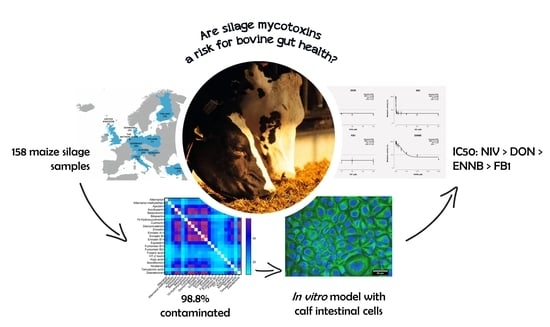
Mycotoxin Occurrence in Maize Silage—A Neglected Risk for Bovine Gut Health?
Abstract
1. Introduction
2. Results
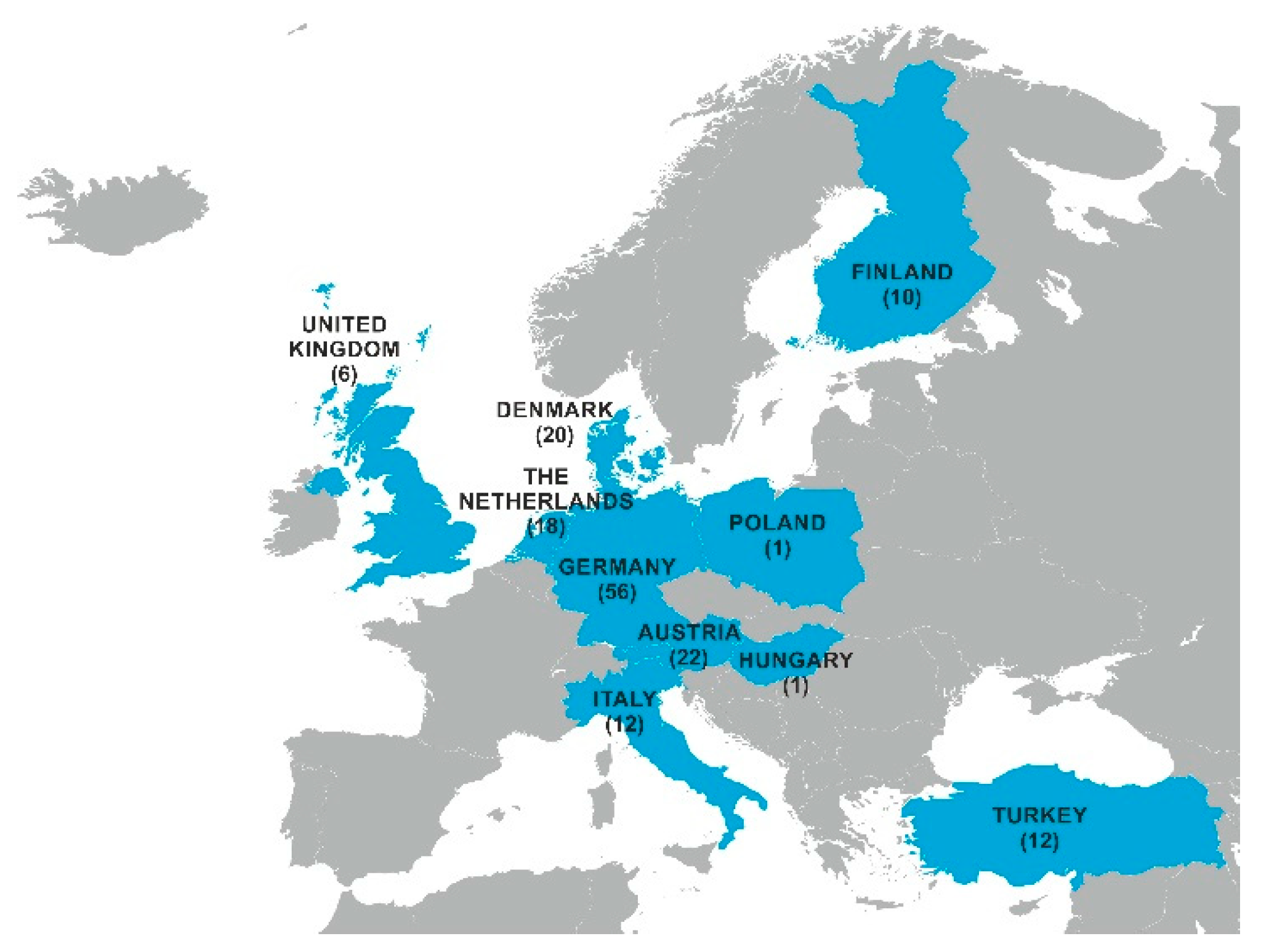
2.1. Mycotoxin Occurrence in Maize Silage
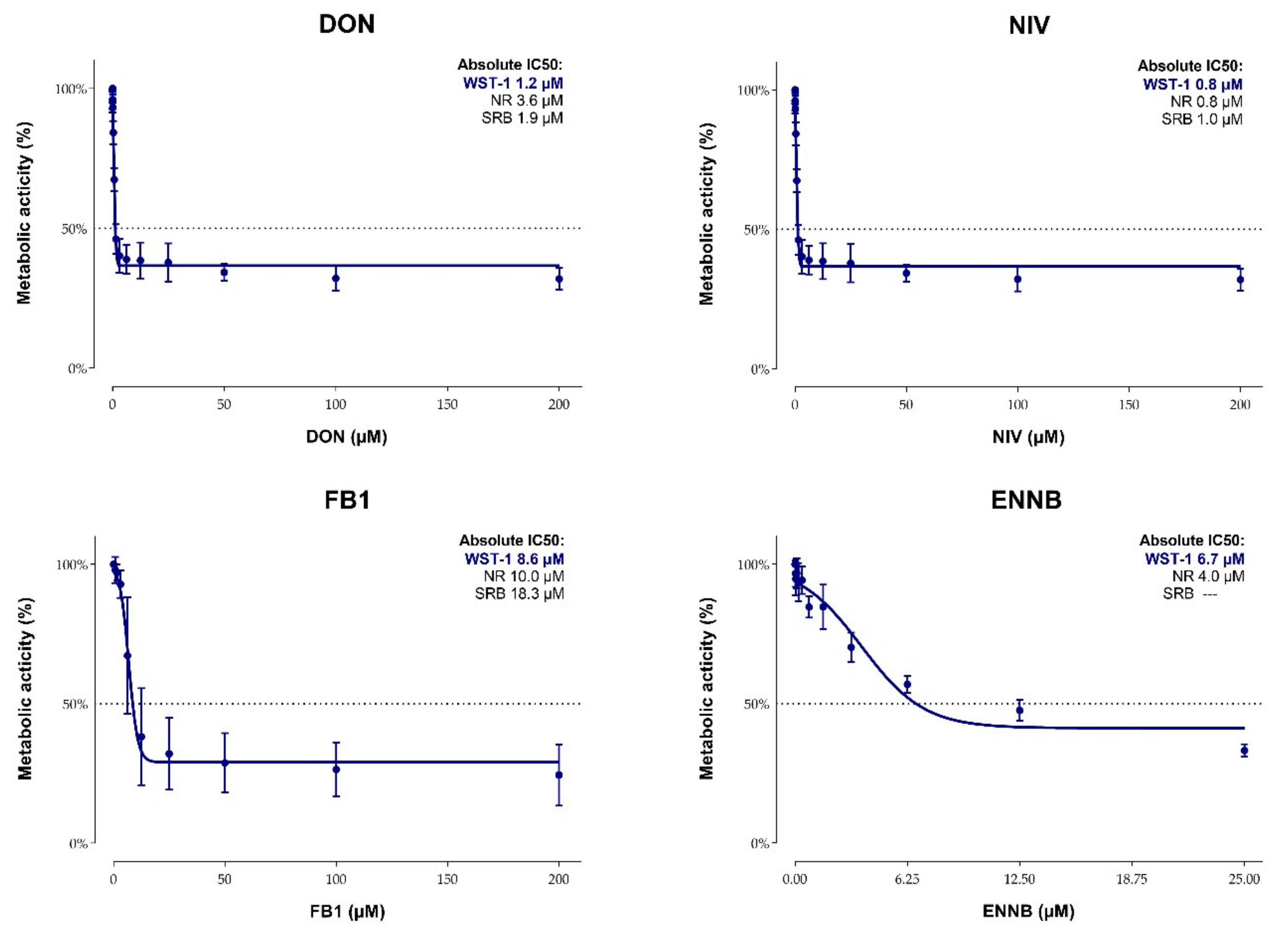
2.2. Cytotoxicity of Mycotoxins on Calf Small Intestinal Epithelial Cells
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Mycotoxin Survey
5.2. In vitro Experiments
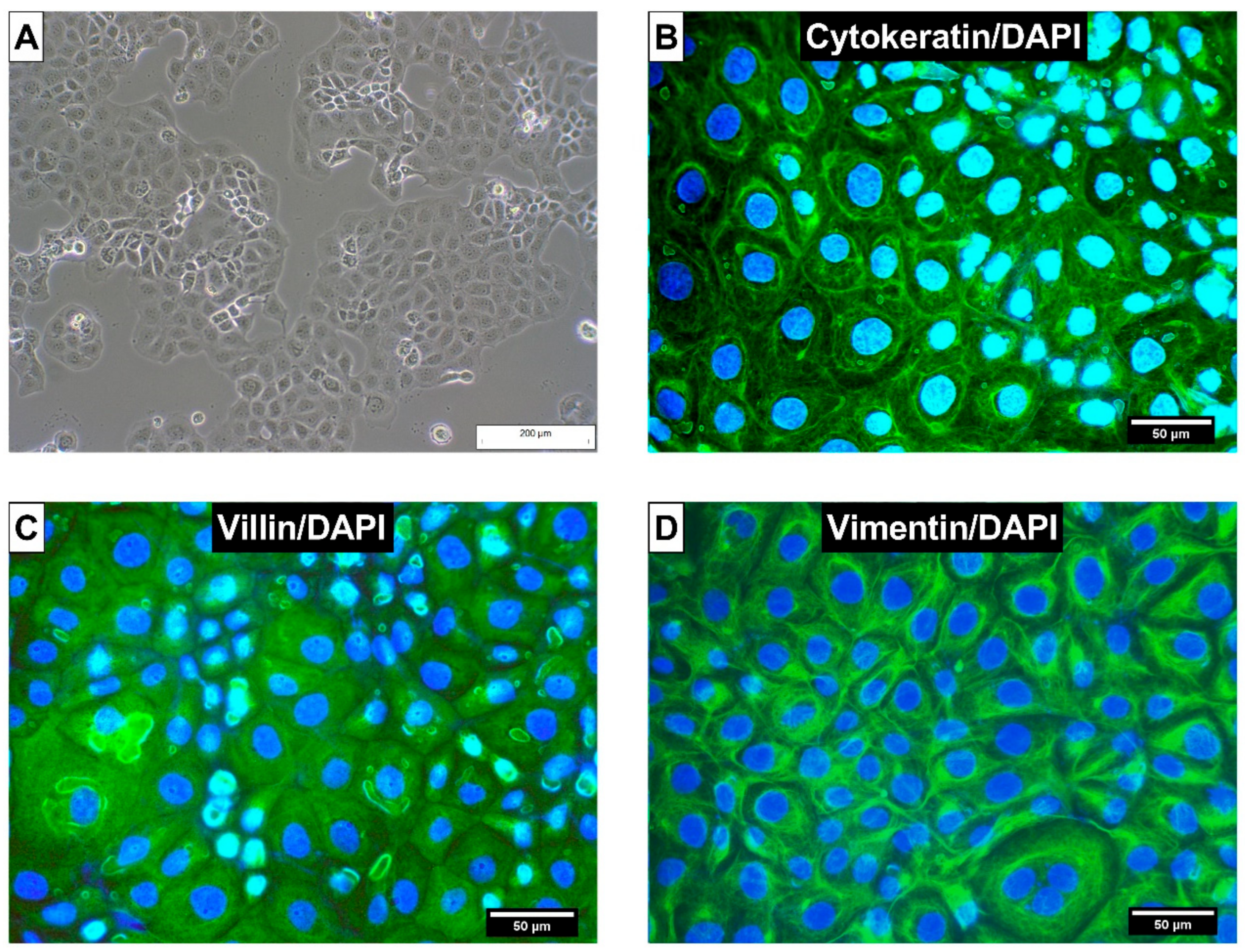
5.2.1. Cell Line
5.2.2. Routine Cultivation of CIEB
5.2.3. Characterization of CIEB by Immunohistochemistry (Cytokeration, Vimentin and Villin)
5.2.4. Cytotoxicity Tests
5.2.5. Sphinganine and Sphingosine Analysis
5.2.6. Data Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Bennett, J.W.; Klich, M. Mycotoxins. Clin. Microbiol. Rev. 2003, 16, 497–516. [Google Scholar] [CrossRef] [PubMed]
- Grenier, B.; Applegate, T. Modulation of intestinal functions following mycotoxin ingestion: Meta-analysis of published experiments in animals. Toxins 2013, 5, 396–430. [Google Scholar] [CrossRef] [PubMed]
- Robert, H.; Payros, D.; Pinton, P.; Théodorou, V.; Mercier-Bonin, M.; Oswald, I.P. Impact of mycotoxins on the intestine: Are mucus and microbiota new targets? J. Toxicol. Environ. Health B Crit. Rev. 2017, 20, 249–275. [Google Scholar] [CrossRef]
- European Commission. Directive 2002/32/EC of the European Parliament and of the Council of 7 May 2002 on undesirable substances in animal feed. Off. J. Eur. Union 2002, L140, 10–22. [Google Scholar]
- European Commission. Commission recommendation of of 17 August 2006 on the presence of deoxynivalenol, zearalenone, ochratoxin a, T-2 and Ht-2 and fumonisins in products intended for animal feeding. Off. J. Eur. Union 2006, L229, 7–9. [Google Scholar]
- Vaclavikova, M.; Malachova, A.; Veprikova, Z.; Dzuman, Z.; Zachariasova, M.; Hajslova, J. ‘Emerging’mycotoxins in cereals processing chains: Changes of enniatins during beer and bread making. Food Chem. 2013, 136, 750–757. [Google Scholar] [CrossRef]
- Gruber-Dorninger, C.; Novak, B.; Nagl, V.; Berthiller, F. Emerging mycotoxins: Beyond traditionally determined food contaminants. J. Agric. Food Chem. 2016. [Google Scholar] [CrossRef]
- Fink-Gremmels, J. Mycotoxins in cattle feeds and carry-over to dairy milk: A review. Food Addit. Contam. 2008, 25, 172–180. [Google Scholar] [CrossRef]
- Gallo, A.; Giuberti, G.; Frisvad, J.; Bertuzzi, T.; Nielsen, K. Review on mycotoxin issues in ruminants: Occurrence in forages, effects of mycotoxin ingestion on health status and animal performance and practical strategies to counteract their negative effects. Toxins 2015, 7, 3057–3111. [Google Scholar] [CrossRef]
- Driehuis, F.; Spanjer, M.; Scholten, J.; Te Giffel, M. Occurrence of mycotoxins in maize, grass and wheat silage for dairy cattle in the Netherlands. Food Addit. Contam. 2008, 1, 41–50. [Google Scholar] [CrossRef]
- Alonso, V.A.; Pereyra, C.M.; Keller, L.A.M.; Dalcero, A.M.; Rosa, C.; Chiacchiera, S.M.; Cavaglieri, L.R. Fungi and mycotoxins in silage: An overview. J. Appl. Microbiol. 2013, 115, 637–643. [Google Scholar] [CrossRef] [PubMed]
- Eurostat. Green Maize by Area, Production and Humidity. Available online: https://ec.europa.eu/eurostat/databrowser/view/tag00101/default/table?lang=en (accessed on 13 August 2019).
- Hedman, R.; Pettersson, H. Transformation of nivalenol by gastrointestinal microbes. Arch. Tierernahr. 1997, 50, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Seeling, K.; Dänicke, S.; Valenta, H.; Van Egmond, H.P.; Schothorst, R.C.; Jekel, A.A.; Lebzien, P.; Schollenberger, M.; Razzazi-Fazeli, E.; Flachowsky, G. Effects of fusarium toxin-contaminated wheat and feed intake level on the biotransformation and carry-over of deoxynivalenol in dairy cows. Food Addit. Contam. 2006, 23, 1008–1020. [Google Scholar] [CrossRef] [PubMed]
- Valgaeren, B.; Théron, L.; Croubels, S.; Devreese, M.; De Baere, S.; Van Pamel, E.; Daeseleire, E.; De Boevre, M.; De Saeger, S.; Vidal, A.; et al. The role of roughage provision on the absorption and disposition of the mycotoxin deoxynivalenol and its acetylated derivatives in calves: From field observations to toxicokinetics. Arch. Toxicol. 2018, 93, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Debevere, S.; De Baere, S.; Haesaert, G.; Rychlik, M.; Croubels, S.; Fievez, V. In vitro rumen simulations show a lower disappearance of deoxynivalenol, nivalenol, zearalenone and enniatin B at conditions of rumen acidosis and at dry conditions. Manuscript in preparation.
- Enemark, J.M. The monitoring, prevention and treatment of sub-acute ruminal acidosis (SARA): A review. Vet. J. 2008, 176, 32–43. [Google Scholar] [CrossRef]
- Prosperini, A.; Berrada, H.; Ruiz, M.J.; Caloni, F.; Coccini, T.; Spicer, L.J.; Perego, M.C.; Lafranconi, A. A review of the mycotoxin enniatin b. Front. Public Health 2017, 5, 304. [Google Scholar] [CrossRef] [PubMed]
- Caloni, F.; Spotti, M.; Auerbach, H.; den Camp, H.O.; Gremmels, J.F.; Pompa, G. In vitro metabolism of fumonisin b1 by ruminal microflora. Vet. Res. Commun. 2000, 24, 379–387. [Google Scholar] [CrossRef] [PubMed]
- Gurung, N.; Rankins, J.D.; Shelby, R. In vitro ruminal disappearance of fumonisin B1 and its effects on in vitro dry matter disappearance. Vet. Hum. Toxicol. 1999, 41, 196–199. [Google Scholar]
- European Commission. 2013/165/eu: Commission recommendation of 27 March 2013 on the presence of t-2 and ht-2 toxin in cereals and cereal products. Off. J. Eur. Union 2013, L91, 12–15. [Google Scholar]
- Jouany, J.P. Methods for preventing, decontaminating and minimizing the toxicity of mycotoxins in feeds. Anim. Feed Sci. Technol. 2007, 137, 342–362. [Google Scholar] [CrossRef]
- Driehuis, F.; Spanjer, M.; Scholten, J.; Te Giffel, M. Occurrence of mycotoxins in feedstuffs of dairy cows and estimation of total dietary intakes. J. Dairy Sci. 2008, 91, 4261–4271. [Google Scholar] [CrossRef] [PubMed]
- Gruber-Dorninger, C.; Jenkins, T.; Schatzmayr, G. Global mycotoxin occurrence in feed: A ten-year survey. Toxins 2019, 11, 375. [Google Scholar] [CrossRef] [PubMed]
- Storm, I.M.; Rasmussen, R.R.; Rasmussen, P.H. Occurrence of pre-and post-harvest mycotoxins and other secondary metabolites in Danish maize silage. Toxins 2014, 6, 2256–2269. [Google Scholar] [CrossRef] [PubMed]
- Panasiuk, L.; Jedziniak, P.; Pietruszka, K.; Piatkowska, M.; Bocian, L. Frequency and levels of regulated and emerging mycotoxins in silage in Poland. Mycotoxin Res. 2018, 35, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Dagnac, T.; Latorre, A.; Fernández Lorenzo, B.; Llompart, M. Validation and application of a liquid chromatography-tandem mass spectrometry based method for the assessment of the co-occurrence of mycotoxins in maize silages from dairy farms in nw spain. Food Addit. Contam Part A Chem. Anal. Control. Expos. Risk Asess. 2016, 33, 1850–1863. [Google Scholar] [CrossRef] [PubMed]
- Sørensen, J.L.; Nielsen, K.F.; Rasmussen, P.H.; Thrane, U. Development of a LC-MS/MS method for the analysis of enniatins and beauvericin in whole fresh and ensiled maize. J. Agric. Food Chem. 2008, 56, 10439–10443. [Google Scholar] [CrossRef] [PubMed]
- Fraeyman, S.; Croubels, S.; Devreese, M.; Antonissen, G. Emerging fusarium and alternaria mycotoxins: Occurrence, toxicity and toxicokinetics. Toxins 2017, 9, 228. [Google Scholar] [CrossRef]
- Springler, A.; Vrubel, G.-J.; Mayer, E.; Schatzmayr, G.; Novak, B. Effect of fusarium-derived metabolites on the barrier integrity of differentiated intestinal porcine epithelial cells (IPEC-J2). Toxins 2016, 8, 345. [Google Scholar] [CrossRef]
- Shimshoni, J.; Cuneah, O.; Sulyok, M.; Krska, R.; Galon, N.; Sharir, B.; Shlosberg, A. Mycotoxins in corn and wheat silage in Israel. Food Addit. Contam Part A Chem. Anal. Control. Expos. Risk Asess. 2013, 30, 1614–1625. [Google Scholar] [CrossRef]
- Woelflingseder, L.; Warth, B.; Vierheilig, I.; Schwartz-Zimmermann, H.; Hametner, C.; Nagl, V.; Novak, B.; Šarkanj, B.; Berthiller, F.; Adam, G. The fusarium metabolite culmorin suppresses the in vitro glucuronidation of deoxynivalenol. Arch. Toxicol. 2019, 93, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Latorre, A.; Dagnac, T.; Lorenzo, B.F.; Llompart, M. Occurrence and stability of masked fumonisins in corn silage samples. Food Chem. 2015, 189, 38–44. [Google Scholar] [CrossRef] [PubMed]
- Dall’Asta, C.; Falavigna, C.; Galaverna, G.; Dossena, A.; Marchelli, R. In vitro digestion assay for determination of hidden fumonisins in maize. J. Agric. Food Chem. 2010, 58, 12042–12047. [Google Scholar] [CrossRef] [PubMed]
- Kosicki, R.; Błajet-Kosicka, A.; Grajewski, J.; Twarużek, M. Multiannual mycotoxin survey in feed materials and feedingstuffs. Anim. Feed Sci. Technol. 2016, 215, 165–180. [Google Scholar] [CrossRef]
- Berthiller, F.; Crews, C.; Dall’Asta, C.; Saeger, S.D.; Haesaert, G.; Karlovsky, P.; Oswald, I.P.; Seefelder, W.; Speijers, G.; Stroka, J. Masked mycotoxins: A review. Mol. Nutr. Food Res. 2013, 57, 165–186. [Google Scholar] [CrossRef] [PubMed]
- Rasmussen, R.R.; Storm, I.M.L.D.; Rasmussen, P.H.; Smedsgaard, J.; Nielsen, K.F. Multi-mycotoxin analysis of maize silage by lc-ms/ms. Anal. Bioanal. Chem. 2010, 397, 765–776. [Google Scholar] [CrossRef]
- Winkler, J.; Kersten, S.; Meyer, U.; Engelhardt, U.; Dänicke, S. Residues of zearalenone (ZEN), deoxynivalenol (DON) and their metabolites in plasma of dairy cows fed fusarium contaminated maize and their relationships to performance parameters. Food Chem. Toxicol. 2014, 65, 196–204. [Google Scholar] [CrossRef] [PubMed]
- Dänicke, S.; Winkler, J.; Meyer, U.; Frahm, J.; Kersten, S. Haematological, clinical–chemical and immunological consequences of feeding fusarium toxin contaminated diets to early lactating dairy cows. Mycotoxin Res. 2017, 33, 1–13. [Google Scholar] [CrossRef]
- Cheli, F.; Giromini, C.; Baldi, A. Mycotoxin mechanisms of action and health impact: ‘In vitro’or ‘in vivo’ tests, that is the question. World Mycotoxin J. 2015, 8, 573–589. [Google Scholar] [CrossRef]
- Steube, K.G.; Koelz, A.-L.; Uphoff, C.C.; Drexler, H.G.; Kluess, J.; Steinberg, P. The necessity of identity assessment of animal intestinal cell lines: A case report. Cytotechnology 2012, 64, 373–378. [Google Scholar] [CrossRef]
- International Cell Line Authentication Committee. Register of Misidentified Cell Lines. Available online: https://iclac.org/databases/cross-contaminations/ (accessed on 13 August 2019).
- Chatterjee, R. Cell biology: Cases of mistaken identity. Science 2007, 16, 928–931. [Google Scholar] [CrossRef] [PubMed]
- Ordóñez, N.G. Broad-Spectrum immunohistochemical epithelial markers: A review. Hum. Pathol. 2013, 44, 1195–1215. [Google Scholar] [CrossRef] [PubMed]
- George, S.P.; Wang, Y.; Mathew, S.; Srinivasan, K.; Khurana, S. Dimerization and actin-bundling properties of villin and its role in the assembly of epithelial cell brush borders. J. Biol. Chem. 2007, 282, 26528–26541. [Google Scholar] [CrossRef] [PubMed]
- Miyazawa, K.; Hondo, T.; Kanaya, T.; Tanaka, S.; Takakura, I.; Itani, W.; Rose, M.T.; Kitazawa, H.; Yamaguchi, T.; Aso, H. Characterization of newly established bovine intestinal epithelial cell line. Histochem. Cell Biol. 2010, 133, 125–134. [Google Scholar] [CrossRef] [PubMed]
- Chang, Y.; Li, H.; Guo, Z. Mesenchymal stem cell-like properties in fibroblasts. Cell Physiol. Biochem. 2014, 34, 703–714. [Google Scholar] [CrossRef]
- Zakrzewski, S.S.; Richter, J.F.; Krug, S.M.; Jebautzke, B.; Lee, I.-F.M.; Rieger, J.; Sachtleben, M.; Bondzio, A.; Schulzke, J.D.; Fromm, M. Improved cell line IPEC-J2, characterized as a model for porcine jejunal epithelium. PLoS ONE 2013, 8, e79643. [Google Scholar] [CrossRef] [PubMed]
- Loret, S.; Rusu, D.; El Moualij, B.; Taminiau, B.; Heinen, E.; Dandrifosse, G.; Mainil, J. Preliminary characterization of jejunocyte and colonocyte cell lines isolated by enzymatic digestion from adult and young cattle. Res. Vet. Sci. 2009, 87, 123–132. [Google Scholar] [CrossRef]
- Nielsen, C.; Casteel, M.; Didier, A.; Dietrich, R.; Märtlbauer, E. Trichothecene-induced cytotoxicity on human cell lines. Mycotoxin Res. 2009, 25, 77–84. [Google Scholar] [CrossRef]
- Alassane-Kpembi, I.; Puel, O.; Oswald, I.P. Toxicological interactions between the mycotoxins deoxynivalenol, nivalenol and their acetylated derivatives in intestinal epithelial cells. Arch. Toxicol. 2014, 89, 1–10. [Google Scholar] [CrossRef]
- Wan, L.Y.M.; Turner, P.C.; El-Nezami, H. Individual and combined cytotoxic effects of fusarium toxins (deoxynivalenol, nivalenol, zearalenone and fumonisins B1) on swine jejunal epithelial cells. Food Chem. Toxicol. 2013, 57, 276–283. [Google Scholar] [CrossRef]
- Dänicke, S.; Hegewald, A.-K.; Kahlert, S.; Kluess, J.; Rothkötter, H.-J.; Breves, G.; Döll, S. Studies on the toxicity of deoxynivalenol (DON), sodium metabisulfite, don-sulfonate (DONS) and de-epoxy-don for porcine peripheral blood mononuclear cells and the intestinal porcine epithelial cell lines IPEC-J1 and IPEC-J2, and on effects of don and dons on piglets. Food Chem. Toxicol. 2010, 48, 2154–2162. [Google Scholar] [PubMed]
- Vejdovszky, K.; Warth, B.; Sulyok, M.; Marko, D. Non-synergistic cytotoxic effects of fusarium and alternaria toxin combinations in caco-2 cells. Toxicol. Lett. 2016, 241, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Springler, A.; Hessenberger, S.; Reisinger, N.; Kern, C.; Nagl, V.; Schatzmayr, G.; Mayer, E. Deoxynivalenol and its metabolite deepoxy-deoxynivalenol: Multi-parameter analysis for the evaluation of cytotoxicity and cellular effects. Mycotoxin Res. 2017, 33, 25–37. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Blanco, C.; Font, G.; Ruiz, M.-J. Interaction effects of enniatin B, deoxinivalenol and alternariol in Caco-2 cells. Toxicol. Lett. 2016, 241, 38–48. [Google Scholar] [CrossRef] [PubMed]
- Ivanova, L.; Egge-Jacobsen, W.; Solhaug, A.; Thoen, E.; Fæste, C. Lysosomes as a possible target of enniatin b-induced toxicity in Caco-2 cells. Chem. Res. Toxicol. 2012, 25, 1662–1674. [Google Scholar] [CrossRef] [PubMed]
- Prosperini, A.; Juan-García, A.; Font, G.; Ruiz, M. Reactive oxygen species involvement in apoptosis and mitochondrial damage in caco-2 cells induced by enniatins A, A1, B and B1. Toxicol. Lett. 2013, 222, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Tonshin, A.A.; Teplova, V.V.; Andersson, M.A.; Salkinoja-Salonen, M.S. The fusarium mycotoxins enniatins and beauvericin cause mitochondrial dysfunction by affecting the mitochondrial volume regulation, oxidative phosphorylation and ion homeostasis. Toxicology 2010, 276, 49–57. [Google Scholar] [CrossRef] [PubMed]
- Wentzel, J.F.; Lombard, M.J.; Du Plessis, L.H.; Zandberg, L. Evaluation of the cytotoxic properties, gene expression profiles and secondary signalling responses of cultured cells exposed to fumonisin B1, deoxynivalenol and zearalenone mycotoxins. Arch. Toxicol. 2017, 91, 2265–2282. [Google Scholar] [CrossRef] [PubMed]
- Creppy, E.E.; Chiarappa, P.; Baudrimont, I.; Borracci, P.; Moukha, S.; Carratù, M.R. Synergistic effects of fumonisin B1 and ochratoxin A: Are in vitro cytotoxicity data predictive of in vivo acute toxicity? Toxicology 2004, 201, 115–123. [Google Scholar] [CrossRef]
- Kouadio, J.H.; Mobio, T.A.; Baudrimont, I.; Moukha, S.; Dano, S.D.; Creppy, E.E. Comparative study of cytotoxicity and oxidative stress induced by deoxynivalenol, zearalenone or fumonisin B1 in human intestinal cell line Caco-2. Toxicology 2005, 213, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Sobral, M.M.C.; Faria, M.A.; Cunha, S.C.; Ferreira, I.M. Toxicological interactions between mycotoxins from ubiquitous fungi: Impact on hepatic and intestinal human epithelial cells. Chemosphere 2018, 202, 538–548. [Google Scholar] [CrossRef] [PubMed]
- Bouhet, S.; Hourcade, E.; Loiseau, N.; Fikry, A.; Martinez, S.; Roselli, M.; Galtier, P.; Mengheri, E.; Oswald, I.P. The mycotoxin fumonisin B1 alters the proliferation and the barrier function of porcine intestinal epithelial cells. Toxicol. Sci. 2004, 77, 165–171. [Google Scholar] [CrossRef] [PubMed]
- Bouhet, S.; Oswald, I.P. The intestine as a possible target for fumonisin toxicity. Mol. Nutr. Food Res. 2007, 51, 925–931. [Google Scholar] [CrossRef] [PubMed]
- Loiseau, N.; Debrauwer, L.; Sambou, T.; Bouhet, S.; Miller, J.D.; Martin, P.G.; Viadère, J.-L.; Pinton, P.; Puel, O.; Pineau, T. Fumonisin b1 exposure and its selective effect on porcine jejunal segment: Sphingolipids, glycolipids and trans-epithelial passage disturbance. Biochem. Pharmacol. 2007, 74, 144–152. [Google Scholar] [CrossRef]
- Grenier, B.; Schwartz-Zimmermann, H.E.; Caha, S.; Moll, W.D.; Schatzmayr, G.; Applegate, T.J. Dose-dependent effects on sphingoid bases and cytokines in chickens fed diets prepared with fusarium verticillioides culture material containing fumonisins. Toxins 2015, 7, 1253–1272. [Google Scholar] [CrossRef]
- Fink-Gremmels, J.; Malekinejad, H. Clinical effects and biochemical mechanisms associated with exposure to the mycoestrogen zearalenone. Anim. Feed Sci. Technol. 2007, 137, 326–341. [Google Scholar] [CrossRef]
- Liew, W.-P.-P.; Mohd-Redzwan, S. Mycotoxin: Its impact on gut health and microbiota. Front. Cell. Infect. Microbiol. 2018, 8, 60. [Google Scholar] [CrossRef]
- Cetin, Y.; Bullerman, L.B. Cytotoxicity of fusarium mycotoxins to mammalian cell cultures as determined by the mtt bioassay. Food Chem. Toxicol. 2005, 43, 755–764. [Google Scholar] [CrossRef]
- Kovalsky Paris, M.P.; Schweiger, W.; Hametner, C.; Stückler, R.; Muehlbauer, G.J.; Varga, E.; Krska, R.; Berthiller, F.; Adam, G. Zearalenone-16-O-glucoside: A new masked mycotoxin. J. Agric. Food Chem. 2014, 62, 1181–1189. [Google Scholar] [CrossRef]
- Malachová, A.; Sulyok, M.; Beltrán, E.; Berthiller, F.; Krska, R. Optimization and validation of a quantitative liquid chromatography—Tandem mass spectrometric method covering 295 bacterial and fungal metabolites including all regulated mycotoxins in four model food matrices. J. Chromatogr. 2014, 1362, 145–156. [Google Scholar] [CrossRef]
- Malachová, A.; Sulyok, M.; Beltran, E.; Berthiller, F.; Krska, R. Multi-toxin determination in food-the power of “dilute and shoot” approaches in LC-MS/MS. LC GC Eur. 2015, 28, 542–555. [Google Scholar]
- Reisinger, N.; Dohnal, I.; Nagl, V.; Schaumberger, S.; Schatzmayr, G.; Mayer, E. Fumonisin B1 (FB1) induces lamellar separation and alters sphingolipid metabolism of in vitro cultured hoof explants. Toxins 2016, 8, 89. [Google Scholar] [CrossRef] [PubMed]
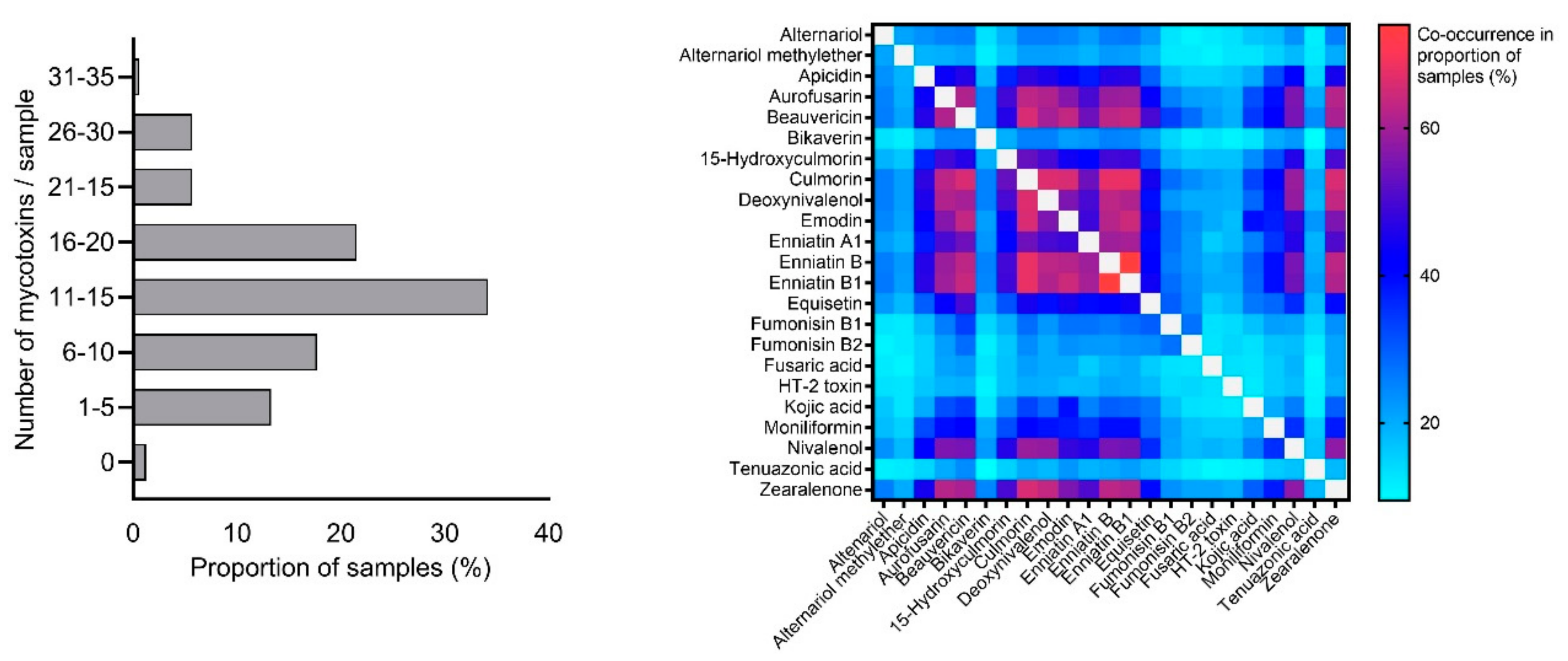
| Mycotoxin | Positive Samples 1 (n) | Positive Samples 1 (%) | Median Concentration 2 (µg/kg) | 75th Percentile 2 (µg/kg) | 95th Percentile2 (µg/kg) | Maximum Concentration (µg/kg) |
|---|---|---|---|---|---|---|
| Regulated mycotoxins (except ergot alkaloids) 3 | ||||||
| Aflatoxin B1 4 | 0 | 0.0 | - | - | - | - |
| Deoxynivalenol | 107 | 67.7 | 303 | 556 | 1490 | 3060 |
| Fumonisin B1 | 55 | 34.8 | 60.0 | 147 | 262 | 553 |
| Fumonisin B2 | 46 | 29.1 | 20.4 | 34.4 | 101 | 133 |
| Ochratoxin A | 4 | 2.5 | 2.38 | 2.51 | 2.62 | 2.65 |
| Zearalenone | 107 | 67.7 | 15.2 | 61 | 1110 | 1670 |
| Ergot alkaloids 3 | ||||||
| Ergine | 0 | 0.0 | - | - | - | - |
| Ergocornine | 0 | 0.0 | - | - | - | - |
| Ergocorninin | 0 | 0.0 | - | - | - | - |
| Ergocristine | 0 | 0.0 | - | - | - | - |
| Ergocristinine | 0 | 0.0 | - | - | - | - |
| Ergocryptine | 2 | 1.3 | 5.41 | 7.77 | 9.65 | 10.12 |
| Ergocryptinine | 0 | 0.0 | - | - | - | - |
| Ergometrine | 1 | 0.6 | 49.6 | 49.6 | 49.6 | 49.6 |
| Ergometrinine | 1 | 0.6 | 3.20 | 3.20 | 3.20 | 3.20 |
| Ergosin | 2 | 1.3 | 1.89 | 1.90 | 1.91 | 1.91 |
| Ergosinin | 0 | 0.0 | - | - | - | - |
| Ergotamine | 1 | 0.6 | 1.54 | 1.54 | 1.54 | 1.54 |
| Ergotaminine | 0 | 0.0 | - | - | - | - |
| Ergovalin | 0 | 0.0 | - | - | - | - |
| Type-A trichothecenes | ||||||
| Diacetoxyscirpenol | 0 | 0.0 | - | - | - | - |
| HT-2 toxin | 34 | 21.5 | 14.7 | 21.4 | 51.9 | 90.2 |
| Monoacetoxyscirpenol | 4 | 2.5 | 9.91 | 15.9 | 29.5 | 32.9 |
| Neosolaniol | 0 | 0.0 | - | - | - | - |
| T-2 toxin | 6 | 3.8 | 2.55 | 2.89 | 3.79 | 4.08 |
| Type-B trichothecenes | ||||||
| 3-Acetyldeoxynivalenol | 0 | 0.0 | - | - | - | - |
| 15-Acetyldeoxynivalenol | 8 | 5.1 | 274 | 480 | 624 | 687 |
| Nivalenol | 94 | 59.5 | 113 | 237 | 623 | 5770 |
| Modified mycotoxins | ||||||
| Deoxynivalenol-3-glucoside | 40 | 25.3 | 17.1 | 49.2 | 121 | 129 |
| HT-2-toxin-3-glucoside 5 | 1 | 0.6 | 6.28 | 6.28 | 6.28 | 6.28 |
| Nivalenol-3-glucoside 5 | 5 | 3.2 | 6.01 | 6.06 | 9.68 | 10.6 |
| α-zearalenol | 12 | 7.6 | 4.84 | 6.93 | 18.1 | 22.2 |
| β-zearalenol | 8 | 5.1 | 4.90 | 6.66 | 12.4 | 12.6 |
| Emerging mycotoxins | ||||||
| Alternariol | 45 | 28.5 | 3.11 | 4.45 | 12.1 | 48.1 |
| Alternariol methylether | 37 | 23.4 | 1.95 | 3.46 | 5.79 | 30.8 |
| Apicidin | 79 | 50.0 | 9.49 | 25.0 | 102 | 175 |
| Aurofusarin | 108 | 68.4 | 97.8 | 307 | 3840 | 4710 |
| Beauvericin | 120 | 76.0 | 9.16 | 19.0 | 75.9 | 214 |
| Bikaverin | 42 | 26.6 | 20.3 | 58.8 | 248 | 415 |
| Butenolid | 30 | 19.0 | 28.9 | 70.9 | 249 | 583 |
| Culmorin | 125 | 79.1 | 190 | 719 | 2930 | 6680 |
| 5-Hydroxyculmorin | 19 | 12.0 | 571 | 989 | 1400 | 1480 |
| 15-Hydroxyculmorin | 84 | 53.2 | 229 | 504 | 1520 | 1670 |
| 15-Hydroxyculmoron | 22 | 13.9 | 204 | 396 | 441 | 484 |
| Emodin | 131 | 82.9 | 4.38 | 14.1 | 211 | 1640 |
| Enniatin A | 30 | 19.0 | 2.45 | 5.23 | 32.5 | 50.1 |
| Enniatin A1 | 98 | 62.0 | 2.70 | 8.73 | 25.2 | 173.9 |
| Enniatin B | 121 | 76.6 | 7.07 | 13.8 | 47.4 | 429 |
| Enniatin B1 | 124 | 78.5 | 5.68 | 15.5 | 46.7 | 555 |
| Enniatin B2 | 8 | 5.1 | 3.40 | 5.49 | 16.0 | 20.7 |
| Enniatin B3 | 0 | 0.0 | - | - | - | - |
| Equisetin | 86 | 54.4 | 4.75 | 8.42 | 17.4 | 45.4 |
| Fusaproliferin | 4 | 2.5 | 170 | 286 | 316 | 322.3 |
| Fusaric acid | 35 | 22.2 | 229 | 998 | 1800 | 4120 |
| Kojic acid | 67 | 42.4 | 96.3 | 185 | 876 | 25,930 |
| Moniliformin | 71 | 44.9 | 7.84 | 18.5 | 61.6 | 113 |
| Mycophenolic Acid | 9 | 5.7 | 14.8 | 80 | 262 | 352 |
| Roquefortine C | 7 | 4.4 | 11.7 | 21.3 | 326 | 454 |
| Sterigmatocystin | 3 | 1.9 | 2.38 | 5.89 | 8.65 | 9.35 |
| Tenuazonic acid | 42 | 26.6 | 60.6 | 182 | 574 | 727 |
| FB1 (µM) | Sa (ng/mL) | So (ng/mL) | Sa/So |
|---|---|---|---|
| 0 | 0.21 ± 0.81 a | 1.40 ± 0.40 | 0.15 ± 0.02 a |
| 0.781 | 0.28 ± 0.10 a | 1.42 ± 0.33 | 0.20 ± 0.03 a |
| 1.563 | 0.53 ± 0.25 a | 1.70 ± 0.47 | 0.31 ± 0.06 a |
| 3.125 | 3.89 ± 1.51 a | 1.61 ± 0.25 | 2.36 ± 0.55 a |
| 6.25 | 15.60 ± 5.91 a | 1.53 ± 0.25 | 9.98 ± 2.20 b |
| 12.5 | 33.61 ± 13.29 a | 1.96 ± 0.64 | 16.96 ± 2.11 b |
| 25 | 44.57 ± 17.19 b | 2.36 ± 1.00 | 19.11 ± 1.71 b |
| 50 | 50.64 ± 25.01 b | 2.49 ± 1.11 | 20.08 ± 0.86 b |
| 100 | 53.57 ± 23.27 b | 2.29 ± 0.90 | 23.25 ± 1.15 b |
| 200 | 56.13 ± 26.26 b | 2.16 ± 0.83 | 25.58 ± 2.11 b |
| p-value | <0.0001 | 0.0462 | <0.0001 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reisinger, N.; Schürer-Waldheim, S.; Mayer, E.; Debevere, S.; Antonissen, G.; Sulyok, M.; Nagl, V. Mycotoxin Occurrence in Maize Silage—A Neglected Risk for Bovine Gut Health? Toxins 2019, 11, 577. https://doi.org/10.3390/toxins11100577
Reisinger N, Schürer-Waldheim S, Mayer E, Debevere S, Antonissen G, Sulyok M, Nagl V. Mycotoxin Occurrence in Maize Silage—A Neglected Risk for Bovine Gut Health? Toxins. 2019; 11(10):577. https://doi.org/10.3390/toxins11100577
Chicago/Turabian StyleReisinger, Nicole, Sonja Schürer-Waldheim, Elisabeth Mayer, Sandra Debevere, Gunther Antonissen, Michael Sulyok, and Veronika Nagl. 2019. "Mycotoxin Occurrence in Maize Silage—A Neglected Risk for Bovine Gut Health?" Toxins 11, no. 10: 577. https://doi.org/10.3390/toxins11100577
APA StyleReisinger, N., Schürer-Waldheim, S., Mayer, E., Debevere, S., Antonissen, G., Sulyok, M., & Nagl, V. (2019). Mycotoxin Occurrence in Maize Silage—A Neglected Risk for Bovine Gut Health? Toxins, 11(10), 577. https://doi.org/10.3390/toxins11100577