Effect of Cyclic Stretch on Tissue Maturation in Myoblast-Laden Hydrogel Fibers
Abstract
1. Introduction
2. Materials and Methods
2.1. Cells
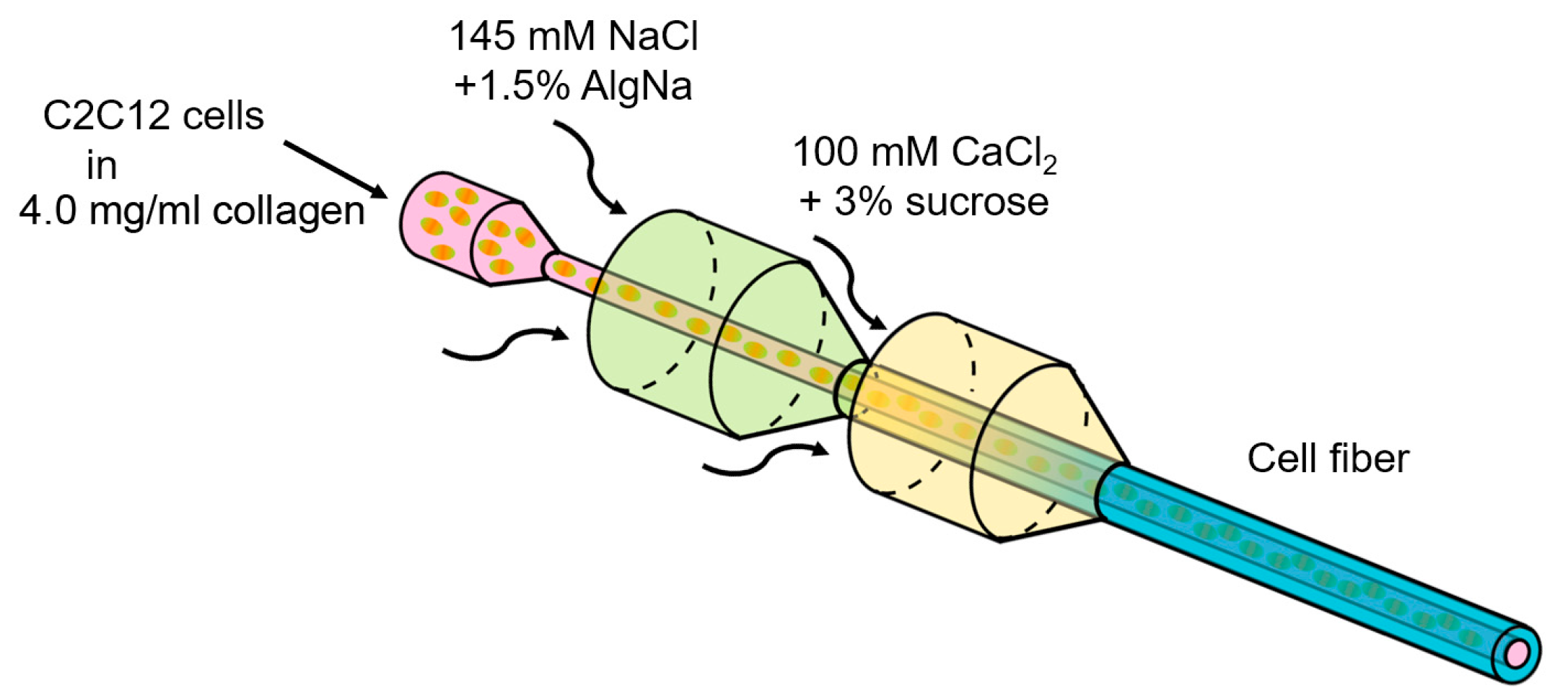
2.2. Formation of Core-Shell Hydrogel Microfibers
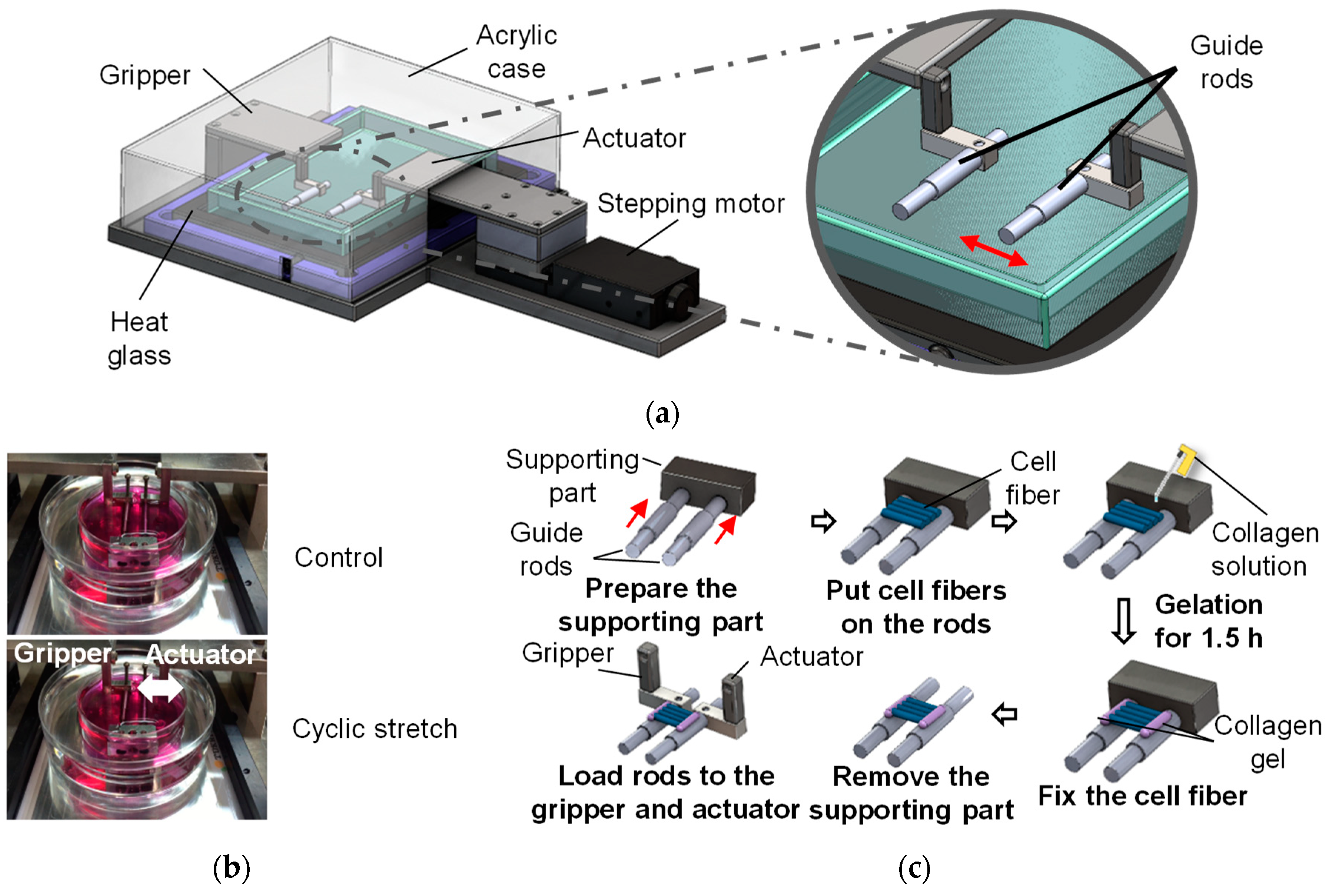
2.3. Three-Dimensional Cell Culture with Cyclic Stretch
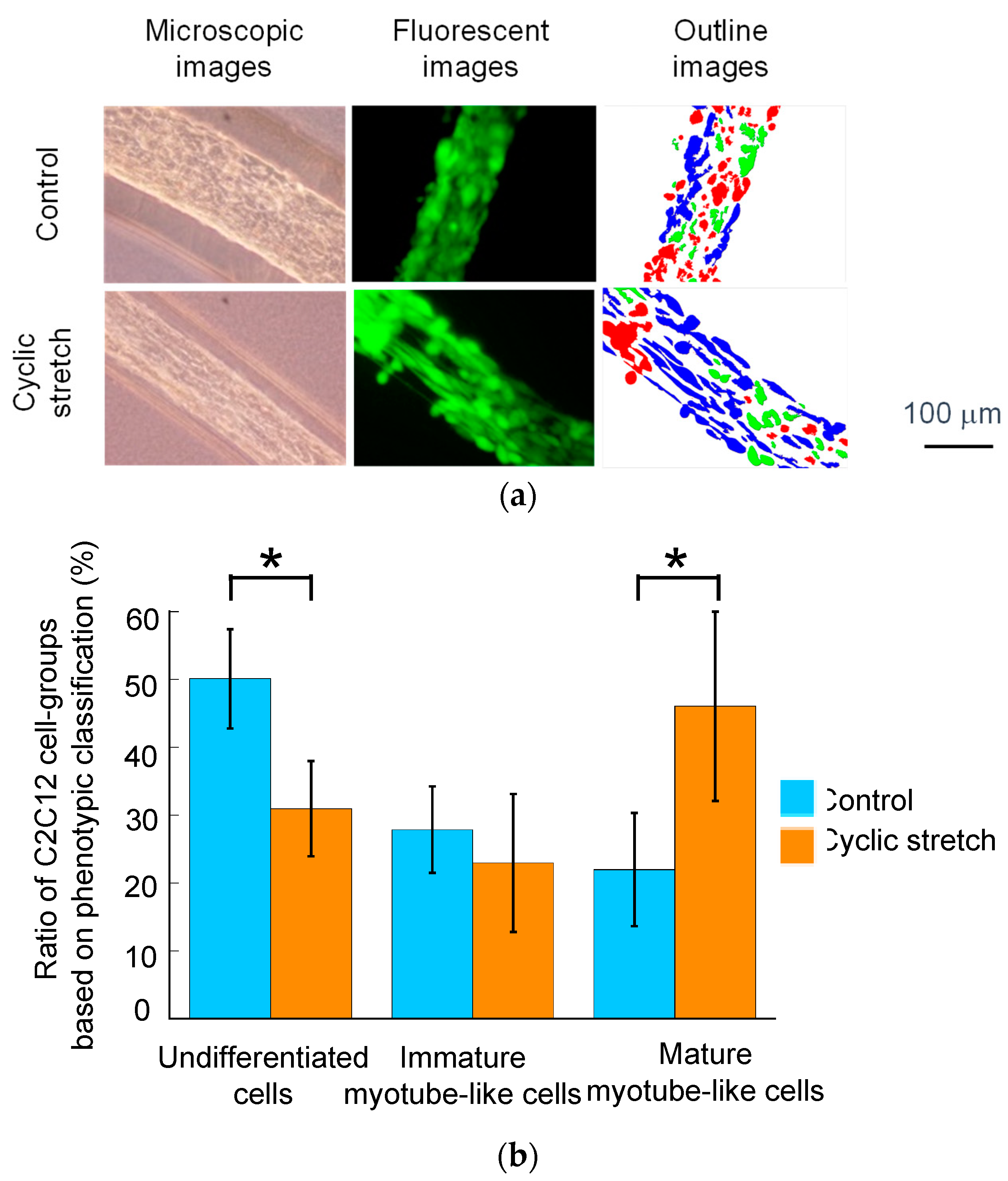
2.4. Microscopy and Image-Based Analysis
3. Results and Discussion
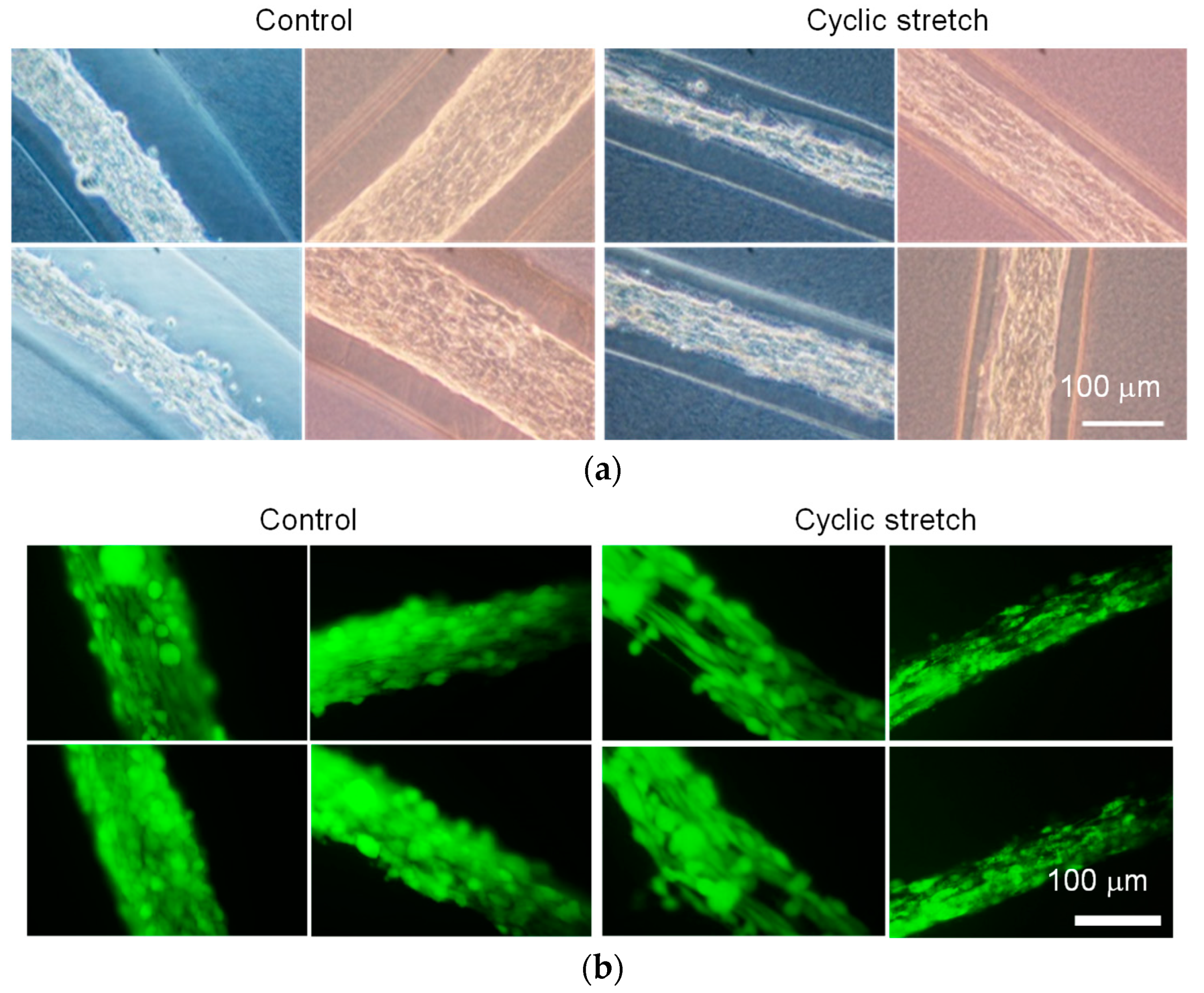
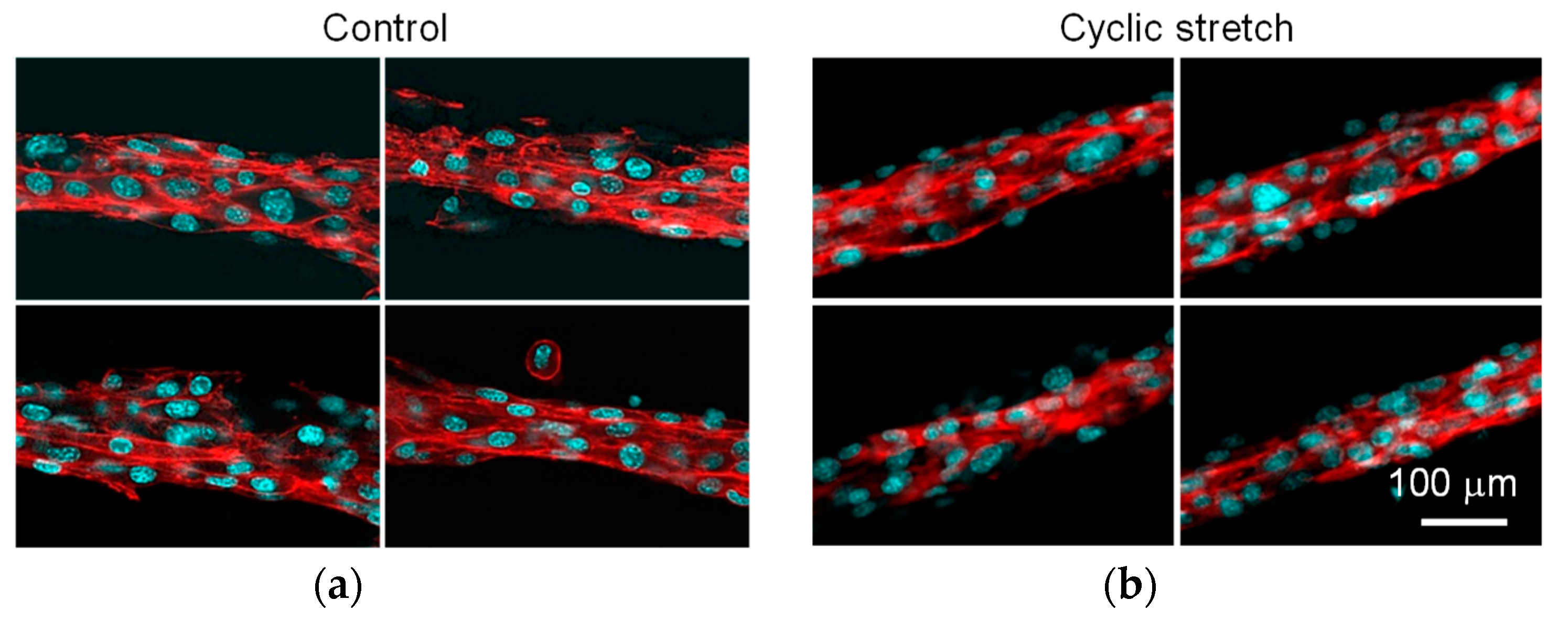
3.1. Difference in Tissue Remodeling in the Cell Fibers and in the Two-Dimensional Culture
3.2. Effect of Cyclic Stretch on Tissue-Reconstruction in the Cell Fibers
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Shah, R.; Knowles, J.C.; Hunt, N.P.; Lewis, M.P. Development of a novel smart scaffold for human 2 skeletal muscle regeneration. J. Tissue Eng. Regen. Med. 2016, 10, 162–171. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, H.; Okano, T. Cell Sheet-Based Tissue Engineering for Organizing Anisotropic Tissue Constructs Produced Using Microfabricated Thermoresponsive Substrates. Adv. Healthc. Mater. 2015, 4, 2388–2407. [Google Scholar] [CrossRef] [PubMed]
- Chimenti, I.; Gaetani, R.; Barile, L.; Forte, E.; Ionta, V.; Angelini, F.; Frati, G.; Messina, E.; Giacomello, A. Isolation and Expansion of Adult Cardiac Stem/Progenitor Cells in the Form of Cardiospheres from Human Cardiac Biopsies and Murine Hearts; Humana Press: Totowa, NJ, USA, 2012; pp. 327–338. [Google Scholar]
- Luo, B.; Tian, L.; Chen, N.; Ramakrishna, S.; Thakor, N.; Yang, I.H. Electrospun nanofibers facilitate better alignment, differentiation, and long-term culture in an in vitro model of the neuromuscular junction (NMJ). Biomater. Sci. 2018, 6, 3262–3272. [Google Scholar] [CrossRef] [PubMed]
- Fee, T.; Surianarayanan, S.; Downs, C.; Zhou, Y.; Berry, J. Nanofiber Alignment Regulates NIH3T3 Cell Orientation and Cytoskeletal Gene Expression on Electrospun PCL+ Gelatin Nanofibers. PLoS ONE 2016, 11. [Google Scholar] [CrossRef] [PubMed]
- Li, T.-S.; Cheng, K.; Lee, S.-T.; Matsushita, S.; Davis, D.; Malliaras, K.; Zhang, Y.; Matsushita, N.; Smith, R.R.; Marbán, E. Cardiospheres Recapitulate a Niche-Like Microenvironment Rich in Stemness and Cell-Matrix Interactions, Rationalizing Their Enhanced Functional Potency for Myocardial Repair. Stem Cells 2010, 28, 2088–2098. [Google Scholar] [CrossRef] [PubMed]
- Vandenburgh, H. High-Content Drug Screening with Engineered Musculoskeletal Tissues. Tissue Eng. Part B Rev. 2010, 16, 55–64. [Google Scholar] [CrossRef] [PubMed]
- Vandenburgh, H.; Shansky, J.; Benesch-Lee, F.; Barbata, V.; Reid, J.; Thorrez, L.; Valentini, R.; Crawford, G. Drug-screening platform based on the contractility of tissue-engineered muscle. Muscle Nerve 2008, 37, 438–447. [Google Scholar] [CrossRef]
- Kim, W.; Kim, J.; Park, H.-S.; Jeon, J.; Kim, W.; Kim, J.; Park, H.-S.; Jeon, J.S. Development of Microfluidic Stretch System for Studying Recovery of Damaged Skeletal Muscle Cells. Micromachines 2018, 9, 671. [Google Scholar] [CrossRef]
- Tchao, J.; Kim, J.J.; Lin, B.; Salama, G.; Lo, C.W.; Yang, L.; Tobita, K. Engineered Human Muscle Tissue from Skeletal Muscle Derived Stem Cells and Induced Pluripotent Stem Cell Derived Cardiac Cells. Int. J. Tissue Eng. 2013, 2013, 198762. [Google Scholar] [CrossRef]
- Liu, H.; Gong, X.; Jing, X.; Ding, X.; Yao, Y.; Huang, Y.; Fan, Y. Shear stress with appropriate time-step and amplification enhances endothelial cell retention on vascular grafts. J. Tissue Eng. Regen. Med. 2017, 11, 2965–2978. [Google Scholar] [CrossRef]
- Kaunas, R.; Nguyen, P.; Usami, S.; Chien, S. From the Cover: Cooperative effects of Rho and mechanical stretch on stress fiber organization. Proc. Natl. Acad. Sci. USA 2005, 102, 15895–15900. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Shan, S.; Wang, C.; Wang, J.; Li, J.; Hu, G.; Dai, K.; Li, Q.; Zhang, X. Mechanical stimulation promote the osteogenic differentiation of bone marrow stromal cells through epigenetic regulation of Sonic Hedgehog. Exp. Cell Res. 2017, 352, 346–356. [Google Scholar] [CrossRef] [PubMed]
- Oortgiesen, D.A.W.; Yu, N.; Bronckers, A.L.J.J.; Yang, F.; Walboomers, X.F.; Jansen, J.A. A three-dimensional cell culture model to study the mechano-biological behavior in periodontal ligament regeneration. Tissue Eng. Part C Methods 2012, 18, 81–89. [Google Scholar] [CrossRef]
- Kaji, H.; Ishibashi, T.; Nagamine, K.; Kanzaki, M.; Nishizawa, M. Electrically induced contraction of C2C12 myotubes cultured on a porous membrane-based substrate with muscle tissue-like stiffness. Biomaterials 2010, 31, 6981–6986. [Google Scholar] [CrossRef]
- Grossi, A.; Lametsch, R.; Karlsson, A.H.; Lawson, M.A. Mechanical stimuli on C2C12 myoblasts affect myoblast differentiation, focal adhesion kinase phosphorylation and galectin-1 expression: A proteomic approach. Cell Biol. Int. 2011, 35, 579–586. [Google Scholar] [CrossRef] [PubMed]
- Kasper, A.M.; Turner, D.C.; Martin, N.R.W.; Sharples, A.P. Mimicking exercise in three-dimensional bioengineered skeletal muscle to investigate cellular and molecular mechanisms of physiological adaptation. J. Cell. Physiol. 2018, 233, 1985–1998. [Google Scholar] [CrossRef] [PubMed]
- Hornberger, T.A.; Armstrong, D.D.; Koh, T.J.; Burkholder, T.J.; Esser, K.A. Intracellular signaling specificity in response to uniaxial vs. multiaxial stretch: Implications for mechanotransduction. Am. J. Physiol. Cell Physiol. 2005, 288, C185–C194. [Google Scholar] [CrossRef] [PubMed]
- Asano, S.; Ito, S.; Morosawa, M.; Furuya, K.; Naruse, K.; Sokabe, M.; Yamaguchi, E.; Hasegawa, Y. Cyclic stretch enhances reorientation and differentiation of 3-D culture model of human airway smooth muscle. Biochem. Biophys. Rep. 2018, 16, 32–38. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, T.D.; Liang, R.; Woo, S.L.-Y.; Burton, S.D.; Wu, C.; Almarza, A.; Sacks, M.S.; Abramowitch, S. Effects of cell seeding and cyclic stretch on the fiber remodeling in an extracellular matrix-derived bioscaffold. Tissue Eng. Part A 2009, 15, 957–963. [Google Scholar] [CrossRef] [PubMed]
- Onoe, H.; Okitsu, T.; Itou, A.; Kato-Negishi, M.; Gojo, R.; Kiriya, D.; Sato, K.; Miura, S.; Iwanaga, S.; Kuribayashi-Shigetomi, K.; et al. Metre-long cell-laden microfibres exhibit tissue morphologies and functions. Nat. Mater. 2013, 12, 584–590. [Google Scholar] [CrossRef] [PubMed]
- Li, R.H.; Altreuter, D.H.; Gentile, F.T. Transport characterization of hydrogel matrices for cell encapsulation. Biotechnol. Bioeng. 1996, 50, 365–373. [Google Scholar] [CrossRef]
- Park, H.; Bhalla, R.; Saigal, R.; Radisic, M.; Watson, N.; Langer, R.; Vunjak-Novakovic, G. Effects of electrical stimulation in C2C12 muscle constructs. J. Tissue Eng. Regen. Med. 2008, 2, 279–287. [Google Scholar] [CrossRef] [PubMed]
- Yamasaki, K.; Hayashi, H.; Nishiyama, K.; Kobayashi, H.; Uto, S.; Kondo, H.; Hashimoto, S.; Fujisato, T. Control of myotube contraction using electrical pulse stimulation for bio-actuator. J. Artif. Organs 2009, 12, 131–137. [Google Scholar] [CrossRef] [PubMed]
- Fujita, H.; Endo, A.; Shimizu, K.; Nagamori, E. Evaluation of serum-free differentiation conditions for C2C12 myoblast cells assessed as to active tension generation capability. Biotechnol. Bioeng. 2010, 107, 894–901. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.-J.; Chen, Y.-J.; Huang, C.-W.; Fan, S.-C.; Huang, B.-M.; Chang, W.-T.; Tsai, Y.-S.; Su, F.-C.; Wu, C.-C. Cyclic Stretch Facilitates Myogenesis in C2C12 Myoblasts and Rescues Thiazolidinedione-Inhibited Myotube Formation. Front. Bioeng. Biotechnol. 2016, 4, 27. [Google Scholar] [CrossRef] [PubMed]
- Evans, D.J.; Britland, S.; Wigmore, P.M. Differential response of fetal and neonatal myoblasts to topographical guidance cues in vitro. Dev. Genes Evol. 1999, 209, 438–442. [Google Scholar] [CrossRef] [PubMed]
- Lam, M.T.; Sim, S.; Zhu, X.; Takayama, S. The effect of continuous wavy micropatterns on silicone substrates on the alignment of skeletal muscle myoblasts and myotubes. Biomaterials 2006, 27, 4340–4347. [Google Scholar] [CrossRef] [PubMed]
- Guex, A.G.; Birrer, D.L.; Fortunato, G.; Tevaearai, H.T.; Giraud, M.-N. Anisotropically oriented electrospun matrices with an imprinted periodic micropattern: A new scaffold for engineered muscle constructs. Biomed. Mater. 2013, 8, 021001. [Google Scholar] [CrossRef] [PubMed]
- Lam, M.T.; Huang, Y.-C.; Birla, R.K.; Takayama, S. Microfeature guided skeletal muscle tissue engineering for highly organized 3-dimensional free-standing constructs. Biomaterials 2009, 30, 1150–1155. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Hattori-Aramaki, N.; Sunohara, A.; Okabe, K.; Sakamoto, Y.; Ochiai, H.; Hayashi, R.; Kishi, K. Alignment of Skeletal Muscle Cells Cultured in Collagen Gel by Mechanical and Electrical Stimulation. Int. J. Tissue Eng. 2014, 2014, 621529. [Google Scholar] [CrossRef]
- Ikeda, K.; Nagata, S.; Okitsu, T.; Takeuchi, S. Cell fiber-based three-dimensional culture system for highly efficient expansion of human induced pluripotent stem cells. Sci. Rep. 2017, 7, 2850. [Google Scholar] [CrossRef] [PubMed]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bansai, S.; Morikura, T.; Onoe, H.; Miyata, S. Effect of Cyclic Stretch on Tissue Maturation in Myoblast-Laden Hydrogel Fibers. Micromachines 2019, 10, 399. https://doi.org/10.3390/mi10060399
Bansai S, Morikura T, Onoe H, Miyata S. Effect of Cyclic Stretch on Tissue Maturation in Myoblast-Laden Hydrogel Fibers. Micromachines. 2019; 10(6):399. https://doi.org/10.3390/mi10060399
Chicago/Turabian StyleBansai, Shinako, Takashi Morikura, Hiroaki Onoe, and Shogo Miyata. 2019. "Effect of Cyclic Stretch on Tissue Maturation in Myoblast-Laden Hydrogel Fibers" Micromachines 10, no. 6: 399. https://doi.org/10.3390/mi10060399
APA StyleBansai, S., Morikura, T., Onoe, H., & Miyata, S. (2019). Effect of Cyclic Stretch on Tissue Maturation in Myoblast-Laden Hydrogel Fibers. Micromachines, 10(6), 399. https://doi.org/10.3390/mi10060399




