3D Printing Breast Tissue Models: A Review of Past Work and Directions for Future Work
Abstract
1. Introduction
2. Materials and Methods
2.1. Review Methods
2.2. Bioprinting Methods
3. Results
3.1. Using Computer Aided Design (CAD) and Computer Aided Manufacturing (CAM) Technology to Assist Surgeons with Breast Reconstruction
3.2. Generating a Bioink from Adipose Tissue to 3D-Bioprint an Implantable Porous Cell-Laden Structure Resembling Breast Tissue
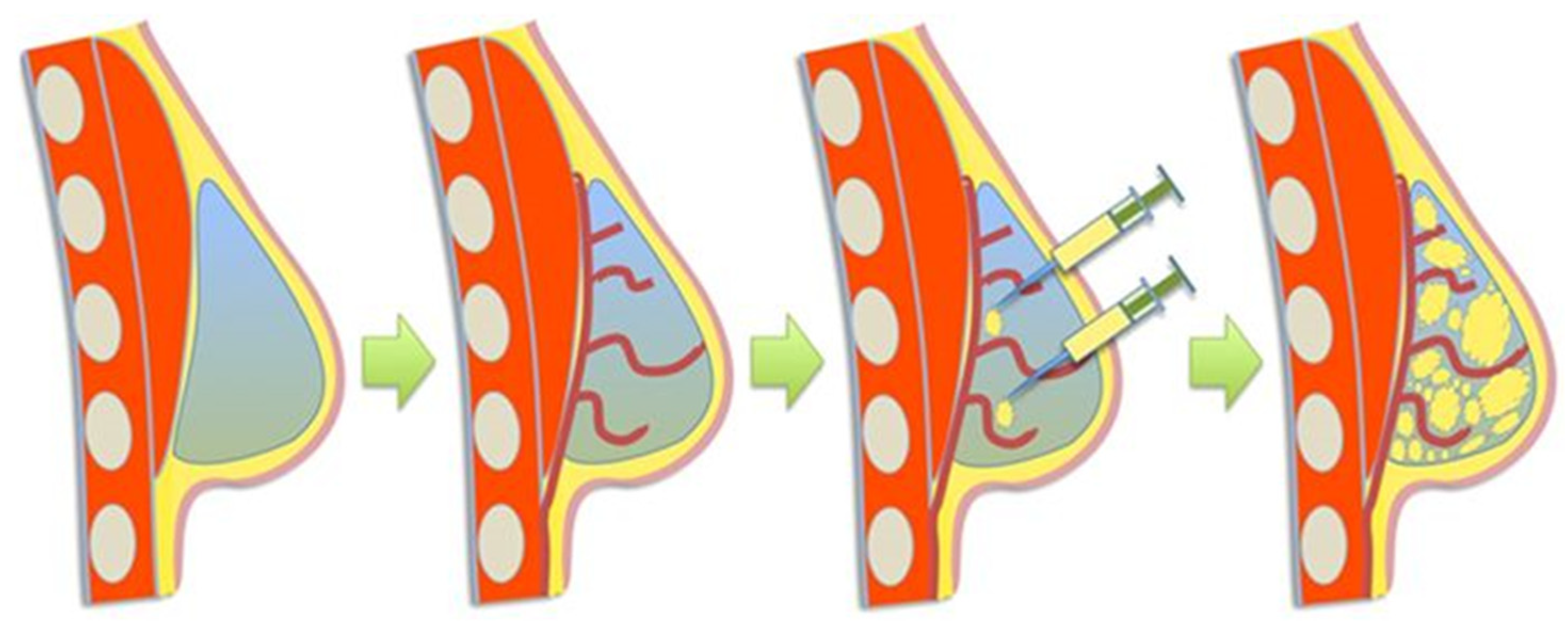
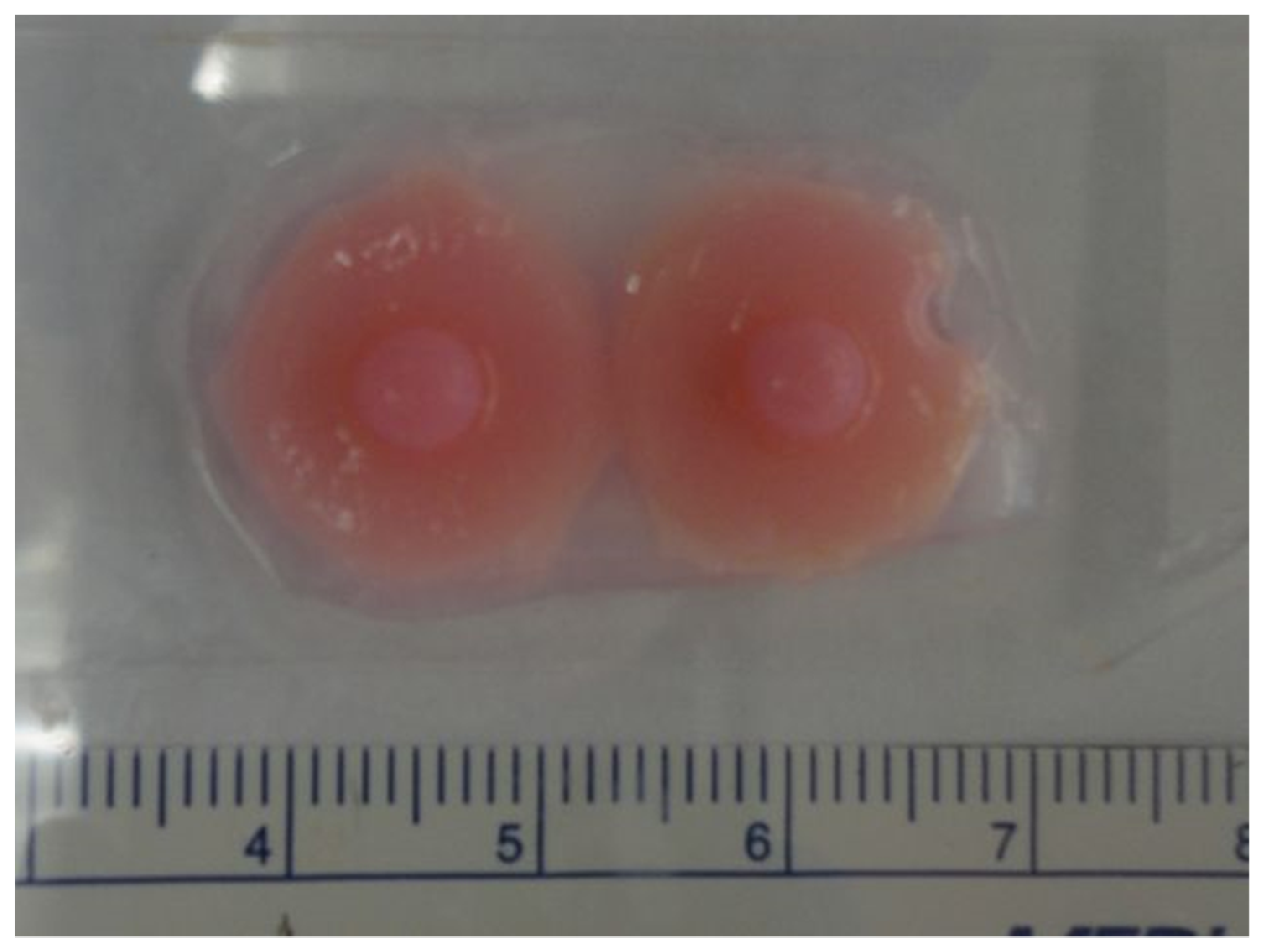
3.3. Implanting Large Polymer Scaffolds Combined with Autologous Adipose Tissue in Minipigs
3.4. The Role of 3D-Printing for Nipple Reconstruction
4. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: Globocan estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [PubMed]
- Siegel, R.L.; Miller, K.D.; Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 2019, 69, 7–34. [Google Scholar] [CrossRef] [PubMed]
- Spronk, I.; Schellevis, F.G.; Burgers, J.S.; de Bock, G.H.; Korevaar, J.C. Incidence of isolated local breast cancer recurrence and contralateral breast cancer: A systematic review. Breast 2018, 39, 70–79. [Google Scholar] [CrossRef] [PubMed]
- McGuire, K.P.; Santillan, A.A.; Kaur, P.; Meade, T.; Parbhoo, J.; Mathias, M.; Shamehdi, C.; Davis, M.; Ramos, D.; Cox, C.E. Are mastectomies on the rise? A 13-year trend analysis of the selection of mastectomy versus breast conservation therapy in 5865 patients. Ann. Surg. Oncol. 2009, 16, 2682–2690. [Google Scholar] [CrossRef] [PubMed]
- Marin-Gutzke, M.; Sanchez-Olaso, A. Reconstructive surgery in young women with breast cancer. Breast Cancer Res. Treat. 2010, 123 (Suppl. S1), 67–74. [Google Scholar] [CrossRef]
- Champaneria, M.C.; Wong, W.W.; Hill, M.E.; Gupta, S.C. The evolution of breast reconstruction: A historical perspective. World J. Surg. 2012, 36, 730–742. [Google Scholar] [CrossRef]
- Kanchwala, S.K.; Glatt, B.S.; Conant, E.F.; Bucky, L.P. Autologous fat grafting to the reconstructed breast: The management of acquired contour deformities. Plast. Reconstr. Surg. 2009, 124, 409–418. [Google Scholar] [CrossRef]
- Howes, B.H.; Fosh, B.; Watson, D.I.; Yip, J.M.; Eaton, M.; Smallman, A.; Dean, N.R. Autologous fat grafting for whole breast reconstruction. Plast. Reconstr. Surg. Glob. Open 2014, 2, e124. [Google Scholar] [CrossRef]
- Spear, S.L.; Mesbahi, A.N. Implant-based reconstruction. Clin. Plast. Surg. 2007, 34, 63–73. [Google Scholar] [CrossRef]
- Steligo, K. The Breast Reconstruction Guidebook: Issues and Answers from Research to Recovery; JHU Press: Baltimore, MD, USA, 2017. [Google Scholar]
- Visscher, L.E.; Cheng, M.; Chhaya, M.; Hintz, M.L.; Schantz, J.T.; Tran, P.; Ung, O.; Wong, C.; Hutmacher, D.W. Breast augmentation and reconstruction from a regenerative medicine point of view: State of the art and future perspectives. Tissue Eng. Part B Rev. 2017, 23, 281–293. [Google Scholar] [CrossRef]
- Jewell, M.L. Silicone gel breast implants at 50: The state of the science. Aesthetic Surg. J. 2012, 32, 1031–1034. [Google Scholar] [CrossRef] [PubMed]
- Combellack, E.J.; Jessop, Z.M.; Naderi, N.; Griffin, M.; Dobbs, T.; Ibrahim, A.; Evans, S.; Burnell, S.; Doak, S.H.; Whitaker, I.S. Adipose regeneration and implications for breast reconstruction: Update and the future. Gland Surg. 2016, 5, 227–241. [Google Scholar] [PubMed]
- Matsumoto, D.; Sato, K.; Gonda, K.; Takaki, Y.; Shigeura, T.; Sato, T.; Aiba-Kojima, E.; Iizuka, F.; Inoue, K.; Suga, H.; et al. Cell-assisted lipotransfer: Supportive use of human adipose-derived cells for soft tissue augmentation with lipoinjection. Tissue Eng. 2006, 12, 3375–3382. [Google Scholar] [CrossRef] [PubMed]
- Miana, V.V.; Gonzalez, E.A.P. Adipose tissue stem cells in regenerative medicine. Ecancermedicalscience 2018, 12, 822. [Google Scholar] [CrossRef] [PubMed]
- Fischer, J.P.; Nelson, J.A.; Cleveland, E.; Sieber, B.; Rohrbach, J.I.; Serletti, J.M.; Kanchwala, S. Breast reconstruction modality outcome study: A comparison of expander/implants and free flaps in select patients. Plast. Reconstr. Surg. 2013, 131, 928–934. [Google Scholar] [CrossRef] [PubMed]
- Arshad, Z.; Karmen, L.; Choudhary, R.; Smith, J.A.; Branford, O.A.; Brindley, D.A.; Pettitt, D.; Davies, B.M. Cell assisted lipotransfer in breast augmentation and reconstruction: A systematic review of safety, efficacy, use of patient reported outcomes and study quality. JPRAS Open 2016, 10, 5–20. [Google Scholar] [CrossRef]
- Eterno, V.; Zambelli, A.; Pavesi, L.; Villani, L.; Zanini, V.; Petrolo, G.; Manera, S.; Tuscano, A.; Amato, A. Adipose-derived mesenchymal stem cells (ascs) may favour breast cancer recurrence via hgf/c-met signaling. Oncotarget 2014, 5, 613–633. [Google Scholar] [CrossRef] [PubMed]
- Sakurai, M.; Miki, Y.; Takagi, K.; Suzuki, T.; Ishida, T.; Ohuchi, N.; Sasano, H. Interaction with adipocyte stromal cells induces breast cancer malignancy via s100a7 upregulation in breast cancer microenvironment. Breast Cancer Res. 2017, 19, 70. [Google Scholar] [CrossRef]
- Schweizer, R.; Tsuji, W.; Gorantla, V.S.; Marra, K.G.; Rubin, J.P.; Plock, J.A. The role of adipose-derived stem cells in breast cancer progression and metastasis. Stem Cells Int. 2015, 2015, 120949. [Google Scholar] [CrossRef]
- Ackova, D.G.; Kanjevac, T.; Rimondini, L.; Bosnakovski, D. Perspectives in engineered mesenchymal stem/stromal cells based anti- cancer drug delivery systems. Recent Pat. Anticancer Drug Discov. 2016, 11, 98–111. [Google Scholar] [CrossRef]
- Scioli, M.G.; Artuso, S.; D’Angelo, C.; Porru, M.; D’Amico, F.; Bielli, A.; Gentile, P.; Cervelli, V.; Leonetti, C.; Orlandi, A. Adipose-derived stem cell-mediated paclitaxel delivery inhibits breast cancer growth. PLoS ONE 2018, 13, e0203426. [Google Scholar] [CrossRef] [PubMed]
- Saltzman, W.M. Tissue Engineering: Engineering Principles for the Design of Replacement Organs and Tissues; Oxford University Press: Oxford, UK, 2004. [Google Scholar]
- Rambhia, K.J.; Ma, P.X. Controlled drug release for tissue engineering. J. Control. Release 2015, 219, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Chae, M.P.; Hunter-Smith, D.J.; Murphy, S.V.; Findlay, M.W. 3D bioprinting adipose tissue for breast reconstruction. In 3D Bioprinting for Reconstructive Surgery: Techniques and Applications; Woodhead Publishing: Cambridge, UK, 2018; pp. 305–353. [Google Scholar]
- Sokol, E.S.; Miller, D.H.; Breggia, A.; Spencer, K.C.; Arendt, L.M.; Gupta, P.B. Growth of human breast tissues from patient cells in 3d hydrogel scaffolds. Breast Cancer Res. 2016, 18, 19. [Google Scholar] [CrossRef] [PubMed]
- Findlay, M.W.; Dolderer, J.H.; Trost, N.; Craft, R.O.; Cao, Y.; Cooper-White, J.; Stevens, G.; Morrison, W.A. Tissue-engineered breast reconstruction: Bridging the gap toward large-volume tissue engineering in humans. Plast. Reconstr. Surg. 2011, 128, 1206–1215. [Google Scholar] [CrossRef] [PubMed]
- Morrison, W.A.; Marre, D.; Grinsell, D.; Batty, A.; Trost, N.; O’Connor, A.J. Creation of a large adipose tissue construct in humans using a tissue-engineering chamber: A step forward in the clinical application of soft tissue engineering. EBioMedicine 2016, 6, 238–245. [Google Scholar] [CrossRef] [PubMed]
- Mazzocchi, A.; Soker, S.; Skardal, A. 3d bioprinting for high-throughput screening: Drug screening, disease modeling, and precision medicine applications. Appl. Phys. Rev. 2019, 6, 011302. [Google Scholar] [CrossRef]
- Chae, M.P.; Hunter-Smith, D.J.; Murphy, S.V.; Rozen, W.M. 3d bioprinting in nipple-areola complex reconstruction. In Nipple-areolar Complex Reconstruction: Principles and Clinical Techniques; Shiffman, M.A., Ed.; Springer International Publishing: Cham, Switzerland, 2018; pp. 587–606. [Google Scholar]
- Jang, J.; Yi, H.-G.; Cho, D.-W. 3d printed tissue models: Present and future. ACS Biomater. Sci. Eng. 2016, 2, 1722–1731. [Google Scholar] [CrossRef]
- Ding, S.; Feng, L.; Wu, J.; Zhu, F.; Tan, Z.E.; Yao, R. Bioprinting of stem cells: Interplay of bioprinting process, bioinks, and stem cell properties. ACS Biomater. Sci. Eng. 2018, 4, 3108–3124. [Google Scholar] [CrossRef]
- Derakhshanfar, S.; Mbeleck, R.; Xu, K.; Zhang, X.; Zhong, W.; Xing, M. 3d bioprinting for biomedical devices and tissue engineering: A review of recent trends and advances. Bioact. Mater. 2018, 3, 144–156. [Google Scholar] [CrossRef]
- Kačarević, Ž.; Rider, P.; Alkildani, S.; Retnasingh, S.; Smeets, R.; Jung, O.; Ivanišević, Z.; Barbeck, M. An introduction to 3d bioprinting: Possibilities, challenges and future aspects. Materials 2018, 11, 2199. [Google Scholar] [CrossRef]
- Tourlomousis, F.; Jia, C.; Karydis, T.; Mershin, A.; Wang, H.; Kalyon, D.M.; Chang, R.C. Machine learning metrology of cell confinement in melt electrowritten three-dimensional biomaterial substrates. Microsyst. Nanoeng. 2019, 5, 15. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Wang, Y.; Liu, J. Bioprinting of 3D tissues/organs combined with microfluidics. RSC Advances. 2018, 3, 21712–21727. [Google Scholar] [CrossRef]
- Cui, X.; Boland, T.; DD’Lima, D.; K Lotz, M. Thermal inkjet printing in tissue engineering and regenerative medicine. Recent Pat. Drug Deliv. Formul. 2012, 6, 149–155. [Google Scholar] [CrossRef] [PubMed]
- Lepock, J.R.; Kruuv, J. Thermotolerance as a possible cause of the critical temperature at 43 in mammalian cells. Cancer Res. 1980, 40, 4485–4488. [Google Scholar] [PubMed]
- Zhang, Y.; Zhan, X.; Xiong, J.; Peng, S.; Huang, W.; Joshi, R.; Cai, Y.; Liu, Y.; Li, R.; Yuan, K. Temperature-dependent cell death patterns induced by functionalized gold nanoparticle photothermal therapy in melanoma cells. Sci. Rep. 2018, 8, 8720. [Google Scholar] [CrossRef]
- Ashraf, H.; Meer, B.; Naz, R.; Saeed, A.; Sajid, U.; Nisar, K.; Aslam, Z.; Anwar, P. 3d-bioprinting: A stepping stone towards enhanced medical approaches. Adv. Life Sci. 2018, 5, 143–153. [Google Scholar]
- Ali, S.; Majeed, S. Advancement of bio inks in three dimensional bioprinting. Biomed. J. Sci. Tech. Res. 2018, 11, 4. [Google Scholar]
- Ji, S.; Guvendiren, M. Recent advances in bioink design for 3d bioprinting of tissues and organs. Front. Bioeng. Biotechnol. 2017, 5, 23. [Google Scholar] [CrossRef]
- Donderwinkel, I.; van Hest, J.C.; Cameron, N.R. Bio-inks for 3d bioprinting: Recent advances and future prospects. Polym. Chem. 2017, 8, 4451–4471. [Google Scholar] [CrossRef]
- Hölzl, K.; Lin, S.; Tytgat, L.; Van Vlierberghe, S.; Gu, L.; Ovsianikov, A. Bioink properties before, during and after 3d bioprinting. Biofabrication 2016, 8, 032002. [Google Scholar] [CrossRef]
- Abelseth, E.; Abelseth, L.; De la Vega, L.; Beyer, S.T.; Wadsworth, S.J.; Willerth, S.M. 3d printing of neural tissues derived from human induced pluripotent stem cells using a fibrin-based bioink. ACS Biomater. Sci. Eng. 2018, 5, 234–243. [Google Scholar] [CrossRef]
- Montgomery, A.; Wong, A.; Gabers, N.; Willerth, S.M. Engineering personalized neural tissue by combining induced pluripotent stem cells with fibrin scaffolds. Biomater. Sci. 2015, 3, 401–413. [Google Scholar] [CrossRef] [PubMed]
- Robinson, M.; Douglas, S.; Willerth, S.M. Mechanically stable fibrin scaffolds promote viability and induce neurite outgrowth in neural aggregates derived from human induced pluripotent stem cells. Sci. Rep. 2017, 7, 6250. [Google Scholar] [CrossRef] [PubMed]
- De la Vega, L.; A Rosas Gómez, D.; Abelseth, E.; Abelseth, L.; Allisson da Silva, V.; Willerth, S. 3d bioprinting human induced pluripotent stem cell-derived neural tissues using a novel lab-on-a-printer technology. Appl. Sci. 2018, 8, 2414. [Google Scholar] [CrossRef]
- Melchels, F.; Wiggenhauser, P.S.; Warne, D.; Barry, M.; Ong, F.R.; Chong, W.S.; Hutmacher, D.W.; Schantz, J.T. Cad/cam-assisted breast reconstruction. Biofabrication 2011, 3, 034114. [Google Scholar] [CrossRef] [PubMed]
- Pati, F.; Jang, J.; Ha, D.H.; Won Kim, S.; Rhie, J.W.; Shim, J.H.; Kim, D.H.; Cho, D.W. Printing three-dimensional tissue analogues with decellularized extracellular matrix bioink. Nat. Commun. 2014, 5, 3935. [Google Scholar] [CrossRef] [PubMed]
- Pati, F.; Ha, D.H.; Jang, J.; Han, H.H.; Rhie, J.W.; Cho, D.W. Biomimetic 3d tissue printing for soft tissue regeneration. Biomaterials 2015, 62, 164–175. [Google Scholar] [CrossRef] [PubMed]
- Chhaya, M.P.; Balmayor, E.R.; Hutmacher, D.W.; Schantz, J.T. Transformation of breast reconstruction via additive biomanufacturing. Sci. Rep. 2016, 6, 28030. [Google Scholar] [CrossRef]
- Lee, H.J.; Ock, J.J. How to improve projection in nipple reconstruction: A modified method using acellular dermal matrix disk and fragments. Plast. Reconstr. Surg. 2019, 143, 698e–706e. [Google Scholar] [CrossRef]
- Thomas, D.J. 3d bioprinting as a solution for engineering the nipple areola complex for breast cancer reconstruction. Int. J. Surg. 2017, 41, 14–15. [Google Scholar] [CrossRef]
- Khoo, D.; Ung, O.; Blomberger, D.; Hutmacher, D.W. Nipple reconstruction: A regenerative medicine approach using 3d-printed tissue scaffolds. Tissue Eng. Part B Rev. 2019, 25, 126–134. [Google Scholar] [CrossRef] [PubMed]
- Gabrick, K.S.; Markov, N.P.; Chouairi, F.; Wu, R.; Persing, S.M.; Abraham, P.; Avraham, T.; Alperovich, M. A predictive model for determining permanent implant size during 2-stage implant breast reconstruction. Plast. Reconstr. Surg. Glob. Open 2018, 6, e1790. [Google Scholar] [CrossRef] [PubMed]
- Patrick, C.W.; Uthamanthil, R.; Beahm, E.; Frye, C. Animal models for adipose tissue engineering. Tissue Eng. Part B Rev. 2008, 14, 167–178. [Google Scholar] [CrossRef] [PubMed]
- Akhtar, A. The flaws and human harms of animal experimentation. Camb. Q. Healthc. Ethics 2015, 24, 407–419. [Google Scholar] [CrossRef] [PubMed]
- Schnitt, S.J.; Connolly, J.L.; Harris, J.R.; Cohen, R.B. Radiation-induced changes in the breast. Hum. Pathol. 1984, 15, 545–550. [Google Scholar] [CrossRef]
- Moore, G.H.; Schiller, J.E.; Moore, G.K. Radiation-induced histopathologic changes of the breast: The effects of time. Am. J. Surg. Pathol. 2004, 28, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Girling, A.; Hanby, A.; Millis, R. Radiation and other pathological changes in breast tissue after conservation treatment for carcinoma. J. Clin. Pathol. 1990, 43, 152–156. [Google Scholar] [CrossRef] [PubMed]
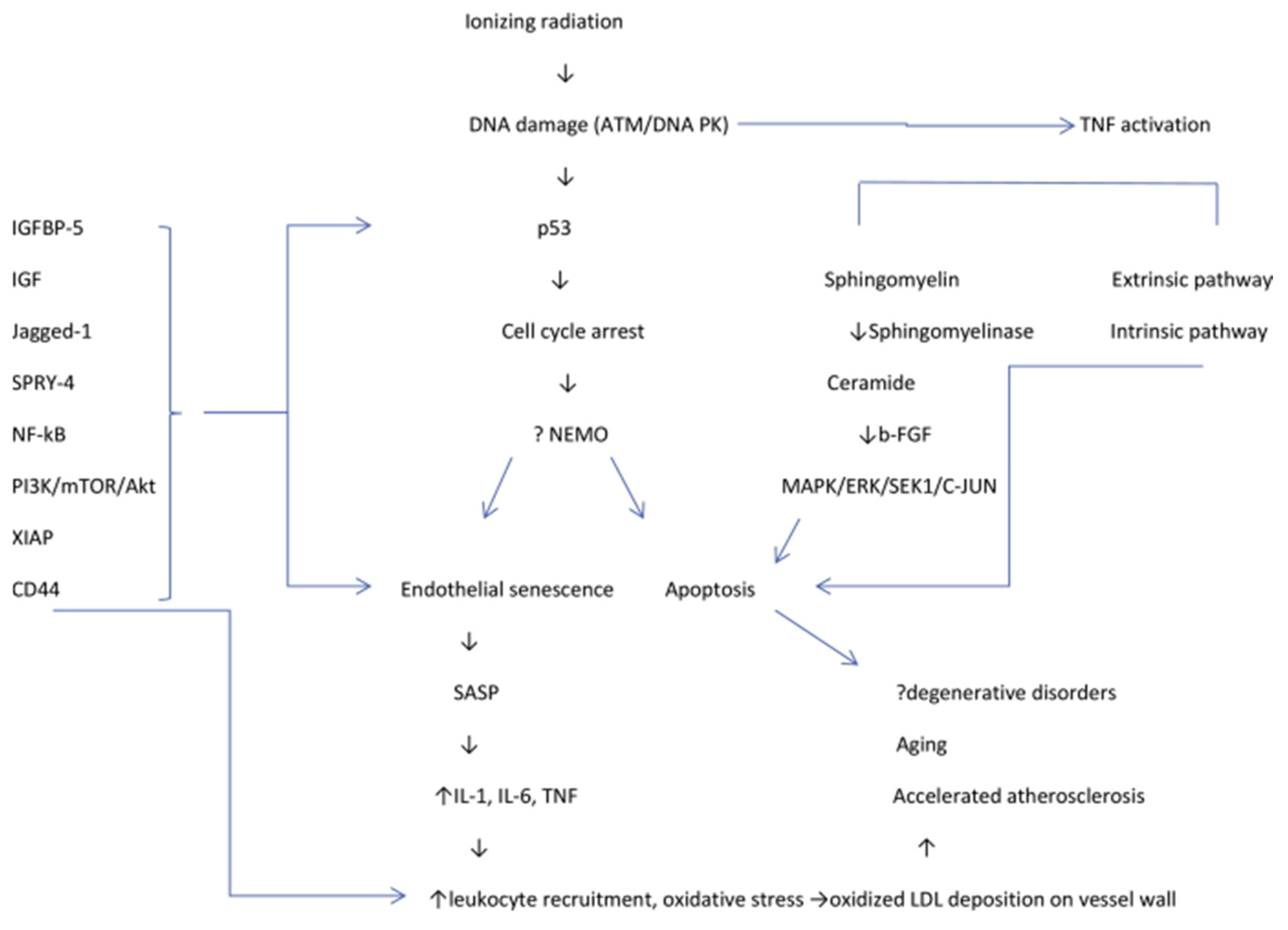
- Venkatesulu, B.P.; Mahadevan, L.S.; Aliru, M.L.; Yang, X.; Bodd, M.H.; Singh, P.K.; Yusuf, S.W.; Abe, J.I.; Krishnan, S. Radiation-induced endothelial vascular injury: A review of possible mechanisms. JACC Basic Transl. Sci. 2018, 3, 563–572. [Google Scholar] [CrossRef]
- Coyne, J.; Parkinson, D.; Baildam, A. Membranous fat necrosis of the breast. Histopathology 1996, 28, 61–64. [Google Scholar] [CrossRef]
- Clarke, D.; Curtis, J.L.; Martinez, A.; Fajardo, L.; Goffinet, D. Fat necrosis of the breast simulating recurrent carcinoma after primary radiotherapy in the management of early stage breast carcinoma. Cancer 1983, 52, 442–445. [Google Scholar] [CrossRef]
- Mondal, D.; Griffith, M.; Venkatraman, S.S. Polycaprolactone-based biomaterials for tissue engineering and drug delivery: Current scenario and challenges. Int. J. Polym. Mater. Polym. Biomater. 2016, 65, 255–265. [Google Scholar] [CrossRef]
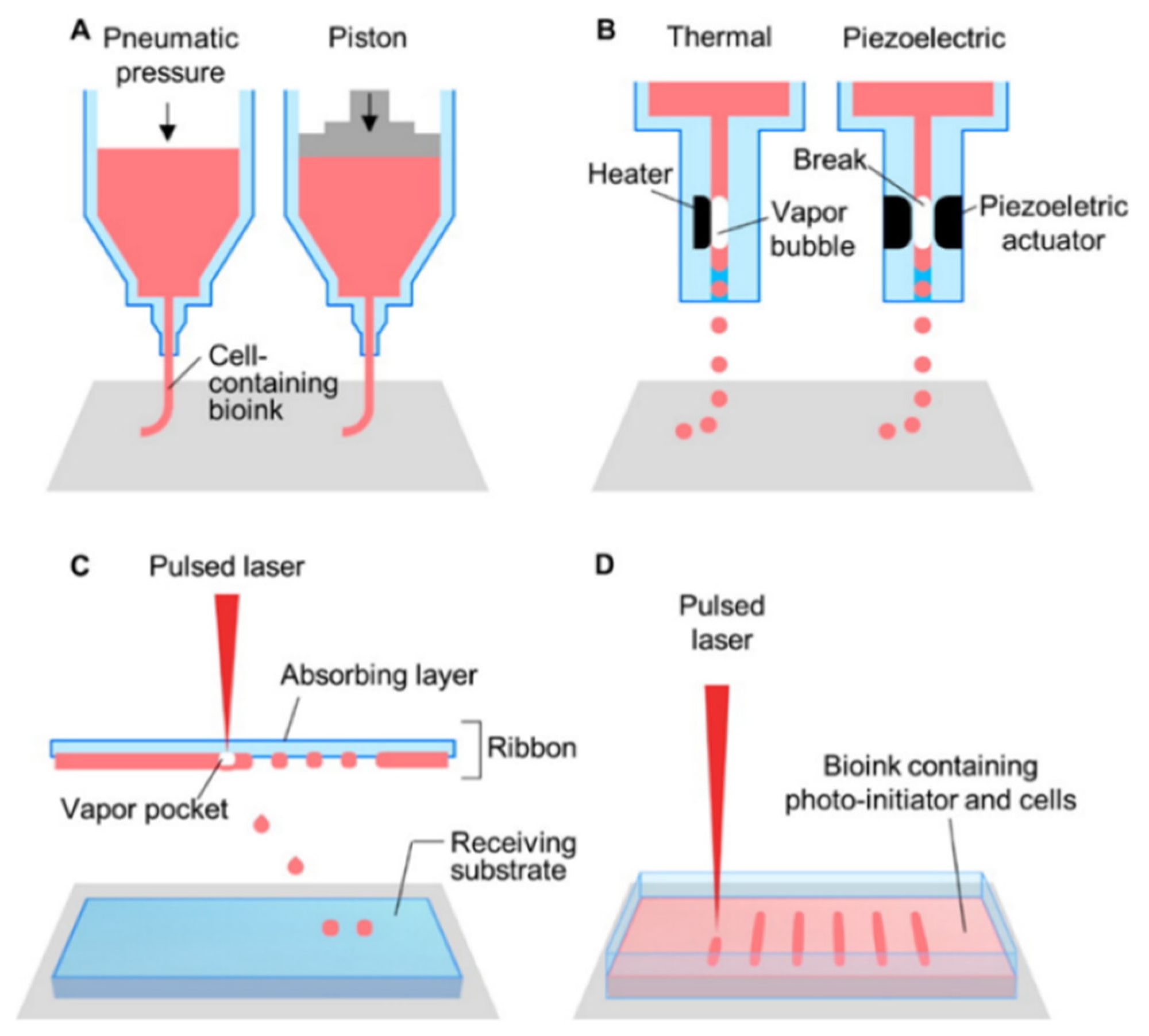
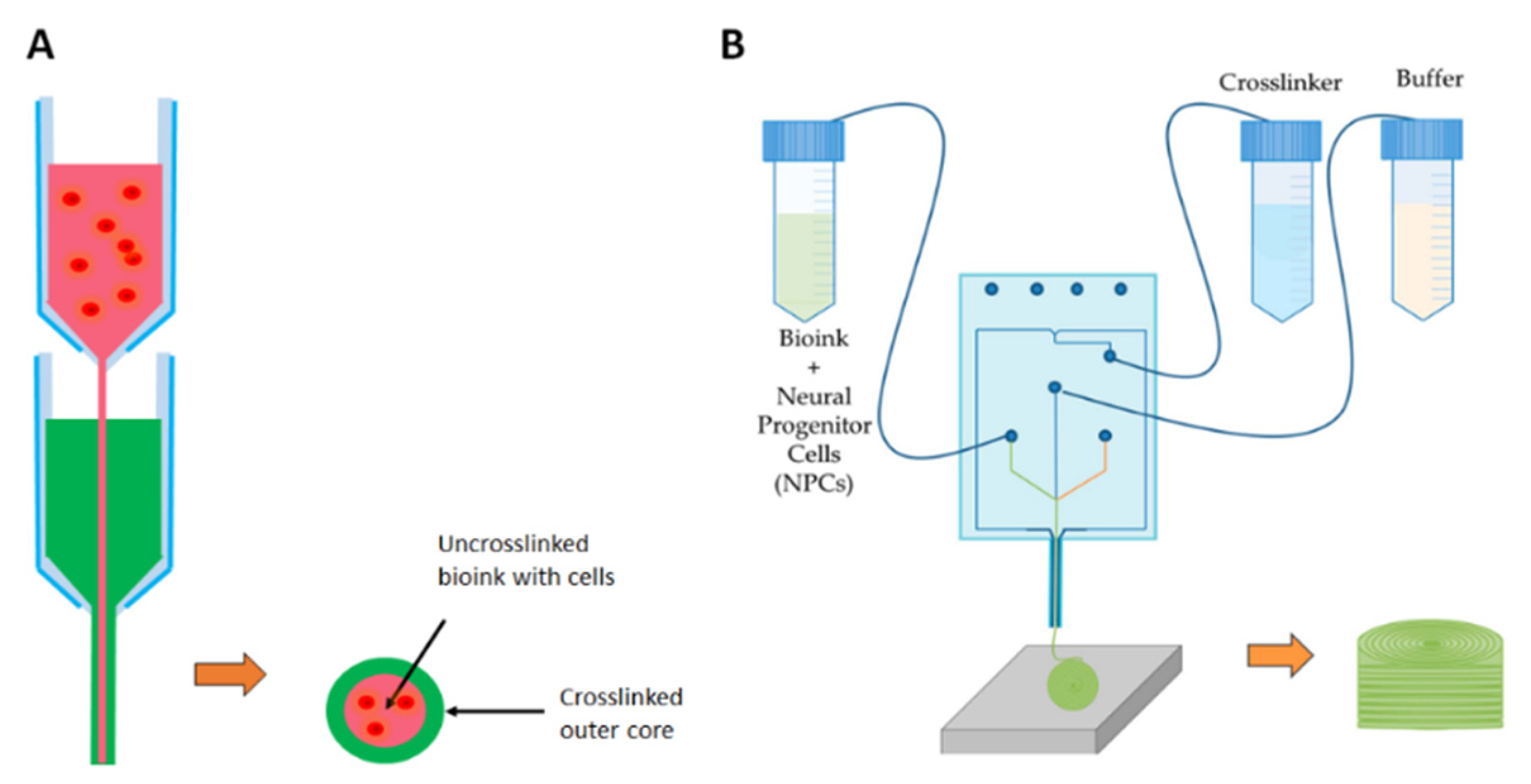
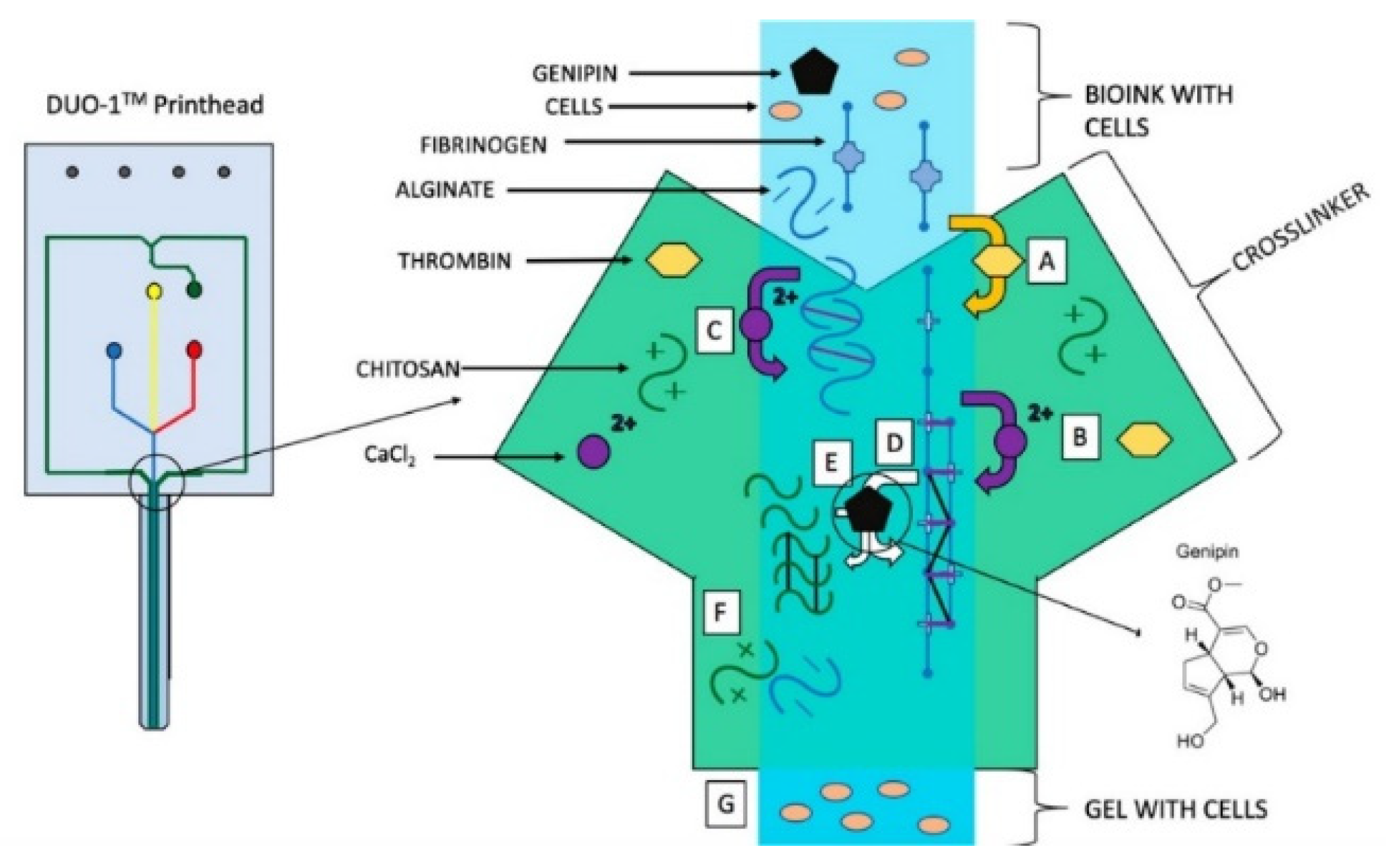
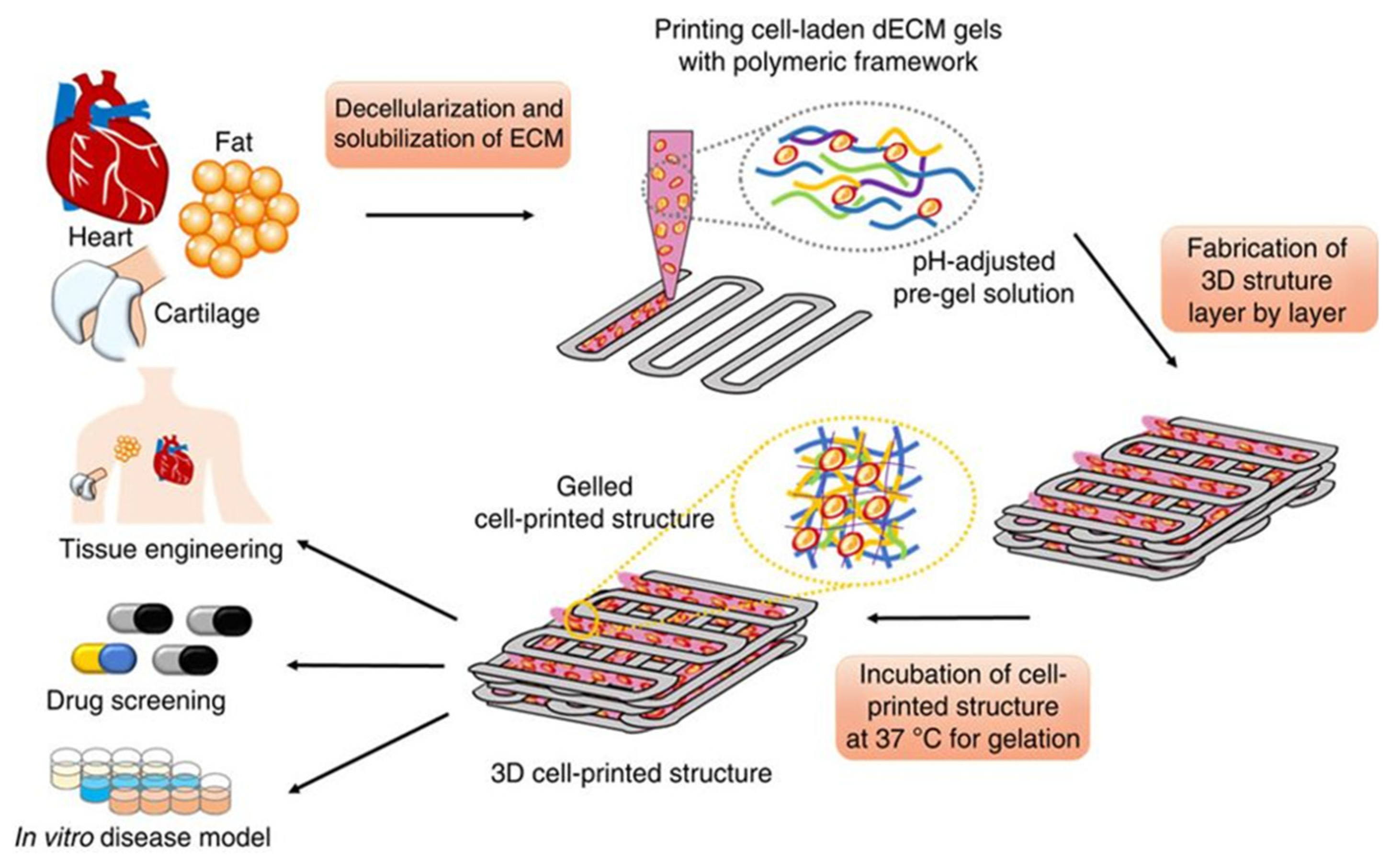
| Technique | Advantages | Disadvantages |
|---|---|---|
| Extrusion-based | Utilize high cell densities, high viscosity bioinks, print multiple bioinks simultaneously | Moderate resolution (~100 µm), slow speed, reduced cell viability secondary to shear stress, moderate cost |
| Advances: Melt writing Core-shell Microfluidic | Precise High cell viability High cell viability and resolution | Requires cell seeding post-print, moderate resolution (~100 µm) -- -- |
| Inkjet | High resolution (50 µm), fast speed, low cost, widely available | Not suited for viscous bioink, limited to non-complex architecture, reduced cell viability secondary to heat or shear stress, low cell density |
| Laser-assisted | No shear stress, viscous or solid bioink compatibility, high resolution (1–50 µm) | Reduced cell viability secondary to heat, medium speed, limited scalability, high cost |
| Stereolithography | No shear stress, high resolution (3–300 µm), high cell viability, fast speed, low cost | Require photo-curable bioink, UV toxicity to cells, poor hollow-structure capabilities |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cleversey, C.; Robinson, M.; Willerth, S.M. 3D Printing Breast Tissue Models: A Review of Past Work and Directions for Future Work. Micromachines 2019, 10, 501. https://doi.org/10.3390/mi10080501
Cleversey C, Robinson M, Willerth SM. 3D Printing Breast Tissue Models: A Review of Past Work and Directions for Future Work. Micromachines. 2019; 10(8):501. https://doi.org/10.3390/mi10080501
Chicago/Turabian StyleCleversey, Chantell, Meghan Robinson, and Stephanie M. Willerth. 2019. "3D Printing Breast Tissue Models: A Review of Past Work and Directions for Future Work" Micromachines 10, no. 8: 501. https://doi.org/10.3390/mi10080501
APA StyleCleversey, C., Robinson, M., & Willerth, S. M. (2019). 3D Printing Breast Tissue Models: A Review of Past Work and Directions for Future Work. Micromachines, 10(8), 501. https://doi.org/10.3390/mi10080501






