Comprehensive Evaluation of the Efficiency of Yeast Cell Wall Extract to Adsorb Ochratoxin A and Mitigate Accumulation of the Toxin in Broiler Chickens
Abstract
1. Introduction
2. Results
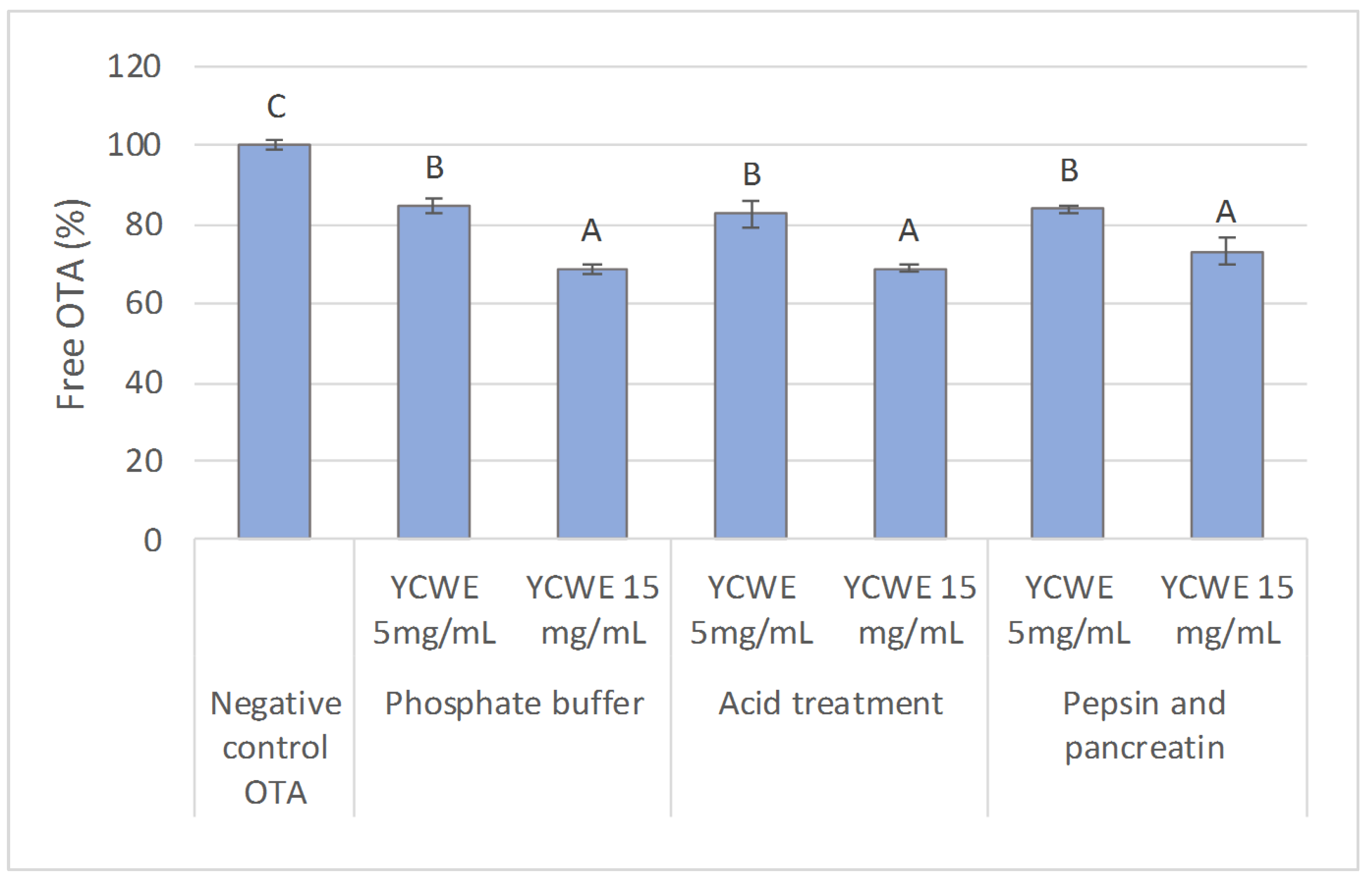
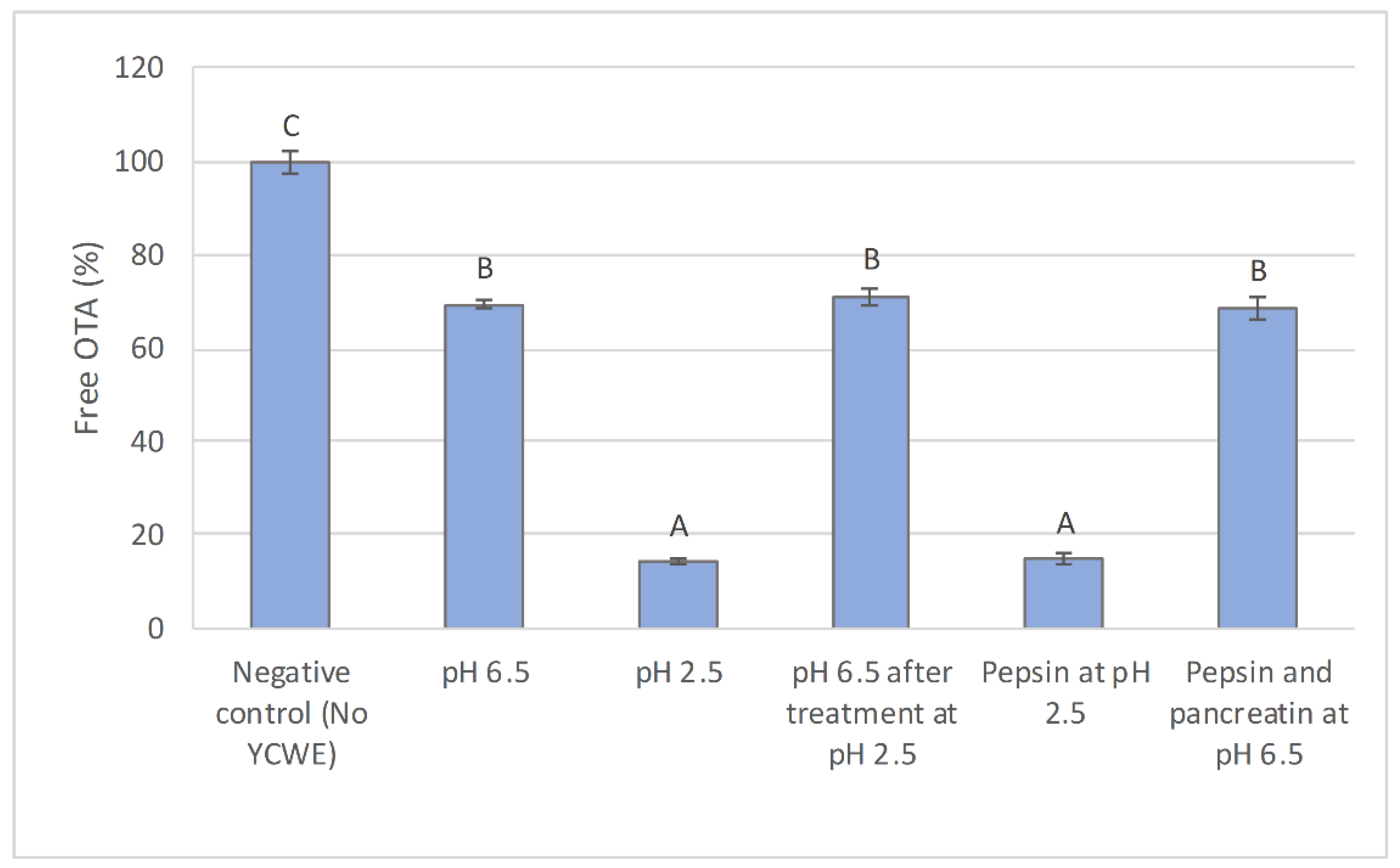
2.1. In Vitro Evaluation of the Influence of Simulated Digestive Conditions on OTA Sequestration
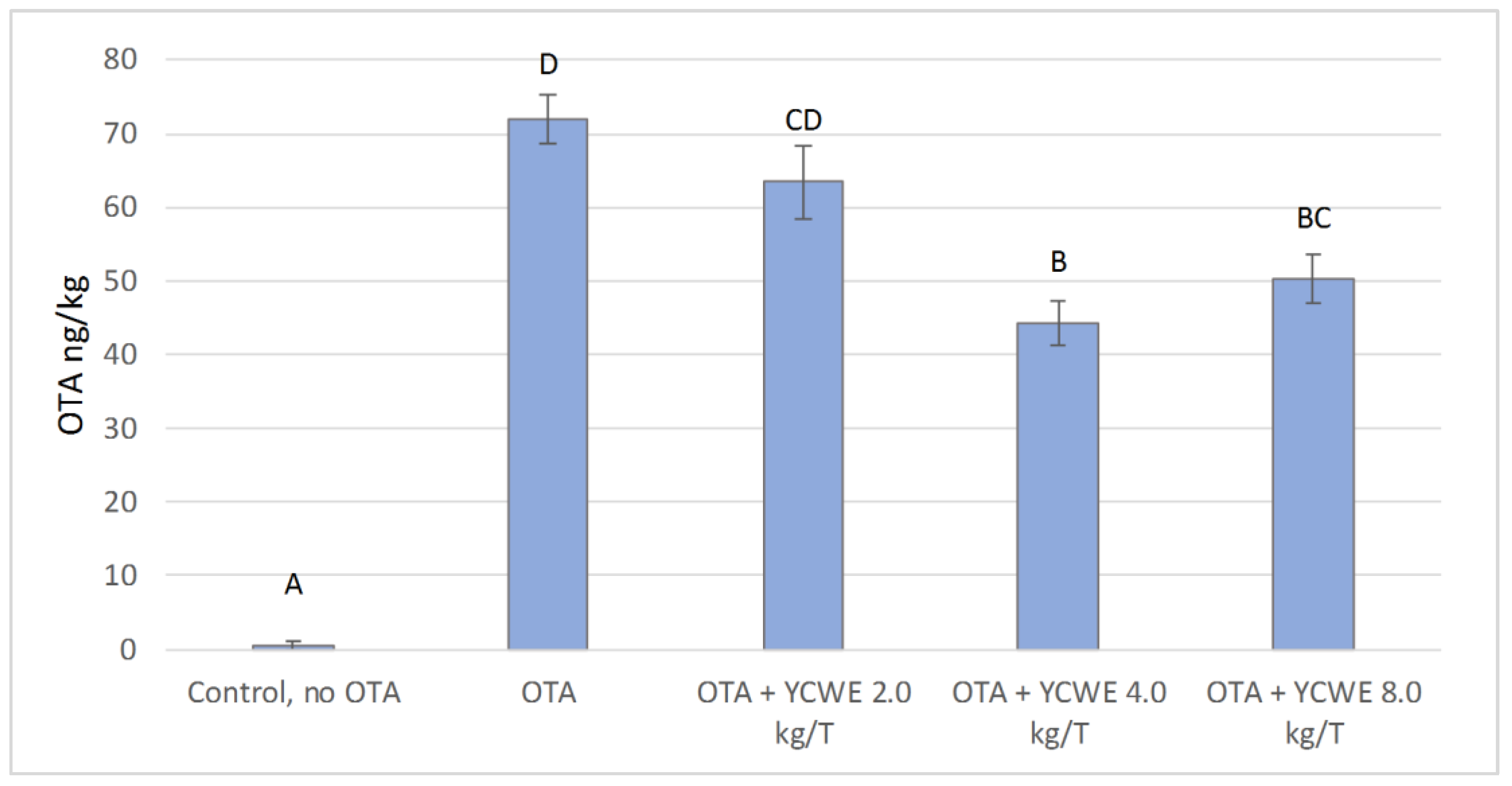
2.2. In Vitro OTA Sequestration by YCWE
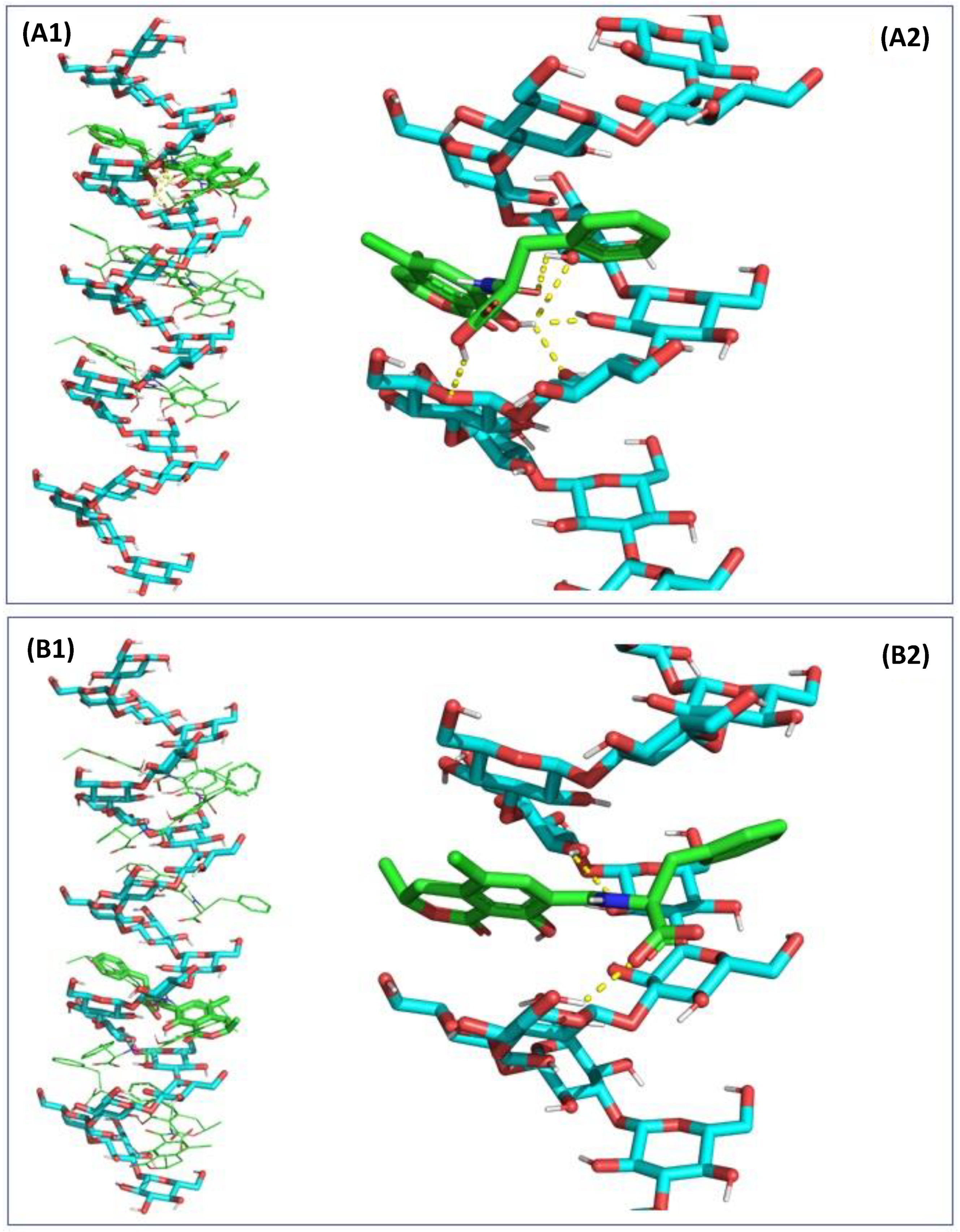
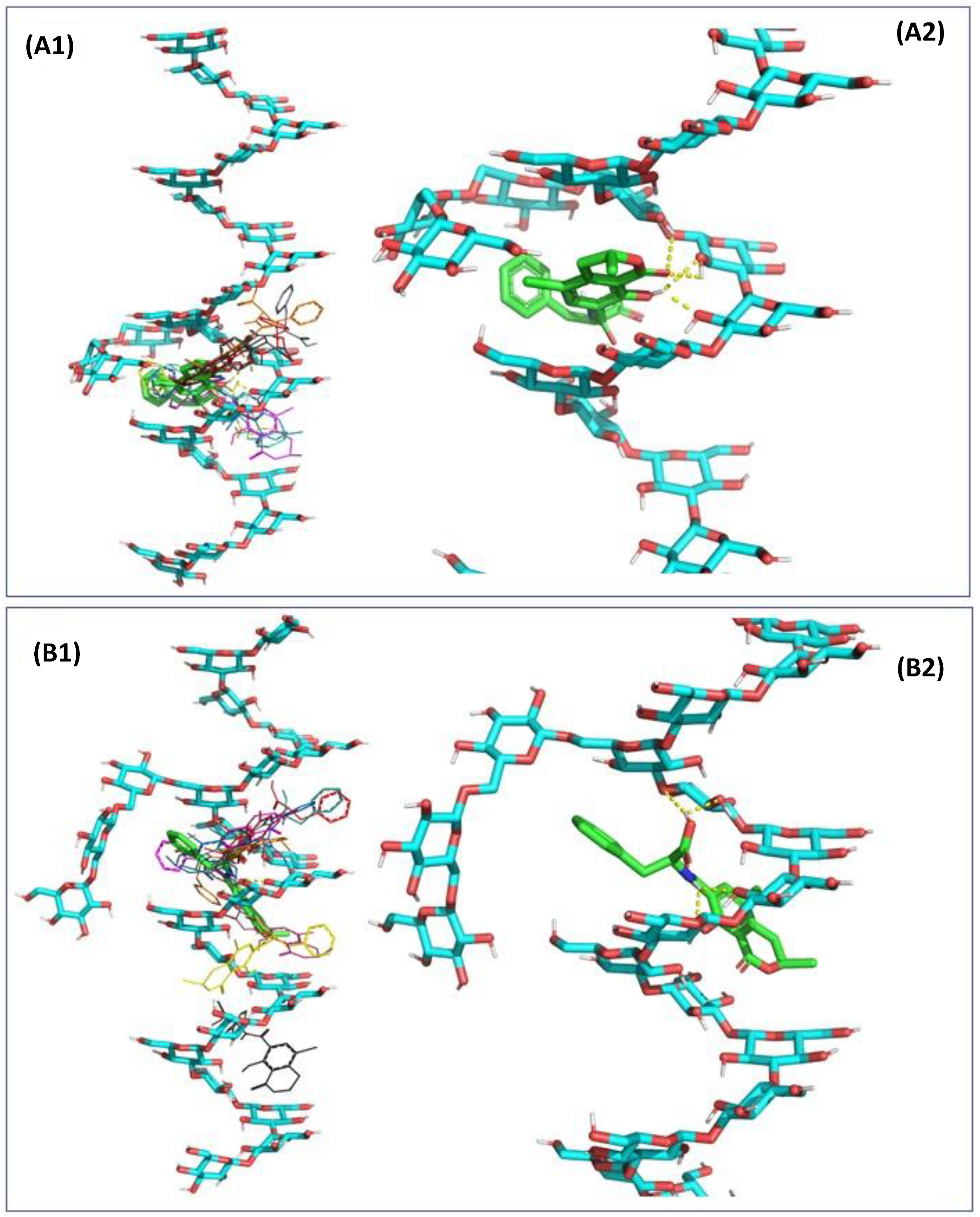
2.3. In Silico Assessment of the Sequestration Properties Investigated by Molecular Mechanics
2.4. In Silico Assessment of the Sequestration Properties Investigated by Molecular Dynamics
2.5. In Vivo Broiler Chicken Feeding Trial
2.6. Analysis of OTA Deposits in Broiler Livers
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. In Vitro Assessment of OTA Sequestration by YCWE
5.2. In Vitro Digestive Simulation Conditions and OTA Sequestration Activity of YCWE
5.3. In Silico Assessment of the Sequestration Properties Investigated by Means of Molecular Mechanics and Dynamics
5.4. In Vivo Dietary Treatments
5.5. Animal Trial
5.6. Mycotoxin Analysis of Dietary Treatments
5.7. OTA Analysis of Liver Tissues
5.8. Data Analysis
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
References
- Ramos, A.; Labernia, N.; Marı́n, S.; Sanchis, V.; Magan, N. Effect of water activity and temperature on growth and ochratoxin production by three strains of Aspergillus ochraceus on a barley extract medium and on barley grains. Int. J. Food Microbiol. 1998, 44, 133–140. [Google Scholar] [CrossRef]
- Cairns-Fuller, V.; Aldred, D.; Magan, N. Water, temperature and gas composition interactions affect growth and ochratoxin A production by isolates of Penicillium verrucosum on wheat grain. J. Appl. Microbiol. 2005, 99, 1215–1221. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Commission Recommendation 2006/576/EC of 17 August 2006 on the presence of deoxynivalenol, zearalenone, ochratoxin A, T-2 and HT-2 and fumonisins in products intended for animal feeding. Off. J. Eur. Union L 2006, 229, 7–9. [Google Scholar]
- Swamy, H.V.L.N.; Smith, T.K.; Karrow, N.A.; Boermans, H.J. Effects of Feeding Blends of Grains Naturally Contaminated with Fusarium. Poult. Sci. 2004, 83, 533–543. [Google Scholar] [CrossRef] [PubMed]
- Moran, C.A.; Apajalahti, J.; Yiannikouris, A.; Ojanpera, S.; Kettunen, H. Effects of low dietary aflatoxin B1 on broiler liver concentration without and with Mycosorb® toxin binder. J. Appl. Anim. Nutr. 2013, 2. [Google Scholar] [CrossRef]
- Moran, C.A.; Kettunen, H.; Yiannikouris, A.; Ojanpera, S.; Pennala, E.; Helander, I.M.; Apajalahti, J. A dairy cow model to assess aflatoxin transmission from feed into milk—Evaluating efficacy of the mycotoxin binder Mycosorb®. J. Appl. Anim. Nutr. 2013, 2. [Google Scholar] [CrossRef]
- Battacone, G.; Nudda, A.; Pulina, G. Effects of ochratoxin a on livestock production. Toxins 2010, 2, 1796–1824. [Google Scholar] [CrossRef]
- Tao, Y.; Xie, S.; Xu, F.; Liu, A.; Wang, Y.; Chen, D.; Pan, Y.; Huang, L.; Peng, D.; Wang, X.; et al. Ochratoxin A: Toxicity, oxidative stress and metabolism. Food Chem. Toxicol. 2018, 112, 320–331. [Google Scholar] [CrossRef]
- Pfohl-Leszkowicz, A.; Manderville, R.A. Ochratoxin A: An overview on toxicity and carcinogenicity in animals and humans. Mol. Nutr. Food Res. 2007, 51, 61–99. [Google Scholar] [CrossRef]
- Kumagai, S.; Aibara, K. Intestinal absorption and secretion of ochratoxin A in the rat. Toxicol. Appl. Pharm. 1982, 64, 94–102. [Google Scholar] [CrossRef]
- Galtier, P.; Alvinerie, M.; Charpenteau, J.L. The pharmacokinetic profiles of ochratoxin A in pigs, rabbits and chickens. Food Cosmet. Toxicol. 1981, 19, 735–738. [Google Scholar] [CrossRef]
- Hagelberg, S.; Hult, K.; Fuchs, R. Toxicokinetics of ochratoxin A in several species and its plasma-binding properties. J. Appl. Toxicol. 1989, 9, 91–96. [Google Scholar] [CrossRef] [PubMed]
- Vettorazzi, A.; González-Peñas, E.; de Cerain, A.L. Ochratoxin A kinetics: A review of analytical methods and studies in rat model. Food Chem. Toxicol. 2014, 72, 273–288. [Google Scholar] [CrossRef] [PubMed]
- Galtier, P.; Charpenteau, J.; Alvinerie, M.; Labouche, C. The pharmacokinetic profile of ochratoxin A in the rat after oral and intravenous administration. Drug Metab. Dispos. 1979, 7, 429–434. [Google Scholar]
- Vila-Donat, P.; Marín, S.; Sanchis, V.; Ramos, A.J. A review of the mycotoxin adsorbing agents, with an emphasis on their multi-binding capacity, for animal feed decontamination. Food Chem. Toxicol. 2018, 114, 246–259. [Google Scholar] [CrossRef]
- Yiannikouris, A.; Kettunen, H.; Apajalahti, J.; Pennala, E.; Moran, C.A. Comparison of the sequestering properties of yeast cell wall extract and hydrated sodium calcium aluminosilicate in three in vitro models accounting for the animal physiological bioavailability of zearalenone. Food Addit. Contam.-Part A 2013, 30, 1641–1650. [Google Scholar] [CrossRef]
- Oh, S.Y.; Quinton, V.M.; Boermans, H.J.; Swamy, H.V.L.N.; Karrow, N.A. In vitro exposure of Penicillium mycotoxins with or without a modified yeast cell wall extract (mYCWE) on bovine macrophages (BoMacs). Mycotoxin Res. 2015, 31, 167–175. [Google Scholar] [CrossRef]
- Yiannikouris, A.; François, J.; Poughon, L.; Dussap, C.G.; Bertin, G.; Jeminet, G.; Jouany, J.P. Alkali extraction of β-d-glucans from Saccharomyces cerevisiae cell wall and study of their adsorptive properties toward zearalenone. J. Agric. Food Chem. 2004, 52, 3666–3673. [Google Scholar] [CrossRef]
- European Food Safety Authority (EFSA). Guidance for the preparation of dossiers for technological additives. EFSA J. 2012, 10, 2528. [Google Scholar] [CrossRef]
- Sacranie, A.; Svihus, B.; Denstadli, V.; Moen, B.; Iji, P.A.; Choct, M. The effect of insoluble fiber and intermittent feeding on gizzard development, gut motility, and performance of broiler chickens. Poult. Sci. 2012, 91, 693–700. [Google Scholar] [CrossRef]
- Bazin, I.; Faucet-Marquis, V.; Monje, M.-C.; El Khoury, M.; Marty, J.-L.; Pfohl-Leszkowicz, A.; Bazin, I.; Faucet-Marquis, V.; Monje, M.-C.; El Khoury, M.; et al. Impact of pH on the Stability and the Cross-Reactivity of Ochratoxin A and Citrinin. Toxins 2013, 5, 2324–2340. [Google Scholar] [CrossRef] [PubMed]
- Rotter, R.G.; Frohlich, A.A.; Marquardt, R.R. Influence of dietary charcoal on ochratoxin A toxicity in Leghorn chicks. Can. J. Vet. Res. 1989, 53, 449–453. [Google Scholar] [PubMed]
- García, A.R.; Avila, E.; Rosiles, R.; Petrone, V.M. Evaluation of Two mycotoxin binders to reduce toxicity of broiler diets containing ochratoxin A and T-2 toxin contaminated grain. Avian Dis. 2003, 47, 691–699. [Google Scholar] [CrossRef]
- Santin, E.; Paulillo, A.; Nakagui, L.; Alessi, A.; Maiorka, A. Evaluation of yeast cell wall on the performance of broiles fed diets with or without mycotoxins. Braz. J. Poult. Sci. 2006, 8, 221–225. [Google Scholar] [CrossRef][Green Version]
- Politis, I.; Fegeros, K.; Nitsch, S.; Schatzmayr, G.; Kantas, D. Use of Trichosporon mycotoxinivorans to suppress the effects of ochratoxicosis on the immune system of broiler chicks. Br. Poult. Sci. 2005, 46, 58–65. [Google Scholar] [CrossRef]
- Oh, S.Y.; Cedergreen, N.; Yiannikouris, A.; Swamy, H.V.L.N.; Karrow, N.A. Assessing interactions of binary mixtures of Penicillium mycotoxins (PMs) by using a bovine macrophage cell line (BoMacs). Toxicol. Appl. Pharm. 2017, 318, 33–40. [Google Scholar] [CrossRef]
- Haighton, L.A.; Lynch, B.S.; Magnuson, B.A.; Nestmann, E.R. A reassessment of risk associated with dietary intake of ochratoxin A based on a lifetime exposure model. Crit. Rev. Toxicol. 2012, 42, 147–168. [Google Scholar] [CrossRef]
- Iqbal, S.Z.; Nisar, S.; Asi, M.R.; Jinap, S. Natural incidence of aflatoxins, ochratoxin A and zearalenone in chicken meat and eggs. Food Control 2014, 43, 98–103. [Google Scholar] [CrossRef]
- Hanif, N.Q.; Muhammad, G.; Muhammad, K.; Tahira, I.; Raja, G.K. Reduction of ochratoxin A in broiler serum and tissues by Trichosporon mycotoxinivorans. Res. Vet. Sci. 2012, 93, 795–797. [Google Scholar] [CrossRef]
- Joo, Y.D.; Kang, C.W.; An, B.K.; Ahn, J.S.; Borutova, R. Effects of ochratoxin a and preventive action of a mycotoxin-deactivation product in broiler chickens. Vet. Ir. Zootech. 2013, 61, 22–29. [Google Scholar]
- Stoev, S.D.; Stefanov, M.; Denev, S.; Radic, B.; Domijan, A.M.; Peraica, M. Experimental mycotoxicosis in chickens induced by ochratoxin A and penicillic acid and intervention with natural plant extracts. Vet. Res. Commun. 2004, 28, 727–746. [Google Scholar] [CrossRef] [PubMed]
- Pfohl-Leszkowicz, A.; Hadjeba-Medjdoub, K.; Ballet, N.; Schrickx, J.; Fink-Gremmels, J. Assessment and characterisation of yeast-based products intended to mitigate ochratoxin exposure using in vitro and in vivo models. Food Addit. Contam.-Part A Chem. Anal. Control. Expo. Risk Assess. 2015, 32, 604–616. [Google Scholar] [CrossRef] [PubMed]
- Pozzo, L.; Cavallarin, L.; Antoniazzi, S.; Guerre, P.; Biasibetti, E.; Capucchio, M.T.; Schiavone, A. Feeding a diet contaminated with ochratoxin A for broiler chickens at the maximum level recommended by the EU for poultry feeds (0.1 mg/kg). 2. Effects on meat quality, oxidative stress, residues and histological traits. J. Anim. Physiol. Anim. Nutr. 2013, 97, 23–31. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, I.; Naehrer, K.; Rodrigues, I.; Naehrer, K. A Three-Year Survey on the Worldwide Occurrence of Mycotoxins in Feedstuffs and Feed. Toxins 2012, 4, 663–675. [Google Scholar] [CrossRef]
- Peng, W.-X.; Marchal, J.L.M.; van der Poel, A.F.B. Strategies to prevent and reduce mycotoxins for compound feed manufacturing. Anim. Feed Sci. Technol. 2018, 237, 129–153. [Google Scholar] [CrossRef]
- Yiannikouris, A.; André, G.; Buleon, A.; Jeminet, G.; Canet, I.; François, J.; Bertin, G.; Jouany, J.-P. Comprehensive Conformational Study of Key Interactions Involved in Zearalenone Complexation with beta-d-Glucans. Biomacromolecules 2004, 7, 2176–2185. [Google Scholar] [CrossRef]
- Trott, O.; Olson, A.J. AutoDock Vina: Improving the speed and accuracy of docking with a new scoring function, efficient optimization and multithreading, J. Comput. Chem. 2010, 31, 455–461. [Google Scholar] [CrossRef]
- Yiannikouris, A.; André, G.; Poughon, L.; François, J.; Dussap, C.-G.; Jeminet, G.; Bertin, G.; Jouany, J.-P. Chemical and Conformational Study of the Interactions Involved in Mycotoxin Complexation with beta-D-Glucans. Biomacromolecules 2006, 7, 1147–1155. [Google Scholar] [CrossRef]
- Danne, R.; Poojari, C.; Martinez-Seara, H. doGlycans-Tools for Preparing Carbohydrate Structures for Atomistic Simulations of Glycoproteins, Glycolipids, and Carbohydrate Polymers for GROMACS. J. Chem. Inf. Model. 2017, 57, 2401–2406. [Google Scholar] [CrossRef]
- Jorgensen, W.L.; Tirado-Rives, J. Potential energy functions for atomic-level simulations of water and organic and biomolecular systems. Proc. Natl. Acad. Sci. USA 2005, 102, 6665–6670. [Google Scholar] [CrossRef]
- Dodda, L.S.; Vilseck, J.Z.; Tirado-Rives, J.; Jorgensen, W.L.J. 1.14* CM1A-LBCC: Localized Bond-Charge Corrected CM1A Charges for Condensed-Phase Simulations. Phys. Chem. B 2017, 121, 3864–3870. [Google Scholar] [CrossRef] [PubMed]
- Dodda, L.S.; Cabeza de Vaca, I.; Tirado-Rives, J.; Jorgensen, W.L. LigParGen web server: An automatic OPLS-AA parameter generator for organic ligands. Nucleic Acids Res. 2017, 45, W331–W336. [Google Scholar] [CrossRef] [PubMed]
- Abraham, M.J.; Murtola, T.; Schulz, R.; Páll, S.; Smith, J.C.; Hess, B.; Lindahl, E. GROMACS: High Performance Molecular Simulations Through Multi-Level Parallelism from Laptops to Supercomputers. SoftwareX 2015, 1–2, 19–25. [Google Scholar] [CrossRef]
- Berendsen, H.J.C.; van der Spoel, D.; van Drunen, R. GROMACS: A message-passing parallel molecular dynamics implementation. Comput. Phys. Commun. 1995, 91, 43–56. [Google Scholar] [CrossRef]
- Lindahl, E.; Hess, B.; van der Spoel, D. GROMACS 3.0: A package for molecular simulation and trajectory analysis. Mol. Model. Annu. 2001, 7, 306–317. [Google Scholar] [CrossRef]
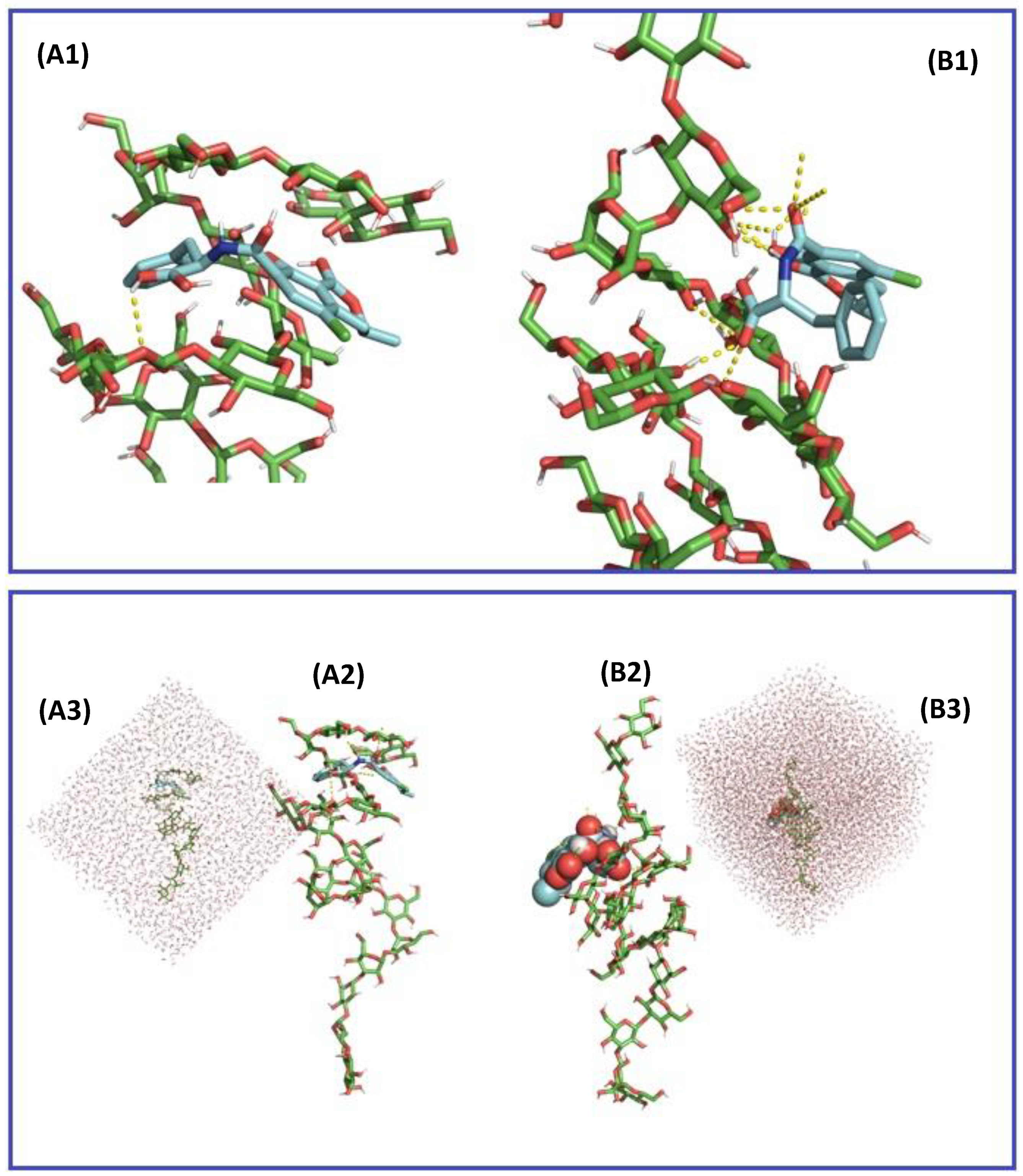
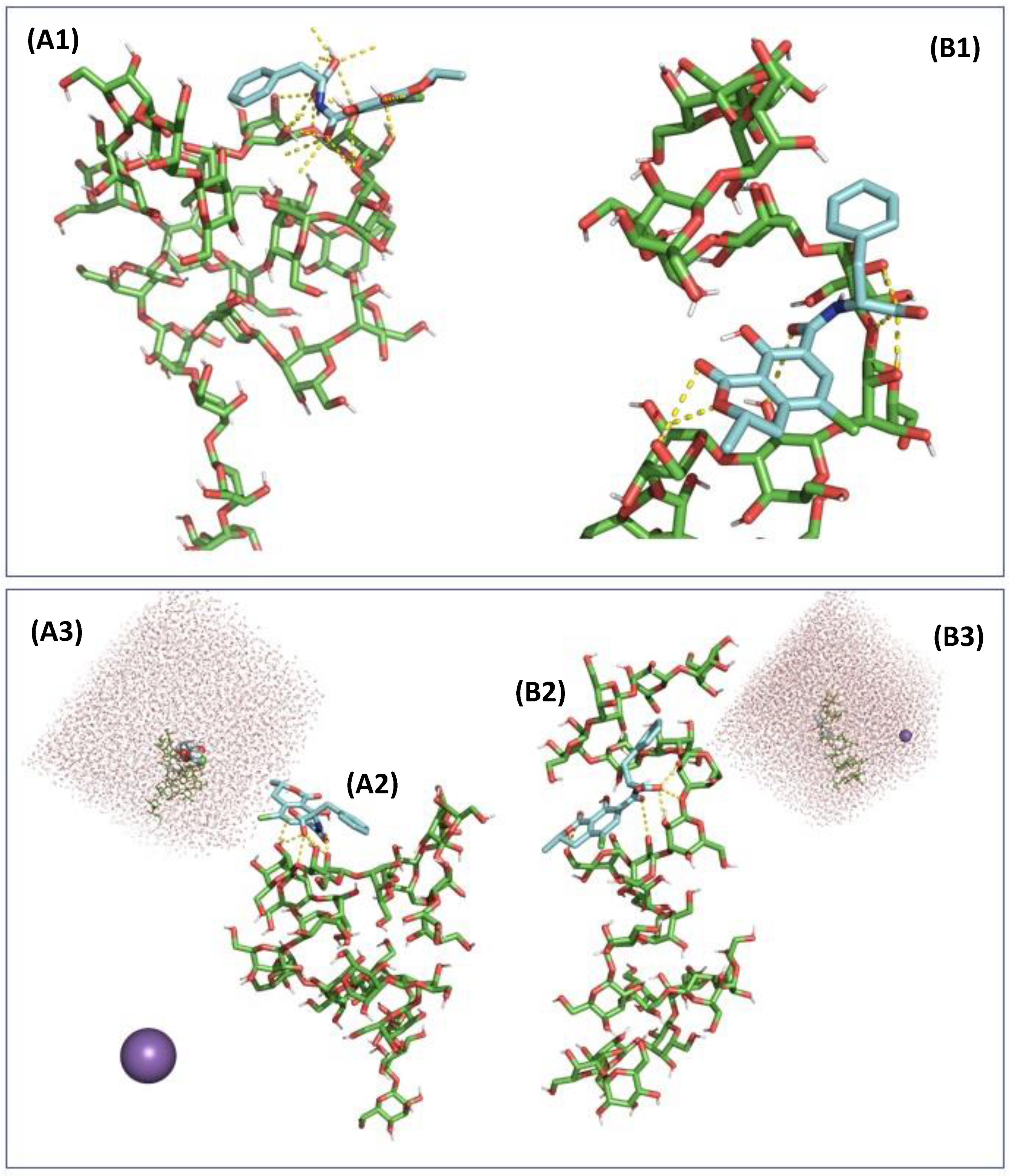
| pH | OTA State | Energy | (kJ/mol) | Average H-Bond G3-OTA | Average H-Bond OTA-Water |
|---|---|---|---|---|---|
| Constrained Receptor | |||||
| pH 2.5 | Protonated OTA and glucan chain Energy | Coulombic Energy | −47.5541 ± 3.0 | 2.218 | 3.861 |
| Lennard-Jones energy | −114.6890 ± 3.9 | ||||
| Total | −162.2431 | ||||
| pH 6.5 | Singly deprotonated OTA and glucan chain energy | Coulombic Energy | −104.4470 ± 4.6 | 3.010 | 5.624 |
| Lennard-Jones energy | −47.3817 ± 4.4 | ||||
| Total | −151.8287 | ||||
| Unconstrainted Receptor | |||||
| pH 2.5 | Protonated OTA and glucan chain Energy | Coulombic Energy | −58.1255 ± 2.4 | 0.911 | 4.316 |
| Lennard-Jones energy | −87.1747 ± 2.9 | ||||
| Total | −145.3002 | ||||
| pH 6.5 | Singly deprotonated OTA and glucan chain energy | Coulombic Energy | −101.9410 ± 9.2 | 2.891 | 6.604 |
| Lennard-Jones energy | −80.0512 ± 6.7 | ||||
| Total | −181.9922 | ||||
| Diet | Body Weight (g) | Mortality (%) | FCR Day 1–21 | |
|---|---|---|---|---|
| Day 1 | Day 21 | |||
| Control | 45.9 ± 0.7 | 856 ± 33 | 2.1 ± 2.1 | 1.62 ± 0.12 |
| + OTA | 46.5 ± 0.6 | 841 ± 37 | 4.2 ± 4.2 | 1.48 ± 0.03 |
| + OTA + YCWE 2.0 kg/T | 45.0 ± 0.6 | 782 ± 22 | 6.3 ± 3.0 | 1.73 ± 0.15 |
| + OTA + YCWE 4.0 kg/T | 45.6 ± 0.5 | 832 ± 22 | 2.1 ± 2.1 | 1.49 ± 0.05 |
| + OTA + YCWE 8.0 kg/T | 46.0 ± 0.9 | 832 ± 45 | 2.1 ± 2.1 | 1.50 ± 0.04 |
| Ingredient | % |
|---|---|
| Wheat | 60.31 |
| Soybean meal | 31.60 |
| Rapeseed oil | 4.0 |
| Monocalcium phosphate | 1.70 |
| Limestone | 1.30 |
| NaCl | 0.40 |
| Mineral premix 1 | 0.20 |
| Vitamin premix 2 | 0.20 |
| Methionine | 0.10 |
| Lysine | 0.09 |
| Threonine | 0.10 |
| Total | 100.00 |
| Diet | Target Dose of OTA (mg/kg) | Analysed Dose of OTA (mg/kg) 1 |
|---|---|---|
| Control | 0.0 | <0.001 |
| + OTA | 0.090 | 0.110 |
| + OTA + YCWE 2.0 kg/T | 0.090 | 0.099 |
| + OTA + YCWE 4.0 kg/T | 0.090 | 0.140 |
| + OTA + YCWE 8.0 kg/T | 0.090 | 0.110 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vartiainen, S.; Yiannikouris, A.; Apajalahti, J.; Moran, C.A. Comprehensive Evaluation of the Efficiency of Yeast Cell Wall Extract to Adsorb Ochratoxin A and Mitigate Accumulation of the Toxin in Broiler Chickens. Toxins 2020, 12, 37. https://doi.org/10.3390/toxins12010037
Vartiainen S, Yiannikouris A, Apajalahti J, Moran CA. Comprehensive Evaluation of the Efficiency of Yeast Cell Wall Extract to Adsorb Ochratoxin A and Mitigate Accumulation of the Toxin in Broiler Chickens. Toxins. 2020; 12(1):37. https://doi.org/10.3390/toxins12010037
Chicago/Turabian StyleVartiainen, Suvi, Alexandros Yiannikouris, Juha Apajalahti, and Colm A. Moran. 2020. "Comprehensive Evaluation of the Efficiency of Yeast Cell Wall Extract to Adsorb Ochratoxin A and Mitigate Accumulation of the Toxin in Broiler Chickens" Toxins 12, no. 1: 37. https://doi.org/10.3390/toxins12010037
APA StyleVartiainen, S., Yiannikouris, A., Apajalahti, J., & Moran, C. A. (2020). Comprehensive Evaluation of the Efficiency of Yeast Cell Wall Extract to Adsorb Ochratoxin A and Mitigate Accumulation of the Toxin in Broiler Chickens. Toxins, 12(1), 37. https://doi.org/10.3390/toxins12010037





