A Sea Anemone Lebrunia neglecta Venom Fraction Decreases Boar Sperm Cells Capacitation: Possible Involvement of HVA Calcium Channels
Abstract
:1. Introduction
2. Results and Discussion
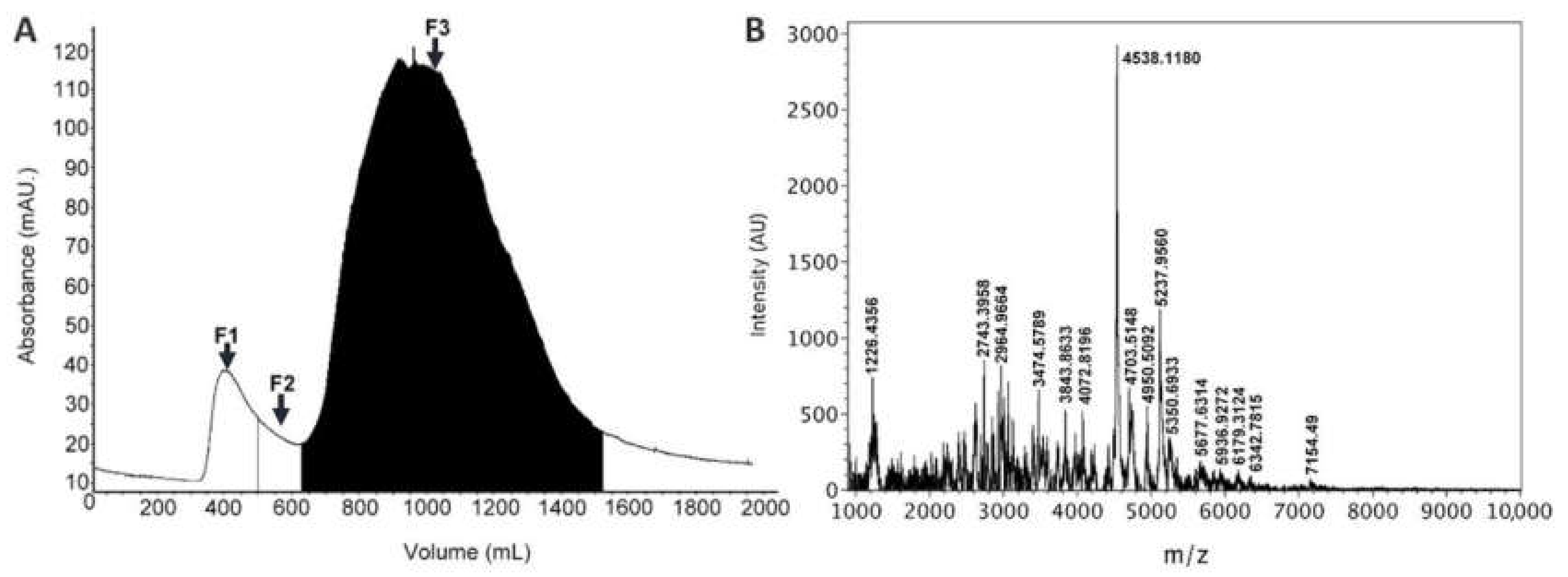
2.1. Venom Extraction and Fractionation
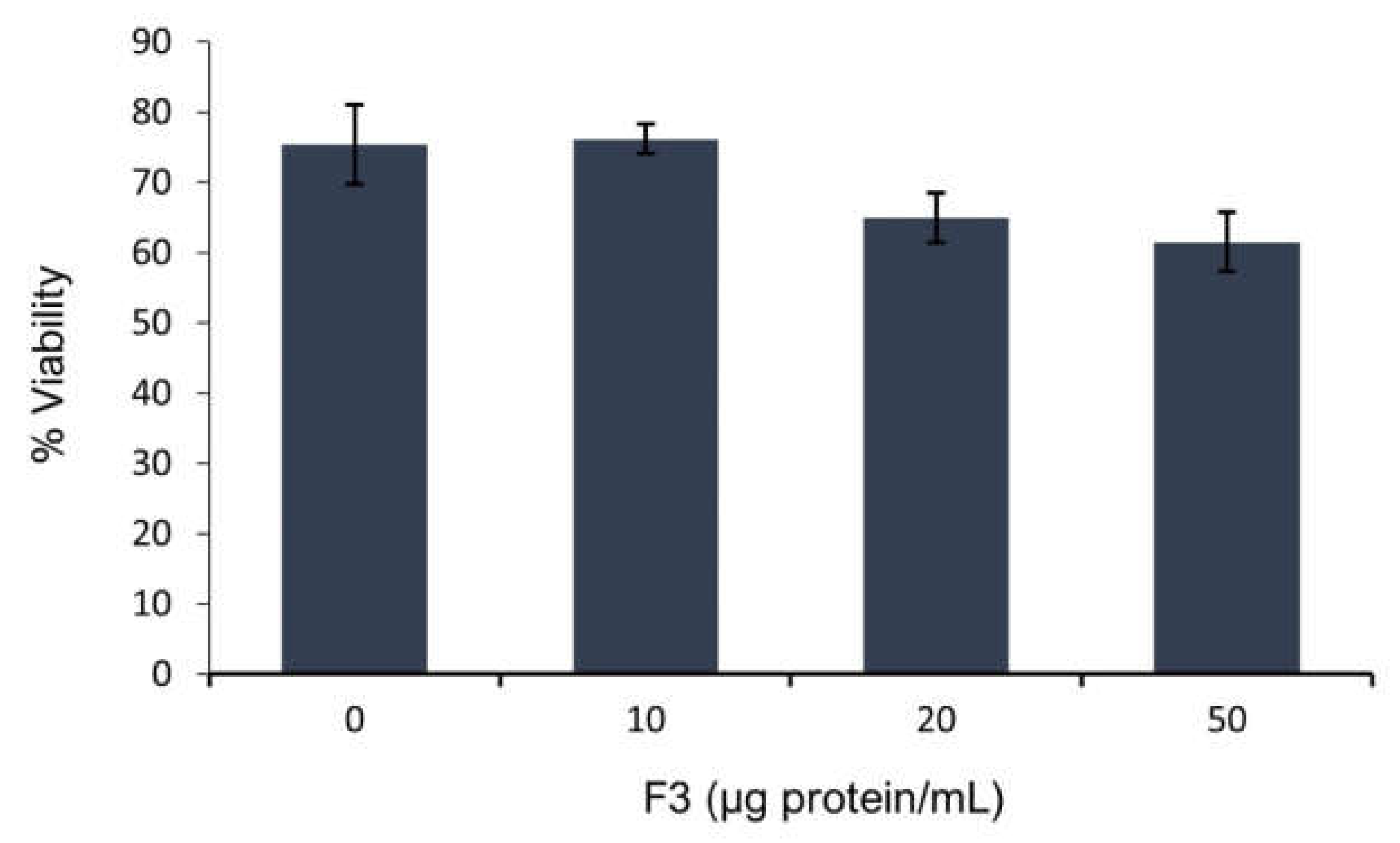
2.2. Sperm Viability
2.3. Sperm Motility
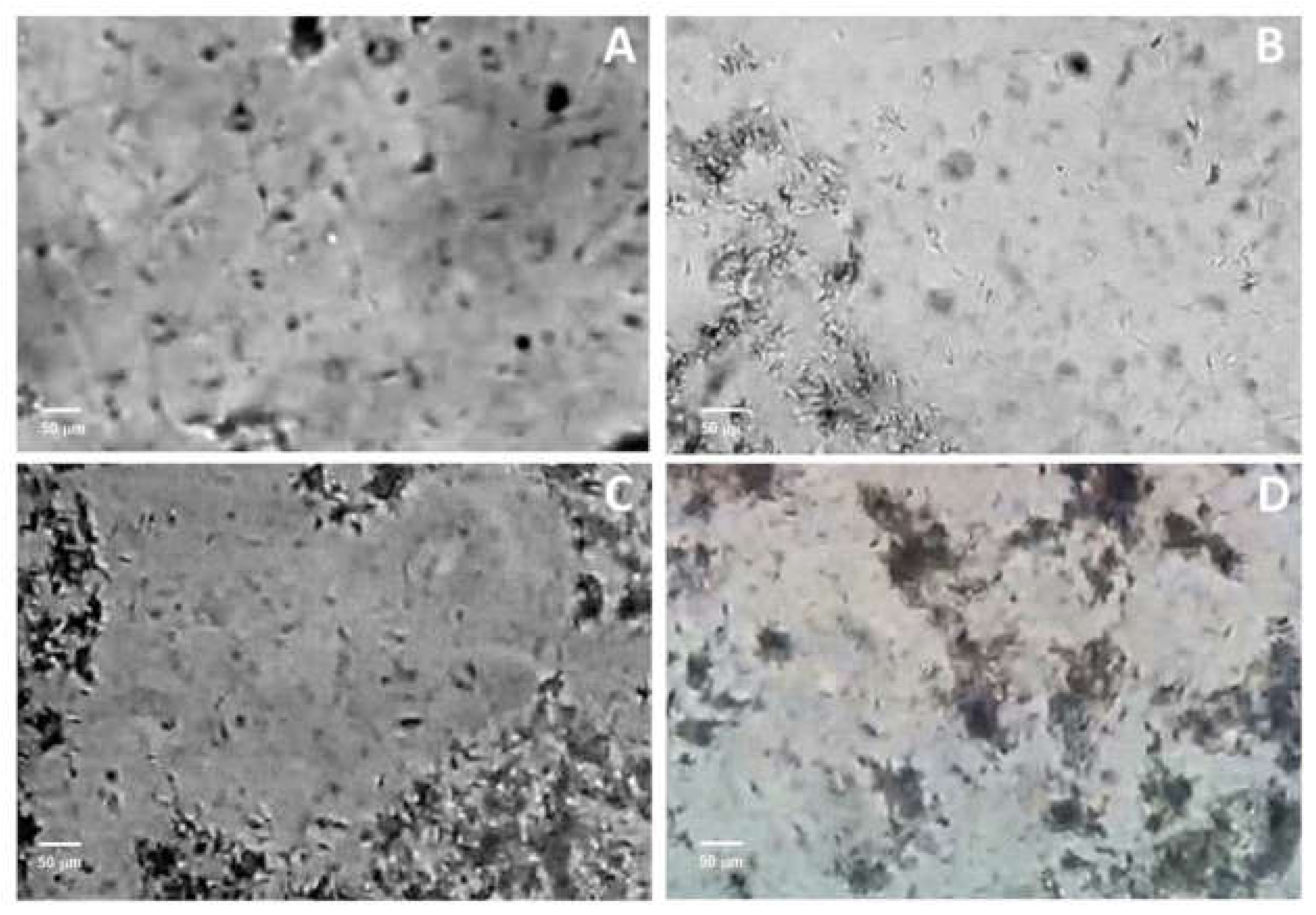
2.4. Capacitation
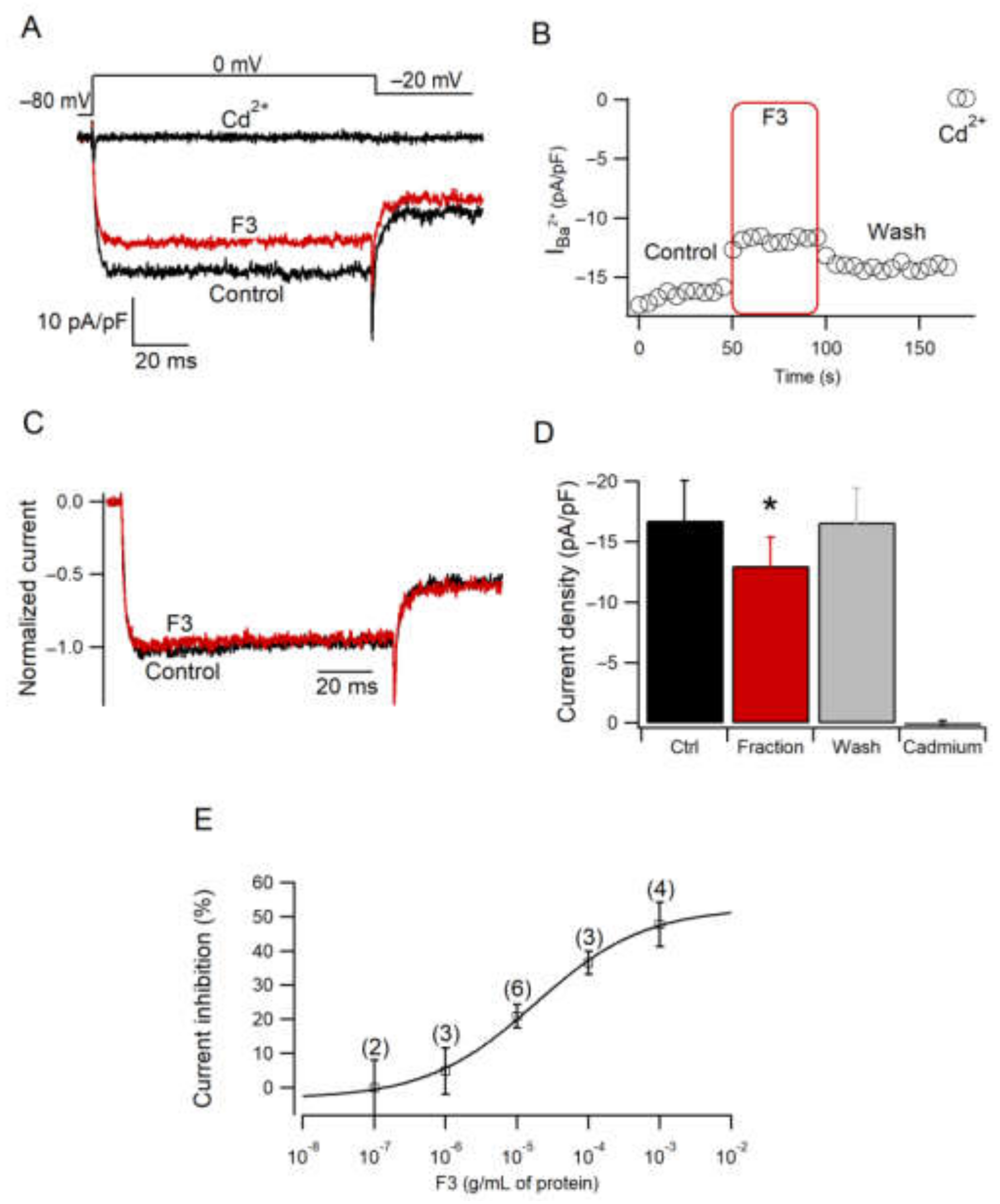
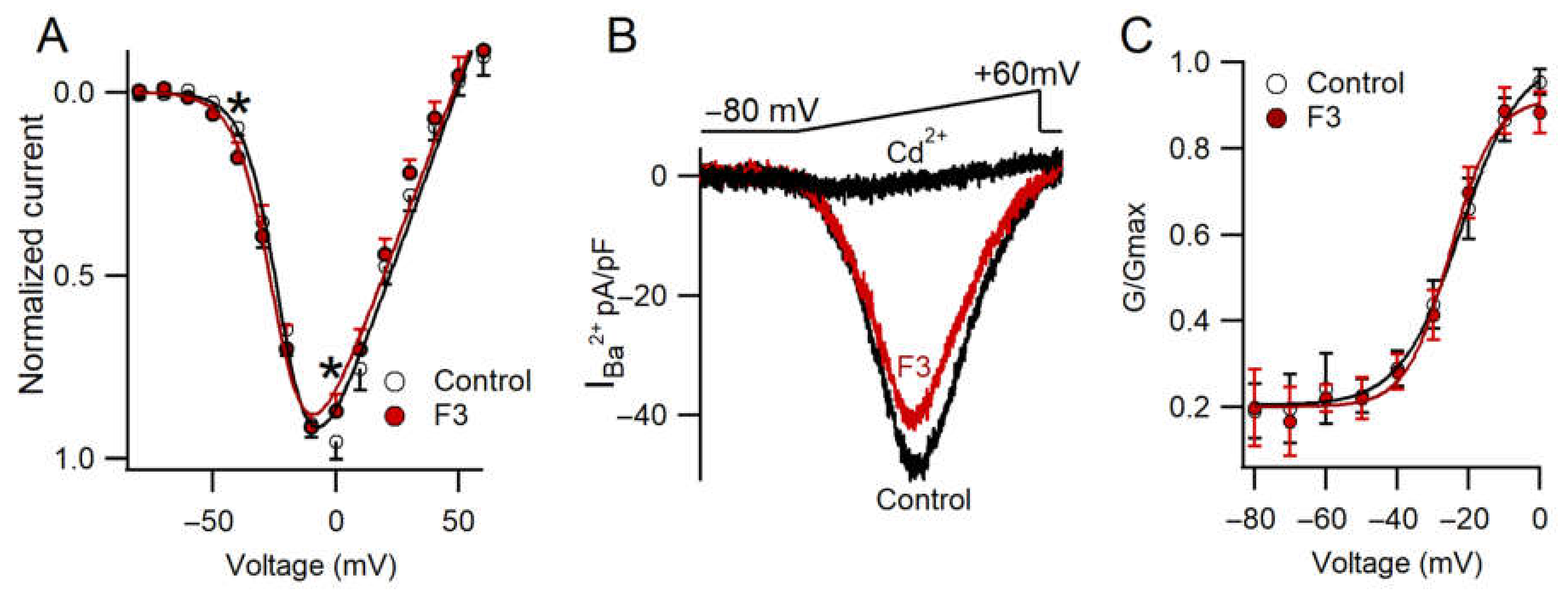
2.5. Effect of F3 on Calcium Current Amplitude
2.6. F3 Inhibits Calcium Currents without Changes in Voltage Dependence
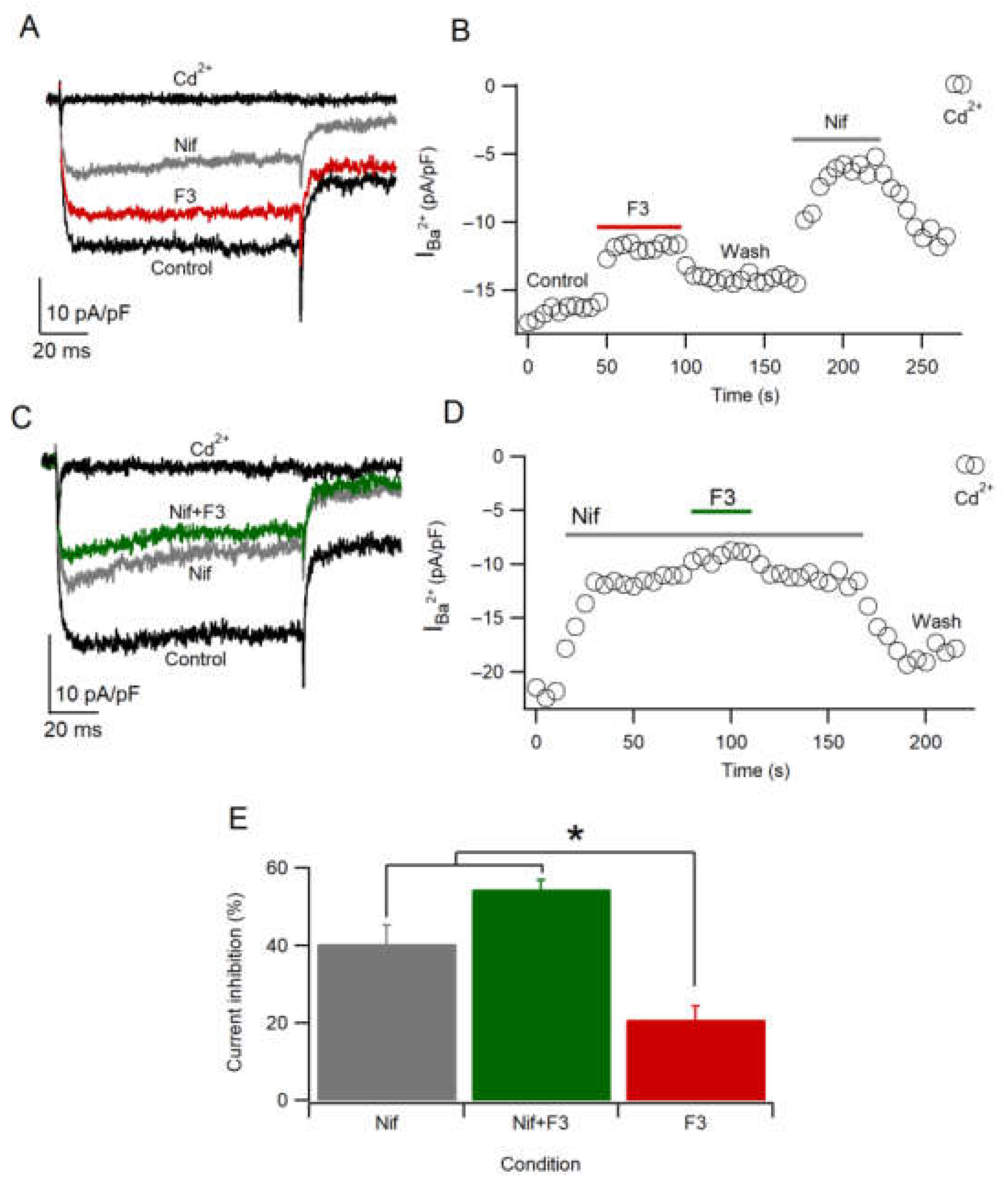
2.7. F3 Does Not Inhibit L-Type Calcium Channels Only
3. Materials and Methods
3.1. Venom Extraction and Fractionation
3.2. Protein Quantification
3.3. Mass Spectrometry Analysis
3.4. Collection and Semen Sample Preparation
3.5. Capacitation
3.6. Toxin Exposure
3.7. Chromaffin Cells Culture
3.8. Electrophysiological Recording
3.9. Statistical Analysis
4. Ethical Statement and Animal Care
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kvetkina, A.; Kostina, E.; Gladkikh, I.; Chausova, V.; Yurchenko, E.; Bakunina, I.; Pivkin, M.; Anastyuk, S.; Popov, R.; Monastyrnaya, M.; et al. Deep-Sea Anemones Are Prospective Source of New Antimicrobial and Cytotoxic Compounds. Mar. Drugs 2021, 19, 654. [Google Scholar] [CrossRef] [PubMed]
- Madio, B.; King, G.F.; Undheim, E.A.B. Sea Anemone Toxins: A Structural Overview. Mar. Drugs 2019, 17, 325. [Google Scholar] [CrossRef] [Green Version]
- Loret, E.P.; Luis, J.; Nuccio, C.; Villard, C.; Mansuelle, P.; Lebrun, R.; Villard, P.H. A Low Molecular Weight Protein from the Sea Anemone Anemonia Viridis with an Anti-Angiogenic Activity. Mar. Drugs 2018, 16, 134. [Google Scholar] [CrossRef] [Green Version]
- Tarcha, E.J.; Chi, V.; Muñoz-Elías, E.J.; Bailey, D.; Londono, L.M.; Upadhyay, S.K.; Norton, K.; Banks, A.; Tjong, I.; Nguyen, H.; et al. Durable Pharmacological Responses from the Peptide ShK-186, a Specific Kv1.3 Channel Inhibitor That Suppresses T Cell Mediators of Autoimmune Disease. J. Pharmacol. Exp. Ther. 2012, 342, 642–653. [Google Scholar] [CrossRef] [Green Version]
- Mariottini, G.L.; Pane, L. Cytotoxic and Cytolytic Cnidarian Venoms. A Review on Health Implications and Possible Therapeutic Applications. Toxins 2013, 6, 108–151. [Google Scholar] [CrossRef] [Green Version]
- Mariottini, G.L.; Grice, I.D. Antimicrobials from Cnidarians. A New Perspective for Anti-Infective Therapy? Mar. Drugs 2016, 14, 48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiménez, I.; González-Márquez, H.; Ortiz, R.; Herrera, J.A.; Garcií, A.; Betancourt, M.; Fierro, R. Changes in the Distribution of Lectin Receptors during Capacitation and Acrosome Reaction in Boar Spermatozoa. Theriogenology 2003, 59, 1171–1180. [Google Scholar] [CrossRef]
- Visconti, P.E.; Krapf, D.; de la Vega-Beltrán, J.L.; Acevedo, J.J.; Darszon, A. Ion Channels, Phosphorylation and Mammalian Sperm Capacitation. Asian J. Androl. 2011, 13, 395–405. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vicente-Carrillo, A.; Álvarez-Rodríguez, M.; Rodríguez-Martínez, H. The CatSper Channel Modulates Boar Sperm Motility during Capacitation. Reprod. Biol. 2017, 17, 69–78. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, A.; Mishra, A.K.; Swain, D.K.; Singh, V.; Yadav, S.; Saxena, A. Role of Transient Receptor Potential Channels in Regulating Spermatozoa Functions: A Mini-Review. Vet. World 2018, 11, 1618–1623. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Darszon, A.; Nishigaki, T.; Wood, C.; Treviño, C.L.; Felix, R.; Beltrán, C. Calcium Channels and Ca2+ Fluctuations in Sperm Physiology. In International Review of Cytology; Academic Press: Boston, USA, 2005; Volume 243, pp. 79–172. [Google Scholar]
- Darszon, A.; Hernández-Cruz, A. T-Type Ca2+ Channels in Spermatogenic Cells and Sperm. Pflug. Arch. 2014, 466, 819–831. [Google Scholar] [CrossRef]
- Nowicka-Bauer, K.; Szymczak-Cendlak, M. Structure and Function of Ion Channels Regulating Sperm Motility—An Overview. Int. J. Mol. Sci. 2021, 22, 3259. [Google Scholar] [CrossRef] [PubMed]
- López-González, I.; Olamendi-Portugal, T.; De la Vega-Beltrán, J.L.; Van der Walt, J.; Dyason, K.; Possani, L.D.; Felix, R.; Darszon, A. Scorpion Toxins That Block T-Type Ca2+ Channels in Spermatogenic Cells Inhibit the Sperm Acrosome Reaction. Biochem. Biophys. Res. Commun. 2003, 300, 408–414. [Google Scholar] [CrossRef]
- Romero, F.; Cunha, M.A.; Sanchez, R.; Ferreira, A.T.; Schor, N.; Oshiro, M.E.M. Effects of Arachnotoxin on Intracellular PH and Calcium in Human Spermatozoa. Fertil. Steril. 2007, 87, 1345–1349. [Google Scholar] [CrossRef] [PubMed]
- Parodi, J.; Navarrete, P.; Marconi, M.; Gutiérrez, R.S.; Martínez-Torres, A.; Mejías, F.R. Tetraethylammonium-Sensitive K+ Current in the Bovine Spermatozoa and Its Blocking by the Venom of the Chilean Latrodectus Mactans. Syst. Biol. Reprod. Med. 2010, 56, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Escoffier, J.; Couvet, M.; de Pomyers, H.; Ray, P.F.; Sève, M.; Lambeau, G.; De Waard, M.; Arnoult, C. Snake Venoms as a Source of Compounds Modulating Sperm Physiology: Secreted Phospholipases A2 from Oxyuranus Scutellatus Scutellatus Impact Sperm Motility, Acrosome Reaction and in Vitro Fertilization in Mice. Biochimie 2010, 92, 826–836. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gonzalez-Muñoz, R.; Simões, N.; Sanchez-Rodriguez, J.; Rodriguez, E.; Segura-Puertas, L. First Inventory of Sea Anemones (Cnidaria: Actiniaria) of the Mexican Caribbean. Zootaxa 2012, 3556, 1–38. [Google Scholar] [CrossRef]
- Ocaña Vicente, O.; Moro, L.; Ortea Rato, J.; Espinosa, J.; Caballer Gutiérrez, M. Guía Visual de La Biodiversidad Marina de Guanahacabibes. I -Anémonas (Anthozoa: Actiniaria, Corallimorpharia, Ceriantharia y Zoanthidea) Visual Guide of the Marine Biodiversity of Guanahacabibes. I -Anemones (Anthozoa: Actiniaria, Corallimorpharia, Ceriantharia & Zoanthidea). Avicennia Revista de biodiversidad Tropical 2007, 19, 129–138. [Google Scholar]
- Sánchez-Rodríguez, J.; Cruz-Vazquez, K. Isolation and Biological Characterization of Neurotoxic Compounds from the Sea Anemone Lebrunia Danae (Duchassaing and Michelotti, 1860). Arch. Toxicol. 2006, 80, 436–441. [Google Scholar] [CrossRef]
- Lazcano-Pérez, F.; Castro, H.; Arenas, I.; García, D.E.; González-Muñoz, R.; Arreguín-Espinosa, R. Activity of Palythoa Caribaeorum Venom on Voltage-Gated Ion Channels in Mammalian Superior Cervical Ganglion Neurons. Toxins 2016, 8, 135. [Google Scholar] [CrossRef] [Green Version]
- Maravilla-Galván, R.; Fierro, R.; González-Márquez, H.; Gómez-Arroyo, S.; Jiménez, I.; Betancourt, M. Effects of Atrazine and Fenoxaprop-Ethyl on Capacitation and the Acrosomal Reaction in Boar Sperm. Int. J. Toxicol. 2009, 28, 24–32. [Google Scholar] [CrossRef] [PubMed]
- Petters, R.M. Transgenic Livestock as Genetic Models of Human Disease. Reprod. Fertil. Dev. 1994, 6, 643–645. [Google Scholar] [CrossRef] [PubMed]
- Zhang, F.; Dai, J.; Chen, T. Role of Lactobacillus in Female Infertility Via Modulating Sperm Agglutination and Immobilization. Front. Cell. Infect. Microbiol. 2021, 10, 620529. [Google Scholar] [CrossRef] [PubMed]
- Ruggieri, G.D. Echinoderm Toxins—II. Animalizing Action in Sea Urchin Development. Toxicon 1965, 3, 157–160. [Google Scholar] [CrossRef]
- Kaur, K.; Prabha, V. Impairment by Sperm Agglutinating Factor Isolated from Escherichia Coli: Receptor Specific Interactions. Biomed. Res. Int. 2013, 2013, 548497. [Google Scholar] [CrossRef] [Green Version]
- Fenton-Navarro, B.; Arreguín-L, B.; García-Hernández, E.; Heimer, E.; Aguilar, M.B.; Rodríguez-A, C.; Arreguín-Espinosa, R. Purification and Structural Characterization of Lectins from the Cnidarian Bunodeopsis Antillienis. Toxicon 2003, 42, 525–532. [Google Scholar] [CrossRef]
- Publicover, S.; Harper, C.V.; Barratt, C. [Ca2+]i Signalling in Sperm—Making the Most of What You’ve Got. Nat. Cell Biol. 2007, 9, 235–242. [Google Scholar] [CrossRef] [PubMed]
- Darszon, A.; Nishigaki, T.; Beltran, C.; Treviño, C.L. Calcium Channels in the Development, Maturation, and Function of Spermatozoa. Physiol. Rev. 2011, 91, 1305–1355. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Darszon, A.; Treviño, C.L.; Wood, C.; Galindo, B.; Rodríguez-Miranda, E.; Acevedo, J.J.; Hernandez-González, E.O.; Beltrán, C.; Martínez-López, P.; Nishigaki, T. Ion Channels in Sperm Motility and Capacitation. Soc. Reprod. Fertil. Suppl. 2007, 65, 229–244. [Google Scholar] [PubMed]
- García, A.G.; García-De-Diego, A.M.; Gandía, L.; Borges, R.; García-Sancho, J. Calcium Signaling and Exocytosis in Adrenal Chromaffin Cells. Physiol. Rev. 2006, 86, 1093–1131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bosmans, F.; Tytgat, J. Sea Anemone Venom as a Source of Insecticidal Peptides Acting on Voltage-Gated Na+ Channels. Toxicon 2007, 49, 550–560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Niklas, B.; Jankowska, M.; Gordon, D.; Béress, L.; Stankiewicz, M.; Nowak, W. Interactions of Sea Anemone Toxins with Insect Sodium Channel—Insights from Electrophysiology and Molecular Docking Studies. Molecules 2021, 26, 1302. [Google Scholar] [CrossRef] [PubMed]
- Orts, D.J.B.; Moran, Y.; Cologna, C.T.; Peigneur, S.; Madio, B.; Praher, D.; Quinton, L.; De Pauw, E.; Bicudo, J.E.P.W.; Tytgat, J.; et al. BcsTx3 Is a Founder of a Novel Sea Anemone Toxin Family of Potassium Channel Blocker. FEBS J. 2013, 280, 4839–4852. [Google Scholar] [CrossRef] [PubMed]
- Prakriya, M.; Lingle, C.J. BK Channel Activation by Brief Depolarizations Requires Ca2+ Influx through L- and Q-Type Ca2+ Channels in Rat Chromaffin Cells. J. Neurophysiol. 1999, 81, 2267–2278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schackmann, R.W.; Christen, R.; Shapiro, B.M. Membrane Potential Depolarization and Increased Intracellular PH Accompany the Acrosome Reaction of Sea Urchin Sperm. Proc. Natl. Acad. Sci. USA 1981, 78, 6066–6070. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marcantoni, A.; Baldelli, P.; Hernandez-Guijo, J.M.; Comunanza, V.; Carabelli, V.; Carbone, E. L-Type Calcium Channels in Adrenal Chromaffin Cells: Role in Pace-Making and Secretion. Cell Calcium 2007, 42, 397–408. [Google Scholar] [CrossRef]
- Helton, T.D.; Xu, W.; Lipscombe, D. Neuronal L-Type Calcium Channels Open Quickly and Are Inhibited Slowly. J. Neurosci. 2005, 25, 10247–10251. [Google Scholar] [CrossRef]
- Wang, Y.; Tang, S.; Harvey, K.E.; Salyer, A.E.; Li, T.A.; Rantz, E.K.; Lill, M.A.; Hockerman, G.H. Molecular Determinants of the Differential Modulation of Cav1.2 and Cav1.3 by Nifedipine and FPL 64176. Mol. Pharmacol. 2018, 94, 973–983. [Google Scholar] [CrossRef] [Green Version]
- Garner, D.L.; Hafez, E.S.E. Spermatozoa and Seminal Plasma. In Reproduction in Farm Animals; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2000; pp. 96–109. ISBN 978-1-119-26530-6. [Google Scholar]
- Oseguera-López, I.; Pérez-Cerezales, S.; Ortiz-Sánchez, P.B.; Mondragon-Payne, O.; Sánchez-Sánchez, R.; Jiménez-Morales, I.; Fierro, R.; González-Márquez, H. Perfluorooctane Sulfonate (PFOS) and Perfluorohexane Sulfonate (PFHxS) Alters Protein Phosphorylation, Increase ROS Levels and DNA Fragmentation during In Vitro Capacitation of Boar Spermatozoa. Animals 2020, 10, 1934. [Google Scholar] [CrossRef]
| Parameter | F3 (μg Protein/mL) | |||
|---|---|---|---|---|
| 0.0 | 10 | 20 | 50 | |
| Motility | +++++ | ++++ | ++ | + |
| Agglutination | ++ | ++ | ++++ | +++++ |
| F3 (μg Protein/mL) | Spermatozoa Status (%) | |
|---|---|---|
| Non-Capacitated | Capacitated | |
| 0.0 | 3.54 | 6.36 |
| 10 | 1.41 | 9.19 |
| 20 | 1.41 | 3.54 |
| 50 | 5.66 | 2.12 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lazcano-Pérez, F.; Bermeo, K.; Castro, H.; Salazar Campos, Z.; Arenas, I.; Zavala-Moreno, A.; Chávez-Villela, S.N.; Jiménez, I.; Arreguín-Espinosa, R.; Fierro, R.; et al. A Sea Anemone Lebrunia neglecta Venom Fraction Decreases Boar Sperm Cells Capacitation: Possible Involvement of HVA Calcium Channels. Toxins 2022, 14, 261. https://doi.org/10.3390/toxins14040261
Lazcano-Pérez F, Bermeo K, Castro H, Salazar Campos Z, Arenas I, Zavala-Moreno A, Chávez-Villela SN, Jiménez I, Arreguín-Espinosa R, Fierro R, et al. A Sea Anemone Lebrunia neglecta Venom Fraction Decreases Boar Sperm Cells Capacitation: Possible Involvement of HVA Calcium Channels. Toxins. 2022; 14(4):261. https://doi.org/10.3390/toxins14040261
Chicago/Turabian StyleLazcano-Pérez, Fernando, Karina Bermeo, Héctor Castro, Zayil Salazar Campos, Isabel Arenas, Ariana Zavala-Moreno, Sheila Narayán Chávez-Villela, Irma Jiménez, Roberto Arreguín-Espinosa, Reyna Fierro, and et al. 2022. "A Sea Anemone Lebrunia neglecta Venom Fraction Decreases Boar Sperm Cells Capacitation: Possible Involvement of HVA Calcium Channels" Toxins 14, no. 4: 261. https://doi.org/10.3390/toxins14040261
APA StyleLazcano-Pérez, F., Bermeo, K., Castro, H., Salazar Campos, Z., Arenas, I., Zavala-Moreno, A., Chávez-Villela, S. N., Jiménez, I., Arreguín-Espinosa, R., Fierro, R., González-Márquez, H., Garcia, D. E., & Sánchez-Rodríguez, J. (2022). A Sea Anemone Lebrunia neglecta Venom Fraction Decreases Boar Sperm Cells Capacitation: Possible Involvement of HVA Calcium Channels. Toxins, 14(4), 261. https://doi.org/10.3390/toxins14040261






