully clamped circular sensor can be obtained in a purely analytical way. By differentiating the bending solution for a simply supported plate and forcing the slope to nullify at the boundary, as imposed by (17), it is possible to calculate the bending moments along the plate edges. The final solution can be calculated by adding the deflection produced by the moments along the edges to the initial simply supported bending equation, obtaining the following expression:
2.1.3. Modelling Results Comparison
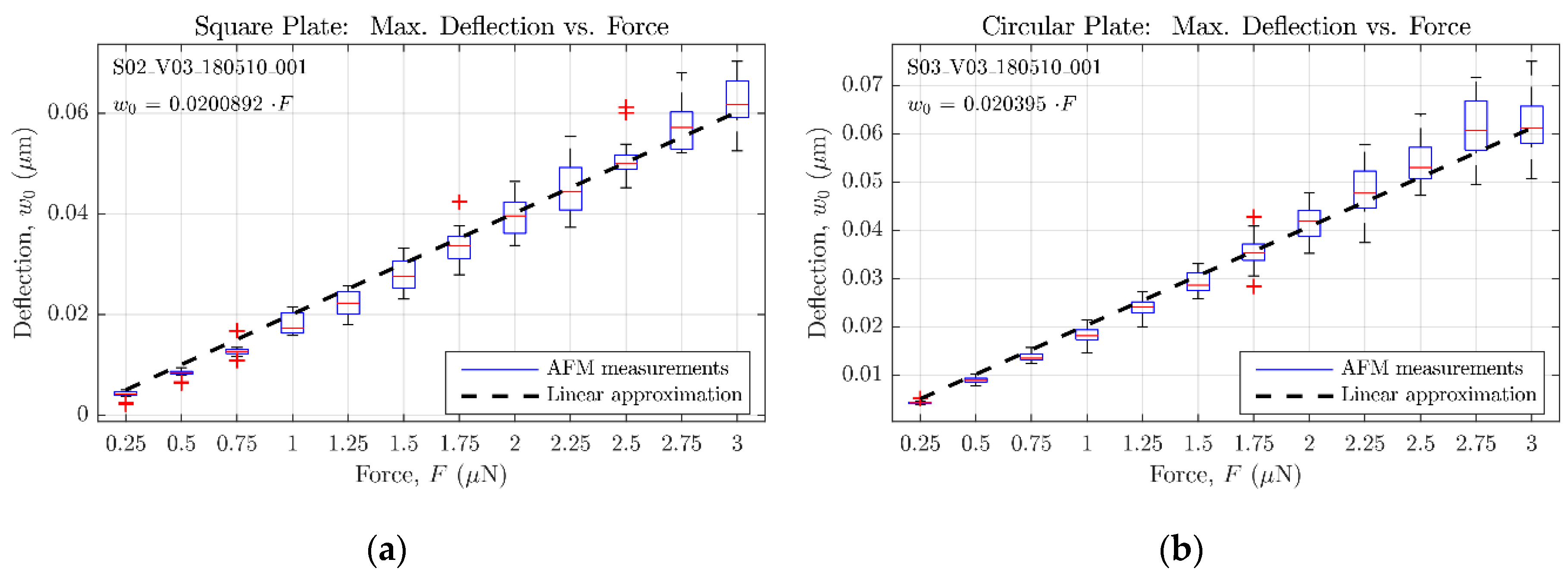
In order to characterize the accuracy of the bending solutions presented in the previous subsections, the responses provided by analytical equations have been compared with the ones obtained from equivalent finite elements (FE) models; so that the full scale error (FSE) between both modelling approaches could be calculated.
Two capacitive polysilicon-based
MEMS pressure sensors, circular and square-shaped, have been designed. A self-developed computed aided design (CAD) tool described in
Section 2.2 has been used for this purpose. An initial design constraint has been imposed to both sensors, forcing their dimensioning to reach an equal nominal capacitance of
.
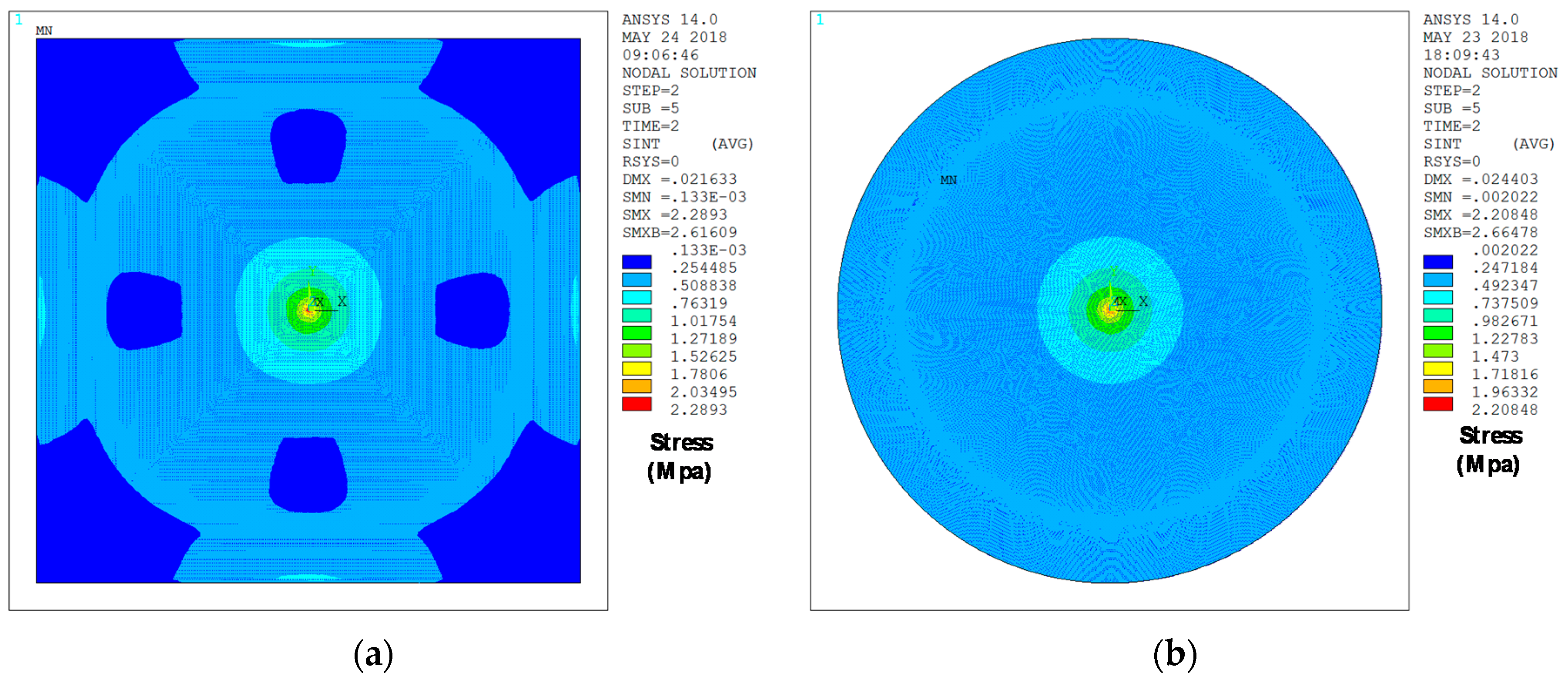
As can be seen in
Table 2 and
Table 3, two different FE models with variable complexity have been developed in ANSYS, in order to perform displacement versus force simulations and compare the resulting bending data with the response anticipated by the analytical expressions (14) and (18). A first FE model, referred as “simple”, consists of a flat square or circular plate fully clamped along its edges. This model requires low computational time in ANSYS to achieve a complete bending characterization of the sensor, because of its relatively low complexity. On the other hand, the second FE model, named “complex”, presents the exact same topology as the prototype sensor fabricated in PolyMUMPS technology. In this case, hole and dimple elements have been added because of the requirement imposed by the manufacturer for diaphragms larger than
[
13]. The former elements are square-shaped through-holes with a side length of
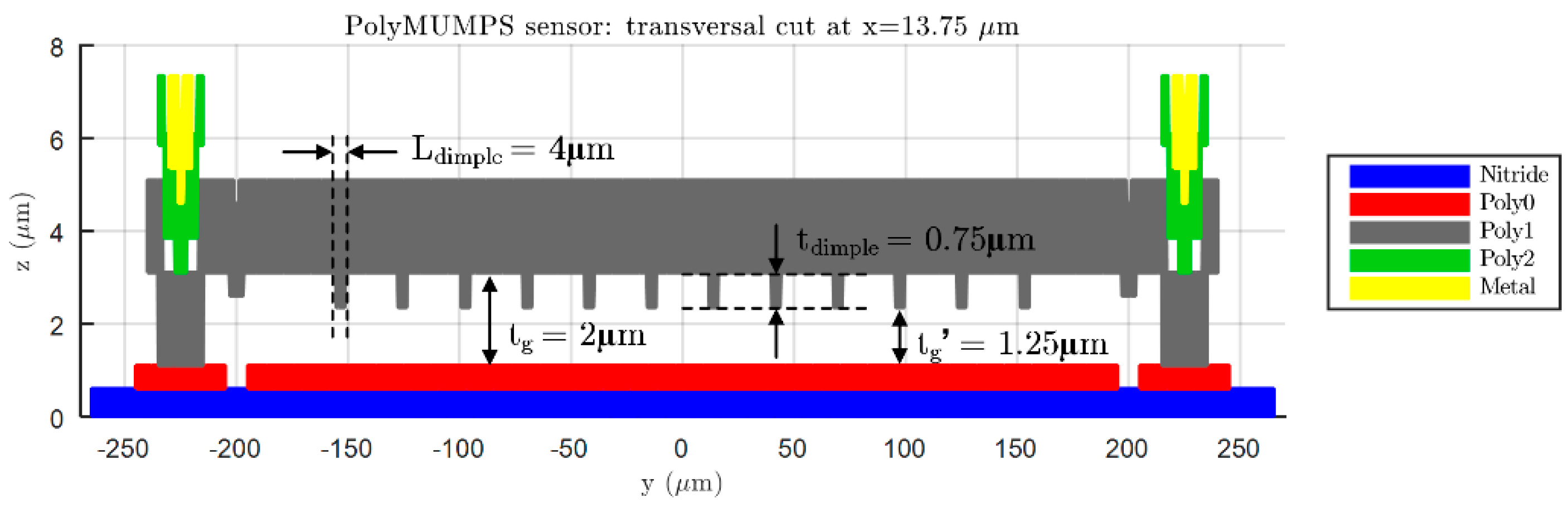
, required to provide shorter release etch paths for the removal of the sacrificial layer, as can be seen in
Figure 5. The latter elements are polysilicon elements of
height, placed under the suspended diaphragm in order to limit the contact surface and reduce the plate stiction occurrence [
13], as showcased in
Figure 6. Moreover, a lateral opening of side
has been added to the top plate anchoring structure, in order to facilitate the bottom plate electrical routing to the sensor bonding PADS. As expected, the greater complexity of this later model provokes a significant increase of the simulation time required to characterize its bending versus force behavior in ANSYS. However, the “complex” FE model produces more accurate results, showcasing an increased sensor sensitivity to the applied load, caused by the presence of hole cavities on the top plate; as well as a realistic contact point between the plates obtained under the presence of dimple elements, which reduce the plates gap in
[
13].
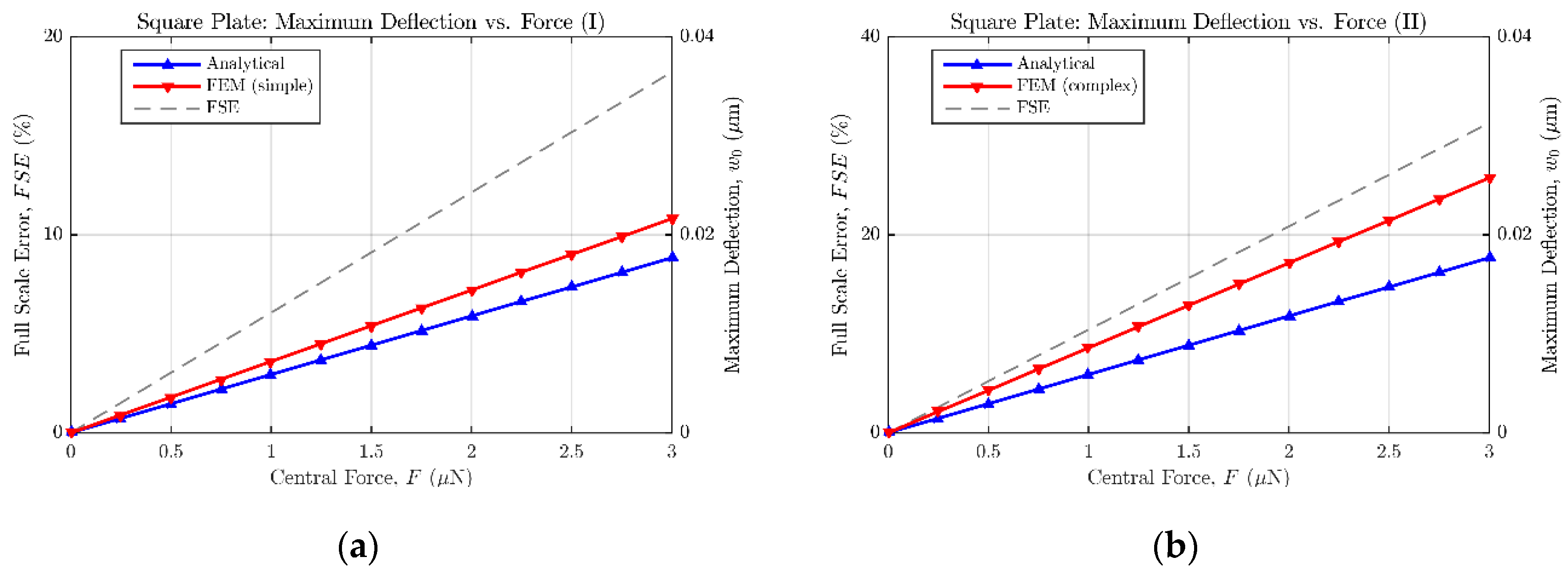
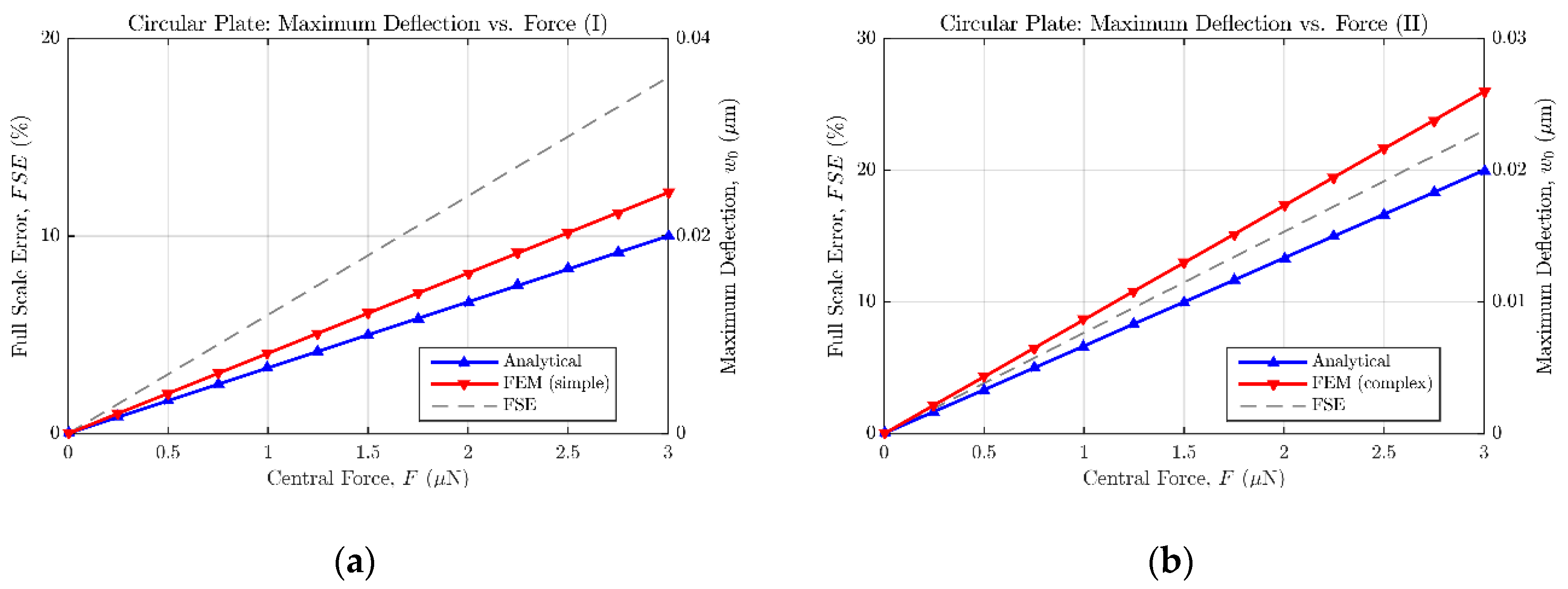
As a result of the aforementioned modelling,
Figure 7 and
Figure 8 show the maximum bending estimation against the concentrated force applied to the sensors described in
Table 2 and
Table 3, respectively. Both the analytical and FE responses are presented in each figure to ease the comparison between models. Additionally, the full scale error value is provided to quantify the mismatch between both models. According to the collected results, it can be stated that the analytical expressions (14) and (18) tend to underestimate the bending of the plates, when compared with the results of the FE models. However, because of their easy translation to the mathematical software (Matlab) and their fast evaluation, the analytical approach can still be considered an accurate-enough method to estimate the sensor behavior in an efficient way, previous to the FE model implementation in ANSYS. Alternative numerical approximation methods can be used to enhance the achieved exactitude, at the expense of immediacy, increased complexity, and higher computing requirements.
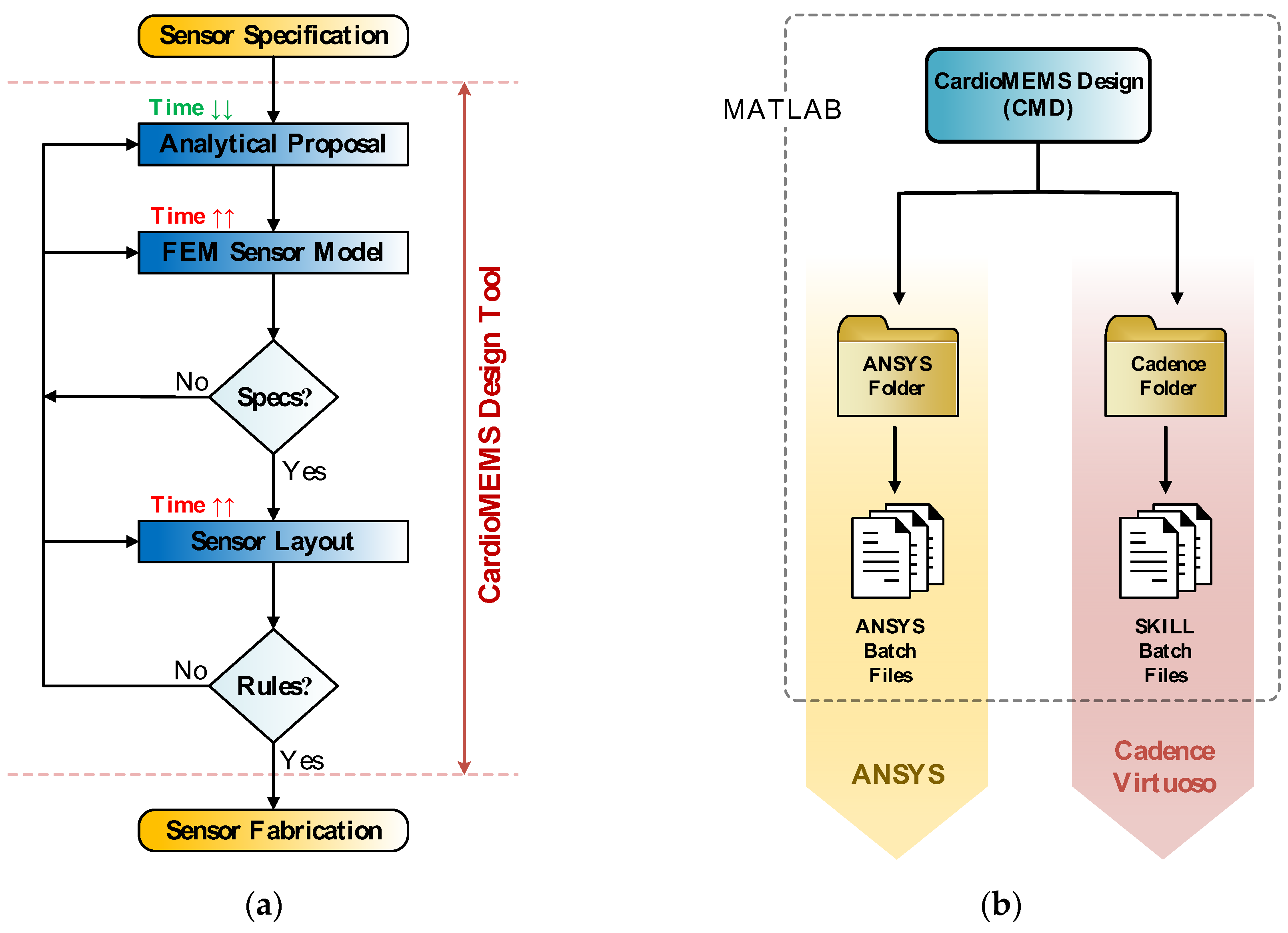
2.2. CardioMEMS Design Tool
MEMS sensor design flow, as shown in
Figure 9a, comprises three main stages. First, a rough sensor design proposal is obtained by the evaluation of a set of analytical expressions, while satisfying the initial design constraints. Second, the sensor realistic 3D structure is implemented in a modelling software for FE analysis; in order to perform the necessary simulations to achieve an accurate characterization of the device behavior. Finally, the third stage involves the sensor layout description in the corresponding technology layers being sent to the manufacturer. CardioMEMS Design (CMD) is a Matlab-based computer aided design (CAD) tool developed with the aim of automating the aforementioned design steps, as well as providing a friendly interface to guide the user through the sensor design process [
14,
15]. Thus, as exposed in
Figure 9b, CMD provides as outputs the set of files required to export the designed sensor to ANSYS and Cadence Virtuoso working environments, to perform different FE analyses and build the sensor layer description, respectively.
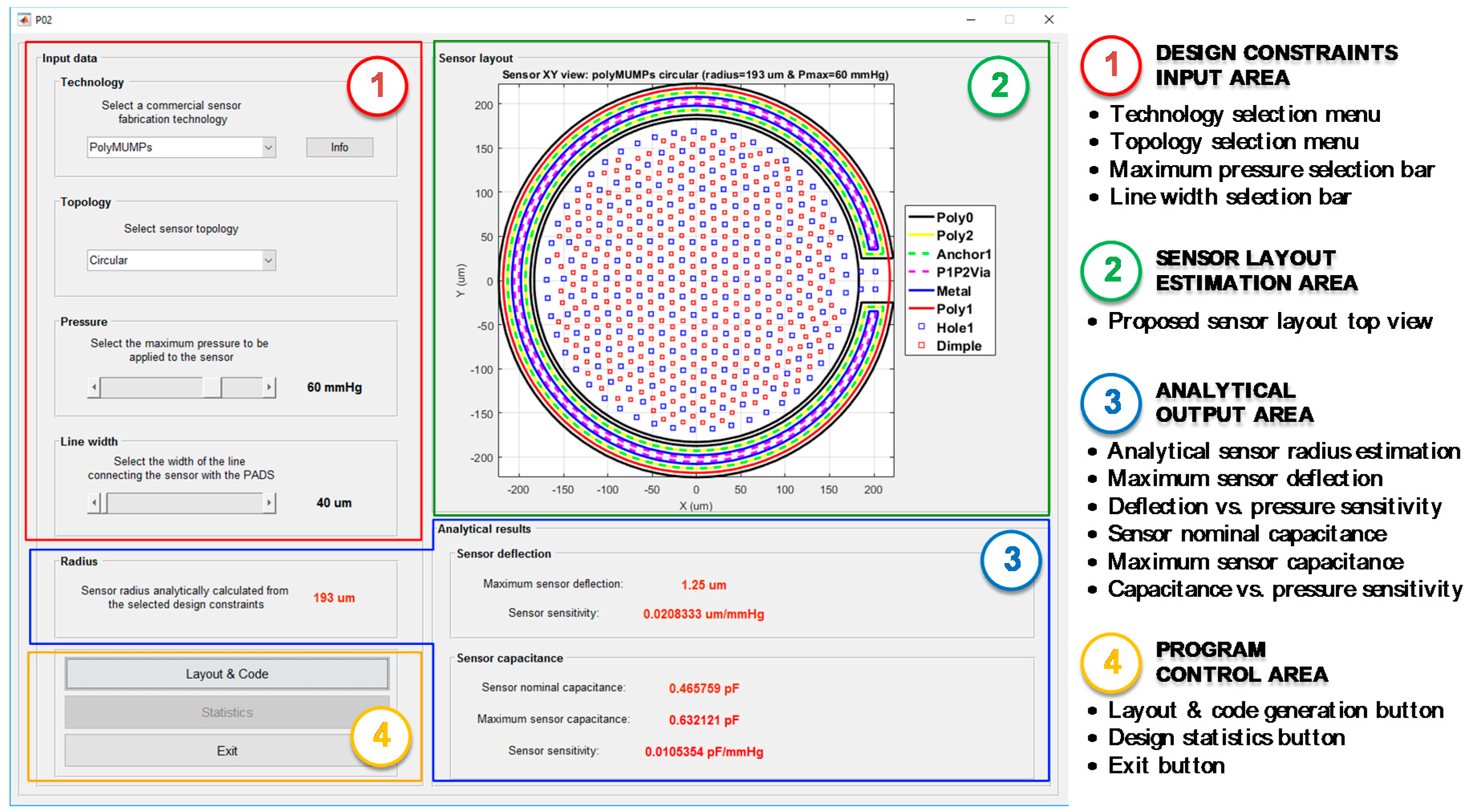
The use of CMD to perform the complete design of a capacitive MEMS pressure sensor for ISR-monitoring iStents is described next, in order to present its functionalities and features. First, as can be seen in
Figure 10, CMD requires the definition of various input parameters to provide an initial sensor design, including the selection of a fabrication technology included in the database, the preferred sensor topology, the maximum pressure to be borne by the transducer, and the desired width of the line used to physically connect the sensor to the bonding PADs.
In the case of our prototype sensors, the selected MEMS technology has been PolyMUMPS by MEMSCAP, a mature and reliable surface-micromachining fabrication process, developed to accommodate a wide variety of MEMS structures. PolyMUMPS uses eight masks to define the topology of seven physical layers: three polysilicon layers, a metal (Au) layer, and two phospho-silicate glass (PSG) sacrificial layers [
13]. It is important to keep in mind that the information about any new fabrication process must be added to the CMD database to allow for the use of that particular technology. Second, any MEMS pressure sensors intended to be used for mild ISR detection in a distal ramification of the pulmonary artery must face pressures in the range of
[
16], while presenting the maximum achievable sensitivity to pressure changes. Thus, a circular sensor topology has been chosen initially, because of its higher sensitivity compared with a squared-shaped sensor of the same area [
14]. Finally, a 40 µm wide Polysilicon line has been selected to physically connect the sensor to the bonding PADs.
Table 4 includes a summary of the input parameters introduced in CMD’s Design Constraints Area.
Once the input parameters have been defined, CMD determines the optimum sensor radius
by evaluation of the analytical equations for the deflection of a circular or square plate under a uniformly applied pressure. Internally, the program considers that the peak deflection, the contact between the suspended top plate and the fixed back plate, is achieved when the maximum pressure is applied to the sensor, thus imposing an additional constraint of
. As can be seen in
Figure 10, the main performance parameters for the proposed sensor are displayed in the analytical output area of the program, including the sensor radius (
), its maximum deflection (
) and deflection versus pressure sensitivity (
), together with its nominal (
) and maximum capacitances (
) and the capacitance versus pressure sensitivity (
). Because of the relative simplicity of the analytical models used, every time one of the input parameters is modified, the output parameters are quickly recalculated and shown to the user. This allow the designer to evaluate different sensor technologies, topologies, and pressure ranges, getting an initial estimation of their performance in an efficient and agile way.
The Layout & Code bottom in the CMD main screen becomes active right after the initial design proposal has been presented to the user. By clicking on it, CMD internally performs a design adjustment; now taking into account the set of design rules provided by the technology manufacturer. Additionally, CMD considers the functional limitations from both ANSYS and Cadence Virtuoso while performing the design rearrangement, in order to guarantee its compatibility with both environments.
In the case of PolyMUMPS sensors, the optimum location for both holes and dimples is selected by CMD through the evaluation of a geometric distribution algorithm, based on Delaunay triangulation theory [
17]. This algorithm minimizes the number of elements added to the structure, while guarantying the fulfillment of the design rules imposed by the technology. The rearranged sensor design is presented to the user in the sensor Layout Estimation Area, providing a top view of the layout displaying all the required technology layers for its fabrication, as shown in
Figure 10.
Additionally, CMD saves relevant statistical information about the layers geometry. This information is accessible by the
Statistics button in the program control area, as indicated in
Figure 10, only after the
Layout & Code operation has concluded.
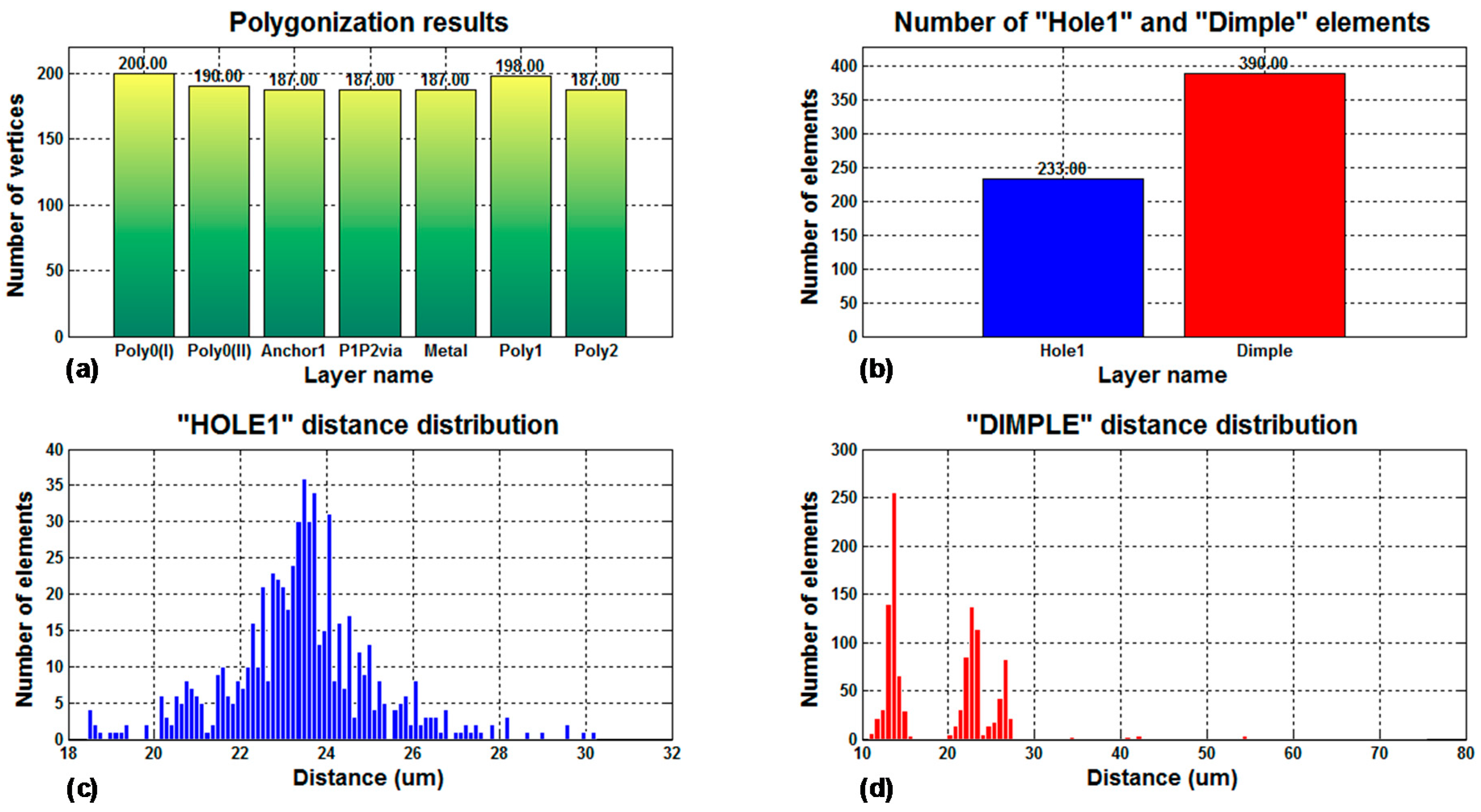
Figure 11 includes a summary of the statistical data analyzed by the program for a PolyMUMPS-based sensor design. More concretely,
Figure 11a provides the number of vertices used to polygonise the sensor defining layouts, which is a Cadence Virtuoso limiting operation factor. As can be noted, CMD fixes the number of vertices to a maximum value of 200 per layer, so Cadence Virtuoso constraints are not infringed. Moreover, CMD takes into account the spacing limitations between hole and dimple elements, defined in the provided PolyMUMPS documentation as
and
, respectively [
13]. The distance distribution for each type of element is displayed in
Figure 11c,d; while
Figure 11b indicates the total number of elements added to the basic sensor structure. This set of data is provided to the designer in order to quickly check that the design restrictions established by both the manufacturer and the software suites are thoroughly satisfied.
As previously mentioned, and illustrated in
Figure 9b, one of the main goals of CMD is to facilitate the sensor design translation to ANSYS and Cadence Virtuoso, so that the final stages of the design flow can be completed. Hence, after each design completion, CMD generates two output folders, denoted as
codeAnsys and
codeCadence, comprising a series of files suitable to automatically export the sensor design to those platforms.
The
codeAnsys folder contains seven files, with three of them being auto-executable files, while the remaining four files have an auxiliary purpose. Those three main batch code files, once individually loaded in ANSYS, build the 3D solid model of the sensor, optimize its FE meshing, and perform different simulations to completely characterize the device behavior. For instance, one of the files configures the program solver to calculate the sensor deflection and equivalent capacitance for a uniformly applied pressure in the intended operation range of
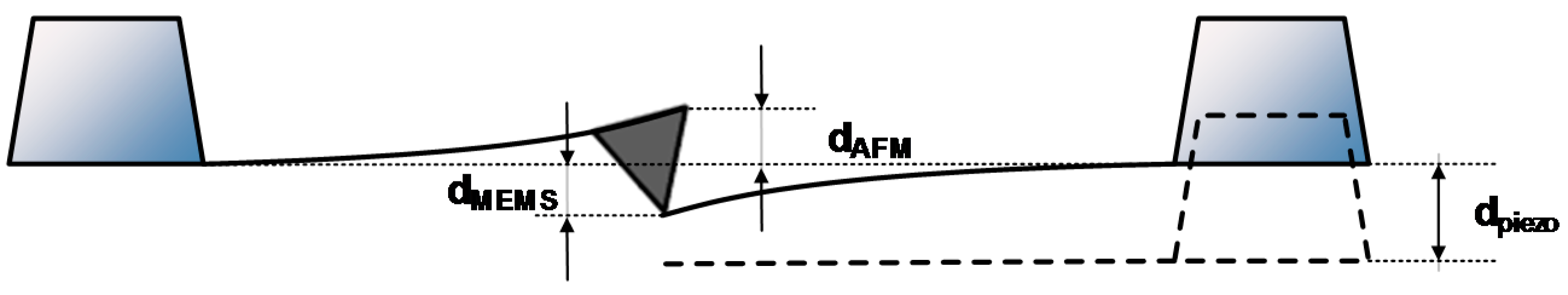
Similarly, a second main file sets the solver to evaluate the sensor deflection and capacitance under the presence of a central force load (
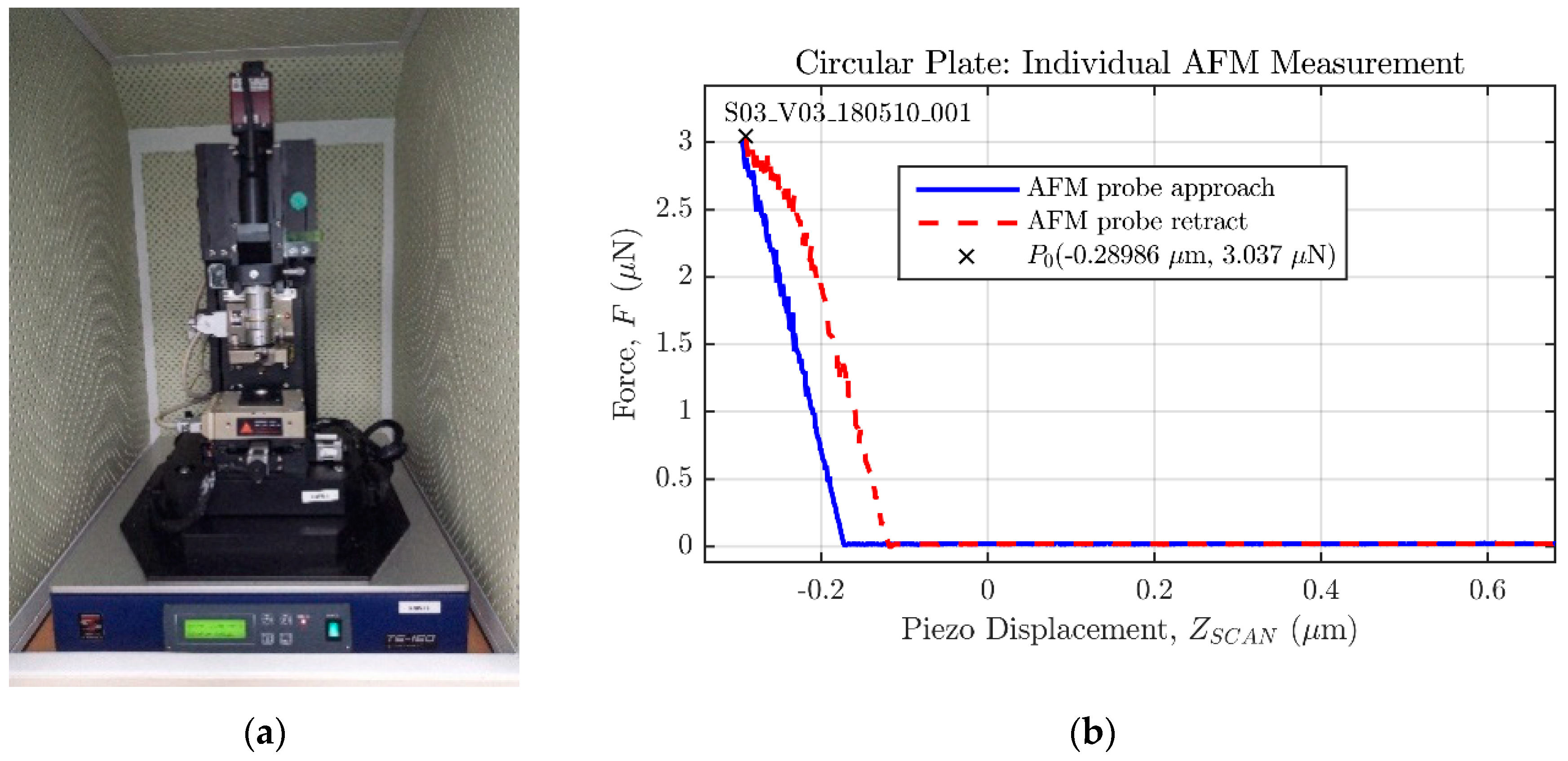
), similar to the one that can be applied by an AFM microscope operating in contact mode [
6,
7,
8,
9]; and described in detail in
Section 2.1. Finally, the last main code file arranges a modal analysis to determine the natural frequencies of the structure.
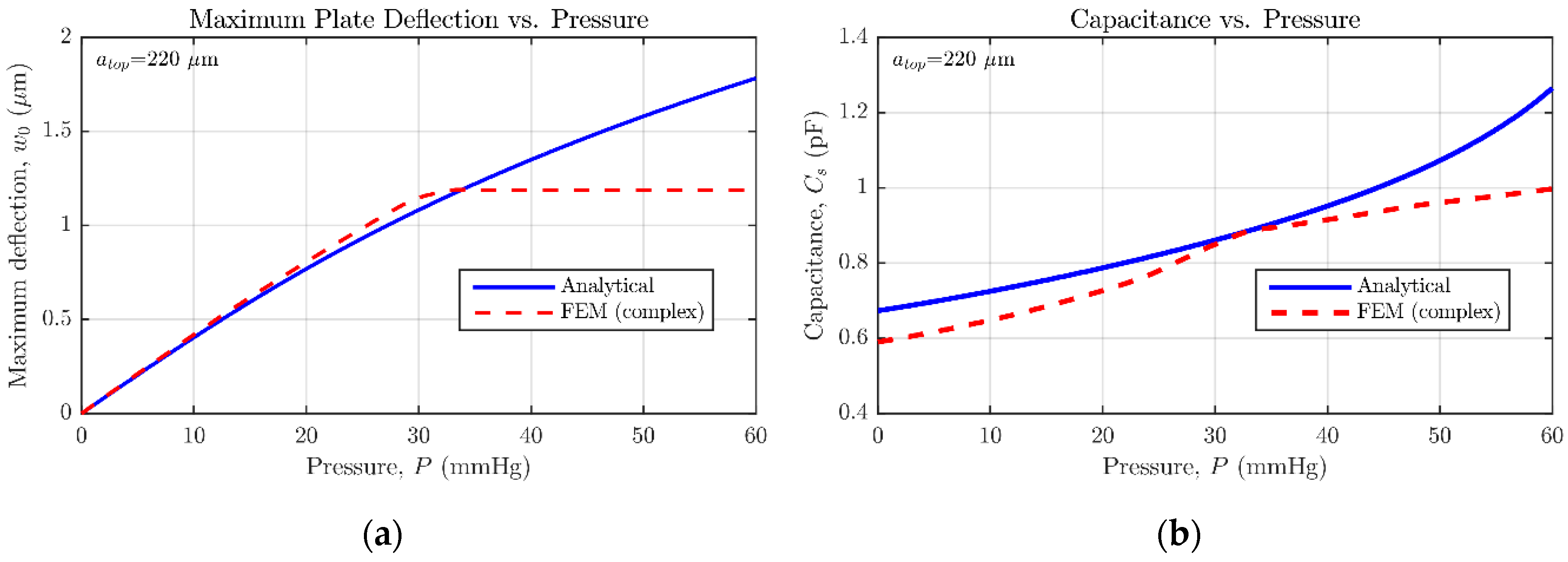
In
Figure 12, the central deflection and capacitance versus pressure simulation results are summed up. As can be observed, the simulation results given by ANSYS diverge slightly from those anticipated by the set of analytical expressions [
14]. It must be acknowledged by the user that CMD proposes an initial rough sensor geometry based on the evaluation of analytical/numerical equations, because of their high computational efficiency. On the other hand, the CMD-rearranged design built in ANSYS presents higher complexity, mainly due to the addition of hole and dimple elements, responsible for reducing the stiffness of the movable plate [
14] and limiting the effective gap distance to
, respectively. Furthermore, as detailed in
Figure 5 and
Figure 6, the realistic sensor model requires a moderately smaller backplate compared with the suspended one, which negatively affects the sensor nominal capacitance (
Figure 12b). It can be perceived how the increased complexity of the structure contributes to locating the contact point in the range of measurable pressures; meaning that the initial sensor proposal underestimates the device sensitivity. However, for some applications, it can be desired to force the sensor operation entirely in contact mode, so a lineal capacitance response can be achieved [
18]. The designer can take advantage of this behavior, being able to get exponential or linear capacitance versus pressure responses just by properly selecting the maximum detectable pressure at the beginning of the design flow.
The
codeCadence folder contains a unique auto executable SKILL batch file, supported by eighteen auxiliary files used to individually define the geometry of each fabrication layer. After being loaded in Cadence Virtuoso, the main batch file conducts the drawing of the necessary layers to create an adequate GDSII stream format file to be sent to the manufacturer.
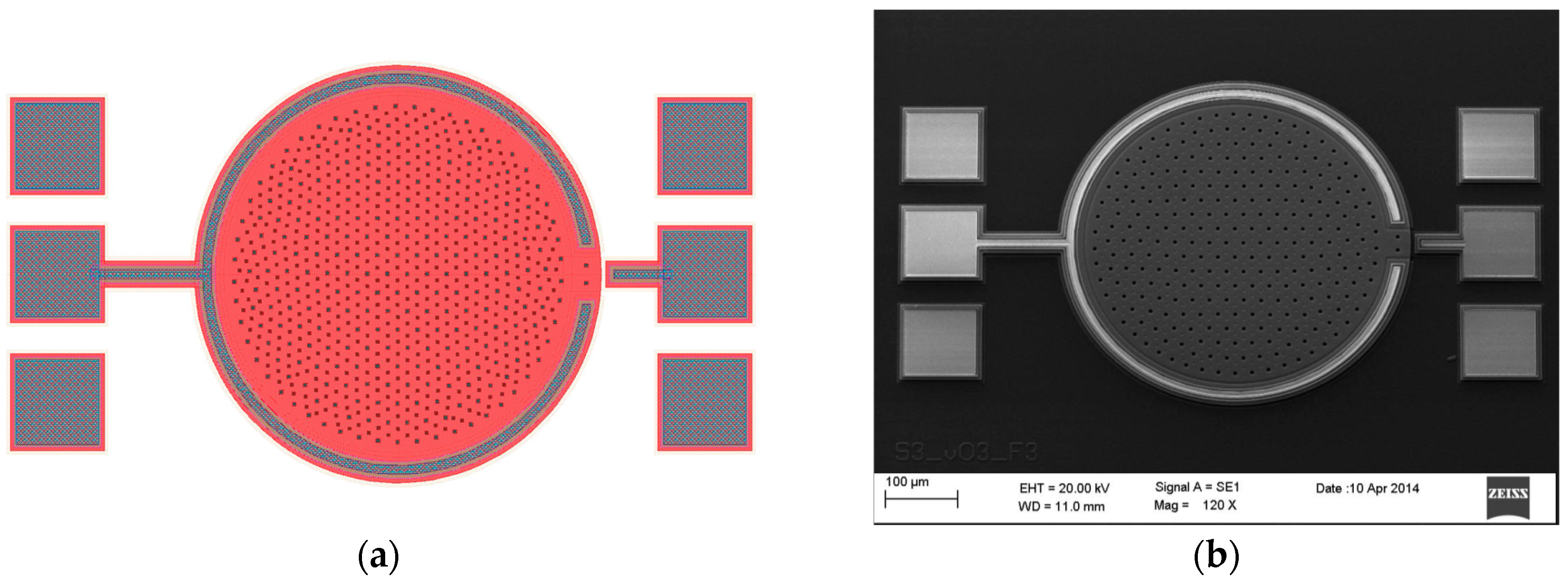
Figure 13a shows the layout view of the prototype sensor automatically built in Candence Virtuoso’s Layout Suite; where the bonding PADs are the only elements manually added by the designer. Besides, for comparison purposes,
Figure 13b presents a scanning electron microscope (SEM) image of the fabricated sensor, defined by the layout in
Figure 13a.