Safety of Corn and Corn-Based Products Intended for Human Consumption Concerning Fumonisins from a Brazilian Processing Plant
Abstract
1. Introduction
2. Results and Discussion
3. Material and Methods
3.1. Sampling
3.2. Fumonisin Determination
3.3. Food Consumption Data
3.4. Estimation of Fumonisin Probable Daily Intake
3.5. Statistical Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Agência Brasileira de Milho (ABIMILHO). O cereal que enriquece a alimentação humana. 2016. Available online: http://www.abimilho.com.br/milho/cereal (accessed on 12 January 2018).
- Companhia Nacional de Abastecimento (CONAB). Acompanhamento da safra brasileira de grãos, v. 5—Safra 2017/18, n. 4—Quarto levantamento, janeiro 2018. 2018. Available online: http://www.conab.gov.br/OlalaCMS/uploads/arquivos/18_01_11_14_17_49_graos_4o_levantamento (accessed on 12 January 2018).
- Council for Agricultural Science and Technology (CAST). Mycotoxins: Risks in Plant, Animal and Human Systems; Task Force Report; Council for Agricultural Science and Technology (CAST): Ames, IA, USA, 2003. [Google Scholar]
- Chuturgoon, A.; Phulukdaree, A.; Moodley, D. Fumonisin B1 induces global DNA hypomethylation in HepG2 cells—An alternative mechanism of action. Toxicology 2014, 315, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Marshall, A.-L.; Venuti, D.J.; Eastman, D.J. Fumonisin exposure in Guatemalan women of child-bearing age: A potential link to the observed high incidence of frontoethmoidal encephalocele. Ann. Glob. Health 2017, 83, 3–11. [Google Scholar] [CrossRef]
- Wang, S.-K.; Wang, T.-T.; Huang, G.-L.; Shi, R.-F.; Yang, L.-G.; Sun, G.-J. Stimulation of the proliferation of human normal esophageal epithelial cells by fumonisin B1 and its mechanism. Exp. Ther. Med. 2014, 7, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Szabó, A.; Szabó-Fodor, J.; Fébel, H.; Mézes, M.; Repa, I.; Kovács, M. Acute hepatic effects of low-dose fumonisin B1 in rats. Acta Vet. Hung. 2016, 64, 436–448. [Google Scholar] [CrossRef] [PubMed]
- Vendruscolo, C.P.; Frias, N.C.; Carvalho, C.B.; Sá, L.R.M.; Belli, C.B.; Baccarin, R.Y.A. Leukoencephalomalacia outbreak in horses due to consumption of contaminated hay. J. Vet. Intern. Med. 2016, 30, 1879–1881. [Google Scholar] [CrossRef] [PubMed]
- International Agency for Research on Cancer (IARC). Some Traditional Herbal Medicines, Some Mycotoxins, Naphthalene and Styrene IARC Monographs on the Evaluation of Carcinogenic Risks to Humans; International Agency for Research on Cancer (IARC): Lyon, France, 2002; Volume 82. [Google Scholar]
- World and Health Organization (WHO). Fumonisins. Evaluations of the Joint FAO/WHO Expert Committee on Food Additives (JECFA); World and Health Organization (WHO): Geneva, Switzerland, 2011. [Google Scholar]
- Bullerman, L.B.; Bianchini, A. Good food- processing techniques: Stability of mycotoxins in processed maize-based foods. In Mycotoxin Reduction in Grain Chains; Leslie, J.F., Logrieco, A., Eds.; John Wiley and Sons, Inc.: Ames, IA, USA, 2014; pp. 92–97. [Google Scholar]
- SCOOP—Scientific Cooperation Task of the European Commission. Collection of Occurrence data of Fusarium Toxins in Food and Assessment of Dietary Intake by the Population of EU Member States; Final Report; Directorate—General Health and Consumer Protection; European Commission: Brussels, Belgium, 2003. [Google Scholar]
- International Programme on Chemical Safety (IPCS). Principles and Methods for the Risk Assessment of Chemicals in Food; Environmental Health Criteria: Stuttgart, Germany, 2009. [Google Scholar]
- European Food Safety Authority (EFSA). Risk assessment of contaminants in food and feed. EFSA J. 2012, 10, 1–12. [Google Scholar]
- European Food Safety Authority (EFSA). International frameworks dealing with human risk assessment of combined exposure to multiple chemicals. EFSA J. 2013, 11, 1–69. [Google Scholar]
- World and Health Organization (WHO). WHO Human Health Risk Assessment Toolkit: Chemical Hazards; World and Health Organization (WHO): Geneva, Switzerland, 2010. [Google Scholar]
- Bordin, K.; Rosim, R.E.; Neef, D.V.; Rottinghaus, G.E.; Oliveira, C.A.F. Assessment of dietary intake of fumonisin B1 in São Paulo, Brazil. Food Chem. 2014, 155, 174–178. [Google Scholar] [CrossRef]
- Caldas, E.D.; Silva, A.C.S. Mycotoxins in corn-based food products consumed in Brazil: An exposure assessment for fumonisins. J. Agric. Food Chem. 2007, 55, 7974–7980. [Google Scholar] [CrossRef]
- Martins, F.A.; Ferreira, F.M.D.; Ferreira, F.D.; Bando, E.; Nerilo, S.B.; Hirooka, E.Y.; Machinski, M., Jr. Daily intake estimates of fumonisins in corn-based food products in the population of Parana, Brazil. Food Control 2012, 26, 614–618. [Google Scholar] [CrossRef]
- Marín, S.; Homedes, V.; Sanchis, V.; Ramos, A.J.; Magan, N. Impact of Fusarium moniliforme and F. proliferatum colonisation of maize on calorific losses and fumonisin production under different environmental conditions. J. Stored Prod. Res. 1999, 35, 15–26. [Google Scholar] [CrossRef]
- Mogensen, J.M.; Nielsen, K.F.; Samson, R.A.; Frisvad, J.C.; Thrane, U. Effect of temperature and water activity on the production of fumonisins by Aspergillus niger and different Fusarium species. BMC Microbiol. 2009, 9, 281. [Google Scholar] [CrossRef] [PubMed]
- Pitt, J.I.; Hocking, A.D. The ecology of food fungal spoilage. In Fungi and Food Spoilage, 3rd ed.; Pitt, J.I., Hocking, A.D., Eds.; Springer: London, UK, 2009; pp. 3–10. [Google Scholar]
- Leistner, L.; Rödel, W. The Significance of Water Activity for Micro-Organisms in Meats; Duckworth, R.B., Ed.; Academic Press Inc.: London, UK, 2012; pp. 309–324. [Google Scholar]
- Aldred, D.; Olsen, M.; Magan, N. The use of HACCP in the control of mycotoxins. In Mycotoxin in Food: Detection and Control; Magan, N., Olsen, M., Eds.; Woodhead Publishing: Cambridge, UK, 2004; pp. 139–173. [Google Scholar]
- Oliveira, M.S.; Rocha, A.; Sulyok, M.; Krska, R.; Mallmann, C.A. Natural mycotoxin contamination of maize (Zea mays L.) in the South region of Brazil. Food Control 2017, 73, 127–132. [Google Scholar] [CrossRef]
- Ono, E.Y.S.; Silva, M.; Hashimoto, E.H.; Vizoni, E.; Kawamura, O.; Sugiura, Y.; Hirooka, E.Y. Mycotoxicological quality evaluation of corn samples used by processing industries in the Northern region of Paraná State, Brazil. Food Addit. Contam. 2008, 25, 1392–1399. [Google Scholar] [CrossRef] [PubMed]
- European Commission. Commission Regulation (EC) No. 1126/2007, of 28 September 2007 amending Regulation (EC) No 1881/2006 setting maximum levels for certain contaminants in foodstuffs as regards Fusarium toxins in maize and maize products. Off. J. Eur. Union 2007, 255, 14–17. [Google Scholar]
- Brasil. Resolução RDC n°7, 18 de fevereiro de 2011. In A Agência Nacional de Vigilância Sanitária Aprova o Regulamento técnico sobre Limites máximos Tolerados para Micotoxinas em Alimentos; Diário Oficial da União da República Federativa do Brasil: Brasília, Distrito Federal, Brasil, 2011; p. 72. [Google Scholar]
- Bordini, J.G.; Ono, M.A.; Garcia, G.T.; Fazani, V.H.F.; Vizoni, E.; Rodrigues, K.C.B.; Hirooka, E.Y.; Ono, E.Y.S. Impact of industrial dry-milling on fumonisin redistribution in non-transgenic corn in Brazil. Food Chem. 2017, 220, 438–443. [Google Scholar] [CrossRef] [PubMed]
- Brera, C.; Debegnach, F.; Grossi, S.; Miraglia, M. Effect of industrial processing on the distribution of fumonisin B1 in dry-milling corn fractions. J. Food Prot. 2004, 67, 1261–1266. [Google Scholar] [CrossRef] [PubMed]
- Castells, M.; Marín, S.; Sanchis, V.; Ramos, A.J. Distribution of fumonisins and aflatoxins in corn fraction during industrial cornflake processing. Int. J. Food Microbiol. 2008, 123, 81–87. [Google Scholar] [CrossRef]
- Caldas, E.D.; Jardim, A.N.O. Exposure to toxic chemicals in the diet: Is the Brazilian population at risk? J. Expo. Sci. Environ. Epidemiol. 2012, 22, 1–15. [Google Scholar] [CrossRef]
- Instituto Brasileiro de Geografia e Estatística (IBGE). Pesquisa de Orçamentos Familiares 2008–2009, Aquisição Alimentar Domiciliar Per Capita no Brasil; Instituto Brasileiro de Geografia e Estatística (IBGE): Rio de Janeiro, Brasil, 2011. [Google Scholar]
- European Food Safety Authority (EFSA). Management of left-censored data in dietary exposure assessment of chemical substances. EFSA J. 2010, 8, 1–96. [Google Scholar]
- International Programme on Chemical Safety (IPCS/GEMS). Reliable Evaluation of Low-Level Contamination of Food; Workshop in the Frame of GEMS/Food-Euro, Appendix 5; IPCS/GEMS: Kulmbach, Germany, 1995. [Google Scholar]
- Food and Agriculture Organization of the United Nations (FAO). Food Security. Indicators from Household Surveys (Gender, Area, Socioeconomics). 2013. Available online: http://www.fao.org/faostat/en/#data/HS (accessed on 25 May 2017).
- Shephard, G.S.; Marasas, W.F.O.; Burger, H.M.; Somdyala, N.I.M.; Rheeder, J.P.; Van der Westhuizen, L.; Gatyeni, P.; Van Schalkwyk, D.J. Exposure assessment for fumonisins in the former Transkei region of South Africa. Food Addit. Contam. 2007, 24, 621–629. [Google Scholar] [CrossRef] [PubMed]
- Shephard, G.S.; Sydenham, E.W.; Thiel, P.G.; Gelderblom, W.C.A. Quantitative determination of fumonisins B1 and B2 by high performance liquid chromatography with fluorescence detection. J. Liq. Chromatogr. 1990, 13, 2077–2087. [Google Scholar] [CrossRef]
- Ueno, Y.; Aoyama, S.; Sugiura, Y.; Wang, D.S.; Lee, U.S.; Hirooka, E.Y.; Yu, S.-Z. A limited survey of fumonisins in corn and corn-based products in Asian countries. Mycotoxin Res. 1993, 9, 27–34. [Google Scholar] [CrossRef]
- Bissoqui, L.Y.; Frehse, M.S.; Freire, R.L.; Ono, M.A.; Bordini, J.G.; Hirozawa, M.T.; Oliveira, A.J.; Ono, E.Y.S. Exposure assessment of dogs to mycotoxins through consumption of dry feed. J. Sci. Food Agric. 2016, 6, 4135–4142. [Google Scholar] [CrossRef] [PubMed]
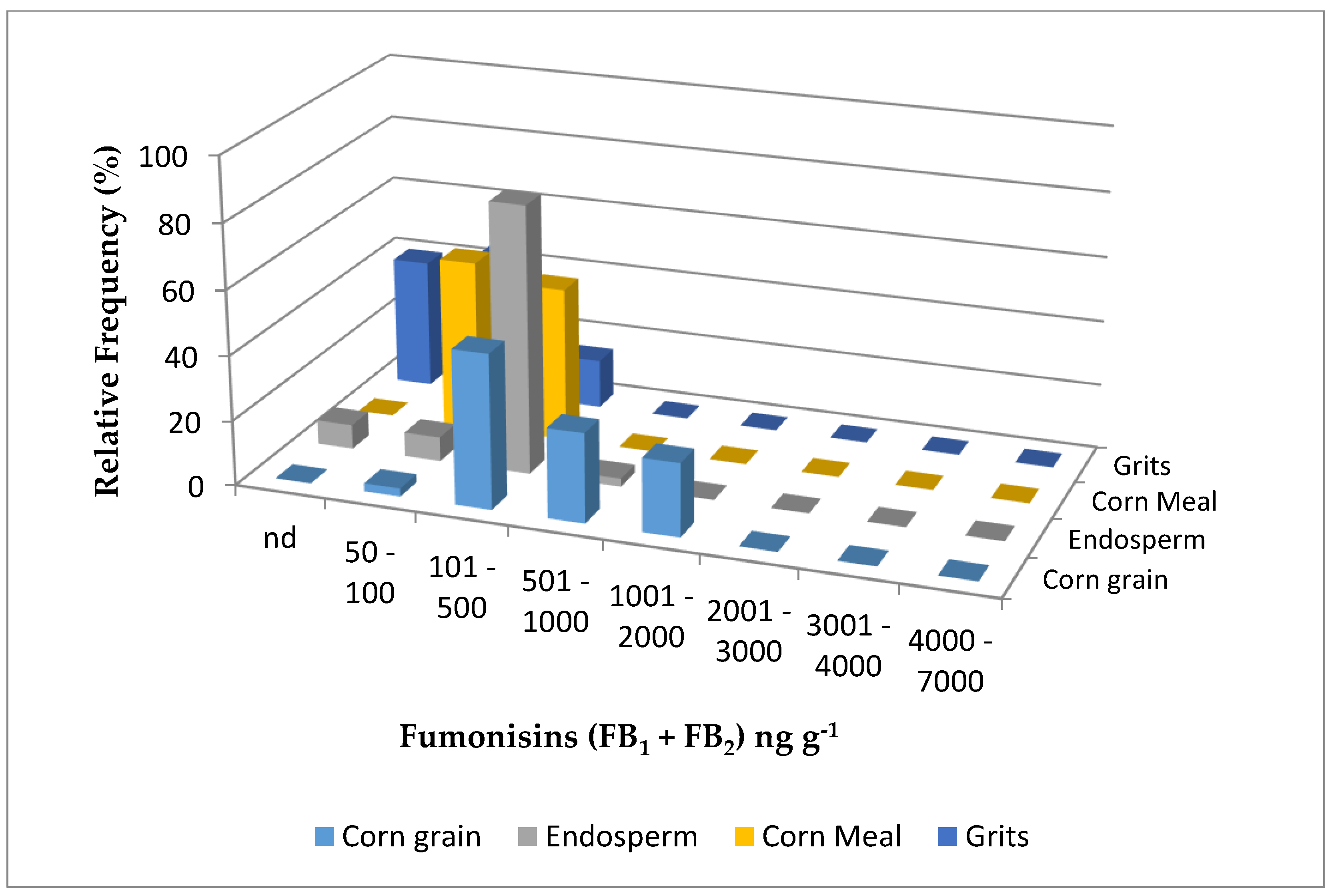
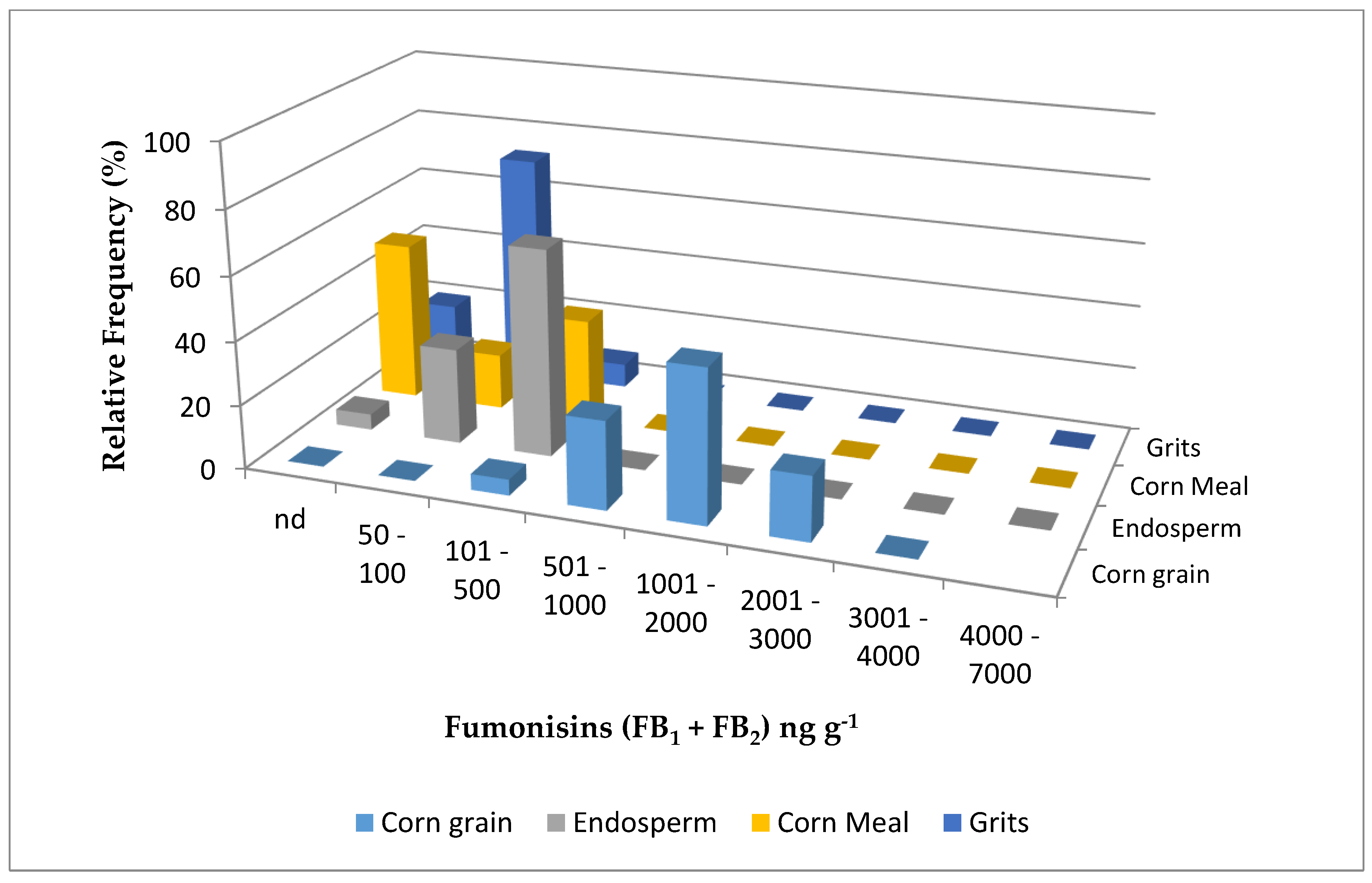
| Year | Sample | N | Water Activity (aw) | Fumonisin B1 | Fumonisin B2 | Fumonisin B1 + B2 | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mean x | Positive Samples (%) | Mean y (ng g−1) | Range (ng g−1) | Positive Samples (%) | Mean y (ng g−1) | Range (ng g−1) | Mean y (ng g−1) | Range (ng g−1) | |||
| 2015 | |||||||||||
| Corn grain | 40 | 0.62 abB | 100 | 480 aB | 121–1441 | 53 | 283 aB | 142–568 | 631 aB | 121–1821 | |
| Endosperm | 40 | 0.64 aA | 93 | 198 bA | 95.0–569 | - | - | - | 198 bA | 95.0–569 | |
| Cornmeal | 40 | 0.61 bB | 100 | 118 cB | 41.0–287 | 5 | 53 b | 50.0–55.0 | 121 cA | 41.0–389 | |
| Corn grits | 40 | 0.60 bB | 60 | 86 cA | 36.0–208 | - | - | - | 86 cA | 36.0–208 | |
| 2016 | |||||||||||
| Corn grain | 40 | 0.65 aA | 100 | 1080 aA | 303–2144 | 88 | 385 A | 77.0–785 | 1417 aA | 303–2863 | |
| Endosperm | 40 | 0.65 aA | 95 | 182 bA | 125–395 | - | - | - | 182 bA | 125–395 | |
| Cornmeal | 40 | 0.66 aA | 50 | 160 bA | 24.0–389 | - | - | - | 160 bA | 24.0–389 | |
| Corn grits | 40 | 0.65 aA | 80 | 54.0 cB | 22.0–256 | - | - | - | 54.0 cB | 22.0–256 | |
| 2015 and | |||||||||||
| 2016 z | Corn grain | 80 | 0.63 a | 100 | 783 a | 155–2140 | 70 | 346 | 77.0–785 | 1026 a | 121–2730 |
| Endosperm | 80 | 0.64 a | 94 | 191 b | 54.0–569 | - | - | - | 191 b | 54.0–569 | |
| Cornmeal | 80 | 0.63 a | 75 | 137 b | 41.0–389 | 2.5 | 53 | 50.0–55.0 | 137 b | 41.0–389 | |
| Corn grits | 80 | 0.63 a | 70 | 70.6 c | 36.0–256 | - | - | - | 70.6 c | 36.0–256 | |
| Sample Type | Mean Fumonisin Levels (ng g−1) | Average Consumption (g/person/day) | Probably Daily Intake (ng kg−1 b.w. day−1) | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Brazil | Europe | Malaysia | Angola | Brazil | Europe | Malaysia | Angola | ||||||||||
| United Kingdom | France | Germany | Netherlands | Italy | United Kingdom | France | Germany | Netherlands | Italy | ||||||||
| Corn | 1026 | 5.80 | 10.4 | 2.44 | 7.48 | 3.0 | 11.5 | 43.3 | 106 | 85.0 | 152 | 35.7 | 109 | 43.9 | 168 | 634 | 1553 |
| Endosperm | 191 | 0.59 | 1.20 | 18.4 | 1.50 | 1.62 | 3.29 | 50.4 | 4.11 | ||||||||
| Cornmeal | 99.9 a1 | 6.63 | 14.9 | 3.99 | 1.00 | 9.46 b1 | 21.2 b1 | 5.69 b1 | 1.42 b1 | ||||||||
| 107 a2 | 10.3 b2 | 22.7 b2 | 6.09 b2 | 1.52 b2 | |||||||||||||
| Grits | 47.1 | 0.02 | 11.6 | 0.01 | 7.80 | ||||||||||||
| Total | 13.0 | 96.9 c | 177 c | 35.7 | 173 c | 43.9 | 173 c | 634 | 1553 | ||||||||
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bordini, J.G.; Ono, M.A.; Hirozawa, M.T.; Garcia, G.T.; Vizoni, E.; Ono, E.Y.S. Safety of Corn and Corn-Based Products Intended for Human Consumption Concerning Fumonisins from a Brazilian Processing Plant. Toxins 2019, 11, 33. https://doi.org/10.3390/toxins11010033
Bordini JG, Ono MA, Hirozawa MT, Garcia GT, Vizoni E, Ono EYS. Safety of Corn and Corn-Based Products Intended for Human Consumption Concerning Fumonisins from a Brazilian Processing Plant. Toxins. 2019; 11(1):33. https://doi.org/10.3390/toxins11010033
Chicago/Turabian StyleBordini, Jaqueline Gozzi, Mario Augusto Ono, Melissa Tiemi Hirozawa, Glauco Tironi Garcia, Edio Vizoni, and Elisabete Yurie Sataque Ono. 2019. "Safety of Corn and Corn-Based Products Intended for Human Consumption Concerning Fumonisins from a Brazilian Processing Plant" Toxins 11, no. 1: 33. https://doi.org/10.3390/toxins11010033
APA StyleBordini, J. G., Ono, M. A., Hirozawa, M. T., Garcia, G. T., Vizoni, E., & Ono, E. Y. S. (2019). Safety of Corn and Corn-Based Products Intended for Human Consumption Concerning Fumonisins from a Brazilian Processing Plant. Toxins, 11(1), 33. https://doi.org/10.3390/toxins11010033