Surface Engineering of Carbon-Based Microelectrodes for High-Performance Microsupercapacitors
Abstract
1. Introduction
2. Experimental
2.1. Preparation of the Developer
2.2. Microfabrication process of Microsupercapacitor (MSC) with Carbon Nanotubes (CNTs) Modification
2.3. Characterization and Electrochemical Performance Measurements
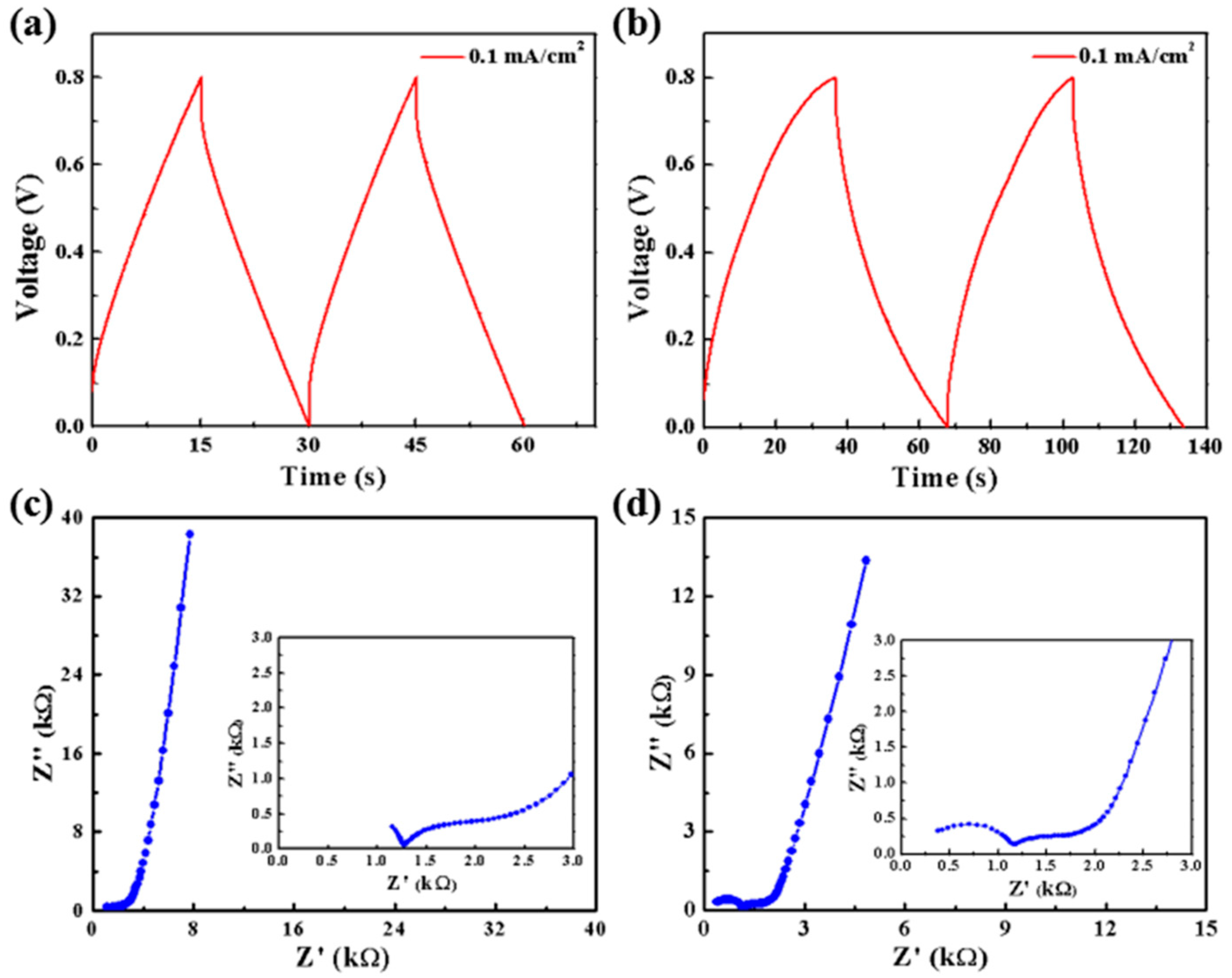
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Simon, P.; Gogotsi, Y. Materials for electrochemical capacitors. Nat. Mater. 2008, 7, 845. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Zou, W.; Quan, B.; Yu, A.; Wu, H.; Jiang, P.; Wei, Z. An All-Solid-State Flexible Micro-supercapacitor on a Chip. Adv. Energy Mater. 2011, 1, 1068. [Google Scholar] [CrossRef]
- Yang, P.; Mai, W. Flexible solid-state electrochemical supercapacitors. Nano Energy 2014, 8, 274. [Google Scholar] [CrossRef]
- Yu, Z.; Tetard, L.; Zhai, L.; Thomas, J. Supercapacitor electrode materials: nanostructures from 0 to 3 dimensions. Energy Environ. Sci. 2015, 8, 702. [Google Scholar] [CrossRef]
- Brousse, T.; Bélanger, D.; Long, J.W. To be or not to be pseudocapacitive? J. Electrochem. Soc. 2015, 162, A5185. [Google Scholar] [CrossRef]
- Jiang, S.; Shi, T.; Zhan, X.; Xi, S.; Long, H.; Gong, B.; Li, J.; Cheng, S.; Huang, Y.; Tang, Z. Scalable fabrication of carbon-based MEMS/NEMS and their applications: a review. J. Micromech. Microeng. 2015, 25, 113001. [Google Scholar] [CrossRef]
- Xiao, Y.; Huang, L.; Zhang, Q.; Xu, S.; Chen, Q.; Shi, W. Gravure printing of hybrid MoS2@ S-rGO interdigitated electrodes for flexible microsupercapacitors. Appl. Phys. Lett. 2015, 107, 013906. [Google Scholar] [CrossRef]
- Beidaghi, M.; Gogotsi, Y. Capacitive energy storage in micro-scale devices: recent advances in design and fabrication of micro-supercapacitors. Energy Environ. Sci. 2014, 7, 867. [Google Scholar] [CrossRef]
- Eustache, E.; Douard, C.; Retoux, R.; Lethien, C.; Brousse, T. MnO2 thin films on 3D scaffold: microsupercapacitor electrodes competing with “bulk” carbon electrodes. Adv. Energy Mater. 2015, 5, 1500680. [Google Scholar] [CrossRef]
- Hsia, B.; Kim, M.S.; Vincent, M.; Carraro, C.; Maboudian, R. Photoresist-derived porous carbon for on-chip micro-supercapacitors. Carbon 2013, 57, 395. [Google Scholar] [CrossRef]
- Ranganathan, S.; McCreery, R.; Majji, S.M.; Madou, M. Photoresist-derived carbon for microelectromechanical systems and electrochemical applications. J. Electrochem. Soc. 2000, 147, 277. [Google Scholar] [CrossRef]
- Wang, S.; Hsia, B.; Carraro, C.; Maboudian, R. High-performance all solid-state micro-supercapacitor based on patterned photoresist-derived porous carbon electrodes and an ionogel electrolyte. J. Mater. Chem. A 2014, 2, 7997. [Google Scholar] [CrossRef]
- Jung, H.; Cheah, C.V.; Jeong, N.; Lee, J. Direct printing and reduction of graphite oxide for flexible supercapacitors. Appl. Phys. Lett. 2014, 105, 053902. [Google Scholar] [CrossRef]
- Beidaghi, M.; Wang, C. Supercapacitors: Micro-Supercapacitors Based on Interdigital Electrodes of Reduced Graphene Oxide and Carbon Nanotube Composites with Ultrahigh Power Handling Performance. Adv. Funct. Mater. 2012, 22, 4500. [Google Scholar] [CrossRef]
- Gao, W.; Singh, N.; Song, L.; Liu, Z.; Reddy, A.L.M.; Ci, L.; Vajtai, R.; Zhang, Q.; Wei, B.; Ajayan, P.M. Direct laser writing of micro-supercapacitors on hydrated graphite oxide films. Nat. Nano 2011, 6, 496. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.; Wang, X.; Zhang, K.; Zou, J.; Zhang, Q. Metal-free SWNT/carbon/MnO2 hybrid electrode for high performance coplanar micro-supercapacitors. Nano Energy 2016, 22, 11. [Google Scholar] [CrossRef]
- Hu, H.; Pei, Z.; Ye, C. Recent advances in designing and fabrication of planar micro-supercapacitors for on-chip energy storage. Energy Storage Mater. 2015, 1, 82. [Google Scholar] [CrossRef]
- Wu, B.; Kumar, A. Extreme ultraviolet lithography and three dimensional integrated circuit—A review. Appl. Phys. Rev. 2014, 1, 011104. [Google Scholar] [CrossRef]
- Kwon, S.; Jung, D.; Lim, H.; Kim, G.; Choi, K.B.; Lee, J. Laser-assisted selective lithography of reduced graphene oxide for fabrication of graphene-based out-of-plane tandem microsupercapacitors with large capacitance. Appl. Phys. Lett. 2017, 111, 143903. [Google Scholar] [CrossRef]
- Zhou, P.; Yang, X.; He, L.; Hao, Z.; Luo, W.; Xiong, B.; Xu, X.; Niu, C.; Yan, M.; Mai, L. The Young’s modulus of high-aspect-ratio carbon/carbon nanotube composite microcantilevers by experimental and modeling validation. Appl. Phys. Lett. 2015, 106, 111908. [Google Scholar] [CrossRef]
- An, Z.; He, L.; Toda, M.; Yamamoto, G.; Hashida, T.; Ono, T. Microstructuring of carbon nanotubes-nickel nanocomposite. Nanotechnology 2015, 26, 195601. [Google Scholar] [CrossRef]
- He, L.; Toda, M.; Kawai, Y.; Miyashita, H.; Omori, M.; Hashida, T.; Berger, R.; Ono, T. Fabrication of CNT-carbon composite microstructures using Si micromolding and pyrolysis. Microsyst. Technol. 2014, 20, 201. [Google Scholar] [CrossRef]
- Hong, X.; He, L.; Ma, X.; Yang, W.; Chen, Y.; Zhang, L.; Yan, H.; Li, Z.; Mai, L. Microstructuring of carbon/tin quantum dots via a novel photolithography and pyrolysis-reduction process. Nano Res. 2017, 10, 3743. [Google Scholar] [CrossRef]
- Kim, M.S.; Hsia, B.; Carraro, C.; Maboudian, R. Flexible micro-supercapacitors with high energy density from simple transfer of photoresist-derived porous carbon electrodes. Carbon 2014, 74, 163. [Google Scholar] [CrossRef]
- Yang, W.; He, L.; Tian, X.; Yan, M.; Yuan, H.; Liao, X.; Meng, J.; Hao, Z.; Mai, L. Carbon-MEMS-Based Alternating Stacked MoS2@ rGO-CNT Micro-Supercapacitor with High Capacitance and Energy Density. Small 2017, 13, 1700639. [Google Scholar] [CrossRef]
- Yang, Y.; He, L.; Tang, C.; Hu, P.; Hong, X.; Yan, M.; Dong, Y.; Tian, X.; Wei, Q.; Mai, L. Improved conductivity and capacitance of interdigital carbon microelectrodes through integration with carbon nanotubes for micro-supercapacitors. Nano Res. 2016, 9, 2510. [Google Scholar] [CrossRef]
- Yin, C.; He, L.; Wang, Y.; Liu, Z.; Zhang, G.; Zhao, K.; Tang, C.; Yan, M.; Han, Y.; Mai, L. Pyrolyzed carbon with embedded NiO/Ni nanospheres for applications in microelectrodes. RSC Adv. 2016, 6, 43436. [Google Scholar] [CrossRef]
- Ma, X.; Feng, S.; He, L.; Yan, M.; Tian, X.; Li, Y.; Tang, C.; Hong, X.; Mai, L. Rapid, all dry microfabrication of three-dimensional Co3O4/Pt nanonetworks for high-performance microsupercapacitors. Nanoscale 2017, 9, 11765. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Chen, G.; Du, Y.; Xu, J.; Wu, S.; Qu, Y.; Zhu, Y. Plasmonic-enhanced Raman scattering of graphene on growth substrates and its application in SERS. Nanoscale 2014, 6, 13754. [Google Scholar] [CrossRef]
- Wang, H.; Peng, C.; Zheng, J.; Peng, F.; Yu, H. Design, synthesis and the electrochemical performance of MnO2/C@ CNT as supercapacitor material. Mater. Res. Bull. 2013, 48, 3389. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, Y.; Cheng, T.; Lai, W.; Pang, H.; Huang, W. Flexible supercapacitors based on paper substrates: a new paradigm for low-cost energy storage. Chem. Soc. Rev. 2015, 44, 5181. [Google Scholar] [CrossRef]
- Park, B.Y.; Taherabadi, L.; Wang, C.; Zoval, J.; Madou, M.J. Electrical properties and shrinkage of carbonized photoresist films and the implications for carbon microelectromechanical systems devices in conductive media. J. Electrochem. Soc. 2005, 152, J136. [Google Scholar] [CrossRef]
- Xu, G.; Zheng, C.; Zhang, Q.; Huang, J.; Zhao, M.; Nie, J.; Wang, X.; Wei, F. Binder-free activated carbon/carbon nanotube paper electrodes for use in supercapacitors. Nano Res. 2011, 4, 870. [Google Scholar] [CrossRef]
- Yun, J.; Kim, D.; Lee, G.; Ha, J.S. All-solid-state flexible micro-supercapacitor arrays with patterned graphene/MWNT electrodes. Carbon 2014, 79, 156. [Google Scholar] [CrossRef]
- Xiao, H.; Wu, Z.S.; Chen, L.; Zhou, F.; Zheng, S.; Ren, W.; Cheng, H.M.; Bao, X. One-step device fabrication of phosphorene and graphene interdigital micro-supercapacitors with high energy density. ACS Nano 2017, 11, 7284. [Google Scholar] [CrossRef] [PubMed]
- Pal, R.K.; Kundu, S.C.; Yadavalli, V.K. Fabrication of Flexible, Fully Organic, Degradable Energy Storage Devices Using Silk Proteins. ACS Appl. Mater. Interfaces 2018, 10, 9620. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Chen, H.; Chen, X.; Wu, H.; Guo, H.; Cheng, X.; Meng, B.; Zhang, H. All-in-one piezoresistive-sensing patch integrated with micro-supercapacitor. Nano Energy 2018, 53, 189. [Google Scholar] [CrossRef]
- Ye, J.; Tan, H.; Wu, S.; Ni, K.; Pan, F.; Liu, J.; Tao, Z.; Qu, Y.; Ji, H.; Simon, P.; Zhu, Y. Direct Laser Writing of Graphene Made from Chemical Vapor Deposition for Flexible, Integratable Micro-Supercapacitors with Ultrahigh Power Output. Adv. Mater. 2018, 30, e1801384. [Google Scholar] [CrossRef] [PubMed]
- Lochmann, S.; Grothe, J.; Eckhardt, K.; Leistenschneider, D.; Borchardt, L.; Kaskel, S. Nanoimprint lithography of nanoporous carbon materials for micro-supercapacitor architectures. Nanoscale 2018, 10, 10109. [Google Scholar] [CrossRef] [PubMed]




| Fabrication Method | Specific Capacitance (mF/cm2) | Materials | Ref. |
|---|---|---|---|
| This work | 4.80 | Pyrolyzed carbon/CNTs | - |
| Mask-assisted filtration | 9.8 | Phosphorene/graphene | [35] |
| Lithography | 5.9 | Carbon | [36] |
| Preset filling | 0.249 | CNTs | [37] |
| Laser-assisted method | 0.0627 | Graphene | [38] |
| Nanoimprint lithography | 0.008 | Carbon | [39] |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
He, L.; Hong, T.; Huang, Y.; Xiong, B.; Hong, X.; Tahir, M.; Haider, W.A.; Han, Y. Surface Engineering of Carbon-Based Microelectrodes for High-Performance Microsupercapacitors. Micromachines 2019, 10, 307. https://doi.org/10.3390/mi10050307
He L, Hong T, Huang Y, Xiong B, Hong X, Tahir M, Haider WA, Han Y. Surface Engineering of Carbon-Based Microelectrodes for High-Performance Microsupercapacitors. Micromachines. 2019; 10(5):307. https://doi.org/10.3390/mi10050307
Chicago/Turabian StyleHe, Liang, Tianjiao Hong, Yue Huang, Biao Xiong, Xufeng Hong, Muhammad Tahir, Waqas Ali Haider, and Yulai Han. 2019. "Surface Engineering of Carbon-Based Microelectrodes for High-Performance Microsupercapacitors" Micromachines 10, no. 5: 307. https://doi.org/10.3390/mi10050307
APA StyleHe, L., Hong, T., Huang, Y., Xiong, B., Hong, X., Tahir, M., Haider, W. A., & Han, Y. (2019). Surface Engineering of Carbon-Based Microelectrodes for High-Performance Microsupercapacitors. Micromachines, 10(5), 307. https://doi.org/10.3390/mi10050307





