Electromagnetic Tracking System for Medical Micro Devices: A Review
Abstract
1. Introduction
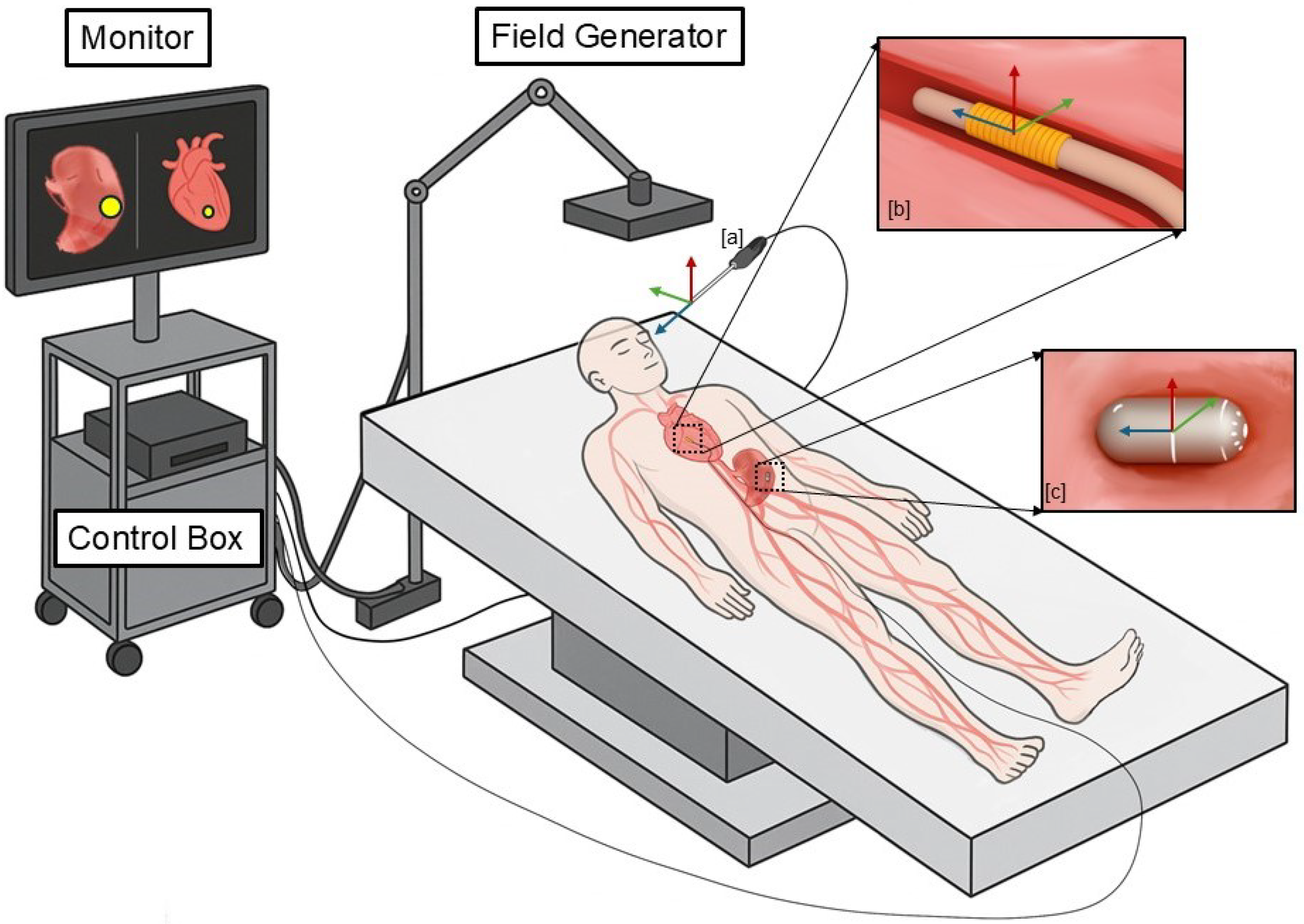
2. Working Principle of Electromagnetic Tracking System
3. Components of Electromagnetic Tracking System
3.1. Field Generators
3.2. Sensors
3.3. Source of Error
4. Current Research Process
4.1. Induction Principle
4.2. Integrated Magnetic Sensors
4.3. Magnetic Field Camera
5. Commercial Device and Clinical Application
5.1. Commercial Device
5.2. Clinical Application
5.3. Comparison

| Field Generator | Sensor | Accuracy | |||||
|---|---|---|---|---|---|---|---|
| Type | Shape | Work Space/mm | Size/mm | Degree of Freedom | Update Rate/Hz | Position/mm | Orientation/° |
| Aurora [100] | |||||||
| Planar FG | Cube | 500 × 500 × 500 | 0.3 × 2.5 | 5 | 40 | 0.70 | 0.20 |
| Dome | 660 | 0.3 × 2.5 | 5 | 40 | 1.10 | 0.20 | |
| Planar FG | Cube | 500 × 500 × 500 | 1.8 × 9 | 6 | 40 | 0.48 | 0.30 |
| Dome | 660 | 1.8 × 9 | 6 | 40 | 0.70 | 0.30 | |
| Tabletop FG | Dome | 600 | 0.3 × 2.5 | 5 | 40 | 1.20 | 0.50 |
| Dome | 600 | 1.8 × 9 | 6 | 40 | 0.80 | ||
| Window FG | Cylinder | 250 | 0.3 × 2.5 | 5 | 40 | N/A | N/A |
| Dome | 600 | 0.3 × 2.5 | 5 | 40 | N/A | N/A | |
| Window FG | Cylinder | 250 | 1.8 × 9 | 6 | 40 | N/A | N/A |
| Dome | 600 | 1.8 × 9 | 6 | 40 | N/A | N/A | |
| 3D Guidance [101] | |||||||
| Mid-Range | Cube | 560 × 460 × 600 | 0.56 × 12 0.9 × 7.25 1.5 × 7.7 2.0 × 9.9 7.9 × 8 × 19.8 | 6 | 80 | 1.40 | 0.50° |
| Short-Range | Cube | 560 × 460 × 600 | 0.56 × 12 0.9 × 7.25 1.5 × 7.7 2.0 × 9.9 | 6 | 80 | N/A | N/A |
| Polhemus [75] | |||||||
| Viper | N/A | 1820 | 1.8 | 6 | 240 | 0.38 (static) | (static) |
| Fastrak | N/A | N/A | 1.8 | 6 | 120 | 0.76 (static) | (static) |
| Patriot | N/A | N/A | 1.8 | 6 | 60 | 1.52 (static) | (static) |
| LIBERTY | N/A | N/A | 1.8 | 6 | 240 | 0.76 (static) | (static) |
| G4 | N/A | N/A | 1.8 | 6 | 120 | 2.0 (static) | (static) |
| Aimooe [76] | |||||||
| Magpilot | Cube | 300 × 300 × 300 | 0.45 × 8.0 0.45 × 5.0 1.5 × 10.0 | 5 5 6 | 80 | 2.0 | N/A |
| OMMO [78] | |||||||
| Orbit + Axon | Sphere | 650 | 21.20 × 5.30 × 4.10 26.30 × 5.30 × 4.10 | 5 6 | N/A | 0.2 0.17 | 0.10° 0.08° |
6. Discussion
- 1.
- Simultaneous localization and navigation: Future research should focus on developing the simultaneous localization and navigation system with high accuracy performance. In some areas where expensive image-guided modalities are not available, some difficult minimally invasive surgeries can be performed with the convenience of EMTS.
- 2.
- Sensor Fusion: EMTS integrated with other image-guided modalities could enable high-accuracy localization and trajectory guidance. This will require advanced sensor fusion strategies and algorithms to maintain robustness in anatomically complex and dynamic surgical environments.
- 3.
- Distortion compensate algorithms: There remains a critical requirement for adaptive, real-time algorithms to compensate magnetic field distortions induced by external disturbances. Emerging approaches enhance localization accuracy and robustness, including both hardware and software solutions.
- 4.
- A standardized evaluation protocol: A standardized evaluation protocol should incorporate well-defined test phantoms, consistent reference frames, and harmonized performance metrics to enable reproducible benchmarking across systems and institutions. Establishing such elements would not only facilitate objective comparison but also accelerate regulatory approval and clinical translation of EMTS.
- 5.
- Cost-effective: Currently, the overall cost of EMTS remains high. Achieving economic viability will require the optimization of hardware architectures and sensor designs while maintaining performance standards. Balancing performance and affordability is critical to facilitate widespread implementation across diverse clinical scenarios.
7. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| EMTS | Electromagnetic Tracking System |
| MIS | Minimally invasive surgery |
| CT | Computed tomography |
| MRI | Magnetic Resonance Imaging |
| US | Ultrasound |
| OCT | Optical Coherence Tomography |
| IVUS | Intravascular Ultrasound |
References
- Fuchs, K.H. Minimally Invasive Surgery. Endoscopy 2002, 34, 154–159. [Google Scholar] [CrossRef]
- Konda, R.; Brumfiel, T.A.; Bercu, Z.L.; Grossberg, J.A.; Desai, J.P. Robotically steerable guidewires—Current trends and future directions. Sci. Robot. 2025, 10, eadt7461. [Google Scholar] [CrossRef]
- Teman, N.R.; Hawkins, R.B.; Charles, E.J.; Mehaffey, J.H.; Speir, A.M.; Quader, M.A.; Ailawadi, G. Minimally Invasive vs. Open Coronary Surgery: A Multi-Institutional Analysis of Cost and Outcomes. Ann. Thorac. Surg. 2021, 111, 1478–1484. [Google Scholar] [CrossRef]
- Matsuda, H.; Sawa, Y.; Takahashi, T.; Hirata, N.; Ohtake, S. Minimally Invasive Cardiac Surgery: Current Status and Perspective. Artif. Organs 1998, 22, 759–764. [Google Scholar] [CrossRef] [PubMed]
- Sierink, J.C.; Treskes, K.; Edwards, M.J.R.; Beuker, B.J.A.; den Hartog, D.; Hohmann, J.; Dijkgraaf, M.G.W.; Luitse, J.S.K.; Beenen, L.F.M.; Hollmann, M.W.; et al. Immediate total-body CT scanning versus conventional imaging and selective CT scanning in patients with severe trauma (REACT-2): A randomised controlled trial. Lancet 2016, 388, 673–683. [Google Scholar] [CrossRef] [PubMed]
- Rocca, M.A.; Preziosa, P.; Barkhof, F.; Brownlee, W.; Calabrese, M.; De Stefano, N.; Granziera, C.; Ropele, S.; Toosy, A.T.; Vidal-Jordana, À.; et al. Current and future role of MRI in the diagnosis and prognosis of multiple sclerosis. Lancet Reg. Health Eur. 2024, 44, 100978. [Google Scholar] [CrossRef] [PubMed]
- Cooperberg, P.L. Clinical ultrasound: A comprehensive guide. Lancet 2002, 359, 87. [Google Scholar] [CrossRef]
- Cleary, K.; Peters, T.M. Image-Guided Interventions: Technology Review and Clinical Applications. Annu. Rev. Biomed. Eng. 2010, 12, 119–142. [Google Scholar] [CrossRef] [PubMed]
- Aumann, S.; Donner, S.; Fischer, J.; Müller, F. Optical Coherence Tomography (OCT): Principle and Technical Realization. In High Resolution Imaging in Microscopy and Ophthalmology: New Frontiers in Biomedical Optics; Bille, J.F., Ed.; Springer International Publishing: Cham, Switzerland, 2019; pp. 59–85. [Google Scholar]
- Xu, J.; Lo, S. Fundamentals and role of intravascular ultrasound in percutaneous coronary intervention. Cardiovasc. Diagn. Ther. 2020, 10, 1358. [Google Scholar] [CrossRef]
- Cavaliere, M.; McVeigh, O.; Jaeger, H.A.; Hinds, S.; O’Donoghue, K.; Cantillon-Murphy, P. Inductive Sensor Design for Electromagnetic Tracking in Image Guided Interventions. IEEE Sens. J. 2020, 20, 8623–8630. [Google Scholar] [CrossRef]
- Qureshi, A.; Vergis, A.; Jimenez, C.; Green, J.; Pryor, A.; Schlachta, C.M.; Okrainec, A. MIS training in Canada: A national survey of general surgery residents. Surg. Endosc. 2011, 25, 3057–3065. [Google Scholar] [CrossRef]
- Sherman, W.R.; Craig, A.B. Chapter 4—Input: Interfacing the Participant(s) with the Virtual World. In Understanding Virtual Reality (Second Edition), 2nd ed.; Sherman, W.R., Craig, A.B., Eds.; The Morgan Kaufmann Series in Computer Graphics; Morgan Kaufmann: Boston, MA, USA, 2018; pp. 190–256. [Google Scholar]
- Peng, C.; Cai, Q.; Chen, M.; Jiang, X. Recent Advances in Tracking Devices for Biomedical Ultrasound Imaging Applications. Micromachines 2022, 13, 1855. [Google Scholar] [CrossRef]
- Bi, S.; Gu, Y.; Zou, J.; Wang, L.; Zhai, C.; Gong, M. High Precision Optical Tracking System Based on near Infrared Trinocular Stereo Vision. Sensors 2021, 21, 2528. [Google Scholar] [CrossRef]
- Andria, G.; Attivissimo, F.; Di Nisio, A.; Lanzolla, A.M.L.; Larizza, P.; Selicato, S. Development and performance evaluation of an electromagnetic tracking system for surgery navigation. Measurement 2019, 148, 106916. [Google Scholar] [CrossRef]
- Monahan, E.; Shimada, K. Computer-aided navigation for arthroscopic hip surgery using encoder linkages for position tracking. Int. J. Med. Robot. Comput. Assist. Surg. 2006, 2, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Falster, C.; Mørkenborg, M.D.; Thrane, M.; Clausen, J.; Arvig, M.; Brockhattingen, K.; Biesenbach, P.; Paludan, L.; Nielsen, R.W.; Nhi Huynh, T.A.; et al. Utility of ultrasound in the diagnostic work-up of suspected pulmonary embolism: An open-label multicentre randomized controlled trial (the PRIME study). Lancet Reg. Health Eur. 2024, 42, 100941. [Google Scholar] [CrossRef]
- Christ, R.D.; Wernli, R.L. Chapter 16—Acoustic Positioning. In The ROV Manual, 2nd ed.; Christ, R.D., Wernli, R.L., Eds.; Butterworth-Heinemann: Oxford, UK, 2014; pp. 425–451. [Google Scholar]
- Kanlić, E.M.; DeLaRosa, F.; Pirela-Cruz, M. Computer Assisted Orthopaedic Surgery—CAOS. Biomol. Biomed. 2006, 6, 7–14. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Freyschlag, C.F.; Kerschbaumer, J.; Eisner, W.; Pinggera, D.; Brawanski, K.R.; Petr, O.; Bauer, M.; Grams, A.E.; Bodner, T.; Seiz, M.; et al. Optical Neuronavigation without Rigid Head Fixation During Awake Surgery. World Neurosurg. 2017, 97, 669–673. [Google Scholar] [CrossRef]
- Pasku, V.; De Angelis, A.; De Angelis, G.; Arumugam, D.D.; Dionigi, M.; Carbone, P.; Moschitta, A.; Ricketts, D.S. Magnetic Field-Based Positioning Systems. IEEE Commun. Surv. Tutor. 2017, 19, 2003–2017. [Google Scholar] [CrossRef]
- Poulin, F.; Amiot, L.P. Interference during the use of an electromagnetic tracking system under OR conditions. J. Biomech. 2002, 35, 733–737. [Google Scholar] [CrossRef]
- Nelson, B.J.; Bendok, B.R.; Turcotte, E.L.; Batjer, H.H. Remote magnetic navigation enables precision telesurgery. Sci. Robot. 2024, 9, eado3187. [Google Scholar] [CrossRef] [PubMed]
- Yaniv, Z.; Wilson, E.; Lindisch, D.; Cleary, K. Electromagnetic tracking in the clinical environment. Med. Phys. 2009, 36, 876–892. [Google Scholar] [CrossRef]
- Floridi, C.; Radaelli, A.; Abi-Jaoudeh, N.; Grass, M.; De Lin, M.; Chiaradia, M.; Geschwind, J.F.; Kobeiter, H.; Squillaci, E.; Maleux, G.; et al. C-arm cone-beam computed tomography in interventional oncology: Technical aspects and clinical applications. Radiol. Medica 2014, 119, 521–532. [Google Scholar] [CrossRef]
- Modarai, B.; Haulon, S.; Ainsbury, E.; Böckler, D.; Vano-Carruana, E.; Dawson, J.; Farber, M.; Van Herzeele, I.; Hertault, A.; van Herwaarden, J.; et al. Editor’s Choice—European Society for Vascular Surgery (ESVS) 2023 Clinical Practice Guidelines on Radiation Safety. Eur. J. Vasc. Endovasc. Surg. 2023, 65, 171–222. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.H.; Choi, H.S.; Keum, B.; Chun, H.J. Robotics in Gastrointestinal Endoscopy. Appl. Sci. 2021, 11, 11351. [Google Scholar] [CrossRef]
- Fu, Y.; Li, Y.; Wu, X.; Liu, M.; Guo, Y.X. Wireless Electromagnetic Tracking System for Wireless Capsule Endoscope. IEEE Trans. Instrum. Meas. 2025, 74, 1–13. [Google Scholar] [CrossRef]
- Wang, M.; Song, S.; Liu, J.; Meng, M.Q.H. Multipoint Simultaneous Tracking of Wireless Capsule Endoscope Using Magnetic Sensor Array. IEEE Trans. Instrum. Meas. 2021, 70, 1–10. [Google Scholar] [CrossRef]
- Franz, A.M.; Haidegger, T.; Birkfellner, W.; Cleary, K.; Peters, T.M.; Maier-Hein, L. Electromagnetic Tracking in Medicine—A Review of Technology, Validation, and Applications. IEEE Trans. Med. Imaging 2014, 33, 1702–1725. [Google Scholar] [CrossRef]
- Sorriento, A.; Porfido, M.B.; Mazzoleni, S.; Calvosa, G.; Tenucci, M.; Ciuti, G.; Dario, P. Optical and Electromagnetic Tracking Systems for Biomedical Applications: A Critical Review on Potentialities and Limitations. IEEE Rev. Biomed. Eng. 2020, 13, 212–232. [Google Scholar] [CrossRef] [PubMed]
- Than, T.D.; Alici, G.; Zhou, H.; Li, W. A Review of Localization Systems for Robotic Endoscopic Capsules. IEEE Trans. Biomed. Eng. 2012, 59, 2387–2399. [Google Scholar] [CrossRef] [PubMed]
- Sauer, B.C.; Dürrbeck, C.; Bert, C. Electromagnetic tracking in interstitial brachytherapy: A systematic review. Front. Phys. 2022, 10, 956983. [Google Scholar] [CrossRef]
- Vergne, C.; Madec, M.; Quirin, T.; Guzman, R.; Hemm, S.; Pascal, J. Electromagnetic Tracking System for Position and Orientation Detection of Deep Brain Stimulation Electrodes During Surgery. IEEE Trans. Biomed. Eng. 2025, 72, 1973–1982. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Guo, S.; Shen, B.; Lyu, C.; Ding, M.; Yang, P.; Zhang, Y.; Zhang, Y.; Liu, J. 5-DOF Microcoil Positioning System Utilizing Single-Axis Electromagnetic Transmitter. IEEE/ASME Trans. Mechatron. 2025, 1–12. [Google Scholar] [CrossRef]
- Sharma, S.; Ramadi, K.B.; Poole, N.H.; Srinivasan, S.S.; Ishida, K.; Kuosmanen, J.; Jenkins, J.; Aghlmand, F.; Swift, M.B.; Shapiro, M.G.; et al. Location-aware ingestible microdevices for wireless monitoring of gastrointestinal dynamics. Nat. Electron. 2023, 6, 242–256. [Google Scholar] [CrossRef]
- Dürrbeck, C.; Gulde, S.; Abu-Hossin, N.; Fietkau, R.; Strnad, V.; Bert, C. Influence and compensation of patient motion in electromagnetic tracking based quality assurance in interstitial brachytherapy of the breast. Med. Phys. 2022, 49, 2652–2662. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Xu, H.; Cao, S.D.; Wei, J.; Rhodes, D.; Siewerdsen, J.H.; Gonzalez, L.F.; Uneri, A. Guiding endovascular catheters using electromagnetic tracking and path-based registration for brain aneurysm treatment. In Proceedings of the Medical Imaging 2024: Image-Guided Procedures, Robotic Interventions, and Modeling, SPIE, San Diego, CA, USA, 19–22 February 2024; Siewerdsen, J.H., Rettmann, M.E., Eds.; International Society for Optics and Photonics: Bellingham, WA, USA, 2024; Volume 12928, p. 129280D. [Google Scholar]
- Esteves, D.S.; Melo, A.; Alves, S.; Durães, N.; Paiva, M.C.; Sequeiros, E.W. Magnetic Field-Assisted Orientation and Positioning of Magnetite for Flexible and Electrically Conductive Sensors. Micromachines 2025, 16, 68. [Google Scholar] [CrossRef] [PubMed]
- Cavaliere, M.; Jaeger, H.A.; O’Donoghue, K.; Cantillon-Murphy, P. Planar Body-Mounted Sensors for Electromagnetic Tracking. Sensors 2021, 21, 2822. [Google Scholar] [CrossRef]
- Barak, R.; Greenburg, B. Coil Geometry for an Electromagnetic Tracking System. US Patent Application No. 18/579,404, 10 October 2024. [Google Scholar]
- Peng, C.; Liu, L. Capsule Endoscope System and Its Magnetic Positioning Method. US Patent Application No. 18/550,992, 6 June 2024. [Google Scholar]
- Yang, L.; Yu, J.; Yang, S.; Wang, B.; Nelson, B.J.; Zhang, L. A Survey on Swarm Microrobotics. IEEE Trans. Robot. 2022, 38, 1531–1551. [Google Scholar] [CrossRef]
- Coey, J.M.D. Magnetism and Magnetic Materials; Cambridge University Press: Cambridge, UK, 2010. [Google Scholar]
- Gervasoni, S.; Pedrini, N.; Rifai, T.; Fischer, C.; Landers, F.C.; Mattmann, M.; Dreyfus, R.; Viviani, S.; Veciana, A.; Masina, E.; et al. A Human-Scale Clinically Ready Electromagnetic Navigation System for Magnetically Responsive Biomaterials and Medical Devices. Adv. Mater. 2024, 36, 2310701. [Google Scholar] [CrossRef]
- Teng, G.; Dong, S.; Zhou, J.; Liu, D.; Zeng, Y.; Wang, S.; Wang, H. Active Electromagnetic Tracking System with Directionally Uniform and High Accuracy Using 2-DoF Orientation Control of the Magnetic Source. IEEE Trans. Instrum. Meas. 2025, 74, 7509612. [Google Scholar] [CrossRef]
- Song, S.; Hu, C.; Li, B.; Li, X.; Meng, M.Q.H. An Electromagnetic Localization and Orientation Method Based on Rotating Magnetic Dipole. IEEE Trans. Magn. 2013, 49, 1274–1277. [Google Scholar] [CrossRef]
- Luo, L.; Guo, X.; Cheng, B.; Zhang, Z.; Jiang, D. Design of electromagnetic tracking system for puncture needle of an intelligent puncture robot. Am. J. Transl. Res. 2023, 15, 6404–6412. [Google Scholar]
- von Arx, D.; Nelson, B.J.; Boehler, Q. Orthogonal Pulse-Width-Modulation for Combined Electromagnetic Actuation and Localization. IEEE Robot. Autom. Lett. 2025, 10, 6424–6431. [Google Scholar] [CrossRef]
- Brauer, J.R. Hall Effect and Magnetoresistive Sensors. In Magnetic Actuators and Sensors; John Wiley & Sons: Hoboken, NJ, USA, 2014; pp. 175–199. [Google Scholar]
- Yao, S.; Luo, P.; Liu, L.; Yan, H.; Meng, M.Q.H. Fast-Adaptive Permanent Magnetic Positioning-based Navigation Framework for Continuum Robots in Colonoscopic Biopsy. IEEE/ASME Trans. Mechatron. 2025, 1–13. [Google Scholar] [CrossRef]
- Fischer, C.; Boehler, Q.; Nelson, B.J. Using Magnetic Fields to Navigate and Simultaneously Localize Catheters in Endoluminal Environments. IEEE Robot. Autom. Lett. 2022, 7, 7217–7223. [Google Scholar] [CrossRef]
- von Arx, D.; Fischer, C.; Torlakcik, H.; Pané, S.; Nelson, B.J.; Boehler, Q. Simultaneous Localization and Actuation Using Electromagnetic Navigation Systems. IEEE Trans. Robot. 2024, 40, 1292–1308. [Google Scholar] [CrossRef]
- Sharma, S.; Telikicherla, A.; Ding, G.; Aghlmand, F.; Talkhooncheh, A.H.; Shapiro, M.G.; Emami, A. Wireless 3D Surgical Navigation and Tracking System with 100 μm Accuracy Using Magnetic-Field Gradient-Based Localization. IEEE Trans. Med. Imaging 2021, 40, 2066–2079. [Google Scholar] [CrossRef]
- Dai, H.; Song, S.; Zeng, X.; Su, S.; Lin, M.; Meng, M.Q.H. 6-D Electromagnetic Tracking Approach Using Uniaxial Transmitting Coil and Tri-Axial Magneto-Resistive Sensor. IEEE Sens. J. 2018, 18, 1178–1186. [Google Scholar] [CrossRef]
- Vergne, C.; Féry, C.; Quirin, T.; Nicolas, H.; Madec, M.; Hemm, S.; Pascal, J. Low-Field Electromagnetic Tracking Using 3-D Magnetometer for Assisted Surgery. IEEE Trans. Magn. 2023, 59, 5400205. [Google Scholar] [CrossRef]
- Zhang, M.; Yang, L.; Zhang, C.; Yang, Z.; Zhang, L. A 5-D Large-Workspace Magnetic Localization and Actuation System Based on an Eye-in-Hand Magnetic Sensor Array and Mobile Coils. IEEE Trans. Instrum. Meas. 2023, 72, 7501411. [Google Scholar] [CrossRef]
- Aguilera Saiz, L.; Groen, H.C.; Heerink, W.J.; Ruers, T.J.M. The influence of the da Vinci surgical robot on electromagnetic tracking in a clinical environment. J. Robot. Surg. 2024, 18, 54. [Google Scholar] [CrossRef] [PubMed]
- Li, M.; Hansen, C.; Rose, G. A software solution to dynamically reduce metallic distortions of electromagnetic tracking systems for image-guided surgery. Int. J. Comput. Assist. Radiol. Surg. 2017, 12, 1621–1633. [Google Scholar] [CrossRef]
- Andria, G.; Attivissimo, F.; Di Nisio, A.; Lanzolla, A.M.L.; Ragolia, M.A. Assessment of Position Repeatability Error in an Electromagnetic Tracking System for Surgical Navigation. Sensors 2020, 20, 961. [Google Scholar] [CrossRef] [PubMed]
- O’Donoghue, K.; Cantillon-Murphy, P. Planar Magnetic Shielding for Use with Electromagnetic Tracking Systems. IEEE Trans. Magn. 2015, 51, 8500112. [Google Scholar] [CrossRef]
- Jaeger, H.A.; Cantillon-Murphy, P. Distorter Characterisation Using Mutual Inductance in Electromagnetic Tracking. Sensors 2018, 18, 3059. [Google Scholar] [CrossRef]
- Sadjadi, H.; Hashtrudi-Zaad, K.; Fichtinger, G. Simultaneous Electromagnetic Tracking and Calibration for Dynamic Field Distortion Compensation. IEEE Trans. Biomed. Eng. 2016, 63, 1771–1781. [Google Scholar] [CrossRef]
- Liu, S.; Wang, H. A Wireless 6-DoF Pose Tracking System Using a Triaxially Anisotropic Soft Magnet. IEEE/ASME Trans. Mechatron. 2025, 30, 1224–1235. [Google Scholar] [CrossRef]
- da Veiga, T.; Pittiglio, G.; Brockdorff, M.; Chandler, J.H.; Valdastri, P. Six-Degree-of-Freedom Localization Under Multiple Permanent Magnets Actuation. IEEE Robot. Autom. Lett. 2023, 8, 3422–3429. [Google Scholar] [CrossRef]
- Xu, Y.; Li, K.; Zhao, Z.; Meng, M.Q.H. A Novel System for Closed-Loop Simultaneous Magnetic Actuation and Localization of WCE Based on External Sensors and Rotating Actuation. IEEE Trans. Autom. Sci. Eng. 2021, 18, 1640–1652. [Google Scholar] [CrossRef]
- Fischer, F.; Gletter, C.; Jeong, M.; Qiu, T. Magneto-oscillatory localization for small-scale robots. npj Robot. 2024, 2, 1. [Google Scholar] [CrossRef]
- Seitel, A.; Groener, D.; Eisenmann, M.; Aguilera Saiz, L.; Pekdemir, B.; Sridharan, P.; Nguyen, C.T.; Häfele, S.; Feldmann, C.; Everitt, B.; et al. Miniaturized electromagnetic tracking enables efficient ultrasound-navigated needle insertions. Sci. Rep. 2024, 14, 14161. [Google Scholar] [CrossRef]
- de Ruiter, Q.M.B.; Xu, S.; Li, M.; Pritchard, W.F.; Starost, M.F.; Filie, A.; Mikhail, A.S.; Mauda-Havakuk, M.; Esparza-Trujillo, J.A.; Bakhutashvili, I.; et al. Electromagnetic Tracking and Optical Molecular Imaging Guidance for Liver Biopsy and Point-of-Care Tissue Assessment in Phantom and Woodchuck Hepatocellular Carcinoma. Cardiovasc. Interv. Radiol. 2021, 44, 1439–1447. [Google Scholar] [CrossRef]
- O’Donoghue, K.; Jaeger, H.A.; Cantillon-Murphy, P. A Radiolucent Electromagnetic Tracking System for Use with Intraoperative X-ray Imaging. Sensors 2021, 21, 3357. [Google Scholar] [CrossRef]
- Anderson, E.E.; Royer, N.C.; Kokes, B.; Dech, J.; Maiara, J. Magnetic Positioning Systems and Methods for Dental Procedures and Applications. US Patent Application No. 18/900,740, 3 April 2025. [Google Scholar]
- Dickhans, W.J. Systems and Methods for Active Tracking of Electromagnetic Navigation Bronchoscopy Tools with Single Guide Sheaths. US Patent Application No. 18/430,192, 19 September 2024. [Google Scholar]
- Northern Digital Inc. Available online: https://www.ndigital.com/ (accessed on 8 August 2025).
- Polhemus. Available online: https://polhemus.com/ (accessed on 8 August 2025).
- Aimooe. Available online: https://www.aimooe.net/ (accessed on 8 August 2025).
- Amisco. Available online: http://www.amisco.cn/ (accessed on 8 August 2025).
- OMMO. Available online: https://www.ommo.co/ (accessed on 8 August 2025).
- Groen, H.C.; Wit, E.M.; Heerink, W.J.; Kuhlmann, K.F.; Nijkamp, J.A.; van Veen, R.; Schoots, I.G.; Balduzzi, S.; Zijlmans, H.J.; van Leeuwen, P.J.; et al. Surgical navigation for targeted retroperitoneal lymph-node removal: A randomised, controlled, phase 3 trial. eClinicalMedicine 2024, 74, 102754. [Google Scholar] [CrossRef] [PubMed]
- Janssen, N.N.; Brastianos, H.; Akingbade, A.; Olding, T.; Vaughan, T.; Ungi, T.; Lasso, A.; Joshi, C.; Korzeniowski, M.; Falkson, C.; et al. Electromagnetic (EM) catheter path tracking in ultrasound-guided brachytherapy of the breast. Int. J. Comput. Assist. Radiol. Surg. 2020, 15, 1645–1652. [Google Scholar] [CrossRef] [PubMed]
- Saiz, L.A.; Heerink, W.J.; Groen, H.C.; Hiep, M.A.; van der Poel, H.G.; Wit, E.M.; Nieuwenhuijzen, J.A.; Roeleveld, T.A.; Vis, A.N.; Donswijk, M.L.; et al. Feasibility of Image-guided Navigation with Electromagnetic Tracking During Robot-assisted Sentinel Node Biopsy: A Prospective Study. Eur. Urol. 2025, 87, 482–490. [Google Scholar] [CrossRef] [PubMed]
- D’Agostino, G.R.; Mancosu, P.; Di Brina, L.; Franzese, C.; Pasini, L.; Iftode, C.; Comito, T.; De Rose, F.; Guazzoni, G.F.; Scorsetti, M. Stereotactic Body Radiation Therapy for Intermediate-risk Prostate Cancer with VMAT and Real-time Electromagnetic Tracking: A Phase II Study. Am. J. Clin. Oncol. 2020, 43, 628–635. [Google Scholar] [CrossRef]
- Gao, Y.; Qin, C.; Tao, B.; Hu, J.; Wu, Y.; Chen, X. An electromagnetic tracking implantation navigation system in dentistry with virtual calibration. Int. J. Med. Robot. Comput. Assist. Surg. 2021, 17, e2215. [Google Scholar] [CrossRef] [PubMed]
- Burchiel, K.J.; Kinsman, M.; Mansfield, K.; Mitchell, A. Verification of the Deep Brain Stimulation Electrode Position Using Intraoperative Electromagnetic Localization. Stereotact. Funct. Neurosurg. 2020, 98, 37–42. [Google Scholar] [CrossRef]
- Jia, F.; Wang, S.; Pham, V.T. A Hybrid Catheter Localisation Framework in Echocardiography Based on Electromagnetic Tracking and Deep Learning Segmentation. Comput. Intell. Neurosci. 2022, 2022, 2119070. [Google Scholar] [CrossRef]
- Lee, K.H.; Fu, K.C.D.; Guo, Z.; Dong, Z.; Leong, M.C.W.; Cheung, C.L.; Lee, A.P.W.; Luk, W.; Kwok, K.W. MR Safe Robotic Manipulator for MRI-Guided Intracardiac Catheterization. IEEE/ASME Trans. Mechatron. 2018, 23, 586–595. [Google Scholar] [CrossRef]
- Ivashchenko, O.V.; Kuhlmann, K.F.; van Veen, R.; Pouw, B.; Kok, N.F.M.; Hoetjes, N.J.; Smit, J.N.; Klompenhouwer, E.G.; Nijkamp, J.; Ruers, T.J.M. CBCT-based navigation system for open liver surgery: Accurate guidance toward mobile and deformable targets with a semi-rigid organ approximation and electromagnetic tracking of the liver. Med. Phys. 2021, 48, 2145–2159. [Google Scholar] [CrossRef]
- Xia, Z.; Wang, H.; Men, Y.; Ma, H.; Cao, Z.; Wang, W.; Du, Z. Kalman Filter-Based EM-Optical Sensor Fusion for Bone Needle Position Tracking. IEEE Sens. J. 2024, 24, 11166–11173. [Google Scholar] [CrossRef]
- Zhu, C.; Chen, Y.; Xu, Q.; Luo, Y.; Cen, J.; Wang, G. Electromagnetic tracking system-assisted ultrasound guidance for percutaneous transhepatic one-step biliary fistulation combined with rigid choledochoscopy in the treatment of hepatolithiasis. Surg. Endosc. 2025, 39, 4357–4364. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Li, J.; Yeerbulati, M.; Liang, S.; Zhang, Y.; Xu, Q. Collaborative Multirobot Navigation-Assisted Magnetic Catheter Guidance and Shape Perception with Vascular Ultrasound and Electromagnetic Tracking. Adv. Intell. Syst. 2025, 2500193. [Google Scholar] [CrossRef]
- Huang, Y.; Deng, X.; Zhao, X.; Zhang, T.; Feng, Y.; Yuan, Z.; Zhao, J. A Localization System Utilizing Maxwell Coil Gradient Magnetic Fields Integrated with Inertial Measurement Unit for Surgical Instrument. IEEE Sens. J. 2025, 25, 29688–29699. [Google Scholar] [CrossRef]
- Koyrakh, L.; Morgan, S.; Brown, A.; Jafari, F.; Miel, I.L.F.; Weisenberger, M.R. Fluoroscopic Imaging-Compatible and X-Ray Dose Reducing Electromagnetic Field Generator for Electromagnetic Tracking. US Patent Application No. 12,193,169, 7 January 2025. [Google Scholar]
- Liu, Z.H.; Po-Chi, H.; Tsai, I.C.; Lin, C.C. Control Method and Controller Related to Electromagnetic Tracking. US Patent Application No. 12,235,136, 25 February 2025. [Google Scholar]
- Sramek, C.; Kintz, G.J.; Romo, E.; Sidki, N.; Yu, A. Surgical Tools Having Electromagnetic Tracking Components. US Patent Application No. 12,064,229, 2 March 2024. [Google Scholar]
- LaVallee, S.; Rousseau, S.; Huguel, L.; Chave, M. Real-Time Electromagnetic Localization System. US Patent Application No. 18/721,221, 19 December 2024. [Google Scholar]
- Wang, H.; Zhou, Z.; Gong, J.; Shen, L.; Sun, Y.; Liang, B. Medical Catheter and Three-Dimensional Magnetic Positioning System. US Patent Application No. 18/551,799, 6 June 2024. [Google Scholar]
- Ragolia, M.A.; Attivissimo, F.; Nisio, A.D.; Maria Lucia Lanzolla, A. Evaluation of Position RMS Error from Magnetic Field Gradient for Surgical EM Tracking Systems. In Proceedings of the 2020 IEEE International Instrumentation and Measurement Technology Conference (I2MTC), Dubrovnik, Croatia, 25–28 May 2020; pp. 1–6. [Google Scholar]
- O’Donoghue, K.; Eustace, D.; Griffiths, J.; O’Shea, M.; Power, T.; Mansfield, H.; Cantillon-Murphy, P. Catheter Position Tracking System Using Planar Magnetics and Closed Loop Current Control. IEEE Trans. Magn. 2014, 50, 5100209. [Google Scholar] [CrossRef]
- Popek, K.M.; Schmid, T.; Abbott, J.J. Six-Degree-of-Freedom Localization of an Untethered Magnetic Capsule Using a Single Rotating Magnetic Dipole. IEEE Robot. Autom. Lett. 2016, 2, 305–312. [Google Scholar] [CrossRef]
- Aurora. Available online: https://www.ndigital.com/electromagnetic-tracking-technology/aurora/ (accessed on 3 September 2025).
- 3D Guidance. Available online: https://www.ndigital.com/electromagnetic-tracking-technology/3d-guidance/ (accessed on 3 September 2025).
- Li, Z.; Xu, Q. Multi-Section Magnetic Soft Robot with Multirobot Navigation System for Vasculature Intervention. Cyborg Bionic Syst. 2024, 5, 0188. [Google Scholar] [CrossRef] [PubMed]
- Zhu, K.; Nguyen, C.C.; Sharma, B.; Phan, P.T.; Hoang, T.T.; Davies, J.; Ji, A.; Nicotra, E.; Wan, J.; Pruscino, P.; et al. Development of a Bioinspired Soft Robotic System for Teleoperated Endoscopic Surgery. Cyborg Bionic Syst. 2025, 6, 0289. [Google Scholar] [CrossRef] [PubMed]










| Feature | Induction Principle | Integrated Magnetic Sensors | Magnetic Field Camera |
|---|---|---|---|
| Advantages |
|
|
|
| Disadvantages |
|
|
|
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, M.; Zhu, A.; Yang, L. Electromagnetic Tracking System for Medical Micro Devices: A Review. Micromachines 2025, 16, 1175. https://doi.org/10.3390/mi16101175
He M, Zhu A, Yang L. Electromagnetic Tracking System for Medical Micro Devices: A Review. Micromachines. 2025; 16(10):1175. https://doi.org/10.3390/mi16101175
Chicago/Turabian StyleHe, Mingshan, Aoji Zhu, and Lidong Yang. 2025. "Electromagnetic Tracking System for Medical Micro Devices: A Review" Micromachines 16, no. 10: 1175. https://doi.org/10.3390/mi16101175
APA StyleHe, M., Zhu, A., & Yang, L. (2025). Electromagnetic Tracking System for Medical Micro Devices: A Review. Micromachines, 16(10), 1175. https://doi.org/10.3390/mi16101175







