Postbiotics and Kidney Disease
Abstract
:1. The Global Burden of Kidney Disease
2. The Gut Microbiota: A Key Modifier of Kidney Disease and Health
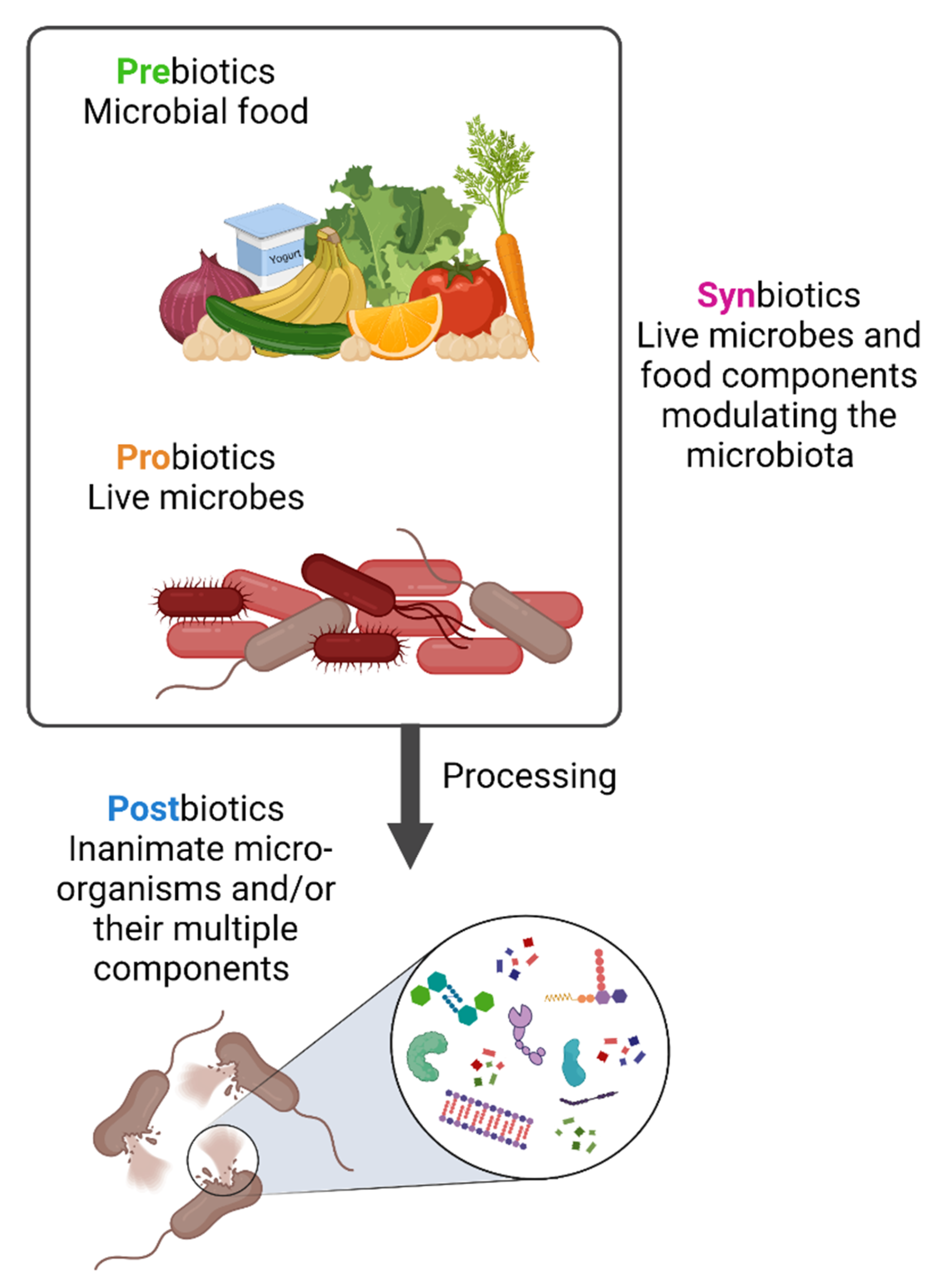
3. Prebiotics, Probiotics, Synbiotics and Postbiotics
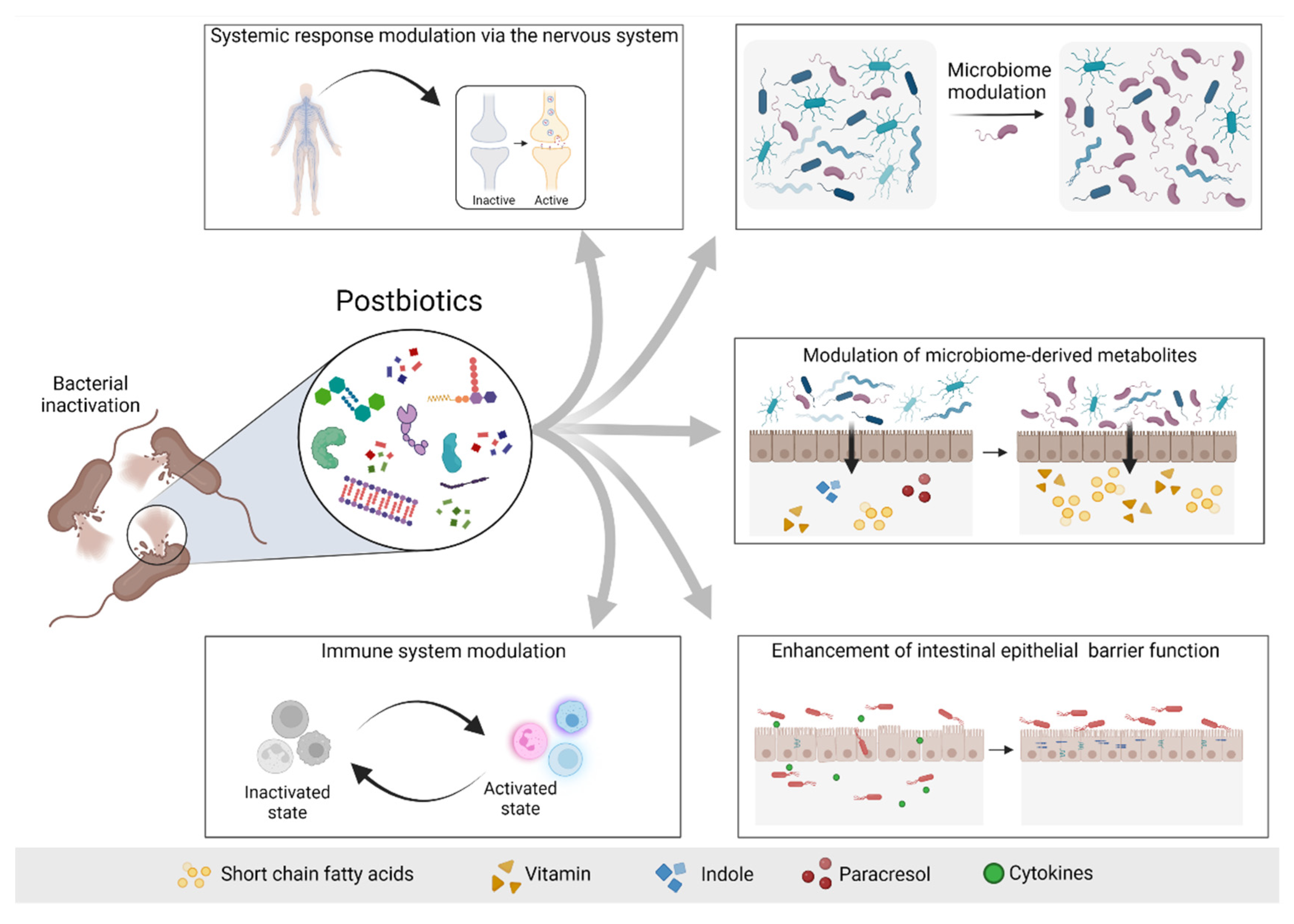
4. The 2019 Concept of Postbiotic: What Is and What Is Not a Postbiotic
5. Postbiotics in Non-Kidney Disease
6. Postbiotics in Kidney Disease
7. Postbiotics in Hyperoxaluria: Oxalobacter formigenes Lysates
8. Diet-Induced Kidney Injury
9. Postbiotics in Nephrotoxic AKI
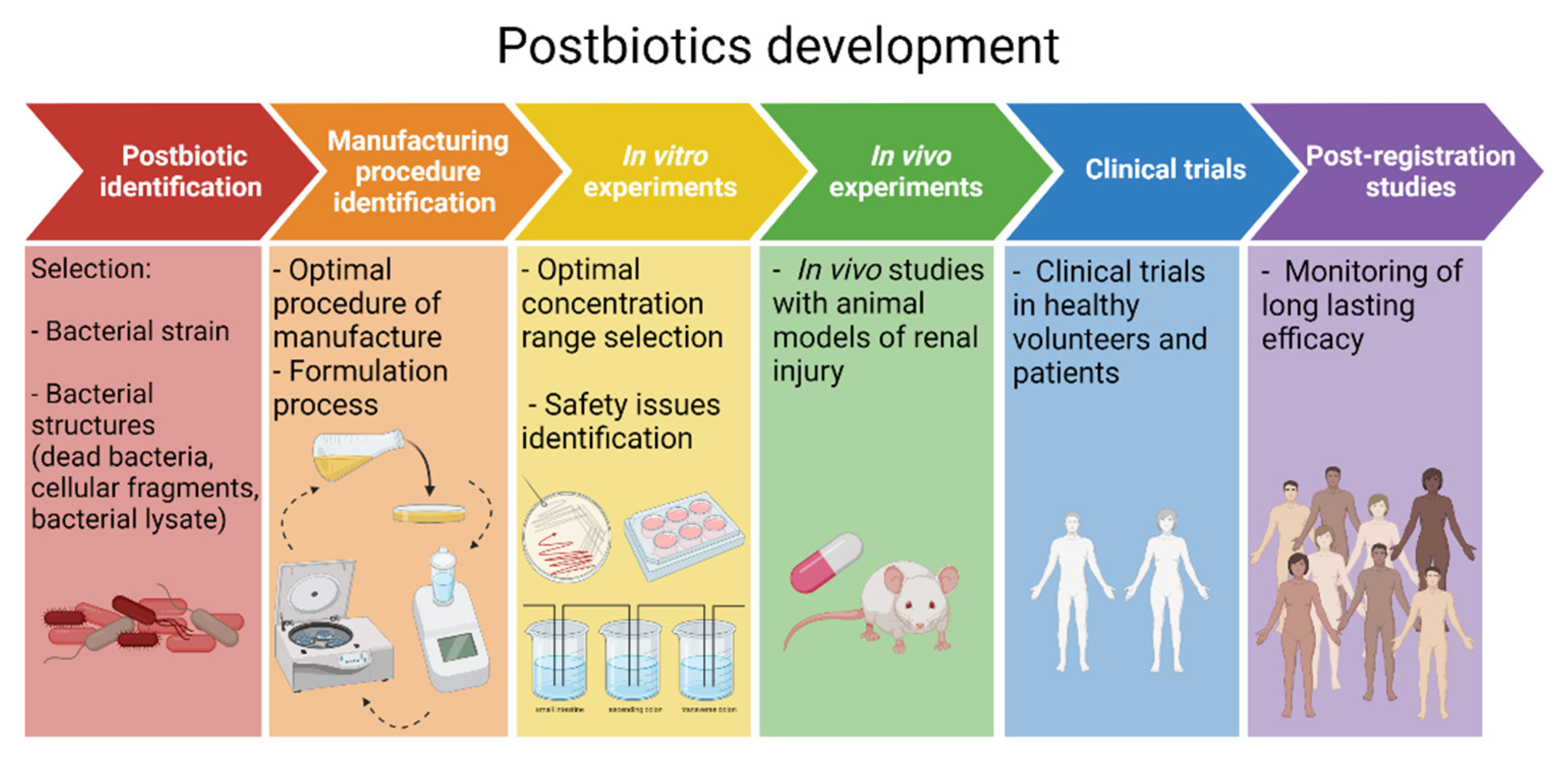
10. A Roadmap towards Postbiotic Therapy for Kidney Disease
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- Perez-Gomez, M.V.; Bartsch, L.-A.; Castillo-Rodriguez, E.; Fernandez-Prado, R.; Fernandez-Fernandez, B.; Martin-Cleary, C.; Gracia-Iguacel, C.; Ortiz, A. Clarifying the Concept of Chronic Kidney Disease for Non-Nephrologists. Clin. Kidney J. 2019, 12, 258–261. [Google Scholar] [CrossRef] [PubMed]
- Ortiz, A.; Roger, M.; Jiménez, V.M.; Perez, J.C.R.; Furlano, M.; Atxer, L.S.; Zurro, D.G.; Casabona, C.M.R.; Gómez, C.G.; Bermúdez, P.P.; et al. RICORS2040: The need for collaborative research in chronic kidney disease. Clin. Kidney J. 2021, 15, 372–387. [Google Scholar] [CrossRef] [PubMed]
- Douglas, A.E.; Werren, J.H. Holes in the Hologenome: Why Host-Microbe Symbioses Are Not Holobionts. mBio 2016, 7, e02099. [Google Scholar] [CrossRef] [PubMed]
- Maier, L.; Pruteanu, M.; Kuhn, M.; Zeller, G.; Telzerow, A.; Anderson, E.E.; Brochado, A.R.; Fernandez, K.C.; Dose, H.; Mori, H.; et al. Extensive Impact of Non-Antibiotic Drugs on Human Gut Bacteria. Nature 2018, 555, 623–628. [Google Scholar] [CrossRef] [PubMed]
- Salminen, S.; Collado, M.C.; Endo, A.; Hill, C.; Lebeer, S.; Quigley, E.M.M.; Sanders, M.E.; Shamir, R.; Swann, J.R.; Szajewska, H.; et al. The International Scientific Association of Probiotics and Prebiotics (ISAPP) Consensus Statement on the Definition and Scope of Postbiotics. Nat. Rev. Gastroenterol. Hepatol. 2021, 18, 649–667. [Google Scholar] [CrossRef]
- Zhang, P.; Zou, J.-Z.; Chen, J.; Tan, X.; Xiang, F.-F.; Shen, B.; Hu, J.-C.; Wang, J.-L.; Wang, Y.-Q.; Yu, J.-B.; et al. Association of Trimethylamine N-Oxide with Cardiovascular and All-Cause Mortality in Hemodialysis Patients. Ren. Fail. 2020, 42, 1004–1014. [Google Scholar] [CrossRef]
- Zhao, J.; Ning, X.; Liu, B.; Dong, R.; Bai, M.; Sun, S. Specific Alterations in Gut Microbiota in Patients with Chronic Kidney Disease: An Updated Systematic Review. Ren. Fail. 2021, 43, 102–112. [Google Scholar] [CrossRef]
- Zsom, L.; Zsom, M.; Salim, S.A.; Fülöp, T. Estimated Glomerular Filtration Rate in Chronic Kidney Disease: A Critical Review of Estimate-Based Predictions of Individual Outcomes in Kidney Disease. Toxins 2022, 14, 127. [Google Scholar] [CrossRef]
- Cigarran Guldris, S.; González Parra, E.; Cases Amenós, A. Gut Microbiota in Chronic Kidney Disease. Nefrologia 2017, 37, 9–19. [Google Scholar] [CrossRef]
- Favero, C.; Carriazo, S.; Cuarental, L.; Fernandez-Prado, R.; Gomá-Garcés, E.; Perez-Gomez, M.V.; Ortiz, A.; Fernandez-Fernandez, B.; Sanchez-Niño, M.D. Phosphate, Microbiota and CKD. Nutrients 2021, 13, 1273. [Google Scholar] [CrossRef]
- Chi, M.; Ma, K.; Wang, J.; Ding, Z.; Li, Y.; Zhu, S.; Liang, X.; Zhang, Q.; Song, L.; Liu, C. The Immunomodulatory Effect of the Gut Microbiota in Kidney Disease. J. Immunol. Res. 2021, 2021, 5516035. [Google Scholar] [CrossRef]
- Castillo-Rodriguez, E.; Fernandez-Prado, R.; Esteras, R.; Perez-Gomez, M.V.; Gracia-Iguacel, C.; Fernandez-Fernandez, B.; Kanbay, M.; Tejedor, A.; Lazaro, A.; Ruiz-Ortega, M.; et al. Impact of Altered Intestinal Microbiota on Chronic Kidney Disease Progression. Toxins 2018, 10, 300. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Prado, R.; Esteras, R.; Perez-Gomez, M.V.; Gracia-Iguacel, C.; Gonzalez-Parra, E.; Sanz, A.B.; Ortiz, A.; Sanchez-Niño, M.D. Nutrients Turned into Toxins: Microbiota Modulation of Nutrient Properties in Chronic Kidney Disease. Nutrients 2017, 9, 489. [Google Scholar] [CrossRef] [PubMed]
- Poveda, J.; Sanchez-Niño, M.D.; Glorieux, G.; Sanz, A.B.; Egido, J.; Vanholder, R.; Ortiz, A. P-Cresyl Sulphate Has pro-Inflammatory and Cytotoxic Actions on Human Proximal Tubular Epithelial Cells. Nephrol. Dial. Transplant. 2014, 29, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Sun, C.-Y.; Chang, S.-C.; Wu, M.-S. Suppression of Klotho Expression by Protein-Bound Uremic Toxins Is Associated with Increased DNA Methyltransferase Expression and DNA Hypermethylation. Kidney Int. 2012, 81, 640–650. [Google Scholar] [CrossRef]
- Gut Microbiota in Chronic Kidney Disease—PubMed. Available online: https://pubmed.ncbi.nlm.nih.gov/27553986/ (accessed on 11 July 2022).
- Mills, S.; Stanton, C.; Lane, J.A.; Smith, G.J.; Ross, R.P. Precision Nutrition and the Microbiome, Part I: Current State of the Science. Nutrients 2019, 11, 923. [Google Scholar] [CrossRef]
- Kim, S.-K.; Guevarra, R.B.; Kim, Y.-T.; Kwon, J.; Kim, H.; Cho, J.H.; Kim, H.B.; Lee, J.-H. Role of Probiotics in Human Gut Microbiome-Associated Diseases. J. Microbiol. Biotechnol. 2019, 29, 1335–1340. [Google Scholar] [CrossRef]
- Markowiak, P.; Śliżewska, K. Effects of Probiotics, Prebiotics, and Synbiotics on Human Health. Nutrients 2017, 9, 1021. [Google Scholar] [CrossRef]
- Holscher, H.D. Dietary Fiber and Prebiotics and the Gastrointestinal Microbiota. Gut Microbes 2017, 8, 172–184. [Google Scholar] [CrossRef] [PubMed]
- Muralitharan, R.R.; Jama, H.A.; Xie, L.; Peh, A.; Snelson, M.; Marques, F.Z. Microbial Peer Pressure: The Role of the Gut Microbiota in Hypertension and Its Complications. Hypertension 2020, 76, 1674–1687. [Google Scholar] [CrossRef]
- Vrzáčková, N.; Ruml, T.; Zelenka, J. Postbiotics, Metabolic Signaling, and Cancer. Molecules 2021, 26, 1528. [Google Scholar] [CrossRef]
- Rhys-Jones, D.; Climie, R.E.; Gill, P.A.; Jama, H.A.; Head, G.A.; Gibson, P.R.; Kaye, D.M.; Muir, J.G.; Marques, F.Z. Microbial Interventions to Control and Reduce Blood Pressure in Australia (MICRoBIA): Rationale and Design of a Double-Blinded Randomised Cross-over Placebo Controlled Trial. Trials 2021, 22, 496. [Google Scholar] [CrossRef]
- Canducci, F.; Armuzzi, A.; Cremonini, F.; Cammarota, G.; Bartolozzi, F.; Pola, P.; Gasbarrini, G.; Gasbarrini, A. A Lyophilized and Inactivated Culture of Lactobacillus Acidophilus Increases Helicobacter Pylori Eradication Rates. Aliment. Pharmacol. Ther. 2000, 14, 1625–1629. [Google Scholar] [CrossRef] [PubMed]
- Tarrerias, A.L.; Costil, V.; Vicari, F.; Létard, J.C.; Adenis-Lamarre, P.; Aisène, A.; Batistelli, D.; Bonnaud, G.; Carpentier, S.; Dalbiès, P.; et al. The Effect of Inactivated Lactobacillus LB Fermented Culture Medium on Symptom Severity: Observational Investigation in 297 Patients with Diarrhea-Predominant Irritable Bowel Syndrome. Dig. Dis. 2011, 29, 588–591. [Google Scholar] [CrossRef] [PubMed]
- Andresen, V.; Gschossmann, J.; Layer, P. Heat-Inactivated Bifidobacterium Bifidum MIMBb75 (SYN-HI-001) in the Treatment of Irritable Bowel Syndrome: A Multicentre, Randomised, Double-Blind, Placebo-Controlled Clinical Trial. Lancet Gastroenterol. Hepatol. 2020, 5, 658–666. [Google Scholar] [CrossRef]
- Xiao, S.-D.; Zhang, D.Z.; Lu, H.; Jiang, S.H.; Liu, H.Y.; Wang, G.S.; Xu, G.M.; Zhang, Z.B.; Lin, G.J.; Wang, G.L. Multicenter, Randomized, Controlled Trial of Heat-Killed Lactobacillus Acidophilus LB in Patients with Chronic Diarrhea. Adv. Ther. 2003, 20, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.; Badger, S.A.; Regan, M.; Clements, B.W.; Diamond, T.; Parks, R.W.; Taylor, M.A. Modulation of Gut Barrier Function in Patients with Obstructive Jaundice Using Probiotic LP299v. Eur. J. Gastroenterol. Hepatol. 2013, 25, 1424–1430. [Google Scholar] [CrossRef] [PubMed]
- Takiishi, T.; Korf, H.; Van Belle, T.L.; Robert, S.; Grieco, F.A.; Caluwaerts, S.; Galleri, L.; Spagnuolo, I.; Steidler, L.; Van Huynegem, K.; et al. Reversal of Autoimmune Diabetes by Restoration of Antigen-Specific Tolerance Using Genetically Modified Lactococcus Lactis in Mice. J. Clin. Investig. 2012, 122, 1717–1725. [Google Scholar] [CrossRef] [PubMed]
- Nishida, K.; Sawada, D.; Kuwano, Y.; Tanaka, H.; Rokutan, K. Health Benefits of Lactobacillus Gasseri CP2305 Tablets in Young Adults Exposed to Chronic Stress: A Randomized, Double-Blind, Placebo-Controlled Study. Nutrients 2019, 11, 1859. [Google Scholar] [CrossRef]
- Hoffman, J.R.; Hoffman, M.W.; Zelicha, H.; Gepner, Y.; Willoughby, D.S.; Feinstein, U.; Ostfeld, I. The Effect of 2 Weeks of Inactivated Probiotic Bacillus Coagulans on Endocrine, Inflammatory, and Performance Responses During Self-Defense Training in Soldiers. J. Strength Cond. Res. 2019, 33, 2330–2337. [Google Scholar] [CrossRef]
- Montané, E.; Barriocanal, A.M.; Arellano, A.L.; Valderrama, A.; Sanz, Y.; Perez-Alvarez, N.; Cardona, P.; Vilaplana, C.; Cardona, P.-J. Pilot, Double-Blind, Randomized, Placebo-Controlled Clinical Trial of the Supplement Food Nyaditum Resae® in Adults with or without Latent TB Infection: Safety and Immunogenicity. PLoS ONE 2017, 12, e0171294. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Guo, S.; Li, C.; Jiang, X. Therapeutic Effects of Inhaled Inactivated Mycobacterium Phlei in Adult Patients with Moderate Persistent Asthma. Immunotherapy 2012, 4, 383–387. [Google Scholar] [CrossRef]
- Tandon, M.K.; Phillips, M.; Waterer, G.; Dunkley, M.; Comans, P.; Clancy, R. Oral Immunotherapy with Inactivated Nontypeable Haemophilus Influenzae Reduces Severity of Acute Exacerbations in Severe COPD. Chest 2010, 137, 805–811. [Google Scholar] [CrossRef] [PubMed]
- Zagólski, O.; Stręk, P.; Kasprowicz, A.; Białecka, A. Effectiveness of Polyvalent Bacterial Lysate and Autovaccines Against Upper Respiratory Tract Bacterial Colonization by Potential Pathogens: A Randomized Study. Med. Sci. Monit. 2015, 21, 2997–3002. [Google Scholar] [CrossRef] [PubMed]
- Braido, F.; Melioli, G.; Cazzola, M.; Fabbri, L.; Blasi, F.; Moretta, L.; Canonica, G.W.; AIACE Study Group. Sub-Lingual Administration of a Polyvalent Mechanical Bacterial Lysate (PMBL) in Patients with Moderate, Severe, or Very Severe Chronic Obstructive Pulmonary Disease (COPD) According to the GOLD Spirometric Classification: A Multicentre, Double-Blind, Randomised, Controlled, Phase IV Study (AIACE Study: Advanced Immunological Approach in COPD Exacerbation). Pulm. Pharmacol. Ther. 2015, 33, 75–80. [Google Scholar] [CrossRef] [PubMed]
- Braido, F.; Melioli, G.; Candoli, P.; Cavalot, A.; Di Gioacchino, M.; Ferrero, V.; Incorvaia, C.; Mereu, C.; Ridolo, E.; Rolla, G.; et al. The Bacterial Lysate Lantigen B Reduces the Number of Acute Episodes in Patients with Recurrent Infections of the Respiratory Tract: The Results of a Double Blind, Placebo Controlled, Multicenter Clinical Trial. Immunol. Lett. 2014, 162, 185–193. [Google Scholar] [CrossRef]
- Krusteva, E.; Hristova, S.; Damyanov, D.; Bogdanov, A.; Altaparmakov, I.; Pacelli, E. Clinical Study of the Effect of the Preparation DEODAN on Leukopenia, Induced by Cytostatics. Int. J. Immunopharmacol. 1997, 19, 487–492. [Google Scholar] [CrossRef]
- Indrio, F.; Ladisa, G.; Mautone, A.; Montagna, O. Effect of a Fermented Formula on Thymus Size and Stool PH in Healthy Term Infants. Pediatr. Res. 2007, 62, 98–100. [Google Scholar] [CrossRef]
- Morisset, M.; Aubert-Jacquin, C.; Soulaines, P.; Moneret-Vautrin, D.-A.; Dupont, C. A Non-Hydrolyzed, Fermented Milk Formula Reduces Digestive and Respiratory Events in Infants at High Risk of Allergy. Eur. J. Clin. Nutr. 2011, 65, 175–183. [Google Scholar] [CrossRef]
- Mullié, C.; Yazourh, A.; Thibault, H.; Odou, M.-F.; Singer, E.; Kalach, N.; Kremp, O.; Romond, M.-B. Increased Poliovirus-Specific Intestinal Antibody Response Coincides with Promotion of Bifidobacterium Longum-Infantis and Bifidobacterium Breve in Infants: A Randomized, Double-Blind, Placebo-Controlled Trial. Pediatr. Res. 2004, 56, 791–795. [Google Scholar] [CrossRef] [Green Version]
- Thibault, H.; Aubert-Jacquin, C.; Goulet, O. Effects of Long-Term Consumption of a Fermented Infant Formula (with Bifidobacterium Breve C50 and Streptococcus Thermophilus 065) on Acute Diarrhea in Healthy Infants. J. Pediatr. Gastroenterol. Nutr. 2004, 39, 147–152. [Google Scholar] [CrossRef] [PubMed]
- Roy, P.; Aubert-Jacquin, C.; Avart, C.; Gontier, C. Benefits of a thickened infant formula with lactase activity in the management of benign digestive disorders in newborns. Arch. Pediatr. 2004, 11, 1546–1554. [Google Scholar] [CrossRef] [PubMed]
- Campeotto, F.; Suau, A.; Kapel, N.; Magne, F.; Viallon, V.; Ferraris, L.; Waligora-Dupriet, A.-J.; Soulaines, P.; Leroux, B.; Kalach, N.; et al. A Fermented Formula in Pre-Term Infants: Clinical Tolerance, Gut Microbiota, down-Regulation of Faecal Calprotectin and up-Regulation of Faecal Secretory IgA. Br. J. Nutr. 2011, 105, 1843–1851. [Google Scholar] [CrossRef] [PubMed]
- Liévin-Le Moal, V.; Sarrazin-Davila, L.E.; Servin, A.L. An Experimental Study and a Randomized, Double-Blind, Placebo-Controlled Clinical Trial to Evaluate the Antisecretory Activity of Lactobacillus Acidophilus Strain LB against Nonrotavirus Diarrhea. Pediatrics 2007, 120, e795–e803. [Google Scholar] [CrossRef]
- Salazar-Lindo, E.; Figueroa-Quintanilla, D.; Caciano, M.I.; Reto-Valiente, V.; Chauviere, G.; Colin, P.; Lacteol Study Group. Effectiveness and Safety of Lactobacillus LB in the Treatment of Mild Acute Diarrhea in Children. J. Pediatr. Gastroenterol. Nutr. 2007, 44, 571–576. [Google Scholar] [CrossRef]
- Simakachorn, N.; Pichaipat, V.; Rithipornpaisarn, P.; Kongkaew, C.; Tongpradit, P.; Varavithya, W. Clinical Evaluation of the Addition of Lyophilized, Heat-Killed Lactobacillus Acidophilus LB to Oral Rehydration Therapy in the Treatment of Acute Diarrhea in Children. J. Pediatr. Gastroenterol. Nutr. 2000, 30, 68–72. [Google Scholar] [CrossRef]
- Kaila, M.; Isolauri, E.; Saxelin, M.; Arvilommi, H.; Vesikari, T. Viable versus Inactivated Lactobacillus Strain GG in Acute Rotavirus Diarrhoea. Arch. Dis. Child. 1995, 72, 51–53. [Google Scholar] [CrossRef]
- Nocerino, R.; Paparo, L.; Terrin, G.; Pezzella, V.; Amoroso, A.; Cosenza, L.; Cecere, G.; De Marco, G.; Micillo, M.; Albano, F.; et al. Cow’s Milk and Rice Fermented with Lactobacillus Paracasei CBA L74 Prevent Infectious Diseases in Children: A Randomized Controlled Trial. Clin. Nutr. 2017, 36, 118–125. [Google Scholar] [CrossRef]
- Corsello, G.; Carta, M.; Marinello, R.; Picca, M.; De Marco, G.; Micillo, M.; Ferrara, D.; Vigneri, P.; Cecere, G.; Ferri, P.; et al. Preventive Effect of Cow’s Milk Fermented with Lactobacillus Paracasei CBA L74 on Common Infectious Diseases in Children: A Multicenter Randomized Controlled Trial. Nutrients 2017, 9, 669. [Google Scholar] [CrossRef]
- Sharieff, W.; Bhutta, Z.; Schauer, C.; Tomlinson, G.; Zlotkin, S. Micronutrients (Including Zinc) Reduce Diarrhoea in Children: The Pakistan Sprinkles Diarrhoea Study. Arch. Dis. Child. 2006, 91, 573–579. [Google Scholar] [CrossRef] [Green Version]
- Kirjavainen, P.V.; Salminen, S.J.; Isolauri, E. Probiotic Bacteria in the Management of Atopic Disease: Underscoring the Importance of Viability. J. Pediatr. Gastroenterol. Nutr. 2003, 36, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Peng, G.-C.; Hsu, C.-H. The Efficacy and Safety of Heat-Killed Lactobacillus Paracasei for Treatment of Perennial Allergic Rhinitis Induced by House-Dust Mite. Pediatr. Allergy Immunol. 2005, 16, 433–438. [Google Scholar] [CrossRef] [PubMed]
- Rampengan, N.H.; Manoppo, J.; Warouw, S.M. Comparison of Efficacies between Live and Killed Probiotics in Children with Lactose Malabsorption. Southeast Asian J. Trop. Med. Public Health 2010, 41, 474–481. [Google Scholar] [PubMed]
- Szajewska, H.; Skórka, A.; Pieścik-Lech, M. Fermented Infant Formulas without Live Bacteria: A Systematic Review. Eur. J. Pediatr. 2015, 174, 1413–1420. [Google Scholar] [CrossRef]
- Keshari, S.; Wang, Y.; Herr, D.R.; Wang, S.-M.; Yang, W.-C.; Chuang, T.-H.; Chen, C.-L.; Huang, C.-M. Skin Cutibacterium Acnes Mediates Fermentation to Suppress the Calcium Phosphate-Induced Itching: A Butyric Acid Derivative with Potential for Uremic Pruritus. J. Clin. Med. 2020, 9, 312. [Google Scholar] [CrossRef]
- Lin, W.-Y.; Lin, J.-H.; Kuo, Y.-W.; Chiang, P.-F.R.; Ho, H.-H. Probiotics and Their Metabolites Reduce Oxidative Stress in Middle-Aged Mice. Curr. Microbiol. 2022, 79, 104. [Google Scholar] [CrossRef]
- Díaz Cano, J.V.; Argente, M.-J.; García, M.-L. Effect of Postbiotic Based on Lactic Acid Bacteria on Semen Quality and Health of Male Rabbits. Animals 2021, 11, 1007. [Google Scholar] [CrossRef]
- Notification to the CHMP/EMA Secretariat of a Referral under Article 31 of Directive 2001/83/EC. Available online: https://www.ema.europa.eu/en/documents/referral/bacterial-lysate-medicines-article-31-referral-notification_en.pdf (accessed on 5 June 2022).
- EMA/351772/2019 Bacterial Lysate Medicines for Respiratory Conditions to Be Used Only for Prevention of Recurrent Infections. Available online: https://www.ema.europa.eu/en/documents/press-release/bacterial-lysate-medicines-respiratory-conditions-be-used-only-prevention-recurrent-infections_en.pdf (accessed on 5 June 2022).
- Pivniouk, V.; Pivniouk, O.; DeVries, A.; Uhrlaub, J.L.; Michael, A.; Pivniouk, D.; VanLinden, S.R.; Conway, M.Y.; Hahn, S.; Malone, S.P.; et al. The OM-85 Bacterial Lysate Inhibits SARS-CoV-2 Infection of Epithelial Cells by Downregulating SARS-CoV-2 Receptor Expression. J. Allergy Clin. Immunol. 2022, 149, 923–933.e6. [Google Scholar] [CrossRef]
- Bacchetta, J.; Wood, K.D. Primary Hyperoxaluria Type 1: Time for Prime Time? Clin. Kidney J. 2022, 15, i1–i3. [Google Scholar] [CrossRef]
- Robijn, S.; Hoppe, B.; Vervaet, B.A.; D’Haese, P.C.; Verhulst, A. Hyperoxaluria: A Gut-Kidney Axis? Kidney Int. 2011, 80, 1146–1158. [Google Scholar] [CrossRef] [Green Version]
- Witting, C.; Langman, C.B.; Assimos, D.; Baum, M.A.; Kausz, A.; Milliner, D.; Tasian, G.; Worcester, E.; Allain, M.; West, M.; et al. Pathophysiology and Treatment of Enteric Hyperoxaluria. Clin. J. Am. Soc. Nephrol. 2021, 16, 487–495. [Google Scholar] [CrossRef]
- Demoulin, N.; Aydin, S.; Gillion, V.; Morelle, J.; Jadoul, M. Pathophysiology and Management of Hyperoxaluria and Oxalate Nephropathy: A Review. Am. J. Kidney Dis. 2022, 79, 717–727. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.; Somers, M.J.G.; Baum, M.A. Treatment of Primary Hyperoxaluria Type 1. Clin. Kidney J. 2022, 15, i9–i13. [Google Scholar] [CrossRef]
- Dejban, P.; Lieske, J.C. New Therapeutics for Primary Hyperoxaluria Type 1. Curr. Opin. Nephrol. Hypertens. 2022, 31, 344–350. [Google Scholar] [CrossRef] [PubMed]
- Arvans, D.; Jung, Y.-C.; Antonopoulos, D.; Koval, J.; Granja, I.; Bashir, M.; Karrar, E.; Roy-Chowdhury, J.; Musch, M.; Asplin, J.; et al. Oxalobacter Formigenes-Derived Bioactive Factors Stimulate Oxalate Transport by Intestinal Epithelial Cells. J. Am. Soc. Nephrol. 2017, 28, 876–887. [Google Scholar] [CrossRef] [PubMed]
- Daniel, S.L.; Moradi, L.; Paiste, H.; Wood, K.D.; Assimos, D.G.; Holmes, R.P.; Nazzal, L.; Hatch, M.; Knight, J. Forty Years of Oxalobacter Formigenes, a Gutsy Oxalate-Degrading Specialist. Appl. Environ. Microbiol. 2021, 87, e0054421. [Google Scholar] [CrossRef] [PubMed]
- Sidhu, H.; Schmidt, M.E.; Cornelius, J.G.; Thamilselvan, S.; Khan, S.R.; Hesse, A.; Peck, A.B. Direct Correlation between Hyperoxaluria/Oxalate Stone Disease and the Absence of the Gastrointestinal Tract-Dwelling Bacterium Oxalobacter Formigenes: Possible Prevention by Gut Recolonization or Enzyme Replacement Therapy. J. Am. Soc. Nephrol. 1999, 10, S334–S340. [Google Scholar] [PubMed]
- Kleinschmidt, K.; Mahlmann, A.; Hautmann, R. Microbial Degradation of Dietary Oxalate in the Human Gut and Urinary Oxalate Concentrations in Patients with Calcium Oxalate Urolithiasis and Control Persons. Investig. Urol. 1994, 5, 222–224. [Google Scholar]
- Liu, M.; Devlin, J.C.; Hu, J.; Volkova, A.; Battaglia, T.W.; Ho, M.; Asplin, J.R.; Byrd, A.; Loke, P.; Li, H.; et al. Microbial Genetic and Transcriptional Contributions to Oxalate Degradation by the Gut Microbiota in Health and Disease. Elife 2021, 10, e63642. [Google Scholar] [CrossRef]
- Hatch, M.; Cornelius, J.; Allison, M.; Sidhu, H.; Peck, A.; Freel, R.W. Oxalobacter Sp. Reduces Urinary Oxalate Excretion by Promoting Enteric Oxalate Secretion. Kidney Int. 2006, 69, 691–698. [Google Scholar] [CrossRef]
- Verhulst, A.; Dehmel, B.; Lindner, E.; Akerman, M.E.; D’Haese, P.C. Oxalobacter Formigenes Treatment Confers Protective Effects in a Rat Model of Primary Hyperoxaluria by Preventing Renal Calcium Oxalate Deposition. Urolithiasis 2022, 50, 119–130. [Google Scholar] [CrossRef] [PubMed]
- Nazzal, L.; Francois, F.; Henderson, N.; Liu, M.; Li, H.; Koh, H.; Wang, C.; Gao, Z.; Perez, G.P.; Asplin, J.R.; et al. Effect of Antibiotic Treatment on Oxalobacter Formigenes Colonization of the Gut Microbiome and Urinary Oxalate Excretion. Sci. Rep. 2021, 11, 16428. [Google Scholar] [CrossRef]
- Hiremath, S.; Viswanathan, P. Oxalobacter Formigenes: A New Hope as a Live Biotherapeutic Agent in the Management of Calcium Oxalate Renal Stones. Anaerobe 2022, 75, 102572. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.W.; Choy, D.; Penniston, K.L.; Lange, D. Inhibition of Urinary Stone Disease by a Multi-Species Bacterial Network Ensures Healthy Oxalate Homeostasis. Kidney Int. 2019, 96, 180–188. [Google Scholar] [CrossRef]
- Ticinesi, A.; Milani, C.; Guerra, A.; Allegri, F.; Lauretani, F.; Nouvenne, A.; Mancabelli, L.; Lugli, G.A.; Turroni, F.; Duranti, S.; et al. Understanding the Gut-Kidney Axis in Nephrolithiasis: An Analysis of the Gut Microbiota Composition and Functionality of Stone Formers. Gut 2018, 67, 2097–2106. [Google Scholar] [CrossRef] [PubMed]
- Whittamore, J.M.; Hatch, M. The Role of Intestinal Oxalate Transport in Hyperoxaluria and the Formation of Kidney Stones in Animals and Man. Urolithiasis 2017, 45, 89–108. [Google Scholar] [CrossRef] [PubMed]
- Hatch, M. Induction of Enteric Oxalate Secretion by Oxalobacter Formigenes in Mice Does Not Require the Presence of Either Apical Oxalate Transport Proteins Slc26A3 or Slc26A6. Urolithiasis 2020, 48, 1–8. [Google Scholar] [CrossRef]
- Hoppe, B.; Pellikka, P.A.; Dehmel, B.; Banos, A.; Lindner, E.; Herberg, U. Effects of Oxalobacter Formigenes in Subjects with Primary Hyperoxaluria Type 1 and End-Stage Renal Disease: A Phase II Study. Nephrol. Dial. Transplant. 2021, 36, 1464–1473. [Google Scholar] [CrossRef]
- Pape, L.; Ahlenstiel-Grunow, T.; Birtel, J.; Krohne, T.U.; Hoppe, B. Oxalobacter Formigenes Treatment Combined with Intensive Dialysis Lowers Plasma Oxalate and Halts Disease Progression in a Patient with Severe Infantile Oxalosis. Pediatr. Nephrol. 2020, 35, 1121–1124. [Google Scholar] [CrossRef]
- Ariceta, G.; Collard, L.; Abroug, S.; Moochhala, S.H.; Gould, E.; Boussetta, A.; Ben Hmida, M.; De, S.; Hunley, T.E.; Jarraya, F.; et al. EPHex: A Phase 3, Double-Blind, Placebo-Controlled, Randomized Study to Evaluate Long-Term Efficacy and Safety of Oxalobacter Formigenes in Patients with Primary Hyperoxaluria. Pediatr. Nephrol. 2022. [Google Scholar] [CrossRef]
- Milliner, D.; Hoppe, B.; Groothoff, J. A Randomised Phase II/III Study to Evaluate the Efficacy and Safety of Orally Administered Oxalobacter Formigenes to Treat Primary Hyperoxaluria. Urolithiasis 2018, 46, 313–323. [Google Scholar] [CrossRef] [Green Version]
- Hoppe, B.; Groothoff, J.W.; Hulton, S.-A.; Cochat, P.; Niaudet, P.; Kemper, M.J.; Deschênes, G.; Unwin, R.; Milliner, D. Efficacy and Safety of Oxalobacter Formigenes to Reduce Urinary Oxalate in Primary Hyperoxaluria. Nephrol. Dial. Transplant. 2011, 26, 3609–3615. [Google Scholar] [CrossRef]
- Osman, A.; El-Gazzar, N.; Almanaa, T.N.; El-Hadary, A.; Sitohy, M. Lipolytic Postbiotic from Lactobacillus Paracasei Manages Metabolic Syndrome in Albino Wistar Rats. Molecules 2021, 26, 472. [Google Scholar] [CrossRef]
- Son, M.; Oh, S.; Lee, H.S.; Choi, J.; Lee, B.-J.; Park, J.-H.; Park, C.H.; Son, K.H.; Byun, K. Gamma-Aminobutyric Acid-Salt Attenuated High Cholesterol/High Salt Diet Induced Hypertension in Mice. Korean J. Physiol. Pharmacol. 2021, 25, 27–38. [Google Scholar] [CrossRef]
- Hayakawa, K.; Kimura, M.; Kasaha, K.; Matsumoto, K.; Sansawa, H.; Yamori, Y. Effect of a Gamma-Aminobutyric Acid-Enriched Dairy Product on the Blood Pressure of Spontaneously Hypertensive and Normotensive Wistar-Kyoto Rats. Br. J. Nutr. 2004, 92, 411–417. [Google Scholar] [CrossRef]
- Inoue, K.; Shirai, T.; Ochiai, H.; Kasao, M.; Hayakawa, K.; Kimura, M.; Sansawa, H. Blood-Pressure-Lowering Effect of a Novel Fermented Milk Containing Gamma-Aminobutyric Acid (GABA) in Mild Hypertensives. Eur. J. Clin. Nutr. 2003, 57, 490–495. [Google Scholar] [CrossRef]
- Lee, H.; Ji, S.Y.; Hwangbo, H.; Kim, M.Y.; Kim, D.H.; Park, B.S.; Park, J.-H.; Lee, B.-J.; Kim, G.-Y.; Jeon, Y.-J.; et al. Protective Effect of Gamma Aminobutyric Acid against Aggravation of Renal Injury Caused by High Salt Intake in Cisplatin-Induced Nephrotoxicity. Int. J. Mol. Sci. 2022, 23, 502. [Google Scholar] [CrossRef]
- Shankaranarayanan, D.; Raj, D.S. Gut Microbiome and Kidney Disease: Reconciling Optimism and Skepticism. Clin. J. Am. Soc. Nephrol. 2022. online ahead of print. [Google Scholar] [CrossRef]
- Aguilera-Correa, J.-J.; Madrazo-Clemente, P.; Martínez-Cuesta, M.D.C.; Peláez, C.; Ortiz, A.; Sánchez-Niño, M.D.; Esteban, J.; Requena, T. Lyso-Gb3 Modulates the Gut Microbiota and Decreases Butyrate Production. Sci. Rep. 2019, 9, 12010. [Google Scholar] [CrossRef]
- Chamberlain, C.A.; Hatch, M.; Garrett, T.J. Extracellular Vesicle Analysis by Paper Spray Ionization Mass Spectrometry. Metabolites 2021, 11, 308. [Google Scholar] [CrossRef]
- Chamberlain, C.A.; Hatch, M.; Garrett, T.J. Oxalobacter Formigenes Produces Metabolites and Lipids Undetectable in Oxalotrophic Bifidobacterium Animalis. Metabolomics 2020, 16, 122. [Google Scholar] [CrossRef] [PubMed]
- Joshi, S.; Goldfarb, D.S. The Use of Antibiotics and Risk of Kidney Stones. Curr. Opin. Nephrol. Hypertens. 2019, 28, 311–315. [Google Scholar] [CrossRef] [PubMed]

| Intervention Group | Postbiotic | Reference |
|---|---|---|
| Helicobacter pylori | Inactivated culture of Lactobacillus | [24] |
| Irritable bowel syndrome and diarrhea | Lacteol, inactivated L. acidophilus LB plus fermented culture medium | [25] |
| Irritable bowel syndrome | Non-viable, heat-inactivated Bifidobacterium bifidum MIMBb75 | [26] |
| Chronic diarrhea | Heat-killed L. acidophilus LB (Lacteol Fort) | [27] |
| Obstructive jaundice | Inactivated Lactiplantibacillus plantarum | [28] |
| Stress response in undergraduate medical students | Heat-inactivated L. gasseri strain CP2305 | [29] |
| Chronic responses in medical students | Heat-inactivated L. gasseri strain CP2305 | [30] |
| Response (endocrine, inflammation, performance) during self-defense training in soldiers | Inactivated Bacillus coagulans | [31] |
| Latent tuberculosis | Heat-killed Mycobacterium manresensis | [32] |
| Asthma | Inactivated Mycobacterium phlei | [33] |
| Chronic obstructive pulmonary disease | Inactivated, non-typable H. influenzae | [34] |
| Bacterial colonization of nose and throat | Lysate containing S. aureus, Streptococcus mitis, S. pyogenes, S. pneumoniae, K. pneumoniae, M. catarrhalis, H. influenzae | [35] |
| Chronic obstructive pulmonary disease | Lyophilized bacterial fragments derived from S. aureus, Streptococcus viridans, S. Pneumoniae, Klebsiella ozaenae, M. catarrhalis, H. influenzae | [36] |
| Recurrent respiratory tract infectious | Lantigen B, suspension of bacterial antigens obtained from S. pneumoniae type 3, S. pyrogenes group A, B. catarrhalis, S. aureus, H. influenzae type B and K. pneumoniae | [37] |
| Cancer and leukopenia following chemotherapy | DEODAN, lysozyme lysates of Lactobacillus bulgaricus | [38] |
| Intervention Group | Postbiotic | References |
|---|---|---|
| Fermented formula (healthy infants) | Fermented formula with BB C50 and ST065 | [39,40,41,42,43] |
| Fermented formula in preterm infants | Heat-inactivated fermented formula with BB C50 and ST 065 | [44] |
| Acute gastroenteritis | Heat-killed Lactobacillus LB | [45,46,47,48] |
| Prevention of common infectious diseases | Heat-inactivated L. paracasei CBA L74 or L. acidophilus | [49,50,51] |
| Atopic eczema and cow’s milk allergy | Live or heat-inactivated L. rhamnosus | [52] |
| Allergic rhinitis | Live or heat-killed L. paracasei 33 | [53] |
| Lactose malabsorption | Killed and live Lactobacillus helveticus R-52 and L. rhamnosus R-11 | [54] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Favero, C.; Giordano, L.; Mihaila, S.M.; Masereeuw, R.; Ortiz, A.; Sanchez-Niño, M.D. Postbiotics and Kidney Disease. Toxins 2022, 14, 623. https://doi.org/10.3390/toxins14090623
Favero C, Giordano L, Mihaila SM, Masereeuw R, Ortiz A, Sanchez-Niño MD. Postbiotics and Kidney Disease. Toxins. 2022; 14(9):623. https://doi.org/10.3390/toxins14090623
Chicago/Turabian StyleFavero, Chiara, Laura Giordano, Silvia Maria Mihaila, Rosalinde Masereeuw, Alberto Ortiz, and Maria Dolores Sanchez-Niño. 2022. "Postbiotics and Kidney Disease" Toxins 14, no. 9: 623. https://doi.org/10.3390/toxins14090623
APA StyleFavero, C., Giordano, L., Mihaila, S. M., Masereeuw, R., Ortiz, A., & Sanchez-Niño, M. D. (2022). Postbiotics and Kidney Disease. Toxins, 14(9), 623. https://doi.org/10.3390/toxins14090623






