2.1. Design and Operating Principle
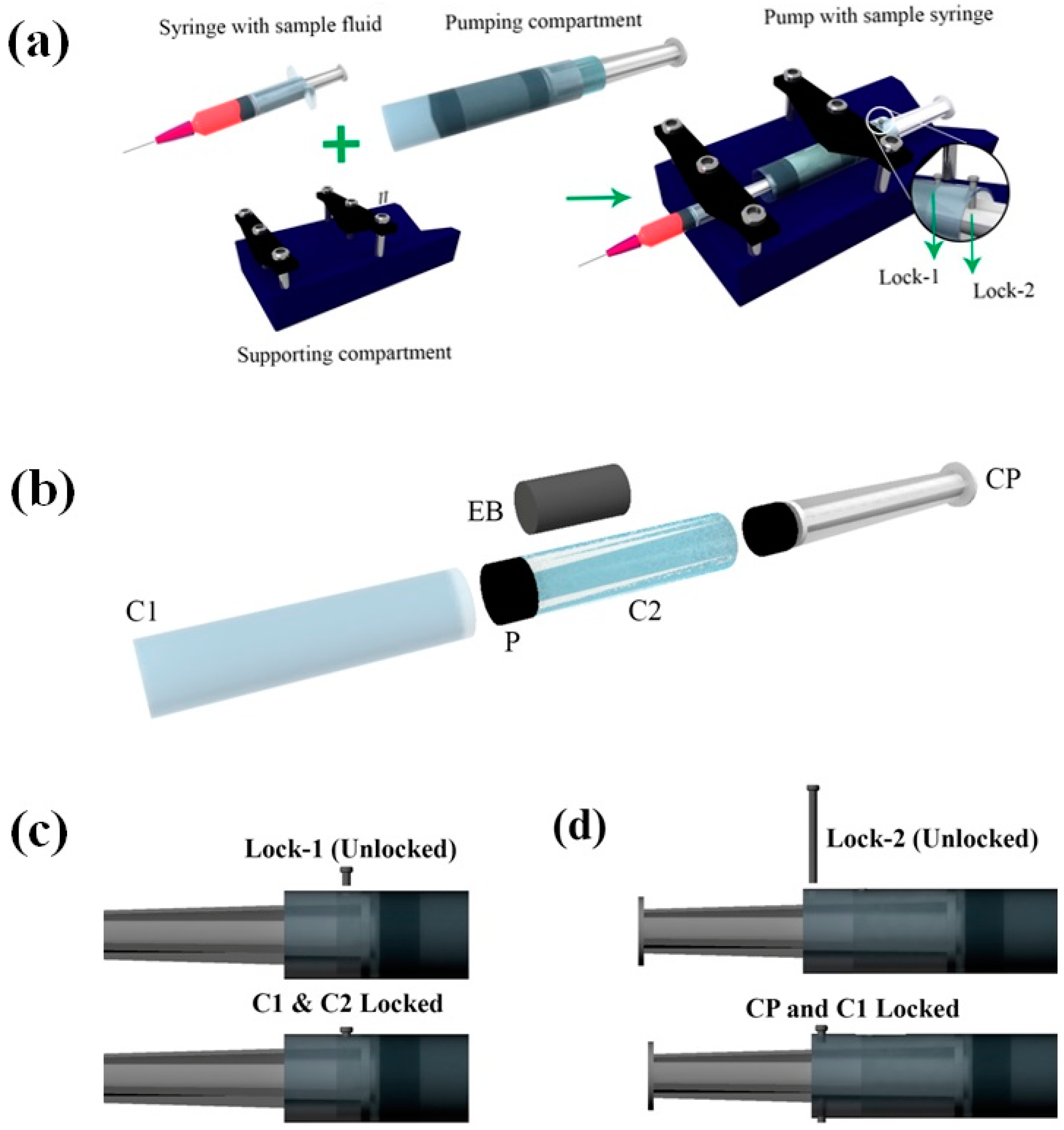
Figure 1a shows the schematic of the pump developed. The pump design can be broadly divided into two parts, namely pumping compartment and supporting compartment. The supporting compartment serves the purpose of holding steadily the pumping compartment and the syringe with sample fluid as shown in
Figure 1a. The pumping compartment consists of one large cylindrical tube that is open at both ends (C1), a small cylindrical tube (C2) that is closed at the front end with a piston rubber (P), an elastic block (EB), a compressor (CP), and a locking–unlocking mechanism. The respective arrangement is shown in
Figure 1b,c. The elastic block EB gets into the smaller cylindrical tube C2. C2 lies inside the larger tube C1 and is free to move along the walls. C2 can be locked or unlocked to C1 using a small pin that goes through aligned holes punched to the two tubes (lock-1). At the rear side of the EB lies the compressor. This compressor CP can be locked to C1 using lock-2 that employs a similar mechanism as that of lock-1. On the front side of P lies the piston of the syringe that contains the sample fluid to be pumped.
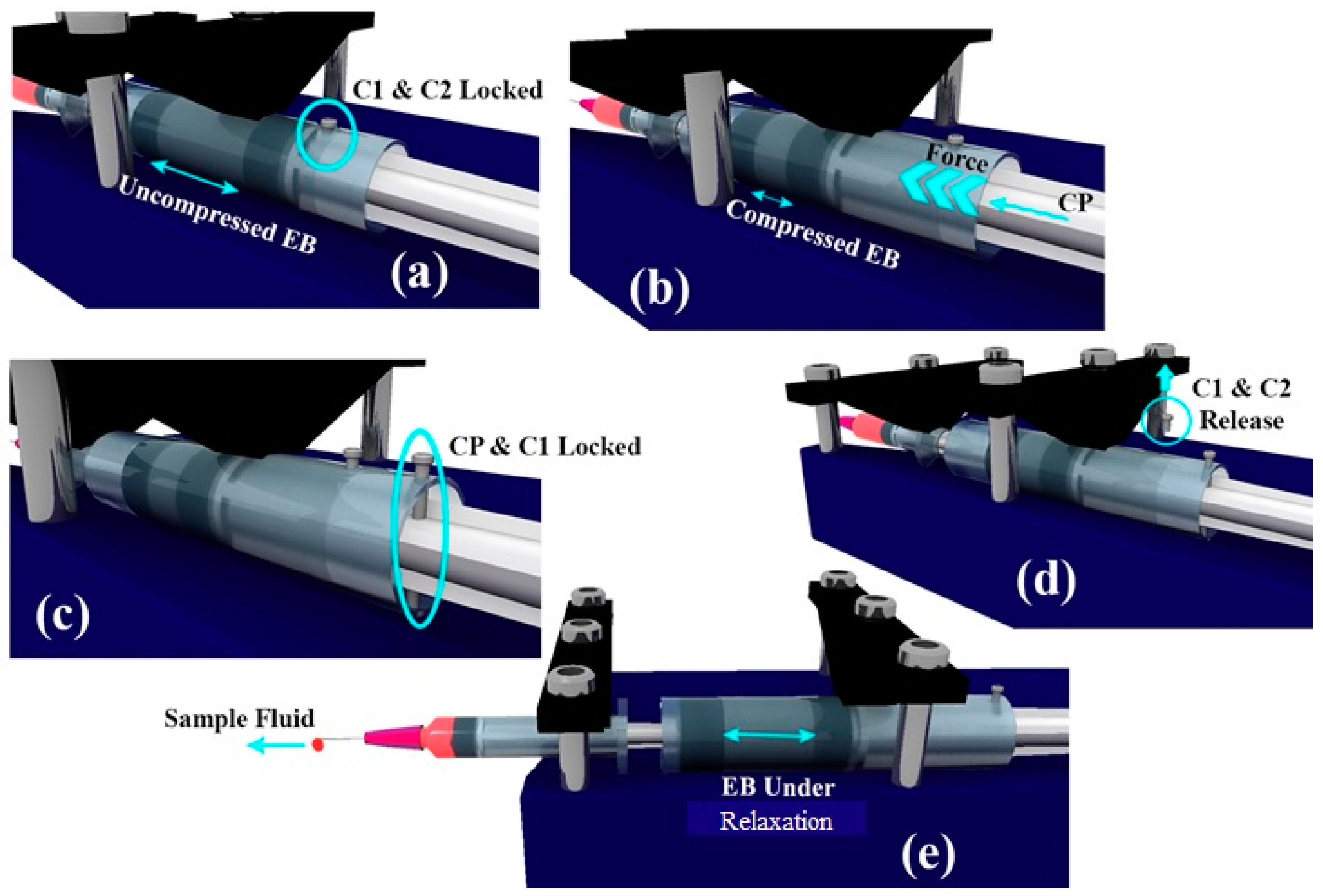
EB has a diameter of De and height he, both being smaller than that of C2. CP has an outer diameter matching the inner diameter of the C2. The pump in its non-pumping condition will have C2 locked with respect to C1, while freely allowing CP to move. The CP, when pushed against the EB and locked with respect to C1, will lead to the development of compressive stresses within EB.
If the locking between C2 and C1 is released, EB pushes C2 in the direction of the sample syringe piston to relieve the stored compressive stresses within the EB. This, in turn, pushes the sample syringe piston leading to the pumping of the sample fluid into the microfluidic device connected. The schematic depicting the working of the pump is shown in
Figure 2.
2.2. Theory and Modeling
The force applied by the elastic block on the elastic container at any moment is given by the product of stress (
St) the elastic block is experiencing and its area of cross-section (π ×
De2/4). Presuming negligible frictional forces, the same force gets transferred to the sample fluid inside the syringe. Hence, the pressure acting on the sample fluid is given by:
where
De is the diameter of the elastic block,
Ds is the sample syringe inner diameter, and
St is the stress that the elastic block is experiencing. This can be further simplified as:
If the syringe is attached to a microfluidic device, the overall hydrodynamic resistance the sample fluid feels for its flow is a combination of resistances offered by the syringe needle, tubing, and the microfluidic device. Typically, the microfluidic device hydrodynamic resistance (Rh) will be much larger than the hydrodynamic resistances of the syringe needle and tubing, hence it can be taken as the effective resistance the fluid experiences for its flow. Whereas, the fluid at the inlet experiences a pressure that is due to the elastic block and the atmosphere. Presuming the device outlet stays at the same height as that of the inlet (which is typically the case), the outlet also feels the same quantity of atmospheric pressure. Hence, the net pressure drop acting on the sample fluid is only due to the elastic block and is given by Equation (2).
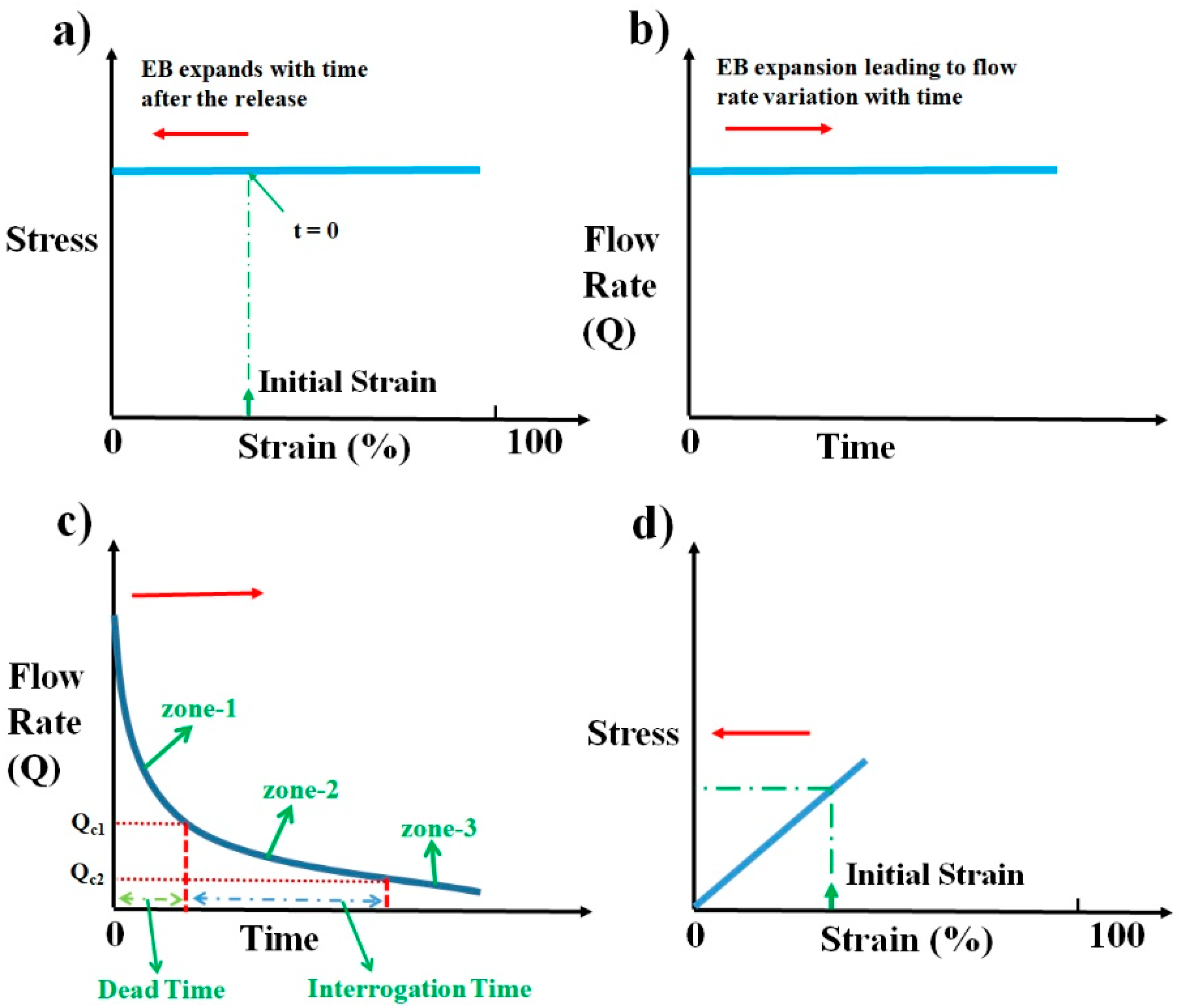
As the elastic block relaxes, depending upon the nature of elastic material, the stress the block exerts on the sample syringe changes. If the elastic block has stress–strain characteristics as shown in
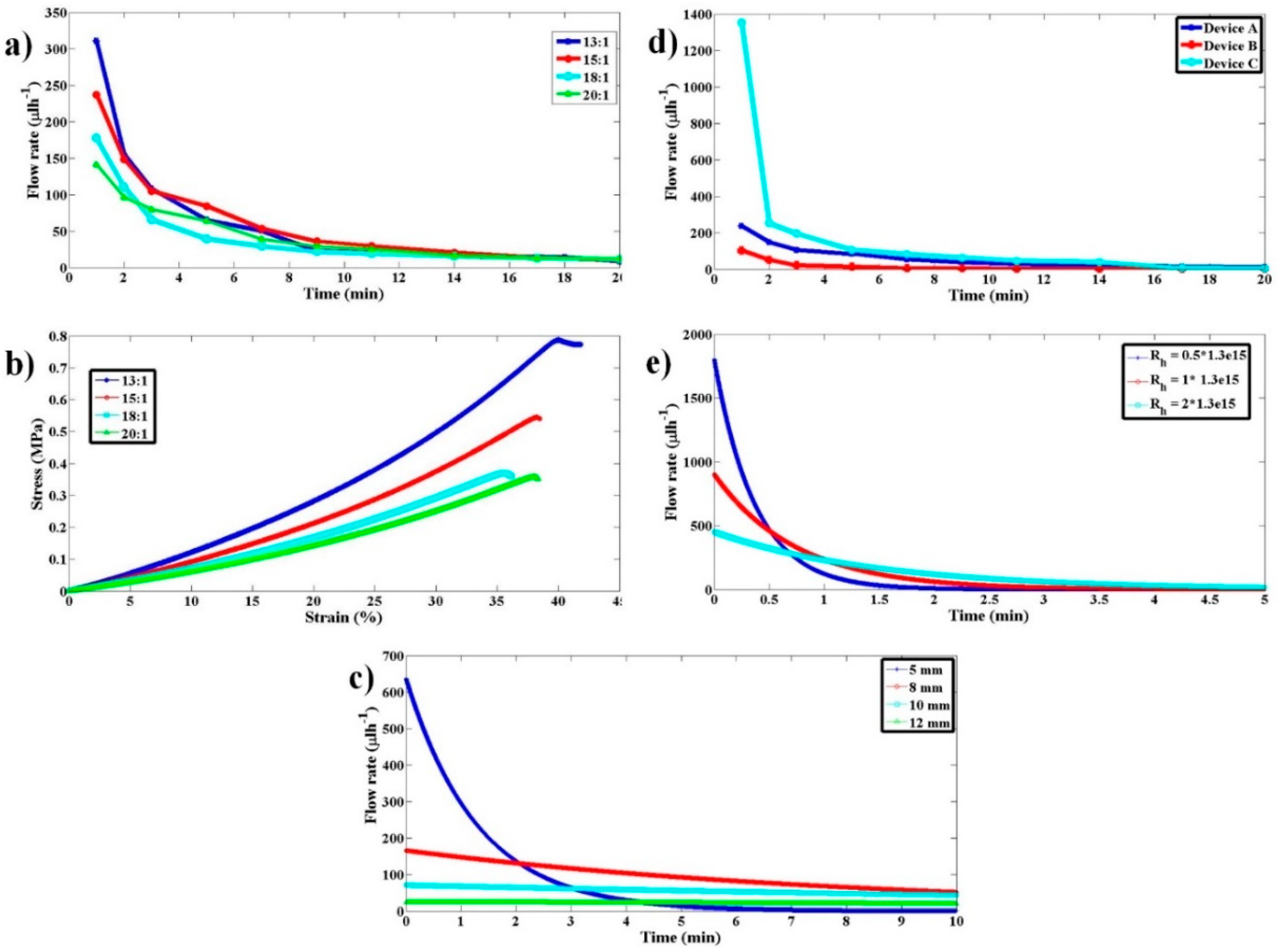
Figure 3a, irrespective of the initial strain induced, it will produce constant stress throughout the relaxation period. The pressure drops thereby produced across the device will be constant as given by Equation (2). Under constant pressure across the device, we can presume flow becomes steady quickly (≈sec) as compared to the time duration of operation (≈min) and the flow rate can be computed as per the steady-state Equation (3).
As
De,
Ds, and
Rh are constants for a given device, the sample syringe, and pump, the flow rate
Q at any moment is dependent upon the elastic stress
St that the block exerts on the fluid, as shown in
Figure 3b. This constant flow rate of pumping is the standard laboratory process followed in microfluidics using pumps such as syringe pumps.
Even though this is what is being followed, it is not always a desired way of pumping from the perspective of gaining a time advantage while performing microfluidic POCD tests. A typical microfluidic POCD test involves the pumping of the sample fluid into the microfluidic device through the tubing. After the sample fluid reaches the microfluidic device, sample interrogation starts. This time lag between the start of the pumping and the sample fluid reaching the microfluidic device is the dead time involved in the diagnosis. This dead time is one of the critical times to be optimized to have an overall shorter duration of operation for any microfluidic POCD device. As an illustration, for imaging flow cytometric applications, the typical flow rate of operation will be around 100 µL·h
−1 [
25]. For a polyethylene tubing with an inner diameter of 0.38 mm and a length of 10 cm, the dead time of operation is around 7 min, which is very huge compared to the requirement of having the overall time of diagnosis to within 2–3 min. This clearly shows that providing a constant flow rate of operation as is conventionally followed in laboratory settings may not be an advantageous way of pumping from the perspective of overall diagnosis time. One of the ways to reduce this dead time is to have a very high flow rate of pumping at the beginning of pump operation, which continuously decreases to a lower desired range of flow rates as shown in
Figure 3c. This high flow rate of operation ensures that the sample fluid crosses the tubing quickly and reaches the microfluidic device. Continuous reduction in flow rate from the initial value ensures that the sample fluid continues to decelerate and reaches the optimum flow rate desired for imaging when it reaches the device. This dead time zone is represented as zone-1 in
Figure 3c. Dead time can be decreased to lower values if the initial flow rate of operation is very large and the rate of reduction is very steep. This is one of the desired features of an ideal pump for microfluidic POCD devices. Once the sample reaches the device, the flow rate of operation should be either constant or slowly vary within a certain flow rate range to avoid any reduction in throughput. The reduction in throughput can happen if the flow rate varies widely to such an extent that either some of the sample particles move at a higher speed (flow rate >
Qc1), thereby causing motion blur, or some move very slowly (flow rate <
Qc2), such that images of the same particle are captured multiple times. This can be avoided if the flow rate varies within an optimum range (between
Qc1 and
Qc2) during the interrogation. This time zone of interrogation is represented as zone-2 in the schematic shown in
Figure 3c. The time zone represented as zone-3 in
Figure 3c is the flow rate variation with time after the interrogation, which is not a critical time zone to scrutinize. The flow rate within that zone can decrease, which is in natural tune with how the flow rate is changing since the start of pumping. This will help to smoothly stop the process of pumping. These desired flow rate behaviors can be achieved from this pump using an elastomeric block that shows a linear stress–strain plot, as shown in
Figure 3d.
In general, most of the materials show a stress–strain behavior that is linear at lower values of compressive strain, as shown in
Figure 3d. Such materials, when compressed and released, show a stress that keeps decreasing as the material relaxes toward its natural length. If such a material is used as an elastomeric block for the pump, it results in decreasing stress and decreasing pressure difference across the microfluidic device as the block relaxes. This results in a variable flow rate of pumping as a function of time. The schematic illustrating the flow rate variation with time is shown in
Figure 3c. The exact solution for this problem requires solving a coupled Navier–Stokes Equation whose input pressure difference depends on the output flow rate, which is computationally highly expensive. For slow variations of pressure difference across the device, the flow can be presumed to reach steady-state instantaneously and applying the steady-state Equation (3) to compute the flow rate at every value of pressure difference across the device. The above process of simulating the flow would be a reasonable approximation to gain good insights into the qualitative behavior of the flow.
So far, the discussion neglected the presence of frictional forces, which, in practice, cannot be avoided altogether in the working of the pump. The major source of the friction comes from the movement of the piston rubber P that is in contact with C1 and from the movement of the sample syringe piston into the sample syringe. Presuming the friction coefficients are similar in both cases (due to the similar materials used), the net frictional forces acting on the sample syringe piston in the direction opposite to its movement is given by:
wherein the first term is a contribution coming from piston rubber P and the latter is due to the sample syringe piston. γ is the friction coefficient between the piston rubber P and C1, and the sample syringe piston rubber and sample syringe.
Dpi and
Dpl are the inner diameter of C1 and the length of the piston rubber P, respectively.
Ds and
Dsl are the inner diameter of the sample syringe and the length of the sample syringe piston rubber, respectively. Hence, the net pressure the
Pnet and the fluid experiences and the associated flow rate
Qeff are given by:
where
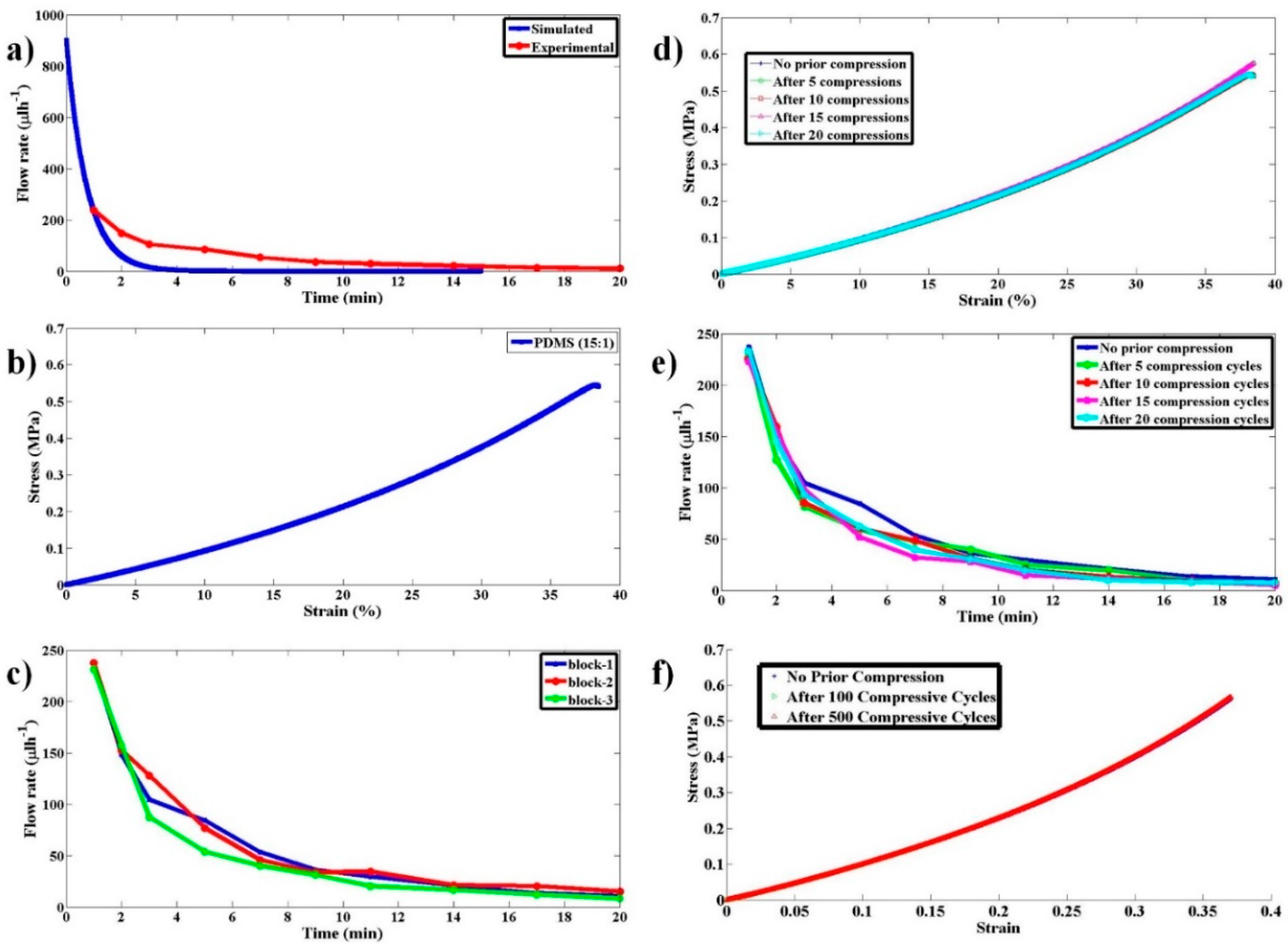
P is given by Equation (2). Simulations were performed in MATLAB (R2014a) to determine the flow rate of pumping as a function of time as per the above model for experimentally used parameters. As the frictional coefficient γ is unknown, its value was chosen such that the simulated flow rate matches with experimental observations at 1 min from the start of pumping.
Elastomers, such as polydimethylsiloxane (PDMS), possess a compressive stress–strain curve that is linear for smaller values of strain, like the one shown in
Figure 3d. By varying the amount of curing agent that is added to the fixed quantity of liquid PDMS to facilitate polymerization, the stiffness of the resulting solid PDMS can be controlled. We express this variable amount addition of curing agent as the ratio of PDMS to curing agent in this paper. The choice of PDMS as EB is due to the wide stiffness range it can offer and its ease of availability and fabrication. To validate the proposed pumping mechanism and characterize the fabricated pump, experiments were conducted with red blood cell (RBC) samples. The use of red blood sample solution serves a dual purpose; the first being a method to estimate the flow rate, while the other demonstrates the biological compatibility. The procedure for EB fabrication using PDMS, cost analysis, sample preparation, and experimental procedure and analysis are detailed in the following sections.
2.3. Materials and Fabrication
Pumping compartment was mostly made using readily available materials in lab, and the suitable support structure (or support compartment) was designed using computer-aided design and drafting software, SolidWorks. The support structure design was exported as STL (Standard Tessellation Language) file and was sliced using MakerBot slicing software prior to giving this as an input to the 3D printer. The 3D printer (Wanhao Duplicator desktop 3D printer) that works on the principle of fused deposition modeling based dual extrusion was used to print the parts, with Polylactic acid as build material. Extruder temperature of 210 °C and build base temperature of 60 °C was maintained throughout the print.
The support structure consists of a base of dimension 8 cm × 15 cm with a V-shape groove (along the longer edge) and slots (along the shorter edge) designed to hold the cylinder (C1) and sample syringe firmly in place. The support structure also includes 2 V-shaped locks that were held together to the support base using standard M6 screws. This combination of V-shaped locks and base structure ensures easy and reproducible attachment of the pumping compartment to the support structure. The support structure built for the pump is shown in
Figure 4a.
A 25 mL Vac-Lok syringe was cut (from the side of the needle) to produce a two-sided opening needed for the outer cylindrical tube C1. Two 3 mm holes were drilled at two different positions for locking C1 with C2 and CP. A 15 mL Vac-Lok syringe was cut from its rear end (away from the needle placing end) and was used as cylinder C2. A Luer connector was designed, 3D printed, and attached to the tip of C2. This Luer connector connects the tip of C2 to the piston rubber P that was taken from the 25 mL Vac-Lok piston to ensure the smooth movement of C2 inside C1. The compressor CP was designed and 3D printed. The CP diameter was matched to the inner diameter of C2 to facilitate smooth compression. The various parts involved in making up the pumping compartment are shown in
Figure 4b. The whole of the pump along with the sample syringe is shown in
Figure 4c.
Several single-use molds were prepared with an inner diameter of 13 mm and a height of 27 mm. These molds were used to prepare PDMS elastic blocks of varied stiffness. PDMS was mixed with curing agents in varying ratios (13:1, 15:1, 18:1, and 20:1), poured into the molds, and were cured overnight at 65 °C. The solidified PDMS blocks were carefully cut open from the molds without damaging them. The PDMS devices with three different hydrodynamic resistances used for testing were fabricated from the three pre-fabricated SU-8 Masters, using the standard process of soft lithography. The computed hydrodynamic resistances of the three Masters (say A, B, and C) are 2 × 1013 Pa·s/m3, 1.3 × 1015 Pa·s/m3, and 2.5 × 1015 Pa·s/m3, respectively.
2.8. Flow Rate Estimation Methodology
The procedure that was followed for flow rate determination involves finding the velocity of RBC in the flow as a function of its position across the channel cross-section inside the microfluidic device and subsequently computing flow rate based on an analytical expression that relates flow rate with velocity at a particular position. Velocity determination requires the acquisition of an RBC image at least twice as it flows. To ensure this, the frame rate of the camera was chosen high enough (1030 frames per second) during the video capture.
As the RBC flows along the length of the channel (z-direction), let us say the first captured image of the RBC has coordinates (x, y, z). As the channel does not have any tilt in the xz-plane, the next image of the RBC will have the same x-coordinate
x, but a different z-coordinate
z + δ
z. The difference in z-coordinates, divided by magnification
M, and the time difference (δ
t) between the two acquired images yields the velocity of the cell at time
t, given by the expression:
The time difference between the acquired images is equal to the inverse of the frame rate as two consecutive frames of a video were used for analysis. The steady-state analytical expression for fluid velocity at (x, y, z) inside a microfluidic channel of width
w (along x-direction), height
h (along y-direction), and length
L is given by:
where
,
, and
z is the direction of flow. Choosing the plane of observation to be close to the center of the channel, i.e.,
y = 0 and for
h <
w, we can approximate the above expression to first order, and can be written as:
Substituting the experimentally determined value of velocity from Equation (7) along with channel dimensions and the x-location of the cell, the analytical Equation (9), for velocity, yields the unknown
Peff/(
μL). This computed value of the unknown is substituted in the following analytical Equation (10) to estimate the flow rate. This analytical expression for flow rate gives accurate results within 10% error for
h/
w ≤ 0.7 [
26].